TULA-Family Regulators of Platelet Activation
Abstract
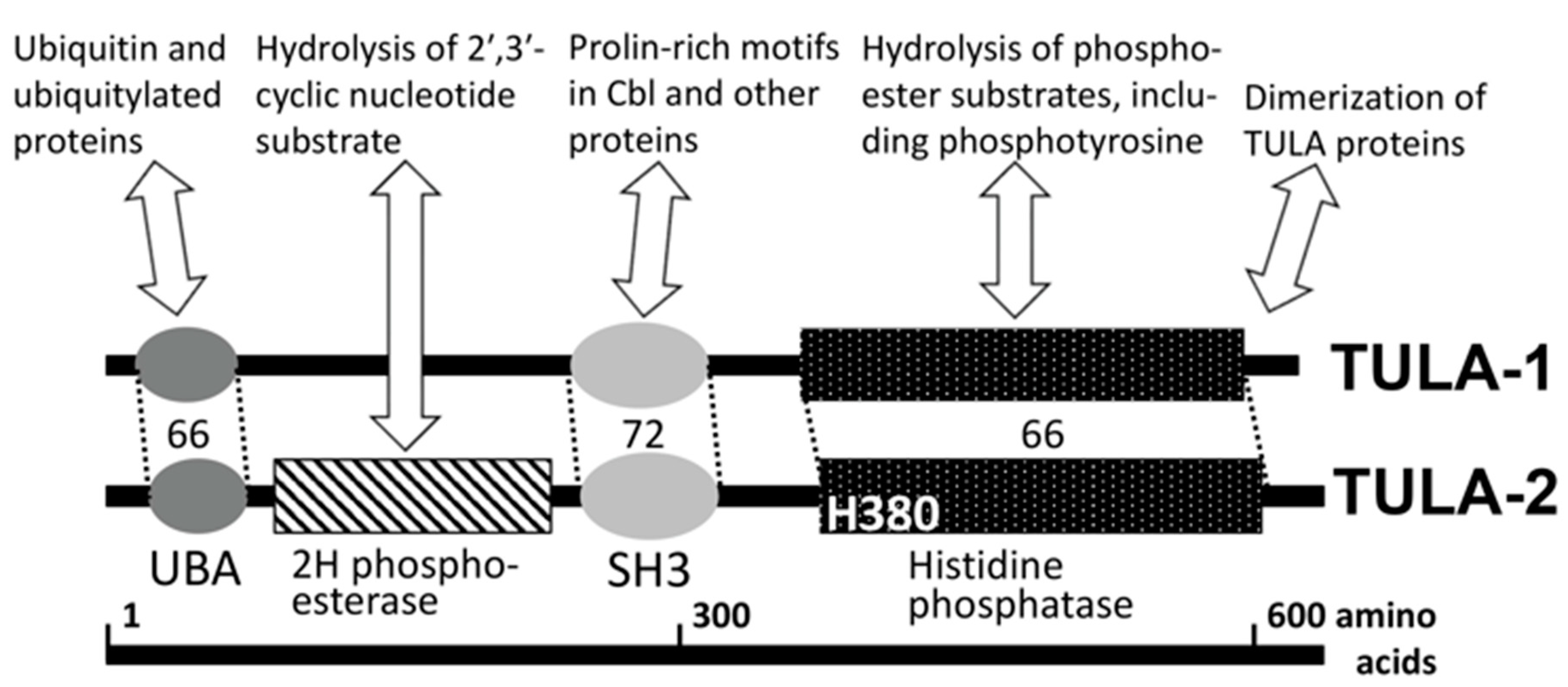
1. Novel Family of Atypical Protein Tyrosine Phosphatases
2. PTP Activities of TULA-Family Proteins and Their Possible PTP-Independent Functions
3. Regulatory Effect of TULA-2 on Platelet Signaling and Activation
3.1. Effects of TULA-Family Proteins in Cells Other Than Platelets
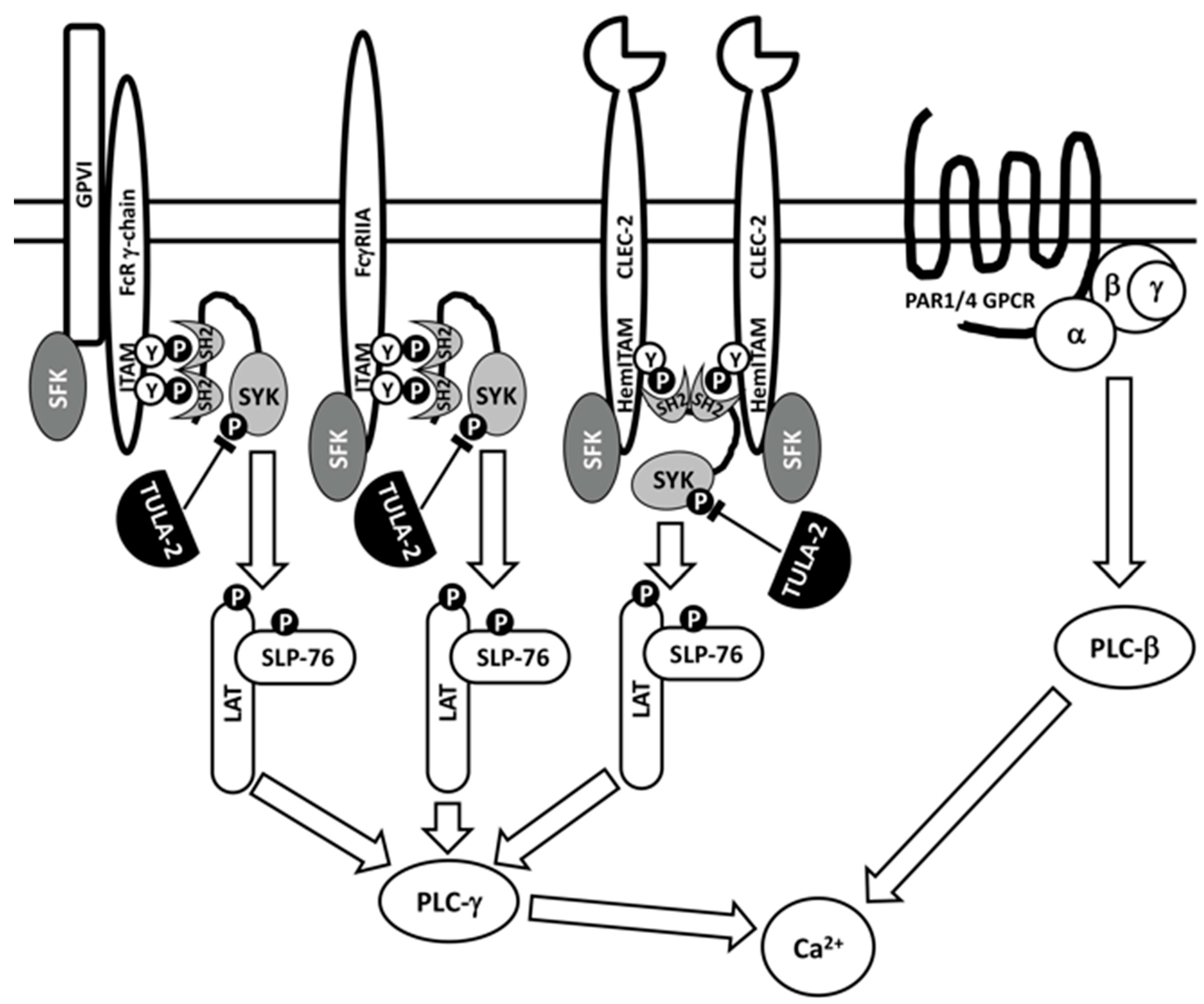
3.2. Effects of TULA-2 on Signaling Mediated by the Glycoprotein VI (GPVI) Collagen Receptor
3.3. Effects of TULA-2 on Signaling through FcγRIIA, A Receptor for the Fc Fragment of IgG
3.4. Effects of TULA-2 on Signaling through the C-Type Lectin-like (CLEC)-2 Receptor
3.5. Physiological Consequences of TULA-2-Mediated Signaling Regulation
4. Molecular Basis of the Regulatory Effect of TULA-2 on Platelet Signaling and Activation
5. Regulation of TULA-2 Level and Activity in Platelets
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wattenhofer, M.; Shibuya, K.; Kudoh, J.; Lyle, R.; Michaud, J.; Rossier, C.; Kawasaki, K.; Asakawa, S.; Minoshima, S.; Berry, A.; et al. Isolation and characterization of the UBASH3A gene on 21q22.3 encoding a potential nuclear protein with a novel combination of domains. Hum. Genet. 2001, 108, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Carpino, N.; Kobayashi, R.; Zang, H.; Takahashi, Y.; Jou, S.T.; Feng, J.; Nakajima, H.; Ihle, J.N. Identification, cDNA cloning, and targeted deletion of p70, a novel, ubiquitously expressed SH3 domain-containing protein. Mol. Cell Biol. 2002, 22, 7491–7500. [Google Scholar] [CrossRef] [PubMed]
- Carpino, N.; Turner, S.; Mekala, D.; Takahashi, Y.; Zang, H.; Geiger, T.L.; Doherty, P.; Ihle, J.N. Regulation of ZAP-70 activation and TCR signaling by two related proteins, Sts-1 and Sts-2. Immunity 2004, 20, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Feshchenko, E.A.; Smirnova, E.V.; Swaminathan, G.; Teckchandani, A.M.; Agrawal, R.; Band, H.; Zhang, X.; Annan, R.S.; Carr, S.A.; Tsygankov, A.Y. TULA: An SH3- and UBA-containing protein that binds to c-Cbl and ubiquitin. Oncogene 2004, 23, 4690–4706. [Google Scholar] [CrossRef] [PubMed]
- Kowanetz, K.; Crosetto, N.; Haglund, K.; Schmidt, M.H.; Heldin, C.H.; Dikic, I. Suppressors of T-cell receptor signaling Sts-1 and Sts-2 bind to Cbl and inhibit endocytosis of receptor tyrosine kinases. J. Biol. Chem. 2004, 279, 32786–32795. [Google Scholar] [CrossRef]
- Tsygankov, A. Multi-domain STS/TULA protein are novel cellular regulators. IUBMB Life 2008, 60, 224–231. [Google Scholar] [CrossRef]
- Tsygankov, A.Y. TULA-family proteins: An odd couple. Cell Mol. Life Sci. 2009, 66, 2949–2952. [Google Scholar] [CrossRef]
- Tsygankov, A.Y. TULA-family proteins: A new class of cellular regulators. J. Cell Physiol. 2013, 228, 43–49. [Google Scholar] [CrossRef]
- Tsygankov, A.Y. TULA-family proteins: Jacks of many trades and then some. J. Cell Physiol. 2019, 234, 274–288. [Google Scholar] [CrossRef]
- Tsygankov, A.Y. TULA proteins as signaling regulators. Cell Signal 2020, 65, 109424. [Google Scholar] [CrossRef]
- Rigden, D.J. The histidine phosphatase superfamily: Structure and function. Biochem. J. 2008, 409, 333–348. [Google Scholar] [CrossRef] [PubMed]
- Mikhailik, A.; Ford, B.; Keller, J.; Chen, Y.; Nassar, N.; Carpino, N. A phosphatase activity of Sts-1 contributes to the suppression of TCR signaling. Mol. Cell 2007, 27, 486–497. [Google Scholar] [CrossRef]
- Tonks, N.K. Protein tyrosine phosphatases--from housekeeping enzymes to master regulators of signal transduction. FEBS J. 2013, 280, 346–378. [Google Scholar] [CrossRef]
- Tiganis, T.; Bennett, A.M. Protein tyrosine phosphatase function: The substrate perspective. Biochem. J. 2007, 402, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Bertelsen, V.; Breen, K.; Sandvig, K.; Stang, E.; Madshus, I.H. The Cbl-interacting protein TULA inhibits dynamin-dependent endocytosis. Exp. Cell Res. 2007, 313, 1696–1709. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Paisie, T.K.; Chen, S.; Concannon, P. UBASH3A Regulates the Synthesis and Dynamics of TCR-CD3 Complexes. J. Immunol. 2019, 203, 2827–2836. [Google Scholar] [CrossRef]
- Hoeller, D.; Crosetto, N.; Blagoev, B.; Raiborg, C.; Tikkanen, R.; Wagner, S.; Kowanetz, K.; Breitling, R.; Mann, M.; Stenmark, H.; et al. Regulation of ubiquitin-binding proteins by monoubiquitination. Nat. Cell Biol. 2006, 8, 163–169. [Google Scholar] [CrossRef]
- Smirnova, E.V.; Collingwood, T.S.; Bisbal, C.; Tsygankova, O.M.; Bogush, M.; Meinkoth, J.L.; Henderson, E.E.; Annan, R.S.; Tsygankov, A.Y. TULA proteins bind to ABCE-1, a host factor of HIV-1 assembly, and inhibit HIV-1 biogenesis in a UBA-dependent fashion. Virology 2008, 372, 10–23. [Google Scholar] [CrossRef]
- Krupina, K.; Kleiss, C.; Metzger, T.; Fournane, S.; Schmucker, S.; Hofmann, K.; Fischer, B.; Paul, N.; Porter, I.M.; Raffelsberger, W.; et al. Ubiquitin Receptor Protein UBASH3B Drives Aurora B Recruitment to Mitotic Microtubules. Dev. Cell 2016, 36, 63–78. [Google Scholar] [CrossRef]
- Kong, M.S.; Hashimoto-Tane, A.; Kawashima, Y.; Sakuma, M.; Yokosuka, T.; Kometani, K.; Onishi, R.; Carpino, N.; Ohara, O.; Kurosaki, T.; et al. Inhibition of T cell activation and function by the adaptor protein CIN. Sci. Signal 2019, 12, 567. [Google Scholar] [CrossRef]
- Ge, Y.; Paisie, T.K.; Newman, J.R.B.; McIntyre, L.M.; Concannon, P. UBASH3A Mediates Risk for Type 1 Diabetes Through Inhibition of T-Cell Receptor-Induced NF-kappaB Signaling. Diabetes 2017, 66, 2033–2043. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Frank, D.; Zhou, W.; Kaur, N.; French, J.B.; Carpino, N. An unexpected 2-histidine phosphoesterase activity of suppressor of T-cell receptor signaling protein 1 contributes to the suppression of cell signaling. J. Biol. Chem. 2020, 295, 8514–8523. [Google Scholar] [CrossRef] [PubMed]
- Kleinman, H.; Ford, B.; Keller, J.; Carpino, N.; Nassar, N. Crystallization and initial crystal characterization of the C-terminal phosphoglycerate mutase homology domain of Sts-1. Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun. 2006, 62, 218–220. [Google Scholar] [CrossRef]
- Chen, Y.; Jakoncic, J.; Carpino, N.; Nassar, N. Structural and functional characterization of the 2H-phosphatase domain of Sts-2 reveals an acid-dependent phosphatase activity. Biochemistry 2009, 48, 1681–1690. [Google Scholar] [CrossRef]
- Chen, Y.; Jakoncic, J.; Parker, K.A.; Carpino, N.; Nassar, N. Structures of the phosphorylated and VO(3)-bound 2H-phosphatase domain of Sts-2. Biochemistry 2009, 48, 8129–8135. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Yin, Y.; Weinheimer, A.S.; Kaur, N.; Carpino, N.; French, J.B. Structural and Functional Characterization of the Histidine Phosphatase Domains of Human Sts-1 and Sts-2. Biochemistry 2017, 56, 4637–4645. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, R.; Carpino, N.; Tsygankov, A. TULA proteins regulate activity of the protein tyrosine kinase Syk. J. Cell Biochem. 2008, 104, 953–964. [Google Scholar] [CrossRef]
- San Luis, B.; Sondgeroth, B.; Nassar, N.; Carpino, N. Sts-2 is a phosphatase that negatively regulates zeta-associated protein (ZAP)-70 and T cell receptor signaling pathways. J. Biol. Chem. 2011, 286, 15943–15954. [Google Scholar] [CrossRef]
- Chen, X.; Ren, L.; Kim, S.; Carpino, N.; Daniel, J.L.; Kunapuli, S.P.; Tsygankov, A.Y.; Pei, D. Determination of the substrate specificity of protein-tyrosine phosphatase TULA-2 and identification of Syk as a TULA-2 substrate. J. Biol. Chem. 2010, 285, 31268–31276. [Google Scholar] [CrossRef]
- Levkowitz, G.; Waterman, H.; Ettenberg, S.A.; Katz, M.; Tsygankov, A.Y.; Alroy, I.; Lavi, S.; Iwai, K.; Reiss, Y.; Ciechanover, A.; et al. Ubiquitin ligase activity and tyrosine phosphorylation underlie suppression of growth factor signaling by c-Cbl/Sli-1. Mol. Cell 1999, 4, 1029–1040. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yeung, Y.G.; Stanley, E.R. CSF-1 stimulated multiubiquitination of the CSF-1 receptor and of Cbl follows their tyrosine phosphorylation and association with other signaling proteins. J. Cell Biochem. 1999, 72, 119–134. [Google Scholar] [CrossRef]
- Yokouchi, M.; Kondo, T.; Houghton, A.; Bartkiewicz, M.; Horne, W.C.; Zhang, H.; Yoshimura, A.; Baron, R. Ligand-induced ubiquitination of the epidermal growth factor receptor involves the interaction of the c-Cbl RING finger and UbcH. J. Biol. Chem. 1999, 274, 31707–31712. [Google Scholar] [CrossRef] [PubMed]
- Paolini, R.; Molfetta, R.; Beitz, L.O.; Zhang, J.; Scharenberg, A.M.; Piccoli, M.; Frati, L.; Siraganian, R.; Santoni, A. Activation of Syk tyrosine kinase is required for c-Cbl-mediated ubiquitination of Fcepsilon RI and Syk in RBL cells. J. Biol. Chem. 2002, 277, 36940–36947. [Google Scholar] [CrossRef] [PubMed]
- Dangelmaier, C.A.; Quinter, P.G.; Jin, J.; Tsygankov, A.Y.; Kunapuli, S.P.; Daniel, J.L. Rapid ubiquitination of Syk following GPVI activation in platelets. Blood 2005, 105, 3918–3924. [Google Scholar] [CrossRef]
- Rao, N.; Ghosh, A.K.; Ota, S.; Zhou, P.; Reddi, A.L.; Hakezi, K.; Druker, B.K.; Wu, J.; Band, H. The non-receptor tyrosine kinase Syk is a target of Cbl-mediated ubiquitylation upon B-cell receptor stimulation. EMBO J. 2001, 20, 7085–7095. [Google Scholar] [CrossRef] [PubMed]
- Carpino, N.; Chen, Y.; Nassar, N.; Oh, H.W. The Sts proteins target tyrosine phosphorylated, ubiquitinated proteins within TCR signaling pathways. Mol. Immunol. 2009, 46, 3224–3231. [Google Scholar] [CrossRef] [PubMed]
- Newman, T.N.; Liverani, E.; Ivanova, E.; Russo, G.L.; Carpino, N.; Ganea, D.; Safadi, F.; Kunapuli, S.P.; Tsygankov, A.Y. Members of the novel UBASH3/STS/TULA family of cellular regulators suppress T-cell-driven inflammatory responses in vivo. Immunol. Cell Biol. 2014, 92, 837–850. [Google Scholar] [CrossRef]
- Collingwood, T.S.; Smirnova, E.V.; Bogush, M.; Carpino, N.; Annan, R.S.; Tsygankov, A.Y. T-cell ubiquitin ligand affects cell death through a functional interaction with apoptosis-inducing factor, a key factor of caspase-independent apoptosis. J. Biol. Chem. 2007, 282, 30920–30928. [Google Scholar] [CrossRef] [PubMed]
- Okabe, N.; Ohmura, K.; Katayama, M.; Akizuki, S.; Carpino, N.; Murakami, K.; Nakashima, R.; Hashimoto, M.; Imura, Y.; Yoshifuji, H.; et al. Suppressor of TCR signaling-2 (STS-2) suppresses arthritis development in mice. Mod. Rheumatol. 2018, 28, 626–636. [Google Scholar] [CrossRef] [PubMed]
- Frank, D.; Naseem, S.; Russo, G.L.; Li, C.; Parashar, K.; Konopka, J.B.; Carpino, N. Phagocytes from mice lacking the Sts phosphatases have an enhanced antifungal response to Candida albicans. mBio 2018, 9, e00782-18. [Google Scholar] [CrossRef]
- Parashar, K.; Carpino, N. A role for the Sts phosphatases in negatively regulating IFNgamma-mediated production of nitric oxide in monocytes. Immun. Inflamm. Dis. 2020, 8, 523–533. [Google Scholar] [CrossRef] [PubMed]
- Back, S.H.; Adapala, N.S.; Barbe, M.F.; Carpino, N.C.; Tsygankov, A.Y.; Sanjay, A. TULA-2, a novel histidine phosphatase, regulates bone remodeling by modulating osteoclast function. Cell Mol. Life Sci. 2013, 70, 1269–1284. [Google Scholar] [CrossRef] [PubMed]
- De Castro, R.O.; Zhang, J.; Groves, J.R.; Barbu, E.A.; Siraganian, R.P. Once phosphorylated, tyrosines in carboxyl terminus of protein-tyrosine kinase Syk interact with signaling proteins, including TULA-2, a negative regulator of mast cell degranulation. J. Biol. Chem. 2012, 287, 8194–8204. [Google Scholar] [CrossRef] [PubMed]
- Au-Yeung, B.B.; Shah, N.H.; Shen, L.; Weiss, A. ZAP-70 in Signaling, Biology, and Disease. Annu. Rev. Immunol. 2017, 36, 127–156. [Google Scholar] [CrossRef]
- Geahlen, R.L. Syk and pTyr’d: Signaling through the B cell antigen receptor. Biochim. Biophys. Acta 2009, 1793, 1115–1127. [Google Scholar] [CrossRef] [PubMed]
- Mocsai, A.; Ruland, J.; Tybulewicz, V.L. The SYK tyrosine kinase: A crucial player in diverse biological functions. Nat. Rev. Immunol. 2010, 10, 387–402. [Google Scholar] [CrossRef] [PubMed]
- Tsygankov, A.Y. Non-receptor protein tyrosine kinases. Front. Biosci. 2003, 8, s595–s635. [Google Scholar] [CrossRef]
- Thomas, D.H.; Getz, T.M.; Newman, T.N.; Dangelmaier, C.A.; Carpino, N.; Kunapuli, S.P.; Tsygankov, A.Y.; Daniel, J.L. A novel histidine tyrosine phosphatase, TULA-2, associates with Syk and negatively regulates GPVI signaling in platelets. Blood 2010, 116, 2570–2578. [Google Scholar] [CrossRef] [PubMed]
- Lim, C.K.; Hwang, W.Y.; Aw, S.E.; Sun, L. Study of gene expression profile during cord blood-associated megakaryopoiesis. Eur. J. Haematol. 2008, 81, 196–208. [Google Scholar] [CrossRef]
- Clark, E.A.; Shattil, S.J.; Brugge, J.S. Regulation of protein tyrosine kinases in platelets. Trends Biochem. Sci. 1994, 19, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Clark, E.A.; Shattil, S.J.; Ginsberg, M.H.; Bolen, J.; Brugge, J.S. Regulation of the protein tyrosine kinase pp72(syk) by platelet agonists and the integrin alpha(IIb)beta(3). J. Biol. Chem. 1994, 269, 28859–28864. [Google Scholar] [CrossRef] [PubMed]
- Yanaga, F.; Poole, A.; Asselin, J.; Blake, R.; Schieven, G.L.; Clark, E.A.; Law, C.L.; Watson, S.P. Syk interacts with tyrosine-phosphorylated proteins in human platelets activated by collagen and cross-linking of the Fc gamma-IIA receptor. Biochem. J. 1995, 311, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Law, D.A.; Nannizzi-Alaimo, L.; Ministri, K.; Hughes, P.E.; Forsyth, J.; Turner, M.; Shattil, S.J.; Ginsberg, M.H.; Tybulewicz, V.L.; Phillips, D.R. Genetic and pharmacological analyses of Syk function in alphaIIbbeta3 signaling in platelets. Blood 1999, 93, 2645–2652. [Google Scholar] [CrossRef] [PubMed]
- Watson, S.P. Collagen receptor signaling in platelets and megakaryocytes. Thromb. Haemost. 1999, 82, 365–376. [Google Scholar] [CrossRef] [PubMed]
- Bhavaraju, K.; Kim, S.; Daniel, J.L.; Kunapuli, S.P. Evaluation of [3-(1-methyl-1H-indol-3-yl-methylene)-2-oxo-2, 3-dihydro-1H-indole-5-sulfonamide] (OXSI-2), as a Syk-selective inhibitor in platelets. Eur. J. Pharmacol. 2008, 580, 285–290. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Getz, T.M.; Mayanglambam, A.; Daniel, J.L.; Kunapuli, S.P. Go6976 abrogates GPVI-mediated platelet functional responses in human platelets through inhibition of Syk. J. Thromb. Haemost. 2011, 9, 608–610. [Google Scholar] [CrossRef] [PubMed]
- Manne, B.K.; Badolia, R.; Dangelmaier, C.; Eble, J.A.; Ellmeier, W.; Kahn, M.; Kunapuli, S.P. Distinct pathways regulate Syk protein activation downstream of immune tyrosine activation motif (ITAM) and hemITAM receptors in platelets. J. Biol. Chem. 2015, 290, 11557–11568. [Google Scholar] [CrossRef]
- Zeiler, M.; Moser, M.; Mann, M. Copy number analysis of the murine platelet proteome spanning the complete abundance range. Mol. Cell Proteom. 2014, 13, 3435–3445. [Google Scholar] [CrossRef] [PubMed]
- Burkhart, J.M.; Vaudel, M.; Gambaryan, S.; Radau, S.; Walter, U.; Martens, L.; Geiger, J.; Sickmann, A.; Zahedi, R.P. The first comprehensive and quantitative analysis of human platelet protein composition allows the comparative analysis of structural and functional pathways. Blood 2012, 120, e73–e82. [Google Scholar] [CrossRef] [PubMed]
- Reppschlager, K.; Gosselin, J.; Dangelmaier, C.A.; Thomas, D.H.; Carpino, N.; McKenzie, S.E.; Kunapuli, S.P.; Tsygankov, A.Y. TULA-2 Protein Phosphatase Suppresses Activation of Syk through the GPVI Platelet Receptor for Collagen by Dephosphorylating Tyr(P)346, a Regulatory Site of Syk. J. Biol. Chem. 2016, 291, 22427–22441. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Abraham, S.; Andre, P.; Edelstein, L.C.; Shaw, C.A.; Dangelmaier, C.A.; Tsygankov, A.Y.; Kunapuli, S.P.; Bray, P.F.; McKenzie, S.E. Anti-miR-148a regulates platelet FcgammaRIIA signaling and decreases thrombosis in vivo in mice. Blood 2015, 126, 2871–2881. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Abraham, S.; Renna, S.; Edelstein, L.C.; Dangelmaier, C.A.; Tsygankov, A.Y.; Kunapuli, S.P.; Bray, P.F.; McKenzie, S.E. TULA-2 (T-Cell Ubiquitin Ligand-2) Inhibits the Platelet Fc Receptor for IgG IIA (FcgammaRIIA) Signaling Pathway and Heparin-Induced Thrombocytopenia in Mice. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 2315–2323. [Google Scholar] [CrossRef]
- Kostyak, J.C.; Mauri, B.R.; Dangelmaier, C.; Patel, A.; Zhou, Y.; Eble, J.A.; Tsygankov, A.Y.; McKenzie, S.E.; Kunapuli, S.P. TULA-2 Deficiency Enhances Platelet Functional Responses to CLEC-2 Agonists. TH Open 2018, 2, e411–e419. [Google Scholar] [CrossRef]
- Hughes, C.E.; Pollitt, A.Y.; Mori, J.; Eble, J.A.; Tomlinson, M.G.; Hartwig, J.H.; O’Callaghan, C.A.; Futterer, K.; Watson, S.P. CLEC-2 activates Syk through dimerization. Blood 2010, 115, 2947–2955. [Google Scholar] [CrossRef] [PubMed]
- Stefanini, L.; Bergmeier, W. Negative regulators of platelet activation and adhesion. J. Thromb. Haemost. 2018, 16, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.J.; Yankee, T.M.; Harrison, M.L.; Geahlen, R.L. Regulation of signaling in B cells through the phosphorylation of Syk on linker region tyrosines. A mechanism for negative signaling by the Lyn tyrosine kinase. J. Biol. Chem. 2002, 277, 31703–31714. [Google Scholar] [CrossRef]
- Zhang, J.; Berenstein, E.; Siraganian, R.P. Phosphorylation of Tyr342 in the linker region of Syk is critical for Fc epsilon RI signaling in mast cells. Mol. Cell Biol. 2002, 22, 8144–8154. [Google Scholar] [CrossRef]
- Sanderson, M.P.; Wex, E.; Kono, T.; Uto, K.; Schnapp, A. Syk and Lyn mediate distinct Syk phosphorylation events in FcvarepsilonRI-signal transduction: Implications for regulation of IgE-mediated degranulation. Mol. Immunol. 2010, 48, 171–178. [Google Scholar] [CrossRef]
- Bohnenberger, H.; Oellerich, T.; Engelke, M.; Hsiao, H.H.; Urlaub, H.; Wienands, J. Complex phosphorylation dynamics control the composition of the Syk interactome in B cells. Eur. J. Immunol. 2011, 41, 1550–1562. [Google Scholar] [CrossRef]
- Caruso, J.A.; Carruthers, N.; Shin, N.; Gill, R.; Stemmer, P.M.; Rosenspire, A. Mercury alters endogenous phosphorylation profiles of SYK in murine B cells. BMC Immunol. 2017, 18, 37. [Google Scholar] [CrossRef]
- Couture, C.; Baier, G.; Altman, A.; Mustelin, T. p56lck-independent activation and tyrosine phosphorylation of p72syk by T-cell antigen receptor/CD3 stimulation. Proc. Natl. Acad. Sci. USA 1994, 91, 5301–5305. [Google Scholar] [CrossRef] [PubMed]
- Kurosaki, T.; Johnson, S.A.; Pao, L.; Sada, K.; Yamamura, H.; Cambier, J.C. Role of the Syk autophosphorylation site and SH2 domains in B cell antigen receptor signaling. J. Exp. Med. 1995, 182, 1815–1823. [Google Scholar] [CrossRef]
- Mansueto, M.S.; Reens, A.; Rakhilina, L.; Chi, A.; Pan, B.S.; Miller, J.R. A reevaluation of the spleen tyrosine kinase (SYK) activation mechanism. J. Biol. Chem. 2019, 294, 7658–7668. [Google Scholar] [CrossRef] [PubMed]
- Simon, M.; Vanes, L.; Geahlen, R.L.; Tybulewicz, V.L. Distinct roles for the linker region tyrosines of Syk in FcepsilonRI signaling in primary mast cells. J. Biol. Chem. 2005, 280, 4510–4517. [Google Scholar] [CrossRef] [PubMed]
- Groesch, T.D.; Zhou, F.; Mattila, S.; Geahlen, R.L.; Post, C.B. Structural basis for the requirement of two phosphotyrosine residues in signaling mediated by Syk tyrosine kinase. J. Mol. Biol. 2006, 356, 1222–1236. [Google Scholar] [CrossRef] [PubMed]
- Tsang, E.; Giannetti, A.M.; Shaw, D.; Dinh, M.; Tse, J.K.; Gandhi, S.; Ho, H.; Wang, S.; Papp, E.; Bradshaw, J.M. Molecular mechanism of the Syk activation switch. J. Biol. Chem. 2008, 283, 32650–32659. [Google Scholar] [CrossRef]
- Gradler, U.; Schwarz, D.; Dresing, V.; Musil, D.; Bomke, J.; Frech, M.; Greiner, H.; Jakel, S.; Rysiok, T.; Muller-Pompalla, D.; et al. Structural and biophysical characterization of the Syk activation switch. J. Mol. Biol. 2013, 425, 309–333. [Google Scholar] [CrossRef]
- Sada, K.; Zhang, J.; Siraganian, R.P. Point mutation of a tyrosine in the linker region of Syk results in a gain of function. J. Immunol. 2000, 164, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Lupher, M.L., Jr.; Rao, N.; Lill, N.L.; Andoniou, C.E.; Miyake, S.; Clark, E.A.; Druker, B.; Band, H. Cbl-mediated negative regulation of the Syk tyrosine kinase. A critical role for Cbl phosphotyrosine-binding domain binding to Syk phosphotyrosine. J. Biol. Chem. 1998, 273, 35273–35281. [Google Scholar] [CrossRef] [PubMed]
- Ota, S.; Hazeki, K.; Rao, N.; Lupher, M.L., Jr.; Andoniou, C.E.; Druker, B.; Band, H. The RING finger domain of Cbl is essential for negative regulation of the Syk tyrosine kinase. J. Biol. Chem. 2000, 275, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Tsygankov, A.; Bolen, J. The Src Family of Tyrosine Protein Kinases in Hemopoietic Signal Transduction. Stem Cells 1993, 11, 371–380. [Google Scholar] [CrossRef]
- Guse, A.H.; Tsygankov, A.Y.; Weber, K.; Mayr, G.W. Transient tyrosine phosphorylation of human ryanodine receptor upon T cell stimulation. J. Biol. Chem. 2001, 276, 34722–34727. [Google Scholar] [CrossRef] [PubMed]
- Hasham, M.G.; Tsygankov, A.Y. Tip, an Lck-interacting protein of Herpesvirus saimiri, causes Fas- and Lck-dependent apoptosis of T lymphocytes. Virology 2004, 320, 313–329. [Google Scholar] [CrossRef][Green Version]
- Chari, R.; Kim, S.; Murugappan, S.; Sanjay, A.; Daniel, J.L.; Kunapuli, S.P. Lyn, PKC-delta, SHIP-1 interactions regulate GPVI-mediated platelet-dense granule secretion. Blood 2009, 114, 3056–3063. [Google Scholar] [CrossRef]
- Dorsam, R.T.; Kim, S.; Murugappan, S.; Rachoor, S.; Shankar, H.; Jin, J.; Kunapuli, S.P. Differential requirements for calcium and Src family kinases in platelet GPIIb/IIIa activation and thromboxane generation downstream of different G-protein pathways. Blood 2005, 105, 2749–2756. [Google Scholar] [CrossRef][Green Version]
- Quek, L.S.; Pasquet, J.M.; Hers, I.; Cornall, R.; Knight, G.; Barnes, M.; Hibbs, M.L.; Dunn, A.R.; Lowell, C.A.; Watson, S.P. Fyn and Lyn phosphorylate the Fc receptor gamma chain downstream of glycoprotein VI in murine platelets, and Lyn regulates a novel feedback pathway. Blood 2000, 96, 4246–4253. [Google Scholar] [CrossRef] [PubMed]
- Lowell, C.A. Src-family and Syk kinases in activating and inhibitory pathways in innate immune cells: Signaling cross talk. Cold Spring Harb. Perspect Biol. 2011, 3, a002352. [Google Scholar] [CrossRef]
- Ma, Y.C.; Huang, J.; Ali, S.; Lowry, W.; Huang, X.Y. Src tyrosine kinase is a novel direct effector of G proteins. Cell 2000, 102, 635–646. [Google Scholar] [CrossRef] [PubMed]
- Luis, B.S.; Carpino, N. Insights into the suppressor of T-cell receptor (TCR) signaling-1 (Sts-1)-mediated regulation of TCR signaling through the use of novel substrate-trapping Sts-1 phosphatase variants. FEBS J. 2014, 281, 696–707. [Google Scholar] [CrossRef] [PubMed]
- Dunster, J.L.; Mazet, F.; Fry, M.J.; Gibbins, J.M.; Tindall, M.J. Regulation of early steps of GPVI signal transduction by phosphatases: A Systems biology approach. PLoS Comput. Biol. 2015, 11, e1004589. [Google Scholar] [CrossRef] [PubMed]
- Beck, F.; Geiger, J.; Gambaryan, S.; Solari, F.A.; Dell’Aica, M.; Loroch, S.; Mattheij, N.J.; Mindukshev, I.; Potz, O.; Jurk, K.; et al. Temporal quantitative phosphoproteomics of ADP stimulation reveals novel central nodes in platelet activation and inhibition. Blood 2017, 129, e1–e12. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.E.; White, F.M. Quantitative analysis of phosphotyrosine signaling networks triggered by CD3 and CD28 costimulation in Jurkat cells. J. Immunol. 2006, 176, 2833–2843. [Google Scholar] [CrossRef] [PubMed]
- Babur, O.; Melrose, A.R.; Cunliffe, J.M.; Klimek, J.; Pang, J.; Sepp, A.I.; Zilberman-Rudenko, J.; Tassi Yunga, S.; Zheng, T.; Parra-Izquierdo, I.; et al. Phosphoproteomic quantitation and causal analysis reveal pathways in GPVI/ITAM-mediated platelet activation programs. Blood 2020, 136, 2346–2358. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kunapuli, S.P.; Tsygankov, A.Y. TULA-Family Regulators of Platelet Activation. Int. J. Mol. Sci. 2022, 23, 14910. https://doi.org/10.3390/ijms232314910
Kunapuli SP, Tsygankov AY. TULA-Family Regulators of Platelet Activation. International Journal of Molecular Sciences. 2022; 23(23):14910. https://doi.org/10.3390/ijms232314910
Chicago/Turabian StyleKunapuli, Satya P., and Alexander Y. Tsygankov. 2022. "TULA-Family Regulators of Platelet Activation" International Journal of Molecular Sciences 23, no. 23: 14910. https://doi.org/10.3390/ijms232314910
APA StyleKunapuli, S. P., & Tsygankov, A. Y. (2022). TULA-Family Regulators of Platelet Activation. International Journal of Molecular Sciences, 23(23), 14910. https://doi.org/10.3390/ijms232314910






