A Discovery-Based Metabolomic Approach Using UPLC-Q-TOF-MS/MS Reveals Potential Antimalarial Compounds Present in Artemisia annua L.
Abstract
:1. Introduction
2. Results
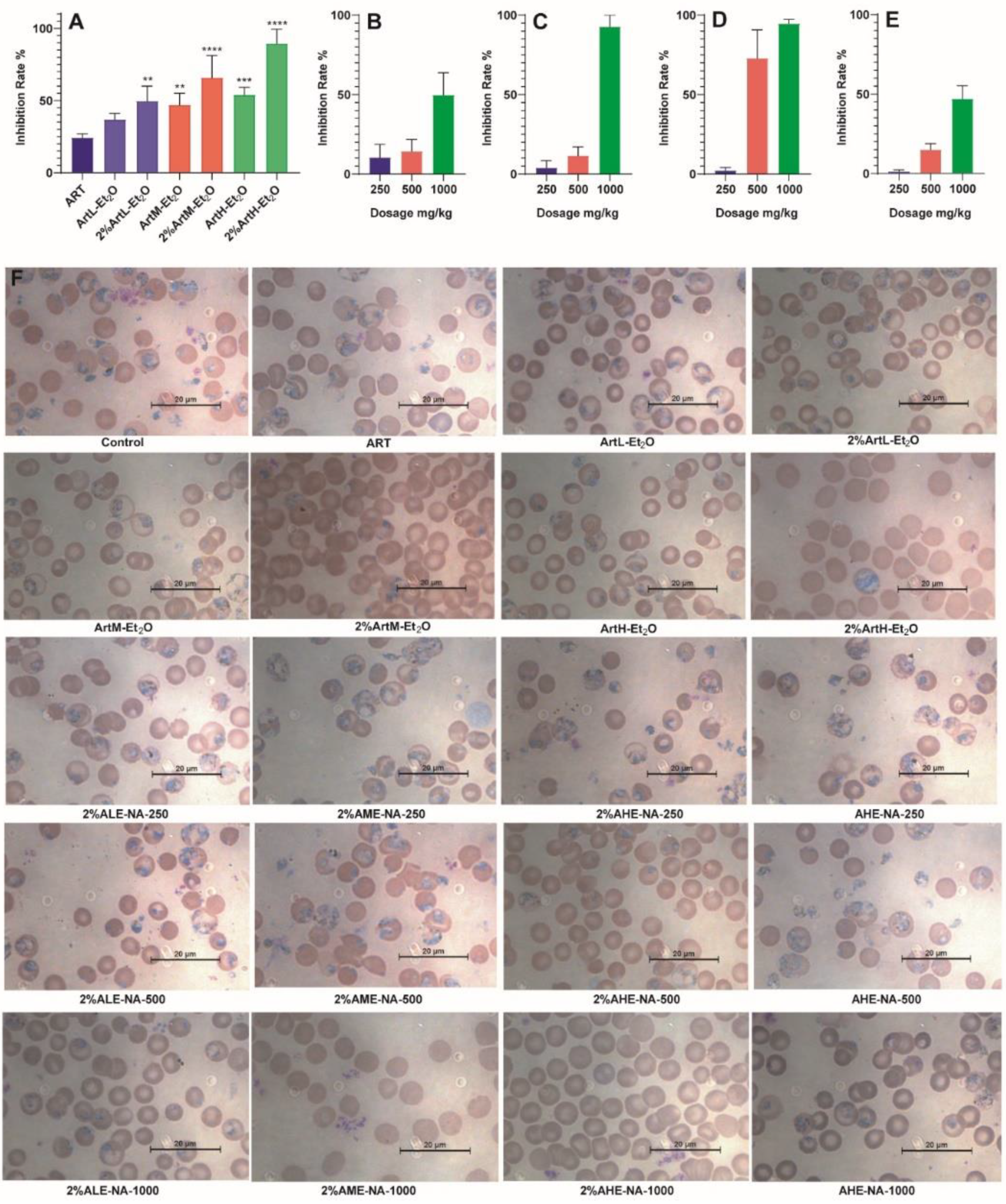
2.1. Antimalarial Activity of Different Extracts of Artemisia annua L.
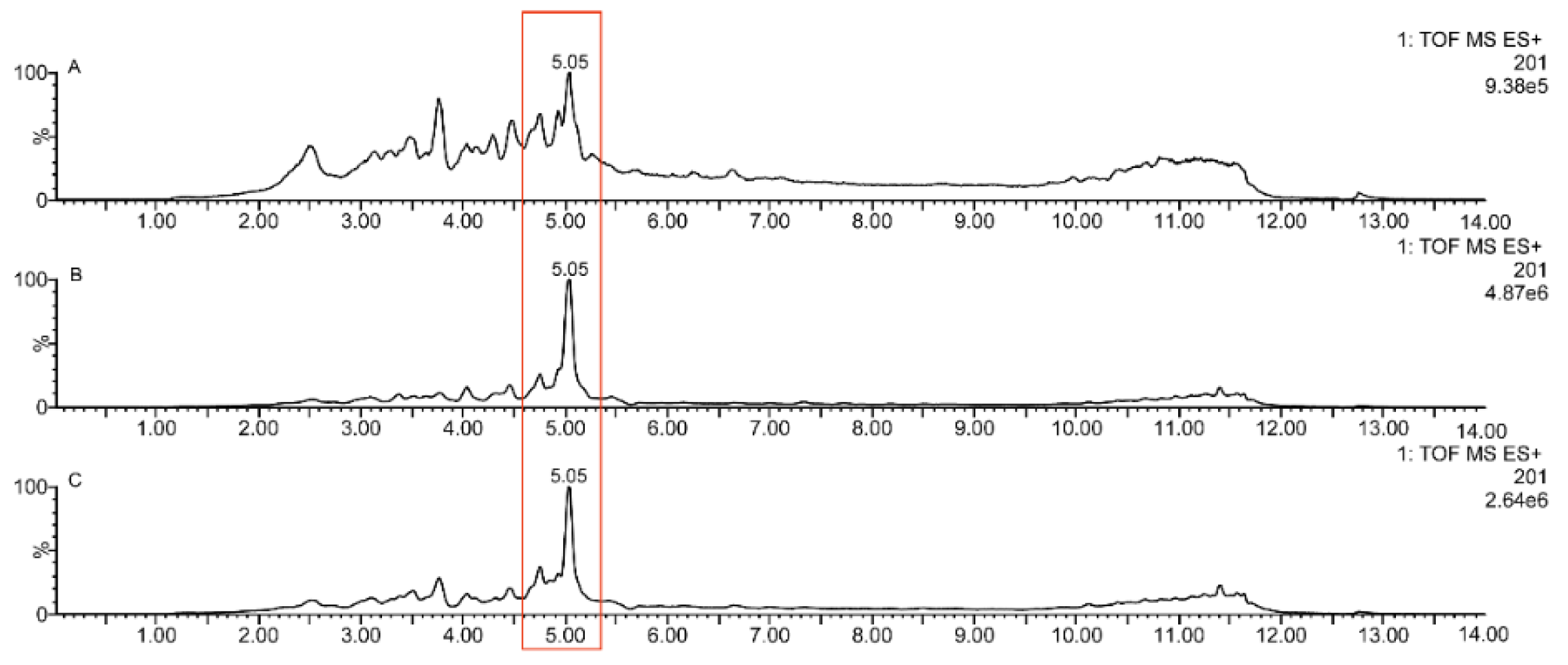
2.2. UPLC-Q-TOF-MSE Qualitative Analysis
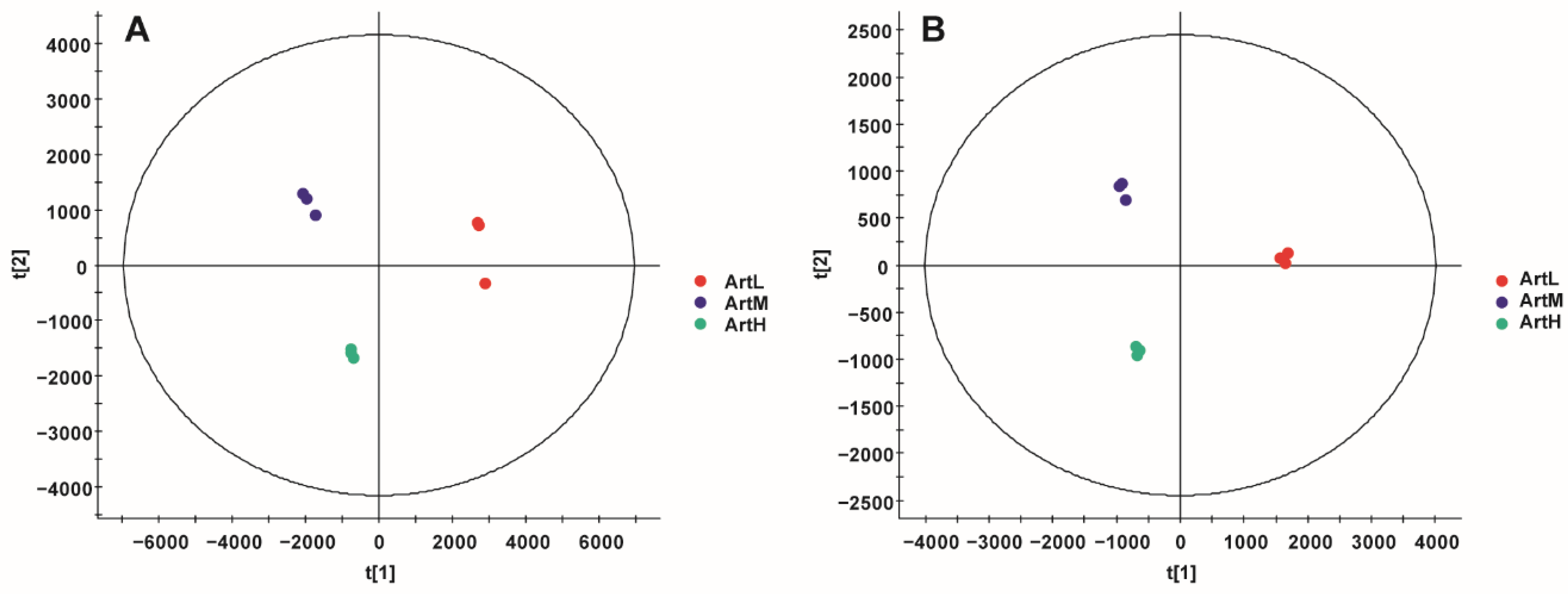
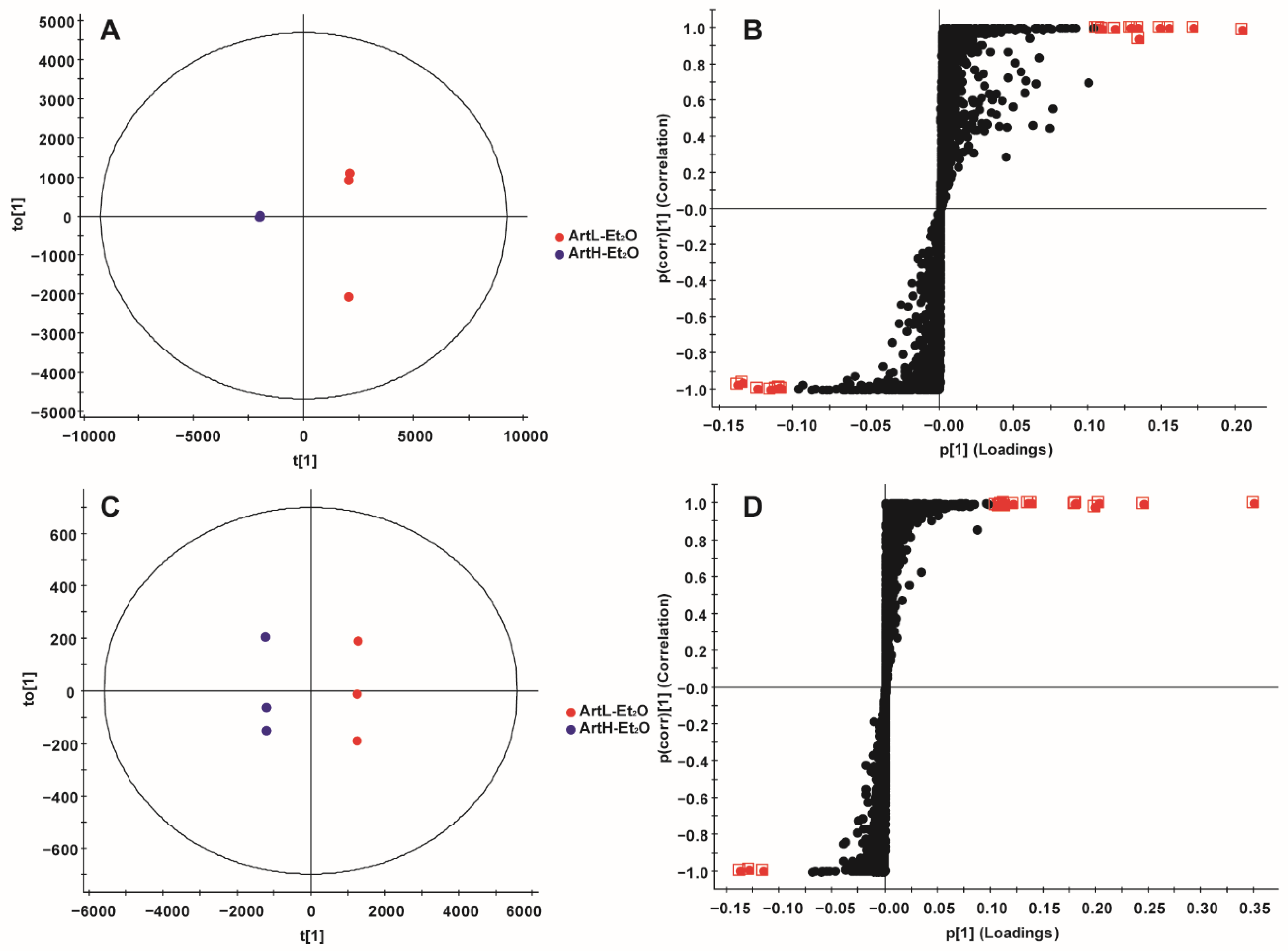
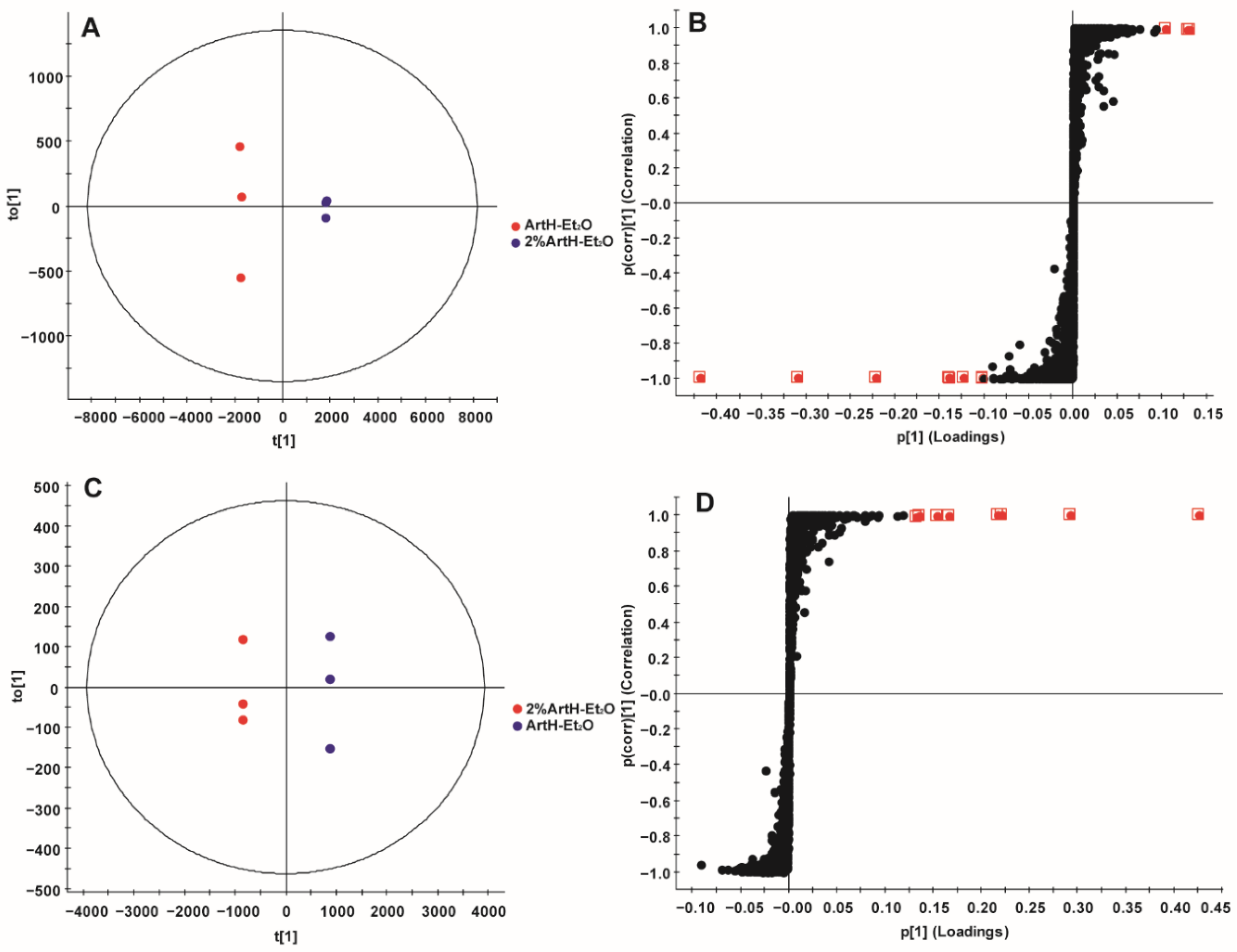
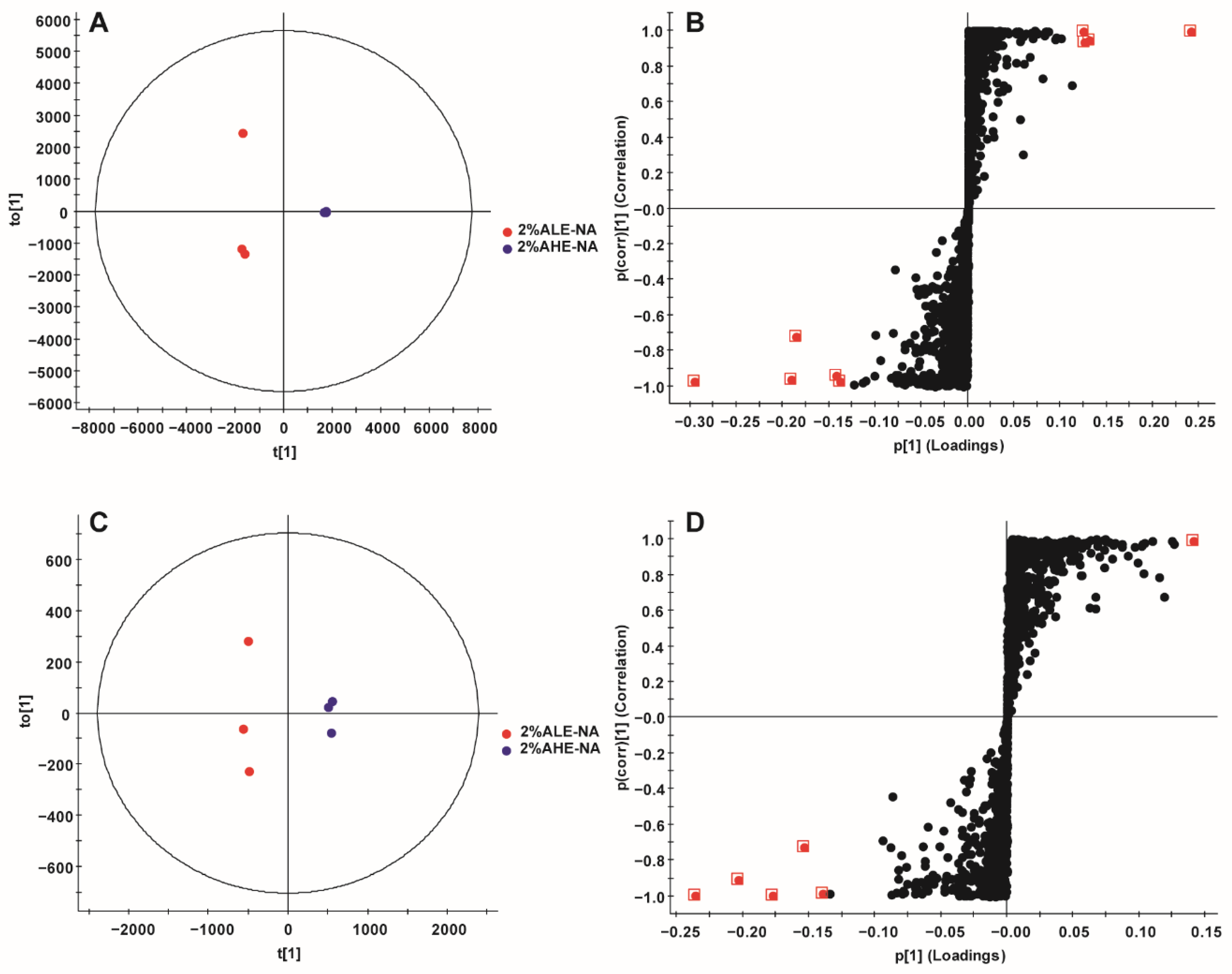
2.3. Multivariate Statistical Analysis
2.3.1. PCA
2.3.2. OPLS-DA
3. Materials and Methods
3.1. Plant Material
3.2. Chemicals
3.3. Sample Preparation
3.3.1. Ether Extracts
3.3.2. Ether Neutral Dry
3.3.3. Ether Neutral Dry without Artemisinin
3.4. Animal Experiment
3.5. UPLC-Q-TOF-MS Measurement
3.6. Data Analysis by UNIFI Platform
3.7. Multivariate Statistical Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACTs | Artemisinin-based combination therapies |
| ART | Artemisinin |
| ArtL(M/H) | Artemisia annua L. with low/medium/high content of artemisinin |
| ArtL(M/H)-Et2O | Ether extract of Artemisia annua L. with low/medium/high content of artemisinin |
| 2% ArtL(M/H)-Et2O | Neutral dry of ArtL(M/H)-Et2O |
| 2% AL(M/H)E-NA | Neutral dry of 2% ArtL(M/H)-Et2O without artemisinin |
| AHE-NA | ArtH-Et2O without artemisinin |
| PE | Petroleum ether |
| EA | Ethyl acetate |
References
- World Health Organization. Antimalarial Drug Combination Therapy: Report of a WHO Technical Consultation. 2001. Available online: https://apps.who.int/iris/handle/10665/66952 (accessed on 30 April 2022).
- World Health Organization. Guidelines for the Treatment of Malaria. WHO Guidelines for the Treatment of Malaria. 2006. Available online: europa.eu (accessed on 30 April 2022).
- World Health Organization. Guidelines for the Treatment of Malaria, 2nd ed.; WHO: Geneva, Switzerland, 2010.
- World Health Organization. Guidelines for the Treatment of Malaria, 3rd ed.; WHO: Geneva, Switzerland, 2015. Available online: 9789241549127_eng.pdf (accessed on 30 April 2022).
- World Health Organization. World Malaria Report; WHO: Geneva, Switzerland, 2015.
- World Health Organization. Global Malaria Programme: Artemisinin and Artemisinin-Based Combination Therapy Resistance; WHO: Geneva, Switzerland, 2017.
- World Health Organization. World Malaria Report; WHO: Geneva, Switzerland, 2021.
- Liu, J.M.; Ni, M.Y.; Fan, J.F.; Tu, Y.Y.; Wu, Z.H.; Wu, Y.L.; Zhou, W.S. Structure and Reaction of Arteanniun. Huaxue Xuebao 1979, 37, 129–143. [Google Scholar]
- Wang, M.Y. Tu Youyou and Artemisinins; People’s Medical Publishing House: Beijing, China, 2015. [Google Scholar]
- Gruessner, B.M.; Cornet-Vernet, L.; Desrosiers, M.R.; Lutgen, P.; Towler, M.J.; Weathers, P.J. It is not just artemisinin: Artemisia sp. for treating diseases including malaria and schistosomiasis. Phytochem. Rev. 2019, 18, 1509–1527. [Google Scholar] [CrossRef] [PubMed]
- Tu, Y.Y. From Artemisia annua L. to Artemisinins; Chapter III; Chemical Industry Press: Beijing, China, 2009. (In Chinese) [Google Scholar]
- Brown, G.D. Cadinanes from Artemisia annua that may be intermediates in the biosynthesis of artemisinin. Phytochemistry 1994, 36, 637–641. [Google Scholar] [CrossRef]
- Brown, G.D.; Liang, G.-Y.; Sy, L.-K. Terpenoids from the seeds of Artemisia annua. Phytochemistry 2003, 64, 303–323. [Google Scholar] [CrossRef] [PubMed]
- Hui, S.-M.; Ngo, K.-S.; Brown, G.D. Epimerization in acid degradation products of artemisinin. J. Chem. Soc. Perkin Trans. 1 1997, 22, 3435–3442. [Google Scholar] [CrossRef]
- Ma, C.; Wang, H.; Lu, X.; Xu, G.; Liu, B. Metabolic fingerprinting investigation of Artemisia annua L. in different stages of development by gas chromatography and gas chromatography—Mass spectrometry. J. Chromatogr. A 2008, 1186, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Sy, L.-K.; Brown, G.D.; Haynes, R. A novel endoperoxide and related sesquiterpenes from Artemisia annua which are possibly derived from allylic hydroperoxides. Tetrahedron 1998, 54, 4345–4356. [Google Scholar] [CrossRef]
- Sy, L.-K.; Brown, G.D. Three sesquiterpenes from Artemisia annua. Phytochemistry 1998, 48, 1207–1211. [Google Scholar] [CrossRef]
- Kamchonwongpaisan, S.; Meshnick, S.R. The mode of action of the antimalarial artemisinin and its derivatives. Gen. Pharmacol. Vasc. Syst. 1996, 27, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Ruan, J.; Liu, Z.; Qiu, F.; Shi, H.; Wang, M. Simultaneous Quantification of Five Sesquiterpene Components after Ultrasound Extraction in Artemisia annua L. by an Accurate and Rapid UPLC–PDA Assay †. Molecules 2019, 24, 1530. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dong, X.; Zhang, X.; Wang, M.; Gu, L.; Li, J.; Gong, M. Heparin-decorated nanostructured lipid carriers of artemether-protoporphyrin IX-transferrin combination for therapy of malaria. Int. J. Pharm. 2021, 605, 120813. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, C.; Gong, M.; Wang, M. Combination of artemisinin-based natural compounds from Artemisia annua L. for the treatment of malaria: Pharmacodynamic and pharmacokinetic studies. Phytother Res. 2018, 32, 1415–1420. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.D. The Biosynthesis of Artemisinin (Qinghaosu) and the Phytochemistry of Artemisia annua L. (Qinghao). Molecules 2010, 15, 7603–7698. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, C.; Qiu, F.; Li, J.; Wang, M.Y.; Gong, M.X. The effects of arteannuin B, arteannuic acid and scopoletinon phar-macokinetics of artemisinin in mice infected by Plasmodium yoelii. Global Tradit. Chin. Med. 2016, 9, 394–398. [Google Scholar] [CrossRef]
- Wei, S.; Ji, H.; Yang, B.; Ma, L.; Bei, Z.; Li, X.; Dang, H.; Yang, X.; Liu, C.; Wu, X.; et al. Impact of chrysosplenetin on the pharmacokinetics and anti-malarial efficacy of artemisinin against Plasmodium berghei as well as in vitro CYP450 enzymatic activities in rat liver microsome. Malar. J. 2015, 14, 432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, J.; Ji, H.Y.; Wei, S.J.; Fu, X.Y.; Liu, M.Y.; Wu, X.L. Determination of chrysosplenetin, metabolic inhibitor of artemisinin, in rat plasma by UPLC-MS/MS and study on its pharmacokinetics. China J. Chin. Mater. Med. 2013, 8, 3363–3367. [Google Scholar] [CrossRef]
| Group | Mean Content/% | RSD/% |
|---|---|---|
| ArtL-Et2O | 1.84 | 0.18 |
| 2% ArtL-Et2O | 0.62 | 0.29 |
| ArtM-Et2O | 13.60 | 2.68 |
| 2% ArtM-Et2O | 11.77 | 1.77 |
| ArtH-Et2O | 16.69 | 2.58 |
| 2% ArtH-Et2O | 10.23 | 3.93 |
| NO. | Component Name | Neutral Mass (Da) | Observed m/z | RT (min) | MS/MS |
|---|---|---|---|---|---|
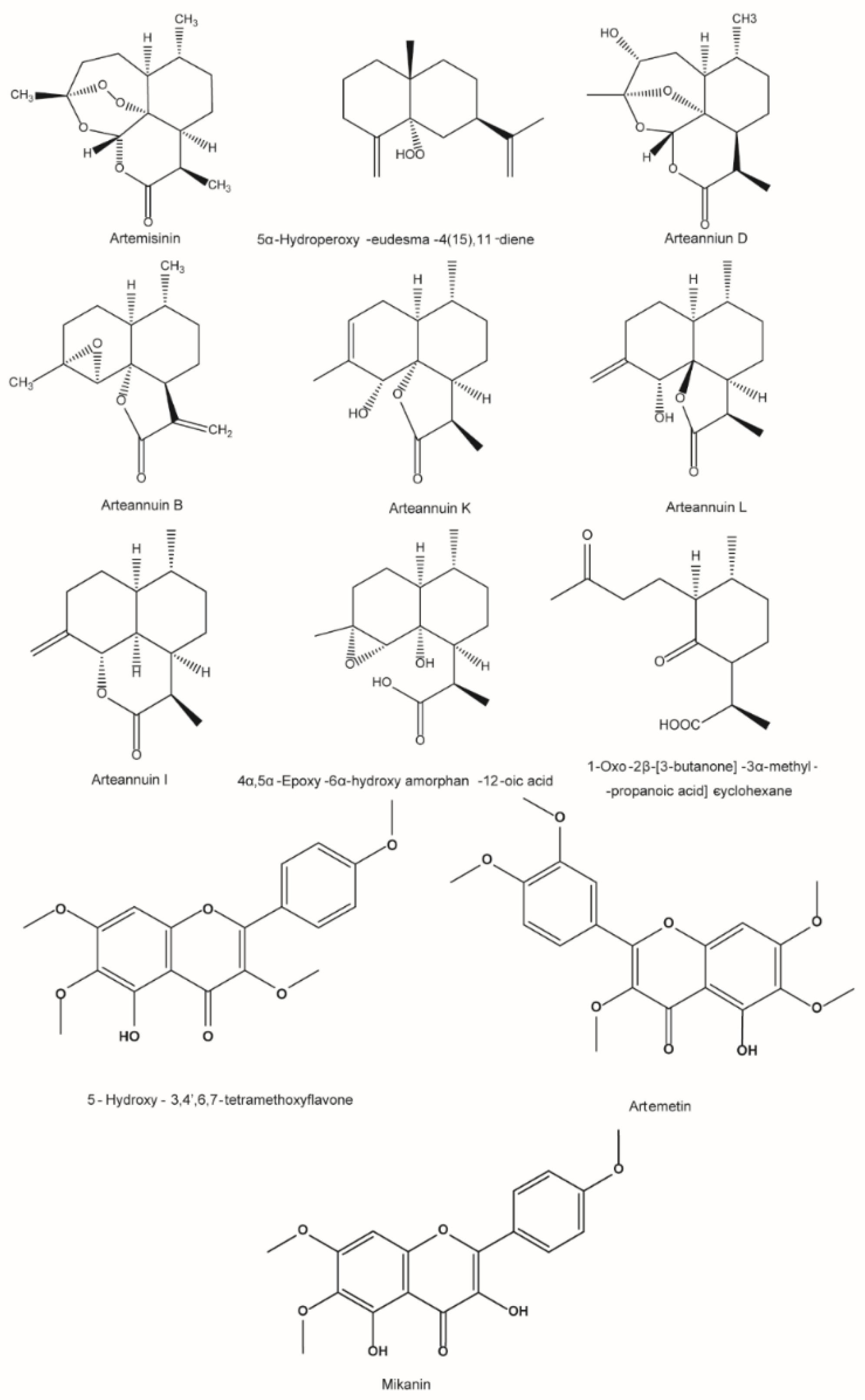
| 1 | Artenniun D | 282.1467 | 283.1655 | 2.70 | 265.1551, 247.1409, 219.1470 |
| 281.1600 * | 265.1625, 251.1826, 181.0640 | ||||
| 2 | Chrysospleno D | 360.0845 | 361.0916 | 3.07 | 345.0744, 303.0629 |
| 359.1078 * | 344.0792, 329.0523 | ||||
| 3 | Casticin | 374.1002 | 375.1073 | 3.90 | 360.1020, 345.0782, 317.0796 |
| 373.1233 * | 358.0995, 343.0695 | ||||
| 4 | 1-Oxo-2-[3-butanone]-3-methyl-6-[2-propanoic acid]-cyclohexane methyl ester | 268.1675 | 291.1691 | 4.26 | 219.1500, 191.1509, 173.1407, 145.1078 |
| 5 | Artenniun B | 248.1412 | 249.1589 | 4.47 | 231.1490, 185.1413 |
| 6 | Artemisinin | 282.1467 | 283.1655 | 5.10 | 265.1551, 247.1441, 237.1582, 229.1470, 219.1500 |
| 7 | Artenniun C | 266.1518 | 267.1716 | 5.24 | 249.1589, 231.1490, 207.1467 |
| 265.1659 * | 116.9371 | ||||
| 8 | Artenniun I | 234.1620 | 235.1800 | 7.21 | 217.1672, 189.1727, 161.1393 |
| 233.1733 * | 116.9371 | ||||
| 9 | Dihydroartemisinic acid | 236.1776 | 237.1960 | 8.53 | 219.1833, 201.1731, 163.1562 |
| 235.1883 * | |||||
| 10 | Artemisinic acid | 234.1620 | 235.1800 | 9.20 | 217.1672, 199.1551, 189.1727 |
| 233.1733 * |
| NO. | Component Name | Neutral Mass (Da) | Observed m/z | Mass Error (mDa) | RT (min) | MS/MS |
|---|---|---|---|---|---|---|
| Flavonoids | ||||||
| 1 | Patuletin-3-O-glucoside | 494.1060 | 495.1092 | −4.2 | 1.24 | 333.0584, 318.0336, 287.0535 |
| 2 | Astragalin | 448.1006 | 447.1125 * | 0.6 | 1.27 | 285.1470, 257.1145 |
| 3 | Quercetin | 302.0427 | 303.0488 | −1.1 | 2.18 | 287.0543 |
| 4 | 4H-1-Benzopyran-4-one, 5,7,8-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-3-methoxy- | 346.0689 | 347.0757 | −0.5 | 2.41 | 317.0650 |
| 5 | Patuletin | 332.0532 | 333.0591 | −1.4 | 2.22 | 318.0346, 287.0545 |
| 332.0532 | 331.0459 * | 0.0 | 2.25 | 285.0410 | ||
| 6 | Kaempferol | 286.0477 | 287.0541 | −0.9 | 2.67 | 153.0173, 229.0476 |
| 286.0477 | 285.0400 * | −0.5 | 2.73 | 151.0031 | ||
| 7 | Chrysoeriol | 300.0634 | 301.0699 | −0.8 | 2.70 | 286.0465, 258.0513, 257.0406 |
| 8 | Rhamnetin | 316.0583 | 317.0645 | −1.1 | 2.77 | 302.042, 270.0513 |
| 316.0583 | 315.0504 * | −0.7 | 2.82 | 299.01, 243.0306 | ||
| 9 | Axillarin | 346.0689 | 347.0753 | −0.8 | 2.84 | 289.1403 |
| 346.0689 | 345.0615 * | 0.0 | 2.86 | 314.0454, 299.0194 | ||
| 10 | 2,4′,5′-Trihydroxy-5′6,7-trimethoxyflavone | 360.0845 | 361.0916 | −0.2 | 3.07 | 345.0604, 328.0575 |
| 11 | Chrysosplenol D | 360.0845 | 361.0916 | −0.2 | 3.07 | 361.1007, 362.1008, 363.1022, 345.0730, 328.0613 |
| 12 | Eupatin | 360.0845 | 361.0913 | −0.5 | 3.62 | 331.0854, 291.1586, 233.1570 |
| 13 | 4H-1-Benzopyran-4-one, 2-(2,4-dihydroxyphenyl)-5-hydroxy-6,7-dimethoxy- | 330.0740 | 331.0805 | −0.7 | 3.63 | 331.0854, 316.0571 |
| 14 | Penduletin | 344.0896 | 345.0963 | −0.6 | 3.76 | 328.0634 |
| 344.0896 | 343.0824 * | 0.1 | 3.79 | |||
| 15 | Casticin | 374.1002 | 375.1073 | −0.2 | 3.90 | 377.1171, 376.1163, 375.1168 |
| 16 | Chrysosplenetin | 374.1002 | 397.0887 | −0.7 | 3.91 | 397.0939, 375.1168, 317.0657 |
| 17 | Quercetagetin-6,7,3′,4′-tetramethylether | 374.1002 | 375.1076 | 0.1 | 4.00 | |
| 18 | Cirsimaritin | 314.0790 | 315.0846 | −1.7 | 4.38 | 300.0607, 285.0376 |
| 19 | Mikanin | 344.0896 | 345.0967 | −0.2 | 4.53 | 330.0771, 271.1332, 205.1633 |
| 344.0896 | 343.0819 * | −0.4 | 4.57 | 328.0585, 313.0351, 285.0435 | ||
| 20 | Artemetin | 388.1158 | 389.1231 | 0.0 | 4.76 | 331.0806, 313.0702283.0598, 267.0644, 135.0436 |
| 21 | Rhamnocitrin | 300.0634 | 301.0705 | −0.2 | 5.64 | |
| 22 | 5-Hydroxy-3,4′,6,7-tetramethoxyflavone | 358.1053 | 359.1125 | 0.0 | 5.64 | 359.1201 |
| 23 | 4H-1-Benzopyran-4-one, 3-hydroxy-6,7-dimethoxy-2-(4-methoxyphenyl)- | 328.0947 | 329.1020 | 0.0 | 6.92 | 314.0840 |
| Sesquiterpenoids | ||||||
| 1 | Arteannuin M | 268.1675 | 291.1563 | −0.4 | 2.27 | 235.1312, 217.1199, 207.1371 |
| 2 | Artemisinin B | 266.1518 | 265.1445 * | −0.1 | 2.41 | 251.1292, 241.1448, 235.1341 |
| 3 | Artenniun D | 282.1467 | 281.1389 * | −0.5 | 2.61 | 219.1348, 191.1049, 172.8616 |
| 4 | Artemisin | 262.1205 | 263.1262 | −1.6 | 2.68 | 231.1366, 203.1422, 161.0951 |
| 5 | seco-Cadinane | 266.1518 | 311.1493 * | −0.7 | 2.71 | 237.1488, 219.1002, 175.1120 |
| 6 | Dihydroxycadinanolide | 266.1518 | 265.1447 * | 0.1 | 2.75 | 247.1341, 231.1396, 219.1021 |
| 7 | 1-Oxo-2β-[3-butanone]-3α-methyl-6β-[2-propanoic acid]-cyclohexane | 254.1518 | 277.1409 | −0.1 | 2.83 | 237.1582, 209.1525, 151.1111 |
| 254.1518 | 253.1445 * | 0.0 | 2.86 | 183.1025, 109.0670, 235.1340 | ||
| 8 | α-Hydroxysantonin | 262.1205 | 263.1213 | −6.5 | 3.16 | 157.0635, 107.0488 |
| 9 | Arteannuin L | 250.1569 | 273.1456 | −0.5 | 3.24 | 233.1528, 189.1261, 187.1474 |
| 10 | Dihydroartemisinic acid hydroperoxide | 268.1675 | 267.1600 * | −0.1 | 3.27 | 249.1496, 221.1547, 203.1440 |
| 11 | Arteannuin G | 282.1467 | 283.1504 | −3.6 | 3.39 | 233.1131, 163.0741, 125.0936 |
| 12 | 4α,5α-Epoxy-6α-hydroxy amorphan-12-oic acid | 268.1675 | 267.1597 * | −0.5 | 3.46 | 251.1648, 223.1343, 205.1593 |
| 13 | Norannuic acid formyl ester 15-nor-10-Hydroxy-oplopan-4-oic acid | 240.1725 | 285.1708 * | 0.1 | 3.65 | 223.1343, 177.0919, 211.1345 |
| 14 | Arteannuin K | 250.1569 | 249.1487 * | −0.9 | 3.80 | 231.1391, 217.1231, 202.0994 |
| 15 | 4-Amorphene-3,7-diol (3α,7α), acetate- | 280.2038 | 303.1906 | −2.5 | 4.04 | 249.1478, 231.1362, 203.1417 |
| 16 | 4-Amorphene-3,7-diol (3α,7α) | 238.1933 | 261.1807 | −1.8 | 4.10 | 209.1517, 202.1641, 175.1106 |
| 17 | 1-Oxo-2-[3-butanone]-3-methyl-6-[2-propanoic acid]-cyclohexane methyl ester | 268.1675 | 267.1599 * | −0.3 | 4.17 | 253.1449, 235.1339, 183.1024 |
| 18 | 4-Amorphen,3,11-diol | 238.1933 | 261.1825 | 0.0 | 4.22 | 187.1473, 159.1164, 129.0690 |
| 19 | α-Epoxy-dihydroartemisinic | 252.1725 | 251.1351 * | −0.2 | 4.26 | 237.1491, 205.1596, 191.1440 |
| 20 | Arteannuin E | 250.1569 | 251.1634 | −0.8 | 4.29 | 233.1510, 215.1402, 191.1041 |
| 21 | Artenniun B | 248.1412 | 271.1300 | −0.4 | 4.47 | 205.1581 |
| 22 | 3α,15-Dihydroxy cedrane | 238.1933 | 261.1831 | 0.6 | 4.60 | 209.1516, 175.1107, 147.1162 |
| 23 | Unknown | 232.1463 | 233.1531 | −0.6 | 4.83 | 215.1416, 187.1472, 145.1004 |
| 24 | Arteannuin F | 250.1569 | 249.1488 * | −0.8 | 4.84 | 235.1340, 231.1385, 209.1179 |
| 25 | Abscisic acid | 264.1362 | 265.1433 | −0.2 | 4.85 | |
| 264.1362 | 263.1288 * | −0.1 | 4.85 | |||
| 26 | 5α-Hydroperoxy-eudesma-4(15),11-diene | 236.1776 | 237.1842 | −0.7 | 5.05 | 221.1514, 219.1833, 201.1731 |
| 27 | Cubenol | 222.1984 | 261.1658 | 4.3 | 5.11 | 145.1007, 128.0618, 105.0697 |
| 28 | Artemisinin | 282.1467 | 281.1395 * | 0.0 | 5.17 | 265.1451, 253.1448, 219.1389 |
| 29 | Arteanniun A | 206.1307 | 207.1375 | −0.5 | 5.24 | 193.1578, 161.1315, 133.1007 |
| 30 | β-copaen-4α-ol | 234.1984 | 273.1659 | 4.3 | 5.25 | 203.1420, 185.1314, 159.1162 |
| 31 | 3α-Hydroxy-4α,5α-epoxy-7-oxo-(8[7→6]-abeo-amorphane | 238.1569 | 239.1632 | −1.0 | 5.26 | 203.1421, 179.1056, 133.1007 |
| 238.1569 | 283.1549 * | −0.2 | 5.16 | 219.1389, 183.1025, 149.0969 | ||
| 32 | Artenniun C | 266.1518 | 265.1444 * | −0.1 | 5.30 | 247.134, 233.1175, 203.1435 |
| 33 | α-Epoxyartemisinic acid | 250.1569 | 249.1494 * | −0.3 | 5.35 | 205.1596 |
| 34 | artemisitene | 280.1311 | 281.1353 | −3.1 | 5.44 | 247.1314, 209.1523 |
| 35 | Norannuic acid | 224.1412 | 247.1321 | 1.6 | 5.79 | 209.1525, 163.1107, 131.0848 |
| 36 | Arteannuin H | 250.1569 | 273.1436 | −2.5 | 6.31 | 203.1415, 189.1258, 147.1158 |
| 37 | Nortaylorione | 220.1463 | 221.1526 | −1.0 | 6.62 | 163.1105, 139.1106, 135.1156 |
| 38 | Deoxyarteannuin B | 232.1463 | 233.1531 | −0.6 | 6.69 | |
| 39 | Annulide | 232.1463 | 233.1531 | −0.6 | 7.00 | 215.1514, 187.1572 |
| 40 | Spathulenol | 220.1827 | 221.1887 | −1.3 | 7.12 | 203.1789, 175.1476, 123.1156 |
| 41 | Artenniun I | 234.1620 | 233.1548 * | 0.1 | 7.22 | 176.0819 |
| 42 | Dihydro-deoxyarteannuin B | 234.1620 | 235.1689 | −0.4 | 7.26 | 189.1628, 161.1322 |
| 43 | Arteannuin J | 234.1620 | 235.1690 | −0.3 | 7.61 | 217.1577, 189.1625, 161.1320 |
| 44 | Dihydroartemisinic acid | 236.1776 | 259.1662 | −0.7 | 7.63 | 219.1736, 189.1625, 161.1320 |
| 45 | 3-Isobutylcadin-4-en-11-ol | 308.2351 | 307.2275 * | −0.4 | 7.70 | 277.2167, 251.1658, 195.1385 |
| 46 | Nootkatone | 218.1671 | 219.1736 | −0.7 | 8.51 | 203.1415, 163.1103, 123.0791 |
| 47 | trans-α-bergamotyl acetic anhydride | 276.1725 | 277.1785 | −1.3 | 8.81 | 233.1520, 205.1572, 235.1676 |
| 48 | Elemyl acetate | 264.2089 | 265.2144 | −1.8 | 9.06 | 233.1522, 205.1572, 191.1410 |
| 49 | Artemisinic acid | 234.1620 | 235.1675 | −1.8 | 9.19 | 175.1103 |
| 50 | Artemisinic aldehyde | 218.1671 | 219.1732 | −1.1 | 9.34 | 202.1265 |
| 51 | Artemisinic acid methyl ester | 248.1776 | 293.1794 * | 3.5 | 9.99 | 221.1545, 177.1279 |
| 52 | Occidentalolacetate | 246.1984 | 285.1657 | 4.2 | 10.04 | 189.1627, 163.1109 |
| 53 | α-Aromadendrene | 204.1878 | 205.1941 | −1.0 | 10.15 | |
| 54 | 6,7-dehydroartemisinic acid | 232.1463 | 233.1531 | −0.5 | 10.25 | 205.1576, 187.1472, 175.1102 |
| 55 | 14-Hydroxy-α-humulene | 262.1933 | 307.1920 * | 0.5 | 10.28 | 221.1549, 191.1069, 151.1126 |
| 56 | 4(15),5,11-Cadinatriene | 202.1722 | 203.1789 | −0.5 | 10.32 | 177.1627, 175.1468, 173.1313 |
| 57 | 14-Hydroxy-δ-cadinene | 220.1827 | 221.1890 | −1.0 | 10.32 | 203.1788, 173.1316, 161.1316 |
| 58 | α-Longipinene | 204.1878 | 205.1963 | 1.2 | 10.33 | 191.1799, 175.1473, 161.1316 |
| 59 | β-Longipinene | 204.1878 | 205.1945 | −0.6 | 10.57 | 187.1470, 158.1039 |
| 60 | cis-Calamenene | 202.1722 | 203.1789 | −0.5 | 10.71 | 173.0881, 145.1004, 115.0537 |
| 61 | α-Cubebene | 204.1878 | 205.1945 | −0.6 | 10.99 | 159.11651, 145.10085, 131.0852 |
| 62 | Cedrylacetate | 264.2089 | 287.1962 | −1.9 | 11.11 | 109.0644 |
| 63 | Cedra-8(15)-en-9α-ol acetate | 262.1933 | 285.1824 | −0.1 | 11.30 | 231.1364, 219.1331, 173.1317 |
| 64 | Guaiazulene | 198.1409 | 199.1462 | −1.9 | 11.40 | 185.1308, 143.0842, 128.0613 |
| 65 | α-Ylangene | 204.1878 | 227.1760 | −1.1 | 11.41 | 161.1318, 147.1163, 129.0689 |
| 66 | Pregeijerene | 162.1409 | 185.1310 | 1.0 | 11.47 | 131.0846, 119.08495, 105.0694 |
| 67 | Cedra-8-en-13-ol, acetate | 248.1776 | 271.1640 | −2.8 | 11.54 | 189.1250, 159.1163, 157.1003 |
| 68 | 1β-Hydroxy-4(15),5-eudesmadiene | 220.1827 | 221.1901 | 0.1 | 11.73 | 207.1731, 180.1464, 146.0806 |
| Others | ||||||
| 1 | 3-Cyclohexene-1-methanol 2-hydroxy-α,α,4-trimethyl-, 1-acetate | 212.1412 | 235.1313 | 0.9 | 5.11 | 105.0697 |
| 2 | trans-5-Hydroxy-2-isopropenyl-5-methylhex-3-en-1-ol | 170.1307 | 193.1204 | 0.5 | 5.26 | 157.1072, 121.1004, 107.0849 |
| 3 | 2,6-Octadien-1-ol, 2,6-dimethyl-8-[(tetrahydro-2H-pyran-2-yl)oxy]- | 254.1882 | 253.1810 * | 0.0 | 8.71 | 235.1700, 219.1872, 205.1598 |
| 4 | 1,10-Oxy-α-myrcene hydroxide | 168.1150 | 191.1039 | −0.3 | 10.25 | 119.0850, 105.0695 |
| 5 | 2-Cyclohexen-1-ol, 3-methyl-6-(1-methylethylidene)- | 152.1201 | 175.1112 | 1.9 | 10.25 | 135.1221, 119.0850, 109.1001 |
| 6 | 2,6-Dimethyl-1,3,5,7-octatetraene | 134.1096 | 135.1155 | −1.3 | 10.27 | 107.0850, 105.0695 |
| 7 | 2-Butenoic acid, 3-methyl-,(1S,2R,4S)-1,7,7-trimethylbicyclo [2.2.1] hept-2-yl ester | 236.1776 | 235.1709 * | 0.5 | 10.30 | 221.1547, 179.1078, 137.0968 |
| 8 | p-Menth-3-ene | 138.1409 | 161.1319 | 1.8 | 10.34 | 123.1158 |
| 9 | Menthol | 156.1514 | 195.1143 | −0.3 | 10.75 | 143.0886, 135.1221, 109.1006 |
| 10 | Borneol isobutyrate | 224.1776 | 247.1683 | 1.5 | 11.11 | 139.0745, 123.0778, 123.0422 |
| 11 | Myrtenal | 150.1045 | 173.0941 | 0.4 | 11.16 | 121.1004, 105.0696 |
| 12 | Stigmasterol | 412.3705 | 413.3770 | −0.8 | 11.32 | 217.1570, 199.1466, 147.1159, 128.0613 |
| 13 | 2-Cyclohexen-1-one, 2-methyl-5-(1-methylcyclopropyl)- | 164.1201 | 187.1091 | −0.2 | 11.33 | |
| 14 | Lavandulylacetate | 196.1463 | 219.1333 | −2.2 | 11.37 | 122.0793 |
| 15 | Sabinene | 136.1252 | 159.1158 | 1.4 | 11.39 | 105.0694 |
| 16 | β-Amyrin acetate | 468.3967 | 491.3903 | 4.4 | 11.44 | 437.3354, 201.1628, 133.1008, 119.0854 |
| 17 | Bornylacetate | 196.1463 | 219.1344 | −1.1 | 11.55 | |
| 18 | Carvone | 150.1045 | 173.0938 | 0.1 | 11.57 | 135.1198, 119.0909 |
| 19 | Fenchol | 154.1358 | 177.1250 | 0.0 | 11.62 | 137.1312, 122.0808 |
| 20 | (2E)-Hexadecene | 280.3130 | 303.3021 | −0.1 | 11.86 | 163.1466, 135.1158, 121.1004, 107.0851 |
| 21 | Taraxasterone | 424.3705 | 425.3740 | −3.8 | 12.84 | 409.3410, 201.1623, 161.1314, 133.1003 |
| NO. | Primary ID | Neutral Mass (Da) | m/z | Rt (min) | Identification | VIP [1 + 1 + 0] |
|---|---|---|---|---|---|---|
| Positive ion mode | ||||||
| 1 | 7.26_178.1355n | 178.1355 | 161.1322 | 7.26 | Arteanniun I | 6.80 |
| 2 | 7.26_234.1612n | 234.1612 | 235.1684 | 7.26 | Arteanniun I | 6.67 |
| 3 | 5.11_282.1456n | 282.1456 | 265.1427 | 5.11 | Artemisinin | 6.09 |
| 4 | 2.85_276.1329n | 254.15181 | 277.1402 | 2.85 | 1-Oxo-2β-[3-butanone]-3α-methyl-6β-[2-propanoic acid]-cyclohexane | 5.64 |
| 5 | 5.10_246.1244n | 246.1244 | 247.1322 | 5.10 | Artemisinin | 5.44 |
| 6 | 4.53_345.0959m/z | 345.0959 | 4.53 | Mikanin | 5.34 | |
| 7 | 5.10_236.1408n | 236.1408 | 219.1375 | 5.10 | Artemisinin | 5.27 |
| Negative ion mode | ||||||
| 8 | 2.55_283.1527m/z | 283.1527 | 2.55 | Arteanniun D | 7.54 | |
| 9 | 3.44_267.1582m/z | 267.1582 | 3.44 | 4α,5α-Epoxy-6α-hydroxy amorphan-12-oic acid | 7.11 | |
| 10 | 2.88_254.1498n | 254.1498 | 253.1435 | 2.88 | 1-Oxo-2β-[3-butanone]-3α-methyl-6β-[2-propanoic acid]-cyclohexane | 6.35 |
| NO. | Primary ID | Neutral Mass (Da) | m/z | Rt/min | Identification | VIP [1 + 1 + 0] |
|---|---|---|---|---|---|---|
| 1 | 7.26_234.1610n | 234.1610 | 235.1682 | 7.26 | Arteannuin I | 6.76 |
| 2 | 7.26_178.1353n | 178.1353 | 161.1321 | 7.26 | 6.67 | |
| 3 | 3.86_250.1558n | 250.1558 | 251.1631 | 3.86 | Arteannuin K | 5.35 |
| NO. | Primary ID | Neutral Mass (Da) | m/z | RT (min) | Identification | VIP [1 + 1 + 0] |
|---|---|---|---|---|---|---|
| 1 | 5.64_358.1045n | 358.1045 | 359.1117 | 5.64 | 5-Hydroxy-3,4′,6,7- tetramethoxyflavone | 10.27 |
| 2 | 3.25_232.1452n | 233.1525 | 3.25 | Arteannuin L | 5.50 | |
| 3 | 3.25_187.1471 m/z | 187.1471 | 3.25 | 5.71 | ||
| 4 | 4.75_388.1152n | 388.1152 | 389.1225 | 4.75 | Artemetin | 5.34 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, H.; Wang, Z.; Xu, F.; Li, J.; Li, J.; Wang, M. A Discovery-Based Metabolomic Approach Using UPLC-Q-TOF-MS/MS Reveals Potential Antimalarial Compounds Present in Artemisia annua L. Int. J. Mol. Sci. 2022, 23, 14903. https://doi.org/10.3390/ijms232314903
Shi H, Wang Z, Xu F, Li J, Li J, Wang M. A Discovery-Based Metabolomic Approach Using UPLC-Q-TOF-MS/MS Reveals Potential Antimalarial Compounds Present in Artemisia annua L. International Journal of Molecular Sciences. 2022; 23(23):14903. https://doi.org/10.3390/ijms232314903
Chicago/Turabian StyleShi, Henan, Zhuqing Wang, Fujie Xu, Jialin Li, Jing Li, and Manyuan Wang. 2022. "A Discovery-Based Metabolomic Approach Using UPLC-Q-TOF-MS/MS Reveals Potential Antimalarial Compounds Present in Artemisia annua L." International Journal of Molecular Sciences 23, no. 23: 14903. https://doi.org/10.3390/ijms232314903




