Interactions of the Fungal Community in the Complex Patho-System of Esca, a Grapevine Trunk Disease
Abstract
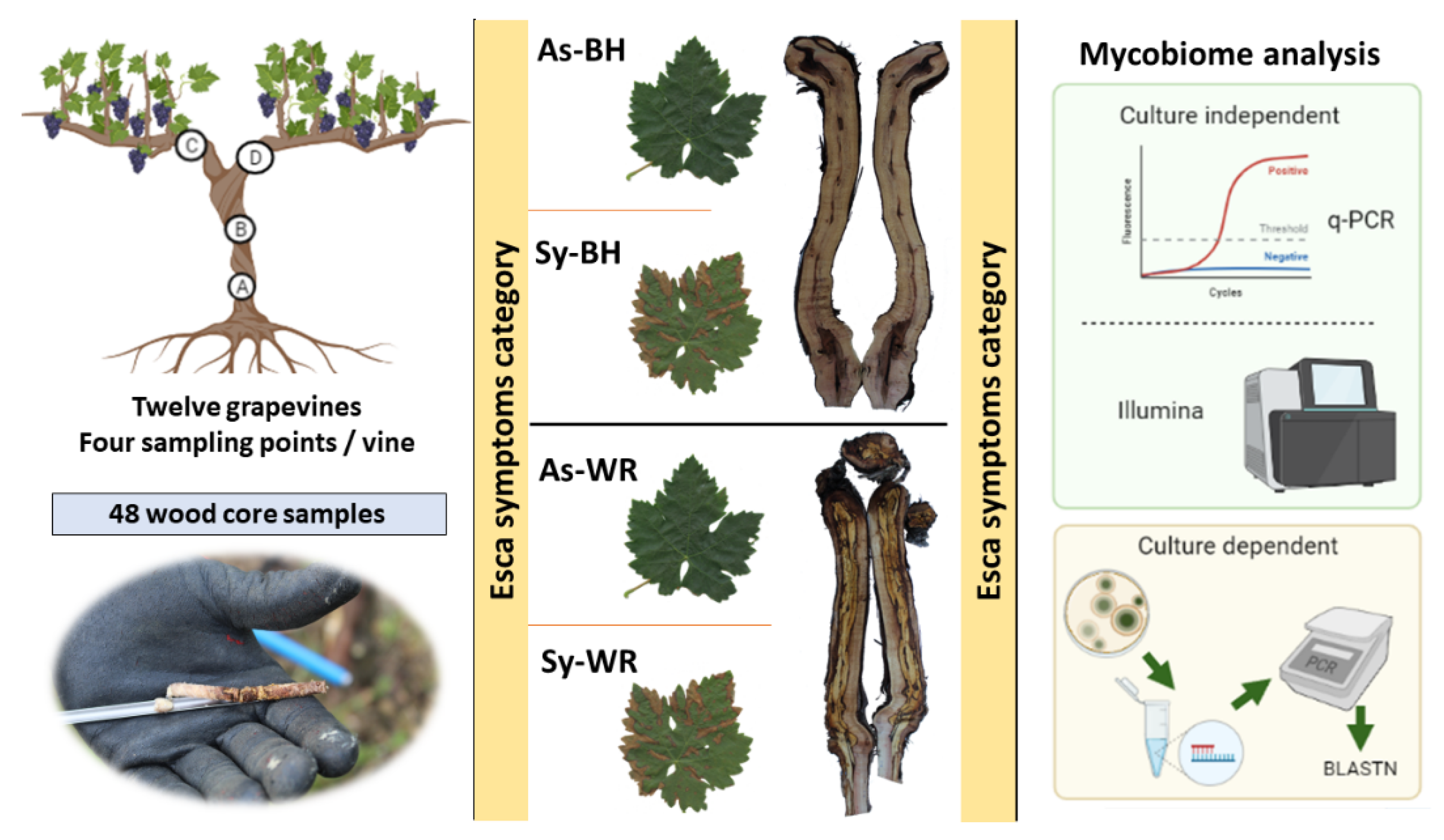
:1. Introduction
- (i)
- To elucidate differences in the fungal community inhabiting little degraded (healthy + black necrosis) or highly degraded (white rot) wood of grapevines showing esca foliar symptoms (symptomatic, Sy) or not (asymptomatic, As).
- (ii)
- To determine the most informative point for detection of fungal infection within a vine. To this end, a comparison of the fungal population at four different parts (above the graft, A, strain cross, B, and right arm, C and left arm, D) of the vine was completed.
- (iii)
- To compare both culture-dependent and culture-independent methods to identify and quantify the fungal species which colonized the grapevine wood. We characterized the wood mycobiome composition by ITS1 sequencing on an Illumina MiSeq instrument. Moreover, quantification of the fungus Phaeomoniella chlamydospora was achieved by using TaqMan qPCR. Molecular results were compared with the cultivable community of fungi to establish the most efficient methodology.
2. Results
2.1. Culture-Dependent Microbiological Analysis
2.2. Culture-Independent Fungal Community Analysis
2.2.1. Illumina® NGS (Next Generation Sequencing) Results
2.2.2. Results of P. chlamydospora Quantification by q-PCR
2.3. Biological Conclusions
3. Discussion
3.1. Comparison of Both Culture-Dependent and Culture-Independent Methods
3.2. Pathogenetic Role of P. chlamydospora and Botryosphaeriaceae, Fungi Associated to GTDs
3.3. Basidiomycota and Hymenochataceae Family as Key of Pathotrophs
3.4. New Insights on Biocontrol and Microbiome Interaction
4. Materials and Methods
4.1. Vineyard
4.2. Wood Sampling
4.3. Culture-Dependent Microbiological Analysis
4.4. Culture-Independent Fungal Community Analysis (qPCR and Illumina Technology)
4.4.1. DNA Extraction
4.4.2. Amplification, Library, Illumina Sequencing and Bioinformatics Processing
4.4.3. Quantification of the Esca Causal Agent Phaeomoniella chlamydospora by qPCR
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- OIV. 2020 Wine Production. OIV First Estimates. 2020. Available online: https://www.oiv.int/public/medias/7541/en-oiv-2020-world-wine-production-first-estimates.pdf (accessed on 1 September 2022).
- Cobos, R.; Ibañez, A.; Diez-Galán, A.; Calvo-Peña, C.; Ghoreshizadeh, S.; Coque, J.J.R. The Grapevine Microbiome to the Rescue: Implications for the Biocontrol of Trunk Diseases. Plants 2022, 11, 840. [Google Scholar] [CrossRef] [PubMed]
- Gramaje, D.; Úrbez-Torres, J.R.; Sosnowski, M.R.; Urbez-Torres, J.R.; Sosnowski, M.R. Managing Grapevine Trunk Diseases with Respect to Etiology and Epidemiology: Current Strategies and Future Prospects. Plant Dis. 2018, 102, 12–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hofstetter, V.; Buyck, B.; Croll, D.; Viret, O.; Couloux, A.; Gindro, K. What if esca disease of grapevine were not a fungal disease? Fungal Divers. 2012, 54, 51–67. [Google Scholar] [CrossRef] [Green Version]
- Bertsch, C.; Ramírez-Suero, M.; Magnin-Robert, M.; Larignon, P.; Chong, J.; Abou-Mansour, E.; Spagnolo, A.; Clément, C.; Fontaine, F.; Clé, C.; et al. Grapevine trunk diseases: Complex and still poorly understood. Plant Pathol. 2013, 62, 243–265. [Google Scholar] [CrossRef] [Green Version]
- Mugnai, L.; Graniti, A.; Surico, G. Esca (black measles) and brown wood-streaking: Two old and elusive diseases of grapevines. Plant Dis. 1999, 83, 404–418. [Google Scholar] [CrossRef] [Green Version]
- Surico, G. Towards a redefinition of the diseases within the esca complex of grapevine. Phytopathol. Mediterr. 2009, 48, 5–10. [Google Scholar] [CrossRef]
- Del Frari, G.; Oliveira, H.; Boavida Ferreira, R. White Rot Fungi (Hymenochaetales) and Esca of Grapevine: Insights from Recent Microbiome Studies. J. Fungi 2021, 7, 770. [Google Scholar] [CrossRef]
- Mondello, V.; Songy, A.; Battiston, E.; Pinto, C.; Coppin, C.; Trotel-Aziz, P.; Clément, C.; Mugnai, L.; Fontaine, F. Grapevine trunk diseases: A review of fifteen years of trials for their control with chemicals and biocontrol agents. Plant Dis. 2018, 102, 1189–1217. [Google Scholar] [CrossRef] [Green Version]
- Pinto, C.; Pinho, D.; Sousa, S.; Pinheiro, M.; Egas, C.; Gomes, A.C. Unravelling the diversity of grapevine microbiome. PLoS ONE 2014, 9, e85622. [Google Scholar] [CrossRef] [Green Version]
- Dissanayake, A.J.; Purahong, W.; Wubet, T.; Hyde, K.D.; Zhang, W.; Xu, H.; Zhang, G.; Fu, C.; Liu, M.; Xing, Q.; et al. Direct comparison of culture-dependent and culture-independent molecular approaches reveal the diversity of fungal endophytic communities in stems of grapevine (Vitis vinifera). Fungal Divers. 2018, 90, 85–107. [Google Scholar] [CrossRef]
- Jayawardena, R.S.; Purahong, W.; Zhang, W.; Wubet, T.; Li, X.; Liu, M.; Zhao, W.; Hyde, K.D.; Liu, J.; Yan, J. Biodiversity of fungi on Vitis vinifera L. revealed by traditional and high-resolution culture-independent approaches. Fungal Divers. 2018, 90, 1–84. [Google Scholar] [CrossRef] [Green Version]
- Ortiz-Álvarez, R.; Ortega-Arranz, H.; Ontiveros, V.J.; de Celis, M.; Ravarani, C.; Acedo, A.; Belda, I. Network properties of local fungal communities reveal the anthropogenic disturbance consequences of farming practices in vineyard soils. mSystems 2021, 6, e00344-21. [Google Scholar] [CrossRef] [PubMed]
- Bruez, E.; Vallance, J.; Gerbore, J.; Lecomte, P.; Da Costa, J.-P.; Guérin-Dubrana, L.; Rey, P. Analyses of the Temporal Dynamics of Fungal Communities Colonizing the Healthy Wood Tissues of Esca Leaf-Symptomatic and Asymptomatic Vines. PLoS ONE 2014, 9, e95928. [Google Scholar] [CrossRef] [PubMed]
- Del Frari, G.; Gobbi, A.; Aggerbeck, M.R.; Oliveira, H.; Hansen, L.H.; Ferreira, R.B. Characterization of the Wood Mycobiome of Vitis vinifera in a Vineyard Affected by Esca. Spatial Distribution of Fungal Communities and Their Putative Relation with Leaf Symptoms. Front. Plant Sci. 2019, 10, 910. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Põlme, S.; Abarenkov, K.; Henrik Nilsson, R.; Lindahl, B.D.; Clemmensen, K.E.; Kauserud, H.; Nguyen, N.; Kjøller, R.; Bates, S.T.; Baldrian, P.; et al. Fungal Traits: A user-friendly traits database of fungi and fungus-like stramenopiles. Fungal Divers. 2020, 105, 1–16. [Google Scholar] [CrossRef]
- Nguyen, N.H.; Song, Z.; Bates, S.T.; Branco, S.; Tedersoo, L.; Menke, J.; Schilling, J.S.; Kennedy, P.G. FUNGuild: An open annotation tool for parsing fungal community datasets by ecological guild. Fungal Ecol. 2016, 20, 241–248. [Google Scholar] [CrossRef]
- González, V.; Tello, M.L. The endophytic mycota associated with Vitis vinifera in central Spain. Fungal Divers. 2011, 47, 29–42. [Google Scholar] [CrossRef]
- Martín, M.T.; Cobos, R.; Martín, L.; López-Enríquez, L. Real-Time PCR Detection of Phaeomoniella chlamydospora and Phaeoacremonium aleophilum. Appl. Environ. Microbiol. 2012, 78, 3985–3991. [Google Scholar] [CrossRef] [Green Version]
- Mostert, L.; Groenewald, J.Z.; Summerbell, R.C.; Gams, W.; Crous, P.W. Taxonomy and Pathology of Togninia (Diaporthales) and its Phaeoacremonium Anamorphs. Stud. Mycol. 2006, 54, 1–113. [Google Scholar] [CrossRef] [Green Version]
- Martín, L.; Martín, M.T. Multi-gene sequence analysis and phenotypic diversity of Phaeoacremonium species isolated from grapevines in Spain. Eur. J. Plant Pathol. 2013, 137, 343–361. [Google Scholar] [CrossRef]
- Phillips, A.J.L.; Alves, A.; Abdollahzadeh, J.; Slippers, B.; Wingfield, M.J.; Groenewald, J.Z.; Crous, P.W. The Botryosphaeriaceae: Genera and species known from culture. Stud. Mycol. 2013, 76, 51–167. [Google Scholar] [CrossRef] [Green Version]
- Yang, T.; Groenewald, J.Z.; Cheewangkoon, R.; Jami, F.; Abdollahzadeh, J.; Lombard, L.; Crous, P.W. Families, genera, and species of Botryosphaeriales. Fungal Biol. 2017, 121, 322–346. [Google Scholar] [CrossRef]
- Fischer, M.; González García, V. An annotated checklist of European basidiomycetes related to white rot of grapevine (Vitis vinifera). Phytopathol. Mediterr. 2015, 54, 281–298. [Google Scholar] [CrossRef]
- Bortolami, G.; Gambetta, G.A.; Cassan, C.; Dayer, S.; Farolfi, E.; Ferrer, N.; Gibon, Y.; Jolivet, J.; Lecomte, P.; Delmas, C.E.L. Grapevines under drought do not express esca leaf symptoms. Proc. Natl. Acad. Sci. USA 2021, 118, e2112825118. [Google Scholar] [CrossRef]
- Maher, N.; Piot, J.; Bastien, S.; Vallance, J.; Rey, P.; Guérin-Dubrana, L. Wood necrosis in esca-affected vines: Types, relationships and possible links with foliar symptom expression. J. Int. Sci. Vigne Vin 2012, 46, 15–27. [Google Scholar] [CrossRef]
- Bustin, S.-A.; Benes, V.; Garson, J.-A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, J.-L.; et al. The MIQE guidelines: Minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef] [Green Version]
- Bruez, E.; Larignon, P.; Bertsch, C.; Robert-Siegwald, G.; Lebrun, M.-H.; Rey, P.; Fontaine, F. Impacts of Sodium Arsenite on Wood Microbiota of Esca-Diseased Grapevines. J. Fungi 2021, 7, 498. [Google Scholar] [CrossRef]
- Pacetti, A.; Moretti, S.; Pinto, C.; Compant, S.; Farine, S.; Bertsch, C.; Mugnai, L. Trunk surgery as a tool to reduce foliar symptoms in diseases of the esca complex and its influence on vine wood microbiota. J. Fungi 2021, 7, 521. [Google Scholar] [CrossRef] [PubMed]
- Pinto, C.; Custódio, V.; Nunes, M.; Songy, A.; Rabenoelina, F.; Courteaux, B.; Clément, C.; Gomes, A.C.; Fontaine, F. Understand the Potential Role of Aureobasidium pullulans, a Resident Microorganism From Grapevine, to Prevent the Infection Caused by Diplodia seriata. Front. Microbiol. 2018, 9, 3047. [Google Scholar] [CrossRef] [PubMed]
- Bekris, F.; Vasileiadis, S.; Papadopoulou, E.; Samaras, A.; Testempasis, S.; Gkizi, D.; Tavlaki, G.; Tzima, A.; Paplomatas, E.; Markakis, E.; et al. Grapevine wood microbiome analysis identifies key fungal pathogens and potential interactions with the bacterial community implicated in grapevine trunk disease appearance. Environ. Microbiol. 2021, 16, 23. [Google Scholar] [CrossRef] [PubMed]
- Haidar, R.; Yacoub, A.; Vallance, J.; Compant, S.; Antonielli, L.; Saad, A.; Habenstein, B.; Kauffmann, B.; Grélard, A.; Loquet, A.; et al. Bacteria associated with wood tissues of Esca-diseased grapevines: Functional diversity and synergy with Fomitiporia mediterranea to degrade wood components. Environ. Microbiol. 2021, 23, 6104–6121. [Google Scholar] [CrossRef] [PubMed]
- Kottek, M.; Grieser, J.; Beck, C.; Rudolf, B.; Rubel, F. World map of the Koppen-Geiger climate classification updated. Meteorol. Z. 2006, 15, 259–263. [Google Scholar] [CrossRef] [PubMed]
- McKinney, H.H. A new system of grading plant diseases. J. Agric. Res. 1923, 26, 195–218. [Google Scholar]
- Crous, P.W.; Gams, W. Phaeomoniella chlamydospora Gen. et Comb. Nov., a Causal Organism of Petri Grapevine Decline and Esca. Phytopathol. Mediterr. 2000, 39, 112–118. [Google Scholar]
- Guarnaccia, V.; Groenewald, J.Z.; Woodhall, J.; Armengol, J.; Cinelli, T.; Eichmeier, A.; Ezra, D.; Fontaine, F.; Gramaje, D.; Gutierrez-Aguirregabiria, A.; et al. Diaporthe diversity and pathogenicity revealed from a broad survey of grapevine diseases in Europe. Pers. Mol. Phylogeny Evol. Fungi 2018, 40, 135–153. [Google Scholar] [CrossRef] [Green Version]
- Luque, J.; Garcia-Figueres, F.; Legorburu, F.J.; Muruamendiaraz, A.; Armengol, J.; Trouillas, F.P. Species of Diatrypaceae associated with grapevine trunk diseases in Eastern Spain. Phytopathol. Mediterr. 2012, 51, 528–540. [Google Scholar]
- White, T.J.; Bruns, T.D.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar] [CrossRef]
- Alves, A.; Phillips, A.J.L.; Henriques, I.; Correia, A. Evaluation of amplified ribosomal DNA restriction analysis as a method for the identification of Botryosphaeria species. FEMS Microbiol. Lett. 2005, 245, 221–229. [Google Scholar] [CrossRef]
- Carbone, I.; Kohn, L.M. A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 1999, 91, 553–556. [Google Scholar] [CrossRef]
- Tegli, S.; Surico, G.; Bertelli, E. Sequence Analysis of ITS Ribosomal DNA in Five” Phaeoacremonium” Species and Development of a PCR-Based Assay for the Detection of” P. chlamydosporum” and” P. aleophilum” in Grapevine Tissue. Phytopathol. Mediterr. 2000, 39, 1000–1016. [Google Scholar]
- Liao, J.; Xu, Q.; Xu, H.; Huang, D. Natural farming improves soil quality and alters microbial diversity in a cabbage field in Japan. Sustainability 2019, 11, 3131. [Google Scholar] [CrossRef] [Green Version]
- Gobbi, A.; Santini, R.G.; Filippi, E.; Ellegaard-Jensen, L.; Jacobsen, C.S.; Hansen, L.H. Quantitative and qualitative evaluation of the impact of the G2 enhancer, bead sizes and lysing tubes on the bacterial community composition during DNA extraction from recalcitrant soil core samples based on community sequencing and qPCR. PLoS ONE 2019, 14, e0200979. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Edgar, R.C.; Flyvbjerg, H. Error filtering, pair assembly and error correction for next-generation sequencing reads. Bioinformatics 2015, 31, 3476–3482. [Google Scholar] [CrossRef] [PubMed]
- Mahé, F.; Czech, L.; Stamatakis, A.; Quince, C.; de Vargas, C.; Dunthorn, M.; Rognes, T. Swarm v3: Towards tera-scale amplicon clustering. Bioinformatics 2021, 38, 267–269. [Google Scholar] [CrossRef]
- Edgar, R.C.; Haas, B.J.; Clemente, J.C.; Quince, C.; Knight, R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar] [CrossRef] [Green Version]
- Glöckner, F.O.; Yilmaz, P.; Quast, C.; Gerken, J.; Beccati, A.; Ciuprina, A.; Bruns, G.; Yarza, P.; Peplies, J.; Westram, R.; et al. 25 years of serving the community with ribosomal RNA gene reference databases and tools. J. Biotechnol. 2017, 261, 169–176. [Google Scholar] [CrossRef]
- Nilsson, R.H.; Larsson, K.-H.; Taylor, A.F.S.; Bengtsson-Palme, J.; Jeppesen, T.S.; Schigel, D.; Kennedy, P.; Picard, K.; Glöckner, F.O.; Tedersoo, L.; et al. The UNITE database for molecular identification of fungi: Handling dark taxa and parallel taxonomic classifications. Nucleic Acids Res. 2019, 47, 259–264. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; Stevens, M.H.H.; Wagner, H.H. Vegan: Community Ecology Package. R Package v.2.4-10. 2016. Available online: https://CRAN.R-project.org/package=vegan (accessed on 26 May 2022).
- De Caceres, M.; Legendre, P. Associations between species and groups of sites: Indices and statistical inference. Ecology 2009, 90, 3566. [Google Scholar] [CrossRef]
- Dufrêne, M.; Legendre, P. Species assemblages and indicator species: The need for a flexible asymmetrical approach. Ecol. Monogr. 1997, 67, 345–366. [Google Scholar] [CrossRef]





| Symptoms | S | H’ | J’ |
|---|---|---|---|
| As-BH | 213.25 ± 32.99 b | 2.33 ± 0.35 ab | 0.45 ± 0.06 ab |
| As-WR | 138.00 ± 12.88 b | 1.82 ± 0.12 b | 0.37 ± 0.02 b |
| Sy-BH | 405.09 ± 57.92 a | 3.41 ± 0.32 a | 0.57 ± 0.05 a |
| Sy-WR | 202.50 ± 39.12 b | 2.67 ± 0.30 ab | 0.52 ± 0.06 ab |
| ANOVA (p-values) | 8.64 (0.0001) | 5.40 (0.0030) | 3.71 (0.0182) |
| Symptoms | C (%) | N (%) |
|---|---|---|
| As-BH | 27.20 ± 1.99 | 0.17 ± 0.06 |
| As-WR | 36.03 ± 5.46 | 0.23 ± 0.03 |
| Sy-BH | 27.33 ± 1.30 | 0.19 ± 0.02 |
| Sy-WR | 28.17 ± 2.69 | 0.20 ± 0.03 |
| ANOVA (p-values) | 1.70 (0.244) | 0.42 (0.747) |
| Symptoms Category | Foliar Esca Symptoms | Wood Symptoms Observed in Trunk | GC | SP |
|---|---|---|---|---|
| As-BH | Asymptomatic | Healthy wood and black necrosis | C04 | A, B, C, D |
| C26 | A, B, C, D | |||
| D86 | A, B, C, D | |||
| As-WR | Asymptomatic | White rot | D43 | A, B, C, D |
| E08 | A, B, C, D | |||
| E60 | A, B, C, D | |||
| Sy-BH | Symptomatic | Healthy wood and black necrosis | D96 | A, B, C, D |
| D80 | A, B, C, D | |||
| C01 | A, B, C, D | |||
| Sy-WR | Symptomatic | White rot | C40 | A, B, C, D |
| C16 | A, B, C, D | |||
| C41 | A, B, C, D |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martín, L.; García-García, B.; Alguacil, M.d.M. Interactions of the Fungal Community in the Complex Patho-System of Esca, a Grapevine Trunk Disease. Int. J. Mol. Sci. 2022, 23, 14726. https://doi.org/10.3390/ijms232314726
Martín L, García-García B, Alguacil MdM. Interactions of the Fungal Community in the Complex Patho-System of Esca, a Grapevine Trunk Disease. International Journal of Molecular Sciences. 2022; 23(23):14726. https://doi.org/10.3390/ijms232314726
Chicago/Turabian StyleMartín, Laura, Blanca García-García, and María del Mar Alguacil. 2022. "Interactions of the Fungal Community in the Complex Patho-System of Esca, a Grapevine Trunk Disease" International Journal of Molecular Sciences 23, no. 23: 14726. https://doi.org/10.3390/ijms232314726





