MicroRNAs as the Sentinels of Redox and Hypertrophic Signalling
Abstract
1. Introduction
1.1. Challenging the Oxidative Stress Theory of Muscular Function Decline
1.2. miRNA Memory Cloud Perpetuates the Exercise-Induced Adaptive Phenotype
1.3. Role of miRNAs in Myogenesis—The Orchestration of the Metabolic Symphony
1.4. The Role of miRNA Networks in Muscle and Metabolic Health—The Context Matters as Much as the Content
1.5. Muscle miRNA Networks in Metabolic Disease, Ageing and Exercise Training—Fine-Tuning Thermostat of Redox and Anabolic Sensitivity
1.6. Chronic Endurance Training-Induced miRNA Cloud and the Adaptation-Permissive Epigenetic Landscape—Revised Molecular Basis of Exercise Adaptation Model
1.7. Circulating miRNAs as the Universal Language of the Hormesis Response—Biomarkers of Systemic Metabolic Health and Adaptability
2. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bassett, D.R.; Howley, E.T. Maximal oxygen uptake: “Classical” versus “contemporary” viewpoints. Med. Sci. Sport. Exerc. 1997, 29, 591–603. [Google Scholar] [CrossRef]
- Joyner, M.J.; Coyle, E.F. Endurance exercise performance: The physiology of champions. J. Physiol. 2008, 586, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Zhang, X.L. Physiological determinants of VO2max and the methods to evaluate it: A critical review. Sci. Sport. 2021, 36, 259–271. [Google Scholar] [CrossRef]
- Bassett, D.R.; Howley, E.T. Limiting factors for maximum oxygen uptake and determinants of endurance performance. Med. Sci. Sport. Exerc. 2000, 32, 70–84. [Google Scholar] [CrossRef] [PubMed]
- van der Zwaard, S.; de Ruiter, C.J.; Noordhof, D.A.; Sterrenburg, R.; Bloemers, F.W.; de Koning, J.J.; Jaspers, R.T.; van der Laarse, W.J. Maximal oxygen uptake is proportional to muscle fiber oxidative capacity, from chronic heart failure patients to professional cyclists. J. Appl. Physiol. 2016, 121, 636–645. [Google Scholar] [CrossRef] [PubMed]
- Myers, J.; Prakash, M.; Froelicher, V.; Do, D.; Partington, S.; Atwood, J.E. Exercise capacity and mortality among men referred for exercise testing. N. Engl. J. Med. 2002, 346, 793–801. [Google Scholar] [CrossRef] [PubMed]
- Rogers, M.A.; Hagberg, J.M.; Martin, W.H.; Ehsani, A.A.; Holloszy, J.O. Decline in VO2max with aging in master athletes and sedentary men. J. Appl. Physiol. 1990, 68, 2195–2199. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, T.; Spina, R.J.; Martin, W.H.; Kohrt, W.M.; Schechtman, K.B.; Holloszy, J.O.; Ehsani, A.A. Effects of aging, sex, and physical training on cardiovascular responses to exercise. Circulation 1992, 86, 494–503. [Google Scholar] [CrossRef]
- Neufer, P.D. The effect of detraining and reduced training on the physiological adaptations to aerobic exercise training. Sport. Med. 1989, 8, 302–321. [Google Scholar] [CrossRef]
- Tatarková, Z.; Kuka, S.; Racay, P.; Lehotsky, J.; Dobrota, D.; Mistuna, D.; Kaplan, P. Effects of aging on activities of mitochondrial electron transport chain complexes and oxidative damage in rat heart. Physiol. Rese 2011, 60, 281–289. [Google Scholar] [CrossRef]
- Picard, M.; Ritchie, D.; Thomas, M.M.; Wright, K.J.; Hepple, R.T. Alterations in intrinsic mitochondrial function with aging are fiber type-specific and do not explain differential atrophy between muscles. Aging Cell 2011, 10, 1047–1055. [Google Scholar] [CrossRef]
- Alway, S.E.; Mohamed, J.S.; Myers, M.J. Mitochondria initiate and regulate sarcopenia. Exerc. Sport Sci. Rev. 2017, 45, 58–69. [Google Scholar] [CrossRef]
- Steinbacher, P.; Eckl, P. Impact of oxidative stress on exercising skeletal muscle. Biomolecules 2015, 5, 356–377. [Google Scholar] [CrossRef] [PubMed]
- Margaritelis, N.V.; Paschalis, V.; Theodorou, A.A.; Kyparos, A.; Nikolaidis, M.G. Redox basis of exercise physiology. Redox Biol. 2020, 35, 101499. [Google Scholar] [CrossRef]
- Horn, A.; Van der Meulen, J.H.; Defour, A.; Hogarth, M.; Sreetama, S.C.; Reed, A.; Scheffer, L.; Chandel, N.S.; Jaiswal, J.K. Mitochondrial redox signaling enables repair of injured skeletal muscle cells. Sci. Signal. 2017, 10, eajj1978. [Google Scholar] [CrossRef]
- Romanello, V.; Sandri, M. Mitochondrial quality control and muscle mass maintenance. Front. Physiol. 2016, 6, 422. [Google Scholar] [CrossRef] [PubMed]
- Sies, H.; Berndt, C.; Jones, D.P. Oxidative stress. Annu. Rev. Biochem. 2017, 86, 715–748. [Google Scholar] [CrossRef] [PubMed]
- Monserrat-Mesquida, M.; Quetglas-Llabrés, M.; Capó, X.; Bouzas, C.; Mateos, D.; Pons, A.; Tur, J.A.; Sureda, A. Metabolic syndrome is associated with oxidative stress and proinflammatory state. Antioxidants 2020, 9, 236. [Google Scholar] [CrossRef] [PubMed]
- Forman, H.J.; Zhang, H. Targeting oxidative stress in disease: Promise and limitations of antioxidant therapy. Nat. Rev. Drug Discov. 2021, 20, 689–709. [Google Scholar] [CrossRef] [PubMed]
- Sies, H. Oxidative eustress: On constant alert for redox homeostasis. Redox Biology 2021, 41, 101867. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Puigserver, P.; Andersson, U.; Zhang, C.; Adelmant, G.; Mootha, V.; Troy, A.; Cinti, S.; Lowell, B.; Scarpulla, R.C.; et al. Mechanisms controlling mitochondrial biogenesis and respiration through the thermogenic coactivator PGC-1. Cell 1999, 98, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Done, A.J.; Gage, M.J.; Nieto, N.C.; Traustadóttir, T. Exercise-induced nrf2-signaling is impaired in aging. Free Radic. Biol. Med. 2016, 96, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Gureev, A.P.; Shaforostova, E.A.; Popov, V.N. Regulation of mitochondrial biogenesis as a way for active longevity: Interaction between the Nrf2 and PGC-1α signaling pathways. Front. Genet. 2019, 10, 435. [Google Scholar] [CrossRef] [PubMed]
- Romanello, V.; Scalabrin, M.; Albiero, M.; Blaauw, B.; Scorrano, L.; Sandri, M. Inhibition of the fission machinery mitigates opa1 impairment in adult skeletal muscles. Cells 2019, 8, 597. [Google Scholar] [CrossRef]
- Sligar, J.; DeBruin, D.A.; Saner, N.J.; Philp, A.M.; Philp, A. The importance of mitochondrial quality control for maintaining skeletal muscle function across health span. Am. J. Physiol. Cell Physiol. 2022, 322, C461–C467. [Google Scholar] [CrossRef] [PubMed]
- Fritzen, A.; Thøgersen, F.; Thybo, K.; Vissing, C.; Krag, T.; Ruiz-Ruiz, C.; Risom, L.; Wibrand, F.; Høeg, L.; Kiens, B.; et al. Adaptations in mitochondrial enzymatic activity occurs independent of genomic dosage in response to aerobic exercise training and deconditioning in human skeletal muscle. Cells 2019, 8, 237. [Google Scholar] [CrossRef]
- Mikkola, J.; Rusko, H.; Izquierdo, M.; Gorostiaga, E.; Häkkinen, K. Neuromuscular and cardiovascular adaptations during concurrent strength and endurance training in untrained men. Int. J. Sport. Med. 2012, 33, 702–710. [Google Scholar] [CrossRef]
- Milanović, Z.; Sporiš, G.; Weston, M. Effectiveness of high-intensity interval training (hit) and continuous endurance training for VO2MAX improvements: A systematic review and meta-analysis of controlled trials. Sport. Med. 2015, 45, 1469–1481. [Google Scholar] [CrossRef]
- Layec, G.; Haseler, L.J.; Hoff, J.; Hart, C.R.; Liu, X.; Le Fur, Y.; Jeong, E.-K.; Richardson, R.S. Short-term training alters the control of mitochondrial respiration rate before maximal oxidative ATP synthesis. Acta Physiol. 2013, 208, 376–386. [Google Scholar] [CrossRef]
- Hughes, D.C.; Ellefsen, S.; Baar, K. Adaptations to endurance and strength training. Cold Spring Harb. Perspect. Med. 2018, 8, a029769. [Google Scholar] [CrossRef] [PubMed]
- Koopman, R.; van Loon, L.J. Aging, exercise, and muscle protein metabolism. J. Appl. Physiol. 2009, 106, 2040–2048. [Google Scholar] [CrossRef] [PubMed]
- Murton, A.J.; Greenhaff, P.L. Resistance exercise and the mechanisms of muscle mass regulation in humans: Acute effects on muscle protein turnover and the gaps in our understanding of chronic resistance exercise training adaptation. Int. J. Biochem. Cell Biol. 2013, 45, 2209–2214. [Google Scholar] [CrossRef]
- Johnson, M.L.; Irving, B.A.; Lanza, I.R.; Vendelbo, M.H.; Konopka, A.R.; Robinson, M.M.; Henderson, G.C.; Klaus, K.A.; Morse, D.M.; Heppelmann, C.; et al. Differential effect of endurance training on mitochondrial protein damage, degradation, and acetylation in the context of aging. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2014, 70, 1386–1393. [Google Scholar] [CrossRef]
- Carter, H.N.; Kim, Y.; Erlich, A.T.; Zarrin-khat, D.; Hood, D.A. Autophagy and mitophagy flux in young and aged skeletal muscle following chronic contractile activity. J. Physiol. 2018, 596, 3567–3584. [Google Scholar] [CrossRef]
- Haran, P.H.; Rivas, D.A.; Fielding, R.A. Role and potential mechanisms of anabolic resistance in sarcopenia. J. Cachexia Sarcopenia Muscle 2012, 3, 157–162. [Google Scholar] [CrossRef]
- Fritzen, A.M.; Andersen, S.P.; Qadri, K.A.; Thøgersen, F.D.; Krag, T.; Ørngreen, M.C.; Vissing, J.; Jeppesen, T.D. Effect of aerobic exercise training and deconditioning on oxidative capacity and muscle mitochondrial enzyme machinery in young and elderly individuals. J. Clin. Med. 2020, 9, 3113. [Google Scholar] [CrossRef]
- Suetta, C.; Frandsen, U.; Mackey, A.L.; Jensen, L.; Hvid, L.G.; Bayer, M.L.; Petersson, S.J.; Schrøder, H.D.; Andersen, J.L.; Aagaard, P.; et al. Ageing is associated with diminished muscle re-growth and myogenic precursor cell expansion early after immobility-induced atrophy in human skeletal muscle. J. Physiol. 2013, 591, 3789–3804. [Google Scholar] [CrossRef] [PubMed]
- Egan, B.; O’Connor, P.L.; Zierath, J.R.; O’Gorman, D.J. Time course analysis reveals gene-specific transcript and protein kinetics of adaptation to short-term aerobic exercise training in human skeletal muscle. PLoS ONE 2013, 8, e74098. [Google Scholar] [CrossRef]
- Bokes, P.; Hojcka, M.; Singh, A. MicroRNA based feedforward control of intrinsic gene expression noise. IEEE/ACM Trans. Comput. Biol. Bioinform. 2019, 18, 272–282. [Google Scholar] [CrossRef]
- Kumar, V.; Selby, A.; Rankin, D.; Patel, R.; Atherton, P.; Hildebrandt, W.; Williams, J.; Smith, K.; Seynnes, O.; Hiscock, N.; et al. Age-related differences in the dose-response relationship of muscle protein synthesis to resistance exercise in young and old men. J. Physiol. 2009, 587, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Brook, M.S.; Wilkinson, D.J.; Mitchell, W.K.; Lund, J.N.; Phillips, B.E.; Szewczyk, N.J.; Greenhaff, P.L.; Smith, K.; Atherton, P.J. Synchronous deficits in cumulative muscle protein synthesis and ribosomal biogenesis underlie age-related anabolic resistance to exercise in humans. J. Physiol. 2016, 594, 7399–7417. [Google Scholar] [CrossRef]
- Hart, C.R.; Ryan, Z.C.; Pfaffenbach, K.T.; Dasari, S.; Parvizi, M.; Lalia, A.Z.; Lanza, I.R. Attenuated activation of the unfolded protein response following exercise in skeletal muscle of older adults. Aging 2019, 11, 7587–7604. [Google Scholar] [CrossRef]
- Kim, J.-S.; Kosek, D.J.; Petrella, J.K.; Cross, J.M.; Bamman, M.M. Resting and load-induced levels of myogenic gene transcripts differ between older adults with demonstrable sarcopenia and young men and women. J. Appl. Physiol. 2005, 99, 2149–2158. [Google Scholar] [CrossRef]
- Kosek, D.J.; Kim, J.-S.; Petrella, J.K.; Cross, J.M.; Bamman, M.M. Efficacy of 3 days/WK resistance training on myofiber hypertrophy and myogenic mechanisms in young vs. older adults. J. Appl. Physiol. 2006, 101, 531–544. [Google Scholar] [CrossRef]
- Fritzen, A.M.; Thøgersen, F.D.; Qadri, K.A.; Krag, T.; Sveen, M.-L.; Vissing, J.; Jeppesen, T.D. Preserved capacity for adaptations in strength and muscle regulatory factors in elderly in response to resistance exercise training and deconditioning. J. Clin. Med. 2020, 9, 2188. [Google Scholar] [CrossRef] [PubMed]
- Sjögren, R.J.; Lindgren Niss, M.H.; Krook, A. Skeletal muscle microRNAs: Roles in differentiation, disease and exercise. In Hormones, Metabolism and the Benefits of Exercise. Research and Perspectives in Endocrine Interactions; Spiegelman, B., Ed.; Springer: Cham, Switzerland, 2017; pp. 67–81. [Google Scholar] [CrossRef]
- Archacka, K.; Ciemerych, M.A.; Florkowska, A.; Romanczuk, K. Non-coding RNAs as regulators of myogenesis and postexercise muscle regeneration. Int. J. Mol. Sci. 2021, 22, 11568. [Google Scholar] [CrossRef]
- Kehl, T.; Backes, C.; Kern, F.; Fehlmann, T.; Ludwig, N.; Meese, E.; Lenhof, H.-P.; Keller, A. About Mirnas, MIRNA seeds, target genes and target pathways. Oncotarget 2017, 8, 107167–107175. [Google Scholar] [CrossRef]
- Riolo, G.; Cantara, S.; Marzocchi, C.; Ricci, C. MIRNA targets: From prediction tools to experimental validation. Methods Protoc. 2020, 4, 1. [Google Scholar] [CrossRef]
- Goljanek-Whysall, K.; Sweetman, D.; Abu-Elmagd, M.; Chapnik, E.; Dalmay, T.; Hornstein, E.; Münsterberg, A. MicroRNA regulation of the paired-box transcription factor PAX3 confers robustness to developmental timing of myogenesis. Proc. Natl. Acad. Sci. USA 2011, 108, 11936–11941. [Google Scholar] [CrossRef]
- Houzelle, A.; Dahlmans, D.; Nascimento, E.B.; Schaart, G.; Jörgensen, J.A.; Moonen-Kornips, E.; Kersten, S.; Wang, X.; Hoeks, J. MicroRNA-204-5p modulates mitochondrial biogenesis in C2C12 myotubes and associates with oxidative capacity in humans. J. Cell. Physiol. 2020, 235, 9851–9863. [Google Scholar] [CrossRef]
- Zhou, C.; Hu, Y.; Duan, S.; Gu, M.; Jiang, D.; Wang, Y.; Deng, Z.; Chen, J.; Chen, S.; Wang, L. Oxidative stress-induced mitophagy is suppressed by the mir-106b-93-25 cluster in a protective manner. Cell Death Dis. 2021, 12, 209. [Google Scholar] [CrossRef]
- Goljanek-Whysall, K.; Soriano-Arroquia, A.; McCormick, R.; Chinda, C.; McDonagh, B. Mir-181a regulates p62/SQSTM1, Parkin, and protein DJ-1 promoting mitochondrial dynamics in skeletal muscle aging. Aging Cell 2020, 19, e13140. [Google Scholar] [CrossRef] [PubMed]
- Fields, C.J.; Li, L.; Hiers, N.M.; Li, T.; Sheng, P.; Huda, T.; Shan, J.; Gay, L.; Gu, T.; Bian, J.; et al. Sequencing of argonaute-bound MIRNA/mRNA hybrids reveals regulation of the unfolded protein response by microRNA-320a. PLoS Genet. 2021, 17, e1009934. [Google Scholar] [CrossRef] [PubMed]
- Russell, A.P.; Lamon, S.; Boon, H.; Wada, S.; Güller, I.; Brown, E.L.; Chibalin, A.V.; Zierath, J.R.; Snow, R.J.; Stepto, N.; et al. Regulation of miRNAs in human skeletal muscle following acute endurance exercise and short-term endurance training. J. Physiol. 2013, 591, 4637–4653. [Google Scholar] [CrossRef]
- Ruijter, A.J.M.; Gennip, A.H.; Caron, H.N.; Kemp, S.; Kuilenburg, A.B.P. Histone Deacetylases (HDACS): Characterization of the classical HDAC family. Biochem. J. 2003, 370, 737–749. [Google Scholar] [CrossRef]
- Tian, H.; Liu, S.; Ren, J.; Lee, J.K.; Wang, R.; Chen, P. Role of histone deacetylases in skeletal muscle physiology and systemic energy homeostasis: Implications for metabolic diseases and therapy. Front. Physiol. 2020, 11, 949. [Google Scholar] [CrossRef]
- Bodine, S.C.; Latres, E.; Baumhueter, S.; Lai, V.K.-M.; Nunez, L.; Clarke, B.A.; Poueymirou, W.T.; Panaro, F.J.; Na, E.; Dharmarajan, K.; et al. Identification of ubiquitin ligases required for skeletal muscle atrophy. Science 2001, 294, 1704–1708. [Google Scholar] [CrossRef]
- Cohen, T.J.; Waddell, D.S.; Barrientos, T.; Lu, Z.; Feng, G.; Cox, G.A.; Bodine, S.C.; Yao, T.-P. The histone deacetylase HDAC4 connects neural activity to muscle transcriptional reprogramming. J. Biol. Chem. 2007, 282, 33752–33759. [Google Scholar] [CrossRef]
- Zahn, J.M.; Sonu, R.; Vogel, H.; Crane, E.; Mazan-Mamczarz, K.; Rabkin, R.; Davis, R.W.; Becker, K.G.; Owen, A.B.; Kim, S.K. Transcriptional profiling of aging in human muscle reveals a common aging signature. PLoS Genet. 2006, 2, e115. [Google Scholar] [CrossRef]
- Soci, U.P.; Melo, S.F.; Gomes, J.L.; Silveira, A.C.; Nóbrega, C.; de Oliveira, E.M. Exercise training and epigenetic regulation: Multilevel modification and regulation of gene expression. Adv. Exp. Med. Biol. 2017, 1000, 281–322. [Google Scholar] [CrossRef]
- Williams, A.H.; Valdez, G.; Moresi, V.; Qi, X.; McAnally, J.; Elliott, J.L.; Bassel-Duby, R.; Sanes, J.R.; Olson, E.N. MicroRNA-206 delays ALS progression and promotes regeneration of neuromuscular synapses in mice. Science 2009, 326, 1549–1554. [Google Scholar] [CrossRef]
- Oikawa, S.; Lee, M.; Akimoto, T. Conditional deletion of dicer in adult mice impairs skeletal muscle regeneration. Int. J. Mol. Sci. 2019, 20, 5686. [Google Scholar] [CrossRef]
- Oikawa, S.; Lee, M.; Motohashi, N.; Maeda, S.; Akimoto, T. An inducible knockout of Dicer in adult mice does not affect endurance exercise-induced muscle adaptation. Am. J. Physiol. Cell Physiol. 2019, 316, C285–C292. [Google Scholar] [CrossRef]
- Markus, I.; Constantini, K.; Hoffman, J.R.; Bartolomei, S.; Gepner, Y. Exercise-induced muscle damage: Mechanism, assessment and nutritional factors to accelerate recovery. Eur. J. Appl. Physiol. 2021, 121, 969–992. [Google Scholar] [CrossRef] [PubMed]
- Margolis, L.M.; Hatch-McChesney, A.; Allen, J.T.; DiBella, M.N.; Carrigan, C.T.; Murphy, N.E.; Karl, J.P.; Gwin, J.A.; Hennigar, S.R.; McClung, J.P.; et al. Circulating and skeletal muscle microrna profiles are more sensitive to sustained aerobic exercise than energy balance in males. J. Physiol. 2022, 600, 3951–3963. [Google Scholar] [CrossRef] [PubMed]
- Kolodziej, F.; McLysaght, A.; Goljanek-Whysall, K. Micro(RNA)-cloud can perpetuate physiological adaptation of skeletal muscle to exercise and energy imbalance. J. Physiol. 2022; in press. [Google Scholar] [CrossRef] [PubMed]
- Langer, H.T.; West, D.; Senden, J.; Spuler, S.; van Loon, L.J.; Baar, K. Myofibrillar protein synthesis rates are increased in chronically exercised skeletal muscle despite decreased anabolic signaling. Sci. Rep. 2022, 12, 7553. [Google Scholar] [CrossRef] [PubMed]
- Drummond, M.J.; McCarthy, J.J.; Fry, C.S.; Esser, K.A.; Rasmussen, B.B. Aging differentially affects human skeletal muscle microRNA expression at rest and after an anabolic stimulus of resistance exercise and essential amino acids. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E1333–E1340. [Google Scholar] [CrossRef]
- Abreu, P.; Mendes, S.V.; Ceccatto, V.M.; Hirabara, S.M. Satellite cell activation induced by aerobic muscle adaptation in response to endurance exercise in humans and rodents. Life Sci. 2017, 170, 33–40. [Google Scholar] [CrossRef]
- Murach, K.A.; Dungan, C.M.; von Walden, F.; Wen, Y. Epigenetic evidence for distinct contributions of resident and acquired Myonuclei during long-term exercise adaptation using timed in vivo myonuclear labeling. Am. J. Physiol.-Cell Physiol. 2022, 322, C86–C93. [Google Scholar] [CrossRef]
- Brook, M.S.; Wilkinson, D.J.; Smith, K.; Atherton, P.J. It’s not just about protein turnover: The role of ribosomal biogenesis and satellite cells in the regulation of skeletal muscle hypertrophy. Eur. J. Sport Sci. 2019, 19, 952–963. [Google Scholar] [CrossRef] [PubMed]
- Murach, K.A.; Fry, C.S.; Dupont-Versteegden, E.E.; McCarthy, J.J.; Peterson, C.A. Fusion and beyond: Satellite cell contributions to loading-induced skeletal muscle adaptation. FASEB J. 2021, 35, e21893. [Google Scholar] [CrossRef]
- Murach, K.A.; Mobley, C.B.; Zdunek, C.J.; Frick, K.K.; Jones, S.R.; McCarthy, J.J.; Peterson, C.A.; Dungan, C.M. Muscle memory: Myonuclear accretion, maintenance, morphology, and MIRNA levels with training and detraining in adult mice. J. Cachexia Sarcopenia Muscle 2020, 11, 1705–1722. [Google Scholar] [CrossRef]
- O’Rourke, J.R.; Georges, S.A.; Seay, H.R.; Tapscott, S.J.; McManus, M.T.; Goldhamer, D.J.; Swanson, M.S.; Harfe, B.D. Essential role for dicer during skeletal muscle development. Dev. Biol. 2007, 311, 359–368. [Google Scholar] [CrossRef] [PubMed]
- Horak, M.; Novak, J.; Bienertova-Vasku, J. Muscle-specific microRNAs in skeletal muscle development. Dev. Biol. 2016, 410, 2692. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Chen, X.; Chen, D.; Yu, B.; Li, M.; He, J.; Huang, Z. Regulation of skeletal myogenesis by microRNAs. J. Cell. Physiol. 2019, 235, 87–104. [Google Scholar] [CrossRef]
- Buckingham, M.; Rigby, P.W.J. Gene regulatory networks and transcriptional mechanisms that control myogenesis. Dev. Cell 2014, 28, 225–238. [Google Scholar] [CrossRef]
- Sweetman, D.; Goljanek, K.; Rathjen, T.; Oustanina, S.; Braun, T.; Dalmay, T.; Münsterberg, A. Specific requirements of Mrfs for the expression of muscle specific micrornas, Mir-1, Mir-206 and Mir-133. Dev. Biol. 2008, 321, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Schüler, S.C.; Hüttner, S.S.; von Eyss, B.; von Maltzahn, J. Adult stem cells at work: Regenerating skeletal muscle. Cell. Mol. Life Sci. 2019, 76, 2559–2570. [Google Scholar] [CrossRef]
- Dell’Orso, S.; Juan, A.H.; Ko, K.-D.; Naz, F.; Gutierrez-Cruz, G.; Feng, X.; Sartorelli, V. Single-cell analysis of adult skeletal muscle stem cells in homeostatic and regenerative conditions. Development 2019, 146, dev174177. [Google Scholar] [CrossRef]
- Ryall, J.G.; Dell’Orso, S.; Derfoul, A.; Juan, A.; Zare, H.; Feng, X.; Clermont, D.; Koulnis, M.; Gutierrez-Cruz, G.; Fulco, M.; et al. The nad+-dependent SIRT1 deacetylase translates a metabolic switch into regulatory epigenetics in skeletal muscle stem cells. Cell Stem Cell 2015, 16, 171–183. [Google Scholar] [CrossRef] [PubMed]
- Pala, F.; Di Girolamo, D.; Mella, S.; Yennek, S.; Chatre, L.; Ricchetti, M.; Tajbakhsh, S. Distinct metabolic states govern skeletal muscle stem cell fates during prenatal and postnatal myogenesis. J. Cell Sci. 2018, 131, jcs212977. [Google Scholar] [CrossRef] [PubMed]
- Hori, S.; Hiramuki, Y.; Nishimura, D.; Sato, F.; Sehara-Fujisawa, A. PDH-mediated metabolic flow is critical for skeletal muscle stem cell differentiation and myotube formation during regeneration in mice. FASEB J. 2019, 33, 8094–8109. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, D.; Scimè, A. Mitochondrial function in muscle stem cell fates. Front. Cell Dev. Biol. 2020, 8, 480. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Kim, J.-S.; Yoon, Y.; Santiago, M.C.; Brown, M.D.; Park, J.-Y. Inhibition of DRP1-dependent mitochondrial division impairs myogenic differentiation. Am. J. Physiol. -Regul. Integr. Comp. Physiol. 2013, 305, R927–R938. [Google Scholar] [CrossRef]
- Sin, J.; Andres, A.M.; Taylor, D.J.; Weston, T.; Hiraumi, Y.; Stotland, A.; Kim, B.J.; Huang, C.; Doran, K.S.; Gottlieb, R.A. Mitophagy is required for mitochondrial biogenesis and myogenic differentiation of c2c12 myoblasts. Autophagy 2016, 12, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Lee, Y.S.; Sivaprasad, U.; Malhotra, A.; Dutta, A. Muscle-specific microRNA mir-206 promotes muscle differentiation. J. Cell Biol. 2006, 174, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Dey, B.K.; Gagan, J.; Dutta, A. Mir-206 and -486 induce myoblast differentiation by downregulating PAX7. Mol. Cell. Biol. 2011, 31, 203–214. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-F.; Tao, Y.; Li, J.; Deng, Z.; Yan, Z.; Xiao, X.; Wang, D.-Z. MicroRNA-1 and microRNA-206 regulate skeletal muscle satellite cell proliferation and differentiation by repressing PAX7. J. Cell Biol. 2010, 190, 867–879. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.; Li, L.; Mubarokah, S.N.; Meech, R. Wnt/β-catenin signaling induces the myomiRs miR-133b and miR-206 to suppress pax7 and induce the myogenic differentiation program. J. Cell. Biochem. 2019, 120, 12740–12751. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-F.; Mandel, E.M.; Thomson, J.M.; Wu, Q.; Callis, T.E.; Hammond, S.M.; Conlon, F.L.; Wang, D.-Z. The role of microRNA-1 and microRNA-133 in skeletal muscle proliferation and differentiation. Nat. Genet. 2005, 38, 228–233. [Google Scholar] [CrossRef]
- Chen, X.; Wang, K.; Chen, J.; Guo, J.; Yin, Y.; Cai, X.; Guo, X.; Wang, G.; Yang, R.; Zhu, L.; et al. In vitro evidence suggests that mir-133a-mediated regulation of uncoupling protein 2 (UCP2) is an indispensable step in myogenic differentiation. J. Biol. Chem. 2009, 284, 5362–5369. [Google Scholar] [CrossRef] [PubMed]
- Cadenas, S. Mitochondrial uncoupling, Ros Generation and cardioprotection. Biochim. Biophys. Acta Bioenerg. 2018, 1859, 940–950. [Google Scholar] [CrossRef] [PubMed]
- Small, E.M.; O’Rourke, J.R.; Moresi, V.; Sutherland, L.B.; McAnally, J.; Gerard, R.D.; Richardson, J.A.; Olson, E.N. Regulation of PI3-kinase/Akt signaling by muscle-enriched microRNA-486. Proc. Natl. Acad. Sci. USA 2010, 107, 4218–4223. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Chen, X.; Chen, D.; Yu, B.; Huang, Z. Foxo1: A novel insight into its molecular mechanisms in the regulation of skeletal muscle differentiation and fiber type specification. Oncotarget 2016, 8, 10662–10674. [Google Scholar] [CrossRef]
- Glass, D.J. Pi3 kinase regulation of skeletal muscle hypertrophy and atrophy. Curr. Top. Microbiol. Immunol. 2010, 346, 267–278. [Google Scholar] [CrossRef] [PubMed]
- van Rooij, E.; Quiat, D.; Johnson, B.A.; Sutherland, L.B.; Qi, X.; Richardson, J.A.; Kelm, R.J.; Olson, E.N. A family of microRNAs encoded by myosin genes governs myosin expression and muscle performance. Dev. Cell 2009, 17, 662–673. [Google Scholar] [CrossRef] [PubMed]
- Bjorkman, K.K.; Guess, M.G.; Harrison, B.C.; Polmear, M.M.; Peter, A.K.; Leinwand, L.A. Mir-206 enforces a slow muscle phenotype. J. Cell Sci. 2020, 133, jcs243162. [Google Scholar] [CrossRef] [PubMed]
- van Rooij, E.; Sutherland, L.B.; Qi, X.; Richardson, J.A.; Hill, J.; Olson, E.N. Control of stress-dependent cardiac growth and gene expression by a MicroRNA. Science 2007, 316, 575–579. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liang, X.; Zhou, D.; Lai, L.; Xiao, L.; Liu, L.; Fu, T.; Kong, Y.; Zhou, Q.; Vega, R.B.; et al. Coupling of mitochondrial function and skeletal muscle fiber type by a miR-499/FNIP1/AMPK circuit. EMBO Mol. Med. 2016, 8, 1212–1228. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Ji, J.; Yan, X.-H. Cross-talk between AMPK and mTOR in regulating energy balance. Crit. Rev. Food Sci. Nutr. 2012, 52, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Ramirez Reyes, J.M.; Cuesta, R.; Pause, A. Folliculin: A regulator of transcription through AMPK and mTOR signaling pathways. Front. Cell Dev. Biol. 2021, 9, 667311. [Google Scholar] [CrossRef]
- Carrer, M.; Liu, N.; Grueter, C.E.; Williams, A.H.; Frisard, M.I.; Hulver, M.W.; Bassel-Duby, R.; Olson, E.N. Control of mitochondrial metabolism and systemic energy homeostasis by microRNAs 378 and 378*. Proc. Natl. Acad. Sci. USA 2012, 109, 15330–15335. [Google Scholar] [CrossRef] [PubMed]
- Machado, I.F.; Teodoro, J.S.; Castela, A.C.; Palmeira, C.M.; Rolo, A.P. Mir-378A-3P participates in metformin’s mechanism of action on C2C12 cells under hyperglycemia. Int. J. Mol. Sci. 2021, 22, 541. [Google Scholar] [CrossRef] [PubMed]
- Hostalek, U.; Gwilt, M.; Hildemann, S. Therapeutic use of metformin in prediabetes and diabetes prevention. Drugs 2015, 75, 1071–1094. [Google Scholar] [CrossRef]
- Konopka, A.R.; Laurin, J.L.; Schoenberg, H.M.; Reid, J.J.; Castor, W.M.; Wolff, C.A.; Musci, R.V.; Safairad, O.D.; Linden, M.A.; Biela, L.M.; et al. Metformin inhibits mitochondrial adaptations to aerobic exercise training in older adults. Aging Cell 2018, 18, e12880. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Cabañas, A.; Morales-Palomo, F.; Alvarez-Jimenez, L.; Ortega, J.F.; Mora-Rodriguez, R. Effects of chronic metformin treatment on training adaptations in men and women with hyperglycemia: A prospective study. Obesity 2022, 30, 1219–1230. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Jiang, J.; Liu, W.; Wang, H.; Zhao, L.; Liu, S.; Li, P.; Zhang, S.; Sun, C.; Wu, Y.; et al. MicroRNA-378 promotes autophagy and inhibits apoptosis in skeletal muscle. Proc. Natl. Acad. Sci. USA 2018, 115, E10849–E10858. [Google Scholar] [CrossRef] [PubMed]
- Mallat, Y.; Tritsch, E.; Ladouce, R.; Winter, D.L.; Friguet, B.; Li, Z.; Mericskay, M. Proteome modulation in H9C2 cardiac cells by microRNAs Mir-378 and Mir-378. Mol. Cell. Proteom. 2014, 13, 18–29. [Google Scholar] [CrossRef] [PubMed]
- Pan, D.; Mao, C.; Quattrochi, B.; Friedline, R.H.; Zhu, L.J.; Jung, D.Y.; Kim, J.K.; Lewis, B.; Wang, Y.-X. MicroRNA-378 controls classical brown fat expansion to counteract obesity. Nat. Commun. 2014, 5, 4725. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Li, H.; Zhang, B.; Li, C.; Dong, D.; Lan, X.; Huang, Y.; Bai, Y.; Lin, F.; Zhao, X.; et al. Mir-378A-3P promotes differentiation and inhibits proliferation of myoblasts by targeting HDAC4 in skeletal muscle development. RNA Biol. 2016, 13, 1300–1309. [Google Scholar] [CrossRef]
- Ju, H.; Yang, Y.; Sheng, A.; Qi, Y. MicroRNA-378 promotes myogenic differentiation by targeting BMP4. Mol. Med. Rep. 2016, 13, 2194–2200. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Backs, J. Cam kinase II selectively signals to histone deacetylase 4 during cardiomyocyte hypertrophy. J. Clin. Investig. 2006, 116, 1853–1864. [Google Scholar] [CrossRef] [PubMed]
- Terada, K.; Misao, S.; Katase, N.; Nishimatsu, S.-I.; Nohno, T. Interaction of Wnt signaling with BMP/Smad signaling during the transition from cell proliferation to myogenic differentiation in mouse myoblast-derived cells. Int. J. Cell Biol. 2013, 201, 616294. [Google Scholar] [CrossRef] [PubMed]
- Cohen, T.J.; Choi, M.-C.; Kapur, M.; Lira, V.A.; Yan, Z.; Yao, T.-P. HDAC4 regulates muscle fiber type-specific gene expression programs. Mol. Cells 2015, 38, 343–348. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.; Martin, S.C.; Parkington, J.; Cadena, S.M.; Zhu, J.; Ibebunjo, C.; Summermatter, S.; Londraville, N.; Patora-Komisarska, K.; Widler, L.; et al. HDAC4 controls muscle homeostasis through deacetylation of myosin heavy chain, PGC-1A, and HSC70. Cell Rep. 2019, 29, 749–763.e12. [Google Scholar] [CrossRef]
- Ji, J.; Tsika, G.L.; Rindt, H.; Schreiber, K.L.; McCarthy, J.J.; Kelm, R.J.; Tsika, R. PURA and PURΒ collaborate with SP3 to negatively regulate β-myosin heavy chain gene expression during skeletal muscle inactivity. Mol. Cell. Biol. 2007, 27, 1531–1543. [Google Scholar] [CrossRef]
- McCarthy, J.J.; Esser, K.A.; Peterson, C.A.; Dupont-Versteegden, E.E. Evidence of myomiR network regulation of β-myosin heavy chain gene expression during skeletal muscle atrophy. Physiol. Genom. 2009, 39, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Boon, H.; Sjögren, R.J.; Massart, J.; Egan, B.; Kostovski, E.; Iversen, P.O.; Hjeltnes, N.; Chibalin, A.V.; Widegren, U.; Zierath, J.R. MicroRNA-208b progressively declines after spinal cord injury in humans and is inversely related to myostatin expression. Physiol. Rep. 2015, 3, e12622. [Google Scholar] [CrossRef] [PubMed]
- Gan, Z.; Rumsey, J.; Hazen, B.C.; Lai, L.; Leone, T.C.; Vega, R.B.; Xie, H.; Conley, K.E.; Auwerx, J.; Smith, S.R.; et al. Nuclear receptor/microRNA circuitry links muscle fiber type to energy metabolism. J. Clin. Investig. 2013, 123, 2564–2575. [Google Scholar] [CrossRef]
- Sager, R.A.; Woodford, M.R.; Backe, S.J.; Makedon, A.M.; Baker-Williams, A.J.; DiGregorio, B.T.; Loiselle, D.R.; Haystead, T.A.; Zachara, N.E.; Prodromou, C.; et al. Post-translational regulation of FNIP1 creates a rheostat for the molecular chaperone hsp90. Cell Rep. 2019, 26, 1344–1356.e5. [Google Scholar] [CrossRef]
- Reinle, K.; Mogk, A.; Bukau, B. The diverse functions of small heat shock proteins in the proteostasis network. J. Mol. Biol. 2022, 434, 167157. [Google Scholar] [CrossRef]
- Dubińska-Magiera, M.; Jabłońska, J.; Saczko, J.; Kulbacka, J.; Jagla, T.; Daczewska, M. Contribution of small heat shock proteins to muscle development and function. FEBS Lett. 2014, 588, 517–530. [Google Scholar] [CrossRef] [PubMed]
- Archer, A.E.; Von Schulze, A.T.; Geiger, P.C. Exercise, heat shock proteins and insulin resistance. Philos. Trans. R. Soc. B Biol. Sci. 2017, 373, 20160529. [Google Scholar] [CrossRef]
- Dimauro, I.; Mercatelli, N.; Caporossi, D. Exercise-induced ROS in heat shock proteins response. Free. Radic. Biol. Med. 2016, 98, 46–55. [Google Scholar] [CrossRef]
- Collier, M.P.; Benesch, J.L.P. Small heat-shock proteins and their role in mechanical stress. Cell Stress Chaperones 2020, 25, 601–613. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Shen, Z.; Yu, D.; Zhao, C.; Zou, H.; Ma, B.; Dong, W.; Chen, W.; Huang, D.; Yu, Z. NRF2 contributes to the benefits of exercise interventions on age-related skeletal muscle disorder via regulating DRP1 stability and mitochondrial fission. Free. Radic. Biol. Med. 2022, 178, 59–75. [Google Scholar] [CrossRef]
- Sabouny, R.; Fraunberger, E.; Geoffrion, M.; Ng, A.C.-H.; Baird, S.D.; Screaton, R.A.; Milne, R.; McBride, H.M.; Shutt, T.E. The KEAP1–Nrf2 stress response pathway promotes mitochondrial hyperfusion through degradation of the mitochondrial fission protein DRP1. Antioxid. Redox Signal. 2017, 27, 1447–1459. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.-D.; Fan, S.-D.; Chen, X.-Y.; Yan, X.-L.; Zhang, X.-Z.; Ma, B.-W.; Yu, D.-Y.; Xiao, W.-Y.; Zhuang, C.-L.; Yu, Z. NRF2 deficiency exacerbates frailty and sarcopenia by impairing skeletal muscle mitochondrial biogenesis and dynamics in an age-dependent manner. Exp. Gerontol. 2019, 119, 61–73. [Google Scholar] [CrossRef]
- Safdar, A.; deBeer, J.; Tarnopolsky, M.A. Dysfunctional NRF2–KEAP1 redox signaling in skeletal muscle of the sedentary old. Free. Radic. Biol. Med. 2010, 49, 1487–1493. [Google Scholar] [CrossRef] [PubMed]
- Cobley, J.N.; Moult, P.R.; Burniston, J.G.; Morton, J.P.; Close, G.L. Exercise improves mitochondrial and redox-regulated stress responses in the elderly: Better late than never! Biogerontology 2014, 16, 249–264. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.; Sinha, I.; Tan, K.Y.; Rosner, B.; Dreyfuss, J.M.; Gjata, O.; Tran, P.; Shoelson, S.E.; Wagers, A.J. Age-associated NF-ΚB signaling in myofibers alters the satellite cell niche and Re-strains muscle stem cell function. Aging 2016, 8, 2871–2896. [Google Scholar] [CrossRef]
- Proto, J.D.; Lu, A.; Dorronsoro, A.; Scibetta, A.; Robbins, P.D.; Niedernhofer, L.J.; Huard, J. Inhibition of NF-ΚB improves the stress resistance and myogenic differentiation of mdspcs isolated from naturally aged mice. PLoS ONE 2017, 12, e0179270. [Google Scholar] [CrossRef] [PubMed]
- Vasilaki, A.; Iwanejko, L.M.; McArdle, F.; Broome, C.S.; Jackson, M.J.; McArdle, A. Skeletal muscles of aged male mice fail to adapt following contractile activity. Biochem. Soc. Trans. 2003, 31, 455–456. [Google Scholar] [CrossRef] [PubMed]
- Vasilaki, A.; Jackson, M.J.; McArdle, A. Attenuated hsp70 response in skeletal muscle of aged rats following contractile activity. Muscle Nerve 2002, 25, 902–905. [Google Scholar] [CrossRef] [PubMed]
- Vasilaki, A.; McArdle, F.; Iwanejko, L.M.; McArdle, A. Adaptive responses of mouse skeletal muscle to contractile activity: The effect of age. Mech. Ageing Dev. 2006, 127, 830–839. [Google Scholar] [CrossRef]
- Tantiwong, P.; Shanmugasundaram, K.; Monroy, A.; Ghosh, S.; Li, M.; DeFronzo, R.A.; Cersosimo, E.; Sriwijitkamol, A.; Mohan, S.; Musi, N. NF-ΚB activity in muscle from obese and type 2 diabetic subjects under basal and exercise-stimulated conditions. Am. J. Physiol. Endocrinol. Metab. 2010, 299, E794–E801. [Google Scholar] [CrossRef] [PubMed]
- Rajasekaran, N.S.; Shelar, S.B.; Jones, D.P.; Hoidal, J.R. Reductive stress impairs myogenic differentiation. Redox Biol. 2020, 34, 101492. [Google Scholar] [CrossRef]
- Alway, S.E.; McCrory, J.L.; Kearcher, K.; Vickers, A.; Frear, B.; Gilleland, D.L.; Bonner, D.E.; Thomas, J.M.; Donley, D.A.; Lively, M.W.; et al. Resveratrol enhances exercise-induced cellular and functional adaptations of skeletal muscle in older men and women. J. Gerontol. Ser. A 2017, 72, 1595–1606. [Google Scholar] [CrossRef]
- Morrison, D.; Hughes, J.; Della Gatta, P.A.; Mason, S.; Lamon, S.; Russell, A.P.; Wadley, G.D. Vitamin C and E supplementation prevents some of the cellular adaptations to endurance-training in humans. Free. Radic. Biol. Med. 2015, 89, 852–862. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, G.; Cumming, K.T.; Holden, G.; Hallén, J.; Rønnestad, B.R.; Sveen, O.; Skaug, A.; Paur, I.; Bastani, N.E.; Østgaard, H.N.; et al. Vitamin C and E supplementation hampers cellular adaptation to endurance training in humans: A double-blind, randomised, controlled trial. J. Physiol. 2014, 592, 1887–1901. [Google Scholar] [CrossRef] [PubMed]
- Ristow, M.; Zarse, K.; Oberbach, A.; Klöting, N.; Birringer, M.; Kiehntopf, M.; Stumvoll, M.; Kahn, C.R.; Blüher, M. Antioxidants Prevent Health-Promoting Effects of Physical Exercise in Humans. Proc. Natl. Acad. Sci. USA 2009, 106, 8665–8670. [Google Scholar] [CrossRef]
- Cobley, J.N.; Sakellariou, G.K.; Owens, D.J.; Murray, S.; Waldron, S.; Gregson, W.; Fraser, W.D.; Burniston, J.G.; Iwanejko, L.A.; McArdle, A.; et al. Lifelong training preserves some redox-regulated adaptive responses after an acute exercise stimulus in aged human skeletal muscle. Free Radic. Biol. Med. 2014, 70, 23–32. [Google Scholar] [CrossRef]
- Iversen, N.; Krustrup, P.; Rasmussen, H.N.; Rasmussen, U.F.; Saltin, B.; Pilegaard, H. Mitochondrial biogenesis and angiogenesis in skeletal muscle of the elderly. Exp. Gerontol. 2011, 46, 670–678. [Google Scholar] [CrossRef]
- Cobley, J.N.; Bartlett, J.D.; Kayani, A.; Murray, S.W.; Louhelainen, J.; Donovan, T.; Waldron, S.; Gregson, W.; Burniston, J.G.; Morton, J.P.; et al. PGC-1α transcriptional response and mitochondrial adaptation to acute exercise is maintained in skeletal muscle of sedentary elderly males. Biogerontology 2012, 13, 621–631. [Google Scholar] [CrossRef]
- Rivas, D.A.; Lessard, S.J.; Rice, N.P.; Lustgarten, M.S.; So, K.; Goodyear, L.J.; Parnell, L.D.; Fielding, R.A. Diminished skeletal muscle microRNA expression with aging is associated with attenuated muscle plasticity and inhibition of IGF-1 signaling. FASEB J. 2014, 28, 4133–4147. [Google Scholar] [CrossRef]
- D’Souza, R.F.; Zeng, N.; Markworth, J.F.; Figueiredo, V.C.; Hedges, C.P.; Petersen, A.C.; Della Gatta, P.A.; Cameron-Smith, D.; Mitchell, C.J. Whey Protein Supplementation Post Resistance exercise in elderly men induces changes in muscle miRNAs compared to resistance exercise alone. Front. Nutr. 2019, 6, 91. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Muthusamy, S.; Liang, R.; Sarojini, H.; Wang, E. Increased expression of mir-34a and mir-93 in rat liver during aging, and their impact on the expression of MGST1 and SIRT1. Mech. Ageing Dev. 2011, 132, 75–85. [Google Scholar] [CrossRef]
- Smit-McBride, Z.; Forward, K.I.; Nguyen, A.T.; Bordbari, M.H.; Oltjen, S.L.; Hjelmeland, L.M. Age-dependent increase in miRNA-34a expression in the posterior pole of the mouse eye. Mol. Vis. 2014, 20, 1569–1578. [Google Scholar]
- Csiszar, A.; Gautam, T.; Sosnowska, D.; Tarantini, S.; Banki, E.; Tucsek, Z.; Toth, P.; Losonczy, G.; Koller, A.; Reglodi, D.; et al. Caloric restriction confers persistent anti-oxidative, pro-angiogenic, and anti-inflammatory effects and promotes anti-aging MIRNA expression profile in cerebromicrovascular endothelial cells of aged rats. Am. J. Physiol. Heart Circ. Physiol. 2014, 307, H292–H306. [Google Scholar] [CrossRef]
- Yuan, H.; Xu, Y.; Luo, Y.; Wang, N.-X.; Xiao, J.-H. Role of Nrf2 in cell senescence regulation. Mol. Cell. Biochem. 2020, 476, 247–259. [Google Scholar] [CrossRef]
- Ruan, L.; Mendhe, B.; Parker, E.; Kent, A.; Isales, C.M.; Hill, W.D.; McGee-Lawrence, M.; Fulzele, S.; Hamrick, M.W. Long non-coding RNA MALAT1 is depleted with age in skeletal muscle in vivo and MALAT1 silencing increases expression of TGF-β1 in vitro. Front. Physiol. 2022, 12, 742004. [Google Scholar] [CrossRef] [PubMed]
- Fulzele, S.; Mendhe, B.; Khayrullin, A.; Johnson, M.; Kaiser, H.; Liu, Y.; Isales, C.M.; Hamrick, M.W. Muscle-derived mir-34a increases with age in circulating extracellular vesicles and induces senescence of bone marrow stem cells. Aging 2019, 11, 1791–1803. [Google Scholar] [CrossRef]
- Soriano-Arroquia, A.; House, L.; Tregilgas, L.; Canty-Laird, E.; Goljanek-Whysall, K. The functional consequences of age-related changes in microRNA expression in skeletal muscle. Biogerontology 2016, 17, 641–654. [Google Scholar] [CrossRef] [PubMed]
- Hofer, T.; Marzetti, E.; Xu, J.; Seo, A.Y.; Gulec, S.; Knutson, M.D.; Leeuwenburgh, C.; Dupont-Versteegden, E.E. Increased iron content and RNA oxidative damage in skeletal muscle with aging and disuse atrophy. Exp. Gerontol. 2008, 43, 563–570. [Google Scholar] [CrossRef]
- Chen, X.; Yu, H.; Li, Z.; Ye, W.; Liu, Z.; Gao, J.; Wang, Y.; Li, X.; Zhang, L.; Alenina, N.; et al. Oxidative RNA damage in the pathogenesis and treatment of type 2 diabetes. Front. Physiol. 2022, 13, 725919. [Google Scholar] [CrossRef] [PubMed]
- Seok, H.; Lee, H.; Lee, S.; Ahn, S.H.; Lee, H.-S.; Kim, G.-W.D.; Peak, J.; Park, J.; Cho, Y.K.; Jeong, Y.; et al. Position-specific oxidation of mir-1 encodes cardiac hypertrophy. Nature 2020, 584, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Ciesielska, S.; Slezak-Prochazka, I.; Bil, P.; Rzeszowska-Wolny, J. Micro RNAs in regulation of cellular redox homeostasis. Int. J. Mol. Sci. 2021, 22, 6022. [Google Scholar] [CrossRef]
- Soriano-Arroquia, A.; Gostage, J.; Xia, Q.; Bardell, D.; McCormick, R.; McCloskey, E.; Bellantuono, I.; Clegg, P.; McDonagh, B.; Goljanek-Whysall, K. Mir-24 and its target gene PRDX6 regulate viability and senescence of myogenic progenitors during aging. Aging Cell 2021, 20, e13475. [Google Scholar] [CrossRef] [PubMed]
- Mailloux, R.J. Protein S-glutathionylation reactions as a global inhibitor of cell metabolism for the desensitization of hydrogen peroxide signals. Redox Biol. 2020, 32, 101472. [Google Scholar] [CrossRef]
- Nielsen, S.; Scheele, C.; Yfanti, C.; Åkerström, T.; Nielsen, A.R.; Pedersen, B.K.; Laye, M. Muscle specific microRNAs are regulated by endurance exercise in human skeletal muscle. J. Physiol. 2010, 588, 4029–4037. [Google Scholar] [CrossRef]
- Egan, B.; Zierath, J.R. Exercise metabolism and the molecular regulation of skeletal muscle adaptation. Cell Metab. 2013, 17, 162–184. [Google Scholar] [CrossRef] [PubMed]
- Ogasawara, R.; Akimoto, T.; Umeno, T.; Sawada, S.; Hamaoka, T.; Fujita, S. MicroRNA expression profiling in skeletal muscle reveals different regulatory patterns in high and low responders to resistance training. Physiol. Genom. 2016, 48, 320–324. [Google Scholar] [CrossRef]
- Davidsen, P.K.; Gallagher, I.J.; Hartman, J.W.; Tarnopolsky, M.A.; Dela, F.; Helge, J.W.; Timmons, J.A.; Phillips, S.M. High responders to resistance exercise training demonstrate differential regulation of skeletal muscle microRNA expression. J. Appl. Physiol. 2011, 110, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Machado, I.F.; Teodoro, J.S.; Palmeira, C.M.; Rolo, A.P. Mir-378A: A new emerging microRNA in metabolism. Cell. Mol. Life Sci. 2020, 77, 1947–1958. [Google Scholar] [CrossRef] [PubMed]
- Fyfe, J.J.; Bishop, D.J.; Zacharewicz, E.; Russell, A.P.; Stepto, N.K. Concurrent exercise incorporating high-intensity interval or continuous training modulates mtorc1 signaling and microrna expression in human skeletal muscle. Am. J. Physiol. -Regul. Integr. Comp. Physiol. 2016, 310, R1297–R1311. [Google Scholar] [CrossRef]
- Renzini, A.; Marroncelli, N.; Noviello, C.; Moresi, V.; Adamo, S. HDAC4 regulates skeletal muscle regeneration via soluble factors. Front. Physiol. 2018, 9, 1387. [Google Scholar] [CrossRef]
- Pigna, E.; Renzini, A.; Greco, E.; Simonazzi, E.; Fulle, S.; Mancinelli, R.; Moresi, V.; Adamo, S. HDAC4 preserves skeletal muscle structure following long-term denervation by mediating distinct cellular responses. Skelet. Muscle 2018, 8, 6. [Google Scholar] [CrossRef]
- Colleluori, G.; Aguirre, L.; Phadnis, U.; Fowler, K.; Armamento-Villareal, R.; Sun, Z.; Brunetti, L.; Hyoung Park, J.; Kaipparettu, B.A.; Putluri, N.; et al. Aerobic plus resistance exercise in obese older adults improves muscle protein synthesis and preserves myocellular quality despite weight loss. Cell Metab. 2019, 30, 261–273. [Google Scholar] [CrossRef]
- Mueller, M.; Breil, F.A.; Lurman, G.; Klossner, S.; Flück, M.; Billeter, R.; Däpp, C.; Hoppeler, H. Different molecular and structural adaptations with eccentric and conventional strength training in elderly men and women. Gerontology 2011, 57, 528–538. [Google Scholar] [CrossRef]
- Rivas, D.A.; Peng, F.; Benard, T.; Ramos da Silva, A.S.; Fielding, R.A.; Margolis, L.M. Mir-19B-3P is associated with a diametric response to resistance exercise in older adults and regulates skeletal muscle anabolism via PTEN inhibition. Am. J. Physiol. Cell Physiol. 2021, 321, C977–C991. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.; Danielson, K.M.; Benton, M.C.; Ziegler, O.; Shah, R.; Stubbs, R.S.; Das, S.; Macartney-Coxson, D. MIRNA signatures of insulin resistance in obesity. Obesity 2017, 25, 1734–1744. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Koh, H.W.; Zhou, L.; Cheng, H.; Loh, T.P.; Parvaresh Rizi, E.; Toh, S.A.; Ronnett, G.V.; Huang, B.E.; Khoo, C.M. Plasma protein and microRNA biomarkers of insulin resistance: A network-based integrative -omics analysis. Front. Physiol. 2019, 10, 379. [Google Scholar] [CrossRef]
- Morris, C.; Grada, C.O.; Ryan, M.; Roche, H.M.; De Vito, G.; Gibney, M.J.; Gibney, E.R.; Brennan, L. The relationship between aerobic fitness level and metabolic profiles in healthy adults. Mol. Nutr. Food Res. 2013, 57, 1246–1254. [Google Scholar] [CrossRef] [PubMed]
- Castro, A.; Duft, R.G.; Ferreira, M.L.; Andrade, A.L.; Gáspari, A.F.; Silva, L.D.; Oliveira-Nunes, S.G.; Cavaglieri, C.R.; Ghosh, S.; Bouchard, C.; et al. Association of skeletal muscle and serum metabolites with maximum power output gains in response to continuous endurance or high-intensity interval training programs: The Times Study—A randomized controlled trial. PLoS ONE 2019, 14, e0212115. [Google Scholar] [CrossRef] [PubMed]
- Soares, R.J.; Cagnin, S.; Chemello, F.; Silvestrin, M.; Musaro, A.; De Pitta, C.; Lanfranchi, G.; Sandri, M. Involvement of microRNAs in the regulation of muscle wasting during catabolic conditions. J. Biol. Chem. 2014, 289, 21909–21925. [Google Scholar] [CrossRef]
- Rome, S.; Forterre, A.; Mizgier, M.L.; Bouzakri, K. Skeletal muscle-released extracellular vesicles: State of the art. Front. Physiol. 2019, 10, 929. [Google Scholar] [CrossRef]
- Whitham, M.; Parker, B.L.; Friedrichsen, M.; Hingst, J.R.; Hjorth, M.; Hughes, W.E.; Egan, C.L.; Cron, L.; Watt, K.I.; Kuchel, R.P.; et al. Extracellular vesicles provide a means for tissue crosstalk during exercise. Cell Metab. 2018, 27, 237–251. [Google Scholar] [CrossRef] [PubMed]
- Groot, M.; Lee, H. Sorting mechanisms for microRNAs into extracellular vesicles and their associated diseases. Cells 2020, 9, 1044. [Google Scholar] [CrossRef]
- Vechetti, I.J.; Valentino, T.; Mobley, C.B.; McCarthy, J.J. The role of extracellular vesicles in skeletal muscle and systematic adaptation to exercise. J. Physiol. 2020, 599, 845–861. [Google Scholar] [CrossRef]
- Ostrom, E.L.; Valencia, A.P.; Marcinek, D.J.; Traustadóttir, T. High intensity muscle stimulation activates a systemic Nrf2-mediated redox stress response. Free. Radic. Biol. Med. 2021, 172, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Vechetti, I.J.; Peck, B.D.; Wen, Y.; Walton, R.G.; Valentino, T.R.; Alimov, A.P.; Dungan, C.M.; Van Pelt, D.W.; von Walden, F.; Alkner, B.; et al. Mechanical overload-induced muscle-derived extracellular vesicles promote adipose tissue lipolysis. FASEB J. 2021, 35, e21644. [Google Scholar] [CrossRef] [PubMed]
- Margolis, L.M.; Lessard, S.J.; Ezzyat, Y.; Fielding, R.A.; Rivas, D.A. Circulating microRNA are predictive of aging and acute adaptive response to resistance exercise in men. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2016, 72, 1319–1326. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.; Sun, B.; Yin, X.; Guo, X.; Chao, D.; Zhang, C.; Zhang, C.-Y.; Chen, X.; Ma, J. Time-course responses of circulating microRNAs to three resistance training protocols in healthy young men. Sci. Rep. 2017, 7, 2203. [Google Scholar] [CrossRef]
- D’Souza, R.F.; Woodhead, J.S.; Zeng, N.; Blenkiron, C.; Merry, T.L.; Cameron-Smith, D.; Mitchell, C.J. Circulatory exosomal MIRNA following intense exercise is unrelated to muscle and plasma MIRNA abundances. Am. J. Physiol. Endocrinol. Metab. 2018, 315, E723–E733. [Google Scholar] [CrossRef]
- Silver, J.L.; Alexander, S.E.; Dillon, H.T.; Lamon, S.; Wadley, G.D. Extracellular vesicular MIRNA expression is not a proxy for skeletal muscle MIRNA expression in males and females following acute, moderate intensity exercise. Physiol. Rep. 2020, 8, e14520. [Google Scholar] [CrossRef]
- Nederveen, J.P.; Warnier, G.; Di Carlo, A.; Nilsson, M.I.; Tarnopolsky, M.A. Extracellular vesicles and exosomes: Insights from exercise science. Front. Physiol. 2021, 11, 604274. [Google Scholar] [CrossRef]
- Nair, V.D.; Ge, Y.; Li, S.; Pincas, H.; Jain, N.; Seenarine, N.; Amper, M.A.; Goodpaster, B.H.; Walsh, M.J.; Coen, P.M.; et al. Sedentary and trained older men have distinct circulating exosomal microRNA profiles at baseline and in response to acute exercise. Front. Physiol. 2020, 11, 605. [Google Scholar] [CrossRef]
- Garai, K.; Adam, Z.; Herczeg, R.; Banfai, K.; Gyebrovszki, A.; Gyenesei, A.; Pongracz, J.E.; Wilhelm, M.; Kvell, K. Physical activity as a preventive lifestyle intervention acts through specific exosomal Mirna species—Evidence from human short- and long-term pilot studies. Front. Physiol. 2021, 12, 658218. [Google Scholar] [CrossRef]
- Ramos, A.E.; Lo, C.; Estephan, L.E.; Tai, Y.-Y.; Tang, Y.; Zhao, J.; Sugahara, M.; Gorcsan, J.; Brown, M.G.; Lieberman, D.E.; et al. Specific circulating microRNAs display dose-dependent responses to variable intensity and duration of endurance exercise. Am. J. Physiol. Heart Circ. Physiol. 2018, 315, H273–H283. [Google Scholar] [CrossRef]
- Castaño, C.; Mirasierra, M.; Vallejo, M.; Novials, A.; Párrizas, M. Delivery of muscle-derived exosomal miRNAs induced by HIIT improves insulin sensitivity through down-regulation of hepatic FOXO1 in mice. Proc. Natl. Acad. Sci. USA 2020, 117, 30335–30343. [Google Scholar] [CrossRef] [PubMed]
- Siqueira, I.R.; Palazzo, R.P.; Cechinel, L.R. Circulating extracellular vesicles delivering beneficial cargo as key players in exercise effects. Free. Radic. Biol. Med. 2021, 172, 273–285. [Google Scholar] [CrossRef] [PubMed]
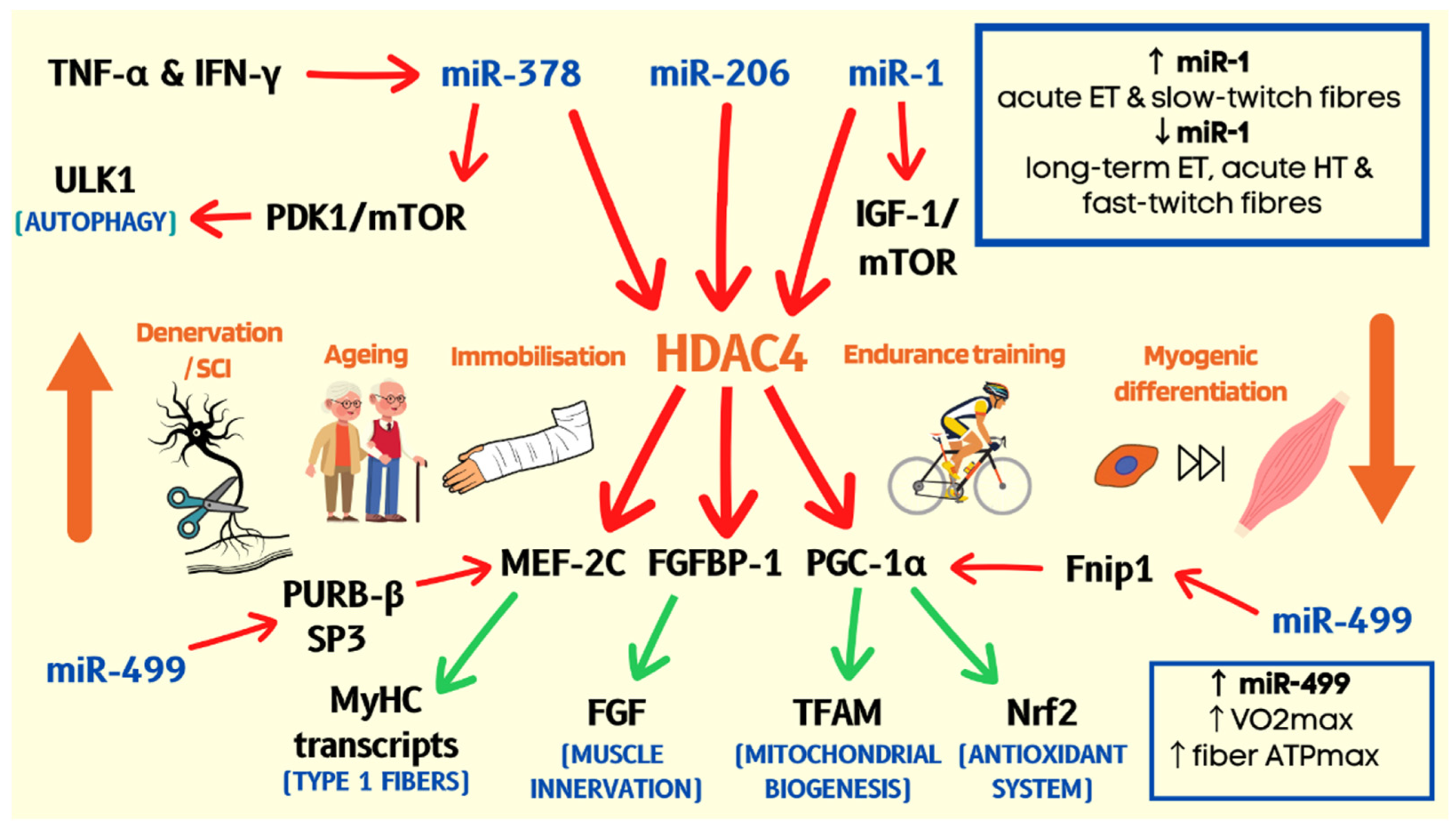
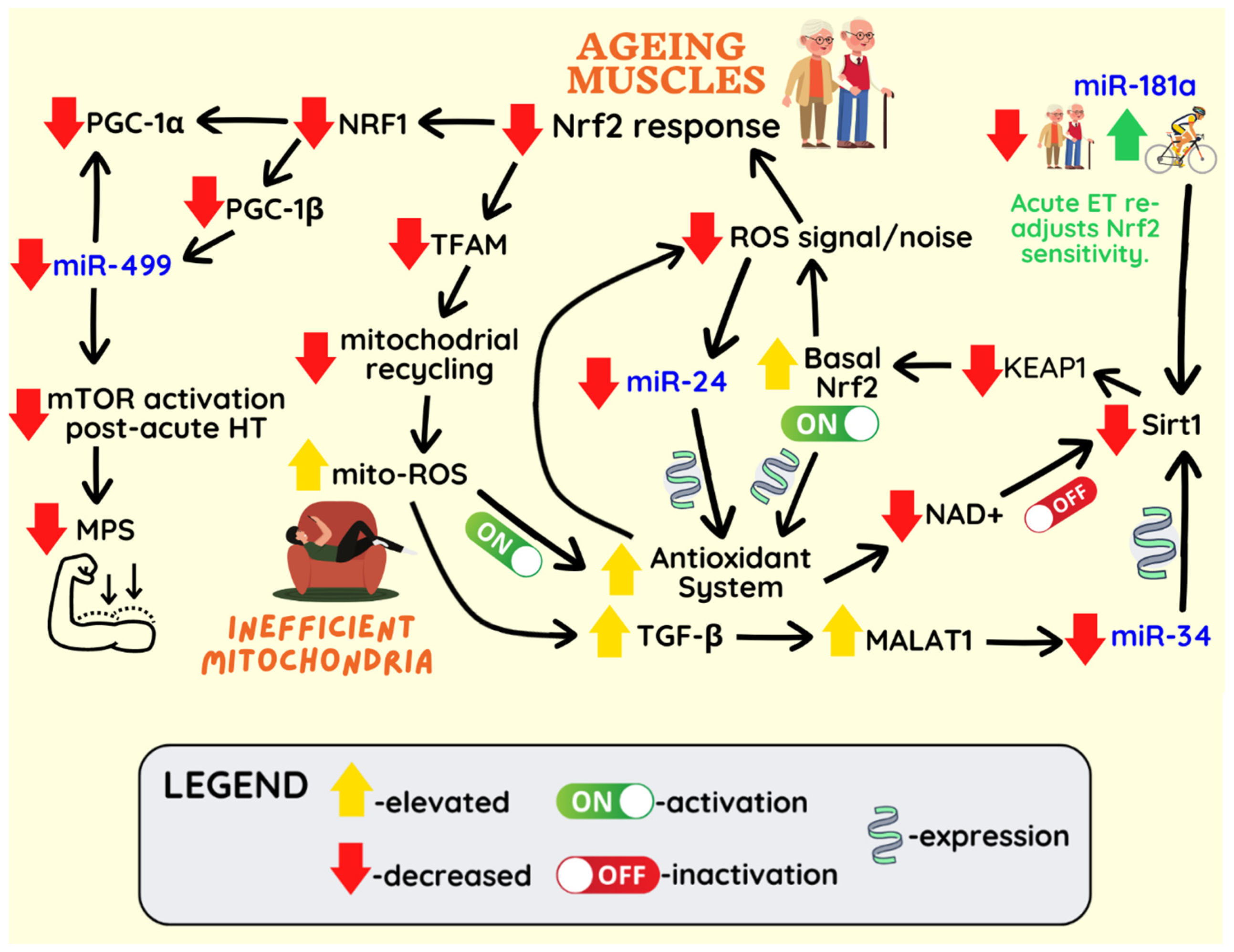

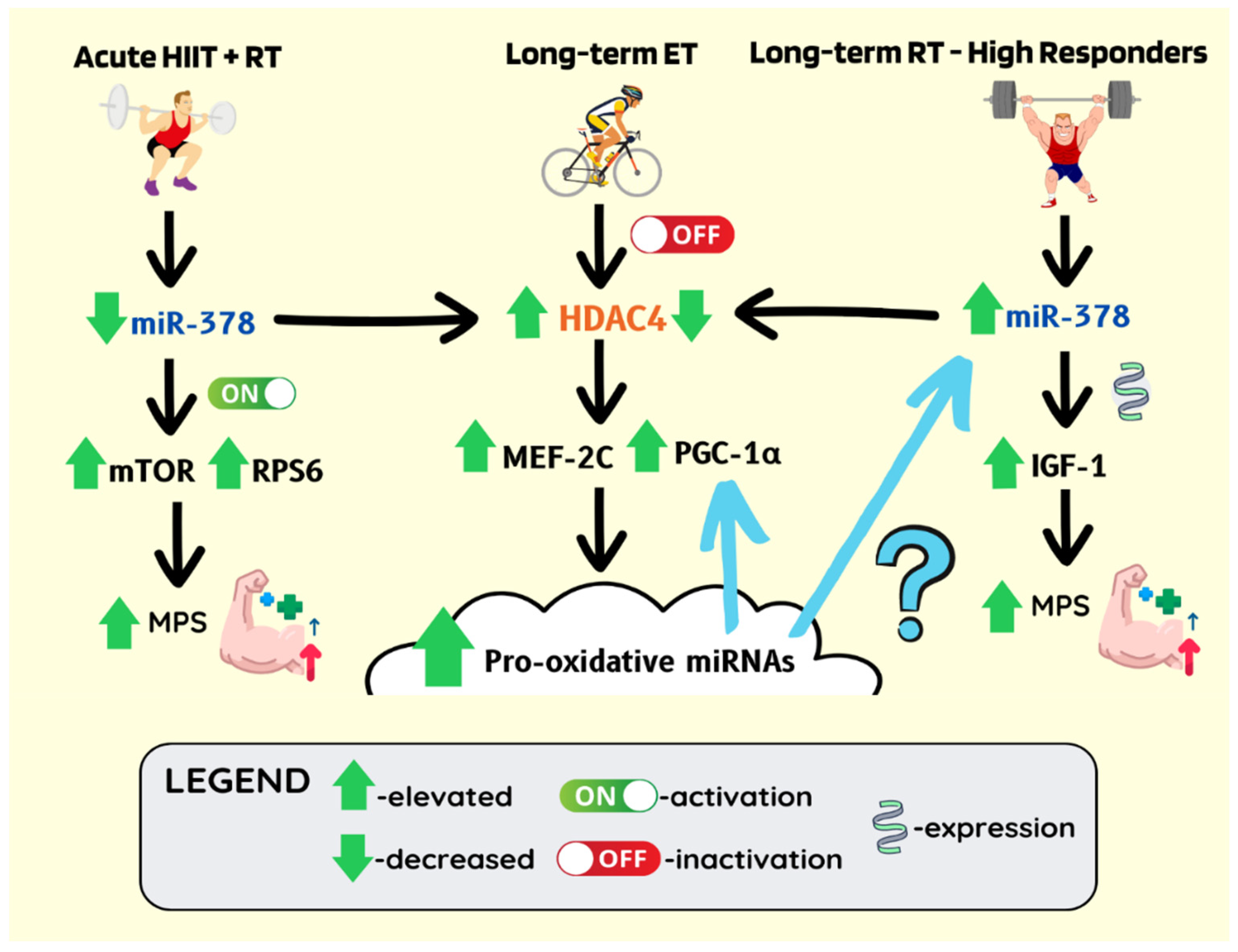
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kolodziej, F.; McDonagh, B.; Burns, N.; Goljanek-Whysall, K. MicroRNAs as the Sentinels of Redox and Hypertrophic Signalling. Int. J. Mol. Sci. 2022, 23, 14716. https://doi.org/10.3390/ijms232314716
Kolodziej F, McDonagh B, Burns N, Goljanek-Whysall K. MicroRNAs as the Sentinels of Redox and Hypertrophic Signalling. International Journal of Molecular Sciences. 2022; 23(23):14716. https://doi.org/10.3390/ijms232314716
Chicago/Turabian StyleKolodziej, Filip, Brian McDonagh, Nicole Burns, and Katarzyna Goljanek-Whysall. 2022. "MicroRNAs as the Sentinels of Redox and Hypertrophic Signalling" International Journal of Molecular Sciences 23, no. 23: 14716. https://doi.org/10.3390/ijms232314716
APA StyleKolodziej, F., McDonagh, B., Burns, N., & Goljanek-Whysall, K. (2022). MicroRNAs as the Sentinels of Redox and Hypertrophic Signalling. International Journal of Molecular Sciences, 23(23), 14716. https://doi.org/10.3390/ijms232314716






