Tirzepatide: A Systematic Update
Abstract
1. Introduction
2. Tirzepatide in the SURPASS Trials
3. Tirzepatide and Obesity: The SURMOUNT-1 Trial
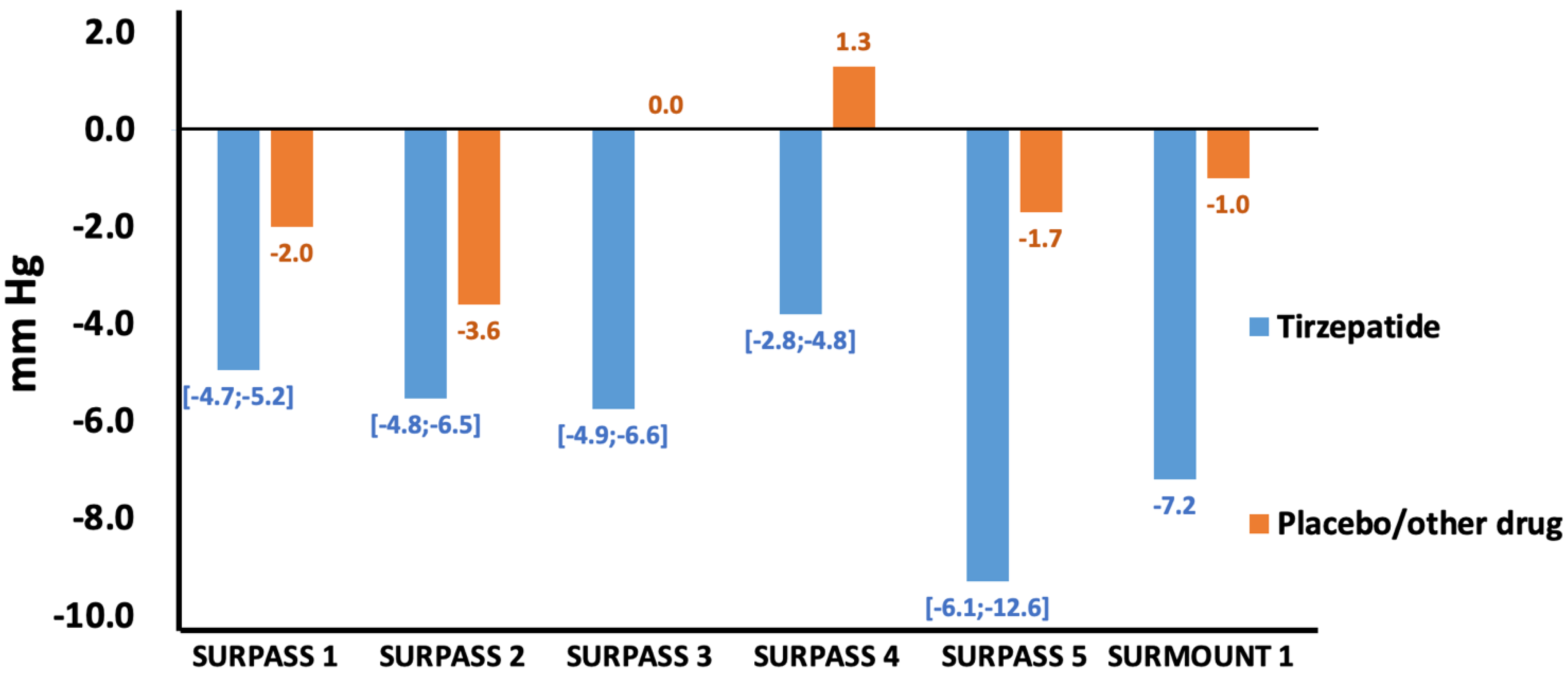
4. Tirzepatide and Hypertension
5. Data from Meta-Analyses
6. New Perspectives
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Visseren, F.L.J.; Mach, F.; Smulders, Y.M.; Carballo, D.; Koskinas, K.C.; Back, M.; Benetos, A.; Biffi, A.; Boavida, J.M.; Capodanno, D.; et al. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice: Developed by the Task Force for cardiovascular disease prevention in clinical practice with representatives of the European Society of Cardiology and 12 medical societies with the special contribution of the European Association of Preventive Cardiology (EAPC). Rev. Esp. Cardiol. 2022, 75, 429. [Google Scholar] [CrossRef] [PubMed]
- Joseph, J.J.; Deedwania, P.; Acharya, T.; Aguilar, D.; Bhatt, D.L.; Chyun, D.A.; Di Palo, K.E.; Golden, S.H.; Sperling, L.S.; American Heart Association Diabetes Committee of the Council on Lifestyle and Cardiometabolic Health; et al. Comprehensive Management of Cardiovascular Risk Factors for Adults with Type 2 Diabetes: A Scientific Statement from the American Heart Association. Circulation 2022, 145, e722–e759. [Google Scholar] [CrossRef] [PubMed]
- Bulum, T. Nephroprotective Properties of the Glucose-Dependent Insulinotropic Polypeptide (GIP) and Glucagon-like Peptide-1 (GLP-1) Receptor Agonists. Biomedicines 2022, 10, 2586. [Google Scholar] [CrossRef] [PubMed]
- Rehfeld, J.F. The Origin and Understanding of the Incretin Concept. Front. Endocrinol. 2018, 9, 387. [Google Scholar] [CrossRef] [PubMed]
- Holst, J.J. From the Incretin Concept and the Discovery of GLP-1 to Today’s Diabetes Therapy. Front. Endocrinol. 2019, 10, 260. [Google Scholar] [CrossRef] [PubMed]
- Nauck, M.A.; Quast, D.R.; Wefers, J.; Pfeiffer, A.F.H. The evolving story of incretins (GIP and GLP-1) in metabolic and cardiovascular disease: A pathophysiological update. Diabetes Obes. Metab. 2021, 23 (Suppl. 3), 5–29. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Nian, C.; McIntosh, C.H. GIP increases human adipocyte LPL expression through CREB and TORC2-mediated trans-activation of the LPL gene. J. Lipid Res. 2010, 51, 3145–3157. [Google Scholar] [CrossRef]
- Baggio, L.L.; Drucker, D.J. Biology of incretins: GLP-1 and GIP. Gastroenterology 2007, 132, 2131–2157. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.S.; Seeley, R.J.; Sandoval, D.A. Signalling from the periphery to the brain that regulates energy homeostasis. Nat. Rev. Neurosci. 2018, 19, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Wang, L.; Zhou, S.; Zhou, C.; Xie, L.; Chen, J.; Tang, D.; Tian, X.; Xie, D.; Ding, J.; et al. A double-blind, randomized, placebo and positive-controlled study in healthy volunteers to evaluate pharmacokinetic and pharmacodynamic properties of multiple oral doses of cetagliptin. Br. J. Clin. Pharmacol 2022, 88, 2946–2958. [Google Scholar] [CrossRef] [PubMed]
- Chai, S.; Zhang, R.; Zhang, Y.; Carr, R.D.; Zheng, Y.; Rajpathak, S.; Ji, L. Effect of dipeptidyl peptidase-4 inhibitors on postprandial glucagon level in patients with type 2 diabetes mellitus: A systemic review and meta-analysis. Front. Endocrinol. 2022, 13, 994944. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, M.P.; Pratley, R.E. GLP-1 Analogs and DPP-4 Inhibitors in Type 2 Diabetes Therapy: Review of Head-to-Head Clinical Trials. Front. Endocrinol. 2020, 11, 178. [Google Scholar] [CrossRef] [PubMed]
- Sanada, J.; Kimura, T.; Shimoda, M.; Tomita, A.; Fushimi, Y.; Kinoshita, T.; Obata, A.; Okauchi, S.; Hirukawa, H.; Kohara, K.; et al. Switching from Daily DPP-4 Inhibitor to Once-Weekly GLP-1 Receptor Activator Dulaglutide Significantly Ameliorates Glycemic Control in Subjects with Poorly Controlled Type 2 Diabetes Mellitus: A Retrospective Observational Study. Front. Endocrinol. 2021, 12, 714447. [Google Scholar] [CrossRef] [PubMed]
- Tan, Q.; Akindehin, S.E.; Orsso, C.E.; Waldner, R.C.; DiMarchi, R.D.; Muller, T.D.; Haqq, A.M. Recent Advances in Incretin-Based Pharmacotherapies for the Treatment of Obesity and Diabetes. Front. Endocrinol. 2022, 13, 838410. [Google Scholar] [CrossRef] [PubMed]
- Samms, R.J.; Coghlan, M.P.; Sloop, K.W. How May GIP Enhance the Therapeutic Efficacy of GLP-1? Trends Endocrinol. Metab. 2020, 31, 410–421. [Google Scholar] [CrossRef] [PubMed]
- Novikoff, A.; O’Brien, S.L.; Bernecker, M.; Grandl, G.; Kleinert, M.; Knerr, P.J.; Stemmer, K.; Klingenspor, M.; Zeigerer, A.; DiMarchi, R.; et al. Spatiotemporal GLP-1 and GIP receptor signaling and trafficking/recycling dynamics induced by selected receptor mono- and dual-agonists. Mol. Metab. 2021, 49, 101181. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.K.; Nikooienejad, A.; Bray, R.; Cui, X.; Wilson, J.; Duffin, K.; Milicevic, Z.; Haupt, A.; Robins, D.A. Dual GIP and GLP-1 Receptor Agonist Tirzepatide Improves Beta-cell Function and Insulin Sensitivity in Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2021, 106, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Baggio, L.L.; Drucker, D.J. Glucagon-like peptide-1 receptor co-agonists for treating metabolic disease. Mol. Metab. 2021, 46, 101090. [Google Scholar] [CrossRef] [PubMed]
- Portron, A.; Jadidi, S.; Sarkar, N.; DiMarchi, R.; Schmitt, C. Pharmacodynamics, pharmacokinetics, safety and tolerability of the novel dual glucose-dependent insulinotropic polypeptide/glucagon-like peptide-1 agonist RG7697 after single subcutaneous administration in healthy subjects. Diabetes Obes. Metab. 2017, 19, 1446–1453. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, C.; Portron, A.; Jadidi, S.; Sarkar, N.; DiMarchi, R. Pharmacodynamics, pharmacokinetics and safety of multiple ascending doses of the novel dual glucose-dependent insulinotropic polypeptide/glucagon-like peptide-1 agonist RG7697 in people with type 2 diabetes mellitus. Diabetes Obes. Metab. 2017, 19, 1436–1445. [Google Scholar] [CrossRef] [PubMed]
- Norregaard, P.K.; Deryabina, M.A.; Tofteng Shelton, P.; Fog, J.U.; Daugaard, J.R.; Eriksson, P.O.; Larsen, L.F.; Jessen, L. A novel GIP analogue, ZP4165, enhances glucagon-like peptide-1-induced body weight loss and improves glycaemic control in rodents. Diabetes Obes. Metab. 2018, 20, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Finan, B.; Ma, T.; Ottaway, N.; Muller, T.D.; Habegger, K.M.; Heppner, K.M.; Kirchner, H.; Holland, J.; Hembree, J.; Raver, C.; et al. Unimolecular dual incretins maximize metabolic benefits in rodents, monkeys, and humans. Sci. Transl. Med. 2013, 5, 209ra151. [Google Scholar] [CrossRef] [PubMed]
- Fisman, E.Z.; Tenenbaum, A. The dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist tirzepatide: A novel cardiometabolic therapeutic prospect. Cardiovasc. Diabetol. 2021, 20, 225. [Google Scholar] [CrossRef] [PubMed]
- Samms, R.J.; Christe, M.E.; Collins, K.A.; Pirro, V.; Droz, B.A.; Holland, A.K.; Friedrich, J.L.; Wojnicki, S.; Konkol, D.L.; Cosgrove, R.; et al. GIPR agonism mediates weight-independent insulin sensitization by tirzepatide in obese mice. J. Clin. Investig. 2021, 131, e146353. [Google Scholar] [CrossRef] [PubMed]
- Frias, J.P.; Bastyr, E.J., 3rd; Vignati, L.; Tschop, M.H.; Schmitt, C.; Owen, K.; Christensen, R.H.; DiMarchi, R.D. The Sustained Effects of a Dual GIP/GLP-1 Receptor Agonist, NNC0090-2746, in Patients with Type 2 Diabetes. Cell Metab. 2017, 26, 343–352.e342. [Google Scholar] [CrossRef]
- Frias, J.P.; Nauck, M.A.; Van, J.; Kutner, M.E.; Cui, X.; Benson, C.; Urva, S.; Gimeno, R.E.; Milicevic, Z.; Robins, D.; et al. Efficacy and safety of LY3298176, a novel dual GIP and GLP-1 receptor agonist, in patients with type 2 diabetes: A randomised, placebo-controlled and active comparator-controlled phase 2 trial. Lancet 2018, 392, 2180–2193. [Google Scholar] [CrossRef] [PubMed]
- Frias, J.P.; Nauck, M.A.; Van, J.; Benson, C.; Bray, R.; Cui, X.; Milicevic, Z.; Urva, S.; Haupt, A.; Robins, D.A. Efficacy and tolerability of tirzepatide, a dual glucose-dependent insulinotropic peptide and glucagon-like peptide-1 receptor agonist in patients with type 2 diabetes: A 12-week, randomized, double-blind, placebo-controlled study to evaluate different dose-escalation regimens. Diabetes Obes. Metab. 2020, 22, 938–946. [Google Scholar] [CrossRef] [PubMed]
- Frias, J.P. Tirzepatide: A glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) dual agonist in development for the treatment of type 2 diabetes. Expert Rev. Endocrinol. Metab. 2020, 15, 379–394. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Willard, F.S.; Feng, D.; Alsina-Fernandez, J.; Chen, Q.; Vieth, M.; Ho, J.D.; Showalter, A.D.; Stutsman, C.; Ding, L.; et al. Structural determinants of dual incretin receptor agonism by tirzepatide. Proc. Natl. Acad. Sci. USA 2022, 119, e2116506119. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Zhou, Q.; Cong, Z.; Hang, K.; Zou, X.; Zhang, C.; Chen, Y.; Dai, A.; Liang, A.; Ming, Q.; et al. Structural insights into multiplexed pharmacological actions of tirzepatide and peptide 20 at the GIP, GLP-1 or glucagon receptors. Nat. Commun. 2022, 13, 1057. [Google Scholar] [CrossRef] [PubMed]
- Coskun, T.; Sloop, K.W.; Loghin, C.; Alsina-Fernandez, J.; Urva, S.; Bokvist, K.B.; Cui, X.; Briere, D.A.; Cabrera, O.; Roell, W.C.; et al. LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: From discovery to clinical proof of concept. Mol. Metab. 2018, 18, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Willard, F.S.; Douros, J.D.; Gabe, M.B.; Showalter, A.D.; Wainscott, D.B.; Suter, T.M.; Capozzi, M.E.; van der Velden, W.J.; Stutsman, C.; Cardona, G.R.; et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight 2020, 5, e140532. [Google Scholar] [CrossRef] [PubMed]
- Santulli, G. Tirzepatide versus Semaglutide Once Weekly in Type 2 Diabetes. N. Engl. J. Med. 2022, 386, e17. [Google Scholar] [CrossRef] [PubMed]
- De Block, C.; Bailey, C.; Wysham, C.; Hemmingway, A.; Allen, S.E.; Peleshok, J. Tirzepatide for the treatment of adults with type 2 diabetes: An endocrine perspective. Diabetes Obes. Metab. 2022. [Google Scholar] [CrossRef] [PubMed]
- Min, T.; Bain, S.C. The Role of Tirzepatide, Dual GIP and GLP-1 Receptor Agonist, in the Management of Type 2 Diabetes: The SURPASS Clinical Trials. Diabetes Ther. 2021, 12, 143–157. [Google Scholar] [CrossRef] [PubMed]
- Greenhill, C. Testing a novel dual receptor agonist for treatment of type 2 diabetes mellitus. Nat. Rev. Endocrinol. 2018, 14, 687. [Google Scholar] [CrossRef] [PubMed]
- Stumvoll, M.; Tschop, M. Twice the benefits with twincretins? Lancet 2018, 392, 2142–2144. [Google Scholar] [CrossRef] [PubMed]
- Rosenstock, J.; Wysham, C.; Frias, J.P.; Kaneko, S.; Lee, C.J.; Fernandez Lando, L.; Mao, H.; Cui, X.; Karanikas, C.A.; Thieu, V.T. Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): A double-blind, randomised, phase 3 trial. Lancet 2021, 398, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Paneni, F.; Patrono, C. Is tirzepatide in the surpass lane over GLP-1 receptor agonists for the treatment of diabetes? Eur. Heart J. 2021, 42, 4211–4212. [Google Scholar] [CrossRef] [PubMed]
- Tan, T.M.; Khoo, B. Tirzepatide and the new era of twincretins for diabetes. Lancet 2021, 398, 95–97. [Google Scholar] [CrossRef] [PubMed]
- Frias, J.P.; Davies, M.J.; Rosenstock, J.; Perez Manghi, F.C.; Fernandez Lando, L.; Bergman, B.K.; Liu, B.; Cui, X.; Brown, K.; SURPASS-2 Investigators. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N. Engl. J. Med. 2021, 385, 503–515. [Google Scholar] [CrossRef] [PubMed]
- Chiu, N.; Aggarwal, R.; Bhatt, D.L. Generalizability of the SURPASS-2 Trial and Effect of Tirzepatide on US Diabetes and Obesity Control. J. Am. Heart Assoc. 2022, 11, e026297. [Google Scholar] [CrossRef] [PubMed]
- Vadher, K.; Patel, H.; Mody, R.; Levine, J.A.; Hoog, M.; Cheng, A.Y.; Pantalone, K.M.; Sapin, H. Efficacy of tirzepatide 5, 10 and 15 mg versus semaglutide 2 mg in patients with type 2 diabetes: An adjusted indirect treatment comparison. Diabetes Obes. Metab. 2022, 24, 1861–1868. [Google Scholar] [CrossRef] [PubMed]
- Ludvik, B.; Giorgino, F.; Jodar, E.; Frias, J.P.; Fernandez Lando, L.; Brown, K.; Bray, R.; Rodriguez, A. Once-weekly tirzepatide versus once-daily insulin degludec as add-on to metformin with or without SGLT2 inhibitors in patients with type 2 diabetes (SURPASS-3): A randomised, open-label, parallel-group, phase 3 trial. Lancet 2021, 398, 583–598. [Google Scholar] [CrossRef]
- Bailey, C.J. Tirzepatide: A new low for bodyweight and blood glucose. Lancet Diabetes Endocrinol. 2021, 9, 646–648. [Google Scholar] [CrossRef]
- Battelino, T.; Bergenstal, R.M.; Rodriguez, A.; Fernandez Lando, L.; Bray, R.; Tong, Z.; Brown, K. Efficacy of once-weekly tirzepatide versus once-daily insulin degludec on glycaemic control measured by continuous glucose monitoring in adults with type 2 diabetes (SURPASS-3 CGM): A substudy of the randomised, open-label, parallel-group, phase 3 SURPASS-3 trial. Lancet Diabetes Endocrinol. 2022, 10, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Gastaldelli, A.; Cusi, K.; Fernandez Lando, L.; Bray, R.; Brouwers, B.; Rodriguez, A. Effect of tirzepatide versus insulin degludec on liver fat content and abdominal adipose tissue in people with type 2 diabetes (SURPASS-3 MRI): A substudy of the randomised, open-label, parallel-group, phase 3 SURPASS-3 trial. Lancet Diabetes Endocrinol. 2022, 10, 393–406. [Google Scholar] [CrossRef] [PubMed]
- Targher, G. Tirzepatide adds hepatoprotection to its armoury. Lancet Diabetes Endocrinol. 2022, 10, 374–375. [Google Scholar] [CrossRef]
- Heise, T.; Mari, A.; DeVries, J.H.; Urva, S.; Li, J.; Pratt, E.J.; Coskun, T.; Thomas, M.K.; Mather, K.J.; Haupt, A.; et al. Effects of subcutaneous tirzepatide versus placebo or semaglutide on pancreatic islet function and insulin sensitivity in adults with type 2 diabetes: A multicentre, randomised, double-blind, parallel-arm, phase 1 clinical trial. Lancet Diabetes Endocrinol. 2022, 10, 418–429. [Google Scholar] [CrossRef]
- Scheen, A.J. Add-on value of tirzepatide versus semaglutide. Lancet Diabetes Endocrinol. 2022, 10, 377–378. [Google Scholar] [CrossRef]
- Starling, S. GIP-GLP1 receptor agonist shows promise. Nat. Rev. Endocrinol. 2022, 18, 391. [Google Scholar] [CrossRef] [PubMed]
- Del Prato, S.; Kahn, S.E.; Pavo, I.; Weerakkody, G.J.; Yang, Z.; Doupis, J.; Aizenberg, D.; Wynne, A.G.; Riesmeyer, J.S.; Heine, R.J.; et al. Tirzepatide versus insulin glargine in type 2 diabetes and increased cardiovascular risk (SURPASS-4): A randomised, open-label, parallel-group, multicentre, phase 3 trial. Lancet 2021, 398, 1811–1824. [Google Scholar] [CrossRef] [PubMed]
- Singh, M. In type 2 diabetes with increased CV risk, tirzepatide reduced HbA1c vs. glargine at 52 wk. Ann. Intern. Med. 2022, 175, JC34. [Google Scholar] [CrossRef] [PubMed]
- Khoo, B.; Tan, T.M. Surpassing insulin glargine in type 2 diabetes with tirzepatide. Lancet 2021, 398, 1779–1781. [Google Scholar] [CrossRef] [PubMed]
- Heerspink, H.J.L.; Sattar, N.; Pavo, I.; Haupt, A.; Duffin, K.L.; Yang, Z.; Wiese, R.J.; Tuttle, K.R.; Cherney, D.Z.I. Effects of tirzepatide versus insulin glargine on kidney outcomes in type 2 diabetes in the SURPASS-4 trial: Post-hoc analysis of an open-label, randomised, phase 3 trial. Lancet Diabetes Endocrinol. 2022, 10, 774–785. [Google Scholar] [CrossRef] [PubMed]
- Dahl, D.; Onishi, Y.; Norwood, P.; Huh, R.; Bray, R.; Patel, H.; Rodriguez, A. Effect of Subcutaneous Tirzepatide vs Placebo Added to Titrated Insulin Glargine on Glycemic Control in Patients with Type 2 Diabetes: The SURPASS-5 Randomized Clinical Trial. JAMA 2022, 327, 534–545. [Google Scholar] [CrossRef] [PubMed]
- Chipkin, S.R. Tirzepatide for Patients with Type 2 Diabetes. JAMA 2022, 327, 529–530. [Google Scholar] [CrossRef]
- Inagaki, N.; Takeuchi, M.; Oura, T.; Imaoka, T.; Seino, Y. Efficacy and safety of tirzepatide monotherapy compared with dulaglutide in Japanese patients with type 2 diabetes (SURPASS J-mono): A double-blind, multicentre, randomised, phase 3 trial. Lancet Diabetes Endocrinol. 2022, 10, 623–633. [Google Scholar] [CrossRef]
- Yabe, D.; Kawamori, D.; Seino, Y.; Oura, T.; Takeuchi, M. Change of pharmacodynamics parameters following once-weekly tirzepatide treatment versus dulaglutide in Japanese patients with type 2 diabetes (SURPASS J-mono sub-study). Diabetes Obes. Metab. 2022. [Google Scholar] [CrossRef]
- Kadowaki, T.; Chin, R.; Ozeki, A.; Imaoka, T.; Ogawa, Y. Safety and efficacy of tirzepatide as an add-on to single oral antihyperglycaemic medication in patients with type 2 diabetes in Japan (SURPASS J-combo): A multicentre, randomised, open-label, parallel-group, phase 3 trial. Lancet Diabetes Endocrinol. 2022, 10, 634–644. [Google Scholar] [CrossRef] [PubMed]
- Emerging Risk Factors, C.; Wormser, D.; Kaptoge, S.; Di Angelantonio, E.; Wood, A.M.; Pennells, L.; Thompson, A.; Sarwar, N.; Kizer, J.R.; Lawlor, D.A.; et al. Separate and combined associations of body-mass index and abdominal adiposity with cardiovascular disease: Collaborative analysis of 58 prospective studies. Lancet 2011, 377, 1085–1095. [Google Scholar] [CrossRef] [PubMed]
- Siren, R.; Eriksson, J.G.; Vanhanen, H. Waist circumference a good indicator of future risk for type 2 diabetes and cardiovascular disease. BMC Public Health 2012, 12, 631. [Google Scholar] [CrossRef] [PubMed]
- Csige, I.; Ujvarosy, D.; Szabo, Z.; Lorincz, I.; Paragh, G.; Harangi, M.; Somodi, S. The Impact of Obesity on the Cardiovascular System. J. Diabetes Res. 2018, 2018, 3407306. [Google Scholar] [CrossRef] [PubMed]
- Lazzaroni, E.; Ben Nasr, M.; Loretelli, C.; Pastore, I.; Plebani, L.; Lunati, M.E.; Vallone, L.; Bolla, A.M.; Rossi, A.; Montefusco, L.; et al. Anti-diabetic drugs and weight loss in patients with type 2 diabetes. Pharmacol. Res. 2021, 171, 105782. [Google Scholar] [CrossRef] [PubMed]
- Powell-Wiley, T.M.; Poirier, P.; Burke, L.E.; Despres, J.P.; Gordon-Larsen, P.; Lavie, C.J.; Lear, S.A.; Ndumele, C.E.; Neeland, I.J.; Sanders, P.; et al. Obesity and Cardiovascular Disease: A Scientific Statement from the American Heart Association. Circulation 2021, 143, e984–e1010. [Google Scholar] [CrossRef] [PubMed]
- Jastreboff, A.M.; Aronne, L.J.; Ahmad, N.N.; Wharton, S.; Connery, L.; Alves, B.; Kiyosue, A.; Zhang, S.; Liu, B.; Bunck, M.C.; et al. Tirzepatide Once Weekly for the Treatment of Obesity. N. Engl. J. Med. 2022, 387, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Ryan, D.H.; Yockey, S.R. Weight Loss and Improvement in Comorbidity: Differences at 5%, 10%, 15%, and Over. Curr. Obes. Rep. 2017, 6, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Davidson, M.B. In adults with obesity without diabetes, adding tirzepatide to a lifestyle intervention increased weight loss at 72 wk. Ann. Intern. Med. 2022, 175, JC116. [Google Scholar] [CrossRef] [PubMed]
- Hindson, J. Tirzepatide to treat obesity: Phase III results. Nat. Rev. Gastroenterol. Hepatol. 2022, 19, 488. [Google Scholar] [CrossRef] [PubMed]
- Tysoe, O. Tirzepatide highly effective for weight loss. Nat. Rev. Endocrinol. 2022, 18, 520. [Google Scholar] [CrossRef]
- Frachon, I.; Etienne, Y.; Jobic, Y.; Le Gal, G.; Humbert, M.; Leroyer, C. Benfluorex and unexplained valvular heart disease: A case-control study. PLoS ONE 2010, 5, e10128. [Google Scholar] [CrossRef] [PubMed]
- Ruckert, I.M.; Baumert, J.; Schunk, M.; Holle, R.; Schipf, S.; Volzke, H.; Kluttig, A.; Greiser, K.H.; Tamayo, T.; Rathmann, W.; et al. Blood Pressure Control Has Improved in People with and without Type 2 Diabetes but Remains Suboptimal: A Longitudinal Study Based on the German DIAB-CORE Consortium. PLoS ONE 2015, 10, e0133493. [Google Scholar] [CrossRef] [PubMed]
- Trimarco, V.; Izzo, R.; Mone, P.; Lembo, M.; Manzi, M.V.; Pacella, D.; Falco, A.; Gallo, P.; Esposito, G.; Morisco, C.; et al. Therapeutic concordance improves blood pressure control in patients with resistant hypertension. Pharmacol. Res. 2022, 106557. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.M.; Lin, Y.; Luo, M.J.; Considine, G.; Cox, A.L.; Bowsman, L.M.; Robins, D.A.; Haupt, A.; Duffin, K.L.; Ruotolo, G. The dual glucose-dependent insulinotropic polypeptide and glucagon-like peptide-1 receptor agonist tirzepatide improves cardiovascular risk biomarkers in patients with type 2 diabetes: A post hoc analysis. Diabetes Obes. Metab. 2022, 24, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Frias, J.P.; Fernandez Lando, L.; Brown, K. Tirzepatide versus Semaglutide Once Weekly in Type 2 Diabetes. Reply. N. Engl. J. Med. 2022, 386, e17. [Google Scholar] [CrossRef]
- Sattar, N.; McGuire, D.K.; Pavo, I.; Weerakkody, G.J.; Nishiyama, H.; Wiese, R.J.; Zoungas, S. Tirzepatide cardiovascular event risk assessment: A pre-specified meta-analysis. Nat. Med. 2022, 28, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Karagiannis, T.; Avgerinos, I.; Liakos, A.; Del Prato, S.; Matthews, D.R.; Tsapas, A.; Bekiari, E. Management of type 2 diabetes with the dual GIP/GLP-1 receptor agonist tirzepatide: A systematic review and meta-analysis. Diabetologia 2022, 65, 1251–1261. [Google Scholar] [CrossRef] [PubMed]
- Bhagavathula, A.S.; Vidyasagar, K.; Tesfaye, W. Efficacy and Safety of Tirzepatide in Patients with Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis of Randomized Phase II/III Trials. Pharmaceuticals 2021, 14, 991. [Google Scholar] [CrossRef] [PubMed]
- Dutta, D.; Surana, V.; Singla, R.; Aggarwal, S.; Sharma, M. Efficacy and safety of novel twincretin tirzepatide a dual GIP and GLP-1 receptor agonist in the management of type-2 diabetes: A Cochrane meta-analysis. Indian J. Endocrinol. Metab. 2021, 25, 475–489. [Google Scholar] [CrossRef]
- Guan, R.; Yang, Q.; Yang, X.; Du, W.; Li, X.; Ma, G. Efficacy and safety of tirzepatide in patients with type 2 diabetes mellitus: A bayesian network meta-analysis. Front. Pharmacol. 2022, 13, 998816. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Hu, G.; Yin, S.; Yang, X.; Zhou, M.; Jian, W. Optimal dose of tirzepatide for type 2 diabetes mellitus: A meta-analysis and trial sequential analysis. Front. Cardiovasc. Med. 2022, 9, 990182. [Google Scholar] [CrossRef] [PubMed]
- Permana, H.; Yanto, T.A.; Hariyanto, T.I. Efficacy and safety of tirzepatide as novel treatment for type 2 diabetes: A systematic review and meta-analysis of randomized clinical trials. Diabetes Metab. Syndr. 2022, 16, 102640. [Google Scholar] [CrossRef] [PubMed]
- Lisco, G.; De Tullio, A.; Disoteo, O.; De Geronimo, V.; Piazzolla, G.; De Pergola, G.; Giagulli, V.A.; Jirillo, E.; Guastamacchia, E.; Sabba, C.; et al. Basal insulin intensification with GLP-1RA and dual GIP and GLP-1RA in patients with uncontrolled type 2 diabetes mellitus: A rapid review of randomized controlled trials and meta-analysis. Front. Endocrinol. 2022, 13, 920541. [Google Scholar] [CrossRef] [PubMed]
- Patoulias, D.; Doumas, M.; Papadopoulos, C. Meta-Analysis Assessing the Effect of Tirzepatide on the Risk for Atrial Fibrillation in Patients with Type 2 Diabetes Mellitus. Am. J. Cardiol. 2022, 173, 157–158. [Google Scholar] [CrossRef] [PubMed]
- Patoulias, D.; Papadopoulos, C.; Fragakis, N.; Doumas, M. Updated Meta-Analysis Assessing the Cardiovascular Efficacy of Tirzepatide. Am. J. Cardiol. 2022, 181, 139–140. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela-Vallejo, L.; Guatibonza-Garcia, V.; Mantzoros, C.S. Recent guidelines for Non-Alcoholic Fatty Liver disease (NAFLD)/Fatty Liver Disease (FLD): Are they already outdated and in need of supplementation? Metabolism 2022, 136, 155248. [Google Scholar] [CrossRef] [PubMed]
- Moura, F.A.; Scirica, B.M.; Ruff, C.T. Tirzepatide for diabetes: On track to SURPASS current therapy. Nat. Med. 2022, 28, 450–451. [Google Scholar] [CrossRef] [PubMed]
- Hartman, M.L.; Sanyal, A.J.; Loomba, R.; Wilson, J.M.; Nikooienejad, A.; Bray, R.; Karanikas, C.A.; Duffin, K.L.; Robins, D.A.; Haupt, A. Effects of Novel Dual GIP and GLP-1 Receptor Agonist Tirzepatide on Biomarkers of Nonalcoholic Steatohepatitis in Patients with Type 2 Diabetes. Diabetes Care 2020, 43, 1352–1355. [Google Scholar] [CrossRef] [PubMed]
- Pirro, V.; Roth, K.D.; Lin, Y.; Willency, J.A.; Milligan, P.L.; Wilson, J.M.; Ruotolo, G.; Haupt, A.; Newgard, C.B.; Duffin, K.L. Effects of Tirzepatide, a Dual GIP and GLP-1 RA, on Lipid and Metabolite Profiles in Subjects with Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2022, 107, 363–378. [Google Scholar] [CrossRef] [PubMed]
- Ferrannini, E. Tirzepatide as an Insulin Sensitizer. J. Clin. Endocrinol. Metab. 2022, 107, e1752–e1753. [Google Scholar] [CrossRef] [PubMed]
- Koufakis, T.; Maltese, G.; Kotsa, K. Toward a new model for the management of type 2 diabetes: The mountain is there and there is no other option than to climb it. Pharmacol. Res. 2022, 184, 106443. [Google Scholar] [CrossRef] [PubMed]
- Schnell, O.; Battelino, T.; Bergenstal, R.; Bluher, M.; Bohm, M.; Brosius, F.; Carr, R.D.; Ceriello, A.; Forst, T.; Giorgino, F.; et al. Report from the CVOT Summit 2021: New cardiovascular, renal, and glycemic outcomes. Cardiovasc. Diabetol. 2022, 21, 50. [Google Scholar] [CrossRef] [PubMed]
- Gallwitz, B. Clinical perspectives on the use of the GIP/GLP-1 receptor agonist tirzepatide for the treatment of type-2 diabetes and obesity. Front. Endocrinol. 2022, 13, 1004044. [Google Scholar] [CrossRef] [PubMed]
- Slomski, A. Tirzepatide Trial Demonstrates Substantial Weight Loss. JAMA 2022, 328, 322. [Google Scholar] [CrossRef] [PubMed]
- Mullard, A. Lilly’s tirzepatide secures first approval in diabetes, paving path for dual-acting hormone mimetics. Nat. Rev. Drug Discov. 2022, 21, 480. [Google Scholar] [CrossRef] [PubMed]
- Syed, Y.Y. Tirzepatide: First Approval. Drugs 2022, 82, 1213–1220. [Google Scholar] [CrossRef]
- Knerr, P.J.; Mowery, S.A.; Douros, J.D.; Premdjee, B.; Hjollund, K.R.; He, Y.; Kruse Hansen, A.M.; Olsen, A.K.; Perez-Tilve, D.; DiMarchi, R.D.; et al. Next generation GLP-1/GIP/glucagon triple agonists normalize body weight in obese mice. Mol. Metab. 2022, 63, 101533. [Google Scholar] [CrossRef] [PubMed]
- Capozzi, M.E.; DiMarchi, R.D.; Tschop, M.H.; Finan, B.; Campbell, J.E. Targeting the Incretin/Glucagon System With Triagonists to Treat Diabetes. Endocr. Rev. 2018, 39, 719–738. [Google Scholar] [CrossRef] [PubMed]
- Finan, B.; Yang, B.; Ottaway, N.; Smiley, D.L.; Ma, T.; Clemmensen, C.; Chabenne, J.; Zhang, L.; Habegger, K.M.; Fischer, K.; et al. A rationally designed monomeric peptide triagonist corrects obesity and diabetes in rodents. Nat. Med. 2015, 21, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Knerr, P.J.; Mowery, S.A.; Finan, B.; Perez-Tilve, D.; Tschop, M.H.; DiMarchi, R.D. Selection and progression of unimolecular agonists at the GIP, GLP-1, and glucagon receptors as drug candidates. Peptides 2020, 125, 170225. [Google Scholar] [CrossRef] [PubMed]
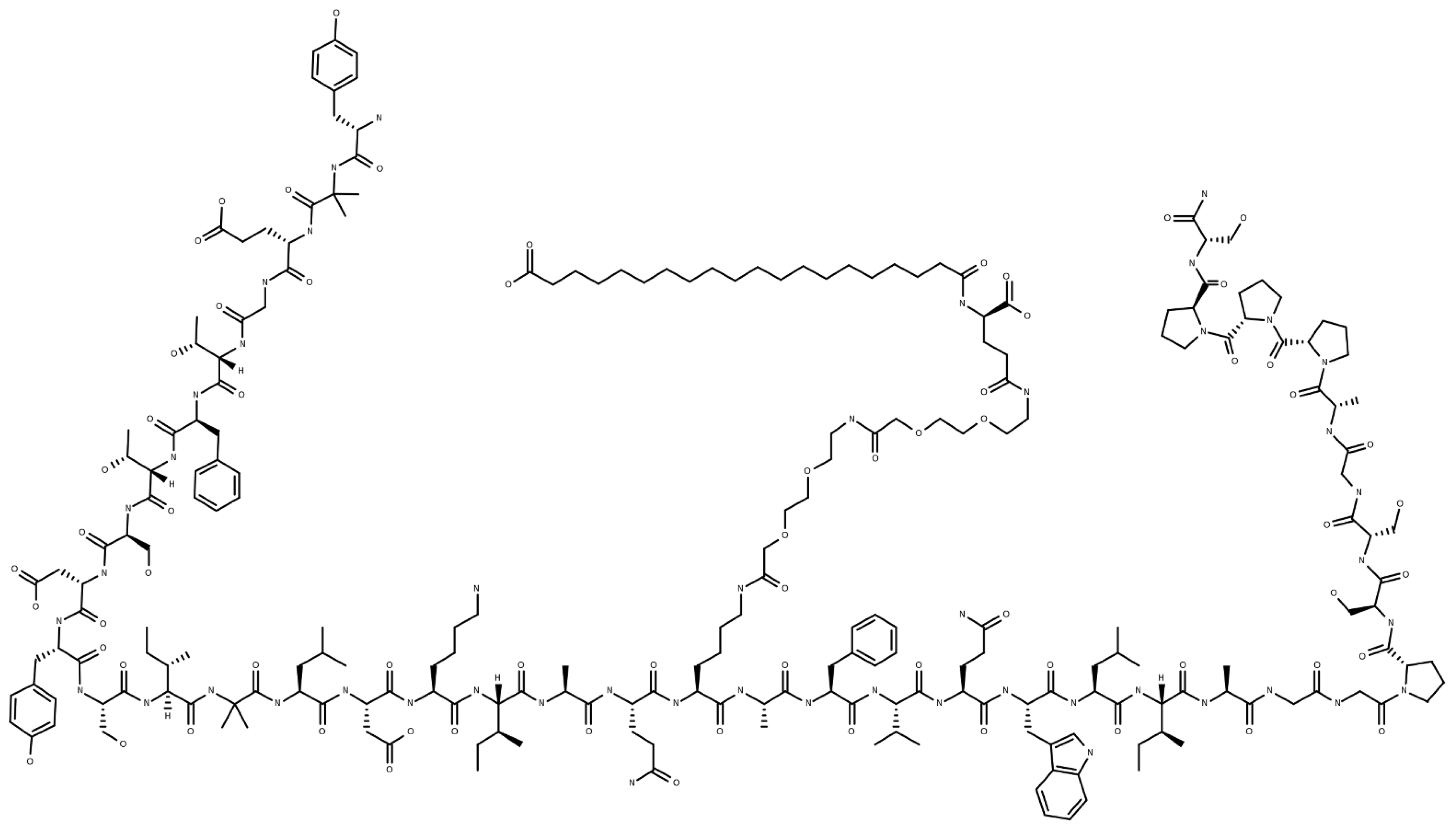
| Outcomes | Tirzepatide 5 mg (n = 121) | Tirzepatide 10 mg (n = 121) | Tirzepatide 15 mg (n = 120) | Placebo (n = 113) | |
|---|---|---|---|---|---|
| HbA1c (%) | Baseline | 7.97 | 7.88 | 7.88 | 8.08 |
| From baseline | −1.87 * | −1.89 * | −2.07 * | 0.04 ** | |
| Versus placebo | −1.91 * | −1.93 * | −2.11 * | - | |
| Weight (Kg) | From baseline | −7.0 * | −7.8 * | −9.5 * | −0.7 *** |
| Versus placebo | −6.3 * | −7.1 * | −8.8 * | - | |
| Outcomes | Tirzepatide 5 mg (n = 470) | Tirzepatide 10 mg (n = 469) | Tirzepatide 15 mg (n = 470) | Semaglutide (n = 469) | |
|---|---|---|---|---|---|
| HbA1c (%) | Baseline | 8.32 | 8.30 | 8.26 | 8.25 |
| From baseline | −2.01 | −2.24 | −2.30 | −1.86 | |
| Versus Semaglutide | −0.15 ** | −0.39 * | −0.45 * | - | |
| Weight (Kg) | From baseline | −7.6 | −9.3 | −11.2 | −5.7 |
| Versus Semaglutide | −1.9 * | −3.6 * | −5.5 * | - | |
| Outcomes | Tirzepatide 5 mg (n = 358) | Tirzepatide 10 mg (n = 360) | Tirzepatide 15 mg (n = 358) | Insulin Degludec (n = 359) | |
|---|---|---|---|---|---|
| HbA1c (%) | Baseline | 8.17 | 8.19 | 8.21 | 8.13 |
| From baseline | −1.93 | −2.20 | −2.37 | −1.34 | |
| Versus insulin degludec | −0.59 * | −0.86 * | −1.04 * | - | |
| Weight (Kg) | From baseline | −7.5 | −10.7 | −12.9 | 2.3 |
| Versus insulin degludec | −9.8 * | −13 * | −15.2 * | - | |
| Outcomes | Tirzepatide 5 mg (n = 326) | Tirzepatide 10 mg (n = 321) | Tirzepatide 15 mg (n = 334) | Insulin Glargine (n = 978) | |
|---|---|---|---|---|---|
| HbA1c (%) | Baseline | 8.52 | 8.60 | 8.52 | 8.51 |
| From baseline | −2.24 | −2.43 | −2.58 | −1.44 | |
| Versus insulin glargine | −0.80 * | −0.99 * | −1.14 * | ||
| Weight (Kg) | From baseline | −7.1 | −9.5 | −11.7 | 1.9 |
| Versus insulin glargine | −9.0 * | −11.4 * | −13.5 * | ||
| Outcomes | Tirzepatide 5 mg (n= 116) | Tirzepatide 10 mg (n= 119) | Tirzepatide 15 mg (n= 120) | Placebo (n= 120) | |
|---|---|---|---|---|---|
| HbA1c (%) | Baseline | 8.30 | 8.36 | 8.22 | 8.38 |
| From baseline | −2.11 | −2.40 | −2.34 | −0.86 | |
| Versus placebo | −1.24 * | −1.53 * | −1.47 * | ||
| Weight (Kg) | From baseline | −5.4 | −7.5 | −8.8 | 1.6 |
| Versus placebo | −7.1 * | −9.1 * | −10.5 * | ||
| Outcomes | Tirzepatide 5 mg (n = 630) | Tirzepatide 10 mg (n = 636) | Tirzepatide 15 mg (n = 630) | Placebo (n = 643) | |
|---|---|---|---|---|---|
| Body Weight (Kg) | Baseline | 102.9 | 105.8 | 105.6 | 104.8 |
| From baseline (%) | −15 | −19.5 | −20.9 | −3.1 | |
| Versus placebo (%) | −11.9 * | −16.4 * | −17.8 * | ||
| Waist circumference (cm) | From baseline | −14 | −17.7 | −18.5 | −4 |
| Versus placebo | −10.1 * | −13.8 * | −14.5 * | ||
| Nausea (%) | Diarrhea (%) | Vomiting (%) | |
|---|---|---|---|
| SURPASS-1 | 12–18 | 12–14 | 2–6 |
| SURPASS-2 | 17–22 | 13–16 | 6–10 |
| SURPASS-3 | 12–24 | 15–17 | 6–10 |
| SURPASS-4 | 12–23 | 13–22 | 5–9 |
| SURPASS-5 | 13–18 | 12–21 | 7–13 |
| SURMOUNT-1 | 25–33 | 19–23 | 8–12 |
| SYNERGY-NASH (phase II) | A Randomized, Double-Blind, Placebo-Controlled Phase 2 Study Comparing the Efficacy and Safety of Tirzepatide Versus Placebo in Patients with Nonalcoholic Steatohepatitis (NASH). |
| SURPASS-CVOT (phase III) | The Effect of Tirzepatide Versus Dulaglutide on Major Adverse Cardiovascular Events in Patients with Type 2 Diabetes. |
| SURPASS-PEDS (phase III) | Efficacy, Safety, and Pharmacokinetics/Pharmacodynamics of Tirzepatide in Pediatric and Adolescent Participants with Type 2 Diabetes Mellitus Inadequately Controlled with Metformin, or Basal Insulin, or Both. |
| SURPASS-6 (phase III) | The Effect of the Addition of Tirzepatide Once Weekly Versus Insulin Lispro Three Times Daily in Participants with Type 2 Diabetes Inadequately Controlled on Insulin Glargine (U100) with or without Metformin. |
| SURMOUNT-2 (phase III) | Efficacy and Safety of Tirzepatide Once Weekly in Participants with Type 2 Diabetes Who Have Obesity or Are Overweight. |
| SURMOUNT-3 (phase III) | Efficacy and Safety of Tirzepatide Once Weekly Versus Placebo After an Intensive Lifestyle Program in Participants without Type 2 Diabetes Who Have obesity or Are Overweight with Weight-Related Comorbidities. |
| SURMOUNT-4 (phase III) | Efficacy and Safety of Tirzepatide Once Weekly Versus Placebo for Maintenance of Weight Loss in Participants Without Type 2 Diabetes Who Have Obesity or Are Overweight with Weight-Related Comorbidities: A Randomized, Double-Blind, Placebo-Controlled Trial. |
| SURMOUNT-CN (phase III) | Efficacy and Safety of Tirzepatide Once Weekly in Chinese Participants Without Type 2 Diabetes Who Have Obesity or Are Overweight with Weight-Related Comorbidities: A Randomized, Double-Blind, Placebo-Controlled Trial. |
| SURMOUNT-J (phase III) | Efficacy and Safety of Once-Weekly Tirzepatide in Participants with Obesity Disease. |
| SUMMIT (phase III) | Study Comparing the Efficacy and Safety of Tirzepatide Versus Placebo in Patients with Heart Failure with Preserved Ejection Fraction and Obesity. |
| SURPASS-EARLY (phase IV) | Study to Evaluate the Long-Term Efficacy and Safety of Tirzepatide Compared with Intensified Conventional Care in Adults When Initiating Treatment Early in the Course of Type 2 Diabetes. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Forzano, I.; Varzideh, F.; Avvisato, R.; Jankauskas, S.S.; Mone, P.; Santulli, G. Tirzepatide: A Systematic Update. Int. J. Mol. Sci. 2022, 23, 14631. https://doi.org/10.3390/ijms232314631
Forzano I, Varzideh F, Avvisato R, Jankauskas SS, Mone P, Santulli G. Tirzepatide: A Systematic Update. International Journal of Molecular Sciences. 2022; 23(23):14631. https://doi.org/10.3390/ijms232314631
Chicago/Turabian StyleForzano, Imma, Fahimeh Varzideh, Roberta Avvisato, Stanislovas S. Jankauskas, Pasquale Mone, and Gaetano Santulli. 2022. "Tirzepatide: A Systematic Update" International Journal of Molecular Sciences 23, no. 23: 14631. https://doi.org/10.3390/ijms232314631
APA StyleForzano, I., Varzideh, F., Avvisato, R., Jankauskas, S. S., Mone, P., & Santulli, G. (2022). Tirzepatide: A Systematic Update. International Journal of Molecular Sciences, 23(23), 14631. https://doi.org/10.3390/ijms232314631