Chiroptical Performances in Self-Assembled Hierarchical Nanosegregated Chiral Intermediate Phases Composed of Two Different Achiral Bent-Core Molecules
Abstract
1. Introduction
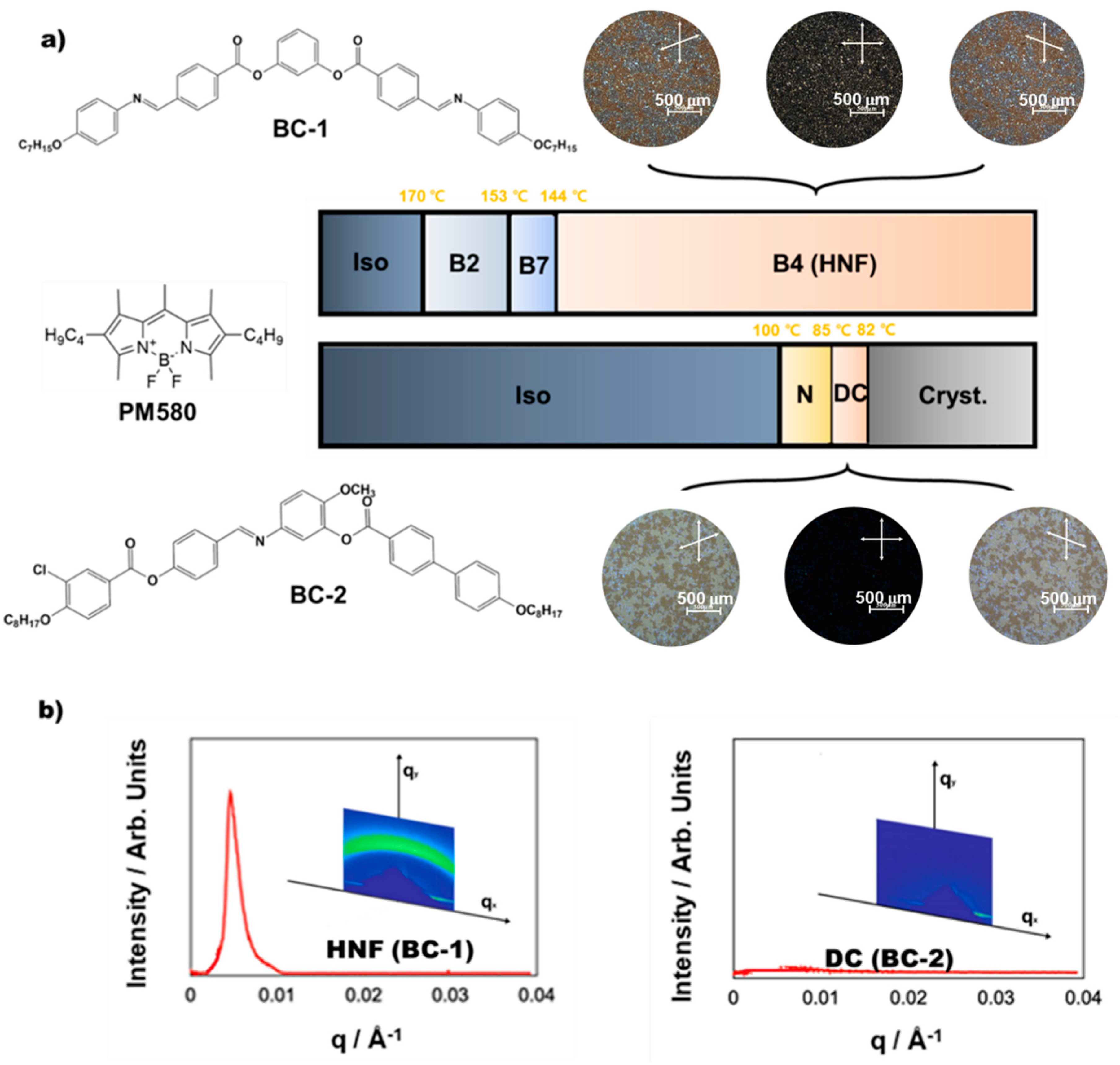
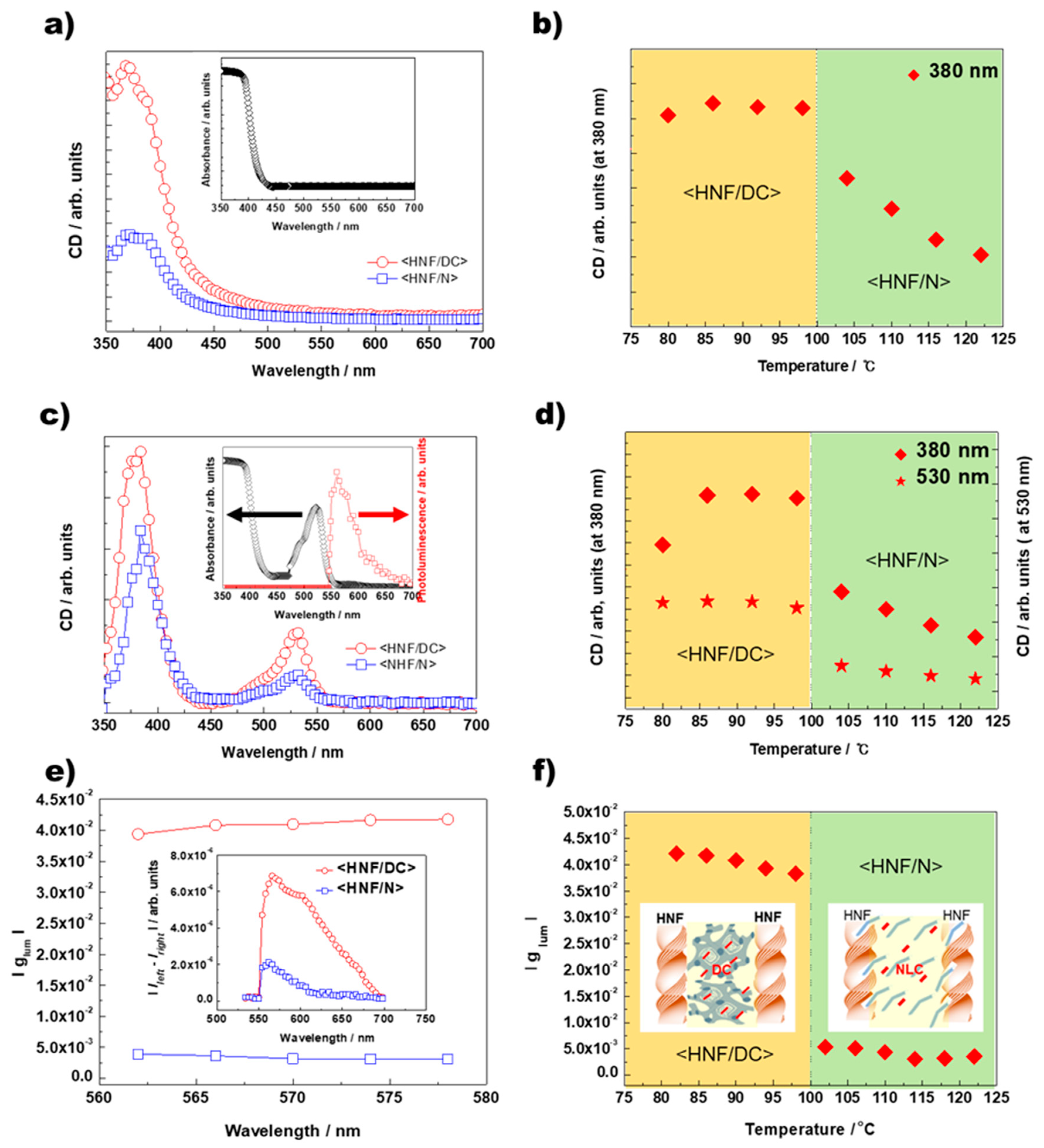
2. Results
3. Materials and Methods
3.1. WAXD and RSoXS Analyses
3.2. CD and CPL
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Pasteur, L. On the Relations Crystalline Form, Chemical Composition and Direction of Polarization Rotatorie. Ann. Chim. Phys. 1848, 24, 442–459. [Google Scholar]
- Park, W.; Wolska, J.M.; Pociecha, D.; Gorecka, E.; Yoon, D.K. Direct Visualization of Optical Activity in Chiral Substances Using a Helical Nanofilament (B4) Liquid Crystal Phase. Adv. Opt. Mater. 2019, 7, 1901399. [Google Scholar] [CrossRef]
- Cao, Y.; Tan, T.; Walba, D.M.; Clark, N.A.; Ungar, G.; Zhu, C.; Zhang, L.; Liu, F. Understanding and Manipulating Helical Nanofilaments in Binary Systems with Achiral Dopants. Nano Lett. 2022, 22, 4569–4575. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, H.; Geng, Z.; Zheng, W.H.; Quan, Y.; Cheng, Y. Inverted Circularly Polarized Luminescence Behavior Induced by Helical Nanofibers through Chiral Co-Assembly from Achiral Liquid Crystal Polymers and Chiral Inducers. ACS Nano 2022, 16, 3173–3181. [Google Scholar] [CrossRef]
- Bae, S.; Noh, Y.W.; Park, D.-W.; Song, M.H.; Choi, S.-W. Development of Colored Perovskite Solar Cells Using Cholesteric Helicoidal Superstructures. Nano Energy 2022, 93, 106801. [Google Scholar] [CrossRef]
- Meyer, R.B.; Liebert, L.; Strzelecki, L.; Keller, P. Ferroelectric Liquid Crystals. J. Phyique Lett. 1975, 36, 69–71. [Google Scholar] [CrossRef]
- Takezoe, H.; Takanishi, Y. Bent-Core Liquid Crystals: Their Mysterious and Attractive World. Jpn. J. Appl. Phys. 2006, 45, 597–625. [Google Scholar] [CrossRef]
- Reddy, R.A.; Tschierske, C. Bent-Core Liquid Crystals: Polar Order, Superstructural Chirality and Spontaneous Desymmetrisation in Soft Matter Systems. J. Mater. Chem. 2006, 16, 907–961. [Google Scholar] [CrossRef]
- Hough, L.E.; Jung, H.T.; Krüerke, D.; Heberling, M.S.; Nakata, M.; Jones, C.D.; Chen, D.; Link, D.R.; Zasadzinski, J.; Heppke, G.; et al. Helical Nanofilament Phases. Science 2009, 325, 456–460. [Google Scholar] [CrossRef]
- Le, K.V.; Takezoe, H.; Araoka, F. Chiral Superstructure Mesophases of Achiral Bent-Shaped Molecules—Hierarchical Chirality Amplification and Physical Properties. Adv. Mater. 2017, 29, 1602737. [Google Scholar] [CrossRef] [PubMed]
- Hough, L.E.; Spannuth, M.; Nakata, M.; Coleman, D.A.; Jones, C.D.; Dantlgraber, G.; Tschierske, C.; Watanabe, J.; Körblova, E.; Walba, D.M.; et al. Chiral Isotropic Liquids from Achiral Molecules. Science 2009, 325, 452–456. [Google Scholar] [CrossRef]
- Kim, B.C.; Choi, H.J.; Lee, J.J.; Araoka, F.; Choi, S.W. Circularly Polarized Luminescence Induced by Chiral Super Nanospaces. Adv. Funct. Mater. 2019, 29, 1903246. [Google Scholar] [CrossRef]
- Takanishi, Y.; Shin, G.J.; Jung, J.C.; Choi, S.W.; Ishikawa, K.; Watanabe, J.; Takezoe, H.; Toledano, P. Observation of Very Large Chiral Domains in a Liquid Crystal Phase Formed by Mixtures of Achiral Bent-Core and Rod Molecules. J. Mater. Chem. 2005, 15, 4020–4024. [Google Scholar] [CrossRef]
- Otani, T.; Araoka, F.; Ishikawa, K.; Takezoe, H. Enhanced Optical Activity by Achiral Rod-Like Molecules Nanosegregated in the B4 Structure of Achiral Bent-Core Molecules. J. Am. Chem. Soc. 2009, 131, 12368–12372. [Google Scholar] [CrossRef]
- Zhu, C.; Chen, D.; Shen, Y.; Jones, C.D.; Glaser, M.A.; MacLennan, J.E.; Clark, N.A. Nanophase Segregation in Binary Mixtures of a Bent-Core and a Rodlike Liquid-Crystal Molecule. Phys. Rev. E 2010, 81, 011704. [Google Scholar] [CrossRef]
- Kim, K.; Kim, H.; Jo, S.Y.; Araoka, F.; Yoon, D.K.; Choi, S.W. Photomodulated Supramolecular Chirality in Achiral Photoresponsive Rodlike Compounds Nanosegregated from the Helical Nanofilaments of Achiral Bent-Core Molecules. ACS Appl. Mater. Interfaces 2015, 7, 22686–22691. [Google Scholar] [CrossRef]
- Jeon, S.W.; Kim, D.Y.; Araoka, F.; Jeong, K.U.; Choi, S.W. Nanosegregated Chiral Materials with Self-Assembled Hierarchical Mesophases: Effect of Thermotropic and Photoinduced Polymorphism in Rodlike Molecules. Chem. A Eur. J. 2017, 23, 17794–17799. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.X.; Liu, C.P.; Zhao, Y.F.; Li, S.C.; Yu, Y.L.; Lv, J.Q.; Chen, L.; Jiang, F.L.; Hong, M.C. White-Light Emission and Circularly Polarized Luminescence from a Chiral Copper(I) Coordination Polymer through Symmetry-Breaking Crystallization. Angew. Chem. Int. Ed. 2022, 61, e202201590. [Google Scholar] [CrossRef]
- Imai, Y.; Nakano, Y.; Kawai, T.; Yuasa, J. A Smart Sensing Method for Object Identification Using Circularly Polarized Luminescence from Coordination-Driven Self-Assembly. Angew. Chem. 2018, 130, 9111–9116. [Google Scholar] [CrossRef]
- Kang, S.; Li, Y.; Bukharina, D.; Kim, M.; Lee, H.; Buxton, M.L.; Han, M.J.; Nepal, D.; Bunning, T.J.; Tsukruk, V.V. Bio-Organic Chiral Nematic Materials with Adaptive Light Emission and On-Demand Handedness. Adv. Mater. 2021, 33, 2103329. [Google Scholar] [CrossRef]
- Shi, Y.; Han, J.; Jin, X.; Miao, W.; Zhang, Y.; Duan, P. Chiral Luminescent Liquid Crystal with Multi-State-Reversibility: Breakthrough in Advanced Anti-Counterfeiting Materials. Adv. Sci. 2022, 9, 2201565. [Google Scholar] [CrossRef]
- Stanciu, C.D.; Hansteen, F.; Kimel, A.V.; Kirilyuk, A.; Tsukamoto, A.; Itoh, A.; Rasing, T. All-Optical Magnetic Recording with Circularly Polarized Light. Phys. Rev. Lett. 2007, 99, 047601. [Google Scholar] [CrossRef]
- Zhang, C.; Diorio, N.; Lavrentovich, O.D.; Jákli, A. Helical Nanofilaments of Bent-Core Liquid Crystals with a Second Twist. Nat. Commun. 2014, 5, 3302. [Google Scholar] [CrossRef]
- Niori, T.; Yamamoto, J.; Yokoyama, H. Dynamics of the Nematic Phase Formed by Achiral Banana-Shaped Materials. Mol. Cryst. Liq. Cryst. 2004, 409, 475–482. [Google Scholar] [CrossRef]
- Kumar, S.; Gowda, A.N. The Chemistry of Bent-Core Molecules Forming Nematic Liquid Crystals. Liq. Cryst. Rev. 2015, 3, 99–145. [Google Scholar] [CrossRef]
- Araoka, F.; Sugiyama, G.; Ishikawa, K.; Takezoe, H. Highly Ordered Helical Nanofilament Assembly Aligned by a Nematic Director Field. Adv. Funct. Mater. 2013, 23, 2701–2707. [Google Scholar] [CrossRef]
- Alaasar, M.; Prehm, M.; Brautzsch, M.; Tschierske, C. Dark Conglomerate Phases of Azobenzene Derived Bent-Core Mesogens—Relationships between the Molecular Structure and Mirror Symmetry Breaking in Soft Matter. Soft Matter 2014, 10, 7285–7296. [Google Scholar] [CrossRef] [PubMed]
- Foley, L.; Park, W.; Yang, M.; Carlson, E.; Korblova, E.; Yoon, D.K.; Walba, D.M. Nanoconfinement of the Low-Temperature Dark Conglomerate: Structural Control from Focal Conics to Helical Nanofilaments. Chem. A Eur. J. 2019, 25, 7438–7442. [Google Scholar] [CrossRef]
- Lee, J.J.; Kim, B.C.; Choi, H.J.; Bae, S.; Araoka, F.; Choi, S.W. Inverse Helical Nanofilament Networks Serving as a Chiral Nanotemplate. ACS Nano 2020, 14, 5243–5250. [Google Scholar] [CrossRef] [PubMed]
- Akutagawa, T.; Matsunaga, Y.; Yasuhara, K. Mesomorphic Behaviour of 1,3-Phenylene Bis[4-(4-Alkoxyphenyliminomethyl)Benzoates] and Related Compounds. Liq. Cryst. 1994, 17, 659–666. [Google Scholar] [CrossRef]
- Kuroda, R.; Harada, T.; Shindo, Y. A Solid-State Dedicated Circular Dichroism Spectrophotometer: Development and Application. Rev. Sci. Instrum. 2001, 72, 3802–3810. [Google Scholar] [CrossRef]
- Hirschmann, M.; Merten, C.; Thiele, C.M. Treating Anisotropic Artefacts in Circular Dichroism Spectroscopy Enables Investigation of Lyotropic Liquid Crystalline Polyaspartate Solutions. Soft Matter 2021, 17, 2849–2856. [Google Scholar] [CrossRef] [PubMed]
- Harada, T.; Kuroda, R.; Moriyama, H. Solid-State Circularly Polarized Luminescence Measurements: Theoretical Analysis. Chem. Phys. Lett. 2012, 530, 126–131. [Google Scholar] [CrossRef]



| BC-1 | BC-2 | Dye | |
|---|---|---|---|
| M1 | 60 | 40 | |
| M2 | 60 | 39.2 | 0.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.-J.; Kim, S.; Nishikawa, H.; Takanishi, Y.; Iwayama, H.; Kim, C.; Choi, S.-W.; Araoka, F. Chiroptical Performances in Self-Assembled Hierarchical Nanosegregated Chiral Intermediate Phases Composed of Two Different Achiral Bent-Core Molecules. Int. J. Mol. Sci. 2022, 23, 14629. https://doi.org/10.3390/ijms232314629
Lee J-J, Kim S, Nishikawa H, Takanishi Y, Iwayama H, Kim C, Choi S-W, Araoka F. Chiroptical Performances in Self-Assembled Hierarchical Nanosegregated Chiral Intermediate Phases Composed of Two Different Achiral Bent-Core Molecules. International Journal of Molecular Sciences. 2022; 23(23):14629. https://doi.org/10.3390/ijms232314629
Chicago/Turabian StyleLee, Jae-Jin, Sangsub Kim, Hiroya Nishikawa, Yoichi Takanishi, Hiroshi Iwayama, Changsoon Kim, Suk-Won Choi, and Fumito Araoka. 2022. "Chiroptical Performances in Self-Assembled Hierarchical Nanosegregated Chiral Intermediate Phases Composed of Two Different Achiral Bent-Core Molecules" International Journal of Molecular Sciences 23, no. 23: 14629. https://doi.org/10.3390/ijms232314629
APA StyleLee, J.-J., Kim, S., Nishikawa, H., Takanishi, Y., Iwayama, H., Kim, C., Choi, S.-W., & Araoka, F. (2022). Chiroptical Performances in Self-Assembled Hierarchical Nanosegregated Chiral Intermediate Phases Composed of Two Different Achiral Bent-Core Molecules. International Journal of Molecular Sciences, 23(23), 14629. https://doi.org/10.3390/ijms232314629








