Effect of Sex Differences in Silicotic Mice
Abstract
1. Introduction
2. Results
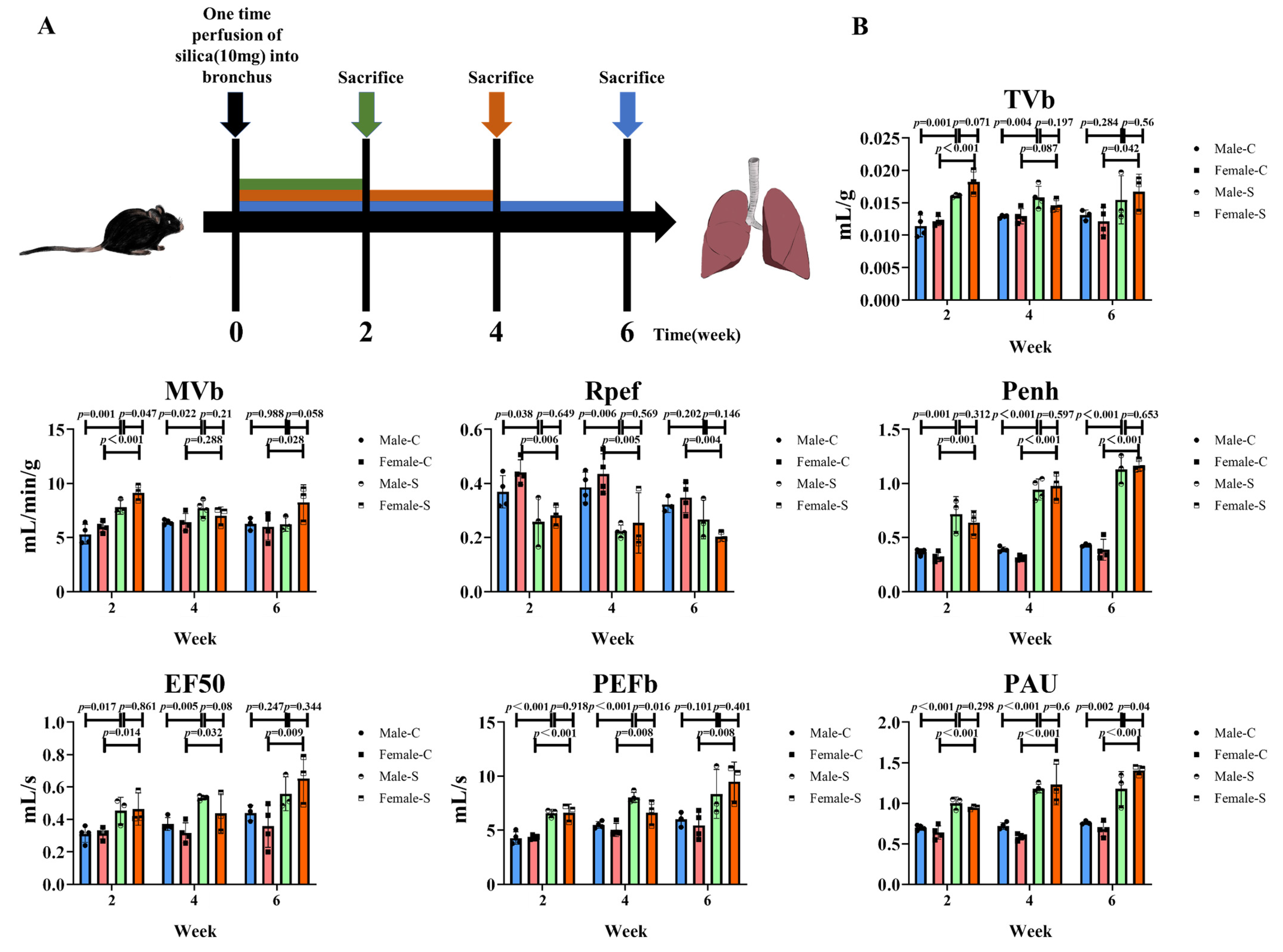
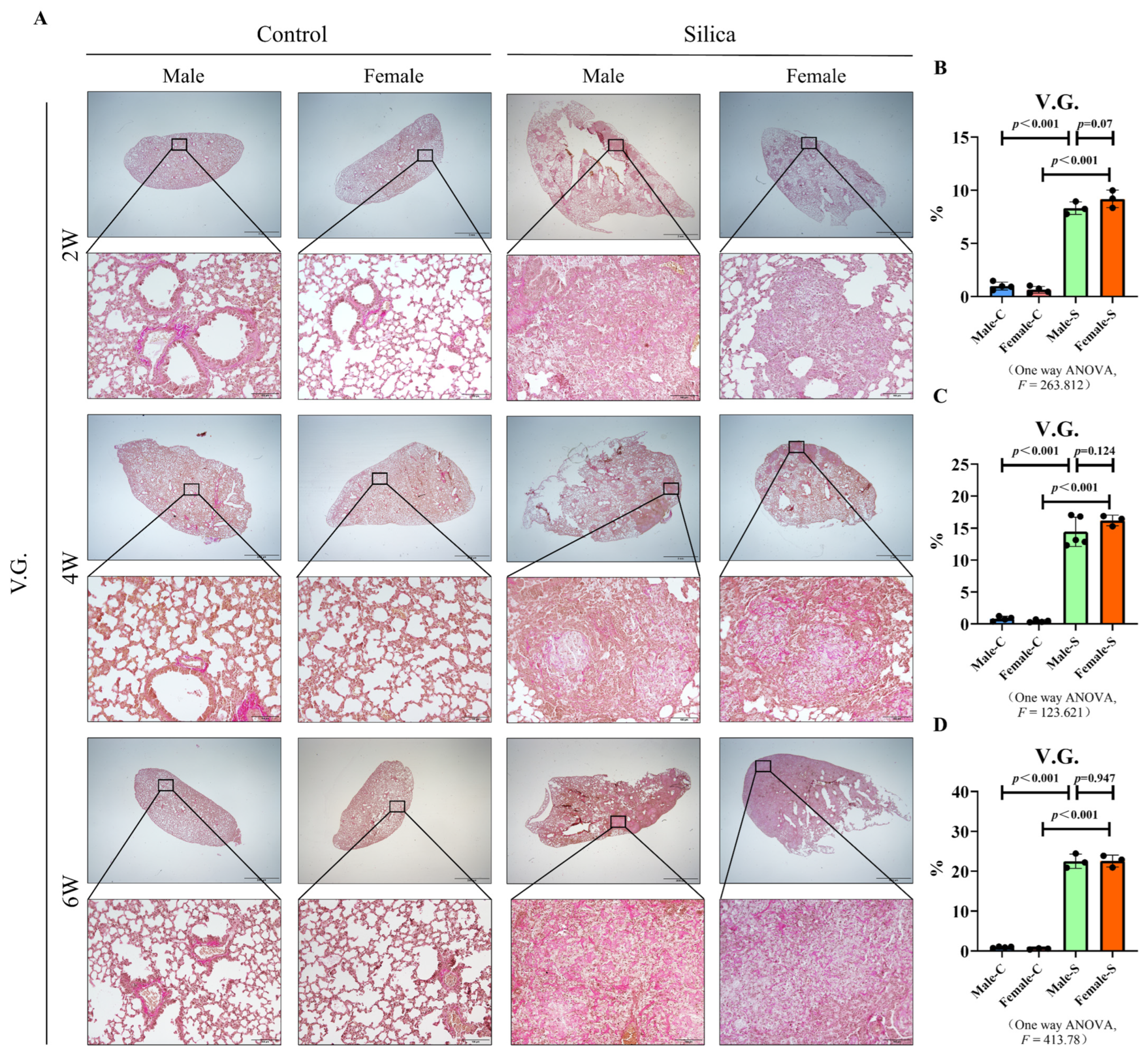
2.1. Effects of Sex on the Silicotic Nodules and Pulmonary Functions
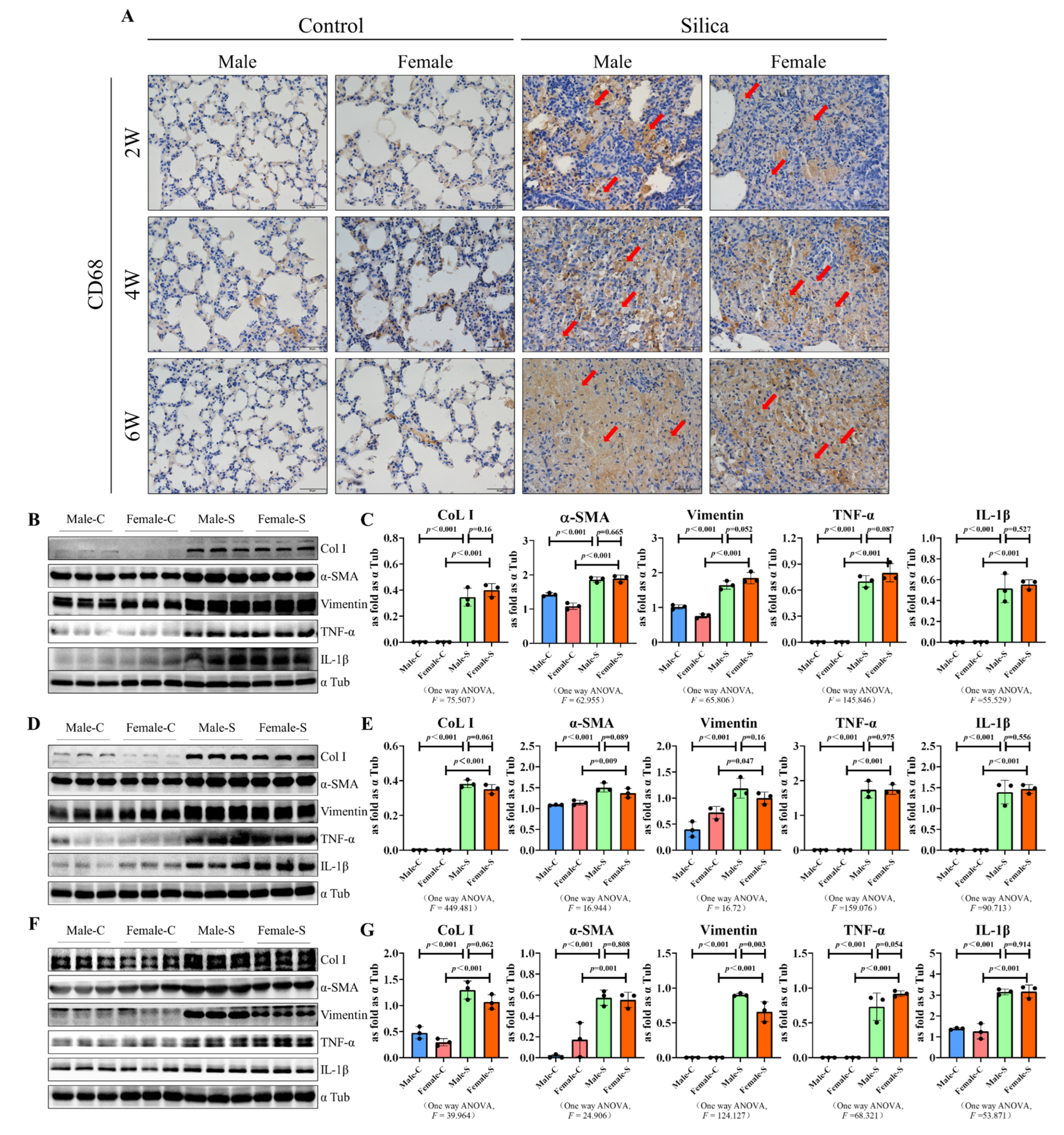
2.2. Effects of Sex on the Collagen Deposition in Silicosis
2.3. Effects of Sex on the Inflammation Response in Silicosis
2.4. Effects of Sex on the TGF-β Signal-Related Factors and Senescence-Related Factors in Silicosis
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Establishment of the Mice Models
4.3. Non-Invasive Measurement of the Pulmonary Function
4.4. H&E Staining
4.5. V.G. Staining
4.6. Western Blot Assay
4.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, J.; Yin, P.; Wang, H.; Wang, L.; You, J.; Liu, J.; Liu, Y.; Wang, W.; Zhang, X.; Niu, P.; et al. The burden of pneumoconiosis in China: An analysis from the Global Burden of Disease Study 2019. BMC Public Health 2022, 22, 1114. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhao, Y.; Sun, D. China’s occupational health challenges. Occup. Med. 2017, 67, 87–90. [Google Scholar] [CrossRef]
- The Lancet Respiratory Medicine. The world is failing on silicosis. Lancet Respir. Med. 2019, 7, 283. [Google Scholar] [CrossRef]
- Li, T.; Yang, X.-Y.; Xu, D.-J.; Gao, Z.-Y.; Gao, Y.-B.; Jin, F.-Y.; Li, Y.-Q.; Liu, S.-P.; Li, S.-F.; Gao, X.-M.; et al. OC-STAMP Overexpression Drives Lung Alveolar Epithelial Cell Type II Senescence in Silicosis. Oxid. Med. Cell. Longev. 2021, 2021, 4158495. [Google Scholar] [CrossRef] [PubMed]
- Jin, F.; Geng, F.; Xu, D.; Li, Y.; Li, T.; Yang, X.; Liu, S.; Zhang, H.; Wei, Z.; Li, S.; et al. Ac-SDKP Attenuates Activation of Lung Macrophages and Bone Osteoclasts in Rats Exposed to Silica by Inhibition of TLR4 and RANKL Signaling Pathways. J. Inflamm. Res. 2021, 14, 1647–1660. [Google Scholar] [CrossRef]
- Mao, N.; Fan, Y.; Liu, W.; Yang, H.; Yang, Y.; Li, Y.; Jin, F.; Li, T.; Yang, X.; Gao, X.; et al. Oxamate Attenuates Glycolysis and ER Stress in Silicotic Mice. Int. J. Mol. Sci. 2022, 23, 3013. [Google Scholar] [CrossRef]
- Li, Y.; Cai, W.; Jin, F.; Wang, X.; Liu, W.; Li, T.; Yang, X.; Liu, H.; Xu, H. Thalidomide Alleviates Pulmonary Fibrosis Induced by Silica in Mice by Inhibiting ER Stress and the TLR4-NF-κB Pathway. Int. J. Mol. Sci. 2022, 23, 5656. [Google Scholar] [CrossRef]
- Geng, F.; Xu, M.; Zhao, L.; Zhang, H.; Li, J.; Jin, F.; Li, Y.; Li, T.; Yang, X.; Li, S.; et al. Quercetin Alleviates Pulmonary Fibrosis in Mice Exposed to Silica by Inhibiting Macrophage Senescence. Front. Pharm. 2022, 13, 912029. [Google Scholar] [CrossRef]
- Gao, X.; Xu, H.; Xu, D.; Li, S.; Wei, Z.; Li, S.; Cai, W.; Mao, N.; Jin, F.; Li, Y.; et al. MiR-411-3p alleviates Silica-induced pulmonary fibrosis by regulating Smurf2/TGF-β signaling. Exp. Cell Res. 2020, 388, 111878. [Google Scholar] [CrossRef]
- Laney, A.S.; Blackley, D.J.; Halldin, C.N. Radiographic disease progression in contemporary US coal miners with progressive massive fibrosis. Occup. Environ. Med. 2017, 74, 517–520. [Google Scholar] [CrossRef]
- Blackley, D.J.; Reynolds, L.E.; Short, C.; Carson, R.; Storey, E.; Halldin, C.N.; Laney, A.S. Progressive massive fibrosis in coal miners from 3 Clinics in Virginia. JAMA 2018, 319, 500–501. [Google Scholar] [CrossRef] [PubMed]
- Maxfield, R.; Alo, C.; Reilly, M.J.; Rosenman, K.; Kalinowski, D.; Stanbury, M.; Valiante, D.J.; Jones, B.; Randolph, S.; Socie, E.; et al. Surveillance for silicosis, 1993–Illinois, Michigan, New Jersey, North Carolina, Ohio, Texas, and Wisconsin. MMWR CDC Surveill. Summ. 1997, 46, 13–28. [Google Scholar] [PubMed]
- Pinkerton, K.E.; Harbaugh, M.; Han, M.K.; Saux, C.J.L.; Winkle, L.S.V.; Martin, W.J.; Kosgei, R.J.; Carter, E.J.; Sitkin, N.; Smiley-Jewell, S.M. Women and Lung Disease. Sex Differences and Global Health Disparities. Am. J. Respir. Crit. Care Med. 2015, 192, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Klein, S.L. Sex differences in immune responses. Nat. Rev. Immunol. 2016, 16, 626–638. [Google Scholar] [CrossRef]
- Ballesteros, B.L.-M.; López-Herranz, M.; Lopez-de-Andrés, A.; Hernandez-Barrera, V.; Jiménez-García, R.; Carabantes-Alarcon, D.; Jiménez-Trujillo, I. Sex Differences in the Incidence and Outcomes of Patients Hospitalized by Idiopathic Pulmonary Fibrosis (IPF) in Spain from 2016 to 2019. J. Clin. Med. 2021, 10, 3474. [Google Scholar] [CrossRef]
- Solopov, P.; Biancatelli, R.M.L.C.; Dimitropoulou, C. Sex-Related Differences in Murine Models of Chemically Induced Pulmonary Fibrosis. Int. J. Mol. Sci. 2021, 22, 5909. [Google Scholar] [CrossRef]
- You, D.J.; Lee, H.Y.; Taylor-Just, A.J.; Linder, K.E. Sex differences in the acute and subchronic lung inflammatory responses of mice to nickel nanoparticles. Nanotoxicology 2020, 14, 1058–1081. [Google Scholar] [CrossRef]
- Li, Y.; Jin, F.; Li, T.; Yang, X.; Cai, W.; Li, S.; Gao, X.; Mao, N.; Liu, H.; Xu, H. Minute Cellular Nodules as Early Lesions in Rats with Silica Exposure via Inhalation. Vet. Sci. 2022, 9, 251. [Google Scholar] [CrossRef]
- Xu, H.; Yang, F.; Sun, Y.; Yuan, Y.; Cheng, H.; Wei, Z.; Li, S.; Cheng, T.; Brann, D. A new antifibrotic target of Ac-SDKP: Inhibition of myofibroblast differentiation in rat lung with silicosis. PLoS ONE 2012, 7, e40301. [Google Scholar] [CrossRef]
- Wu, H.; Yu, Y.; Huang, H.; Hu, Y.; Fu, S.; Wang, Z.; Shi, M.; Zhao, X.; Yuan, J.; Li, J.; et al. Progressive Pulmonary Fibrosis Is Caused by Elevated Mechanical Tension on Alveolar Stem Cells. Cell 2020, 180, 107–121.e17. [Google Scholar] [CrossRef]
- Poinen-Rughooputh, S.; Rughooputh, M.S.; Guo, Y.; Lai, H.; Sun, W. Sex-Related Differences in the Risk of Silicosis Among Chinese Pottery Workers: A Cohort Study. J. Occup. Environ. Med. 2021, 63, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Hoy, R.F. Silica-related diseases in the modern world. Allergy 2020, 75, 2805–2817. [Google Scholar] [CrossRef] [PubMed]
- Bayram, H. Killer jeans and silicosis. Am. J. Respir. Crit. Care Med. 2011, 184, 1322–1324. [Google Scholar] [CrossRef]
- Conti, S.; Harari, S.; Caminati, A.; Zanobetti, A.; Schwartz, J.D.; Bertazzi, P.A.; Cesana, G. The association between air pollution and the incidence of idiopathic pulmonary fibrosis in Northern Italy. Eur. Respir. J. 2018, 51, 1700397. [Google Scholar] [CrossRef] [PubMed]
- Angelidis, I.; Simon, L.M.; Fernandez, I.E.; Strunz, M.; Mayr, C.H.; Greiffo, F.R.; Tsitsiridis, G.; Ansari, M.; Graf, E.; Strom, T.; et al. An atlas of the aging lung mapped by single cell transcriptomics and deep tissue proteomics. Nat. Commun. 2019, 10, 963. [Google Scholar] [CrossRef]
- Deutsch, G.H.; Young, L.R.; Deterding, R.R.; Fan, L.L.; Dell, S.D.; Bean, J.A.; Brody, A.S.; Nogee, L.M.; Trapnell, B.C.; Langston, C.; et al. Diffuse lung disease in young children: Application of a novel classification scheme. Am. J. Respir. Crit. Care Med. 2007, 176, 1120–1128. [Google Scholar] [CrossRef]
- Liptzin, D.R.; Landau, L.I. Sex and the lung: Observations, hypotheses, and future directions. Pediatr. Pulmonol. 2015, 50, 1159–1169. [Google Scholar] [CrossRef]
- Gharaee-Kermani, M.; Hatano, K.; Nozaki, Y.; Phan, S.H. Gender-Based Differences in Bleomycin-Induced Pulmonary Fibrosis. Am. J. Pathol. 2005, 166, 1593–1606. [Google Scholar] [CrossRef]
- Walters, D.M. Mouse models of bleomycin-induced pulmonary fibrosis. Curr. Protoc. Pharmacol. 2008, 40, 5-46. [Google Scholar] [CrossRef]
- Haston, C.K.; Amos, C.I.; King, T.M. Inheritance of susceptibility to bleomycin-induced pulmonary fibrosis in the mouse. Cancer Res. 1996, 56, 2596–2601. [Google Scholar]
- Leung, C.C.; Yu, I.T.S.; Chen, W. Silicosis. Lancet 2012, 379, 2008–2018. [Google Scholar] [CrossRef]
- Lai, Y.-T.; Huang, Y.-S. Modification and Application of a Commercial Whole-body Plethysmograph to Monitor Respiratory Abnormalities in Neonatal Mice. Bio-Protoc. 2017, 7, e2343. [Google Scholar] [CrossRef] [PubMed]
- Shumin, L.; Min, Z.; Peng, W.; Fan, H.; Chen, W.; Yue, Z.; Zhenyang, G.; Xinyu, Y.; Xiaoxiao, H.; Ying, S.; et al. Determination of noninvasive pulmonary function in mice and its significance. Acta Lab. Anim. Sci. Sin. 2018, 26, 548–553. [Google Scholar]
- Pollard, K.M. Perspective: The lung, particles, fibers, nanomaterials, and autoimmunity. Front. Immunol. 2020, 11, 587136. [Google Scholar] [CrossRef] [PubMed]
- Adamcakova, J.; Mokra, D. New Insights into Pathomechanisms and Treatment Possibilities for Lung Silicosis. Int. J. Mol. Sci. 2021, 22, 4162. [Google Scholar] [CrossRef] [PubMed]
- Buskiewicz, I.A.; Huber, S.A.; Fairweather, D. Sex hormone receptor expression in the immune system. In Sex Differences in Physiology; Academic Press: Amsterdam, The Netherlands, 2016; pp. 45–60. [Google Scholar]
- Di Florio, D.N.; Sin, J.; Coronado, M.J.; Atwal, P.S.; Fairweather, D. Sex differences in inflammation, redox biology, mitochondria and autoimmunity. Redox Biol. 2020, 31, 101482. [Google Scholar] [CrossRef]
- He, R.; Yuan, X.; Lv, X.; Liu, Q.; Tao, L.; Meng, J. Caveolin-1 negatively regulates inflammation and fibrosis in silicosis. J. Cell Mol. Med. 2022, 26, 99–107. [Google Scholar] [CrossRef]
- Du, S.; Li, C.; Lu, Y.; Lei, X.; Zhang, Y.; Li, S.; Liu, F.; Chen, Y.; Weng, D.; Chen, J. Dioscin Alleviates Crystalline Silica-Induced Pulmonary Inflammation and Fibrosis through Promoting Alveolar Macrophage Autophagy. Theranostics 2019, 9, 1878–1892. [Google Scholar] [CrossRef]
- Mayeux, J.M.; Escalante, G.M.; Christy, J.M.; Pawar, R.D.; Kono, D.H.; Pollard, K.M. Silicosis and Silica-Induced Autoimmunity in the Diversity Outbred Mouse. Front. Immunol. 2018, 9, 874. [Google Scholar] [CrossRef]
- Budi, E.H.; Schaub, J.R.; Decaris, M.; Turner, S. TGF-β as a driver of fibrosis: Physiological roles and therapeutic opportunities. J. Pathol. 2021, 254, 358–373. [Google Scholar] [CrossRef]
- Li, S.; Wei, Z.; Li, G.; Zhang, Q.; Niu, S.; Xu, D.; Mao, N.; Chen, S.; Gao, X.; Cai, W.; et al. Silica Perturbs Primary Cilia and Causes Myofibroblast Differentiation during Silicosis by Reduction of the KIF3A-Repressor GLI3 Complex. Theranostics 2020, 10, 1719–1732. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Xu, D.; Li, S.; Wei, Z.; Li, S.; Cai, W.; Mao, N.; Jin, F.; Li, Y.; Yi, X.; et al. Pulmonary Silicosis Alters MicroRNA Expression in Rat Lung and miR-411-3p Exerts Anti-fibrotic Effects by Inhibiting MRTF-A/SRF Signaling. Mol. Ther. Nucleic Acids 2020, 20, 851–865. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; Xu, H.; Zhang, B.; Gao, X.; Li, S.; Wei, Z.; Li, S.; Mao, N.; Jin, F.; Li, Y.; et al. Biomedecine, p.; pharmacotherapie, Differential expression of lncRNAs during silicosis and the role of LOC103691771 in myofibroblast differentiation induced by TGF-β1. Biomed. Pharmacother. 2020, 125, 109980. [Google Scholar] [CrossRef] [PubMed]
- Shah, T.A. Unanswered Questions Regarding Sex and BMP/TGF-β Signaling. J. Dev. Biol. 2018, 6, 14. [Google Scholar] [CrossRef] [PubMed]
- Ito, I.; Hanyu, A.; Wayama, M.; Goto, N.; Katsuno, Y.; Kawasaki, S.; Nakajima, Y.; Kajiro, M.; Komatsu, Y.; Fujimura, A.; et al. Estrogen inhibits transforming growth factor beta signaling by promoting Smad2/3 degradation. J. Biol. Chem. 2010, 285, 14747–14755. [Google Scholar] [CrossRef]
- Ziller, N.; Kotolloshi, R.; Esmaeili, M.; Liebisch, M.; Mrowka, R.; Baniahmad, A.; Liehr, T.; Wolf, G. Sex Differences in Diabetes- and TGF-β1-Induced Renal Damage. Cells 2020, 9, 2236. [Google Scholar] [CrossRef]
- Diamond-Stanic, M.K.; You, Y.H. Sugar, sex, and TGF-β in diabetic nephropathy. Semin. Nephrol. 2012, 32, 261–268. [Google Scholar] [CrossRef][Green Version]
- Rocca, W.A.; Rocca, L.G.; Smith, C.Y.; Grossardt, B.R.; Faubion, S.S.; Shuster, L.T.; Kirkland, J.L.; LeBrasseur, N.K.; Schafer, M.J.; Mielke, M.M.; et al. Loss of Ovarian Hormones and Accelerated Somatic and Mental Aging. Physiology 2018, 33, 374–383. [Google Scholar] [CrossRef]
- Farr, J.N.; Rowsey, J.L.; Eckhardt, B.A.; Thicke, B.S.; Fraser, D.G.; Tchkonia, T.; Kirkland, J.L.; Monroe, D.G. Independent Roles of Estrogen Deficiency and Cellular Senescence in the Pathogenesis of Osteoporosis: Evidence in Young Adult Mice and Older Humans. J. Bone Miner. Res. 2019, 34, 1407–1418. [Google Scholar] [CrossRef]
- Wei, Y.; Fu, J.; Wu, W.; Ma, P.; Ren, L.; Wu, J. Estrogen prevents cellular senescence and bone loss through Usp10-dependent p53 degradation in osteocytes and osteoblasts: The role of estrogen in bone cell senescence. Cell Tissue Res. 2021, 386, 297–308. [Google Scholar] [CrossRef]
- Sharpless, N.E. Forging a signature of in vivo senescence. Nat. Rev. Cancer 2015, 15, 397–408. [Google Scholar] [CrossRef] [PubMed]
- Kerget, B.; Araz, O.; Ucar, E.Y.; Karaman, A.; Calik, M.; Alper, F. Female workers’ silicosis diagnosis delayed due to gender bias. Occup. Med. 2019, 69, 219–222. [Google Scholar] [CrossRef] [PubMed]
- Forastiere, F.; Goldsmith, D.F.; Sperati, A.; Rapiti, E.; Miceli, M.; Cavariani, F.; Perucci, C.A. Silicosis and lung function decrements among female ceramic workers in Italy. Am. J. Epidemiol. 2002, 156, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Ndlovu, N.; Richards, G.; Vorajee, N.; Murray, J. Silicosis and pulmonary tuberculosis in deceased female South African miners. Occup. Med. 2019, 69, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Shifeng, L.; Hong, X.; Xue, Y.; Siyu, N.; Qiaodan, Z.; Dingjie, X.; Lijuan, Z.; Zhongqiu, W.; Xuemin, G.; Wenchen, C.; et al. Ac-SDKP increases α-TAT 1 and promotes the apoptosis in lung fibroblasts and epithelial cells double-stimulated with TGF-β1 and silica. Toxicol. Appl. Pharmacol. 2019, 369, 17–29. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, F.; Li, Y.; Wang, X.; Yang, X.; Li, T.; Xu, H.; Wei, Z.; Liu, H. Effect of Sex Differences in Silicotic Mice. Int. J. Mol. Sci. 2022, 23, 14203. https://doi.org/10.3390/ijms232214203
Jin F, Li Y, Wang X, Yang X, Li T, Xu H, Wei Z, Liu H. Effect of Sex Differences in Silicotic Mice. International Journal of Molecular Sciences. 2022; 23(22):14203. https://doi.org/10.3390/ijms232214203
Chicago/Turabian StyleJin, Fuyu, Yaqian Li, Xiaojing Wang, Xinyu Yang, Tian Li, Hong Xu, Zhongqiu Wei, and Heliang Liu. 2022. "Effect of Sex Differences in Silicotic Mice" International Journal of Molecular Sciences 23, no. 22: 14203. https://doi.org/10.3390/ijms232214203
APA StyleJin, F., Li, Y., Wang, X., Yang, X., Li, T., Xu, H., Wei, Z., & Liu, H. (2022). Effect of Sex Differences in Silicotic Mice. International Journal of Molecular Sciences, 23(22), 14203. https://doi.org/10.3390/ijms232214203




