Transcriptome Analysis of Particulate Matter 2.5-Induced Abnormal Effects on Human Sebocytes
Abstract
1. Introduction
2. Results
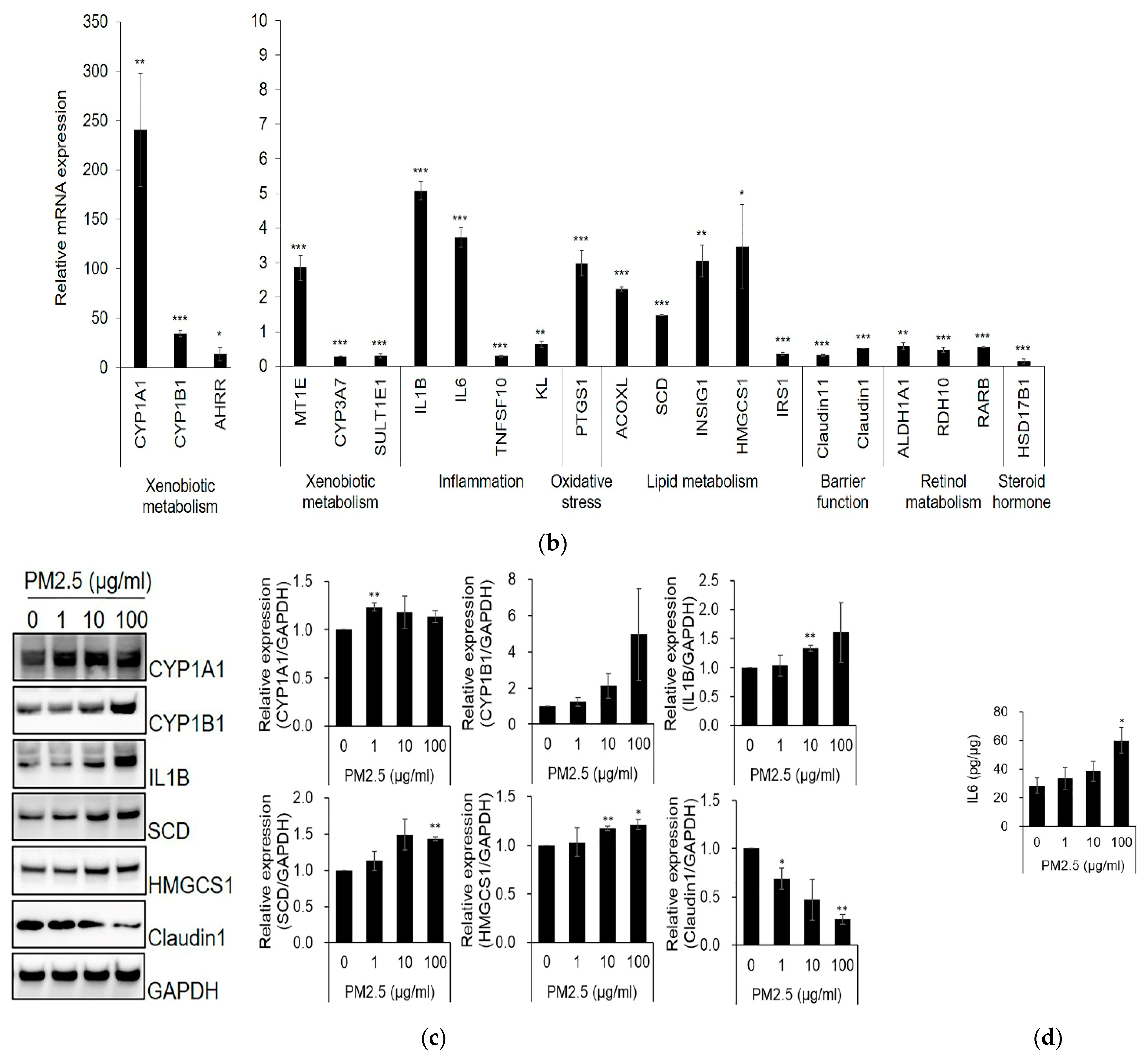
2.1. Differentially Expressed Genes (DEGs) and Gene Ontology (GO)-Based Gene-Set Enrichment Analysis of SZ95 Sebocytes after PM2.5 Treatment
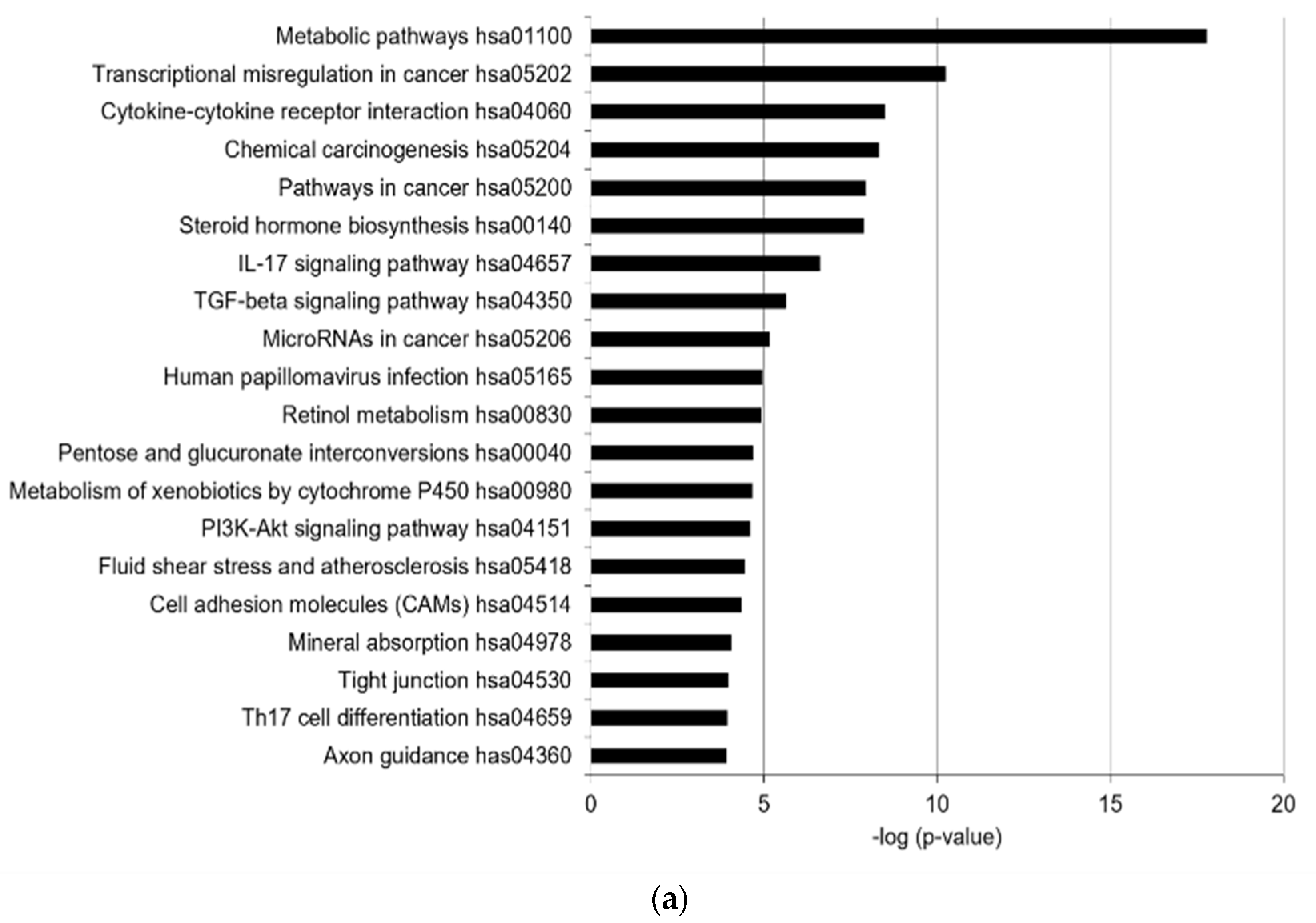
2.2. Kyoto Encyclopedia of Genes and Genomes (KEGG) Terms and Canonical Pathway Prediction Analysis
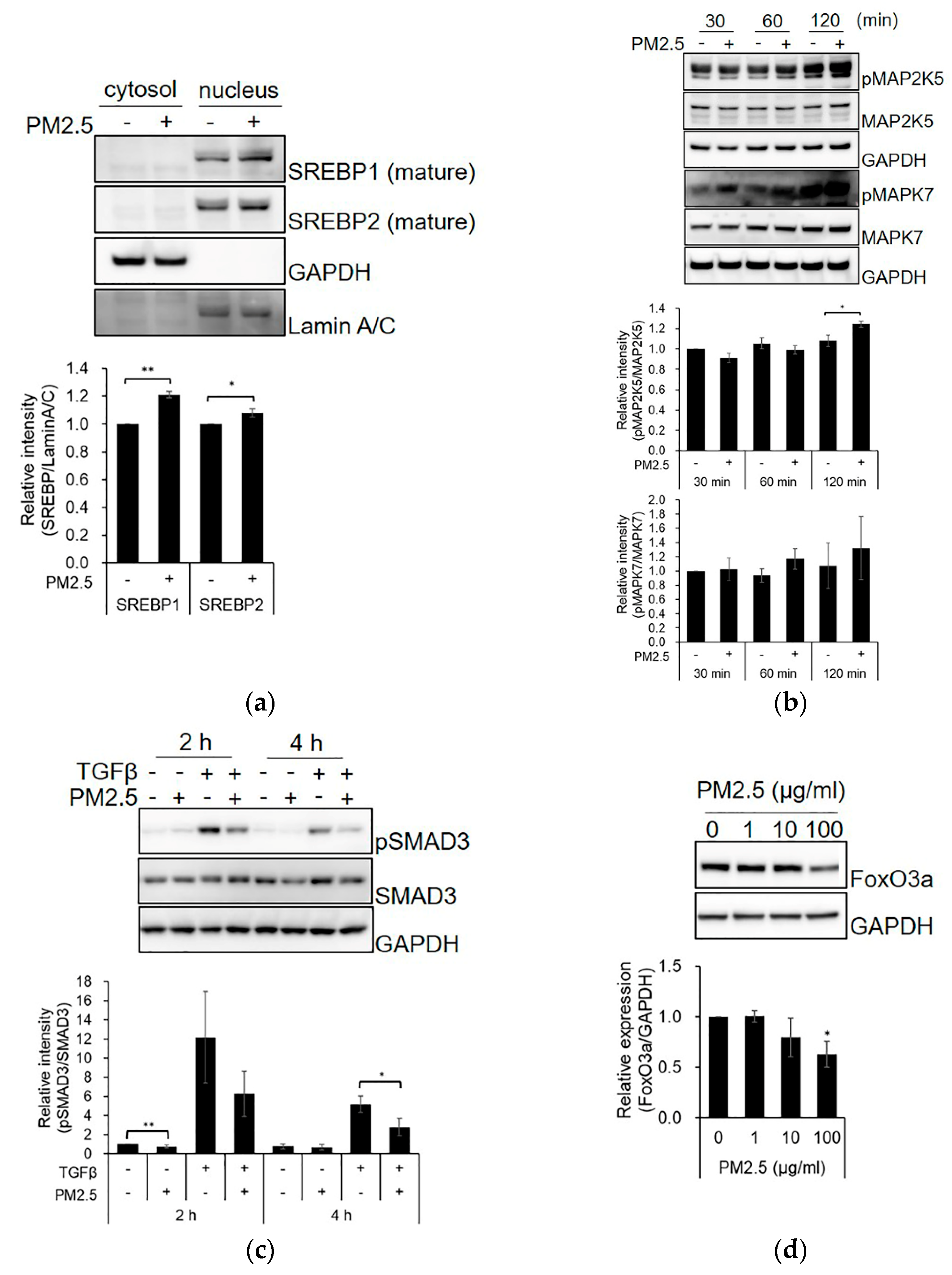
2.3. Upstream Regulator Analysis and Disease and Biological Function Prediction by IPA
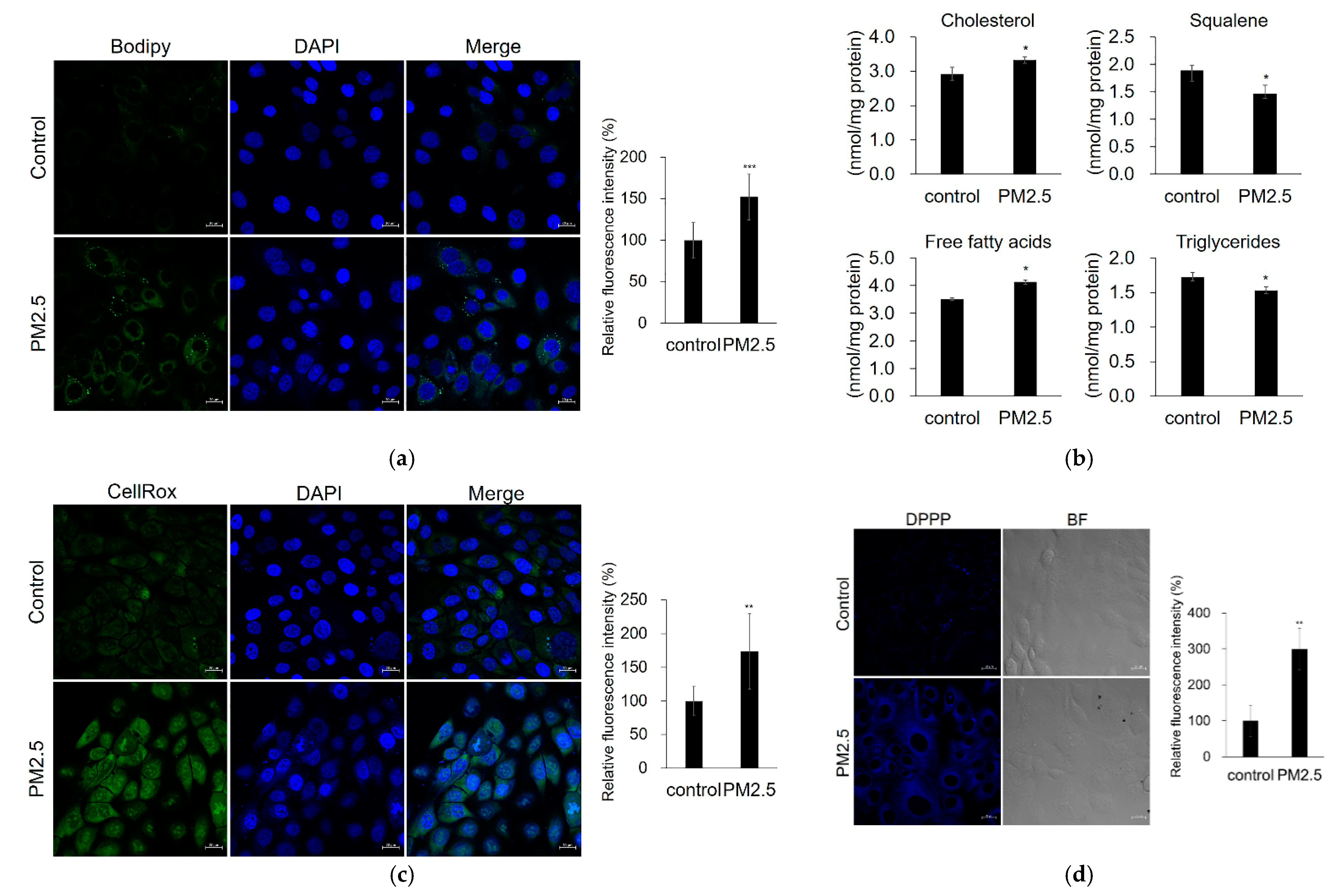
2.4. Effect of PM2.5 on Lipid Production, ROS Generation, and Lipid-Peroxidation in SZ95 Sebocytes
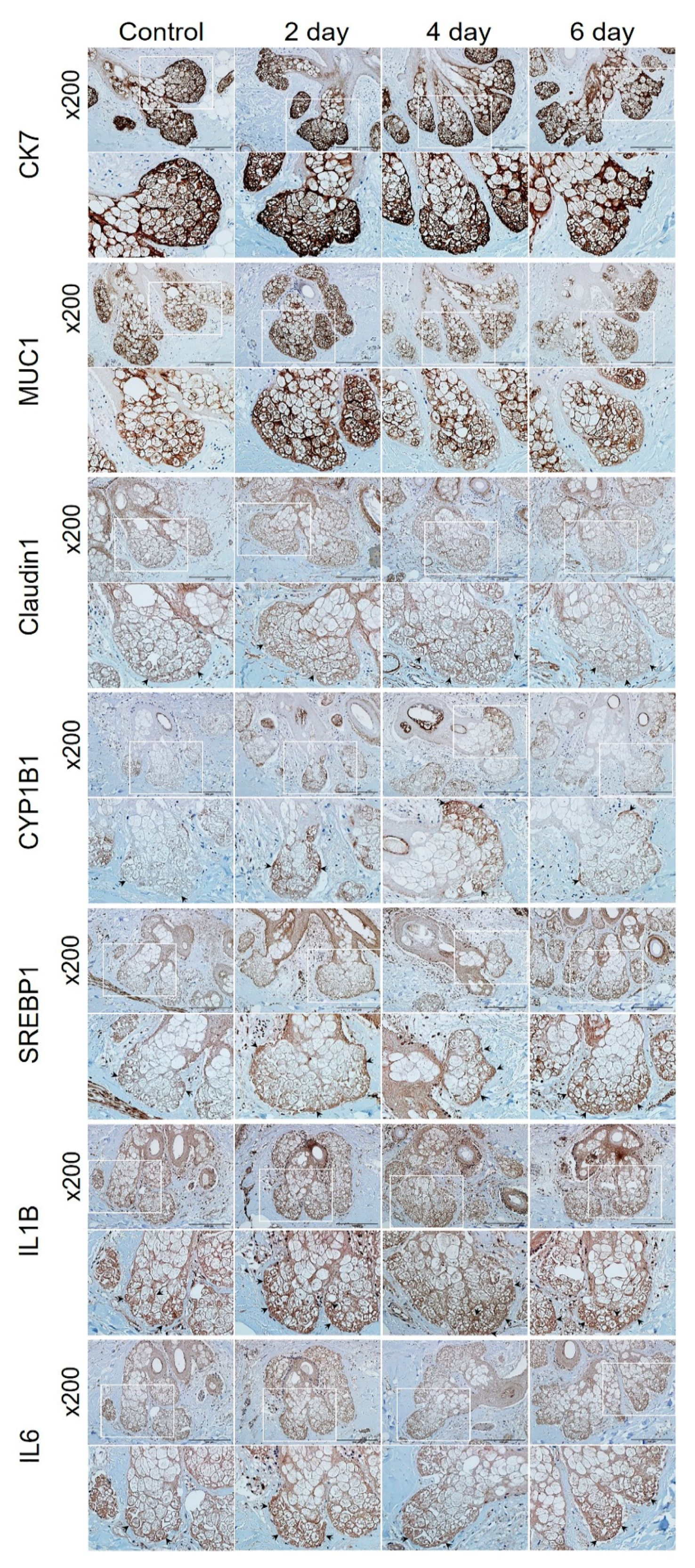
2.5. Confirmation of PM2.5 Effects on Human Skin Tissue
3. Discussion
4. Materials and Methods
4.1. PM2.5 Preparation and Analysis
4.2. Cell Culture
4.3. RT-qPCR Analysis
4.4. Western Blotting of Cell Lysates and ELISA
4.5. Measurement of Lipid Production
4.6. Measurement of ROS and Lipid Peroxidation
4.7. Human Skin Tissue Model and Immunohistochemistry
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. WHO Global Air Quality Guidelines: Particulate Matter (PM2.5 and PM10), Ozone, Nitrogen Dioxide, Sulfur Dioxide and Carbon Monoxide; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- Jeng, H.A. Chemical composition of ambient particulate matter and redox activity. Environ. Monit. Assess. 2010, 169, 597–606. [Google Scholar] [CrossRef]
- Kim, H.J.; Bae, I.H.; Son, E.D.; Park, J.; Cha, N.; Na, H.W.; Jung, C.; Go, Y.S.; Kim, D.Y.; Lee, T.R.; et al. Transcriptome analysis of airborne PM(2.5)-induced detrimental effects on human keratinocytes. Toxicol. Lett. 2017, 273, 26–35. [Google Scholar] [CrossRef]
- Dockery, D.W. Health effects of particulate air pollution. Ann. Epidemiol. 2009, 19, 257–263. [Google Scholar] [CrossRef]
- Xing, Y.F.; Xu, Y.H.; Shi, M.H.; Lian, Y.X. The impact of PM2.5 on the human respiratory system. J. Thorac. Dis. 2016, 8, E69–E74. [Google Scholar] [CrossRef]
- Jin, S.P.; Li, Z.; Choi, E.K.; Lee, S.; Kim, Y.K.; Seo, E.Y.; Chung, J.H.; Cho, S. Urban particulate matter in air pollution penetrates into the barrier-disrupted skin and produces ROS-dependent cutaneous inflammatory response in vivo. J. Dermatol. Sci. 2018, 91, 175–183. [Google Scholar] [CrossRef]
- Lee, E.S.; Kim, S.; Lee, S.W.; Jung, J.; Lee, S.H.; Na, H.W.; Kim, H.J.; Hong, Y.D.; Park, W.S.; Lee, T.G.; et al. Molecule-Resolved Visualization of Particulate Matter on Human Skin Using Multimodal Nonlinear Optical Imaging. Int. J. Mol. Sci. 2021, 22, 5199. [Google Scholar] [CrossRef]
- Zouboulis, C.C. Acne and sebaceous gland function. Clin. Dermatol. 2004, 22, 360–366. [Google Scholar] [CrossRef]
- Plewig, G.; Melnik, B.; Chen, W. Pilosebaceous Follicles: Structure, Biochemistry, and Function. In Plewig and Kligman’s Acne and Rosacea; Springer International Publishing: Cham, Germany, 2019; pp. 1–34. [Google Scholar]
- Picardo, M.; Mastrofrancesco, A.; Bíró, T. Sebaceous gland-a major player in skin homoeostasis. Exp. Dermatol. 2015, 24, 485–486. [Google Scholar] [CrossRef]
- Zouboulis, C.C.; Böhm, M. Neuroendocrine regulation of sebocytes—A pathogenetic link between stress and acne. Exp. Dermatol. 2004, 13 (Suppl. 4), 31–35. [Google Scholar] [CrossRef]
- Niemann, C.; Unden, A.B.; Lyle, S.; Zouboulis Ch, C.; Toftgård, R.; Watt, F.M. Indian hedgehog and beta-catenin signaling: Role in the sebaceous lineage of normal and neoplastic mammalian epidermis. Proc. Natl. Acad. Sci. USA 2003, 100 (Suppl. S1), 11873–11880. [Google Scholar] [CrossRef]
- Gary, G. Optimizing treatment approaches in seborrheic dermatitis. J. Clin. Aesthetic Dermatol. 2013, 6, 44–49. [Google Scholar]
- Liakou, A.I.; Nyengaard, J.R.; Bonovas, S.; Knolle, J.; Makrantonaki, E.; Zouboulis, C.C. Marked Reduction of the Number and Individual Volume of Sebaceous Glands in Psoriatic Lesions. Dermatology 2016, 232, 415–424. [Google Scholar] [CrossRef]
- Zhang, C.; Chinnappan, M.; Prestwood, C.A.; Edwards, M.; Artami, M.; Thompson, B.M.; Eckert, K.M.; Vale, G.; Zouboulis, C.C.; McDonald, J.G.; et al. Interleukins 4 and 13 drive lipid abnormalities in skin cells through regulation of sex steroid hormone synthesis. Proc. Natl. Acad. Sci. USA 2021, 118, e2100749118. [Google Scholar] [CrossRef]
- Kim, K.E.; Cho, D.; Park, H.J. Air pollution and skin diseases: Adverse effects of airborne particulate matter on various skin diseases. Life Sci. 2016, 152, 126–134. [Google Scholar] [CrossRef]
- Wróbel, A.; Seltmann, H.; Fimmel, S.; Müller-Decker, K.; Tsukada, M.; Bogdanoff, B.; Mandt, N.; Blume-Peytavi, U.; Orfanos, C.E.; Zouboulis, C.C. Differentiation and apoptosis in human immortalized sebocytes. J. Investig. Dermatol. 2003, 120, 175–181. [Google Scholar] [CrossRef]
- Xia, L.; Zouboulis, C.C.; Ju, Q. Culture of human sebocytes in vitro. Dermatoendocrinology 2009, 1, 92–95. [Google Scholar] [CrossRef]
- Zouboulis, C.C.; Seltmann, H.; Neitzel, H.; Orfanos, C.E. Establishment and characterization of an immortalized human sebaceous gland cell line (SZ95). J. Investig. Dermatol. 1999, 113, 1011–1020. [Google Scholar] [CrossRef]
- Rendic, S.; Guengerich, F.P. Survey of Human Oxidoreductases and Cytochrome P450 Enzymes Involved in the Metabolism of Xenobiotic and Natural Chemicals. Chem. Res. Toxicol. 2015, 28, 38–42. [Google Scholar] [CrossRef]
- Dijkhoff, I.M.; Drasler, B.; Karakocak, B.B.; Petri-Fink, A.; Valacchi, G.; Eeman, M.; Rothen-Rutishauser, B. Impact of airborne particulate matter on skin: A systematic review from epidemiology to in vitro studies. Part. Fibre Toxicol. 2020, 17, 35. [Google Scholar] [CrossRef]
- Everts, H.B. Endogenous retinoids in the hair follicle and sebaceous gland. Biochim. Biophys. Acta 2012, 1821, 222–229. [Google Scholar] [CrossRef]
- Tien, C.P.; Chen, C.H.; Lin, W.Y.; Liu, C.S.; Liu, K.J.; Hsiao, M.; Chang, Y.C.; Hung, S.C. Ambient particulate matter attenuates Sirtuin1 and augments SREBP1-PIR axis to induce human pulmonary fibroblast inflammation: Molecular mechanism of microenvironment associated with COPD. Aging 2019, 11, 4654–4671. [Google Scholar] [CrossRef] [PubMed]
- Eberlé, D.; Hegarty, B.; Bossard, P.; Ferré, P.; Foufelle, F. SREBP transcription factors: Master regulators of lipid homeostasis. Biochimie 2004, 86, 839–848. [Google Scholar] [CrossRef]
- Cristea, S.; Coles, G.L.; Hornburg, D.; Gershkovitz, M.; Arand, J.; Cao, S.; Sen, T.; Williamson, S.C.; Kim, J.W.; Drainas, A.P.; et al. The MEK5-ERK5 Kinase Axis Controls Lipid Metabolism in Small-Cell Lung Cancer. Cancer Res. 2020, 80, 1293–1303. [Google Scholar] [CrossRef] [PubMed]
- Melnik, B.C. Pro-inflammatory sebocyte growth and survival signalling in acne vulgaris are reversed by pro-apoptotic isotretinoin signalling. Exp. Dermatol. 2016, 25, 676–677. [Google Scholar] [CrossRef] [PubMed][Green Version]
- McNairn, A.J.; Doucet, Y.; Demaude, J.; Brusadelli, M.; Gordon, C.B.; Uribe-Rivera, A.; Lambert, P.F.; Bouez, C.; Breton, L.; Guasch, G. TGFβ signaling regulates lipogenesis in human sebaceous glands cells. BMC Dermatol. 2013, 13, 2. [Google Scholar] [CrossRef] [PubMed]
- Nebert, D.W.; Dalton, T.P. The role of cytochrome P450 enzymes in endogenous signalling pathways and environmental carcinogenesis. Nat. Rev. Cancer 2006, 6, 947–960. [Google Scholar] [CrossRef]
- Belyaeva, O.V.; Adams, M.K.; Popov, K.M.; Kedishvili, N.Y. Generation of Retinaldehyde for Retinoic Acid Biosynthesis. Biomolecules 2019, 10, 5. [Google Scholar] [CrossRef]
- Schiffer, L.; Barnard, L.; Baranowski, E.S.; Gilligan, L.C.; Taylor, A.E.; Arlt, W.; Shackleton, C.H.L.; Storbeck, K.H. Human steroid biosynthesis, metabolism and excretion are differentially reflected by serum and urine steroid metabolomes: A comprehensive review. J. Steroid Biochem. Mol. Biol. 2019, 194, 105439. [Google Scholar] [CrossRef]
- Marom-Haham, L.; Shulman, A. Cigarette smoking and hormones. Curr. Opin. Obstet. Gynecol. 2016, 28, 230–235. [Google Scholar] [CrossRef]
- Lefebvre, M.A.; Pham, D.M.; Boussouira, B.; Bernard, D.; Camus, C.; Nguyen, Q.L. Evaluation of the impact of urban pollution on the quality of skin: A multicentre study in Mexico. Int. J. Cosmet. Sci. 2015, 37, 329–338. [Google Scholar] [CrossRef]
- Lefebvre, M.A.; Pham, D.M.; Boussouira, B.; Qiu, H.; Ye, C.; Long, X.; Chen, R.; Gu, W.; Laurent, A.; Nguyen, Q.L. Consequences of urban pollution upon skin status. A controlled study in Shanghai area. Int. J. Cosmet. Sci. 2016, 38, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Liao, Z.; Nie, J.; Sun, P. The impact of particulate matter (PM2.5) on skin barrier revealed by transcriptome analysis: Focusing on cholesterol metabolism. Toxicol. Rep. 2020, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kwack, M.H.; Ha, D.-L.; Ha, N.; Lee, W.J. Effects of PM10 on Sebocytes, Outer Root Sheath Cells, and Cutibacterium acnes-Pretreated Mice Were Inhibited by Punicalagin. SSRN Electron. J. 2021. [Google Scholar] [CrossRef]
- Zouboulis, C.C.; Jourdan, E.; Picardo, M. Acne is an inflammatory disease and alterations of sebum composition initiate acne lesions. J. Eur. Acad. Dermatol. Venereol. JEADV 2014, 28, 527–532. [Google Scholar] [CrossRef]
- Ottaviani, M.; Camera, E.; Picardo, M. Lipid mediators in acne. Mediat. Inflamm. 2010, 2010, 858176. [Google Scholar] [CrossRef]
- Bowe, W.P.; Logan, A.C. Clinical implications of lipid peroxidation in acne vulgaris: Old wine in new bottles. Lipids Health Dis. 2010, 9, 141. [Google Scholar] [CrossRef]
- Picardo, M.; Ottaviani, M.; Camera, E.; Mastrofrancesco, A. Sebaceous gland lipids. Dermatoendocrinol 2009, 1, 68–71. [Google Scholar] [CrossRef]
- Tan, J.K.L.; Stein Gold, L.F.; Alexis, A.F.; Harper, J.C. Current Concepts in Acne Pathogenesis: Pathways to Inflammation. Semin. Cutan. Med. Surg. 2018, 37, S60–S62. [Google Scholar] [CrossRef]
- Choi, C.W.; Kim, Y.; Kim, J.E.; Seo, E.Y.; Zouboulis, C.C.; Kang, J.S.; Youn, S.W.; Chung, J.H. Enhancement of lipid content and inflammatory cytokine secretion in SZ95 sebocytes by palmitic acid suggests a potential link between free fatty acids and acne aggravation. Exp. Dermatol. 2019, 28, 207–210. [Google Scholar] [CrossRef]
- Fasano, C.; Disciglio, V.; Bertora, S.; Lepore Signorile, M.; Simone, C. FOXO3a from the Nucleus to the Mitochondria: A Round Trip in Cellular Stress Response. Cells 2019, 8, 1110. [Google Scholar] [CrossRef]
- Melnik, B.C. The TRAIL to acne pathogenesis: Let’s focus on death pathways. Exp. Dermatol. 2017, 26, 270–272. [Google Scholar] [CrossRef] [PubMed]
- Niemann, C. Differentiation of the sebaceous gland. Dermatoendocrinology 2009, 1, 64–67. [Google Scholar] [CrossRef] [PubMed]
- Han, G.; Li, A.G.; Liang, Y.Y.; Owens, P.; He, W.; Lu, S.; Yoshimatsu, Y.; Wang, D.; Ten Dijke, P.; Lin, X.; et al. Smad7-induced beta-catenin degradation alters epidermal appendage development. Dev. Cell 2006, 11, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef]
- Kim, D.; Langmead, B.; Salzberg, S.L. HISAT: A fast spliced aligner with low memory requirements. Nat. Methods 2015, 12, 357–360. [Google Scholar] [CrossRef]
- Pertea, M.; Kim, D.; Pertea, G.M.; Leek, J.T.; Salzberg, S.L. Transcript-level expression analysis of RNA-seq experiments with HISAT, StringTie and Ballgown. Nat. Protoc. 2016, 11, 1650–1667. [Google Scholar] [CrossRef]
- Pertea, M.; Pertea, G.M.; Antonescu, C.M.; Chang, T.C.; Mendell, J.T.; Salzberg, S.L. StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat. Biotechnol. 2015, 33, 290–295. [Google Scholar] [CrossRef]
- Krämer, A.; Green, J.; Pollard, J., Jr.; Tugendreich, S. Causal analysis approaches in Ingenuity Pathway Analysis. Bioinformatics 2013, 30, 523–530. [Google Scholar] [CrossRef]
- Kim, B.J.; Lee, N.R.; Lee, C.H.; Lee, Y.B.; Choe, S.J.; Lee, S.; Hwang, H.J.; Kim, E.; Lavery, G.G.; Shin, K.O.; et al. Increased Expression of 11β-Hydroxysteroid Dehydrogenase Type 1 Contributes to Epidermal Permeability Barrier Dysfunction in Aged Skin. Int. J. Mol. Sci. 2021, 22, 5750. [Google Scholar] [CrossRef]
- Lee, Y.; Shin, K.; Shin, K.O.; Yoon, S.; Jung, J.; Hwang, E.; Chung, H.J.; Hossini, A.M.; Zouboulis, C.C.; Baek, M.J.; et al. Topical application of autophagy-activating peptide improved skin barrier function and reduced acne symptoms in acne-prone skin. J. Cosmet. Dermatol. 2021, 20, 1009–1016. [Google Scholar] [CrossRef]
- Hasan, M.; Siegmund, W.; Oswald, S. Rapid LC-MS/MS method for the determination of 4-hydroxycholesterol/cholesterol ratio in serum as endogenous biomarker for CYP3A activity in human and foals. J. Chromatography. B Anal. Technol. Biomed. Life Sci. 2016, 1033–1034, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Navarro, J.; Da Ros, A.; Masuero, D.; Izquierdo-Cañas, P.M.; Hermosín-Gutiérrez, I.; Gómez-Alonso, S.; Mattivi, F.; Vrhovsek, U. LC-MS/MS analysis of free fatty acid composition and other lipids in skins and seeds of Vitis vinifera grape cultivars. Food Res. Int. 2019, 125, 108556. [Google Scholar] [CrossRef] [PubMed]
| Ingenuity Canonical Pathways | −Log (p-Value) | Ratio | z-Score | Molecules |
|---|---|---|---|---|
| Xenobiotic metabolism AHR signaling pathway | 6.41 | 0.23 | 3.578 | ABCG2, AHRR, ALDH1A1, ALDH1L1, ALDH3A1, ALDH3B1, ALDH3B2, ALDH6A1, CYP1A1, CYP1B1, GSTA4, GSTM2, GSTM3, GSTM4, IL1A, IL1B, IL6, UGT1A1, UGT1A3, UGT1A6 |
| Superpathway of cholesterol biosynthesis | 5.17 | 0.345 | 3.162 | ACAT2, CYP51A1, DHCR7, FDFT1, FDPS, HMGCR, HMGCS1, LSS, MSMO1, MVD |
| Cholesterol Biosynthesis I | 3.07 | 0.385 | 2.236 | CYP51A1, DHCR7, FDFT1, LSS, MSMO1 |
| Cholesterol Biosynthesis II (via 24,25-dihydrolanosterol) | 3.07 | 0.385 | 2.236 | CYP51A1, DHCR7, FDFT1, LSS, MSMO1 |
| Cholesterol Biosynthesis III (Via Desmosterol) | 3.07 | 0.385 | 2.236 | CYP51A1, DHCR7, FDFT1, LSS, MSMO1 |
| Role of IL-17A in psoriasis | 2.9 | 0.357 | −2.236 | CXCL1, CXCL6, CXCL8, S100A8, S100A9 |
| Thyroid cancer signaling | 2.4 | 0.152 | −2.887 | CCND1, CXCL8, FOS, IRS1, jun, myc, PIK3R3, RAP2B, RASD2, TCF4, TCF7L1, TP53 |
| Superpathway of Geranylgeranyldiphosphate Biosynthesis I (via mevalonate) | 2.36 | 0.278 | 2.236 | ACAT2, FDPS, HMGCR, HMGCS1, MVD |
| Estrogen-dependent breast cancer signaling | 1.96 | 0.139 | −2.236 | AKR1C1/AKR1C2, CCND1, FOS, HSD17B1, HSD17B14, HSD17B2, HSD17B3, JUN, PIK3R3, RAP2B, RASD2 |
| eNOS signaling | 1.09 | 0.0943 | 2.496 | BDKRB1, CALML5, CAV1, CCNA1, CHRNB4, ESR2, GUCY1B1, HSPA5, KDR, LPAR1, LPAR3, PGF, PIK3R3, PRKAA2, PRKD1 |
| Upstream Regulator | Molecule Type | Predicted Activation State | Activation z-Score | p-Value of Overlap |
|---|---|---|---|---|
| SREBF1 | Transcription regulator | Activated | 4.149 | 1.11 × 10−14 |
| SREBF2 | Transcription regulator | Activated | 4.063 | 1.41 × 10−13 |
| MAPK7 | Kinase | Activated | 2.935 | 1.49 × 10−11 |
| SCAP | Other | Activated | 4.12 | 3.39 × 10−10 |
| MAP2K5 | Kinase | Activated | 3.704 | 1.59 × 10−9 |
| DSCAML1 | Other | Activated | 2.294 | 1.41 × 10−8 |
| EWSR1-FLI1 | Fusion gene/product | Activated | 2.426 | 1.72 × 10−8 |
| CYP7A1 | Enzyme | Activated | 2.333 | 3.62 × 10−7 |
| DSCAM | Other | Activated | 3.286 | 8.32 × 10−7 |
| SH3TC2 | Other | Activated | 2.111 | 1.2 × 10−6 |
| CTNNB1 | Transcription regulator | Inhibited | −2.587 | 4.66 × 10−16 |
| SMAD3 | Transcription regulator | Inhibited | −2.105 | 6.62 × 10−14 |
| WNT3A | Cytokine | Inhibited | −3.324 | 1.21 × 10−12 |
| TGFB1 | Growth factor | Inhibited | −2.179 | 3.34 × 10−12 |
| INSIG1 | Other | Inhibited | −3.761 | 4.37 × 10−11 |
| LRP6 | Transcription regulator | Inhibited | −2.619 | 1.58 × 10−9 |
| MRTFB | Transcription regulator | Inhibited | −2.939 | 8.37 × 10−9 |
| FOXO3 | Transcription regulator | Inhibited | −3.051 | 1.71 × 10−8 |
| PDGF BB | Complex | Inhibited | −2.768 | 3.16 × 10−8 |
| MFSD2A | Transporter | Inhibited | −3.293 | 3.76 × 10−8 |
| Categories | Diseases or Functions Annotation | p-Value | B–H p-Value | Predicted Activation State | Activation z-Score | Bias-Corrected z-Score | No. of Molecules |
|---|---|---|---|---|---|---|---|
| Lipid metabolism, molecular transport, small molecule biochemistry | Concentration of lipid | 1.42 × 10−14 | 1.79 × 10−12 | Increased | 2.871 | 2.767 | 152 |
| dermatological diseases and conditions, organismal injury and abnormalities | Abnormality of skin morphology | 3.81 × 10−8 | 1.66 × 10−6 | Increased | 2.646 | 2.853 | 69 |
| lipid metabolism, small molecule biochemistry | Fatty acid metabolism | 6.95 × 10−15 | 9.14 × 10−13 | Increased | 2.611 | 2.078 | 111 |
| cancer, organismal injury and abnormalities, respiratory disease | Development of lung tumor | 3.6 × 10−10 | 2.3 × 10−8 | Increased | 2.566 | 2.766 | 273 |
| organismal survival | Organismal death | 1.08 × 10−17 | 1.78 × 10−15 | Increased | 2.547 | 4.19 | 380 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Na, H.-W.; Kim, H.S.; Choi, H.; Cha, N.; Seo, Y.R.; Hong, Y.D.; Kim, H.-J. Transcriptome Analysis of Particulate Matter 2.5-Induced Abnormal Effects on Human Sebocytes. Int. J. Mol. Sci. 2022, 23, 11534. https://doi.org/10.3390/ijms231911534
Na H-W, Kim HS, Choi H, Cha N, Seo YR, Hong YD, Kim H-J. Transcriptome Analysis of Particulate Matter 2.5-Induced Abnormal Effects on Human Sebocytes. International Journal of Molecular Sciences. 2022; 23(19):11534. https://doi.org/10.3390/ijms231911534
Chicago/Turabian StyleNa, Hye-Won, Hyun Soo Kim, Hyunjung Choi, Nari Cha, Young Rok Seo, Yong Deog Hong, and Hyoung-June Kim. 2022. "Transcriptome Analysis of Particulate Matter 2.5-Induced Abnormal Effects on Human Sebocytes" International Journal of Molecular Sciences 23, no. 19: 11534. https://doi.org/10.3390/ijms231911534
APA StyleNa, H.-W., Kim, H. S., Choi, H., Cha, N., Seo, Y. R., Hong, Y. D., & Kim, H.-J. (2022). Transcriptome Analysis of Particulate Matter 2.5-Induced Abnormal Effects on Human Sebocytes. International Journal of Molecular Sciences, 23(19), 11534. https://doi.org/10.3390/ijms231911534






