The Potential Roles of Extracellular Vesicles as Biomarkers for Parkinson’s Disease: A Systematic Review
Abstract
1. Introduction
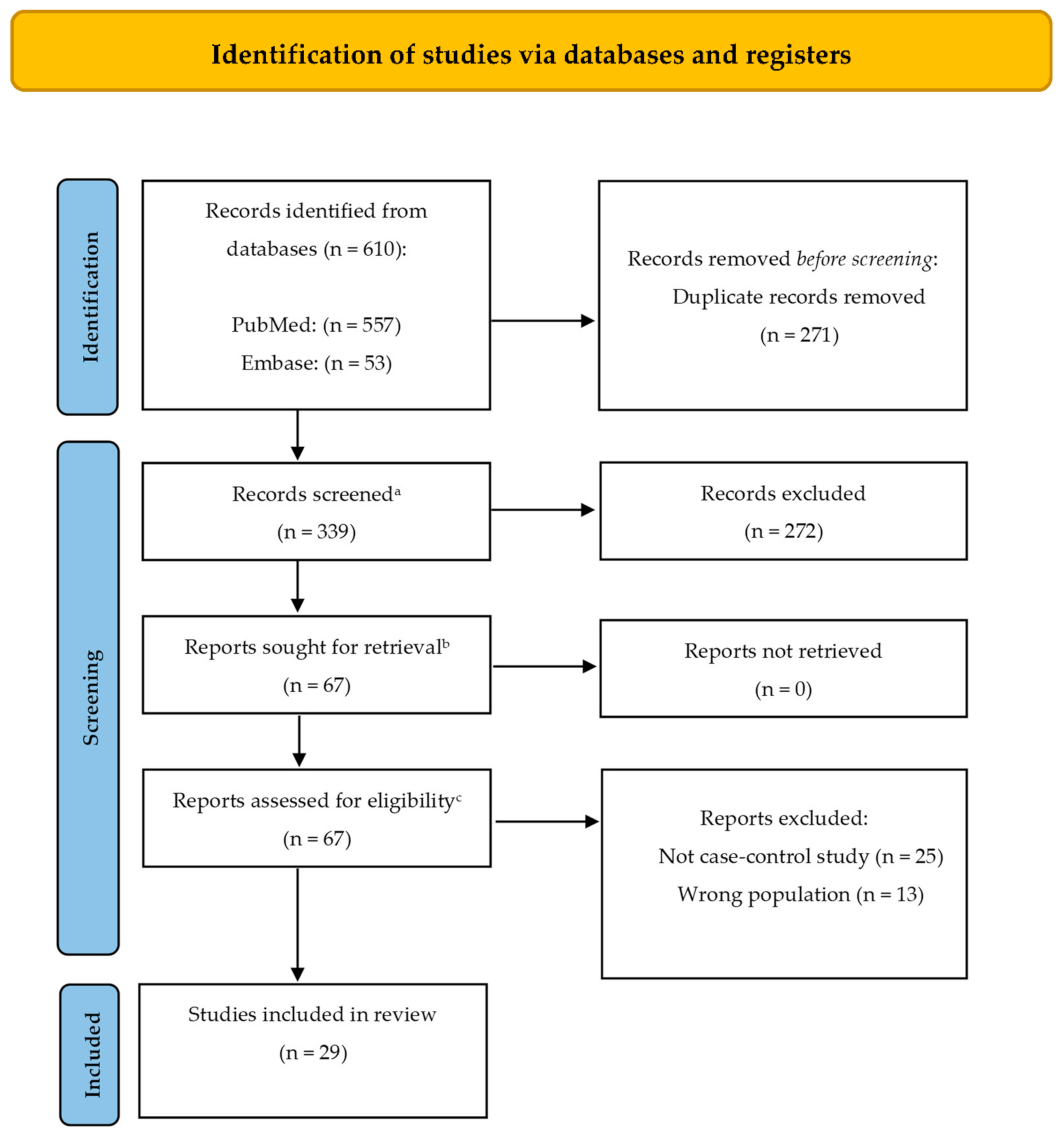
2. Methods
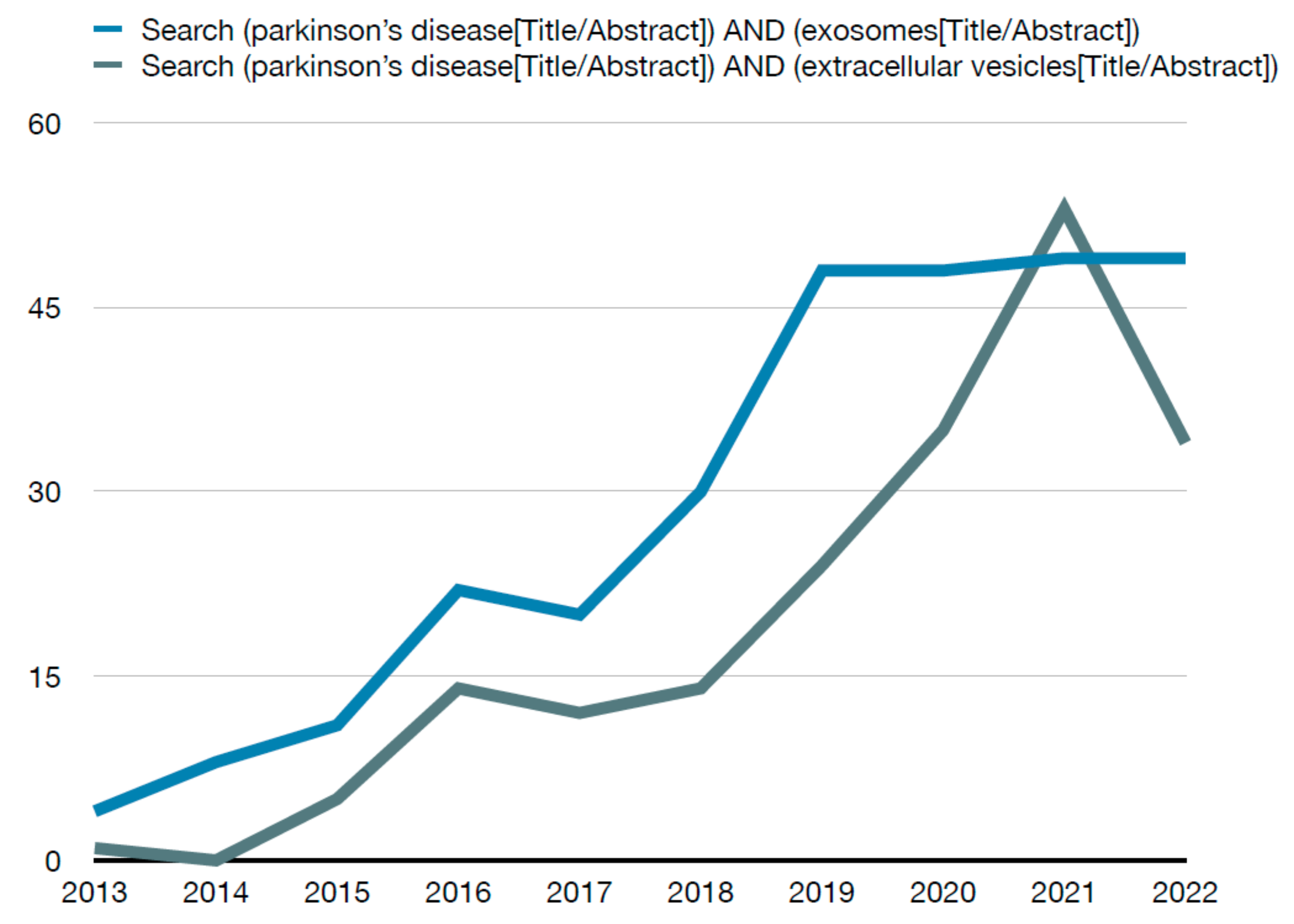
- (exosomes[Title/Abstract]) AND parkinson’s disease[Title/Abstract]
- (exosomes[MeSH Terms]) AND parkinson’s disease[MeSH Terms]
- (extracellular vesicles[Title/Abstract]) AND parkinson’s disease[Title/Abstract]
- (extracellular vesicles[MeSH Terms]) AND parkinson’s disease[MeSH Terms]
- exosomes:ti,ab,kw AND ‘parkinson disease’:ti,ab,kw
- extracellular vesicles:ti,ab,kw AND ‘parkinson disease’:ti,ab,kw
3. Results and Discussion
3.1. α-syn and Its Derivatives
3.2. PrPc, DJ-1, OxiDJ-1 and Tau Protein
3.3. RNAs and Micro RNAs
3.4. Neural-Derived Extracellular Vesicles
3.5. Other Potential Biomarkers
3.6. Neuroinflammation and Neurodegeneration
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Kalia, L.V.; Kalia, S.K.; Lang, A.E. Disease-modifying strategies for Parkinson’s disease. Mov. Disord. 2015, 30, 1442–1450. [Google Scholar] [CrossRef] [PubMed]
- Vandendriessche, C.; Bruggeman, A.; van Cauwenberghe, C.; Vandenbroucke, R.E. Extracellular vesicles in Alzheimer’s and Parkinson’s Disease: Small entities with large consequences. Cells 2020, 9, 2485. [Google Scholar] [CrossRef] [PubMed]
- Kalia, L.V.; Kalia, S.K. α-Synuclein and Lewy pathology in Parkinson’s disease. Curr. Opin. Neurol. 2015, 28, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.U.; Akram, M.; Daniyal, M.; Zainab, R. Awareness and current knowledge of Parkinson’s Disease: A neurodegenerative disorder. Int. J. Neurosci. 2019, 129, 55–93. [Google Scholar] [CrossRef]
- Russo, I.; Bubacco, L.; Greggio, E. Exosomes-associated neurodegeneration and progression of Parkinson’s disease. Am. J. Neurodegener. Dis. 2012, 1, 217. [Google Scholar]
- Zhang, T.M.; Yu, S.Y.; Guo, P.; Du, Y.; Hu, Y.; Piao, Y.S.; Zuo, L.J.; Lian, T.H.; Wang, R.D.; Yu, Q.J.; et al. Nonmotor symptoms in patients with Parkinson disease: A cross-sectional observational study. Medicine 2016, 95, e5400. [Google Scholar] [CrossRef]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef]
- Kalluri, R.; LeBleu, V.S. The biology, function, and biomedical applications of exosomes. Science 2020, 367, eaau6977. [Google Scholar] [CrossRef]
- Porro, C.; Panaro, M.A.; Lofrumento, D.D.; Hasalla, E.; Trotta, T. The multiple roles of exosomes in Parkinson’s disease: An overview. Immunopharmacol. Immunotoxicol. 2019, 41, 469–476. [Google Scholar] [CrossRef]
- De la Torre Gomez, C.; Goreham, R.V.; Bech Serra, J.J.; Nann, T.; Kussmann, M. “Exosomics”-A review of biophysics, biology and biochemistry of exosomes with a focus on human breast milk. Front. Genet. 2018, 9, 92. [Google Scholar] [CrossRef]
- Yáñez-Mó, M.; Siljander, P.R.M.; Andreu, Z.; Zavec, A.B.; Borràs, F.E.; Buzas, E.I.; Buzas, K.; Casal, E.; Cappello, F.; Carvalho, J.; et al. Biological properties of extracellular vesicles and their physiological functions. J. Extracell. Vesicles 2015, 4, 27066. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.F. Extracellular vesicles and neurodegenerative diseases. J. Neurosci. 2019, 39, 9269–9273. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Han, C.L.; Wang, K.L.; Sui, Y.P.; Li, Z.B.; Chen, N.; Fan, S.Y.; Shimabukuro, M.; Wang, F.; Meng, F.G. Integrated analysis of exosomal LncRNA and MRNA expression profiles reveals the involvement of Lnc-MKRN2-42:1 in the pathogenesis of Parkinson’s disease. CNS Neurosci. Ther. 2020, 26, 527–537. [Google Scholar] [CrossRef] [PubMed]
- Cerri, S.; Ghezzi, C.; Sampieri, M.; Siani, F.; Avenali, M.; Dornini, G.; Zangaglia, R.; Minafra, B.; Blandini, F. The Exosomal/Total α-Synuclein ratio in plasma is associated with glucocerebrosidase activity and correlates with measures of disease severity in PD patients. Front. Cell. Neurosci. 2018, 12, 125. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An updated guideline for reporting systematic reviews. Syst. Rev. 2021, 10, 89. [Google Scholar] [CrossRef]
- Leng, B.; Sun, H.; Zhao, J.; Liu, Y.; Shen, T.; Liu, W.; Liu, X.; Tan, M.; Li, F.; Zhang, J.; et al. Plasma exosomal prion protein levels are correlated with cognitive decline in PD patients. Neurosci. Lett. 2020, 723, 134866. [Google Scholar] [CrossRef]
- Jiang, R.; Rong, C.; Ke, R.; Meng, S.; Yan, X.; Ke, H.; Wu, S.; Azim, A. Differential proteomic analysis of serum exosomes reveals alterations in progression of Parkinson disease. Medicine 2019, 98, e17478. [Google Scholar] [CrossRef]
- Zhao, Z.H.; Chen, Z.T.; Zhou, R.L.; Zhang, X.; Ye, Q.Y.; Wang, Y.Z. Increased DJ-1 and α-Synuclein in plasma neural-derived exosomes as potential markers for Parkinson’s disease. Front. Aging Neurosci. 2019, 10, 438. [Google Scholar] [CrossRef]
- Fraser, K.B.; Rawlins, A.B.; Clark, R.G.; Alcalay, R.N.; Standaert, D.G.; Liu, N.; West, A.B. Ser(P)-1292 LRRK2 in urinary exosomes is elevated in idiopathic Parkinson’s disease. Mov. Disord. 2016, 31, 1543–1550. [Google Scholar] [CrossRef]
- Gui, Y.X.; Liu, H.; Zhang, L.S.; Lv, W.; Hu, X.Y. Altered MicroRNA profiles in cerebrospinal fluid exosome in Parkinson disease and Alzheimer disease. Oncotarget 2015, 6, 37043–37053. [Google Scholar] [CrossRef]
- Vacchi, E.; Burrello, J.; di Silvestre, D.; Burrello, A.; Bolis, S.; Mauri, P.; Vassalli, G.; Cereda, C.W.; Farina, C.; Barile, L.; et al. Immune profiling of plasma-derived extracellular vesicles identifies Parkinson disease. Neurol-Neuroimmunol. 2020, 7, e866. [Google Scholar] [CrossRef] [PubMed]
- Lamontagne-Proulx, J.; St-Amour, I.; Labib, R.; Pilon, J.; Denis, H.L.; Cloutier, N.; Roux-Dalvai, F.; Vincent, A.T.; Mason, S.L.; Williams-Gray, C.; et al. Portrait of blood-derived extracellular vesicles in patients with Parkinson’s disease. Neurobiol. Dis. 2019, 124, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Gualerzi, A.; Picciolini, S.; Carlomagno, C.; Terenzi, F.; Ramat, S.; Sorbi, S.; Bedoni, M. Raman profiling of circulating extracellular vesicles for the stratification of Parkinson’s patients. Nanomedicine 2019, 22, 102097. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Wu, Y.; Liu, G.; Jiang, Y.; Wang, X.; Wang, Z.; Zhang, J.; Feng, T. α-Synuclein in salivary extracellular vesicles as a potential biomarker of Parkinson’s disease. Neurosci. Lett. 2019, 696, 114–120. [Google Scholar] [CrossRef]
- Niu, M.; Li, Y.; Li, G.; Zhou, L.; Luo, N.; Yao, M.; Kang, W.; Liu, J. A longitudinal study on α-synuclein in plasma neuronal exosomes as a biomarker for Parkinson’s disease development and progression. Eur. J. Neurol. 2020, 27, 967–974. [Google Scholar] [CrossRef]
- Grossi, I.; Radeghieri, A.; Paolini, L.; Porrini, V.; Pilotto, A.; Padovani, A.; Marengoni, A.; Barbon, A.; Bellucci, A.; Pizzi, M.; et al. MicroRNA-34a-5p expression in the plasma and in its extracellular vesicle fractions in subjects with Parkinson’s disease: An exploratory study. Int. J. Mol. Med. 2021, 47, 533–546. [Google Scholar] [CrossRef]
- Ohmichi, T.; Mitsuhashi, M.; Tatebe, H.; Kasai, T.; Ali El-Agnaf, O.M.; Tokuda, T. Quantification of brain-derived extracellular vesicles in plasma as a biomarker to diagnose Parkinson’s and related diseases. Parkinsonism Relat. Disord. 2019, 61, 82–87. [Google Scholar] [CrossRef]
- Zou, J.; Guo, Y.; Wei, L.; Yu, F.; Yu, B.; Xu, A. Long noncoding RNA POU3F3 and α-synuclein in plasma L1CAM exosomes combined with β-glucocerebrosidase activity: Potential predictors of Parkinson’s disease. Neurotherapeutics 2020, 17, 1104–1119. [Google Scholar] [CrossRef]
- Chung, C.C.; Chan, L.; Chen, J.H.; Hung, Y.C.; Hong, C.T. Plasma extracellular vesicle α-synuclein level in patients with Parkinson’s disease. Biomolecules 2021, 11, 744. [Google Scholar] [CrossRef]
- Stuendl, A.; Kunadt, M.; Kruse, N.; Bartels, C.; Moebius, W.; Danzer, K.M.; Mollenhauer, B.; Schneider, A. Induction of α-synuclein aggregate formation by CSF exosomes from patients with Parkinson’s disease and dementia with Lewy bodies. Brain 2016, 139, 481–494. [Google Scholar] [CrossRef]
- Cao, X.Y.; Lu, J.M.; Zhao, Z.Q.; Li, M.C.; Lu, T.; An, X.S.; Xue, L.J. MicroRNA biomarkers of Parkinson’s disease in serum exosome-like microvesicles. Neurosci. Lett. 2017, 644, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Jiang, C.; Tofaris, G.K.; Davis, J.J. Facile Impedimetric analysis of neuronal exosome markers in Parkinson’s disease diagnostics. Anal. Chem. 2020, 92, 13647–13651. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.F.; Qu, M.W.; Li, G.C.; Zhang, F.B.; Rui, H.C. Circulating exosomal MiRNAs as diagnostic biomarkers in Parkinson’s disease. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 5278–5283. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Xie, Z.; Zhang, X.; Mao, J.; Wang, M.; Wei, S.; Fu, Y.; Zheng, H.; He, Y.; Chen, H.; et al. Investigation of α-synuclein species in plasma exosomes and the oligomeric and phosphorylated α-synuclein as potential peripheral biomarker of Parkinson’s disease. Neuroscience 2021, 469, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, Y.; Kojima, M.; Kurosawa, T.; Sasaki, R.; Ichihara, S.; Hiraku, Y.; Tomimoto, H.; Murata, M.; Oikawa, S. Proteomic profiling of exosomal proteins for blood-based biomarkers in Parkinson’s disease. Neuroscience 2018, 392, 121–128. [Google Scholar] [CrossRef]
- Rani, K.; Mukherjee, R.; Singh, E.; Kumar, S.; Sharma, V.; Vishwakarma, P.; Bharti, P.S.; Nikolajeff, F.; Dinda, A.K.; Goyal, V.; et al. Neuronal exosomes in saliva of Parkinson’s disease patients: A pilot study. Parkinsonism Relat. Disord. 2019, 67, 21–23. [Google Scholar] [CrossRef]
- Shi, M.; Kovac, A.; Korff, A.; Cook, T.J.; Ginghina, C.; Bullock, K.M.; Yang, L.; Stewart, T.; Zheng, D.; Aro, P.; et al. CNS Tau efflux via exosomes is likely increased in Parkinson’s disease but not in Alzheimer’s disease. Alzheimer Dement. 2016, 12, 1125–1131. [Google Scholar] [CrossRef]
- Xia, Y.; Zhang, G.; Han, C.; Ma, K.; Guo, X.; Wan, F.; Kou, L.; Yin, S.; Liu, L.; Huang, J.; et al. Microglia as modulators of exosomal alpha-synuclein transmission. Cell Death Dis. 2019, 10, 174. [Google Scholar] [CrossRef]
- Si, X.; Tian, J.; Chen, Y.; Yan, Y.; Pu, J.; Zhang, B. Central nervous system-derived exosomal alpha-synuclein in serum may be a biomarker in Parkinson’s disease. Neuroscience 2019, 413, 308–316. [Google Scholar] [CrossRef]
- Jang, J.; Jeong, S.; Lee, S.I.; Seol, W.; Seo, H.; Son, I.; Ho, D.H. Oxidized DJ-1 levels in urine samples as a putative biomarker for Parkinson’s disease. Parkinsons Dis. 2018, 2018, 1241757. [Google Scholar] [CrossRef]
- Lucien, F.; Benarroch, E.E.; Mullan, A.; Ali, F.; Boeve, B.F.; Mielke, M.M.; Petersen, R.C.; Kim, Y.; Stang, C.; Camerucci, E.; et al. Poly (ADP-Ribose) and α-synuclein extracellular vesicles in patients with Parkinson disease: A possible biomarker of disease severity. PLoS ONE 2022, 17, e0264446. [Google Scholar] [CrossRef] [PubMed]
- Kluge, A.; Bunk, J.; Schaeffer, E.; Drobny, A.; Xiang, W.; Knacke, H.; Bub, S.; Lückstädt, W.; Arnold, P.; Lucius, R.; et al. Detection of neuron-derived pathological α-synuclein in blood. Brain 2022, 145, 3058–3071. [Google Scholar] [CrossRef] [PubMed]
- Rocha, E.M.; de Miranda, B.; Sanders, L.H. Alpha-synuclein: Pathology, mitochondrial dysfunction and neuroinflammation in Parkinson’s DISEase. Neurobiol. Dis. 2018, 109, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Delamarre, A.; Meissner, W.G. Epidemiology, Environmental risk factors and genetics of Parkinson’s disease. Presse Medicale 2017, 46, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Sun, T.; An, J.; Wen, L.; Liu, F.; Bu, Z.; Cui, Y.; Feng, J. Potential roles of exosomes in Parkinson’s Disease: From pathogenesis, diagnosis, and treatment to prognosis. Front. Cell. Dev. Biol. 2020, 8, 86. [Google Scholar] [CrossRef] [PubMed]
- Danzer, K.M.; Kranich, L.R.; Ruf, W.P.; Cagsal-Getkin, O.; Winslow, A.R.; Zhu, L.; Vanderburg, C.R.; McLean, P.J. Exosomal cell-to-cell transmission of alpha synuclein oligomers. Mol. Neurodegener. 2012, 7, 42. [Google Scholar] [CrossRef]
- Li, J.Y.; Englund, E.; Holton, J.L.; Soulet, D.; Hagell, P.; Lees, A.J.; Lashley, T.; Quinn, N.P.; Rehncrona, S.; Björklund, A.; et al. Lewy bodies in grafted neurons in subjects with Parkinson’s Disease suggest host-to-graft disease propagation. Nat. Med. 2008, 14, 501–503. [Google Scholar] [CrossRef]
- Emmanouilidou, E.; Melachroinou, K.; Roumeliotis, T.; Garbis, S.D.; Ntzouni, M.; Margaritis, L.H.; Stefanis, L.; Vekrellis, K. Cell-Produced alpha-synuclein is secreted in a calcium-dependent manner by exosomes and impacts neuronal survival. J. Neurosci. 2010, 30, 6838–6851. [Google Scholar] [CrossRef]
- Stefanis, L.; Emmanouilidou, E.; Pantazopoulou, M.; Kirik, D.; Vekrellis, K.; Tofaris, G.K. How Is alpha-synuclein cleared from the cell? J. Neurochem. 2019, 150, 577–590. [Google Scholar] [CrossRef]
- Kenwrick, S.; Watkins, A.; de Angelis, E. Neural cell recognition molecule L1: Relating biological complexity to human disease mutations. Hum. Mol. Genet. 2000, 9, 879–886. [Google Scholar] [CrossRef]
- Mollenhauer, B.; Batrla, R.; El-Agnaf, O.; Galasko, D.R.; Lashuel, H.A.; Merchant, K.M.; Shaw, L.M.; Selkoe, D.J.; Umek, R.; Vanderstichele, H.; et al. A user’s guide for a-synuclein biomarker studies in biological fluids: Perianalytical considerations. Mov. Dis. 2017, 32, 1117–1130. [Google Scholar] [CrossRef] [PubMed]
- Elkouris, M.; Kouroupi, G.; Vourvoukelis, A.; Papagiannakis, N.; Kaltezioti, V.; Matsas, R.; Stefanis, L.; Xilouri, M.; Politis, P.K. Long non-coding rnas associated with neurodegeneration-linked genes are reduced in Parkinson’s disease patients. Front. Cell. Neurosci. 2019, 13, 58. [Google Scholar] [CrossRef] [PubMed]
- Riva, P.; Ratti, A.; Venturin, M. The long non-coding RNAs in neurodegenerative diseases: Novel mechanisms of pathogenesis. Curr. Alzheimer Res. 2016, 13, 1219–1231. [Google Scholar] [CrossRef]
- Caggiu, E.; Paulus, K.; Mameli, G.; Arru, G.; Sechi, G.P.; Sechi, L.A. Differential expression of MiRNA 155 and MiRNA 146a in Parkinson’s disease patients. eNeurologicalSci 2018, 13, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Zhao, X.; Lu, K.; Cheng, G. LncRNA BDNF-AS Promotes autophagy and apoptosis in MPTP-induced Parkinson’s disease via ablating MicroRNA-125b-5p. Brain Res. Bull. 2020, 157, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Huang, X.; Li, R. LncRNA MALAT1/MiR-205-5p axis regulates MPP+-induced cell apoptosis in MN9D cells by directly targeting LRRK2. Am. J. Transl. Res. 2018, 10, 563. [Google Scholar] [PubMed]
- Calabrese, V.; Santoro, A.; Monti, D.; Crupi, R.; di Paola, R.; Latteri, S.; Cuzzocrea, S.; Zappia, M.; Giordano, J.; Calabrese, E.J.; et al. Aging and Parkinson’s Disease: Inflammaging, neuroinflammation and biological remodeling as key factors in pathogenesis. Free Radic. Biol. Med. 2018, 115, 80–91. [Google Scholar] [CrossRef] [PubMed]
- Ravanidis, S.; Bougea, A.; Papagiannakis, N.; Maniati, M.; Koros, C.; Simitsi, A.M.; Bozi, M.; Pachi, I.; Stamelou, M.; Paraskevas, G.P.; et al. Circulating brain-enriched MicroRNAs for detection and discrimination of idiopathic and genetic Parkinson’s disease. Mov. Disord. 2020, 35, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Gan-Or, Z.; Liong, C.; Alcalay, R.N. GBA-associated Parkinson’s disease and other synucleinopathies. Curr. Neurol. Neurosci. Rep. 2017, 18, 1–10. [Google Scholar] [CrossRef]
- Schapira, A.H.V. Glucocerebrosidase and Parkinson disease: Recent advances. Mol. Cell. Neurosci. 2015, 66, 37–42. [Google Scholar] [CrossRef]
- Manna, I.; Quattrone, A.; de Benedittis, S.; Iaccino, E.; Quattrone, A. Roles of non-coding RNAs as novel diagnostic biomarkers in Parkinson’s disease. J. Parkinsons Dis. 2021, 11, 1475–1489. [Google Scholar] [CrossRef] [PubMed]
- Ngolab, J.; Trinh, I.; Rockenstein, E.; Mante, M.; Florio, J.; Trejo, M.; Masliah, D.; Adame, A.; Masliah, E.; Rissman, R.A. Brain-derived exosomes from dementia with Lewy bodies propagate α-synuclein pathology. Acta Neuropathol. Commun. 2017, 5, 46. [Google Scholar] [CrossRef] [PubMed]
- Norman, M.; Ter-Ovanesyan, D.; Trieu, W.; Lazarovits, R.; Kowal, E.J.K.; Lee, J.H.; Chen-Plotkin, A.S.; Regev, A.; Church, G.M.; Walt, D.R. L1CAM is not associated with extracellular vesicles in human cerebrospinal fluid or plasma. Nat. Methods 2021, 18, 631–634. [Google Scholar] [CrossRef] [PubMed]
- Van de Bovenkamp, F.S.; Dijkstra, D.J.; van Kooten, C.; Gelderman, K.A.; Trouw, L.A. Circulating C1q levels in health and disease, more than just a biomarker. Mol. Immunol. 2021, 140, 206–216. [Google Scholar] [CrossRef]
- Breuer, M.; Guglielmi, L.; Zielonka, M.; Hemberger, V.; Kölker, S.; Okun, J.G.; Hoffmann, G.F.; Carl, M.; Sauer, S.W.; Opladen, T. QDPR homologues in danio rerio regulate melanin synthesis, early gliogenesis, and glutamine homeostasis. PLoS ONE 2019, 14, e0215162. [Google Scholar] [CrossRef]
- Li, Y.; Schrodi, S.; Rowland, C.; Tacey, K.; Catanese, J.; Grupe, A. Genetic evidence for ubiquitin-specific proteases USP24 and USP40 as candidate genes for late-onset Parkinson disease. Hum. Mut. 2006, 27, 1017–1023. [Google Scholar] [CrossRef]
- Ransohoff, R.M. How neuroinflammation contributes to neurodegeneration. Science 2016, 353, 777–783. [Google Scholar] [CrossRef]
- Console, L.; Scalise, M.; Indiveri, C. Exosomes in inflammation and role as biomarkers. Clin. Chim. Acta 2019, 488, 165–171. [Google Scholar] [CrossRef]
- Finkbeiner, S. The autophagy lysosomal pathway and neurodegeneration. Cold Spring Harb. Perspect. Biol. 2020, 12, a033993. [Google Scholar] [CrossRef]
| Object of Study | Sample Size * (Total/PD) | Type of Biological Sample | EVs Isolation Method | Main Outcomes | Ref. |
|---|---|---|---|---|---|
| Plasma exosomal prion proteins | 60/40 | Blood | Centrifugation and ExoQuick Plasma prep and exosome precipitation kit | Plasma exosomal prion protein levels were higher in PD group than in HC. It was also elevated in the PD-CI group compared to the PD-NCI group. Its concentration increased with age in HC, but no correlation was found in the PD group. It was positively associated with impaired cognitive level, visual spatial function, memory, attention and calculation abilities. Thus, plasma exosomal prion protein could be used as a biomarker for cognitive decline in PD patients. | [16] |
| Serum-derived exosome protein content | 30/20 | Blood | Ultracentrifugation | A total of 429 proteins were detected. Among these, 9 proteins were only detected in serum exosomes of patients with severe PD: protein S100, tyrosine protein kinase receptor, lactoferrin, dermcidin, platelet-activating factor acetyl hydrolase and isocitrate dehydrogenase. Clusterin, complement C1r subcomponent, afamin, angiotensinogen variant, apolipoprotein D, gelsolin, and PEDF were progressively upregulated from mild to severe PD. On the other hand, human neuroblastoma full-length cDNA clone CS0DD006YL02, precursor (AA-19 to 113), complement C1q subcomponent, myosin-reactive immunoglobulin kappa chain, Ig kappa chain V-III region, immunoglobulin mu chain, and immunoglobulin kappa variables 1 to 33 were gradually downregulated from mild to severe PD. A proteomic approach could be used to detect variations in the protein profile in different stages of PD development. Larger cohorts are needed. | [17] |
| DJ-1 and α-syn in plasma neural-derived exosomes | 78/39 | Blood | Centrifugation and precipitation | DJ-1 and α-syn levels from plasma neural-derived exosomes so as the ratio of plasma neural-derived exosomal DJ-1/total DJ-1 were significantly higher in the PD group than HC. No differences were observed between DJ-1 and α-syn levels from plasma neural-derived exosomes between PD patients at different stages of disease. They could serve as biomarkers for PD detection. | [18] |
| Exosomal Ser(P)-1292 LRRK2 | 158/79 | Urine | Differential ultracentrifugation | Exosomal Ser(P)-1292 LRRK2 levels were significantly elevated in the PD group compared to HC; it was also positively correlated with multiple nonmotor measures of PD (MoCA, MDS-UPDRS Part I and II, Epworth SS). | [19] |
| Exosomal miRNA in CSF | 102/47 | Cerebrospinal fluid | Of a total of 746 exosomal miRNAs profiled, 27 of them were differentially expressed in CSF from PD patients compared to HC. Among them, 16 were upregulated (hsa-mir-103a, hsa-mir-30b, hsa-mir-16-2, hsa-mir-26a, hsa-mir-331-5p, hsa-mir-153, hsa-mir-132-5p, hsa-mir-485-5p, hsa-mir-127-3p, hsa-mir-409-3p, hsa-mir-433, hsa-mir-370, hsa-let-7g-3p, hsa-mir-873-3p, hsa-mir-136-3p, hsa-mir-10a-5p) and 11 were downregulated (hsa-mir-1, hsa-mir-22, hsa-mir-29, hsa-mir-374, hsa-mir-119a, hsa-mir-126, hsa-mir-151, hsa-mir-28, hsa-mir-301a, hsa-mir-19b-3p, hsa-mir-29c). Through DIANA-miRPath, the dysregulated exosomal miRNA signatures were associated with the neurotrophin signaling pathway, mTOR signaling pathway, ubiquitin-mediated proteolysis, long-term potentiation, axon guidance, cholinergic synapse, gap junction, dopaminergic synapse and glutamatergic synapse. Six miRNAs were selected for further validation (miR-1, miR-153, miR-409-3p, miR-19b-3p, miR-10a-5p, let-7g-3p) confirming that these miRNAs highly discriminated PD patients from HC. | [20] | |
| Immune profiling of plasma-derived EVs | 63/27 | Blood | MACSPlex Human Exosome Kit | Plasma EV concentration was higher in patients with PD. Sixteen markers showed differences between the two PD and HC: CD4, CD19, CD45, CD1c, CD2, CD11c, CD31, CD41, CD42a, CD62, CD146, melanoma-associated chondroitin sulfate proteoglycan (MCSP), CD25, CD40, CD20 and HLA-ABC. Among the association of EV surface antigens as discriminants for PD diagnosis (eleven markers total), six were exclusive of the PD group (CD1c, CD11c, CD19, CD41b, CD45 and CD146). Through supervised machine learning algorithms, the combination of multiple EV specific markers showed a high sensitivity and specificity for the diagnosis of PD (AUC 0.908). Machine learning algorithms, based on EV-specific signature, discriminated patients with PD and MSA. | [21] |
| Profile and protein signature of EV in plasma and EV derived from erythrocytes | 97/60 ** | Blood | Centrifugation/fluorescence-activated cell sorting (FACS, Canto II Special Order Research Product) | A large cohort and patients in early stages of the disease are essential for future study. There was no difference in the number of EEV containing α-Syn and phospho α-Syn (serine 129) nor in the levels of α-Syn between PD patients and HC. Proteomic analyses of EEV of PD patients were performed. A total of 818 proteins were identified by removing hemoglobin from erythrocytes (in contrast to 356 without removing hemoglobin). Eight of these proteins were highly expressed and mapped the disease stage according to UPDRS scores, allowing them to be divided in three groups: Group I were highly expressed in controls (ABHD14B, alpha/beta hydrolase domain-containing protein 14B; AIDA, axin interactor dorsalization-associated protein; NADSYN1, glutamine-dependent NAD(+) synthetase) Group II expressed in mild PD patients (QDPR, quinoid dihydro pteridine reductase; AKR1A1, alcohol dehydrogenase NADP+; NRIP1, cannabinoid receptor-interacting protein 1. Group III predominantly expressed in moderate PD patients (USP24, ubiquitin carboxyl-terminal hydrolase 24; ATP5A1, ATP synthase subunit alpha mitochondrial). | [22] |
| Biochemistry of circulating EVs by Raman spectroscopy (RS) | 40/22 | Blood | Size exclusion chromatography/ultracentrifugation | Raman spectroscopy analysis demonstrated there were biochemical differences between circulating EVs from the PD group vs. HC, in particular, involving proteins, lipids and saccharides. RS could discern between PD patients from HC with an accuracy of 71% (p = 0.013). Blood-derived EVs from PD patients’ biochemical signature can be correlated to clinical scores measured by HY (describes degree of progression) and UPDRS part III (describes motor impairment) scores. | [23] |
| α-syn in salivary exosomes | 134/74 | Saliva | Centrifugation, precipitation and XYCQ Enrichment Kit | α-syn oligomer levels (2.05 pg/ng) and α-syn oligomer/α-syn total ratio (0.18 pg/ng) in salivary exosomes are higher in PD group than in HC group and may serve as a potential diagnostic biomarker for PD. | [24] |
| α-syn in plasma neuronal exosomes | 94/53 | Blood | Antibody-coated superparamagnetic microbeads | α-syn levels in plasma neuronal exosomes were significantly higher in patients with early stage PD compared with HCs (p = 0.007). Moreover, its concentrations had correlation with UPDRS III/(I,II,III) scores, NMSQ scores and SS-16 scores of patients with PD. α-syn levels in plasma neuronal exosomes could distinguish between early stage PD patients and HCs (AUC, 0.8; sensitivity, 100%; specificity, 57.1%). In a longitudinal study (n = 18 early stage PD), an increase in neuronal exosomal α-syn levels was associated with a higher risk of motor progression. | [25] |
| Circulating miR-34a-5p in small extracellular vesicles (SEVs) | 29/15 | Blood | Ultracentrifugation/density gradient centrifugation | miR-34a-5p levels were significantly overexpressed in pure SEVs from the plasma of PD patients compared to controls. In addition, miR-34a-5p expression in pure SEVs revealed a good ability to distinguish PD patients from control subjects (AUC, 0.738) suggesting its potential consideration as a marker of diagnosis at a molecular level. Pure SEVs miR-34a-5p levels were higher in PD patients even at the beginning stage of PD when the disease duration was less than 5 years. High levels of pure SEV miR-34a-5p were detected in PD patients with mild/progressive symptoms of disease and were associated with minimal/absent depression. There is the necessity to consider not only the whole plasma, but each EV subpopulation in order to improve the possibility to identify relevant differences of specific miRNAs levels. For this purpose, the SEVs purification protocol is crucial. | [26] |
| Brain-derived exosomes in plasma | 52/15 | Blood | Centrifugation | Plasma levels of brain-derived exosomes (BDE) were significantly higher in advanced PD compared to the HC group. Plasma levels of neuron-derived exosomes (NDE) and oligodendrocyte-derived exosomes (ODE) were higher even in mild PD compared to the HC group. These results suggested the capability of NDE and ODE as diagnostic biomarkers for PD. ODE levels were significantly higher in moderate to advanced disease, indicating it could be a biomarker for monitoring disease progression. | [27] |
| Linc-POU3F3 and α-syn levels in L1CAM exosomes and GCase activity. | 178/93 | Blood | Ultracentrifugation/antibody-coated superparamagnetic microbeads | The increase of L1CAM exosomal Linc-POU3F3 levels in plasma PD patients was positively correlated with disease severity (H-Y score and UPDRS-III). The L1CAM exosomal α-syn concentration and the ratio of exosomal α-syn to total α-syn (exo/total) were significantly increased in PD patients compared with healthy controls. GCase activity levels were decreased in PD vs. controls and negatively correlated with increased Linc-POU3F3 levels in the exosomal L1CAM of PD patients. The combination of plasma L1CAM exosomal Linc-POU3F3, plasma L1CAM exosomal α-syn and GCase activity showed to be more reliable (AUC 0.824) in distinguishing PD vs. controls than each individually. L1CAM exosomal Linc-POU3F3, α-syn levels in L1CAM exosomes and GCase activity may be potential diagnostic biomarkers and useful tools to evaluate the severity of PD. | [28] |
| Plasma EV α-syn | 162/116 | Blood | Size-exclusion chromatography (exoEasy Maxi kit) | Plasma EV α-syn levels were significantly decreased in the PD vs. control group and showed a negative association with akinetic rigidity syndromes severity in PD patients. Future large cohorts and investigations are necessary. | [29] |
| Exosomal α-syn from CSF | 134/76 | Cerebrospinal fluid | Centrifugation and ultracentrifugation | Total α-syn levels in CSF were significantly lower in the PD group than in the HC group. The CSF exosomal levels of α-syn were lower in the PD group than in the HC group. CSF exosomal levels of α-syn could serve as diagnostic biomarkers. | [30] |
| miRNAs in serum exosomes | 149/109 | Blood | Centrifugation/exosome isolation reagent from body fluids (Invitrogen) | A total of 24 previously reported miRNAs were analyzed in PD patients and an HC group. Among these, three had consistent results. The levels of miR-24 and miR-195 were significantly higher in serum samples from the PD group vs. HC (p < 0.05). On the contrary, miR-19b levels were significantly lower in PD vs. HC serum samples (p < 0.05). The sensitivity and specificity for diagnostic value in PD were: miR-195, 82.6% and 55.0%; miR-19b, 68.8% and 77.5; and miR-24, 81.7% and 85.0%, respectively. The three miRNAs (miR-24, miR-195, miR-19b) together, presented an AUC of 0.946 (95% CI, 0.910–0.981); the specificity was 90.0% and the sensitivity was 85.3%. The levels of miR-195, miR-19b and miR-24 may be useful noninvasive biomarkers for the diagnosis of PD. Future research is needed to identify the biological function of miR-24, miR-195 and miR-19b. | [31] |
| Neuronal exosomal α-syn | 40/20 | Blood | Immunoaffinity-based technology (magnetic beads coated with zwitterionic polymer pCBMA), conjugated with anti-L1CAM antibody. | The concentration of exosomes extracted by the precipitation method was significantly higher compared to the one obtained with ultracentrifugation. pCBMA@Fe3O4 MBs (magnetic beads coated with zwitterionic polymer pCBMA) conjugated with the anti-L1CAM antibody were effective in isolating neuronal-derived exosomes from serum and allowed them to perform electroanalyses with lower levels of serum compared to ELISA or electrochemiluminescence. The total quantification of α-syn using EIS was higher than the one detected by electrochemiluminescence. Levels of α-syn in neuronal exosomes in PD were higher compared to control. Neuronal exosome-associated Synt-1 content did not show any difference between the two groups. | [32] |
| Exosomal miRNAs | 100/52 | Blood | PureExo Exosome Isolation Kit (precipitation) and centrifugation | Exosomal miR-331-5p was significantly higher in PD patients than in the HC group. It was found mainly in exosomes. It is thought that miR-331-5p is transferred to PD-related cells through exosomes, being involved in the pathological process of PD. | [33] |
| Oligomeric α-syn, phosphorylated α-syn, and total α-syn in plasma exosomes | 72/36 | Blood | Precipitation (Total Exosome Isolation kit, Invitrogen) and differential centrifugation | The ratio of plasma exosomal α-syn/total α-syn monomers was significantly lower in PD patients vs. controls compared to higher levels in PD vs. controls in the ratio of α-syn/total α-syn oligomers and p-α-syn/p-α-syn oligomers. Triton X-100 insoluble α-syn and p-α-syn in the PD group was significantly higher than in HC compared to the soluble α-syn components. After treatment with different concentrations of PK, plasma exosomal insoluble α-syn components in the PD group were higher compared to controls. p-α-syn in PD patients’ plasma exosomes was more difficult to degrade by PK than that of the healthy controls. The levels of α-syn oligomer/total α-syn and p-α-syn oligomer/total p-α-syn in plasma exosomes of PD patients were higher than in the HC group. These results may indicate the PD pathological changes. ROC performance of both α-syn oligomer/total α-syn and p-α-syn oligomers/total p-α-syn (0.71, 0.69, respectively) in exosomes was moderate and may be a helpful tool in PD diagnosis. | [34] |
| Plasma-derived exosome protein content | 24 | Blood | Size exclusion chromatography on drip column (EV-Second) | Exosomal apolipoprotein A1 levels in PD patients at HY stage III were significantly decreased compared to HY stage II patients and correlated with the progression of the disease. Apolipoprotein A1, clusterin, complement C1r subcomponent and fibrinogen gamma chain exosomal expression levels may serve as a biomarker for disease progression. | [35] |
| Salivary exosomes from neuronal origin and its α-syn levels | 36/18 | Saliva | Centrifugation | Salivary exosomal phospho α-syn levels were higher in the PD group than in the HC group due to the higher secretion of exosomes from neuronal endings in salivary glands in the PD group. It could serve as a biomarker for early PD detection and a tool to measure disease progression in drug efficacy studies. | [36] |
| Central nervous system (CNS) exosomal tau in peripheral blood | 303/91 | Blood and cerebrospinal fluid | Centrifugation | Mean plasma exosomal tau was significantly higher in the PD group than in the HC group. In the PD group, plasma exosomal tau correlated with CSF t-tau and p-tau. Tau in L1CAM-containing exosomes was associated with disease duration. CNS-derived tau species could be used as PD biomarkers in plasma exosomes. | [37] |
| lncRNA in peripheral exosomes | 14/7 | Blood | Ultracentrifugation | A total of 15 upregulated and 24 downregulated lncRNAs were found. Of those, MSTRG.242001.1 and MSTRG.169261.1 were highly expressed among PD patients and MSTRG.336210.1 and lnc-MKRN2-42:1 among HC. A GO analysis of these lncRNAs showed their involvement in intracellular part, single-organism cellular process and heterocyclic compound binding. lnc-MKRN2-42:1 was found to regulate genes involved in apoptosis, synaptic remodeling, long-term potential, immunity and glutamate neurotransmitter metabolism. Thus, it was selected for further analysis, which showed that its expression was correlated with the MDS-UPDRS III score among PD patients, measuring the severity of dyskinesia and dysarthria. | [13] |
| Plasma exosomal α-syn | 35/20 | Blood | Differential centrifugation/ultracentrifugation | Exosomal total α-syn was elevated in plasma from the PD group compared to HC. Levels of exosomal α-syn oligomers and monomers in plasma were higher in the PD group than in HC group. The identification in plasma exosomal α-syn oligomers could possibly be used as a potential biomarker. | [38] |
| L1CAM-exosomal α-syn from CNS | 77/38 | Blood | Centrifugation and ExoQuick exosome precipitation solution | The mean value of α-syn in L1CAM-containing exosomes was lower in the PD group than in the ET and HC groups. It was lower in NTD-PD (nontremor-dominant group) compared to the TD-PD group (tremor-dominant group). CNS-derived exosomal α-syn in serum may be inverse to the course and severity in PD patients, helping diagnose PD patients and differentiating motor types in early stages. | [39] |
| Exosomal OxiDJ-1 in urine | 55/33 | Urine | Centrifugation and filtration | OxiDJ-1 levels in urine were higher in the PD group than HC. It could serve as a diagnostic biomarker for PD diagnosis. | [40] |
| Plasma exosomal α-syn | 72/39 | Blood | Centrifugation and ultracentrifugation | α-syn concentration in plasma exosomes was higher in PD patients than HC, confirming it is associated with the pathological status. | [14] |
| Poly (ADP-Ribose) and α–syn extracellular vesicles | 117/57 | Blood | Centrifugation/incubation with fluorescent-labelled primary antibodies against total α-syn | Median concentration of α-syn extracellular vesicles was significantly higher in PD patients compared to the other groups (Kruskal–Wallis, p < 0.0001). | [41] |
| α-syn conformers | 80/30 | Blood | Centrifugation and precipitation | The detection of pathological α-syn conformers from neuron-derived extracellular vesicles from blood plasma samples has the potential to evolve into a blood-biomarker of PD | [42] |
| Potential Biomarkers | Specifications | Type of Biological Sample | Ref. |
|---|---|---|---|
| α-syn | Plasma neural-derived exosomes | Blood | [18] |
| α-syn | Plasma neuronal exosomes | Blood | [25] |
| α-syn | Plasma EVs | Blood | [29] |
| α-syn | CSF exosomal levels | Cerebrospinal fluid | [30] |
| α-syn | Neuronal exosomes | Blood | [32] |
| α-syn conformers | Plasma EVs | Blood | [42] |
| Oligomeric α-syn and total α-syn | Salivary EVs | Saliva | [24] |
| Oligomeric α-syn, phosphorylated α-syn and total α-syn | Plasma exosomes | Blood | [34] |
| Phosphorylated α-syn | Neuronal exosomes | Saliva | [35] |
| Poly (ADP-Ribose) and α–syn | Plasma EVs | Blood | [41] |
| PrPc (cell prion protein) | Plasma exosomal prion protein | Blood | [16] |
| DJ-1 | Plasma neural-derived exosome | Blood | [18] |
| OxiDJ-1 | Urine exosomes | Urine | [39] |
| tau protein | L1CAM exosomal tau in plasma and cerebrospinal fluid | Blood and cerebrospinal fluid | [36] |
| miR-1, miR-153, miR-409-3p, miR-19b-3p, miR-10a-5p and let-7g-3p | miRNAs in cerebrospinal fluid (CSF) exosomes | Cerebrospinal fluid | [20] |
| miR-24, miR-195 and miR-19b | miRNAs in serum exosomes | Blood | [31] |
| miR 34a 5p | Circulating miR-34a-5p in small extracellular vesicles (SEVs) | Blood | [26] |
| miR-331-5p and miR-505 | Plasma exosomes circulating miRNAs | Blood | [33] |
| lnc-MKRN2-42:1 | lncRNA in plasma exosomes | Blood and cerebrospinal fluid | [13] |
| Ser(P)-1292 LRRK2 | Autophosphorylated LRRK2 protein in urinary exosomes | Urine | [19] |
| Linc-POU3F3 and α-syn levels and GCase activity. | Plasma L1CAM exosomal levels | Blood | [28] |
| CD1c, CD11c, CD19, CD41b, CD45 and CD146 | EVs surface antigens expression in plasma | Blood | [21] |
| NDEs (neuron-derived exosomes) and ODE (oligodendrocyte-derived exosomes) | Quantification of brain derived exosomes in plasma | Blood | [27] |
| Apolipoprotein A1, clusterin, complement C1r subcomponent and fibrinogen gamma chain | Plasma-derived protein content | Blood | [35] |
| Clusterin, complement C1r subcomponent, afamin, angiotensinogen variant, apolipoprotein D, gelsolin, PEDF, human neuroblastoma full-length cDNA clone CS0DD006YL02, precursor (AA-19 to 113), complement C1q subcomponent, myosin-reactive immunoglobulin kappa chain, Ig kappa chain V-III region, immunoglobulin mu chain and immuno-globulin kappa variables 1 to 33. | Proteomic analysis of serum exosomes | Blood | [17] |
| QDPR, Quinoid Dihydro pteridine Reductase; AKR1A1, alcohol dehydrogenase NADP+; NRIP1, cannabinoid receptor-interacting protein 1; USP24, ubiquitin carboxyl-terminal hydrolase 24 and ATP5A1, ATP synthase subunit alpha mitochondrial | Proteomic analysis of EVs derived from erythrocytes | Blood | [22] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valencia, J.; Ferreira, M.; Merino-Torres, J.F.; Marcilla, A.; Soriano, J.M. The Potential Roles of Extracellular Vesicles as Biomarkers for Parkinson’s Disease: A Systematic Review. Int. J. Mol. Sci. 2022, 23, 11508. https://doi.org/10.3390/ijms231911508
Valencia J, Ferreira M, Merino-Torres JF, Marcilla A, Soriano JM. The Potential Roles of Extracellular Vesicles as Biomarkers for Parkinson’s Disease: A Systematic Review. International Journal of Molecular Sciences. 2022; 23(19):11508. https://doi.org/10.3390/ijms231911508
Chicago/Turabian StyleValencia, Jessica, Marta Ferreira, J. Francisco Merino-Torres, Antonio Marcilla, and Jose M. Soriano. 2022. "The Potential Roles of Extracellular Vesicles as Biomarkers for Parkinson’s Disease: A Systematic Review" International Journal of Molecular Sciences 23, no. 19: 11508. https://doi.org/10.3390/ijms231911508
APA StyleValencia, J., Ferreira, M., Merino-Torres, J. F., Marcilla, A., & Soriano, J. M. (2022). The Potential Roles of Extracellular Vesicles as Biomarkers for Parkinson’s Disease: A Systematic Review. International Journal of Molecular Sciences, 23(19), 11508. https://doi.org/10.3390/ijms231911508







