Visceral Adipose Tissue Molecular Networks and Regulatory microRNA in Pediatric Obesity: An In Silico Approach
Abstract
:1. Introduction
2. Results
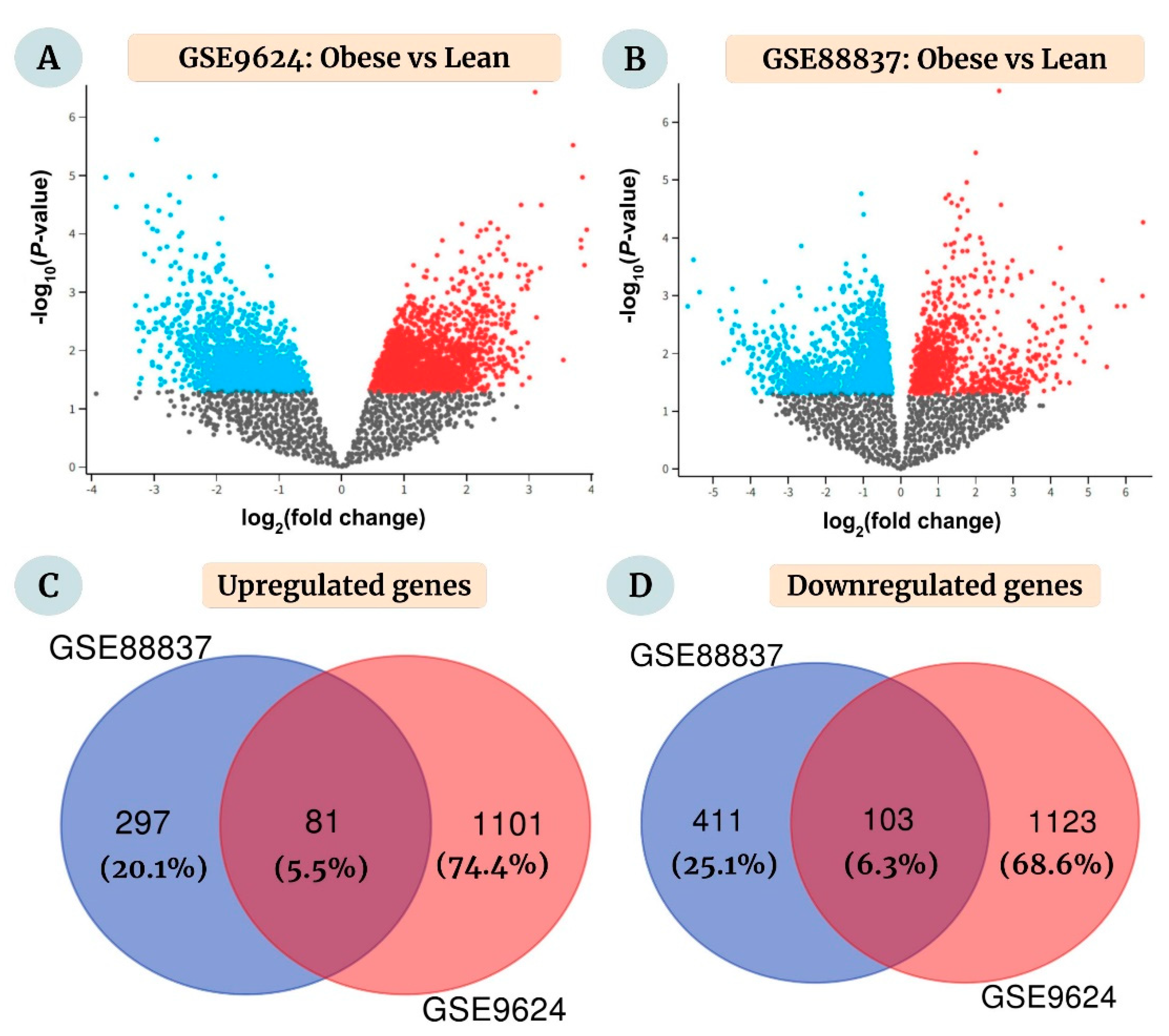
2.1. Identification of DEGs between Obese and Lean Individuals
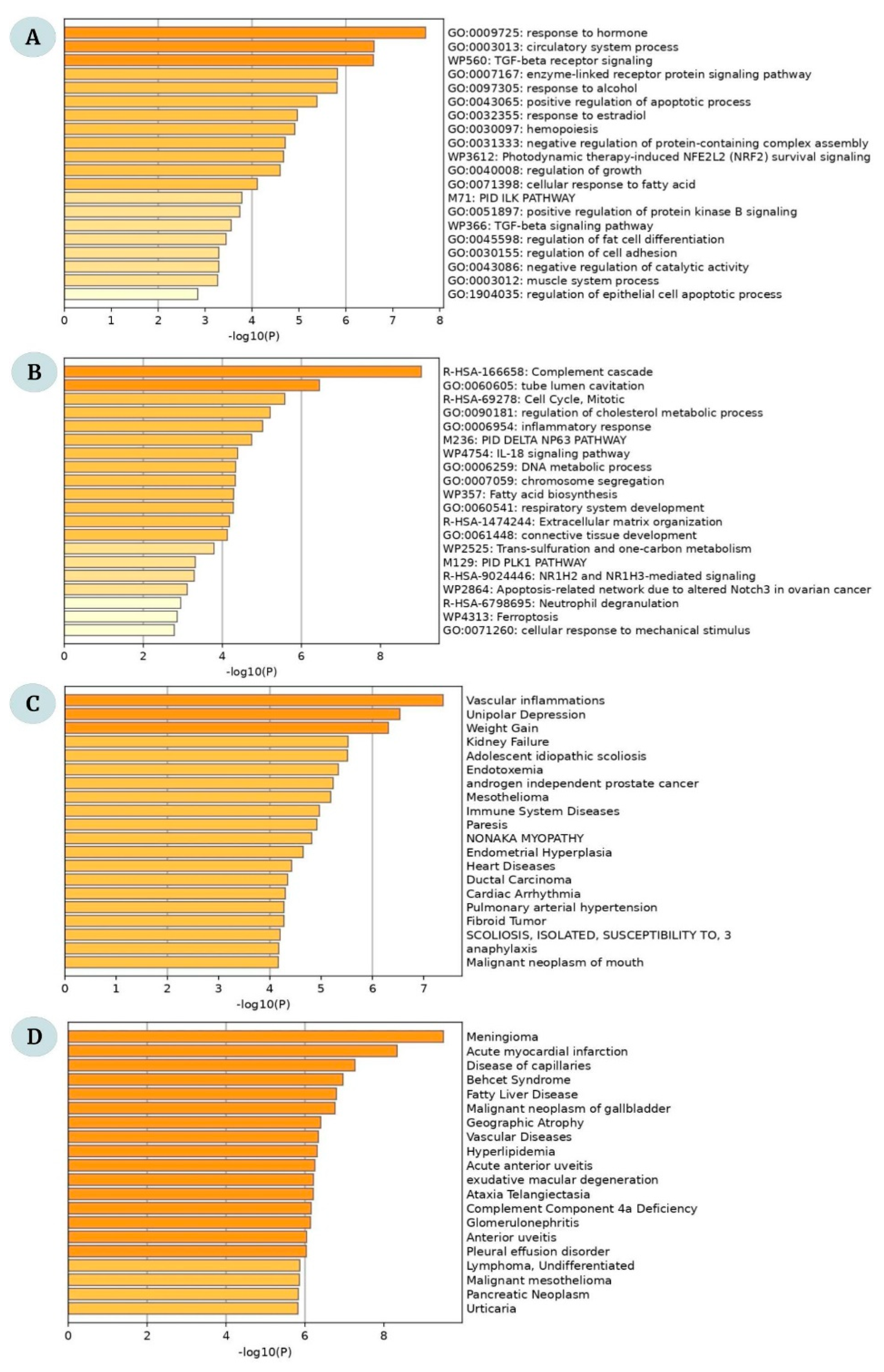
2.2. Functional Enrichment Analysis of DEGs
2.3. PPI Network Construction and Significant Modules
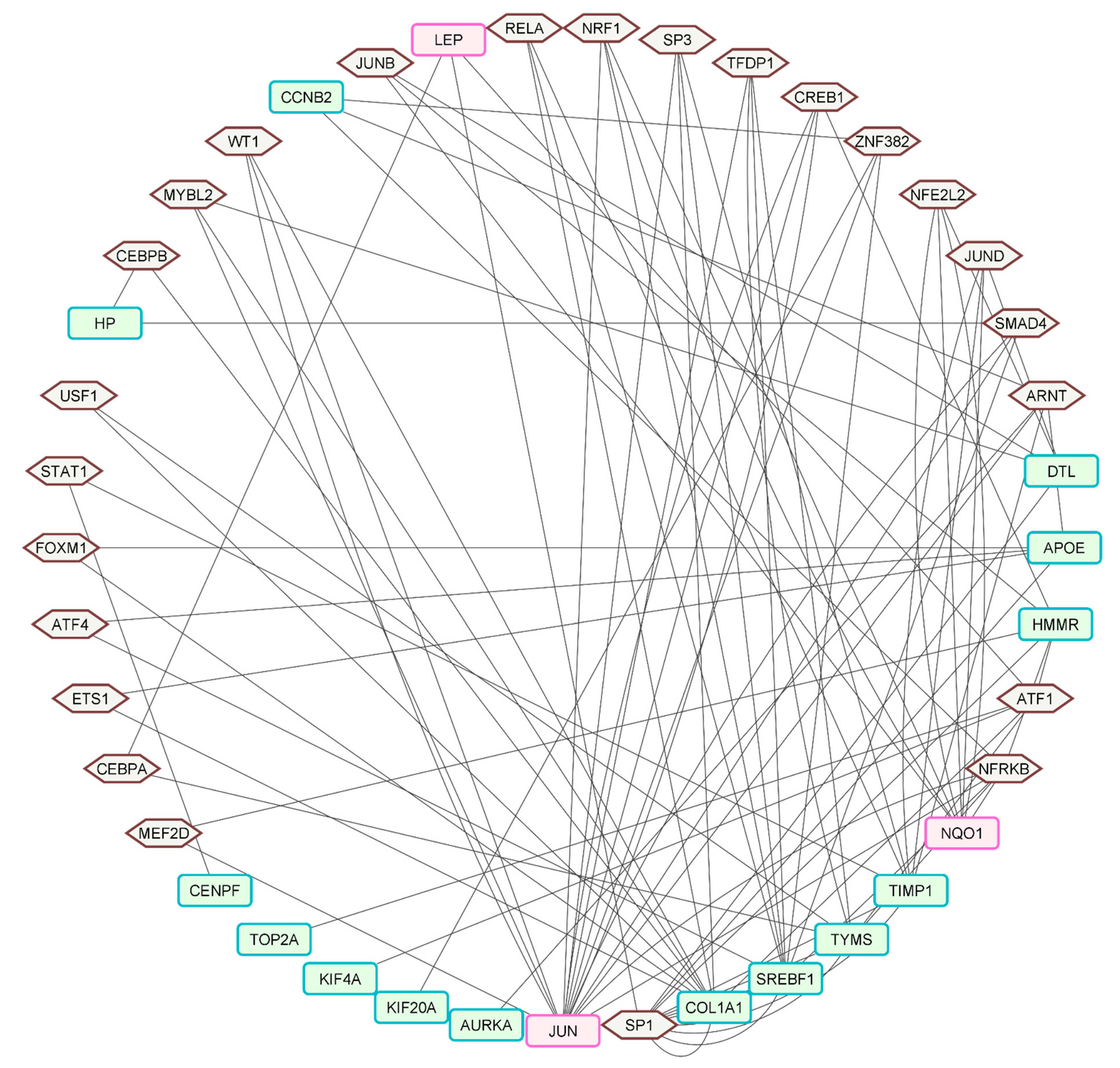
2.4. Hub Gene Identification and TF–Gene Interactions
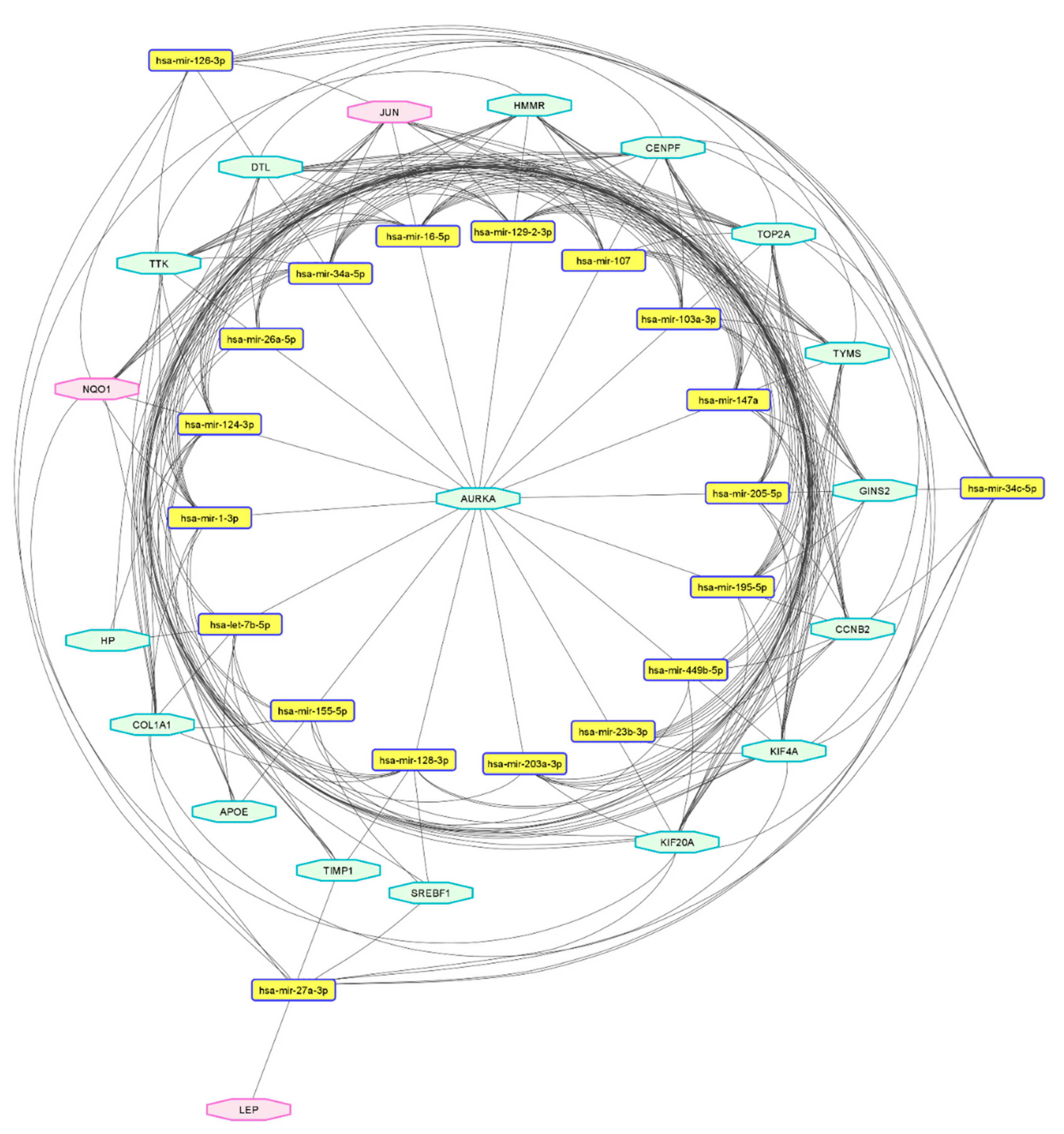
2.5. MicroRNA and Hub Gene Network
3. Discussion
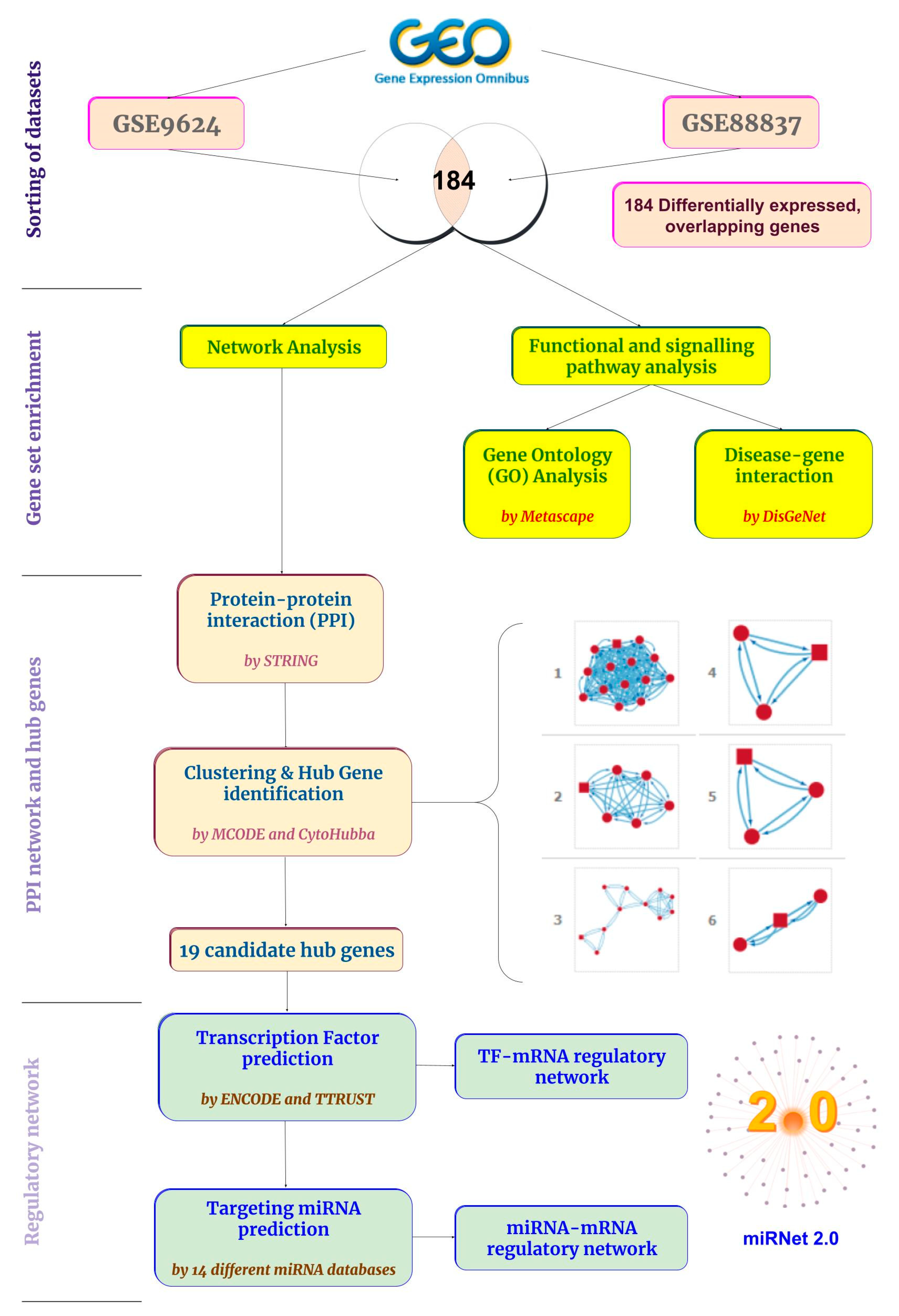
4. Materials and Methods
4.1. Microarray Datasets and Screening of DEGs
4.2. DEG Functional Enrichment and Disease–Gene Interactions
4.3. PPI Network and Hub Gene Identification
4.4. Hub-Gene Targeting TF, miRNA Network, and Functional Enrichment
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ng, M.; Fleming, T.; Robinson, M.; Thomson, B.; Graetz, N.; Margono, C.; Mullany, E.C.; Biryukov, S.; Abbafati, C.; Abera, S.F.; et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2014, 384, 766–781, Erratum in Lancet 2014, 384, 746. [Google Scholar] [CrossRef]
- Monzon, A.D.; Patton, S.R.; Koren, D. Childhood diabetes and sleep. Pediatr Pulmonol. 2022, 57, 1835–1850. [Google Scholar] [CrossRef] [PubMed]
- Weihe, P.; Spielmann, J.; Kielstein, H.; Henning-Klusmann, J.; Weihrauch-Blüher, S. Childhood Obesity and Cancer Risk in Adulthood. Curr. Obes. Rep. 2020, 9, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Maffeis, C.; Morandi, A. Body composition and insulin resistance in children. Eur. J. Clin. Nutr. 2018, 72, 1239–1245. [Google Scholar] [CrossRef] [PubMed]
- Kade, A.; Chabanets, E.; Zanin, S.; Polyakov, P. Sick fat (adiposopathy) as the main contributor to metabolic syndrome. Probl. Nutr. 2022, 91, 27–36. [Google Scholar] [CrossRef]
- Vajravelu, M.E.; Kindler, J.M.; Zemel, B.S.; Jawad, A.; Koren, D.; Brar, P.; Brooks, L.J.; Reiner, J.; Katz, L.E.L. Visceral adiposity is related to insulin sensitivity and inflammation in adolescents with obesity and mild sleep disordered breathing. J. Pediatr. Endocrinol. Metab. 2022, 35, 1069–1077. [Google Scholar] [CrossRef]
- Rydén, M.; Hrydziuszko, O.; Mileti, E.; Raman, A.; Bornholdt, J.; Boyd, M.; Toft, E.; Qvist, V.; Näslund, E.; Thorell, A.; et al. The Adipose Transcriptional Response to Insulin Is Determined by Obesity, Not Insulin Sensitivity. Cell Rep. 2016, 16, 2317–2326. [Google Scholar] [CrossRef]
- Roy, D.; Modi, A.; Khokhar, M.; Sankanagoudar, S.; Yadav, D.; Sharma, S.; Purohit, P.; Sharma, P. MicroRNA 21 Emerging Role in Diabetic Complications: A Critical Update. Curr. Diabetes Rev. 2021, 17, 122–135. [Google Scholar] [CrossRef]
- Khokhar, M.; Roy, D.; Bajpai, N.K.; Bohra, G.K.; Yadav, D.; Sharma, P.; Purohit, P. Metformin mediates MicroRNA-21 regulated circulating matrix metalloproteinase-9 in diabetic nephropathy: An in-silico and clinical study. Arch. Physiol. Biochem. 2021, 1–11, Epub ahead of print. [Google Scholar] [CrossRef]
- Hutny, M.; Hofman, J.; Zachurzok, A.; Matusik, P. MicroRNAs as the promising markers of comorbidities in childhood obesity—A systematic review. Pediatr. Obes. 2021, 17, e12880. [Google Scholar] [CrossRef]
- Strycharz, J.; Wróblewski, A.; Zieleniak, A.; Świderska, E.; Matyjas, T.; Rucińska, M.; Pomorski, L.; Czarny, P.; Szemraj, J.; Drzewoski, J.; et al. Visceral Adipose Tissue of Prediabetic and Diabetic Females Shares a Set of Similarly Upregulated microRNAs Functionally Annotated to Inflammation, Oxidative Stress and Insulin Signaling. Antioxidants 2021, 10, 101. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Luo, Z.; Jin, Z.; Ji, Y.; Ying, W. Adipose Tissue Macrophages Modulate Obesity-Associated β Cell Adaptations through Secreted miRNA-Containing Extracellular Vesicles. Cells 2021, 10, 2451. [Google Scholar] [CrossRef] [PubMed]
- Oses, M.; Medrano, M.; Sanchez, J.M.; Portillo, M.P.; Aguilera, C.M.; Altmäe, S.; Labayen, I. Peripheral blood mononuclear cells-expressed miRNA profiles derived from children with metabolic-associated fatty liver disease and insulin resistance. Pediatr. Obes. 2022, e12966. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, C.M.; Gomez-Llorente, C.; Tofé, I.; Gil-Campos, M.; Cañete, R.; Gil, A. Genome-Wide Expression in Visceral Adipose Tissue from Obese Prepubertal Children. Int. J. Mol. Sci. 2015, 16, 7723–7737. [Google Scholar] [CrossRef]
- Barberio, M.D.; Nadler, E.P.; Sevilla, S.; Lu, R.; Harmon, B.; Hubal, M.J. Comparison of visceral adipose tissue DNA methylation and gene expression profiles in female adolescents with obesity. Diabetol. Metab. Syndr. 2019, 11, 98. [Google Scholar] [CrossRef]
- Osorio-Conles, ȯ.; Vega-Beyhart, A.; Ibarzabal, A.; Balibrea, J.M.; Graupera, I.; Rimola, J.; Vidal, J.; de Hollanda, A. A Distinctive NAFLD Signature in Adipose Tissue from Women with Severe Obesity. Int. J. Mol. Sci. 2021, 22, 10541. [Google Scholar] [CrossRef]
- Siddiqui, J.A.; Pothuraju, R.; Khan, P.; Sharma, G.; Muniyan, S.; Seshacharyulu, P.; Jain, M.; Nasser, M.W.; Batra, S.K. Pathophysiological role of growth differentiation factor 15 (GDF15) in obesity, cancer, and cachexia. Cytokine Growth Factor Rev. 2022, 64, 71–83. [Google Scholar] [CrossRef]
- Medeiros, N.I.; Mattos, R.T.; Menezes, C.A.; Fares, R.C.G.; Talvani, A.; Dutra, W.O.; Rios-Santos, F.; Correa-Oliveira, R.; Gomes, J.A.S. IL-10 and TGF-β unbalanced levels in neutrophils contribute to increase inflammatory cytokine expression in childhood obesity. Eur. J. Nutr. 2017, 57, 2421–2430. [Google Scholar] [CrossRef]
- Koeck, E.S.; Iordanskaia, T.; Sevilla, S.; Ferrante, S.C.; Hubal, M.J.; Freishtat, R.J.; Nadler, E.P. Adipocyte exosomes induce transforming growth factor beta pathway dysregulation in hepatocytes: A novel paradigm for obesity-related liver disease. J. Surg. Res. 2014, 192, 268–275. [Google Scholar] [CrossRef]
- Rao, W.W.; Zong, Q.Q.; Zhang, J.W.; An, F.R.; Jackson, T.; Ungvari, G.S.; Xiang, Y.; Su, Y.Y.; D'Arcy, C.; Xiang, Y.T. Obesity increases the risk of depression in children and adolescents: Results from a systematic review and meta-analysis. J. Affect. Disord. 2020, 267, 78–85. [Google Scholar] [CrossRef]
- Beck, J.J.; Mahan, S.T.; Nowicki, P.; Schreiber, V.M.; Minkowitz, B. What Is New in Pediatric Bone Health. J. Pediatr. Orthop. 2021, 41, e594–e599. [Google Scholar] [CrossRef] [PubMed]
- Sundqvist, A.; Morikawa, M.; Ren, J.; Vasilaki, E.; Kawasaki, N.; Kobayashi, M.; Koinuma, D.; Aburatani, H.; Miyazono, K.; Heldin, C.H.; et al. JUNB governs a feed-forward network of TGFβ signaling that aggravates breast cancer invasion. Nucleic Acids Res. 2017, 46, 1180–1195. [Google Scholar] [CrossRef] [PubMed]
- Costantino, S.; Akhmedov, A.; Melina, G.; Mohammed, S.A.; Othman, A.; Ambrosini, S.; Wijnen, W.J.; Sada, L.; Ciavarella, G.M.; Liberale, L.; et al. Obesity-induced activation of JunD promotes myocardial lipid accumulation and metabolic cardiomyopathy. Eur. Heart J. 2019, 40, 997–1008. [Google Scholar] [CrossRef]
- Modi, A.; Purohit, P.; Roy, D.; Vishnoi, J.R.; Pareek, P.; Elhence, P.; Singh, P.; Sharma, S.; Sharma, P.; Misra, S. FOXM1 mediates GDF-15 dependent stemness and intrinsic drug resistance in breast cancer. Mol. Biol. Rep. 2022, 49, 2877–2888. [Google Scholar] [CrossRef]
- Yamamoto, J.; Imai, J.; Izumi, T.; Takahashi, H.; Kawana, Y.; Takahashi, K.; Kodama, S.; Kaneko, K.; Gao, J.; Uno, K.; et al. Neuronal signals regulate obesity induced β-cell proliferation by FoxM1 dependent mechanism. Nat. Commun. 2017, 8, 1930. [Google Scholar] [CrossRef]
- Zu, L.; Xue, Y.; Wang, J.; Fu, Y.; Wang, X.; Xiao, G.; Hao, M.; Sun, X.; Wang, Y.; Fu, G.; et al. The feedback loop between miR-124 and TGF-β pathway plays a significant role in non-small cell lung cancer metastasis. Carcinogenesis 2016, 37, 333–343. [Google Scholar] [CrossRef]
- Kang, L.; Mokshagundam, S.; Reuter, B.; Lark, D.S.; Sneddon, C.C.; Hennayake, C.; Williams, A.S.; Bracy, D.P.; James, F.D.; Pozzi, A.; et al. Integrin-Linked Kinase in Muscle Is Necessary for the Development of Insulin Resistance in Diet-Induced Obese Mice. Diabetes 2016, 65, 1590–1600. [Google Scholar] [CrossRef] [PubMed]
- Tajbakhsh, A.; Gheibihayat, S.M.; Karami, N.; Savardashtaki, A.; Butler, A.E.; Rizzo, M.; Sahebkar, A. The regulation of efferocytosis signaling pathways and adipose tissue homeostasis in physiological conditions and obesity: Current understanding and treatment options. Obes Rev. 2022, 23, e13487. [Google Scholar] [CrossRef]
- Litwiniuk, A.; Bik, W.; Kalisz, M.; Baranowska-Bik, A. Inflammasome NLRP3 Potentially Links Obesity-Associated Low-Grade Systemic Inflammation and Insulin Resistance with Alzheimer’s Disease. Int. J. Mol. Sci. 2021, 22, 5603. [Google Scholar] [CrossRef]
- Moreno-Navarrete, J.M.; Fernández-Real, J.M. The complement system is dysfunctional in metabolic disease: Evidences in plasma and adipose tissue from obese and insulin resistant subjects. Semin. Cell Dev. Biol. 2019, 85, 164–172. [Google Scholar] [CrossRef]
- Xiang, W.; Ma, Y.L.; Chen, C.; Fu, S.M.; Yang, J.F.; Zhao, S.P.; Guo, D.X.; Zhao, D.C.; Nie, S.; Wang, F.L. Apolipoprotein E gene expression in peripheral blood monocyte in children with obesity. Chin. J. Pediatr. 2003, 41, 755–760. [Google Scholar]
- Kochmanski, J.; Goodrich, J.; Peterson, K.E.; Lumeng, J.C.; Dolinoy, D.C. Neonatal bloodspot DNA methylation patterns are associated with childhood weight status in the Healthy Families Project. Pediatr. Res. 2018, 85, 848–855. [Google Scholar] [CrossRef]
- Akinci, A.; Kara, A.; Özgür, A.; Turkkahraman, D.; Aksu, S. Genomic analysis to screen potential genes and mutations in children with non-syndromic early onset severe obesity: A multicentre study in Turkey. Mol. Biol. Rep. 2021, 49, 1883–1893. [Google Scholar] [CrossRef]
- Castellano-Castillo, D.; Ramos-Molina, B.; Oliva-Olivera, W.; Ocaña-Wilhelmi, L.; Queipo-Ortuño, M.I.; Cardona, F. Genome Profiling of H3k4me3 Histone Modification in Human Adipose Tissue during Obesity and Insulin Resistance. Biomedicines 2021, 9, 1363. [Google Scholar] [CrossRef] [PubMed]
- Meissburger, B.; Stachorski, L.; Röder, E.; Rudofsky, G.; Wolfrum, C. Tissue inhibitor of matrix metalloproteinase 1 (TIMP1) controls adipogenesis in obesity in mice and in humans. Diabetologia 2011, 54, 1468–1479. [Google Scholar] [CrossRef]
- Liu, X.F.; Bera, T.K.; Kahue, C.; Escobar, T.; Fei, Z.; Raciti, G.A.; Pastan, I. ANKRD26 and Its Interacting Partners TRIO, GPS2, HMMR and DIPA Regulate Adipogenesis in 3T3-L1 Cells. PLoS ONE 2012, 7, e38130. [Google Scholar] [CrossRef]
- Rendra, E.; Riabov, V.; Mossel, D.M.; Sevastyanova, T.; Harmsen, M.C.; Kzhyshkowska, J. Reactive oxygen species (ROS) in macrophage activation and function in diabetes. Immunobiology 2018, 224, 242–253. [Google Scholar] [CrossRef] [PubMed]
- Evseeva, M.N.; Balashova, M.S.; Kulebyakin, K.Y.; Rubtsov, Y.P. Adipocyte Biology from the Perspective of In Vivo Research: Review of Key Transcription Factors. Int. J. Mol. Sci. 2021, 23, 322. [Google Scholar] [CrossRef]
- Roy, D.; Modi, A.; Purohit, P. Interactome Profile of Visceral Adipose Tissue in Obesity Links Key Genes to Cancer Pathogenesis. J. Endocr. Soc. 2021, 5, A51–A52. [Google Scholar] [CrossRef]
- Jiang, J.; Guo, Z.; Xu, J.; Sun, T.; Zheng, X. Identification of Aurora Kinase A as a Biomarker for Prognosis in Obesity Patients with Early Breast Cancer. OncoTargets Ther. 2020, 13, 4971–4985. [Google Scholar] [CrossRef]
- Sun, N.; Meng, F.; Zhao, J.; Li, X.; Li, R.; Han, J.; Chen, X.; Cheng, W.; Yang, X.; Kou, Y.; et al. Aurka deficiency in the intestinal epithelium promotes age-induced obesity via propionate-mediated AKT activation. Int. J. Biol. Sci. 2021, 17, 1302–1314. [Google Scholar] [CrossRef] [PubMed]
- Khokhar, M.; Roy, D.; Tomo, S.; Gadwal, A.; Sharma, P.; Purohit, P. Novel Molecular Networks and Regulatory MicroRNAs in Type 2 Diabetes Mellitus: Multiomics Integration and Interactomics Study. JMIR Bioinform. Biotechnol. 2022, 3, e32437. [Google Scholar] [CrossRef]
- Duan, J.; Liu, H.; Chen, J.; Li, X.; Li, P.; Zhang, R. Changes in gene expression of adipose tissue CD14+ cells in patients with Type 2 diabetes mellitus and their relationship with environmental factors. J. Cent. South Univ. (Med. Sci.) 2021, 46, 1–10. [Google Scholar]
- Rahman, H.; Peng, S.; Hu, X.; Chen, C.; Rahman, R.; Uddin, S.; Quinn, J.M.; Moni, M.A. A Network-Based Bioinformatics Approach to Identify Molecular Biomarkers for Type 2 Diabetes that Are Linked to the Progression of Neurological Diseases. Int. J. Environ. Res. Public Health 2020, 17, 1035. [Google Scholar] [CrossRef]
- Sørensen, A.; van Poppel, M.; Desoye, G.; Damm, P.; Simmons, D.; Jensen, D.; Dalgaard, L. The DALI Core Investigator Group The Predictive Value of miR-16, -29a and -134 for Early Identification of Gestational Diabetes: A Nested Analysis of the DALI Cohort. Cells 2021, 10, 170. [Google Scholar] [CrossRef]
- Pan, Y.; Hui, H.; Hoo, R.; Feng, T.; Lam, K.S.; Xu, A. miR-34a Aggravates Obesity-Induced Adipose Inflammation and Metabolic Dysfunction via Blocking Polarization of Anti-inflammatory M2 Macrophage. Diabetes 2018, 67, 2025. [Google Scholar] [CrossRef]
- Trajkovski, M.; Hausser, J.; Soutschek, J.; Bhat, B.; Akin, A.; Zavolan, M.; Heim, M.H.; Stoffel, M. MicroRNAs 103 and 107 regulate insulin sensitivity. Nature 2011, 474, 649–653. [Google Scholar] [CrossRef]
- Zhang, Z.; Wu, S.; Muhammad, S.; Ren, Q.; Sun, C. miR-103/107 promote ER stress-mediated apoptosis via targeting the Wnt3a/β-catenin/ATF6 pathway in preadipocytes. J. Lipid Res. 2018, 59, 843–853. [Google Scholar] [CrossRef]
- Khakoo, N.S.; Ioannou, S.; Khakoo, N.S.; Vedantam, S.; Pearlman, M. Impact of Obesity on Inflammatory Bowel Disease. Curr. Gastroenterol. Rep. 2022, 24, 26–36. [Google Scholar] [CrossRef]
- Rasul, T.; Frederiksen, J.L. Link between overweight/obese in children and youngsters and occurrence of multiple sclerosis. J. Neurol. 2018, 265, 2755–2763. [Google Scholar] [CrossRef]
- Karere, G.M.; Cox, L.A.; Bishop, A.C.; South, A.M.; Shaltout, H.A.; Mercado-Deane, M.G.; Cuda, S. Sex Differences in MicroRNA Expression and Cardiometabolic Risk Factors in Hispanic Adolescents with Obesity. J. Pediatr. 2021, 235, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zhou, B.; Pache, L.; Chang, M.; Khodabakhshi, A.H.; Tanaseichuk, O.; Benner, C.; Chanda, S.K. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat. Commun. 2019, 10, 1523. [Google Scholar] [CrossRef]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef] [PubMed]
- Chin, C.H.; Chen, S.H.; Wu, H.H.; Ho, C.W.; Ko, M.T.; Lin, C.Y. cytoHubba: Identifying hub objects and sub-networks from complex interactome. BMC Syst. Biol. 2014, 8 (Suppl. S4), S11. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.; Zhou, G.; Soufan, O.; Xia, J. miRNet 2.0: Network-based visual analytics for miRNA functional analysis and systems biology. Nucleic Acids Res. 2020, 48, W244–W251. [Google Scholar] [CrossRef] [PubMed]



| Sl No. | Official Gene Symbol | Number of Methods Involved |
|---|---|---|
| 1 | TOP2A | 10 |
| 2 | JUN | 9 |
| 3 | APOE | 9 |
| 4 | TIMP1 | 6 |
| 5 | COL1A1 | 5 |
| 6 | HMMR | 5 |
| 7 | KIF4A | 5 |
| 8 | KIF20A | 5 |
| 9 | TYMS | 4 |
| 10 | LEP | 4 |
| 11 | CENPF | 4 |
| 12 | GINS2 | 4 |
| 13 | SREBF1 | 3 |
| 14 | HP | 3 |
| 15 | NQO1 | 3 |
| 16 | CCNB2 | 3 |
| 17 | TTK | 3 |
| 18 | DTL | 3 |
| 19 | AURKA | 3 |
| Candidate Hub Gene | Targeting Transcription Factors | Targeting miRNA |
|---|---|---|
| TOP2A | ATF1 | miR-34a-5p, miR-16-5p, miR-124-3p, miR-103a-3p, miR-107, miR-129-2-3p, miR-1-3p, miR-147a, miR-126-3p, miR-27a-3p, miR-195-5p, miR-205-5p, let-7b-5p, miR-26a-5p, miR-23b-3p, miR-128-3p, miR-449b-5p, miR-34c-5p, miR-203a-3p |
| JUN | CREB1, MYBL2, NFRKB, NRF1, SMAD4, SP3, TFDP1, WT1, ARNT, ZNF382, MEF2D | miR-34a-5p, miR-16-5p, miR-124-3p, miR-103a-3p, miR-107, miR-129-2-3p, miR-1-3p, miR-147a, miR-126-3p, miR-195-5p, miR-26a-5p, miR-23b-3p, miR-203a-3p, miR-155-5p |
| APOE | ARNT, ETS1, ATF4, FOXM1, SP1 | miR-34a-5p, miR-16-5p, miR-1-3p, let-7b-5p, miR-155-5p |
| TIMP1 | ARNT, JUND, RELA, SP1, SP3, STAT1 | miR-34a-5p, miR-124-3p, miR-27a-3p, let-7b-5p, miR-26a-5p, miR-128-3p |
| COL1A1 | ATF1, CEBPB, FOXM1, SP1, USF1, WT1, ETS1, MYBL2, RELA, SP1, SP3 | miR-34a-5p, miR-16-5p, miR-124-3p, miR-103a-3p, miR-107, miR-129-2-3p, miR-1-3p, miR-27a-3p, let-7b-5p, miR-128-3p, miR-34c-5p, miR-155-5p |
| HMMR | ATF1, CREB1, JUNB, MEF2D, NFRKB, SP1 | miR-34a-5p, miR-16-5p, miR-124-3p, miR-103a-3p, miR-107, miR-129-2-3p, miR-1-3p, miR-147a, miR-27a-3p, miR-195-5p, miR-205-5p, miR-23b-3p, let-7b-5p, miR-203a-3p, miR-155-5p |
| KIF4A | ATF1 | miR-34a-5p, miR-16-5p, miR-124-3p, miR-103a-3p, miR-107, miR-129-2-3p, miR-1-3p, miR-147a, miR-126-3p, miR-27a-3p, miR-195-5p, miR-205-5p, miR-26a-5p, miR-23b-3p, miR-449b-5p, miR-34c-5p, miR-203a-3p |
| KIF20A | ZNF382 | miR-34a-5p, miR-16-5p, miR-124-3p, miR-103a-3p, miR-107, miR-129-2-3p, miR-147a, miR-126-3p, miR-27a-3p, miR-195-5p, miR-205-5p, miR-23b-3p, miR-449b-5p, miR-34c-5p, miR-203a-3p, miR-155-5p |
| TYMS | ATF1, CEBPA, NFRKB, SP1, TFDP1, USF1 | miR-34a-5p, miR-16-5p, miR-103a-3p, miR-107, miR-129-2-3p, miR-1-3p, miR-147a, miR-126-3p, miR-195-5p, let-7b-5p, miR-26a-5p, miR-23b-3p, miR-449b-5p, miR-203a-3p, miR-155-5p |
| LEP | ATF1, CEBPA, SP1 | miR-27a-3p |
| CENPF | STAT1 | miR-34a-5p, miR-16-5p, miR-124-3p, miR-103a-3p, miR-107, miR-129-2-3p, miR-1-3p, miR-147a, miR-126-3p, miR-27a-3p, miR-195-5p, miR-205-5p, miR-26a-5p, miR-23b-3p, miR-128-3p, miR-449b-5p, miR-34c-5p |
| GINS2 | - | miR-34a-5p, miR-16-5p, miR-124-3p, miR-103a-3p, miR-107, miR-129-2-3p, miR-1-3p, miR-147a, miR-195-5p, miR-205-5p, miR-34c-5p, miR-203a-3p |
| SREBF1 | ATF4, NFRKB, NRF1, RELA, SMAD4, SP3, TFDP1, ZNF382, SP1 | miR-16-5p, miR-27a-3p, miR-128-3p, miR-155-5p |
| HP | CEBPB, SMAD4 | miR-124-3p, miR-147a, let-7b-5p |
| NQO1 | NFRKB, NRF1, JUNB, JUND, NFE2L2 | miR-34a-5p, miR-124-3p, miR-103a-3p, miR-107, miR-129-2-3p, miR-1-3p, miR-147a, miR-126-3p, miR-27a-3p, miR-205-5p, miR-128-3p |
| CCNB2 | ZNF382, ARNT, NFRKB | miR-34a-5p, miR-16-5p, miR-124-3p, miR-103a-3p, miR-107, miR-129-2-3p, miR-147a, miR-195-5p, miR-205-5p, miR-126-3p, miR-23b-3p, let-7b-5p, miR-449b-5p, miR-34c-5p |
| TTK | - | miR-34a-5p, miR-16-5p, miR-124-3p, miR-103a-3p, miR-107, miR-129-2-3p, miR-1-3p, miR-147a, miR-126-3p, miR-195-5p, miR-205-5p, miR-26a-5p, miR-128-3p, miR-449b-5p, miR-34c-5p |
| DTL | JUNB, JUND, MYBL2, NFE2L2, SP1 | miR-34a-5p, miR-16-5p, miR-124-3p, miR-103a-3p, miR-107, miR-129-2-3p, miR-1-3p, miR-147a, miR-126-3p, miR-195-5p, miR-205-5p, miR-26a-5p, miR-128-3p, miR-449b-5p, miR-34c-5p |
| AURKA | ARNT, NFRKB, ZNF382 | miR-34a-5p, miR-16-5p, miR-124-3p, miR-103a-3p, miR-107, miR-129-2-3p, miR-1-3p, miR-147a, miR-195-5p, miR-205-5p, miR-26a-5p, miR-23b-3p, let-7b-5p, miR-128-3p, miR-449b-5p, miR-203a-3p, miR-155-5p |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roy, D.; Modi, A.; Ghosh, R.; Ghosh, R.; Benito-León, J. Visceral Adipose Tissue Molecular Networks and Regulatory microRNA in Pediatric Obesity: An In Silico Approach. Int. J. Mol. Sci. 2022, 23, 11036. https://doi.org/10.3390/ijms231911036
Roy D, Modi A, Ghosh R, Ghosh R, Benito-León J. Visceral Adipose Tissue Molecular Networks and Regulatory microRNA in Pediatric Obesity: An In Silico Approach. International Journal of Molecular Sciences. 2022; 23(19):11036. https://doi.org/10.3390/ijms231911036
Chicago/Turabian StyleRoy, Dipayan, Anupama Modi, Ritwik Ghosh, Raghumoy Ghosh, and Julián Benito-León. 2022. "Visceral Adipose Tissue Molecular Networks and Regulatory microRNA in Pediatric Obesity: An In Silico Approach" International Journal of Molecular Sciences 23, no. 19: 11036. https://doi.org/10.3390/ijms231911036
APA StyleRoy, D., Modi, A., Ghosh, R., Ghosh, R., & Benito-León, J. (2022). Visceral Adipose Tissue Molecular Networks and Regulatory microRNA in Pediatric Obesity: An In Silico Approach. International Journal of Molecular Sciences, 23(19), 11036. https://doi.org/10.3390/ijms231911036






