Prospects and Challenges of Electrospun Cell and Drug Delivery Vehicles to Correct Urethral Stricture
Abstract
1. Introduction
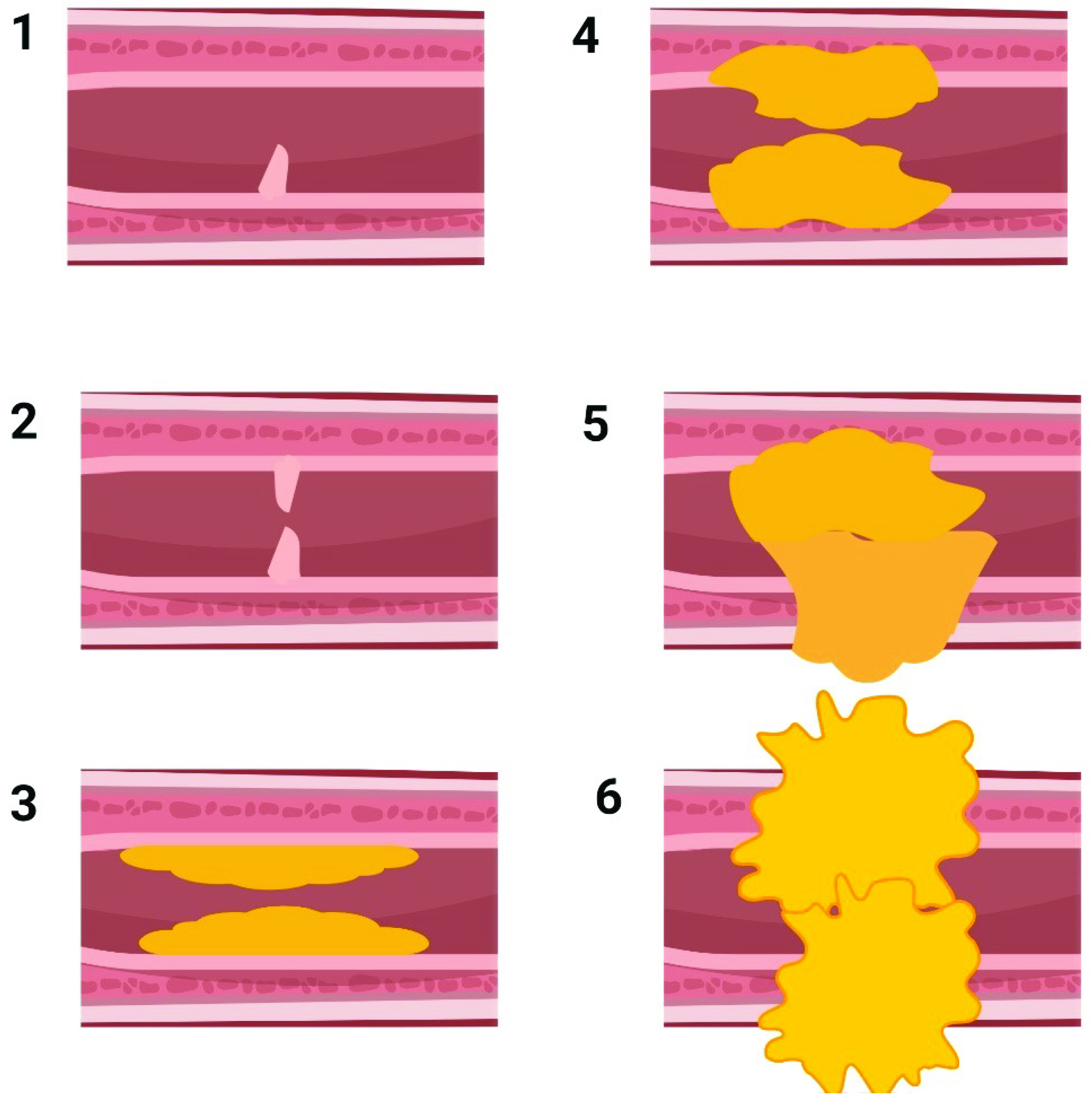
2. Pathophysiology of US, Its Etiology, and Current Treatment Options
3. Pros and Cons of Potential Cell Sources to Repair Urethra Defects
4. Bioactive Agents That Can Prevent US
5. General Characteristics of Cell and Drug Delivery Systems to Treat Urethra Defects
| Polymer/s. | Delivery System | Cell/Drug Type | Fabrication Method | Cell Type/s for In Vitro Study | In Vivo Model | Experimental Results | References |
|---|---|---|---|---|---|---|---|
| Polycaprolactone and fibrin | Hydrogel | Urothelial cells and smooth muscle cells | 3D printing | Urothelial cells and smooth muscle cells | - | The produced delivery system had comparable mechanical strength with rabbit urethra and supported cell viability up to 7 days after printing | [138] |
| Propylene glycol | Hydrogel | Mitomycin C | Cross-linking | - | Clinical trial | Mitomycin C-loaded hydrogel could significantly reduce the recurrence of US after internal urethrotomy | [139] |
| Gelatin methacrylate and pure collagen | Bioprinted scaffold | Bladder smooth muscle cells | Bioprinting | Bladder smooth muscle cells | - | Cells stayed viable in the printed scaffolds, and cell density increased over time | [140] |
| Poly l lactic acid, poly D,L-lactic-co-glycolic acid, and poly(L-lactic acid-co-ε-caprolactone) | Porous sponge | Adipose-derived stem cells | Lyophilization | Adipose-derived stem cells | New Zealand white rabbit model of urethra defect | Hypoxia-preconditioned stem cells delivered via the porous scaffolds could successfully repair urethral defects and induced a robust angiogenesis | [141] |
| Poly [N-isopropyl acry-lamide-co-n-butyl methacrylate] poly [NIPAAm-co-BMA]) and hydrophilic blocks (polyethylene glycol) | Hydrogel | Buccal epithelial cells | Sol-gel transition was obtained by changing the temperature. | Buccal epithelial cells | Clinical trial | The treated patients void well with a normal mean peak flow rate. Two of the six patients showed recurrent stricture at 18 and 24 months after treatment | [142] |
| TGP | Hydrogel | Buccal mucosal epithelial cells | Thermo-reversible gelation | Buccal mucosal epithelial cells | Japanese white male rabbit model of urethra defect | Cells stayed viable in the hydrogel system and differentiated into fibroblast-like cells. Cell-loaded hydrogel system repaired urethra defects and cells engrafted at the injury site | [143] |
| No materials were used | tubular scaffold | Human mesenchymal stem cells | Self-assembly | Human mesenchymal stem cells | Nude rat model | scaffolds showed two layers of cells and no stricture after implantation into the nude rat | [144] |
| Natural ECM | Decellularized bladder matrices obtained from lamina propria | Autologous bladder epithelial and smooth muscle cells | Decellularization | Autologous bladder epithelial and smooth muscle cells | Rabbit model of anterior penile urethra defect | Cell-seeded tubular matrices showed a wide urethral caliber with no strictures. In addition, a transitional cell layer was formed in the cell-seeded matrix group, and the newly developed urethra showed contractility | [145] |
| Silk fibroin and a nanoporous bacterial cellulose | Porous Bilayer scaffold | Lingual Keratinocytes and muscle cells | Freeze-drying and self-assembling | Lingual keratinocytes and muscle cells | Canine model of urethra defect | Microstructure studies showed that the seeded cells could adhere to the scaffolds. Cell-seeded urethral grafts showed superior healing function over cell-free ones | [146] |
| Bacterial cellulose | 3D porous scaffold | Lingual keratinocytes | Biosynthesis via bacterial species | Lingual keratinocytes | New Zealand White male rabbit model of urethra defect | In scaffolds seeded with lingual keratinocytes, the caliber of the urethras was wide open and a continuous epithelium was formed | [147] |
| Natural ECM | Decellularized human amniotic scaffolds | Allogeneic bone marrow mesenchymal cells and/or endothelial progenitor cells | Decellularization | Bone marrow mesenchymal cells and/or endothelial progenitor cells | Canine model of circumferential urethral defect | Animals treated with cell-seeded scaffolds showed unhindered urination and wide open urethra caliber. Furthermore, extensive vascularization was observed in this group | [60] |
| Natural ECM | 3-D porous small intestinal submucosa | Urothelial and smooth muscle cells that were produced from the differentiation of urine-derived stem cells. | Decellularization | Urine-derived stem cells differentiated into urothelial cells and smooth muscle cells. | Cell-seeded scaffolds were implanted into Athymic mice | The seeded cells developed uniform layers on the scaffold and penetrated deep into the inner parts | [148] |
| Poly-D,L-lactide-co-εcaprolactone | Bilayer polymeric matrix | Allogenic mesenchymal stem cells | Casting and air drying | Mesenchymal stem cells | Chinchilla rabbit model of urethra defect | Cell-seeded scaffolds showed integration with the urethra tissue with no adverse tissue reactions. Delivered cells expressed cytokeratin marker AE1/AE3, implying their potential differentiation into neo-urothelium | [149] |
| Natural ECM | Acellular matrix | Endothelial progenitor cells that secrete antibiotic peptide LL37 | Decellularization | Endothelial progenitor cells | New Zealand white Male rabbit model of urethra defect | Antipoetic-delivering cells seeded on the acellular matrix could successfully repair critical-sized urethra defects | [150] |
| Gelatin, poly l lactic acid, and silk fibroin | Porous tubular scaffolds | Mitomycin C and epidermal growth factor | Freeze drying | Urethral epithelial cells and urethral scar-derived fibroblast cells | - | The proportion of Urethral epithelial cells was significantly increased when cultured on the drug-loaded system | [151] |
| Poly-l-lactic acid and poly-dl-lactic acid | Tubular scaffold | Paclitaxel | Casting | - | Male rabbit model of urethra defect | The drug-eluting stent could successfully prevent US, reduced inflammation, and alleviated fibrotic reactions | [152] |
| Poly-L189 lactide-co-caprolactone (PLC) and Polyethylene glycol diacrylate (PEGDA) | Polyurethane double pig-tailed ureteric stent spray-coated with Mitomycin C-loaded PLC and overlaid with PEGDA hydrogel. | Mitomycin C | Spray coating and cross-linking | HBdSF cells | Porcine model | The developed system released the loaded drug sustainably and could deliver the drug to urothelium with no adverse effects | [153] |
| Collagen | A synthetic catheter coated with collagen | Insulin-like growth factor 1 (IGF-1) | Coating on a synthetic catheter | HUEpCs cell line | Japanese white rabbit model of urethra defect | Animals treated with IGF-1/collagen-impregnated catheters had significantly bigger urethra caliber than other groups | [154] |
6. Principles of Electrospinning
7. Principles of Cell Delivery via Electrospun Scaffolds
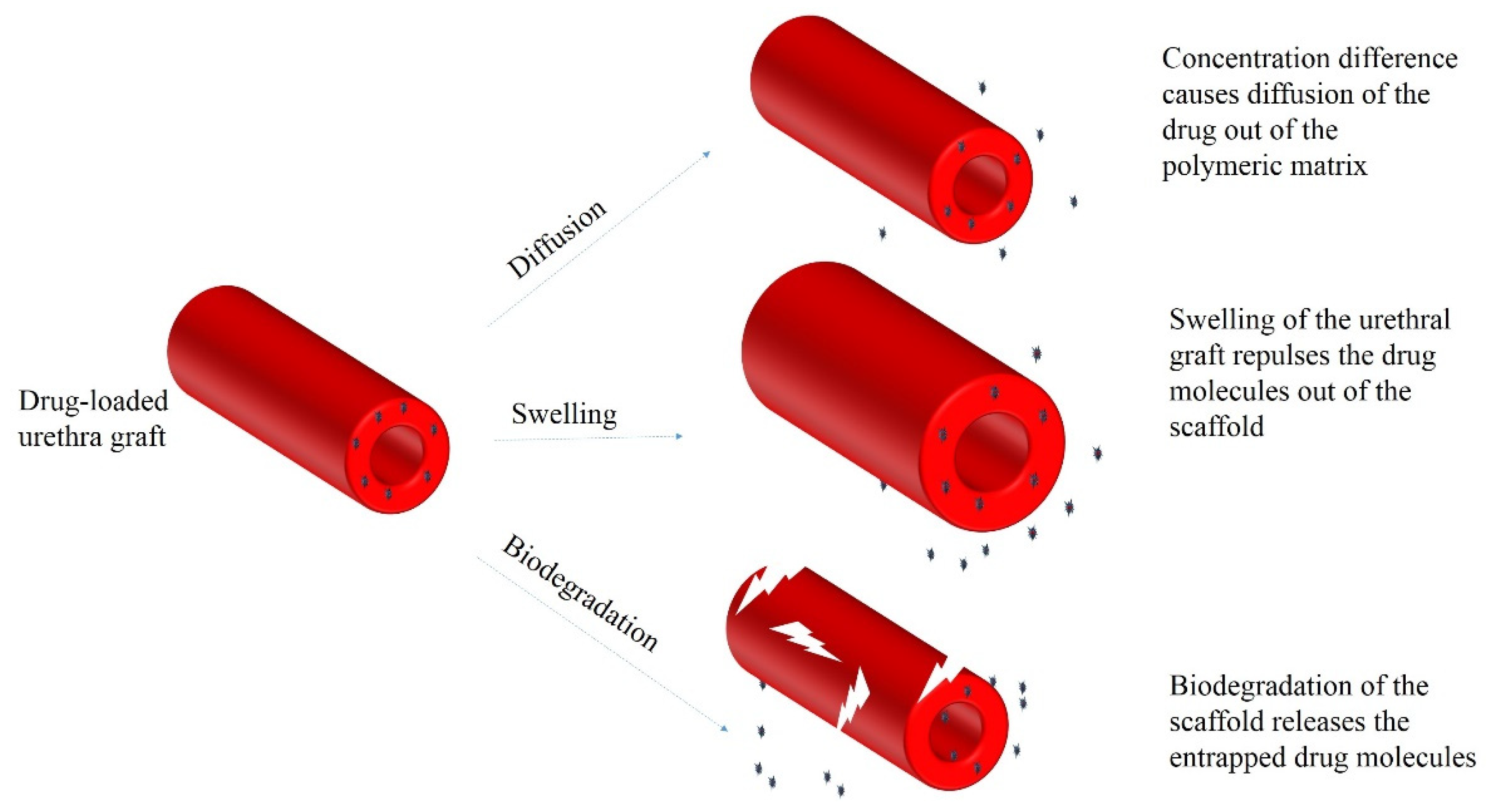
8. Principles of Drug Delivery with Electrospun Scaffolds
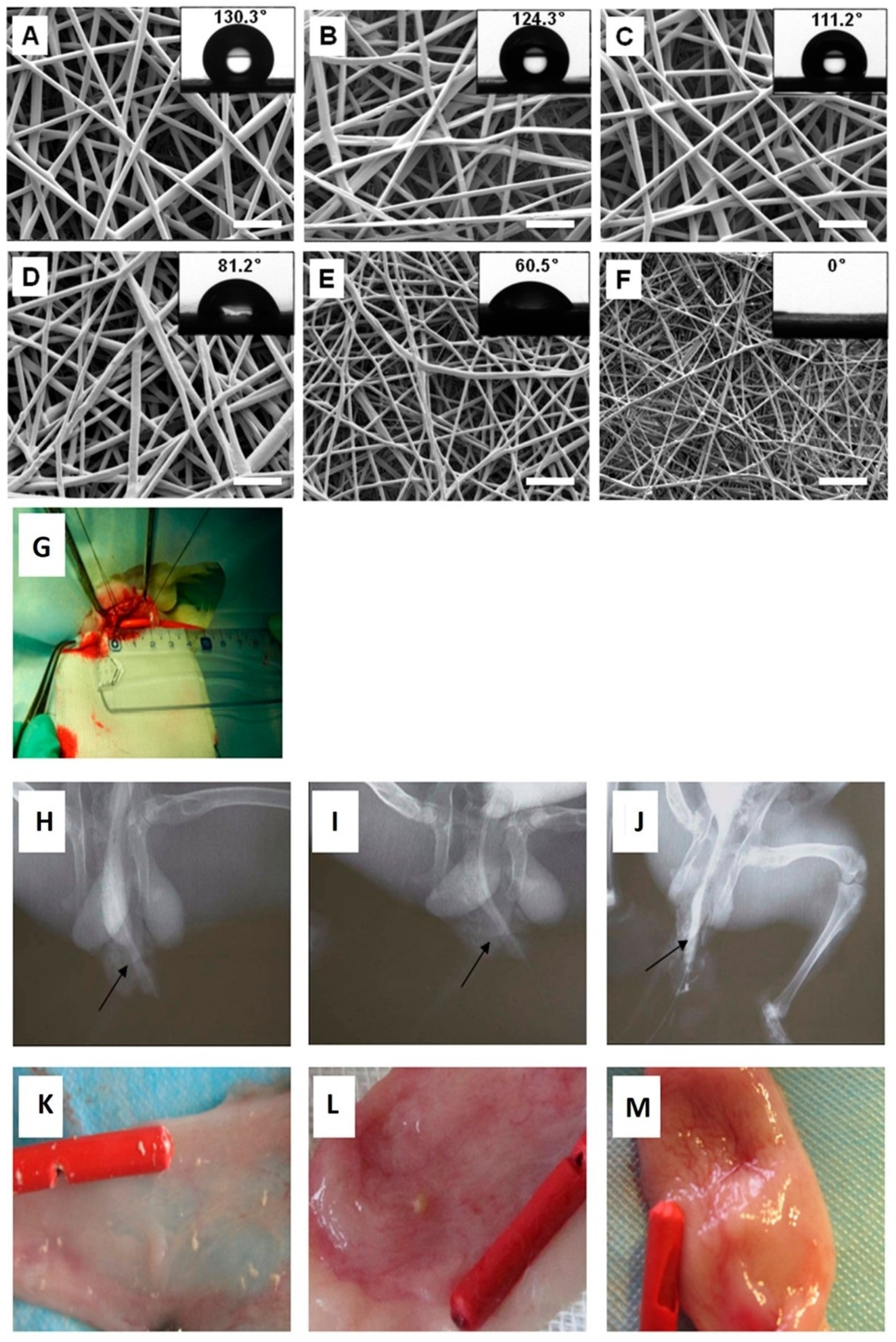
9. Previous Applications of Electrospun Cell Delivery Systems to Treat Urethra Defects
10. Previous Applications of Drug-Loaded Electrospun Delivery Systems to Treat Urethral Defects
11. Challenges and Potential Mitigation Strategies
12. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Simsek, A.; Aldamanhori, R.; Chapple, C.R.; MacNeil, S. Overcoming Scarring in the Urethra: Challenges for Tissue Engineering. Asian J. Urol. 2018, 5, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Lazzeri, M.; Sansalone, S.; Guazzoni, G.; Barbagli, G. Incidence, Causes, and Complications of Urethral Stricture Disease. Eur. Urol. Suppl. 2016, 15, 2–6. [Google Scholar] [CrossRef]
- Cheng, L.; Li, S.; Wang, Z.; Huang, B.; Lin, J. A Brief Review on Anterior Urethral Strictures. Asian J. Urol. 2017, 5, 88–93. [Google Scholar] [CrossRef] [PubMed]
- King, C.; Rourke, K.F. Urethral Stricture is Frequently a Morbid Condition: Incidence and Factors Associated with Complications Related to Urethral Stricture. Urology 2019, 132, 189–194. [Google Scholar] [CrossRef]
- Gild, P.; Kluth, L.A.; Vetterlein, M.W.; Engel, O.; Chun, F.K.; Fisch, M. Adult Iatrogenic Ureteral Injury and Stricture-Incidence and Treatment Strategies. Asian J. Urol. 2018, 5, 101–106. [Google Scholar] [CrossRef]
- Hampson, L.A.; McAninch, J.W.; Breyer, B.N. Male Urethral Strictures and Their Management. Nat. Rev. Urol. 2013, 11, 43–50. [Google Scholar] [CrossRef]
- Gallegos, M.A.; Santucci, R.A. Advances in Urethral Stricture Management. F1000Research 2016, 5, 2913. [Google Scholar] [CrossRef]
- Zamani, M.; Shakhssalim, N.; Ramakrishna, S.; Naji, M. Electrospinning: Application and Prospects for Urologic Tissue Engineering. Front. Bioeng. Biotechnol. 2020, 8, 579925. [Google Scholar] [CrossRef]
- Wisenbaugh, E.S.; Gelman, J. The Use of Flaps and Grafts in the Treatment of Urethral Stricture Disease. Adv. Urol. 2015, 2015. [Google Scholar] [CrossRef]
- Mangir, N.; Wilson, K.J.; Osman, N.I.; Chapple, C.R. Current State of Urethral Tissue Engineering. Curr. Opin. Urol. 2019, 29, 385–393. [Google Scholar] [CrossRef]
- Rashidbenam, Z.; Jasman, M.H.; Hafez, P.; Tan, G.H.; Goh, E.H.; Fam, X.I.; Ho, C.C.K.; Zainuddun, Z.M.; Rajan, R.; Nor, F.M.; et al. Overview of Urethral Reconstruction by Tissue Engineering: Current Strategies, Clinical Status and Future Direction. Tissue Eng. Regen. Med. 2019, 16, 365–384. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Meng, Q.; Jin, X.; Liu, F.; Yu, J. Biodegradable Scaffolds for Urethra Tissue Engineering Based on 3D Printing. ACS Appl. Bio. Mater. 2020, 3, 2007–2016. [Google Scholar] [CrossRef]
- Chan, Y.Y.; Bury, M.I.; Yura, E.M.; Hofer, M.D.; Cheng, E.Y.; Sharma, A.K. The Current State of Tissue Engineering in the Management of Hypospadias. Nat. Rev. Urol. 2020, 17, 162–175. [Google Scholar] [CrossRef] [PubMed]
- Adamowicz, J.; Kuffel, B.; Van Breda, S.V.; Pokrwczynska, M.; Drewa, T.; Pokrywczynska, M. Reconstructive Urology and Tissue Engineering: Converging Developmental Paths. J. Tissue Eng. Regen. Med. 2019, 13, 522–533. [Google Scholar] [CrossRef] [PubMed]
- Rahmati, M.; Mills, D.K.; Urbanska, A.M.; Saeb, M.R.; Venugopal, J.R.; Ramakrishna, S.; Mozafari, M. Electrospinning for Tissue Engineering Applications. Prog. Mater. Sci. 2020, 117, 100721. [Google Scholar] [CrossRef]
- Tan, G.Z.; Zhou, Y. Electrospinning of Biomimetic Fibrous Scaffolds for Tissue Engineering: A Review. Int. J. Polym. Mater. Polym. Biomater. 2020, 69, 947–960. [Google Scholar] [CrossRef]
- Ye, K.; Kuang, H.; You, Z.; Morsi, Y.; Mo, X. Electrospun Nanofibers for Tissue Engineering with Drug Loading and Release. Pharmaceutics 2019, 11, 182. [Google Scholar] [CrossRef]
- Luraghi, A.; Peri, F.; Moroni, L. Electrospinning for drug delivery applications: A review. J. Control. Release 2021, 334, 463–484. [Google Scholar] [CrossRef]
- Stoddard, N.; Leslie, S.W. Histology, Male Urethra; StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Tretiakova, M. Urethra Pathology, Practical Genitourinary Pathology; Springer: Berlin/Heidelberg, Germany, 2021; pp. 117–132. [Google Scholar]
- Grimes, M.D.; Tesdahl, B.A.; Schubbe, M.; Dahmoush, L.; Pearlman, A.M.; Kreder, K.J.; Erickson, B.A. Histopathology of Anterior Urethral Strictures: Toward a Better Understanding of Stricture Pathophysiology. J. Urol. 2019, 202, 748–756. [Google Scholar] [CrossRef]
- Chua, K.J.; Mikhail, M.; Patel, H.V.; Tabakin, A.L.; Doppalapudi, S.K.; Sterling, J.; Tunuguntla, H.S. Treatment of Urethral Stricture Disease in Women: Nonsystematic Review of Surgical Techniques and Intraoperative Considerations. Res. Rep. Urol. 2021, 13, 381–406. [Google Scholar] [CrossRef]
- Cavalcanti, A.; Schul, A.; Restreppo, C.F.; Aldred, J.E. Pathophysiology of Urethral Stricture: Optimizing Urethroplasty Outcomes. In Scientific Advances in Reconstructive Urology and Tissue Engineering; Elsevier: Amsterdam, The Netherlands, 2022; pp. 177–195. [Google Scholar] [CrossRef]
- Zelefsky, M.; Wallner, K.; Ling, C. Urethral Stricture Disease: Etiology, Pathophysiology and Its Economic Impact 17. Atlas Urethroplasty 2014, 17, 16. [Google Scholar]
- Verla, W.; Oosterlinck, W.; Spinoit, A.F.; Waterloos, M. A Comprehensive Review Emphasizing Anatomy, Etiology, Diagnosis, And Treatment of Male Urethral Stricture Disease. Bio. Med. Res. Int. 2019, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Kinnaird, A.S.; Levine, M.A.; Ambati, D.; Zorn, J.D.; Rourke, K.F. Stricture Length and Etiology as Preoperative Independent Predictors of Recurrence After Urethroplasty: A Multivariate Analysis Of 604 Urethroplasties. Can. Urol. Assoc. J. 2014, 8, E296. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cotter, K.J.; Hahn, A.E.; Voelzke, B.B.; Myers, J.B.; Smith, T.G., 3rd; Elliott, S.P.; Alsikafi, N.F.; Breyer, B.N.; Vanni, A.J.; Buckley, J.C.; et al. Trends in Urethral Stricture Disease Etiology and Urethroplasty Technique from A Multi-Institutional Surgical Outcomes Research Group. Urology 2019, 130, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Alwaal, A.; Blaschko, S.D.; McAninch, J.W.; Breyer, B.N. Epidemiology of Urethral Strictures. Transl. Androl. Urol. 2014, 3, 209–213. [Google Scholar] [CrossRef]
- Jacobs, M.E.; de Kemp, V.F.; Albersen, M.; de Kort, L.M.; de Graaf, P. The Use of Local Therapy in Preventing Urethral Strictures: A Systematic Review. PLoS ONE 2021, 16, e0258256. [Google Scholar] [CrossRef]
- Belsante, M.J.; Selph, J.P.; Peterson, A.C. The Contemporary Management of Urethral Strictures in Men Resulting from Lichen Sclerosus. Transl. Androl. Urol. 2015, 4, 22–28. [Google Scholar] [CrossRef]
- Smith, T.G., III. Current Management of Urethral Stricture Disease. Indian J. Urol. IJU J. Urol. Soc. India 2016, 32, 27. [Google Scholar] [CrossRef]
- Hu, Z.; Ma, X.; Li, H.-Z.; Wang, C.; Ye, D.-W.; Gong, D.-J.; Zhang, X. A Radially Expanding Sheath for Urethral Dilation. Med. Hypotheses 2009, 73, 585–586. [Google Scholar] [CrossRef]
- Agochukwu-Mmonu, N.; Srirangapatanam, S.; Cohen, A.; Breyer, B. Female Urethral Strictures: Review of Diagnosis, Etiology, And Management. Curr. Urol. Rep. 2019, 20, 1–6. [Google Scholar] [CrossRef]
- Dubey, D. The Current Role of Direct Vision Internal Urethrotomy and Self-Catheterization for Anterior Urethral Strictures. Indian J. Urol. 2011, 27, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Zhu, Z.; Lin, L.; Lv, T.; Cai, T.; Lin, J. Efficacy of Mitomycin C Combined with Direct Vision Internal Urethrotomy for Urethral Strictures: A Systematic Review and Meta-Analysis. Urol. Int. 2021, 25, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Yenice, M.G.; Seker, K.G.; Sam, E.; Colakoglu, Y.; Atar, F.A.; Sahin, S.; Simsek, A.; Tugcu, V. Comparison of Cold-Knife Optical Internal Urethrotomy and Holmium: Yag Laser Internal Urethrotomy in Bulbar Urethral Strictures. Central Eur. J. Urol. 2018, 71, 114–120. [Google Scholar] [CrossRef]
- Kluth, L.A.; Ernst, L.; Vetterlein, M.W.; Meyer, C.P.; Reiss, C.P.; Fisch, M.; Rosenbaum, C.M. Direct Vision Internal Urethrotomy for Short Anterior Urethral Strictures and Beyond: Success Rates, Predictors of Treatment Failure, and Recurrence Management. Urology 2017, 106, 210–215. [Google Scholar] [CrossRef]
- Aydemir, H.; Sağlam, H.S.; Köse, O.; Erdik, A.; Halis, F.; Gökçe, A. The Effect of Recurrent Direct Vision Internal Urethrotomy for Short Anterior Urethral Strictures on the Disease Course and the Predictors of Treatment Failure. Can. Urol. Assoc. J. 2018, 13, E366–E370. [Google Scholar] [CrossRef]
- Iqbal, M.; Khawaja, U.A. Management of Recurrent Urethral Strictures: A Therapeutic Challenge. Cureus 2020, 12, 223–232. [Google Scholar] [CrossRef]
- Lee, L.C.; Schröder, A.; Bägli, D.J.; Lorenzo, A.J.; Farhat, W.A.; Koyle, M.A. Stent-Related Complications After Hypospadias Repair: A Prospective Trial Comparing Silastic Tubing and Koyle Urethral Stents. J. Pediatr. Urol. 2014, 14, 423.e1–423.e5. [Google Scholar] [CrossRef]
- Bryk, D.J.; Yamaguchi, Y.; Zhao, L.C. Tissue Transfer Techniques in Reconstructive Urology. Korean J. Urol. 2015, 56, 478–486. [Google Scholar] [CrossRef]
- Suh, J.-G.; Choi, W.S.; Paick, J.-S.; Kim, S.W. Surgical Outcome of Excision and End-to-End Anastomosis for Bulbar Urethral Stricture. Korean J. Urol. 2013, 54, 442–447. [Google Scholar] [CrossRef][Green Version]
- Barratt, R.; Chan, G.; La Rocca, R.; Dimitropoulos, K.; Martins, F.E.; Campos-Juanatey, F.; Geenwell, T.J.; Waterloos, M.; Riechardt, S.; Osman, N.I.; et al. Free Graft Augmentation Urethroplasty for Bulbar Urethral Strictures: Which Technique Is Best? A Systematic Review. Eur. Urol. 2021, 80, 57–68. [Google Scholar] [CrossRef]
- Mangera, A.; Patterson, J.M.; Chapple, C.R. A Systematic Review of Graft Augmentation Urethroplasty Techniques for the Treatment of Anterior Urethral Strictures. Eur. Urol. 2011, 59, 797–814. [Google Scholar] [CrossRef] [PubMed]
- Culenova, M.; Ziaran, S.; Danisovic, L. Cells Involved in Urethral Tissue Engineering: Systematic Review. Cell Transplant. 2019, 28, 1106–1115. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Gupta, D. Tissue Engineering and Stem Cell Therapy in Pediatric Urology. J. Indian Assoc. Pediatr. Surg. 2019, 24, 237–246. [Google Scholar] [CrossRef]
- Caneparo, C.; Chabaud, S.; Bolduc, S. Challenges and Perspectives in Male Anterior Urethra Reconstruction Using Tissue Engineering. Res. Ther. J. 2019, 2, 127. [Google Scholar]
- Han, Y.; Li, X.; Zhang, Y.; Han, Y.; Chang, F.; Ding, J. Mesenchymal Stem Cells for Regenerative Medicine. Cells 2019, 8, 886. [Google Scholar] [CrossRef]
- Shammaa, R.; El-Kadiry, A.E.-H.; Abusarah, J.; Rafei, M. Mesenchymal Stem Cells Beyond Regenerative Medicine. Front. Cell Dev. Biol. 2020, 8, 72. [Google Scholar] [CrossRef]
- Liang, Y.C.; Wu, Y.P.; Li, X.D.; Chen, S.H.; Ye, X.J.; Xue, X.Y.; Xu, N. TNF-α-induced exosomal miR-146a mediates mesenchymal stem cell-dependent suppression of urethral stricture. J. Cell. Physiol. 2019, 234, 23243–23255. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.; Wang, Q.; Jiang, D. The Preventative Effect of Bone Marrow-Derived Mesenchymal Stem Cell Exosomes on Urethral Stricture in Rats. Transl. Androl. Urol. 2020, 9, 2071–2081. [Google Scholar] [CrossRef]
- Farzamfar, S.; Naseri-Nosar, M.; Ghanavatinejad, A.; Vaez, A.; Zarnani, A.H.; Salehi, M. Sciatic Nerve Regeneration by Transplantation of Menstrual Blood-Derived Stem Cells. Mol. Biol. Rep. 2017, 44, 407–412. [Google Scholar] [CrossRef]
- Lanza, R.; Langer, R.; Vacanti, J.P.; Atala, A. (Eds.) Principles of Tissue Engineering; Academic Press: Cambridge, MA, USA, 2020. [Google Scholar]
- Castiglione, F.; Dewulf, K.; Hakim, L.; Weyne, E.; Montorsi, F.; Russo, A.; Boeti, L.; Bivalacqua, T.J.; Rider, D.D.; Juniau, S.; et al. Adipose-Derived Stem Cells Counteract Urethral Stricture Formation in Rats. Eur. Urol. 2016, 70, 1032–1041. [Google Scholar] [CrossRef]
- Luo, J.; Zhao, S.; Wang, J.; Luo, L.; Li, E.; Zhu, Z.; Liu, Y.; Kang, R.; Zhao, Z. Bone Marrow Mesenchymal Stem Cells Reduce Ureteral Stricture Formation in a Rat Model Via the Paracrine Effect of Extracellular Vesicles. J. Cell. Mol. Med. 2018, 22, 4449–4459. [Google Scholar] [CrossRef] [PubMed]
- Nassar, W.; El-Ansary, M.; Sabry, D.; Mostafa, M.A.; Fayad, T.; Kotb, E.; Temraz, M.; Saad, A.-N.; Essa, W.; Adel, H. Umbilical Cord Mesenchymal Stem Cells Derived Extracellular Vesicles Can Safely Ameliorate the Progression of Chronic Kidney Diseases. Biomater. Res. 2016, 20, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Chopra, H.H.M.K.; Hung, M.K.; Kwong, D.L.; Zhang, C.F.; Pow, E.H.N. Insights into Endothelial Progenitor Cells: Origin, Classification, Potentials, and Prospects. Stem Cells Int. 2018, 230–241. [Google Scholar] [CrossRef]
- Peters, E.B. Endothelial Progenitor Cells for the Vascularization of Engineered Tissues. Tissue Eng. Part B Rev. 2018, 24, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-H.; Xu, Q.; Shen, T.; Li, J.-K.; Sheng, J.-Y.; Shi, H.-J. Prevention of In-Stent Restenosis with Endothelial Progenitor Cell (EPC) Capture Stent Placement Combined with Regional EPC Transplantation: An Atherosclerotic Rabbit Model. Cardiol. J. 2019, 26, 283–291. [Google Scholar] [CrossRef]
- Chen, C.; Zheng, S.; Zhang, X.; Dai, P.; Gao, Y.; Nan, L.; Zhang, Y. Transplantation of Amniotic Scaffold-Seeded Mesenchymal Stem Cells and/or Endothelial Progenitor Cells from Bone Marrow to Efficiently Repair 3-cm Circumferential Urethral Defect in Model Dogs. Tissue Eng. Part A 2018, 24, 47–56. [Google Scholar] [CrossRef]
- Chapple, C. Tissue Engineering of the Urethra: Where Are We in 2019? World J. Urol. 2019, 38, 2101–2105. [Google Scholar] [CrossRef] [PubMed]
- Coguplugil, A.E.; Ebiloglu, T.; Sarikaya, S.; Yilmaz, S.; Topuz, B.; Gurdal, M. Ventral Onlay Buccal Mucosa Graft Urethroplasty for Female Urethral Stricture. Int. J. Urol. 2021, 28, 538–543. [Google Scholar] [CrossRef] [PubMed]
- Daneshvar, M.; Simhan, J.; Blakely, S.; Angulo, J.C.; Lucas, J.; Hunter, C.; Chee, J.; Alvarado, D.L.; Perez, E.A.R.; Madala, A.; et al. Transurethral Ventral Buccal Mucosa Graft Inlay for Treatment of Distal Urethral Strictures: International Multi-Institutional Experience. World J. Urol. 2020, 38, 2601–2607. [Google Scholar] [CrossRef]
- Selim, M.; Salem, S.; Elsherif, E.; Badawy, A.; Elshazely, M.; Gawish, M. Outcome of Staged Buccal Mucosal Graft for Repair of Long Segment Anterior Urethral Stricture. BMC Urol. 2019, 19, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, S.; Yadav, S.S.; Tomar, V. Outcome of Buccal Mucosa and Lingual Mucosa Graft Urethroplasty in the Management of Urethral Strictures: A Comparative Study. Urol. Ann. 2016, 8, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Xie, M.; Xu, Y.; Song, L.; Wang, J.; Lv, X.; Zhang, Y. Tissue-Engineered Buccal Mucosa Using Silk Fibroin Matrices for Urethral Reconstruction in a Canine Model. J. Surg. Res. 2014, 188, 1–7. [Google Scholar] [CrossRef]
- Rowe, R.G.; Daley, G.Q. Induced Pluripotent Stem Cells in Disease Modelling and Drug Discovery. Nat. Rev. Genet. 2019, 20, 377–388. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, R.E.; Anzai, T.; Chanthra, N.; Uosaki, H. A Brief Review of Current Maturation Methods for Human Induced Pluripotent Stem Cells-Derived Cardiomyocytes. Front. Cell Dev. Biol. 2020, 8, 178. [Google Scholar] [CrossRef] [PubMed]
- Hirschi, K.K.; Li, S.; Roy, K. Induced Pluripotent Stem Cells for Regenerative Medicine. Annu. Rev. Biomed. Eng. 2014, 16, 277–294. [Google Scholar] [CrossRef]
- Li, Y.; Wen, Y.; Wang, Z.; Wei, Y.; Wani, P.; Green, M.; Swaminathan, G.; Ramamurthi, A.; Pera, R.R.; Chen, B. Smooth Muscle Progenitor Cells Derived from Human Pluripotent Stem Cells Induce Histologic Changes in Injured Urethral Sphincter. STEM CELLS Transl. Med. 2016, 5, 1719–1729. [Google Scholar] [CrossRef]
- Toh, W.S.; Lee, E.H.; Cao, T. Potential of Human Embryonic Stem Cells in Cartilage Tissue Engineering and Regenerative Medicine. Stem Cell Rev. Rep. 2010, 7, 544–559. [Google Scholar] [CrossRef]
- Eguizabal, C.; Aran, B.; Lopes, S.M.C.D.S.; Geens, M.; Heindryckx, B.; Panula, S.; Popovic, M.; Vassena, R.; Veiga, A. Two Decades of Embryonic Stem Cells: A Historical Overview. Hum. Reprod. Open 2019, 2019, hoy024. [Google Scholar] [CrossRef]
- Wang, Z.; Wen, Y.; Li, Y.H.; Wei, Y.; Green, M.; Wani, P.; Zhang, P.; Pera, R.R.; Chen, B. Smooth Muscle Precursor Cells Derived from Human Pluripotent Stem Cells for Treatment of Stress Urinary Incontinence. Stem Cells Dev. 2016, 25, 453–461. [Google Scholar] [CrossRef]
- Subramaniam, R.; Hinley, J.; Stahlschmidt, J.; Southgate, J. Tissue Engineering Potential of Urothelial Cells from Diseased Bladders. J. Urol. 2011, 186, 2014–2020. [Google Scholar] [CrossRef]
- Singh, A.; Bivalacqua, T.J.; Sopko, N. Urinary Tissue Engineering: Challenges and Opportunities. Sex. Med. Rev. 2018, 6, 35–44. [Google Scholar] [CrossRef]
- Atala, A. Experimental and Clinical Experience with Tissue Engineering Techniques for Urethral Reconstruction. Urol. Clin. N. Am. 2002, 29, 485–492. [Google Scholar] [CrossRef]
- Sievert, K.-D.; Daum, L.; Maurer, S.; Toomey, P.; Vaegler, M.; Aufderklamm, S.; Amend, B. Urethroplasty Performed with an Autologous Urothelium-Vegetated Collagen Fleece to Treat Urethral Stricture in the Minipig Model. World J. Urol. 2019, 38, 2123–2131. [Google Scholar] [CrossRef] [PubMed]
- de Kemp, V.; de Graaf, P.; Fledderus, J.O.; Ruud Bosch, J.L.H.; de Kort, L.M. Tissue Engineering for Human Urethral Reconstruction: Systematic Review of Recent Literature. PLoS ONE 2015, 10, e0118653. [Google Scholar] [CrossRef]
- Liu, G.; Fu, M.; Li, F.; Fu, W.; Zhao, Z.; Xia, H.; Niu, Y. Tissue-Engineered PLLA/Gelatine Nanofibrous Scaffold Promoting the Phenotypic Expression of Epithelial and Smooth Muscle Cells for Urethral Reconstruction. Mater. Sci. Eng. C 2020, 111, 110810. [Google Scholar] [CrossRef]
- Pokrywczynska, M.; Balcerczyk, D.; Jundzill, A.; Gagat, M.; Czapiewska, M.; Kloskowski, T.; Nowacki, M.; Gastecka, A.M.; Bodnar, M.; Grzanka, A.; et al. Isolation, Expansion and Characterization of Porcine Urinary Bladder Smooth Muscle Cells for Tissue Engineering. Biol. Proced. Online 2016, 18, 17. [Google Scholar] [CrossRef]
- Da Silva, L.F.A.; Micol, L.; Tiemessen, D.; Van Kuppevelt, T.H.; Frey, P.; Oosterwijk, E.; Geutjes, P.; Feitz, W.F. Is There a Need for Smooth Muscle Cell Transplantation in Urethral Reconstruction? Tissue Eng. Part A 2014, 20, 1542–1549. [Google Scholar] [CrossRef]
- Barbalias, D.; Lappas, G.; Ravazoula, P.; Liourdi, D.; Kyriazis, I.; Liatsikos, E.; Kallidonis, P. Evaluation of the Distribution of Paclitaxel After Application of a Paclitaxel-Coated Balloon in the Rabbit Urethra. J. Endourol. 2018, 32, 381–386. [Google Scholar] [CrossRef]
- Pranata, F.H.; Hidayatullah, F.; Kloping, Y.P.; Rahman, Z.A.; Rizaldi, F.; Soebadi, D.M. The Efficacy and Safety of Mitomycin C Intra Urethral Injection to Prevent Recurrent Urethral Stricture: A Systematic Review and Meta-Analysis. Ann. Med. Surg. 2022, 77, 103576. [Google Scholar] [CrossRef]
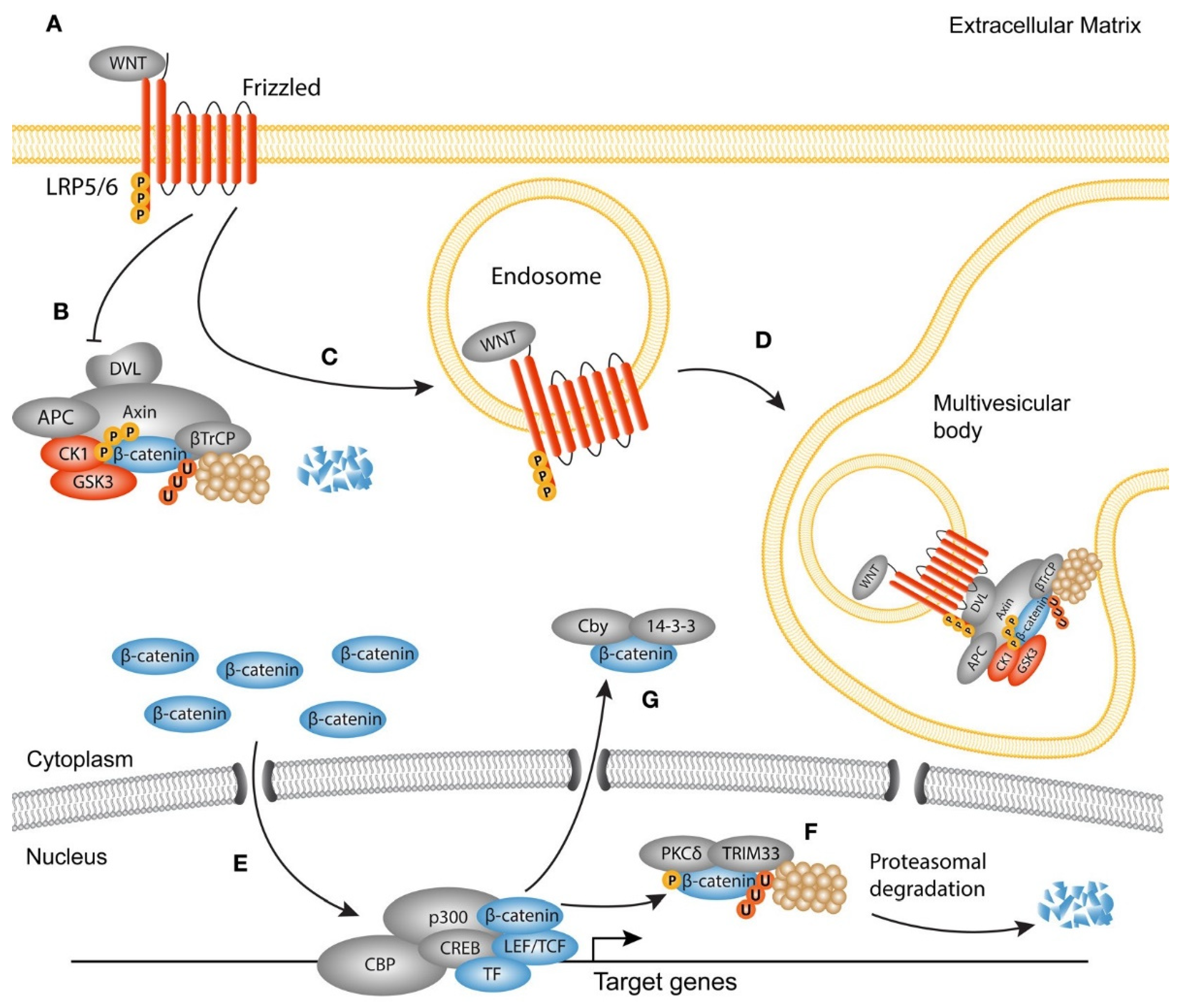
- Choi, K.H.; Kim, D.K.; Kim, A.R.; Lee, S. Prevention of Urethral Fibrosis Induced by Transforming Growth Factor Beta 1 Using Selective Wnt/Β-Catenin Signaling Inhibitors in A Rat Model. Int. J. Urol. 2022, 29, 764–771. [Google Scholar] [CrossRef]
- Han, K.; Park, J.-H.; Yang, S.-G.; Lee, D.H.; Tsauo, J.; Kim, K.Y.; Kim, M.T.; Gang, S.G.; Kim, D.-K.; Kim, D.-H.; et al. EW-7197 Eluting Nanofiber Covered Self-Expandable Metallic Stent to Prevent Granulation Tissue Formation in A Canine Urethral Model. PLoS ONE 2018, 13, e0192430. [Google Scholar] [CrossRef]
- Srilakshmi, N.; Hegre, P.S. Prospective Comparative Study of Visual Internal Urethrotomy and Visual Internal Urethrotomy with Intralesional Triamcinolone and Mitomycin C in Treatment of Stricture Urethra. Eur. J. Mol. Clin. Med. 2022, 9, 198–205. [Google Scholar]
- Abdallah, H.M.; Abuelnaga, M.; Rasmy, A.G. Intraurethral Steroid Injection Following Visual Internal Uerthrotomy in the Management of Anterior Urethral Strictures. QJM Int. J. Med. 2020, 113 (Suppl. S1), hcaa070. [Google Scholar] [CrossRef]
- Pang, K.H.; Chapple, C.R.; Chatters, R.; Downey, A.P.; Harding, C.K.; Hind, D.; Watkin, N.; Osman, N.I. A Systematic Review and Meta-analysis of Adjuncts to Minimally Invasive Treatment of Urethral Stricture in Men. Eur. Urol. 2021, 80, 467–479. [Google Scholar] [CrossRef]
- Mangir, N.; Chapple, C. Recent Advances in Treatment of Urethral Stricture Disease in Men. F1000Research 2020, 9, 330. [Google Scholar] [CrossRef]
- Zhang, D.; Sun, L.; Xian, W.; Liu, F.; Ling, G.; Xiao, L.; Liu, Y.; Peng, Y.; Haruna, Y.; Kanwar, Y.S. Low-Dose Paclitaxel Ameliorates Renal Fibrosis in Rat UUO Model by Inhibition of TGF-Β/Smad Activity. Lab. Investig. 2010, 90, 436–447. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Zhang, L.; Yang, R.; Xu, X.; Chen, J.; Wang, S.; He, Z.; Dong, Z.; Yang, J.; Zhao, Y.; et al. Paclitaxel Attenuates Renal Interstitial Fibroblast Activation and Interstitial Fibrosis by Inhibiting STAT3 Signaling. Drug Des. Dev. Ther. 2015, 9, 2139–2148. [Google Scholar] [CrossRef] [PubMed]
- Virasoro, R.; Delong, J.M.; Mann, R.A.; Estrella, R.E.; Pichardo, M.; Lay, R.R.; Espino, G.; Roth, J.D.; Elliott, S.P. A Drug-Coated Balloon Treatment for Urethral Stricture Disease: Interim Results from the ROBUST I Study. Can. Urol. Assoc. J. 2020, 14, 187–191. [Google Scholar] [CrossRef]
- Calori, I.R.; Braga, G.; de Jesus, P.D.C.C.; Bi, H.; Tedesco, A.C. Polymer Scaffolds as Drug Delivery Systems. Eur. Polym. J. 2020, 129, 109621. [Google Scholar] [CrossRef]
- Wisselink, D.D.; Braakhuis, L.L.; Gallo, G.; van Grevenstein, W.M.; van Dieren, S.; Kok, N.F.; de Reuver, P.R.; Tanis, P.J.; de Hingh, I.H. Systematic Review of Published Literature on Oxaliplatin and Mitomycin C As Chemotherapeutic Agents for Hyperthermic Intraperitoneal Chemotherapy in Patients with Peritoneal Metastases from Colorectal Cancer. Crit. Rev. Oncol. Hematol. 2019, 142, 119–129. [Google Scholar] [CrossRef]
- Gabizon, A.; Shmeeda, H.; Tahover, E.; Kornev, G.; Patil, Y.; Amitay, Y.; Ohana, P.; Sapir, E.; Zalipsky, S. Development of Promitil®, A Lipidic Prodrug of Mitomycin C In Pegylated Liposomes: From Bench to Bedside. Adv. Drug Deliv. Rev. 2020, 154, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Mazdak, H.; Meshki, I.; Ghassami, F. Effect of Mitomycin C on Anterior Urethral Stricture Recurrence after Internal Ure-Throtomy. Eur. Urol. 2007, 51, 1089–1092. [Google Scholar] [CrossRef] [PubMed]
- Zuo, Y.; Liu, Y. New Insights into The Role and Mechanism of Wnt/Β-Catenin Signalling in Kidney Fibrosis. Nephrology 2018, 23, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Miao, J.; Liu, J.; Niu, J.; Zhang, Y.; Shen, W.; Luo, C.; Liu, Y.; Li, C.; Li, H.; Yang, P.; et al. Wnt/β-catenin/RAS Signaling Mediates Age-Related Renal Fibrosis and Is Associated with Mitochondrial Dysfunction. Aging Cell 2019, 18, e13004. [Google Scholar] [CrossRef] [PubMed]
- Piersma, B.; Bank, R.A.; Boersema, M. Signaling in Fibrosis: TGF-β, WNT, and YAP/TAZ Converge. Front. Med. 2015, 2, 59. [Google Scholar] [CrossRef]
- Lv, X.; Li, J.; Hu, Y.; Wang, S.; Yang, C.; Li, C.; Zhong, G. Overexpression of miR-27b-3p Targeting Wnt3a Regulates the Signaling Pathway of Wnt/β-Catenin and Attenuates Atrial Fibrosis in Rats with Atrial Fibrillation. Oxidative Med. Cell. Longev. 2019, 2019, 1–13. [Google Scholar] [CrossRef]
- Sun, Z.; Yang, Z.; Wang, M.; Huang, C.; Ren, Y.; Zhang, W.; Gao, F.; Cao, L.; Li, L.; Nie, S. Paraquat Induces Pulmonary Fibrosis Through Wnt/Β-Catenin Signaling Pathway and Myofibroblast Differentiation. Toxicol. Lett. 2020, 333, 170–183. [Google Scholar] [CrossRef]
- Binabaj, M.M.; Asgharzadeh, F.; Avan, A.; Rahmani, F.; Soleimani, A.; Parizadeh, M.R.; Ferns, G.A.; Ryzhikov, M.R.; Khazaei, M. Hassanian, S.M. EW-7197 Prevents Ulcerative Colitis-Associated Fibrosis and Inflammation. J. Cell. Physiol. 2019, 234, 11654–11661. [Google Scholar] [CrossRef] [PubMed]
- Rockson, S.G. Inhibition of Fibrosis to Combat Lymphedema; Mary Ann Liebert, Inc.: New York, NY, USA, 2020; p. 399. [Google Scholar]
- Tabassi, K.T.; Yarmohamadi, A.; Mohammadi, S. Triamcinolone Injection Following Internal Urethrotomy for Treatment of Urethral Stricture. Urol. J. 2011, 8, 132. [Google Scholar]
- Mazdak, H.; Izadpanahi, M.H.; Ghalamkari, A.; Kabiri, M.; Khorrami, M.-H.; Nouri-Mahdavi, K.; Alizadeh, F.; Zargham, M.; Tadayyon, F.; Mohammadi, A.; et al. Internal Urethrotomy and Intraurethral Submucosal Injection of Triamcinolone in Short Bulbar Urethral Strictures. Int. Urol. Nephrol. 2009, 42, 565–568. [Google Scholar] [CrossRef]
- Zhou, W.; Yu, Q.; Ma, J.; Xu, C.; Wu, D.; Li, C. Triamcinolone Acetonide Combined With 5-Fluorouracil Suppresses Urethral Scar Fibroblasts Autophagy and Fibrosis by Increasing Mir-192-5p Expression. Am. J. Transl. Res. 2021, 13, 5956–5968. [Google Scholar] [PubMed]
- Rowe, C.K.; Foster, C.; Ruan, W.; Burke, K.A. Mp08-16 Novel Biomaterial for Slow-Release Drug Delivery to the Urethra. J. Urol. 2022, 207 (Suppl. 5), e131. [Google Scholar] [CrossRef]
- Tenney, J.; Hudson, N.; Alnifaidy, H.; Li, J.T.C.; Fung, K.H. Risk Factors for Aquiring Multidrug-Resistant Organisms in Urinary Tract Infections: A Systematic Literature Review. Saudi Pharm. J. 2018, 26, 678–684. [Google Scholar] [CrossRef] [PubMed]
- Murphy, A.M.; Wong, A.L.; Bezuhly, M. Modulation of Angiotensin II Signaling in The Prevention of Fibrosis. Fibrogenesis Tissue Repair 2015, 8, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kurniawan, W.; Soesatyo, M.H.N.E.; Aryandono, T. The Effects of Docetaxel And/Or Captopril in Expression Of TGF-Β1, MMP-1, CTGF, And PAI-1 As Markers of Anterior Urethral Stricture in an Animal Model. Ther. Adv. Urol. 2020, 12, 1756287220927994. [Google Scholar] [CrossRef]
- Shirazi, M.; Khezri, A.; Mohammadi-Samani, S.; Monabati, A.; Kojoori, J.; Hassanpour, A. Effect of Intraurethral Captopril Gel on the Recurrence of Urethral Stricture After Direct Vision Internal Urethrotomy: Phase II Clinical Trial. Int. J. Urol. 2007, 14, 203–208. [Google Scholar] [CrossRef]
- Eltom, A.; Zhong, G.; Muhammad, A. Scaffold Techniques and Designs in Tissue Engineering Functions and Purposes: A Review. Adv. Mater. Sci. Eng. 2019, 2019, 3429527. [Google Scholar] [CrossRef]
- Zhang, Y.; Yoo, J.J.; Atala, A. Tissue Engineering: Bladder and Urethra. Princ. Tissue Eng. 2020, 16, 845–862. [Google Scholar] [CrossRef]
- Gnecchi, M.; Danieli, P.; Malpasso, G.; Ciuffreda, M.C. Paracrine Mechanisms of Mesenchymal Stem Cells in Tissue Repair. Methods Mol. Biol. 2016, 1416, 123–146. [Google Scholar] [CrossRef]
- Farzamfar, S.; Nazeri, N.; Salehi, M.; Valizadeh, A.; Marashi, S.M.; Kouzehkonan, G.S.; Ghanbari, H. Will Nanotechnology Bring New Hope for Stem Cell Therapy? Cells Tissues Organs 2018, 206, 229–241. [Google Scholar] [CrossRef]
- Golchin, A.; Hosseinzadeh, S.; Roshangar, L. The Role of Nanomaterials in Cell Delivery Systems. Med. Mol. Morphol. 2017, 51, 1–12. [Google Scholar] [CrossRef]
- Bartold, P.M.; Xiao, Y.; Lyngstaadas, S.P.; Paine, M.L.; Snead, M.L. Principles and Applications of Cell Delivery Systems for Periodontal Regeneration. Periodontology 2000 2006, 41, 123–135. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Lu, M.; Wang, T.; Dian, D.; Zhong, Y.; Aleahmad, M. Human Amniotic Membrane as a Delivery Vehicle for Stem Cell-Based Therapies. Life Sci. 2021, 272, 119157. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Shi, L.; Duan, Z.; Rohani, S. Hyaluronic Acid Hydrogels, as a Biological Macromolecule-Based Platform for Stem Cells Delivery and Their Fate Control: A Review. Int. J. Biol. Macromol. 2021, 189, 554–566. [Google Scholar] [CrossRef]
- Li, Z.; Wang, Y.; Ding, Y.; Repp, L.; Kwon, G.S.; Hu, Q. Cell-Based Delivery Systems: Emerging Carriers for Immunotherapy. Adv. Funct. Mater. 2021, 31, 2100088. [Google Scholar] [CrossRef]
- Mooney, D.J.; Vandenburgh, H. Cell Delivery Mechanisms for Tissue Repair. Cell Stem Cell 2008, 2, 205–213. [Google Scholar] [CrossRef]
- Ullah, S.; Chen, X. Fabrication, Applications and Challenges of Natural Biomaterials in Tissue Engineering. Appl. Mater. Today 2020, 20, 100656. [Google Scholar] [CrossRef]
- Keshvardoostchokami, M.; Majidi, S.S.; Huo, P.; Ramachandran, R.; Chen, M.; Liu, B. Electrospun Nanofibers of Natural and Synthetic Polymers as Artificial Extracellular Matrix for Tissue Engineering. Nanomaterials 2021, 11, 21. [Google Scholar] [CrossRef]
- Nemati, S.; Kim, S.-J.; Shin, Y.M.; Shin, H. Current Progress in Application of Polymeric Nanofibers to Tissue Engineering. Nano Converg. 2019, 6, 1–16. [Google Scholar] [CrossRef]
- Abbasian, M.; Massoumi, B.; Mohammad-Rezaei, R.; Samadian, H.; Jaymand, M. Scaffolding Polymeric Biomaterials: Are Naturally Occurring Biological Macromolecules More Appropriate for Tissue Engineering? Int. J. Biol. Macromol. 2019, 134, 673–694. [Google Scholar] [CrossRef]
- Li, H.; Liu, Y. The Hopes and Hypes of Plant and Bacteria-Derived Cellulose Application in Stem Cell Technology. Cellulose 2022, 29, 3035–3058. [Google Scholar] [CrossRef]
- Abbas, T.O.; Ali, T.A.; Uddin, S. Urine as a Main Effector in Urological Tissue Engineering—A Double-Edged Sword. Cells 2020, 9, 538. [Google Scholar] [CrossRef] [PubMed]
- George, D. Cells in Urothelium Tissue Engineering, Stem Cells and Regenerative Medicine. In Proceedings of the 9th International Europe-China Symposium, Strasbourg, France, 2–4 October 2019; p. 110. [Google Scholar]
- Zhang, F.; King, M.W. Biodegradable Polymers as the Pivotal Player in the Design of Tissue Engineering Scaffolds. Adv. Health Mater. 2020, 9, e1901358. [Google Scholar] [CrossRef]
- Aguilar, L.M.C.; Silva, S.M.; Moulton, S.E. Growth Factor Delivery: Defining the Next Generation Platforms for Tissue Engineering. J. Control. Release 2019, 306, 40–58. [Google Scholar] [CrossRef]
- Zeng, Y.; Hoque, J.; Varghese, S. Biomaterial-Assisted Local and Systemic Delivery of Bioactive Agents for Bone Repair. Acta Biomater. 2019, 93, 152–168. [Google Scholar] [CrossRef]
- Ferracini, R.; Herreros, I.M.; Russo, A.; Casalini, T.; Rossi, F.; Perale, G. Scaffolds as Structural Tools for Bone-Targeted Drug Delivery. Pharmaceutics 2018, 10, 122. [Google Scholar] [CrossRef]
- Sarigol-Calamak, E.; Hascicek, C. Tissue Scaffolds as a Local Drug Delivery System for Bone Regeneration. Cut. Edge Enabling Technol. Regen. Med. 2018, 1078, 475–493. [Google Scholar] [CrossRef]
- Mouriño, V. Scaffolds with Drug Delivery Capability. In Tissue Engineering Using Ceramics and Polymers; Elsevier: Amsterdam, The Netherlands, 2022; pp. 817–840. [Google Scholar]
- Cao, S.; Deng, Y.; Zhang, L.; Aleahmad, M. Chitosan Nanoparticles, As Biological Macromolecule-Based Drug Delivery Systems to Improve the Healing Potential of Artificial Neural Guidance Channels: A Review. Int. J. Biol. Macromol. 2022, 201, 569–579. [Google Scholar] [CrossRef]
- Narayanaswamy, R.; Torchilin, V.P. Hydrogels and Their Applications in Targeted Drug Delivery. Molecules 2019, 24, 603. [Google Scholar] [CrossRef]
- Das, M.; Giri, T.K. Hydrogels Based on Gellan Gum in Cell Delivery and Drug Delivery. J. Drug Deliv. Sci. Technol. 2020, 56, 101586. [Google Scholar] [CrossRef]
- Zhang, K.; Fu, Q.; Yoo, J.; Chen, X.; Chandra, P.; Mo, X.; Song, L.; Atala, A.; Zhao, W. 3D Bioprinting of Urethra with PCL/PLCL Blend and Dual Autologous Cells in Fibrin Hydrogel: An In Vitro Evaluation of Biomimetic Mechanical Property and Cell Growth Environment. Acta Biomater. 2017, 50, 154–164. [Google Scholar] [CrossRef] [PubMed]
- Moradi, M.; Derakhshandeh, K.; Karimian, B.; Fasihi, M. Safety and Efficacy of Intraurethral Mitomycin C Hydrogel for Prevention of Post-Traumatic Anterior Urethral Stricture Recurrence After Internal Urethrotomy. J. Inj. Violence Res. 2016, 8, 75–79. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wan, Z.; Zhang, P.; Liu, Y.; Lv, L.; Zhou, Y. Four-dimensional bioprinting: Current developments and applications in bone tissue engineering. Acta Biomater. 2020, 101, 26–42. [Google Scholar] [CrossRef] [PubMed]
- Wan, X.; Xie, M.-K.; Xu, H.; Wei, Z.-W.; Yao, H.-J.; Wang, Z.; Zheng, D.-C. Hypoxia-Preconditioned Adipose-Derived Stem Cells Combined with Scaffold Promote Urethral Reconstruction by Upregulation of Angiogenesis and Glycolysis. Stem Cell Res. Ther. 2020, 11, 1–16. [Google Scholar] [CrossRef]
- Vaddi, S.P.; Reddy, V.B.; Abraham, S.J. Buccal Epithelium Expanded and Encapsulated in Scaffold-Hybrid Approach to Urethral Stricture (BEES-HAUS) Procedure: A Novel Cell Therapy-Based Pilot Study. Int. J. Urol. 2019, 26, 253–257. [Google Scholar] [CrossRef]
- Horiguchi, A.; Ojima, K.; Shinchi, M.; Kushibiki, T.; Mayumi, Y.; Miyai, K.; Katoh, S.; Takeda, M.; Iwasaki, M.; Prakash, V.S.; et al. Successful Engraftment of Epithelial Cells Derived from Autologous Rabbit Buccal Mucosal Tissue, Encapsulated in a Polymer Scaffold in a Rabbit Model of a Urethral Stricture, Transplanted Using the Transurethral Approach. Regen. Ther. 2021, 18, 127–132. [Google Scholar] [CrossRef]
- Yamamoto, T.; Funahashi, Y.; Mastukawa, Y.; Tsuji, Y.; Mizuno, H.; Nakayama, K.; Gotoh, M. Mp19-17 Human Urethra-Engineered with Human Mesenchymal Stem Cell with Maturation by Rearrangement of Cells for Self-Organization-Newly Developed Scaffold-Free Three-Dimensional Bio-Printer. J. Urol. 2015, 193, e221–e222. [Google Scholar] [CrossRef]
- De Filippo, R.E.; Kornitzer, B.S.; Yoo, J.J.; Atala, A. Penile Urethra Replacement with Autologous Cell-Seeded Tubularized Collagen Matrices. J. Tissue Eng. Regen. Med. 2012, 9, 257–264. [Google Scholar] [CrossRef]
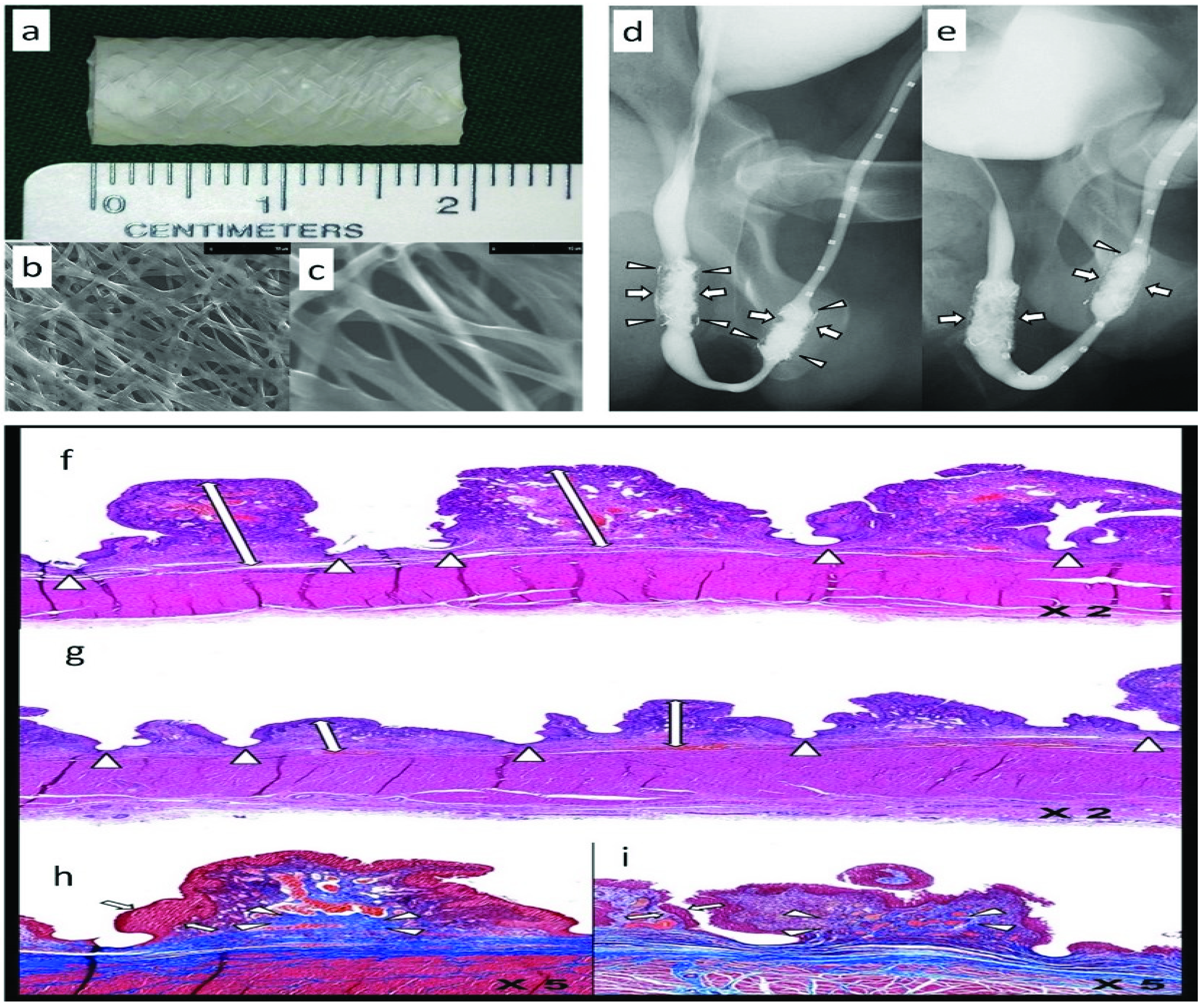
- Lu, X.; Feng, C.; Liu, Y.; Peng, X.; Chen, S.; Xiao, D.; Wang, H.; Li, Z.; Xu, Y.; Lu, M. A Smart Bilayered Scaffold Supporting Keratinocytes and Muscle Cells in Micro/Nanoscale for Urethral Reconstruction. Theranostics 2018, 8, 3153. [Google Scholar] [CrossRef]
- Huang, J.-W.; Lv, X.-G.; Li, Z.; Song, L.-J.; Feng, C.; Xie, M.-K.; Li, C.; Li, H.-B.; Wang, J.-H.; Zhu, W.-D.; et al. Urethral Reconstruction with A 3D Porous Bacterial Cellulose Scaffold Seeded with Lingual Keratinocytes in a Rabbit Model. Biomed. Mater. 2015, 10, 055005. [Google Scholar] [CrossRef]
- Wu, S.; Liu, Y.; Bharadwaj, S.; Atala, A.; Zhang, Y. Human Urine-Derived Stem Cells Seeded in a Modified 3D Porous Small Intestinal Submucosa Scaffold for Urethral Tissue Engineering. Biomaterials 2011, 32, 1317–1326. [Google Scholar] [CrossRef]
- Yudintceva, N.M.; Nashchekina, Y.A.; Mikhailova, N.A.; Vinogradova, T.I.; Yablonsky, P.K.; Gorelova, A.A.; Muraviov, A.N.; Gorelov, A.V.; Samusenko, I.A.; Nikolaev, B.P.; et al. Urethroplasty with a bilayered poly-D, L-lactide-co-ε-caprolactone scaffold seeded with allogenic mesenchymal stem cells. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020, 108, 1010–1021. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wu, J.; Feng, F.; Men, C.; Yang, D.; Gao, Z.; Zhu, Z.; Cui, Y.; Zhao, H. A Preclinical Study of Cell-seeded Tubularized Scaffolds Specially Secreting LL37 for Reconstruction of Long Urethral Defects. Anticancer Res. 2017, 37, 4295–4301. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhu, J.; Yang, F.; He, F.; Tian, X.; Tang, S.; Chen, X. A Tubular Gelatin Scaffold Capable of the Time-Dependent Controlled Release of Epidermal Growth Factor and Mitomycin C. Colloids Surfaces B Biointerfaces 2015, 135, 416–424. [Google Scholar] [CrossRef]
- Wang, Z.; Li, Q.; Wang, P.; Yang, M. Biodegradable Drug-Eluting Urethral Stent in Limiting Urethral Stricture Formation After Urethral Injury: An Experimental Study in Rabbit. J. Bioact. Compat. Polym. 2020, 35, 378–388. [Google Scholar] [CrossRef]
- Lim, W.S.; Chen, K.; Chong, T.W.; Xiong, G.M.; Birch, W.R.; Pan, J.; Lee, B.H.; Er, P.S.; Salvekar, A.V.; Venkatraman, S.S.; et al. A Bilayer Swellable Drug-Eluting Ureteric Stent: Localized Drug Delivery to Treat Urothelial Diseases. Biomaterials 2018, 165, 25–38. [Google Scholar] [CrossRef] [PubMed]
- Shinchi, M.; Kushibiki, T.; Mayumi, Y.; Ito, K.; Asano, T.; Ishihara, M.; Horiguchi, A. Insulin-Like Growth Factor 1 Sustained-Release Collagen on Urethral Catheter Prevents Stricture After Urethral Injury in a Rabbit Model. Int. J. Urol. 2019, 26, 572–577. [Google Scholar] [CrossRef] [PubMed]
- Ehterami, A.; Salehi, M.; Farzamfar, S.; Vaez, A.; Samadian, H.; Sahrapeyma, H.; Mirzaii, M.; Ghorbani, S.; Goodarzi, A. In Vitro and In Vivo Study Of PCL/COLL Wound Dressing Loaded with Insulin-Chitosan Nanoparticles on Cutaneous Wound Healing in Rats Model. Int. J. Biol. Macromol. 2018, 117, 601–609. [Google Scholar] [CrossRef]
- Samadian, H.; Salehi, M.; Farzamfar, S.; Vaez, A.; Ehterami, A.; Sahrapeyma, H.; Goodarzi, A.; Ghorbani, S. In Vitro And In Vivo Evaluation of Electrospun Cellulose Acetate/Gelatin/Hydroxyapatite Nanocomposite Mats for Wound Dressing Applications. Artif. Cells Nanomed. Biotechnol. 2018, 46, 964–974. [Google Scholar] [CrossRef] [PubMed]
- Farzamfar, S.; Naseri-Nosar, M.; Vaez, A.; Esmaeilpour, F.; Ehterami, A.; Sahrapeyma, H.; Samadian, H.; Hamidieh, A.-A.; Ghorbani, S.; Goodarzi, A.; et al. Neural Tissue Regeneration by a Gabapentin-Loaded Cellulose Acetate/Gelatin Wet-Electrospun Scaffold. Cellulose 2017, 25, 1229–1238. [Google Scholar] [CrossRef]
- Blackstone, B.; Gallentine, S.; Powell, H. Collagen-Based Electrospun Materials for Tissue Engineering: A Systematic Review. Bioengineering 2021, 8, 39. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Liu, C.; Mao, X.; Dong, J.; Fan, W.; Zhi, C.; Qin, X.; Sun, R. Multiple-Jet Needleless Electrospinning Approach via a Linear Flume Spinneret. Polymers 2019, 11, 2052. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Mukherjee, S.; Liu, Y.; Ramakrishna, S. Recent Studies on Electrospinning Preparation of Patterned, Core–Shell, and Aligned Scaffolds. J. Appl. Polym. Sci. 2018, 135, 46570. [Google Scholar] [CrossRef]
- Bombin, A.D.J.; Dunne, N.J.; McCarthy, H.O. Electrospinning of Natural Polymers for the Production of Nanofibres for Wound Healing Applications. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 114, 110994. [Google Scholar] [CrossRef]
- Tiwari, S.K.; Venkatraman, S.S. Importance of Viscosity Parameters in Electrospinning: Of Monolithic and Core–Shell Fibers. Mater. Sci. Eng. C 2012, 32, 1037–1042. [Google Scholar] [CrossRef]
- Li, Z.; Wang, C. Effects of Working Parameters on Electrospinning. In One-Dimensional Nanostructures: Electrospinning Technique and Unique Nanofibers; Li, A., Wang, C., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 15–28. [Google Scholar]
- Blachowicz, T.; Ehrmann, A. Conductive Electrospun Nanofiber Mats. Materials 2019, 13, 152. [Google Scholar] [CrossRef]
- Liu, Y.; Dong, L.; Fan, J.; Wang, R.; Yu, J.-Y. Effect of Applied Voltage on Diameter and Morphology of Ultrafine Fibers in Bubble Electrospinning. J. Appl. Polym. Sci. 2010, 120, 592–598. [Google Scholar] [CrossRef]
- Eslamian, M.; Khorrami, M.; Yi, N.; Majd, S.; Abidian, M.R. Electrospinning of Highly Aligned Fibers for Drug Delivery Applications. J. Mater. Chem. B 2019, 7, 224–232. [Google Scholar] [CrossRef]
- Xue, J.; Wu, T.; Dai, Y.; Xia, Y. Electrospinning and Electrospun Nanofibers: Methods, Materials, and Applications. Chem. Rev. 2019, 119, 5298–5415. [Google Scholar] [CrossRef]
- Islam, M.S.; Ang, B.C.; Andriyana, A.; Afifi, A.M. A Review on Fabrication of Nanofibers Via Electrospinning and Their Applications. SN Appl. Sci. 2019, 1, 1248. [Google Scholar] [CrossRef]
- Dziemidowicz, K.; Sang, Q.; Wu, J.; Zhang, Z.; Zhou, F.; Lagaron, J.M.; Mo, X.-M.; Parker, G.J.M.; Yu, D.-G.; Zhu, L.-M.; et al. Electrospinning for Healthcare: Recent Advancements. J. Mater. Chem. B 2020, 9, 939–951. [Google Scholar] [CrossRef] [PubMed]
- Mailley, D.; Hébraud, A.; Schlatter, G. A Review on the Impact of Humidity during Electrospinning: From the Nanofiber Structure Engineering to the Applications. Macromol. Mater. Eng. 2021, 306, 2100115. [Google Scholar] [CrossRef]
- De Vrieze, S.; Van Camp, T.; Nelvig, A.; Hagström, B.; Westbroek, P.; De Clerck, K. The Effect of Temperature and Humidity on Electrospinning. J. Mater. Sci. 2009, 44, 1357–1362. [Google Scholar] [CrossRef]
- Tucker, N.; Stanger, J.J.; Staiger, M.; Razzaq, H.; Hofman, K. The History of the Science and Technology of Electrospinning from 1600 to 1995. J. Eng. Fibers Fabr. 2012, 7 (Suppl. S2). [Google Scholar] [CrossRef]
- Singh, A.; Yadav, C.; Tabassum, N.; Bajpeyee, A.; Verma, V. Stem Cell Niche: Dynamic Neighbor of Stem Cells. Eur. J. Cell Biol. 2018, 98, 65–73. [Google Scholar] [CrossRef]
- Tenney, R.M.; Discher, D.E. Stem Cells, Microenvironment Mechanics, And Growth Factor Activation. Curr. Opin. Cell Biol. 2009, 21, 630–635. [Google Scholar] [CrossRef]
- McCarthy, N.; Kraiczy, J.; Shivdasani, R.A. Cellular and Molecular Architecture of the Intestinal Stem Cell Niche. Nat. Cell Biol. 2020, 22, 1033–1041. [Google Scholar] [CrossRef]
- Chaudhuri, O.; Cooper-White, J.; Janmey, P.A.; Mooney, D.J.; Shenoy, V.B. Effects of Extracellular Matrix Viscoelasticity on Cellular Behaviour. Nature 2020, 584, 535–546. [Google Scholar] [CrossRef] [PubMed]
- Walma, D.A.C.; Yamada, K.M. The Extracellular Matrix in Development. Development 2020, 147, dev175596. [Google Scholar] [CrossRef]
- Hussey, G.S.; Dziki, J.L.; Badylak, S.F. Extracellular Matrix-Based Materials for Regenerative Medicine. Nat. Rev. Mater. 2018, 3, 159–173. [Google Scholar] [CrossRef]
- Zhao, X.; Niu, Y.; Mi, C.; Gong, H.; Yang, X.; Cheng, J.; Zhou, Z.; Liu, J.; Peng, X.; Wei, D. Electrospinning Nanofibers of Microbial Polyhydroxyalkanoates for Applications in Medical Tissue Engineering. J. Appl. Polym. Sci. 2021, 59, 1994–2013. [Google Scholar] [CrossRef]
- Kular, J.K.; Basu, S.; I Sharma, R. The Extracellular Matrix: Structure, Composition, Age-Related Differences, Tools for Analysis and Applications for Tissue Engineering. J. Tissue Eng. 2014, 5, 2041731414557112. [Google Scholar] [CrossRef] [PubMed]
- Perestrelo, T.; Correia, M.; Ramalho-Santos, J.; Wirtz, D. Metabolic and Mechanical Cues Regulating Pluripotent Stem Cell Fate. Trends Cell Biol. 2018, 28, 1014–1029. [Google Scholar] [CrossRef] [PubMed]
- Fahy, N.; Alini, M.; Stoddart, M.J. Mechanical Stimulation of Mesenchymal Stem Cells: Implications for Cartilage Tissue Engineering. J. Orthop. Res. 2018, 36, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Chandra, P.K.; Soker, S.; Atala, A. Tissue Engineering: Current Status and Future Perspectives. In Principles of Tissue Engineering; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1–35. [Google Scholar] [CrossRef]
- McLaughlin, K.A.; Levin, M. Bioelectric Signaling in Regeneration: Mechanisms of Ionic Controls of Growth and Form. Dev. Biol. 2018, 433, 177–189. [Google Scholar] [CrossRef]
- Burnstine-Townley, A.; Eshel, Y.; Amdursky, N. Conductive Scaffolds for Cardiac and Neuronal Tissue Engineering: Governing Factors and Mechanisms. Adv. Funct. Mater. 2020, 30, 1901369. [Google Scholar] [CrossRef]
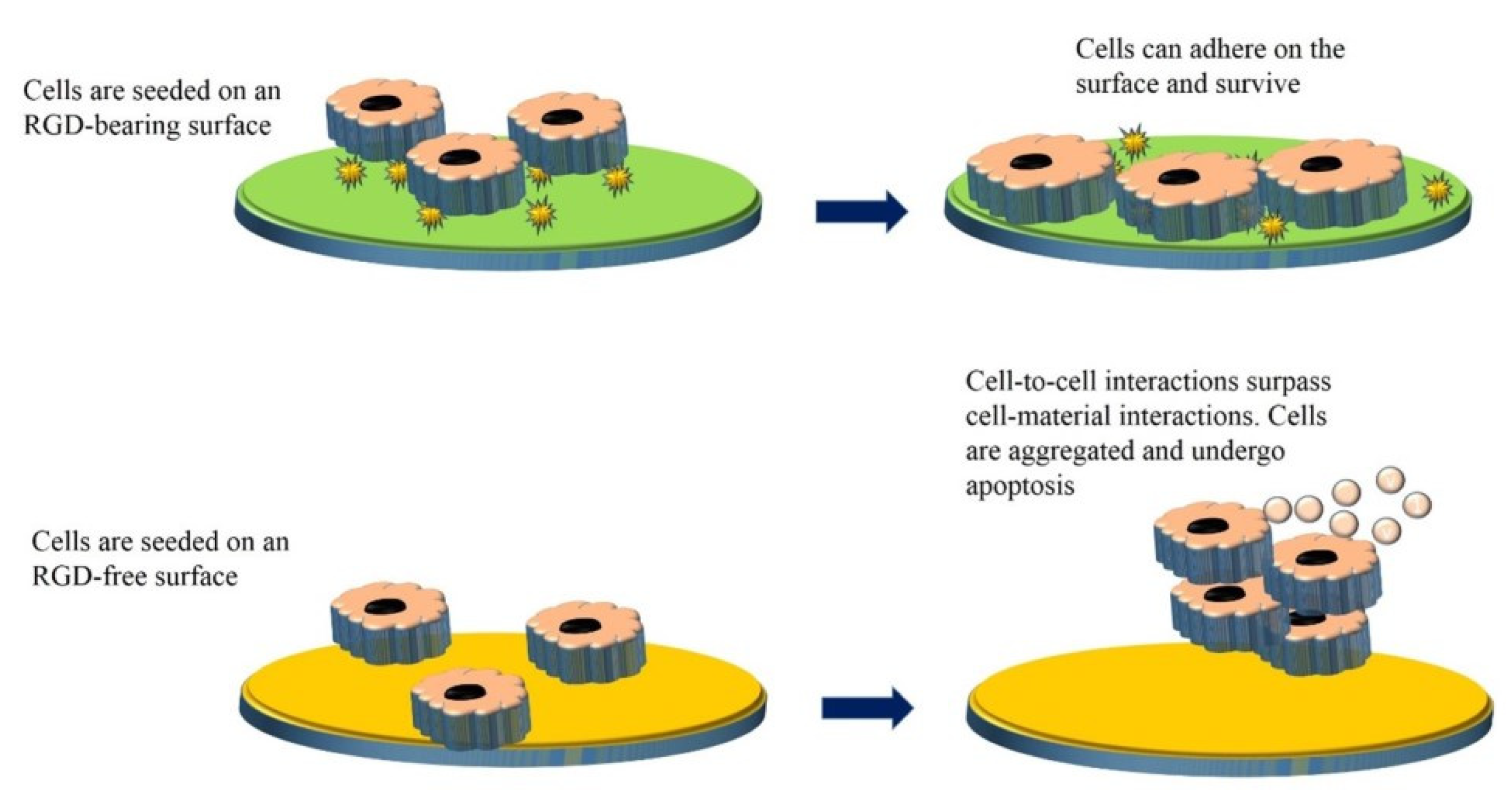
- Amani, H.; Arzaghi, H.; Bayandori, M.; Dezfuli, A.S.; Pazoki-Toroudi, H.; Shafiee, A.; Moradi, L. Controlling Cell Behavior through the Design of Biomaterial Surfaces: A Focus on Surface Modification Techniques. Adv. Mater. Interfaces 2019, 6, 48. [Google Scholar] [CrossRef]
- Ferrari, M.; Cirisano, F.; Morán, M.C. Mammalian Cell Behavior on Hydrophobic Substrates: Influence of Surface Properties. Colloids Interfaces 2019, 3, 48. [Google Scholar] [CrossRef]
- Wang, S.; Li, J.; Zhou, Z.; Zhou, S.; Hu, Z. Micro-/Nano-Scales Direct Cell Behavior on Biomaterial Surfaces. Molecules 2018, 24, 75. [Google Scholar] [CrossRef]
- Fedele, C.; Mäntylä, E.; Belardi, B.; Hamkins-Indik, T.; Cavalli, S.; Netti, P.A.; Fletcher, D.A.; Nymark, S.; Priimagi, A.; Ihalainen, T.O. Azobenzene-Based Sinusoidal Surface Topography Drives Focal Adhesion Confinement and Guides Collective Migration of Epithelial Cells. Sci. Rep. 2020, 10, 15329. [Google Scholar] [CrossRef]
- Farzamfar, S.; Hasanpour, A.; Nazeri, N.; Razavi, H.; Salehi, M.; Shafei, S.; Nooshabadi, V.T.; Vaez, A.; Ehterami, A.; Sahrapeyma, H.; et al. Extracellular Micro/Nanovesicles Rescue Kidney from Ischemia-Reperfusion Injury. J. Cell. Physiol. 2019, 234, 12290–12300. [Google Scholar] [CrossRef] [PubMed]
- Nooshabadi, V.T.; Khanmohamadi, M.; Valipour, E.; Mahdipour, S.; Salati, A.; Malekshahi, Z.V.; Shafei, S.; Amini, E.; Farzamfar, S.; Ai, J. Impact of Exosome-Loaded Chitosan Hydrogel in Wound Repair and Layered Dermal Reconstitution in Mice Animal Model. J. Biomed. Mater. Res. Part A 2020, 108, 2138–2149. [Google Scholar] [CrossRef] [PubMed]
- Shafei, S.; Khanmohammadi, M.; Heidari, R.; Ghanbari, H.; Taghdiri Nooshabadi, V.; Farzamfar, S.; Akbariqomi, M.; Sanikhani, N.S.; Absalan, M.; Tavoosidana, G. Exosome Loaded Alginate Hydrogel Promotes Tissue Regeneration in Full-Thickness Skin Wounds: An In Vivo Study. J. Biomed. Mater. Res. Part A 2020, 108, 545–556. [Google Scholar] [CrossRef]
- Keshtkar, S.; Azarpira, N.; Ghahremani, M.H. Mesenchymal Stem Cell-Derived Extracellular Vesicles: Novel Frontiers in Regenerative Medicine. Stem Cell Res. Ther. 2018, 9, 63. [Google Scholar] [CrossRef] [PubMed]
- Ghosal, K.; Agatemor, C.; Špitálsky, Z.; Thomas, S.; Kny, E. Electrospinning Tissue Engineering and Wound Dressing Scaffolds from Polymer-Titanium Dioxide Nanocomposites. Chem. Eng. J. 2019, 358, 1262–1278. [Google Scholar] [CrossRef]
- Zhang, Y.; Luo, J.; Zhang, Q.; Deng, T. Growth Factors, As Biological Macromolecules in Bioactivity Enhancing of Electrospun Wound Dressings for Diabetic Wound Healing: A Review. Int. J. Biol. Macromol. 2021, 193, 205–218. [Google Scholar] [CrossRef]
- Farzamfar, S.; Naseri-Nosar, M.; Samadian, H.; Mahakizadeh, S.; Tajerian, R.; Rahmati, M.; Veaz, A.; Salehi, M. Taurine-Loaded Poly (Ε-Caprolactone)/Gelatin Electrospun Mat as a Potential Wound Dressing Material: In Vitro and In Vivo Evaluation. J. Bioact. Compat. Polym. 2018, 33, 282–294. [Google Scholar] [CrossRef]
- Salehi, M.; Shahporzadeh, K.; Ehterami, A.; Yeganehfard, H.; Ziaei, H.; Azizi, M.M.; Farzamfar, S.; Tahersoltani, A.; Goodarzi, A.; Ai, J.; et al. Electrospun Poly (ε-caprolactone)/Gelatin Nanofibrous Mat Containing Selenium as a Potential Wound Dressing Material: In Vitro and In Vivo Study. Fibers Polym. 2020, 21, 1713–1721. [Google Scholar] [CrossRef]
- Begines, B.; Ortiz, T.; Pérez-Aranda, M.; Martínez, G.; Merinero, M.; Argüelles-Arias, F.; Alcudia, A. Polymeric Nanoparticles for Drug Delivery: Recent Developments and Future Prospects. Nanomaterials 2020, 10, 1403. [Google Scholar] [CrossRef]
- Naseri-Nosar, M.; Farzamfar, S.; Sahrapeyma, H.; Ghorbani, S.; Bastami, F.; Vaez, A.; Salehi, M. Cerium Oxide Nanoparticle-Containing Poly (Ε-Caprolactone)/Gelatin Electrospun Film as a Potential Wound Dressing Material: In Vitro and In Vivo Evaluation. Mater. Sci. Eng. C 2017, 81, 366–372. [Google Scholar] [CrossRef]
- Zhang, S.; Xing, M.; Li, B. Biomimetic Layer-By-Layer Self-Assembly of Nanofilms, Nanocoatings, and 3D Scaffolds for Tissue Engineering. Int. J. Mol. Sci. 2018, 19, 1641. [Google Scholar] [CrossRef] [PubMed]
- Easton, C.D.; Bullock, A.J.; Gigliobianco, G.; McArthur, S.L.; MacNeil, S. Application of Layer-By-Layer Coatings to Tissue Scaffolds–Development of an Angiogenic Biomaterial. J. Mater. Chem. B 2014, 2, 5558–5568. [Google Scholar] [CrossRef] [PubMed]
- Ariga, K.; Lvov, Y.M.; Kawakami, K.; Ji, Q.; Hill, J.P. Layer-By-Layer Self-Assembled Shells for Drug Delivery. Adv. Drug Deliv. Rev. 2011, 63, 762–771. [Google Scholar] [CrossRef] [PubMed]
- Wohl, B.M.; Engbersen, J.F. Responsive Layer-By-Layer Materials for Drug Delivery. J. Control. Release 2012, 158, 2–14. [Google Scholar] [CrossRef]
- Yoo, H.S.; Kim, T.G.; Park, T.G. Surface-Functionalized Electrospun Nanofibers for Tissue Engineering and Drug Delivery. Adv. Drug Deliv. Rev. 2009, 61, 1033–1042. [Google Scholar] [CrossRef]
- Ning, Y.; Shen, W.; Ao, F. Application of Blocking and Immobilization of Electrospun Fiber in the Biomedical Field. RSC Adv. 2020, 10, 37246–37265. [Google Scholar] [CrossRef]
- Ding, Y.; Floren, M.; Tan, W. Mussel-Inspired Polydopamine for Bio-Surface Functionalization. Biosurface Biotribology 2016, 2, 121–136. [Google Scholar] [CrossRef]
- Ambekar, R.S.; Kandasubramanian, B. A Polydopamine-Based Platform for Anti-Cancer Drug Delivery. Biomater. Sci. 2019, 7, 1776–1793. [Google Scholar] [CrossRef]
- Lee, J.; Yoo, J.J.; Atala, A.; Lee, S.J. The Effect of Controlled Release of PDGF-BB From Heparin-Conjugated Electrospun PCL/Gelatin Scaffolds on Cellular Bioactivity and Infiltration. Biomaterials 2012, 33, 6709–6720. [Google Scholar] [CrossRef]
- Wang, C.; Chen, C.; Guo, M.; Li, B.; Han, F.; Chen, W. Stretchable Collagen-Coated Polyurethane-Urea Hydrogel Seeded with Bladder Smooth Muscle Cells for Urethral Defect Repair in a Rabbit Model. J. Mater. Sci. Mater. Med. 2019, 30, 135. [Google Scholar] [CrossRef]
- Joshi, V.S.; Lei, N.Y.; Walthers, C.M.; Wu, B.; Dunn, J.C. Macroporosity Enhances Vascularization of Electrospun Scaffolds. J. Surg. Res. 2013, 183, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.; Liu, G.; Chen, C.; Fu, M.; Fu, W.; Zhao, Z.; Xia, H.; Stadler, F.J. Urethral Reconstruction Using an Amphiphilic Tissue-Engineered Autologous Polyurethane Nanofiber Scaffold with Rapid Vascularization Function. Biomater. Sci. 2020, 8, 2164–2174. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.; Liu, G.; Fu, M.; Chen, C.; Fu, W.; Zhang, Z.; Xia, H.; Stadler, F.J. Designing a Multifaceted Bio-Interface Nanofiber Tissue-Engineered Tubular Scaffold Graft to Promote Neo-Vascularization for Urethral Regeneration. J. Mater. Chem. B 2020, 8, 1748–1758. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Jakus, A.E.; Kural, M.; Qian, H.; Engler, A.; Ghaedi, M.; Shah, R.; Steinbacher, D.M.; Niklason, L.E. Vascularization of Natural and Synthetic Bone Scaffolds. Cell Transplant. 2018, 27, 1269–1280. [Google Scholar] [CrossRef]
- Lindhorst, D.; Tavassol, F.; Von See, C.; Schumann, P.; Laschke, M.W.; Harder, Y.; Bormann, k.H.; Essig, H.; Kokemüller, H.; Kampmann, A.; et al. Effects of VEGF Loading on Scaffold-Confined Vascularization. J. Biomed. Mater. Res. Part A 2010, 95, 783–792. [Google Scholar] [CrossRef]
- Singh, S.; Wu, B.M.; Dunn, J.C. Accelerating Vascularization in Polycaprolactone Scaffolds by Endothelial Progenitor Cells. Tissue Eng. Part A 2011, 17, 1819–1830. [Google Scholar] [CrossRef]
- Miana, V.V.; González, E.A.P. Adipose Tissue Stem Cells in Regenerative Medicine. Ecancermedicalscience 2018, 12, 822. [Google Scholar] [CrossRef]
- Wang, D.-J.; Li, M.-Y.; Huang, W.-T.; Lu, M.-H.; Hu, C.; Li, K.; Qiu, J.-G.; Gao, X. Repair of Urethral Defects with Polylactid Acid Fibrous Membrane Seeded with Adipose-Derived Stem Cells in a Rabbit Model. Connect. Tissue Res. 2015, 56, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Salehi, M.; Naseri-Nosar, M.; Ebrahimi-Barough, S.; Nourani, M.; Khojasteh, A.; Hamidieh, A.A.; Amani, A.; Farzamfar, S.; Ai, J. Sciatic Nerve Regeneration by Transplantation of Schwann Cells Via Erythropoietin Controlled-Releasing Polylactic Acid/Multiwalled Carbon Nanotubes/Gelatin Nanofibrils Neural Guidance Conduit. J. Biomed. Mater. Res. Part B Appl. Biomater. 2018, 106, 1463–1476. [Google Scholar] [CrossRef]
- Lu, X.; Guo, Q.; Han, F.; Chen, C.; Ling, C.; Chen, W.; Li, B. Electrospun Poly (L-Lactide)/Poly (Ethylene Glycol) Scaffolds Seeded with Human Amniotic Mesenchymal Stem Cells for Urethral Epithelium Repair. Int. J. Mol. Sci. 2016, 17, 1262. [Google Scholar] [CrossRef]
- Jing, H.; He, X.; Zheng, J. Exosomes and Regenerative Medicine: State of the Art and Perspectives. Transl. Res. 2018, 196, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Cheng, W.; Zhu, J.; Li, W.; Li, D.; Yang, X.; Zhao, W.; Ren, M.; Ren, J.; Mo, X.; et al. Electrospun Nanoyarn and Exosomes of Adipose-Derived Stem Cells for Urethral Regeneration: Evaluations In Vitro and In Vivo. Colloids Surfaces B Biointerfaces 2021, 209, 112218. [Google Scholar] [CrossRef]
- Lee, J.Y.; Kim, H.S. Extracellular Vesicles in Regenerative Medicine: Potentials and Challenges. Tissue Eng. Regen. Med. 2021, 18, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Masoumipour, M.; Abbaspanah, B.; Mousavi, S.H. Extracellular Vesicles: Regenerative Medicine Prospect in Hematological Malignancies. Cell Biol. Int. 2021, 45, 2031–2044. [Google Scholar] [CrossRef] [PubMed]
- Langer, R.; Vacanti, J. Advances in Tissue Engineering. J. Pediatr. Surg. 2016, 51, 8–12. [Google Scholar] [CrossRef]
- Sun, W.; Gregory, D.A.; Tomeh, M.A.; Zhao, X. Silk Fibroin as a Functional Biomaterial for Tissue Engineering. Int. J. Mol. Sci. 2021, 22, 1499. [Google Scholar] [CrossRef]
- Xie, M.; Song, L.; Wang, J.; Fan, S.; Zhang, Y.; Xu, Y. Evaluation of Stretched Electrospun Silk Fibroin Matrices Seeded with Urothelial Cells for Urethra Reconstruction. J. Surg. Res. 2013, 184, 774–781. [Google Scholar] [CrossRef] [PubMed]
- Boni, B.O.O.; Bakadia, B.M.; Osi, A.R.; Shi, Z.; Chen, H.; Gauthier, M.; Yang, G. Immune Response to Silk Sericin–Fibroin Composites: Potential Immunogenic Elements and Alternatives for Immunomodulation. Macromol. Biosci. 2021, 22, 2100292. [Google Scholar] [CrossRef]
- Jana, S.; Lerman, A. In Vivo Tissue Engineering of a Trilayered Leaflet-Shaped Tissue Construct. Regen. Med. 2020, 15, 1177–1192. [Google Scholar] [CrossRef]
- Hu, J.; Ai, B.; Zhu, S.; Wang, Z.; Xia, H.; Jia, W. Electrospun PLGA and PLGA/Gelatin Scaffolds for Tubularized Urethral Replacement: Studies In Vitro and In Vivo. J. Biomater. Appl. 2022, 36, 956–964. [Google Scholar] [CrossRef]
- Niu, Y.; Stadler, F.J.; Yang, X.; Deng, F.; Liu, G.; Xia, H. HA-Coated Collagen Nanofibers for Urethral Regeneration Via in Situ Polarization of M2 Macrophages. J. Nanobiotechnol. 2021, 19, 283. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Zhi, Y.; He, M.; Ahmadzadeh, B.; Rohani, S. Cellulose Acetate/Plerixafor Wound Dressings for Transplantation of Menstrual Blood Stem Cells: Potential Treatment Modality for Diabetic Wounds. J. Drug Deliv. Sci. Technol. 2022, 71, 103319. [Google Scholar] [CrossRef]
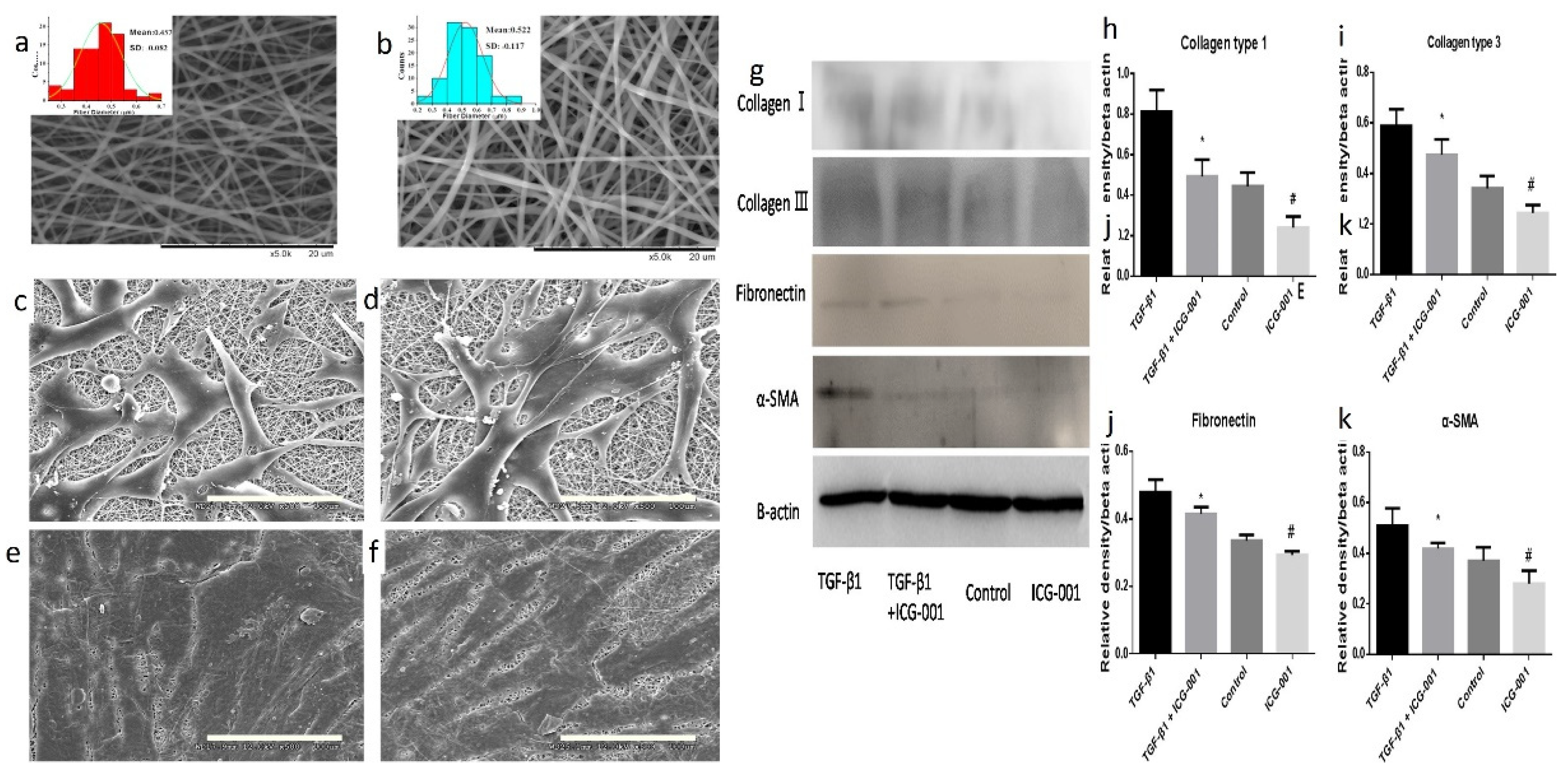
- Zhang, K.; Guo, X.; Zhao, W.; Niu, G.; Mo, X.; Fu, Q. Application of Wnt Pathway Inhibitor Delivering Scaffold for Inhibiting Fibrosis in Urethra Strictures: In Vitro and in Vivo Study. Int. J. Mol. Sci. 2015, 16, 27659–27676. [Google Scholar] [CrossRef] [PubMed]
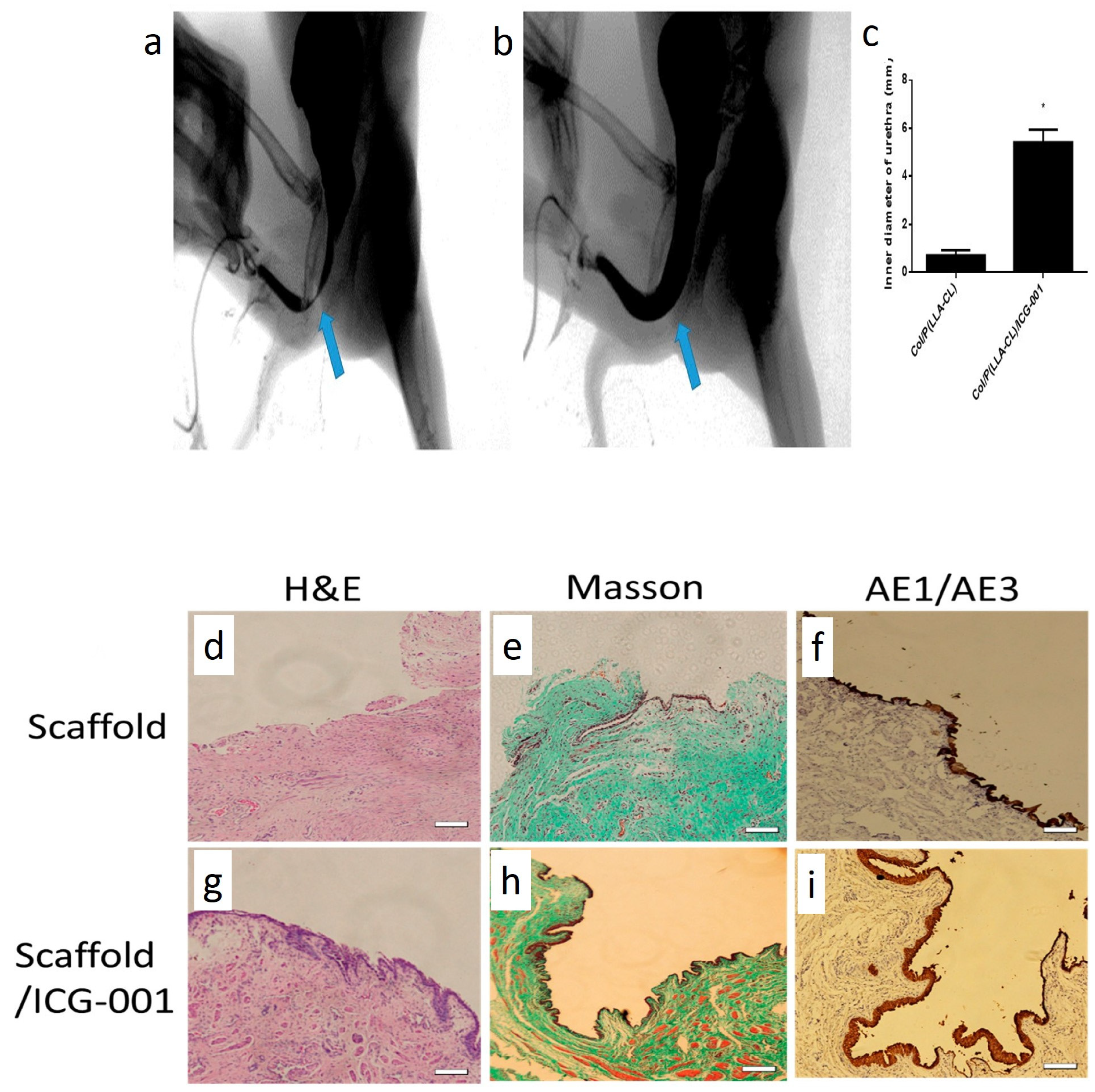
- Zhang, K.; Fang, X.; Zhu, J.; Yang, R.; Wang, Y.; Zhao, W.; Mo, X.; Fu, Q. Effective Reconstruction of Functional Urethra Promoted With ICG-001 Delivery Using Core-Shell Collagen/Poly(Llactide-co-caprolactone) [P(LLA-CL)] Nanoyarn-Based Scaffold: A Study in Dog Model. Front. Bioeng. Biotechnol. 2020, 8, 774. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Zhang, K.; El-Aassar, M.; Wang, N.; El-Hamshary, H.; El-Newehy, M.; Fu, Q.; Mo, X. The Comparison of the Wnt Signaling Pathway Inhibitor Delivered Electrospun Nanoyarn Fabricated with Two Methods for the Application of Urethroplasty. Front. Mater. Sci. 2016, 10, 346–357. [Google Scholar] [CrossRef]
- Hirakawa, T.; Nasu, K.; Miyabe, S.; Kouji, H.; Katoh, A.; Uemura, N.; Narahara, H. β-Catenin Signaling Inhibitors ICG-001 and C-82 Improve Fibrosis in Preclinical Models of Endometriosis. Sci. Rep. 2019, 9, 20056. [Google Scholar] [CrossRef]
- Chung, J.Y.-F.; Chan, M.K.-K.; Li, J.S.; Chan, A.; Tang, P.; Leung, K.-T.; To, K.-F.; Lan, H.-Y.; Tang, P. TGF-β Signaling: From Tissue Fibrosis to Tumor Microenvironment. Int. J. Mol. Sci. 2021, 22, 7575. [Google Scholar] [CrossRef]
- Tsauo, J.; Song, H.Y.; Choi, E.Y.; Kim, D.K.; Kim, K.Y.; Park, J.H.; Yoon, S.H.; Lim, Y.J. EW-7197, An Oral Transforming Growth Factor Β Type I Receptor Kinase Inhibitor, for Preventing Peritoneal Adhesion Formation in a Rat Model. Surgery 2018, 164, 1100–1108. [Google Scholar] [CrossRef]
- Kim, J.H.; Song, H.-Y.; Park, J.-H.; Yoon, H.-J.; Park, H.G.; Kim, D.-K. IN-1233, an ALK-5 Inhibitor: Prevention of Granulation Tissue Formation after Bare Metallic Stent Placement in a Rat Urethral Model 1. Radiology 2010, 255, 75–82. [Google Scholar] [CrossRef]
- Lang, Q.; Liu, Q.; Xu, N.; Qian, K.-L.; Qi, J.-H.; Sun, Y.-C.; Xiao, L.; Shi, X.-F. The Antifibrotic Effects Of TGF-Β1 Sirna on Hepatic Fibrosis in Rats. Biochem. Biophys. Res. Commun. 2011, 409, 448–453. [Google Scholar] [CrossRef]
- Xu, C.; Liu, Y.; Li, C.; Zhou, W.; Huang, S.; Le, W.; Hong, Z.; Wu, D. Urethral Reconstruction with RNA Interference and Polycaprolactone/Silk Fibroin/Collagen Electrospun Fiber in Rabbits. J. Biomater. Tissue Eng. 2018, 7, 858–862. [Google Scholar] [CrossRef]
- Alshaer, W.; Zureigat, H.; Al Karaki, A.; Al-Kadash, A.; Gharaibeh, L.; Ma’mon, M.H.; Aljabali, A.A.; Awidi, A. Sirna: Mechanism of Action, Challenges, and Therapeutic Approaches. Eur. J. Pharmacol. 2021, 905, 174178. [Google Scholar] [CrossRef] [PubMed]
- Kitaori, T.; Ito, H.; Schwarz, E.M.; Tsutsumi, R.; Yoshitomi, H.; Oishi, S.; Nagasawa, T.; Nakamura, T. Stromal Cell–Derived Factor 1/CXCR4 Signaling Is Critical for the Recruitment of Mesenchymal Stem Cells to The Fracture Site During Skeletal Repair in a Mouse Model. Arthritis Rheum. Off. J. Am. Coll. Rheumatol. 2009, 60, 813–823. [Google Scholar] [CrossRef] [PubMed]
- Xiao Ling, K.; Peng, L.; Jian Feng, Z.; Wei, C.; Wei Yan, Y.; Nan, S.; Cheng Qi, G.; Zhi Wei, W. Stromal Derived Factor-1/CXCR4 Axis Involved in Bone Marrow Mesenchymal Stem Cells Recruitment to Injured Liver. Stem Cells Int. 2016, 206–215. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, L.; Yuan, W.; Zhang, D.; Gu, Y.; Huang, J.; Murphy, S.; Ali, M.; Zhang, Y.; Song, L. Sustained Release of Stromal Cell–Derived Factor-1 Alpha from Silk Fibroin Microfiber Promotes Urethral Reconstruction in Rabbits. J. Biomed. Mater. Res. Part A 2020, 108, 1760–1773. [Google Scholar] [CrossRef]
- Horzum, N.; Demir, M.M.; Muñoz-Espí, R.; Crespy, D. (Eds.) Green Electrospinning; De Gruyter: Berlin, Germany, 2019; Volume 1, pp. 1–11. [Google Scholar]
- Soares, R.M.; Siqueira, N.M.; Prabhakaram, M.P.; Ramakrishna, S. Electrospinning and Electrospray of Bio-Based and Natural Polymers for Biomaterials Development. Mater. Sci. Eng. C 2018, 92, 969–982. [Google Scholar] [CrossRef]
- Oryan, A.; Kamali, A.; Moshiri, A.; Baharvand, H.; Daemi, H. Chemical Crosslinking of Biopolymeric Scaffolds: Current Knowledge and Future Directions of Crosslinked Engineered Bone Scaffolds. Int. J. Biol. Macromol. 2018, 107, 678–688. [Google Scholar] [CrossRef]
- Bose, S.; Koski, C.; Vu, A.A. Additive Manufacturing of Natural Biopolymers and Composites for Bone Tissue Engineering. Mater. Horiz. 2020, 7, 2011–2027. [Google Scholar] [CrossRef]
- Lyu, X.; Hu, M.; Peng, J.; Zhang, X.; Sanders, Y.Y. HDAC Inhibitors as Antifibrotic Drugs in Cardiac and Pulmonary Fibrosis. Ther. Adv. Chronic Dis. 2019, 10, 2040622319862697. [Google Scholar] [CrossRef]
- Dai, Z.; Ronholm, J.; Tian, Y.; Sethi, B.; Cao, X. Sterilization Techniques for Biodegradable Scaffolds in Tissue Engineering Applications. J. Tissue Eng. 2016, 7, 2041731416648810. [Google Scholar] [CrossRef]
- Rediguieri, C.F.; Sassonia, R.C.; Dua, K.; Kikuchi, I.S.; Pinto, T.D.J.A. Impact of Sterilization Methods on Electrospun Scaffolds for Tissue Engineering. Eur. Polym. J. 2016, 82, 181–195. [Google Scholar] [CrossRef]
- Ahmed, F.E.; Lalia, B.S.; Hashaikeh, R. A Review on Electrospinning for Membrane Fabrication: Challenges and Applications. Desalination 2015, 356, 15–30. [Google Scholar] [CrossRef]
- Akhmetova, A.; Heinz, A. Electrospinning Proteins for Wound Healing Purposes: Opportunities and Challenges. Pharmaceutics 2020, 13, 4. [Google Scholar] [CrossRef] [PubMed]
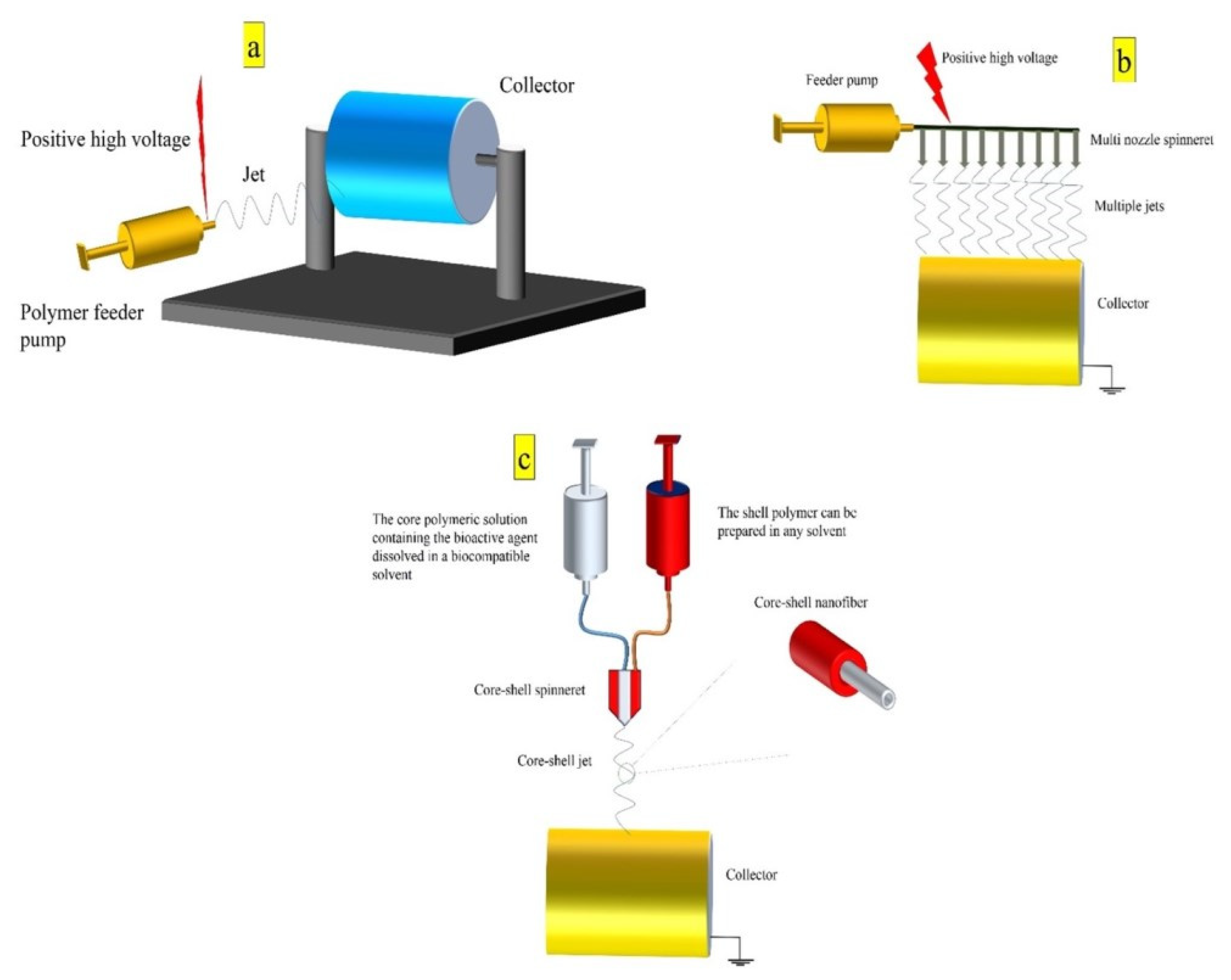
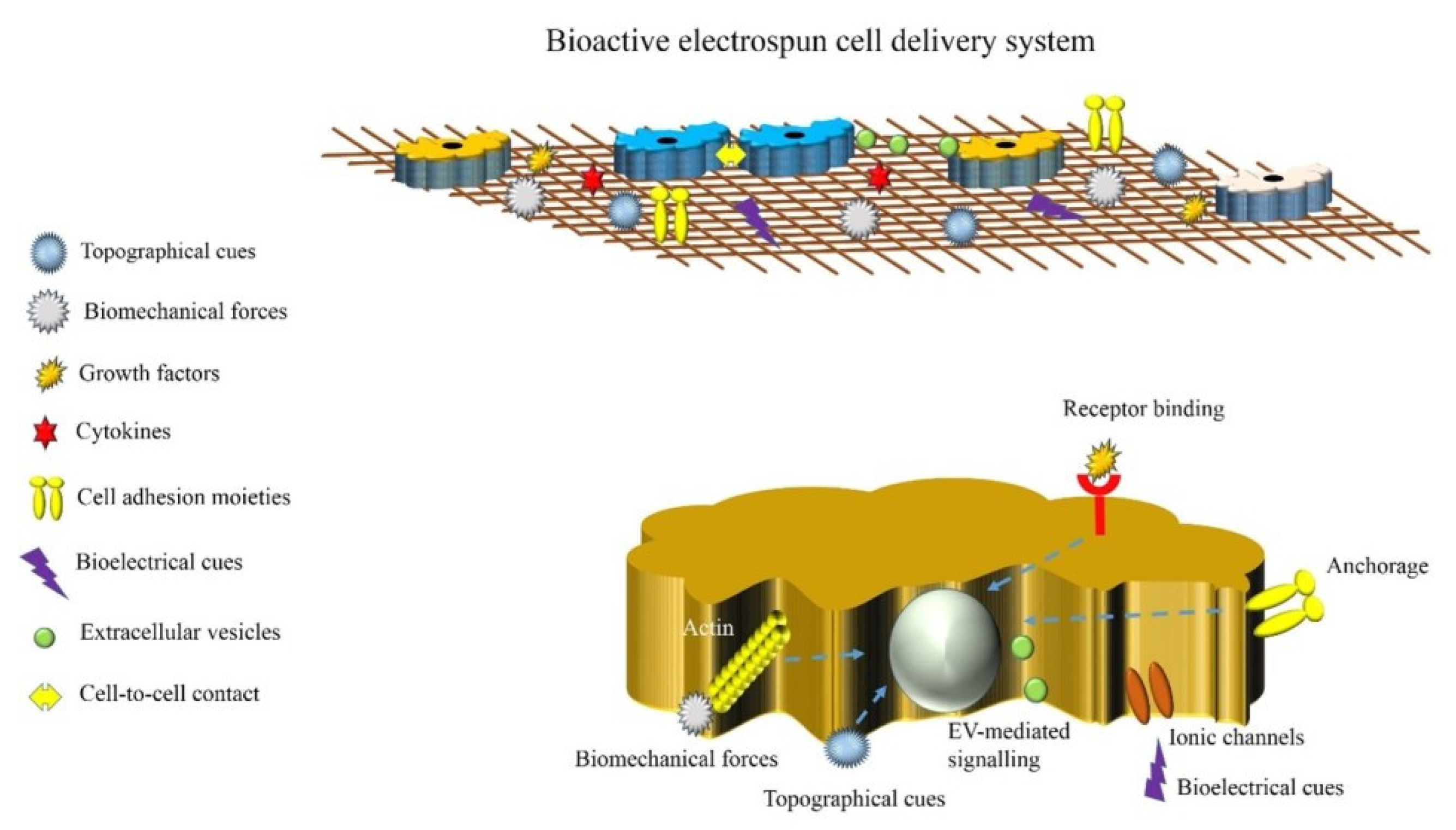
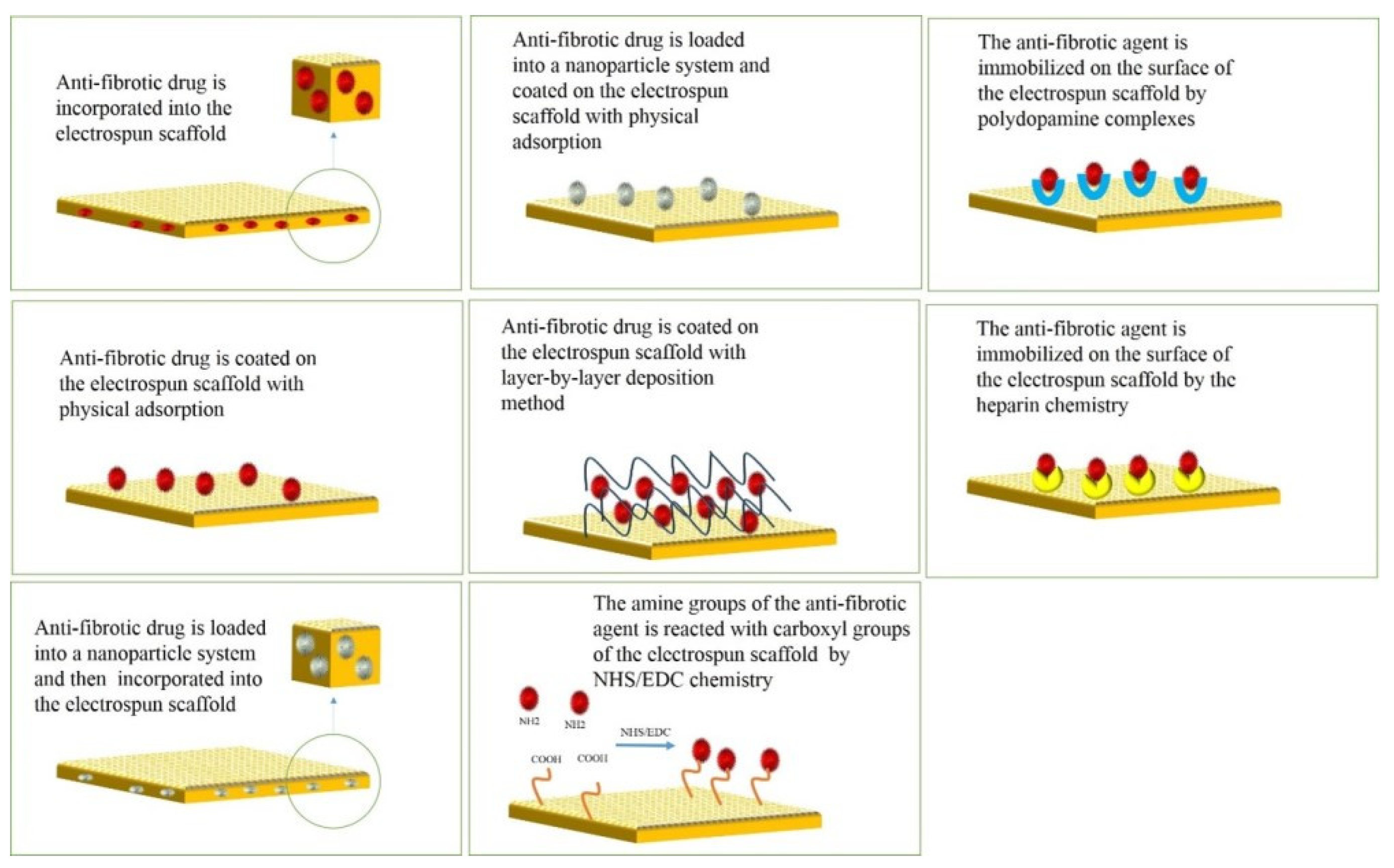

| Parameter | Effects | References |
|---|---|---|
| The molecular mass of the polymer | Polymers with low molecular mass tend to form beads, while higher molecular mass results in fibers with a more uniform structure | [161] |
| Viscosity | The higher molecular mass and polymer concentration increase the viscosity of the polymeric solution. As a result, viscose solutions tend to produce thicker fibers | [162] |
| Surface tension | Increasing the polymer concentration reduces the surface tension and leads to fibers with a continuous and uniform structure. Changing the solvent system or adding surfactant can also alter surface tension | [163] |
| Conductivity | The polymeric solution’s conductivity affects the fibers’ mean fiber size and morphology. Incorporation of salts or polyelectrolytes can improve conductivity | [164] |
| Applied voltage | High voltages decrease the average fiber diameter. Furthermore, high voltages stretch the fibers and align the polymeric chains, increasing fibers’ crystallinity | [165] |
| Solution flow rate | The average fiber diameter will increase with higher flow rates and vice versa | [166] |
| Needle to collector distance | The increase in the fiber receiving distance to a certain extent will produce ultrafine fibers. Therefore, the distance should be optimized for every polymeric solution; otherwise, disintegrated or beady fibers will be produced | [167] |
| Properties of the receiver | The architecture and morphology of the fibers can be determined by using different collectors. Increasing the collector’s rotation rate increases the alignment of fibers. In addition, 3D electrospun matrices can be produced when fibers are spun into a liquid coagulation bath | [168,169] |
| Humidity | Humidity can alter the solvent’s humidity and cause fibers’ fusion | [170] |
| Temperature | Temperature can alter viscosity, solvents’ volatility, and surface tension. In addition, high-temperature results in rapid solvent evaporation and reduces the flying time, thereby increasing fibers’ diameter | [171] |
| Air pressure | The air pressure affects the solvent’s volatility and jet stability. However, fibers with a uniform structure and consistency can be produced when spun at a constant air pressure | [172] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farzamfar, S.; Elia, E.; Chabaud, S.; Naji, M.; Bolduc, S. Prospects and Challenges of Electrospun Cell and Drug Delivery Vehicles to Correct Urethral Stricture. Int. J. Mol. Sci. 2022, 23, 10519. https://doi.org/10.3390/ijms231810519
Farzamfar S, Elia E, Chabaud S, Naji M, Bolduc S. Prospects and Challenges of Electrospun Cell and Drug Delivery Vehicles to Correct Urethral Stricture. International Journal of Molecular Sciences. 2022; 23(18):10519. https://doi.org/10.3390/ijms231810519
Chicago/Turabian StyleFarzamfar, Saeed, Elissa Elia, Stéphane Chabaud, Mohammad Naji, and Stéphane Bolduc. 2022. "Prospects and Challenges of Electrospun Cell and Drug Delivery Vehicles to Correct Urethral Stricture" International Journal of Molecular Sciences 23, no. 18: 10519. https://doi.org/10.3390/ijms231810519
APA StyleFarzamfar, S., Elia, E., Chabaud, S., Naji, M., & Bolduc, S. (2022). Prospects and Challenges of Electrospun Cell and Drug Delivery Vehicles to Correct Urethral Stricture. International Journal of Molecular Sciences, 23(18), 10519. https://doi.org/10.3390/ijms231810519






