The Toxicity, Sublethal Effects, and Biochemical Mechanism of β-Asarone, a Potential Plant-Derived Insecticide, against Bemisia tabaci
Abstract
:1. Introduction
2. Results
2.1. Baseline Susceptibility to β-Asarone and Cross Resistance Pattern
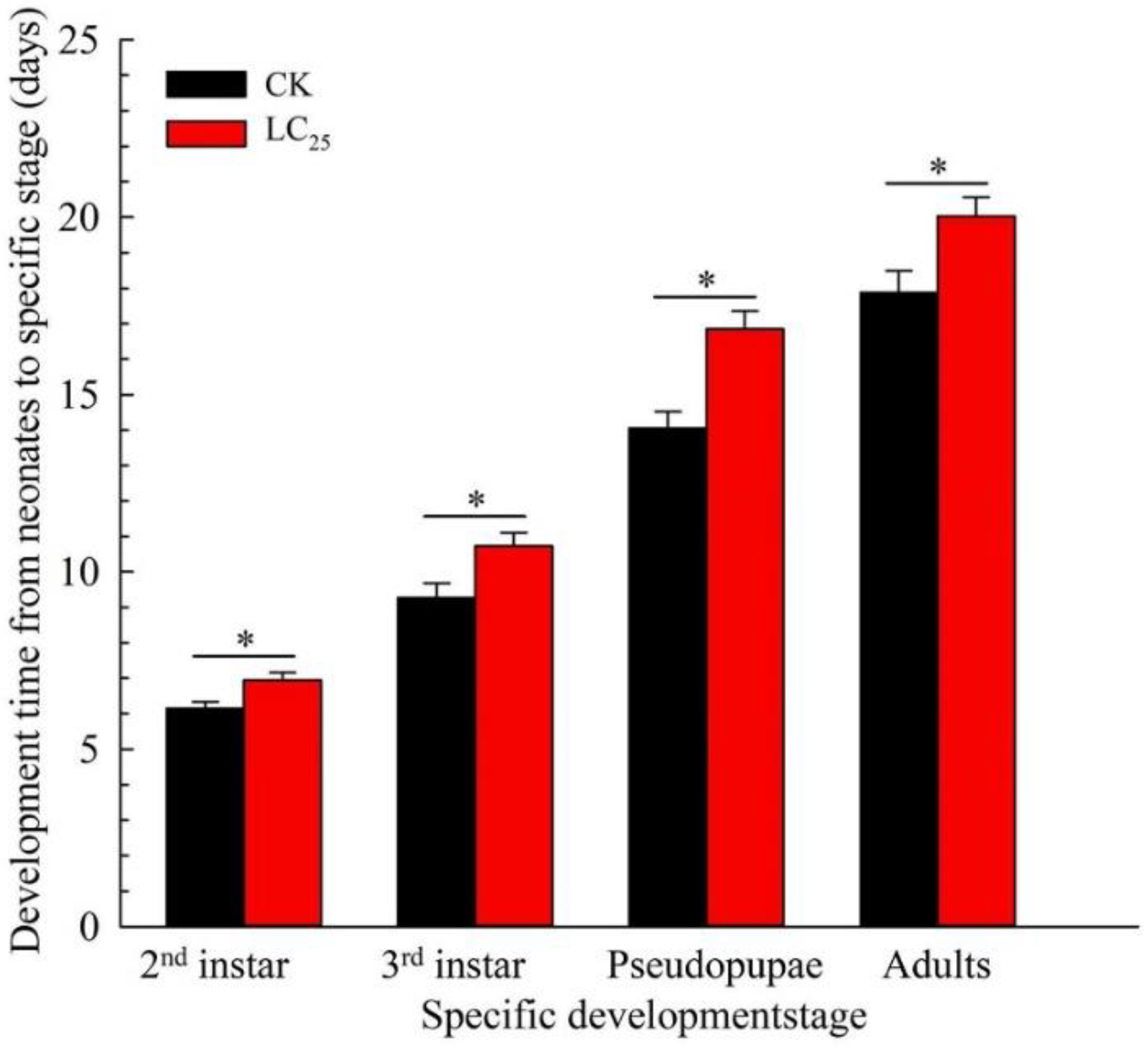
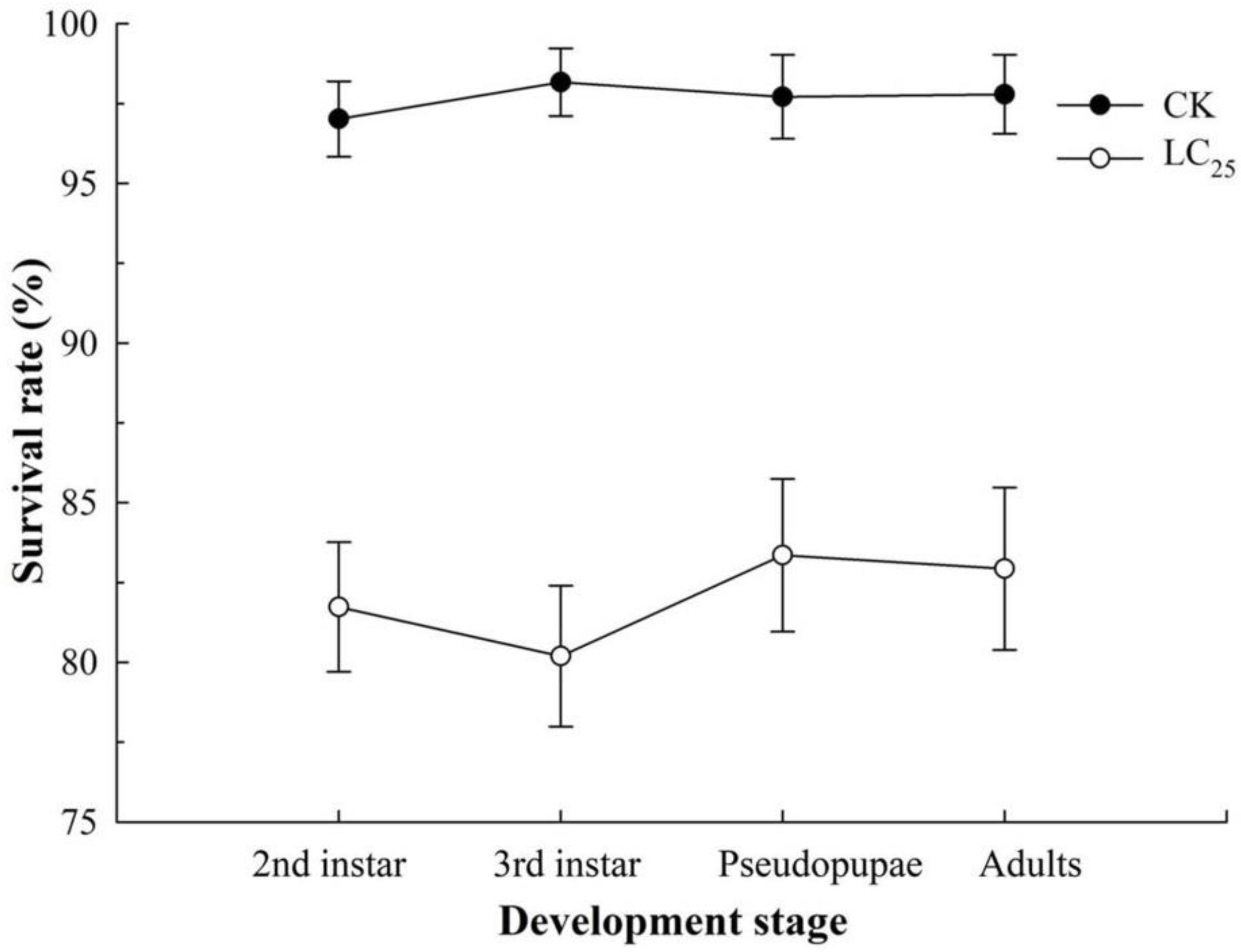
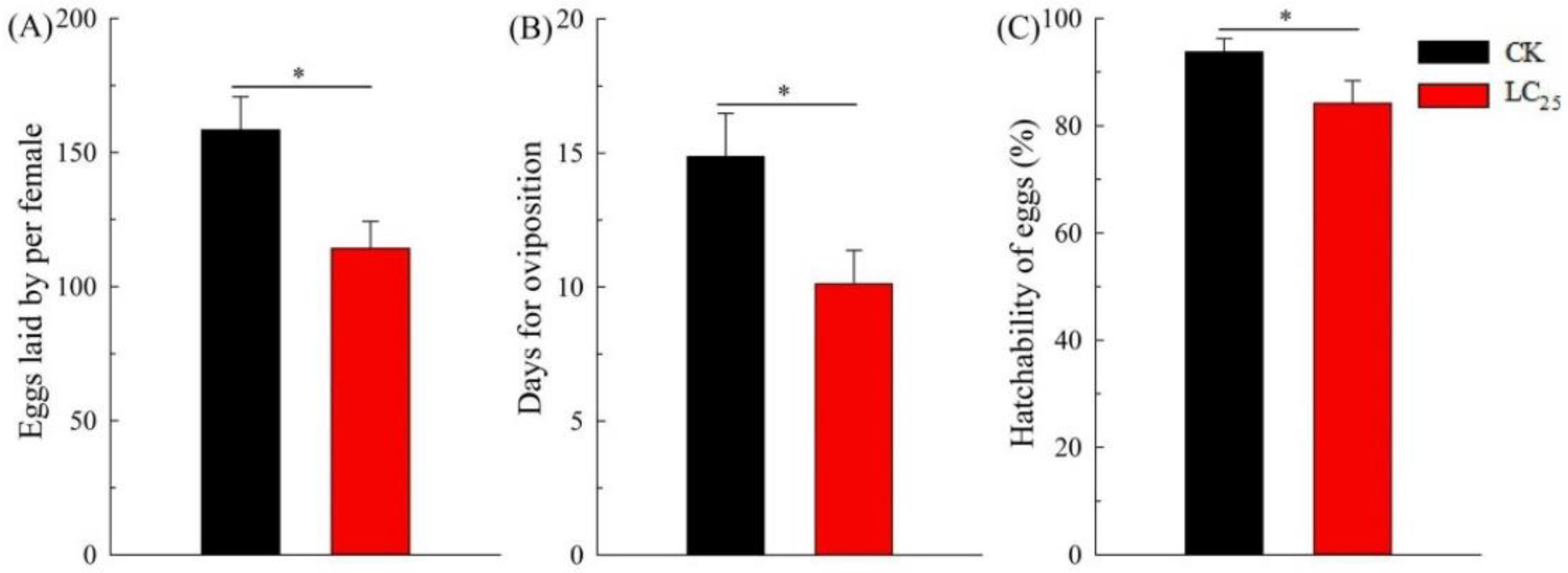
2.2. Sublethal Effects of β-Asarone on B. tabaci
2.3. Biochemical Mechanism of Response to LC25 of β-Asarone
3. Discussion
4. Materials and Methods
4.1. Insects
4.2. Insecticides and Chemicals
4.3. Toxicity of β-Asarone on B. tabaci
4.4. Sublethal Effects of β-Asarone on B. tabaci
4.5. Metabolic Enzyme Assays and Expression Patterns of Detoxification Related to P450s
4.6. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, X.W.; Li, P.; Liu, S.S. Whitefly interactions with plants. Curr. Opin. Insect Sci. 2017, 19, 70–75. [Google Scholar] [CrossRef]
- Horowitz, A.R.; Ghanim, M.; Roditakis, E.; Nauen, R.; Ishaaya, I. Insecticide resistance and its management in Bemisia tabaci species. J. Pest Sci. 2020, 93, 893–910. [Google Scholar] [CrossRef]
- Wei, J.; He, Y.Z.; Guo, Q.; Guo, T.; Liu, Y.Q.; Zhou, X.P.; Liu, S.S.; Wang, X.W. Vector development and vitellogenin determine the transovarial transmission of begomoviruses. Proc. Natl. Acad. Sci. USA 2017, 114, 6746–6751. [Google Scholar] [CrossRef]
- Bass, C.; Denholm, I.; Williamson, M.S.; Nauen, R. The global status of insect resistance to neonicotinoid insecticides. Pestic. Biochem. Physiol. 2015, 121, 78–87. [Google Scholar] [CrossRef]
- Zheng, H.X.; Xie, W.; Wang, S.L.; Wu, Q.J.; Zhou, X.M.; Zhang, Y.J. Dynamic monitoring (B versus Q) and further resistance status of Q type Bemisia tabaci in China. Crop Prot. 2017, 94, 115–121. [Google Scholar] [CrossRef]
- Wang, S.L.; Zhang, Y.J.; Yang, X.; Xie, W.; Wu, Q.J. Resistance monitoring for eight insecticides on the sweet potato whitefly Bemisia tabaci in China. J. Econ. Entomol. 2017, 110, 660–666. [Google Scholar] [CrossRef]
- Wang, R.; Fang, Y.; Mu, C.Q.; Qu, C.; Li, F.Q.; Wang, Z.Y.; Luo, C. Baseline susceptibility and cross-resistance of cycloxaprid, a novel cis-nitromethylene neonicotinoid insecticide, in Bemisia tabaci MED from China. Crop Prot. 2018, 110, 283–287. [Google Scholar] [CrossRef]
- Wang, R.; Wang, J.D.; Che, W.N.; Sun, Y.; Li, W.X.; Luo, C. Characterization of field-evolved resistance to cyantraniliprole in Bemisia tabaci MED from China. J. Integr. Agric. 2019, 18, 2571–2578. [Google Scholar] [CrossRef]
- Wang, R.; Wang, J.D.; Che, W.N.; Fang, Y.; Luo, C. Baseline susceptibility and biochemical mechanism of resistance to flupyradifurone in Bemisia tabaci. Crop Prot. 2020, 132, 105132. [Google Scholar] [CrossRef]
- Wang, R.; Fang, Y.; Che, W.; Zhang, Q.; Wang, J.; Luo, C. Metabolic resistance in abamectin-resistant Bemisia tabaci Mediterranean from northern China. Toxins 2022, 14, 424. [Google Scholar] [CrossRef]
- Adfa, M.; Livandri, F.; Meita, N.P.; Manaf, S.; Ninomiya, M.; Gustian, I.; Putranto, A.M.H.; Supriati, R.; Koketsu, M. Termiticidal activity of Acorus calamus Linn. rhizomes and its main constituents against Coptotermes curvignathus Holmgren. J. Asia Pac. Entomol. 2015, 18, 47–50. [Google Scholar] [CrossRef]
- Imai, T.; Masuda, R. Insecticidal activities of methyleugenol and β-asarone, from the herbal medicines Saishin and Sekishōkon, and other alkoxy-propenyl-benzene derivatives against the cigarette beetle Lasioderma serricorne (Coleoptera: Anobiidae). Appl. Entomol. Zool. 2016, 52, 183–188. [Google Scholar] [CrossRef]
- Arasu, M.V.; Viayaraghavan, P.; Ilavenil, S.; Al-Dhabi, N.A.; Choi, K.C. Essential oil of four medicinal plants and protective properties in plum fruits against the spoilage bacteria and fungi. Ind. Crop. Prod. 2019, 133, 54–62. [Google Scholar] [CrossRef]
- Chen, H.P.; Yang, K.; Zheng, L.S.; You, C.X.; Cai, Q.; Wang, C.F. Repellant and insecticidal activities of shyobunone and isoshyobunone derived from the essential oil of Acorus calamus rhizomes. Pharmacogn. Mag. 2015, 11, 675–681. [Google Scholar] [PubMed]
- Kumar, R.; Sharma, S.; Sharma, S.; Kumari, A.; Kumar, D.; Nadda, G.; Padwad, Y.; Ogra, R.K.; Kumar, N. Chemical composition, cytotoxicity and insecticidal activities of Acorus calamus accessions from the western Himalayas. Ind. Crop. Prod. 2016, 94, 520–527. [Google Scholar] [CrossRef]
- Yao, Y.J.; Cai, W.L.; Yang, C.J.; Hua, H.X. Supercritical fluid CO2 extraction of Acorus calamus L. (Arales: Araceae) and its contact toxicity to Sitophilus zeamais Motschusky (Coleoptera: Curculionidae). Nat. Prod. Res. 2012, 26, 1498–1503. [Google Scholar] [CrossRef] [PubMed]
- Yooboon, T.; Kuramitsu, K.; Bullangpoti, V.; Kainoh, Y.; Furukawa, S. Cytotoxic effects of β-asarone on Sf9 insect cells. Arch. Insect Biochem. Physiol. 2019, 102, e21596. [Google Scholar] [CrossRef] [PubMed]
- Isman, M.B. Botanical insecticides, deterrents, and repellents in modern agriculture and an increasingly regulated world. Annu. Rev. Entomol. 2006, 51, 45–66. [Google Scholar] [CrossRef]
- Velasques, J.; Cardoso, M.H.; Abrantes, G.; Frihling, B.E.; Franco, O.L.; Migliolo, L. The rescue of botanical insecticides: A bioinspiration for new niches and needs. Pestic. Biochem. Physiol. 2017, 143, 14–25. [Google Scholar] [CrossRef]
- Chowański, S.; Chudzińska, E.; Lelario, F.; Ventrella, E.; Marciniak, P.; Miądowicz-Kobielska, M.; Spochacz, M.; Szymczak, M.; Scrano, L.; Bufo, S.A.; et al. Insecticidal properties of Solanum nigrum and Armoracia rusticana extracts on reproduction and development of Drosophila melanogaster. Ecotoxicol. Environ. Saf. 2018, 162, 454–463. [Google Scholar] [CrossRef]
- Campos, E.V.R.; Proença, P.L.F.; Oliveira, J.L.; Melville, C.C.; Della Vechia, J.F.; de Andrade, D.J.; Fraceto, L.F. Chitosan nanoparticles functionalized with β-cyclodextrin: A promising carrier for botanical pesticides. Sci. Rep. 2018, 8, 2067. [Google Scholar] [CrossRef]
- Fang, Y.; Wang, J.D.; Luo, C.; Wang, R. Lethal and sublethal effects of clothianidin on the development and reproduction of Bemisia tabaci (Hemiptera: Aleyrodidae) MED and MEAM1. J. Insect Sci. 2018, 18, 37. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Zhang, W.; Che, W.N.; Qu, C.; Li, F.Q.; Desneux, N.; Luo, C. Lethal and sublethal effects of cyantraniliprole, a new anthranilic diamide insecticide, on Bemisia tabaci (Hemiptera: Aleyrodidae) MED. Crop Prot. 2017, 91, 108–113. [Google Scholar] [CrossRef]
- Wang, R.; Zheng, H.X.; Qu, C.; Wang, Z.H.; Kong, Z.Q.; Luo, C. Lethal and sublethal effects of a novel cis-nitromethylene neonicotinoid insecticide, cycloxaprid, on Bemisia tabaci. Crop Prot. 2016, 83, 15–19. [Google Scholar] [CrossRef]
- Qu, C.; Zhang, W.; Li, F.Q.; Tetreau, G.; Luo, C.; Wang, R. Lethal and sublethal effects of dinotefuran on two invasive whiteflies, Bemisia tabaci (Hemiptera: Aleyrodidae). J. Asia-Pac. Entomol. 2017, 20, 325–330. [Google Scholar] [CrossRef]
- Zhou, X.; Zhang, Z.; Zheng, H.; Zhang, Q.; Gong, J.; Li, C.; Wang, R. Physiological and biochemical responses to sublethal concentrations of the novel pyropene insecticide, afidopyropen, in whitefly Bemisia tabaci MED. Agronomy 2021, 11, 2260. [Google Scholar] [CrossRef]
- Xu, X.; Li, X.; Wen, D.; Zhao, C.; Fan, L.; Wu, C.; Liu, Z.; Han, K.; Zhao, M.; Zhang, S.; et al. Sublethal and transgenerational effects of a potential plant-derived insecticide, β-asarone, on population fitness of brown planthopper, Nilaparvata lugens. Entomol. Exp. Appl. 2022, 170, 555–564. [Google Scholar] [CrossRef]
- Desneux, N.; Decourtye, A.; Delpuech, J.M. The sublethal effects of pesticides on beneficial arthropods. Annu. Rev. Entomol. 2007, 52, 81–106. [Google Scholar] [CrossRef]
- Saxena, B.P.; Koul, O.; Tikku, K.; Atal, C.K. A new insect chemosterilant isolated from Acorus calamus L. Nature 1977, 270, 512–513. [Google Scholar] [CrossRef]
- Perrett, S.; Whitfield, P.J. Anthelmintic and pesticidal activity of Acorus gramineus (Araceae) is associated with phenylpropanoid asarones. Phytother. Res. 1995, 9, 405–409. [Google Scholar] [CrossRef]
- Müller, T.; Römer, C.I.; Müller, C. Parental sublethal insecticide exposure prolongs mating response and decreases reproductive output in offspring. J. Appl. Ecol. 2019, 56, 1528–1537. [Google Scholar] [CrossRef]
- Piri, A.; Sahebzadeh, N.; Zibaee, A.; Sendi, J.J.; Shamakhi, L.; Shahriari, M. Toxicity and physiological effects of ajwain (Carum copticum, Apiaceae) essential oil and its major constituents against Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). Chemosphere 2020, 256, 127103. [Google Scholar] [CrossRef] [PubMed]
- Toledo, P.F.S.; Ferreira, T.P.; Bastos, I.M.A.S.; Rezende, S.M.; Jumbo, L.O.V.; Didonet, J.; Andrade, B.S.; Melo, T.S.; Smagghe, G.; Oliveira, E.E.; et al. Essential oil from Negramina (Siparuna guianensis) plants controls aphids without impairing survival and predatory abilities of non-target ladybeetles. Environ. Pollut. 2020, 255, 113153. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Zhang, Q.; Zhou, X.; Zhang, M.; Yang, Q.; Su, Q.; Luo, C. Characterization of field-evolved resistance to afidopyropen, a novel insecticidal toxin developed from microbial secondary metabolites, in Bemisia tabaci. Toxins 2022, 14, 453. [Google Scholar] [CrossRef]
- Wang, R.; Wang, J.D.; Zhang, J.S.; Che, W.N.; Luo, C. Characterization of flupyradifurone resistance in the whitefly Bemisia tabaci Mediterranean (Q biotype). Pest Manag. Sci. 2020, 76, 4286–4292. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Che, W.; Qu, C.; Wang, J.; Luo, C. Identification and detection of CYP4G68 overexpression associated with cyantraniliprole resistance in Bemisia tabaci from China. Front. Environ. Sci. 2022, 10, 914636. [Google Scholar] [CrossRef]
- Wang, Q.; Wang, M.; Jia, Z.; Ahmat, T.; Xie, L.; Jiang, W. Resistance to neonicotinoid insecticides and expression changes of eighteen cytochrome P450 genes in field populations of Bemisia tabaci from Xinjiang, China. Entomol. Res. 2020, 50, 205–211. [Google Scholar] [CrossRef]
- Zhou, C.S.; Cao, Q.; Li, G.Z.; Ma, D.Y. Role of Several CytochromeP450s in the Resistance and Cross-Resistance against Imidacloprid and Acetamiprid of Bemisia tabaci (Hemiptera: Aleyrodidae) MEAM1 Cryptic Species in Xinjiang, China. Pestic. Biochem. Physiol. 2020, 163, 209–215. [Google Scholar] [CrossRef]
- Xu, X.; Li, X.; Wang, F.; Han, K.; Liu, Z.; Fan, L.; Hua, H.; Cai, W.; Yao, Y. Candidate detoxification-related genes in brown planthopper, Nilaparvata lugens, in response to beta-asarone based on transcriptomic analysis. Ecotox. Environ. Saf. 2019, 185, 109735. [Google Scholar] [CrossRef]
- Xu, X.; Li, X.; Liu, Z.; Wang, F.; Fan, L.; Cai, W.; Yao, Y. Knockdown of CYP301B1 and CYP6AX1v2 increases the susceptibility of the brown planthopper to beta-asarone, a potential plant-derived insecticide. Int. J. Biol. Macromol. 2021, 171, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.P.; Chu, D.; Ge, D.Q.; Wang, S.L.; Wu, Q.J.; Xie, W.; Jiao, X.G.; Liu, B.M.; Yang, X.; Yang, N.N.; et al. Further spread of and domination by Bemisia tabaci (Hemiptera: Aleyrodidae) biotype Q on field crops in China. J. Econ. Entomol. 2011, 104, 978–985. [Google Scholar] [CrossRef]
- Wang, R.; Che, W.; Wang, J.; Qu, C.; Luo, C. Cross-resistance and biochemical mechanism of resistance to cyantraniliprole in a near-isogenic line of whitefly Bemisia tabaci Mediterranean (Q Biotype). Pestic. Biochem. Physiol. 2020, 167, 104590. [Google Scholar] [CrossRef]
- Luo, C.; Yao, Y.; Wang, R.; Yan, F.; Hu, D.; Zhang, Z. The use of mitochondrial cytochrome oxidase mt COI gene sequencesfor the identification of biotypes of Bemisia tabaci (Gennadius) in China. Acta Entomol. Sin. 2002, 45, 759–763. [Google Scholar]
- Liang, J.; Yang, J.; Hu, J.; Fu, B.; Gong, P.; Du, T.; Xue, H.; Wei, X.; Liu, S.; Huang, M.; et al. Cytpchrome P450 CYP4G68 is associated with imidacloprid and thiamethoxam resistance in field whitefly, Bemisia tabaci (Hemiptera: Gennadius). Agriculture 2022, 12, 473. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- LeOra Software. Polo Plus; A user’s guide to probit or logit analysis; LeOra Software: Berkeley, CA, USA, 2002. [Google Scholar]
- SPSS. Release 13.0 Version for Windows; SPSS: Chicago, IL, USA, 2011. [Google Scholar]

| Population | N a | LC50 (95% CL) (mg L−1) b | Slope ± SE | χ2 (df) | RR c |
|---|---|---|---|---|---|
| MED-S | 732 | 15.39 (12.82–18.23) | 1.31 ± 0.12 | 1.78 (3) | |
| LY | 727 | 13.19 (10.50–16.34) | 1.02 ± 0.12 | 1.43 (3) | 0.9 |
| CY | 730 | 17.14 (13.69–23.04) | 1.24 ± 0.13 | 2.35 (3) | 1.1 |
| HD | 723 | 24.36 (20.40–29.06) | 1.27 ± 0.12 | 2.07 (3) | 1.6 |
| TZ | 736 | 31.90 (26.25–37.87) | 1.32 ± 0.12 | 1.40 (3) | 2.1 |
| WQ | 729 | 22.60 (18.52–27.39) | 1.14 ± 0.12 | 1.26 (3) | 1.5 |
| JH | 739 | 35.77 (30.20–42.17) | 1.35 ± 0.12 | 1.90 (3) | 2.3 |
| ZJK | 721 | 29.78 (25.19–34.43) | 1.77 ± 0.14 | 1.64 (3) | 1.9 |
| BD | 735 | 11.78 (9.62–14.36) | 1.11 ± 0.12 | 2.33 (3) | 0.8 |
| ZZ | 718 | 26.10 (21.42–30.88) | 1.44 ± 0.13 | 1.82 (3) | 1.7 |
| XZ | 731 | 42.80 (34.52–56.23) | 1.14 ± 0.12 | 2.89 (3) | 2.8 |
| JN | 727 | 16.06 (13.14–20.34) | 1.12 ± 0.12 | 2.58 (3) | 1.0 |
| TA | 734 | 21.61 (17.00–29.59) | 1.01 ± 0.12 | 0.51 (3) | 1.4 |
| Insecticide | Strain | N a | LC50 (95% CL) (mg/L) b | Slope ± SE | χ2 (df) | RR c |
|---|---|---|---|---|---|---|
| β-Asarone | MED-S | 721 | 15.51 (13.71–17.47) | 2.02 ± 0.14 | 2.32 (3) | |
| HD-Afi | 732 | 12.56 (9.97–15.13) | 1.39 ± 0.13 | 1.64 (3) | 0.8 | |
| CYAN-R | 722 | 14.33 (12.29–16.46) | 1.73 ± 0.14 | 1.92 (3) | 0.9 | |
| FLU-SEL | 724 | 16.74 (13.74–19.72) | 1.62 ± 0.14 | 2.66 (3) | 1.1 | |
| Afidopyropen | MED-S | 725 | 13.13 (10.73–16.11) | 1.10 ± 0.12 | 1.06 (3) | |
| HD-Afi | 737 | 702.38 (595.07–835.88) | 1.33 ± 0.12 | 0.75 (3) | 53.5 | |
| CYAN-R | 711 | 15.06 (12.04–18.22) | 1.21 ± 0.12 | 2.77 (3) | 1.1 | |
| FLU-SEL | 714 | 12.70 (9.58–15.87) | 1.07 ± 0.12 | 2.27 (3) | 1.0 | |
| Cyantraniliprole | MED-S | 715 | 1.57 (1.32–1.88) | 1.29 ± 0.12 | 1.31 (3) | |
| HD-Afi | 721 | 1.85 (1.55–2.15) | 1.72 ± 0.14 | 2.45 (3) | 1.2 | |
| CYAN-R | 719 | 131.08 (107.30–156.33) | 1.29 ± 0.12 | 2.86 (3) | 83.5 | |
| FLU-SEL | 706 | 1.28 (1.06–1.50) | 1.60 ± 0.13 | 0.85 (3) | 0.8 | |
| Flupyradifurone | MED-S | 712 | 15.93 (12.65–19.40) | 1.15 ± 0.12 | 1.25 (3) | |
| HD-Afi | 726 | 14.61 (12.09–17.36) | 1.30 ± 0.12 | 2.49 (3) | 0.9 | |
| CYAN-R | 711 | 12.44 (9.91–15.11) | 1.17 ± 0.12 | 2.05 (3) | 0.8 | |
| FLU-SEL | 721 | 1541.67 (1286.02–1895.20) | 1.25 ± 0.12 | 1.71 (3) | 96.8 |
| Treatment | P450 Activity | EST Activity | GST Activity | |||
|---|---|---|---|---|---|---|
| pmol min−1 mg−1 | Ratio b | nmol min−1 mg−1 | Ratio b | nmol min−1 mg−1 | Ratio b | |
| CK | 0.68 ± 0.16 | 311.06 ± 18.93 | 40.26 ± 10.31 | |||
| LC25 | 1. 32 ± 0.24 * | 1.9 | 329.75 ± 25.32 | 1.1 | 51.07 ± 11.12 | 1.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, R.; Fang, Y.; Che, W.; Zhang, Q.; Wang, J.; Luo, C. The Toxicity, Sublethal Effects, and Biochemical Mechanism of β-Asarone, a Potential Plant-Derived Insecticide, against Bemisia tabaci. Int. J. Mol. Sci. 2022, 23, 10462. https://doi.org/10.3390/ijms231810462
Wang R, Fang Y, Che W, Zhang Q, Wang J, Luo C. The Toxicity, Sublethal Effects, and Biochemical Mechanism of β-Asarone, a Potential Plant-Derived Insecticide, against Bemisia tabaci. International Journal of Molecular Sciences. 2022; 23(18):10462. https://doi.org/10.3390/ijms231810462
Chicago/Turabian StyleWang, Ran, Yong Fang, Wunan Che, Qinghe Zhang, Jinda Wang, and Chen Luo. 2022. "The Toxicity, Sublethal Effects, and Biochemical Mechanism of β-Asarone, a Potential Plant-Derived Insecticide, against Bemisia tabaci" International Journal of Molecular Sciences 23, no. 18: 10462. https://doi.org/10.3390/ijms231810462
APA StyleWang, R., Fang, Y., Che, W., Zhang, Q., Wang, J., & Luo, C. (2022). The Toxicity, Sublethal Effects, and Biochemical Mechanism of β-Asarone, a Potential Plant-Derived Insecticide, against Bemisia tabaci. International Journal of Molecular Sciences, 23(18), 10462. https://doi.org/10.3390/ijms231810462








