GhBOP1 as a Key Factor of Ribosomal Biogenesis: Development of Wrinkled Leaves in Upland Cotton
Abstract
1. Introduction
2. Results
2.1. GhBOP1 Bioinformatics and Expression Analysis
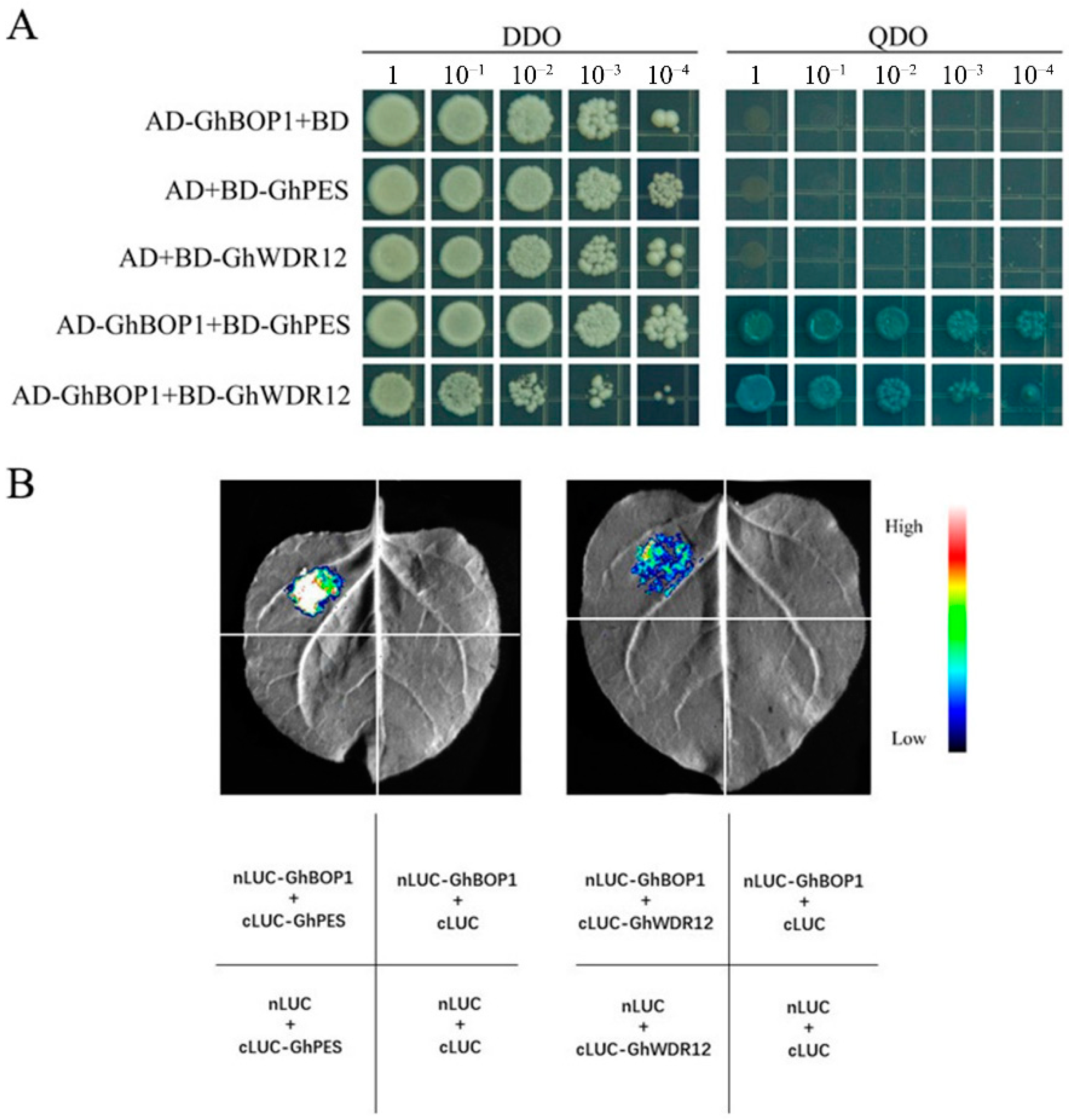
2.2. Interaction of GhBOP1 with GhPES and GhWDR12
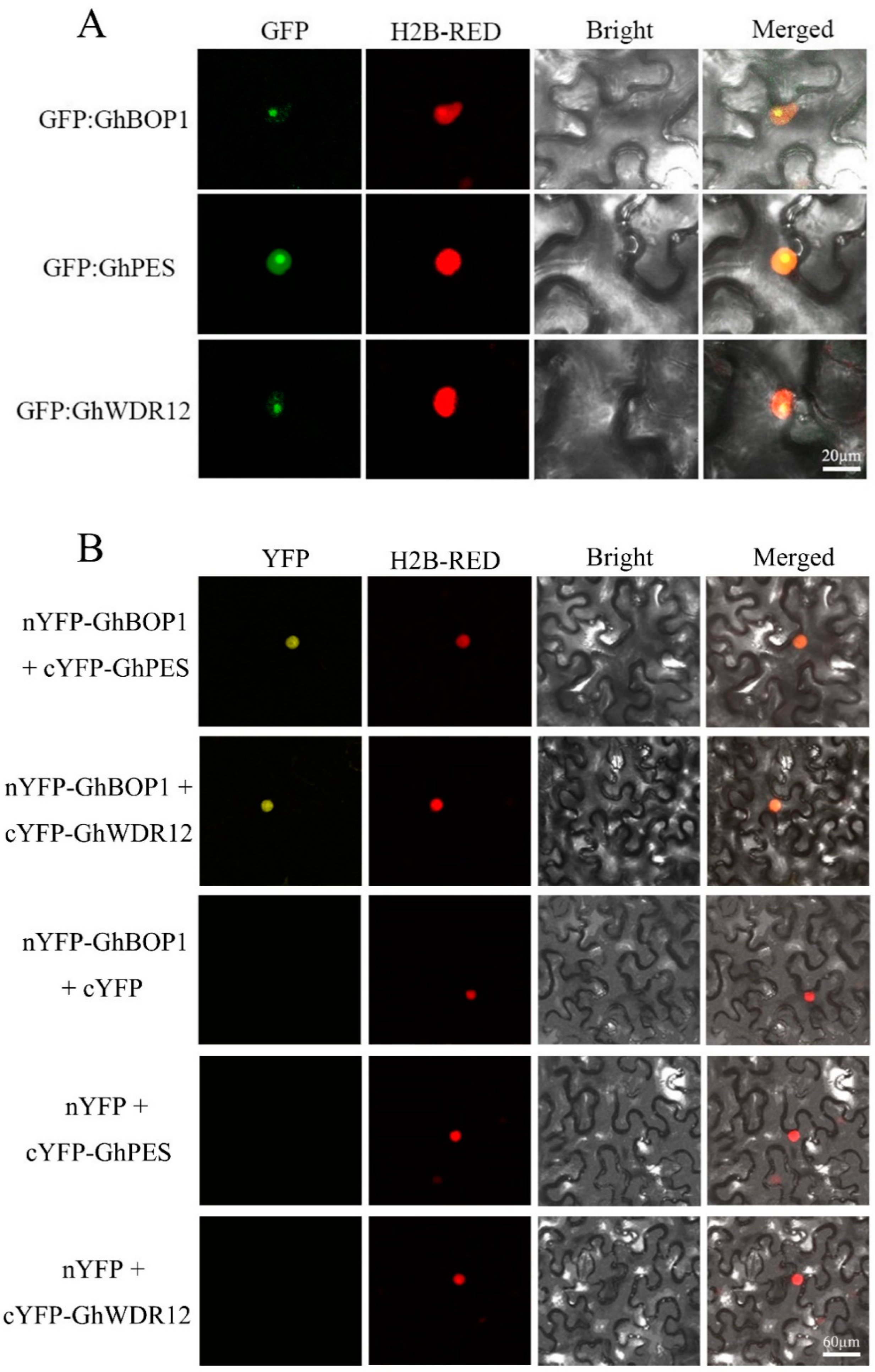
2.3. Subcellular Localization of GhBOP1 and GhPES/GhWDR12
2.4. Phenotype of VIGS in GhBOP1 in Cotton
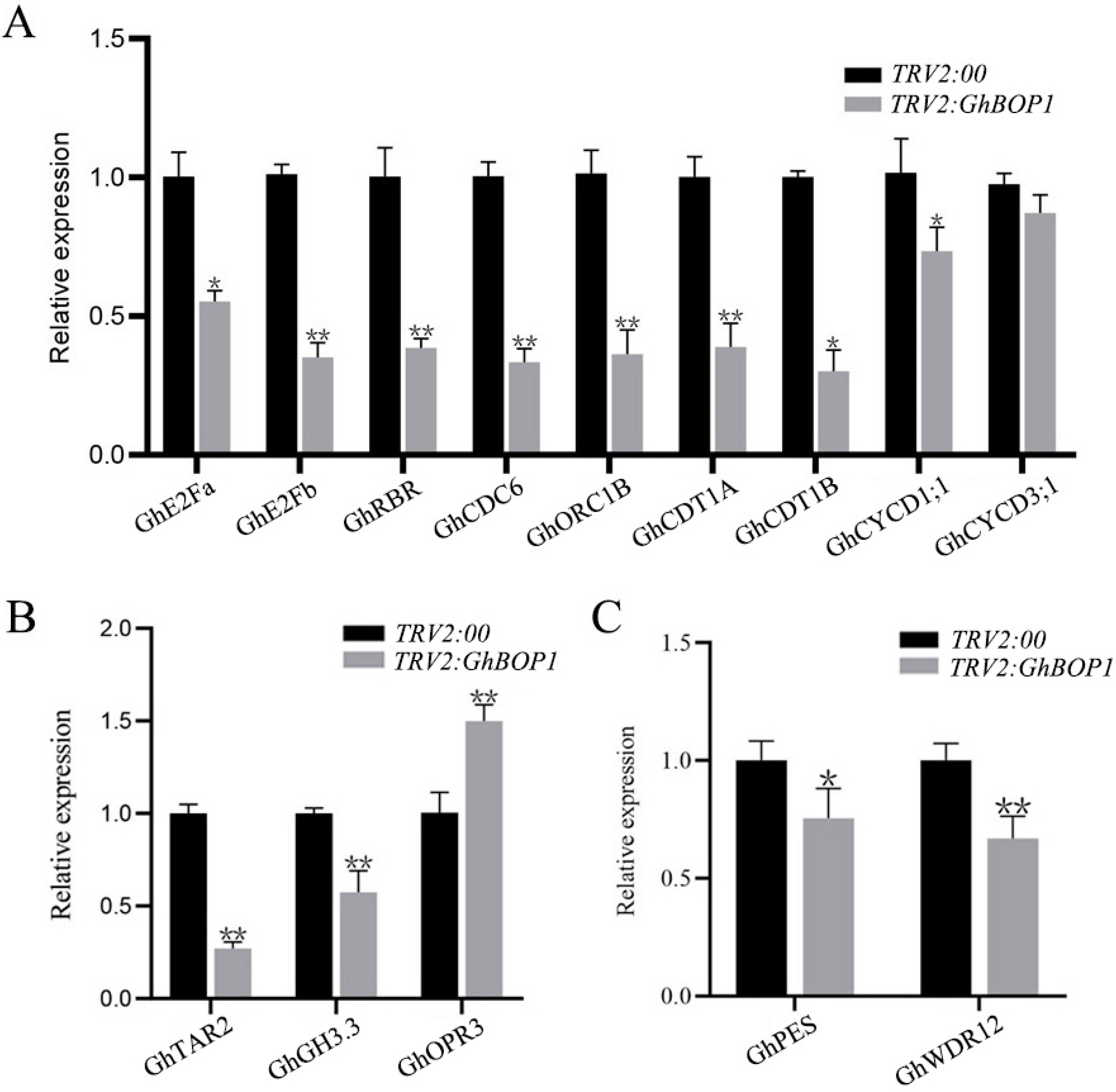
2.5. Expression of Jasmonic Acid, Cell Cycle, and Auxin Related Genes in GhBOP1-Silenced Lines
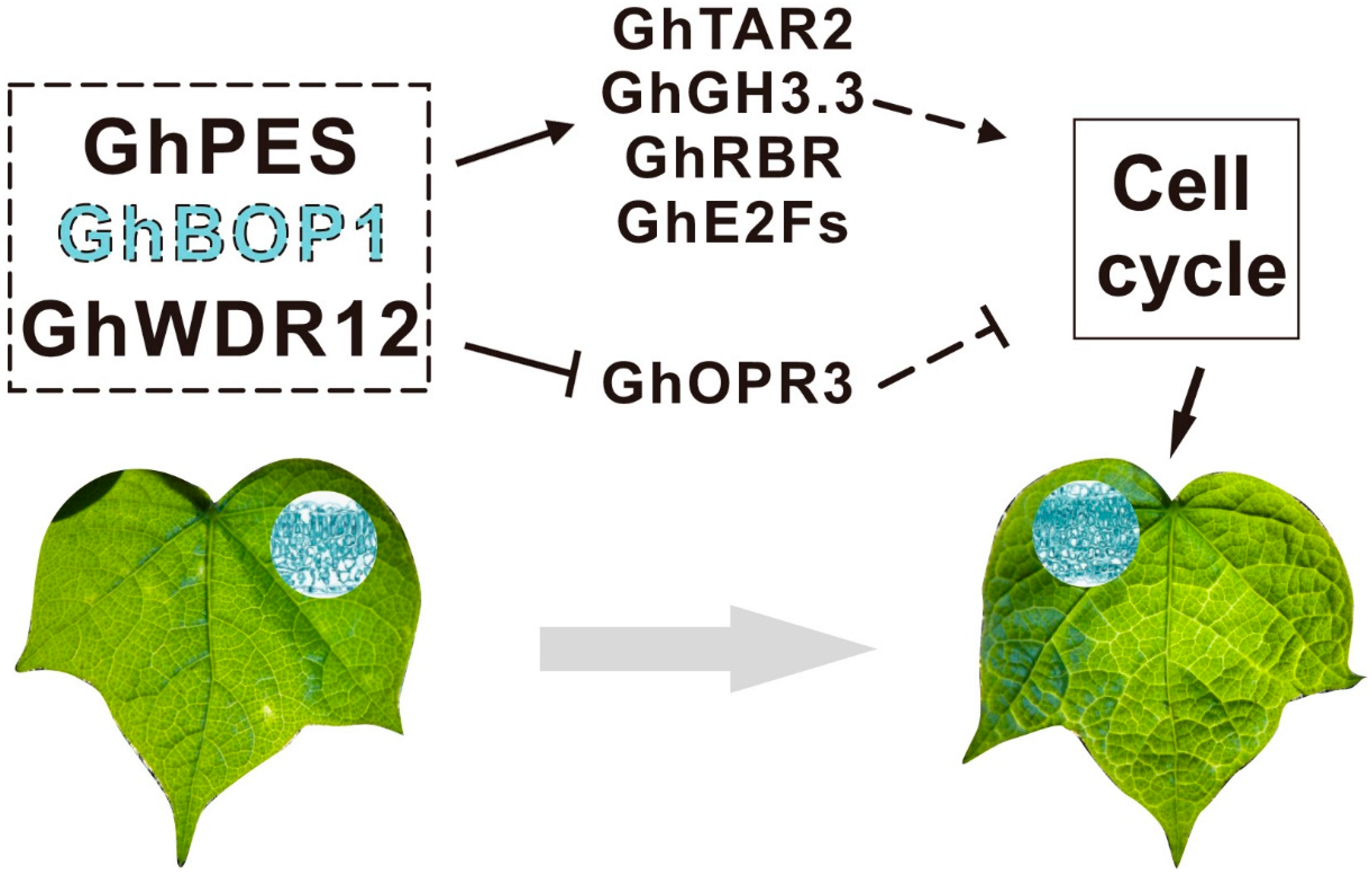
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Growth Conditions
4.2. Bioinformatics Analysis
4.3. Yeast Two-Hybrid Assays
4.4. Split Luciferase Complementation Assay (Split-LUC)
4.5. Bimolecular Fluorescence Complementation (BiFC)
4.6. Subcellular Localization of GhBOP1 and GhWDR12/GhPES
4.7. Virus-Induced Gene Silencing of BOP1 in Cotton and Paraffin Section of Leaf
4.8. Real-Time Quantitative PCR Detecting
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Strezoska, Z.; Pestov, D.G.; Lau, L.F. Bop1 Is a Mouse WD40 Repeat Nucleolar Protein Involved in 28S and 5.8S RRNA Processing and 60S Ribosome Biogenesis. Mol. Cell. Biol. 2000, 20, 5516–5528. [Google Scholar] [CrossRef] [PubMed]
- Degenhardt, R.F.; Bonham-Smith, P.C. Arabidopsis Ribosomal Proteins RPL23aA and RPL23aB Are Differentially Targeted to the Nucleolus and Are Disparately Required for Normal Development. Plant Physiol. 2008, 147, 128–142. [Google Scholar] [CrossRef] [PubMed]
- Tzafrir, I.; Pena-Muralla, R.; Dickerman, A.; Berg, M.; Rogers, R.; Hutchens, S.; Sweeney, T.C.; McElver, J.; Aux, G.; Patton, D.; et al. Identification of Genes Required for Embryo Development in Arabidopsis. Plant Physiol. 2004, 135, 1206–1220. [Google Scholar] [CrossRef] [PubMed]
- Weijers, D.; Dijk, M.F.V.; Vencken, R.J.; Quint, A.; Hooykaas, P.; Offringa, R. An Arabidopsis Minute-like Phenotype Caused by a Semi-dominant Mutation in a Ribosomal Protein S5 Gene. Development 2001, 128, 4289–4299. [Google Scholar] [CrossRef]
- Spence, J.; Gali, R.R.; Dittmar, G.; Sherman, F.; Karin, M.; Finley, D. Cell Cycle–Regulated Modification of the Ribosome by a Variant Multiubiquitin Chain. Cell Press 2000, 102, 67–76. [Google Scholar] [CrossRef]
- Kressler, D.; Hurt, E.; Bassler, J. Driving Ribosome Assembly. Biochim. Biophys. Acta 2010, 1803, 673–683. [Google Scholar] [CrossRef]
- Kressler, D.; Linder, P.; Cruz, J.D. Protein Trans-Acting Factors Involved in Ribosome Biogenesis in Saccharomyces Cerevisiae. Mol. Cell. Biol. 1999, 19, 7897–7912. [Google Scholar] [CrossRef]
- Pestov, D.G.; Stockelman, M.G.; Strezoska, Z.; Lau, L.F. ERB1, the Yeast Homolog of Mammalian Bop1, Is an Essential Gene Required for Maturation of the 25S and 5.8S Ribosomal RNAs. Nucleic Acids Res. 2001, 29, 3621–3630. [Google Scholar] [CrossRef]
- Ahn, C.S.; Cho, H.K.; Lee, D.H.; Sim, H.J.; Kim, S.G.; Pai, H.S. Functional Characterization of the Ribosome Biogenesis Factors PES, BOP1, and WDR12 (PeBoW), and Mechanisms of Defective Cell Growth and Proliferation Caused by PeBoW Deficiency in Arabidopsis. J. Exp. Bot. 2016, 67, 5217–5232. [Google Scholar] [CrossRef]
- Pestov, D.G.; Grzeszkiewicz, T.M.; Lau, L.F. Isolation of Growth Suppressors from a cDNA Expression Library. Oncogene 1998, 17, 3187–3197. [Google Scholar] [CrossRef]
- Yang, Y.P.; Qin, R.H.; Zhao, J.J.; Qin, X.Y. BOP1 Silencing Suppresses Gastric Cancer Proliferation through p53 Modulation. Curr. Med. Sci. 2021, 41, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, S.D.; Chatterjee, M.; Coleman, L.; Clancy, M.A.; Folta, K.M. Analysis of Block of Cell Proliferation 1 (BOP1) Activity in Strawberry and Arabidopsis. Plant Sci. 2016, 245, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.K.; Ahn, C.S.; Lee, H.S.; Kim, J.K.; Pai, H.S. Pescadillo Plays an Essential Role in Plant Cell Growth and Survival by Modulating Ribosome Biogenesis. Plant J. 2013, 76, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Yu, X.; Shen, R.; He, Y. HYL1 Gene Maintains Venation and Polarity of Leaves. Planta 2005, 221, 231–242. [Google Scholar] [CrossRef]
- Liu, Z.; Jia, L.; Wang, H.; He, Y. HYL1 Regulates the Balance between Adaxial and Abaxial Identity for Leaf Flattening via miRNA-mediated Pathways. J. Exp. Bot. 2011, 62, 4367–4381. [Google Scholar] [CrossRef]
- Petricka, J.J.; Nelson, T.M. Arabidopsis Nucleolin Affects Plant Development and Patterning. Plant Physiol. 2007, 144, 173–186. [Google Scholar] [CrossRef]
- Weis, B.L.; Kovacevic, J.; Missbach, S.; Schleiff, E. Plant-Specific Features of Ribosome Biogenesis. Trends Plant Sci. 2015, 20, 729–740. [Google Scholar] [CrossRef]
- Abbasi, N.; Kim, H.B.; Park, N.I.; Kim, H.S.; Kim, Y.K.; Park, Y.I.; Choi, S.B. APUM23, a Nucleolar Puf Domain Protein, Is Involved in Pre-ribosomal RNA Processing and Normal Growth Patterning in Arabidopsis. Plant J. 2010, 64, 960–976. [Google Scholar] [CrossRef]
- Baβler, J.; Kallas, M.; Pertschy, B.; Ulbrich, C.; Thoms, M.; Hurt, E. The AAA-ATPase Rea1 Drives Removal of Biogenesis Factors During Multiple Stages of 60S Ribosome Assembly. Mol. Cell 2010, 38, 712–721. [Google Scholar]
- Lange, H.; Sement, F.M.; Gagliardi, D. MTR4, a Putative RNA Helicase and Exosome Co-factor, Is Required for Proper rRNA Biogenesis and Development in Arabidopsis Thaliana. Plant J. 2011, 68, 51–63. [Google Scholar] [CrossRef]
- Weis, B.L.; Missbach, S.; Marzi, J.; Bohnsack, M.T.; Schleiff, E. The 60S Associated Ribosome Biogenesis Factor LSG1-2 Is Required for 40S Maturation in Arabidopsis Thaliana. Plant J. 2014, 80, 1043–1056. [Google Scholar] [CrossRef] [PubMed]
- Pestov, D.G.; Strezoska, Z.; Lau, L.F. Evidence of p53-dependent cross-talk between ribosome biogenesis and the cell cycle: Effects of nucleolar protein Bop1 on G(1)/S transition. Mol. Cell Biol. 2001, 21, 4246–4255. [Google Scholar] [CrossRef]
- Holzel, M.; Rohrmoser, M.; Schlee, M.; Grimm, T.; Harasim, T.; Malamoussi, A.; Gruber-Eber, A.; Kremmer, E.; Hiddemann, W.; Bornkamm, G.W.; et al. Mammalian WDR12 Is a Novel Member of the Pes1-Bop1 Complex and Is Required for Ribosome Biogenesis and Cell Proliferation. J. Cell Biol. 2005, 170, 367–378. [Google Scholar] [CrossRef] [PubMed]
- Du, F.; Guan, C.; Jiao, Y. Molecular Mechanisms of Leaf Morphogenesis. Mol. Plant 2018, 11, 1117–1134. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Khan, N.; Xie, L. Molecular and Hormonal Regulation of Leaf Morphogenesis in Arabidopsis. Int. J. Mol. Sci. 2020, 21, 5132. [Google Scholar] [CrossRef] [PubMed]
- Li, L.C.; Kang, D.M.; Chen, Z.L.; Qu, L.J. Hormonal Regulation of Leaf Morphogenesis in Arabidopsis. J. Integr. Plant Biol. 2007, 49, 75–80. [Google Scholar] [CrossRef]
- Horiguchi, G.; Molla-Morales, A.; Perez-Perez, J.M.; Kojima, K.; Robles, P.; Ponce, M.R.; Micol, J.L.; Tsukaya, H. Differential Contributions of Ribosomal Protein Genes to Arabidopsis Thaliana Leaf Development. Plant J. 2011, 65, 724–736. [Google Scholar] [CrossRef]
- Serrano-Cartagena, J.; Robles, P.; Ponce, M.R.; Micol, J.L. Genetic Analysis of Leaf form Mutants from the Arabidopsis Information Service collection. Mol. Gen. Genet. 1999, 261, 725–739. [Google Scholar] [CrossRef]
- Liu, Z.; Jia, L.; Mao, Y.; He, Y. Classification and Quantification of Leaf Curvature. J. Exp. Bot. 2010, 61, 2757–2767. [Google Scholar] [CrossRef]
- Pulungan, S.I.; Yano, R.; Okabe, Y.; Ichino, T.; Kojima, M.; Takebayashi, Y.; Sakakibara, H.; Ariizumi, T.; Ezura, H. SlLAX1 is Required for Normal Leaf Development Mediated by Balanced Adaxial and Abaxial Pavement Cell Growth in Tomato. Plant Cell Physiol. 2018, 59, 1170–1186. [Google Scholar] [CrossRef]
- Gotoh, E.; Suetsugu, N.; Higa, T.; Matsushita, T.; Tsukaya, H.; Wada, M. Palisade Cell Shape Affects the Light-induced Chloroplast Movements and Leaf Photosynthesis. Sci. Rep. 2018, 8, 1472. [Google Scholar] [CrossRef] [PubMed]
- David, K.M.; Couch, D.; Braun, N.; Brown, S.; Grosclaude, J.; Perrot-Rechenmann, C. The Auxin-binding Protein 1 Is Essential for the Control of Cell Cycle. Plant J. 2007, 50, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Massonnet, C.; Vile, D.; Fabre, J.; Hannah, M.A.; Caldana, C.; Lisec, J.; Beemster, G.T.; Meyer, R.C.; Messerli, G.; Gronlund, J.T.; et al. Probing the Reproducibility of Leaf Growth and Molecular Phenotypes: A Comparison of Three Arabidopsis Accessions Cultivated in Ten Laboratories. Plant Physiol. 2010, 152, 2142–2157. [Google Scholar] [CrossRef] [PubMed]
- Perrot-Rechenmann, C. Cellular Responses to Auxin: Division Versus Expansion. C.S.H. Perspect. Biol. 2010, 2, a001446. [Google Scholar] [CrossRef] [PubMed]
- Tromas, A.; Braun, N.; Muller, P.; Khodus, T.; Paponov, I.A.; Palme, K.; Ljung, K.; Lee, J.Y.; Benfey, P.; Murray, J.A.; et al. The Auxin Binding Protein 1 Is Required for Differential Auxin Responses Mediating Root Growth. PLoS ONE 2009, 4, e6648. [Google Scholar] [CrossRef]
- Li, Z.; Wang, X.; Cao, X.; Chen, B.; Ma, C.; Lv, J.; Sun, Z.; Qiao, K.; Zhu, L.; Zhang, C.; et al. GhTULP34, a Member of Tubby-like Proteins, Interacts with GhSKP1A to Negatively Regulate Plant Osmotic Stress. Genomics 2021, 113, 462–474. [Google Scholar] [CrossRef]
- Braun, N.; Wyrzykowska, J.; Muller, P.; David, K.; Couch, D.; Perrot-Rechenmann, C.; Fleming, A.J. Conditional Repression of Auxin Binding Protein 1 Reveals That It Coordinates Cell Division and Cell Expansion during Postembryonic Shoot Development in Arabidopsis and Tobacco. Plant Cell 2008, 20, 2746–2762. [Google Scholar] [CrossRef]
- Himanen, K.; Boucheron, E.; Vanneste, S.; Almeida Engler, J.; Inze, D.; Beeckman, T. Auxin-mediated Cell Cycle Activation during Early Lateral Root Initiation. Plant Cell 2002, 14, 2339–2351. [Google Scholar] [CrossRef]
- Magyar, Z.; De Veylder, L.; Atanassova, A.; Bako, L.; Inze, D.; Bogre, L. The Role of the Arabidopsis E2FB Transcription Factor in Regulating Auxin-Dependent Cell Division. Plant Cell 2005, 17, 2527–2541. [Google Scholar] [CrossRef]
- Świątek, A.; Lenjou, M.; Van Bockstaele, D.; Inzé, D.; Van Onckelen, H. Differential Effect of Jasmonic Acid and Abscisic Acid on Cell Cycle Progression in Tobacco BY-2 Cells. Plant Physiol. 2002, 128, 201–211. [Google Scholar] [CrossRef]
- Noir, S.; Bomer, M.; Takahashi, N.; Ishida, T.; Tsui, T.L.; Balbi, V.; Shanahan, H.; Sugimoto, K.; Devoto, A. Jasmonate Controls Leaf Growth by Repressing Cell Proliferation and the Onset of Endoreduplication while Maintaining a Potential Stand-by Mode. Plant Physiol. 2013, 161, 1930–1951. [Google Scholar] [CrossRef] [PubMed]
- De Veylder, L.; Beeckman, T.; Inze, D. The Ins and Outs of the Plant Cell Cycle. Nat. Rev. Mol. Cell Biol. 2007, 8, 655–665. [Google Scholar] [CrossRef] [PubMed]
- Inzé, D. Green Light for the Cell Cycle. EMBO J. 2005, 24, 657–662. [Google Scholar] [CrossRef] [PubMed]
- Inze, D.; De Veylder, L. Cell Cycle Regulation in Plant Development. Annu. Rev. Genet. 2006, 40, 77–105. [Google Scholar] [CrossRef]
- Sablowski, R.; Carnier Dornelas, M. Interplay between Cell Growth and Cell Cycle in Plants. J. Exp. Bot. 2014, 65, 2703–2714. [Google Scholar] [CrossRef]
- Gutzat, R.; Borghi, L.; Gruissem, W. Emerging Roles of Retinoblastoma-related Proteins in Evolution and Plant Development. Trends Plant Sci. 2012, 17, 139–148. [Google Scholar] [CrossRef]
- Dewitte, W.; Murray, J.A. The Plant Cell Cycle. Annu. Rev. Plant Biol. 2003, 54, 235–264. [Google Scholar] [CrossRef]
- Egelkrout, E.M.; Mariconti, L.; Settlage, S.B.; Cella, R.; Robertson, D.; Hanley-Bowdoin, L. Two E2F Elements Regulate the Proliferating Cell Nuclear Antigen Promoter Differently during Leaf Development. Plant Cell 2002, 14, 3225–3236. [Google Scholar] [CrossRef][Green Version]
- Ramirez-Parra, E.; Fründt, C.; Gutierrez, C. A Genome-wide Identification of E2F-regulated Genes in Arabidopsis. Plant J. 2003, 33, 801–811. [Google Scholar] [CrossRef]
- Sun, Z.; Wang, X.; Qiao, K.; Fan, S.; Ma, Q. Genome-wide Analysis of JMJ-C Histone Demethylase Family Involved in Salt-tolerance in Gossypium hirsutum L. Plant Physiol. Bioch. 2021, 158, 420–433. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-time Quantitative PCR and the 2-ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Sun, Z.; Wang, L.; Chen, L.; Ma, L.; Lv, J.; Qiao, K.; Fan, S.; Ma, Q. GhBOP1 as a Key Factor of Ribosomal Biogenesis: Development of Wrinkled Leaves in Upland Cotton. Int. J. Mol. Sci. 2022, 23, 9942. https://doi.org/10.3390/ijms23179942
Wang Y, Sun Z, Wang L, Chen L, Ma L, Lv J, Qiao K, Fan S, Ma Q. GhBOP1 as a Key Factor of Ribosomal Biogenesis: Development of Wrinkled Leaves in Upland Cotton. International Journal of Molecular Sciences. 2022; 23(17):9942. https://doi.org/10.3390/ijms23179942
Chicago/Turabian StyleWang, Yanwen, Zhimao Sun, Long Wang, Lingling Chen, Lina Ma, Jiaoyan Lv, Kaikai Qiao, Shuli Fan, and Qifeng Ma. 2022. "GhBOP1 as a Key Factor of Ribosomal Biogenesis: Development of Wrinkled Leaves in Upland Cotton" International Journal of Molecular Sciences 23, no. 17: 9942. https://doi.org/10.3390/ijms23179942
APA StyleWang, Y., Sun, Z., Wang, L., Chen, L., Ma, L., Lv, J., Qiao, K., Fan, S., & Ma, Q. (2022). GhBOP1 as a Key Factor of Ribosomal Biogenesis: Development of Wrinkled Leaves in Upland Cotton. International Journal of Molecular Sciences, 23(17), 9942. https://doi.org/10.3390/ijms23179942




