18F-Fluoroethylcholine PET/CT Radiomic Analysis for Newly Diagnosed Prostate Cancer Patients: A Monocentric Study
Abstract
:1. Introduction
2. Results
3. Discussion
3.1. Conventional Semiquantitative PET Parameters
3.2. Advanced Radiomic Features
3.3. Limitations and Solutions
4. Materials and Methods
4.1. Acquisition Protocol and Image Analysis
- Maximum and mean Standardized Uptake Value, which indirectly estimates the maximum and mean values of 18F-FeCh concentration within the VOI by the normalization with the patient’s body weight (tSUVmax and tSUVmean, respectively);
- Metabolic Tumor Volume (tMTV), which represents the volume involving all the 18F-FECh counts with at least 40% of SUVmax value;
- Total Lesion Activity (tTLA as expression of tSUVmean × tMTV);
- First-order radiomic features, such as tSkewness and tKurtosis, which describe the asimmetricity and the shape of distribution of 18F-FECh values within the VOI, respectively.
4.2. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mottet, N.; Bellmunt, J.; Bolla, M.; Briers, E.; Cumberbatch, M.G.; De Santis, M.; Fossati, N.; Gross, T.; Henry, A.M.; Joniau, S.; et al. EAU-ESTRO-SIOG Guidelines on Prostate Cancer. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur. Urol. 2017, 71, 618–629. [Google Scholar] [CrossRef] [PubMed]
- Hull, G.W.; Rabbani, F.; Abbas, F.; Wheeler, T.M.; Kattan, M.W.; Scardino, P.T. Cancer control with radical prostatectomy alone in 1000 consecutive patients. J. Urol. 2002, 167, 528–534. [Google Scholar] [CrossRef]
- Kupelian, P.; Katcher, J.; Levin, H.; Zippe, C.; Klein, E. Correlation of clinical and pathologic factors with rising prostate-specific antigen profiles after radical prostatectomy alone for clinically localized prostate cancer. Urology 1996, 48, 249–260. [Google Scholar] [CrossRef]
- Stephenson, A.J.; Scardino, P.T.; Eastham, J.A.; Bianco, F.J., Jr.; Dotan, Z.A.; Fearn, P.A.; Kattan, M.W. Preoperative nomogram predicting the 10-year probability of prostate cancer recurrence after radical prostatectomy. J. Natl. Cancer Inst. 2006, 98, 715–717. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, D.J.; Nielsen, M.E.; Han, M.; Partin, A.W. Contemporary evaluation of the D’amico risk classification of prostate cancer. Urology 2007, 70, 931–935. [Google Scholar] [CrossRef] [PubMed]
- Hatt, M.; Tixier, F.; Pierce, L.; Kinahan, P.E.; Le Rest, C.C.; Visvikis, D. Characterization of PET/CT images using texture analysis: The past, the present… any future? Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 151–165. [Google Scholar] [CrossRef] [PubMed]
- Annunziata, S.; Pizzuto, D.A.; Treglia, G. Diagnostic Performance of PET Imaging Using Different Radiopharmaceuticals in Prostate Cancer According to Published Meta-Analyses. Cancers 2020, 12, 2153. [Google Scholar] [CrossRef]
- Lin, C.Y.; Lee, M.T.; Lin, C.L.; Kao, C.H. Comparing the Staging/Restaging Performance of 68Ga-Labeled Prostate-Specific Membrane Antigen and 18F-Choline PET/CT in Prostate Cancer: A Systematic Review and Meta-analysis. Clin. Nucl. Med. 2019, 44, 365–376. [Google Scholar] [CrossRef]
- Evangelista, L.; Zattoni, F.; Guttilla, A.; Saladini, G.; Zattoni, F.; Colletti, P.M.; Rubello, D. Choline PET or PET/CT and biochemical relapse of prostate cancer: A systematic review and meta-analysis. Clin Nucl Med. 2013, 38, 305–314. [Google Scholar] [CrossRef]
- Parekh, V.; Jacobs, M.A. Radiomics: A new application from established techniques. Expert Rev. Precis. Med. Drug Dev. 2016, 1, 207–226. [Google Scholar] [CrossRef]
- Alongi, P.; Stefano, A.; Comelli, A.; Laudicella, R.; Scalisi, S.; Arnone, G.; Barone, S.; Spada, M.; Purpura, P.; Bartolotta, T.V.; et al. Radiomics analysis of 18F-Choline PET/CT in the prediction of disease outcome in high-risk prostate cancer: An explorative study on machine learning feature classification in 94 patients. Eur. Radiol. 2021, 31, 4595–4605. [Google Scholar] [CrossRef] [PubMed]
- Zanoni, L.; Mei, R.; Bianchi, L.; Giunchi, F.; Maltoni, L.; Pultrone, C.V.; Nanni, C.; Bossert, I.; Matti, A.; Schiavina, R.; et al. The Role of [18F]Fluciclovine PET/CT in the Characterization of High-Risk Primary Prostate Cancer: Comparison with [11C]Choline PET/CT and Histopathological Analysis. Cancers 2021, 13, 1575. [Google Scholar] [CrossRef]
- Gandaglia, G.; Abdollah, F.; Schiffmann, J.; Trudeau, V.; Shariat, S.F.; Kim, S.P.; Perrotte, P.; Montorsi, F.; Briganti, A.; Trinh, Q.D.; et al. Distribution of metastatic sites in patients with prostate cancer: A population-based analysis. Prostate 2014, 74, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Dejust, S.; Messaoud, L.; Jallerat, P.; Marical, V.; Morland, D. Hepatic Metastases from Prostatic Adenocarcinoma without Elevated 18F-Choline Activity. Clin. Nucl. Med. 2018, 43, 780–781. [Google Scholar] [CrossRef]
- Schaefferkoetter, J.D.; Wang, Z.; Stephenson, M.C.; Roy, S.; Conti, M.; Eriksson, L.; Townsend, D.W.; Thamboo, T.; Chiong, E. Quantitative (18)F-fluorocholine positron emission tomography for prostate cancer: Correlation between kinetic parameters and Gleason scoring. EJNMMI Res. 2017, 7, 25. [Google Scholar] [CrossRef] [PubMed]
- Castellucci, P.; Ceci, F.; Fanti, S. Imaging of Prostate Cancer Using (11)C-Choline PET/Computed Tomography. Urol. Clin. N. Am. 2018, 45, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Tu, S.J.; Tran, V.T.; Teo, J.M.; Chong, W.C.; Tseng, J.R. Utility of radiomic zones for risk classification and clinical outcome predictions using supervised machine learning during simultaneous (11) C-choline PET/MRI acquisition in prostate cancer patients. Med. Phys. 2021, 48, 5192–5201. [Google Scholar] [CrossRef]
- Zamboglou, C.; Carles, M.; Fechter, T.; Kiefer, S.; Reichel, K.; Fassbender, T.F.; Bronsert, P.; Koeber, G.; Schilling, O.; Ruf, J.; et al. Radiomic features from PSMA PET for non-invasive intraprostatic tumor discrimination and characterization in patients with intermediate- and high-risk prostate cancer—A comparison study with histology reference. Theranostics 2019, 9, 2595–2605. [Google Scholar] [CrossRef]
- Kasivisvanathan, V.; Rannikko, A.S.; Borghi, M.; Panebianco, V.; Mynderse, L.A.; Vaarala, M.H.; Briganti, A.; Budäus, L.; Hellawell, G.; Hindley, R.G.; et al. MRI-Targeted or Standard Biopsy for Prostate-Cancer Diagnosis. N. Engl. J. Med. 2018, 378, 1767–1777. [Google Scholar] [CrossRef]
- Hartenbach, M.; Hartenbach, S.; Bechtloff, W.; Danz, B.; Kraft, K.; Klemenz, B.; Sparwasser, C.; Hacker, M. Combined PET/MRI improves diagnostic accuracy in patients with prostate cancer: A prospective diagnostic trial. Clin. Cancer Res. 2014, 20, 3244–3253. [Google Scholar] [CrossRef]
- Zwanenburg, A.; Vallières, M.; Abdalah, M.A.; Aerts, H.; Andrearczyk, V.; Apte, A.; Ashrafinia, S.; Bakas, S.; Beukinga, R.J.; Boellaard, R.; et al. The Image Biomarker Standardization Initiative: Standardized Quantitative Radiomics for High-Throughput Image-based Phenotyping. Radiology 2020, 295, 328–338. [Google Scholar] [CrossRef] [PubMed]
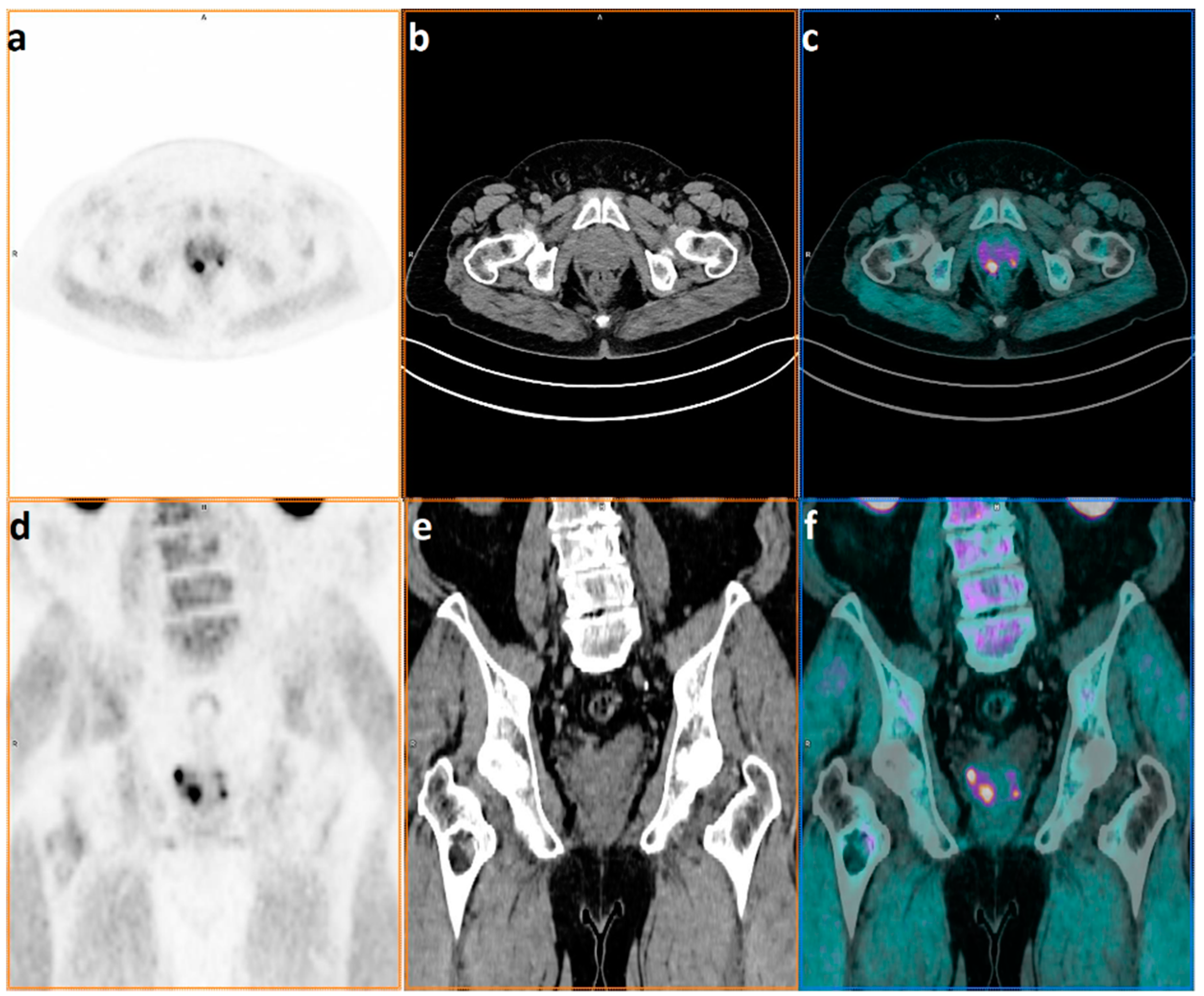
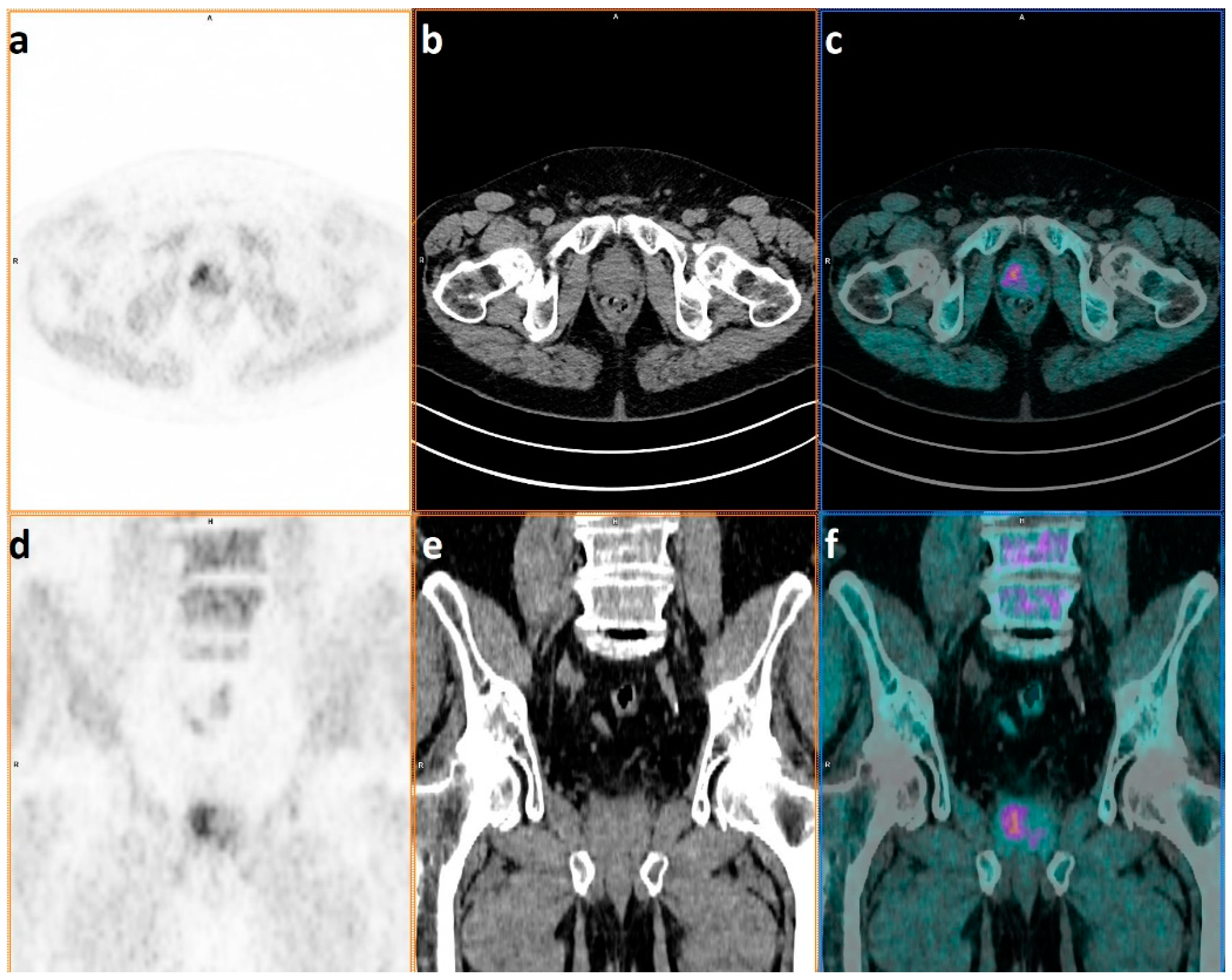
| n = 67 | |
|---|---|
| Age in years (mean ± sd) | 70.1 ± 7.1 |
| PSA in ng/mL (mean ± sd) | 15.0 ± 13.0 |
| Digital Rectal Examination (n, %) | |
| Suspicious | 26 (41.3%) |
| Negative | 37 (58.7%) |
| Missing data | 4 (6.0%) |
| Gleason score (n, %) | |
| 6 = 3 + 3 | 1 (1.5%) |
| 7 = 3 + 4 | 8 (11.9%) |
| 7 = 4 + 3 | 20 (29.9%) |
| 8 = 3 + 5 | 1 (1.5%) |
| 8 = 4 + 4 | 19 (28.3%) |
| 9 = 4 + 5 | 17 (25.4%) |
| 9 = 5 + 4 | 1 (1.5%) |
| PET-CT system (n, %) | |
| Gemini XL | 39 (58.2%) |
| Biograph mCT | 22 (32.8%) |
| Biograph Vision V600 | 6 (9.0%) |
| PET parameters (mean ± sd) | |
| tSUVmax | 10.4 ± 4.4 |
| tSUVmean | 3.9 ± 1.5 |
| tMTV (mL) | 16.0 ± 11.7 |
| tTLA | 60.6 ± 51.6 |
| tSkewness | 1.1 ± 0.7 |
| tKurtosis | 2.2 ± 3.7 |
| TBR | 2.7 ± 1.1 |
| SNR | 15.3 ± 6.4 |
| PSA > Median Value (9.3 ng/mL) | GS 6-7 vs. 8-9 | Digital Rectal Examination Results | ||
|---|---|---|---|---|
| Univariate | Univariate | Univariate | Multivariate | |
| tSUVmax | 0.285 | 0.664 | p = 0.701 | - |
| tSUVmean | 0.074 | 0.306 | p = 0.047 * 1.52 [1.01; 2.29] | p = 0.13 |
| tMTV (mL) | 0.210 | 0.447 | p = 0.867 | - |
| tTLA | 0.195 | 0.954 | p = 0.400 | - |
| tSkewness | 0.345 | 0.188 | p = 0.007 * 0.21 [0.07; 0.65] | Rejected |
| tKurtosis | 0.196 | 0.135 | p = 0.018 * 0.60 [0.39; 0.91] | p = 0.03 * 0.64 [0.42; 0.96] |
| TBR | 0.319 | 0.378 | p = 0.313 | - |
| SNR | 0.091 | 0.723 | p = 0.752 | - |
| cov.1.cov.1 | cov.2.cov.2 | p.Value.1 | sd.p.Value.1 | p.Value.2 | sd.p.Value.2 | AUC | sd.AUC | |
|---|---|---|---|---|---|---|---|---|
| PSA | szm_2.5D.z.entr | cm.clust.tend | 0.079 | 0.035 | 0.132 | 0.075 | 0.829 | 0.195 |
| DRE | stat.kurt | Stat.entropy | 0.136 | 0.048 | 0.371 | 0.145 | 0.787 | 0.097 |
| GS | cm.info.corr.1 | rlm.hgre | 0.410 | 0.214 | 0.219 | 0.114 | 0.812 | 0.118 |
| Philips Gemini XL | Siemens Biograph mCT | Siemens Biograph Vision V600 | |
|---|---|---|---|
| Low dose CT scan | 120 kV, 40–50 mAs | 120 kV, 40–50 mAs | 120 kV, 40–50 mAs |
| Acquisition time and modality | 2 min per bed | 2 min per bed | PET continuous bed motion: 1–2 mm/sec |
| Image Reconstruction | LOR RAMLA reconstruction without PSF and TOF (3 iterations and 33 subsets, voxel size: 4 × 4 × 4 mm3) | UltraHD-PET: line-of- response row-action maximum likelihood algorithm 3D OSEM reconstruction + PSF modeling + TOF (2 iterations, 21 subsets, voxel size: 3.2 × 3.2 × 5 mm3), | UltraHD-PET: line-of- response row-action maximum likelihood algorithm 3D OSEM reconstruction + PSF modeling + TOF (4 iterations and 5 subsets, voxel size of 1.8 × 1.8 × 5 mm3), |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pizzuto, D.A.; Triumbari, E.K.A.; Morland, D.; Boldrini, L.; Gatta, R.; Treglia, G.; Bientinesi, R.; De Summa, M.; De Risi, M.; Caldarella, C.; et al. 18F-Fluoroethylcholine PET/CT Radiomic Analysis for Newly Diagnosed Prostate Cancer Patients: A Monocentric Study. Int. J. Mol. Sci. 2022, 23, 9120. https://doi.org/10.3390/ijms23169120
Pizzuto DA, Triumbari EKA, Morland D, Boldrini L, Gatta R, Treglia G, Bientinesi R, De Summa M, De Risi M, Caldarella C, et al. 18F-Fluoroethylcholine PET/CT Radiomic Analysis for Newly Diagnosed Prostate Cancer Patients: A Monocentric Study. International Journal of Molecular Sciences. 2022; 23(16):9120. https://doi.org/10.3390/ijms23169120
Chicago/Turabian StylePizzuto, Daniele Antonio, Elizabeth Katherine Anna Triumbari, David Morland, Luca Boldrini, Roberto Gatta, Giorgio Treglia, Riccardo Bientinesi, Marco De Summa, Marina De Risi, Carmelo Caldarella, and et al. 2022. "18F-Fluoroethylcholine PET/CT Radiomic Analysis for Newly Diagnosed Prostate Cancer Patients: A Monocentric Study" International Journal of Molecular Sciences 23, no. 16: 9120. https://doi.org/10.3390/ijms23169120
APA StylePizzuto, D. A., Triumbari, E. K. A., Morland, D., Boldrini, L., Gatta, R., Treglia, G., Bientinesi, R., De Summa, M., De Risi, M., Caldarella, C., Scarciglia, E., Totaro, A., & Annunziata, S. (2022). 18F-Fluoroethylcholine PET/CT Radiomic Analysis for Newly Diagnosed Prostate Cancer Patients: A Monocentric Study. International Journal of Molecular Sciences, 23(16), 9120. https://doi.org/10.3390/ijms23169120










