Stem Cell-Based Therapies for Inflammatory Bowel Disease
Abstract
1. Introduction
2. The Pathogenesis of Inflammatory Bowel Disease
2.1. Genetic Factors
2.2. Environmental Factors and Lifestyle
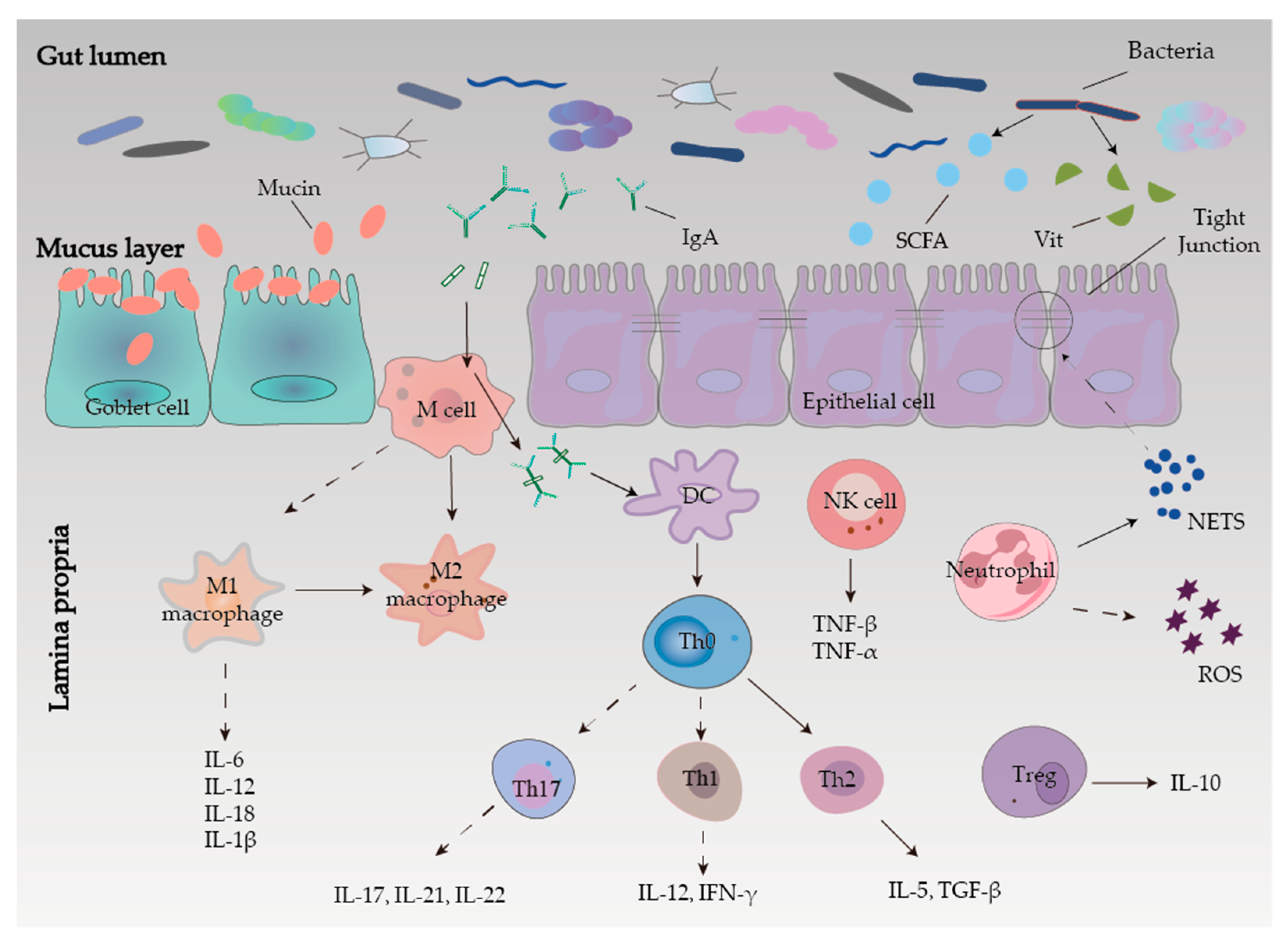
2.3. Intestinal Mucosal Barrier
2.3.1. Mechanical Barrier
2.3.2. Chemical Barrier
2.3.3. Microbial Barrier
2.3.4. Immune Barrier
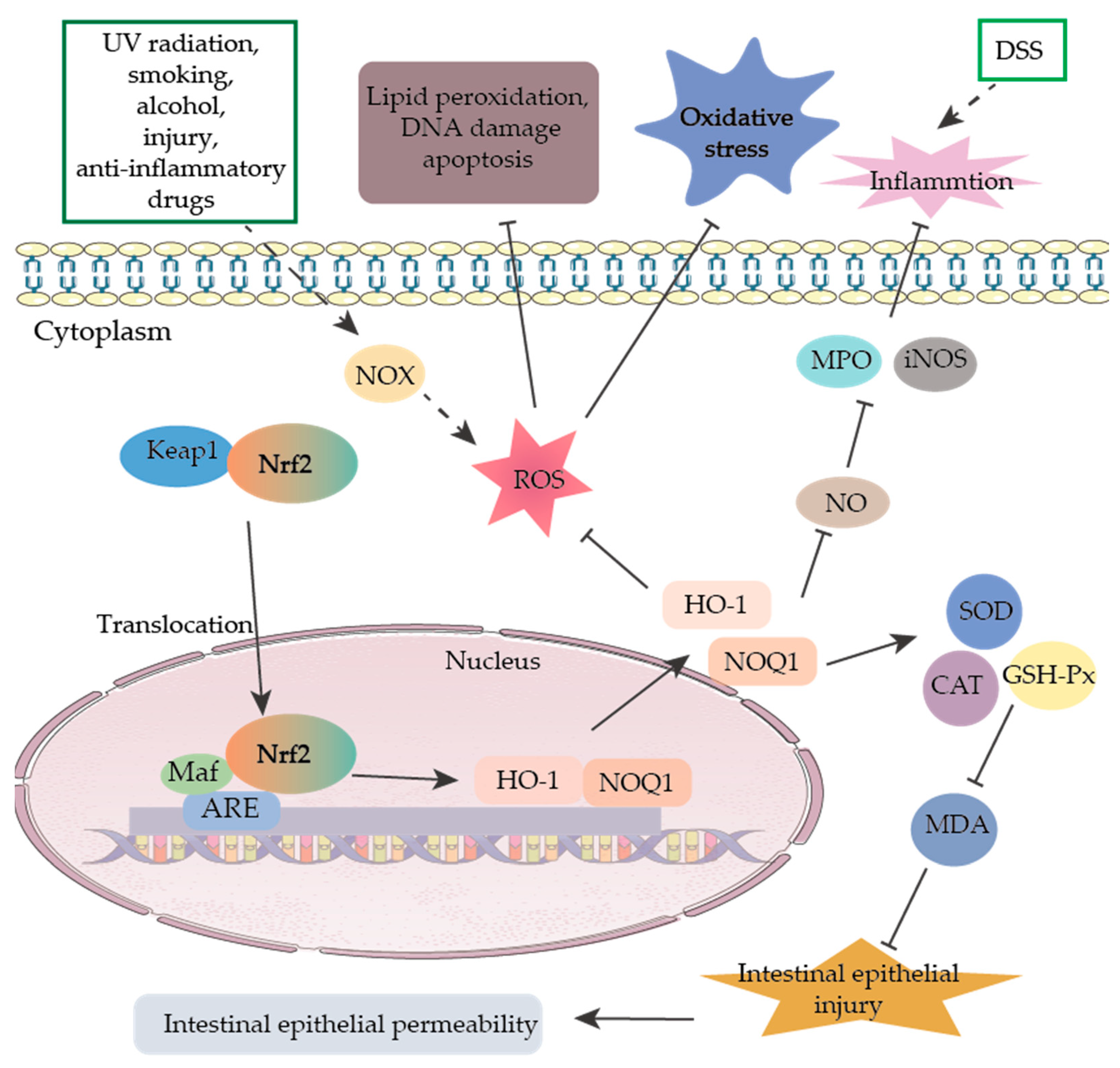
2.4. Oxidative Stress
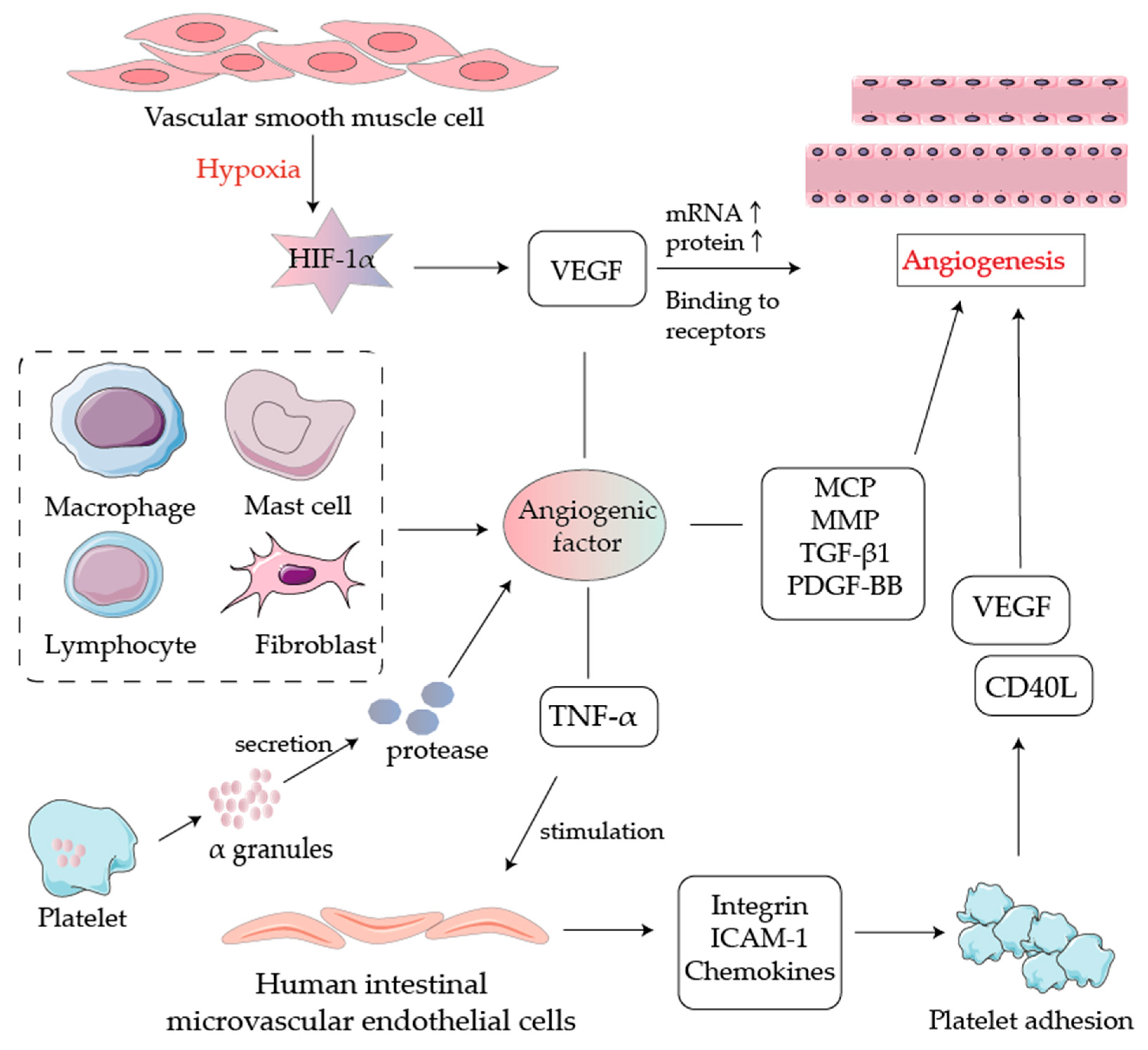
2.5. Angiogenesis
3. Pharmacological Treatments of Inflammatory Bowel Disease
3.1. Small Molecule Medications
3.1.1. Amino Salicylate
3.1.2. Glucocorticoids
3.1.3. Immunomodulators
3.2. Biological Agents
3.2.1. Tumor Necrosis Factor Inhibitors
3.2.2. Integrin Receptor Antagonists
3.2.3. Interleukin Antagonists
4. Stem Cell Therapy for IBD
4.1. HSCT and IBD
4.1.1. Mechanism and Application of HSCT
4.1.2. Safety of HSCT
4.2. Mesenchymal Stem Cells and IBD
4.2.1. Mechanism of MSCs for IBD
Local Microcirculation Construction
Fixation Repair
Immunomodulation
- Macrophages
- Dendritic Cells
- T-cell subsets
- MSCs-derived exosomes
4.2.2. Safety of Mesenchymal Stem Cells
4.3. Embryonic Stem Cells and IBD
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Viola, A.; Fiorino, G.; Costantino, G.; Fries, W. Epidemiology and clinical course of late onset inflammatory bowel disease. Minerva Gastroenterol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Senger, P.C.; Zacharias, P.; Quaresma, A.B.; Kotze, P.G. Inflammatory bowel disease epidemiology data from a prospective registry in Cordoba, Argentina: Raising the bar for future studies in Latin America. Dig. Liver Dis. 2021, 53, 1212–1213. [Google Scholar] [CrossRef] [PubMed]
- Goodman, W.A.; Erkkila, I.P.; Pizarro, T.T. Sex matters: Impact on pathogenesis, presentation and treatment of inflammatory bowel disease. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 740–754. [Google Scholar] [CrossRef] [PubMed]
- Nishida, A.; Inoue, R.; Inatomi, O.; Bamba, S.; Naito, Y.; Andoh, A. Gut microbiota in the pathogenesis of inflammatory bowel disease. Clin. J. Gastroenterol. 2018, 11, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.Y.; Luo, Q.; Xu, J.R.; Bai, J.; Mu, L.M.; Yan, Y.; Duan, J.L.; Cui, Y.N.; Su, Z.B.; Xie, Y.; et al. Regulating Stem Cell-Related Genes Induces the Plastic Differentiation of Cancer Stem Cells to Treat Breast Cancer. Mol. Ther. Oncolytics 2020, 18, 396–408. [Google Scholar] [CrossRef] [PubMed]
- Drakos, P.E.; Nagler, A.; Or, R. Case of Crohn’s disease in bone marrow transplantation. Am. J. Hematol. 1993, 43, 157–158. [Google Scholar] [CrossRef] [PubMed]
- Kashyap, A.; Forman, S.J. Autologous bone marrow transplantation for non-Hodgkin’s lymphoma resulting in long-term remission of coincidental Crohn’s disease. Br. J. Haematol. 1998, 103, 651–652. [Google Scholar] [CrossRef] [PubMed]
- Oyama, Y.; Craig, R.M.; Traynor, A.E.; Quigley, K.; Statkute, L.; Halverson, A.; Brush, M.; Verda, L.; Kowalska, B.; Krosnjar, N.; et al. Autologous hematopoietic stem cell transplantation in patients with refractory Crohn’s disease. Gastroenterology 2005, 128, 552–563. [Google Scholar] [CrossRef]
- Jostins, L.; Ripke, S.; Weersma, R.K.; Duerr, R.H.; McGovern, D.P.; Hui, K.Y.; Lee, J.C.; Schumm, L.P.; Sharma, Y.; Anderson, C.A.; et al. Host-microbe interactions have shaped the genetic architecture of inflammatory bowel disease. Nature 2012, 491, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Juyal, G.; Negi, S.; Sood, A.; Gupta, A.; Prasad, P.; Senapati, S.; Zaneveld, J.; Singh, S.; Midha, V.; van Sommeren, S.; et al. Genome-wide association scan in north Indians reveals three novel HLA-independent risk loci for ulcerative colitis. Gut 2015, 64, 571–579. [Google Scholar] [CrossRef]
- Liu, J.Z.; van Sommeren, S.; Huang, H.; Ng, S.C.; Alberts, R.; Takahashi, A.; Ripke, S.; Lee, J.C.; Jostins, L.; Shah, T.; et al. Association analyses identify 38 susceptibility loci for inflammatory bowel disease and highlight shared genetic risk across populations. Nat. Genet. 2015, 47, 979–986. [Google Scholar] [CrossRef] [PubMed]
- Khor, B.; Gardet, A.; Xavier, R.J. Genetics and pathogenesis of inflammatory bowel disease. Nature 2011, 474, 307–317. [Google Scholar] [CrossRef] [PubMed]
- Barrett, J.C.; Hansoul, S.; Nicolae, D.L.; Cho, J.H.; Duerr, R.H.; Rioux, J.D.; Brant, S.R.; Silverberg, M.S.; Taylor, K.D.; Barmada, M.M.; et al. Genome-wide association defines more than 30 distinct susceptibility loci for Crohn’s disease. Nat. Genet. 2008, 40, 955–962. [Google Scholar] [CrossRef]
- Agus, A.; Planchais, J.; Sokol, H. Gut Microbiota Regulation of Tryptophan Metabolism in Health and Disease. Cell Host Microbe 2018, 23, 716–724. [Google Scholar] [CrossRef] [PubMed]
- Severs, M.; van Erp, S.J.; van der Valk, M.E.; Mangen, M.J.; Fidder, H.H.; van der Have, M.; van Bodegraven, A.A.; de Jong, D.J.; van der Woude, C.J.; Romberg-Camps, M.J.; et al. Smoking is Associated with Extra-intestinal Manifestations in Inflammatory Bowel Disease. J. Crohns Colitis 2016, 10, 455–461. [Google Scholar] [CrossRef]
- Lang, B.M.; Biedermann, L.; van Haaften, W.T.; de Valliere, C.; Schuurmans, M.; Begre, S.; Zeitz, J.; Scharl, M.; Turina, M.; Greuter, T.; et al. Genetic polymorphisms associated with smoking behaviour predict the risk of surgery in patients with Crohn’s disease. Aliment. Pharmacol. Ther. 2018, 47, 55–66. [Google Scholar] [CrossRef]
- Lee, D.; Albenberg, L.; Compher, C.; Baldassano, R.; Piccoli, D.; Lewis, J.D.; Wu, G.D. Diet in the pathogenesis and treatment of inflammatory bowel diseases. Gastroenterology 2015, 148, 1087–1106. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.D.; Abreu, M.T. Diet as a Trigger or Therapy for Inflammatory Bowel Diseases. Gastroenterology 2017, 152, 398–414.e6. [Google Scholar] [CrossRef] [PubMed]
- Mouli, V.P.; Ananthakrishnan, A.N. Review article: Vitamin D and inflammatory bowel diseases. Aliment. Pharmacol. Ther. 2014, 39, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Chieppa, M.; Rescigno, M.; Huang, A.Y.; Germain, R.N. Dynamic imaging of dendritic cell extension into the small bowel lumen in response to epithelial cell TLR engagement. J. Exp. Med. 2006, 203, 2841–2852. [Google Scholar] [CrossRef] [PubMed]
- Epple, H.J.; Zeitz, M. HIV infection and the intestinal mucosal barrier. Ann. N. Y. Acad. Sci. 2012, 1258, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H. Intestinal permeability regulation by tight junction: Implication on inflammatory bowel diseases. Intest. Res. 2015, 13, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Takizawa, Y.; Kishimoto, H.; Tomita, M.; Hayashi, M. Changes in the expression levels of tight junction components during reconstruction of tight junction from mucosal lesion by intestinal ischemia/reperfusion. Eur. J. Drug Metab. Pharmacokinet. 2014, 39, 211–220. [Google Scholar] [CrossRef]
- Paone, P.; Cani, P.D. Mucus barrier, mucins and gut microbiota: The expected slimy partners? Gut 2020, 69, 2232–2243. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Pineiro, A.M.; Bergstrom, J.H.; Ermund, A.; Gustafsson, J.K.; Schutte, A.; Johansson, M.E.; Hansson, G.C. Studies of mucus in mouse stomach, small intestine, and colon. II. Gastrointestinal mucus proteome reveals Muc2 and Muc5ac accompanied by a set of core proteins. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 305, G348–G356. [Google Scholar] [CrossRef] [PubMed]
- Albert-Bayo, M.; Paracuellos, I.; Gonzalez-Castro, A.M.; Rodriguez-Urrutia, A.; Rodriguez-Lagunas, M.J.; Alonso-Cotoner, C.; Santos, J.; Vicario, M. Intestinal Mucosal Mast Cells: Key Modulators of Barrier Function and Homeostasis. Cells 2019, 8, 135. [Google Scholar] [CrossRef]
- Wang, W.; Chen, L.; Zhou, R.; Wang, X.; Song, L.; Huang, S.; Wang, G.; Xia, B. Increased proportions of Bifidobacterium and the Lactobacillus group and loss of butyrate-producing bacteria in inflammatory bowel disease. J. Clin. Microbiol. 2014, 52, 398–406. [Google Scholar] [CrossRef]
- Singh, N.; Gurav, A.; Sivaprakasam, S.; Brady, E.; Padia, R.; Shi, H.; Thangaraju, M.; Prasad, P.D.; Manicassamy, S.; Munn, D.H.; et al. Activation of Gpr109a, receptor for niacin and the commensal metabolite butyrate, suppresses colonic inflammation and carcinogenesis. Immunity 2014, 40, 128–139. [Google Scholar] [CrossRef] [PubMed]
- Amagase, K.; Kimura, Y.; Wada, A.; Yukishige, T.; Murakami, T.; Nakamura, E.; Takeuchi, K. Prophylactic effect of monosodium glutamate on NSAID-induced enteropathy in rats. Curr. Pharm. Des. 2014, 20, 2783–2790. [Google Scholar] [CrossRef]
- Zhao, H.M.; Xu, R.; Huang, X.Y.; Cheng, S.M.; Huang, M.F.; Yue, H.Y.; Wang, X.; Zou, Y.; Lu, A.P.; Liu, D.Y. Curcumin Suppressed Activation of Dendritic Cells via JAK/STAT/SOCS Signal in Mice with Experimental Colitis. Front. Pharmacol. 2016, 7, 455. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Yin, Y.T.; Zhao, H.M.; Wang, H.Y.; Zhong, Y.B.B.; Long, J.; Liu, D.Y. Sishen Pill Treatment of DSS-Induced Colitis via Regulating Interaction with Inflammatory Dendritic Cells and Gut Microbiota. Front. Physiol. 2020, 11, 801. [Google Scholar] [CrossRef]
- Wera, O.; Lancellotti, P.; Oury, C. The Dual Role of Neutrophils in Inflammatory Bowel Diseases. J. Clin. Med. 2016, 5, 118. [Google Scholar] [CrossRef]
- Kumar, S.; Gupta, E.; Kaushik, S.; Jyoti, A. Neutrophil Extracellular Traps: Formation and Involvement in Disease Progression. Iran. J. Allergy Asthma Immunol. 2018, 17, 208–220. [Google Scholar]
- Lin, E.Y.; Lai, H.J.; Cheng, Y.K.; Leong, K.Q.; Cheng, L.C.; Chou, Y.C.; Peng, Y.C.; Hsu, Y.H.; Chiang, H.S. Neutrophil Extracellular Traps Impair Intestinal Barrier Function during Experimental Colitis. Biomedicines 2020, 8, 275. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Shen, H.; Gu, P.Q.; Liu, Y.J.; Zhang, L.; Cheng, J.F. Baicalin alleviates TNBS-induced colitis by inhibiting PI3K/AKT pathway activation. Exp. Ther. Med. 2020, 20, 581–590. [Google Scholar] [CrossRef]
- Zhang, T.; Jiang, J.; Liu, J.; Xu, L.; Duan, S.; Sun, L.; Zhao, W.; Qian, F. MK2 Is Required for Neutrophil-Derived ROS Production and Inflammatory Bowel Disease. Front. Med. 2020, 7, 207. [Google Scholar] [CrossRef] [PubMed]
- Mills, C.D. Anatomy of a discovery: M1 and m2 macrophages. Front. Immunol. 2015, 6, 212. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Yang, X.; Yue, W.; Xu, X.; Li, B.; Zou, L.; He, R. Chemerin aggravates DSS-induced colitis by suppressing M2 macrophage polarization. Cell Mol. Immunol. 2014, 11, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Kamada, N.; Hisamatsu, T.; Okamoto, S.; Chinen, H.; Kobayashi, T.; Sato, T.; Sakuraba, A.; Kitazume, M.T.; Sugita, A.; Koganei, K.; et al. Unique CD14 intestinal macrophages contribute to the pathogenesis of Crohn disease via IL-23/IFN-gamma axis. J. Clin. Invest. 2008, 118, 2269–2280. [Google Scholar]
- Fujino, S.; Andoh, A.; Bamba, S.; Ogawa, A.; Hata, K.; Araki, Y.; Bamba, T.; Fujiyama, Y. Increased expression of interleukin 17 in inflammatory bowel disease. Gut 2003, 52, 65–70. [Google Scholar] [CrossRef]
- Teng, M.W.; Bowman, E.P.; McElwee, J.J.; Smyth, M.J.; Casanova, J.L.; Cooper, A.M.; Cua, D.J. IL-12 and IL-23 cytokines: From discovery to targeted therapies for immune-mediated inflammatory diseases. Nat. Med. 2015, 21, 719–729. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Huang, H.; Liu, D.; Fang, S.; Xian, Y.; Zhou, J.; Zuo, Y.; Wang, F.; Huang, O.; He, M. Evaluation of 14-3-3 protein family levels and associated receptor expression of estrogen and progesterone in human uterine leiomyomas. Gynecol. Endocrinol. 2012, 28, 665–668. [Google Scholar] [CrossRef]
- Ramanan, D.; Tang, M.S.; Bowcutt, R.; Loke, P.; Cadwell, K. Bacterial sensor Nod2 prevents inflammation of the small intestine by restricting the expansion of the commensal Bacteroides vulgatus. Immunity 2014, 41, 311–324. [Google Scholar] [CrossRef] [PubMed]
- Cleynen, I.; Boucher, G.; Jostins, L.; Schumm, L.P.; Zeissig, S.; Ahmad, T.; Andersen, V.; Andrews, J.M.; Annese, V.; Brand, S.; et al. Inherited determinants of Crohn’s disease and ulcerative colitis phenotypes: A genetic association study. Lancet 2016, 387, 156–167. [Google Scholar] [CrossRef]
- Ghorpade, D.S.; Sinha, A.Y.; Holla, S.; Singh, V.; Balaji, K.N. NOD2-nitric oxide-responsive microRNA-146a activates Sonic hedgehog signaling to orchestrate inflammatory responses in murine model of inflammatory bowel disease. J. Biol. Chem. 2013, 288, 33037–33048. [Google Scholar] [CrossRef] [PubMed]
- Pierik, M.; Joossens, S.; Van Steen, K.; Van Schuerbeek, N.; Vlietinck, R.; Rutgeerts, P.; Vermeire, S. Toll-like receptor-1, -2, and -6 polymorphisms influence disease extension in inflammatory bowel diseases. Inflamm. Bowel Dis. 2006, 12, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Lakatos, P.L.; Lakatos, L.; Szalay, F.; Willheim-Polli, C.; Osterreicher, C.; Tulassay, Z.; Molnar, T.; Reinisch, W.; Papp, J.; Mozsik, G.; et al. Toll-like receptor 4 and NOD2/CARD15 mutations in Hungarian patients with Crohn’s disease: Phenotype-genotype correlations. World J. Gastroenterol. 2005, 11, 1489–1495. [Google Scholar] [CrossRef]
- Meena, N.K.; Ahuja, V.; Meena, K.; Paul, J. Association of TLR5 gene polymorphisms in ulcerative colitis patients of north India and their role in cytokine homeostasis. PLoS ONE 2015, 10, e0120697. [Google Scholar] [CrossRef] [PubMed]
- O’Shea, J.J.; Holland, S.M.; Staudt, L.M. JAKs and STATs in immunity, immunodeficiency, and cancer. N. Engl. J. Med. 2013, 368, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.J.; Shin, J.S.; Noh, Y.S.; Cho, Y.W.; Hong, S.J.; Park, J.H.; Lee, J.Y.; Lee, J.Y.; Lee, K.T. Anti-inflammatory effects of methanol extract of Patrinia scabiosaefolia in mice with ulcerative colitis. J. Ethnopharmacol. 2011, 136, 428–435. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Chattopadhyay, R.; Mitra, S.; Crowe, S.E. Oxidative stress: An essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiol. Rev. 2014, 94, 329–354. [Google Scholar] [CrossRef] [PubMed]
- Muller, F. The nature and mechanism of superoxide production by the electron transport chain: Its relevance to aging. J. Am. Aging Assoc. 2000, 23, 227–253. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Williams, E.; Cadenas, E. Mitochondrial respiratory chain-dependent generation of superoxide anion and its release into the intermembrane space. Biochem. J. 2001, 353 Pt 2, 411–416. [Google Scholar] [CrossRef]
- Boveris, A.; Chance, B. The mitochondrial generation of hydrogen peroxide. General properties and effect of hyperbaric oxygen. Biochem. J. 1973, 134, 707–716. [Google Scholar] [CrossRef]
- Piechota-Polanczyk, A.; Fichna, J. Review article: The role of oxidative stress in pathogenesis and treatment of inflammatory bowel diseases. Naunyn Schmiedebergs Arch. Pharmacol 2014, 387, 605–620. [Google Scholar] [CrossRef]
- Achitei, D.; Ciobica, A.; Balan, G.; Gologan, E.; Stanciu, C.; Stefanescu, G. Different profile of peripheral antioxidant enzymes and lipid peroxidation in active and non-active inflammatory bowel disease patients. Dig. Dis. Sci. 2013, 58, 1244–1249. [Google Scholar] [CrossRef] [PubMed]
- Tonelli, C.; Chio, I.I.C.; Tuveson, D.A. Transcriptional Regulation by Nrf2. Antioxid Redox Signal. 2018, 29, 1727–1745. [Google Scholar] [CrossRef]
- Lee, J.S.; An, J.M.; Kang, E.A.; Han, Y.M.; Kim, Y.S.; Lee, H.J.; Kim, K.J.; Surh, Y.J.; Hahm, K.B. Host nuclear factor erythroid 2-related factor-2 defense system determines the outcome of dextran sulfate sodium-induced colitis in mice. J. Physiol. Pharmacol. 2018, 69, 755–767. [Google Scholar]
- Xiao, X.; Song, D.; Cheng, Y.; Hu, Y.; Wang, F.; Lu, Z.; Wang, Y. Biogenic nanoselenium particles activate Nrf2-ARE pathway by phosphorylating p38, ERK1/2, and AKT on IPEC-J2 cells. J. Cell Physiol. 2019, 234, 11227–11234. [Google Scholar] [CrossRef]
- Kageyama, S.; Saito, T.; Obata, M.; Koide, R.H.; Ichimura, Y.; Komatsu, M. Negative Regulation of the Keap1-Nrf2 Pathway by a p62/Sqstm1 Splicing Variant. Mol. Cell Biol. 2018, 38, e00642-17. [Google Scholar] [CrossRef]
- Fan, W.; Tang, Z.; Chen, D.; Moughon, D.; Ding, X.; Chen, S.; Zhu, M.; Zhong, Q. Keap1 facilitates p62-mediated ubiquitin aggregate clearance via autophagy. Autophagy 2010, 6, 614–621. [Google Scholar] [CrossRef]
- Harada, S.; Nakagawa, T.; Yokoe, S.; Edogawa, S.; Takeuchi, T.; Inoue, T.; Higuchi, K.; Asahi, M. Autophagy Deficiency Diminishes Indomethacin-Induced Intestinal Epithelial Cell Damage through Activation of the ERK/Nrf2/HO-1 Pathway. J. Pharmacol. Exp. Ther. 2015, 355, 353–361. [Google Scholar] [CrossRef]
- Scaldaferri, F.; Vetrano, S.; Sans, M.; Arena, V.; Straface, G.; Stigliano, E.; Repici, A.; Sturm, A.; Malesci, A.; Panes, J.; et al. VEGF-A links angiogenesis and inflammation in inflammatory bowel disease pathogenesis. Gastroenterology 2009, 136, 585–595.e5. [Google Scholar] [CrossRef] [PubMed]
- Pousa, I.D.; Mate, J.; Gisbert, J.P. Angiogenesis in inflammatory bowel disease. Eur. J. Clin. Investig. 2008, 38, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Tolstanova, G.; Deng, X.; Khomenko, T.; Garg, P.; Paunovic, B.; Chen, L.; Sitaraman, S.V.; Shiloach, J.; Szabo, S.; Sandor, Z. Role of anti-angiogenic factor endostatin in the pathogenesis of experimental ulcerative colitis. Life Sci. 2011, 88, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Italiano, J.E., Jr.; Richardson, J.L.; Patel-Hett, S.; Battinelli, E.; Zaslavsky, A.; Short, S.; Ryeom, S.; Folkman, J.; Klement, G.L. Angiogenesis is regulated by a novel mechanism: Pro- and antiangiogenic proteins are organized into separate platelet alpha granules and differentially released. Blood 2008, 111, 1227–1233. [Google Scholar] [CrossRef]
- Rutella, S.; Vetrano, S.; Correale, C.; Graziani, C.; Sturm, A.; Spinelli, A.; De Cristofaro, R.; Repici, A.; Malesci, A.; Danese, S. Enhanced platelet adhesion induces angiogenesis in intestinal inflammation and inflammatory bowel disease microvasculature. J. Cell Mol. Med. 2011, 15, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Milkiewicz, M.; Brown, M.D.; Egginton, S.; Hudlicka, O. Association between shear stress, angiogenesis, and VEGF in skeletal muscles in vivo. Microcirculation 2001, 8, 229–241. [Google Scholar] [CrossRef]
- Cane, G.; Moal, V.L.; Pages, G.; Servin, A.L.; Hofman, P.; Vouret-Craviari, V. Up-regulation of intestinal vascular endothelial growth factor by Afa/Dr diffusely adhering Escherichia coli. PLoS ONE 2007, 2, e1359. [Google Scholar] [CrossRef]
- Punchard, N.A.; Greenfield, S.M.; Thompson, R.P. Mechanism of action of 5-arninosalicylic acid. Mediat. Inflamm. 1992, 1, 151–165. [Google Scholar] [CrossRef]
- Truelove, S.C.; Witts, L.J. Cortisone in ulcerative colitis; preliminary report on a therapeutic trial. Br. Med. J. 1954, 2, 375–378. [Google Scholar] [CrossRef] [PubMed]
- Ford, A.C.; Bernstein, C.N.; Khan, K.J.; Abreu, M.T.; Marshall, J.K.; Talley, N.J.; Moayyedi, P. Glucocorticosteroid therapy in inflammatory bowel disease: Systematic review and meta-analysis. Am. J. Gastroenterol. 2011, 106, 590–599. [Google Scholar] [CrossRef] [PubMed]
- Kornbluth, A.; Sachar, D.B.; Practice Parameters Committee of the American College of Gastroenterology. Ulcerative colitis practice guidelines in adults: American College of Gastroenterology, Practice Parameters Committee. Am. J. Gastroenterol. 2010, 105, 501–523. [Google Scholar] [CrossRef] [PubMed]
- Feagan, B.G.; Fedorak, R.N.; Irvine, E.J.; Wild, G.; Sutherland, L.; Steinhart, A.H.; Greenberg, G.R.; Koval, J.; Wong, C.J.; Hopkins, M.; et al. A comparison of methotrexate with placebo for the maintenance of remission in Crohn’s disease. North American Crohn’s Study Group Investigators. N. Engl. J. Med. 2000, 342, 1627–1632. [Google Scholar] [CrossRef]
- Lichtenstein, G.R.; Abreu, M.T.; Cohen, R.; Tremaine, W.; American Gastroenterological, A. American Gastroenterological Association Institute technical review on corticosteroids, immunomodulators, and infliximab in inflammatory bowel disease. Gastroenterology 2006, 130, 940–987. [Google Scholar] [CrossRef]
- Zhou, Z.; Dai, C.; Liu, W.X. Anti-TNF-A therapy about infliximab and adalimamab for the effectiveness in ulcerative colitis compared with conventional therapy: A meta-analysis. Hepatogastroenterology 2015, 62, 309–318. [Google Scholar]
- Asgharpour, A.; Cheng, J.; Bickston, S.J. Adalimumab treatment in Crohn’s disease: An overview of long-term efficacy and safety in light of the EXTEND trial. Clin. Exp. Gastroenterol. 2013, 6, 153–160. [Google Scholar]
- Altwegg, R.; Vincent, T. TNF blocking therapies and immunomonitoring in patients with inflammatory bowel disease. Mediators Inflamm. 2014, 2014, 172821. [Google Scholar] [CrossRef][Green Version]
- Targownik, L.E.; Bernstein, C.N. Infectious and malignant complications of TNF inhibitor therapy in IBD. Am. J. Gastroenterol. 2013, 108, 1835–1842. [Google Scholar] [CrossRef]
- Park, S.C.; Jeen, Y.T. Anti-integrin therapy for inflammatory bowel disease. World J. Gastroenterol. 2018, 24, 1868–1880. [Google Scholar] [CrossRef]
- Sands, B.E.; Peyrin-Biroulet, L.; Loftus, E.V., Jr.; Danese, S.; Colombel, J.F.; Toruner, M.; Jonaitis, L.; Abhyankar, B.; Chen, J.; Rogers, R.; et al. Vedolizumab versus Adalimumab for Moderate-to-Severe Ulcerative Colitis. N. Engl. J. Med. 2019, 381, 1215–1226. [Google Scholar] [CrossRef] [PubMed]
- Weaver, K.N.; Gregory, M.; Syal, G.; Hoversten, P.; Hicks, S.B.; Patel, D.; Christophi, G.; Beniwal-Patel, P.; Isaacs, K.L.; Raffals, L.; et al. Ustekinumab Is Effective for the Treatment of Crohn’s Disease of the Pouch in a Multicenter Cohort. Inflamm. Bowel Dis. 2019, 25, 767–774. [Google Scholar] [CrossRef] [PubMed]
- Na, S.Y.; Moon, W. Perspectives on Current and Novel Treatments for Inflammatory Bowel Disease. Gut Liver 2019, 13, 604–616. [Google Scholar] [CrossRef]
- Murray, A.; Nguyen, T.M.; Parker, C.E.; Feagan, B.G.; MacDonald, J.K. Oral 5-aminosalicylic acid for induction of remission in ulcerative colitis. Cochrane Database Syst. Rev. 2020, 8, CD000543. [Google Scholar]
- van Dieren, J.M.; Kuipers, E.J.; Samsom, J.N.; Nieuwenhuis, E.E.; van der Woude, C.J. Revisiting the immunomodulators tacrolimus, methotrexate, and mycophenolate mofetil: Their mechanisms of action and role in the treatment of IBD. Inflamm. Bowel Dis 2006, 12, 311–327. [Google Scholar] [CrossRef]
- Sands, B.E.; Sandborn, W.J.; Panaccione, R.; O’Brien, C.D.; Zhang, H.; Johanns, J.; Adedokun, O.J.; Li, K.; Peyrin-Biroulet, L.; Van Assche, G.; et al. Ustekinumab as Induction and Maintenance Therapy for Ulcerative Colitis. N. Engl. J. Med. 2019, 381, 1201–1214. [Google Scholar] [CrossRef]
- Salem, G.A.; Selby, G.B. Stem cell transplant in inflammatory bowel disease: A promising modality of treatment for a complicated disease course. Stem. Cell Investig. 2017, 4, 95. [Google Scholar] [CrossRef]
- Kavanagh, D.P.; Kalia, N. Hematopoietic stem cell homing to injured tissues. Stem. Cell Rev. Rep. 2011, 7, 672–682. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Montiel Mdel, P.; Gomez-Gomez, G.J.; Flores, A.I. Therapy with stem cells in inflammatory bowel disease. World J. Gastroenterol. 2014, 20, 1211–1227. [Google Scholar] [CrossRef] [PubMed]
- Burt, R.K.; Craig, R.M.; Milanetti, F.; Quigley, K.; Gozdziak, P.; Bucha, J.; Testori, A.; Halverson, A.; Verda, L.; de Villiers, W.J.; et al. Autologous nonmyeloablative hematopoietic stem cell transplantation in patients with severe anti-TNF refractory Crohn disease: Long-term follow-up. Blood 2010, 116, 6123–6132. [Google Scholar] [CrossRef] [PubMed]
- Clerici, M.; Cassinotti, A.; Onida, F.; Trabattoni, D.; Annaloro, C.; Della Volpe, A.; Rainone, V.; Lissoni, F.; Duca, P.; Sampietro, G.; et al. Immunomodulatory effects of unselected haematopoietic stem cells autotransplantation in refractory Crohn’s disease. Dig. Liver Dis. 2011, 43, 946–952. [Google Scholar] [CrossRef] [PubMed]
- Hasselblatt, P.; Drognitz, K.; Potthoff, K.; Bertz, H.; Kruis, W.; Schmidt, C.; Stallmach, A.; Schmitt-Graeff, A.; Finke, J.; Kreisel, W. Remission of refractory Crohn’s disease by high-dose cyclophosphamide and autologous peripheral blood stem cell transplantation. Aliment. Pharmacol. Ther. 2012, 36, 725–735. [Google Scholar] [CrossRef] [PubMed]
- Hawkey, C.J.; Allez, M.; Clark, M.M.; Labopin, M.; Lindsay, J.O.; Ricart, E.; Rogler, G.; Rovira, M.; Satsangi, J.; Danese, S.; et al. Autologous Hematopoetic Stem Cell Transplantation for Refractory Crohn Disease: A Randomized Clinical Trial. JAMA 2015, 314, 2524–2534. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, M.A.; Junior, R.L.K.; Piron-Ruiz, L.; Saran, P.S.; Castiglioni, L.; de Quadros, L.G.; Pinho, T.S.; Burt, R.K. Medical, ethical, and legal aspects of hematopoietic stem cell transplantation for Crohn’s disease in Brazil. World J. Stem. Cells 2020, 12, 1113–1123. [Google Scholar] [CrossRef]
- Copelan, E.A. Hematopoietic stem-cell transplantation. N. Engl. J. Med. 2006, 354, 1813–1826. [Google Scholar] [CrossRef]
- Snowden, J.A.; Saccardi, R.; Allez, M.; Ardizzone, S.; Arnold, R.; Cervera, R.; Denton, C.; Hawkey, C.; Labopin, M.; Mancardi, G.; et al. Haematopoietic SCT in severe autoimmune diseases: Updated guidelines of the European Group for Blood and Marrow Transplantation. Bone Marrow Transplant. 2012, 47, 770–790. [Google Scholar] [CrossRef]
- Christophi, G.P.; Rong, R.; Holtzapple, P.G.; Massa, P.T.; Landas, S.K. Immune markers and differential signaling networks in ulcerative colitis and Crohn’s disease. Inflamm. Bowel Dis. 2012, 18, 2342–2356. [Google Scholar] [CrossRef]
- Khalil, P.N.; Weiler, V.; Nelson, P.J.; Khalil, M.N.; Moosmann, S.; Mutschler, W.E.; Siebeck, M.; Huss, R. Nonmyeloablative stem cell therapy enhances microcirculation and tissue regeneration in murine inflammatory bowel disease. Gastroenterology 2007, 132, 944–954. [Google Scholar] [CrossRef]
- Muraro, P.A.; Douek, D.C.; Packer, A.; Chung, K.; Guenaga, F.J.; Cassiani-Ingoni, R.; Campbell, C.; Memon, S.; Nagle, J.W.; Hakim, F.T.; et al. Thymic output generates a new and diverse TCR repertoire after autologous stem cell transplantation in multiple sclerosis patients. J. Exp. Med. 2005, 201, 805–816. [Google Scholar] [CrossRef]
- Alexander, T.; Thiel, A.; Rosen, O.; Massenkeil, G.; Sattler, A.; Kohler, S.; Mei, H.; Radtke, H.; Gromnica-Ihle, E.; Burmester, G.R.; et al. Depletion of autoreactive immunologic memory followed by autologous hematopoietic stem cell transplantation in patients with refractory SLE induces long-term remission through de novo generation of a juvenile and tolerant immune system. Blood 2009, 113, 214–223. [Google Scholar] [CrossRef]
- Hossein-Khannazer, N.; Torabi, S.; Hosseinzadeh, R.; Shahrokh, S.; Asadzadeh Aghdaei, H.; Memarnejadian, A.; Kadri, N.; Vosough, M. Novel cell-based therapies in inflammatory bowel diseases: The established concept, promising results. Hum. Cell 2021, 34, 1289–1300. [Google Scholar] [CrossRef] [PubMed]
- Pockley, A.G.; Lindsay, J.O.; Foulds, G.A.; Rutella, S.; Gribben, J.G.; Alexander, T.; Snowden, J.A. Immune Reconstitution After Autologous Hematopoietic Stem Cell Transplantation in Crohn’s Disease: Current Status and Future Directions. A Review on Behalf of the EBMT Autoimmune Diseases Working Party and the Autologous Stem Cell Transplantation In Refractory CD-Low Intensity Therapy Evaluation Study Investigators. Front. Immunol. 2018, 9, 646. [Google Scholar] [PubMed]
- Leung, Y.; Geddes, M.; Storek, J.; Panaccione, R.; Beck, P.L. Hematopoietic cell transplantation for Crohn’s disease; is it time? World J. Gastroenterol. 2006, 12, 6665–6673. [Google Scholar] [CrossRef] [PubMed]
- Kotlarz, D.; Beier, R.; Murugan, D.; Diestelhorst, J.; Jensen, O.; Boztug, K.; Pfeifer, D.; Kreipe, H.; Pfister, E.D.; Baumann, U.; et al. Loss of interleukin-10 signaling and infantile inflammatory bowel disease: Implications for diagnosis and therapy. Gastroenterology 2012, 143, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Le Blanc, K.; Mougiakakos, D. Multipotent mesenchymal stromal cells and the innate immune system. Nat. Rev. Immunol. 2012, 12, 383–396. [Google Scholar] [CrossRef]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Uccelli, A.; Laroni, A.; Freedman, M.S. Mesenchymal stem cells for the treatment of multiple sclerosis and other neurological diseases. Lancet Neurol. 2011, 10, 649–656. [Google Scholar] [CrossRef]
- Burdon, T.J.; Paul, A.; Noiseux, N.; Prakash, S.; Shum-Tim, D. Bone marrow stem cell derived paracrine factors for regenerative medicine: Current perspectives and therapeutic potential. Bone Marrow Res. 2011, 2011, 207326. [Google Scholar] [CrossRef]
- Melief, S.M.; Zwaginga, J.J.; Fibbe, W.E.; Roelofs, H. Adipose tissue-derived multipotent stromal cells have a higher immunomodulatory capacity than their bone marrow-derived counterparts. Stem. Cells Transl. Med. 2013, 2, 455–463. [Google Scholar] [CrossRef]
- Duijvestein, M.; Wildenberg, M.E.; Welling, M.M.; Hennink, S.; Molendijk, I.; van Zuylen, V.L.; Bosse, T.; Vos, A.C.; de Jonge-Muller, E.S.; Roelofs, H.; et al. Pretreatment with interferon-gamma enhances the therapeutic activity of mesenchymal stromal cells in animal models of colitis. Stem Cells 2011, 29, 1549–1558. [Google Scholar] [CrossRef]
- Liang, J.; Zhang, H.; Wang, D.; Feng, X.; Wang, H.; Hua, B.; Liu, B.; Sun, L. Allogeneic mesenchymal stem cell transplantation in seven patients with refractory inflammatory bowel disease. Gut 2012, 61, 468–469. [Google Scholar] [CrossRef]
- Forbes, G.M.; Sturm, M.J.; Leong, R.W.; Sparrow, M.P.; Segarajasingam, D.; Cummins, A.G.; Phillips, M.; Herrmann, R.P. A phase 2 study of allogeneic mesenchymal stromal cells for luminal Crohn’s disease refractory to biologic therapy. Clin. Gastroenterol. Hepatol. 2014, 12, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Olmo, D.; Garcia-Arranz, M.; Garcia, L.G.; Cuellar, E.S.; Blanco, I.F.; Prianes, L.A.; Montes, J.A.; Pinto, F.L.; Marcos, D.H.; Garcia-Sancho, L. Autologous stem cell transplantation for treatment of rectovaginal fistula in perianal Crohn’s disease: A new cell-based therapy. Int J. Colorectal. Dis. 2003, 18, 451–454. [Google Scholar] [CrossRef]
- Garcia-Olmo, D.; Herreros, D.; Pascual, I.; Pascual, J.A.; Del-Valle, E.; Zorrilla, J.; De-La-Quintana, P.; Garcia-Arranz, M.; Pascual, M. Expanded adipose-derived stem cells for the treatment of complex perianal fistula: A phase II clinical trial. Dis. Colon Rectum 2009, 52, 79–86. [Google Scholar] [CrossRef]
- Guadalajara, H.; Herreros, D.; De-La-Quintana, P.; Trebol, J.; Garcia-Arranz, M.; Garcia-Olmo, D. Long-term follow-up of patients undergoing adipose-derived adult stem cell administration to treat complex perianal fistulas. Int. J. Colorectal Dis. 2012, 27, 595–600. [Google Scholar] [CrossRef] [PubMed]
- Herreros, M.D.; Garcia-Arranz, M.; Guadalajara, H.; De-La-Quintana, P.; Garcia-Olmo, D.; Group, F.C. Autologous expanded adipose-derived stem cells for the treatment of complex cryptoglandular perianal fistulas: A phase III randomized clinical trial (FATT 1: Fistula Advanced Therapy Trial 1) and long-term evaluation. Dis. Colon Rectum 2012, 55, 762–772. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.H.; Li, Q.Q.; Zhu, X.Q.; Li, Z.A.; Cai, X.M.; Pang, R.Q.; Ruan, G.P. Mechanism and therapeutic effect of umbilical cord mesenchymal stem cells in inflammatory bowel disease. Sci. Rep. 2019, 9, 17646. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Li, Y.; Yang, M.; Zou, Y.; Liu, H.; Liang, Z.; Yin, Y.; Niu, G.; Yan, Z.; Zhang, B. Efficient Differentiation of Bone Marrow Mesenchymal Stem Cells into Endothelial Cells in Vitro. Eur. J. Vasc. Endovasc. Surg. 2018, 55, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Tao, H.; Han, Z.; Han, Z.C.; Li, Z. Proangiogenic Features of Mesenchymal Stem Cells and Their Therapeutic Applications. Stem. Cells Int. 2016, 2016, 1314709. [Google Scholar] [CrossRef]
- Brittan, M.; Hunt, T.; Jeffery, R.; Poulsom, R.; Forbes, S.J.; Hodivala-Dilke, K.; Goldman, J.; Alison, M.R.; Wright, N.A. Bone marrow derivation of pericryptal myofibroblasts in the mouse and human small intestine and colon. Gut 2002, 50, 752–757. [Google Scholar] [CrossRef]
- Soontararak, S.; Chow, L.; Johnson, V.; Coy, J.; Wheat, W.; Regan, D.; Dow, S. Mesenchymal Stem Cells (MSC) Derived from Induced Pluripotent Stem Cells (iPSC) Equivalent to Adipose-Derived MSC in Promoting Intestinal Healing and Microbiome Normalization in Mouse Inflammatory Bowel Disease Model. Stem. Cells Transl. Med. 2018, 7, 456–467. [Google Scholar] [CrossRef] [PubMed]
- Yabana, T.; Arimura, Y.; Tanaka, H.; Goto, A.; Hosokawa, M.; Nagaishi, K.; Yamashita, K.; Yamamoto, H.; Adachi, Y.; Sasaki, Y.; et al. Enhancing epithelial engraftment of rat mesenchymal stem cells restores epithelial barrier integrity. J. Pathol. 2009, 218, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, A.; Kojima, Y.; Ikarashi, S.; Seino, S.; Watanabe, Y.; Kawata, Y.; Terai, S. Clinical trials using mesenchymal stem cells in liver diseases and inflammatory bowel diseases. Inflamm. Regen. 2017, 37, 16. [Google Scholar] [CrossRef] [PubMed]
- Song, W.J.; Li, Q.; Ryu, M.O.; Ahn, J.O.; Ha Bhang, D.; Chan Jung, Y.; Youn, H.Y. TSG-6 Secreted by Human Adipose Tissue-derived Mesenchymal Stem Cells Ameliorates DSS-induced colitis by Inducing M2 Macrophage Polarization in Mice. Sci. Rep. 2017, 7, 5187. [Google Scholar] [CrossRef] [PubMed]
- Djouad, F.; Charbonnier, L.M.; Bouffi, C.; Louis-Plence, P.; Bony, C.; Apparailly, F.; Cantos, C.; Jorgensen, C.; Noel, D. Mesenchymal stem cells inhibit the differentiation of dendritic cells through an interleukin-6-dependent mechanism. Stem Cells 2007, 25, 2025–2032. [Google Scholar] [CrossRef]
- Zhang, Y.; Ge, X.H.; Guo, X.J.; Guan, S.B.; Li, X.M.; Gu, W.; Xu, W.G. Bone Marrow Mesenchymal Stem Cells Inhibit the Function of Dendritic Cells by Secreting Galectin-1. Biomed Res. Int. 2017, 2017, 3248605. [Google Scholar] [CrossRef] [PubMed]
- Alves, V.B.F.; de Sousa, B.C.; Fonseca, M.T.C.; Ogata, H.; Caliari-Oliveira, C.; Yaochite, J.N.U.; Rodrigues Junior, V.; Chica, J.E.L.; da Silva, J.S.; Malmegrim, K.C.R.; et al. A single administration of human adipose tissue-derived mesenchymal stromal cells (MSC) induces durable and sustained long-term regulation of inflammatory response in experimental colitis. Clin. Exp. Immunol. 2019, 196, 139–154. [Google Scholar] [CrossRef] [PubMed]
- Mounayar, M.; Kefaloyianni, E.; Smith, B.; Solhjou, Z.; Maarouf, O.H.; Azzi, J.; Chabtini, L.; Fiorina, P.; Kraus, M.; Briddell, R.; et al. PI3kalpha and STAT1 Interplay Regulates Human Mesenchymal Stem Cell Immune Polarization. Stem Cells 2015, 33, 1892–1901. [Google Scholar] [CrossRef] [PubMed]
- Chehimi, M.; Vidal, H.; Eljaafari, A. Pathogenic Role of IL-17-Producing Immune Cells in Obesity, and Related Inflammatory Diseases. J. Clin. Med. 2017, 6, 68. [Google Scholar] [CrossRef]
- Fu, Z.W.; Zhang, Z.Y.; Ge, H.Y. Mesenteric injection of adipose-derived mesenchymal stem cells relieves experimentally-induced colitis in rats by regulating Th17/Treg cell balance. Am. J. Transl. Res. 2018, 10, 54–66. [Google Scholar]
- Terraza-Aguirre, C.; Campos-Mora, M.; Elizondo-Vega, R.; Contreras-Lopez, R.A.; Luz-Crawford, P.; Jorgensen, C.; Djouad, F. Mechanisms behind the Immunoregulatory Dialogue between Mesenchymal Stem Cells and Th17 Cells. Cells 2020, 9, 1660. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.B.; Luo, M.M.; Chen, Z.Y.; He, B.H. The Function and Role of the Th17/Treg Cell Balance in Inflammatory Bowel Disease. J. Immunol. Res. 2020, 2020, 8813558. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Shan, Q.; Sang, X.; Zhu, R.; Chen, X.; Cao, G. Total Glycosides of Peony Protects Against Inflammatory Bowel Disease by Regulating IL-23/IL-17 Axis and Th17/Treg Balance. Am. J. Chin. Med. 2019, 47, 177–201. [Google Scholar] [CrossRef] [PubMed]
- Andoh, A.; Bamba, S.; Brittan, M.; Fujiyama, Y.; Wright, N.A. Role of intestinal subepithelial myofibroblasts in inflammation and regenerative response in the gut. Pharmacol. Ther. 2007, 114, 94–106. [Google Scholar] [CrossRef]
- Gong, W.; Guo, M.; Han, Z.; Wang, Y.; Yang, P.; Xu, C.; Wang, Q.; Du, L.; Li, Q.; Zhao, H.; et al. Mesenchymal stem cells stimulate intestinal stem cells to repair radiation-induced intestinal injury. Cell Death Dis. 2016, 7, e2387. [Google Scholar] [CrossRef] [PubMed]
- Halim, N.S.; Aizat, W.M.; Yahaya, B.H. The effect of mesenchymal stem cell-secreted factors on airway epithelial repair. Regen. Med. 2019, 14, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, R.; Yajima, T.; Yamazaki, M.; Kanai, T.; Mukai, M.; Okamoto, S.; Ikeda, Y.; Hibi, T.; Inazawa, J.; Watanabe, M. Damaged epithelia regenerated by bone marrow-derived cells in the human gastrointestinal tract. Nat. Med. 2002, 8, 1011–1017. [Google Scholar] [CrossRef] [PubMed]
- Bain, C.C.; Mowat, A.M. Macrophages in intestinal homeostasis and inflammation. Immunol. Rev. 2014, 260, 102–117. [Google Scholar] [CrossRef] [PubMed]
- Song, W.J.; Li, Q.; Ryu, M.O.; Ahn, J.O.; Bhang, D.H.; Jung, Y.C.; Youn, H.Y. TSG-6 released from intraperitoneally injected canine adipose tissue-derived mesenchymal stem cells ameliorate inflammatory bowel disease by inducing M2 macrophage switch in mice. Stem. Cell Res. Ther. 2018, 9, 91. [Google Scholar] [CrossRef]
- Mao, F.; Wu, Y.; Tang, X.; Kang, J.; Zhang, B.; Yan, Y.; Qian, H.; Zhang, X.; Xu, W. Exosomes Derived from Human Umbilical Cord Mesenchymal Stem Cells Relieve Inflammatory Bowel Disease in Mice. Biomed. Res. Int. 2017, 2017, 5356760. [Google Scholar] [CrossRef] [PubMed]
- Vasandan, A.B.; Jahnavi, S.; Shashank, C.; Prasad, P.; Kumar, A.; Prasanna, S.J. Human Mesenchymal stem cells program macrophage plasticity by altering their metabolic status via a PGE2-dependent mechanism. Sci. Rep. 2016, 6, 38308. [Google Scholar] [CrossRef] [PubMed]
- Rescigno, M.; Di Sabatino, A. Dendritic cells in intestinal homeostasis and disease. J. Clin. Investig. 2009, 119, 2441–2450. [Google Scholar] [CrossRef] [PubMed]
- Hostmann, A.; Kapp, K.; Beutner, M.; Ritz, J.P.; Loddenkemper, C.; Ignatius, R.; Duchmann, R.; Daum, S.; Grone, J.; Hotz, H.; et al. Dendritic cells from human mesenteric lymph nodes in inflammatory and non-inflammatory bowel diseases: Subsets and function of plasmacytoid dendritic cells. Immunology 2013, 139, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Larabi, A.; Barnich, N.; Nguyen, H.T.T. New insights into the interplay between autophagy, gut microbiota and inflammatory responses in IBD. Autophagy 2020, 16, 38–51. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Jung, J.; Cho, K.J.; Choi, J.H.; Lee, H.S.; Kim, G.J.; Lee, S.G. Immunomodulatory Effects of Placenta-derived Mesenchymal Stem Cells on T Cells by Regulation of FoxP3 Expression. Int. J. Stem. Cells 2018, 11, 196–204. [Google Scholar] [CrossRef]
- Watanabe, Y.; Tsuchiya, A.; Terai, S. The development of mesenchymal stem cell therapy in the present, and the perspective of cell-free therapy in the future. Clin. Mol. Hepatol. 2021, 27, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Ocansey, D.K.W.; Zhang, L.; Wang, Y.; Yan, Y.; Qian, H.; Zhang, X.; Xu, W.; Mao, F. Exosome-mediated effects and applications in inflammatory bowel disease. Biol. Rev. 2020, 95, 1287–1307. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Zhu, Q.; Zhang, Y.; Bian, Q.; Hong, Y.; Shen, Z.; Xu, H.; Rui, K.; Yin, K.; Wang, S. Olfactory Ecto-Mesenchymal Stem Cell-Derived Exosomes Ameliorate Experimental Colitis via Modulating Th1/Th17 and Treg Cell Responses. Front. Immunol. 2020, 11, 598322. [Google Scholar] [CrossRef]
- Bassi, G.; Pacelli, L.; Carusone, R.; Zanoncello, J.; Krampera, M. Adipose-derived stromal cells (ASCs). Transfus. Apher. Sci. 2012, 47, 193–198. [Google Scholar] [CrossRef]
- Nasuno, M.; Arimura, Y.; Nagaishi, K.; Isshiki, H.; Onodera, K.; Nakagaki, S.; Watanabe, S.; Idogawa, M.; Yamashita, K.; Naishiro, Y.; et al. Mesenchymal stem cells cancel azoxymethane-induced tumor initiation. Stem. Cells 2014, 32, 913–925. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; He, X.; He, X.; Chen, X.; Lin, X.; Zou, Y.; Wu, X.; Lan, P. Bone marrow mesenchymal stem cells ameliorate colitis-associated tumorigenesis in mice. Biochem. Biophys. Res. Commun. 2014, 450, 1402–1408. [Google Scholar] [CrossRef] [PubMed]
- Lalu, M.M.; McIntyre, L.; Pugliese, C.; Fergusson, D.; Winston, B.W.; Marshall, J.C.; Granton, J.; Stewart, D.J.; Canadian Critical Care Trials, G. Safety of cell therapy with mesenchymal stromal cells (SafeCell): A systematic review and meta-analysis of clinical trials. PLoS ONE 2012, 7, e47559. [Google Scholar] [CrossRef] [PubMed]
- Young, R.A. Control of the embryonic stem cell state. Cell 2011, 144, 940–954. [Google Scholar] [CrossRef]
- Sanchez, L.; Gutierrez-Aranda, I.; Ligero, G.; Rubio, R.; Munoz-Lopez, M.; Garcia-Perez, J.L.; Ramos, V.; Real, P.J.; Bueno, C.; Rodriguez, R.; et al. Enrichment of human ESC-derived multipotent mesenchymal stem cells with immunosuppressive and anti-inflammatory properties capable to protect against experimental inflammatory bowel disease. Stem. Cells 2011, 29, 251–262. [Google Scholar] [CrossRef]
- Srivastava, A.S.; Feng, Z.; Mishra, R.; Malhotra, R.; Kim, H.S.; Carrier, E. Embryonic stem cells ameliorate piroxicam-induced colitis in IL10-/- KO mice. Biochem. Biophys. Res. Commun. 2007, 361, 953–959. [Google Scholar] [CrossRef] [PubMed]
- Murry, C.E.; Keller, G. Differentiation of embryonic stem cells to clinically relevant populations: Lessons from embryonic development. Cell 2008, 132, 661–680. [Google Scholar] [CrossRef] [PubMed]
| Treatments | Molecule Group | Compound | Mechanisms of Action | Limitations | References |
|---|---|---|---|---|---|
| Small molecule medications | Amino Salicylate | 5-ASA | Antibacterial action Inhibits prostaglandin and NF-κB activity Scavenge reactive oxygen metabolites | Adverse gastrointestinal events: nausea, and vomiting | [70] |
| Glucocorticoids | Prednisone Hydrocortisone | Interact with glucocorticoid receptors in the nucleus | Osteoporosis, systemic edema, infections, and a high drug dependence | [71,72,73] | |
| Inhibit the expression of adhesion molecules | |||||
| Prevent the transport of inflammatory cells to the intestine | |||||
| Immunomodulators | Methotrexate | Inhibit several enzymes involved in the folate metabolic pathway | Nausea, vomiting, fatigue, diarrhea, leucopenia, liver fibrosis, allergic pneumonia, teratogenicity, and certain nephrotoxicity | [74,75] | |
| Play an anti-inflammatory role by inhibiting the production of IL-1, IL-2, IL-6, and IL-8 | |||||
| Biological agents | Tumor necrosis factor inhibitors | Infliximab Adalimumab Golimumab Certolizumab pegol | Affect the downstream effects of TNF on cytokines, chemokines, acute phase reactants, apoptosis, and inflammation | Some patients present primary resistance, and may increase the risk of certain uncommon classes of infections and malignancies | [76,77,78,79] |
| Integrin receptor antagonists | Natalizumab | Induce and maintaining clinical remission of CD through anti-α4 integrins interacts with VCAM-1 | Affect cerebral antiviral immunity, and can cause a fatal brain infection due to the reactivation of the JC virus | [80,81] | |
| Interleukin antagonists | Ustekinumab | Bind to the p40 subunit shared by both and preventing its binding to the IL-12β1 receptor on the cell surface | Potential risks are not known | [82,83] | |
| Inhibit pro-inflammatory cytokines | |||||
| Reduce T-cell activation |
| Types | Mechanism | Advantage | Deficiency | References |
|---|---|---|---|---|
| Autologous HSCT | The removal of reactive T-lymphocytes by chemotherapy and replacing them with more immune-tolerant lymphocytes to rebuild the immune system | Provide long-term remission | It is not possible to change the patient’s genetic susceptibility at the genetic level | [101,102] |
| Allogeneic HSCT | A complete replacement of the recipient’s immune cells with the donor’s immune cells | Alter the patient’s susceptibility to IBD at the genetic level | High lethality, and increases the set of risks of graft rejection | [101,103] |
| Biological Effects | Mechanism of Action | References |
|---|---|---|
| Local microcirculation construction | Differentiation into vascular endothelial cells, and form vessel like structures in vitro | [118] |
| Induce EC proliferation by producing multiple angiogenic factors | [119] | |
| Directly secrete and paracrine a variety of cytokines | [120] | |
| Fixation repair | Activate the Wnt/β-catenin signaling pathway, increase the number of Lgr5+ ISCs | [121] |
| Repair the integrity of the intestinal epithelial barrier through recombinant cellular | [122] | |
| Inhibit apoptosis | [123] | |
| Immunomodulation | Alter the macrophage phenotype from M1 to M2 | [124] |
| Upregulate Gal-1 expression, inhibit DC maturation, in-crease anti-inflammatory factors, diminish T-cell activation | [125,126] | |
| Regulate the ratio of Th1 to Th2 | [127,128] | |
| Adjust the balance between Th17/Tregs | [129,130,131,132,133] |
| Disease | Clinical Indication | Source of Stem Cells | Number of Patients Enrolled | Follow-Up Period | Outcome | Author and Time | Reference |
|---|---|---|---|---|---|---|---|
| Refractory CD | Have failed treatment with corticosteroids, mesalamine, met- ronidazole, azathioprine (or 6-mercaptopurine), and mono- clonal antibody to TNF receptor (infliximab). | Autologous HSCT | 12 | 7–37 months | 11 patients entered a sustained remission. After a median follow-up of 18.5 months, only 1 patient has developed a recurrence of active CD, which occurred 15 months after HSCT | Oyama, Y. et al., 2005 | [8] |
| Severe CD | Refractory to conventional therapies including anti-TNF inhibitor | Autologous nonmyeloablative HSCT | 24 | 1–5 years | Eighteen of 24 patients are 5 or more years after transplantation | Burt, R.K. et al., 2010 | [90] |
| Active moderate-severe CD | Refractory or intolerant to various conventional treatment schedules including corticosteroids and at least 2 immunosuppressors | Autologous HSCT | 7 | 1 year | Most of whom showed clinical and endoscopic complete remission and were maintained for one year without further treatment | Clerici, M. et al., 2011 | [91] |
| Refractory CD | Intolerance or failure of conventional therapies including immunosuppressors and at least one anti-TNF antibody | High-dose immunosuppression and autologous peripheral blood stem cell transplantation (autoPBSCT) | 12 | 0.5–10.3 years | 5 patients achieved a clinical and endoscopic remission within 6 months after autoPBSCT. However, relapses occurred in 7/9 patients during follow-up, but disease activity could be controlled by low-dose corticosteroids and conventional immunosuppressive therapy | Hasselblatt, P. et al., 2012 | [92] |
| Refractory CD | Treatment with 3 or more immunosuppressants or biological agents and corticosteroids leads to impaired quality of life and is not suitable for surgery | Autologous HSCT | 45 | 1 year | Compared with conventional therapy, did not result in a statistically significant improvement in sustained disease remission at 1 year and was associated with significant toxicity | Hawkey, C.J. et al., 2015 | [93] |
| Early onset IBD | IL-10 gene defect | Allogeneic HSCT | 66 | 2 years | Allogeneic HSCT was performed in 5 patients to induce sustained clinical remission | Kotlarz, D. et al., 2012 | [104] |
| Refractory IBD | 4 CD, 3 UC | Allogeneic MSCT | 7 | Mean 19 mouths | Diarrhea frequency and abdominal pain/ cramps gradually improved in all the seven patients, accompanied by a significant reduction in CD Activity Index scores in CD patients and Clinical Activity Index scores in UC patients | Liang, J. et al., | [111] |
| Luminal CD | With infliximab- or adalimumab-refractory, endoscopically confirmed | Allogeneic MSCT | 16 | 42 days | In a phase 2 study, administration of allogeneic MSCs reduced CDAI and CDEIS scores in patients with luminal CD refractory to biologic therapy | Forbes, G.M. et al., 2013 | [112] |
| CD | Had a recurrent rectovaginal fistula | ASCs | 1 | 3 mouths | Since the surgical procedure 3 month ago the patient has not experienced vaginal flatus or fecal incontinence through her vagina | Garcia-Olmo, D. et al., 2003 | [113] |
| Complex perianal fistulas | Had a complex perianal fistula (either of cryptoglandular origin or associated with CD) with a visible external opening. | Administration of expanded ASCs in combination with fibrin glue | 24 | 1 year | Combination therapy appears to achieve higher rates of healing than fibrin glue alone | Garcia-Olmo, D. et al., 2009 | [114] |
| perianal fistulas | Had received at least one dose of treatment (ASCs plus fibrin glue or fibrin glue alone) | ASCs plus fibrin glue or fibrin glue alone | 49 | 3 years | A low proportion of the stem cell-treated patients with closure after the procedure remained free of recurrence after more than 3 years of follow-up | Guadalajara, H. et al., 2011 | [115] |
| complex fistula-in-ano | Complex fistula-in-ano | A dose of 20 or 60 million ASCs alone or in combination with fibrin glue | 200 | 1 year | Achieving healing rates of approximately 40% at 6 months and of more than 50% at 1-year follow-up | Herreros, M.D. et al., 2012 | [116] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, H.-M.; Yuan, S.; Meng, H.; Hou, X.-T.; Li, J.; Xue, J.-C.; Li, Y.; Wang, Q.; Nan, J.-X.; Jin, X.-J.; et al. Stem Cell-Based Therapies for Inflammatory Bowel Disease. Int. J. Mol. Sci. 2022, 23, 8494. https://doi.org/10.3390/ijms23158494
Zhang H-M, Yuan S, Meng H, Hou X-T, Li J, Xue J-C, Li Y, Wang Q, Nan J-X, Jin X-J, et al. Stem Cell-Based Therapies for Inflammatory Bowel Disease. International Journal of Molecular Sciences. 2022; 23(15):8494. https://doi.org/10.3390/ijms23158494
Chicago/Turabian StyleZhang, Hua-Min, Shuo Yuan, Huan Meng, Xiao-Ting Hou, Jiao Li, Jia-Chen Xue, You Li, Qi Wang, Ji-Xing Nan, Xue-Jun Jin, and et al. 2022. "Stem Cell-Based Therapies for Inflammatory Bowel Disease" International Journal of Molecular Sciences 23, no. 15: 8494. https://doi.org/10.3390/ijms23158494
APA StyleZhang, H.-M., Yuan, S., Meng, H., Hou, X.-T., Li, J., Xue, J.-C., Li, Y., Wang, Q., Nan, J.-X., Jin, X.-J., & Zhang, Q.-G. (2022). Stem Cell-Based Therapies for Inflammatory Bowel Disease. International Journal of Molecular Sciences, 23(15), 8494. https://doi.org/10.3390/ijms23158494






