Dynamic Changes in Seed Germination under Low-Temperature Stress in Maize
Abstract
:1. Introduction
2. Results
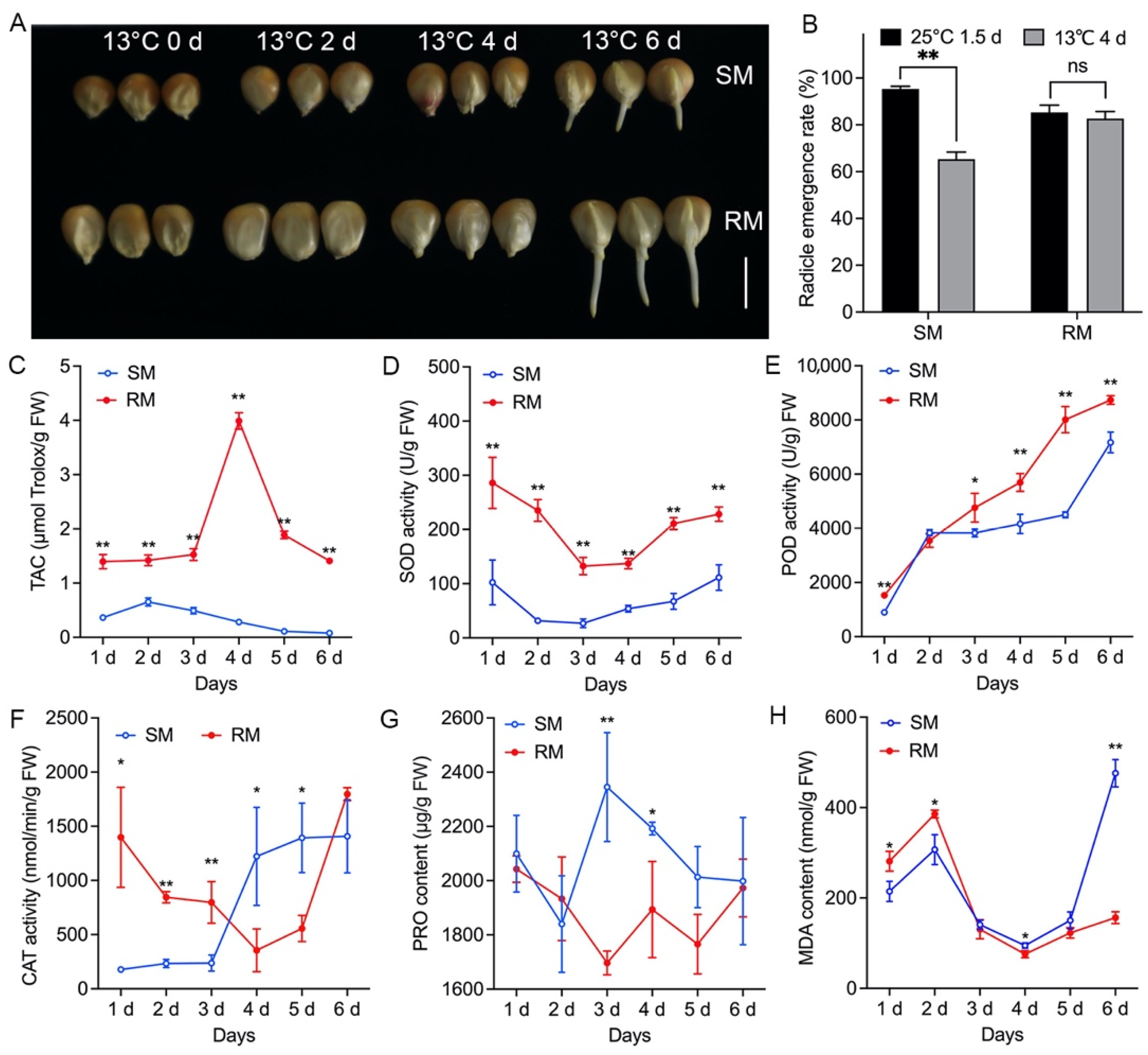
2.1. Effects of Low-Temperature Stress on Seed Germination in Two Maize Inbred Lines
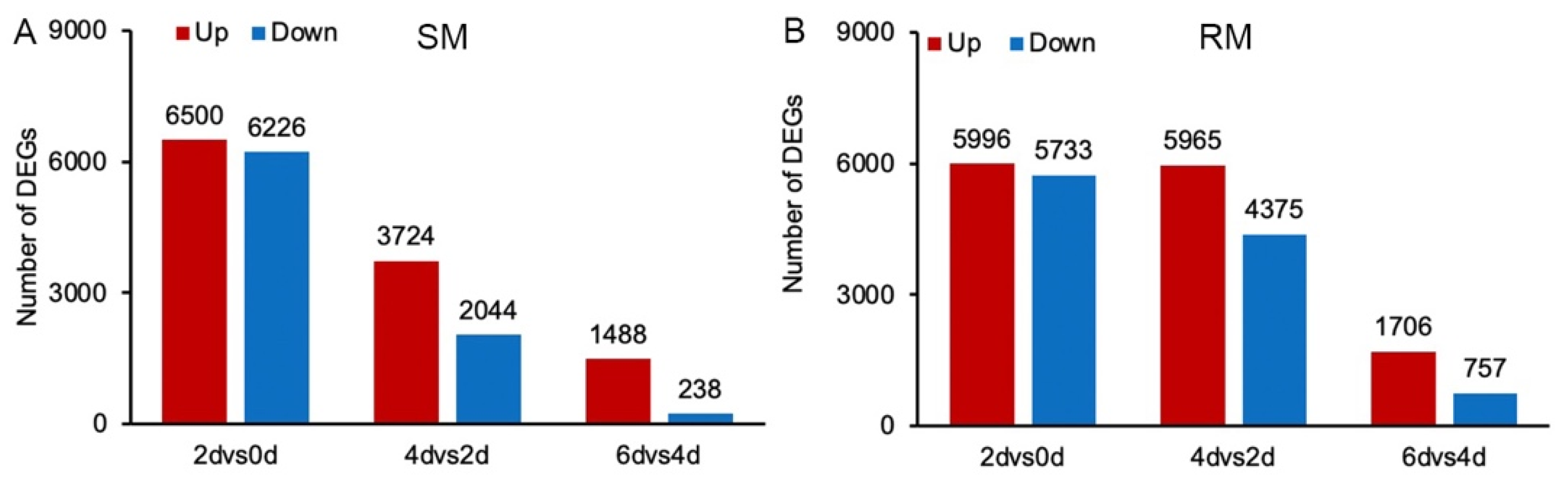
2.2. Transcriptome Analysis of Seed Germination in Two Maize Inbred Lines under Low-Temperature Stress
2.3. Peroxidase Activity-Related Genes Are Involved in Response to Low-Temperature Stress during Seed Germination
2.4. Photosystem II (PSII) and Heat Shock Protein Binding-Related GO Terms in the SM Line Are More Vulnerable to Low-Temperature Stress at the Early Stage of Seed Germination
2.5. Low-Temperature Stress May Delay Seed Germination by the Downregulation of Ribosome-Related Genes
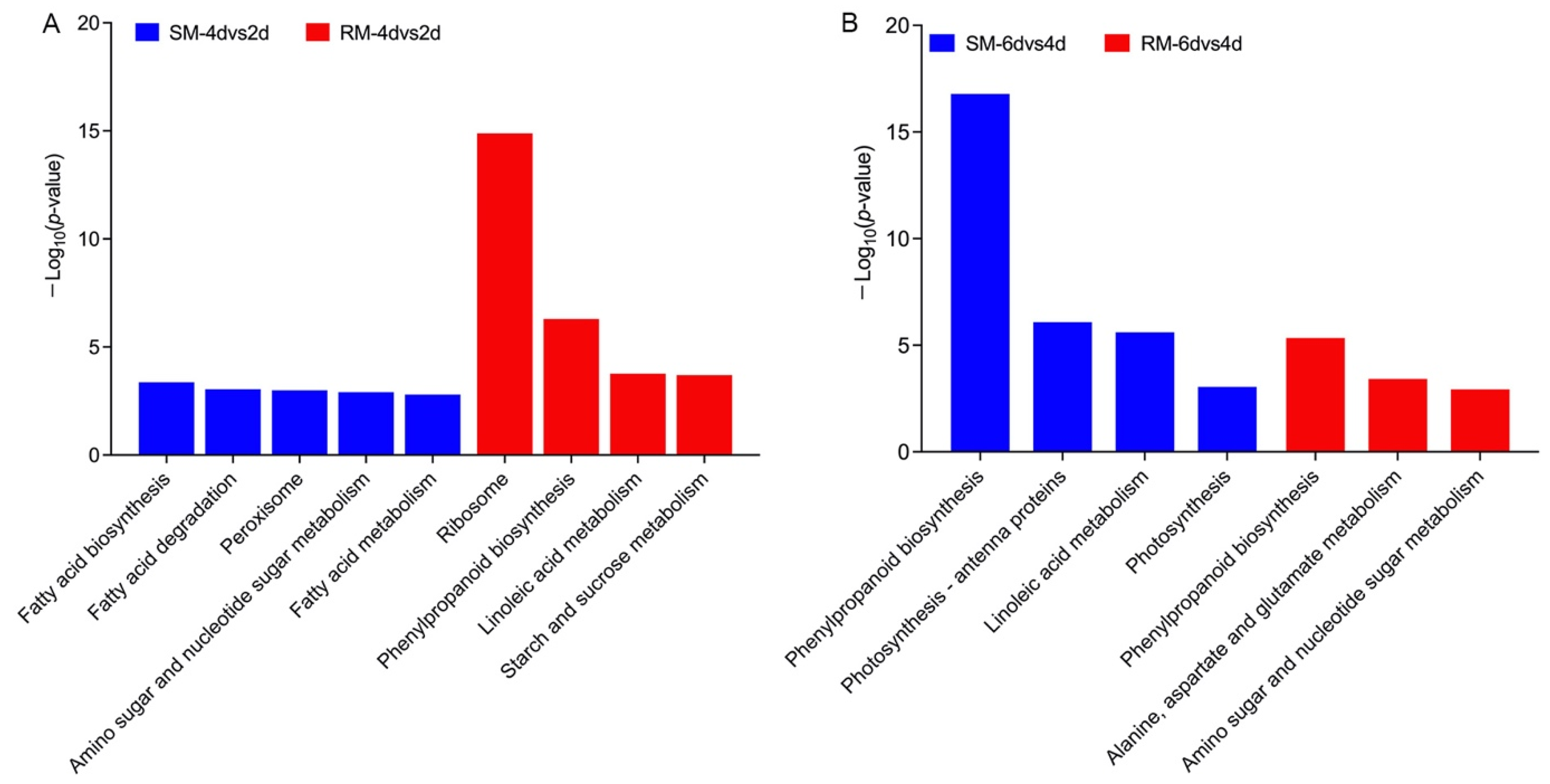
2.6. Kyoto Encyclopedia of Genes and Genomes (KEGG) Enrichment Analysis of the DEGs in Dynamic Changes in Seed Germination under Low-Temperature Stress
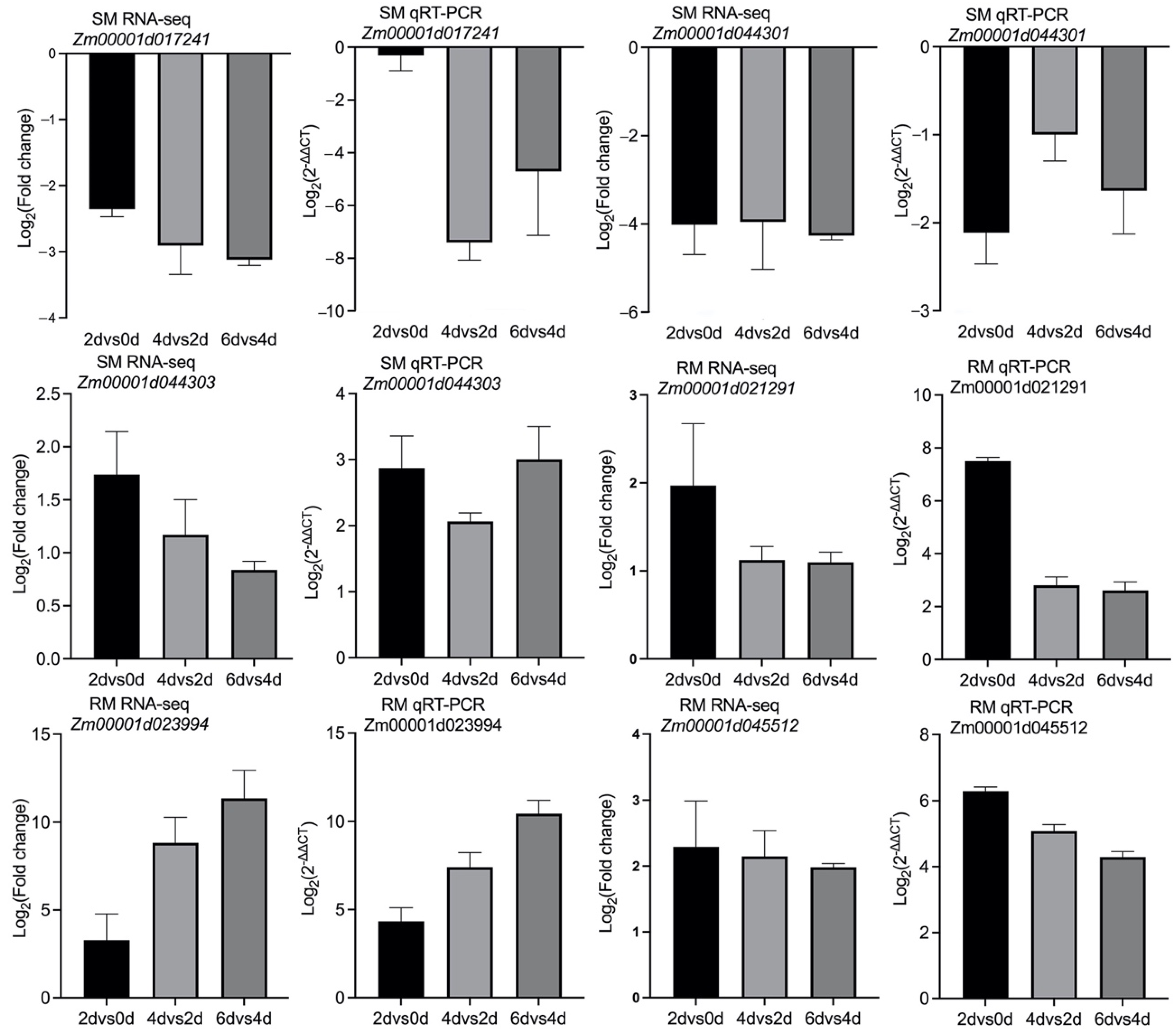
2.7. Verification of the RNA-seq Data by Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR) Analysis
3. Discussion
4. Materials and Methods
4.1. Plant Materials
4.2. Determination of Antioxidant Enzyme Activities, PRO Content, and Lipid Peroxidation
4.2.1. Measurement of TAC
4.2.2. Measurement of SOD Activity
4.2.3. Measurement of POD Activity
4.2.4. Measurement of CAT Activity
4.2.5. Measurement of PRO Content
4.2.6. Determination of Lipid Peroxidation
4.3. RNA-seq and Data Analysis
4.4. Verification of DEGs by a qRT-PCR Analysis
4.5. IBM SPSS Statistical Version 21.0 Software
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| RM | low-temperature-resistant maize |
| SM | low-temperature-sensitive maize |
| MDA | Malondialdehyde |
| GO | Gene ontology |
| POD | peroxidase |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| DEGs | differentially expressed genes |
| ROS | reactive oxygen species |
| SOD | superoxide dismutase |
| POD | peroxidase |
| CAT | catalase |
| GPX | glutathione peroxidase |
| VB6 | vitamin B6 |
| PRO | proline |
| TAC | total antioxidant capacity |
| qRT-PCR | quantitative real-time polymerase chain reaction |
| PSII | Photosystem II |
| HSP | Heat shock proteins |
| ABTS | 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt |
References
- Walbot, V. 10 Reasons to be Tantalized by the B73 Maize Genome. PLoS Genet. 2009, 5, e1000723. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- June, C.; Page, S.E.E.L.; Pasternak, S.; Liang, C.; Zhang, J.; Fulton, L.; Graves, T.A.; Minx, P.; Reily, A.D.; Courtney, L.; et al. The B73 Maize Genome: Complexity, Diversity, and Dynamics. Science 2009, 1912, 1112–1115. [Google Scholar]
- Tenaillon, M.I.; Charcosset, A. A European perspective on maize history. Comptes Rendus-Biol. 2011, 334, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Zeng, R.; Li, Z.; Shi, Y.; Fu, D.; Yin, P.; Cheng, J.; Jiang, C.; Yang, S. Natural variation in a type—A response regulator confers maize chilling tolerance. Nat Commun. 2021, 12, 4713. [Google Scholar] [CrossRef]
- Sowiński, P.; Rudzińska-Langwald, A.; Adamczyk, J.; Kubica, I.; Fronk, J. Recovery of maize seedling growth, development and photosynthetic efficiency after initial growth at low temperature. J Plant Physiol. 2005, 162, 67–80. [Google Scholar] [CrossRef]
- Grzybowski, M.; Adamczyk, J.; Jończyk, M.; Sobkowiak, A.; Szczepanik, J.; Frankiewicz, K.; Fronk, J.; Sowiński, P. Increased photosensitivity at early growth as a possible mechanism of maize adaptation to cold springs. J Exp Botany 2019, 70, 2887–2904. [Google Scholar] [CrossRef] [Green Version]
- Bewley, J.D. Seed germination and dormancy. Plant Cell 1997, 9, 1055–1066. [Google Scholar] [CrossRef] [Green Version]
- Foyer, C.H.; Vanacker, H.; Gomez, L.D.; Harbinson, J. Regulation of photosynthesis and antioxidant metabolism in maize leaves at optimal and chilling temperatures: Review. Plant Physiol. Biochem. 2002, 40, 659–668. [Google Scholar] [CrossRef]
- Kołodziejczyk, I.; Kaźmierczak, A.; Posmyk, M.M. Melatonin application modifies antioxidant defense and induces endoreplication in maize seeds exposed to chilling stress. Int. J. Mol. Sci. 2021, 22, 8628. [Google Scholar] [CrossRef]
- Greaves, J.A. Improving suboptimal temperature tolerance in maize—The search for variation. J. Exp. Botany 1996, 47, 307–323. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, J.; Xu, Q.; Wang, D.; Di, H.; Huang, J.; Yang, X.; Wang, Z.; Zhang, L.; Dong, L.; et al. Identification of candidate tolerance genes to low-temperature during maize germination by GWAS and RNA-seq approaches. BMC Plant Biol. 2020, 20, 333. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Liu, D.; Chong, K. Cold signaling in plants: Insights into mechanisms and regulation. J. Integr. Plant Biol. 2018, 60, 745–756. [Google Scholar] [CrossRef] [Green Version]
- Crèvecoeur, M.; Deltour, R.; Bronchart, R. Effects of subminimal temperature on physiology and ultrastructure of Zea mays embryo during germination. Can. J. Botany 1983, 61, 1117–1125. [Google Scholar] [CrossRef]
- Bafana, A.; Dutt, S.; Kumar, S.; Ahuja, P.S. Superoxide dismutase: An industrial perspective. Crit. Rev. Biotechnol. 2011, 31, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Mittler, R.; Vanderauwera, S.; Gollery, M.; Van Breusegem, F. Reactive oxygen gene network of plants. Trends Plant Sci. 2004, 9, 490–498. [Google Scholar] [CrossRef]
- Filiz, E.; Tombuloğlu, H. Genome-wide distribution of superoxide dismutase (SOD) gene families in Sorghum bicolor. Turk. J. Biology 2015, 39, 49–59. [Google Scholar] [CrossRef]
- Denslow, S.A.; Rueschhoff, E.E.; Daub, M.E. Regulation of the Arabidopsis thaliana vitamin B6 biosynthesis genes by abiotic stress. Plant Physiol. Biochem. 2007, 45, 152–161. [Google Scholar] [CrossRef]
- Browning, K.S.; Bailey-Serres, J. Mechanism of Cytoplasmic mRNA Translation. Arab. Book 2015, 13, 1–39. [Google Scholar] [CrossRef] [Green Version]
- Sanchez, C.G.; Teixeira, F.K.; Czech, B.; Preall, J.B.; Zamparini, A.L.; Seifert, J.R.K.; Malone, C.D.; Hannon, G.J.; Lehmann, R. Regulation of Ribosome Biogenesis and Protein Synthesis Controls Germline Stem Cell Differentiation. Cell Stem Cell 2016, 18, 276–290. [Google Scholar] [CrossRef] [Green Version]
- Sinturel, F.; Gerber, A.; Mauvoisin, D.; Wang, J.; Gatfield, D.; Stubblefield, J.J.; Green, C.B.; Gachon, F.; Schibler, U. Diurnal Oscillations in Liver Mass and Cell Size Accompany Ribosome Assembly Cycles. Cell 2017, 169, 651–663. [Google Scholar] [CrossRef] [Green Version]
- Ohbayashi, I.; Konishi, M.; Ebine, K.; Sugiyama, M. Genetic identification of Arabidopsis RID2 as an essential factor involved in pre-rRNA processing. Plant J. 2011, 67, 49–60. [Google Scholar] [CrossRef]
- Al Refaii, A.; Alix, J.H. Ribosome biogenesis is temperature-dependent and delayed in Escherichia coli lacking the chaperones DnaK or DnaJ. Mol. Microbiol. 2009, 71, 748–762. [Google Scholar] [CrossRef] [PubMed]
- Hang, R.; Wang, Z.; Deng, X.; Liu, C.; Yan, B.; Yang, C.; Song, X.; Mo, B.; Cao, X. Ribosomal RNA Biogenesis and Its Response to Chilling Stress in Oryza sativa. Plant Physiol. 2018, 177, 381–397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, T.; Zhang, J.; Cao, J.; Cai, Q.; Li, X.; Sun, Y.; Li, S.; Li, Y.; Hu, G.; Cao, S.; et al. Leaf transcriptomic response mediated by cold stress in two maize inbred lines with contrasting tolerance levels. Genomics 2021, 113, 782–794. [Google Scholar] [CrossRef] [PubMed]
- Meng, A.; Wen, D.; Zhang, C. Maize Seed Germination under Low-Temperature Stress Impacts Seedling Growth under Normal Temperature by Modulating Photosynthesis and Antioxidant Metabolism. Front. Plant Sci. 2022, 13, 843033. [Google Scholar] [CrossRef]
- Gechev, T.; Willekens, H.; Van Montagu, M.; Inzé, D.; Van Camp, W.; Toneva, V.; Minkov, I. Different responses of tobacco antioxidant enzymes to light and chilling stress. J. Plant Physiol. 2003, 160, 509–515. [Google Scholar] [CrossRef]
- Li, Y.; Wang, X.; Ban, Q.; Zhu, X.; Jiang, C.; Wei, C.; Bennetzen, J.L. Comparative transcriptomic analysis reveals gene expression associated with cold adaptation in the tea plant Camellia sinensis. BMC Genom. 2019, 20, 624. [Google Scholar] [CrossRef] [Green Version]
- Møller, I.M.; Jensen, P.E.; Hansson, A. Oxidative modifications to cellular components in plants. Ann. Rev. Plant Biol. 2007, 58, 459–481. [Google Scholar] [CrossRef] [Green Version]
- Chakraborty, A.; Bhattacharjee, S. Differential competence of redox-regulatory mechanism under extremes of temperature determines growth performances and cross tolerance in two indica rice cultivars. J. Plant Physiol. 2015, 176, 65–77. [Google Scholar] [CrossRef]
- Denslow, S.A.; Walls, A.A.; Daub, M.E. Regulation of biosynthetic genes and antioxidant properties of vitamin B6 vitamers during plant defense responses. Phys. Mol. Plant Pathol. 2005, 66, 244–255. [Google Scholar] [CrossRef]
- Huang, S.H.; Zhang, J.Y.; Wang, L.H.; Huang, L.Q. Effect of abiotic stress on the abundance of different vitamin B6 vitamers in tobacco plants. Plant Phys. Biochem. 2013, 66, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Aminaka, R.; Taira, Y.; Kashino, Y.; Koike, H.; Satoh, K. Acclimation to the growth temperature and thermosensitivity of photosystem II in a mesophilic cyanobacterium, Synechocystis sp. PCC6803. Plant Cell Physiol. 2006, 47, 1612–1621. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rowland, J.G.; Pang, X.; Suzuki, I.; Murata, N.; Simon, W.J.; Slabas, A.R. Identification of components associated with thermal acclimation of photosystem II in Synechocystis sp. PCC6803. PLoS ONE 2010, 5, e10511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chauhan, H.; Khurana, N.; Nijhavan, A.; Khurana, J.P.; Khurana, P. The wheat chloroplastic small heat shock protein (sHSP26) is involved in seed maturation and germination and imparts tolerance to heat stress. Plant Cell Environ. 2012, 35, 1912–1931. [Google Scholar] [CrossRef]
- Jiang, C.; Xu, J.; Zhang, H.; Zhang, X.; Shi, J.; Li, M.; Ming, F. A cytosolic class i small heat shock protein, RcHSP17.8, of Rosa chinensis confers resistance to a variety of stresses to Escherichia coli, yeast and Arabidopsis thaliana. Plant Cell Environ. 2009, 32, 1046–1059. [Google Scholar] [CrossRef]
- Sun, L.; Liu, Y.; Kong, X.; Zhang, D.; Pan, J.; Zhou, Y.; Wang, L.; Li, D.; Yang, X. ZmHSP16.9, a cytosolic class I small heat shock protein in maize (Zea mays), confers heat tolerance in transgenic tobacco. Plant Cell Rep. 2012, 31, 1473–1484. [Google Scholar] [CrossRef]
- Xu, J.; Xue, C.; Xue, D.; Zhao, J.; Gai, J.; Guo, N.; Xing, H. Overexpression of GmHsp90s, a Heat Shock Protein 90 (Hsp90) Gene Family Cloning from Soybean, Decrease Damage of Abiotic Stresses in Arabidopsis thaliana. PLoS ONE 2013, 8, e69810. [Google Scholar] [CrossRef]
- Kong, F.; Deng, Y.; Zhou, B.; Wang, G.; Wang, Y.; Meng, Q. A chloroplast-targeted DnaJ protein contributes to maintenance of photosystem II under chilling stress. J. Exp. Botany 2014, 65, 143–158. [Google Scholar] [CrossRef] [Green Version]
- Cheong, B.E.; Beine-Golovchuk, O.; Gorka, M.; Ho, W.W.H.; Martinez-Seidel, F.; Firmino, A.A.P.; Skirycz, A.; Roessner, U.; Kopka, J. Arabidopsis REI-LIKE proteins activate ribosome biogenesis during cold acclimation. Sci. Rep. 2021, 11, 2410. [Google Scholar] [CrossRef]
- Wen, D.; Hou, H.; Meng, A.; Meng, J.; Xie, L.; Zhang, C. Rapid evaluation of seed vigor by the absolute content of protein in seed within the same crop. Sci. Rep. 2018, 8, 5569. [Google Scholar] [CrossRef]
- Scalzo, J.; Politi, A.; Pellegrini, N.; Mezzetti, B.; Battino, M. Plant genotype affects total antioxidant capacity and phenolic contents in fruit. Nutrition 2005, 21, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Giannopolitis, C.N.; Ries, S.K. Superoxide Dismutases I. Occurrence in Higher Plants. Plant Physiol. 1977, 59, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Nakano, Y.; Asada, K. Hydrogen Peroxide is Scavenged by Ascorbate-specific Peroxidase in Spinach Chloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar] [CrossRef]
- Havir, E.A.; McHale, N.A. Biochemical and Developmental Characterization of Multiple Forms of Catalase in Tobacco Leaves. Plant Physiol. 1987, 84, 450–455. [Google Scholar] [CrossRef] [Green Version]
- Bates, L.S.; Waldren, R.P.; Teare, I.D. Rapid determination of free proline for water-stress studies Summary. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Checchio, M.V.; de Cássia Alves, R.; de Oliveira, K.R.; Moro, G.V.; dos Santos, D.M.M.; Gratão, P.L. Enhancement of salt tolerance in corn using Azospirillum brasilense: An approach on antioxidant systems. J. Plant Res. 2021, 134, 1279–1289. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, Y.; Yan, Y.; Peng, H.; Long, Y.; Zhang, Y.; Jiang, Z.; Liu, P.; Zou, C.; Peng, H.; et al. Transcriptome sequencing analysis of maize embryonic callus during early redifferentiation. BMC Genom. 2019, 20, 159. [Google Scholar] [CrossRef] [Green Version]
- Stamm, K.; Tomita-Mitchell, A.; Bozdag, S. GSEPD: A Bioconductor package for RNA-seq gene set enrichment and projection display. BMC Bioinform. 2019, 20, 115. [Google Scholar] [CrossRef] [Green Version]
- Harris, M.A.; Deegan, J.I.; Ireland, A.; Lomax, J.; Ashburner, M.; Tweedie, S.; Carbon, S.; Lewis, S.; Mungall, C.; Day-Richter, J.; et al. The Gene Ontology project in 2008. Nucleic Acids Res. 2008, 36, 440–444. [Google Scholar] [CrossRef] [Green Version]
- Yu, G.; Wang, L.G.; Han, Y.; He, Q.Y. ClusterProfiler: An R package for comparing biological themes among gene clusters. OMICS A J. Integr. Biol. 2012, 16, 284–287. [Google Scholar] [CrossRef]
- Zhang, Z.; Yang, J.; Wu, Y. Transcriptional regulation of zein gene expression in maize through the additive and synergistic action of opaque2, prolamine-box binding factor, and O2 heterodimerizing proteins. Plant Cell. 2015, 27, 1162–1172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meng, A.; Wen, D.; Zhang, C. Dynamic Changes in Seed Germination under Low-Temperature Stress in Maize. Int. J. Mol. Sci. 2022, 23, 5495. https://doi.org/10.3390/ijms23105495
Meng A, Wen D, Zhang C. Dynamic Changes in Seed Germination under Low-Temperature Stress in Maize. International Journal of Molecular Sciences. 2022; 23(10):5495. https://doi.org/10.3390/ijms23105495
Chicago/Turabian StyleMeng, Aiju, Daxing Wen, and Chunqing Zhang. 2022. "Dynamic Changes in Seed Germination under Low-Temperature Stress in Maize" International Journal of Molecular Sciences 23, no. 10: 5495. https://doi.org/10.3390/ijms23105495
APA StyleMeng, A., Wen, D., & Zhang, C. (2022). Dynamic Changes in Seed Germination under Low-Temperature Stress in Maize. International Journal of Molecular Sciences, 23(10), 5495. https://doi.org/10.3390/ijms23105495




