Physiological Role of ATPase for GABAA Receptor Resensitization
Abstract
1. Introduction
2. Results
2.1. Reapplication of GABA Causes Desensitization of GABAAR-Mediated Cl− Influx
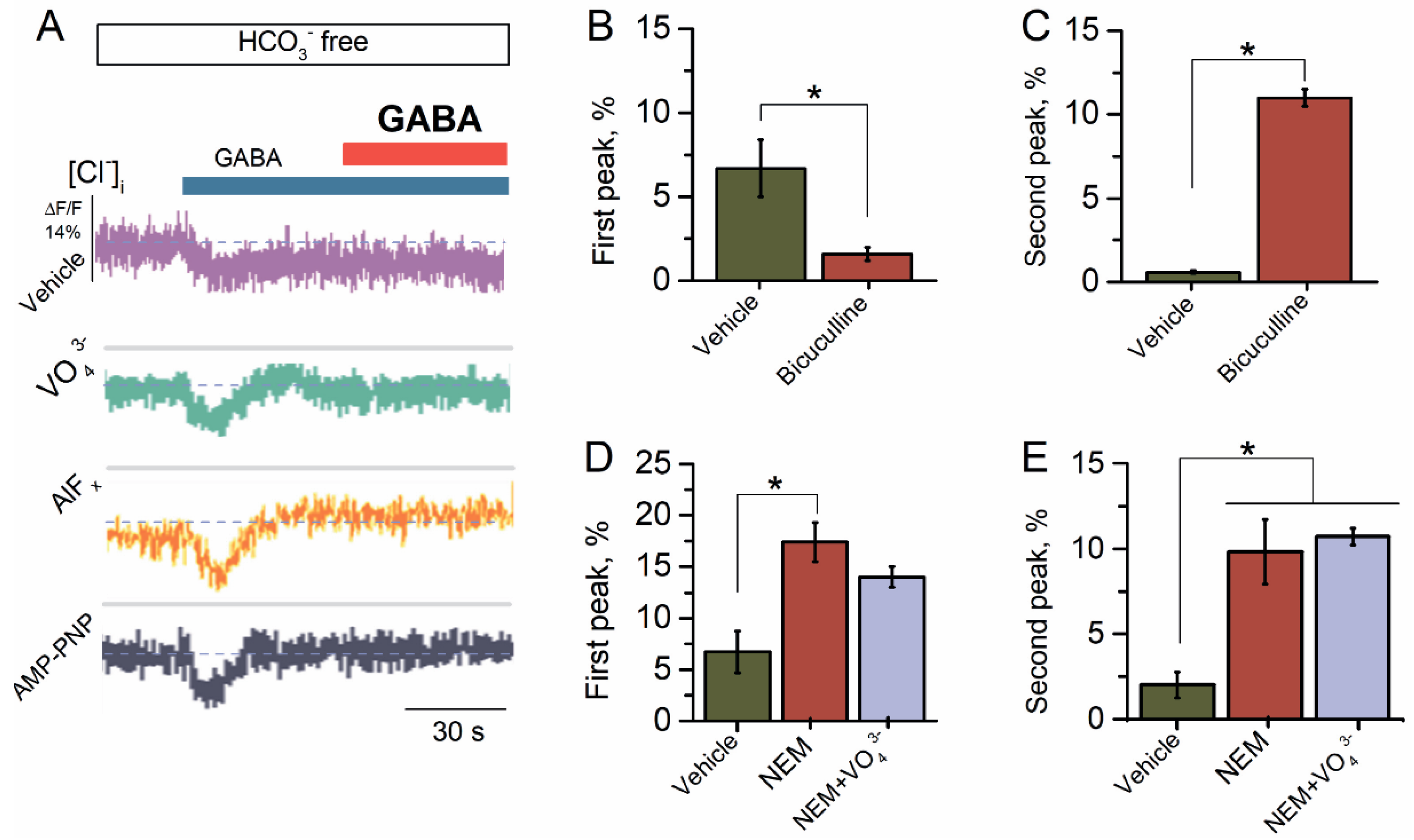
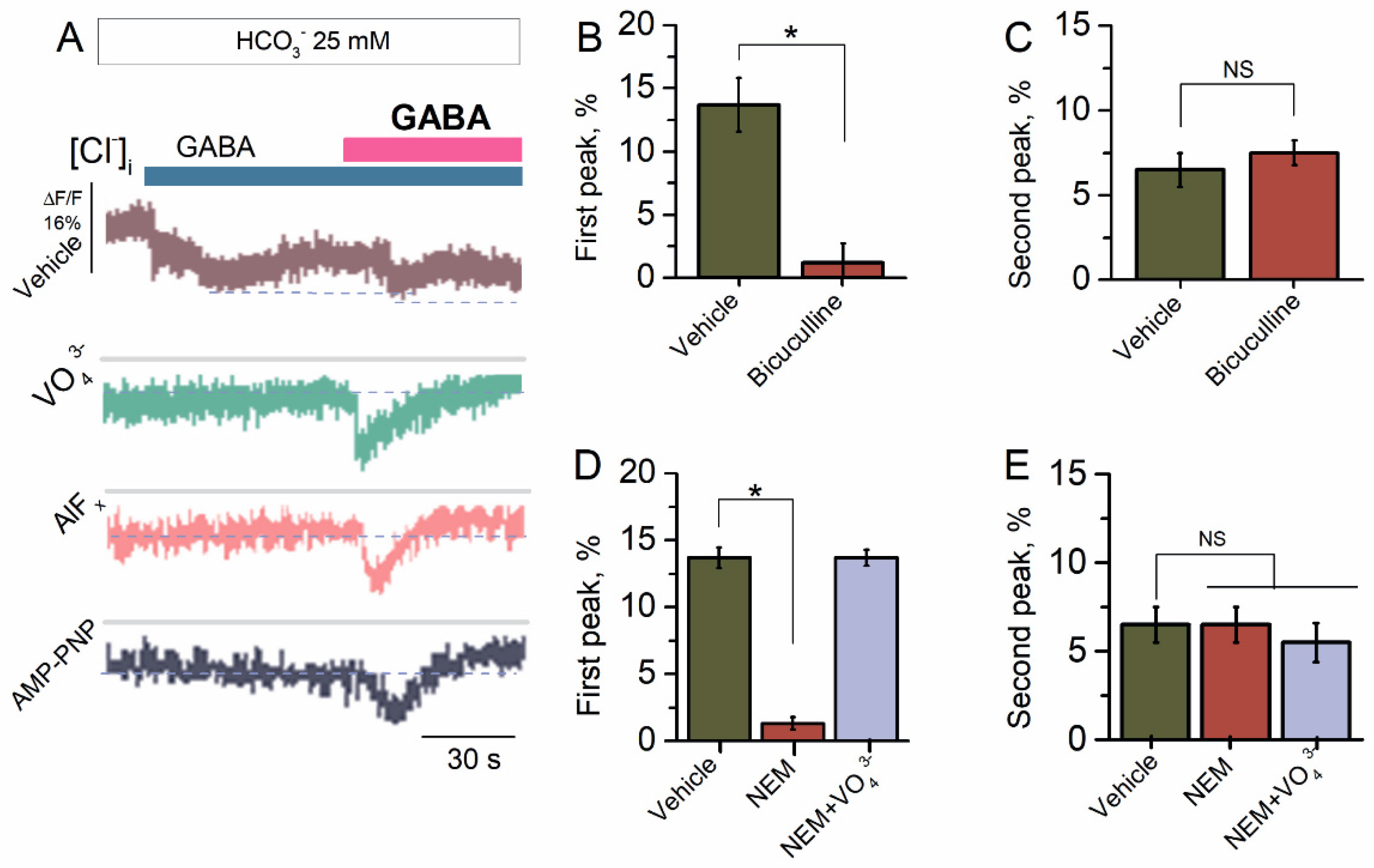
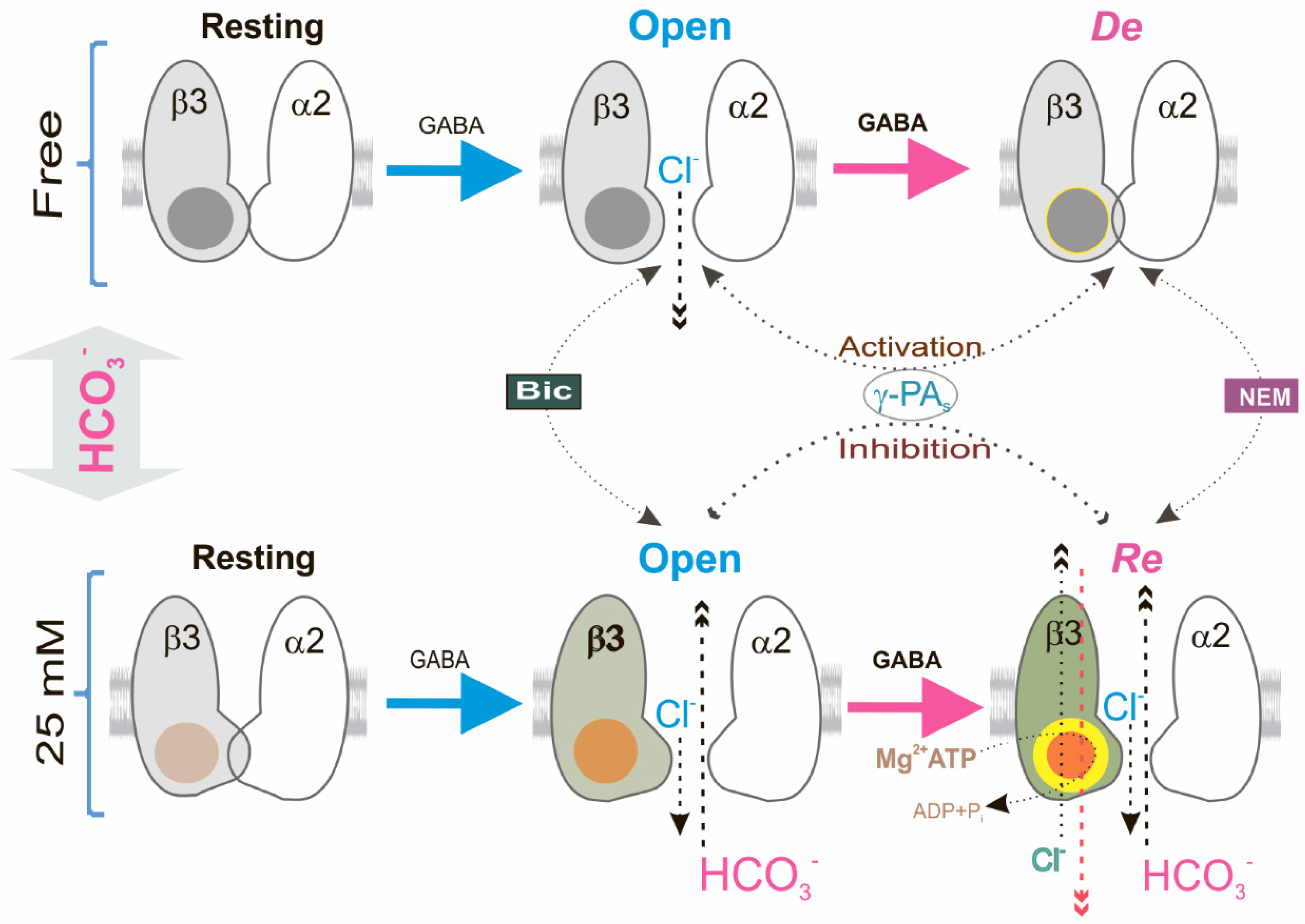
2.2. Bicarbonate Determines a Transition from Desensitization to Resensitization State of GABAAR-Mediated Cl− Influx
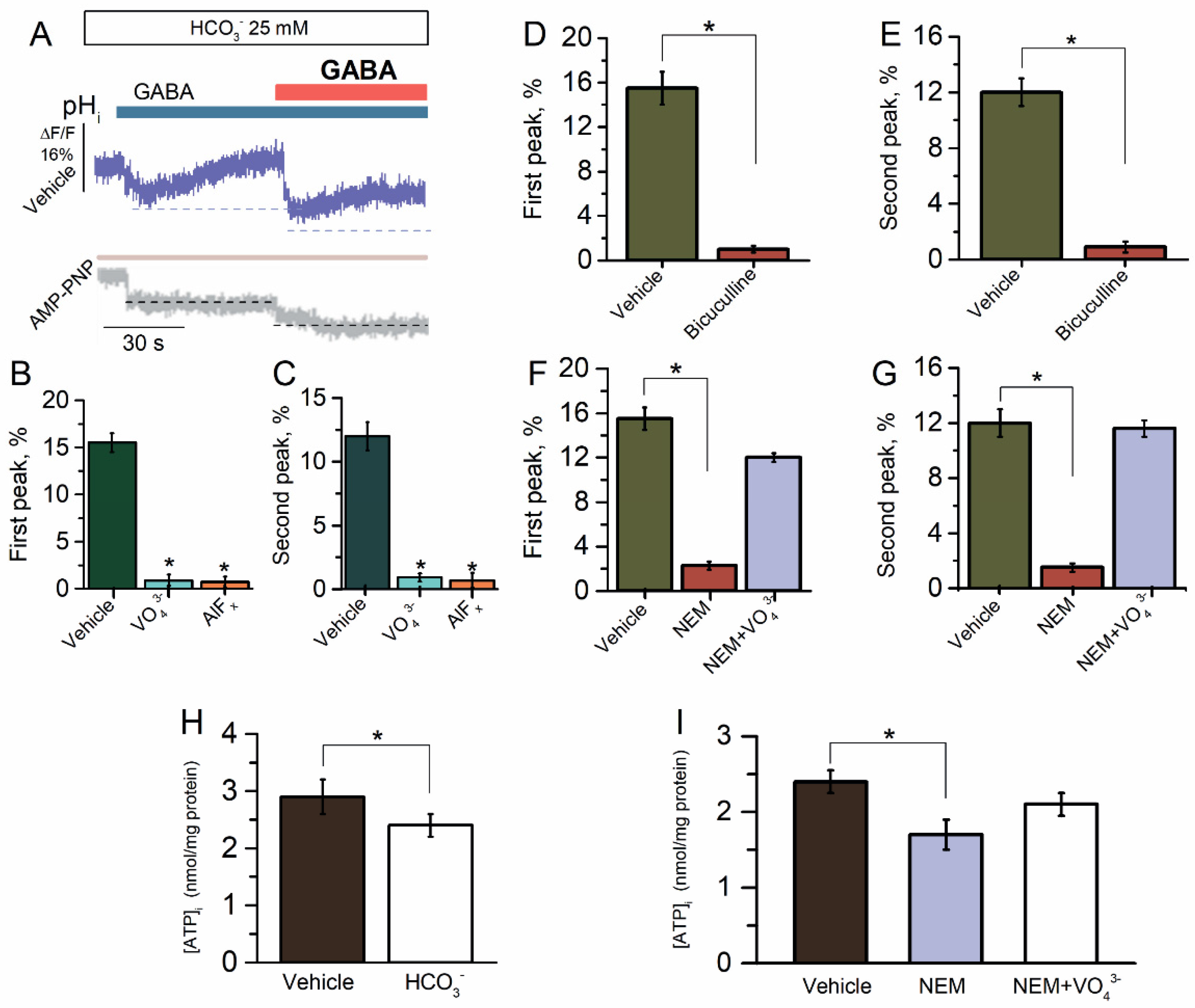
2.3. ATP-Dependent Recovery of GABAAR-Mediated pHi Changes
2.4. β3 Subunit Is Responsible for GABAAR Resensitization in HEK 293FT Cells
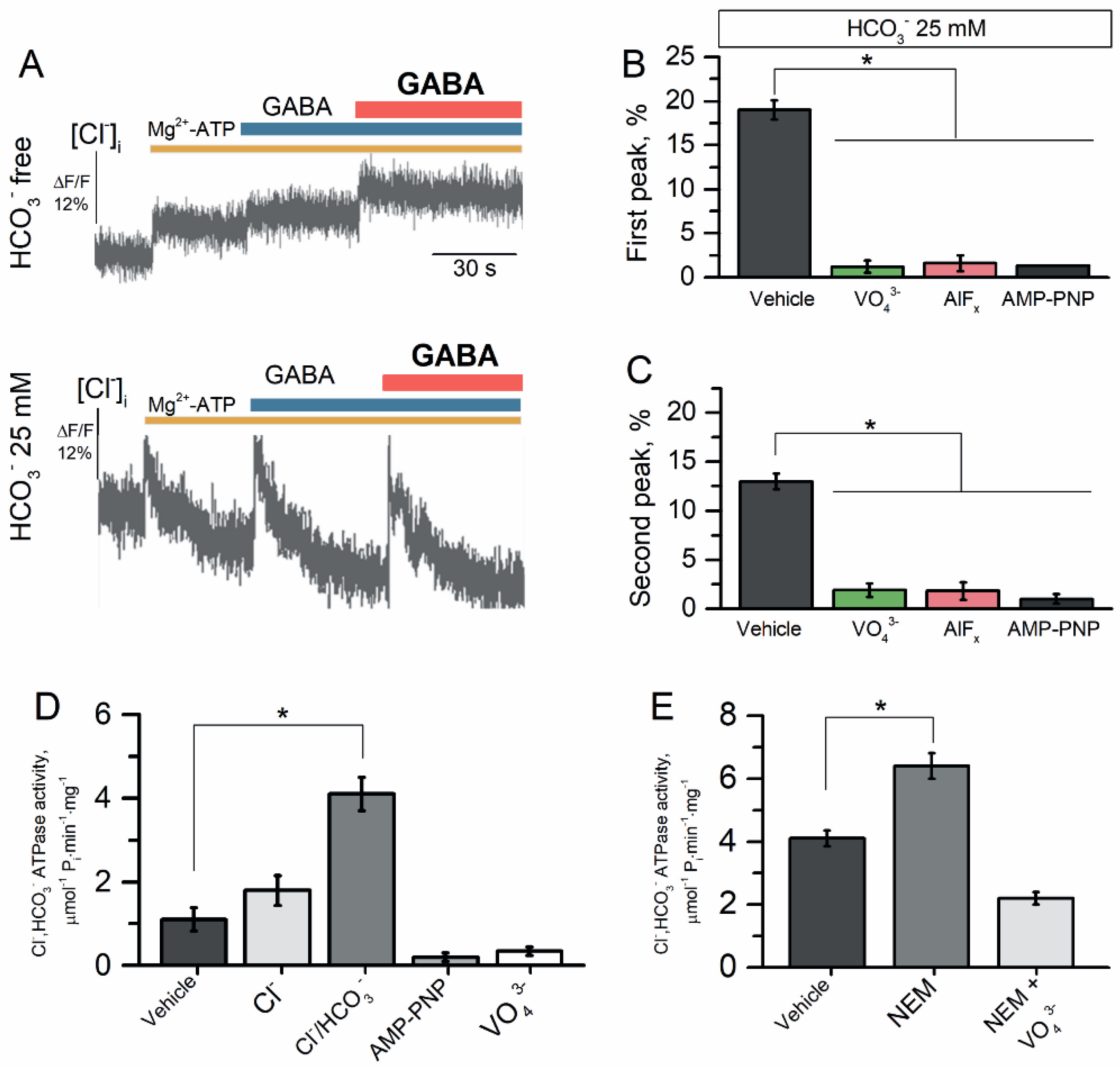
2.5. The Reconstituted Cl−, HCO3− ATPase Responsible for GABAAR Resensitization
3. Discussion
4. Materials and Methods
4.1. Animals and Housing
4.2. Synaptoneurosomes (SNs) Preparation
4.3. Plasma Membrane (PM) Preparation
4.4. Cell Cultures and Transfection
4.5. Molecular Biology
4.6. Transfection
4.7. VLP Production
4.8. Cl− and HCO3−-Transport Assays
4.9. [ATP]i Measurement
4.10. ATPase Activation Assay
4.11. Western Blot Analysis
4.12. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Farrant, M.; Kaila, K. The cellular, molecular and ionic basis of GABAA receptor signalling. Prog. Brain Res. 2007, 160, 59–87. [Google Scholar] [PubMed]
- Sallard, E.; Letourneur, D.; Legendre, P. Electrophysiology of ionotropic GABA receptors. Cell Mol. Life Sci. 2021, 78, 5341–5370. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, M.T.; Macdonald, R.L. Slow phases of GABAA receptor desensitization: Structural determinants and possible relevance for synaptic function. J. Physiol. 2002, 544, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Ghansah, E.; Chen, Y.; Ye, J.; Weiss, D.S. Desensitization mechanism of GABA receptors revealed by single oocyte binding and receptor function. J. Neurosci. 2002, 22, 7982–7990. [Google Scholar] [CrossRef]
- Field, M.; Dorovykh, V.; Thomas, P.; Smart, T.G. Physiological role for GABAA receptor desensitization in the induction of long-term potentiation at inhibitory synapses. Nat. Commun. 2021, 12, 2112. [Google Scholar] [CrossRef]
- Gielen, M.; Barilone, N.; Corringer, P.J. The desensitization pathway of GABAA receptors, one subunit at a time. Nat. Commun. 2020, 11, 5369. [Google Scholar] [CrossRef]
- Jones, M.V.; Westbrook, G.L. Desensitized states prolong GABAA channel responses to brief agonist pulses. Neuron 1995, 15, 181–191. [Google Scholar] [CrossRef]
- Tia, S.; Wang, J.F.; Kotchabhakdi, N.; Vicini, S. Distinct deactivation and desensitization kinetics of recombinant GABAA receptors. Neuropharmacology 1996, 35, 1375–1382. [Google Scholar] [CrossRef]
- Kang, Y.; Saito, M.; Toyoda, H. Molecular and Regulatory Mechanisms of Desensitization and Resensitization of GABAA Receptors with a Special Reference to Propofol/Barbiturate. Int. J. Mol. Sci. 2020, 21, 563. [Google Scholar] [CrossRef]
- Hinkle, D.J.; Macdonald, R.L. β subunit phosphorylation selectively increases fast desensitization and prolongs deactivation of α1β1γ2L and α1β3γ2L GABAA receptor currents. J. Neurosci. 2003, 23, 11698–11710. [Google Scholar] [CrossRef]
- Karlsson, U.; Druzin, M.; Johansson, S. Cl− concentration changes and desensitization of GABAA and glycine receptors. J. Gen. Physiol. 2011, 138, 609–626. [Google Scholar] [CrossRef]
- Florence, L.; Lassi, D.L.S.; Kortas, G.T.; Lima, D.R.; de Azevedo-Marques Périco, C.; Andrade, A.G.; Torales, J.; Ventriglio, A.; De Berardis, D.; De Aquino, J.P.; et al. Brain Correlates of the Alcohol Use Disorder Pharmacotherapy Response: A Systematic Review of Neuroimaging Studies. Brain Sci. 2022, 12, 386. [Google Scholar] [CrossRef]
- Martinotti, G.; Lupi, M.; Sarchione, F.; Santacroce, R.; Salone, A.; De Berardis, D.; Serroni, N.; Cavuto, M.; Signorelli, M.; Aguglia, E.; et al. The potential of pregabalin in neurology, psychiatry and addiction: A qualitative overview. Curr. Pharm. Des. 2013, 19, 6367–6374. [Google Scholar] [CrossRef]
- Gravielle, M.C.C. Regulation of GABAA receptors by prolonged exposure to endogenous and exogenous ligands. Neurochem. Int. 2018, 118, 96–104. [Google Scholar] [CrossRef]
- Chen, L.; Wang, H.; Vicini, S.; Olsen, R.W. The γ-aminobutyric acid type A (GABAA) receptor-associated protein (GABARAP) promotes GABAA receptor clustering and modulates the channel kinetics. Proc. Natl. Acad. Sci. USA 2000, 97, 11557–11562. [Google Scholar] [CrossRef]
- Sugasawa, Y.; Cheng, W.W.; Bracamontes, J.R.; Chen, Z.W.; Wang, L.; Germann, A.L.; Pierce, S.R.; Senneff, T.C.; Krishnan, K.; Reichert, D.E.; et al. Site-specific effects of neurosteroids on GABAA receptor activation and desensitization. elife 2020, 9, e55331. [Google Scholar] [CrossRef]
- Gielen, M.; Corringer, P.-J. The dual-gate model for pentameric ligand-gated ion channels activation and desensitization. J. Physiol. 2018, 596, 1873–1902. [Google Scholar] [CrossRef]
- Kaczor, P.T.; Wolska, A.D.; Mozrzymas, J.W. α1 Subunit Histidine 55 at the Interface between Extracellular and Transmembrane Domains Affects Preactivation and Desensitization of the GABAA Receptor. ACS Chem. Neurosci. 2021, 12, 562–572. [Google Scholar] [CrossRef]
- Amico, C.; Cupello, A.; Fossati, C.; Robello, M. Involvement of phosphatase activities in the run-down of GABAA receptor function in rat cerebellar granule cells in culture. Neuroscience 1998, 84, 529–535. [Google Scholar] [CrossRef]
- Moss, S.J.; Gorrie, G.H.; Amato, A.; Smart, T.G. Modulation of GABAA receptors by tyrosine phosphorylation. Nature 1995, 377, 344–348. [Google Scholar] [CrossRef]
- Liu, J.; Wang, Y.T. Allosteric modulation of GABAA receptors by extracellular ATP. Mol. Brain 2014, 24, 6. [Google Scholar] [CrossRef]
- Stelzer, A.; Kay, A.R.; Wong, R.K. GABAA-receptor function in hippocampal cells is maintained by phosphorylation factors. Science 1988, 241, 339–341. [Google Scholar] [CrossRef]
- Harata, N.; Wu, J.; Ishibashi, H.; Ono, K.; Akaike, N. Run-down of the GABAA response under experimental ischaemia in acutely dissociated CA1 pyramidal neurones of the rat. J. Physiol. 1997, 500, 673–688. [Google Scholar] [CrossRef]
- Shirasaki, T.; Aibara, K.; Akaike, N. Direct modulation of GABAA receptor by intracellular ATP in dissociated nucleus tractus solitarii neurones of rat. J. Physiol. 1992, 449, 551–572. [Google Scholar] [CrossRef]
- Li, G.; Yang, K.; Zheng, C.; Liu, Q.; Chang, Y.; Kerrigan, J.F.; Wu, J. Functional rundown of gamma-aminobutyric acidA receptors in human hypothalamic hamartomas. Ann. Neurol. 2011, 69, 664–672. [Google Scholar] [CrossRef]
- Cifelli, P.; Di Angelantonio, S.; Alfano, V.; Morano, A.; De Felice, E.; Aronica, E.; Ruffolo, G.; Palma, E. Dissecting the Molecular Determinants of GABAA Receptors Current Rundown, a Hallmark of Refractory Human Epilepsy. Brain Sci. 2021, 11, 441. [Google Scholar] [CrossRef]
- Menzikov, S.A.; Zaichenko, D.M.; Moskovtsev, A.A.; Morozov, S.G.; Kubatiev, A.A. Ectopic GABAA receptor β3 subunit determines Cl−/HCO3−-ATPase and chloride transport in HEK 293FT cells. FEBS J. 2020, 288, 699–712. [Google Scholar] [CrossRef]
- Menzikov, S.A.; Morozov, S.G.; Kubatiev, A.A. Intricacies of GABAA Receptor Function: The Critical Role of the β3 Subunit in Norm and Pathology. Int. J. Mol. Sci. 2021, 22, 1457. [Google Scholar] [CrossRef]
- Menzikov, S.A.; Ruzhinskaia, N.N.; Menzikova, O.V. Mg2+-ATPase in the fish brain and its ultrastructural localization. J. Evol. Biochem. Physiol. 2000, 36, 346–352. [Google Scholar] [CrossRef]
- Lacabanne, D.; Wiegand, T.; Wili, N.; Kozlova, M.I.; Cadalbert, R.; Klose, D.; Mulkidjanian, A.Y.; Meier, B.H.; Böckmann, A. ATP Analogues for Structural Investigations: Case Studies of a DnaB Helicase and an ABC Transporter. Molecules 2020, 25, 5268. [Google Scholar] [CrossRef]
- Pinto, J.G.; Hornby, K.R.; Jones, D.G.; Murphy, K.M. Developmental changes in GABAergic mechanisms in human visual cortex across the lifespan. Front. Cell Neurosci. 2010, 10, 16. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Segawa, K.; Fukuda, H. Functional diversity of GABAA receptor ligand-gated chloride channels in rat synaptoneurosomes. Synapse 1995, 19, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Francis, J.; Mihic, S.J.; Sneddon, W.B.; Burnham, W.M. The effect of anticonvulsant drugs on GABA-stimulated chloride uptake. Can. J. Neurol. Sci. 1994, 21, 3–8. [Google Scholar] [CrossRef]
- LoPachin, R.M.; Barber, D.S. Synaptic cysteine sulfhydryl groups as targets of electrophilic neurotoxicants. Toxicol. Sci. 2006, 94, 240–255. [Google Scholar] [CrossRef]
- Conway, L.C.; Cardarelli, R.A.; Moore, Y.E.; Jones, K.; McWilliams, L.J.; Baker, D.J.; Burnham, M.; Bürli, R.W.; Wang, Q.; Brandon, N.; et al. N-Ethylmaleimide increases KCC2 cotransporter activity by modulating transporter phosphorylation. J. Biol. Chem. 2017, 292, 21253–21263. [Google Scholar] [CrossRef]
- Morishita, W.; Alger, B.E. Evidence for endogenous excitatory amino acids as mediators in DSI of GABAAergic transmission in hippocampal CA1. J. Neurophysiol. 1999, 82, 2556–2564. [Google Scholar] [CrossRef]
- Kirmse, K.; Kirischuk, S. N-ethylmaleimide increases release probability at GABAergic synapses in layer I of the mouse visual cortex. Eur. J. Neurosci. 2006, 24, 2741–2748. [Google Scholar] [CrossRef]
- Morishita, W.; Kirov, S.A.; Pitler, T.A.; Martin, L.A.; Lenz, R.A.; Alger, B.E. N-ethylmaleimide blocks depolarization-induced suppression of inhibition and enhances GABA release in the rat hippocampal slice in vitro. J. Neurosci. 1997, 17, 941–950. [Google Scholar] [CrossRef]
- Beau, F.E.; Alger, B.E. Transient suppression of GABAA-receptor-mediated IPSPs after epileptiform burst discharges in CA1 pyramidal cells. J. Neurophysiol. 1998, 79, 659–669. [Google Scholar] [CrossRef][Green Version]
- Perkins, K.L.; Wong, R.K. Ionic basis of the postsynaptic depolarizing GABA response in hippocampal pyramidal cells. J. Neurophysiol. 1996, 76, 3886–3894. [Google Scholar] [CrossRef]
- Kaila, K. Ionic basis of GABAA receptor channel function in the nervous system. Prog. Neurobiol. 1994, 42, 489–537. [Google Scholar] [CrossRef]
- Do-Young, K.; Fenoglio, K.A.; Kerrigan, J.F.; Rho, J.M. Bicarbonate contributes to GABAA receptor-mediated neuronal excitation in surgically resected human hypothalamic hamartomas. Epilepsy Res. 2009, 83, 89–93. [Google Scholar]
- Staley, K.J.; Proctor, W.R. Modulation of mammalian dendritic GABAA receptor function by the kinetics of Cl− and HCO3− transport. J. Physiol. Lond. 1999, 519, 693–712. [Google Scholar] [CrossRef]
- Lückermann, M.; Trapp, S.; Ballanyi, K. GABA- and glycine-mediated fall of intracellular pH in rat medullary neurons in situ. J. Neurophysiol. 1997, 77, 1844–1852. [Google Scholar] [CrossRef]
- Pan, Z.H.; Zhang, X.; Lipton, S.A. Redox modulation of recombinant human GABA(A) receptors. Neuroscience 2000, 98, 333–338. [Google Scholar] [CrossRef]
- Nakamura, Y.; Darnieder, L.M.; Deeb, T.Z.; Moss, S.J. Regulation of GABAARs by phosphorylation. Adv. Pharmacol. 2015, 72, 97–146. [Google Scholar]
- Hübner, C.A.; Holthoff, K. Anion transport and GABA signaling. Front. Cell Neurosci. 2013, 7, 177. [Google Scholar] [CrossRef]
- Raimondo, J.V.; Richards, B.A.; Woodin, M.A. Neuronal chloride and excitability-the big impact of small changes. Curr. Opin. Neurobiol. 2017, 43, 35–42. [Google Scholar] [CrossRef]
- Rivera, C.; Voipio, J.; Kaila, K. Two developmental switches in GABAergic signalling: The K+–Cl− cotransporter KCC2 and carbonic anhydrase CAVII. J. Physiol. 2005, 562, 27–36. [Google Scholar] [CrossRef]
- Phillips, I.; Martin, K.F.; Thompson, K.S.; Heal, D.J. GABA-evoked depolarisations in the rat cortical wedge: Involvement of GABAA receptors and HCO3− ions. Brain Res. 1998, 798, 330–332. [Google Scholar] [CrossRef]
- Sato, S.S.; Artoni, P.; Landi, S.; Cozzolino, O.; Parra, R.; Pracucci, E.; Trovato, F.; Szczurkowska, J.; Luin, S.; Arosio, D.; et al. Simultaneous two-photon imaging of intracellular chloride concentration and pH in mouse pyramidal neurons in vivo. Proc. Natl. Acad. Sci. USA 2017, 114, 8770–8779. [Google Scholar]
- Lombardi, A.; Jedlicka, P.; Luhmann, H.J.; Kilb, W. Interactions between Membrane Resistance, GABA-A Receptor Properties, Bicarbonate Dynamics and Cl−-Transport Shape Activity-Dependent Changes of Intracellular Cl− Concentration. Int. J. Mol. Sci. 2019, 20, 1416. [Google Scholar] [CrossRef]
- Kühlbrandt, W. Biology, structure and mechanism of P-type ATPases. Nat. Rev. Mol. Cell Biol. 2004, 5, 282–295. [Google Scholar] [CrossRef]
- Mächtel, R.; Narducci, A.; Griffith, D.A.; Cordes, T.; Orelle, C. An integrated transport mechanism of the maltose ABC importer. Res. Microbiol. 2019, 170, 321–337. [Google Scholar] [CrossRef]
- Prieß, M.; Göddeke, H.; Groenhof, G.; Schäfer, L.V. Molecular Mechanism of ATP Hydrolysis in an ABC Transporter. ACS Cent. Sci. 2018, 4, 1334–1343. [Google Scholar] [CrossRef]
- Clausen, J.D.; Bublitz, M.; Arnou, B.; Olesen, C.; Andersen, J.P.; Mølle, J.V.; Nissen, P. Crystal Structure of the Vanadate-Inhibited Ca2+-ATPase. Structure 2016, 24, 617–623. [Google Scholar] [CrossRef]
- Cornelius, F.; Mahmmoud, Y.A.; Toyoshima, C. Metal fluoride complexes of Na+, K+-ATPase: Characterization of fluoride-stabilized phosphoenzyme analogues and their interaction with cardiotonic steroids. J. Biol. Chem. 2011, 286, 29882–29892. [Google Scholar] [CrossRef]
- Locher, K.P. Mechanistic diversity in ATP-binding cassette (ABC) transporters. Nat. Struct. Mol. Biol. 2016, 23, 487–493. [Google Scholar] [CrossRef]
- Sieghart, W.; Sperk, G. Subunit composition, distribution and function of GABA-A receptor subtypes. Curr. Top. Med. Chem. 2002, 2, 795–816. [Google Scholar] [CrossRef]
- Connolly, C.N.; Wooltorton, J.R.; Smart, T.G.; Moss, S.J. Subcellular localization of gamma-aminobutyric acid type A receptors is determined by receptor beta subunits. Proc. Natl. Acad. Sci. USA 1996, 93, 9899–9904. [Google Scholar] [CrossRef]
- Nguyen, Q.A.; Nicoll, R.A. The GABAA Receptor β Subunit Is Required for Inhibitory Transmission. Neuron 2018, 98, 718–725. [Google Scholar] [CrossRef]
- Le Foll, F.; Soriani, O.; Vaudry, H.; Cazin, L. Contribution of changes in the chloride driving force to the fading of IGABA in frog melanotrophs. Am. J. Physiol. Endocrinol. Metab. 2000, 278, E430–E443. [Google Scholar] [CrossRef]
- Heynen, A.J.; Quinlan, E.M.; Bae, D.C.; Bear, M.F. Bidirectional, activity-dependent regulation of glutamate receptors in the adult hippocampus in vivo. Neuron 2000, 28, 527–536. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Menzikov, S.A.; Zaichenko, D.M.; Moskovtsev, A.A.; Morozov, S.G.; Kubatiev, A.A. Physiological Role of ATPase for GABAA Receptor Resensitization. Int. J. Mol. Sci. 2022, 23, 5320. https://doi.org/10.3390/ijms23105320
Menzikov SA, Zaichenko DM, Moskovtsev AA, Morozov SG, Kubatiev AA. Physiological Role of ATPase for GABAA Receptor Resensitization. International Journal of Molecular Sciences. 2022; 23(10):5320. https://doi.org/10.3390/ijms23105320
Chicago/Turabian StyleMenzikov, Sergey A., Danila M. Zaichenko, Aleksey A. Moskovtsev, Sergey G. Morozov, and Aslan A. Kubatiev. 2022. "Physiological Role of ATPase for GABAA Receptor Resensitization" International Journal of Molecular Sciences 23, no. 10: 5320. https://doi.org/10.3390/ijms23105320
APA StyleMenzikov, S. A., Zaichenko, D. M., Moskovtsev, A. A., Morozov, S. G., & Kubatiev, A. A. (2022). Physiological Role of ATPase for GABAA Receptor Resensitization. International Journal of Molecular Sciences, 23(10), 5320. https://doi.org/10.3390/ijms23105320




