Nitric Oxide as a Target for Phytochemicals in Anti-Neuroinflammatory Prevention Therapy
Abstract
1. Introduction
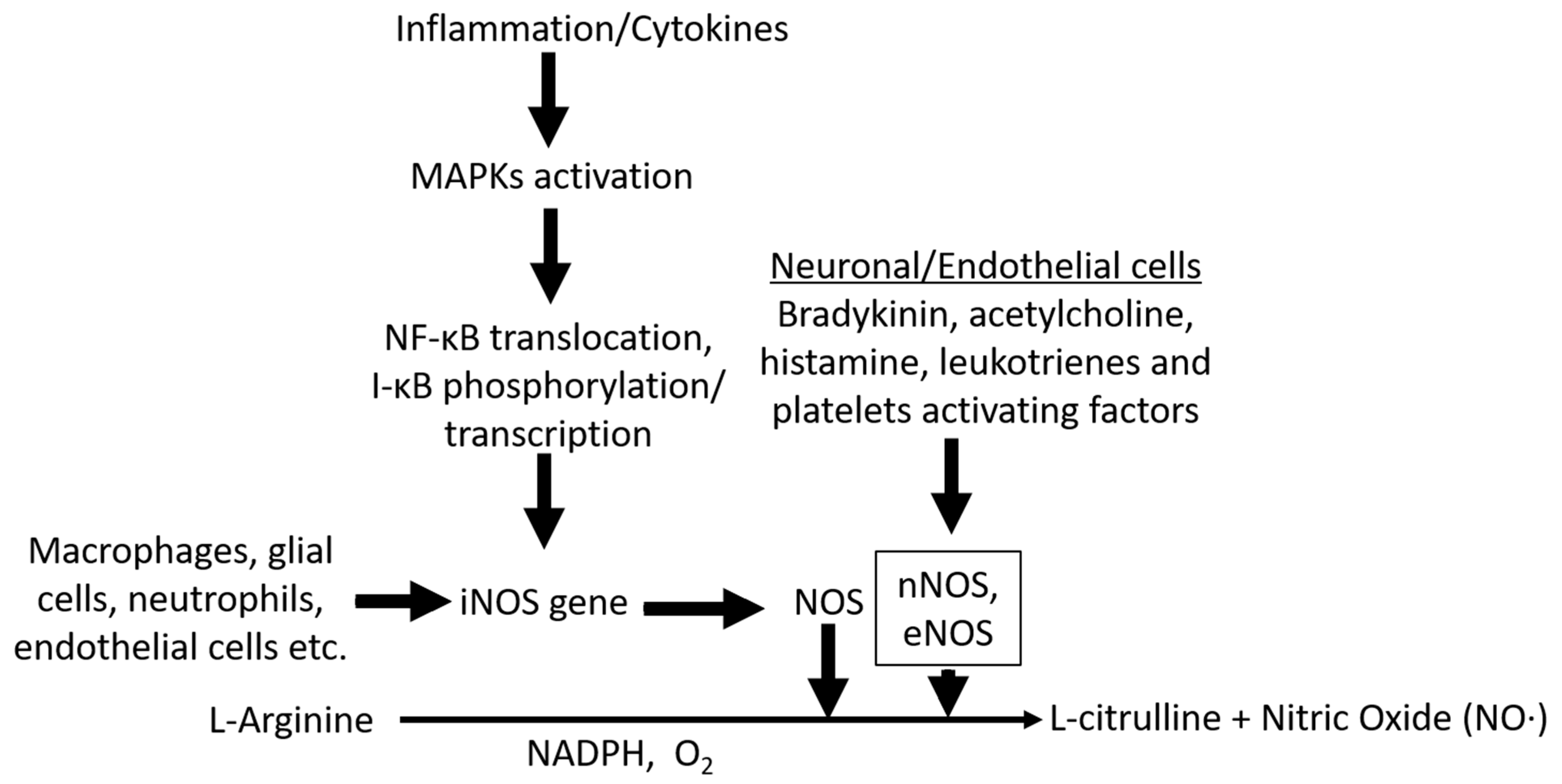
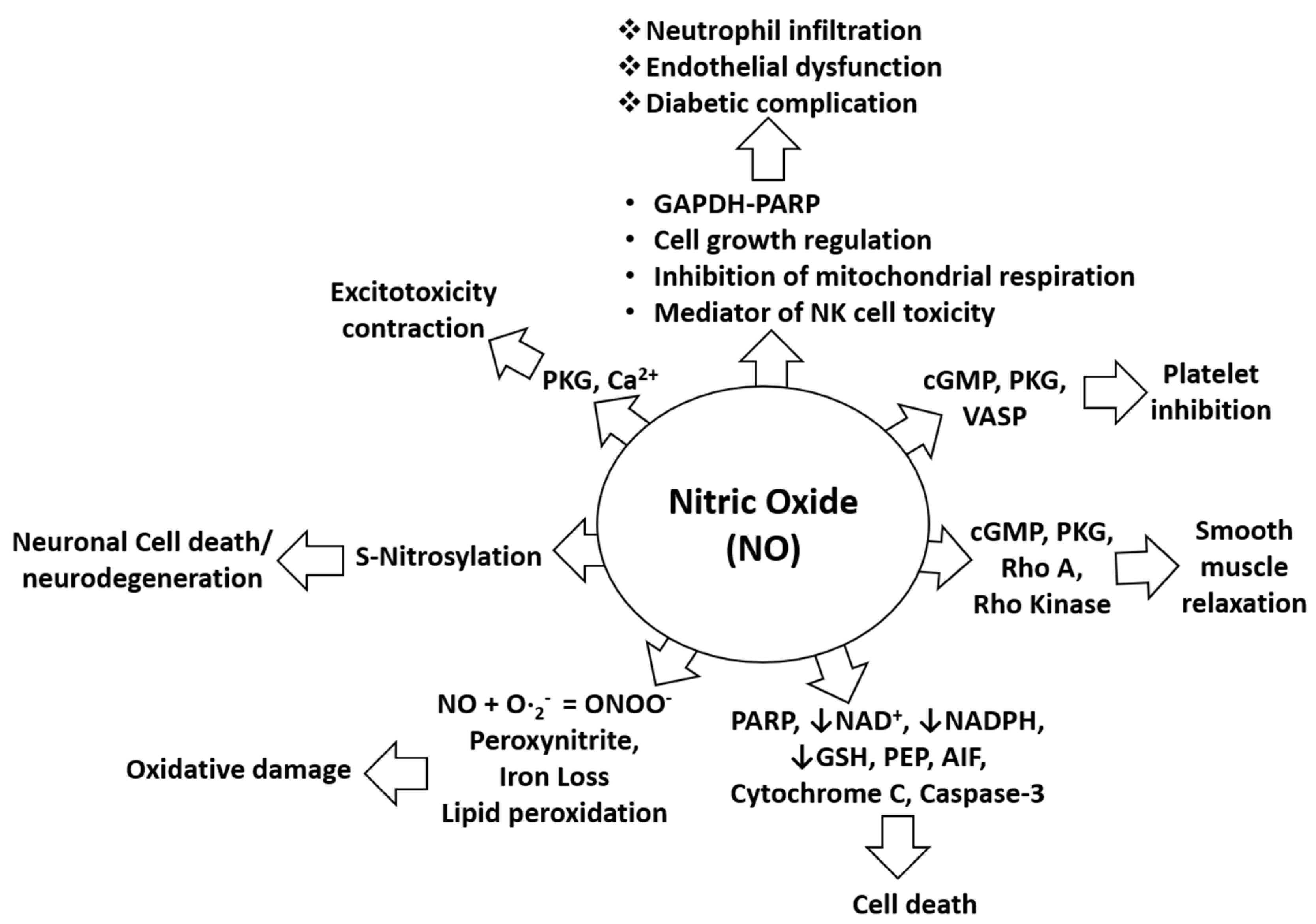
1.1. The role of NO in Various Neurological Disorders
| Stimulator | Cell Type | References |
|---|---|---|
| LPS | Murine microglia (BV2), primary microglia, and RAW cells | [49,50] |
| TNF-α | Macrophages | [51] |
| IL-1β | Murine N9 microglia | [52] |
| Estrogen | Human umbilical vein cells (HUVEC) and human neuroblastoma cells (SK-N-SH) | [53] |
| IFN-γ | Macrophages | [51] |
| Hypoxia | Rat insulinoma cell line (INS-1), rat islet cells | [54] |
| Ethanol | Hypothalamic neuronal cell cultures | [55] |
| Advanced glycation end products | BV2 cells, endothelial cells | [56] |
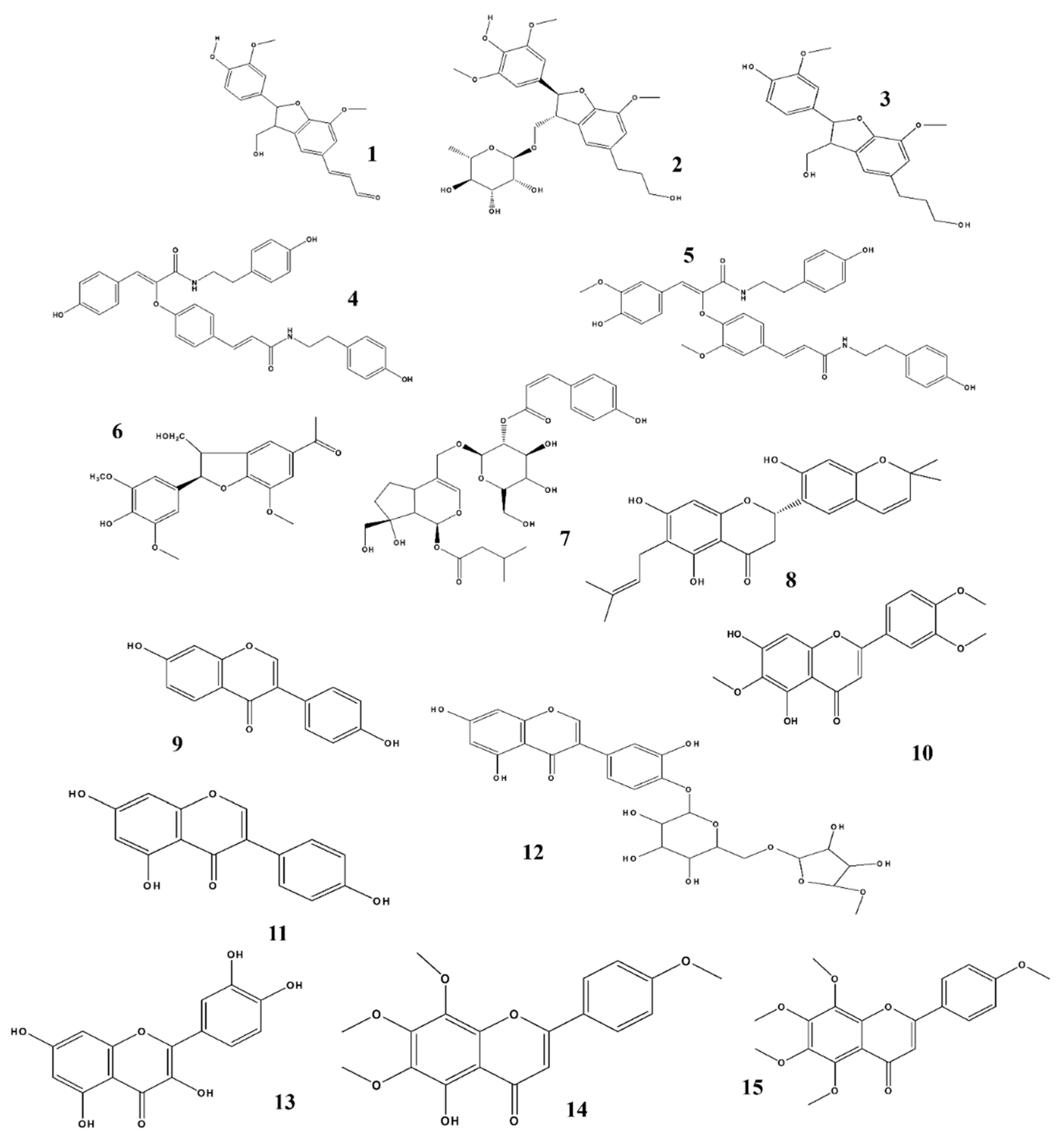
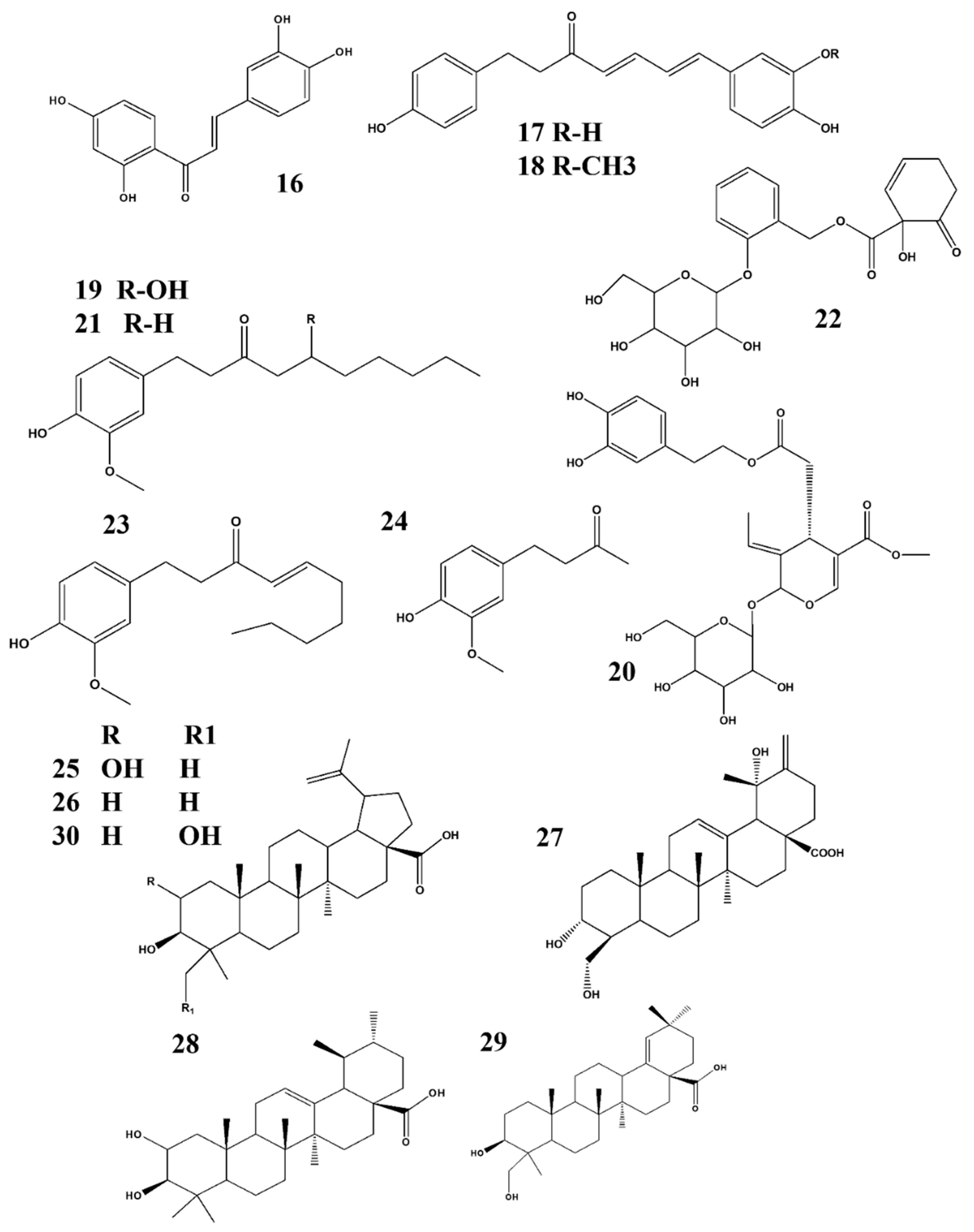
1.2. Anti-Neuroinflammatory and NO-Inhibiting Activity of Lignans and Neolignans
1.3. Anti-Neuroinflammatory and NO-Inhibiting Effects of Flavonoids
1.4. Anti-Neuroinflammatory and NO-Inhibiting Activity of Polyphenols
1.5. Anti-Neuroinflammatory and NO-Inhibiting Activity of Triterpenoids
1.6. Anti-Neuroinflammatory and NO-Inhibiting Activity of Phytoestrogens
1.7. Anti-Neuroinflammatory and NO-Inhibiting Activity of Coumarins
1.8. Anti-Inflammatory and NO-Inhibiting Activity of other Compounds
| Scientific Name | Compound | IC50 | Class | Pharmacological Target | Pharmacological Effects | References |
|---|---|---|---|---|---|---|
| Abies holophylla | Holophyllane A | 12.75 | Triterpenoid | NO | Anti-inflammatory Anticancer | [89] |
| Holophyllane B | 18.96 | |||||
| Artemisia argyi | Sesquiterpene dimer | NA | Terpenoid | NO, PGE2, TNF-α, IL-1β, GM-CSF, MIP-1α, p38 MAPKs., JNK, JAK2/STAT3 | Anti-inflammation Anticancer Antioxidant Antidiabetic | [93] |
| Artemisia asiatica | Eupatilin | NA | Flavone | NF-κB | Anti-inflammatory Neuroprotective | [68] |
| Betula schmidtii | 2α-O-benzoyl-3β,19α-dihydroxy-urs-12-en-28-oic acid | 4.92 | Triterpenoids | NO | Anti-inflammatory | [86,102] |
| 2α-O-benzoyl-19α-hydroxy-3-oxo-urs-12-en-28-oic acid | 9.68 | |||||
| Corosolic acid 11-Oxo-erythrodiol | 12.93 16.58 | NO | ||||
| Maslinic acid | 4.46 | TNF-α, NF-κB, a COX-2, NO | ||||
| Morolic acid 3-O-caffeate | 8.62 | |||||
| Ambradiolic acid | ||||||
| Isotachioside 4-allyl-2-hydrophenyl 1-O-β- | 23.9 | Phenolic derivatives | ||||
| D-apiosyl-(1 → 6)- β -D-glucopyranoside | 25.3 | |||||
| Genistein 5-O-β-D-glucoside | 28.8 | |||||
| Prunetinoside | 34.0 | |||||
| Brassica olifera | Sulforaphane | 5.85 | isothiocyanate | Anti-inflammatory Antioxidant Anti-amnesic Neuroprotective | [56,103,104] | |
| Bupleurum falcatum | Saikosaponins | NA | Terpenoid | NO, TNF-α, IL-6, IL-1β, iNOS, NF-κB |
Immune modulator Antibacterial Anti-inflammatory Antihepatoma | [92] |
| Capsella Bursa-pastoris | Sesquilignan glycoside | 75.13 | Phenolic glycosides | Anti-inflammatory | [105] | |
| 7S, 8R, 8′R-(−)-lariciresinol-4,4′-bis-O-glucopyranoside | 48.80 | |||||
| Lariciresinol4′-O-β-d-glucoside | 30.70 | |||||
| (+)-Pinoresinol-β-d-glucoside | 17.80 | |||||
| Salidroside | 31.14 | |||||
| 3-(4-β-D-glucopyranosyloxy-3,5-dimethoxy)-phenyl-2E-propanol | 62.21 | |||||
| β-Hydroxy-propiovanillone 3-O-β-d-glucopyranoside | 27.91 | |||||
| Coniferin | 49.21 | |||||
| Chaenomeles sinensis | 3β-O-cis-feruloyl-2α,19α- dihydroxyurs-12-en-28-oic acid | 37.1 | Triterpenoids | NO | Anti-inflammatory | [106] |
| Maslinic acid | 17.8 | |||||
| 2α,3α-Dihydroxyolean-12-en-28-oic acid | 21.6 | |||||
| 2α-Hydroxyursolic acid | 47.1 | |||||
| Betulinic acid | 4.5 | NF-κB, NO, prostaglandin E2, TNF-α, IL-1β, IL-6, COX-2 | ||||
| Alphitolic acid | 14.5 | |||||
| 3-O-cis-caffeoylbetulinic | 13.4 | |||||
| Ilekudinol C | 25.5 | |||||
| Chaenomin | 48.37 | |||||
| Berbekorin A | 49.29 | |||||
| Aucuparin | 50.15 | NO | ||||
| 2′-Hydroxyaucuparin | 38.06 | |||||
| 2′-Methoxyaucuparin | 28.09 | |||||
| 2′,4′-Dimethoxyaucuparin | 39.64 | |||||
| ε-Cotonefuran | 17.78 | NO | ||||
| Chaenomeles speciosa | Chaenomin B | 283.33 | Biphenyl derivatives | Anticancer Anti-inflammatory Neuroprotective | [88] | |
| Chaenomin A | 86.02 | |||||
| 2′,4′-Dimethoxyaucuparin | 272.72 | |||||
| Aucuparin | 20.04 | |||||
| 2′-Methoxyaucuparin | 22.02 | |||||
| Maslinic acid | 26.96 | |||||
| Dihydrotomentosolic acid | 19.41 | |||||
| Ilexgenin B, | 12.72 | |||||
| Betulinic acid | 2.38 | NF-κB, NO, prostaglandin E2, TNF-α, IL-1β, IL-6, COX-2 | ||||
| 23-Hydroxybetulinic acid and pycarenic acid | 6.70 | |||||
| Pycarenic acid | 67.50 | |||||
| Citrus aurantium | Tangeretin | NA | Flavone | Anti-neuroinflammatory | [73] | |
| Citrus unshiu | Citrusin XI | 70 | Cyclopeptide | NO, iNOS, NF-κB | Anti-inflammatory Anti-fungal Anti-bacterial Antioxidant | [98] |
| Clematis armandii | (7R, 8S)-dehydrodiconferyl alcohol | 9.3 | Lignan | NO, COX-2, iNOS, NF-κB, AKT, JNK, src, fak | Anti-aging Anti-inflammatory | [59,60] |
| Cudrania tricuspidata Bureau | Cudraflavanone A | 22.2 | Flavonoid | NO, PGE2, iNOS, NF-κB, P38, JNK MAPK | Anti-inflammatory Neuroprotective Antioxidant | [64] |
| Dioscorea nipponica | Tsaokarylone | 13.36 | Phenols | NO | Anti-diabetic Anti-inflammatory | [75] |
| (4E,6E)-1-(3ʹ,4ʹ-dihydroxyphenyl)-7-(4ʹʹ-hydroxyphenyl)-hepta-4, 6-dien-3-one | 14.36 | |||||
| Firmiana simplex | 4-[(1S,2R)-1,3-dihydroxy-2-[4-[(1E)-3-hydroxy-1-propenyl]-2- methoxyphenoxy] propyl]-2-methoxyphenyl β-D-glucopyranoside | 59.83 | Lignan glycosides | NO, PGE2, TNF-α, IL-1β, COX2, MAPKs, ERK, JNK, p38 MAPK | Anti-neuroinflammatory Anti-inflammatory Neuroprotective | [107] |
| Balanophonin | 10.25 | Neolignan derivative | [57] | |||
| Firmianols A | 35.39 | Lignan derivatives | ||||
| Firmianols B | >500 | |||||
| (+)-Piperitol | 32.65 | |||||
| (+)-Pinoresinol | 25.1 | |||||
| (+)-Syringaresinol | 27.53 | |||||
| Buddlenol E | 19.22 | |||||
| (+)-Sesamin | 26.26 | |||||
| (−)-Pinoresinol | 31.1 | |||||
| (+)-7′-Methoxylariciresinol | 32.99 | |||||
| (−)-5-Methoxybalanophonin | 10.0 | |||||
| Buddlenol A Threo-(7R,8R)- | 15.23 | |||||
| Guaiacylglycerol-β-coniferyl aldehyde ether | 1.05 | |||||
| Erythro-(7S,8R)-Guaiacylglycerol-β-coniferyl aldehyde ether | 0.929 | |||||
| Threo-Guaiacylglycerol-8-O-4′-sinapyl alcohol ether | 9.14 | |||||
| Erythro-Syringylglycerol-8-O-4′-coniferyl alcohol ether | 9.14 | |||||
| Threo-Guaiacylglycerol-8-O-4′-coniferyl alcohol ether | 32.56 | |||||
| Threo-Guaiacylglycerol 8′-vanillin ether | 47.59 | |||||
| Fraxinus rhynchophylla | Oleuropein | NO, ROS, ERK/p38/NF-κB | [77] | |||
| Glycine max | Daidzein | 93.15 | Isoflavones | NO, AMP kinase, NF-κB | Anti-inflammatory Neuroprotective | [65] |
| Daidzein | 14.09 | |||||
| Genistein | 137.50 | |||||
| Genistein | 10.63 | |||||
| Equol | 3.45 | |||||
| Impatiens balsamina | Balsamisides A | 33.33 | Biflavonoid glycosides | NO | Anti-neurodegenerative | [71] |
| Balsamisides B | 56.86 | |||||
| Balsamisides C | 39.16 | |||||
| Balsamisides D | 31.02 | |||||
| Kaempferol | 8.86 | |||||
| Kaempferol 3-O-β-D-glucopyranoside, | 23.50 | |||||
| Kaempferol3-O-α-L-rhamnopyranosyl-(1→2)-β-D-glucopyranoside | 31.73 | |||||
| Kaempferol3-O-α-L-rhamnopyranosyl-(1→6)-β-Dglucopyranoside | 44.44 | |||||
| Kaempferol 3-O-β-D-allopyranoside | 80.35 19.11 | |||||
| Quercetin | 19.11 | |||||
| Quercetin3-O-β-D-glucospyranoide, | 55.59 | |||||
| Quercetin3-O-α-L-rhamnopyranosyl-(1→6)-β-D-glucopyranoside | 24.29 | |||||
| Dihydromyricetin | 32.66 | |||||
| Lagerstroemia indica | Pterospermin A | 21.4 | Phenolic derivative | Anti-inflammatory | [108] | |
| Dihydrodehydroconiferyl alcohol | 14.6 | |||||
| Alatusol | 35.4 | |||||
| Ficusol | 36.0 | |||||
| Evofolin-B | 22.0 | |||||
| Marphenol | 44.9 | |||||
| Ligustrum obtusifolium | Obtusifolisides A | 33.85 | Secoiridoid glycosides | LPS-activated microglia neuroinflammation | [109] | |
| Obtusifolisides B | 5.45 | |||||
| Oleoside-11-methylester | 38.67 | |||||
| Oleoside-7,11-dimethylester 10-Hydroxyoleuropein | 38.89 11.17 | |||||
| Oleuropein | 14.62 | |||||
| Ligstroside | 61.25 | |||||
| (2″R)-2″- methoxyoleuropein | 15.45 | |||||
| Neonuezhenide | 14.96 | |||||
| 4,4′-(1E,1′E,3E,3′E)-3,3′-(hydrazine-1,2-diylidene) bis-(prop-1-ene-1-yl-3-ylidene) bis-(2-methoxyphenol) | 12.47 | Azine derivative | [110] | |||
| Lindera glauca | Eudeglaucone | 15.90 | Eudesmane sesquiterpene | Anti-inflammatory | [87,111] | |
| (+)-Faurinone | 3.67 | |||||
| 3α-Hydroxycostic acid | 26.48 | |||||
| Ilicic acid | 14.92 | |||||
| γ-Costic acid | 24.44 | |||||
| Costic acid | 12.13 | |||||
| Lindera neesiana | Koaburaside | NA | Phenolic glycoside | NO, MAPK, COX-2, INOS | Anti-inflammatory, Neuroprotection | [99] |
| Medicago sativa Linn | Coumestrol | NA | Phytoestrogen | NO, 1 (IRF-1), stat1, MCP-1, IL-6 | Antidiabetic Lower liver cholesterol | [95] |
| Phaeanthus vietnamensis | (7S,8R,8′R)-9,9′-epoxy-3,5,3′,5′-tetramethoxylignan-4,4′,7-triol | 65.2 | Alkaloids | Anti-inflammatory | [94] | |
| 8R,8′R-bishydrosyringenin | 25.3 | |||||
| (+)-5,5’-Dimethoxylariciresinol | 73.9 | |||||
| (+)-Pinoresinol | 60 | |||||
| 8α-Hydroxyoplop-11(12)-en-14-one | 46.3 | |||||
| Spathulenol | 15.7 | NO | ||||
| 1αH,5βH-aromandendrane-4β,10α-diol | 29.3 | |||||
| 1αH,5βH-aromandendrane-4α,10α-diol10α-diol | 23.0 | |||||
| 1βH,5βH-aromandendrane-4α,10β-diol | 22.6 | |||||
| 3α,4β-dihydroxybisabola-1,10-diene | 39.9 | |||||
| Nerolidol | 50.8 | |||||
| (1R,2S,4S)-4-acetyl-2-[(E)-(cinnamoyloxy)]-1-methylcyclohexan-1-ol | 45.7 | |||||
| Pinus koraiensis | Koraiensides E | 24.1 | Diterpenoid Glycosides | Anti-inflammatory Neuroprotective | [112] | |
| Raphanus sativus | Sinapoyl desulfoglucoraphenin | 45.36 | 4-Methylthio-butanyl derivatives | NO | Anti-inflammatory Anti-proliferative | [100] |
| Genistein | 137.50 | |||||
| Genistein | 10.63 | |||||
| Equol | 3.45 | |||||
| Salix glandulosa | Saliglandin | 120.18 | Salicin derivatives | NO | Anti-inflammatory | [79] |
| 6′-O-(Z)-p-coumaroylsalicin | 31.55 | |||||
| Salicin | 85.40 | |||||
| 2′-O-acetylsalicin | 123.36 | |||||
| 3′-O-acetylsalicin | 27.27 | |||||
| Fragilin | 206.12 | |||||
| Trumuloidin | 114.30 | |||||
| 2′-O-(E)-p-coumaroylsalicin 2′-O-(Z)-p-coumaroylsalicin | 29.77 25.47 | |||||
| 6′-O-(E)-p-coumaroylsalicin | 38.25 | |||||
| Salicortin | 13.57 | NO | ||||
| 2′-O-acetylsalicortin | 14.61 | |||||
| 3′-O-acetylsalicortin | 18.27 | |||||
| 6′-O-acetylsalicortin | 22.78 | |||||
| Tremulacin | 18.59 | |||||
| Cochinchiside A | 23.40 | |||||
| Sambucus williamsii | Sambucasinol B | 0.9 | New iridoid glycosides Lignan | NO | Anti-inflammatory Neuroprotective | [63] |
| Sambucuside C | 1.3 | |||||
| Sambucuside E | 1.2 | |||||
| Sambucasinol A | 6.82 | |||||
| Lariciresinol | 72.58 | |||||
| (7αH,8′αH)-4,4′,8α,9-tetrahydroxy-3,3′-dimethoxy-7,9′-epoxylignan | >500 | |||||
| Berchemol | 215.41 | |||||
| 7-Hydroxylariciresinol | 128.97 | |||||
| (−)-Medioresinol | 45.59 | |||||
| (−)-Pinoresinol | 34.25 | |||||
| 7R,8S-dihydrodehydrodiconiferyl alcohol | 39.97 | |||||
| Selagginella tamariscina | (2R, 3S) dihydro-2-(3,5-dimethoxy-4-hydroxyphenyl)-7-methoxy-5-acetyl-benzofuran | 32.3 | Lignanoside | NO, iNOS, TNF-α, and NF-κB | Anti-inflammatory | [62] |
| Tamariscinoside E | NA | |||||
| Smenospongia cerebriformis | Smenohaimiens A | 30.13 | Sesquiterpene derivatives | Anti-inflammatory | [90] | |
| Smenohaimiens B | 28.33 | |||||
| Smenohaimiens C | >40 | |||||
| Smenohaimiens D | >40 | |||||
| Smenohaimiens E 19-Hydroxy-polyfibrospongol | 24.37 24.44 | |||||
| Ilimaquinone, | 10.40 | NO | ||||
| Dictyoceratin C, | >40 | |||||
| Polyfibrospongol A, | >40 | |||||
| Polyfibrospongol B | 30.43 | |||||
| Solanum melongena L | Melongenamide C | 16.4 | Lignanamides | NO | Anti-inflammatory | [61] |
| Cannabisin F | 16.2 | |||||
| Sophora alopecuroides | Sophoraflavanone G | NA | Flavonoid | NO, PGE2, TNF-α, IL-1β, IL-6, iNOS, COX-2, JAK/STAT, P13K/AKT, MAPKs | Anti-neuroinflammatory | [72] |
| Sorbus commixta | Sorcomisides A | NA | Phenolic glycosides | Anti-inflammatory Neuroprotective Anticancer | [113] | |
| Sorcomisides B | ||||||
| Sorcomic acid | 180.12 | Fatty Acids | Anti-inflammatory Neuroprotective Anticancer | [114] | ||
| Methyl (3S,5S)-3,5-dihydroxyhexanoate | 165.03 | |||||
| (S)-(E)-4-hydroxy-2-nonenoic acid | >500 | |||||
| 3(R)-hydroxyoctanoic acid | >500 | |||||
| 9-Hydroxynonanoic acid | 187.87 | |||||
| Methyl 9-hydroxynonanoate | 196.67 | |||||
| Azelaric acid | 150.22 | |||||
| Monomethyl azelate | >500 | |||||
| (9S,12S,13R)-(E)-9,12,13-trihydroxy-10-octadecaenoic acid | 186.67 | |||||
| (9S,12R,13R)-(E)-9,12,13-trihydroxy-10-octadecaenoic acid | 71.25 | |||||
| Tilia amurensis | Orobol4’-O-β-D-apiofuranosyl-(1→6)-β-D-glucopyranoside | 23.42 | Isoflavonoid glycoside | NO | Anti-neuroinflammatory Anti-proliferative | [70] |
| Pratensein-7-O-β- D-glucoside | 32.23 | |||||
| Orobol 7-O-β-D-glucoside | 31.85 | |||||
| Orobol 4′-O-β-glucopyranoside | >200 | |||||
| Kelampayoside A | >200 | |||||
| Osmanthuside H | 25.99 | |||||
| Salidroside | 35.64 | |||||
| Dihydroconiferin | 158.49 | |||||
| Isotachioside | 50.29 | |||||
| Tachioside | 116.66 | |||||
| Koaburside | >200 | |||||
| 2-Methoxyhydroquinone | 58.87 | |||||
| Scopoletin | 37.69 | |||||
| Scopolin | 148.92 | |||||
| Fraxin | 30.02 | |||||
| n-Butyl β-D-glucopyranoside | >200 | |||||
| Adenosine | >200 | |||||
| Toddaliae asiaticae | Omphalocarpin | 7.4 | Phytoestrogen | NO, TNF-α, IL-1β, COX-2 | Anti-inflammatory | [96] |
| Toxicodendron verniciflumm | Butein | 11.68 | Phenols | NO | Cytotoxic Anti-inflammatory | [74] |
| Ulmus davidiana | Fraxetin | 18.72 | chromane derivative | Anti-inflammatory Neuroprotective | [115] | |
| (+)-lyoniresinol | 12.31 | |||||
| 4-O-β-D-glucopyranosyl vanillic acid | 21.40 | |||||
| Wasabi japonica | Allyl isothiocyanate | 20.10 | Aliphatic isothiocyanate | LPS-activated microglia neuroinflammation | [116] | |
| Zanthoxylum schinifolium | Zanthplanispine | 36.8 | Lignan | NO | Anti-inflammatory | [101] |
| Zinginber officinale | Gingerols | NA | Polyphenols | NO, NF-κB, IL-6, IL-1β, TNF-α | Anti-inflammatory Neuroprotective | [76] |
| Gingerone | NA | |||||
| Shogaol | NA | NO | ||||
| Zingerone | NA | IL-1β, IL-6, TNF-α, NF-κB, TLR4, MyD88, TRIF | ||||
| Paradol | NA | TNF-α, iNOS | ||||
| Honokiol | 17.0 | Pyrazole analogs | Anti-inflammatory | [117] | ||
| Positive control | L-NMMA | 20.53 | Methyl-arginine | Anti-inflammatory | [89] |
2. Discussion and Future Prospective
Author Contributions
Funding
Conflicts of Interest
References
- Kuriyama, K.; Ohkuma, S. Role of nitric oxide in central synaptic transmission: Effects on neurotransmitter release. Jpn. J. Pharmacol. 1995, 69, 1–8. [Google Scholar] [CrossRef]
- Luiking, Y.C.; Engelen, M.P.; Deutz, N.E. Regulation of nitric oxide production in health and disease. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Forstermann, U.; Sessa, W.C. Nitric oxide synthases: Regulation and function. Eur. Heart J. 2012, 33, 829–837. [Google Scholar] [CrossRef] [PubMed]
- Toda, N.; Ayajiki, K.; Okamura, T. Cerebral blood flow regulation by nitric oxide in neurological disorders. Can. J. Physiol. Pharmacol. 2009, 87, 581–594. [Google Scholar] [CrossRef] [PubMed]
- Esplugues, J.V. NO as a signalling molecule in the nervous system. Br. J. Pharmacol. 2002, 135, 1079–1095. [Google Scholar] [CrossRef]
- Garthwaite, J. Concepts of neural nitric oxide-mediated transmission. Eur. J. Neurosci. 2008, 27, 2783–2802. [Google Scholar] [CrossRef]
- Ashina, M.; Bendtsen, L.; Jensen, R.; Olesen, J. Nitric oxide-induced headache in patients with chronic tension-type headache. Brain 2000, 123, 1830–1837. [Google Scholar] [CrossRef]
- Olesen, J. The role of nitric oxide (NO) in migraine, tension-type headache and cluster headache. Pharmacol. Ther. 2008, 120, 157–171. [Google Scholar] [CrossRef]
- Gupta, S.; Nahas, S.J.; Peterlin, B.L. Chemical mediators of migraine: Preclinical and clinical observations. Headache 2011, 51, 1029–1045. [Google Scholar] [CrossRef]
- Sharma, J.N.; Al-Omran, A.; Parvathy, S.S. Role of nitric oxide in inflammatory diseases. Inflammopharmacology 2007, 15, 252–259. [Google Scholar] [CrossRef]
- Gaire, B.P. Herbal medicine in ischemic stroke: Challenges and prospective. Chin. J. Integr. Med. 2018, 24, 243–246. [Google Scholar] [CrossRef] [PubMed]
- Gaire, B.P.; Subedi, L. Medicinal plant diversity and their pharmacological aspects of Nepal Himalayas. Pharm. J. 2011, 3, 6–17. [Google Scholar] [CrossRef]
- Gaire, B.P.; Choi, J.W. Sphingosine 1-phosphate receptors in cerebral ischemia. Neuromol. Med. 2020, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Togo, T.; Katsuse, O.; Iseki, E. Nitric oxide pathways in Alzheimer’s disease and other neurodegenerative dementias. Neurol. Res. 2004, 26, 563–566. [Google Scholar] [CrossRef] [PubMed]
- Balez, R.; Ooi, L. Getting to NO Alzheimer’s Disease: Neuroprotection versus Neurotoxicity Mediated by Nitric Oxide. Oxid. Med. Cell. Longev. 2016, 2016. [Google Scholar] [CrossRef]
- Austin, S.A.; Santhanam, A.V.; Hinton, D.J.; Choi, D.S.; Katusic, Z.S. Endothelial nitric oxide deficiency promotes Alzheimer’s disease pathology. J. Neurochem. 2013, 127, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Montagne, A.; Barnes, S.R.; Sweeney, M.D.; Halliday, M.R.; Sagare, A.P.; Zhao, Z.; Toga, A.W.; Jacobs, R.E.; Liu, C.Y.; Amezcua, L.; et al. Blood-brain barrier breakdown in the aging human hippocampus. Neuron 2015, 85, 296–302. [Google Scholar] [CrossRef]
- Mattson, M.P. Oxidative stress, perturbed calcium homeostasis, and immune dysfunction in Alzheimer’s disease. J. Neurovirol. 2002, 8, 539–550. [Google Scholar] [CrossRef]
- Javed, H.; Khan, M.M.; Ahmad, A.; Vaibhav, K.; Ahmad, M.E.; Khan, A.; Ashafaq, M.; Islam, F.; Siddiqui, M.S.; Safhi, M.M.; et al. Rutin prevents cognitive impairments by ameliorating oxidative stress and neuroinflammation in rat model of sporadic dementia of Alzheimer type. Neuroscience 2012, 210, 340–352. [Google Scholar] [CrossRef]
- Spuch, C.; Ortolano, S.; Navarro, C. New insights in the amyloid-Beta interaction with mitochondria. J. Aging Res. 2012, 2012. [Google Scholar] [CrossRef]
- Zhang, L.; Dawson, V.L.; Dawson, T.M. Role of nitric oxide in Parkinson’s disease. Pharmacol. Ther. 2006, 109, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Aquilano, K.; Baldelli, S.; Rotilio, G.; Ciriolo, M.R. Role of nitric oxide synthases in Parkinson’s disease: A review on the antioxidant and anti-inflammatory activity of polyphenols. Neurochem. Res. 2008, 33, 2416–2426. [Google Scholar] [CrossRef] [PubMed]
- Yacoubian, T.A.; Standaert, D.G. Targets for neuroprotection in Parkinson’s disease. Biochim. Biophys. Acta 2009, 1792, 676–687. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.J.; Lassmann, H. The role of nitric oxide in multiple sclerosis. Lancet Neurol. 2002, 1, 232–241. [Google Scholar] [CrossRef]
- Lan, M.; Tang, X.; Zhang, J.; Yao, Z. Insights in pathogenesis of multiple sclerosis: Nitric oxide may induce mitochondrial dysfunction of oligodendrocytes. Rev. Neurosci. 2018, 29, 39–53. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.Q.; Mou, R.T.; Feng, D.X.; Wang, Z.; Chen, G. The role of nitric oxide in stroke. Med. Gas Res. 2017, 7, 194–203. [Google Scholar] [PubMed]
- Castillo, J.; Rama, R.; Davalos, A. Nitric oxide-related brain damage in acute ischemic stroke. Stroke 2000, 31, 852–857. [Google Scholar] [CrossRef] [PubMed]
- Urushitani, M.; Shimohama, S. The role of nitric oxide in amyotrophic lateral sclerosis. Amyotroph. Lateral. Scler. Other. Motor. Neuron. Disord. 2001, 2, 71–81. [Google Scholar]
- Yui, K.; Kawasaki, Y.; Yamada, H.; Ogawa, S. Oxidative Stress and Nitric Oxide in Autism Spectrum Disorder and Other Neuropsychiatric Disorders. CNS Neurol. Disord. Drug Targets 2016, 15, 587–596. [Google Scholar] [CrossRef]
- Steinert, J.R.; Chernova, T.; Forsythe, I.D. Nitric oxide signaling in brain function, dysfunction, and dementia. Neuroscientist 2010, 16, 435–452. [Google Scholar] [CrossRef]
- Ahlawat, A.; Rana, A.; Goyal, N.; Sharma, S. Potential role of nitric oxide synthase isoforms in pathophysiology of neuropathic pain. Inflammopharmacology 2014, 22, 269–278. [Google Scholar] [CrossRef]
- Naik, A.K.; Tandan, S.K.; Kumar, D.; Dudhgaonkar, S.P. Nitric oxide and its modulators in chronic constriction injury-induced neuropathic pain in rats. Eur. J. Pharmacol. 2006, 530, 59–69. [Google Scholar] [CrossRef]
- Olesen, J.; Thomsen, L.L.; Iversen, H. Nitric oxide is a key molecule in migraine and other vascular headaches. Trends Pharmacol. Sci. 1994, 15, 149–153. [Google Scholar] [CrossRef]
- Neeb, L.; Reuter, U. Nitric oxide in migraine. CNS Neurol. Disord. Drug Targets 2007, 6, 258–264. [Google Scholar] [CrossRef]
- Van der Kuy, P.H.; Lohman, J.J. The role of nitric oxide in vascular headache. Pharm. World Sci. 2003, 25, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, M.; Harandizadeh, F.; Niazmand, S.; Soukhtanloo, M.; Faizpour, A.; Ghasemabady, M. The role for nitric oxide on the effects of hydroalcoholic extract of Achillea wilhelmsii on seizure. Avic. J. Phytomed. 2014, 4, 251–259. [Google Scholar]
- Faradji, H.; Rousset, C.; Debilly, G.; Vergnes, M.; Cespuglio, R. Sleep and epilepsy: A key role for nitric oxide? Epilepsia 2000, 41, 794–801. [Google Scholar] [CrossRef]
- Levy, D.; Zochodne, D.W. NO pain: Potential roles of nitric oxide in neuropathic pain. Pain Pract. 2004, 4, 11–18. [Google Scholar] [CrossRef]
- Prabhakar, S.S. Role of nitric oxide in diabetic nephropathy. Semin. Nephrol. 2004, 24, 333–344. [Google Scholar] [CrossRef]
- Zheng, L.; Kern, T.S. Role of nitric oxide, superoxide, peroxynitrite and PARP in diabetic retinopathy. Front. Biosci. 2009, 14, 3974–3987. [Google Scholar] [CrossRef]
- Torre, D.; Pugliese, A.; Speranza, F. Role of nitric oxide in HIV-1 infection: Friend or foe? Lancet Infect. Dis. 2002, 2, 273–280. [Google Scholar] [CrossRef]
- Liu, X.; Jana, M.; Dasgupta, S.; Koka, S.; He, J.; Wood, C.; Pahan, K. Human immunodeficiency virus type 1 (HIV-1) tat induces nitric-oxide synthase in human astroglia. J. Biol. Chem. 2002, 277, 39312–39319. [Google Scholar] [CrossRef] [PubMed]
- Seiler, K.P.; Vavrin, Z.; Eichwald, E.; Hibbs, J.B., Jr.; Weis, J.J. Nitric oxide production during murine Lyme disease: Lack of involvement in host resistance or pathology. Infect. Immun. 1995, 63, 3886–3895. [Google Scholar] [CrossRef] [PubMed]
- Brundin, L.; Svenungsson, E.; Morcos, E.; Andersson, M.; Olsson, T.; Lundberg, I.; Wiklund, N.P. Central nervous system nitric oxide formation in cerebral systemic lupus erythematosus. Ann. Neurol. 1998, 44, 704–706. [Google Scholar] [CrossRef]
- Kwak, E.K.; Kim, J.W.; Kang, K.S.; Lee, Y.H.; Hua, Q.H.; Park, T.I.; Park, J.Y.; Sohn, Y.K. The role of inducible nitric oxide synthase following spinal cord injury in rat. J. Korean Med. Sci. 2005, 20, 663–669. [Google Scholar] [CrossRef]
- Tardivo, V.; Crobeddu, E.; Pilloni, G.; Fontanella, M.; Spena, G.; Panciani, P.P.; Berjano, P.; Ajello, M.; Bozzaro, M.; Agnoletti, A.; et al. Say “no” to spinal cord injury: Is nitric oxide an option for therapeutic strategies? Int. J. Neurosci. 2015, 125, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Vaziri, N.D. Potential risks of nitric oxide inhibition in spinal cord injury. J. Spinal. Cord Med. 2007, 30, 3–4. [Google Scholar] [CrossRef][Green Version]
- Lee, E.-J.; Choi, M.-J.; Lee, G.; Gaire, B.P.; Choi, J.W.; Kim, H.-S. Regulation of neuroinflammation by matrix metalloproteinase-8 inhibitor derivatives in activated microglia and astrocytes. Oncotarget 2017, 8, 78677–78690. [Google Scholar] [CrossRef]
- Han, Q.; Yuan, Q.; Meng, X.; Huo, J.; Bao, Y.; Xie, G. 6-Shogaol attenuates LPS-induced inflammation in BV2 microglia cells by activating PPAR-gamma. Oncotarget 2017, 8, 42001–42006. [Google Scholar] [CrossRef] [PubMed]
- Chi, D.S.; Qui, M.; Krishnaswamy, G.; Li, C.; Stone, W. Regulation of nitric oxide production from macrophages by lipopolysaccharide and catecholamines. Nitric Oxide 2003, 8, 127–132. [Google Scholar] [CrossRef]
- Salim, T.; Sershen, C.L.; May, E.E. Investigating the Role of TNF-alpha and IFN-gamma Activation on the Dynamics of iNOS Gene Expression in LPS Stimulated Macrophages. PLoS ONE 2016, 11, e0153289. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, R.; Xapelli, S.; Santos, T.; Silva, A.P.; Cristovao, A.; Cortes, L.; Malva, J.O. Neuropeptide Y modulation of interleukin-1{beta} (IL-1{beta})-induced nitric oxide production in microglia. J. Biol. Chem. 2010, 285, 41921–41934. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Krukoff, T.L. Estrogen induces nitric oxide production via activation of constitutive nitric oxide synthases in human neuroblastoma cells. Endocrinology 2004, 145, 4550–4557. [Google Scholar] [CrossRef] [PubMed]
- Ko, S.H.; Ryu, G.R.; Kim, S.; Ahn, Y.B.; Yoon, K.H.; Kaneto, H.; Ha, H.; Kim, Y.S.; Song, K.H. Inducible nitric oxide synthase-nitric oxide plays an important role in acute and severe hypoxic injury to pancreatic beta cells. Transplantation 2008, 85, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Boyadjieva, N.I.; Sarkar, D.K. Microglia play a role in ethanol-induced oxidative stress and apoptosis in developing hypothalamic neurons. Alcohol Clin. Exp. Res. 2013, 37, 252–262. [Google Scholar] [CrossRef]
- Subedi, L.; Lee, J.H.; Gaire, B.P.; Kim, S.Y. Sulforaphane Inhibits MGO-AGE-Mediated Neuroinflammation by Suppressing NF-κB, MAPK, and AGE–RAGE Signaling Pathways in Microglial Cells. Antioxidants 2020, 9, 792. [Google Scholar] [CrossRef]
- Lim, S.Y.; Subedi, L.; Shin, D.; Kim, C.S.; Lee, K.R.; Kim, S.Y. A New Neolignan Derivative, Balanophonin Isolated from Firmiana simplex Delays the Progress of Neuronal Cell Death by Inhibiting Microglial Activation. Biomol. Ther. 2017, 25, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.S.; Subedi, L.; Kim, S.Y.; Choi, S.U.; Kim, K.H.; Lee, K.R. Lignan Glycosides from the Twigs of Chaenomeles sinensis and Their Biological Activities. J. Nat. Prod. 2015, 78, 1174–1178. [Google Scholar] [CrossRef]
- Xiong, J.; Bui, V.B.; Liu, X.H.; Hong, Z.L.; Yang, G.X.; Hu, J.F. Lignans from the stems of Clematis armandii (“Chuan-Mu-Tong”) and their anti-neuroinflammatory activities. J. Ethnopharmacol. 2014, 153, 737–743. [Google Scholar] [CrossRef]
- Pan, L.L.; Zhang, Q.Y.; Luo, X.L.; Xiong, J.; Xu, P.; Liu, S.Y.; Hu, J.F.; Liu, X.H. (7R,8S)-9-acetyl-dehydrodiconiferyl alcohol inhibits inflammation and migration in lipopolysaccharide-stimulated macrophages. Phytomedicine 2016, 23, 541–549. [Google Scholar] [CrossRef]
- Sun, J.; Gu, Y.F.; Su, X.Q.; Li, M.M.; Huo, H.X.; Zhang, J.; Zeng, K.W.; Zhang, Q.; Zhao, Y.F.; Li, J.; et al. Anti-inflammatory lignanamides from the roots of Solanum melongena L. Fitoterapia 2014, 98, 110–116. [Google Scholar] [CrossRef]
- Dat, L.D.; Zhao, B.T.; Hung, N.D.; Lee, J.H.; Min, B.S.; Woo, M.H. Lignan derivatives from Selaginella tamariscina and their nitric oxide inhibitory effects in LPS-stimulated RAW 264.7 cells. Bioorg. Med. Chem. Lett. 2017, 27, 524–529. [Google Scholar] [CrossRef] [PubMed]
- Suh, W.S.; Kim, C.S.; Subedi, L.; Kim, S.Y.; Choi, S.U.; Lee, K.R. Iridoid Glycosides from the Twigs of Sambucus williamsii var. coreana and Their Biological Activities. J. Nat. Prod. 2017, 80, 2502–2508. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.W.; Quang, T.H.; Ko, W.; Kim, D.C.; Yoon, C.S.; Oh, H.; Kim, Y.C. Anti-neuroinflammatory effects of cudraflavanone A isolated from the chloroform fraction of Cudrania tricuspidata root bark. Pharm. Biol. 2018, 56, 192–200. [Google Scholar] [CrossRef]
- Subedi, L.; Ji, E.; Shin, D.; Jin, J.; Yeo, J.H.; Kim, S.Y. Equol, a Dietary Daidzein Gut Metabolite Attenuates Microglial Activation and Potentiates Neuroprotection In Vitro. Nutrients 2017, 9, 207. [Google Scholar] [CrossRef] [PubMed]
- Chinta, S.J.; Ganesan, A.; Reis-Rodrigues, P.; Lithgow, G.J.; Andersen, J.K. Anti-inflammatory role of the isoflavone diadzein in lipopolysaccharide-stimulated microglia: Implications for Parkinson’s disease. Neurotox. Res. 2013, 23, 145–153. [Google Scholar] [CrossRef]
- Scuro, L.S.; Simioni, P.U.; Grabriel, D.L.; Saviani, E.E.; Modolo, L.V.; Tamashiro, W.M.; Salgado, I. Suppression of nitric oxide production in mouse macrophages by soybean flavonoids accumulated in response to nitroprusside and fungal elicitation. BMC Biochem. 2004, 5, 5. [Google Scholar] [CrossRef]
- Sapkota, A.; Gaire, B.P.; Cho, K.S.; Jeon, S.J.; Kwon, O.W.; Jang, D.S.; Kim, S.Y.; Ryu, J.H.; Choi, J.W. Eupatilin exerts neuroprotective effects in mice with transient focal cerebral ischemia by reducing microglial activation. PLoS ONE 2017, 12, e0171479. [Google Scholar] [CrossRef]
- Ji, G.; Zhang, Y.; Yang, Q.; Cheng, S.; Hao, J.; Zhao, X.; Jiang, Z. Genistein suppresses LPS-induced inflammatory response through inhibiting NF-kappaB following AMP kinase activation in RAW 264.7 macrophages. PLoS ONE 2012, 7, e53101. [Google Scholar] [CrossRef]
- Kim, K.H.; Moon, E.; Cha, J.M.; Lee, S.; Yu, J.S.; Kim, C.S.; Kim, S.Y.; Choi, S.U.; Lee, K.R. Antineuroinflammatory and Antiproliferative Activities of Constituents from Tilia amurensis. Chem. Pharm. Bull. 2015, 63, 837–842. [Google Scholar] [CrossRef]
- Kim, C.S.; Bae, M.; Oh, J.; Subedi, L.; Suh, W.S.; Choi, S.Z.; Son, M.W.; Kim, S.Y.; Choi, S.U.; Oh, D.C.; et al. Anti-Neurodegenerative Biflavonoid Glycosides from Impatiens balsamina. J. Nat. Prod. 2017, 80, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Yang, L.; Wan, C.X.; Xia, Y.Z.; Zhang, C.; Chen, M.H.; Wang, Z.D.; Li, Z.R.; Li, X.M.; Geng, Y.D.; et al. Anti-neuroinflammatory effect of Sophoraflavanone G from Sophora alopecuroides in LPS-activated BV2 microglia by MAPK, JAK/STAT and Nrf2/HO-1 signaling pathways. Phytomedicine 2016, 23, 1629–1637. [Google Scholar] [CrossRef] [PubMed]
- Shu, Z.; Yang, B.; Zhao, H.; Xu, B.; Jiao, W.; Wang, Q.; Wang, Z.; Kuang, H. Tangeretin exerts anti-neuroinflammatory effects via NF-kappaB modulation in lipopolysaccharide-stimulated microglial cells. Int. Immunopharmacol. 2014, 19, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Moon, E.; Choi, S.U.; Pang, C.; Kim, S.Y.; Lee, K.R. Identification of cytotoxic and anti-inflammatory constituents from the bark of Toxicodendron vernicifluum (Stokes) F.A. Barkley. J. Ethnopharmacol. 2015, 162, 231–237. [Google Scholar] [CrossRef]
- Woo, K.W.; Moon, E.; Kwon, O.W.; Lee, S.O.; Kim, S.Y.; Choi, S.Z.; Son, M.W.; Lee, K.R. Anti-neuroinflammatory diarylheptanoids from the rhizomes of Dioscorea nipponica. Bioorg. Med. Chem. Lett. 2013, 23, 3806–3809. [Google Scholar] [CrossRef]
- Ho, S.C.; Chang, K.S.; Lin, C.C. Anti-neuroinflammatory capacity of fresh ginger is attributed mainly to 10-gingerol. Food Chem. 2013, 141, 3183–3191. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Min, J.S.; Chae, U.; Lee, J.Y.; Song, K.S.; Lee, H.S.; Lee, H.J.; Lee, S.R.; Lee, D.S. Anti-inflammatory effect of oleuropein on microglia through regulation of Drp1-dependent mitochondrial fission. J. Neuroimmunol. 2017, 306, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Gaire, B.P.; Kwon, O.W.; Park, S.H.; Chun, K.H.; Kim, S.Y.; Shin, D.Y.; Choi, J.W. Neuroprotective effect of 6-paradol in focal cerebral ischemia involves the attenuation of neuroinflammatory responses in activated microglia. PLoS ONE 2015, 10, e0120203. [Google Scholar] [CrossRef]
- Kim, C.S.; Subedi, L.; Park, K.J.; Kim, S.Y.; Choi, S.U.; Kim, K.H.; Lee, K.R. Salicin derivatives from Salix glandulosa and their biological activities. Fitoterapia 2015, 106, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Azam, F.; Amer, A.M.; Abulifa, A.R.; Elzwawi, M.M. Ginger components as new leads for the design and development of novel multi-targeted anti-Alzheimer’s drugs: A computational investigation. Drug Des. Devel. Ther. 2014, 8, 2045–2059. [Google Scholar] [CrossRef]
- Ruangsuriya, J.; Budprom, P.; Viriyakhasem, N.; Kongdang, P.; Chokchaitaweesuk, C.; Sirikaew, N.; Chomdej, S.; Nganvongpanit, K.; Ongchai, S. Suppression of Cartilage Degradation by Zingerone Involving the p38 and JNK MAPK Signaling Pathway. Planta Med. 2017, 83, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Fan, H.J.; Li, H.; Ding, H.; Lv, Q.; Hou, S.K. Zingerone ameliorates lipopolysaccharide-induced acute kidney injury by inhibiting Toll-like receptor 4 signaling pathway. Eur. J. Pharmacol. 2016, 772, 108–114. [Google Scholar] [CrossRef]
- Raju, R.; Gunawardena, D.; Ahktar, M.A.; Low, M.; Reddell, P.; Munch, G. Anti-Inflammatory Chemical Profiling of the Australian Rainforest Tree Alphitonia petriei (Rhamnaceae). Molecules 2016, 21, 1521. [Google Scholar] [CrossRef]
- Mesaik, A.M.; Poh, H.W.; Bin, O.Y.; Elawad, I.; Alsayed, B. In Vivo Anti-Inflammatory, Anti-Bacterial and Anti-Diarrhoeal Activity of Ziziphus Jujuba Fruit Extract. Open Access Maced. J. Med. Sci. 2018, 6, 757–766. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.S.; Lee, D.S.; Kim, D.C.; Yoon, C.S.; Ko, W.; Oh, H.; Kim, Y.C. Anti-Inflammatory Effects and Mechanisms of Action of Coussaric and Betulinic Acids Isolated from Diospyros kaki in Lipopolysaccharide-Stimulated RAW 264.7 Macrophages. Molecules 2016, 21, 1206. [Google Scholar] [CrossRef]
- Park, K.J.; Subedi, L.; Kim, S.Y.; Choi, S.U.; Lee, K.R. Bioactive triterpenoids from twigs of Betula schmidtii. Bioorg. Chem. 2018, 77, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.S.; Baek, J.; Park, H.B.; Moon, E.; Kim, S.Y.; Choi, S.U.; Kim, K.H. A new rearranged eudesmane sesquiterpene and bioactive sesquiterpenes from the twigs of Lindera glauca (Sieb. et Zucc.) Blume. Arch. Pharm. Res. 2016, 39, 1628–1634. [Google Scholar] [CrossRef]
- Suh, W.S.; Park, K.J.; Kim, D.H.; Subedi, L.; Kim, S.Y.; Choi, S.U.; Lee, K.R. A biphenyl derivative from the twigs of Chaenomeles speciosa. Bioorg. Chem. 2017, 72, 156–160. [Google Scholar] [CrossRef]
- Kim, C.S.; Oh, J.; Subedi, L.; Kim, S.Y.; Choi, S.U.; Lee, K.R. Holophyllane A: A Triterpenoid Possessing an Unprecedented B-nor-3,4-seco-17,14-friedo-lanostane Architecture from Abies holophylla. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Kiem, P.V.; Huyen, L.T.; Hang, D.T.; Nhiem, N.X.; Tai, B.H.; Anh, H.L.; Cuong, P.V.; Quang, T.H.; Minh, C.V.; Dau, N.V.; et al. Sesquiterpene derivatives from marine sponge Smenospongia cerebriformis and their anti-inflammatory activity. Bioorg. Med. Chem. Lett. 2017, 27, 1525–1529. [Google Scholar] [CrossRef]
- Yap, W.H.; Lim, Y.M. Mechanistic Perspectives of Maslinic Acid in Targeting Inflammation. Biochem. Res. Int. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Park, W.H.; Kang, S.; Piao, Y.; Pak, C.J.; Oh, M.S.; Kim, J.; Kang, M.S.; Pak, Y.K. Ethanol extract of Bupleurum falcatum and saikosaponins inhibit neuroinflammation via inhibition of NF-kappaB. J. Ethnopharmacol. 2015, 174, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Zeng, K.W.; Wang, S.; Dong, X.; Jiang, Y.; Tu, P.F. Sesquiterpene dimer (DSF-52) from Artemisia argyi inhibits microglia-mediated neuroinflammation via suppression of NF-kappaB, JNK/p38 MAPKs and Jak2/Stat3 signaling pathways. Phytomedicine 2014, 21, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Nhiem, N.X.; Tuong, N.T.; Ky, P.T.; Subedi, L.; Park, S.J.; Ngoc, T.M.; Yen, P.H.; Tai, B.H.; Quang, T.H.; Kiem, P.V.; et al. Chemical Components from Phaeanthus vietnamensis and Their Inhibitory NO Production in BV2 Cells. Chem. Biodivers. 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Bora, K.S.; Sharma, A. Phytochemical and pharmacological potential of Medicago sativa: A review. Pharm. Biol. 2011, 49, 211–220. [Google Scholar] [CrossRef]
- Tong, L.; Chen, T.; Chen, Z.; Zhang, P.; Pi, H.; Ruan, H.; Wu, J. Anti-inflammatory activity of omphalocarpin isolated from Radix Toddaliae Asiaticae. J. Ethnopharmacol. 2014, 155, 1553–1560. [Google Scholar] [CrossRef]
- Kim, C.S.; Subedi, L.; Kwon, O.K.; Kim, S.Y.; Yeo, E.J.; Choi, S.U.; Lee, K.R. Isolation of bioactive biphenyl compounds from the twigs of Chaenomeles sinensis. Bioorg. Med. Chem. Lett. 2016, 26, 351–354. [Google Scholar] [CrossRef]
- Noh, H.J.; Hwang, D.; Lee, E.S.; Hyun, J.W.; Yi, P.H.; Kim, G.S.; Lee, S.E.; Pang, C.; Park, Y.J.; Chung, K.H.; et al. Anti-inflammatory activity of a new cyclic peptide, citrusin XI, isolated from the fruits of Citrus unshiu. J. Ethnopharmacol. 2015, 163, 106–112. [Google Scholar] [CrossRef]
- Subedi, L.; Gaire, B.P.; Do, M.H.; Lee, T.H.; Kim, S.Y. Anti-neuroinflammatory and neuroprotective effects of the Lindera neesiana fruit in vitro. Phytomedicine 2016, 23, 872–881. [Google Scholar] [CrossRef]
- Kim, K.H.; Moon, E.; Kim, S.Y.; Choi, S.U.; Lee, J.H.; Lee, K.R. 4-Methylthio-butanyl derivatives from the seeds of Raphanus sativus and their biological evaluation on anti-inflammatory and antitumor activities. J. Ethnopharmacol. 2014, 151, 503–508. [Google Scholar] [CrossRef]
- Su, G.Y.; Cheng, Y.C.; Wang, K.W.; Wang, X.Y.; Wu, B. An Unusual Tetrahydrofuran Lignan from the Roots of Zanthoxylum planispinum and the Potential Anti-inflammatory Effects. Chem. Biodivers. 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Park, K.J.; Cha, J.M.; Subedi, L.; Kim, S.Y.; Lee, K.R. Phenolic constituents from the twigs of Betula schmidtii collected in Goesan, Korea. Phytochemistry 2019, 167. [Google Scholar] [CrossRef]
- Subedi, L.; Cho, K.; Park, Y.U.; Choi, H.J.; Kim, S.Y. Sulforaphane-enriched broccoli sprouts pretreated by pulsed electric fields reduces neuroinflammation and ameliorates scopolamine-induced amnesia in mouse brain through its antioxidant ability via Nrf2-HO-1 activation. Oxid. Med. Cell. Longev. 2019, 2019. [Google Scholar] [CrossRef] [PubMed]
- Subedi, L.; Lee, J.H.; Yumnam, S.; Ji, E.; Kim, S.Y. Anti-inflammatory effect of sulforaphane on LPS-activated microglia potentially through JNK/AP-1/NF-κB inhibition and Nrf2/HO-1 activation. Cells 2019, 8, 194. [Google Scholar] [CrossRef]
- Cha, J.M.; Suh, W.S.; Lee, T.H.; Subedi, L.; Kim, S.Y.; Lee, K.R. Phenolic Glycosides from Capsella bursa-pastoris (L.) Medik and Their Anti-Inflammatory Activity. Molecules 2017, 22, 1203. [Google Scholar] [CrossRef]
- Kim, C.S.; Subedi, L.; Oh, J.; Kim, S.Y.; Choi, S.U.; Lee, K.R. Bioactive Triterpenoids from the Twigs of Chaenomeles sinensis. J. Nat. Prod. 2017, 80, 1134–1140. [Google Scholar] [CrossRef] [PubMed]
- Woo, K.W.; Park, J.E.; Cha, J.M.; Subedi, L.; Kim, S.Y.; Lee, K.R. Three New Lignan Glycosides from the Firmiana simplex. Chem. Pharm. Bull. 2019, 67, 18–22. [Google Scholar] [CrossRef]
- Woo, K.W.; Suh, W.S.; Subedi, L.; Kim, S.Y.; Choi, S.U.; Kim, K.H.; Lee, K.R. Phenolic derivatives from the stems of Lagerstroemia indica and their biological activity. Heterocycles 2015, 91, 2355–2366. [Google Scholar]
- Suh, W.S.; Kwon, O.K.; Lee, T.H.; Subedi, L.; Kim, S.Y.; Lee, K.R. Secoiridoid Glycosides from the Twigs of Ligustrum obtusifolium Possess Anti-inflammatory and Neuroprotective Effects. Chem. Pharm. Bull. 2018, 66, 78–83. [Google Scholar] [CrossRef]
- Subedi, L.; Kwon, O.W.; Pak, C.; Lee, G.; Lee, K.; Kim, H.; Kim, S.Y. N,N-disubstituted azines attenuate LPS-mediated neuroinflammation in microglia and neuronal apoptosis via inhibiting MAPK signaling pathways. BMC Neurosci. 2017, 18, 82. [Google Scholar] [CrossRef]
- Wang, S.Y.; Lan, X.Y.; Xiao, J.H.; Yang, J.C.; Kao, Y.T.; Chang, S.T. Antiinflammatory activity of Lindera erythrocarpa fruits. Phytotherapy. Res. 2007, 22, 213–216. [Google Scholar] [CrossRef] [PubMed]
- Park, K.J.; Khan, Z.; Subedi, L.; Kim, S.Y.; Lee, K.R. Antineurodegenerative labdane diterpenoid glycosides from the twigs of Pinus koraiensis. J. Nat. Prod. 2020, 83, 1794–1803. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.S.; Oh, J.; Subedi, L.; Kim, S.Y.; Choi, S.U.; Lee, K.R. Two New Phenolic Glycosides from Sorbus commixta. Chem. Pharm. Bull. 2018, 66, 839–842. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.S.; Suh, W.S.; Subedi, L.; Kim, S.Y.; Choi, S.U.; Lee, K.R. Neuroprotective Fatty Acids from the Stem Bark of Sorbus commixta. Lipids 2016, 51, 989–995. [Google Scholar] [CrossRef]
- So, H.M.; Yu, J.S.; Khan, Z.; Subedi, L.; Ko, Y.-J.; Lee, I.K.; Park, W.S.; Chung, S.J.; Ahn, M.-J.; Kim, S.Y. Chemical constituents of the root bark of Ulmus davidiana var. japonica and their potential biological activities. Bioorg. Chem. 2019, 91. [Google Scholar] [CrossRef]
- Subedi, L.; Venkatesan, R.; Kim, S.Y. Neuroprotective and Anti-Inflammatory Activities of Allyl Isothiocyanate through Attenuation of JNK/NF-kappaB/TNF-alpha Signaling. Int. J. Mol. Sci. 2017, 18, 1423. [Google Scholar] [CrossRef]
- Yuan, Y.; Subedi, L.; Lim, D.; Jung, J.-K.; Kim, S.Y.; Seo, S.-Y. Synthesis and anti-neuroinflammatory activity of N-heterocyclic analogs based on natural biphenyl-neolignan honokiol. Bioorg. Med. Chem. Lett. 2019, 29, 329–333. [Google Scholar] [CrossRef]
- Parveen, A.; Subedi, L.; Kim, H.W.; Khan, Z.; Zahra, Z.; Farooqi, M.Q.; Kim, S.Y. Phytochemicals targeting VEGF and VEGF-related multifactors as anticancer therapy. J. Clin. Med. 2019, 8, 350. [Google Scholar] [CrossRef]
- Parveen, A.; Kim, J.H.; Oh, B.G.; Subedi, L.; Khan, Z.; Kim, S.Y. Phytochemicals: Target-based therapeutic strategies for diabetic retinopathy. Molecules 2018, 23, 1519. [Google Scholar] [CrossRef]
- Parveen, A.; Jin, M.; Kim, S.Y. Bioactive phytochemicals that regulate the cellular processes involved in diabetic nephropathy. Phytomedicine 2018, 39, 146–159. [Google Scholar] [CrossRef]
- Parveen, A.; Farooqi, M.Q.; Kyunn, W.W.; Arshad, M. Phytochemical screening and content determination of different species of genus Caesalpinia belonging to different origin with antidiabetic activity. Pharm. J. 2017, 9. [Google Scholar] [CrossRef]
- Parveen, A.; Farooq, M.A.; Kyunn, W.W. A New Oleanane Type Saponin from the Aerial Parts of Nigella sativa with Anti-Oxidant and Anti-Diabetic Potential. Molecules 2020, 25, 2171. [Google Scholar] [CrossRef]
- Do, M.H.; Lee, J.H.; Cho, K.; Kang, M.C.; Subedi, L.; Parveen, A.; Kim, S.Y. Therapeutic potential of Lespedeza bicolor to prevent methylglyoxal-induced glucotoxicity in familiar diabetic nephropathy. J. Clin. Med. 2019, 8, 1138. [Google Scholar] [CrossRef] [PubMed]
- Parveen, A.; Choi, S.; Kang, J.-H.; Oh, S.H.; Kim, S.Y. Trifostigmanoside I, an Active Compound from Sweet Potato, Restores the Activity of MUC2 and Protects the Tight Junctions through PKCα/β to Maintain Intestinal Barrier Function. Int. J. Mol. Sci. 2021, 22, 291. [Google Scholar] [CrossRef] [PubMed]
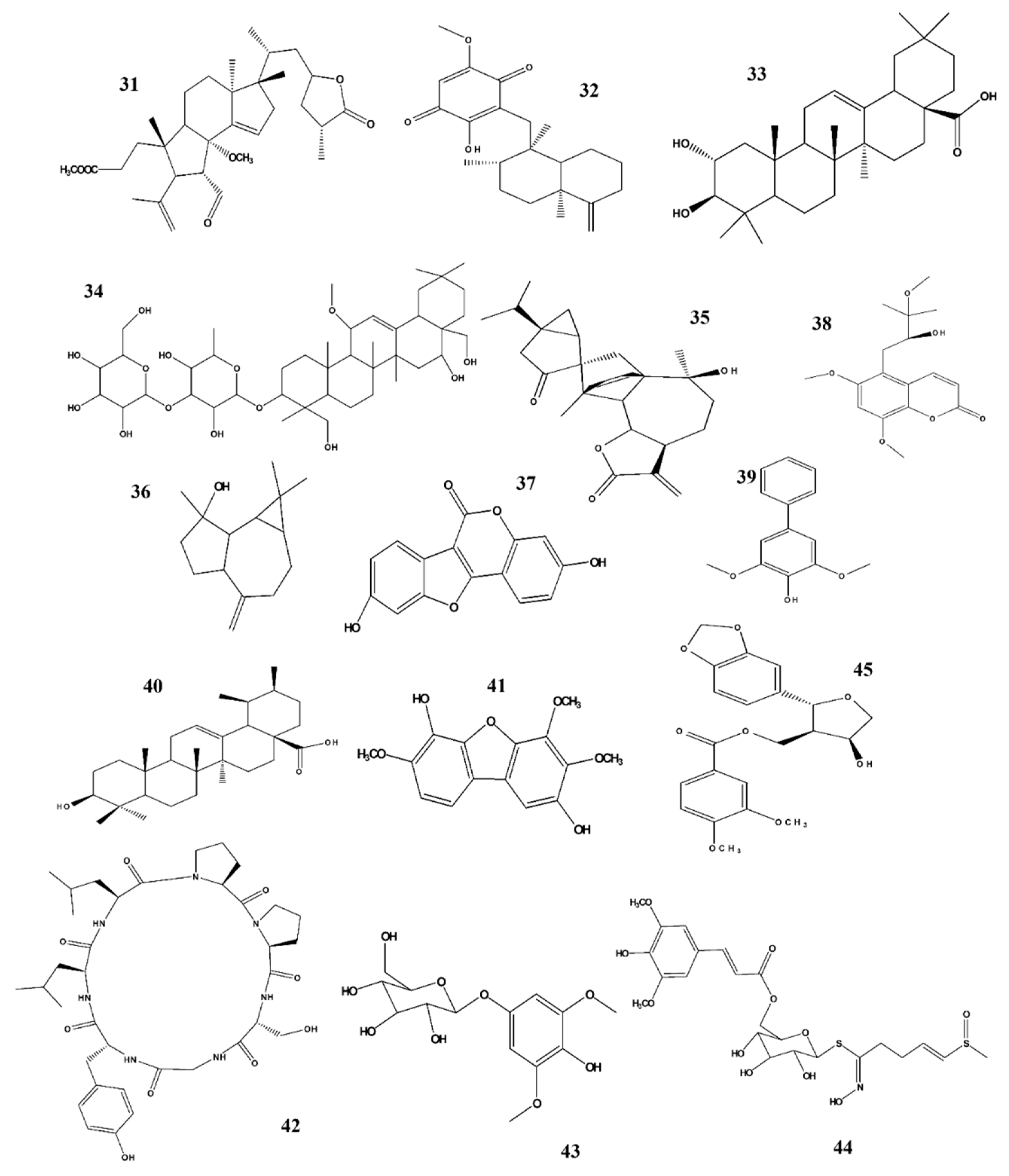
| No. | Potential NO Inhibitor | IC50 (μM) |
|---|---|---|
| 1. | Sambucasinol B | 0.9 |
| 2. | Erythro-(7S,8R)-Guaiacylglycerol-β-coniferyl aldehyde ether | 0.925 |
| 3. | Threo-(7R,8R)-Guaiacylglycerol-β-coniferyl aldehyde ether | 1.05 |
| 4. | sambucuside E | 1.2 |
| 5. | sambucuside C | 1.3 |
| 6. | Betulinic acid | 2.38 |
| 7. | Equol | 3.45 |
| 8. | (+)-faurinone | 3.67 |
| 9. | Maslinic acid | 4.46 |
| 10. | 2α-O-benzoyl-3β,19α-dihydroxy-urs-12-en-28-oic acid | 4.92 |
| 11. | Obtusifolisides B | 5.45 |
| 12. | 23-hydroxybetulinic acid | 6.7 |
| 13. | Sulforaphane | 5.85 |
| 14. | Sambucasinol A | 6.82 |
| 15. | 1-cinnamoyltrichilinin | 7.73 |
| 16. | Morolic acid 3-O-caffeate | 8.62 |
| 17. | kaempferol | 8.86 |
| 18. | Erythro-syringylglycerol-8-O-4′-coniferyl alcohol ether | 9.14 |
| 19. | 2α-O-benzoyl-19α-hydroxy-3-oxo-urs-12-en-28-oic acid | 9.68 |
| 20. | Methoxy-Balanophonin | 10 |
| 21. | Balanophonin | 10.25 |
| 22. | Ilimaquinone | 10.4 |
| 23. | Genistein | 10.63 |
| 24. | Oleuropein | 11.17 |
| 25. | Costic acid | 12.13 |
| 26. | (+)-lyoniresinol | 12.31 |
| 27. | Holophyllane A | 12.74 |
| 28. | Ilexgenin B | 12.72 |
| 29. | 3-O-cis-caffeoylbetulinic | 13.4 |
| 30. | Daidzein | 14.09 |
| 31. | Alphitolic acid | 14.5 |
| 32. | Dihydrodehydroconiferyl alcohol | 14.6 |
| 33. | Neonuezhenide | 14.69 |
| 34. | Ilicic acid | 14.92 |
| 35. | Trichilinin B | 15.28 |
| 36. | Eudeglaucone | 15.9 |
| 37. | (2″R)-2″- methoxyoleuropein | 15.45 |
| 38. | Fraxetin | 18.72 |
| 39. | 1-desacetylnimbolinin B | 18.75 |
| 40. | L-NMMA | 20.53 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Subedi, L.; Gaire, B.P.; Kim, S.-Y.; Parveen, A. Nitric Oxide as a Target for Phytochemicals in Anti-Neuroinflammatory Prevention Therapy. Int. J. Mol. Sci. 2021, 22, 4771. https://doi.org/10.3390/ijms22094771
Subedi L, Gaire BP, Kim S-Y, Parveen A. Nitric Oxide as a Target for Phytochemicals in Anti-Neuroinflammatory Prevention Therapy. International Journal of Molecular Sciences. 2021; 22(9):4771. https://doi.org/10.3390/ijms22094771
Chicago/Turabian StyleSubedi, Lalita, Bhakta Prasad Gaire, Sun-Yeou Kim, and Amna Parveen. 2021. "Nitric Oxide as a Target for Phytochemicals in Anti-Neuroinflammatory Prevention Therapy" International Journal of Molecular Sciences 22, no. 9: 4771. https://doi.org/10.3390/ijms22094771
APA StyleSubedi, L., Gaire, B. P., Kim, S.-Y., & Parveen, A. (2021). Nitric Oxide as a Target for Phytochemicals in Anti-Neuroinflammatory Prevention Therapy. International Journal of Molecular Sciences, 22(9), 4771. https://doi.org/10.3390/ijms22094771







