Effect of Whole Tissue Culture and Basic Fibroblast Growth Factor on Maintenance of Tie2 Molecule Expression in Human Nucleus Pulposus Cells
Abstract
1. Introduction
2. Results
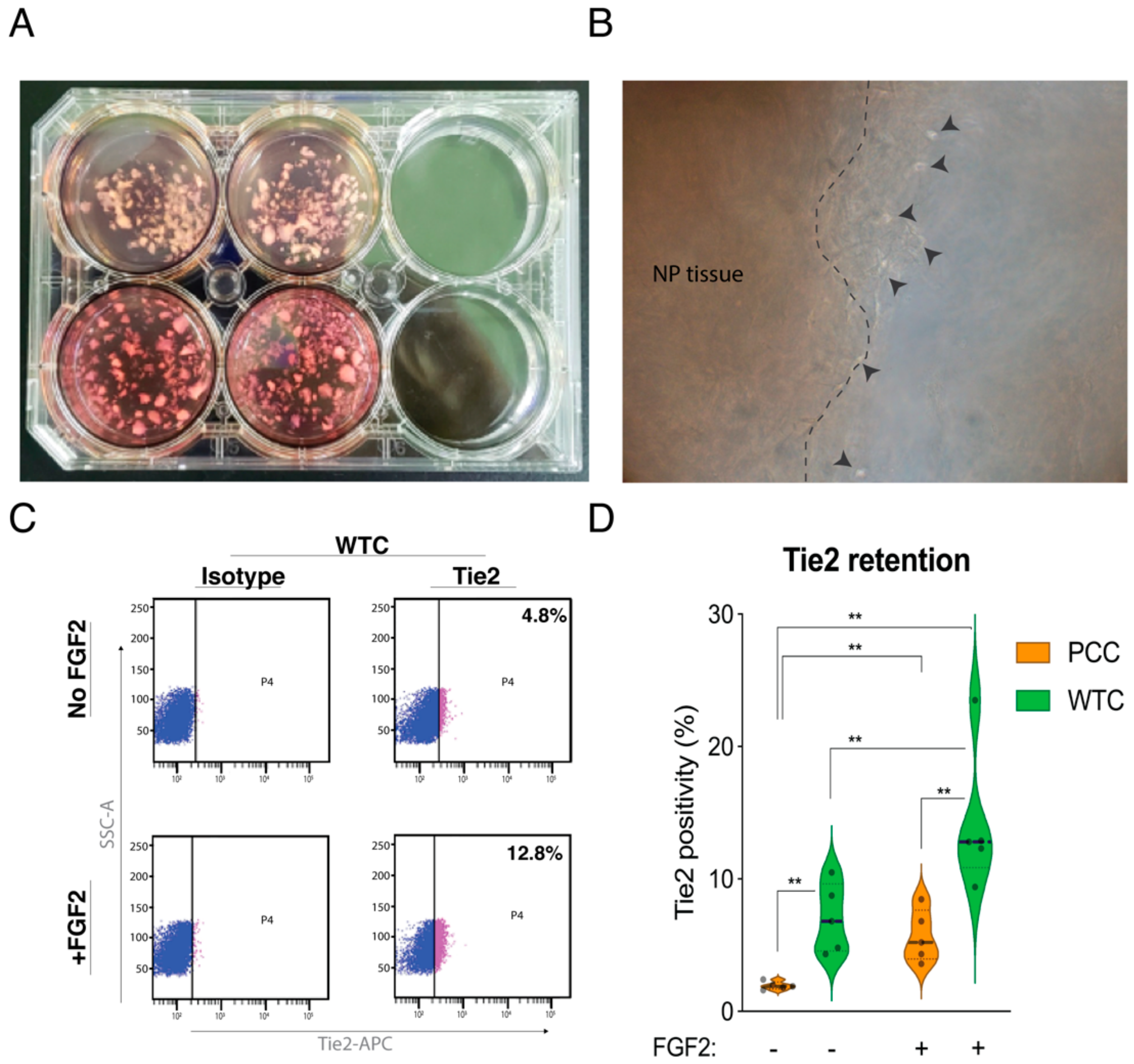
2.1. The Effect of WTC Method and FGF2 on Tie2 Expression Rates
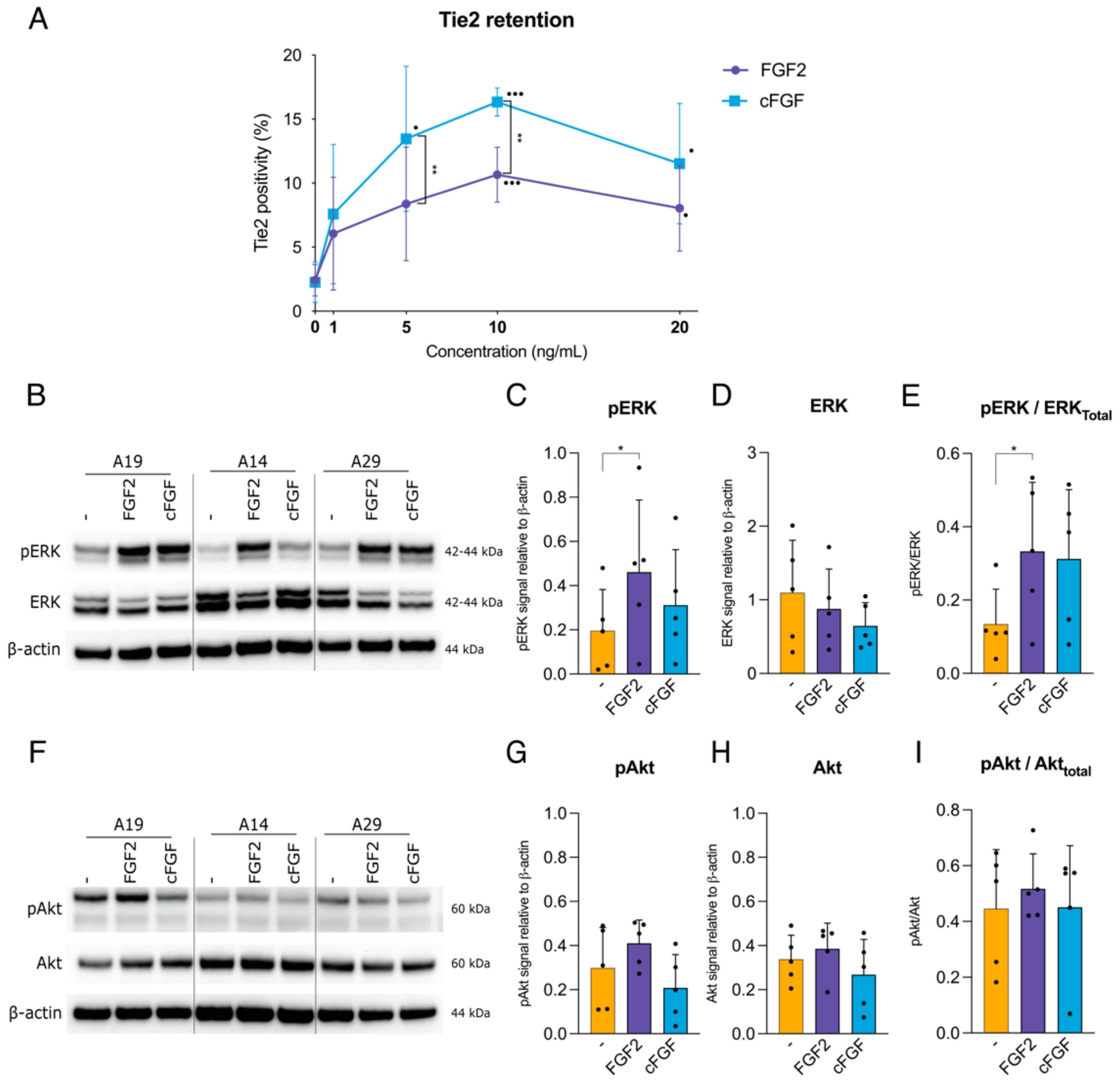
2.2. Tie2 Expression and Signal Transduction Pathway by cFGF
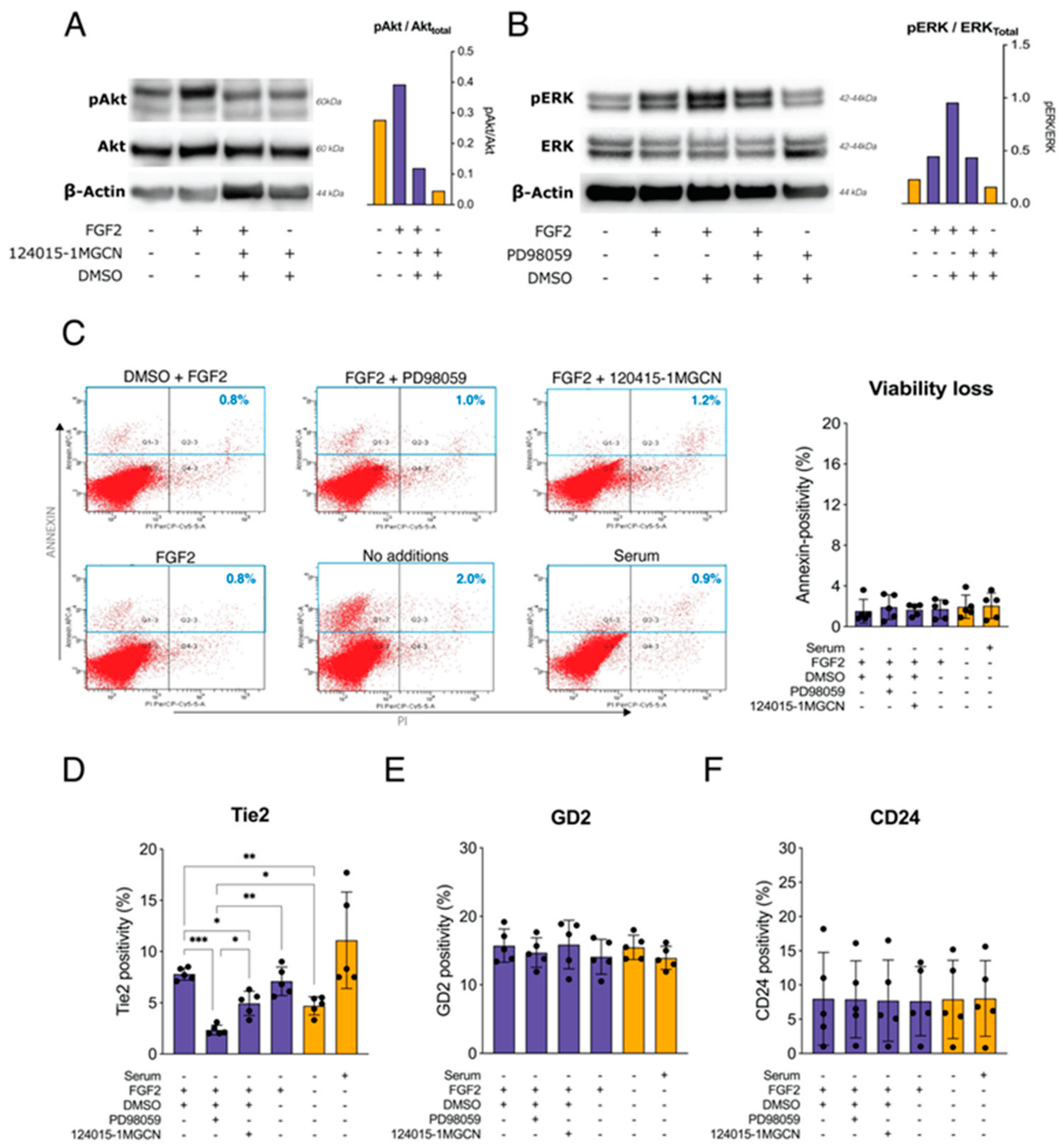
2.3. Effects of MEK, Akt Inhibitors on FGF2 Signaling and Tie2 Expression
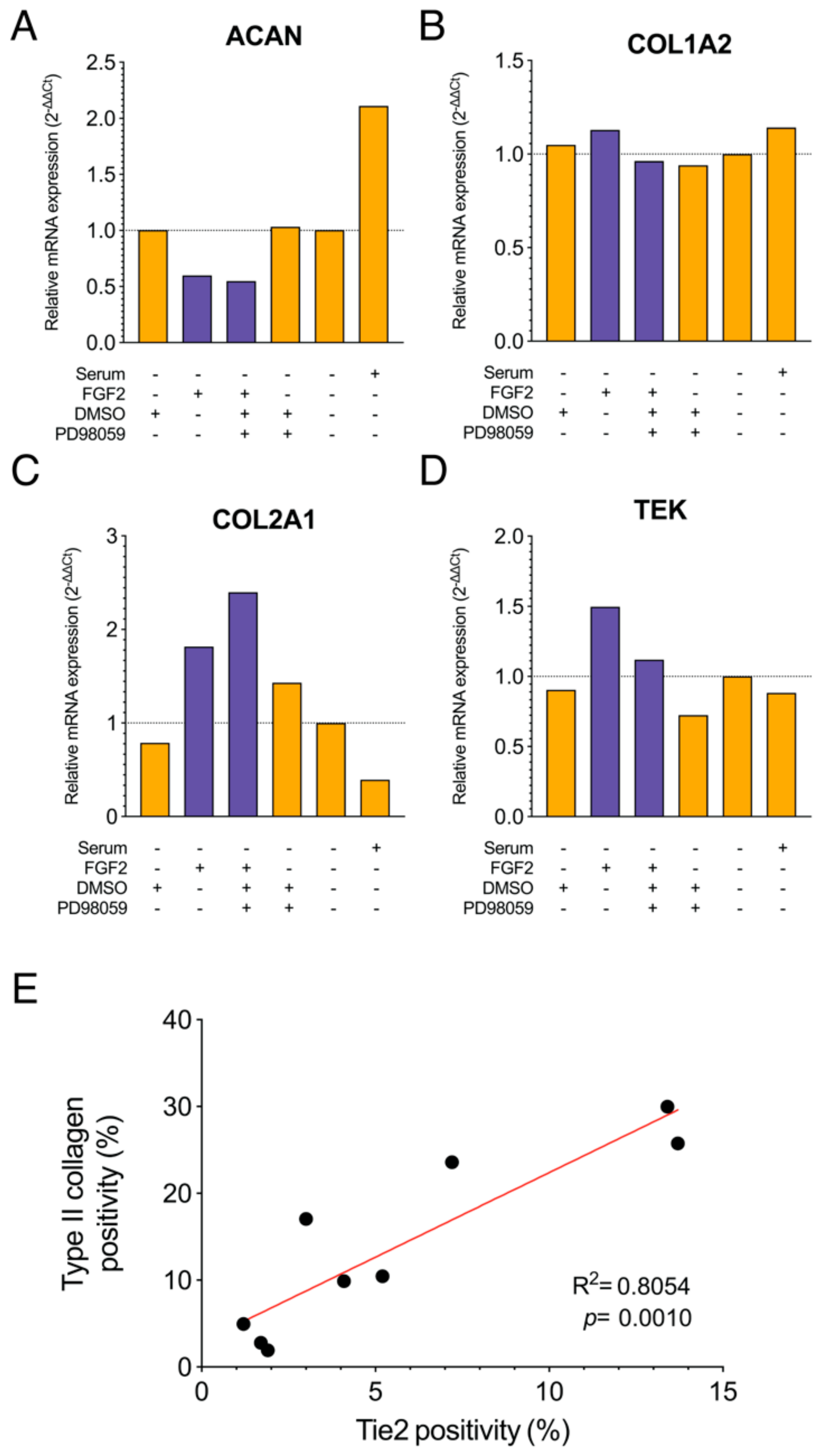
2.4. Correlation Between Tie2 Expression by FGF2 and COL2A1 Expression
3. Discussion
4. Materials and Methods
4.1. Human NP Cell Isolation
4.2. WTC Method
4.3. PCC Method
4.4. Assessment of FGF2 on Tie2 Expression in WTC and PCC
4.5. Role PI3K/Akt and MEK/ERK in FGF2 and cFGF Supported Tie2 Maintenance
4.6. Flow Cytometry and Fluorescence-Activated Cell Sorting
4.7. Western Blotting
4.8. Real-Time Polymerase Chain Reaction Analysis
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Strine, T.W.; Hootman, J.M. US national prevalence and correlates of low back and neck pain among adults. Arthritis Rheum. 2007, 57, 656–665. [Google Scholar] [CrossRef] [PubMed]
- Vos, T.; Allen, C.; Arora, M.; Barber, R.M.; Bhutta, Z.A.; Brown, A.; Carter, A.; Casey, D.C.; Charlson, F.J.; Chen, A.Z.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016, 388, 1545–1602. [Google Scholar] [CrossRef]
- Oichi, T.; Taniguchi, Y.; Oshima, Y.; Tanaka, S.; Saito, T. Pathomechanism of intervertebral disc degeneration. JOR Spine 2020, 3, e1076. [Google Scholar] [CrossRef] [PubMed]
- Frost, B.A.; Camarero-Espinosa, S.; Foster, E.J. Materials for the Spine: Anatomy, Problems, and Solutions. Materials 2019, 12, 253. [Google Scholar] [CrossRef]
- Kepler, C.K.; Ponnappan, R.K.; Tannoury, C.A.; Risbud, M.V.; Anderson, D.G. The molecular basis of intervertebral disc degeneration. Spine J. 2013, 13, 318–330. [Google Scholar] [CrossRef]
- Urban, J.P.; Roberts, S. Degeneration of the intervertebral disc. Arthritis Res. Ther. 2003, 5, 120–130. [Google Scholar] [CrossRef]
- Rustenburg, C.M.E.; Emanuel, K.S.; Peeters, M.; Lems, W.F.; Vergroesen, P.A.; Smit, T.H. Osteoarthritis and intervertebral disc degeneration: Quite different, quite similar. JOR Spine 2018, 1, e1033. [Google Scholar] [CrossRef]
- Krock, E.; Rosenzweig, D.H.; Chabot-Dore, A.J.; Jarzem, P.; Weber, M.H.; Ouellet, J.A.; Stone, L.S.; Haglund, L. Painful, degenerating intervertebral discs up-regulate neurite sprouting and CGRP through nociceptive factors. J. Cell Mol. Med. 2014, 18, 1213–1225. [Google Scholar] [CrossRef]
- Fournier, D.E.; Kiser, P.K.; Shoemaker, J.K.; Battie, M.C.; Seguin, C.A. Vascularization of the human intervertebral disc: A scoping review. JOR Spine 2020, 3, e1123. [Google Scholar] [CrossRef]
- Garcia-Cosamalon, J.; del Valle, M.E.; Calavia, M.G.; Garcia-Suarez, O.; Lopez-Muniz, A.; Otero, J.; Vega, J.A. Intervertebral disc, sensory nerves and neurotrophins: Who is who in discogenic pain? J. Anat. 2010, 217, 1–15. [Google Scholar] [CrossRef]
- Raj, P.P. Intervertebral disc: Anatomy-physiology-pathophysiology-treatment. Pain Pr. 2008, 8, 18–44. [Google Scholar] [CrossRef]
- Evans, C.H.; Huard, J. Gene therapy approaches to regenerating the musculoskeletal system. Nat. Rev. Rheumatol. 2015, 11, 234–242. [Google Scholar] [CrossRef]
- Hodgkinson, T.; Shen, B.; Diwan, A.; Hoyland, J.A.; Richardson, S.M. Therapeutic potential of growth differentiation factors in the treatment of degenerative disc diseases. JOR Spine 2019, 2, e1045. [Google Scholar] [CrossRef]
- Harmon, M.D.; Ramos, D.M.; Nithyadevi, D.; Bordett, R.; Rudraiah, S.; Nukavarapu, S.P.; Moss, I.L.; Kumbar, S.G. Growing a backbone—Functional biomaterials and structures for intervertebral disc (IVD) repair and regeneration: Challenges, innovations, and future directions. Biomater. Sci. 2020, 8, 1216–1239. [Google Scholar] [CrossRef]
- Buckley, C.T.; Hoyland, J.A.; Fujii, K.; Pandit, A.; Iatridis, J.C.; Grad, S. Critical aspects and challenges for intervertebral disc repair and regeneration-Harnessing advances in tissue engineering. JOR Spine 2018, 1, e1029. [Google Scholar] [CrossRef]
- Gullbrand, S.E.; Smith, L.J.; Smith, H.E.; Mauck, R.L. Promise, progress, and problems in whole disc tissue engineering. JOR Spine 2018, 1, e1015. [Google Scholar] [CrossRef]
- Schol, J.; Sakai, D. Cell therapy for intervertebral disc herniation and degenerative disc disease: Clinical trials. Int. Orthop. 2019, 43, 1011–1025. [Google Scholar] [CrossRef]
- Smith, L.J.; Silverman, L.; Sakai, D.; Le Maitre, C.L.; Mauck, R.L.; Malhotra, N.R.; Lotz, J.C.; Buckley, C.T. Advancing cell therapies for intervertebral disc regeneration from the lab to the clinic: Recommendations of the ORS spine section. JOR Spine 2018, 1, e1036. [Google Scholar] [CrossRef]
- Loibl, M.; Wuertz-Kozak, K.; Vadala, G.; Lang, S.; Fairbank, J.; Urban, J.P. Controversies in regenerative medicine: Should intervertebral disc degeneration be treated with mesenchymal stem cells? JOR Spine 2019, 2, e1043. [Google Scholar] [CrossRef]
- Vickers, L.; Thorpe, A.A.; Snuggs, J.; Sammon, C.; Le Maitre, C.L. Mesenchymal stem cell therapies for intervertebral disc degeneration: Consideration of the degenerate niche. JOR Spine 2019, 2, e1055. [Google Scholar] [CrossRef]
- Sakai, D.; Nakamura, Y.; Nakai, T.; Mishima, T.; Kato, S.; Grad, S.; Alini, M.; Risbud, M.V.; Chan, D.; Cheah, K.S.; et al. Exhaustion of nucleus pulposus progenitor cells with ageing and degeneration of the intervertebral disc. Nat. Commun. 2012, 3, 1264. [Google Scholar] [CrossRef]
- Frauchiger, D.A.; Tekari, A.; May, R.D.; Dzafo, E.; Chan, S.C.W.; Stoyanov, J.; Bertolo, A.; Zhang, X.; Guerrero, J.; Sakai, D.; et al. Fluorescence-Activated Cell Sorting Is More Potent to Fish Intervertebral Disk Progenitor Cells Than Magnetic and Beads-Based Methods. Tissue Eng. Part C Methods 2019, 25, 571–580. [Google Scholar] [CrossRef]
- Sakai, D.; Schol, J.; Bach, F.C.; Tekari, A.; Sagawa, N.; Nakamura, Y.; Chan, S.C.W.; Nakai, T.; Creemers, L.B.; Frauchiger, D.A.; et al. Successful fishing for nucleus pulposus progenitor cells of the intervertebral disc across species. JOR Spine 2018, 1, e1018. [Google Scholar] [CrossRef]
- Ishii, T.; Sakai, D.; Schol, J.; Nakai, T.; Suyama, K.; Watanabe, M. Sciatic nerve regeneration by transplantation of in vitro differentiated nucleus pulposus progenitor cells. Regen. Med. 2017, 12, 365–376. [Google Scholar] [CrossRef]
- Li, X.C.; Tang, Y.; Wu, J.H.; Yang, P.S.; Wang, D.L.; Ruan, D.K. Characteristics and potentials of stem cells derived from human degenerated nucleus pulposus: Potential for regeneration of the intervertebral disc. BMC Musculoskelet. Disord. 2017, 18, 242. [Google Scholar] [CrossRef]
- Guerrero, J.; Häckel, S.; Croft, A.S.; Albers, C.E.; Gantenbein, B. The effects of 3D culture on the expansion and maintenance of nucleus pulposus progenitor cell multipotency. JOR Spine 2020, 4, e1131. [Google Scholar] [CrossRef]
- Humphreys, M.D.; Ward, L.; Richardson, S.M.; Hoyland, J.A. An optimized culture system for notochordal cell expansion with retention of phenotype. JOR Spine 2018, 1, e1028. [Google Scholar] [CrossRef]
- Nukaga, T.; Sakai, D.; Schol, J.; Suyama, K.; Nakai, T.; Hiyama, A.; Watanabe, M. Minimal Sustainability of Dedifferentiation by ROCK Inhibitor on Rat Nucleus Pulposus Cells In Vitro. Spine Surg. Relat. Res. 2019, 3, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Tekari, A.; Chan, S.C.W.; Sakai, D.; Grad, S.; Gantenbein, B. Angiopoietin-1 receptor Tie2 distinguishes multipotent differentiation capability in bovine coccygeal nucleus pulposus cells. Stem Cell Res. Ther. 2016, 7, 75. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Guerrero, J.; Croft, A.S.; Albers, C.E.; Hackel, S.; Gantenbein, B. Spheroid-Like Cultures for Expanding Angiopoietin Receptor-1 (aka. Tie2) Positive Cells from the Human Intervertebral Disc. Int. J. Mol. Sci. 2020, 21, 9423. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Guerrero, J.; Croft, A.S.; Oswald, K.A.C.; Albers, C.E.; Häckel, S.; Gantenbein, B. Towards Tissue-Specific Stem Cell Therapy for the Intervertebral Disc: PPARδ Agonist Increases the Yield of Human Nucleus Pulposus Progenitor Cells in Expansion. Surgeries 2021, 2, 92–104. [Google Scholar] [CrossRef]
- Sako, K.; Sakai, D.; Nakamura, Y.; Matsushita, E.; Schol, J.; Warita, T.; Horikita, N.; Sato, M.; Watanabe, M. Optimization of Spheroid Colony Culture and Cryopreservation of Nucleus Pulposus Cells for the Development of Intervertebral Disc Regenerative Therapeutics. Appl. Sci. 2021, 11, 3309. [Google Scholar] [CrossRef]
- Fearing, B.V.; Hernandez, P.A.; Setton, L.A.; Chahine, N.O. Mechanotransduction and cell biomechanics of the intervertebral disc. JOR Spine 2018, 1. [Google Scholar] [CrossRef]
- Motomura, K.; Hagiwara, A.; Komi-Kuramochi, A.; Hanyu, Y.; Honda, E.; Suzuki, M.; Kimura, M.; Oki, J.; Asada, M.; Sakaguchi, N.; et al. An FGF1:FGF2 chimeric growth factor exhibits universal FGF receptor specificity, enhanced stability and augmented activity useful for epithelial proliferation and radioprotection. Biochim. Biophys. Acta 2008, 1780, 1432–1440. [Google Scholar] [CrossRef]
- Nakayama, F.; Hagiwara, A.; Umeda, S.; Asada, M.; Goto, M.; Oki, J.; Suzuki, M.; Imamura, T.; Akashi, M. Post treatment with an FGF chimeric growth factor enhances epithelial cell proliferation to improve recovery from radiation-induced intestinal damage. Int. J. Radiat. Oncol. Biol. Phys. 2010, 78, 860–867. [Google Scholar] [CrossRef]
- Onuma, Y.; Higuchi, K.; Aiki, Y.; Shu, Y.; Asada, M.; Asashima, M.; Suzuki, M.; Imamura, T.; Ito, Y. A stable chimeric fibroblast growth factor (FGF) can successfully replace basic FGF in human pluripotent stem cell culture. PLoS ONE 2015, 10, e0118931. [Google Scholar] [CrossRef]
- Pratsinis, H.; Kletsas, D. PDGF, bFGF and IGF-I stimulate the proliferation of intervertebral disc cells in vitro via the activation of the ERK and Akt signaling pathways. Eur. Spine J. 2007, 16, 1858–1866. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Z.; Chen, P.; Ma, C.Y.; Li, C.; Au, T.Y.K.; Tam, V.; Peng, Y.; Wu, R.; Cheung, K.M.C.; et al. Directed Differentiation of Notochord-like and Nucleus Pulposus-like Cells Using Human Pluripotent Stem Cells. Cell Rep. 2020, 30, 2791–2806.e5. [Google Scholar] [CrossRef]
- Seguin, C.A.; Chan, D.; Dahia, C.L.; Gazit, Z. Latest advances in intervertebral disc development and progenitor cells. JOR Spine 2018, 1, e1030. [Google Scholar] [CrossRef]
- Hiraishi, S.; Schol, J.; Sakai, D.; Nukaga, T.; Erickson, I.; Silverman, L.; Foley, K.; Watanabe, M. Discogenic cell transplantation directly from a cryopreserved state in an induced intervertebral disc degeneration canine model. JOR Spine 2018, 1, e1013. [Google Scholar] [CrossRef]
- US National Library of Medicine—ClinicalTrials.gov 2019. Available online: https://clinicaltrials.gov/ct2/show/NCT03955315 (accessed on 20 March 2021).
- US National Library of Medicine—ClinicalTrials.gov 2017. Available online: https://clinicaltrials.gov/ct2/show/NCT03347708 (accessed on 20 March 2021).
- Thorpe, A.A.; Bach, F.C.; Tryfonidou, M.A.; Le Maitre, C.L.; Mwale, F.; Diwan, A.D.; Ito, K. Leaping the hurdles in developing regenerative treatments for the intervertebral disc from preclinical to clinical. JOR Spine 2018, 1, e1027. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Fukuhara, S.; Sako, K.; Takenouchi, T.; Kitani, H.; Kume, T.; Koh, G.Y.; Mochizuki, N. Angiopoietin-1/Tie2 signal augments basal Notch signal controlling vascular quiescence by inducing delta-like 4 expression through AKT-mediated activation of beta-catenin. J. Biol. Chem. 2011, 286, 8055–8066. [Google Scholar] [CrossRef] [PubMed]
- Augustin, H.G.; Koh, G.Y.; Thurston, G.; Alitalo, K. Control of vascular morphogenesis and homeostasis through the angiopoietin-Tie system. Nat. Rev. Mol. Cell Biol. 2009, 10, 165–177. [Google Scholar] [CrossRef] [PubMed]
- Chislock, E.M.; Ring, C.; Pendergast, A.M. Abl kinases are required for vascular function, Tie2 expression, and angiopoietin-1-mediated survival. Proc. Natl. Acad. Sci. USA 2013, 110, 12432–12437. [Google Scholar] [CrossRef]
- Brazil, D.P.; Hemmings, B.A. Ten years of protein kinase B signalling: A hard Akt to follow. Trends Biochem. Sci. 2001, 26, 657–664. [Google Scholar] [CrossRef]
- Wang, K.; Kang, L.; Liu, W.; Song, Y.; Wu, X.; Zhang, Y.; Hua, W.; Zhao, K.; Li, S.; Tu, J.; et al. Angiopoietin-2 promotes extracellular matrix degradation in human degenerative nucleus pulposus cells. Int. J. Mol. Med. 2018, 41, 3551–3558. [Google Scholar] [CrossRef]
- Wang, K.; Liu, W.; Song, Y.; Wu, X.; Zhang, Y.; Li, S.; Gao, Y.; Tu, J.; Liu, Y.; Yang, C. The role of angiopoietin-2 in nucleus pulposus cells during human intervertebral disc degeneration. Lab. Invest. 2017, 97, 971–982. [Google Scholar] [CrossRef]
- Risbud, M.V.; Fertala, J.; Vresilovic, E.J.; Albert, T.J.; Shapiro, I.M. Nucleus pulposus cells upregulate PI3K/Akt and MEK/ERK signaling pathways under hypoxic conditions and resist apoptosis induced by serum withdrawal. Spine 2005, 30, 882–889. [Google Scholar] [CrossRef]
- Pratsinis, H.; Constantinou, V.; Pavlakis, K.; Sapkas, G.; Kletsas, D. Exogenous and autocrine growth factors stimulate human intervertebral disc cell proliferation via the ERK and Akt pathways. J. Orthop. Res. 2012, 30, 958–964. [Google Scholar] [CrossRef]
- Pratsinis, H.; Kletsas, D. Organotypic Cultures of Intervertebral Disc Cells: Responses to Growth Factors and Signaling Pathways Involved. BioMed Res. Int. 2015, 2015, 427138. [Google Scholar] [CrossRef]
- Eswarakumar, V.P.; Lax, I.; Schlessinger, J. Cellular signaling by fibroblast growth factor receptors. Cytokine Growth Factor Rev. 2005, 16, 139–149. [Google Scholar] [CrossRef]
- Risbud, M.V.; Schoepflin, Z.R.; Mwale, F.; Kandel, R.A.; Grad, S.; Iatridis, J.C.; Sakai, D.; Hoyland, J.A. Defining the phenotype of young healthy nucleus pulposus cells: Recommendations of the Spine Research Interest Group at the 2014 annual ORS meeting. J. Orthop. Res. 2015, 33, 283–293. [Google Scholar] [CrossRef]
| Code | Age (Year) | Sex | Pathology | IVD Tissue (g) | NP Tissue (g) |
|---|---|---|---|---|---|
| E32 | 32 | F | LDH | 4.75 | 4.75 |
| A24 | 24 | M | LDH | 3.65 | 3.30 |
| A20 | 20 | M | LDH | 6.98 | 3.23 |
| T20 | 20 | M | LDH | 1.89 | 1.25 |
| T21 | 21 | F | LDH | 2.82 | 1.81 |
| A25 | 25 | F | LDH | 2.07 | 1.92 |
| T19 | 19 | M | LDH | 8.05 | 7.59 |
| A18 | 18 | M | LDH | 5.88 | 3.73 |
| A14 | 14 | F | LDH | 1.74 | 1.54 |
| A18-2 | 18 | M | LDH | 1.74 | 1.42 |
| A29 | 29 | M | LDH | 2.68 | 1.77 |
| T18 | 18 | M | LDH | 3.07 | 2.70 |
| A18-3 | 18 | M | LDH | 2.16 | 1.26 |
| A21 | 21 | M | LDH | 1.75 | 1.36 |
| A20-2 | 20 | F | LDH | 1.06 | 0.98 |
| A16 | 16 | M | LDH | 1.81 | 1.50 |
| T16 | 16 | M | LDH | 2.03 | 2.03 |
| K30F | 30 | M | LDH | 3.18 | 1.03 |
| K30M | 30 | F | LDH | 4.26 | 2.5 |
| K21 | 21 | M | LDH | 1.79 | 0.85 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sako, K.; Sakai, D.; Nakamura, Y.; Schol, J.; Matsushita, E.; Warita, T.; Horikita, N.; Sato, M.; Watanabe, M. Effect of Whole Tissue Culture and Basic Fibroblast Growth Factor on Maintenance of Tie2 Molecule Expression in Human Nucleus Pulposus Cells. Int. J. Mol. Sci. 2021, 22, 4723. https://doi.org/10.3390/ijms22094723
Sako K, Sakai D, Nakamura Y, Schol J, Matsushita E, Warita T, Horikita N, Sato M, Watanabe M. Effect of Whole Tissue Culture and Basic Fibroblast Growth Factor on Maintenance of Tie2 Molecule Expression in Human Nucleus Pulposus Cells. International Journal of Molecular Sciences. 2021; 22(9):4723. https://doi.org/10.3390/ijms22094723
Chicago/Turabian StyleSako, Kosuke, Daisuke Sakai, Yoshihiko Nakamura, Jordy Schol, Erika Matsushita, Takayuki Warita, Natsumi Horikita, Masato Sato, and Masahiko Watanabe. 2021. "Effect of Whole Tissue Culture and Basic Fibroblast Growth Factor on Maintenance of Tie2 Molecule Expression in Human Nucleus Pulposus Cells" International Journal of Molecular Sciences 22, no. 9: 4723. https://doi.org/10.3390/ijms22094723
APA StyleSako, K., Sakai, D., Nakamura, Y., Schol, J., Matsushita, E., Warita, T., Horikita, N., Sato, M., & Watanabe, M. (2021). Effect of Whole Tissue Culture and Basic Fibroblast Growth Factor on Maintenance of Tie2 Molecule Expression in Human Nucleus Pulposus Cells. International Journal of Molecular Sciences, 22(9), 4723. https://doi.org/10.3390/ijms22094723







