The Antithrombotic Agent Pterostilbene Interferes with Integrin αIIbβ3-Mediated Inside-Out and Outside-In Signals in Human Platelets
Abstract
1. Introduction
2. Results
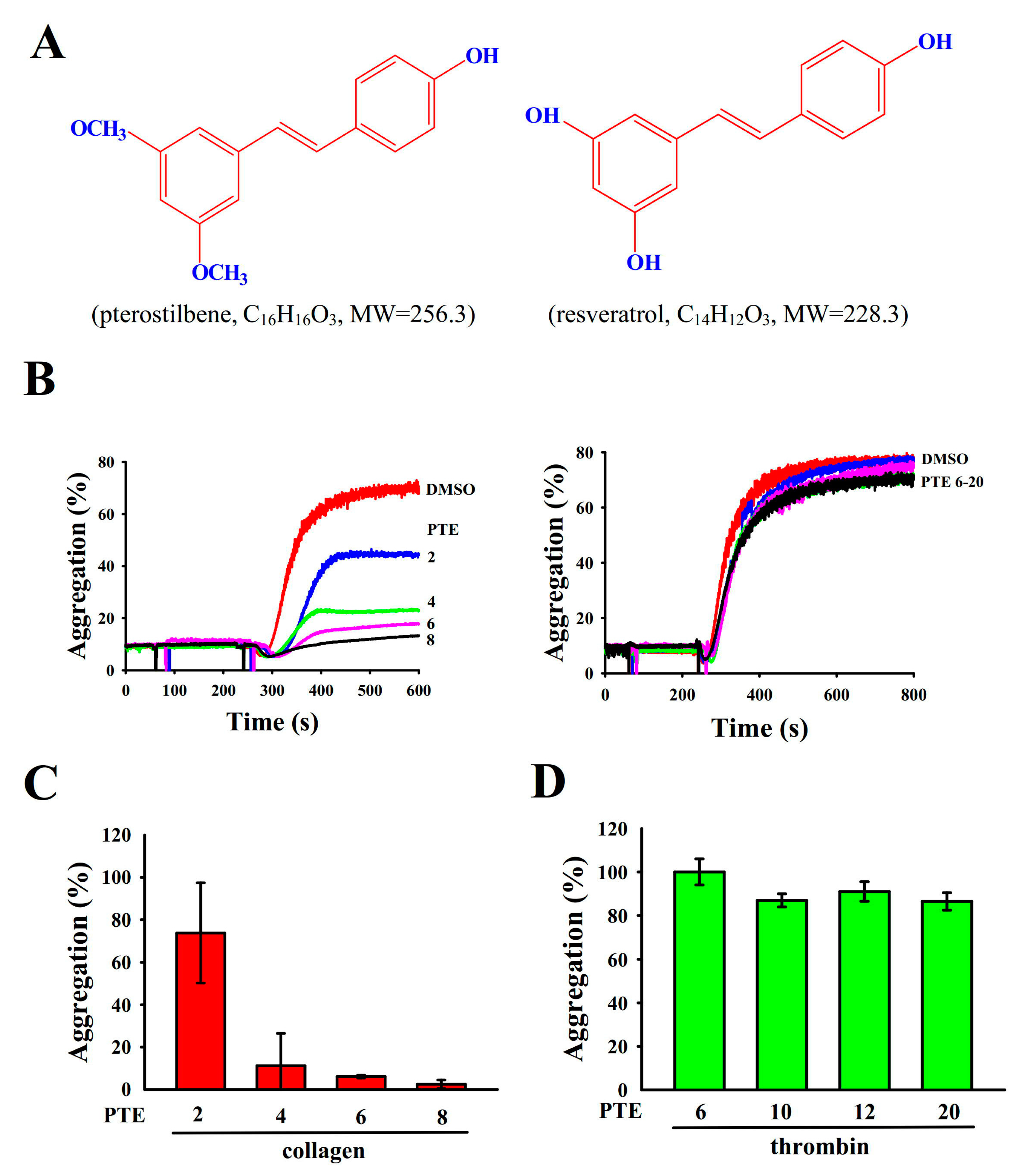
2.1. Inhibitory Profiles of PTE in Platelet Aggregation Stimulated with Collagen and Thrombin in Humans
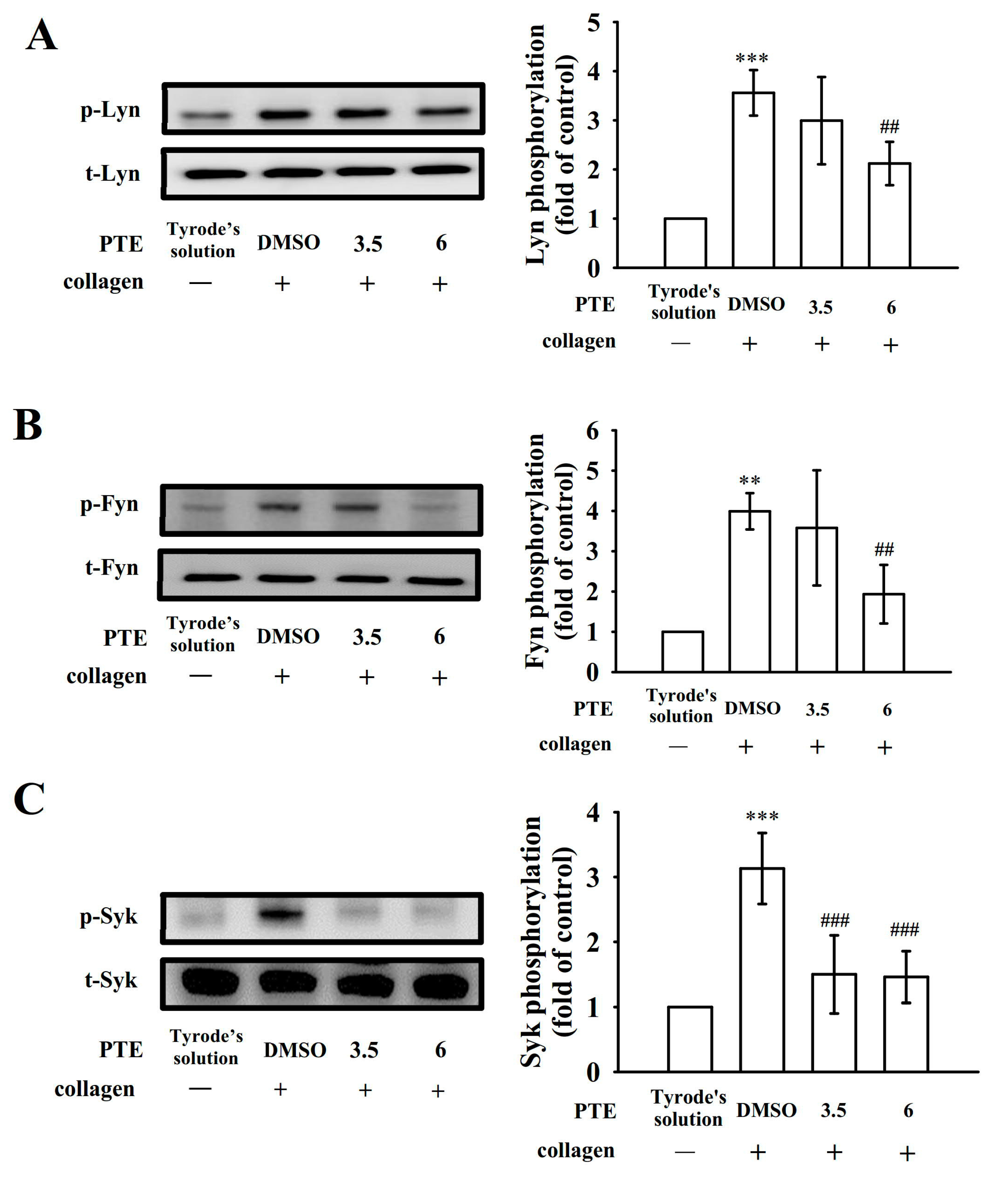
2.2. Effects of PTE on Glycoprotein VI-Mediated Lyn, Fyn, and Syk Phosphorylation
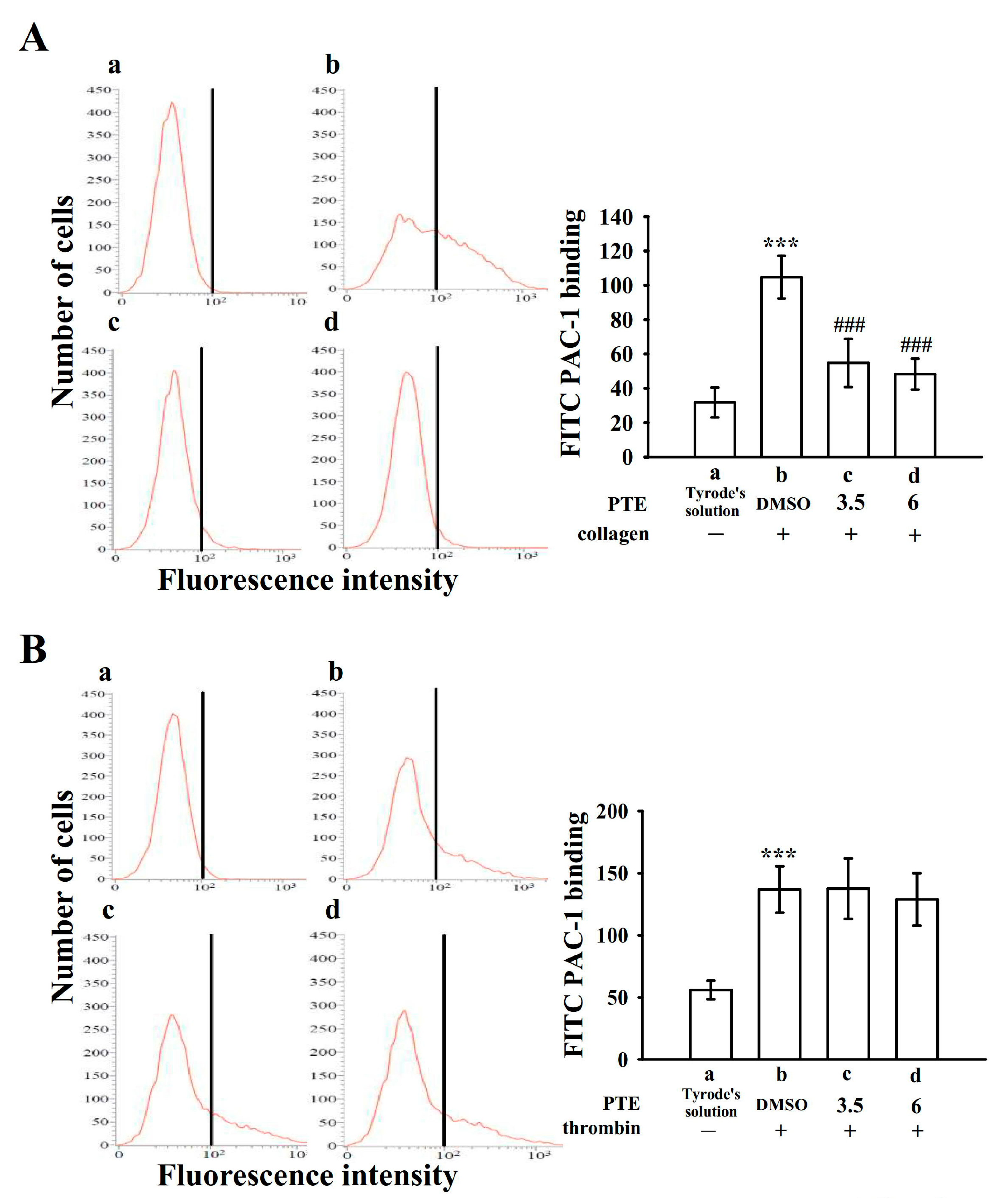
2.3. Effect of PTE on Integrin αIIbβ3 Activation
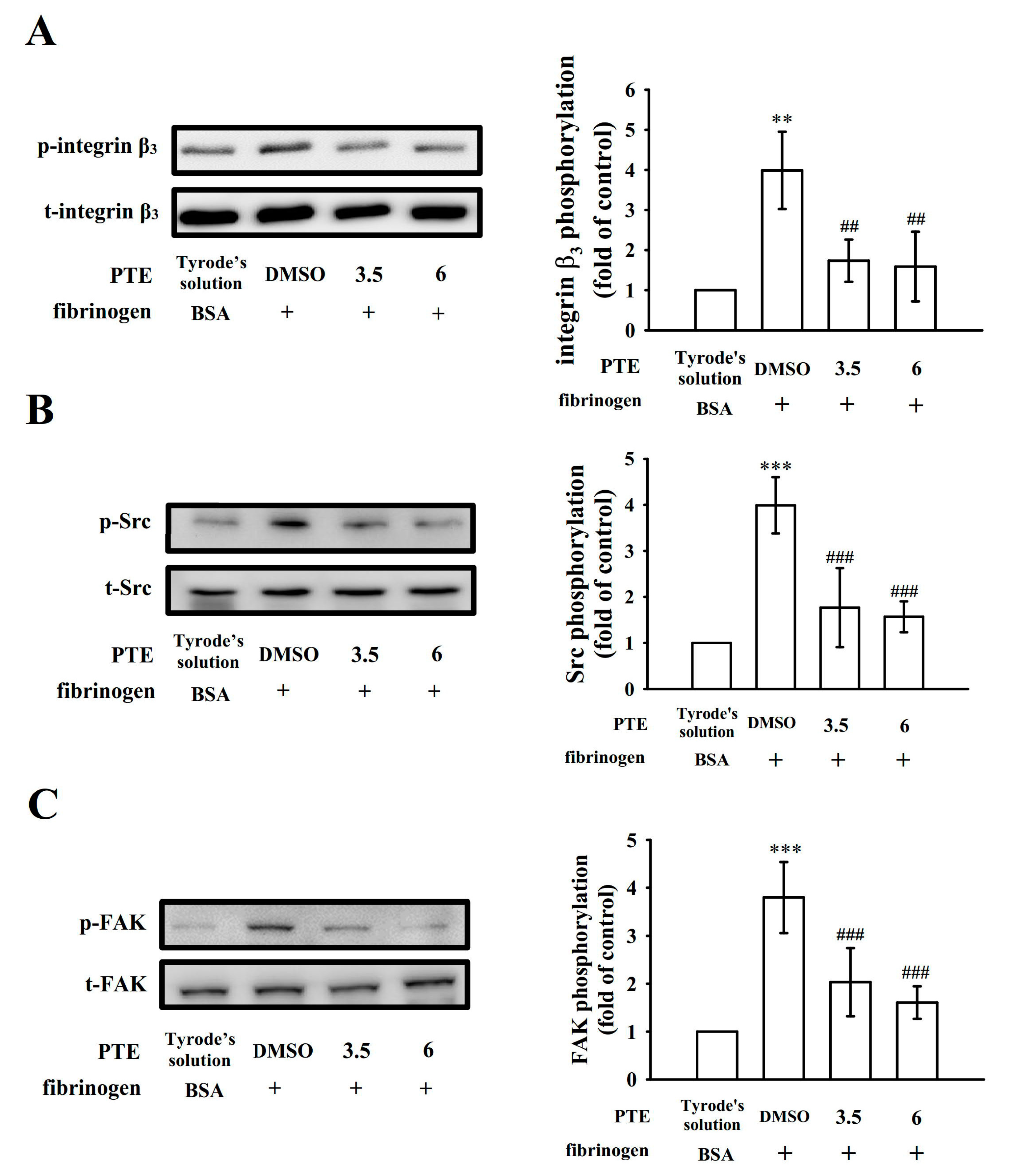
2.4. PTE Restricts Integrin αIIbβ3-Mediated Outside-In Signaling of Protein Kinase Activation
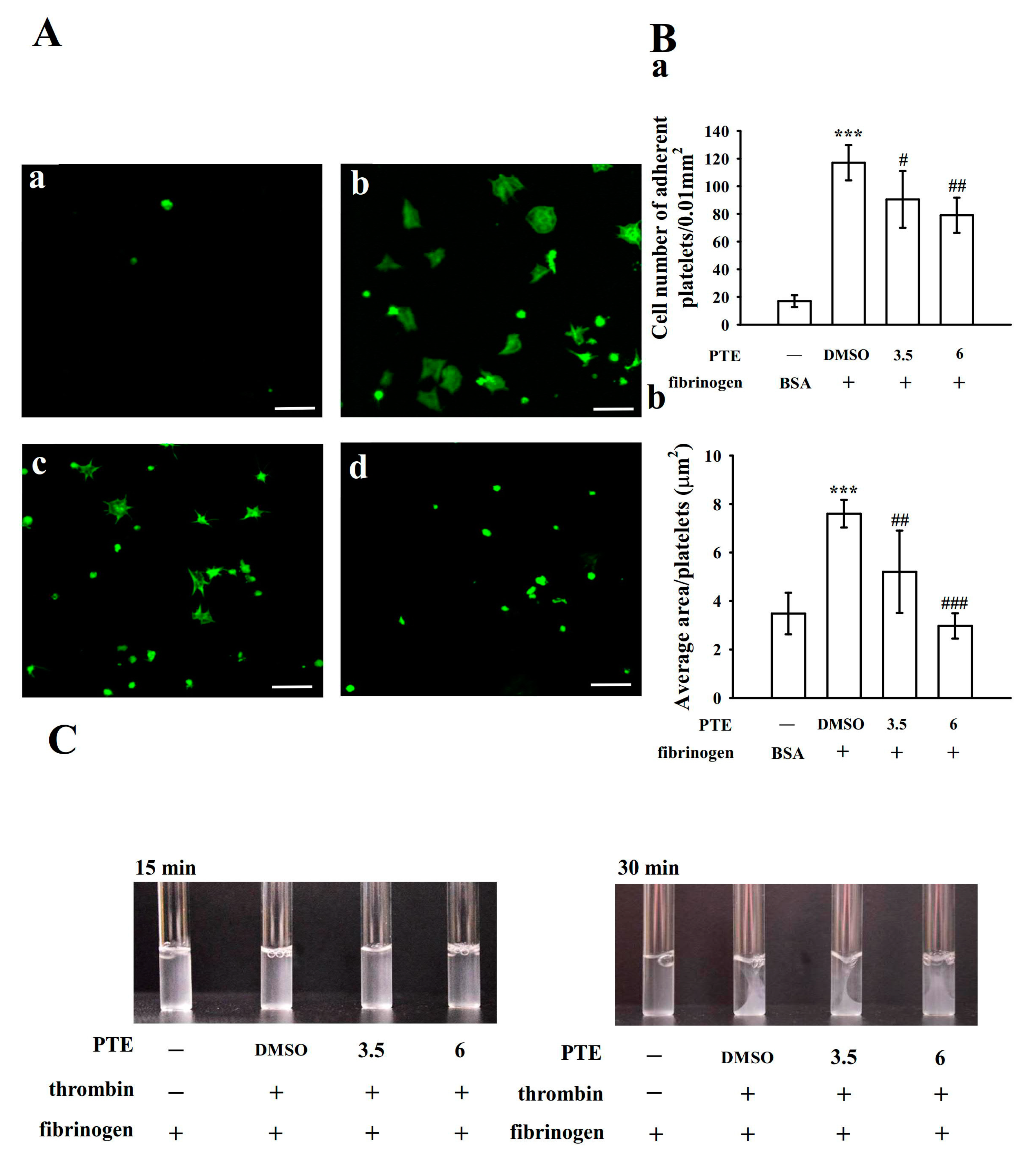
2.5. PTE Limits Integrin αIIbβ3-Mediated Outside-In Signaling of Platelet Adhesion and Spreading As Well As Fibrin Clot Retraction
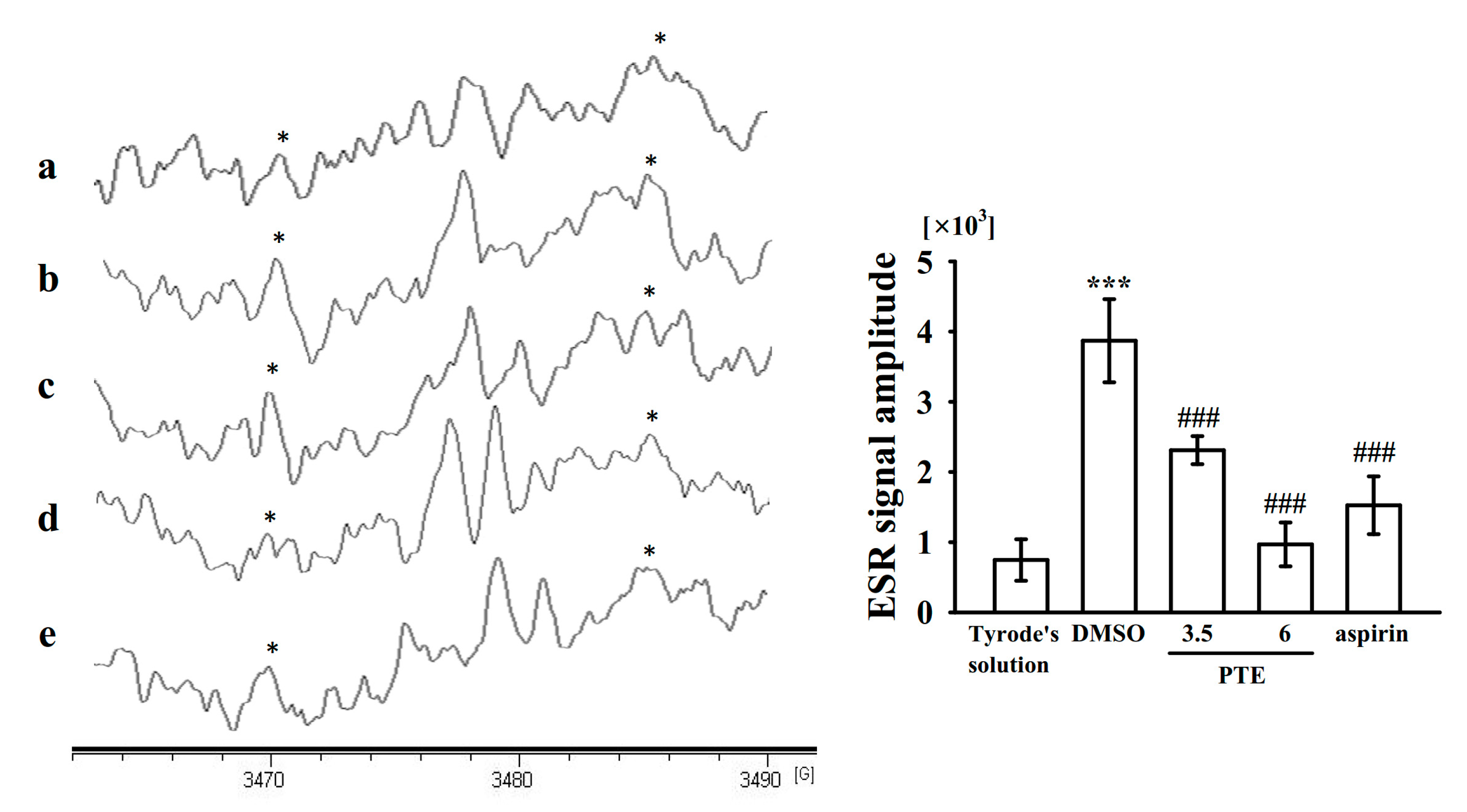
2.6. Regulatory Activities of PTE in Hydroxyl Radical Formation
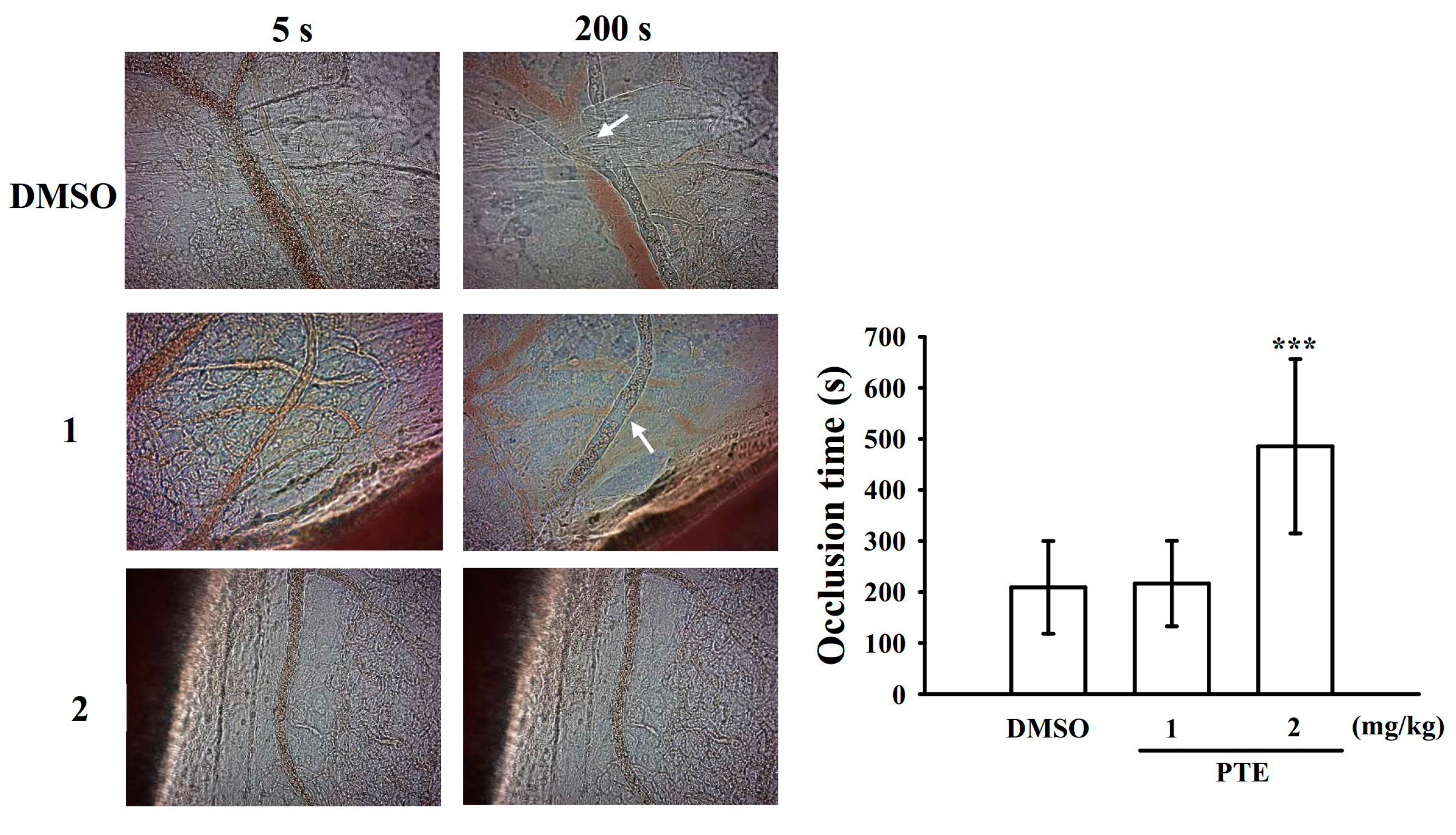
2.7. Effect of PTE in Vascular Thrombus Formation In Vivo
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Platelet Preparation and Aggregation Study
4.3. Study of Binding Activated Integrin αIIbβ3
4.4. Immunoblotting
4.5. Confocal Microscopy Analysis of Platelet Adhesion and Spreading
4.6. Platelet-Mediated Fibrin Clot Retraction
4.7. Measurement of Hydroxyl Radicals Through Electron Spin Resonance Spectrometry
4.8. Measurement of Vascular Thrombus Formation in Mouse Mesenteric Microvessels Irradiated with Sodium Fluorescein
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jennings, L.K. Mechanisms of platelet activation: Need for new strategies to protect against platelet-mediated atherothrombosis. Thromb. Haemost. 2009, 102, 248–257. [Google Scholar] [CrossRef]
- Payrastre, B.; Missy, K.; Trumel, C.; Bodin, S.; Plantavid, M.; Chap, H. The integrin alpha IIb/beta 3 in human platelet signal transduction. Biochem. Pharmacol. 2000, 60, 1069–1074. [Google Scholar] [CrossRef]
- Dvorakova, M.; Landa, P. Anti-inflammatory activity of natural stilbenoids: A review. Pharmacol. Res. 2017, 124, 126–145. [Google Scholar] [CrossRef]
- Zhu, Q.; Tang, T.; Liu, H.; Sun, Y.; Wang, X.; Liu, Q.; Yang, L.; Lei, Z.; Huang, Z.; Chen, Z.; et al. Pterostilbene attenuates cocultured BV-2 microglial inflammation-mediated SH-SY5Y neuronal oxidative injury via SIRT-1 signaling. Oxid. Med. Cell. Longev. 2020. [Google Scholar] [CrossRef]
- Park, E.-S.; Lim, Y.; Hong, J.-T.; Yoo, H.-S.; Lee, C.-K.; Pyo, M.-Y.; Yun, Y.-P. Pterostilbene, a natural dimethylated analog of resveratrol, inhibits rat aortic vascular smooth muscle cell proliferation by blocking Akt-dependent pathway. Vascular Pharmacol. 2010, 53, 61–67. [Google Scholar] [CrossRef]
- Xiong, X.; Lu, W.; Zhang, K.; Zhou, W. Pterostilbene reduces endothelial cell apoptosis by regulation of the Nrf2 mediated TLR 4/MyD88/NF κB pathway in a rat model of atherosclerosis. Exp. Ther. Med. 2020, 20, 2090–2098. [Google Scholar]
- Messina, F.; Guglielmini, G.; Curini, M.; Orsini, S.; Gresele, P.; Marcotullio, M. Effect of substituted stilbenes on platelet function. Fitoterapia 2015, 105, 228–233. [Google Scholar] [CrossRef]
- Shen, M.Y.; Hsiao, G.; Liu, C.L.; Fong, T.H.; Lin, K.H.; Chou, D.S.; Sheu, J.R. Inhibitory mechanisms of resveratrol in platelet activation: Pivotal roles of p38 MAPK and NO/cyclic GMP. Br. J. Haematol. 2007, 139, 475–485. [Google Scholar] [CrossRef]
- Varga-Szabo, D.; Braun, A.; Nieswandt, B. Calcium signaling in platelets. J. Thromb. Haemost. 2009, 7, 1057–1066. [Google Scholar] [CrossRef]
- Cosemans, J.M.; Iserbyt, B.F.; Deckmyn, H.; Heemskerk, J.W. Multiple ways to switch platelet integrins on and off. J. Thromb. Haemost. 2008, 6, 1253–1261. [Google Scholar] [CrossRef] [PubMed]
- Shattil, S.J. The beta3 integrin cytoplasmic tail: Protein scaffold and control freak. J. Thromb. Haemost. 2009, 7, 210–213. [Google Scholar] [CrossRef] [PubMed]
- Stoclet, J.-C.; Chataigneau, T.; Ndiaye, M.; Oak, M.-H.; El Bedoui, J.; Chataigneau, M.; Schini-Kerth, V.B. Vascular protection by dietary polyphenols. Eur. J. Pharmacol. 2004, 500, 299–313. [Google Scholar] [CrossRef]
- Fulda, S.; Debatin, K.M. Resveratrol modulation of signal transduction in apoptosis and cell survival: A mini-review. Cancer Detect. Prev. 2006, 30, 217–223. [Google Scholar] [CrossRef]
- Asensi, M.; Medina, I.; Ortega, A.; Carretero, J.; Baño, M.; Obrador, E.; Estrela, J.M. Inhibition of cancer growth by resveratrol is related to its low bioavailability. Free Radic. Biol. Med. 2002, 33, 387–398. [Google Scholar] [CrossRef]
- Wen, X.; Walle, T. Methylated flavonoids have greatly improved intestinal absorption and metabolic stability. Drug Metab. Dispos. 2006, 34, 1786–1792. [Google Scholar] [CrossRef]
- Lin, H.S.; Yue, B.D.; Ho, P.C. Determination of pterostilbene in rat plasma by a simple HPLC-UV method and its application in pre-clinical pharmacokinetic study. Biomed Chromatogr. 2009, 23, 1308–1315. [Google Scholar] [CrossRef] [PubMed]
- Kapetanovic, I.M.; Muzzio, M.; Huang, Z.; Thompson, T.N.; McCormick, D.L. Pharmacokinetics, oral bioavailability, and metabolic profile of resveratrol and its dimethylether analog, pterostilbene, in rats. Cancer Chemother. Pharmacol. 2011, 68, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.F.; Lee, J.J.; Chang, C.C.; Lin, K.H.; Wang, S.H.; Sheu, J.R. Platelet protease-activated receptor (PAR)4, but not PAR1, associated with neutral sphingomyelinase responsible for thrombin-stimulated ceramide-NF-κB signaling in human platelets. Haematologica 2013, 98, 793–801. [Google Scholar] [CrossRef] [PubMed]
- Jackson, S.P.; Nesbitt, W.S.; Kulkarni, S. Signaling events underlying thrombus formation. J. Thromb. Haemost. 2003, 1, 1602–1612. [Google Scholar] [CrossRef]
- Rubenstein, D.A.; Yin, W. Platelet-Activation Mechanisms and Vascular Remodeling. Compr. Physiol. 2018, 8, 1117–1156. [Google Scholar]
- Niedergang, F.; Alcover, A.; Knight, C.G.; Farndale, R.W.; Barnes, M.J.; Francischetti, I.M.; Bon, C.; Leduc, M. Convulxin binding to platelet receptor GPVI: Competition with collagen related peptides. Biochem. Biophys. Res. Commun. 2000, 273, 246–250. [Google Scholar] [CrossRef]
- Estevez, B.; Du, X. New concepts and mechanisms of platelet activation signaling. Physiology (Bethesda) 2017, 32, 162–177. [Google Scholar] [CrossRef] [PubMed]
- Shattil, S.J.; Cunningham, M.; Hoxie, J.A. Detection of activated platelets in whole blood using activation-dependent monoclonal antibodies and flow cytometry. Blood 1987, 70, 307–315. [Google Scholar] [CrossRef]
- Law, D.A.; DeGuzman, F.R.; Heiser, P.; Ministri-Madrid, K.; Killeen, N.; Phillips, D.R. Integrin cytoplasmic tyrosine motif is required for outside-in alphaIIbbeta3signaling and platelet function. Nature 1999, 401, 808–811. [Google Scholar] [CrossRef]
- Schaller, M.D. Cellular functions of FAK kinases: Insight into molecular mechanisms and novel functions. J. Cell. Sci. 2010, 123, 1007–1013. [Google Scholar] [CrossRef]
- Qiao, J.; Arthur, J.F.; Gardiner, E.E.; Andrews, R.K.; Zeng, L.; Xu, K. Regulation of platelet activation and thrombus formation by reactive oxygen species. Redox Biol. 2018, 14, 126–130. [Google Scholar] [CrossRef]
- Begonja, A.J.; Gambaryan, S.; Geiger, J.; Aktas, B.; Pozgajova, M.; Nieswandt, B.; Walter, U. Platelet NAD(P)H-oxidase-generated ROS production regulates αIIbβ3-integrin activation independent of the NO/cGMP pathway. Blood 2005, 106, 2757–2760. [Google Scholar] [CrossRef]
- Hsiao, G.; Lin, K.H.; Chang, Y.; Chen, T.L.; Tzu, N.H.; Chou, D.S.; Sheu, J.R. Protective mechanisms of inosine in platelet activation and cerebral ischemic damage. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 1998–2004. [Google Scholar] [CrossRef]
- Sheu, J.R.; Hung, W.C.; Wu, C.H.; Ma, M.C.; Kan, Y.C.; Lin, C.H.; Lin, M.-S.; Luk, H.-N.; Yen, M.-H. Reduction in lipopolysaccharide-induced thrombocytopenia by triflavin in a rat model of septicemia. Circulation 1999, 99, 3056–3062. [Google Scholar] [CrossRef]
- Osdoit, S.; Rosa, J.P. Fibrin clot retraction by human platelets correlates with alpha(IIb)beta(3) integrin-dependent protein tyrosine dephosphorylation. J. Biol. Chem. 2001, 276, 6703–6710. [Google Scholar] [CrossRef]
- Chou, D.S.; Hsiao, G.; Shen, M.Y.; Tsai, Y.J.; Chen, T.F.; Sheu, J.R. ESR spin trapping of a carbon-centered free radical from agonist-stimulated human platelets. Free Radic. Biol. Med. 2005, 39, 237–248. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, W.-C.; Lin, K.-C.; Hsia, C.-W.; Hsia, C.-H.; Chen, T.-Y.; Bhavan, P.S.; Sheu, J.-R.; Hou, S.-M. The Antithrombotic Agent Pterostilbene Interferes with Integrin αIIbβ3-Mediated Inside-Out and Outside-In Signals in Human Platelets. Int. J. Mol. Sci. 2021, 22, 3643. https://doi.org/10.3390/ijms22073643
Huang W-C, Lin K-C, Hsia C-W, Hsia C-H, Chen T-Y, Bhavan PS, Sheu J-R, Hou S-M. The Antithrombotic Agent Pterostilbene Interferes with Integrin αIIbβ3-Mediated Inside-Out and Outside-In Signals in Human Platelets. International Journal of Molecular Sciences. 2021; 22(7):3643. https://doi.org/10.3390/ijms22073643
Chicago/Turabian StyleHuang, Wei-Chieh, Kao-Chang Lin, Chih-Wei Hsia, Chih-Hsuan Hsia, Ting-Yu Chen, Periyakali Saravana Bhavan, Joen-Rong Sheu, and Shaw-Min Hou. 2021. "The Antithrombotic Agent Pterostilbene Interferes with Integrin αIIbβ3-Mediated Inside-Out and Outside-In Signals in Human Platelets" International Journal of Molecular Sciences 22, no. 7: 3643. https://doi.org/10.3390/ijms22073643
APA StyleHuang, W.-C., Lin, K.-C., Hsia, C.-W., Hsia, C.-H., Chen, T.-Y., Bhavan, P. S., Sheu, J.-R., & Hou, S.-M. (2021). The Antithrombotic Agent Pterostilbene Interferes with Integrin αIIbβ3-Mediated Inside-Out and Outside-In Signals in Human Platelets. International Journal of Molecular Sciences, 22(7), 3643. https://doi.org/10.3390/ijms22073643






