Genetic Background and Kinetics Define Wound Bed Extracellular Vesicles in a Mouse Model of Cutaneous Injury
Abstract
1. Introduction
2. Results
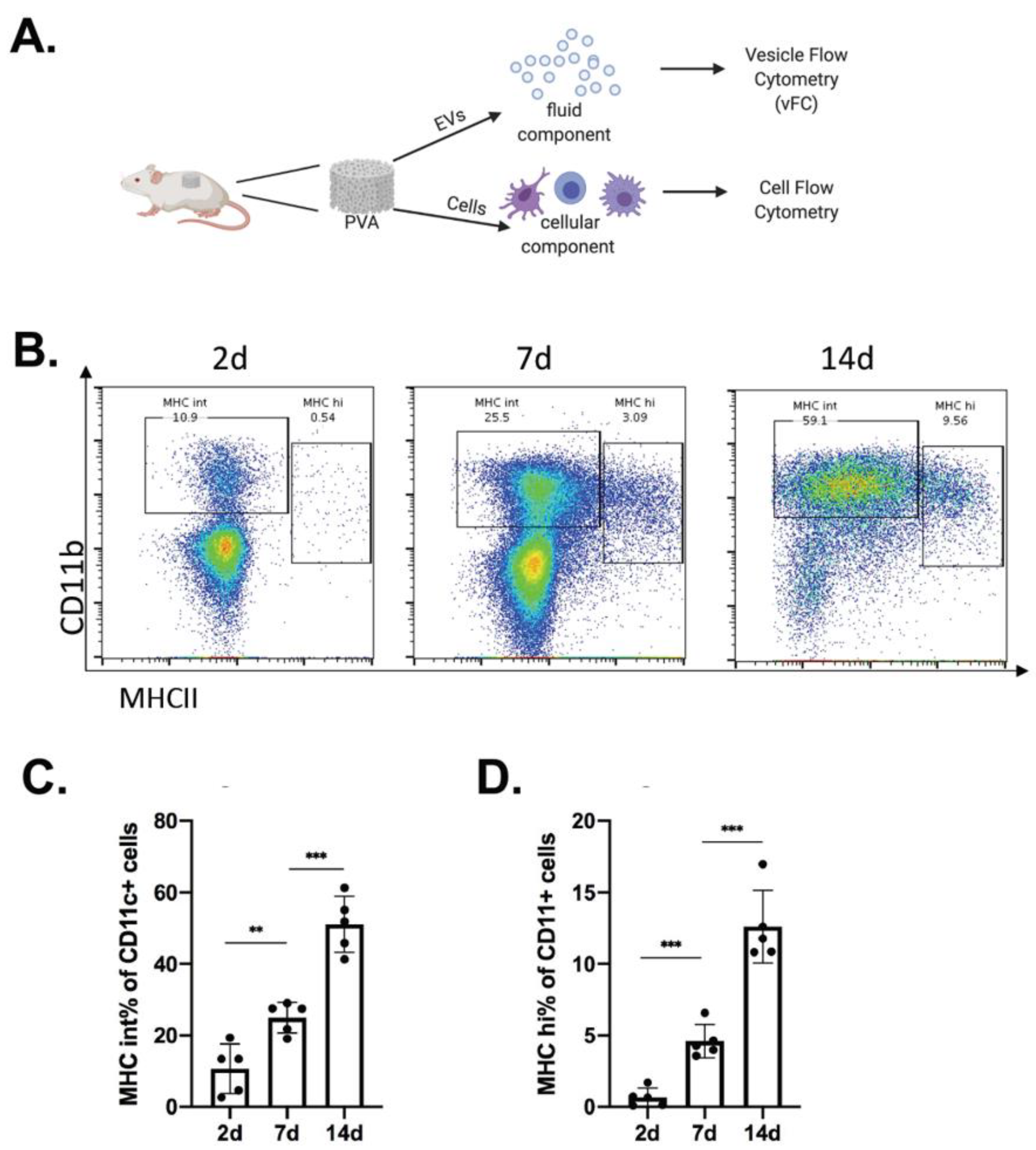
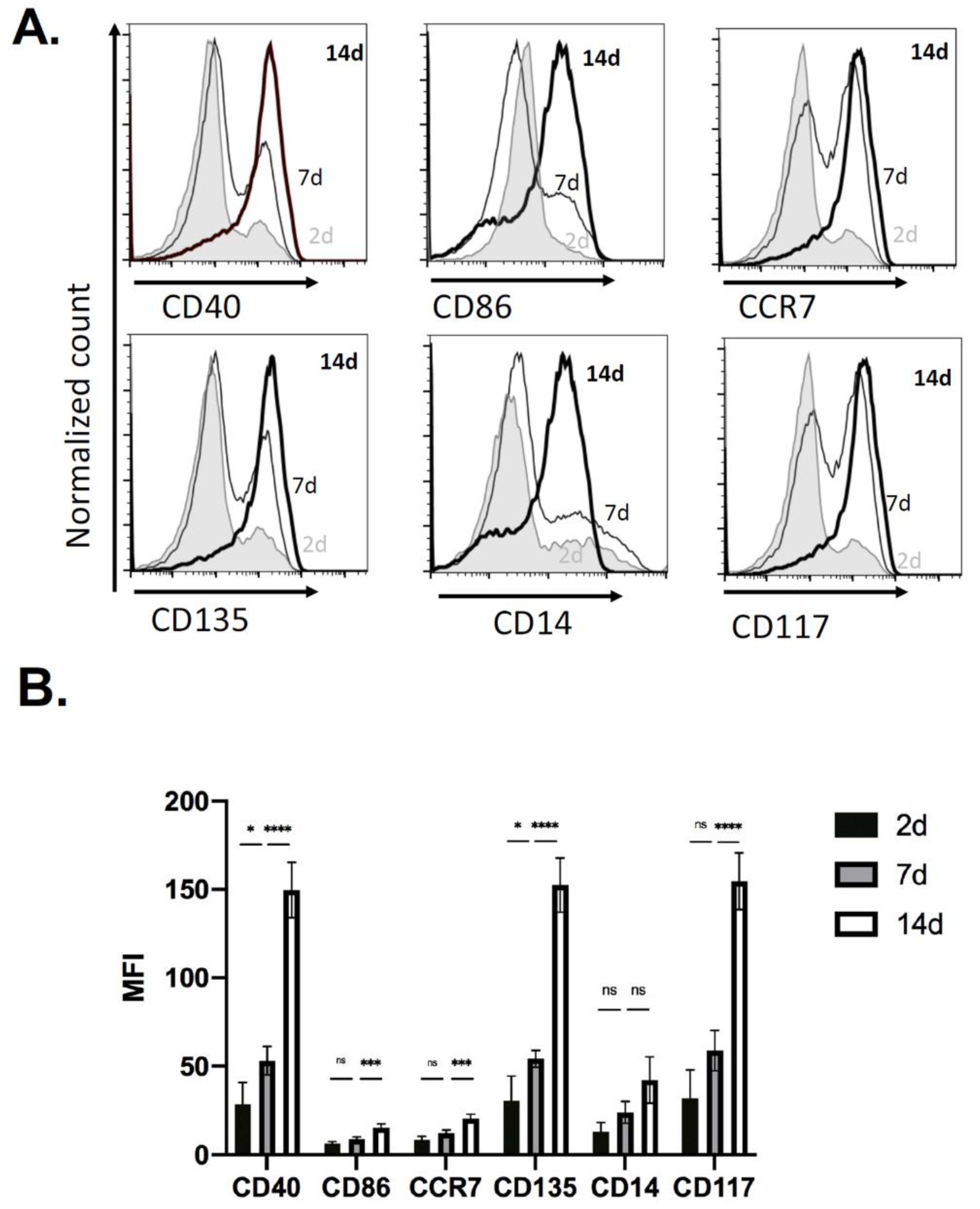
2.1. Immune Cell Recruitment in Immune-Competent Mice
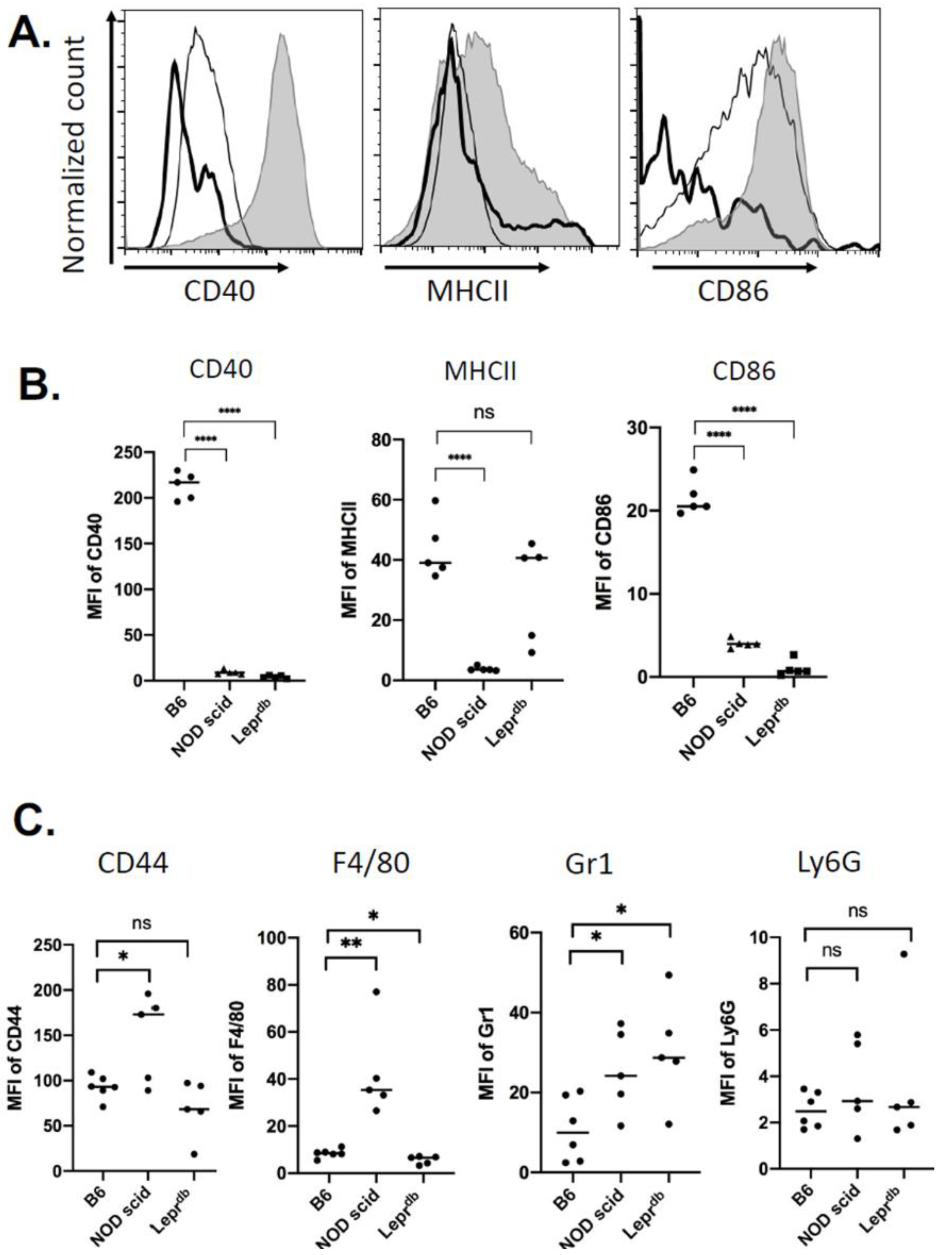
2.2. Immune Cell Recruitment Skewed in Mouse Models of Immuno-Deficiency and Diabetes
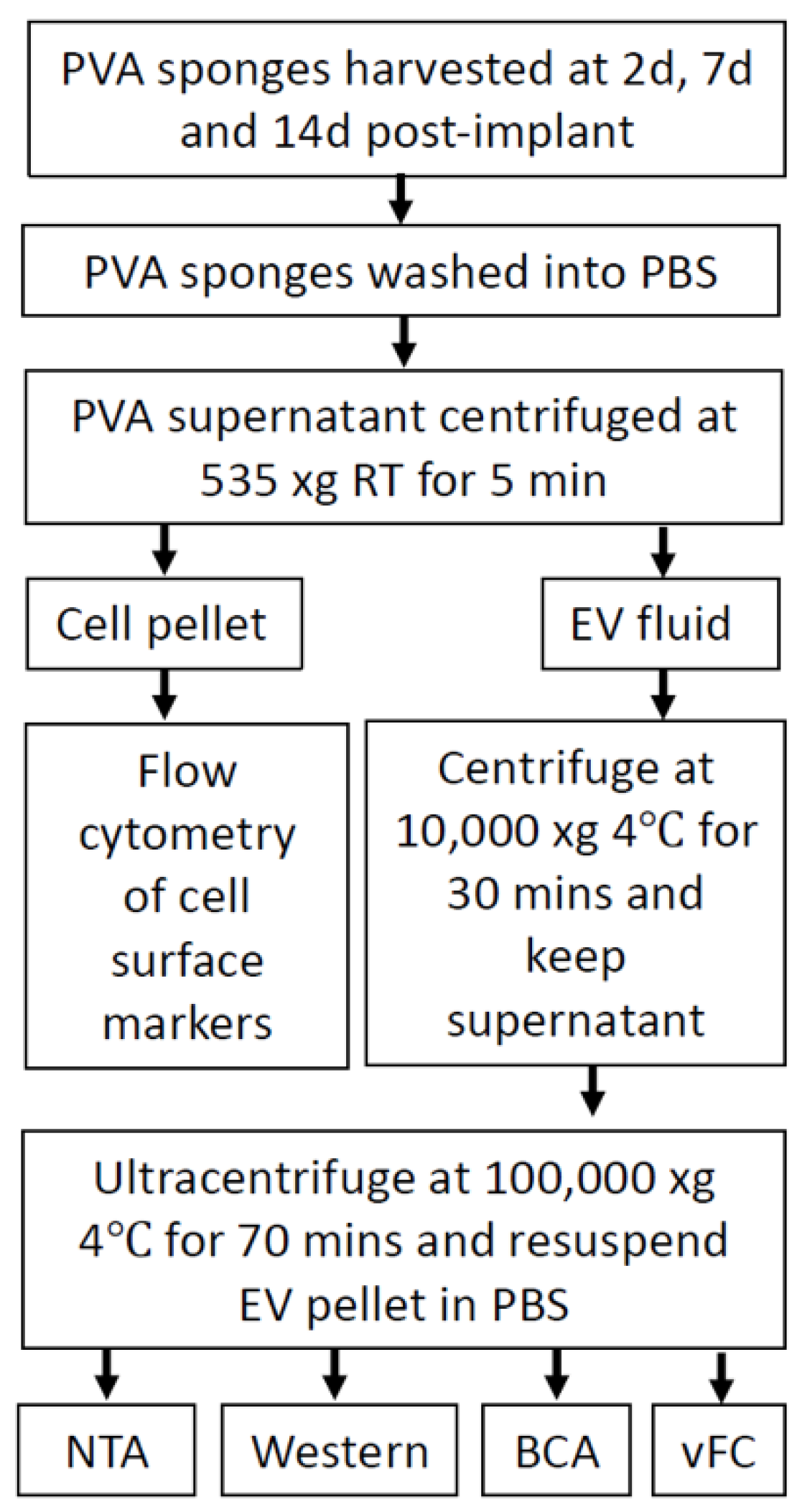
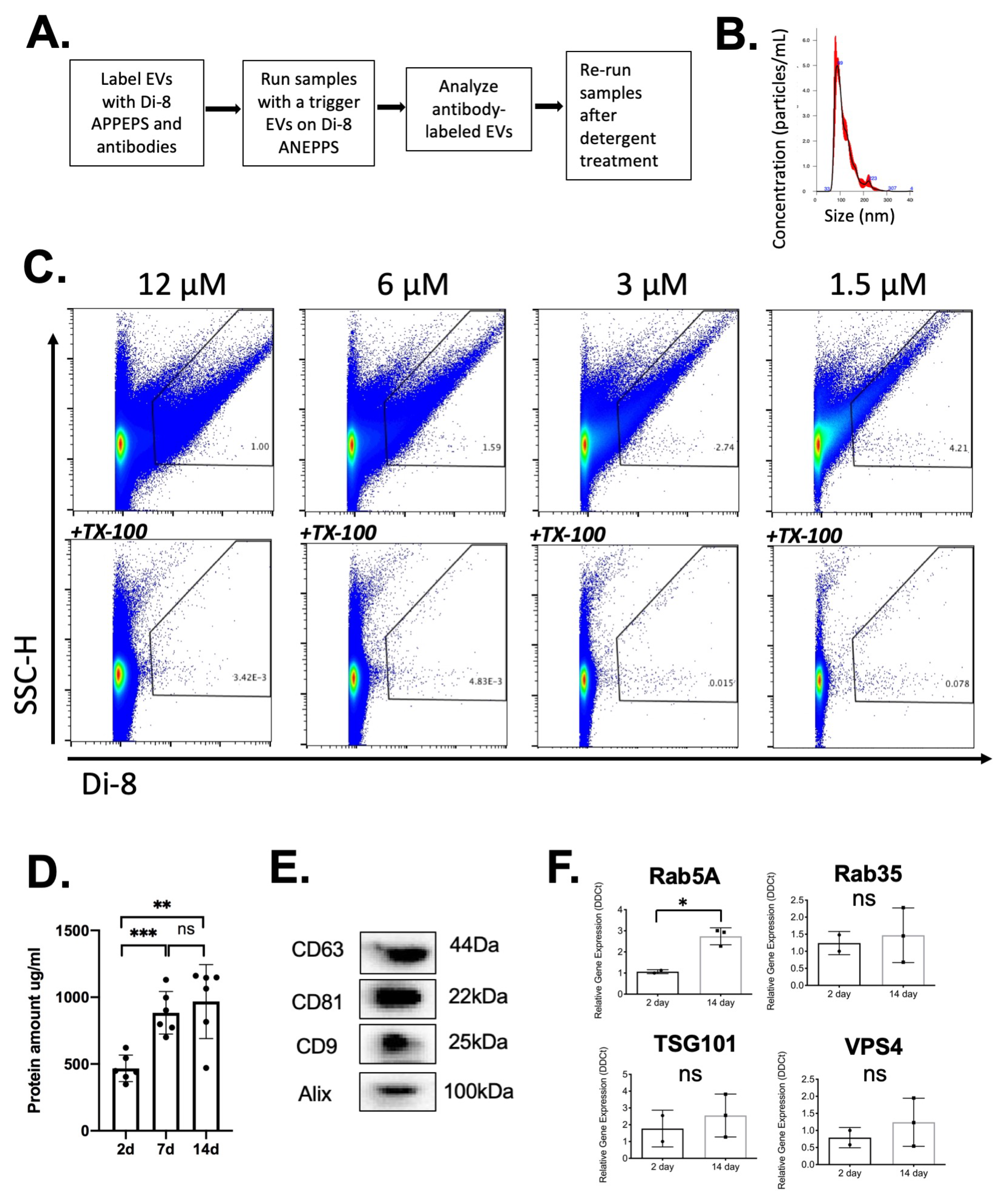
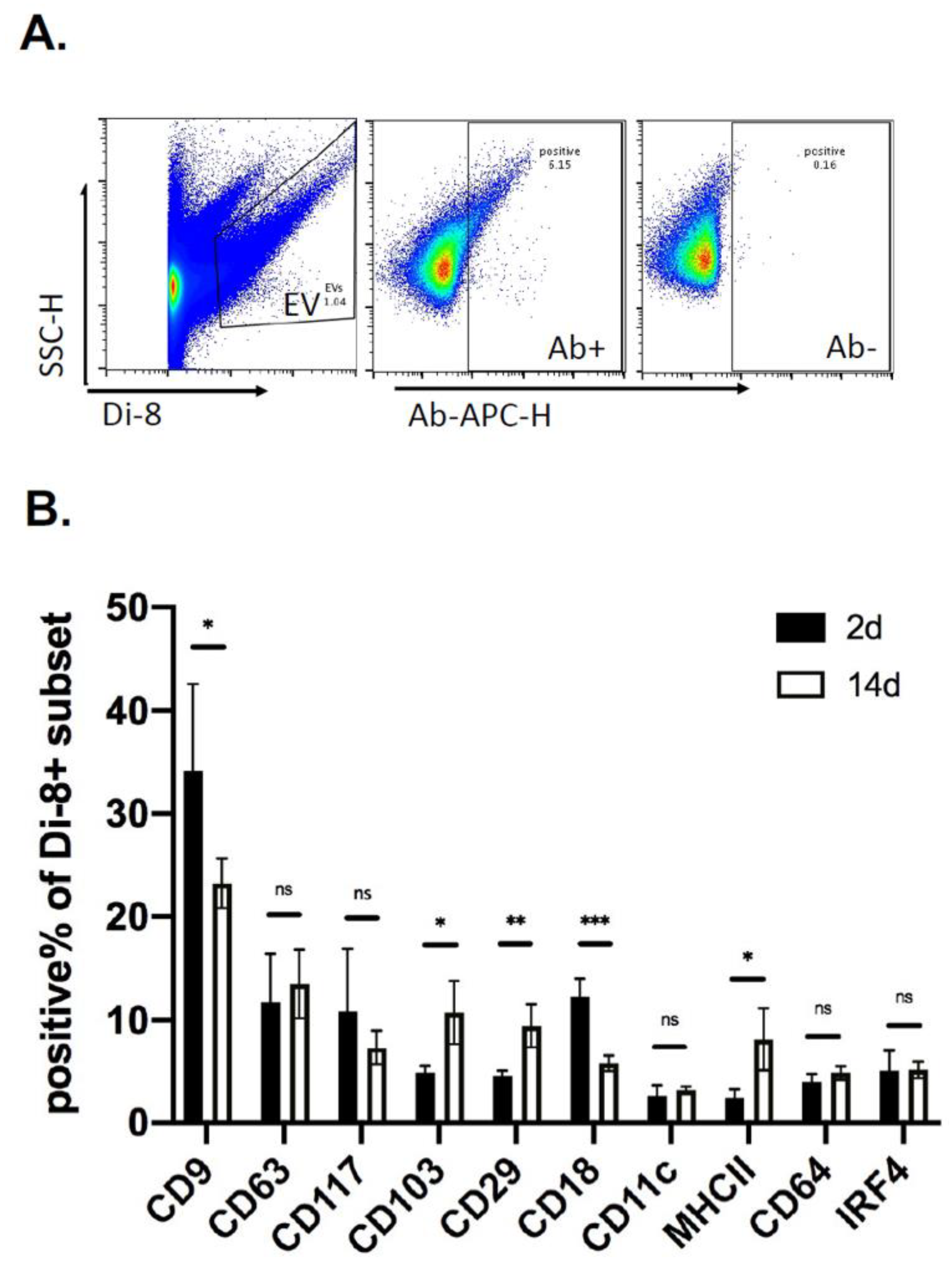
2.3. Analysis of EVs in a Defined Injury Model of Immune Cell Recruitment
2.4. Kinetics of EV Protein Changes in PVA Sponges
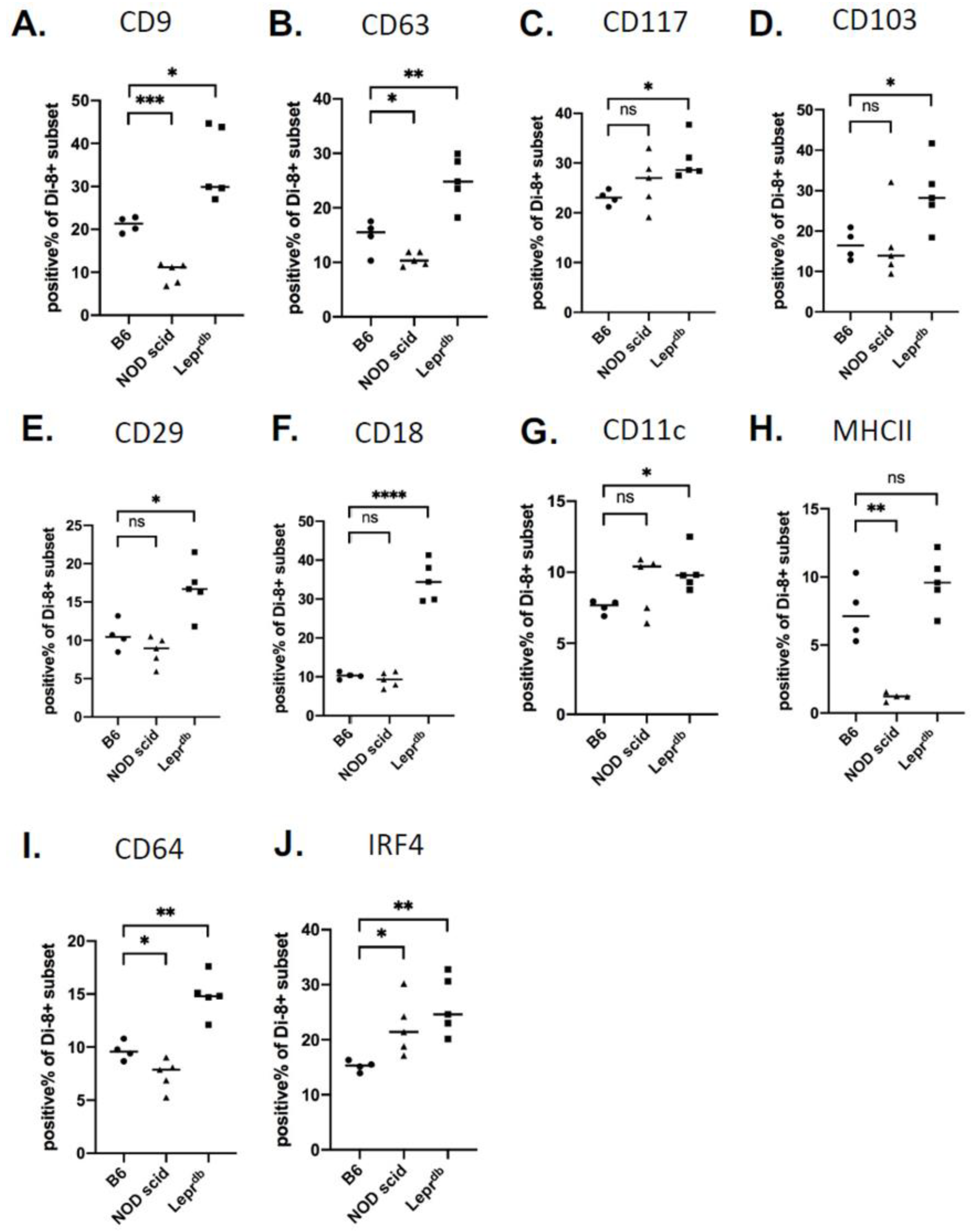
2.5. Changes in Wound EV Proteins in the Wound Bed Immunodeficient and Diabetic Mice
3. Discussion
4. Materials and Methods
4.1. Mice and PVA Sponge Implant and Tissue Harvest
4.2. Enrichment of Extracellular Vesicles from PVA Wound Fluid
4.3. Flow Cytometry Analysis of Cells
4.4. Analysis of EVs by Immunoblotting
4.5. Nanoparticle Tracking Analysis (NTA)
4.6. Quantitative Real-Time PCR
4.7. Vesicle Flow Cytometry
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| EVs | Extracellular vesicles |
| vFC | Vesicle flow cytometry |
| DCs | Dendritic cells |
| PVA | Polyvinyl alcohol |
| NFDM | Nonfat dry milk |
| APCs | Antigen-pressing cells |
| NOD | Non-obese diabetic |
| NTA | Nanoparticle tracking analysis |
References
- Koh, T.J.; DiPietro, L.A. Inflammation and wound healing: The role of the macrophage. Expert Rev. Mol. Med. 2011, 13, e23. [Google Scholar] [CrossRef] [PubMed]
- Minutti, C.M.; Knipper, J.A.; Allen, J.E.; Zaiss, D.M. Tissue-specific contribution of macrophages to wound healing. In Seminars in Cell & Developmental Biology; Elsevier: Amsterdam, The Netherlands, 2017; pp. 3–11. [Google Scholar]
- Eming, S.A.; Martin, P.; Tomic-Canic, M. Wound repair and regeneration: Mechanisms, signaling, and translation. Sci. Transl. Med. 2014, 6, 265sr6. [Google Scholar] [CrossRef]
- Claes, L.; Recknagel, S.; Ignatius, A. Fracture healing under healthy and inflammatory conditions. Nat. Rev. Rheumatol. 2012, 8, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Mietto, B.S.; Mostacada, K.; Martinez, A.M.B. Neurotrauma and inflammation: Cns and pns responses. Mediat. Inflamm. 2015, 2015, 1–14. [Google Scholar] [CrossRef]
- Dlugonska, H.; Gatkowska, J. Exosomes in the context of toxoplasma gondii-host communication. Ann. Parasitol. 2016, 62, 169–174. [Google Scholar] [PubMed]
- Zhang, B.; Yin, Y.; Lai, R.C.; Tan, S.S.; Choo, A.B.H.; Lim, S.K. Mesenchymal stem cells secrete immunologically active exosomes. Stem Cells Dev. 2014, 23, 1233–1244. [Google Scholar] [CrossRef]
- Qin, S.; Dorschner, R.A.; Masini, I.; Lavoie-Gagne, O.; Stahl, P.D.; Costantini, T.W.; Baird, A.; Eliceiri, B.P. Tbc1d3 regulates the payload and biological activity of extracellular vesicles that mediate tissue repair. FASEB J. 2019, 33, 6129–6139. [Google Scholar] [CrossRef]
- Zhang, J.; Guan, J.; Niu, X.; Hu, G.; Guo, S.; Li, Q.; Xie, Z.; Zhang, C.; Wang, Y. Exosomes released from human induced pluripotent stem cells-derived mscs facilitate cutaneous wound healing by promoting collagen synthesis and angiogenesis. J. Transl. Med. 2015, 13, 1–14. [Google Scholar] [CrossRef]
- Shabbir, A.; Cox, A.; Rodriguez-Menocal, L.; Salgado, M.; Badiavas, E.V. Mesenchymal stem cell exosomes induce proliferation and migration of normal and chronic wound fibroblasts, and enhance angiogenesis in vitro. Stem Cells Dev. 2015, 24, 1635–1647. [Google Scholar] [CrossRef]
- Sinha, M.; Sen, C.K.; Singh, K.; Das, A.; Ghatak, S.; Rhea, B.; Blackstone, B.; Powell, H.M.; Khanna, S.; Roy, S. Direct conversion of injury-site myeloid cells to fibroblast-like cells of granulation tissue. Nat. Commun. 2018, 9, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Théry, C.; Duban, L.; Segura, E.; Véron, P.; Lantz, O.; Amigorena, S. Indirect activation of naïve cd4+ t cells by dendritic cell–derived exosomes. Nat. Immunol. 2002, 3, 1156–1162. [Google Scholar] [CrossRef] [PubMed]
- Zitvogel, L.; Regnault, A.; Lozier, A.; Wolfers, J.; Flament, C.; Tenza, D.; Ricciardi-Castagnoli, P.; Raposo, G.; Amigorena, S. Eradication of established murine tumors using a novel cell-free vaccine: Dendritic cell derived exosomes. Nat. Med. 1998, 4, 594–600. [Google Scholar] [CrossRef]
- Raposo, G.; Nijman, H.W.; Stoorvogel, W.; Liejendekker, R.; Harding, C.V.; Melief, C.; Geuze, H.J. B lymphocytes secrete antigen-presenting vesicles. J. Exp. Med. 1996, 183, 1161–1172. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, S.; Schorey, J.S. Exosomes released from infected macrophages contain mycobacterium avium glycopeptidolipids and are proinflammatory. J. Biol. Chem. 2007, 282, 25779–25789. [Google Scholar] [CrossRef] [PubMed]
- Elsner, L.; Muppala, V.; Gehrmann, M.; Lozano, J.; Malzahn, D.; Bickeböller, H.; Brunner, E.; Zientkowska, M.; Herrmann, T.; Walter, L. The heat shock protein hsp70 promotes mouse nk cell activity against tumors that express inducible nkg2d ligands. J. Immunol. 2007, 179, 5523–5533. [Google Scholar] [CrossRef] [PubMed]
- Bretz, N.P.; Ridinger, J.; Rupp, A.-K.; Rimbach, K.; Keller, S.; Rupp, C.; Marmé, F.; Umansky, L.; Umansky, V.; Eigenbrod, T. Body fluid exosomes promote secretion of inflammatory cytokines in monocytic cells via toll-like receptor signaling. J. Biol. Chem. 2013, 288, 36691–36702. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Yin, Y.; Lai, R.C.; Lim, S.K. Immunotherapeutic potential of extracellular vesicles. Front. Immunol. 2014, 5, 518. [Google Scholar] [CrossRef] [PubMed]
- Reis, M.; Mavin, E.; Nicholson, L.; Green, K.; Dickinson, A.M.; Wang, X.-n. Mesenchymal stromal cell-derived extracellular vesicles attenuate dendritic cell maturation and function. Front. Immunol. 2018, 9, 2538. [Google Scholar] [CrossRef]
- Arkhypov, I.; Lasser, S.; Petrova, V.; Weber, R.; Groth, C.; Utikal, J.; Altevogt, P.; Umansky, V. Myeloid cell modulation by tumor-derived extracellular vesicles. Int. J. Mol. Sci. 2020, 21, 6319. [Google Scholar] [CrossRef]
- Withrow, J.; Murphy, C.; Liu, Y.; Hunter, M.; Fulzele, S.; Hamrick, M.W. Extracellular vesicles in the pathogenesis of rheumatoid arthritis and osteoarthritis. Arthritis Res. Ther. 2016, 18, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Alexander, M.; Hu, R.; Runtsch, M.C.; Kagele, D.A.; Mosbruger, T.L.; Tolmachova, T.; Seabra, M.C.; Round, J.L.; Ward, D.M.; O’Connell, R.M. Exosome-delivered micrornas modulate the inflammatory response to endotoxin. Nat. Commun. 2015, 6, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Mardpour, S.; Hassani, S.N.; Mardpour, S.; Sayahpour, F.; Vosough, M.; Ai, J.; Aghdami, N.; Hamidieh, A.A.; Baharvand, H. Extracellular vesicles derived from human embryonic stem cell-mscs ameliorate cirrhosis in thioacetamide-induced chronic liver injury. J. Cell. Physiol. 2018, 233, 9330–9344. [Google Scholar] [CrossRef] [PubMed]
- Romanelli, P.; Bieler, L.; Scharler, C.; Pachler, K.; Kreutzer, C.; Zaunmair, P.; Jakubecova, D.; Mrowetz, H.; Benedetti, B.; Rivera, F.J. Extracellular vesicles can deliver anti-inflammatory and anti-scarring activities of mesenchymal stromal cells after spinal cord injury. Front. Neurol. 2019, 10, 1225. [Google Scholar] [CrossRef] [PubMed]
- Song, T.; Eirin, A.; Zhu, X.; Zhao, Y.; Krier, J.D.; Tang, H.; Jordan, K.L.; Woollard, J.R.; Taner, T.; Lerman, A. Mesenchymal stem cell–derived extracellular vesicles induce regulatory t cells to ameliorate chronic kidney injury. Hypertension 2020, 75, 1223–1232. [Google Scholar] [CrossRef]
- Hu, P.; Yang, Q.; Wang, Q.; Shi, C.; Wang, D.; Armato, U.; Prà, I.D.; Chiarini, A. Mesenchymal stromal cells-exosomes: A promising cell-free therapeutic tool for wound healing and cutaneous regeneration. Burn. Trauma 2019, 7. [Google Scholar] [CrossRef] [PubMed]
- Carrasco, E.; Soto-Heredero, G.; Mittelbrunn, M. The role of extracellular vesicles in cutaneous remodeling and hair follicle dynamics. Int. J. Mol. Sci. 2019, 20, 2758. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.d.F.; Gomes, D.A. Stem cell extracellular vesicles in skin repair. Bioengineering 2019, 6, 4. [Google Scholar] [CrossRef]
- Nolan, J.P. Flow cytometry of extracellular vesicles: Potential, pitfalls, and prospects. Curr. Protoc. Cytom. 2015, 73, 13–14. [Google Scholar] [CrossRef] [PubMed]
- Stoner, S.A.; Duggan, E.; Condello, D.; Guerrero, A.; Turk, J.R.; Narayanan, P.K.; Nolan, J.P. High sensitivity flow cytometry of membrane vesicles. Cytom. Part A 2016, 89, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Nolan, J.P.; Jones, J.C. Detection of platelet vesicles by flow cytometry. Platelets 2017, 28, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Nolan, J.P.; Duggan, E. Analysis of individual extracellular vesicles by flow cytometry. Flow Cytom. Protoc. 2018, 1678, 79–92. [Google Scholar]
- Van Der Vlist, E.J.; Nolte, E.N.; Stoorvogel, W.; Arkesteijn, G.J.; Wauben, M.H. Fluorescent labeling of nano-sized vesicles released by cells and subsequent quantitative and qualitative analysis by high-resolution flow cytometry. Nat. Protoc. 2012, 7, 1311–1326. [Google Scholar] [CrossRef] [PubMed]
- Helft, J.; Böttcher, J.; Chakravarty, P.; Zelenay, S.; Huotari, J.; Schraml, B.U.; Goubau, D.; Reis e Sousa, C. Gm-csf mouse bone marrow cultures comprise a heterogeneous population of cd11c+ mhcii+ macrophages and dendritic cells. Immunity 2015, 42, 1197–1211. [Google Scholar] [CrossRef]
- Prochazka, M.; Gaskins, H.R.; Shultz, L.D.; Leiter, E.H. The nonobese diabetic scid mouse: Model for spontaneous thymomagenesis associated with immunodeficiency. Proc. Natl. Acad. Sci. USA 1992, 89, 3290–3294. [Google Scholar] [CrossRef]
- Lubec, B.; Hermon, M.; Hoeger, H.; Lubec, G. Aromatic hydroxylation in animal models of diabetes mellitus. FASEB J. 1998, 12, 1581–1587. [Google Scholar] [CrossRef] [PubMed]
- Glowacki, J.; Mizuno, S.; Kung, J.; Goff, J.; Epperly, M.; Dixon, T.; Wang, H.; Greenberger, J.S. Effects of mouse genotype on bone wound healing and irradiation-induced delay of healing. In Vivo 2014, 28, 189–196. [Google Scholar] [PubMed]
- Gibran, N.S.; Jang, Y.-C.; Isik, F.F.; Greenhalgh, D.G.; Muffley, L.A.; Underwood, R.A.; Usui, M.L.; Larsen, J.; Smith, D.G.; Bunnett, N. Diminished neuropeptide levels contribute to the impaired cutaneous healing response associated with diabetes mellitus. J. Surg. Res. 2002, 108, 122–128. [Google Scholar] [CrossRef]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K. Minimal information for studies of extracellular vesicles 2018 (misev2018): A position statement of the international society for extracellular vesicles and update of the misev2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef]
- Leoni, G.; Neumann, P.; Sumagin, R.; Denning, T.; Nusrat, A. Wound repair: Role of immune–epithelial interactions. Mucosal Immunol. 2015, 8, 959–968. [Google Scholar] [CrossRef]
- Gutiérrez-Vázquez, C.; Villarroya-Beltri, C.; Mittelbrunn, M.; Sánchez-Madrid, F. Transfer of extracellular vesicles during immune cell-cell interactions. Immunol. Rev. 2013, 251, 125–142. [Google Scholar] [CrossRef]
- Muller, L.; Mitsuhashi, M.; Simms, P.; Gooding, W.E.; Whiteside, T.L. Tumor-derived exosomes regulate expression of immune function-related genes in human t cell subsets. Sci. Rep. 2016, 6, 1–13. [Google Scholar] [CrossRef]
- Veerman, R.E.; Akpinar, G.G.; Eldh, M.; Gabrielsson, S. Immune cell-derived extracellular vesicles–functions and therapeutic applications. Trends Mol. Med. 2019, 25, 382–394. [Google Scholar] [CrossRef] [PubMed]
- Kojima, M.; Gimenes-Junior, J.A.; Chan, T.W.; Eliceiri, B.P.; Baird, A.; Costantini, T.W.; Coimbra, R. Exosomes in postshock mesenteric lymph are key mediators of acute lung injury triggering the macrophage activation via toll-like receptor 4. FASEB J. 2018, 32, 97–110. [Google Scholar] [CrossRef]
- Kojima, M.; Gimenes-Junior, J.A.; Langness, S.; Morishita, K.; Lavoie-Gagne, O.; Eliceiri, B.; Costantini, T.W.; Coimbra, R. Exosomes, not protein or lipids, in mesenteric lymph activate inflammation: Unlocking the mystery of post-shock multiple organ failure. J. Trauma Acute Care Surg. 2017, 82, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.M.; Teixeira, J.H.; Almeida, M.I.; Goncalves, R.M.; Barbosa, M.A.; Santos, S.G. Extracellular vesicles: Immunomodulatory messengers in the context of tissue repair/regeneration. Eur. J. Pharm. Sci. 2017, 98, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Patel, D.B.; Santoro, M.; Born, L.J.; Fisher, J.P.; Jay, S.M. Towards rationally designed biomanufacturing of therapeutic extracellular vesicles: Impact of the bioproduction microenvironment. Biotechnol. Adv. 2018, 36, 2051–2059. [Google Scholar] [CrossRef]
- Baird, A.; Deng, C.; Eliceiri, M.H.; Haghi, F.; Dang, X.; Coimbra, R.; Costantini, T.W.; Torbett, B.E.; Eliceiri, B.P. Mice engrafted with human hematopoietic stem cells support a human myeloid cell inflammatory response in vivo. Wound Repair Regen. 2016, 24, 1004–1014. [Google Scholar] [CrossRef] [PubMed]
- Welsh, J.A.; Van Der Pol, E.; Arkesteijn, G.J.; Bremer, M.; Brisson, A.; Coumans, F.; Dignat-George, F.; Duggan, E.; Ghiran, I.; Giebel, B. Miflowcyt-ev: A framework for standardized reporting of extracellular vesicle flow cytometry experiments. J. Extracell. Vesicles 2020, 9, 1713526. [Google Scholar] [CrossRef] [PubMed]
| Antibody | Clone | Flour | Cat# | Vendor |
|---|---|---|---|---|
| CD9 | MZ3 | APC | 130-102-612 | Miltenyi Biotec |
| CD63 | REA563 | APC | 130-108-924 | Miltenyi Biotec |
| CD103 | 2E7 | APC | 130-102-516 | Miltenyi Biotec |
| CD117 | 3C11 | APC | 130-122-948 | Miltenyi Biotec |
| CD29 | HMβ1-1 | APC | 130-102-557 | Miltenyi Biotec |
| CD18 | M18/2 | APC | 130-104-019 | Miltenyi Biotec |
| CD11c | N418 | APC | 130-119-802 | Miltenyi Biotec |
| MHCII | M5/114.15.2 | APC | 130-123-785 | Miltenyi Biotec |
| CD64 | X54-5/7.1 | APC | 139305 | Biolegend |
| IRF4 | IRF4.3E4 | Alexa Flour 647 | 646407 | Biolegend |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qian, J.; Park, D.J.; Perrott, S.; Patel, P.; Eliceiri, B.P. Genetic Background and Kinetics Define Wound Bed Extracellular Vesicles in a Mouse Model of Cutaneous Injury. Int. J. Mol. Sci. 2021, 22, 3551. https://doi.org/10.3390/ijms22073551
Qian J, Park DJ, Perrott S, Patel P, Eliceiri BP. Genetic Background and Kinetics Define Wound Bed Extracellular Vesicles in a Mouse Model of Cutaneous Injury. International Journal of Molecular Sciences. 2021; 22(7):3551. https://doi.org/10.3390/ijms22073551
Chicago/Turabian StyleQian, Jin, Dong Jun Park, Sophia Perrott, Parth Patel, and Brian P. Eliceiri. 2021. "Genetic Background and Kinetics Define Wound Bed Extracellular Vesicles in a Mouse Model of Cutaneous Injury" International Journal of Molecular Sciences 22, no. 7: 3551. https://doi.org/10.3390/ijms22073551
APA StyleQian, J., Park, D. J., Perrott, S., Patel, P., & Eliceiri, B. P. (2021). Genetic Background and Kinetics Define Wound Bed Extracellular Vesicles in a Mouse Model of Cutaneous Injury. International Journal of Molecular Sciences, 22(7), 3551. https://doi.org/10.3390/ijms22073551







