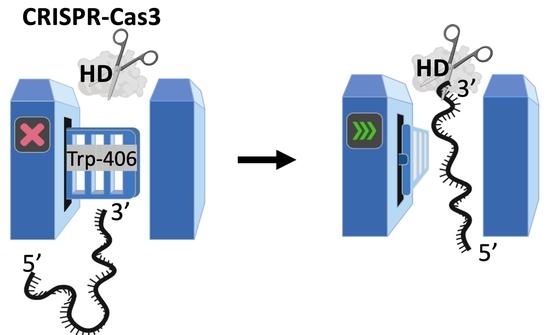
A Tryptophan ‘Gate’ in the CRISPR-Cas3 Nuclease Controls ssDNA Entry into the Nuclease Site, That When Removed Results in Nuclease Hyperactivity
Abstract
1. Introduction
2. Results
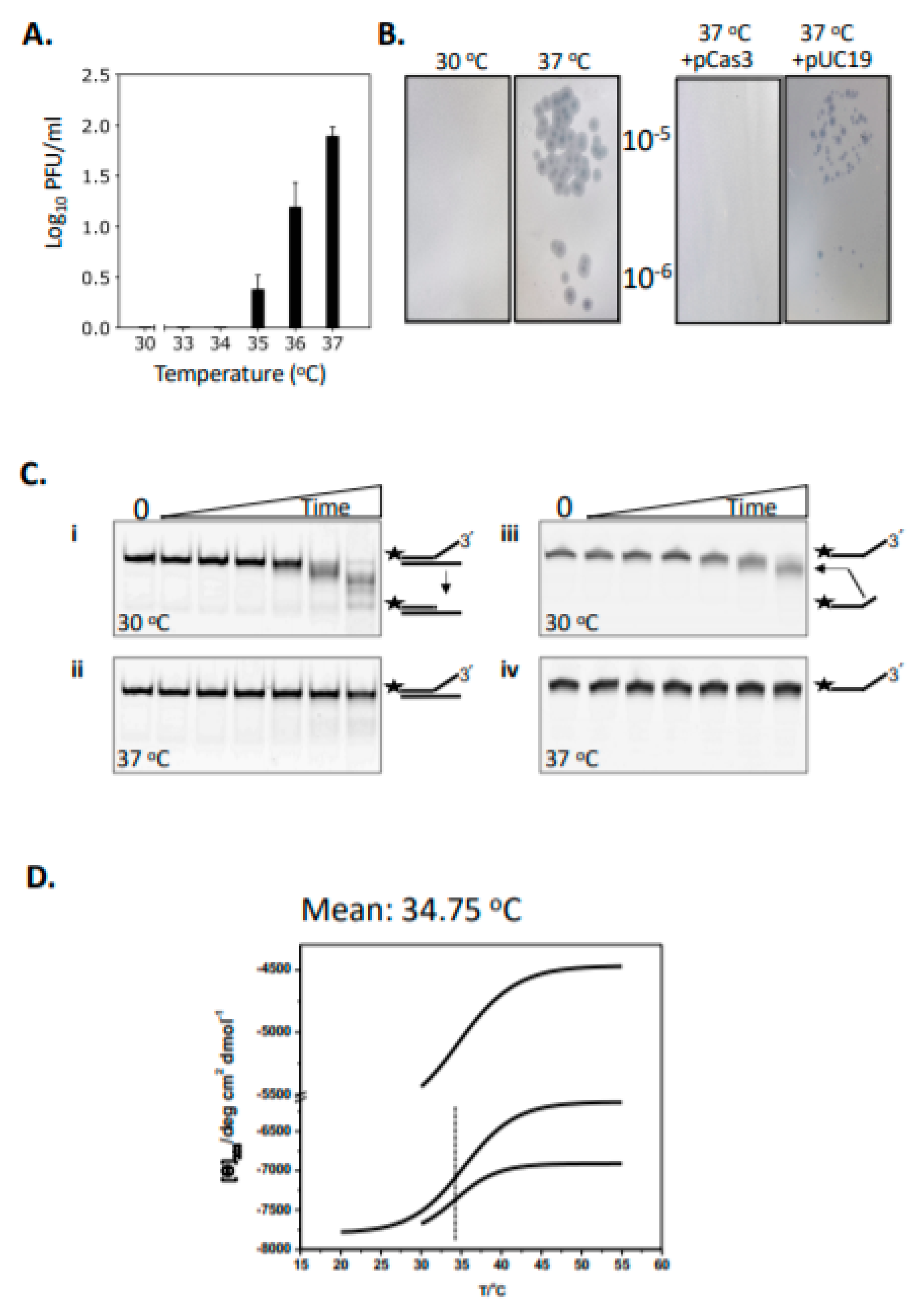
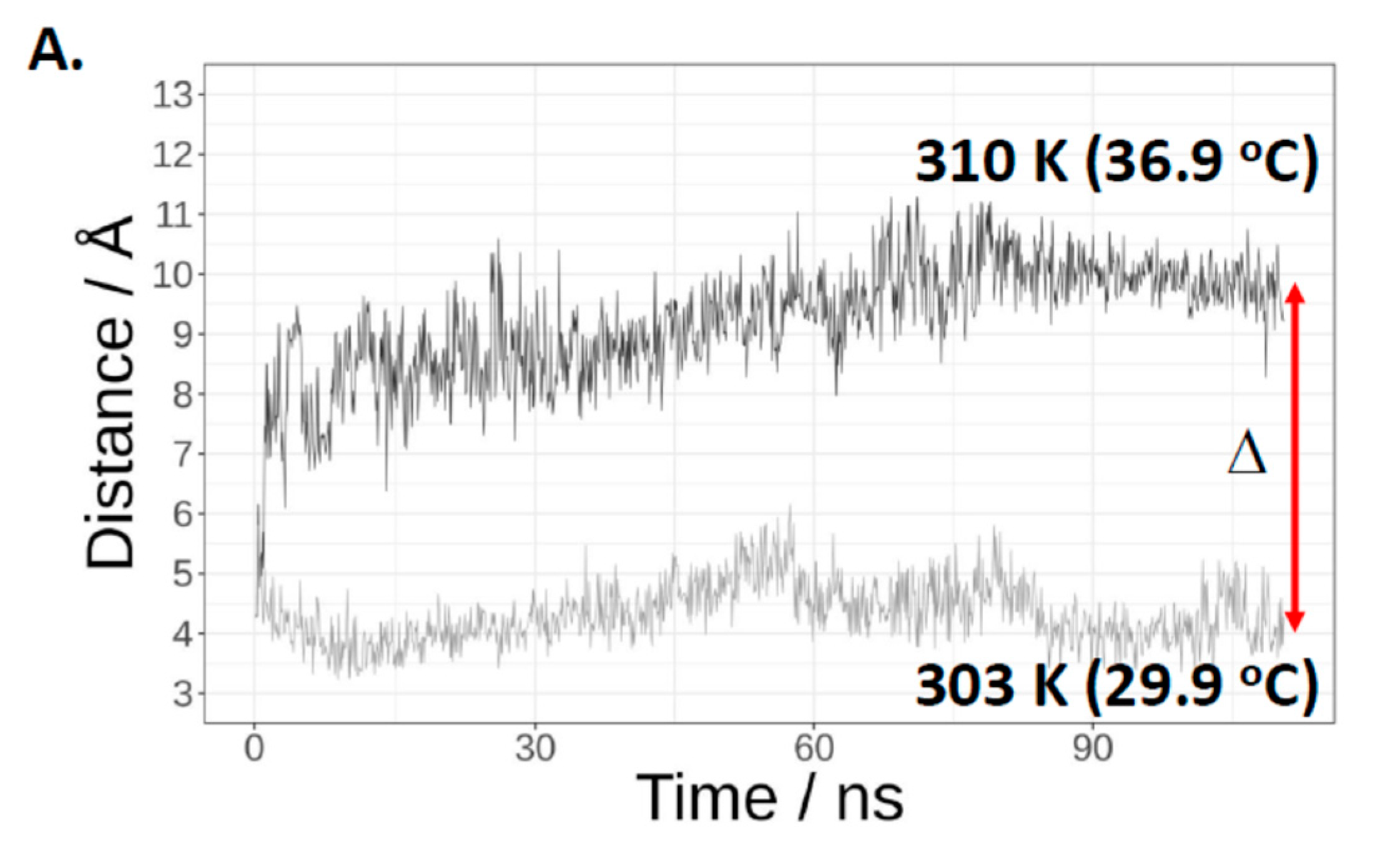
2.1. Cas3 Nuclease Activity Is Modulated by Temperature Changes That Correspond with Conformational Re-Arrangement of Cas3 in Transition from 30 °C to 37 °C
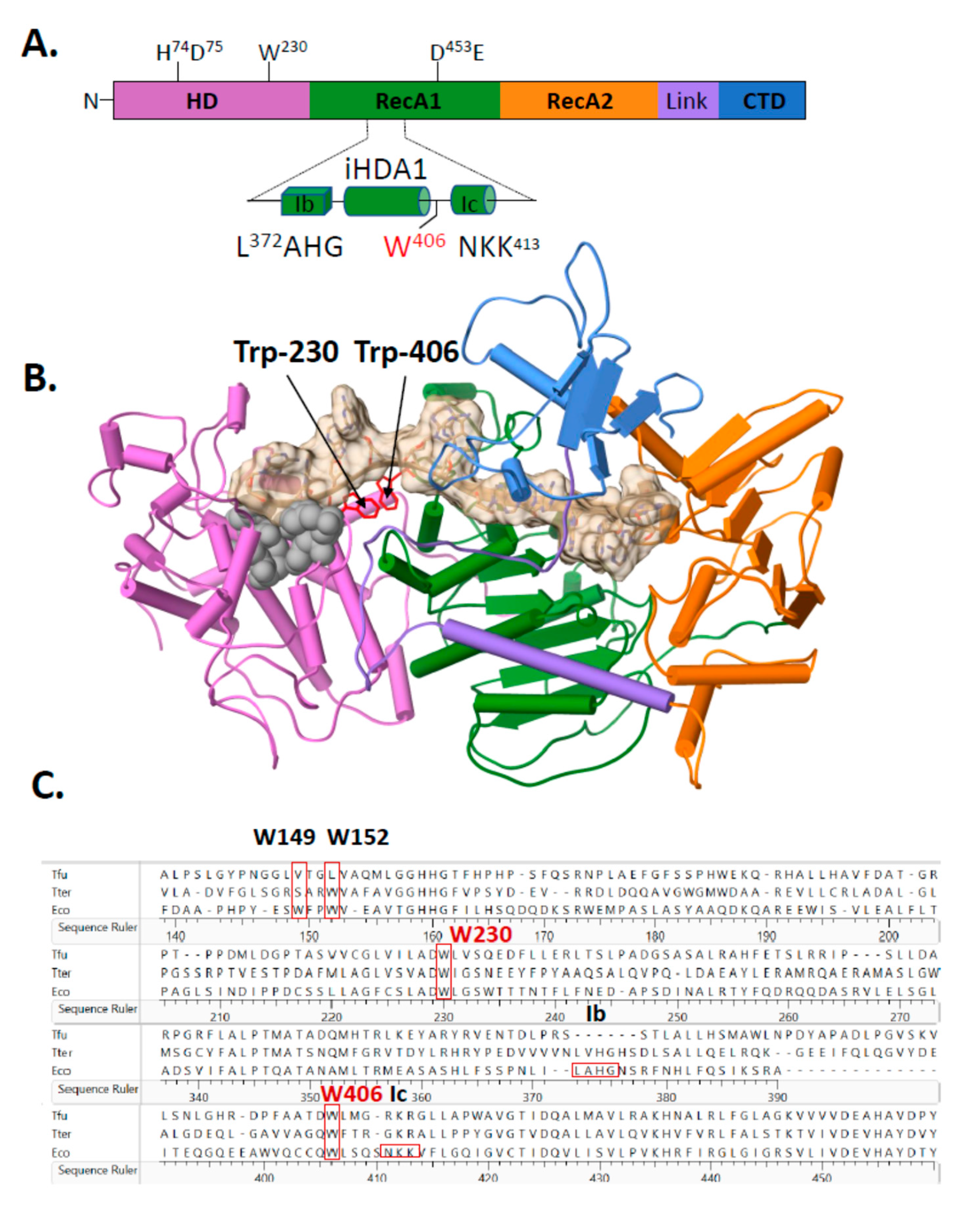
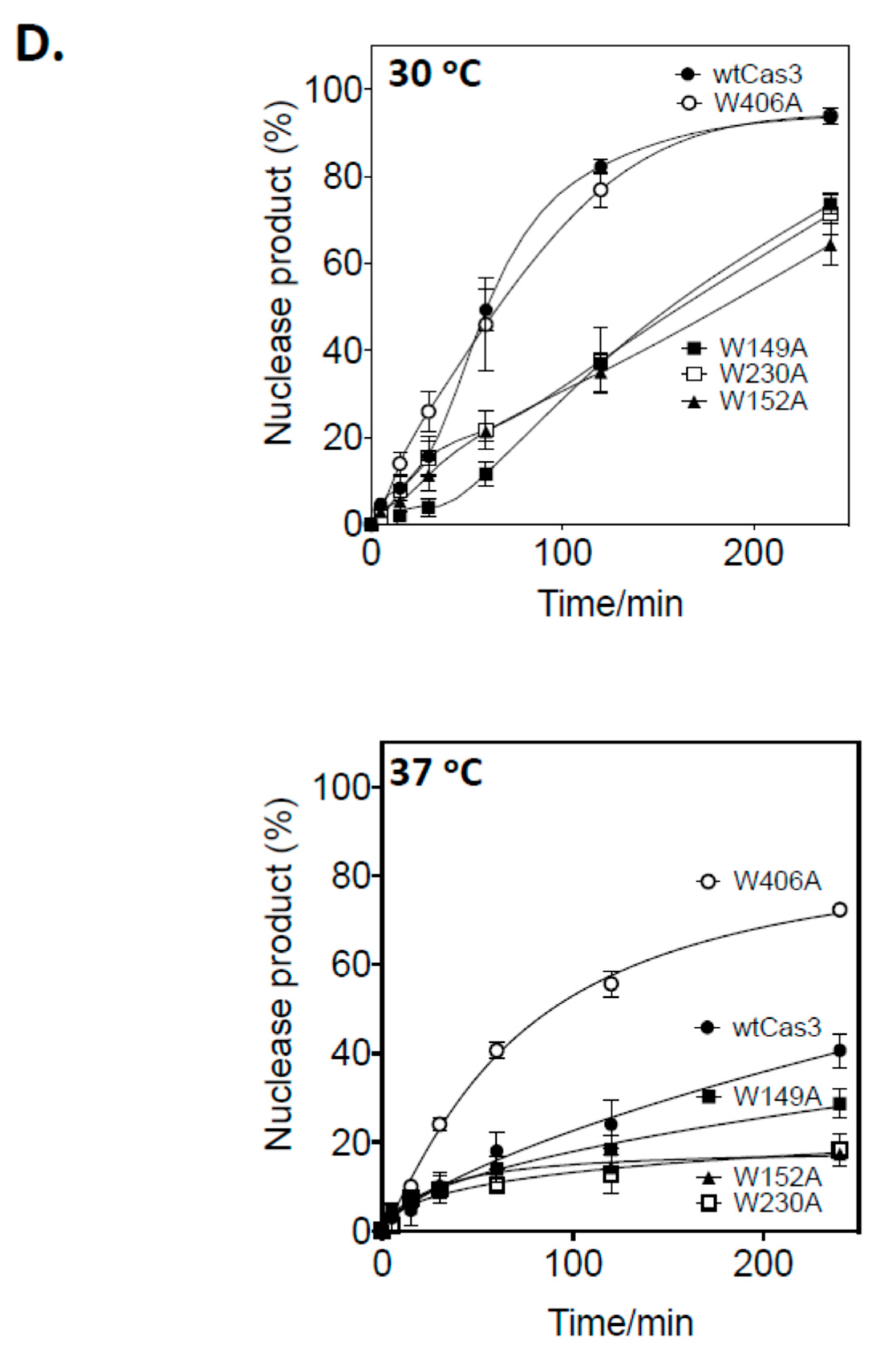
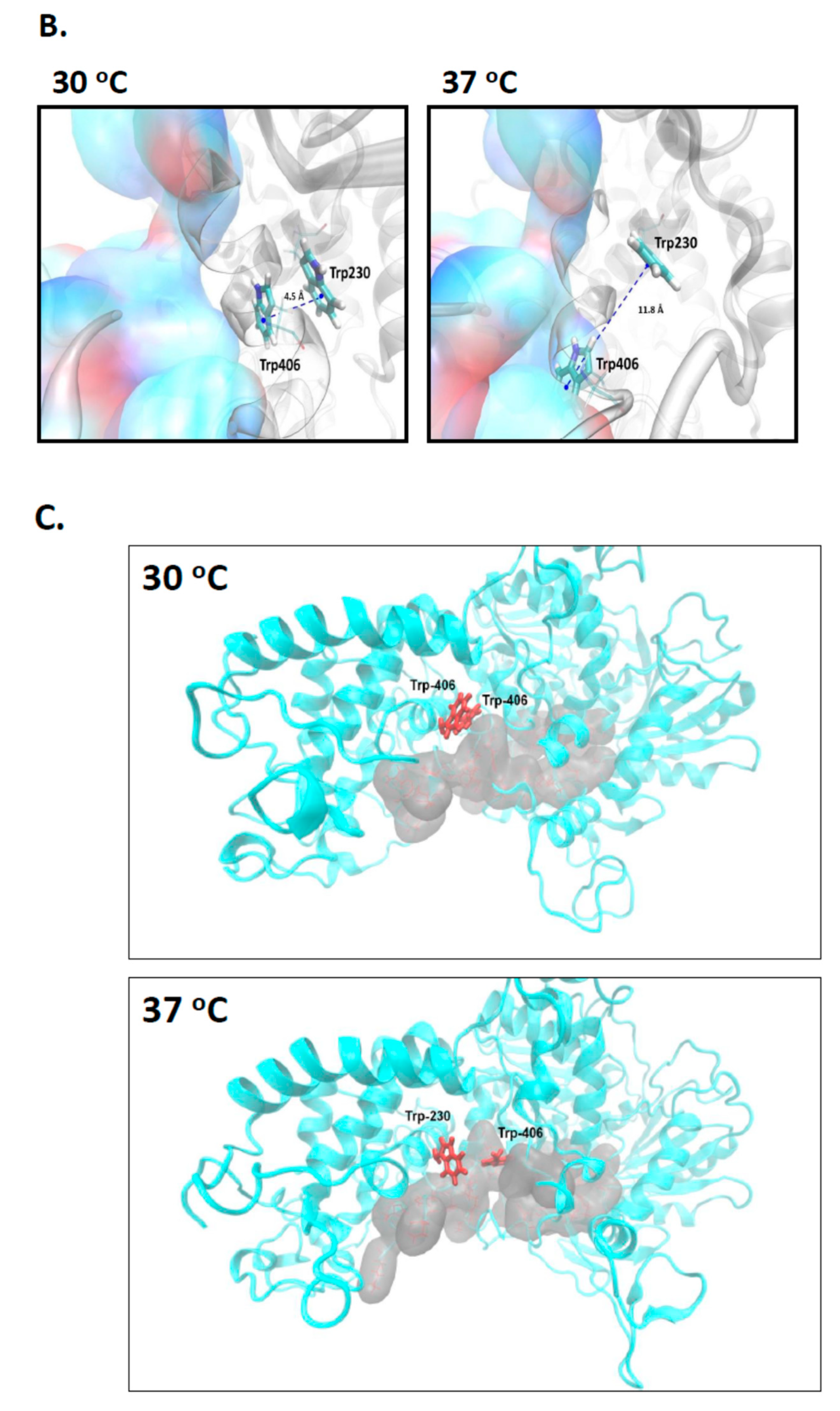
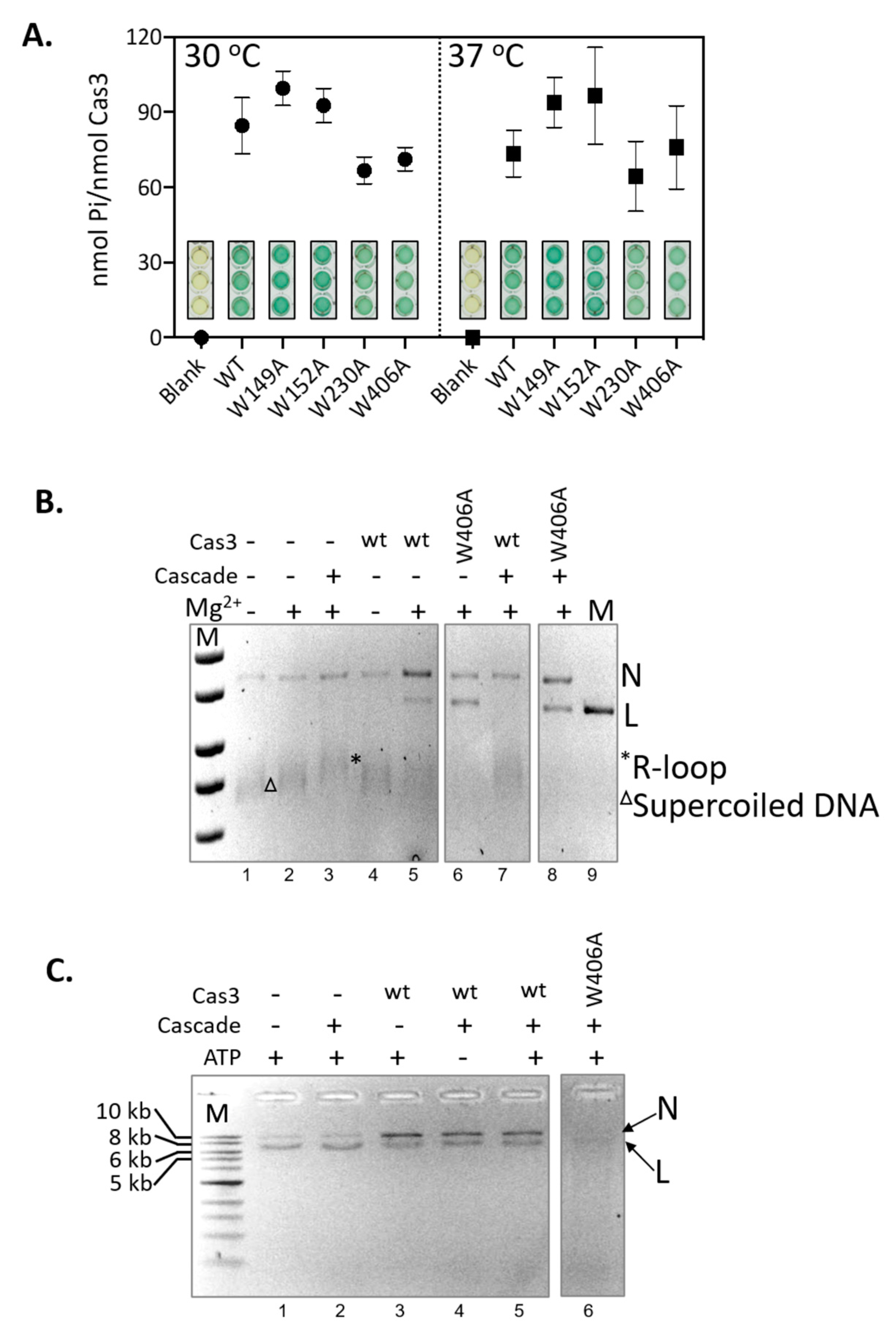
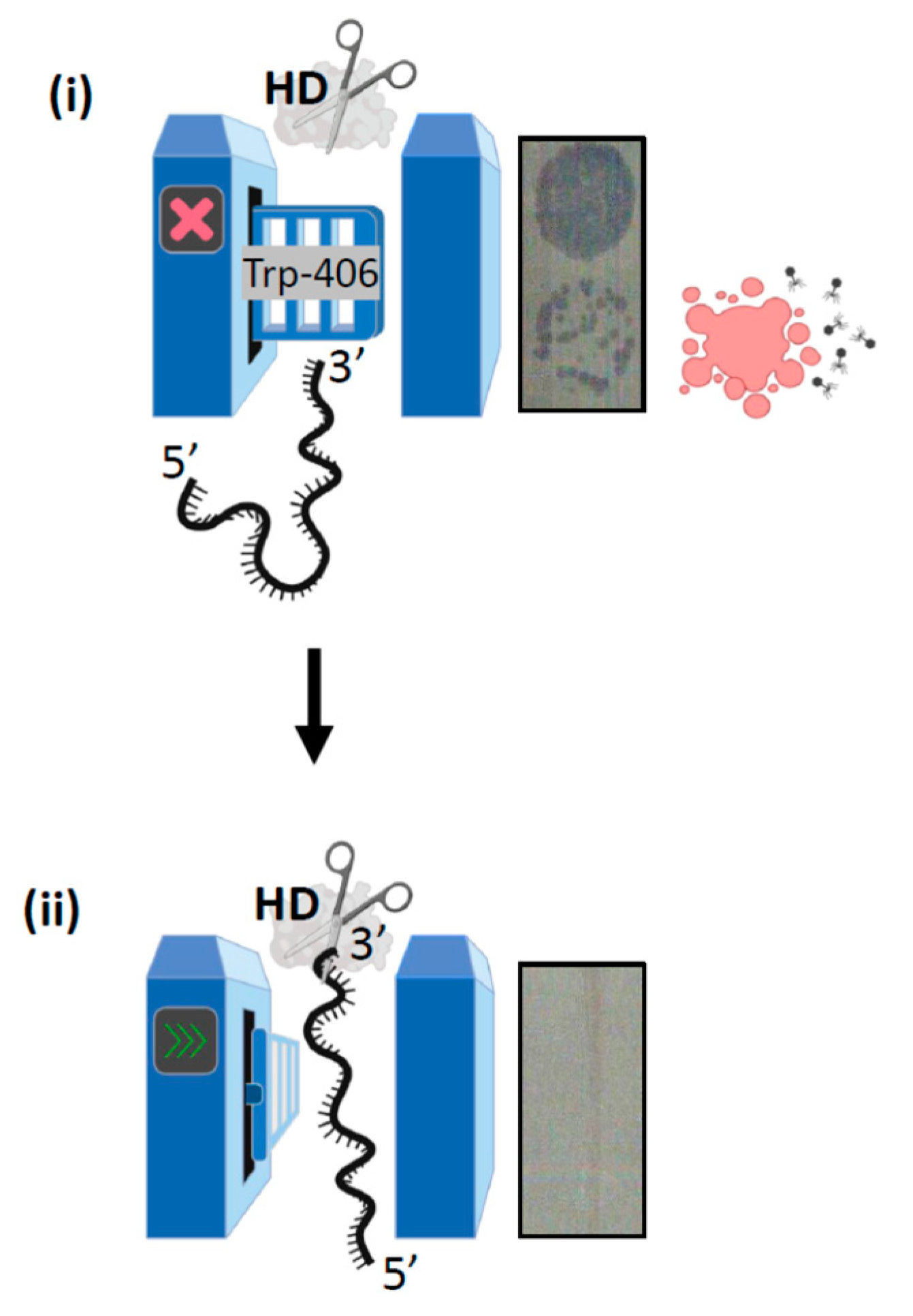
2.2. A Single Tryptophan Residue (Trp-406) Modulates the Cas3 Nuclease Activity
2.3. Evidence That Trp-406 Is a ‘Gate’ in Cas3 That Controls Access of ssDNA to the Nuclease Active Site
2.4. Hyperactivity of Cas3W406A Nuclease Is Independent of Cas3 ATPase Activity and of the Cascade Complex
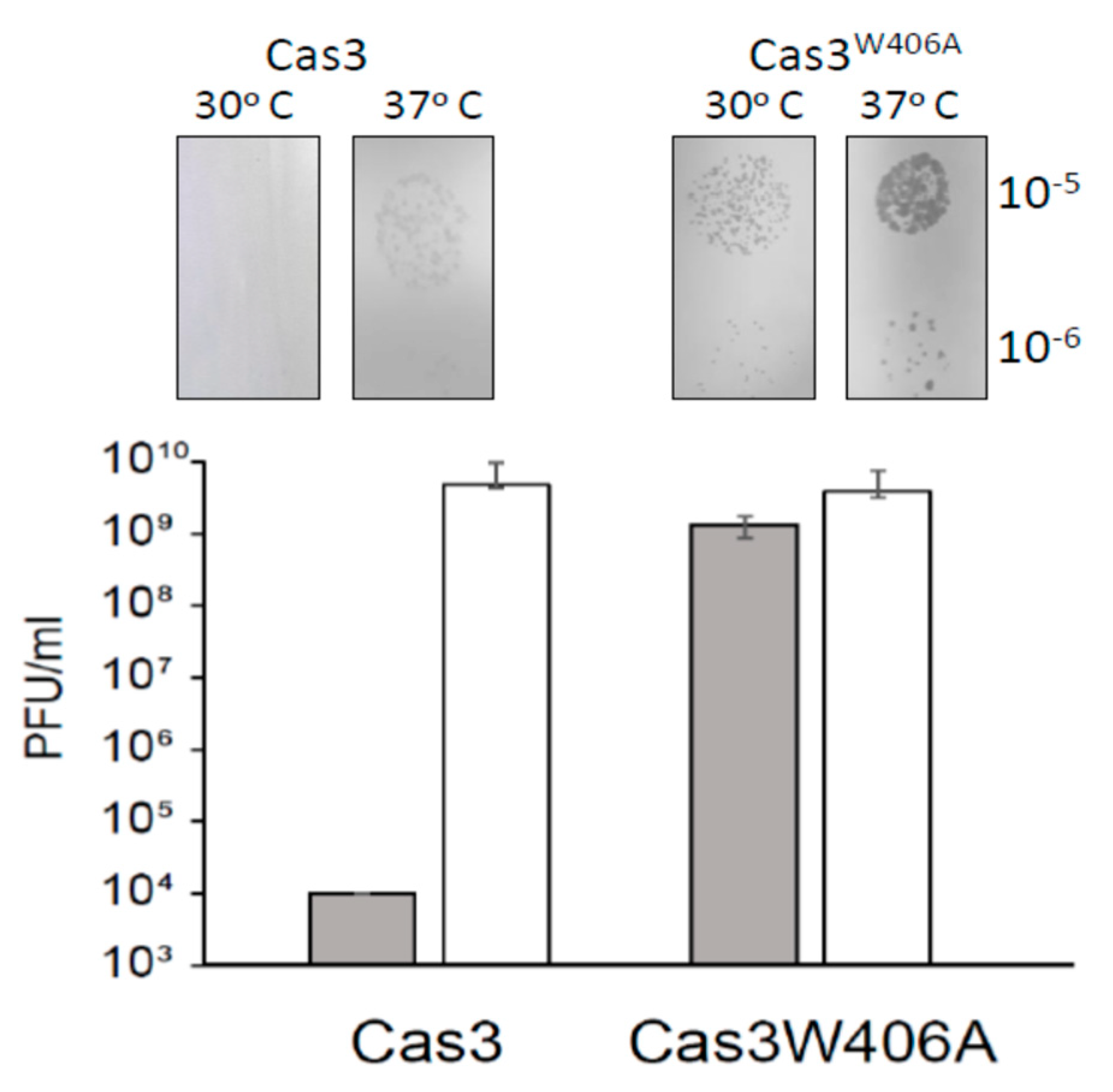
2.5. Cas3W406A Does Not Protect E. coli Cells from Phage Lysis
3. Discussion
4. Materials and Methods
4.1. E. coli Strains, Plasmids and Molecular Cloning
4.2. Protein Expression and Purification
4.3. DNA Substrate Preparation
4.4. Protein-Nucleic Acid Assays
4.5. ATPase Assays
4.6. Phage Sensitivity Assay by Plaque Formation
4.7. Construction of the Chromosomal Cas3 Point Mutant
4.8. Circular Dichroism Measurements and Analysis of Cas3 Secondary Structure
4.9. Molecular Dynamics Simulations of Cas3 Protein
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Knott, G.J.; Doudna, J.A. CRISPR-Cas guides the future of genetic engineering. Science 2018, 361, 866–869. [Google Scholar] [CrossRef] [PubMed]
- Mojica, F.J.; Rodriguez-Valera, F. The discovery of CRISPR in archaea and bacteria. FEBS J. 2016, 283, 3162–3169. [Google Scholar] [CrossRef]
- Westra, E.R.; Levin, B.R. It is unclear how important CRISPR-Cas systems are for protecting natural populations of bacteria against infections by mobile genetic elements. Proc. Natl. Acad. Sci. USA 2020, 117, 27777–27785. [Google Scholar] [CrossRef]
- Makarova, K.S.; Wolf, Y.I.; Iranzo, J.; Shmakov, S.A.; Alkhnbashi, O.S.; Brouns, S.J.J.; Charpentier, E.; Cheng, D.; Haft, D.H.; Horvath, P.; et al. Evolutionary classification of CRISPR-Cas systems: A burst of class 2 and derived variants. Nat. Rev. MicroBiol. 2020, 18, 67–83. [Google Scholar] [CrossRef]
- Brouns, S.J.; Jore, M.M.; Lundgren, M.; Westra, E.R.; Slijkhuis, R.J.; Snijders, A.P.; Dickman, M.J.; Makarova, K.S.; Koonin, E.V.; van der Oost, J. Small CRISPR RNAs guide antiviral defense in prokaryotes. Science 2008, 321, 960–964. [Google Scholar] [CrossRef]
- Plagens, A.; Richter, H.; Charpentier, E.; Randau, L. DNA and RNA interference mechanisms by CRISPR-Cas surveillance complexes. FEMS MicroBiol. Rev. 2015, 39, 442–463. [Google Scholar] [CrossRef] [PubMed]
- Jackson, R.N.; Golden, S.M.; van Erp, P.B.; Carter, J.; Westra, E.R.; Brouns, S.J.; van der Oost, J.; Terwilliger, T.C.; Read, R.J.; Wiedenheft, B. Crystal structure of the CRISPR RNA-guided surveillance complex from Escherichia coli. Science 2014, 345, 1473–1479. [Google Scholar] [CrossRef]
- Dillard, K.E.; Brown, M.W.; Johnson, N.V.; Xiao, Y.; Dolan, A.; Hernandez, E.; Dahlhauser, S.D.; Kim, Y.; Myler, L.R.; Anslyn, E.V.; et al. Assembly and Translocation of a CRISPR-Cas Primed Acquisition Complex. Cell 2018, 175, 934–946 e915. [Google Scholar] [CrossRef]
- Wiedenheft, B.; Lander, G.C.; Zhou, K.; Jore, M.M.; Brouns, S.J.; van der Oost, J.; Doudna, J.A.; Nogales, E. Structures of the RNA-guided surveillance complex from a bacterial immune system. Nature 2011, 477, 486–489. [Google Scholar] [CrossRef]
- Szczelkun, M.D.; Tikhomirova, M.S.; Sinkunas, T.; Gasiunas, G.; Karvelis, T.; Pschera, P.; Siksnys, V.; Seidel, R. Direct observation of R-loop formation by single RNA-guided Cas9 and Cascade effector complexes. Proc. Natl. Acad. Sci. USA 2014, 111, 9798–9803. [Google Scholar] [CrossRef]
- Semenova, E.; Jore, M.M.; Datsenko, K.A.; Semenova, A.; Westra, E.R.; Wanner, B.; van der Oost, J.; Brouns, S.J.; Severinov, K. Interference by clustered regularly interspaced short palindromic repeat (CRISPR) RNA is governed by a seed sequence. Proc. Natl. Acad. Sci. USA 2011, 108, 10098–10103. [Google Scholar] [CrossRef]
- Shah, S.A.; Erdmann, S.; Mojica, F.J.; Garrett, R.A. Protospacer recognition motifs: Mixed identities and functional diversity. RNA Biol. 2013, 10, 891–899. [Google Scholar] [CrossRef]
- Sasnauskas, G.; Siksnys, V. CRISPR adaptation from a structural perspective. Curr. Opin. Struct. Biol. 2020, 65, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Shiriaeva, A.; Fedorov, I.; Vyhovskyi, D.; Severinov, K. Detection of CRISPR adaptation. Biochem. Soc. Tran.s 2020, 48, 257–269. [Google Scholar] [CrossRef]
- Lau, C.H.; Reeves, R.; Bolt, E.L. Adaptation processes that build CRISPR immunity: Creative destruction, updated. Essays Biochem. 2019, 63, 227–235. [Google Scholar] [PubMed]
- Jore, M.M.; Lundgren, M.; van Duijn, E.; Bultema, J.B.; Westra, E.R.; Waghmare, S.P.; Wiedenheft, B.; Pul, U.; Wurm, R.; Wagner, R.; et al. Structural basis for CRISPR RNA-guided DNA recognition by Cascade. Nat. Struct. Mol. Biol. 2012, 18, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Hegazy, Y.A.; Fernando, C.M.; Tran, E.J. The balancing act of R-loop biology: The good, the bad, and the ugly. J. Biol. Chem. 2020, 295, 905–913. [Google Scholar] [CrossRef]
- He, L.; St John James, M.; Radovcic, M.; Ivancic-Bace, I.; Bolt, E.L. Cas3 Protein-A Review of a Multi-Tasking Machine. Genes 2020, 11, 208. [Google Scholar] [CrossRef] [PubMed]
- Kunne, T.; Kieper, S.N.; Bannenberg, J.W.; Vogel, A.I.; Miellet, W.R.; Klein, M.; Depken, M.; Suarez-Diez, M.; Brouns, S.J. Cas3-Derived Target DNA Degradation Fragments Fuel Primed CRISPR Adaptation. Mol. Cell 2016, 63, 852–864. [Google Scholar] [CrossRef] [PubMed]
- Dorman, C.J.; Ni Bhriain, N. CRISPR-Cas, DNA Supercoiling, and Nucleoid-Associated Proteins. Trends MicroBiol. 2020, 28, 19–27. [Google Scholar] [CrossRef]
- Yang, C.D.; Chen, Y.H.; Huang, H.Y.; Huang, H.D.; Tseng, C.P. CRP represses the CRISPR/Cas system in Escherichia coli: Evidence that endogenous CRISPR spacers impede phage P1 replication. Mol. MicroBiol. 2014, 92, 1072–1091. [Google Scholar] [CrossRef]
- Westra, E.R.; Pul, U.; Heidrich, N.; Jore, M.M.; Lundgren, M.; Stratmann, T.; Wurm, R.; Raine, A.; Mescher, M.; Van Heereveld, L.; et al. H-NS-mediated repression of CRISPR-based immunity in Escherichia coli K12 can be relieved by the transcription activator LeuO. Mol. MicroBiol. 2010, 77, 1380–1393. [Google Scholar] [CrossRef]
- Fragel, S.M.; Montada, A.; Heermann, R.; Baumann, U.; Schacherl, M.; Schnetz, K. Characterization of the pleiotropic LysR-type transcription regulator LeuO of Escherichia coli. Nucleic. Acids. Res. 2019, 47, 7363–7379. [Google Scholar] [CrossRef]
- Yang, D.; Wang, Z.; Ma, J.; Fu, Q.; Wu, L.; Wang, H.; Wang, S.; Yan, Y.; Sun, J. Glycine Cleavage System and cAMP Receptor Protein Co-Regulate CRISPR/cas3 Expression to Resist Bacteriophage. Viruses 2020, 12, 90. [Google Scholar] [CrossRef] [PubMed]
- Yosef, I.; Goren, M.G.; Kiro, R.; Edgar, R.; Qimron, U. High-temperature protein G is essential for activity of the Escherichia coli clustered regularly interspaced short palindromic repeats (CRISPR)/Cas system. Proc. Natl. Acad. Sci. USA 2011, 108, 20136–20141. [Google Scholar] [CrossRef]
- Majsec, K.; Bolt, E.L.; Ivancic-Bace, I. Cas3 is a limiting factor for CRISPR-Cas immunity in Escherichia coli cells lacking H-NS. BMC MicroBiol. 2016, 16, 28. [Google Scholar] [CrossRef] [PubMed]
- Huo, Y.; Nam, K.H.; Ding, F.; Lee, H.; Wu, L.; Xiao, Y.; Farchione, M.D., Jr.; Zhou, S.; Rajashankar, K.; Kurinov, I.; et al. Structures of CRISPR Cas3 offer mechanistic insights into Cascade-activated DNA unwinding and degradation. Nat. Struct. Mol. Biol. 2014, 21, 771–777. [Google Scholar] [CrossRef] [PubMed]
- Mitic, D.; Radovcic, M.; Markulin, D.; Ivancic-Bace, I. StpA represses CRISPR-Cas immunity in H-NS deficient Escherichia coli. Biochimie 2020, 174, 136–143. [Google Scholar] [CrossRef]
- Mulepati, S.; Bailey, S. Structural and biochemical analysis of nuclease domain of clustered regularly interspaced short palindromic repeat (CRISPR)-associated protein 3 (Cas3). J. Biol. Chem. 2011, 286, 31896–31903. [Google Scholar] [CrossRef]
- Howard, J.A.; Delmas, S.; Ivancic-Bace, I.; Bolt, E.L. Helicase dissociation and annealing of RNA-DNA hybrids by Escherichia coli Cas3 protein. Biochem. J. 2011, 439, 85–95. [Google Scholar] [CrossRef]
- Greenfield, N.J. Using circular dichroism spectra to estimate protein secondary structure. Nat. Protoc. 2006, 1, 2876–2890. [Google Scholar] [CrossRef] [PubMed]
- Whitmore, L.; Wallace, B.A. DICHROWEB, an online server for protein secondary structure analyses from circular dichroism spectroscopic data. Nucleic. Acids. Res. 2004, 32, W668–W673. [Google Scholar] [CrossRef] [PubMed]
- Micsonai, A.; Wien, F.; Bulyaki, E.; Kun, J.; Moussong, E.; Lee, Y.H.; Goto, Y.; Refregiers, M.; Kardos, J. BeStSel: A web server for accurate protein secondary structure prediction and fold recognition from the circular dichroism spectra. Nucleic. Acids. Res. 2018, 46, W315–W322. [Google Scholar] [CrossRef]
- Provencher, S.W.; Glockner, J. Estimation of globular protein secondary structure from circular dichroism. Biochemistry 1981, 20, 33–37. [Google Scholar] [CrossRef]
- Gong, B.; Shin, M.; Sun, J.; Jung, C.H.; Bolt, E.L.; van der Oost, J.; Kim, J.S. Molecular insights into DNA interference by CRISPR-associated nuclease-helicase Cas3. Proc. Natl. Acad. Sci. USA 2014, 111, 16359–16364. [Google Scholar] [CrossRef] [PubMed]
- Waterhouse, A.; Bertoni, M.; Bienert, S.; Studer, G.; Tauriello, G.; Gumienny, R.; Heer, F.T.; de Beer, T.A.P.; Rempfer, C.; Bordoli, L.; et al. SWISS-MODEL: Homology modelling of protein structures and complexes. Nucleic. Acids. Res. 2018, 46, W296–W303. [Google Scholar] [CrossRef]
- Sinkunas, T.; Gasiunas, G.; Fremaux, C.; Barrangou, R.; Horvath, P.; Siksnys, V. Cas3 is a single-stranded DNA nuclease and ATP-dependent helicase in the CRISPR/Cas immune system. EMBO J. 2011, 30, 1335–1342. [Google Scholar] [CrossRef]
- Bird, L.E.; Hakansson, K.; Pan, H.; Wigley, D.B. Characterization and crystallization of the helicase domain of bacteriophage T7 gene 4 protein. Nucleic. Acids. Res. 1997, 25, 2620–2626. [Google Scholar] [CrossRef]
- Swarts, D.C.; Mosterd, C.; van Passel, M.W.; Brouns, S.J. CRISPR Interference Directs Strand Specific Spacer Acquisition. PLoS ONE 2012, 7, e35888. [Google Scholar] [CrossRef]
- Westra, E.R.; van Erp, P.B.; Kunne, T.; Wong, S.P.; Staals, R.H.; Seegers, C.L.; Bollen, S.; Jore, M.M.; Semenova, E.; Severinov, K.; et al. CRISPR Immunity Relies on the Consecutive Binding and Degradation of Negatively Supercoiled Invader DNA by Cascade and Cas3. Mol. Cell 2012, 46, 595–605. [Google Scholar] [CrossRef]
- Kunne, T.; Swarts, D.C.; Brouns, S.J. Planting the seed: Target recognition of short guide RNAs. Trends MicroBiol. 2014, 22, 74–83. [Google Scholar] [CrossRef]
- Staals, R.H.; Jackson, S.A.; Biswas, A.; Brouns, S.J.; Brown, C.M.; Fineran, P.C. Interference-driven spacer acquisition is dominant over naive and primed adaptation in a native CRISPR-Cas system. Nat. Commun. 2016, 7, 12853. [Google Scholar] [CrossRef]
- Sinkunas, T.; Gasiunas, G.; Siksnys, V. (2015) Cas3 nuclease-helicase activity assays. Methods Mol. Biol. 2015, 1311, 277–291. [Google Scholar]
- Killelea, T.; Hawkins, M.; Howard, J.L.; McGlynn, P.; Bolt, E.L. DNA replication roadblocks caused by Cascade interference complexes are alleviated by RecG DNA repair helicase. RNA Biol. 2019, 16, 543–548. [Google Scholar] [CrossRef]
- Hurme, R.; Rhen, M. Temperature sensing in bacterial gene regulation--what it all boils down to. Mol. MicroBiol. 1998, 30, 1–6. [Google Scholar] [CrossRef]
- Macek, B.; Forchhammer, K.; Hardouin, J.; Weber-Ban, E.; Grangeasse, C.; Mijakovic, I. Protein post-translational modifications in bacteria. Nat. Rev. MicroBiol. 2019, 17, 651–664. [Google Scholar] [CrossRef]
- Cameron, P.; Coons, M.M.; Klompe, S.E.; Lied, A.M.; Smith, S.C.; Vidal, B.; Donohoue, P.D.; Rotstein, T.; Kohrs, B.W.; Nyer, D.B.; et al. Harnessing type I CRISPR-Cas systems for genome engineering in human cells. Nat. Biotechnol. 2019, 37, 1471–1477. [Google Scholar] [CrossRef] [PubMed]
- Dolan, A.E.; Hou, Z.; Xiao, Y.; Gramelspacher, M.J.; Heo, J.; Howden, S.E.; Freddolino, P.L.; Ke, A.; Zhang, Y. Introducing a Spectrum of Long-Range Genomic Deletions in Human Embryonic Stem Cells Using Type I CRISPR-Cas. Mol. Cell 2019, 74, 936–950 e935. [Google Scholar] [CrossRef]
- Morisaka, H.; Yoshimi, K.; Okuzaki, Y.; Gee, P.; Kunihiro, Y.; Sonpho, E.; Xu, H.; Sasakawa, N.; Naito, Y.; Nakada, S.; et al. CRISPR-Cas3 induces broad and unidirectional genome editing in human cells. Nat. Commun. 2019, 10, 5302. [Google Scholar] [CrossRef] [PubMed]
- Verhaart, I.E.C.; Aartsma-Rus, A. Therapeutic developments for Duchenne muscular dystrophy. Nat. Rev. Neurol. 2019, 15, 373–386. [Google Scholar] [CrossRef] [PubMed]
- Link, A.J.; Phillips, D.; Church, G.M. Methods for generating precise deletions and insertions in the genome of wild-type Escherichia coli: Application to open reading frame characterization. J. Bacteriol. 1997, 179, 6228–6237. [Google Scholar] [CrossRef] [PubMed]
- Anandakrishnan, R.; Aguilar, B.; Onufriev, A.V. H++ 3.0: Automating pK prediction and the preparation of biomolecular structures for atomistic molecular modeling and simulations. Nucleic. Acids. Res. 2012, 40, W537–W541. [Google Scholar] [CrossRef]
- Vriend, G. WHAT IF: A molecular modeling and drug design program. J. Mol. Graph. 1990, 8, 52–56. [Google Scholar] [CrossRef]
- Case, D.A.; Ben-Shalom, I.Y.; Brozell, S.R.; Cerutti, D.S.; Cheatham, T.E., III; Cruzeiro, V.W.D.; Darden, T.A.; Duke, R.E.; Giambasu, G.; Gilson, M.K.; et al. AMBER 2020; University of California: San Franciso, CA, USA, 2020. [Google Scholar]
- Kekic, T.; Fulgosi, H.; Vojta, L.; Bertosa, B. Molecular basis of ferredoxin:NADP(+) reductase interactions with FNR binding domains from TROL and Tic62 proteins. J. Mol. Struct. 2020, 1215, 128281. [Google Scholar] [CrossRef]
- Hünenberger, P.H.H. Thermostat algorithms for molecular dynamics simulations. Adv. Polym. Sci. 2005, 173, 105–149. [Google Scholar]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD: Visual molecular dynamics. J. Mol. Graph. 1996, 14, 33–38. [Google Scholar] [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera--a visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef] [PubMed]
- Sehnal, D.; Svobodova Varekova, R.; Berka, K.; Pravda, L.; Navratilova, V.; Banas, P.; Ionescu, C.M.; Otyepka, M.; Koca, J. MOLE 2.0: Advanced approach for analysis of biomacromolecular channels. J. Cheminform 2013, 5, 39. [Google Scholar] [CrossRef] [PubMed]
| Secondary Structure | % Composition in Cas3 at 30 °C | % Composition in Cas3 at 35 °C | Change (Δ) |
|---|---|---|---|
| α-Helix | 18.1 | 14.3 | −3.8 |
| β-antiparallel | 11.7 | 14.9 | 3.2 |
| β-parallel | 0.0 | 0.0 | 0 |
| Turn | 20.5 | 20.5 | 0 |
| Other | 49.7 | 50.3 | 0.6 |
| Secondary Structure | % Composition in Cas3W406A at 30 °C | % Composition in Cas3W406A at 35 °C | Change (Δ) |
|---|---|---|---|
| α-helix | 22.1 | 12.8 | −9.3 |
| β-antiparallel | 22.0 | 22.8 | 0.8 |
| β-parallel | 6.5 | 3.7 | −2.8 |
| Turn | 10.3 | 11.7 | 1.4 |
| Other | 39.1 | 49.0 | 9.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, L.; Matošević, Z.J.; Mitić, D.; Markulin, D.; Killelea, T.; Matković, M.; Bertoša, B.; Ivančić-Baće, I.; Bolt, E.L. A Tryptophan ‘Gate’ in the CRISPR-Cas3 Nuclease Controls ssDNA Entry into the Nuclease Site, That When Removed Results in Nuclease Hyperactivity. Int. J. Mol. Sci. 2021, 22, 2848. https://doi.org/10.3390/ijms22062848
He L, Matošević ZJ, Mitić D, Markulin D, Killelea T, Matković M, Bertoša B, Ivančić-Baće I, Bolt EL. A Tryptophan ‘Gate’ in the CRISPR-Cas3 Nuclease Controls ssDNA Entry into the Nuclease Site, That When Removed Results in Nuclease Hyperactivity. International Journal of Molecular Sciences. 2021; 22(6):2848. https://doi.org/10.3390/ijms22062848
Chicago/Turabian StyleHe, Liu, Zoe Jelić Matošević, Damjan Mitić, Dora Markulin, Tom Killelea, Marija Matković, Branimir Bertoša, Ivana Ivančić-Baće, and Edward L. Bolt. 2021. "A Tryptophan ‘Gate’ in the CRISPR-Cas3 Nuclease Controls ssDNA Entry into the Nuclease Site, That When Removed Results in Nuclease Hyperactivity" International Journal of Molecular Sciences 22, no. 6: 2848. https://doi.org/10.3390/ijms22062848
APA StyleHe, L., Matošević, Z. J., Mitić, D., Markulin, D., Killelea, T., Matković, M., Bertoša, B., Ivančić-Baće, I., & Bolt, E. L. (2021). A Tryptophan ‘Gate’ in the CRISPR-Cas3 Nuclease Controls ssDNA Entry into the Nuclease Site, That When Removed Results in Nuclease Hyperactivity. International Journal of Molecular Sciences, 22(6), 2848. https://doi.org/10.3390/ijms22062848