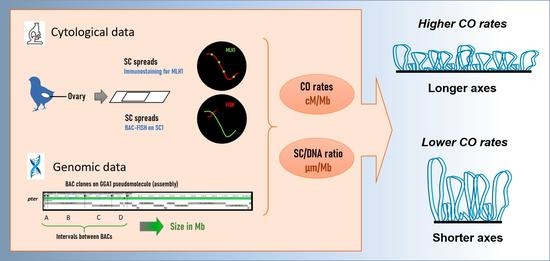
DNA Organization along Pachytene Chromosome Axes and Its Relationship with Crossover Frequencies
Abstract
1. Introduction
2. Results
2.1. DNA Densities in Macro- and MicroSCs
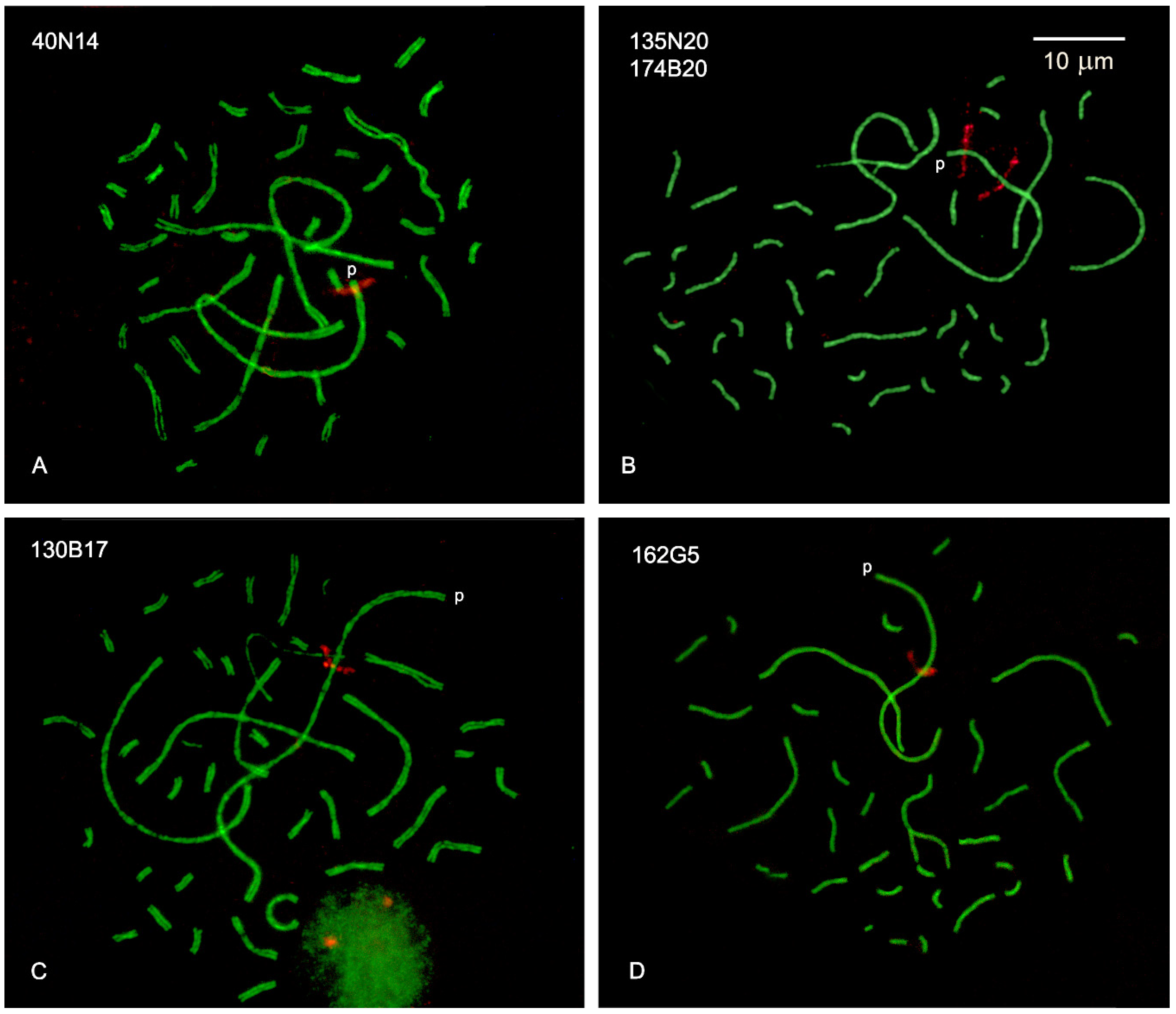
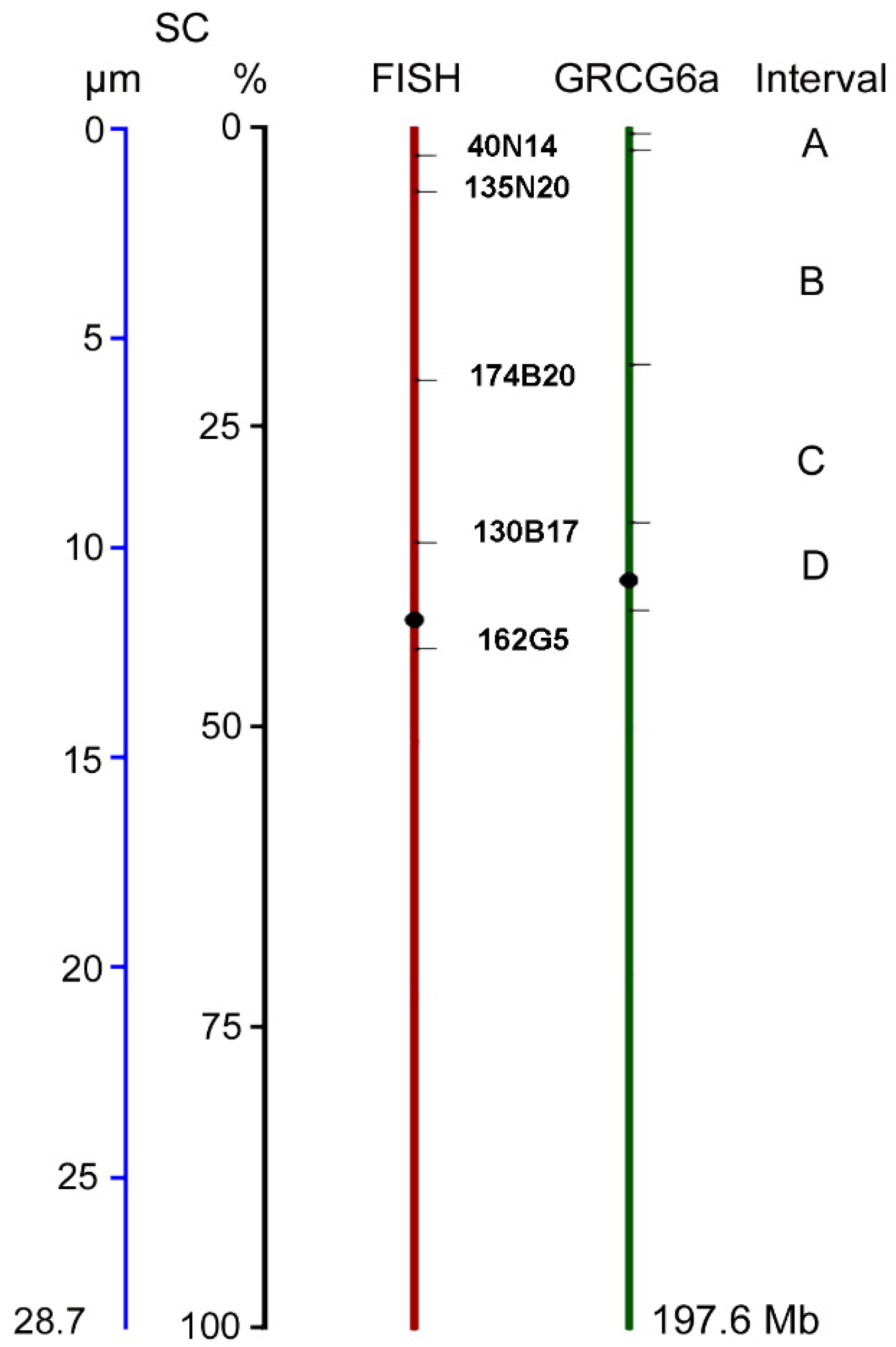
2.2. DNA Density at Specific Intervals of SC1
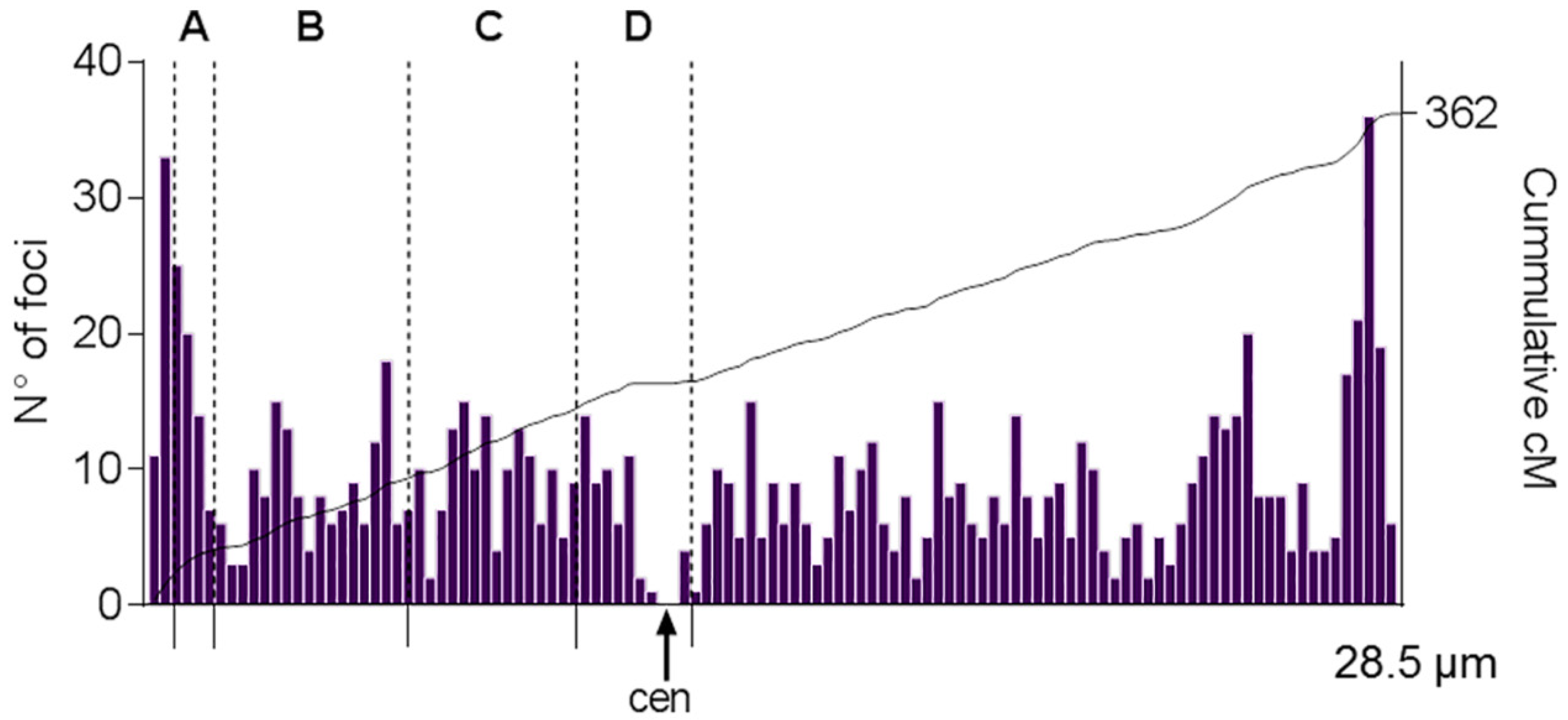
2.3. Crossover Rates and SC/DNA Ratio at Different Intervals on SC1
3. Discussion and Conclusion
4. Materials and Methods
4.1. Biological Material and Synaptonemal Complex Spreads
4.2. Immunostaining and BAC-FISH
4.3. MLH1-Focus Map along SC1
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| SC | Synaptonemal complex |
| CO | Crossover |
| BAC-FISH | Bacterial Artificial Chromosome clones localized by Fluorescent in situ Hybridization |
References
- Page, S.L.; Hawley, R.S. The genetics and molecular biology of the synaptonemal complex. Ann. Rev. Cell Dev. Biol. 2004, 20, 525–558. [Google Scholar] [CrossRef]
- Yang, F.; Wang, P.J. The mammalian synaptonemal complex: A scaffold and beyond. Genome Dyn. 2009, 5, 69–80. [Google Scholar] [PubMed]
- Fraune, J.; Schramm, S.; Alsheimer, M.; Benavente, R. The mammalian synaptonemal complex: Protein components, assembly and role in meiotic recombination. Exp. Cell Res. 2012, 318, 1340–1346. [Google Scholar] [CrossRef] [PubMed]
- Syrjanen, J.L.; Pellegrini, L.; Davies, O.R. A molecular model for the role of SYCP3 in meiotic chromosome organisation. Elife 2014, 3. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Espagne, E.; de Muyt, A.; Zickler, D.; Kleckner, N.E. Interference-mediated synaptonemal complex formation with embedded crossover designation. Proc. Natl. Acad. Sci. USA 2014, 111, E5059–E5068. [Google Scholar] [CrossRef] [PubMed]
- Zickler, D.; Kleckner, N. Recombination, Pairing, and Synapsis of Homologs during Meiosis. Cold Spring Harb. Perspect. Biol. 2015, 7. [Google Scholar] [CrossRef]
- Wang, S.; Hassold, T.; Hunt, P.; White, M.A.; Zickler, D.; Kleckner, N.; Zhang, L. Inefficient Crossover Maturation Underlies Elevated Aneuploidy in Human Female Meiosis. Cell 2017, 168, 977–989.e17. [Google Scholar] [CrossRef]
- Peterson, D.G.; Stack, S.M.; Healy, J.L.; Donohoe, B.S.; Anderson, L.K. The relationship between synaptonemal complex length and genome size in four vertebrate classes (Osteicthyes, Reptilia, Aves, Mammalia). Chromosome Res. 1994, 2, 153–162. [Google Scholar] [CrossRef]
- Anderson, L.K.; Stack, S.M.; Fox, M.H.; Zhang, C.S. The relationship between genome size and synaptonemal complex length in higher plants. Exp. Cell Res. 1985, 156, 367–378. [Google Scholar] [CrossRef]
- Kleckner, N.; Zickler, D.; Jones, G.H.; Dekker, J.; Padmore, R.; Henle, J.; Hutchinson, J. A mechanical basis for chromosome function. Proc. Natl. Acad. Sci. USA 2004, 101, 12592–12597. [Google Scholar] [CrossRef]
- Wang, S.; Liu, Y.; Shang, Y.; Zhai, B.; Yang, X.; Kleckner, N.; Zhang, L. Crossover Interference, Crossover Maturation, and Human Aneuploidy. BioEssays 2019, 41, 1800221. [Google Scholar] [CrossRef] [PubMed]
- Kleckner, N. Chiasma formation: Chromatin/axis interplay and the role(s) of the synaptonemal complex. Chromosoma 2006, 115, 175–194. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Herrera, A.; Vozdova, M.; Fernández, J.; Sebestova, H.; Capilla, L.; Frohlich, J.; Vara, C.; Hernández-Marsal, A.; Sipek, J.; Robinson, T.J.; et al. Recombination correlates with synaptonemal complex length and chromatin loop size in bovids—Insights into mammalian meiotic chromosomal organization. Chromosoma 2017, 126, 615–631. [Google Scholar] [CrossRef] [PubMed]
- Patel, L.; Kang, R.; Rosenberg, S.C.; Qiu, Y.; Raviram, R.; Chee, S.; Hu, R.; Ren, B.; Cole, F.; Corbett, K.D. Dynamic reorganization of the genome shapes the recombination landscape in meiotic prophase. Nat. Struct. Mol. Biol. 2019, 26, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Solari, A.J. Ultrastructure of the synaptic autosomes and the ZW bivalent in chicken oocytes. Chromosoma 1977, 64, 155–165. [Google Scholar] [CrossRef]
- Pigozzi, M.I. The Chromosomes of Birds during Meiosis. Cytogenet. Genome Res. 2016, 150, 128–138. [Google Scholar] [CrossRef]
- Pigozzi, M.I. Relationship between physical and genetic distances along the zebra finch Z chromosome. Chromosome Res. 2008, 16, 839–849. [Google Scholar] [CrossRef]
- Pigozzi, M.I. Localization of single-copy sequences on chicken synaptonemal complex spreads using fluorescence in situ hybridization (FISH). Cytogenet. Genome Res. 2007, 119, 105–112. [Google Scholar] [CrossRef]
- Warren, W.C.; Hillier, L.W.; Tomlinson, C.; Minx, P.; Kremitzki, M.; Graves, T.; Markovic, C.; Bouk, N.; Pruitt, K.D.; Thibaud-Nissen, F.; et al. A New Chicken Genome Assembly Provides Insight into Avian Genome Structure. G3 Genes Genomes Genet. 2017, 7, 109–117. [Google Scholar] [CrossRef]
- Calderón, P.L.; Pigozzi, M.I. MLH1-focus mapping in birds shows equal recombination between sexes and diversity of crossover patterns. Chromosome Res. 2006, 14, 605–612. [Google Scholar] [CrossRef]
- Fröhlich, J.; Vozdova, M.; Kubickova, S.; Cernohorska, H.; Sebestova, H.; Rubes, J. Variation of Meiotic Recombination Rates and MLH1 Foci Distribution in Spermatocytes of Cattle, Sheep and Goats. Cytogenet. Genome Res. 2015, 146, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Basheva, E.A.; Bidau, C.J.; Borodin, P.M. General pattern of meiotic recombination in male dogs estimated by MLH1 and RAD51 immunolocalization. Chromosome Res. 2008, 16, 709–719. [Google Scholar] [CrossRef] [PubMed]
- Froenicke, L.; Anderson, L.K.; Wienberg, J.; Ashley, T. Male Mouse Recombination Maps for Each Autosome Identified by Chromosome Painting. Am. J. Hum. Genet. 2002, 71, 1353–1368. [Google Scholar] [CrossRef] [PubMed]
- Piégu, B.; Arensburger, P.; Guillou, F.; Bigot, Y. But where did the centromeres go in the chicken genome models? Chromosom. Res. 2018, 26, 297–306. [Google Scholar] [CrossRef]
- Stack, S.M. Heterochromatin, the synaptonemal complex and crossing over. J. Cell Sci. 1984, 71, 159–176. [Google Scholar] [PubMed]
- Codina-Pascual, M.; Navarro, J.; Oliver-Bonet, M.; Kraus, J.; Speicher, M.R.; Arango, O.; Egozcue, J.; Benet, J. Behaviour of human heterochromatic regions during the synapsis of homologous chromosomes. Hum. Reprod. 2006, 21, 1490–1497. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Stack, S.M.; Shearer, L.A.; Lohmiller, L.; Anderson, L.K. Meiotic Crossing Over in Maize Knob Heterochromatin. Genetics 2017, 205, 1101–1112. [Google Scholar] [CrossRef]
- Shearer, L.A.; Anderson, L.K.; De Jong, H.; Smit, S.; Goicoechea, J.L.; Roe, B.A.; Hua, A.; Giovannoni, J.J.; Stack, S.M. FluorescenceIn SituHybridization and Optical Mapping to Correct Scaffold Arrangement in the Tomato Genome. G3 Genes Genomes Genet. 2014, 4, 1395–1405. [Google Scholar] [CrossRef]
- Kleckner, N.; Storlazzi, A.; Zickler, D. Coordinate variation in meiotic pachytene SC length and total crossover/chiasma frequency under conditions of constant DNA length. Trends Genet. 2003, 19, 623–628. [Google Scholar] [CrossRef]
- Ellegren, H. The Evolutionary Genomics of Birds. Annu. Rev. Ecol. Evol. Syst. 2013, 44, 239–259. [Google Scholar] [CrossRef]
- Stock, A.; Arrighi, F.; Stefos, K. Chromosome homology in birds: Banding patterns of the chromosomes of the domestic chicken, ring-necked dove, and domestic pigeon. Cytogenet. Genome Res. 1974, 13, 410–418. [Google Scholar] [CrossRef]
- Carlenius, C.; Ryttman, H.; Tegelström, H.; Jasson, H. R-, G- and C-banded chromosomes in the domestic fowl (Gallus domesticus). Hereditas 1981, 94, 5. [Google Scholar] [CrossRef]
- Solari, A.J. Synaptosomal complexes and associated structures in microspread human spermatocytes. Chromosom 1980, 81, 315–337. [Google Scholar] [CrossRef] [PubMed]
- Codina-Pascual, M.; Kraus, J.; Speicher, M.R.; Oliver-Bonet, M.; Murcia, V.; Sarquella, J.; Egozcue, S.; Navarro, J.; Benet, J. Characterization of all human male synaptonemal complexes by subtelomere multiplex-FISH. Cytogenet. Genome Res. 2004, 107, 18–21. [Google Scholar] [CrossRef] [PubMed]
- Heng, H.H.; Chamberlain, J.W.; Shi, X.M.; Spyropoulos, B.; Tsui, L.C.; Moens, P.B. Regulation of meiotic chromatin loop size by chromosomal position. Proc. Natl. Acad. Sci. USA 1996, 93, 2795–2800. [Google Scholar] [CrossRef]
- Møens, P.B.; Pearlman, R.E. Chromatin organization at meiosis. BioEssays 1988, 9, 151–153. [Google Scholar] [CrossRef] [PubMed]
- Zickler, D.; Kleckner, N. Meiotic Chromosomes: Integrating Structure and Function. Annu. Rev. Genet. 1999, 33, 603–754. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Liang, Z.; Hutchinson, J.; Kleckner, N. Crossover Patterning by the Beam-Film Model: Analysis and Implications. PLoS Genet. 2014, 10, e1004042. [Google Scholar] [CrossRef] [PubMed]
- Cahoon, C.K.; Hawley, R.S. Regulating the construction and demolition of the synaptonemal complex. Nat. Struct. Mol. Biol. 2016, 23, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Fraune, J.; Brochier-Armanet, C.; Alsheimer, M.; Volff, J.-N.; Schücker, K.; Benavente, R. Evolutionary history of the mammalian synaptonemal complex. Chromosom 2016, 125, 355–360. [Google Scholar] [CrossRef]
- Zheng, Y.H.; Rengaraj, D.; Choi, J.W.; Park, K.J.; Lee, S.I.; Han, J.Y. Expression pattern of meiosis associated SYCP family members during germline development in chickens. Reproduction 2009, 138, 483–492. [Google Scholar] [CrossRef]
- Sciurano, R.B.; Pigozzi, M.I.; Benavente, R. Disassembly of the synaptonemal complex in chicken oocytes analyzed by super-resolution microscopy. Chromosom 2019, 128, 443–451. [Google Scholar] [CrossRef]
- Rahn, M.I.; Solari, A.J. Recombination nodules in the oocytes of the chicken, Gallus domesticus. Cytogenet. Cell Genet. 1986, 43, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Codina-Pascual, M.; Campillo, M.; Kraus, J.; Speicher, M.; Egozcue, J.; Navarro, J.; Benet, J. Crossover frequency and synaptonemal complex length: Their variability and effects on human male meiosis. Mol. Hum. Reprod. 2006, 12, 123–133. [Google Scholar] [CrossRef] [PubMed]
- McQueen, H.A.; Siriaco, G.; Bird, A. Chicken Microchromosomes Are Hyperacetylated, Early Replicating, and Gene Rich. Genome Res. 1998, 8, 621–630. [Google Scholar] [CrossRef] [PubMed]
- Auer, H.; Mayr, B.; Lambrou, M.; Schleger, W. An extended chicken karyotype, including the NOR chromosome. Cytogenet. Genome Res. 1987, 45, 218–221. [Google Scholar] [CrossRef] [PubMed]
- International Chicken Genome Sequencing Consortium. Sequence and comparative analysis of the chicken genome provide unique perspectives on vertebrate evolution. Nat. Cell Biol. 2004, 432, 695–716. [Google Scholar] [CrossRef]
- Saccone, S.; Cacciò, S.; Perani, P.; Andreozzi, L.; Rapisarda, A.; Motta, S.; Bernardi, G. Compositional Mapping of Mouse Chromosomes and Identification of the Gene-Rich Regions. Chromosom. Res. 1997, 5, 293–300. [Google Scholar] [CrossRef]
- Jabbari, K.; Wirtz, J.; Rauscher, M.; Wiehe, T. A common genomic code for chromatin architecture and recombination landscape. PLoS ONE 2019, 14, e0213278. [Google Scholar] [CrossRef]
- Baier, B.; Hunt, P.; Broman, K.W.; Hassold, T. Variation in Genome-Wide Levels of Meiotic Recombination Is Established at the Onset of Prophase in Mammalian Males. PLoS Genet. 2014, 10, e1004125. [Google Scholar] [CrossRef]
- Pigozzi, M.I.; Solari, A.J. Recombination nodule mapping and chiasma distribution in spermatocytes of the pigeon, Columba livia. Genome 1999, 42, 308–314. [Google Scholar] [PubMed]
- Borodin, P.M.; Karamysheva, T.V.; Belonogova, N.M.; Torgasheva, A.A.; Rubtsov, N.B.; Searle, J.B. Recombination map of the common shrew, Sorex araneus (Eulipotyphla, Mammalia). Genetics 2008, 178, 621–632. [Google Scholar]
- Reeves, A. MicroMeasure: A new computer program for the collection and analysis of cytogenetic data. Genome 2001, 44, 439–443. [Google Scholar] [CrossRef]
- Del Priore, L.; Pigozzi, M.I. Sex-specific recombination maps for individual macrochromosomes in the Japanese quail (Coturnix japonica). Chromosome Res. Int. J. Mol. Supramol. Evol. Asp. Chromosome Biol. 2015, 23, 199–210. [Google Scholar] [CrossRef] [PubMed]
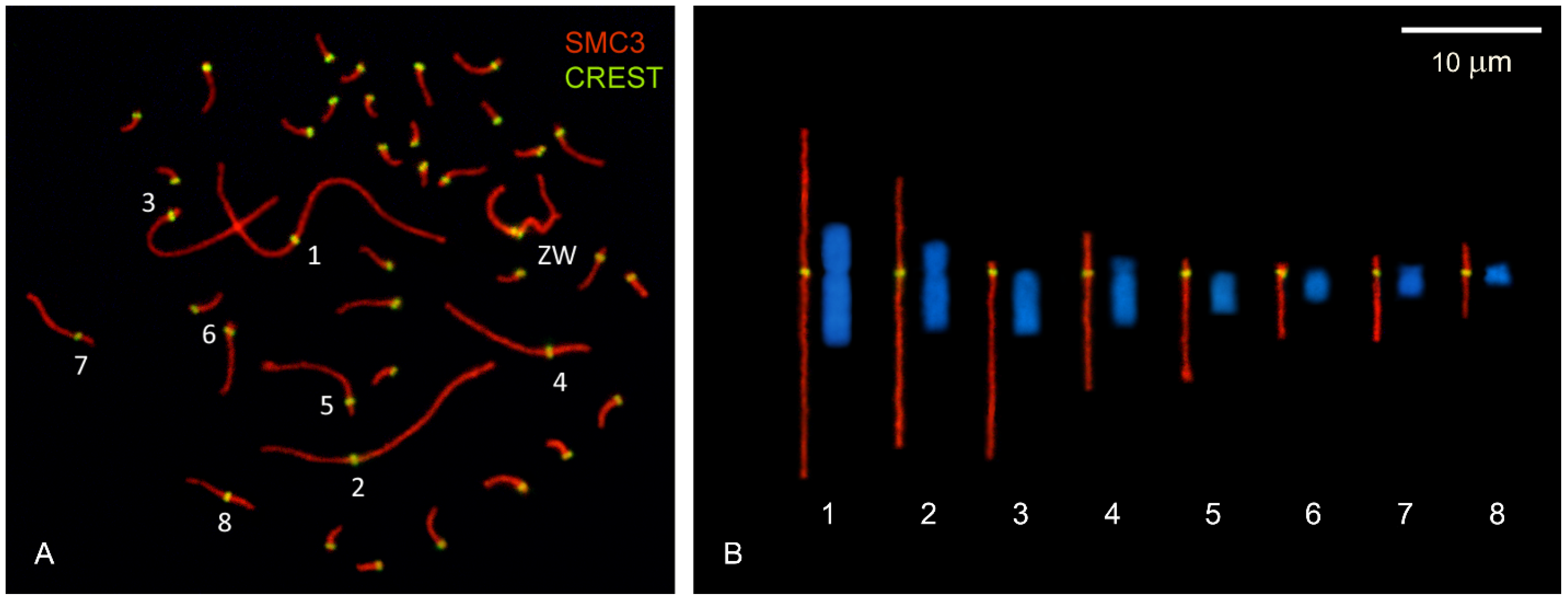
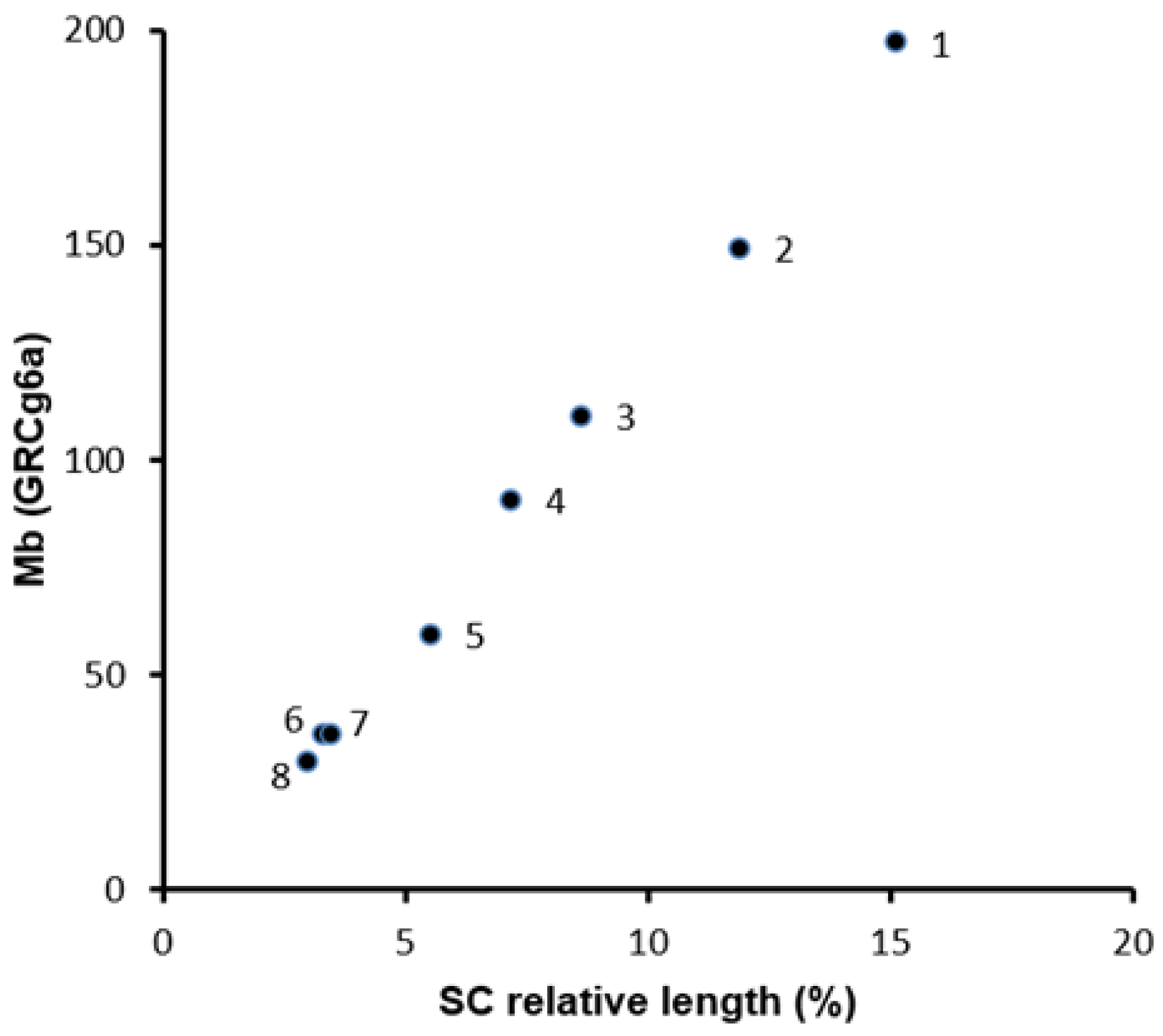
| Chr Size (Mb) a | SC Spread Parameters | DNA Density | |||
|---|---|---|---|---|---|
| Chr | GRCg6a | µm | RL (%) | CI | Mb/µm |
| 1 | 197.7 | 28.7 | 15.3 | 0.41 | 6.9 |
| 2 | 149.7 | 22.2 | 11.8 | 0.35 | 6.7 |
| 3 | 110.8 | 16.2 | 8.6 | 0.04 | 6.8 |
| 4 | 91.3 | 13.5 | 7.1 | 0.24 | 6.8 |
| 5 | 59.8 | 10.3 | 5.4 | 0.09 | 5.7 |
| 6 | 36.4 | 6.1 | 3.3 | 0.08 | 6.0 |
| 7 | 36.7 | 6.5 | 3.5 | 0.24 | 5.6 |
| 8 | 30.2 | 5.6 | 3.0 | 0.38 | 5.4 |
| 9–38 | 436 | 78.5 b | 42 | - | 5.5 |
| Total | 1148 c | 187.6 | - | - | 6.1 |
| GRCg6a | BAC-FISH c | |||||
|---|---|---|---|---|---|---|
| BAC ID | bp a | % b | % | SD | µm | SD |
| 40N14 | 1,274,288 | 0.6 | 2.5 | 0.3 | 0.7 | 0.1 |
| 135N20 | 4,069,830 | 2.0 | 5.5 | 0.5 | 1.6 | 0.1 |
| 174B20 | 39,215,363 | 19.8 | 21.1 | 0.3 | 6.0 | 0.2 |
| 130B17 | 64,984,575 | 32.8 | 34.4 | 1.0 | 9.8 | 0.3 |
| 162G5 | 79,454,082 | 40.2 | 43.4 | 0.8 | 12.3 | 0.3 |
| Interval a | SC Length (µm) | Size (Mb) | SC/DNA Ratio | cM | CO Rate (cM/Mb) |
|---|---|---|---|---|---|
| A | 0.9 | 2.8 | 0.31 | 29 | 10.3 |
| B | 4.4 | 35.1 | 0.13 | 57 | 1.6 |
| C | 3.9 | 25.8 | 0.15 | 52 | 2.0 |
| D | 2.5 | 14.5 | 0.17 | 21 | 1.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
del Priore, L.; Pigozzi, M.I. DNA Organization along Pachytene Chromosome Axes and Its Relationship with Crossover Frequencies. Int. J. Mol. Sci. 2021, 22, 2414. https://doi.org/10.3390/ijms22052414
del Priore L, Pigozzi MI. DNA Organization along Pachytene Chromosome Axes and Its Relationship with Crossover Frequencies. International Journal of Molecular Sciences. 2021; 22(5):2414. https://doi.org/10.3390/ijms22052414
Chicago/Turabian Styledel Priore, Lucía, and María Inés Pigozzi. 2021. "DNA Organization along Pachytene Chromosome Axes and Its Relationship with Crossover Frequencies" International Journal of Molecular Sciences 22, no. 5: 2414. https://doi.org/10.3390/ijms22052414
APA Styledel Priore, L., & Pigozzi, M. I. (2021). DNA Organization along Pachytene Chromosome Axes and Its Relationship with Crossover Frequencies. International Journal of Molecular Sciences, 22(5), 2414. https://doi.org/10.3390/ijms22052414