Increased Expression of Ephrins on Immune Cells of Patients with Relapsing Remitting Multiple Sclerosis Affects Oligodendrocyte Differentiation
Abstract
1. Introduction
2. Results
2.1. The Percentages of Ephrin-A2 and Ephrin-A3-Positive Cells Are Elevated on Immune Cells and Mostly on T Cells of Patients with Relapsing-Remitting Multiple Sclerosis (RR-MS)
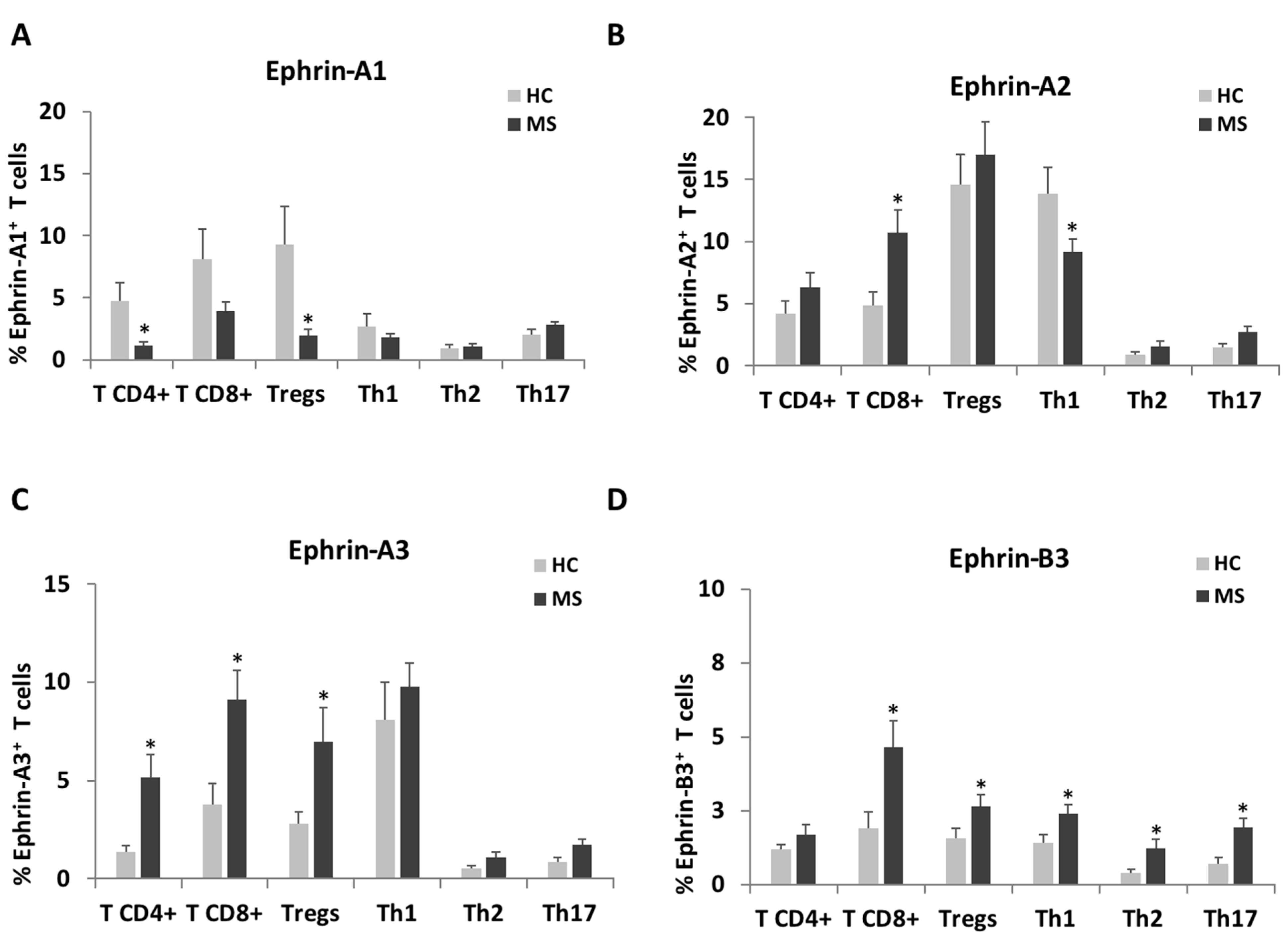
2.2. The Expression of Ephrins-A1, -A2, -A3 and -B3 on T Cell Subpopulations
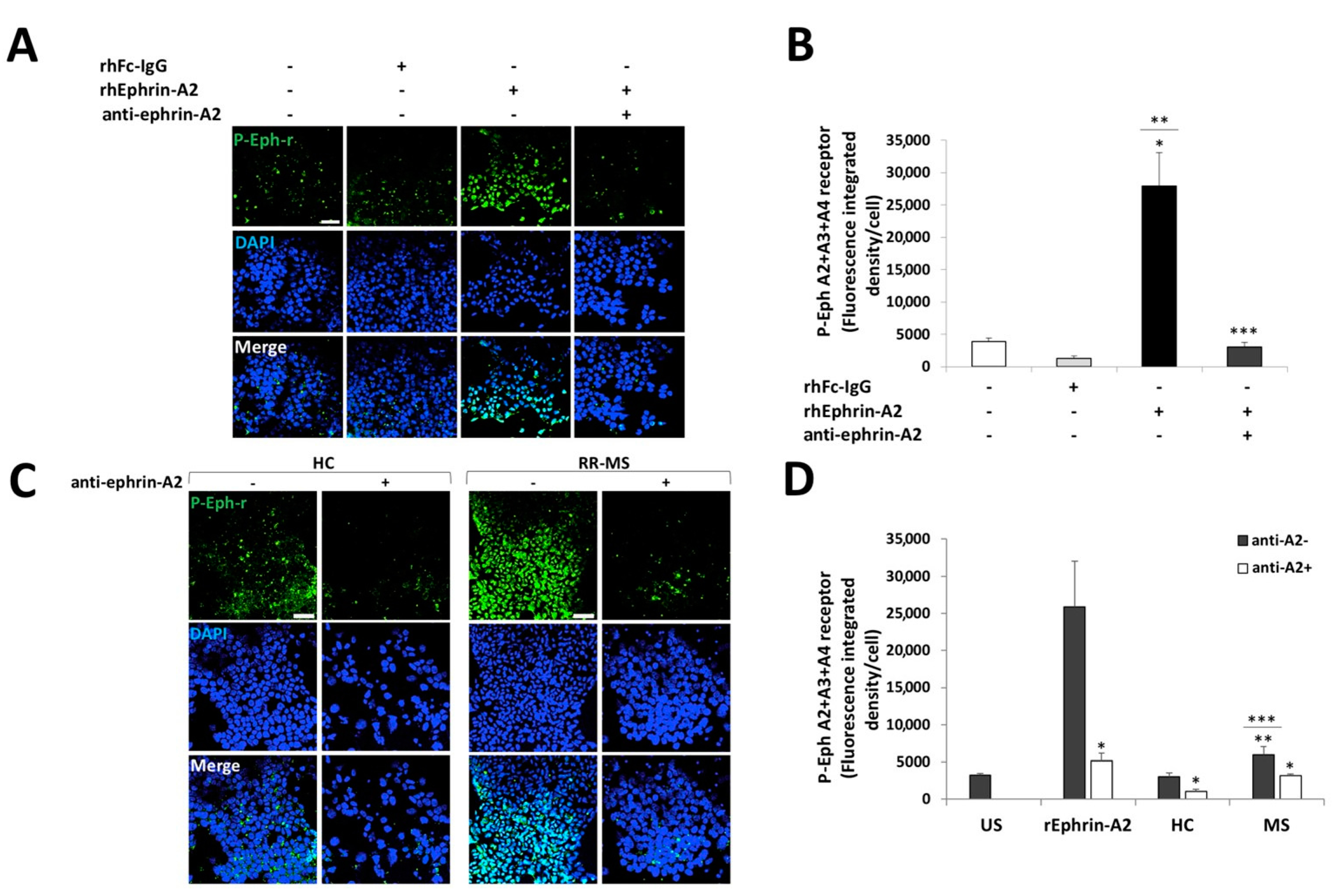
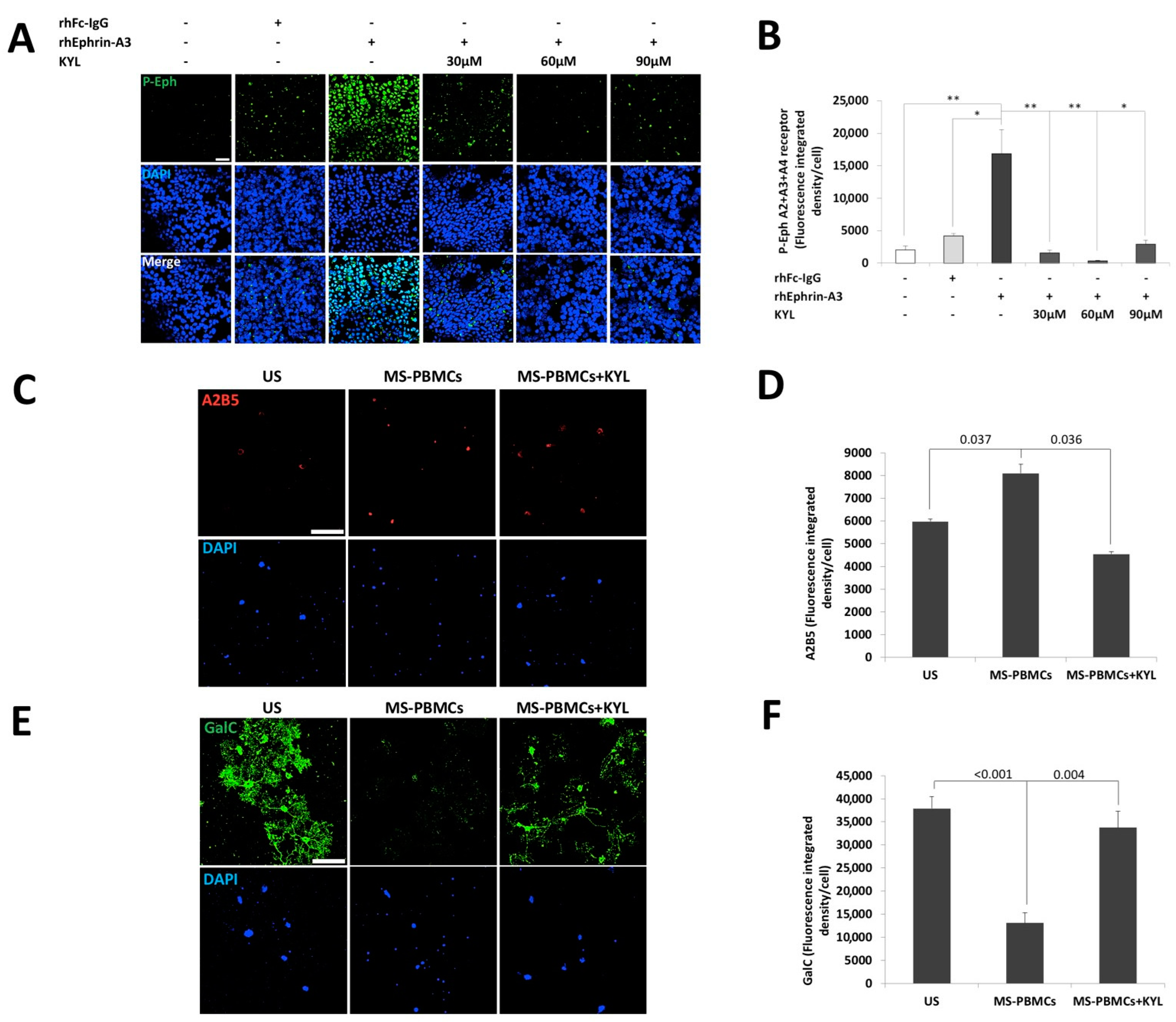
2.3. Ephrins Forward Signaling Is Enhanced When Stimulated with Immune Cells from Patients with RR-MS Compared to Healthy Controls
2.4. Immune Cells of Patients with RR-MS Inhibit the Differentiation of Oligodendrocyte Precursor Cells towards Mature Oligodendrocytes
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Cell Collection and Culture
4.3. Flow Cytometry
4.4. Ephrin Phosphorylation In-Vitro Assay
4.5. Oligodendrocyte Precursor Cells Differentiation
4.6. Immunofluorescence Staining and Confocal Analysis
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Prat, A.; Antel, J. Pathogenesis of multiple sclerosis. Curr. Opin. Neurol. 2005, 18, 225–230. [Google Scholar] [CrossRef]
- Lucchinetti, C.F.; Popescu, B.F.G.; Bunyan, R.F.; Moll, N.M.; Roemer, S.F.; Lassmann, H.; Brück, W.; Parisi, J.E.; Scheithauer, B.W.; Giannini, C.; et al. Inflammatory cortical demyelination in early multiple sclerosis. N. Engl. J. Med. 2011, 365, 2188–2197. [Google Scholar] [CrossRef]
- Lassmann, H. Pathogenic Mechanisms Associated With Different Clinical Courses of Multiple Sclerosis. Front. Immunol. 2018, 9, 3116. [Google Scholar] [CrossRef] [PubMed]
- Dendrou, C.A.; Fugger, L.; Friese, M.A. Immunopathology of multiple sclerosis. Nat. Rev. Immunol. 2015, 15, 545–558. [Google Scholar] [CrossRef] [PubMed]
- Wolswijk, G. Chronic stage multiple sclerosis lesions contain a relatively quiescent population of oligodendrocyte precursor cells. J. Neurosci. 1998, 18, 601–609. [Google Scholar] [CrossRef]
- Lucchinetti, C.; Bruck, W.; Parisi, J.; Scheithauer, B.; Rodriguez, M.; Lassmann, H. A quantitative analysis of oligodendrocytes in multiple sclerosis lesions. A study of 113 cases. Brain 1999, 122, 2279–2295. [Google Scholar] [CrossRef] [PubMed]
- Maeda, Y.; Solanky, M.; Menonna, J.; Chapin, J.; Li, W.; Dowling, P. Platelet-derived growth factor-alpha receptor-positive oligodendroglia are frequent in multiple sclerosis lesions. Ann. Neurol. 2001, 49, 776–785. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.; Tourtellotte, W.W.; Rudick, R.; Trapp, B.D. Premyelinating oligodendrocytes in chronic lesions of multiple sclerosis. N. Engl. J. Med. 2002, 346, 165–173. [Google Scholar] [CrossRef]
- Kuhlmann, T.; Miron, V.; Cui, Q.; Wegner, C.; Antel, J.; Brück, W. Differentiation block of oligodendroglial progenitor cells as a cause for remyelination failure in chronic multiple sclerosis. Brain 2008, 131, 1749–1758. [Google Scholar] [CrossRef] [PubMed]
- Nait-Oumesmar, B.; Picard-Riera, N.; Kerninon, C.; Decker, L.; Seilhean, D.; Höglinger, G.U.; Hirsch, E.C.; Reynolds, R.; Baron-Van Evercooren, A. Activation of the subventricular zone in multiple sclerosis: Evidence for early glial progenitors. Proc. Natl. Acad. Sci. USA 2007, 104, 4694–4699. [Google Scholar] [CrossRef]
- Nait-Oumesmar, B.; Decker, L.; Lachapelle, F.; Avellana-Adalid, V.; Bachelin, C.; Baron-Van Evercooren, A. Progenitor cells of the adult mouse subventricular zone proliferate, migrate and differentiate into oligodendrocytes after demyelination. Eur. J. Neurosci. 1999, 11, 4357–4366. [Google Scholar] [CrossRef] [PubMed]
- Snethen, H.; Love, S.; Scolding, N. Disease-responsive neural precursor cells are present in multiple sclerosis lesions. Regen. Med. 2008, 3, 835–847. [Google Scholar] [CrossRef] [PubMed]
- Prineas, J.W.; Connell, F. Remyelination in multiple sclerosis. Ann. Neurol. 1979, 5, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Raine, C.S.; Wu, E. Multiple sclerosis: Remyelination in acute lesions. J. Neuropathol. Exp. Neurol. 1993, 52, 199–204. [Google Scholar] [CrossRef]
- Patrikios, P.; Stadelmann, C.; Kutzelnigg, A.; Rauschka, H.; Schmidbauer, M.; Laursen, H.; Sorensen, P.S.; Brück, W.; Lucchinetti, C.; Lassmann, H. Remyelination is extensive in a subset of multiple sclerosis patients. Brain 2006, 129, 3165–3172. [Google Scholar] [CrossRef]
- Patani, R.; Balaratnam, M.; Vora, A.; Reynolds, R. Remyelination can be extensive in multiple sclerosis despite a long disease course. Neuropathol. Appl. Neurobiol. 2007, 33, 277–287. [Google Scholar] [CrossRef]
- Kotter, M.R.; Li, W.-W.; Zhao, C.; Franklin, R.J.M. Myelin impairs CNS remyelination by inhibiting oligodendrocyte precursor cell differentiation. J. Neurosci. 2006, 26, 328–332. [Google Scholar] [CrossRef]
- Miller, R.H.; Mi, S. Dissecting demyelination. Nat. Neurosci. 2007, 10, 1351–1354. [Google Scholar] [CrossRef] [PubMed]
- Piaton, G.; Aigrot, M.-S.; Williams, A.; Moyon, S.; Tepavcevic, V.; Moutkine, I.; Gras, J.; Matho, K.S.; Schmitt, A.; Soellner, H.; et al. Class 3 semaphorins influence oligodendrocyte precursor recruitment and remyelination in adult central nervous system. Brain 2011, 134, 1156–1167. [Google Scholar] [CrossRef]
- Frohman, E.M.; Racke, M.K.; Raine, C.S. Multiple sclerosis--the plaque and its pathogenesis. N. Engl. J. Med. 2006, 354, 942–955. [Google Scholar] [CrossRef]
- Lucchinetti, C.F.; Brück, W.; Rodriguez, M.; Lassmann, H. Distinct patterns of multiple sclerosis pathology indicates heterogeneity on pathogenesis. Brain Pathol. 1996, 6, 259–274. [Google Scholar] [CrossRef] [PubMed]
- Correale, J.; Farez, M.F. The Role of Astrocytes in Multiple Sclerosis Progression. Front. Neurol. 2015, 6, 180. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Liang, C.; Yang, K.Y.; Huang, X.; Han, M.Y.; Li, X.; Chan, V.W.; Chan, K.S.; Liu, D.; Huang, Z.-P.; et al. Specific ablation of CD4(+) T-cells promotes heart regeneration in juvenile mice. Theranostics 2020, 10, 8018–8035. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yang, K.Y.; Tam, R.C.Y.; Chan, V.W.; Lan, H.Y.; Hori, S.; Zhou, B.; Lui, K.O. Regulatory T-cells regulate neonatal heart regeneration by potentiating cardiomyocyte proliferation in a paracrine manner. Theranostics 2019, 9, 4324–4341. [Google Scholar] [CrossRef]
- Liang, C.; Yang, K.Y.; Chan, V.W.; Li, X.; Fung, T.H.W.; Wu, Y.; Tian, X.Y.; Huang, Y.; Qin, L.; Lau, J.Y.W.; et al. CD8(+) T-cell plasticity regulates vascular regeneration in type-2 diabetes. Theranostics 2020, 10, 4217–4232. [Google Scholar] [CrossRef]
- Davies, C.L.; Miron, V.E. Distinct origins, gene expression and function of microglia and monocyte-derived macrophages in CNS myelin injury and regeneration. Clin. Immunol. 2018, 189, 57–62. [Google Scholar] [CrossRef]
- Lloyd, A.F.; Miron, V.E. Cellular and Molecular Mechanisms Underpinning Macrophage Activation during Remyelination. Front. Cell Dev. Biol. 2016, 4, 60. [Google Scholar] [CrossRef]
- Miron, V.E.; Franklin, R.J.M. Macrophages and CNS remyelination. J. Neurochem. 2014, 130, 165–171. [Google Scholar] [CrossRef]
- Moore, C.S.; Cui, Q.-L.; Warsi, N.M.; Durafourt, B.A.; Zorko, N.; Owen, D.R.; Antel, J.P.; Bar-Or, A. Direct and indirect effects of immune and central nervous system-resident cells on human oligodendrocyte progenitor cell differentiation. J. Immunol. 2015, 194, 761–772. [Google Scholar] [CrossRef]
- Baxi, E.G.; DeBruin, J.; Tosi, D.M.; Grishkan, I.V.; Smith, M.D.; Kirby, L.A.; Strasburger, H.J.; Fairchild, A.N.; Calabresi, P.A.; Gocke, A.R. Transfer of myelin-reactive th17 cells impairs endogenous remyelination in the central nervous system of cuprizone-fed mice. J. Neurosci. 2015, 35, 8626–8639. [Google Scholar] [CrossRef]
- Bieber, A.J.; Kerr, S.; Rodriguez, M. Efficient central nervous system remyelination requires T cells. Ann. Neurol. 2003, 53, 680–684. [Google Scholar] [CrossRef]
- Dombrowski, Y.; O’Hagan, T.; Dittmer, M.; Penalva, R.; Mayoral, S.R.; Bankhead, P.; Fleville, S.; Eleftheriadis, G.; Zhao, C.; Naughton, M.; et al. Regulatory T cells promote myelin regeneration in the central nervous system. Nat. Neurosci. 2017, 20, 674–680. [Google Scholar] [CrossRef]
- de la Vega Gallardo, N.; Dittmer, M.; Dombrowski, Y.; Fitzgerald, D.C. Regenerating CNS myelin: Emerging roles of regulatory T cells and CCN proteins. Neurochem. Int. 2019, 130, 104349. [Google Scholar] [CrossRef]
- McIntyre, L.L.; Greilach, S.A.; Othy, S.; Sears-Kraxberger, I.; Wi, B.; Ayala-Angulo, J.; Vu, E.; Pham, Q.; Silva, J.; Dang, K.; et al. Regulatory T cells promote remyelination in the murine experimental autoimmune encephalomyelitis model of multiple sclerosis following human neural stem cell transplant. Neurobiol. Dis. 2020, 140, 104868. [Google Scholar] [CrossRef] [PubMed]
- Lassmann, H. Multiple sclerosis: Lessons from molecular neuropathology. Exp. Neurol. 2014, 262, 2–7. [Google Scholar] [CrossRef]
- Mizuno, T.; Zhang, G.; Takeuchi, H.; Kawanokuchi, J.; Wang, J.; Sonobe, Y.; Jin, S.; Takada, N.; Komatsu, Y.; Suzumura, A. Interferon-gamma directly induces neurotoxicity through a neuron specific, calcium-permeable complex of IFN-gamma receptor and AMPA GluR1 receptor. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2008, 22, 1797–1806. [Google Scholar] [CrossRef]
- Neumann, H.; Medana, I.M.; Bauer, J.; Lassmann, H. Cytotoxic T lymphocytes in autoimmune and degenerative CNS diseases. Trends Neurosci. 2002, 25, 313–319. [Google Scholar] [CrossRef]
- Starost, L.; Lindner, M.; Herold, M.; Xu, Y.K.T.; Drexler, H.C.A.; Heß, K.; Ehrlich, M.; Ottoboni, L.; Ruffini, F.; Stehling, M.; et al. Extrinsic immune cell-derived, but not intrinsic oligodendroglial factors contribute to oligodendroglial differentiation block in multiple sclerosis. Acta Neuropathol. 2020, 140, 715–736. [Google Scholar] [CrossRef] [PubMed]
- Chew, L.-J.; King, W.C.; Kennedy, A.; Gallo, V. Interferon-gamma inhibits cell cycle exit in differentiating oligodendrocyte progenitor cells. Glia 2005, 52, 127–143. [Google Scholar] [CrossRef] [PubMed]
- Mausner-Fainberg, K.; Urshansky, N.; Regev, K.; Auriel, E.; Karni, A. Elevated and dysregulated bone morphogenic proteins in immune cells of patients with relapsing-remitting multiple sclerosis. J. Neuroimmunol. 2013, 264, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Urshansky, N.; Mausner-Fainberg, K.; Auriel, E.; Regev, K.; Bornstein, N.M.; Karni, A. Reduced production of noggin by immune cells of patients with relapsing-remitting multiple sclerosis. J. Neuroimmunol. 2011, 232, 171–178. [Google Scholar] [CrossRef]
- Urshansky, N.; Mausner-Fainberg, K.; Auriel, E.; Regev, K.; Karni, A. Low and dysregulated production of follistatin in immune cells of relapsing-remitting multiple sclerosis patients. J. Neuroimmunol. 2011, 238, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Mausner-Fainberg, K.; Kolb, H.; Penn, M.; Regev, K.; Vaknin-Dembinsky, A.; Gadoth, A.; Kestenbaum, M.; Karni, A. Differential screening-selected gene aberrative in neuroblastoma (DAN) is increased in the CSF of patients with MS and may be induced by therapy with interferon-β. J. Neuroimmunol. 2016, 292, 93–96. [Google Scholar] [CrossRef] [PubMed]
- Mausner-Fainberg, K.; Penn, M.; Golan, M.; Benhamou, M.; Wilf-Yarkoni, A.; Gertel, S.; Karni, A. Reduced levels of Coco in sera of multiple sclerosis patients: A potential role in neuro-regeneration failure. J. Neuroimmunol. 2019, 327, 36–40. [Google Scholar] [CrossRef]
- Coulthard, M.G.; Morgan, M.; Woodruff, T.M.; Arumugam, T.V.; Taylor, S.M.; Carpenter, T.C.; Lackmann, M.; Boyd, A.W. Eph/Ephrin signaling in injury and inflammation. Am. J. Pathol. 2012, 181, 1493–1503. [Google Scholar] [CrossRef]
- Pasquale, E.B. Eph-ephrin promiscuity is now crystal clear. Nat. Neurosci. 2004, 7, 417–418. [Google Scholar] [CrossRef] [PubMed]
- Murai, K.K.; Pasquale, E.B. ’Eph’ective signaling: Forward, reverse and crosstalk. J. Cell Sci. 2003, 116, 2823–2832. [Google Scholar] [CrossRef] [PubMed]
- Goldshmit, Y.; McLenachan, S.; Turnley, A. Roles of Eph receptors and ephrins in the normal and damaged adult CNS. Brain Res. Rev. 2006, 52, 327–345. [Google Scholar] [CrossRef]
- Cramer, K.S.; Miko, I.J. Eph-ephrin signaling in nervous system development. F1000Research 2016, 5. [Google Scholar] [CrossRef]
- Wan, Y.; Yang, J.-S.; Xu, L.-C.; Huang, X.-J.; Wang, W.; Xie, M.-J. Roles of Eph/ephrin bidirectional signaling during injury and recovery of the central nervous system. Neural Regen. Res. 2018, 13, 1313–1321. [Google Scholar] [CrossRef] [PubMed]
- Munro, K.M.; Dixon, K.J.; Gresle, M.M.; Jonas, A.; Kemper, D.; Doherty, W.; Fabri, L.J.; Owczarek, C.M.; Pearse, M.; Boyd, A.W.; et al. EphA4 receptor tyrosine kinase is a modulator of onset and disease severity of experimental autoimmune encephalomyelitis (EAE). PLoS ONE 2013, 8, e55948. [Google Scholar] [CrossRef]
- Luo, H.; Broux, B.; Wang, X.; Hu, Y.; Ghannam, S.; Jin, W.; Larochelle, C.; Prat, A.; Wu, J. EphrinB1 and EphrinB2 regulate T cell chemotaxis and migration in experimental autoimmune encephalomyelitis and multiple sclerosis. Neurobiol. Dis. 2016, 91, 292–306. [Google Scholar] [CrossRef] [PubMed]
- Noberini, R.; Koolpe, M.; Peddibhotla, S.; Dahl, R.; Su, Y.; Cosford, N.D.P.; Roth, G.P.; Pasquale, E.B. Small molecules can selectively inhibit ephrin binding to the EphA4 and EphA2 receptors. J. Biol. Chem. 2008, 283, 29461–29472. [Google Scholar] [CrossRef] [PubMed]
- Harboe, M.; Torvund-Jensen, J.; Kjaer-Sorensen, K.; Laursen, L.S. Ephrin-A1-EphA4 signaling negatively regulates myelination in the central nervous system. Glia 2018, 66, 934–950. [Google Scholar] [CrossRef] [PubMed]
- Holmberg, J.; Armulik, A.; Senti, K.-A.; Edoff, K.; Spalding, K.; Momma, S.; Cassidy, R.; Flanagan, J.G.; Frisen, J. Ephrin-A2 reverse signaling negatively regulates neural progenitor proliferation and neurogenesis. Genes Dev. 2005, 19, 462–471. [Google Scholar] [CrossRef]
- Jagielska, A.; Lowe, A.L.; Makhija, E.; Wroblewska, L.; Guck, J.; Franklin, R.J.M.; Shivashankar, G.V.; Van Vliet, K.J. Mechanical Strain Promotes Oligodendrocyte Differentiation by Global Changes of Gene Expression. Front. Cell. Neurosci. 2017, 11, 93. [Google Scholar] [CrossRef] [PubMed]
- Linneberg, C.; Harboe, M.; Laursen, L.S. Axo-Glia Interaction Preceding CNS Myelination Is Regulated by Bidirectional Eph-Ephrin Signaling. ASN Neuro 2015, 7. [Google Scholar] [CrossRef]
- Syed, Y.A.; Zhao, C.; Mahad, D.; Mobius, W.; Altmann, F.; Foss, F.; Gonzalez, G.A.; Senturk, A.; Acker-Palmer, A.; Lubec, G.; et al. Antibody-mediated neutralization of myelin-associated EphrinB3 accelerates CNS remyelination. Acta Neuropathol. 2016, 131, 281–298. [Google Scholar] [CrossRef]
- Vergara-Silva, A.; Schaefer, K.L.; Berg, L.J. Compartmentalized Eph receptor and ephrin expression in the thymus. Gene Expr. Patterns 2002, 2, 261–265. [Google Scholar] [CrossRef]
- Darling, T.K.; Lamb, T.J. Emerging Roles for Eph Receptors and Ephrin Ligands in Immunity. Front. Immunol. 2019, 10, 1473. [Google Scholar] [CrossRef] [PubMed]
- Sobel, R.A. Ephrin A receptors and ligands in lesions and normal-appearing white matter in multiple sclerosis. Brain Pathol. 2005, 15, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Prestoz, L.; Chatzopoulou, E.; Lemkine, G.; Spassky, N.; Lebras, B.; Kagawa, T.; Ikenaka, K.; Zalc, B.; Thomas, J.-L. Control of axonophilic migration of oligodendrocyte precursor cells by Eph-ephrin interaction. Neuron Glia Biol. 2004, 1, 73–83. [Google Scholar] [CrossRef]
- Lamberto, I.; Qin, H.; Noberini, R.; Premkumar, L.; Bourgin, C.; Riedl, S.J.; Song, J.; Pasquale, E.B. Distinctive binding of three antagonistic peptides to the ephrin-binding pocket of the EphA4 receptor. Biochem. J. 2012, 445, 47–56. [Google Scholar] [CrossRef]
- Skaper, S.D. Oligodendrocyte precursor cells as a therapeutic target for demyelinating diseases. In Progress in Brain Research; Elsevier: Amsterdam, The Netherlands, 2019; Volume 245, pp. 119–144. ISBN 9780444642080. [Google Scholar]
- Schwartz, M.; Moalem, G.; Leibowitz-Amit, R.; Cohen, I.R. Innate and adaptive immune responses can be beneficial for CNS repair. Trends Neurosci. 1999, 22, 295–299. [Google Scholar] [CrossRef]
- Kotter, M.R.; Setzu, A.; Sim, F.J.; Van Rooijen, N.; Franklin, R.J. Macrophage depletion impairs oligodendrocyte remyelination following lysolecithin-induced demyelination. Glia 2001, 35, 204–212. [Google Scholar] [CrossRef]
- Miron, V.E. Beyond immunomodulation: The regenerative role for regulatory T cells in central nervous system remyelination. J. Cell Commun. Signal. 2017, 11, 191–192. [Google Scholar] [CrossRef]
- Kirby, L.; Jin, J.; Cardona, J.G.; Smith, M.D.; Martin, K.A.; Wang, J.; Strasburger, H.; Herbst, L.; Alexis, M.; Karnell, J.; et al. Oligodendrocyte precursor cells present antigen and are cytotoxic targets in inflammatory demyelination. Nat. Commun. 2019, 10, 3887. [Google Scholar] [CrossRef] [PubMed]
- Bitsch, A.; Schuchardt, J.; Bunkowski, S.; Kuhlmann, T.; Brück, W. Acute axonal injury in multiple sclerosis. Correlation with demyelination and inflammation. Brain 2000, 123, 1174–1183. [Google Scholar] [CrossRef] [PubMed]
- Ji, Q.; Castelli, L.; Goverman, J.M. MHC class I-restricted myelin epitopes are cross-presented by Tip-DCs that promote determinant spreading to CD8+ T cells. Nat. Immunol. 2013, 14, 254–261. [Google Scholar] [CrossRef]
- Denic, A.; Wootla, B.; Rodriguez, M. CD8(+) T cells in multiple sclerosis. Expert Opin. Ther. Targets 2013, 17, 1053–1066. [Google Scholar] [CrossRef]
- Babbe, H.; Roers, A.; Waisman, A.; Lassmann, H.; Goebels, N.; Hohlfeld, R.; Friese, M.; Schröder, R.; Deckert, M.; Schmidt, S.; et al. Clonal expansions of CD8(+) T cells dominate the T cell infiltrate in active multiple sclerosis lesions as shown by micromanipulation and single cell polymerase chain reaction. J. Exp. Med. 2000, 192, 393–404. [Google Scholar] [CrossRef]
- Höftberger, R.; Aboul-Enein, F.; Brueck, W.; Lucchinetti, C.; Rodriguez, M.; Schmidbauer, M.; Jellinger, K.; Lassmann, H. Expression of major histocompatibility complex class I molecules on the different cell types in multiple sclerosis lesions. Brain Pathol. 2004, 14, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Marik, C.; Felts, P.A.; Bauer, J.; Lassmann, H.; Smith, K.J. Lesion genesis in a subset of patients with multiple sclerosis: A role for innate immunity? Brain 2007, 130, 2800–2815. [Google Scholar] [CrossRef]
- Kaskow, B.J.; Baecher-Allan, C. Effector T Cells in Multiple Sclerosis. Cold Spring Harb. Perspect. Med. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Ujigo, S.; Kamei, N.; Hadoush, H.; Fujioka, Y.; Miyaki, S.; Nakasa, T.; Tanaka, N.; Nakanishi, K.; Eguchi, A.; Sunagawa, T.; et al. Administration of microRNA-210 promotes spinal cord regeneration in mice. Spine (Phila. Pa. 1976) 2014, 39, 1099–1107. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nakashima, S.; Jinnin, M.; Kanemaru, H.; Kajihara, I.; Igata, T.; Okamoto, S.; Tazaki, Y.; Harada, M.; Masuguchi, S.; Fukushima, S.; et al. The role of miR-210, E2F3 and ephrin A3 in angiosarcoma cell proliferation. Eur. J. Dermatol. 2017, 27, 464–471. [Google Scholar] [CrossRef]
- Zhang, X.; Gong, X.; Qiu, J.; Zhang, Y.; Gong, F. MicroRNA-210 contributes to peripheral nerve regeneration through promoting the proliferation and migration of Schwann cells. Exp. Ther. Med. 2017, 14, 2809–2816. [Google Scholar] [CrossRef][Green Version]
- Duffy, P.; Wang, X.; Siegel, C.S.; Tu, N.; Henkemeyer, M.; Cafferty, W.B.J.; Strittmatter, S.M. Myelin-derived ephrinB3 restricts axonal regeneration and recovery after adult CNS injury. Proc. Natl. Acad. Sci. USA 2012, 109, 5063–5068. [Google Scholar] [CrossRef] [PubMed]
- Tsenkina, Y.; Ricard, J.; Runko, E.; Quiala-Acosta, M.M.; Mier, J.; Liebl, D.J. EphB3 receptors function as dependence receptors to mediate oligodendrocyte cell death following contusive spinal cord injury. Cell Death Dis. 2015, 6, e1922. [Google Scholar] [CrossRef] [PubMed]
- Mahnke, Y.D.; Beddall, M.H.; Roederer, M. OMIP-015: Human regulatory and activated T-cells without intracellular staining. Cytometry A 2013, 83, 179–181. [Google Scholar] [CrossRef]
- Mahnke, Y.D.; Beddall, M.H.; Roederer, M. OMIP-017: Human CD4(+) helper T-cell subsets including follicular helper cells. Cytometry A 2013, 83, 439–440. [Google Scholar] [CrossRef] [PubMed]

| Untreated RR-MS Patients | Healthy Controls | |
|---|---|---|
| Number | 43 | 27 |
| Males | 18 | 8 |
| Females | 25 | 19 |
| Age (mean ± SD), years | 37.7 ± 11.86 | 38.9 ± 9.52 |
| Range, years | (19–69) | (19–60) |
| EDSS (mean ± SD) | 2.15 ± 2.16 | |
| Range | (0–7) | |
| Disease duration, years (mean ± SD) | 6.79 ± 7.61 | |
| Range, years | (0–28) |
| Ephrin-A1 | Ephrin-A2 | Ephrin-A3 | Ephrin-B3 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HC | MS | p | HC | MS | p | HC | MS | p | HC | MS | p | |
| PBMCs | 7305 ± 563 | 8630 ± 970 | 0.133 | 6968 ± 599 | 8873 ± 958 | 0.036 | 10,713 ± 1199 | 13,585 ± 1227 | 0.053 | 5974 ± 573 | 8035 ± 972 | 0.022 |
| T cells | 8395 ± 1430 | 15,252 ± 1905 | 0.005 | 6649 ± 1016 | 6517 ± 406 | 0.905 | 6323 ± 858 | 9340 ± 711 | 0.010 | 3540 ± 478 | 3950 ± 267 | 0.458 |
| B cells | 5434 ± 832 | 5670 ± 771 | 0.836 | 8314 ± 1002 | 7403 ± 521 | 0.440 | 5792 ± 781 | 6226 ± 890 | 0.715 | 3610 ± 514 | 6051 ± 1037 | 0.037 |
| Mo | 7852 ± 779 | 8926 ± 642 | 0.272 | 8088 ± 917 | 9855 ± 821 | 0.132 | 11,121 ± 1437 | 13,899 ± 1082 | 0.106 | 6367 ± 794 | 8614 ± 740 | 0.033 |
| Ephrin-A1 | Ephrin-A2 | Ephrin-A3 | Ephrin-B3 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HC | MS | p | HC | MS | p | HC | MS | p | HC | MS | p | |
| CD4+ | 5623 ± 1538 | 8800 ± 1498 | 0.144 | 5699 ± 733 | 5804 ± 350 | 0.898 | 5669 ± 739 | 9422 ± 766 | <0.001 | 2608 ± 326 | 3118 ± 225 | 0.196 |
| CD8+ | 1138 ± 80 | 1211 ± 80 | 0.518 | 2476 ± 253 | 2577 ± 162 | 0.739 | 3237 ± 547 | 4912 ± 596 | 0.042 | 2020 ± 216 | 1709 ± 118 | 0.213 |
| T-regs | 1200 ± 155 | 1793 ± 261 | 0.055 | 2973 ± 231 | 4186 ± 510 | 0.034 | 2414 ± 377 | 4537 ± 465 | <0.001 | 1201 ± 147 | 1719 ± 243 | 0.076 |
| Th1 | 582 ± 87 | 731 ± 134 | 0.357 | 710 ± 48 | 826 ± 88 | 0.254 | 661 ± 71 | 1004 ± 85 | 0.003 | 401 ± 21 | 514 ± 39 | 0.013 |
| Th2 | 676 ± 75 | 3256 ± 1353 | 0.065 | 660 ± 96 | 1070 ± 414 | 0.340 | 846 ± 87 | 1124 ± 133 | 0.078 | 540 ± 72 | 685 ± 60 | 0.117 |
| Th17 | 4268 ± 1870 | 22,841 ± 8471 | 0.038 | 882 ± 149 | 824 ± 149 | 0.785 | 971 ± 166 | 838 ± 118 | 0.518 | 503 ± 74 | 728 ± 159 | 0.207 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Golan, M.; Krivitsky, A.; Mausner-Fainberg, K.; Benhamou, M.; Vigiser, I.; Regev, K.; Kolb, H.; Karni, A. Increased Expression of Ephrins on Immune Cells of Patients with Relapsing Remitting Multiple Sclerosis Affects Oligodendrocyte Differentiation. Int. J. Mol. Sci. 2021, 22, 2182. https://doi.org/10.3390/ijms22042182
Golan M, Krivitsky A, Mausner-Fainberg K, Benhamou M, Vigiser I, Regev K, Kolb H, Karni A. Increased Expression of Ephrins on Immune Cells of Patients with Relapsing Remitting Multiple Sclerosis Affects Oligodendrocyte Differentiation. International Journal of Molecular Sciences. 2021; 22(4):2182. https://doi.org/10.3390/ijms22042182
Chicago/Turabian StyleGolan, Maya, Avivit Krivitsky, Karin Mausner-Fainberg, Moshe Benhamou, Ifat Vigiser, Keren Regev, Hadar Kolb, and Arnon Karni. 2021. "Increased Expression of Ephrins on Immune Cells of Patients with Relapsing Remitting Multiple Sclerosis Affects Oligodendrocyte Differentiation" International Journal of Molecular Sciences 22, no. 4: 2182. https://doi.org/10.3390/ijms22042182
APA StyleGolan, M., Krivitsky, A., Mausner-Fainberg, K., Benhamou, M., Vigiser, I., Regev, K., Kolb, H., & Karni, A. (2021). Increased Expression of Ephrins on Immune Cells of Patients with Relapsing Remitting Multiple Sclerosis Affects Oligodendrocyte Differentiation. International Journal of Molecular Sciences, 22(4), 2182. https://doi.org/10.3390/ijms22042182








