How to Select Firefly Luciferin Analogues for In Vivo Imaging
Abstract
1. Introduction
2. The Development of Luciferin and Luciferase for NIR BLI
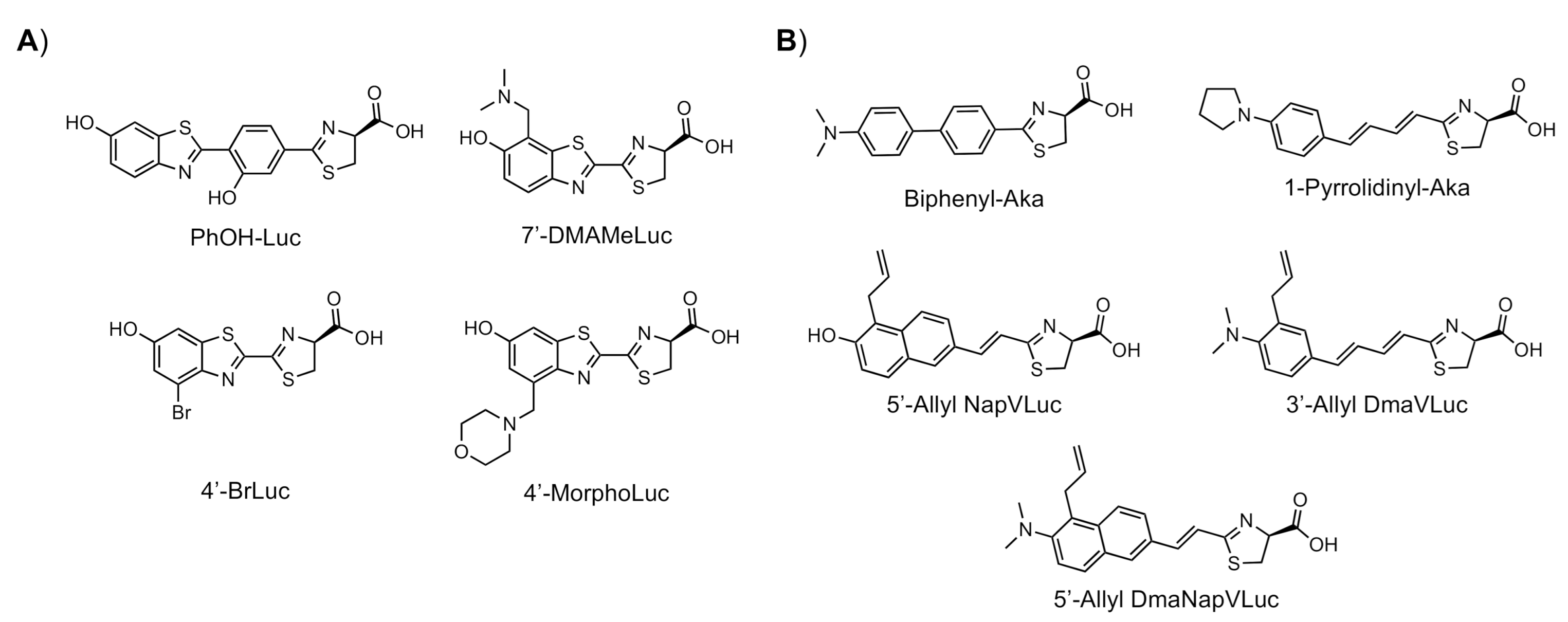
2.1. Luciferin Analogues that Produce NIR Light
2.2. The Development and Evaluation of Combinations of Luciferin Analogues and Luciferase Mutants
3. Chemical and Physical Characteristics of the NIR Luciferin Analogues TokeOni and seMpai
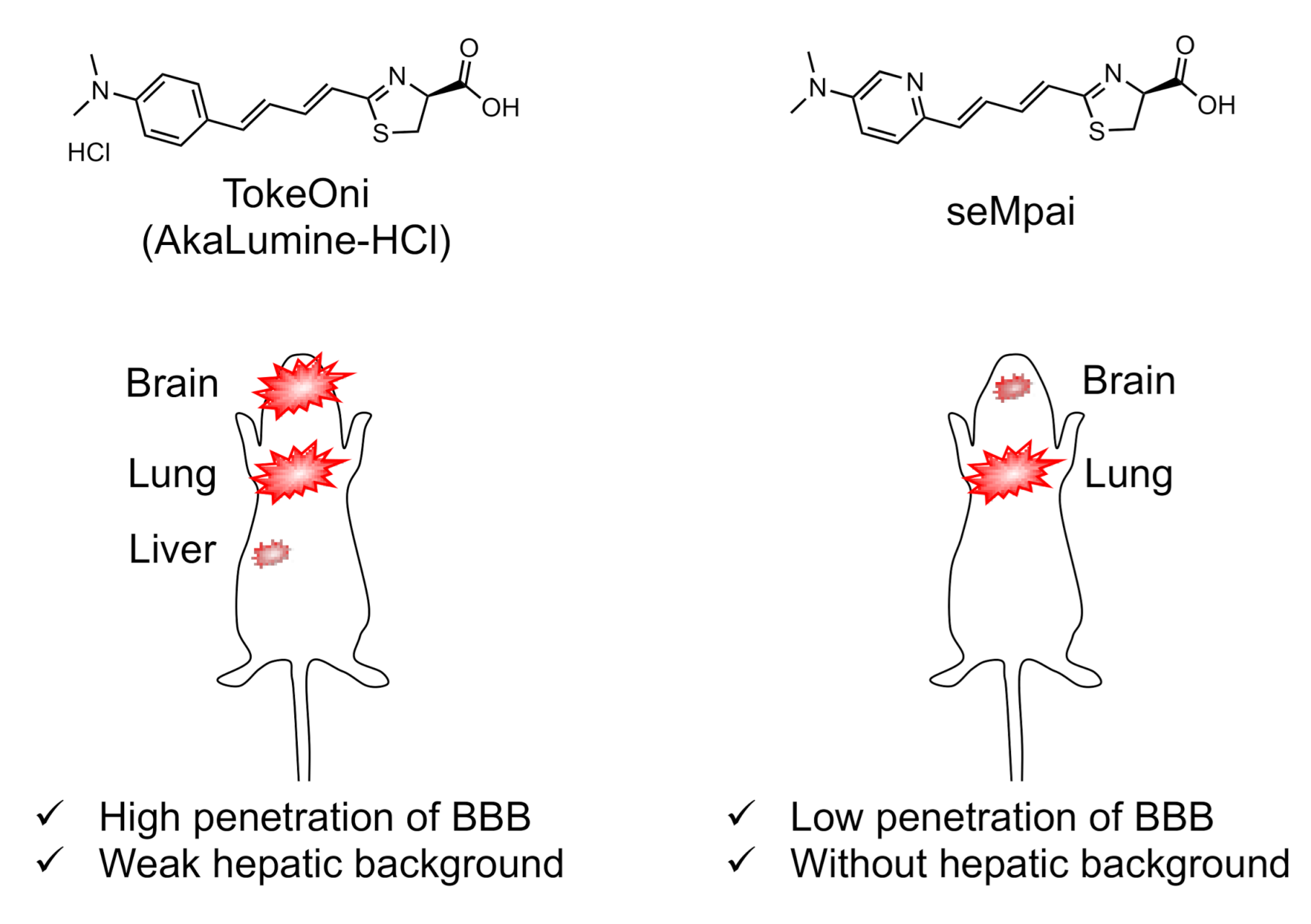
3.1. Development of TokeOni (AkaLumine-HCl)
3.2. Development of seMpai
3.3. Differences between Luciferin Analogue Dynamics In Vivo
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ATP | Adenosine triphosphate |
| BBB | Blood–brain barrier |
| BDNF | Brain-derived neurotrophic factor |
| BLI | Bioluminescence imaging |
| CBR | Click beetle luciferase |
| CTZ | Coelenterazine |
| DLH2 | d-Luciferin |
| Eluc | Emerald luciferase |
| Fluc | Firefly luciferase |
| FMZ | Furimazine |
| FMZ | Furimazine |
| Gluc | Gaussia luciferase |
| iLH2 | Infraluciferin |
| NIR | Near-infrared |
| Oluc | Oplophorus luciferase |
| PBS | Phosphate buffered saline |
| Rluc | Renilla luciferase |
| SLR | Stable luciferase red |
References
- Wilson, T.; Hastings, J.W. Bioluminescence. Annu. Rev. Cell Dev. Biol. 1998, 14, 197–230. [Google Scholar] [CrossRef]
- Shimomura, O. The Fireflies and Luminous Insects. In Bioluminescence; World Scientific: Toh Tuck Link, Singapore, 2012; pp. 1–30. ISBN 978-981-4366-08-3. [Google Scholar] [CrossRef]
- Kaskova, Z.M.; Tsarkova, A.S.; Yampolsky, I.V. 1001 lights: Luciferins, luciferases, their mechanisms of action and applications in chemical analysis, biology and medicine. Chem. Soc. Rev. 2016, 45, 6048–6077. [Google Scholar] [CrossRef]
- Fraga, H. Firefly luminescence: A historical perspective and recent developments. Photochem. Photobiol. Sci. 2008, 7, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Branchini, B.R.; Murtiashaw, M.H.; Magyar, R.A.; Portier, N.C.; Ruggiero, M.C.; Stroh, J.G. Yellow-green and red firefly bioluminescence from 5,5-dimethyloxyluciferin. J. Am. Chem. Soc. 2002, 124, 2112–2113. [Google Scholar] [CrossRef] [PubMed]
- Branchini, B.R.; Southworth, T.L.; Murtiashaw, M.H.; Magyar, R.A.; Gonzalez, S.A.; Ruggiero, M.C.; Stroh, J.G. An alternative mechanism of bioluminescence color determination in firefly luciferase. Biochemistry 2004, 43, 7255–7262. [Google Scholar] [CrossRef] [PubMed]
- Hirano, T.; Hasumi, Y.; Ohtsuka, K.; Maki, S.; Niwa, H.; Yamaji, M.; Hashizume, D. Spectroscopic studies of the light-color modulation mechanism of firefly (Beetle) bioluminescence. J. Am. Chem. Soc. 2009, 131, 2385–2396. [Google Scholar] [CrossRef]
- Shimomura, O.; Masugi, T.; Johnson, F.H.; Haneda, Y. Properties and reaction mechanism of the bioluminescence system of the deep-sea shrimp Oplophorus gracilorostris. Biochemistry 1978, 17, 994–998. [Google Scholar] [CrossRef]
- Inouye, S.; Watanabe, K.; Nakamura, H.; Shimomura, O. Secretional luciferase of the luminous shrimp Oplophorus gracilirostris: cDNA cloning of a novel imidazopyrazinone luciferase. FEBS Lett. 2000, 481, 19–25. [Google Scholar] [CrossRef]
- Krasitskaya, V.V.; Bashmakova, E.E.; Frank, L.A. Coelenterazine-Dependent Luciferases as a Powerful Analytical Tool for Research and Biomedical Applications. Int. J. Mol. Sci. 2020, 21, 7465. [Google Scholar] [CrossRef]
- Oba, Y.; Konishi, K.; Yano, D.; Shibata, H.; Kato, D.; Shirai, T. Resurrecting the ancient glow of the fireflies. Sci. Adv. 2020, 6, eabc5705. [Google Scholar] [CrossRef]
- Thornton, J.W. Resurrecting ancient genes: Experimental analysis of extinct molecules. Nat. Rev. Genet. 2004, 5, 366–375. [Google Scholar] [CrossRef]
- Thorne, N.; Inglese, J.; Auld, D.S. Illuminating insights into firefly luciferase and other bioluminescent reporters used in chemical biology. Chem. Biol. 2010, 17, 646–657. [Google Scholar] [CrossRef]
- Badr, C.E.; Tannous, B.A. Bioluminescence imaging: Progress and applications. Trends Biotechnol. 2011, 29, 624–633. [Google Scholar] [CrossRef]
- Krishnasamy, S.; Weng, Y.-C.; Thammisetty, S.S.; Phaneuf, D.; Lalancette-Hebert, M.; Kriz, J. Molecular imaging of nestin in neuroinflammatory conditions reveals marked signal induction in activated microglia. J. Neuroinflammation 2017, 14, 45. [Google Scholar] [CrossRef] [PubMed]
- Dorsaz, S.; Coste, A.T.; Sanglard, D. Red-shifted firefly luciferase optimized for Candida albicans in vivo bioluminescence imaging. Front. Microbiol. 2017, 8, 1478. [Google Scholar] [CrossRef] [PubMed]
- Tannert, A.; Ramoji, A.; Neugebauer, U.; Popp, J. Photonic monitoring of treatment during infection and sepsis: Development of new detection strategies and potential clinical applications. Anal. Bioanal. Chem. 2018, 410, 773–790. [Google Scholar] [CrossRef] [PubMed]
- Lalancette-Hébert, M.; Faustino, J.; Thammisetty, S.S.; Chip, S.; Vexler, Z.S.; Kriz, J. Live imaging of the innate immune response in neonates reveals differential TLR2 dependent activation patterns in sterile inflammation and infection. Brain. Behav. Immun. 2017, 65, 312–327. [Google Scholar] [CrossRef] [PubMed]
- Rahimian, R.; Cordeau, P.; Kriz, J. Brain response to injuries: When microglia go sexist. Neuroscience 2019, 405, 14–23. [Google Scholar] [CrossRef]
- Cordeau, P.; Kriz, J. Real-Time Imaging after Cerebral Ischemia. In Methods in Enzymology; Elsevier Inc.: Amsterdam, The Netherlands, 2012; Volume 506, pp. 117–133. ISBN 9780123918567. [Google Scholar] [CrossRef]
- Barash, N.R.; Nosala, C.; Pham, J.K.; McInally, S.G.; Gourguechon, S.; McCarthy-Sinclair, B.; Dawson, S.C. Giardia colonizes and encysts in high-density foci in the murine small intestine. mSphere 2017, 2, 1–20. [Google Scholar] [CrossRef]
- Martin, R.M.; Fowler, J.L.; Cromer, M.K.; Lesch, B.J.; Ponce, E.; Uchida, N.; Nishimura, T.; Porteus, M.H.; Loh, K.M. Improving the safety of human pluripotent stem cell therapies using genome-edited orthogonal safeguards. Nat. Commun. 2020, 11, 2713. [Google Scholar] [CrossRef] [PubMed]
- Furuya, T.; Takehara, I.; Shimura, A.; Kishimoto, H.; Yasujima, T.; Ohta, K.; Shirasaka, Y.; Yuasa, H.; Inoue, K. Organic anion transporter 1 (OAT1/SLC22A6) enhances bioluminescence based on d-luciferin–luciferase reaction in living cells by facilitating the intracellular accumulation of d-luciferin. Biochem. Biophys. Res. Commun. 2018, 495, 2152–2157. [Google Scholar] [CrossRef]
- Yamada, K.; Noguchi, K.; Kimitsuki, K.; Kaimori, R.; Saito, N.; Komeno, T.; Nakajima, N.; Furuta, Y.; Nishizono, A. Reevaluation of the efficacy of favipiravir against rabies virus using in vivo imaging analysis. Antiviral Res. 2019, 172, 104641. [Google Scholar] [CrossRef] [PubMed]
- Mehle, A. Fiat luc: Bioluminescence imaging reveals in vivo viral replication dynamics. PLoS Pathog. 2015, 11, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Cook, S.H.; Griffin, D.E. Luciferase imaging of a neurotropic viral infection in intact animals. J. Virol. 2003, 77, 5333–5338. [Google Scholar] [CrossRef] [PubMed]
- Smale, S.T. Luciferase Assay. Cold Spring Harb. Protoc. 2010, 2010, pdb.prot5421. [Google Scholar] [CrossRef]
- Bennett, J.; Duan, D.; Engelhardt, J.F.; Maguire, A.M. Real-time, noninvasive in vivo assessment of adeno-associated virus- mediated retinal transduction. Investig. Ophthalmol. Vis. Sci. 1997, 38, 2857–2863. [Google Scholar]
- Contag, P.R.; Nick Olomu, I.; Stevenson, D.K.; Contag, C.H. Bioluminescent indicators in living mammals. Nat. Med. 1998, 4, 245–247. [Google Scholar] [CrossRef]
- Fleiss, A.; Sarkisyan, K.S. A brief review of bioluminescent systems (2019). Curr. Genet. 2019, 65, 877–882. [Google Scholar] [CrossRef]
- Chen, L.; Chen, Z.; Zheng, S.; Fan, L.; Zhu, L.; Yu, J.; Tang, C.; Liu, Q.; Xiong, Y. Study on mechanism of elemene reversing tumor multidrug resistance based on luminescence pharmacokinetics in tumor cellsin vitroandin vivo. RSC Adv. 2020, 10, 34928–34937. [Google Scholar] [CrossRef]
- Alsawaftah, N.; Farooq, A.; Dhou, S.; Majdalawieh, A.F. Bioluminescence imaging applications in cancer: A comprehensive review. IEEE Rev. Biomed. Eng. 2021, 14, 307–326. [Google Scholar] [CrossRef]
- Nakayama, J.; Ito, E.; Fujimoto, J.; Watanabe, S.; Semba, K. Comparative analysis of gene regulatory networks of highly metastatic breast cancer cells established by orthotopic transplantation and intra-circulation injection. Int. J. Oncol. 2017, 50, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Kuchimaru, T.; Kataoka, N.; Nakagawa, K.; Isozaki, T.; Miyabara, H.; Minegishi, M.; Kadonosono, T.; Kizaka-Kondoh, S. A reliable murine model of bone metastasis by injecting cancer cells through caudal arteries. Nat. Commun. 2018, 9, 2981. [Google Scholar] [CrossRef]
- Kuroiwa, Y.; Nakayama, J.; Adachi, C.; Inoue, T.; Watanabe, S.; Semba, K. Proliferative classification of intracranially injected HER2-positive breast cancer cell lines. Cancers 2020, 12, 1811. [Google Scholar] [CrossRef]
- Han, Y.; Nakayama, J.; Hayashi, Y.; Jeong, S.; Futakuchi, M.; Ito, E.; Watanabe, S.; Semba, K. Establishment and characterization of highly osteolytic luminal breast cancer cell lines by intracaudal arterial injection. Genes Cells 2020, 25, 111–123. [Google Scholar] [CrossRef]
- Yanagihara, K.; Takigahira, M.; Takeshita, F.; Komatsu, T.; Nishio, K.; Hasegawa, F.; Ochiya, T. A photon counting technique for quantitatively evaluating progression of peritoneal tumor dissemination. Cancer Res. 2006, 66, 7532–7539. [Google Scholar] [CrossRef] [PubMed]
- Manni, I.; de Latouliere, L.; Gurtner, A.; Piaggio, G. Transgenic animal models to visualize cancer-related cellular processes by bioluminescence imaging. Front. Pharmacol. 2019, 10, 235. [Google Scholar] [CrossRef]
- Kim, J.-B.; Urban, K.; Cochran, E.; Lee, S.; Ang, A.; Rice, B.; Bata, A.; Campbell, K.; Coffee, R.; Gorodinsky, A.; et al. Non-invasive detection of a small number of bioluminescent cancer cells in vivo. PLoS ONE 2010, 5, e9364. [Google Scholar] [CrossRef] [PubMed]
- Mizui, Y.; Eguchi, M.; Tanaka, M.; Ikeda, Y.; Yoshimura, H.; Ozawa, T.; Citterio, D.; Hiruta, Y. Long-term single cell bioluminescence imaging with C-3 position protected coelenterazine analogues. Org. Biomol. Chem. 2021, 19, 579–586. [Google Scholar] [CrossRef]
- Nishihara, R.; Hoshino, E.; Kakudate, Y.; Kishigami, S.; Iwasawa, N.; Sasaki, S.; Nakajima, T.; Sato, M.; Nishiyama, S.; Citterio, D.; et al. Azide- and dye-conjugated coelenterazine analogues for a multiplex molecular imaging platform. Bioconjug. Chem. 2018, 29, 1922–1931. [Google Scholar] [CrossRef]
- Nishihara, R.; Suzuki, H.; Hoshino, E.; Suganuma, S.; Sato, M.; Saitoh, T.; Nishiyama, S.; Iwasawa, N.; Citterio, D.; Suzuki, K. Bioluminescent coelenterazine derivatives with imidazopyrazinone C-6 extended substitution. Chem. Commun. 2015, 51, 391–394. [Google Scholar] [CrossRef]
- Nishihara, R.; Paulmurugan, R.; Nakajima, T.; Yamamoto, E.; Natarajan, A.; Afjei, R.; Hiruta, Y.; Iwasawa, N.; Nishiyama, S.; Citterio, D.; et al. Highly bright and stable NIR-BRET with blue-shifted coelenterazine derivatives for deep-tissue imaging of molecular events in vivo. Theranostics 2019, 9, 2646–2661. [Google Scholar] [CrossRef] [PubMed]
- Coutant, E.P.; Gagnot, G.; Hervin, V.; Baatallah, R.; Goyard, S.; Jacob, Y.; Rose, T.; Janin, Y.L. Bioluminescence profiling of NanoKAZ/NanoLuc luciferase using a chemical library of coelenterazine analogues. Chem. A Eur. J. 2020, 26, 948–958. [Google Scholar] [CrossRef] [PubMed]
- Belarbi, E.; Legros, V.; Basset, J.; Desprès, P.; Roques, P.; Choumet, V. Bioluminescent ross river rvrus allows live monitoring of acute and long-term alphaviral infection by in vivo imaging. Viruses 2019, 11, 584. [Google Scholar] [CrossRef] [PubMed]
- Gupta, D.; Liang, X.; Pavlova, S.; Wiklander, O.P.B.; Corso, G.; Zhao, Y.; Saher, O.; Bost, J.; Zickler, A.M.; Piffko, A.; et al. Quantification of extracellular vesicles in vitro and in vivo using sensitive bioluminescence imaging. J. Extracell. Vesicles 2020, 9, 1800222. [Google Scholar] [CrossRef]
- Inagaki, S.; Agetsuma, M.; Ohara, S.; Iijima, T.; Yokota, H.; Wazawa, T.; Arai, Y.; Nagai, T. Imaging local brain activity of multiple freely moving mice sharing the same environment. Sci. Rep. 2019, 9, 7460. [Google Scholar] [CrossRef]
- Anindita, P.D.; Sasaki, M.; Nobori, H.; Sato, A.; Carr, M.; Ito, N.; Sugiyama, M.; Orba, Y.; Sawa, H. Generation of recombinant rabies viruses encoding NanoLuc luciferase for antiviral activity assays. Virus Res. 2016, 215, 121–128. [Google Scholar] [CrossRef]
- Hall, M.P.; Unch, J.; Binkowski, B.F.; Valley, M.P.; Butler, B.L.; Wood, M.G.; Otto, P.; Zimmerman, K.; Vidugiris, G.; Machleidt, T.; et al. Engineered luciferase reporter from a deep sea shrimp utilizing a novel imidazopyrazinone substrate. ACS Chem. Biol. 2012, 7, 1848–1857. [Google Scholar] [CrossRef]
- Stacer, A.C.; Nyati, S.; Moudgil, P.; Iyengar, R.; Luker, K.E.; Rehemtulla, A.; Luker, G.D. NanoLuc reporter for dual luciferase imaging in living animals. Mol. Imaging 2013, 12, 457–469. [Google Scholar] [CrossRef]
- Dixon, A.S.; Schwinn, M.K.; Hall, M.P.; Zimmerman, K.; Otto, P.; Lubben, T.H.; Butler, B.L.; Binkowski, B.F.; Machleidt, T.; Kirkland, T.A.; et al. NanoLuc complementation reporter optimized for accurate measurement of protein interactions in cells. ACS Chem. Biol. 2016, 11, 400–408. [Google Scholar] [CrossRef]
- Yeh, H.W.; Xiong, Y.; Wu, T.; Chen, M.; Ji, A.; Li, X.; Ai, H.W. ATP-independent bioluminescent reporter variants to improve in vivo imaging. ACS Chem. Biol. 2019, 14, 959–965. [Google Scholar] [CrossRef] [PubMed]
- Yeh, H.W.; Wu, T.; Chen, M.; Ai, H.W. Identification of factors complicating bioluminescence imaging. Biochemistry 2019, 58, 1689–1697. [Google Scholar] [CrossRef] [PubMed]
- Hikita, T.; Miyata, M.; Watanabe, R.; Oneyama, C. In vivo imaging of long-term accumulation of cancer-derived exosomes using a BRET-based reporter. Sci. Rep. 2020, 10, 16616. [Google Scholar] [CrossRef]
- Jiang, T.; Du, L.; Li, M. Lighting up bioluminescence with coelenterazine: Strategies and applications. Photochem. Photobiol. Sci. 2016, 15, 466–480. [Google Scholar] [CrossRef]
- Love, A.C.; Prescher, J.A. Seeing (and using) the light: Recent developments in bioluminescence technology. Cell Chem. Biol. 2020, 27, 904–920. [Google Scholar] [CrossRef]
- Evans, M.S.; Chaurette, J.P.; Adams, S.T.; Reddy, G.R.; Paley, M.A.; Aronin, N.; Prescher, J.A.; Miller, S.C. A synthetic luciferin improves bioluminescence imaging in live mice. Nat. Methods 2014, 11, 393–395. [Google Scholar] [CrossRef] [PubMed]
- Kuchimaru, T.; Iwano, S.; Kiyama, M.; Mitsumata, S.; Kadonosono, T.; Niwa, H.; Maki, S.; Kizaka-Kondoh, S. A luciferin analogue generating near-infrared bioluminescence achieves highly sensitive deep-tissue imaging. Nat. Commun. 2016, 7, 11856. [Google Scholar] [CrossRef]
- Weissleder, R. A clearer vision for in vivo imaging. Nat. Biotechnol. 2001, 19, 316–317. [Google Scholar] [CrossRef] [PubMed]
- Dawson, J.B.; Barker, D.J.; Ellis, D.J.; Cotterill, J.A.; Grassam, E.; Fisher, G.W.; Feather, J.W. A theoretical and experimental study of light absorption and scattering by in vivo skin. Phys. Med. Biol. 1980, 25, 695–709. [Google Scholar] [CrossRef]
- Lee, K.-H.; Byun, S.S.; Paik, J.-Y.; Lee, S.Y.; Song, S.H.; Choe, Y.S.; Kim, B.-T. Cell uptake and tissue distribution of radioiodine labelled d-luciferin: Implications for luciferase-based gene imaging. Nucl. Med. Commun. 2003, 24, 1003–1009. [Google Scholar] [CrossRef]
- Berger, F.; Paulmurugan, R.; Bhaumik, S.; Gambhir, S.S. Uptake kinetics and biodistribution of 14C-d-luciferin—A radiolabeled substrate for the firefly luciferase catalyzed bioluminescence reaction: Impact on bioluminescence-based reporter gene imaging. Eur. J. Nucl. Med. Mol. Imaging 2008, 35, 2275–2285. [Google Scholar] [CrossRef]
- Miller, S.C.; Mofford, D.M.; Adams, S.T. Lessons learned from luminous luciferins and latent luciferases. ACS Chem. Biol. 2018, 13, 1734–1740. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Ruan, Z.; Zhang, H.; Xu, H. Recent achievements of bioluminescence imaging based on firefly luciferin-luciferase system. Eur. J. Med. Chem. 2021, 211, 113111. [Google Scholar] [CrossRef] [PubMed]
- Kajiyama, N.; Nakano, E. Isolation and characterization of mutants of firefly luciferase which produce different colors of light. Protein Eng. 1991, 4, 691–693. [Google Scholar] [CrossRef]
- Branchini, B.R.; Magyar, R.A.; Murtiashaw, M.H.; Anderson, S.M.; Helgerson, L.C.; Zimmer, M. Site-directed mutagenesis of firefly luciferase active site amino acids: A proposed model for bioluminescence color. Biochemistry 1999, 38, 13223–13230. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, Y.; Kimura, T.; Sugata, K.; Enomoto, T.; Asakawa, A.; Kubota, H.; Ikeda, M.; Ohmiya, Y. Multicolor luciferase assay system: One-step monitoring of multiple gene expressions with a single substrate. Biotechniques 2005, 38, 891–894. [Google Scholar] [CrossRef] [PubMed]
- Viviani, V.R.; Bevilaqua, V.R.; de Souza, D.R.; Pelentir, G.F.; Kakiuchi, M.; Hirano, T. A very bright far-red bioluminescence emitting combination based on engineered railroad worm luciferase and 6′-amino-analogs for bioimaging purposes. Int. J. Mol. Sci. 2020, 22, 303. [Google Scholar] [CrossRef] [PubMed]
- Branchini, B.R.; Ablamsky, D.M.; Rosenberg, J.C. Chemically modified firefly luciferase is an efficient source of near-infrared light. Bioconjug. Chem. 2010, 21, 2023–2030. [Google Scholar] [CrossRef]
- Kojima, R.; Takakura, H.; Ozawa, T.; Tada, Y.; Nagano, T.; Urano, Y. Rational design and development of near-infrared-emitting firefly luciferins available in vivo. Angew. Chemie Int. Ed. 2013, 52, 1175–1179. [Google Scholar] [CrossRef]
- Mikitsh, J.L.; Chacko, A.M. Pathways for small molecule delivery to the central nervous system across the blood-brain barrier. Perspect. Medicin. Chem. 2014, 11–24. [Google Scholar] [CrossRef]
- Harwood, K.R.; Mofford, D.M.; Reddy, G.R.; Miller, S.C. Identification of mutant firefly luciferases that efficiently utilize aminoluciferins. Chem. Biol. 2011, 18, 1649–1657. [Google Scholar] [CrossRef]
- Wu, W.; Su, J.; Tang, C.; Bai, H.; Ma, Z.; Zhang, T.; Yuan, Z.; Li, Z.; Zhou, W.; Zhang, H.; et al. CybLuc: An effective aminoluciferin derivative for deep bioluminescence imaging. Anal. Chem. 2017, 89, 4808–4816. [Google Scholar] [CrossRef]
- Hall, M.P.; Woodroofe, C.C.; Wood, M.G.; Que, I.; van’t Root, M.; Ridwan, Y.; Shi, C.; Kirkland, T.A.; Encell, L.P.; Wood, K.V.; et al. Click beetle luciferase mutant and near infrared naphthyl-luciferins for improved bioluminescence imaging. Nat. Commun. 2018, 9, 132. [Google Scholar] [CrossRef]
- Reddy, G.R.; Thompson, W.C.; Miller, S.C. Robust light emission from cyclic alkylaminoluciferin substrates for firefly luciferase. J. Am. Chem. Soc. 2010, 132, 13586–13587. [Google Scholar] [CrossRef] [PubMed]
- Simonyan, H.; Hurr, C.; Young, C.N. A synthetic luciferin improves in vivo bioluminescence imaging of gene expression in cardiovascular brain regions. Physiol. Genomics 2016, 48, 762–770. [Google Scholar] [CrossRef]
- Ji, X.; Adams, S.T.; Miller, S.C. Bioluminescence Imaging in Mice with Synthetic Luciferin Analogues. In Methods in Enzymology; Elsevier Inc.: Amsterdam, The Netherlands, 2020; Volume 640, pp. 165–183. ISBN 9780128211533. [Google Scholar] [CrossRef]
- Kleinovink, J.W.; Mezzanotte, L.; Zambito, G.; Fransen, M.F.; Cruz, L.J.; Verbeek, J.S.; Chan, A.; Ossendorp, F.; Löwik, C. A dual-color bioluminescence reporter mouse for simultaneous in vivo imaging of T cell localization and function. Front. Immunol. 2019, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Mezzanotte, L.; Iljas, J.D.; Que, I.; Chan, A.; Kaijzel, E.; Hoeben, R.; Löwik, C. Optimized longitudinal monitoring of stem cell grafts in mouse brain using a novel bioluminescent/near infrared fluorescent fusion reporter. Cell Transplant. 2017, 26, 1878–1889. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.T.; Mofford, D.M.; Reddy, G.S.K.K.; Miller, S.C. Firefly luciferase mutants allow substrate-selective bioluminescence imaging in the mouse brain. Angew. Chemie Int. Ed. 2016, 55, 4943–4946. [Google Scholar] [CrossRef] [PubMed]
- Iwano, S.; Sugiyama, M.; Hama, H.; Watakabe, A.; Hasegawa, N.; Kuchimaru, T.; Tanaka, K.Z.; Takahashi, M.; Ishida, Y.; Hata, J.; et al. Single-cell bioluminescence imaging of deep tissue in freely moving animals. Science 2018, 359, 935–939. [Google Scholar] [CrossRef]
- Saito, R.; Kuchimaru, T.; Higashi, S.; Lu, S.W.; Kiyama, M.; Iwano, S.; Obata, R.; Hirano, T.; Kizaka-Kondoh, S.; Maki, S.A. Synthesis and luminescence properties of near-infrared N-heterocyclic luciferin analogues for in vivo optical imaging. Bull. Chem. Soc. Jpn. 2019, 92, 608–618. [Google Scholar] [CrossRef]
- Ikeda, Y.; Nomoto, T.; Hiruta, Y.; Nishiyama, N.; Citterio, D. Ring-fused firefly luciferins: Expanded palette of near-infrared emitting bioluminescent substrates. Anal. Chem. 2020, 92, 4235–4243. [Google Scholar] [CrossRef]
- Iwano, S.; Obata, R.; Miura, C.; Kiyama, M.; Hama, K.; Nakamura, M.; Amano, Y.; Kojima, S.; Hirano, T.; Maki, S.; et al. Development of simple firefly luciferin analogs emitting blue, green, red, and near-infrared biological window light. Tetrahedron 2013, 69, 3847–3856. [Google Scholar] [CrossRef]
- Fukuchi, M.; Izumi, H.; Mori, H.; Kiyama, M.; Otsuka, S.; Maki, S.; Maehata, Y.; Tabuchi, A.; Tsuda, M. Visualizing changes in brain-derived neurotrophic factor (BDNF) expression using bioluminescence imaging in living mice. Sci. Rep. 2017, 7, 4949. [Google Scholar] [CrossRef] [PubMed]
- Fukuchi, M.; Saito, R.; Maki, S.; Hagiwara, N.; Nakajima, Y.; Mitazaki, S.; Izumi, H.; Mori, H. Visualization of activity-regulated BDNF expression in the living mouse brain using non-invasive near-infrared bioluminescence imaging. Mol. Brain 2020, 13, 122. [Google Scholar] [CrossRef] [PubMed]
- Zhong, C.; Kayamori, K.; Koide, S.; Shinoda, D.; Oshima, M.; Nakajima-Takagi, Y.; Nagai, Y.; Mimura, N.; Sakaida, E.; Yamazaki, S.; et al. Efficacy of the novel tubulin polymerization inhibitor PTC-028 for myelodysplastic syndrome. Cancer Sci. 2020, 4336–4347. [Google Scholar] [CrossRef] [PubMed]
- Raikwar, S.P.; Thangavel, R.; Ahmed, M.E.; Selvakumar, G.P.; Kempuraj, D.; Wu, K.; Khan, O.; Bazley, K.; Bussinger, B.; Kukulka, K.; et al. Real-time noninvasive bioluminescence, ultrasound and photoacoustic imaging in NFκB-RE-Luc transgenic mice reveal glia maturation factor-mediated immediate and sustained spatio-temporal activation of NFκB signaling post-traumatic brain injury in a gender-. Cell. Mol. Neurobiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, J.; Saito, R.; Hayashi, Y.; Kitada, N.; Tamaki, S.; Han, Y.; Semba, K.; Maki, S.A. High sensitivity in vivo imaging of cancer metastasis using a near-infrared luciferin analogue seMpai. Int. J. Mol. Sci. 2020, 21, 7896. [Google Scholar] [CrossRef] [PubMed]
- Jathoul, A.P.; Grounds, H.; Anderson, J.C.; Pule, M.A. A dual-color far-red to near-infrared firefly luciferin analogue designed for multiparametric bioluminescence imaging. Angew. Chemie Int. Ed. 2014, 53, 13059–13063. [Google Scholar] [CrossRef]
- Stowe, C.L.; Burley, T.A.; Allan, H.; Vinci, M.; Kramer-Marek, G.; Ciobota, D.M.; Parkinson, G.N.; Southworth, T.L.; Agliardi, G.; Hotblack, A.; et al. Near-infrared dual bioluminescence imaging in mouse models of cancer using infraluciferin. Elife 2019, 8, 1–22. [Google Scholar] [CrossRef]
- Zhang, C.; Yuan, M.; Han, G.; Gao, Y.; Tang, C.; Li, X.; Du, L.; Li, M. Novel caged luciferin derivatives can prolong bioluminescence imaging: In vitro and in vivo. RSC Adv. 2018, 8, 19596–19599. [Google Scholar] [CrossRef]
- Zambito, G.; Gaspar, N.; Ridwan, Y.; Hall, M.P.; Shi, C.; Kirkland, T.A.; Encell, L.P.; Löwik, C.; Mezzanotte, L. Evaluating brightness and spectral properties of click beetle and firefly luciferases using luciferin analogues: Identification of preferred pairings of luciferase and substrate for in vivo bioluminescence imaging. Mol. Imaging Biol. 2020, 22, 1523–1531. [Google Scholar] [CrossRef]
- Yao, Z.; Zhang, B.S.; Steinhardt, R.C.; Mills, J.H.; Prescher, J.A. Multicomponent bioluminescence imaging with a π-extended luciferin. J. Am. Chem. Soc. 2020, 142, 14080–14089. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.A.; Porterfield, W.B.; Rathbun, C.M.; McCutcheon, D.C.; Paley, M.A.; Prescher, J.A. Orthogonal luciferase–luciferin pairs for bioluminescence imaging. J. Am. Chem. Soc. 2017, 139, 2351–2358. [Google Scholar] [CrossRef] [PubMed]
- Rathbun, C.M.; Porterfield, W.B.; Jones, K.A.; Sagoe, M.J.; Reyes, M.R.; Hua, C.T.; Prescher, J.A. Parallel screening for rapid identification of orthogonal bioluminescent tools. ACS Cent. Sci. 2017, 3, 1254–1261. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, Y.; Liu, Y.; Zheng, S.; Wang, X.; Zhao, J.; Yang, F.; Zhang, G.; Wang, C.; Chen, P.R. Time-resolved protein activation by proximal decaging in living systems. Nature 2019, 569, 509–513. [Google Scholar] [CrossRef]
- Chen, Z.; Boyken, S.E.; Jia, M.; Busch, F.; Flores-Solis, D.; Bick, M.J.; Lu, P.; VanAernum, Z.L.; Sahasrabuddhe, A.; Langan, R.A.; et al. Programmable design of orthogonal protein heterodimers. Nature 2019, 565, 106–111. [Google Scholar] [CrossRef]
- Dou, J.; Vorobieva, A.A.; Sheffler, W.; Doyle, L.A.; Park, H.; Bick, M.J.; Mao, B.; Foight, G.W.; Lee, M.Y.; Gagnon, L.A.; et al. De novo design of a fluorescence-activating β-barrel. Nature 2018, 561, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Miura, C.; Kiyama, M.; Iwano, S.; Ito, K.; Obata, R.; Hirano, T.; Maki, S.; Niwa, H. Synthesis and luminescence properties of biphenyl-type firefly luciferin analogs with a new, near-infrared light-emitting bioluminophore. Tetrahedron 2013, 69, 9726–9734. [Google Scholar] [CrossRef]
- Kiyama, M.; Iwano, S.; Otsuka, S.; Lu, S.W.; Obata, R.; Miyawaki, A.; Hirano, T.; Maki, S.A. Quantum yield improvement of red-light-emitting firefly luciferin analogues for in vivo bioluminescence imaging. Tetrahedron 2018, 74, 652–660. [Google Scholar] [CrossRef]
- Kitada, N.; Saitoh, T.; Ikeda, Y.; Iwano, S.; Obata, R.; Niwa, H.; Hirano, T.; Miyawaki, A.; Suzuki, K.; Nishiyama, S.; et al. Toward bioluminescence in the near-infrared region: Tuning the emission wavelength of firefly luciferin analogues by allyl substitution. Tetrahedron Lett. 2018, 59, 1087–1090. [Google Scholar] [CrossRef]
- Kitada, N.; Saito, R.; Obata, R.; Iwano, S.; Karube, K.; Miyawaki, A.; Hirano, T.; Maki, S.A. Development of near-infrared firefly luciferin analogue reacted with wild-type and mutant luciferases. Chirality 2020, 32, 922–931. [Google Scholar] [CrossRef]
- Aswendt, M.; Vogel, S.; Schäfer, C.; Jathoul, A.; Pule, M.; Hoehn, M. Quantitative in vivo dual-color bioluminescence imaging in the mouse brain. Neurophotonics 2019, 6, 025006. [Google Scholar] [CrossRef] [PubMed]
- Doi, M.; Sato, M.; Ohmiya, Y. In vivo simultaneous analysis of gene expression by dual-color luciferases in caenorhabditis elegans. Int. J. Mol. Sci. 2020, 22, 119. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Walker, J.R.; Park, Y.; Smith, T.P.; Liu, L.X.; Hall, M.P.; Labanieh, L.; Hurst, R.; Wang, D.C.; Encell, L.P.; et al. Novel NanoLuc substrates enable bright two-population bioluminescence imaging in animals. Nat. Methods 2020, 17, 852–860. [Google Scholar] [CrossRef] [PubMed]
| Scheme | Luciferase | Species | Co-Factors | References |
|---|---|---|---|---|
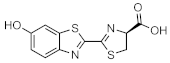 d-Luciferin (DLH2) | Firefly luciferase (Fluc) | Photinus pyralis | ATP, O2, Mg2+ | [1,2,3,4,5,6,7] |
| Click beetleluciferase (CBluc) | Pyrophorus plagiophthalamus | |||
 Coelenterazine (CTZ) | Gaussia luciferase (Gluc) | Gaussia princeps | O2 | [1,2,3,8,9,10] |
| Renilla luciferase (Rluc) | Renilla reniformas | |||
| Oplophorus luciferase (Oluc) | Oplophorus gracilirostris |
| Substrate | Luciferase | Wavelength | Km1 | Relative Intensity 2 | Target Organ | References |
|---|---|---|---|---|---|---|
 d-Luciferin(DLH2) | Firefly luciferase (luc, luc+, luc2) | 560 nm | 107 µM [58], 6.76 µM [72], 22.10 µM [73], 1 µM [74] | 1 | bone, brain, breast, lung, small intestine, subcutaneous | [15,16,18,19,20,21,24,28,29,30,31,32,33,34,35,36,37] |
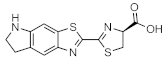 CycLuc1 | Firefly luciferase (luc, luc+, luc2) | 599 nm | 1.06 µM [58] 0.10 µM [72] | 0.7 (in vitro) 8 (brain) | brain | [57,75,76,77,78,79,80] |
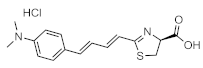 TokeOni (AkaLumine-HCl) | Firefly luciferase (luc, luc+, luc2) | 675 nm | 2.06 µM [58], 2.78 µM [81], 1.3 µM [82], 0.195 µM [83] | 0.05 (in vitro) 40 (subcutaneous) 8.1 (lung) | brain, lung, subcutaneous | [22,54,58,81,84,85,86,87,88] |
| AkaLuc (mutant) | 650 nm | 8.96 µM [81], 4.55 µM [89] | 10 (in cell) 1408 (brain) 52 (lung) | |||
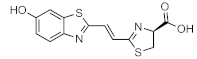 Infraluciferin (iLH2) | Fluc_red S284T Fluc (mutant) | 706 nm | 6.0 µM [90] 4 | 7.28 (intracranial) 4.1 (systemic) 42 (subcutaneous) | brain, lymphoma, subcutaneous | [90,91] |
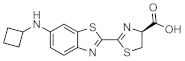 CybLuc | Firefly luciferase (luc, luc+, luc2) | 603 nm | 0.16 µM [73] | 18 (brain) 20 (subcutaneous) | brain, subcutaneous | [73,92] |
 NH2-NpLH2 | Firefly luciferase (luc, luc+, luc2) | No emission | – | – | brain, subcutaneous | [74] |
| CBR2opt (mutant) | 743 nm | 0.5 µM [74] | 0.3 (brain) 0.5 (subcutaneous) | |||
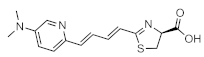 seMpai | Firefly luciferase (luc, luc+, luc2) | 675 nm | 6.2 µM [82] | 0.1 (in vitro) 0.7 (brain) 6 (lung) 1 (subcutaneous) | brain, breast, lung, subcutaneous | [82,86,89] |
| AkaLuc (mutant) | 640 nm | 44.9 µM [89] | 0.2 (in vitro) 3 | |||
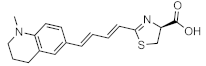 NIRLuc2 | Firefly luciferase (luc, luc+, luc2) | 683 nm | 1.05 µM [83] | 7 (subcutaneous) | subcutaneous | [83] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saito-Moriya, R.; Nakayama, J.; Kamiya, G.; Kitada, N.; Obata, R.; Maki, S.A.; Aoyama, H. How to Select Firefly Luciferin Analogues for In Vivo Imaging. Int. J. Mol. Sci. 2021, 22, 1848. https://doi.org/10.3390/ijms22041848
Saito-Moriya R, Nakayama J, Kamiya G, Kitada N, Obata R, Maki SA, Aoyama H. How to Select Firefly Luciferin Analogues for In Vivo Imaging. International Journal of Molecular Sciences. 2021; 22(4):1848. https://doi.org/10.3390/ijms22041848
Chicago/Turabian StyleSaito-Moriya, Ryohei, Jun Nakayama, Genta Kamiya, Nobuo Kitada, Rika Obata, Shojiro A. Maki, and Hiroshi Aoyama. 2021. "How to Select Firefly Luciferin Analogues for In Vivo Imaging" International Journal of Molecular Sciences 22, no. 4: 1848. https://doi.org/10.3390/ijms22041848
APA StyleSaito-Moriya, R., Nakayama, J., Kamiya, G., Kitada, N., Obata, R., Maki, S. A., & Aoyama, H. (2021). How to Select Firefly Luciferin Analogues for In Vivo Imaging. International Journal of Molecular Sciences, 22(4), 1848. https://doi.org/10.3390/ijms22041848







