A Review of the Current Mammalian Models of Alzheimer’s Disease and Challenges That Need to Be Overcome
Abstract
1. Introduction
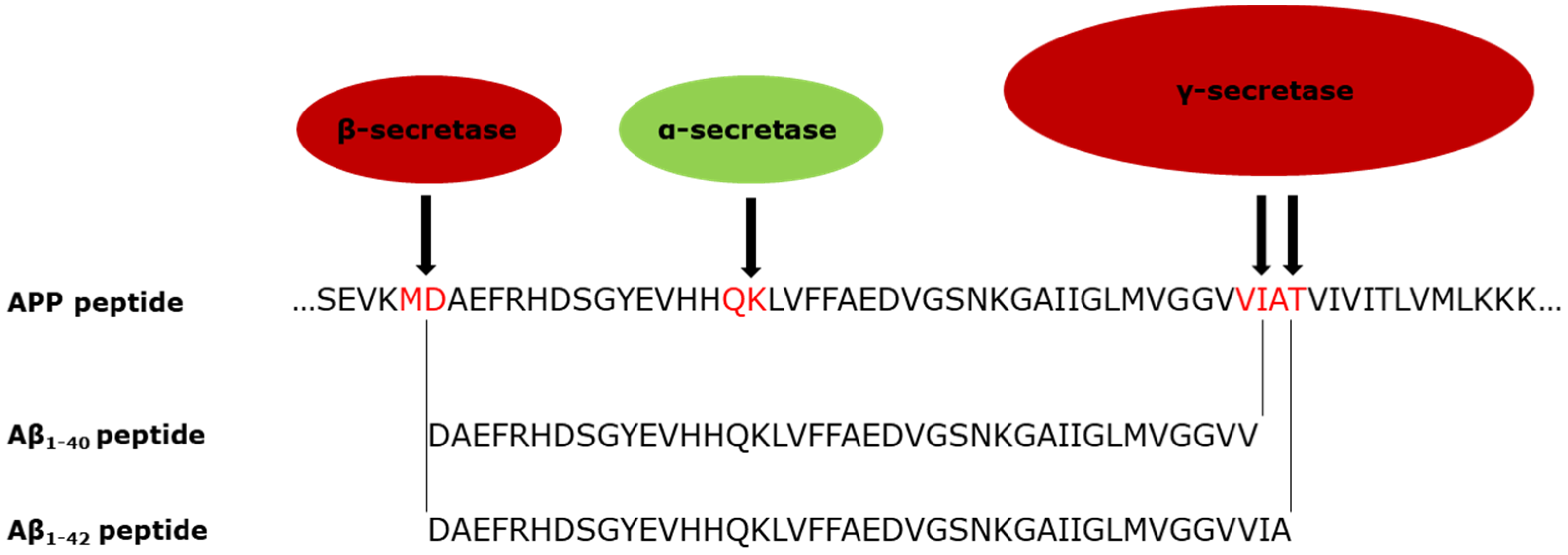
2. The Defining Pathological Features of AD; Plaques and Tangles
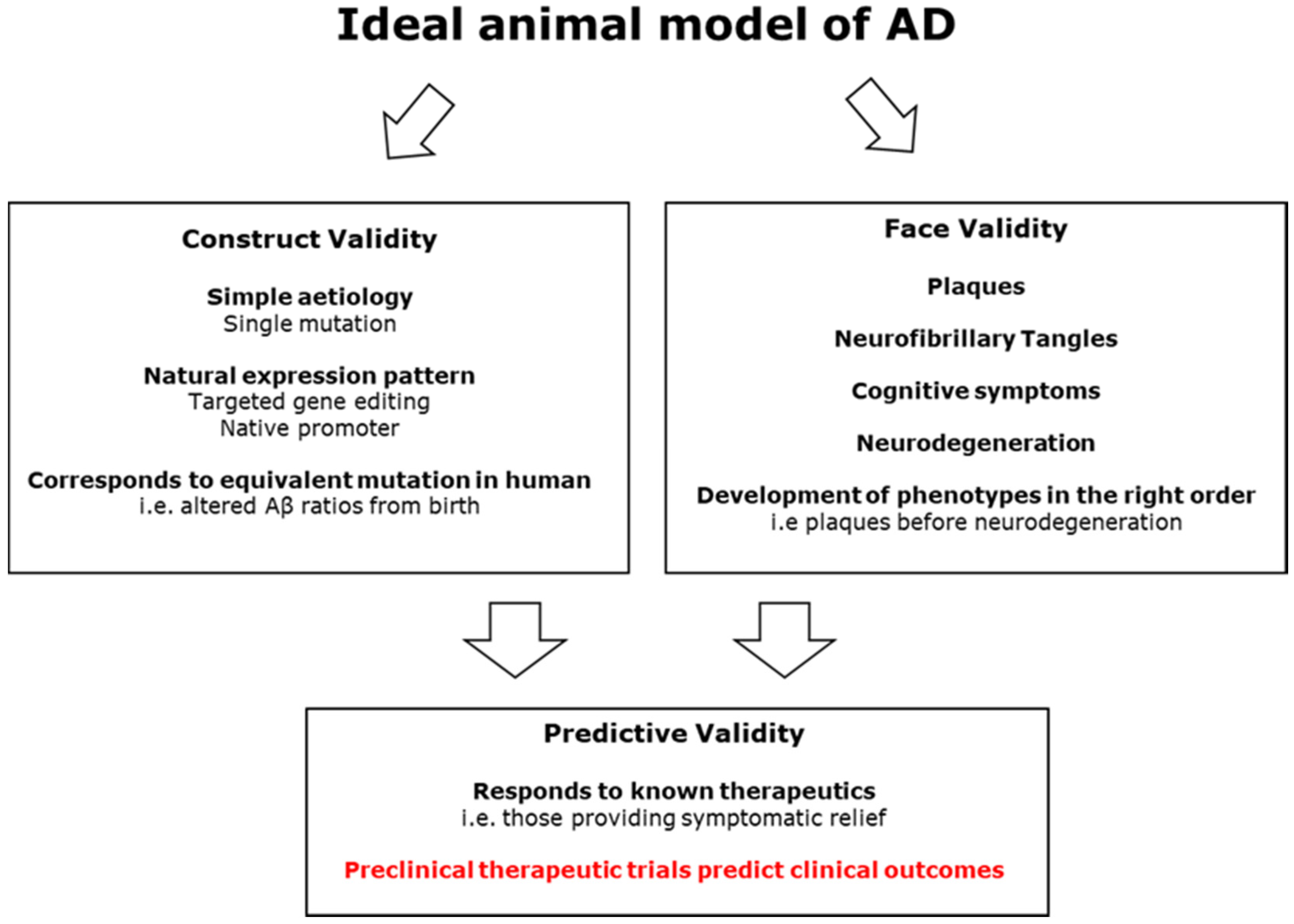
3. Modelling AD in Animals
4. Small Animal Models of AD
4.1. Mouse Models
4.1.1. Plaque Pathology in Mouse Models
4.1.2. Replicating AD Tau Pathology
4.1.3. Construct Validity of Transgenic Mouse Models of AD
4.1.4. Murine APP Knock in Models
4.1.5. Murine PSEN1 Knock in Models
4.1.6. Construct Validity of MAPT Mouse Models
4.1.7. Predictive Validity of Murine Models
4.1.8. Murine Model Summary
4.2. Rat Models of AD
5. Large Animal Models in AD Research
5.1. Primate Models of AD
5.2. Larger Non-Primate Mammalian Models
5.2.1. Larger Companion Animals
5.2.2. Farm Animals
5.2.3. Pigs
5.2.4. Sheep
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khachaturian, Z.S. Diagnosis of Alzheimer’s disease. Arch. Neurol. 1985, 42, 1097–1105. [Google Scholar] [CrossRef]
- Hebert, L.E.; Beckett, L.A.; Scherr, P.A.; Evans, D.A. Annual incidence of Alzheimer disease in the United States projected to the years 2000 through 2050. Alzheimer Dis. Assoc. Disord. 2001, 15, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Sloane, P.D.; Zimmerman, S.; Suchindran, C.; Reed, P.; Wang, L.; Boustani, M.; Sudha, S. The public health impact of Alzheimer’s disease, 2000–2050: Potential implication of treatment advances. Annu. Rev. Public Health 2002, 23, 213–231. [Google Scholar] [CrossRef]
- Fox, P.J.; Kohatsu, N.; Max, W.; Arnsberger, P. Estimating the costs of caring for people with Alzheimer disease in California: 2000–2040. J. Public Health Policy 2001, 22, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Katzman, R.; Fox, P. The World-Wide Impact of Dementia. Projections of Prevalance and Costs. In Epidemiology of Alzheimer’s Disease: From Gene to Prevention; Springer: Berlin, Germany, 1999; pp. 1–17. [Google Scholar]
- Tagarelli, A.; Piro, A.; Tagarelli, G.; Lagonia, P.; Quattrone, A. Alois Alzheimer: A hundred years after the discovery of the eponymous disorder. Int. J. Biomed. Sci. 2006, 2, 196. [Google Scholar]
- Glenner, G.G.; Wong, C.W. Alzheimer’s disease: Initial report of the purification and characterization of a novel cerebrovascular amyloid protein. Biochem. Biophys. Res. Commun. 1984, 120, 885–890. [Google Scholar] [CrossRef]
- Goedert, M.; Spillantini, M.G. A century of Alzheimer’s disease. Science 2006, 314, 777–781. [Google Scholar] [CrossRef]
- Kidd, M. Paired helical filaments in electron microscopy of Alzheimer’s disease. Nature 1963, 197, 192–193. [Google Scholar] [CrossRef]
- Cummings, J.; Lee, G.; Zhong, K.; Fonseca, J.; Taghva, K. Alzheimer’s disease drug development pipeline: 2021. Alzheimer’s Dement. Transl. Res. Clin. Interv. 2021, 7, e12179. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.; Feldman, H.H.; Scheltens, P. The “rights” of precision drug development for Alzheimer’s disease. Alzheimer’s Res. Ther. 2019, 11, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.L.; Morstorf, T.; Zhong, K. Alzheimer’s disease drug-development pipeline: Few candidates, frequent failures. Alzheimer’s Res. Ther. 2014, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Glenner, G.G.; Wong, C.W. Alzheimer’s disease and Down’s syndrome: Sharing of a unique cerebrovascular amyloid fibril protein. Biochem. Biophys. Res. Commun. 1984, 122, 1131–1135. [Google Scholar] [CrossRef]
- Masters, C.L.; Simms, G.; Weinman, N.A.; Multhaup, G.; McDonald, B.L.; Beyreuther, K. Amyloid plaque core protein in Alzheimer disease and Down syndrome. Proc. Natl. Acad. Sci. USA 1985, 82, 4245–4249. [Google Scholar] [CrossRef]
- Masters, C.L.; Beyreuther, K. Pathways to the discovery of the Aβ amyloid of Alzheimer’s disease. J. Alzheimer’s Dis. 2006, 9, 155–161. [Google Scholar] [CrossRef]
- Miller, D.L.; Papayannopoulos, I.A.; Styles, J.; Bobin, S.A.; Lin, Y.Y.; Biemann, K.; Iqbal, K. Peptide compositions of the cerebrovascular and senile plaque core amyloid deposits of Alzheimer’ s disease. Arch. Biochem. Biophys. 1993, 301, 41–52. [Google Scholar] [CrossRef]
- Nunan, J.; Small, D.H. Regulation of APP cleavage by α-, β-and γ-secretases. FEBS Lett. 2000, 483, 6–10. [Google Scholar] [CrossRef]
- Haass, C.; Selkoe, D.J. Soluble protein oligomers in neurodegeneration: Lessons from the Alzheimer’s amyloid [beta]-peptide. Nat. Rev. Mol. Cell Biol. 2007, 8, 101. [Google Scholar] [CrossRef]
- Chow, V.W.; Mattson, M.P.; Wong, P.C.; Gleichmann, M. An overview of APP processing enzymes and products. Neuromol. Med. 2010, 12, 1–12. [Google Scholar] [CrossRef]
- Reid, S.J.; van Roon-Mom, W.M.; Wood, P.C.; Rees, M.I.; Owen, M.J.; Faull, R.L.; Dragunow, M.; Snell, R.G. TBP, a polyglutamine tract containing protein, accumulates in Alzheimer’s disease. Mol. Brain Res. 2004, 125, 120–128. [Google Scholar] [CrossRef]
- Alonso, A.d.C.; Grundke-Iqbal, I.; Iqbal, K. Alzheimer’s disease hyperphosphorylated tau sequesters normal tau into tangles of filaments and disassembles microtubules. Nat. Med. 1996, 2, 783–787. [Google Scholar] [CrossRef] [PubMed]
- Qian, J.; Hyman, B.T.; Betensky, R.A. Neurofibrillary tangle stage and the rate of progression of Alzheimer symptoms: Modeling using an autopsy cohort and application to clinical trial design. JAMA Neurol. 2017, 74, 540–548. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Isla, T.; Hollister, R.; West, H.; Mui, S.; Growdon, J.H.; Petersen, R.C.; Parisi, J.E.; Hyman, B.T. Neuronal loss correlates with but exceeds neurofibrillary tangles in Alzheimer’s disease. Ann. Neurol. Off. J. Am. Neurol. Assoc. Child Neurol. Soc. 1997, 41, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.A.; Higgins, G.A. Alzheimer’s disease: The amyloid cascade hypothesis. Science 1992, 256, 184. [Google Scholar] [CrossRef]
- Hardy, J. Alzheimer’s disease: The amyloid cascade hypothesis: An update and reappraisal. J. Alzheimer’s Dis. 2006, 9, 151–153. [Google Scholar] [CrossRef]
- Hardy, J.; Selkoe, D.J. The amyloid hypothesis of Alzheimer’s disease: Progress and problems on the road to therapeutics. Science 2002, 297, 353–356. [Google Scholar] [CrossRef] [PubMed]
- Campion, D.; Dumanchin, C.; Hannequin, D.; Dubois, B.; Belliard, S.; Puel, M.; Thomas-Anterion, C.; Michon, A.; Martin, C.; Charbonnier, F. Early-onset autosomal dominant Alzheimer disease: Prevalence, genetic heterogeneity, and mutation spectrum. Am. J. Hum. Genet. 1999, 65, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Selkoe, D.J. Alzheimer’s disease: Genes, proteins, and therapy. Physiol. Rev. 2001, 81, 741–766. [Google Scholar] [CrossRef]
- Berchtold, N.C.; Cotman, C.W. Evolution in the conceptualization of dementia and Alzheimer’s disease: Greco-Roman period to the 1960s. Neurobiol. Aging 1998, 19, 173–189. [Google Scholar] [CrossRef]
- Takasugi, N.; Tomita, T.; Hayashi, I.; Tsuruoka, M. The role of presenilin cofactors in the gamma-secretase complex. Nature 2003, 422, 438. [Google Scholar] [CrossRef]
- Wu, L.; Rosa-Neto, P.; Hsiung, G.-Y.R.; Sadovnick, A.D.; Masellis, M.; Black, S.E.; Jia, J.; Gauthier, S. Early-onset familial Alzheimer’s disease (EOFAD). Can. J. Neurol. Sci. 2012, 39, 436–445. [Google Scholar] [CrossRef]
- Levy-Lahad, E.; Bird, T.D. Genetic factors in Alzheimer’s disease: A review of recent advances. Ann. Neurol. 1996, 40, 829–840. [Google Scholar] [CrossRef]
- Scheuner, D.; Eckman, C.; Jensen, M.; Song, X.; Citron, M.; Suzuki, N.; Bird, T.D.; Hardy, J.; Hutton, M.; Kukull, W.; et al. Secreted amyloid beta-protein similar to that in the senile plaques of Alzheimer’s disease is increased in vivo by the presenilin 1 and 2 and APP mutations linked to familial Alzheimer’s disease. Nat. Med. 1996, 2, 864–870. [Google Scholar] [CrossRef] [PubMed]
- Bellenguez, C.; Küçükali, F.; Jansen, I.; Andrade, V.; Moreno-Grau, S.; Amin, N.; Naj, A.C.; Grenier-Boley, B.; Campos-Martin, R.; Holmans, P.A. New insights on the genetic etiology of Alzheimer’s and related dementia. MedRxiv 2020. [Google Scholar] [CrossRef]
- Gatz, M.; Reynolds, C.A.; Fratiglioni, L.; Johansson, B.; Mortimer, J.A.; Berg, S.; Fiske, A.; Pedersen, N.L. Role of genes and environments for explaining Alzheimer disease. Arch. Gen. Psychiatry 2006, 63, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Coon, K.D.; Myers, A.J.; Craig, D.W.; Webster, J.A.; Pearson, J.V.; Lince, D.H.; Zismann, V.L.; Beach, T.G.; Leung, D.; Bryden, L. A high-density whole-genome association study reveals that APOE is the major susceptibility gene for sporadic late-onset Alzheimer’s disease. J. Clin. Psychiatry 2007, 68, 613–618. [Google Scholar] [CrossRef] [PubMed]
- Hollingworth, P.; Harold, D.; Sims, R.; Gerrish, A.; Lambert, J.C.; Carrasquillo, M.M.; Abraham, R.; Hamshere, M.L.; Pahwa, J.S.; Moskvina, V.; et al. Common variants at ABCA7, MS4A6A/MS4A4E, EPHA1, CD33 and CD2AP are associated with Alzheimer’s disease. Nat. Genet. 2011, 43, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Naj, A.C.; Jun, G.; Beecham, G.W.; Wang, L.S.; Vardarajan, B.N.; Buros, J.; Gallins, P.J.; Buxbaum, J.D.; Jarvik, G.P.; Crane, P.K.; et al. Common variants at MS4A4/MS4A6E, CD2AP, CD33 and EPHA1 are associated with late-onset Alzheimer’s disease. Nat. Genet. 2011, 43, 436–441. [Google Scholar] [CrossRef] [PubMed]
- Guerreiro, R.; Wojtas, A.; Bras, J.; Carrasquillo, M.; Rogaeva, E.; Majounie, E.; Cruchaga, C.; Sassi, C.; Kauwe, J.S.; Younkin, S. TREM2 variants in Alzheimer’s disease. N. Engl. J. Med. 2013, 368, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Justice, M.J.; Dhillon, P. Using the Mouse to Model Human Disease: Increasing Validity and Reproducibility; The Company of Biologists Ltd.: Cambridge, UK, 2016. [Google Scholar]
- Toledano, A.; Alvarez, M.I. Lesion-Induced Vertebrate Models of Alzheimer Dementia. Neuromethods 2011, 48, 295–345. [Google Scholar] [CrossRef]
- Beach, T.G.; Potter, P.E.; Kuo, Y.M.; Emmerling, M.R.; Durham, R.A.; Webster, S.D.; Walker, D.G.; Sue, L.I.; Scott, S.; Layne, K.J.; et al. Cholinergic deafferentation of the rabbit cortex: A new animal model of A beta deposition. Neurosci. Lett. 2000, 283, 9–12. [Google Scholar] [CrossRef]
- Wenk, G.L. A Primate Model of Alzheimers-Disease. Behav. Brain Res. 1993, 57, 117–122. [Google Scholar] [CrossRef]
- Ridley, R.M.; Murray, T.K.; Johnson, J.A.; Baker, H.F. Learning Impairment Following Lesion of the Basal Nucleus of Meynert in the Marmoset—Modification by Cholinergic Drugs. Brain Res. 1986, 376, 108–116. [Google Scholar] [CrossRef]
- Coyle, J.T.; Price, D.L.; Delong, M.R. Alzheimers-Disease—A Disorder of Cortical Cholinergic Innervation. Science 1983, 219, 1184–1190. [Google Scholar] [CrossRef]
- Whitehouse, P.J.; Price, D.L.; Struble, R.G.; Clark, A.W.; Coyle, J.T.; Delong, M.R. Alzheimers-Disease and Senile Dementia—Loss of Neurons in the Basal Forebrain. Science 1982, 215, 1237–1239. [Google Scholar] [CrossRef] [PubMed]
- McGonigle, P. Animal models of CNS disorders. Biochem. Pharm. 2014, 87, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Games, D.; Adams, D.; Alessandrini, R.; Barbour, R.; Borthelette, P.; Blackwell, C.; Carr, T.; Clemens, J.; Donaldson, T.; Gillespie, F. Alzheimer-type neuropathology in transgenic mice overexpressing V717F β-amyloid precursor protein. Nature 1995, 373, 523–527. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, K.; Chapman, P.; Nilsen, S.; Eckman, C. Correlative memory deficits, A beta elevation, and amyloid plaques in transgenic mice. Science 1996, 274, 98. [Google Scholar] [CrossRef]
- Chishti, M.A.; Yang, D.-S.; Janus, C.; Phinney, A.L.; Horne, P.; Pearson, J.; Strome, R.; Zuker, N.; Loukides, J.; French, J. Early-onset amyloid deposition and cognitive deficits in transgenic mice expressing a double mutant form of amyloid precursor protein 695. J. Biol. Chem. 2001, 276, 21562–21570. [Google Scholar] [CrossRef]
- Holcomb, L.; Gordon, M.N.; McGowan, E.; Yu, X.; Benkovic, S.; Jantzen, P.; Wright, K.; Saad, I.; Mueller, R.; Morgan, D. Accelerated Alzheimer-type phenotype in transgenic mice carrying both mutant amyloid precursor protein and presenilin 1 transgenes. Nat. Med. 1998, 4, 97–100. [Google Scholar] [CrossRef]
- McGowan, E.; Pickford, F.; Kim, J.; Onstead, L.; Eriksen, J.; Yu, C.; Skipper, L.; Murphy, M.P.; Beard, J.; Das, P. Aβ42 is essential for parenchymal and vascular amyloid deposition in mice. Neuron 2005, 47, 191–199. [Google Scholar] [CrossRef]
- Oakley, H.; Cole, S.L.; Logan, S.; Maus, E.; Shao, P.; Craft, J.; Guillozet-Bongaarts, A.; Ohno, M.; Disterhoft, J.; Van Eldik, L. Intraneuronal β-amyloid aggregates, neurodegeneration, and neuron loss in transgenic mice with five familial Alzheimer’s disease mutations: Potential factors in amyloid plaque formation. J. Neurosci. 2006, 26, 10129–10140. [Google Scholar] [CrossRef]
- Lewis, J.; McGowan, E.; Rockwood, J.; Melrose, H.; Nacharaju, P.; Van Slegtenhorst, M.; Gwinn-Hardy, K.; Murphy, M.P.; Baker, M.; Yu, X. Neurofibrillary tangles, amyotrophy and progressive motor disturbance in mice expressing mutant (P301L) tau protein. Nat. Genet. 2000, 25, 402–405. [Google Scholar] [CrossRef] [PubMed]
- Ramsden, M.; Kotilinek, L.; Forster, C.; Paulson, J.; McGowan, E.; SantaCruz, K.; Guimaraes, A.; Yue, M.; Lewis, J.; Carlson, G.; et al. Age-dependent neurofibrillary tangle formation, neuron loss, and memory impairment in a mouse model of human tauopathy (P301L). J. Neurosci. 2005, 25, 10637–10647. [Google Scholar] [CrossRef] [PubMed]
- Oddo, S.; Caccamo, A.; Kitazawa, M.; Tseng, B.P.; LaFerla, F.M. Amyloid deposition precedes tangle formation in a triple transgenic model of Alzheimer’s disease. Neurobiol. Aging 2003, 24, 1063–1070. [Google Scholar] [CrossRef]
- Lewis, J.; Dickson, D.W.; Lin, W.-L.; Chisholm, L.; Corral, A.; Jones, G.; Yen, S.-H.; Sahara, N.; Skipper, L.; Yager, D. Enhanced neurofibrillary degeneration in transgenic mice expressing mutant tau and APP. Science 2001, 293, 1487–1491. [Google Scholar] [CrossRef]
- Irizarry, M.C.; Soriano, F.; McNamara, M.; Page, K.J.; Schenk, D.; Games, D.; Hyman, B.T. Abeta deposition is associated with neuropil changes, but not with overt neuronal loss in the human amyloid precursor protein V717F (PDAPP) transgenic mouse. J. Neurosci. 1997, 17, 7053–7059. [Google Scholar] [CrossRef] [PubMed]
- King, D.L.; Arendash, G.W. Behavioral characterization of the Tg2576 transgenic model of Alzheimer’s disease through 19 months. Physiol. Behav. 2002, 75, 627–642. [Google Scholar] [CrossRef]
- Westerman, M.A.; Cooper-Blacketer, D.; Mariash, A.; Kotilinek, L.; Kawarabayashi, T.; Younkin, L.H.; Carlson, G.A.; Younkin, S.G.; Ashe, K.H. The relationship between Aβ and memory in the Tg2576 mouse model of Alzheimer’s disease. J. Neurosci. 2002, 22, 1858–1867. [Google Scholar] [CrossRef] [PubMed]
- Kotilinek, L.A.; Bacskai, B.; Westerman, M.; Kawarabayashi, T.; Younkin, L.; Hyman, B.T.; Younkin, S.; Ashe, K.H. Reversible memory loss in a mouse transgenic model of Alzheimer’s disease. J. Neurosci. 2002, 22, 6331–6335. [Google Scholar] [CrossRef]
- Janus, C.; Phinney, A.L.; Chishti, M.A.; Westaway, D. New developments in animal models of Alzheimer’s disease. Curr. Neurol. Neurosci. Rep. 2001, 1, 451–457. [Google Scholar] [CrossRef]
- Borchelt, D.R.; Thinakaran, G.; Eckman, C.B.; Lee, M.K.; Davenport, F.; Ratovitsky, T.; Prada, C.-M.; Kim, G.; Seekins, S.; Yager, D. Familial Alzheimer’s disease–linked presenilin 1 variants elevate Aβ1–42/1–40 ratio in vitro and in vivo. Neuron 1996, 17, 1005–1013. [Google Scholar] [CrossRef]
- Citron, M.; Westaway, D.; Xia, W.; Carlson, G.; Diehl, T.; Levesque, G.; Johnson-Wood, K.; Lee, M.; Seubert, P.; Davis, A.; et al. Mutant presenilins of Alzheimer’s disease increase production of 42-residue amyloid beta-protein in both transfected cells and transgenic mice. Nat. Med. 1997, 3, 67–72. [Google Scholar] [CrossRef]
- Duff, K.; Eckman, C.; Zehr, C.; Yu, X.; Prada, C.-M.; Perez-Tur, J.; Hutton, M.; Buee, L.; Harigaya, Y.; Yager, D. Increased amyloid-β42 (43) in brains of mice expressing mutant presenilin 1. Nature 1996, 383, 710–713. [Google Scholar] [CrossRef]
- Borchelt, D.R.; Ratovitski, T.; Van Lare, J.; Lee, M.K.; Gonzales, V.; Jenkins, N.A.; Copeland, N.G.; Price, D.L.; Sisodia, S.S. Accelerated amyloid deposition in the brains of transgenic mice coexpressing mutant presenilin 1 and amyloid precursor proteins. Neuron 1997, 19, 939–945. [Google Scholar] [CrossRef]
- Jawhar, S.; Trawicka, A.; Jenneckens, C.; Bayer, T.A.; Wirths, O. Motor deficits, neuron loss, and reduced anxiety coinciding with axonal degeneration and intraneuronal Abeta aggregation in the 5XFAD mouse model of Alzheimer’s disease. Neurobiol. Aging 2012, 33, 196.e29–196.e40. [Google Scholar] [CrossRef]
- Chen, F.; David, D.; Ferrari, A.; Gotz, J. Posttranslational modifications of tau—Role in human tauopathies and modeling in transgenic animals. Curr. Drug Targets 2004, 5, 503–515. [Google Scholar] [CrossRef]
- Rademakers, R.; Cruts, M.; van Broeckhoven, C. The role of tau (MAPT) in frontotemporal dementia and related tauopathies. Hum. Mutat. 2004, 24, 277–295. [Google Scholar] [CrossRef] [PubMed]
- Santacruz, K.; Lewis, J.; Spires, T.; Paulson, J.; Kotilinek, L.; Ingelsson, M.; Guimaraes, A.; Deture, M.; Ramsden, M.; McGowan, E. Tau suppression in a neurodegenerative mouse model improves memory function. Science 2005, 309, 476–481. [Google Scholar] [CrossRef]
- Eriksen, J.L.; Janus, C.G. Plaques, tangles, and memory loss in mouse models of neurodegeneration. Behav. Genet. 2007, 37, 79–100. [Google Scholar] [CrossRef]
- Yamada, T.; Sasaki, H.; Furuya, H.; Miyata, T.; Goto, I.; Sakaki, Y. Complementary DNA for the mouse homolog of the human amyloid beta protein precursor. Biochem. Biophys. Res. Commun. 1987, 149, 665–671. [Google Scholar] [CrossRef]
- Liu, K.; Doms, R.W.; Lee, V.M.-Y. Glu11 site cleavage and N-terminally truncated Aβ production upon BACE overexpression. Biochemistry 2002, 41, 3128–3136. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Wang, Y.; McCarthy, D.; Wen, H.; Borchelt, D.R.; Price, D.L.; Wong, P.C. BACE1 is the major [beta]-secretase for generation of A [beta] peptides by neurons. Nat. Neurosci. 2001, 4, 233. [Google Scholar] [CrossRef]
- Kawasumi, M.; Chiba, T.; Yamada, M.; Miyamae-Kaneko, M.; Matsuoka, M.; Nakahara, J.; Tomita, T.; Iwatsubo, T.; Kato, S.; Aiso, S.; et al. Targeted introduction of V642I mutation in amyloid precursor protein gene causes functional abnormality resembling early stage of Alzheimer’s disease in aged mice. Eur. J. Neurosci. 2004, 19, 2826–2838. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Shiojiri, S.; Takahashi, Y.; Kitaguchi, N.; Ito, H.; Kameyama, M.; Kimura, J.; Nakamura, S.; Ueda, K. Tissue-specific expression of three types of beta-protein precursor mRNA: Enhancement of protease inhibitor-harboring types in Alzheimer’s disease brain. Biochem. Biophys. Res. Commun. 1989, 165, 1406–1414. [Google Scholar] [CrossRef]
- Tanaka, S.; Liu, L.; Kimura, J.; Shiojiri, S.; Takahashi, Y.; Kitaguchi, N.; Nakamura, S.; Ueda, K. Age-related changes in the proportion of amyloid precursor protein mRNAs in Alzheimer’s disease and other neurological disorders. Brain Res. Mol. Brain Res. 1992, 15, 303–310. [Google Scholar] [CrossRef]
- Moir, R.D.; Lynch, T.; Bush, A.I.; Whyte, S.; Henry, A.; Portbury, S.; Multhaup, G.; Small, D.H.; Tanzi, R.E.; Beyreuther, K.; et al. Relative increase in Alzheimer’s disease of soluble forms of cerebral Abeta amyloid protein precursor containing the Kunitz protease inhibitory domain. J. Biol. Chem. 1998, 273, 5013–5019. [Google Scholar] [CrossRef]
- Higgins, L.S.; Catalano, R.; Quon, D.; Cordell, B. Transgenic mice expressing human beta-APP751, but not mice expressing beta-APP695, display early Alzheimer’s disease-like histopathology. Ann. N. Y. Acad. Sci. 1993, 695, 224–227. [Google Scholar] [CrossRef]
- Sasahara, M.; Fries, J.W.; Raines, E.W.; Gown, A.M.; Westrum, L.E.; Frosch, M.P.; Bonthron, D.T.; Ross, R.; Collins, T. PDGF B-chain in neurons of the central nervous system, posterior pituitary, and in a transgenic model. Cell 1991, 64, 217–227. [Google Scholar] [CrossRef]
- Caroni, P. Overexpression of growth-associated proteins in the neurons of adult transgenic mice. J. Neurosci. Methods 1997, 71, 3–9. [Google Scholar] [CrossRef]
- Asante, E.A.; Gowland, I.; Linehan, J.M.; Mahal, S.P.; Collinge, J. Expression pattern of a mini human PrP gene promoter in transgenic mice. Neurobiol. Dis. 2002, 10, 1–7. [Google Scholar] [CrossRef]
- Sturchler-Pierrat, C.; Abramowski, D.; Duke, M.; Wiederhold, K.-H.; Mistl, C.; Rothacher, S.; Ledermann, B.; Bürki, K.; Frey, P.; Paganetti, P.A. Two amyloid precursor protein transgenic mouse models with Alzheimer disease-like pathology. Proc. Natl. Acad. Sci. USA 1997, 94, 13287–13292. [Google Scholar] [CrossRef]
- Jankowsky, J.L.; Slunt, H.H.; Gonzales, V.; Savonenko, A.V.; Wen, J.C.; Jenkins, N.A.; Copeland, N.G.; Younkin, L.H.; Lester, H.A.; Younkin, S.G. Persistent amyloidosis following suppression of Aβ production in a transgenic model of Alzheimer disease. PLoS Med. 2005, 2, e355. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, M.; Johnson-Venkatesh, E.M.; Zhang, H.; Parent, J.M.; Sutton, M.A.; Umemori, H. Multiple forms of activity-dependent competition refine hippocampal circuits in vivo. Neuron 2011, 70, 1128–1142. [Google Scholar] [CrossRef]
- Liu, P.; Paulson, J.B.; Forster, C.L.; Shapiro, S.L.; Ashe, K.H.; Zahs, K.R. Characterization of a novel mouse model of Alzheimer’s disease—Amyloid pathology and unique β-Amyloid oligomer profile. PLoS ONE 2015, 10, e126317. [Google Scholar] [CrossRef] [PubMed]
- Reaume, A.G.; Howland, D.S.; Trusko, S.P.; Savage, M.J.; Lang, D.M.; Greenberg, B.D.; Siman, R.; Scott, R.W. Enhanced amyloidogenic processing of the β-amyloid precursor protein in gene-targeted mice bearing the Swedish familial Alzheimer’s disease mutations and a “humanized” Aβ sequence. J. Biol. Chem. 1996, 271, 23380–23388. [Google Scholar] [CrossRef]
- Li, H.; Guo, Q.; Inoue, T.; Polito, V.A.; Tabuchi, K.; Hammer, R.E.; Pautler, R.G.; Taffet, G.E.; Zheng, H. Vascular and parenchymal amyloid pathology in an Alzheimer disease knock-in mouse model: Interplay with cerebral blood flow. Mol. Neurodegener 2014, 9, 28. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Matsuba, Y.; Mihira, N.; Takano, J.; Nilsson, P.; Itohara, S.; Iwata, N.; Saido, T.C. Single App knock-in mouse models of Alzheimer’s disease. Nat. Neurosci. 2014, 17, 661–663. [Google Scholar] [CrossRef]
- Masuda, A.; Kobayashi, Y.; Kogo, N.; Saito, T.; Saido, T.C.; Itohara, S. Cognitive deficits in single App knock-in mouse models. Neurobiol. Learn. Mem. 2016, 135, 73–82. [Google Scholar] [CrossRef]
- Jankowsky, J.L.; Zheng, H. Practical considerations for choosing a mouse model of Alzheimer’s disease. Mol. Neurodegener 2017, 12, 89. [Google Scholar] [CrossRef]
- Anantharaman, M.; Tangpong, J.; Keller, J.N.; Murphy, M.P.; Markesbery, W.R.; Kiningham, K.K.; Clair, D.K.S. β-Amyloid mediated nitration of manganese superoxide dismutase: Implication for oxidative stress in a APPNLH/NLH X PS-1P264L/P264L double knock-in mouse model of Alzheimer’s disease. Am. J. Pathol. 2006, 168, 1608–1618. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; McNeil, E.; Dressler, L.; Siman, R. Long-lasting impairment in hippocampal neurogenesis associated with amyloid deposition in a knock-in mouse model of familial Alzheimer’s disease. Exp. Neurol. 2007, 204, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Matsuba, Y.; Yamazaki, N.; Hashimoto, S.; Saido, T.C. Calpain activation in Alzheimer’s model mice is an artifact of APP and presenilin overexpression. J. Neurosci. 2016, 36, 9933–9936. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, S.; Ishii, A.; Kamano, N.; Watamura, N.; Saito, T.; Ohshima, T.; Yokosuka, M.; Saido, T.C. Endoplasmic reticulum stress responses in mouse models of Alzheimer’s disease: Overexpression paradigm versus knockin paradigm. J. Biol. Chem. 2018, 293, 3118–3125. [Google Scholar] [CrossRef] [PubMed]
- Drummond, E.; Wisniewski, T. Alzheimer’s disease: Experimental models and reality. Acta Neuropathol. 2017, 133, 155–175. [Google Scholar] [CrossRef]
- Dickson, D.W.; Crystal, H.A.; Mattiace, L.A.; Masur, D.M.; Blau, A.D.; Davies, P.; Yen, S.-H.; Aronson, M.K. Identification of normal and pathological aging in prospectively studied nondemented elderly humans. Neurobiol. Aging 1992, 13, 179–189. [Google Scholar] [CrossRef]
- Nakano, Y.; Kondoh, G.; Kudo, T.; Imaizumi, K.; Kato, M.; Miyazaki, J.i.; Tohyama, M.; Takeda, J.; Takeda, M. Accumulation of murine amyloidβ42 in a gene-dosage-dependent manner in PS1 ‘knock-in’mice. Eur. J. Neurosci. 1999, 11, 2577–2581. [Google Scholar] [CrossRef]
- Flood, D.G.; Reaume, A.G.; Dorfman, K.S.; Lin, Y.-G.; Lang, D.M.; Trusko, S.P.; Savage, M.J.; Annaert, W.G.; De Strooper, B.; Siman, R. FAD mutant PS-1 gene-targeted mice: Increased Aβ42 and Aβ deposition without APP overproduction. Neurobiol. Aging 2002, 23, 335–348. [Google Scholar] [CrossRef]
- Holcomb, L.A.; Gordon, M.N.; Jantzen, P.; Hsiao, K.; Duff, K.; Morgan, D. Behavioral changes in transgenic mice expressing both amyloid precursor protein and presenilin-1 mutations: Lack of association with amyloid deposits. Behav. Genet. 1999, 29, 177–185. [Google Scholar] [CrossRef]
- Huang, X.; Yee, B.; Nag, S.; Chan, S.; Tang, F. Behavioral and neurochemical characterization of transgenic mice carrying the human presenilin-1 gene with or without the leucine-to-proline mutation at codon 235. Exp. Neurol. 2003, 183, 673–681. [Google Scholar] [CrossRef]
- Janus, C.; D’Amelio, S.; Amitay, O.; Chishti, M.; Strome, R.; Fraser, P.; Carlson, G.; Roder, J.; George–Hyslop, P.S.; Westaway, D. Spatial learning in transgenic mice expressing human presenilin 1 (PS1) transgenes. Neurobiol. Aging 2000, 21, 541–549. [Google Scholar] [CrossRef]
- Dineley, K.T.; Xia, X.; Bui, D.; Sweatt, J.D.; Zheng, H. Accelerated plaque accumulation, associative learning deficits, and up-regulation of α7 nicotinic receptor protein in transgenic mice co-expressing mutant human presenilin 1 and amyloid precursor proteins. J. Biol. Chem. 2002, 277, 22768–22780. [Google Scholar] [CrossRef] [PubMed]
- Jankowsky, J.L.; Fadale, D.J.; Anderson, J.; Xu, G.M.; Gonzales, V.; Jenkins, N.A.; Copeland, N.G.; Lee, M.K.; Younkin, L.H.; Wagner, S.L. Mutant presenilins specifically elevate the levels of the 42 residue β-amyloid peptide in vivo: Evidence for augmentation of a 42-specific γ secretase. Hum. Mol. Genet. 2004, 13, 159–170. [Google Scholar] [CrossRef]
- Casas, C.; Sergeant, N.; Itier, J.-M.; Blanchard, V.; Wirths, O.; van der Kolk, N.; Vingtdeux, V.; van de Steeg, E.; Ret, G.; Canton, T. Massive CA1/2 neuronal loss with intraneuronal and N-terminal truncated Aβ 42 accumulation in a novel Alzheimer transgenic model. Am. J. Pathol. 2004, 165, 1289–1300. [Google Scholar] [CrossRef]
- Goedert, M.; Spillantini, M.G.; Jakes, R.; Rutherford, D.; Crowther, R.A. Multiple Isoforms of Human Microtubule-Associated Protein-Tau—Sequences and Localization in Neurofibrillary Tangles of Alzheimers-Disease. Neuron 1989, 3, 519–526. [Google Scholar] [CrossRef]
- Hampel, H.; Blennow, K.; Shaw, L.M.; Hoessler, Y.C.; Zetterberg, H.; Trojanowski, J.Q. Total and phosphorylated tau protein as biological markers of Alzheimer’s disease. Exp. Gerontol. 2010, 45, 30–40. [Google Scholar] [CrossRef] [PubMed]
- McMillan, P.; Korvatska, E.; Poorkaj, P.; Evstafjeva, Z.; Robinson, L.; Greenup, L.; Leverenz, J.; Schellenberg, G.D.; D’Souza, I. Tau Isoform Regulation Is Region- and Cell-Specific in Mouse Brain. J. Comp. Neurol. 2008, 511, 788–803. [Google Scholar] [CrossRef]
- Liu, C.; Götz, J. Profiling murine tau with 0N, 1N and 2N isoform-specific antibodies in brain and peripheral organs reveals distinct subcellular localization, with the 1N isoform being enriched in the nucleus. PLoS ONE 2013, 8, e84849. [Google Scholar] [CrossRef]
- Roberson, E.D.; Scearce-Levie, K.; Palop, J.J.; Yan, F.; Cheng, I.H.; Wu, T.; Gerstein, H.; Yu, G.-Q.; Mucke, L. Reducing endogenous tau ameliorates amyloid ß-induced deficits in an Alzheimer’s disease mouse model. Science 2007, 316, 750–754. [Google Scholar] [CrossRef]
- Wegmann, S.; Maury, E.A.; Kirk, M.J.; Saqran, L.; Roe, A.; DeVos, S.L.; Nicholls, S.; Fan, Z.; Takeda, S.; Cagsal-Getkin, O. Removing endogenous tau does not prevent tau propagation yet reduces its neurotoxicity. EMBO J. 2015, 34, 3028–3041. [Google Scholar] [CrossRef] [PubMed]
- Sabbagh, J.J.; Kinney, J.W.; Cummings, J.L. Animal systems in the development of treatments for Alzheimer’s disease: Challenges, methods, and implications. Neurobiol. Aging 2013, 34, 169–183. [Google Scholar] [CrossRef]
- Mullane, K.; Williams, M. Alzheimer’s therapeutics: Continued clinical failures question the validity of the amyloid hypothesis-but what lies beyond? Biochem. Pharm. 2013, 85, 289–305. [Google Scholar] [CrossRef]
- Allen, B.; Ingram, E.; Takao, M.; Smith, M.J.; Jakes, R.; Virdee, K.; Yoshida, H.; Holzer, M.; Craxton, M.; Emson, P.C. Abundant tau filaments and nonapoptotic neurodegeneration in transgenic mice expressing human P301S tau protein. J. Neurosci. 2002, 22, 9340–9351. [Google Scholar] [CrossRef]
- Förstl, H.; Kurz, A. Clinical features of Alzheimer’s disease. Eur. Arch. Psychiatry Clin. Neurosci. 1999, 249, 288–290. [Google Scholar] [CrossRef] [PubMed]
- Perel, P.; Roberts, I.; Sena, E.; Wheble, P.; Briscoe, C.; Sandercock, P.; Macleod, M.; Mignini, L.E.; Jayaram, P.; Khan, K.S. Comparison of treatment effects between animal experiments and clinical trials: Systematic review. Br. Med. J. 2007, 334, 197–200. [Google Scholar] [CrossRef]
- Ioannidis, J.P.A. Extrapolating from Animals to Humans. Sci. Transl. Med. 2012, 4, 151ps15. [Google Scholar] [CrossRef] [PubMed]
- Holmes, C.; Boche, D.; Wilkinson, D.; Yadegarfar, G.; Hopkins, V.; Bayer, A.; Jones, R.W.; Bullock, R.; Love, S.; Neal, J.W.; et al. Long-term effects of A beta(42) immunisation in Alzheimer’s disease: Follow-up of a randomised, placebo-controlled phase I trial. Lancet 2008, 372, 216–223. [Google Scholar] [CrossRef]
- Gilman, S.; Koller, M.; Black, R.S.; Jenkins, L.; Griffith, S.G.; Fox, N.C.; Eisner, L.; Kirby, L.; Rovira, M.B.; Forette, F.; et al. Clinical effects of A beta immunization (AN1792) in patients with AD in an interrupted trial. Neurology 2005, 64, 1553–1562. [Google Scholar] [CrossRef]
- Bard, F.; Cannon, C.; Barbour, R.; Burke, R.L.; Games, D.; Grajeda, H.; Guido, T.; Hu, K.; Huang, J.P.; Johnson-Wood, K.; et al. Peripherally administered antibodies against amyloid beta-peptide enter the central nervous system and reduce pathology in a mouse model of Alzheimer disease. Nat. Med. 2000, 6, 916–919. [Google Scholar] [CrossRef] [PubMed]
- Dodart, J.C.; Bales, K.R.; Gannon, K.S.; Greene, S.J.; DeMattos, R.B.; Mathis, C.; DeLong, C.A.; Wu, S.; Wu, X.; Holtzman, D.M.; et al. Immunization reverses memory deficits without reducing brain A beta burden in Alzheimer’s disease model. Nat. Neurosci. 2002, 5, 452–457. [Google Scholar] [CrossRef]
- Salloway, S.P.; Black, R.; Sperling, R.; Fox, N.; Gilman, S.; Schenk, D.; Grundman, M. A Phase 2 Multiple Ascending Dose Trial of Bapineuzumab in Mild to Moderate Alzheimer Disease Reply. Neurology 2010, 74, 2026–2027. [Google Scholar]
- Farlow, M.; Amold, S.E.; van Dyck, C.H.; Aisen, P.S.; Snider, B.J.; Porsteinsson, A.P.; Friedrich, S.; Dean, R.A.; Gonzales, C.; Sethuraman, G.; et al. Safety and biomarker effects of Solanezumab in patients with Alzheimer’s disease. Alzheimer’s Dement. 2012, 8, 261–271. [Google Scholar] [CrossRef]
- Doody, R.S.; Thomas, R.G.; Farlow, M.; Iwatsubo, T.; Vellas, B.; Joffe, S.; Kieburtz, K.; Raman, R.; Sun, X.Y.; Aisen, P.S.; et al. Phase 3 Trials of Solanezumab for Mild-to-Moderate Alzheimer’s Disease. N. Engl. J. Med. 2014, 370, 311–321. [Google Scholar] [CrossRef] [PubMed]
- Abyadeh, M.; Gupta, V.; Gupta, V.; Chitranshi, N.; Wu, Y.; Amirkhani, A.; Meyfour, A.; Sheriff, S.; Shen, T.; Dhiman, K. Comparative Analysis of Aducanumab, Zagotenemab and Pioglitazone as Targeted Treatment Strategies for Alzheimer’s Disease. Aging Dis. 2022, 12, 1964–1976. [Google Scholar]
- Holtzman, D.M.; Bales, K.R.; Tenkova, T.; Fagan, A.M.; Parsadanian, M.; Sartorius, L.J.; Mackey, B.; Olney, J.; McKeel, D.; Wozniak, D. Apolipoprotein E isoform-dependent amyloid deposition and neuritic degeneration in a mouse model of Alzheimer’s disease. Proc. Natl. Acad. Sci. USA 2000, 97, 2892–2897. [Google Scholar] [CrossRef] [PubMed]
- Jay, T.R.; Hirsch, A.M.; Broihier, M.L.; Miller, C.M.; Neilson, L.E.; Ransohoff, R.M.; Lamb, B.T.; Landreth, G.E. Disease progression-dependent effects of TREM2 deficiency in a mouse model of Alzheimer’s disease. J. Neurosci. 2017, 37, 637–647. [Google Scholar] [CrossRef]
- Lewandowski, C.T.; Weng, J.M.; LaDu, M.J. Alzheimer’s disease pathology in APOE transgenic mouse models: The Who, What, When, Where, Why, and How. Neurobiol. Dis. 2020, 139, 104811. [Google Scholar] [CrossRef]
- Do Carmo, S.; Cuello, A.C. Modeling Alzheimer’s disease in transgenic rats. Mol. Neurodegener 2013, 8, 37. [Google Scholar] [CrossRef]
- Lin, J.H. Species similarities and differences in pharmacokinetics. Drug Metab. Dispos. 1995, 23, 1008–1021. [Google Scholar]
- Jacob, H.J.; Kwitek, A.E. Rat genetics: Attachign physiology and pharmacology to the genome. Nat. Rev. Genet. 2002, 3, 33–42. [Google Scholar] [CrossRef]
- Echeverria, V.; Ducatenzeiler, A.; Alhonen, L.; Janne, J.; Grant, S.M.; Wandosell, F.; Muro, A.; Baralle, F.; Li, H.S.; Duff, K.; et al. Rat transgenic models with a phenotype of intracellular A beta accumulation in hippocampus and cortex. J. Alzheimer’s Dis. 2004, 6, 209–219. [Google Scholar] [CrossRef]
- Leon, W.C.; Canneva, F.; Partridge, V.; Allard, S.; Ferretti, M.T.; DeWilde, A.; Vercauteren, F.; Atifeh, R.; Ducatenzeiler, A.; Klein, W. A novel transgenic rat model with a full Alzheimer’s-like amyloid pathology displays pre-plaque intracellular amyloid-β-associated cognitive impairment. J. Alzheimer’s Dis. 2010, 20, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Flood, D.G.; Lin, Y.-G.; Lang, D.M.; Trusko, S.P.; Hirsch, J.D.; Savage, M.J.; Scott, R.W.; Howland, D.S. A transgenic rat model of Alzheimer’s disease with extracellular Aβ deposition. Neurobiol. Aging 2009, 30, 1078–1090. [Google Scholar] [CrossRef]
- Cohen, R.M.; Rezai-Zadeh, K.; Weitz, T.M.; Rentsendorj, A.; Gate, D.; Spivak, I.; Bholat, Y.; Vasilevko, V.; Glabe, C.G.; Breunig, J.J.; et al. A Transgenic Alzheimer Rat with Plaques, Tau Pathology, Behavioral Impairment, Oligomeric A beta, and Frank Neuronal Loss. J. Neurosci. 2013, 33, 6245–6256. [Google Scholar] [CrossRef]
- Hanes, J.; Zilka, N.; Bartkova, M.; Caletkova, M.; Dobrota, D.; Novak, M. Rat tau proteome consists of six tau isoforms: Implication for animal models of human tauopathies. J. Neurochem. 2009, 108, 1167–1176. [Google Scholar] [CrossRef]
- Kosik, K.S.; Orecchio, L.D.; Bakalis, S.; Neve, R.L. Developmentally regulated expression of specific tau sequences. Neuron 1989, 2, 1389–1397. [Google Scholar] [CrossRef]
- Filipcik, P.; Zilka, N.; Bugos, O.; Kucerak, J.; Koson, P.; Novak, P.; Novak, M. First transgenic rat model developing progressive cortical neurofibrillary tangles. Neurobiol. Aging 2012, 33, 1448–1456. [Google Scholar] [CrossRef] [PubMed]
- Koson, P.; Zilka, N.; Kovac, A.; Kovacech, B.; Korenova, M.; Filipcik, P.; Novak, M. Truncated tau expression levels determine life span of a rat model of tauopathy without causing neuronal loss or correlating with terminal neurofibrillary tangle load. Eur. J. Neurosci. 2008, 28, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Hrnkova, M.; Zilka, N.; Minichova, Z.; Koson, P.; Novak, M. Neurodegeneration caused by expression of human truncated tau leads to progressive neurobehavioural impairment in transgenic rats. Brain Res. 2007, 1130, 206–213. [Google Scholar] [CrossRef]
- Aigner, B.; Renner, S.; Kessler, B.; Klymiuk, N.; Kurome, M.; Wunsch, A.; Wolf, E. Transgenic pigs as models for translational biomedical research. J. Mol. Med. 2010, 88, 653–664. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.L.; Lin, L.; Bolund, L.; Jensen, T.G.; Sorensen, C.B. Genetically modified pigs for biomedical research. J. Inherit. Metab. Dis. 2012, 35, 695–713. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, J.C.; Bawden, C.S.; Rudiger, S.R.; McLaughlan, C.J.; Reid, S.J.; Waldvogel, H.J.; MacDonald, M.E.; Gusella, J.F.; Walker, S.K.; Kelly, J.M. An ovine transgenic Huntington’s disease model. Hum. Mol. Genet. 2010, ddq063. [Google Scholar] [CrossRef]
- Johnstone, E.; Chaney, M.; Norris, F.; Pascual, R.; Little, S. Conservation of the sequence of the Alzheimer’s disease amyloid peptide in dog, polar bear and five other mammals by cross-species polymerase chain reaction analysis. Mol. Brain Res. 1991, 10, 299–305. [Google Scholar] [CrossRef]
- Reid, S.J.; Mckean, N.E.; Henty, K.; Portelius, E.; Blennow, K.; Rudiger, S.R.; Bawden, C.S.; Handley, R.R.; Verma, P.J.; Faull, R.L. Alzheimer’s disease markers in the aged sheep (Ovis aries). Neurobiol. Aging 2017, 58, 112–119. [Google Scholar] [CrossRef]
- Papaioannou, N.; Tooten, P.C.; van Ederen, A.M.; Bohl, J.R.; Rofina, J.; Tsangaris, T.; Gruys, E. Immunohistochemical investigation of the brain of aged dogs. I. Detection of neurofibrillary tangles and of 4-hydroxynonenal protein, an oxidative damage product, in senile plaques. Amyloid 2001, 8, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Rosen, R.F.; Farberg, A.S.; Gearing, M.; Dooyema, J.; Long, P.M.; Anderson, D.C.; Davis-Turak, J.; Coppola, G.; Geschwind, D.H.; Pare, J.F.; et al. Tauopathy with paired helical filaments in an aged chimpanzee. J. Comp. Neurol. 2008, 509, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Gearing, M.; Rebeck, G.W.; Hyman, B.T.; Tigges, J.; Mirra, S.S. Neuropathology and apolipoprotein E profile of aged chimpanzees: Implications for Alzheimer disease. Proc. Natl. Acad. Sci. USA 1994, 91, 9382–9386. [Google Scholar] [CrossRef] [PubMed]
- Gearing, M.; Tigges, J.; Mori, H.; Mirra, S. β-Amyloid (Aβ) deposition in the brains of aged orangutans. Neurobiol. Aging 1997, 18, 139–146. [Google Scholar] [CrossRef]
- Kimura, N.; Nakamura, S.; Goto, N.; Narushima, E.; Hara, I.; Shichiri, S.; Saitou, K.; Nose, M.; Hayashi, T.; Kawamura, S.; et al. Senile plaques in an aged Western lowland gorilla. Exp. Anim. Tokyo 2001, 50, 77–81. [Google Scholar] [CrossRef]
- Perez, S.E.; Raghanti, M.A.; Hof, P.R.; Kramer, L.; Ikonomovic, M.D.; Lacor, P.N.; Erwin, J.M.; Sherwood, C.C.; Mufson, E.J. Alzheimer’s disease pathology in the neocortex and hippocampus of the western lowland gorilla (Gorilla gorilla gorilla). J. Comp. Neurol. 2013, 521, 4318–4338. [Google Scholar] [CrossRef]
- Perez, S.E.; Sherwood, C.C.; Cranfield, M.R.; Erwin, J.M.; Mudakikwa, A.; Hof, P.R.; Mufson, E.J. Early Alzheimer’s disease–type pathology in the frontal cortex of wild mountain gorillas (Gorilla beringei beringei). Neurobiol. Aging 2016, 39, 195–201. [Google Scholar] [CrossRef]
- Nakamura, S.; Kiatipattanasakul, W.; Nakayama, H.; Ono, F.; Sakakibara, I.; Yoshikawa, Y.; Goto, N.; Doi, K. Immunohistochemical characteristics of the constituents of senile plaques and amyloid angiopathy in aged cynomolgus monkeys. J. Med. Primatol. 1996, 25, 294–300. [Google Scholar] [CrossRef]
- Darusman, H.S.; Gjedde, A.; Sajuthi, D.; Schapiro, S.J.; Kalliokoski, O.; Kristianingrum, Y.P.; Handaryani, E.; Hau, J. Amyloid Beta1–42 and the Phoshorylated Tau Threonine 231 in Brains of Aged Cynomolgus Monkeys (Macaca fascicularis). Front. Aging Neurosci. 2014, 6, 313. [Google Scholar] [CrossRef]
- Nakamura, S.i.; Nakayama, H.; Goto, N.; Ono, F.; Sakakibara, I.; Yoshikawa, Y. Histopathological studies of senile plaques and cerebral amyloidosis in cynomolgus monkeys. J. Med. Primatol. 1998, 27, 244–252. [Google Scholar] [CrossRef]
- Nakamura, S.; Kimura, N.; Nishimura, M.; Torii, R.; Terao, K. Neurofibrillary tangles and senile plaques in aged cynomolgus monkeys. In Proceedings of the AFLAS and CALAS, Kyoto, Japan, 20–22 September 2008. [Google Scholar]
- Wisniewski, H.M.; Ghetti, B.; Terry, R.D. Neuritic Senile Plaques and Filamentous Changes in Aged Rhesus-Monkeys. J. Neuropathol. Exp. Neurol. 1973, 32, 566–584. [Google Scholar] [CrossRef]
- Paspalas, C.D.; Carlyle, B.C.; Leslie, S.; Preuss, T.M.; Crimins, J.L.; Huttner, A.J.; van Dyck, C.H.; Rosene, D.L.; Nairn, A.C.; Arnsten, A.F. The aged rhesus macaque manifests Braak stage III/IV Alzheimer’s-like pathology. Alzheimer’s Dement. 2018, 14, 680–691. [Google Scholar] [CrossRef] [PubMed]
- Toledano, A.; Álvarez, M.; López-Rodríguez, A.; Toledano-Díaz, A.; Fernández-Verdecia, C. Does Alzheimer disease exist in all primates? Alzheimer pathology in non-human primates and its pathophysiological implications (II). Neurología 2014, 29, 42–55. [Google Scholar] [CrossRef]
- Latimer, C.S.; Shively, C.A.; Keene, C.D.; Jorgensen, M.J.; Andrews, R.N.; Register, T.C.; Montine, T.J.; Wilson, A.M.; Neth, B.J.; Mintz, A. A nonhuman primate model of early Alzheimer’s disease pathologic change: Implications for disease pathogenesis. Alzheimer’s Dement. 2019, 15, 93–105. [Google Scholar] [CrossRef]
- Ndung’u, M.; Hartig, W.; Wegner, F.; Mwenda, J.M.; Low, R.W.C.; Akinyemi, R.O.; Kalaria, R.N. Cerebral amyloid beta(42) deposits and microvascular pathology in ageing baboons. Neuropathol. Appl. Neurol. 2012, 38, 487–499. [Google Scholar] [CrossRef] [PubMed]
- Schultz, C.; Dehghani, F.; Hubbard, G.B.; Thal, D.R.; Struckhoff, G.; Braak, E.; Braak, H. Filamentous tau pathology in nerve cells, astrocytes, and oligodendrocytes of aged baboons. J. Neuropathol. Exp. Neurol. 2000, 59, 39–52. [Google Scholar] [CrossRef]
- Schultz, C.; Hubbard, G.B.; Rub, U.; Braak, E.; Braak, H. Age-related progression of tau pathology in brains of baboons. Neurobiol. Aging 2000, 21, 905–912. [Google Scholar] [CrossRef]
- Lemere, C.A.; Oh, J.; Stanish, H.A.; Peng, Y.; Pepivani, I.; Fagan, A.M.; Yamaguchi, H.; Westmoreland, S.V.; Mansfield, K.G. Cerebral amyloid-beta protein accumulation with aging in cotton-top tamarins: A model of early Alzheimer’s disease? Rejuvenation Res. 2008, 11, 321–332. [Google Scholar] [CrossRef]
- Giannakopoulos, P.; Silhol, S.; Jallageas, V.; Mallet, J.; Bons, N.; Bouras, C.; Delaere, P. Quantitative analysis of tau protein-immunoreactive accumulations and β amyloid protein deposits in the cerebral cortex of the mouse lemur, Microcebus murinus. Acta Neuropathol. 1997, 94, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Kraska, A.; Dorieux, O.; Picq, J.-L.; Petit, F.; Bourrin, E.; Chenu, E.; Volk, A.; Perret, M.; Hantraye, P.; Mestre-Frances, N. Age-associated cerebral atrophy in mouse lemur primates. Neurobiol. Aging 2011, 32, 894–906. [Google Scholar] [CrossRef]
- Mestre, N.; Bons, N. Age-related cytological changes and neuronal loss in basal forebrain cholinergic neurons in Microcebus murinus (Lemurian primate). Neurodegeneration 1993, 2, 25–32. [Google Scholar]
- Geula, C.; Nagykery, N.; Wu, C.-K. Amyloid-β deposits in the cerebral cortex of the aged common marmoset (Callithrix jacchus): Incidence and chemical composition. Acta Neuropathol. 2002, 103, 48–58. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Callejas, J.D.; Fuchs, E.; Perez-Cruz, C. Evidence of tau hyperphosphorylation and dystrophic microglia in the common marmoset. Front. Aging Neurosci. 2016, 8, 315. [Google Scholar] [CrossRef] [PubMed]
- Elfenbein, H.A.; Rosen, R.F.; Stephens, S.L.; Switzer, R.C.; Smith, Y.; Pare, J.; Mehta, P.D.; Warzok, R.; Walker, L.C. Cerebral beta-amyloid angiopathy in aged squirrel monkeys. Histol. Histopathol. 2007, 22, 155–167. [Google Scholar]
- Walker, L.; Masters, C.; Beyreuther, K.; Price, D. Amyloid in the brains of aged squirrel monkeys. Acta Neuropathol. 1990, 80, 381–387. [Google Scholar] [CrossRef]
- Smith, D.; Chen, X.; Nonaka, M.; Trojanowski, J.; Lee, V.-Y.; Saatman, K.; Leoni, M.; Xu, B.; Wolf, J.; Meaney, D. Accumulation of amyloid β and tau and the formation of neurofilament inclusions following diffuse brain injury in the pig. J. Neuropathol. Exp. Neurol. 1999, 58, 982–992. [Google Scholar] [CrossRef] [PubMed]
- Braak, H.; Braak, E.; Strothjohann, M. Abnormally phosphorylated tau protein related to the formation of neurofibrillary tangles and neuropil threads in the cerebral cortex of sheep and goat. Neurosci. Lett. 1994, 171, 1–4. [Google Scholar] [CrossRef]
- Nelson, P.; Saper, C. Ultrastructure of neurofibrillary tangles in the cerebral cortex of sheep. Neurobiol. Aging 1995, 16, 315–323. [Google Scholar] [CrossRef]
- Nelson, P.T.; Greenberg, S.G.; Saper, C.B. Neurofibrillary tangles in the cerebral cortex of sheep. Neurosci. Lett. 1994, 170, 187–190. [Google Scholar] [CrossRef]
- Nakamura, S.-I.; Nakayama, H.; Uetsuka, K.; Sasaki, N.; Uchida, K.; Goto, N. Senile plaques in an aged two-humped (Bactrian) camel (Camelus bactrianus). Acta Neuropathol. 1995, 90, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Härtig, W.; Klein, C.; Brauer, K.; Schüppel, K.-F.; Arendt, T.; Brückner, G.; Bigl, V. Abnormally phosphorylated protein tau in the cortex of aged individuals of various mammalian orders. Acta Neuropathol. 2000, 100, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Colle, M.-A.; Hauw, J.-J.; Crespeau, F.; Uchihara, T.; Akiyama, H.; Checler, F.; Pageat, P.; Duykaerts, C. Vascular and parenchymal Aβ deposition in the aging dog: Correlation with behavior. Neurobiol. Aging 2000, 21, 695–704. [Google Scholar] [CrossRef]
- Yu, C.H.; Song, G.S.; Yhee, J.Y.; Kim, J.H.; Im, K.S.; Nho, W.G.; Lee, J.H.; Sur, J.H. Histopathological and Immunohistochemical Comparison of the Brain of Human Patients with Alzheimer’s Disease and the Brain of Aged Dogs with Cognitive Dysfunction. J. Comp. Pathol. 2011, 145, 45–58. [Google Scholar] [CrossRef] [PubMed]
- Abey, A.; Davies, D.; Goldsbury, C.; Buckland, M.; Valenzuela, M.; Duncan, T. Distribution of tau hyperphosphorylation in canine dementia resembles early Alzheimer’s disease and other tauopathies. Brain Pathol. 2021, 31, 144–162. [Google Scholar] [CrossRef] [PubMed]
- Ozawa, M.; Chambers, J.K.; Uchida, K.; Nakayama, H. The Relation between canine cognitive dysfunction and age-related brain lesions. J. Vet. Med Sci. 2016, 78, 991–1006. [Google Scholar] [CrossRef]
- Schmidt, F.; Boltze, J.; Jäger, C.; Hofmann, S.; Willems, N.; Seeger, J.; Härtig, W.; Stolzing, A. Detection and quantification of β-amyloid, pyroglutamyl Aβ, and tau in aged canines. J. Neuropathol. Exp. Neurol. 2015, 74, 912–923. [Google Scholar] [CrossRef]
- Smolek, T.; Madari, A.; Farbakova, J.; Kandrac, O.; Jadhav, S.; Cente, M.; Brezovakova, V.; Novak, M.; Zilka, N. Tau hyperphosphorylation in synaptosomes and neuroinflammation are associated with canine cognitive impairment. J. Comp. Neurol. 2016, 524, 874–895. [Google Scholar] [CrossRef]
- Fiock, K.L.; Smith, J.D.; Crary, J.F.; Hefti, M.M. β-amyloid and tau pathology in the aging feline brain. J. Comp. Neurol. 2020, 528, 112–117. [Google Scholar] [CrossRef]
- Sordo Sordo, L. Neuropathology, Diagnosis, and Potential Treatment of Feline Cognitive Dysfunction Syndrome and Its Similarities to Alzheimer’s Disease; University of Edinburgh: Edinburgh, UK, 2021. [Google Scholar]
- Chambers, J.K.; Uchida, K.; Harada, T.; Tsuboi, M.; Sato, M.; Kubo, M.; Kawaguchi, H.; Miyoshi, N.; Tsujimoto, H.; Nakayama, H. Neurofibrillary tangles and the deposition of a beta amyloid peptide with a novel N-terminal epitope in the brains of wild Tsushima leopard cats. PLoS ONE 2012, 7, e46452. [Google Scholar] [CrossRef]
- Cork, L.C.; Powers, R.E.; Selkoe, D.J.; Davies, P.; Geyer, J.J.; Price, D.L. Neurofibrillary tangles and senile plaques in aged bears. J. Neuropathol. Exp. Neurol. 1988, 47, 629–641. [Google Scholar] [CrossRef] [PubMed]
- Uchida, K.; Yoshino, T.; Yamaguchi, R.; Tateyama, S.; Kimoto, Y.; Nakayama, H.; Goto, N. Senile plaques and other senile changes in the brain of an aged American black bear. Vet. Pathol. 1995, 32, 412–414. [Google Scholar] [CrossRef]
- Roertgen, K.E.; Parisi, J.E.; Clark, H.B.; Barnes, D.L.; O’Brien, T.D.; Johnson, K.H. Aβ-associated cerebral angiopathy and senile plaques with neurofibrillary tangles and cerebral hemorrhage in an aged wolverine (Gulo gulo). Neurobiol. Aging 1996, 17, 243–247. [Google Scholar] [CrossRef]
- Takaichi, Y.; Chambers, J.K.; Takahashi, K.; Soeda, Y.; Koike, R.; Katsumata, E.; Kita, C.; Matsuda, F.; Haritani, M.; Takashima, A. Amyloid β and tau pathology in brains of aged pinniped species (sea lion, seal, and walrus). Acta Neuropathol. Commun. 2021, 9, 1–15. [Google Scholar] [CrossRef]
- Finch, C.E. Evolution of the human lifespan and diseases of aging: Roles of infection, inflammation, and nutrition. Proc. Natl. Acad. Sci. USA 2010, 107, 1718–1724. [Google Scholar] [CrossRef]
- Knight, A. The beginning of the end for chimpanzee experiments? Philos. Ethics Humanit. Med. 2008, 3, 1–14. [Google Scholar] [CrossRef]
- Shumaker, R.W.; Wich, S.A.; Perkins, L. Reproductive life history traits of female orangutans (Pongo spp.). In Primate Reproductive Aging; Karger Publishers: Basel, Switzerland, 2008; Volume 36, pp. 147–161. [Google Scholar]
- Nishida, T.; Corp, N.; Hamai, M.; Hasegawa, T.; Hiraiwa-Hasegawa, M.; Hosaka, K.; Hunt, K.D.; Itoh, N.; Kawanaka, K.; Matsumoto-Oda, A. Demography, female life history, and reproductive profiles among the chimpanzees of Mahale. Am. J. Primatol. Off. J. Am. Soc. Primatol. 2003, 59, 99–121. [Google Scholar] [CrossRef] [PubMed]
- Li, H.W.; Zhang, L.; Qin, C. Current state of research on non-human primate models of Alzheimer’s disease. Anim. Models Exp. Med. 2019, 2, 227–238. [Google Scholar] [CrossRef]
- Uno, H. Age-related pathology and biosenescent markers in captive rhesus macaques. Age 1997, 20, 1–13. [Google Scholar] [CrossRef]
- Souder, D.C.; Dreischmeier, I.A.; Smith, A.B.; Wright, S.; Martin, S.A.; Sagar, M.A.K.; Eliceiri, K.W.; Salamat, S.M.; Bendlin, B.B.; Colman, R.J. Rhesus monkeys as a translational model for late-onset Alzheimer’s disease. Aging Cell 2021, 20, e13374. [Google Scholar] [CrossRef]
- Braak, H.; Braak, E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 1991, 82, 239–259. [Google Scholar] [CrossRef] [PubMed]
- Picq, J.-L. Aging affects executive functions and memory in mouse lemur primates. Exp. Gerontol. 2007, 42, 223–232. [Google Scholar] [CrossRef]
- Mestre-Francés, N.; Trouche, S.G.; Fontes, P.; Lautier, C.; Devau, G.; Lasbleiz, C.; Dhenain, M.; Verdier, J.-M. Old Gray Mouse Lemur Behavior, Cognition, and Neuropathology. In Conn’s Handbook of Models for Human Aging; Elsevier: Amsterdam, The Netherlands, 2018; pp. 287–300. [Google Scholar]
- Bons, N.; Mestre, N.; Petter, A. Senile plaques and neurofibrillary changes in the brain of an aged lemurian primate, Microcebus murinus. Neurobiol. Aging 1992, 13, 99–105. [Google Scholar] [CrossRef]
- Bons, N.; Mestre, N.; Ritchie, K.; Petter, A.; Podlisny, M.; Selkoe, D. Identification of Amyloid-Beta Protein in the Brain of the Small, Short-Lived Lemurian Primate Microcebus-Murinus. Neurobiol. Aging 1994, 15, 215–220. [Google Scholar] [CrossRef]
- Bons, N.; Rieger, F.; Prudhomme, D.; Fisher, A.; Krause, K.H. Microcebus murinus: A useful primate model for human cerebral aging and Alzheimer’s disease? Genes Brain Behav. 2006, 5, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Dhenain, M.; Chenu, E.; Hisley, C.K.; Aujard, F.; Volk, A. Regional atrophy in the brain of lissencephalic mouse lemur primates: Measurement by automatic histogram-based segmentation of MR images. Magn. Reson. Med. Off. J. Int. Soc. Magn. Reson. Med. 2003, 50, 984–992. [Google Scholar] [CrossRef] [PubMed]
- Okano, H.; Hikishima, K.; Iriki, A.; Sasaki, E. The common marmoset as a novel animal model system for biomedical and neuroscience research applications. In Seminars in Fetal and Neonatal Medicine; Elsevier: Amsterdam, The Netherlands, 2012; pp. 336–340. [Google Scholar]
- King, A. The search for better animal models of Alzheimer’s disease. Nature 2018, 559, S13. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, M.; Ridley, R.; Baker, H.; Maclean, C.; Honer, W.; Francis, P. Chronic elevation of amyloid precursor protein in the neocortex or hippocampus of marmosets with selective cholinergic lesions. J. Neural Transm. 2001, 108, 809–826. [Google Scholar] [CrossRef]
- Trouche, S.G.; Asuni, A.; Rouland, S.; Wisniewski, T.; Frangione, B.; Verdier, J.-M.; Sigurdsson, E.M.; Mestre-Francés, N. Antibody response and plasma Aβ1-40 levels in young Microcebus murinus primates immunized with Aβ1-42 and its derivatives. Vaccine 2009, 27, 957–964. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Seneca, N.; Cai, L.; Liow, J.-S.; Zoghbi, S.S.; Gladding, R.L.; Hong, J.; Pike, V.W.; Innis, R.B. Brain and whole-body imaging in nonhuman primates with [11C] MeS-IMPY, a candidate radioligand for β-amyloid plaques. Nuclear Med. Biol. 2007, 34, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Liang, S.H.; Holland, J.P.; Stephenson, N.A.; Kassenbrock, A.; Rotstein, B.H.; Daignault, C.P.; Lewis, R.; Collier, L.; Hooker, J.M.; Vasdev, N. PET neuroimaging studies of [18F] CABS13 in a double transgenic mouse model of Alzheimer’s disease and nonhuman primates. ACS Chem. Neurosci. 2015, 6, 535–541. [Google Scholar] [CrossRef]
- Heuer, E.; Jacobs, J.; Du, R.; Wang, S.; Keifer, O.P.; Cintron, A.F.; Dooyema, J.; Meng, Y.; Zhang, X.; Walker, L.C. Amyloid-Related Imaging Abnormalities in an Aged Squirrel Monkey with Cerebral Amyloid Angiopathy. J. Alzheimer’s Dis. 2017, 57, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Stöhr, J.; Watts, J.C.; Mensinger, Z.L.; Oehler, A.; Grillo, S.K.; DeArmond, S.J.; Prusiner, S.B.; Giles, K. Purified and synthetic Alzheimer’s amyloid beta (Aβ) prions. Proc. Natl. Acad. Sci. USA 2012, 109, 11025–11030. [Google Scholar] [CrossRef]
- Goedert, M. Alzheimer’s and Parkinson’s diseases: The prion concept in relation to assembled Aβ, tau, and α-synuclein. Science 2015, 349. [Google Scholar] [CrossRef]
- Ridley, R.; Baker, H.; Windle, C.; Cummings, R. Very long term studies of the seeding of β-amyloidosis in primates. J. Neural Transm. 2006, 113, 1243–1251. [Google Scholar] [CrossRef]
- Beckman, D.; Morrison, J.H. Towards developing a rhesus monkey model of early Alzheimer’s disease focusing on women’s health. Am. J. Primatol. 2021, e23289. [Google Scholar] [CrossRef]
- Forny-Germano, L.; e Silva, N.M.L.; Batista, A.F.; Brito-Moreira, J.; Gralle, M.; Boehnke, S.E.; Coe, B.C.; Lablans, A.; Marques, S.A.; Martinez, A.M.B. Alzheimer’s disease-like pathology induced by amyloid-β oligomers in nonhuman primates. J. Neurosci. 2014, 34, 13629–13643. [Google Scholar] [CrossRef]
- Beckman, D.; Chakrabarty, P.; Ott, S.; Dao, A.; Zhou, E.; Janssen, W.G.; Donis-Cox, K.; Muller, S.; Kordower, J.H.; Morrison, J.H. A novel tau-based rhesus monkey model of Alzheimer’s pathogenesis. Alzheimer’s Dement. 2021, 17, 933–945. [Google Scholar] [CrossRef]
- Seita, Y.; Morimura, T.; Watanabe, N.; Iwatani, C.; Tsuchiya, H.; Nakamura, S.; Suzuki, T.; Yanagisawa, D.; Tsukiyama, T.; Nakaya, M. Generation of transgenic cynomolgus monkeys overexpressing the gene for Amyloid-β precursor protein. J. Alzheimer’s Dis. 2020, 75, 45–60. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Sasaguri, H.; Kumita, W.; Inoue, T.; Kurosaki, Y.; Nagata, K.; Mihira, N.; Sato, K.; Sakuma, T.; Yamamoto, T. A non-human primate model of familial Alzheimer’s disease. bioRxiv 2020. [Google Scholar] [CrossRef]
- Zeiss, C.J. Utility of spontaneous animal models of Alzheimer’s disease in preclinical efficacy studies. Cell Tissue Res. 2020, 380, 273–286. [Google Scholar] [CrossRef]
- Bosch, M.N.; Pugliese, M.; Gimeno-Bayon, J.; Rodriguez, M.J.; Mahy, N. Dogs with Cognitive Dysfunction Syndrome: A Natural Model of Alzheimer’s Disease. Curr. Alzheimer Res. 2012, 9, 298–314. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Head, E. Brain aging in dogs: Parallels with human brain aging and Alzheimer’s disease. Vet. Ther. 2001, 2, 247–260. [Google Scholar] [PubMed]
- Head, E. A canine model of human aging and Alzheimer’s disease. Biochim. Et Biophys. Acta (BBA)-Mol. Basis Dis. 2013, 1832, 1384–1389. [Google Scholar] [CrossRef]
- Prpar Mihevc, S.; Majdič, G. Canine Cognitive Dysfunction and Alzheimer’s Disease–Two Facets of the Same Disease? Front. Neurosci. 2019, 13, 604. [Google Scholar] [CrossRef]
- Insua, D.; Suárez, M.-L.; Santamarina, G.; Sarasa, M.; Pesini, P. Dogs with canine counterpart of Alzheimer’s disease lose noradrenergic neurons. Neurobiol. Aging 2010, 31, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Neilson, J.C.; Hart, B.L.; Cliff, K.D.; Ruehl, W.W. Prevalence of behavioral changes associated with age-related cognitive impairment in dogs. J. Am. Vet. Med. Assoc. 2001, 218, 1787–1791. [Google Scholar] [CrossRef]
- Salvin, H.E.; McGreevy, P.D.; Sachdev, P.S.; Valenzuela, M.J. The canine cognitive dysfunction rating scale (CCDR): A data-driven and ecologically relevant assessment tool. Vet. J. 2011, 188, 331–336. [Google Scholar] [CrossRef]
- Madari, A.; Farbakova, J.; Katina, S.; Smolek, T.; Novak, P.; Weissova, T.; Novak, M.; Zilka, N. Assessment of severity and progression of canine cognitive dysfunction syndrome using the CAnine DEmentia Scale (CADES). Appl. Anim. Behav. Sci. 2015, 171, 138–145. [Google Scholar] [CrossRef]
- Osella, M.; Re, G.; Odore, R.; Girardi, C.; Badino, P.; Barbero, R.; Bergamasco, L. Canine Cognitive Dysfunction: Prevalence, Clinical Signs and Treatment with a Nutraceutical; Purdue University Press: West Lafayette, IN, USA, 2005; pp. 66–72. [Google Scholar]
- Wisniewski, H.; Johnson, A.; Raine, C.; Kay, W.; Terry, R. Senile plaques and cerebral amyloidosis in aged dogs. A histochemical and ultrastructural study. Lab. Investig. 1970, 23, 287–296. [Google Scholar] [PubMed]
- Pugliese, M.; Geloso, M.C.; Carrasco, J.L.; Mascort, J.; Michetti, F.; Mahy, N. Canine cognitive deficit correlates with diffuse plaque maturation and S100β (−) astrocytosis but not with insulin cerebrospinal fluid level. Acta Neuropathol. 2006, 111, 519. [Google Scholar] [CrossRef] [PubMed]
- Butterfield, D.A.; Barone, E.; Di Domenico, F.; Cenini, G.; Sultana, R.; Murphy, M.P.; Mancuso, C.; Head, E. Atorvastatin treatment in a dog preclinical model of Alzheimer’s disease leads to up-regulation of haem oxygenase-1 and is associated with reduced oxidative stress in brain. Int. J. Neuropsychopharmacol. 2012, 15, 981–987. [Google Scholar] [CrossRef]
- Barone, E.; Mancuso, C.; Di Domenico, F.; Sultana, R.; Murphy, M.P.; Head, E.; Butterfield, D.A. Biliverdin reductase-A: A novel drug target for atorvastatin in a dog pre-clinical model of Alzheimer disease. J. Neurochem. 2012, 120, 135–146. [Google Scholar] [CrossRef]
- Di Domenico, F.; Perluigi, M.; Barone, E. Biliverdin Reductase-A correlates with inducible nitric oxide synthasein in atorvastatin treated aged canine brain. Neural Regen. Res. 2013, 8, 1925. [Google Scholar] [PubMed]
- Bosch, M.N.; Bayon, J.G.; Rodriguez, M.J.; Pugliese, M.; Mahy, N. Rapid improvement of canine cognitive dysfunction with immunotherapy designed for Alzheimer’s disease. Curr. Alzheimer Res. 2013, 10, 482–493. [Google Scholar] [CrossRef]
- Head, E.; Moffat, K.; Das, P.; Sarsoza, E.; Poon, W.W.; Landsberg, G.; Cotman, C.W.; Murphy, M.P. beta-amyloid deposition and tau phosphorylation in clinically characterized aged cats. Neurobiol. Aging 2005, 26, 749–763. [Google Scholar] [CrossRef]
- Nakamura, S.-i.; Nakayama, H.; Kiatipattanasakul, W.; Uetsuka, K.; Uchida, K.; Goto, N. Senile plaques in very aged cats. Acta Neuropathol. 1996, 91, 437–439. [Google Scholar] [CrossRef]
- Klug, J.; Snyder, J.M.; Darvas, M.; Imai, D.M.; Church, M.; Latimer, C.; Keene, C.D.; Ladiges, W. Aging pet cats develop neuropathology similar to human Alzheimer’s disease. Aging Pathobiol. Ther. 2020, 2, 120–125. [Google Scholar] [CrossRef]
- Sordo, L.; Gunn-Moore, D.A. Cognitive dysfunction in cats: Update on neuropathological and behavioural changes plus clinical management. Vet. Rec. 2021, 188, e3. [Google Scholar] [CrossRef] [PubMed]
- Gunn-Moore, D.; Moffat, K.; Christie, L.A.; Head, E. Cognitive dysfunction and the neurobiology of ageing in cats. J. Small Anim. Pract. 2007, 48, 546–553. [Google Scholar] [CrossRef]
- Perleberg, C.; Kind, A.; Schnieke, A. Genetically engineered pigs as models for human disease. Dis. Models Mech. 2018, 11, dmm030783. [Google Scholar] [CrossRef] [PubMed]
- Prather, R.S.; Lorson, M.; Ross, J.W.; Whyte, J.J.; Walters, E. Genetically engineered pig models for human diseases. Annu. Rev. Anim. Biosci. 2013, 1, 203–219. [Google Scholar] [CrossRef]
- Walters, E.M.; Agca, Y.; Ganjam, V.; Evans, T. Animal models got you puzzled? Think pig. Ann. N. Y. Acad. Sci. 2011, 1245, 63–64. [Google Scholar] [CrossRef]
- Hoffe, B.; Holahan, M.R. The use of pigs as a translational model for studying neurodegenerative diseases. Front. Physiol. 2019, 10, 838. [Google Scholar] [CrossRef]
- Kragh, P.M.; Nielsen, A.L.; Li, J.; Du, Y.; Lin, L.; Schmidt, M.; Bøgh, I.B.; Holm, I.E.; Jakobsen, J.E.; Johansen, M.G. Hemizygous minipigs produced by random gene insertion and handmade cloning express the Alzheimer’s disease-causing dominant mutation APPsw. Transgenic Res. 2009, 18, 545–558. [Google Scholar] [CrossRef]
- Søndergaard, L.V.; Ladewig, J.; Dagnæs-Hansen, F.; Herskin, M.S.; Holm, I.E. Object recognition as a measure of memory in 1–2 years old transgenic minipigs carrying the APPsw mutation for Alzheimer’s disease. Transgenic Res. 2012, 21, 1341–1348. [Google Scholar] [CrossRef] [PubMed]
- Hall, V.J.; Lindblad, M.M.; Jakobsen, J.E.; Gunnarsson, A.; Schmidt, M.; Rasmussen, M.A.; Volke, D.; Zuchner, T.; Hyttel, P. Impaired APP activity and altered Tau splicing in embryonic stem cell-derived astrocytes obtained from an APPsw transgenic minipig. Dis. Models Mech. 2015, 8, 1265–1278. [Google Scholar]
- Jakobsen, J.E.; Johansen, M.G.; Schmidt, M.; Liu, Y.; Li, R.; Callesen, H.; Melnikova, M.; Habekost, M.; Matrone, C.; Bouter, Y. Expression of the Alzheimer’s disease mutations AβPP695sw and PSEN1M146I in double-transgenic göttingen minipigs. J. Alzheimer’s Dis. 2016, 53, 1617–1630. [Google Scholar] [CrossRef] [PubMed]
- Weber, K.; Pearce, D.A. Large animal models for Batten disease: A review. J. Child Neurol. 2013, 28, 1123–1127. [Google Scholar] [CrossRef] [PubMed]
- Cook, R.; Jolly, R.; Palmer, D.; Tammen, I.; Broom, M.; McKinnon, R. Neuronal ceroid lipofuscinosis in Merino sheep. Aust. Vet. J. 2002, 80, 292–297. [Google Scholar] [CrossRef]
- Jolly, R.; Arthur, D.; Kay, G.; Palmer, D. Neuronal ceroid-lipofuscinosis in Borderdale sheep. N. Z. Vet. J. 2002, 50, 199–202. [Google Scholar] [CrossRef] [PubMed]
- Jolly, R.; Janmaat, A.; West, D.a.; Morrison, I. Ovine ceroid-lipofuscinosis: A model of Batten’s disease. Neuropathol. Appl. Neurol. 1980, 6, 195–209. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.M.; Kleemann, D.O.; Walker, S.K. Enhanced efficiency in the production of offspring from 4-to 8-week-old lambs. Theriogenology 2005, 63, 1876–1890. [Google Scholar] [CrossRef]
- Reid, S.J.; Patassini, S.; Handley, R.R.; Rudiger, S.R.; McLaughlan, C.J.; Osmand, A.; Jacobsen, J.C.; Morton, A.J.; Weiss, A.; Waldvogel, H.J. Further molecular characterisation of the OVT73 transgenic sheep model of Huntington’s disease identifies cortical aggregates. J. Huntingt. Dis. 2013, 2, 279–295. [Google Scholar]
- Handley, R.R.; Reid, S.J.; Brauning, R.; Maclean, P.; Mears, E.R.; Fourie, I.; Patassini, S.; Cooper, G.J.; Rudiger, S.R.; McLaughlan, C.J. Brain urea increase is an early Huntington’s disease pathogenic event observed in a prodromal transgenic sheep model and HD cases. Proc. Natl. Acad. Sci. USA 2017, 201711243. [Google Scholar] [CrossRef] [PubMed]
- Pfister, E.L.; DiNardo, N.; Mondo, E.; Borel, F.; Conroy, F.; Fraser, C.; Gernoux, G.; Han, X.; Hu, D.; Johnson, E. Artificial miRNAs reduce human mutant Huntingtin throughout the striatum in a transgenic sheep model of Huntington’s disease. Hum. Gene Ther. 2018, 29, 663–673. [Google Scholar] [CrossRef]
- Jiang, Y.; Xie, M.; Chen, W.; Talbot, R.; Maddox, J.F.; Faraut, T.; Wu, C.; Muzny, D.M.; Li, Y.; Zhang, W.; et al. The sheep genome illuminates biology of the rumen and lipid metabolism. Science 2014, 344, 1168–1173. [Google Scholar] [CrossRef]
- Kendrick, K.M.; da Costa, A.P.; Leigh, A.E.; Hinton, M.R.; Peirce, J.W. Sheep don’t forget a face. Nature 2001, 414, 165–166. [Google Scholar] [CrossRef]
- Morton, A.J.; Avanzo, L. Executive decision-making in the domestic sheep. PLoS ONE 2011, 6, e15752. [Google Scholar] [CrossRef] [PubMed]
- Perentos, N.; Martins, A.Q.; Watson, T.C.; Bartsch, U.; Mitchell, N.L.; Palmer, D.N.; Jones, M.W.; Morton, A.J. Translational neurophysiology in sheep: Measuring sleep and neurological dysfunction in CLN5 Batten disease affected sheep. Brain 2015, 138, 862–874. [Google Scholar] [CrossRef] [PubMed]
- Sawiak, S.J.; Perumal, S.R.; Rudiger, S.R.; Matthews, L.; Mitchell, N.L.; McLaughlan, C.J.; Bawden, C.S.; Palmer, D.N.; Kuchel, T.; Morton, A.J. Rapid and Progressive Regional Brain Atrophy in CLN6 Batten Disease Affected Sheep Measured with Longitudinal Magnetic Resonance Imaging. PLoS ONE 2015, 10, e0132331. [Google Scholar] [CrossRef] [PubMed]

| Name | Type of Modification | FAD Mutations | MAPT Mutations | Plaques | Tangles | Neurodegeneration | Reference |
|---|---|---|---|---|---|---|---|
| PDAPP | Transgenesis | Indiana in APP | X | [48] | |||
| Tg2576 | Transgenesis | Swedish in APP | X | [49] | |||
| TgCRND8 | Transgenesis | Swedish and Indiana in APP | X | [50] | |||
| PSAPP | Transgenesis | Swedish in APP, M146L in PSEN1 | X | [51] | |||
| BRI-Aβ40 | Transgenesis | Aβ1–40 peptide | [52] | ||||
| BRI-Aβ42 | Transgenesis | Aβ1–42 peptide | X | [52] | |||
| 5XFAD | Transgenesis | Swedish, Florida, London in APP. M146L and L286V in PSEN1 | X | X | [53] | ||
| JNPL3 | Transgenesis | P301L in MAPT | X | X | [54] | ||
| rTg4510 | Transgenesis | P301L in MAPT | X | X | [55] | ||
| 3xTg | Transgenesis | Swedish in APP, M146L in PSEN1 | P301L in MAPT | X | X | X | [56] |
| TAPP | Transgenesis | Swedish in APP | P301L in MAPT | X | X | X | [57] |
| Species | Scientific Name | Plaques | Tangles | Neurodegeneration | References |
|---|---|---|---|---|---|
| Chimpanzee | Pan troglodytes | X | X | [147,148] | |
| Orang-Utan | Pongo spp. | X | [149] | ||
| Western Gorilla | Gorilla | X | X | [150,151] | |
| Eastern Gorilla | Gorilla beringei | X | X | [152] | |
| Cynomolgus Monkey | Macaca fascicularis | X | X | [153,154,155,156] | |
| Rhesus Macaque | Macaca mulattas | X | X | [157,158] | |
| Stump Tailed macaque | Macaca arctoides | X | X | [159] | |
| Vervet Monkey | Chlorocebus aethiops | X | X | [160] | |
| Baboon | Papio hamadryas | X | X | [161,162,163] | |
| Cotton Topped Tamarin | Saguinus oedipus | X | [164] | ||
| Mouse Lemur | Microcebus murinus | X | X | X | [165,166,167] |
| Common Marmoset | Callithrix jacchus | X | X | [168,169] | |
| Squirrel Monkey | Saimiri sciureus | X | [170,171] | ||
| Pigs | Sus domesticus | X * | X * | [172] | |
| Domestic Sheep | Ovis aries | X | X | [145,173,174,175] | |
| Domestic Goat | Capra hircus | X | [173] | ||
| Bactrian Camel | Camelus bactrianus | X | [176] | ||
| Reindeer | Rangifer tarandus | X | [177] | ||
| American Bison | Bison | X | [177] | ||
| Domestic Dog | Canis familiaris | X | X | X | [178,179,180,181,182,183] |
| Domestic Cat | Felis catus | X | X | X | [184,185] |
| Leopard Cat | Prionailurus bengalensis | X | X | [186] | |
| Polar Bear | Ursus maritimus | X | [187] | ||
| Brown Bear | Ursus arctos | X | [187] | ||
| Black Bear | Ursus americanus | X | [188] | ||
| Wolverine | Gulo | X | X | [189] | |
| Harbor Seal species | Phoca largha, Phoca vitulina | X | X | [190] | |
| Sea Lion species | Eumetopias jubatus, Zalophus californianus, Neophoca cinerea | X | X | [190] | |
| Walrus | Odobenus rosmarus | X | X | [190] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mckean, N.E.; Handley, R.R.; Snell, R.G. A Review of the Current Mammalian Models of Alzheimer’s Disease and Challenges That Need to Be Overcome. Int. J. Mol. Sci. 2021, 22, 13168. https://doi.org/10.3390/ijms222313168
Mckean NE, Handley RR, Snell RG. A Review of the Current Mammalian Models of Alzheimer’s Disease and Challenges That Need to Be Overcome. International Journal of Molecular Sciences. 2021; 22(23):13168. https://doi.org/10.3390/ijms222313168
Chicago/Turabian StyleMckean, Natasha Elizabeth, Renee Robyn Handley, and Russell Grant Snell. 2021. "A Review of the Current Mammalian Models of Alzheimer’s Disease and Challenges That Need to Be Overcome" International Journal of Molecular Sciences 22, no. 23: 13168. https://doi.org/10.3390/ijms222313168
APA StyleMckean, N. E., Handley, R. R., & Snell, R. G. (2021). A Review of the Current Mammalian Models of Alzheimer’s Disease and Challenges That Need to Be Overcome. International Journal of Molecular Sciences, 22(23), 13168. https://doi.org/10.3390/ijms222313168






