Impact of Cigarette Smoking on the Expression of Oxidative Stress-Related Genes in Cumulus Cells Retrieved from Healthy Women Undergoing IVF
Abstract
1. Introduction
2. Results
2.1. Patients’ Characteristics
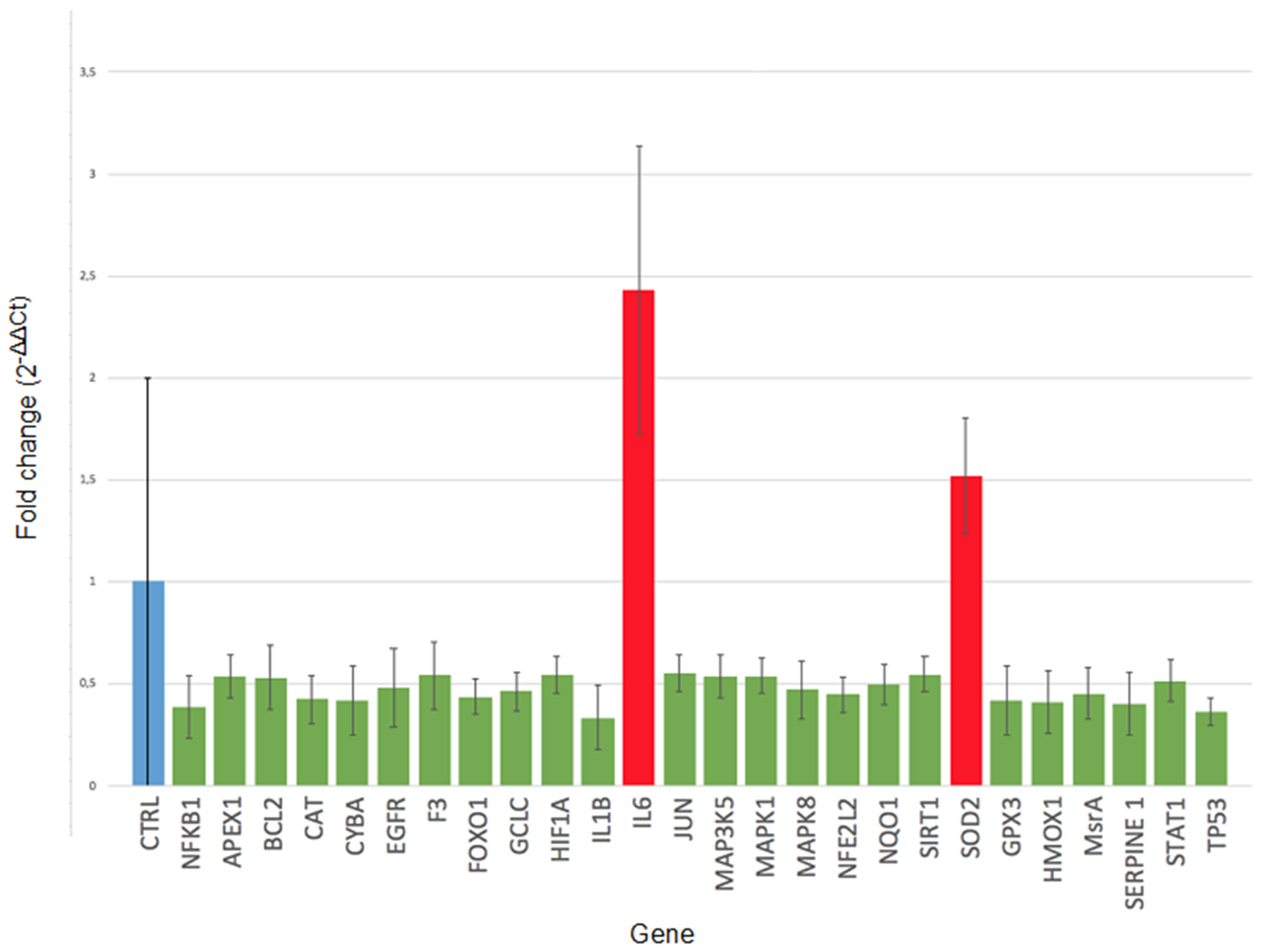
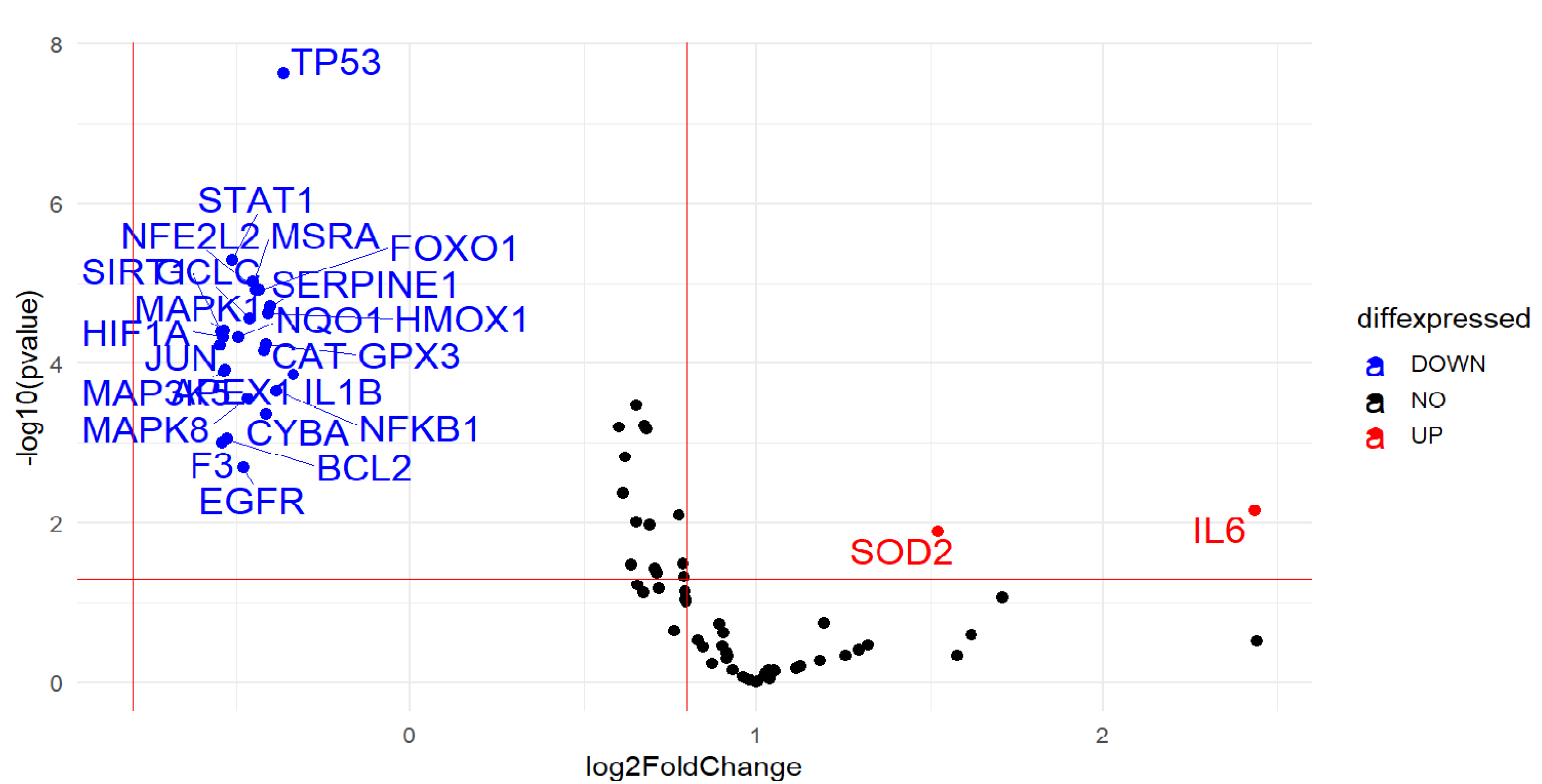
2.2. Gene Expression
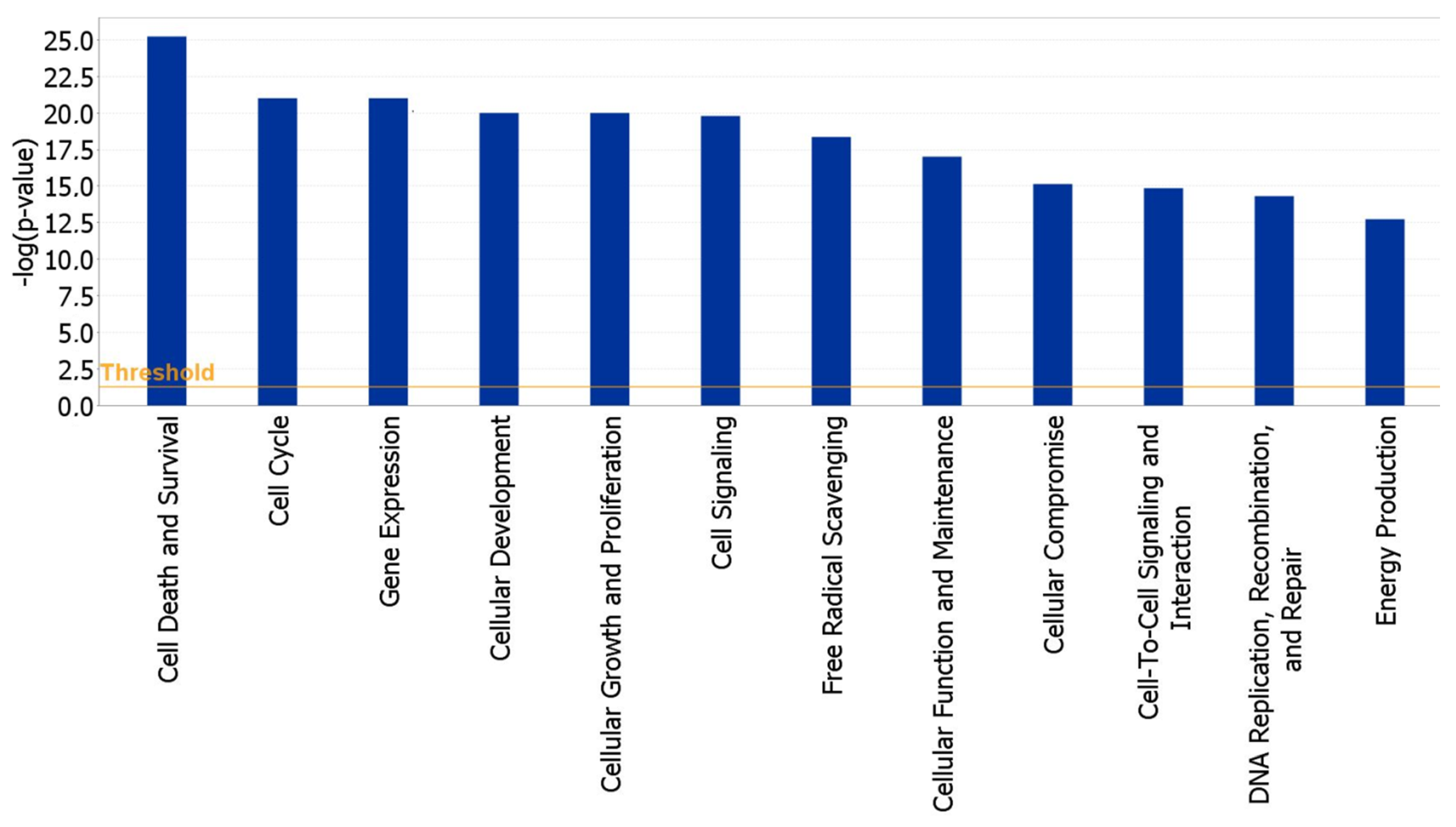
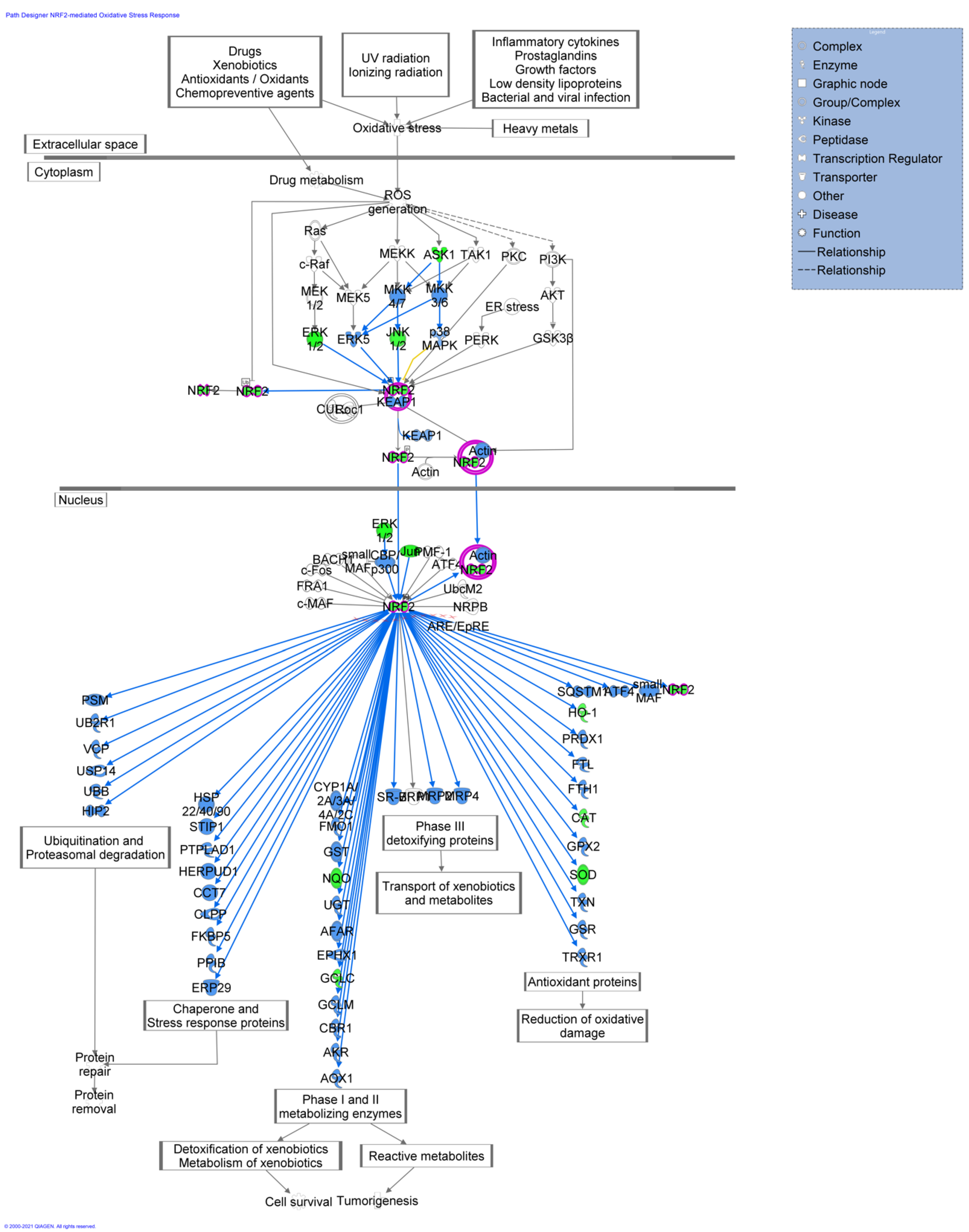
2.3. Functional and Network Analysis
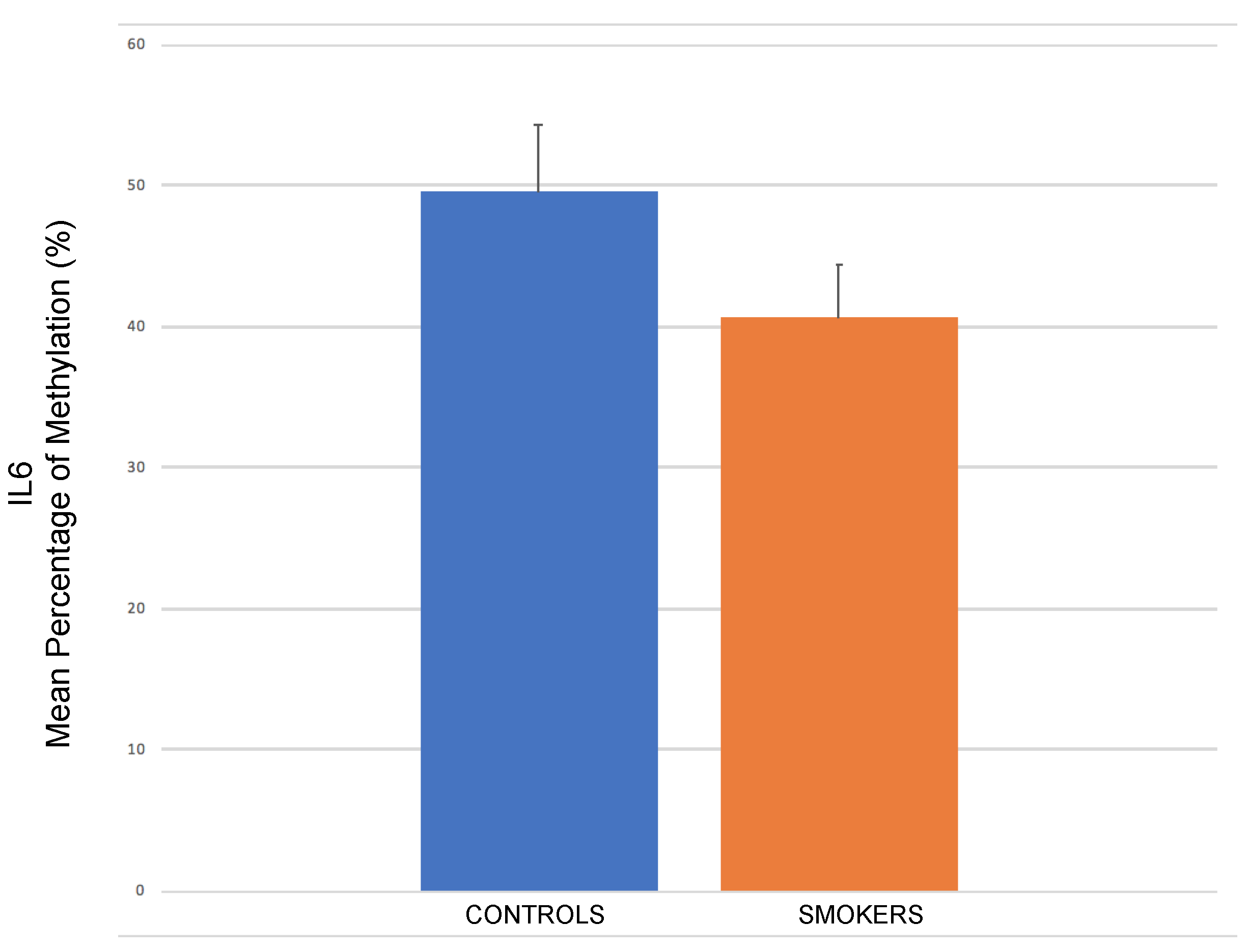
2.4. IL-6 Promoter Methylation through Pyrosequencing
3. Discussion
4. Materials and Methods
4.1. Ethical Approval
4.2. Source of Cumulus Cells and Patients’ Selection Criteria
4.3. Ovarian Stimulation Protocols
4.4. Cumulus Cells Isolation
4.5. Quantitative Real-Time PCR
4.6. IPA-Inferred Biological Networks and Upstream Regulators Analysis
4.7. Bisulfite Conversion and DNA Methylation by Pyrosequencing
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Limitations
References
- CDC, Centers for Disease Control and Prevention. Available online: Cdc.gov (accessed on 26 July 2021).
- Dechanet, C.; Anahory, T.; Daude, J.C.M.; Quantin, X.; Reyftmann, L.; Hamamah, S.; Hedon, B.; Dechaud, H. Effects of cigarette smoking on reproduction. Hum. Reprod. Update 2010, 17, 76–95. [Google Scholar] [CrossRef]
- Sharma, R.; Harlev, A.; Agarwal, A.; Esteves, S. Cigarette Smoking and Semen Quality: A New Meta-analysis Examining the Effect of the 2010 World Health Organization Laboratory Methods for the Examination of Human Semen. Eur. Urol. 2016, 70, 635–645. [Google Scholar] [CrossRef] [PubMed]
- Konstantinidou, F.; Stuppia, L.; Gatta, V. Looking Inside the World of Granulosa Cells: The Noxious Effects of Cigarette Smoke. Biomedicines 2020, 8, 309. [Google Scholar] [CrossRef]
- Budani, M.C.; Tiboni, G.M. Ovotoxicity of cigarette smoke: A systematic review of the literature. Reprod. Toxicol. 2017, 72, 164–181. [Google Scholar] [CrossRef]
- Practice Committee of the American Society for Reproductive Medicine. Smoking and infertility: A committee opinion. Fertil. Steril. 2018, 110, 611–618. [Google Scholar] [CrossRef] [PubMed]
- Budani, M.C.; Fensore, S.; Di Marzio, M.; Tiboni, G.M. Cigarette smoking impairs clinical outcomes of assisted reproductive technologies: A meta-analysis of the literature. Reprod. Toxicol. 2018, 80, 49–59. [Google Scholar] [CrossRef]
- Freour, T.; Masson, D.; Mirallie, S.; Jean, M.; Bach, K.; Dejoie, T.; Barriere, P. Active smoking compromises IVF outcome and affects ovarian reserve. Reprod. Biomed. Online 2008, 16, 96–102. [Google Scholar] [CrossRef]
- Waylen, A.; Metwally, M.; Jones, G.; Wilkinson, A.J.; Ledger, W.L. Effects of cigarette smoking upon clinical outcomes of assisted reproduction: A meta-analysis. Hum. Reprod. Update 2009, 15, 31–44. [Google Scholar] [CrossRef]
- Freour, T.; Masson, D.; Dessolle, L.; Allaoua, D.; Dejoie, T.; Mirallie, S.; Jean, M.; Barriere, P. Ovarian reserve and in vitro fer-tilization cycles outcome according to women smoking status and stimulation regimen. Arch. Gynecol. Obstet. 2012, 285, 1177–1182. [Google Scholar] [CrossRef]
- Ben-Haroush, A.; Ashekenazi, J.; Sapir, O.; Pinkas, H.; Fisch, B.; Farhi, J. High quality embyos maintain high pregnancy rates in passive smokers but not in active smokers. Reprod. Biomed. Online 2011, 22, 44–49. [Google Scholar] [CrossRef]
- Valko, M.; Leibfritz, D.; Moncola, J.; Cronin, M.T.D.; Mazura, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef]
- Sobinoff, A.; Pye, V.; Nixon, B.; Roman, S.; McLaughlin, E. Jumping the gun: Smoking constituent BaP causes premature primordial follicle activation and impairs oocyte fusibility through oxidative stress. Toxicol. Appl. Pharmacol. 2012, 260, 70–80. [Google Scholar] [CrossRef]
- Budani, M.C.; Carletti, E.; Tiboni, G.M. Cigarette smoke is associated with altered expression of antioxidant enzymes in gran-ulosa cells from women undergoing in vitro fertilization. Zygote 2017, 25, 296–303. [Google Scholar] [CrossRef]
- Budani, M.C.; D’Aurora, M.; Stuppia, L.; Gatta, V.; Tiboni, G.M. Whole-body exposure to cigarette smoke alters oocyte miRNAs expression in C57BL/6 mice. Mol. Reprod. Dev. 2019, 86, 1741–1757. [Google Scholar] [CrossRef]
- Lu, J.; Wang, Z.; Cao, J.; Chen, Y.; Dong, Y. A novel and compact review on the role of oxidative stress in female reproduction. Reprod. Biol. Endocrinol. 2018, 16, 80. [Google Scholar] [CrossRef]
- Sugino, N. Roles of reactive oxygen species in the corpus luteum. Anim. Sci. J. 2006, 77, 556–565. [Google Scholar] [CrossRef]
- Cardoso, J.P.; Cocuzza, M.; Elterman, D. Optimizing male fertility: Oxidative stress and the use of antioxidants. World J. Urol. 2019, 37, 1029–1034. [Google Scholar] [CrossRef]
- Siddique, S.; Sadeau, J.C.; Foster, W.G.; Feng, Y.L.; Zhu, J. In vitro exposure to cigarette smoke induces oxidative stress in fol-licular cells of F1 hybrid mice. J. Appl. Toxicol. 2014, 34, 224–226. [Google Scholar] [CrossRef]
- Sobinoff, A.; Beckett, E.; Jarnicki, A.; Sutherland, J.M.; McCluskey, A.; Hansbro, P.M.; McLaughlin, E.A. Scrambled and fried: Cigarette smoke exposure causes antral follicle destruction and oocyte dysfunction through oxidative stress. Toxicol. Appl. Pharmacol. 2013, 271, 156–167. [Google Scholar] [CrossRef]
- Mai, Z.; Lei, M.; Yu, B.; Du, H.; Liu, J. The Effects of Cigarette Smoke Extract on Ovulation, Oocyte Morphology and Ovarian Gene Expression in Mice. PLoS ONE 2014, 9, e95945. [Google Scholar] [CrossRef]
- Huang, Z.; Wells, D. The human oocyte and cumulus cells relationship: New insights from the cumulus cell transcriptome. Mol. Hum. Reprod. 2010, 16, 715–725. [Google Scholar] [CrossRef]
- Liu, Y.; Li, G.P.; Rickords, L.F.; White, K.L.; Sessions, B.R.; Aston, K.I.; Bunch, T.D. Effect of nicotine on in vitro maturation of bovine oocytes. Anim. Reprod. Sci. 2008, 103, 13–24. [Google Scholar] [CrossRef]
- Vrsanská, S.; Nagyová, E.; Mlynarcíková, A.; Ficková, M.; Kolena, J. Components of cigarette smoke inhibit expansion of oocyte-cumulus complexes from porcine follicles. Physiol. Res. 2003, 52, 383–387. [Google Scholar]
- Zong, D.; Liu, X.; Li, J.; Ouyang, R.; Chen, P. The role of cigarette smoke-induced epigenetic alterations in inflammation. Epigenetics Chromatin 2019, 12, 65. [Google Scholar] [CrossRef]
- Zong, D.; Ouyang, R.; Chen, P. Epigenetic mechanisms in chronic obstructive pulmonary disease. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 844–856. [Google Scholar]
- Stuppia, L.; Franzago, M.; Ballerini, P.; Gatta, V.; Antonucci, I. Epigenetics and male reproduction: The consequences of paternal lifestyle on fertility, embryo development, and children lifetime health. Clin. Epigenetics 2015, 7, 120. [Google Scholar] [CrossRef]
- Pruksananonda, K.; Wasinarom, A.; Sereepapong, W.; Sirayapiwat, P.; Rattanatanyong, P.; Mutirangura, A. Epigenetic modification of long interspersed elements-1 in cumulus cells of mature and immature oocytes from patients with polycystic ovary syndrome. Clin. Exp. Reprod. Med. 2016, 43, 82–89. [Google Scholar] [CrossRef]
- Moore, L.D.; Le, T.; Fan, G. DNA Methylation and Its Basic Function. Neuropsychopharmacology 2013, 38, 23–38. [Google Scholar] [CrossRef]
- Shaw, P.; Chattopadhyay, A. Nrf2–ARE signaling in cellular protection: Mechanism of action and the regulatory mechanisms. J. Cell. Physiol. 2020, 235, 3119–3130. [Google Scholar] [CrossRef]
- Russell, D.L.; Gilchrist, R.B.; Brown, H.M.; Thompson, J.G. Bidirectional communication between cumulus cells and the oocyte: Old hands and new players? Theriogenology 2016, 86, 62–68. [Google Scholar] [CrossRef]
- Tatone, C.; Carbone, M.C.; Falone, S.; Aimola, P.; Giardinelli, A.; Caserta, D.; Marci, R.; Pandolfi, A.; Ragnelli, A.M.; Amicarelli, F. Age-dependent changes in the expression of superoxide dismutases and catalase are associated with ultrastructural modi-fications in human granulosa cells. Mol. Hum. Reprod. 2006, 12, 655–660. [Google Scholar] [CrossRef]
- Sticozzi, C.; Cervellati, F.; Muresan, X.M.; Cervellati, C.; Valacchi, G. Resveratrol prevents cigarette smoke-induced keratinocytes damage. Food Funct. 2014, 5, 2348–2356. [Google Scholar] [CrossRef]
- Van Montfoort, A.P.; Geraedts, J.P.; Dumoulin, J.C.; Stassen, A.P.; Evers, J.L.; Ayoubi, T.A. Differential gene expression in cumulus cells as a prognostic indicator of embryo viability: A microarray analysis. Mol. Hum. Reprod. 2008, 14, 157–168. [Google Scholar] [CrossRef]
- Garbin, U.; Fratta Pasini, A.; Stranieri, C.; Cominacini, M.; Pasini, A.; Manfro, S.; Lugoboni, F.; Mozzini, C.; Guidi, G.; Faccini, G.; et al. Cigarette smoking blocks the protective expression of Nrf2/ARE pathway in peripheral mononuclear cells of young heavy smokers favouring inflammation. PLoS ONE 2009, 4, e8225. [Google Scholar] [CrossRef]
- Thimmulappa, R.K.; Lee, H.; Rangasamy, T.; Reddy, S.P.; Yamamoto, M.; Kensler, T.W.; Biswal, S. Nrf2 is a critical regulator of the innate immune response and survival during experimental sepsis. J. Clin. Investig. 2006, 116, 984–995. [Google Scholar] [CrossRef]
- Ma, R.; Liang, W.; Sun, Q.; Qiu, X.; Lin, Y.; Ge, X.; Jueraitetibaike, K.; Xie, M.; Zhou, J.; Huang, X.; et al. Sirt1/Nrf2 pathway is involved in oocyte aging by regulating Cyclin B1. Aging 2018, 10, 2991–3004. [Google Scholar] [CrossRef]
- Loboda, A.; Damulewicz, M.; Pyza, E.; Jozkowicz, A.; Dulak, J. Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: An evolutionarily conserved mechanism. Cell. Mol. Life Sci. 2016, 73, 3221–3247. [Google Scholar] [CrossRef]
- Potteti, H.R.; Reddy, N.M.; Hei, T.K.; Kalvakolanu, D.V.; Reddy, S.P. The NRF2 Activation and Antioxidative Response Are Not Impaired Overall during Hyperoxia-Induced Lung Epithelial Cell Death. Oxid. Med. Cell. Longev. 2013, 2013, 798401. [Google Scholar] [CrossRef]
- Li, Z.; Xu, W.; Su, Y.; Gao, K.; Chen, Y.; Ma, L.; Xie, Y. Nicotine induces insulin resistance via downregulation of Nrf2 in car-diomyocyte. Mol. Cell. Endocrinol. 2019, 495, 110507. [Google Scholar] [CrossRef]
- Tan, Z.; Shi, L.; Schreiber, S.S. Differential Expression of Redox Factor-1 Associated with Beta-Amyloid-Mediated Neurotoxicity. Open Neurosci. J. 2009, 3, 26–34. [Google Scholar] [CrossRef]
- Luo, M.; Delaplane, S.; Jiang, A.; Reed, A.; He, Y.; Fishel, M.; Nyland, R.L., 2nd; Borch, R.F.; Qiao, X.; Georgiadis, M.M..; et al. Role of the multifunctional DNA repair and redox signaling protein Ape1/Ref-1 in cancer and endothelial cells: Small-molecule inhibition of the redox function of Ape1. Antioxid. Redox Signal. 2008, 10, 1853–1867. [Google Scholar] [CrossRef]
- DeVallance, E.; Li, Y.; Jurczak, M.J.; Cifuentes-Pagano, E.; Pagano, P.J. The Role of NADPH Oxidases in the Etiology of Obesity and Metabolic Syndrome: Contribution of Individual Isoforms and Cell Biology. Antioxid. Redox Signal. 2019, 31, 687–709. [Google Scholar] [CrossRef]
- Dong, Q.; Xing, W.; Li, K.; Zhou, X.; Wang, S.; Zhang, H. Tetrahydroxystilbene glycoside improves endothelial dys-function and hypertension in obese rats: The role of omentin-1. Biochem. Pharmacol. 2021, 186, 114489. [Google Scholar] [CrossRef]
- Guan, R.; Cai, Z.; Wang, J.; Ding, M.; Li, Z.; Xu, J.; Li, Y.; Li, J.; Yao, H.; Liu, W.; et al. Hydrogen sulfide attenuates mitochondrial dysfunction-induced cellular senescence and apoptosis in alveolar epithelial cells by upregulating sirtuin 1. Aging 2019, 11, 11844–11864. [Google Scholar] [CrossRef]
- Michan, S.; Sinclair, D. Sirtuins in mammals: Insights into their biological function. Biochem. J. 2007, 404, 1–13. [Google Scholar] [CrossRef]
- Yao, H.; Chung, S.; Hwang, J.W.; Rajendrasozhan, S.; Sundar, I.K.; Dean, D.A.; McBurney, M.W.; Guarente, L.; Gu, W.; Rönty, M.; et al. SIRT1 protects against emphysema via FOXO3-mediated reduction of premature senescence in mice. J. Clin. Investig. 2012, 122, 2032–2045. [Google Scholar] [CrossRef]
- Gao, N.; Wang, Y.; Zheng, C.M.; Gao, Y.L.; Li, H.; Li, Y.; Fu, T.T.; Xu, L.L.; Wang, W.; Ying, S.; et al. β2-Microglobulin participates in development of lung emphysema by inducing lung epithelial cell senescence. Am. J. Physiol. Cell. Mol. Physiol. 2017, 312, L669–L677. [Google Scholar] [CrossRef]
- Di Vincenzo, S.; Heijink, I.H.; Noordhoek, J.A.; Cipollina, C.; Siena, L.; Bruno, A.; Ferraro, M.; Postma, D.S.; Gjomarkaj, M.; Pace, E. SIRT1/FoxO3 axis alteration leads to aberrant immune responses in bronchial epithelial cells. J. Cell. Mol. Med. 2018, 22, 2272–2282. [Google Scholar] [CrossRef]
- Di Emidio, G.; Falone, S.; Vitti, M.; D’Alessandro, A.M.; Vento, M.; DI Pietro, C.S.; Amicarelli, F.; Tatone, C. SIRT1 signalling protects mouse oocytes against oxidative stress and is deregulated during aging. Hum. Reprod. 2014, 29, 2006–2017. [Google Scholar] [CrossRef]
- Meng, L.; Wu, Z.; Zhao, K.; Tao, J.; Chit, T.; Zhang, S.; Wang, C.C.; Teerds, K. Transcriptome Analysis of Porcine Granulosa Cells in Healthy and Atretic Follicles: Role of Steroidogenesis and Oxidative Stress. Antioxidants 2020, 10, 22. [Google Scholar] [CrossRef]
- Liu, Y.; Zuo, H.; Wang, Y.; Tian, L.; Xu, X.; Xiong, J.; Pei, X. Ethanol promotes apoptosis in rat ovarian granulosa cells via the Bcl-2 family dependent intrinsic apoptotic pathway. Cell. Mol. Biol. 2018, 64, 118–125. [Google Scholar] [CrossRef]
- Lingappan, K. NF-κB in Oxidative Stress. Curr. Opin. Toxicol. 2018, 7, 81–86. [Google Scholar] [CrossRef]
- Hsieh, M.; Thao, K.; Conti, M. Genetic dissection of epidermal growth factor receptor signaling during luteinizing hor-mone-induced oocyte maturation. PLoS ONE 2011, 6, e21574. [Google Scholar] [CrossRef]
- Field, S.L.; Dasgupta, T.; Cummings, M.; Orsi, N.M. Cytokines in ovarian folliculogenesis, oocyte maturation and luteinisation. Mol. Reprod. Dev. 2014, 81, 284–314. [Google Scholar] [CrossRef]
- Basini, G.; Baratta, M.; Bussolati, S.; Tamanini, C. Interleukin-1 beta fragment (163-171) modulates bovine granulosa cell pro-liferation in vitro: Dependence on size of follicle. J. Reprod. Immunol. 1998, 37, 139–153. [Google Scholar] [CrossRef]
- Silva, J.; Lima, F.; Souza, A.; Silva, A. Interleukin-1β and TNF-α systems in ovarian follicles and their roles during follicular development, oocyte maturation and ovulation. Zygote 2020, 28, 270–277. [Google Scholar] [CrossRef]
- Ahsan, S.; Lacey, M.; Whitehead, S.A. Interactions between interleukin-1 beta, nitric oxide and prostaglandin E2 in the rat ovary: Effects on steroidogenesis. Eur. J. Endocrinol. 1997, 137, 293–300. [Google Scholar] [CrossRef][Green Version]
- Xu, B.; Zhang, Y.W.; Tong, X.H.; Liu, Y.S. Characterization of microRNA profile in human cumulus granulosa cells: Identifi-cation of microRNAs that regulate Notch signaling and are associated with PCOS. Mol. Cell. Endocrinol. 2015, 404, 26–36. [Google Scholar] [CrossRef]
- Schreiber, M.; Kolbus, A.; Piu, F.; Szabowski, A.; Möhle-Steinlein, U.; Tian, J.; Karin, M.; Angel, P.; Wagner, E.F. Control of cell cycle progression by c-Jun is p53 dependent. Genes Dev. 1999, 13, 607–619. [Google Scholar] [CrossRef]
- Salih, D.A.; Brunet, A. FoxO transcription factors in the maintenance of cellular homeostasis during aging. Curr. Opin. Cell Biol. 2008, 20, 126–136. [Google Scholar] [CrossRef]
- Pisarska, M.D.; Kuo, F.T.; Tang, D.; Zarrini, P.; Khan, S.; Ketefian, A. Expression of forkhead transcription factors in human granulosa cells. Fertil. Steril. 2009, 91, 1392–1394. [Google Scholar] [CrossRef]
- Artini, P.G.; Tatone, C.; Sperduti, S.; D’Aurora, M.; Franchi, S.; Di Emidio, G.; Ciriminna, R.; Vento, M.; Di Pietro, C.; Stuppia, L.; et al. Cumulus cells surrounding oocytes with high developmental competence exhibit down-regulation of phosphoinositol 1.3 kinase/protein kinase B (PI3K/AKT) signalling genes involved in proliferation and survival. Hum. Reprod. 2017, 32, 2474–2484. [Google Scholar] [CrossRef]
- Shen, M.; Jiang, Y.; Guan, Z.; Cao, Y.; Li, L.; Liu, H.; Sun, S.C. Protective mechanism of FSH against oxidative damage in mouse ovarian granulosa cells by repressing autophagy. Autophagy 2017, 13, 1364–1385. [Google Scholar] [CrossRef]
- Turhan, A.; Pereira, M.T.; Schuler, G.; Bleul, U.; Kowalewski, M.P. Hypoxia-inducible factor (HIF1alpha) inhibition modulates cumulus cell function and affects bovine oocyte maturation in vitro†. Biol. Reprod. 2021, 104, 479–491. [Google Scholar] [CrossRef]
- Wei, H.; Wang, J.; Liang, Z. STAT1-p53-p21axis-dependent stress-induced progression of chronic nephrosis in adriamy-cin-induced mouse model. Ann. Transl. Med. 2020, 8, 1002. [Google Scholar] [CrossRef]
- Townsend, P.A.; Scarabelli, T.M.; Davidson, S.M.; Knight, R.A.; Latchman, D.S.; Stephanou, A. STAT-1 interacts with p53 to enhance DNA damage-induced apoptosis. J. Biol. Chem. 2004, 279, 5811–5820. [Google Scholar] [CrossRef]
- Yu, B.Y.; Subudeng, G.; Du, C.G.; Liu, Z.H.; Zhao, Y.F.; Namei, E.; Bai, Y.; Yang, B.X.; Li, H.J. Plasminogen activator, tissue type regulates germinal vesicle breakdown and cumulus expansion of bovine cumulus–oocyte complex in vitro. Biol. Reprod. 2019, 100, 1473–1481. [Google Scholar] [CrossRef]
- Bajou, K.; Peng, H.; Laug, W.E.; Maillard, C.; Noel, A.; Foidart, J.M.; Martial, J.A.; DeClerck, Y.A. Plasminogen activator in-hibitor-1 protects endothelial cells from FasL-mediated apoptosis. Cancer Cell. 2008, 14, 324–334. [Google Scholar] [CrossRef]
- Dosunmu-Ogunbi, A.M.; Wood, K.C.; Novelli, E.M.; Straub, A.C. Decoding the role of SOD2 in sickle cell disease. Blood Adv. 2019, 3, 2679–2687. [Google Scholar] [CrossRef]
- Zhao, X.; Chen, S.; Zhao, C.; Xia, F. Maternal Immune System and State of Inflammation Dictate the Fate and Severity of Disease in Preeclampsia. J. Immunol. Res. 2021, 2021, 9947884. [Google Scholar] [CrossRef]
- Assou, S.; Pourret, E.; Péquignot, M.; Rigau, V.; Kalatzis, V.; Aït-Ahmed, O.; Hamamah, S. Cultured Cells from the Human Oocyte Cumulus Niche Are Efficient Feeders to Propagate Pluripotent Stem Cells. Stem Cells Dev. 2015, 24, 2317–2327. [Google Scholar] [CrossRef]
- Zuo, H.P.; Guo, Y.Y.; Che, L.; Wu, X.Z. Hypomethylation of Interleukin-6 Promoter is Associated with the Risk of Coronary Heart Disease. Arq. Bras. Cardiol. 2016, 107, 131–136. [Google Scholar] [CrossRef]
- Nampoothiri, L.P.; Agarwal, A.; Gupta, S. Effect of co-exposure to lead and cadmium on antioxidant status in rat ovarian granulose cells. Arch. Toxicol. 2007, 81, 145–150. [Google Scholar] [CrossRef]
- Meng, L.; Zhao, K.; Wang, C.; Tao, J.; Wu, Z.; Teerds, K.; Zhang, S. Characterization of Long Non-Coding RNA Profiles in Porcine Granulosa Cells of Healthy and Atretic Antral Follicles: Implications for a Potential Role in Apoptosis. Int. J. Mol. Sci. 2021, 22, 2677. [Google Scholar] [CrossRef]
- Nuñez-Calonge, R.; Rancan, L.; Cortes, S.; Vara, E.; Andres, C.; Caballero, P.; Fernandez-Tresguerres, J. Apoptotic markers and antioxidant enzymes have altered expression in cumulus and granulosa cells of young women with poor response to ovarian stimulation. J. Reprod. Biol. Endocrinol. 2019, 3, 1–6. [Google Scholar]
- Tiboni, G.M.; Bucciarelli, T.; Giampietro, F.; Sulpizio, M.; Di Ilio, C. Influence of cigarette smoking on vitamin E, vitamin A, beta-carotene and lycopene concentrations in human pre-ovulatory follicular fluid. Int. J. Immunopathol. Pharmacol. 2004, 17, 389–393. [Google Scholar] [CrossRef]
- Yang, L.; Chen, Y.; Liu, Y.; Xing, Y.; Miao, C.; Zhao, Y.; Chang, X.; Zhang, Q. The Role of Oxidative Stress and Natural An-tioxidants in Ovarian Aging. Front. Pharmacol. 2021, 11, 617843. [Google Scholar] [CrossRef]
- Sinkó, I.; Mórocz, M.; Zádori, J.; Kokavszky, K.; Raskó, I. Effect of cigarette smoking on DNA damage of human cumulus cells analyzed by comet assay. Reprod. Toxicol. 2005, 20, 65–71. [Google Scholar] [CrossRef]
- Shaeib, F.; Khan, S.N.; Ali, I.; Thakur, M.; Saed, M.G.; Dai, J.; Awonuga, A.O.; Banerjee, J.; Abu-Soud, H.M. The defensive role of cumulus cells against reactive oxygen species insult in metaphase II mouse oocytes. Reprod. Sci. 2016, 23, 498–507. [Google Scholar] [CrossRef]
- Rahman, I.; Morrison, D.; Donaldson, K.; MacNee, W. Systemic oxidative stress in asthma, COPD, and smokers. Am. J. Respir. Crit. Care Med. 1996, 154, 1055–1060. [Google Scholar] [CrossRef]

| Non Smokers (n = 5) | Smokers (n = 5) | p-Value | |
|---|---|---|---|
| Mean n° of cigarettes smoked/daily | - | 8.2 | |
| Age | 30.8 ± 0.8 | 33.2 ± 2.6 | 0.08 |
| AMH | 8.6 ± 2.5 | 9.2 ± 8.3 | 0.9 |
| BMI | 25.3 ± 4.5 | 24.6 ± 5.4 | 0.8 |
| Previous IVF attempts | 1.4 ± 0.5 | 1.4 ± 0.5 | 1 |
| Days of stimulation | 9.4 ± 2.1 | 9.8 ± 0.8 | 0.7 |
| Total dosage of gonadotropin | 1260.0 ± 697.6 | 1695.0 ± 1336.8 | 0.5 |
| Oocytes retrieved at ovum pick-up | 11.6 ± 2.1 | 12.2 ± 8.9 | 0.9 |
| N° of MII oocytes | 8.4 ± 1.9 | 8.8 ± 7.4 | 0.9 |
| ID | Expr Log Ratio | Entrez Gene Name | Location | Type(s) |
|---|---|---|---|---|
| APEX1 | −0.534 | apurinic/apyrimidinic endodeoxyribonuclease 1 | Nucleus | enzyme |
| BCL2 | −0.528 | BCL2 apoptosis regulator | Cytoplasm | transporter |
| CAT | −0.422 | catalase | Cytoplasm | enzyme |
| CYBA | −0.414 | cytochrome b-245 alpha chain | Cytoplasm | enzyme |
| EGFR | −0.480 | epidermal growth factor receptor | Plasma Membrane | kinase |
| F3 | −0.540 | coagulation factor III, tissue factor | Plasma Membrane | transmembrane receptor |
| FOXO1 | −0.435 | forkhead box O1 | Nucleus | transcription regulator |
| GCLC | −0.461 | glutamate-cysteine ligase catalytic subunit | Cytoplasm | enzyme |
| GPX3 | −0.416 | glutathione peroxidase 3 | Extracellular Space | enzyme |
| HIF1A | −0.539 | hypoxia inducible factor 1 subunit alpha | Nucleus | transcription regulator |
| HMOX1 | −0.408 | heme oxygenase 1 | Cytoplasm | enzyme |
| IL1B | −0.335 | interleukin 1 beta | Extracellular Space | cytokine |
| IL6 | 2.432 | interleukin 6 | Extracellular Space | cytokine |
| JUN | −0.548 | Jun proto-oncogene, AP-1 transcription factor subunit | Nucleus | transcription regulator |
| MAP3K5 | −0.533 | mitogen-activated protein kinase kinase kinase 5 | Cytoplasm | kinase |
| MAPK1 | −0.536 | mitogen-activated protein kinase 1 | Cytoplasm | kinase |
| MAPK8 | −0.469 | mitogen-activated protein kinase 8 | Cytoplasm | kinase |
| MSRA | −0.452 | methionine sulfoxide reductase A | Cytoplasm | enzyme |
| NFE2L2 | −0.445 | nuclear factor, erythroid 2 like 2 | Nucleus | transcription regulator |
| NFKB1 | −0.386 | nuclear factor kappa B subunit 1 | Nucleus | transcription regulator |
| NQO1 | −0.495 | NAD(P)H quinone dehydrogenase 1 | Cytoplasm | enzyme |
| SERPINE1 | −0.402 | serpin family E member 1 | Extracellular Space | other |
| SIRT1 | −0.544 | sirtuin 1 | Nucleus | transcription regulator |
| SOD2 | 1.520 | superoxide dismutase 2 | Cytoplasm | enzyme |
| STAT1 | −0.513 | signal transducer and activator of transcription 1 | Nucleus | transcription regulator |
| TP53 | −0.364 | tumor protein p53 | Nucleus | transcription regulator |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Konstantinidou, F.; Budani, M.C.; Sarra, A.; Stuppia, L.; Tiboni, G.M.; Gatta, V. Impact of Cigarette Smoking on the Expression of Oxidative Stress-Related Genes in Cumulus Cells Retrieved from Healthy Women Undergoing IVF. Int. J. Mol. Sci. 2021, 22, 13147. https://doi.org/10.3390/ijms222313147
Konstantinidou F, Budani MC, Sarra A, Stuppia L, Tiboni GM, Gatta V. Impact of Cigarette Smoking on the Expression of Oxidative Stress-Related Genes in Cumulus Cells Retrieved from Healthy Women Undergoing IVF. International Journal of Molecular Sciences. 2021; 22(23):13147. https://doi.org/10.3390/ijms222313147
Chicago/Turabian StyleKonstantinidou, Fani, Maria Cristina Budani, Annalina Sarra, Liborio Stuppia, Gian Mario Tiboni, and Valentina Gatta. 2021. "Impact of Cigarette Smoking on the Expression of Oxidative Stress-Related Genes in Cumulus Cells Retrieved from Healthy Women Undergoing IVF" International Journal of Molecular Sciences 22, no. 23: 13147. https://doi.org/10.3390/ijms222313147
APA StyleKonstantinidou, F., Budani, M. C., Sarra, A., Stuppia, L., Tiboni, G. M., & Gatta, V. (2021). Impact of Cigarette Smoking on the Expression of Oxidative Stress-Related Genes in Cumulus Cells Retrieved from Healthy Women Undergoing IVF. International Journal of Molecular Sciences, 22(23), 13147. https://doi.org/10.3390/ijms222313147






