Evaluation of the Physico-Chemical Properties of Liposomes Assembled from Bioconjugates of Anisic Acid with Phosphatidylcholine
Abstract
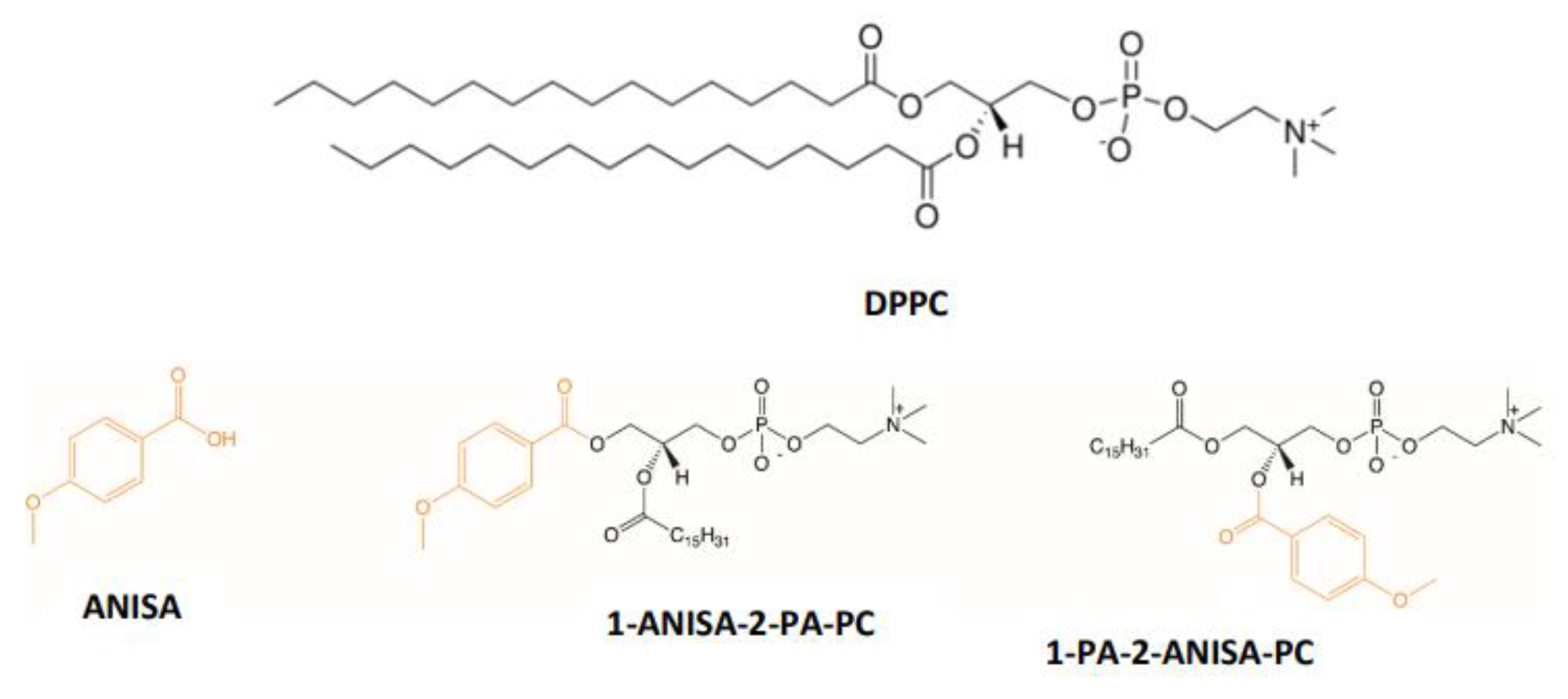
1. Introduction
2. Results and Discussion
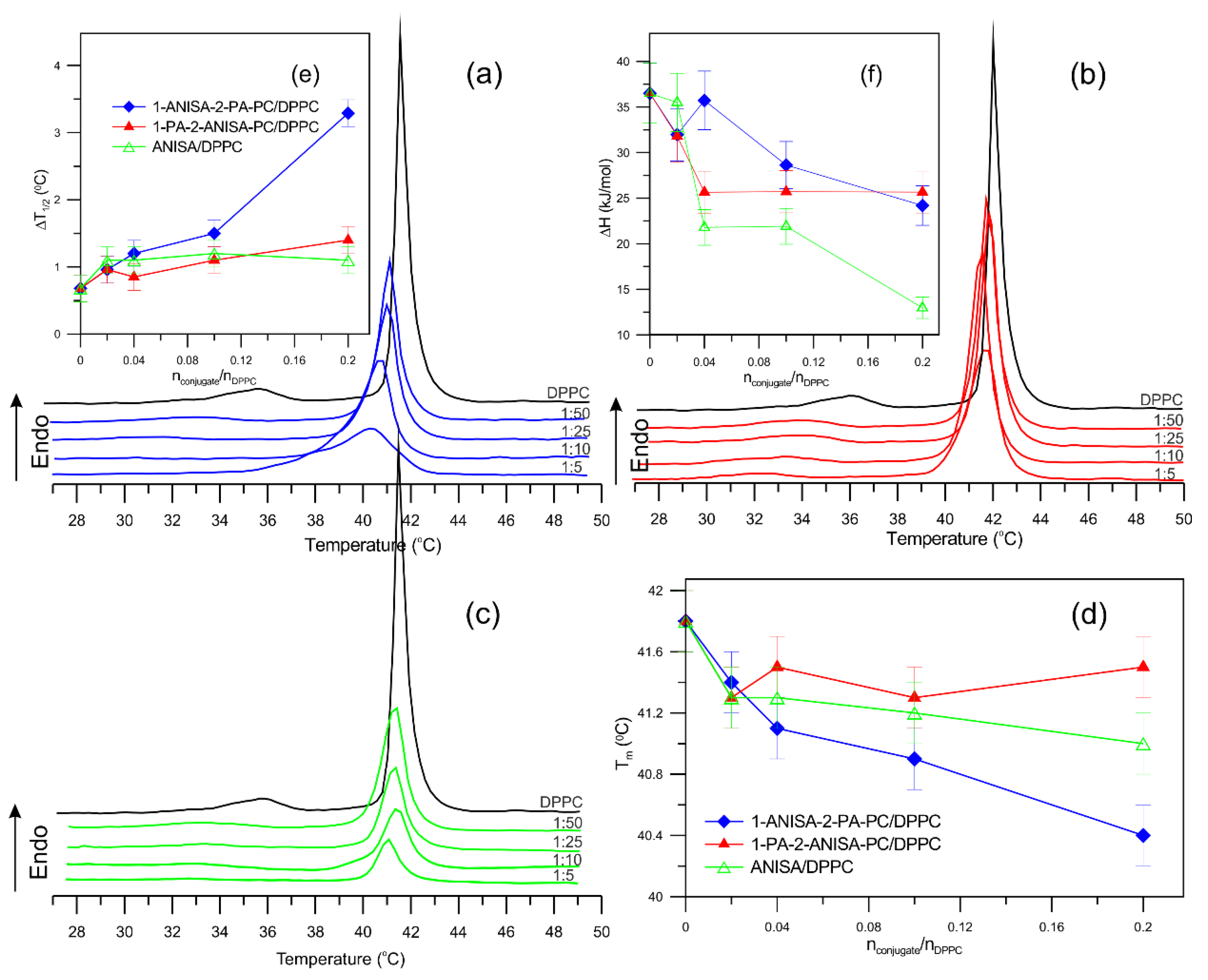
2.1. Differential Scanning Calorimetry (DSC)
2.2. Spectroscopy Methods
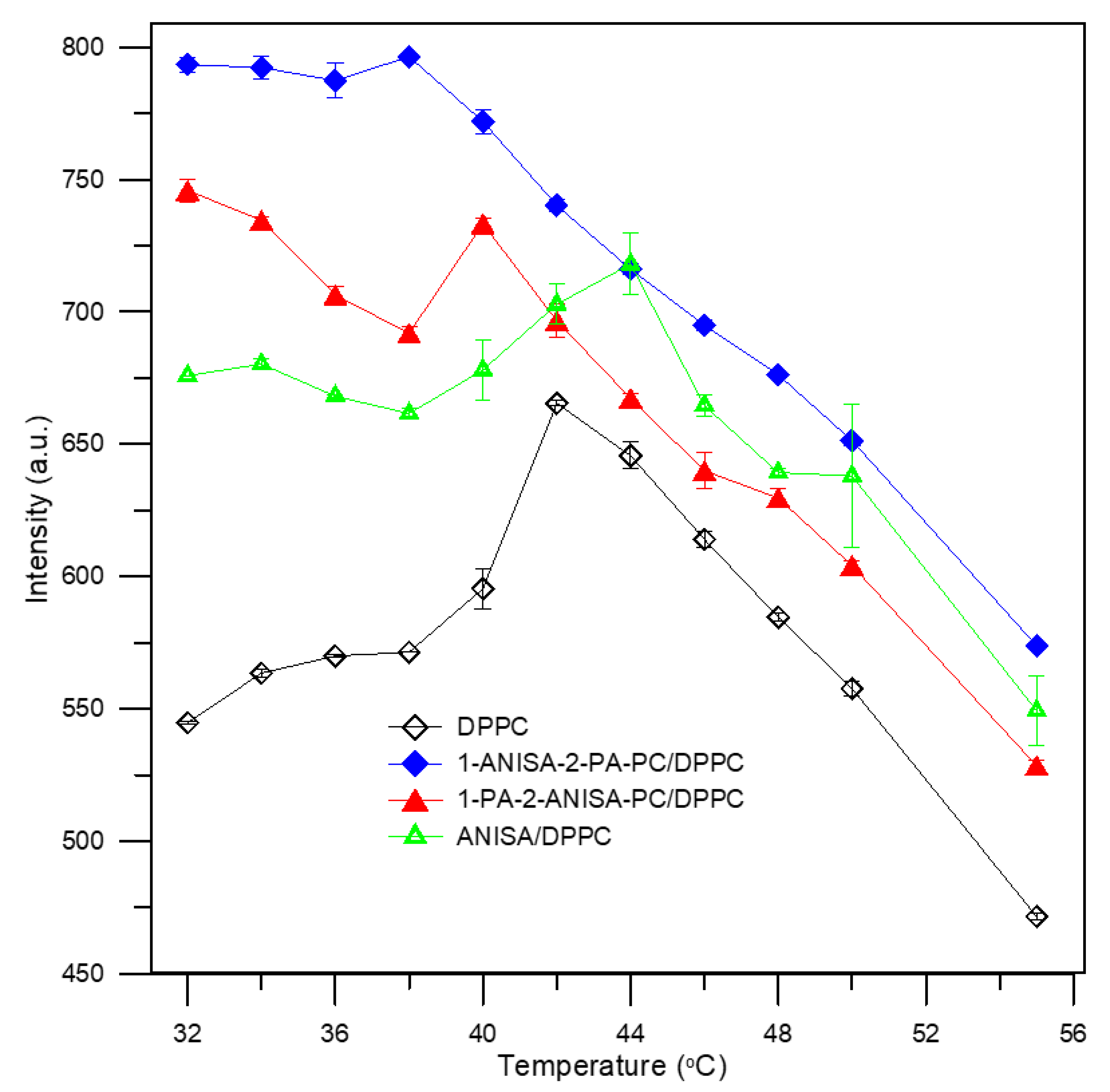
2.2.1. Steady-State Fluorescence Spectroscopy
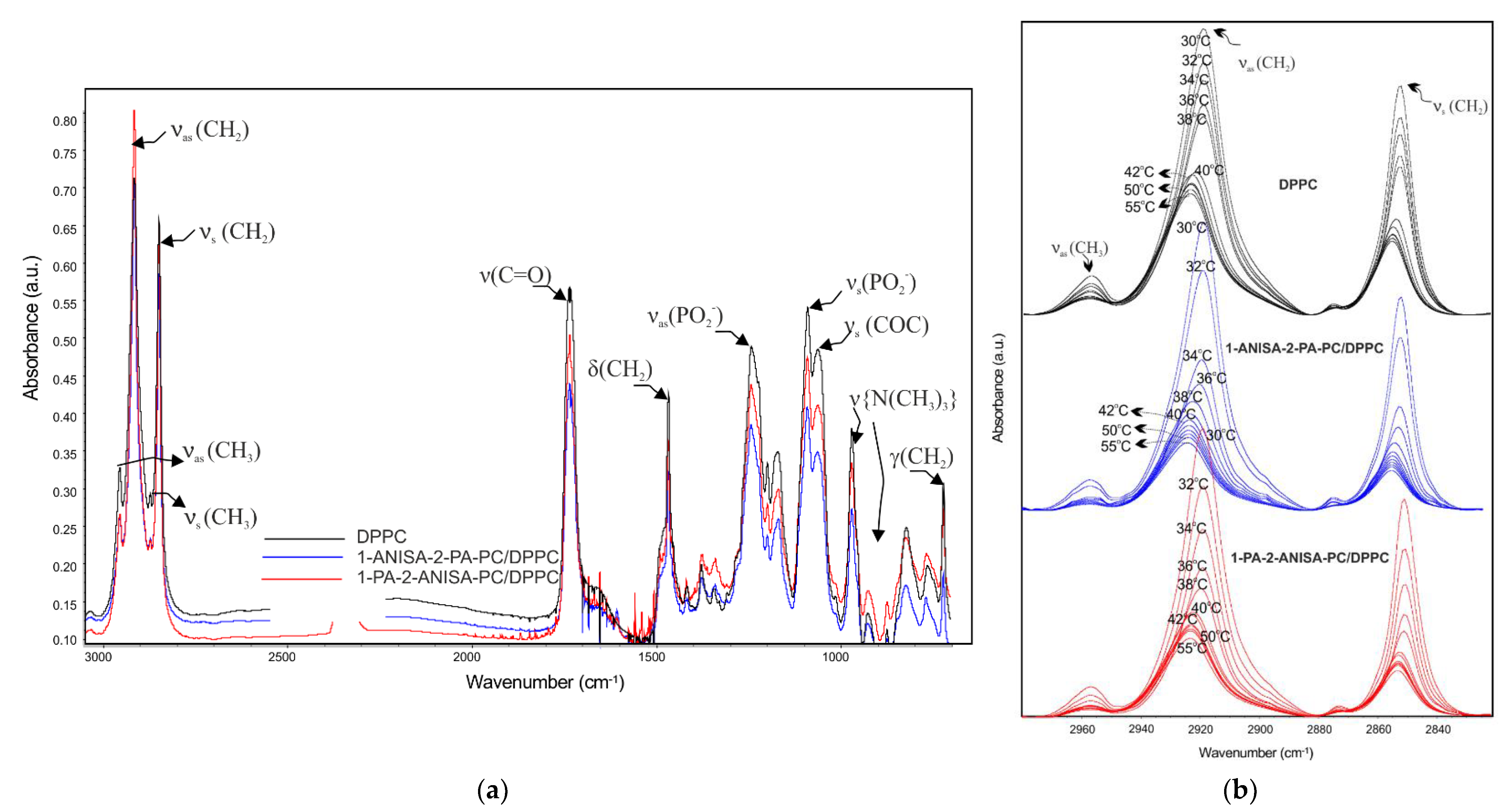
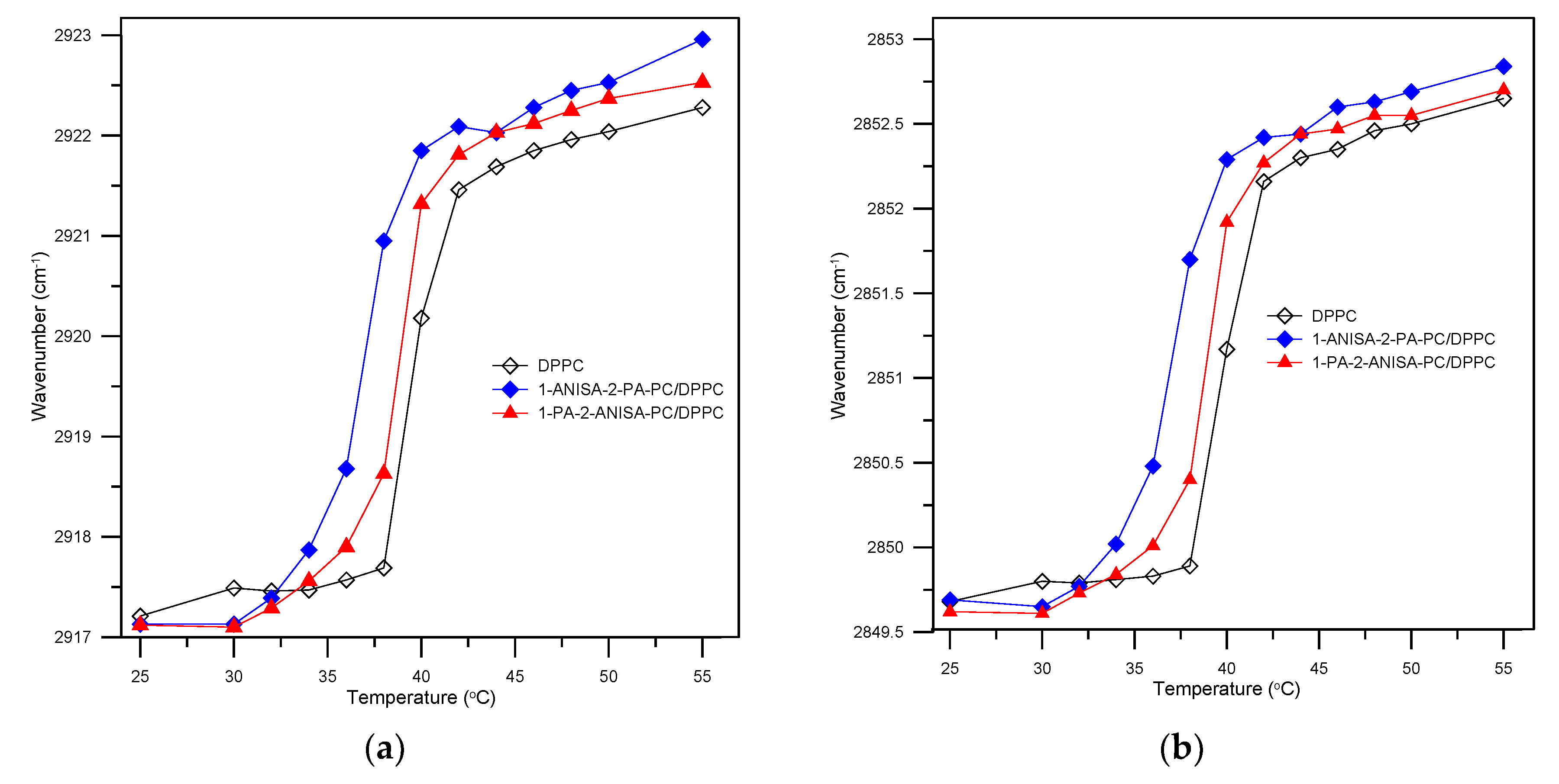
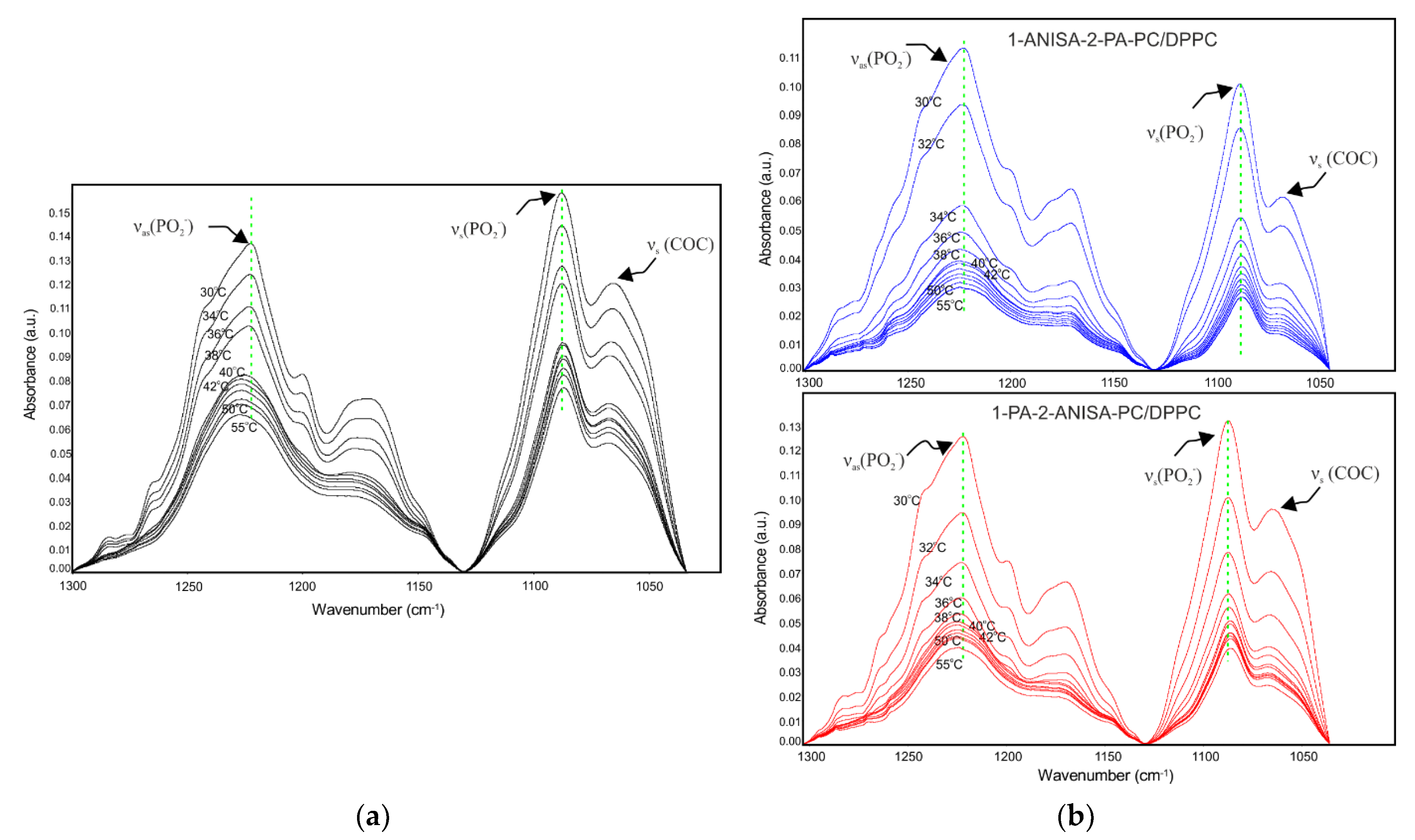
2.2.2. Attenuated Total Reflectance Fourier Transform Infrared Spectroscopy (ATR-FTIR)
3. Summary
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Liposome Preparation
4.3. Differential Scanning Calorimetry (DSC)
4.4. Spectroscopy Methods
4.4.1. Steady-State Fluorescence Spectroscopy
4.4.2. Attenuated Total Reflectance Fourier Transform Infrared Spectroscopy (ATR-FTIR)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bangham, A.D.; Standish, M.M.; Watkins, J.C. Diffusion of univalent ions across the lamellae of swollen phospholipids. J. Mol. Biol. 1965, 13, 238–252. [Google Scholar] [CrossRef]
- Chu, Z.; Zhang, S.; Zhang, B.; Zhang, C.; Fang, C.Y.; Rehor, I.; Cigler, P.; Chang, H.C.; Lin, G.; Liu, R.; et al. Unambiguous observation of shape effects on cellular fate of nanoparticles. Sci. Rep. 2014, 4, 4495. [Google Scholar] [CrossRef]
- Gregoriadis, G.; Ryman, B.E. Liposomes as carriers of enzymes or drugs—New approach to treatment of storage diseases. Biochem. J. 1971, 124, P58. [Google Scholar]
- Jones, M.N. The surface properties of phospholipid liposome systems and their characterisation. Adv. Colloid Interface Sci. 1995, 54, 93–128. [Google Scholar] [CrossRef]
- Chang, H.-I.; Yeh, M.-K. Clinical development of liposome-based drugs: Formulation, characterization, and therapeutic efficacy. Int. J. Nanomed. 2012, 7, 49–60. [Google Scholar]
- Allen, T.M.; Cullis, P.R. Liposomal drug delivery systems: From concept to clinical applications. Adv. Drug Deliv. Rev. 2013, 65, 36–48. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Jaafari, M.R.; Szoka, F.C. Disterolphospholipids: Nonexchangeable lipids and their application to liposomal drug delivery. Angew. Chemie—Int. Ed. 2009, 48, 4146–4149. [Google Scholar] [CrossRef] [PubMed]
- Linderoth, L.; Peters, G.H.; Madsen, R.; Andresen, T.L. Drug delivery by an enzyme-mediated cyclization of a lipid prodrug with unique bilayer-formation properties. Angew. Chemie—Int. Ed. 2009, 48, 1823–1826. [Google Scholar] [CrossRef]
- Fang, S.; Niu, Y.; Zhu, W.; Zhang, Y.; Yu, L.; Li, X. Liposomes assembled from a dual drug-tailed phospholipid for cancer therapy. Chem. Asian J. 2015, 10, 1232–1238. [Google Scholar] [CrossRef]
- Gliszczyńska, A.; Niezgoda, N.; Gładkowski, W.; Czarnecka, M.; Świtalska, M.; Wietrzyk, J. Synthesis and biological evaluation of novel phosphatidylcholine analogues containing monoterpene acids as potent antiproliferative agents. PLoS ONE 2016, 11, e0157278. [Google Scholar] [CrossRef]
- Gliszczyńska, A.; Niezgoda, N.; Gładkowski, W.; Świtalska, M.; Wietrzyk, J. Isoprenoid-phospholipid conjugates as potential therapeutic agents: Synthesis, characterization and antiproliferative studies. PLoS ONE 2017, 12, e0172238. [Google Scholar] [CrossRef]
- Czarnecka, M.; Świtalska, M.; Wietrzyk, J.; Maciejewska, G.; Gliszczyńska, A. Synthesis, characterization, and in vitro cancer cell growth inhibition evaluation of novel phosphatidylcholines with anisic and veratric acids. Molecules 2018, 23, 2022. [Google Scholar] [CrossRef]
- Czarnecka, M.; Świtalska, M.; Wietrzyk, J.; Maciejewska, G.; Gliszczyńska, A. Synthesis and biological evaluation of phosphatidylcholines with cinnamic and 3-methoxycinnamic acids with potent antiproliferative activity. RSC Adv. 2018, 8, 35744–35752. [Google Scholar] [CrossRef]
- Palko-Łabuz, A.; Gliszczyńska, A.; Skonieczna, M.; Poła, A.; Wesołowska, O.; Środa-Pomianek, K. Conjugation with phospholipids as a modification increasing anticancer activity of phenolic acids in metastatic melanoma—In vitro and in silico studies. Int. J. Mol. Sci. 2021, 22, 8397. [Google Scholar] [CrossRef]
- Pentak, D. Evaluation of the physicochemical properties of liposomes as potential carriers of anticancer drugs: Spectroscopic study. J. Nanoparticle Res. 2016, 18, 126. [Google Scholar] [CrossRef]
- Drabik, D.; Chodaczek, G.; Kraszewski, S.; Langner, M. Mechanical Properties Determination of DMPC, DPPC, DSPC, and HSPC Solid-Ordered Bilayers. Langmuir 2020, 36, 3826–3835. [Google Scholar] [CrossRef]
- Tamai, N.; Inazawa, S.; Takeuchi, S.; Goto, M.; Matsuki, H. Phase behavior of binary bilayer membrane of dipalmitoylphosphatidylcholine and stigmasterol. J. Therm. Anal. Calorim. 2019, 135, 2635–2645. [Google Scholar] [CrossRef]
- Arouri, A.; Mouritsen, O.G. Anticancer double lipid prodrugs: Liposomal preparation and characterization. J. Liposome Res. 2011, 21, 296–305. [Google Scholar] [CrossRef]
- Sieber, F. Merocyanine 540. Photochem. Photobiol. 1987, 46, 1035–1042. [Google Scholar] [CrossRef]
- Lagerberg, J.W.M.; VanSteveninck, J.; Dubbelman, T.M.A.R. Effect of hydrogen peroxide on the binding of Merocyanine 540 to human erythrocytes. Cell. Mol. Life Sci. 1997, 53, 257–262. [Google Scholar] [CrossRef]
- Mateašik, A.; Šikurová, L.; Chorvát, D. Interaction of merocyanine 540 with charged membranes. Bioelectrochemistry 2002, 55, 173–175. [Google Scholar] [CrossRef]
- Williamson, P.; Mattocks, K.; Schlegel, R.A. Merocyanine 540, a fluorescent probe sensitive to lipid packing. BBA—Biomembr. 1983, 732, 387–393. [Google Scholar] [CrossRef]
- Langner, M.; Hui, S.W. Merocyanine interaction with phosphatidylcholine bilayers. BBA—Biomembr. 1993, 1149, 175–179. [Google Scholar] [CrossRef]
- Parasassi, T.; Krasnowska, E.K.; Bagatolli, L.; Gratton, E. Laurdan and Prodan as Polarity-Sensitive Fluorescent Membrane Probes. J. Fluoresc. 1998, 8, 365–373. [Google Scholar] [CrossRef]
- Parasassi, T.; De Stasio, G.; Ravagnan, G.; Rusch, R.M.; Gratton, E. Quantitation of lipid phases in phospholipid vesicles by the generalized polarization of Laurdan fluorescence. Biophys. J. 1991, 60, 179–189. [Google Scholar] [CrossRef]
- Engelke, M.; Bojarski, P.; Bloß, R.; Diehl, H. Tamoxifen perturbs lipid bilayer order and permeability: Comparison of DSC, fluorescence anisotropy, Laurdan generalized polarization and carboxyfluorescein leakage studies. Biophys. Chem. 2001, 90, 157–173. [Google Scholar] [CrossRef]
- Pruchnik, H.; Włoch, A.; Żyłka, R.; Oszmiański, J.; Kleszczyńska, H. Interaction of skullcap (Scutellaria baicalensis Georgi) and buckwheat (Fagopyrum esculentum Moench) extracts with lipid bilayers. J. Therm. Anal. Calorim. 2015, 121, 475–484. [Google Scholar] [CrossRef][Green Version]
- Wu, F.G.; Jia, Q.; Wu, R.G.; Yu, Z.W. Regional cooperativity in the phase transitions of dipalmitoylphosphatidylcholine bilayers: The lipid tail triggers the isothermal crystallization process. J. Phys. Chem. B 2011, 115, 8559–8568. [Google Scholar] [CrossRef]
- Pentak, D. In vitro spectroscopic study of piperine-encapsulated nanosize liposomes. Eur. Biophys. J. 2016, 45, 175–186. [Google Scholar] [CrossRef]
- Lee, D.C.; Chapman, D. Infrared spectroscopic studies of biomembranes and model membranes. Biosci. Rep. 1986, 6, 235–256. [Google Scholar] [CrossRef]
- Binder, H. The molecular architecture of lipid membranes—New insights from hydration-tuning infrared linear dichroism spectroscopy. Appl. Spectrosc. Rev. 2003, 38, 15–69. [Google Scholar] [CrossRef]
- Mouritsen, O.G.; Andresen, T.L.; Halperin, A.; Hansen, P.L.; Jakobsen, A.F.; Jensen, U.B.; Jensen, M.; Jørgensen, K.; Kaasgaard, T.; Leidy, C.; et al. Activation of interfacial enzymes at membrane surfaces. J. Phys. Condens. Matter 2006, 18, 1293–1304. [Google Scholar] [CrossRef]
- Arouri, A.; Mouritsen, O.G. Phospholipase A 2-susceptible liposomes of anticancer double lipid-prodrugs. Eur. J. Pharm. Sci. 2012, 45, 408–420. [Google Scholar] [CrossRef]
- Włoch, A.; Stygar, D.; Bahri, F.; Bażanów, B.; Kuropka, P.; Chełmecka, E.; Pruchnik, H.; Gładkowski, W. Antiproliferative, antimicrobial and antiviral activity of β-aryl-δ-iodo-γ-lactones, their effect on cellular oxidative stress markers and biological membranes. Biomolecules 2020, 10, 1594. [Google Scholar] [CrossRef]
- Lakowicz, J.R. Principles of Fluorescence Spectroscopy, 3rd ed.; Lakowicz, J.R., Ed.; Springer: New York, NY, USA, 2006; ISBN 0387312781. [Google Scholar]
- Strugała, P.; Urbaniak, A.; Kuryś, P.; Włoch, A.; Kral, T.; Ugorski, M.; Hof, M.; Gabrielska, J. Antitumor and antioxidant activities of purple potato ethanolic extract and its interaction with liposomes, albumin and plasmid DNA. Food Funct. 2021, 12, 1271–1290. [Google Scholar] [CrossRef]

| ATR Spectra, Wavenumbers (cm−1) | |||
|---|---|---|---|
| Assigned * | DPPC | 1-ANISA-2-PA-PC/DPPC | 1-PA-2-ANISA-PC/DPPC |
| 25 °C | |||
| δ(CH2) | 1467.66 | 1467.53 | 1467.37 |
| γ(CH2) | 720.44 | 720.88 | 721.06 |
| νs(N-C) | 925.39 | 925.39 | 926.07 |
| νas(N-C)ip | 970.31 | 970.93 | 969.85 |
| νs(COP) | 1064.92 | 1066.32 | 1064.65 |
| νs(PO2−) | 1087.80 | 1088.40 | 1088.94 |
| νas(PO2−) | 1221.61 | 1221.47 | 1221.82 |
| ν(C=O) | 1733.94 | 1733.52 | 1733.74 |
| νs(CH2) | 2849.71 | 2849.69 | 2849.62 |
| νs(CH3) | 2872.91 | 2872.75 | 2872.83 |
| νas(CH2) | 2917.26 | 2917.13 | 2917.12 |
| νas(CH3) | 2955.57 | 2955.53 | 2955.53 |
| 38 °C | |||
| δ(CH2) | 1467.73 | 1467.63 | 1467.69 |
| γr(CH2) | 720.44 | 721.08 | 721.06 |
| νs(N-C) | 925.39 | 925.95 | 926.07 |
| νas(N-C)ip | 969.61 | 971.90 | 969.85 |
| νs(COP) | 1066.61 | 1068.58 | 1068.44 |
| νs(PO2−) | 1087.69 | 1087.49 | 1087.24 |
| νas(PO2−) | 1221.86 | 1225.89 | 1221.27 |
| ν(C=O) | 1736.55 | 1735.46 | 1733.50 |
| νs(CH2) | 2849.87 | 2851.71 | 2850.40 |
| νs(CH3) | 2872.73 | 2872.63 | 2872.68 |
| νas(CH2) | 2917.71 | 2920.99 | 2918.63 |
| νas(CH3) | 2956.57 | 2956.53 | 2956.31 |
| 50 °C | |||
| δ(CH2) | 1465.32 | 1464.78 | 1464.88 |
| γr(CH2) | ND | ND | ND |
| νs(N-C) | ND | ND | ND |
| νas N-C)ip | 967.77 | 971.81 | 969.85 |
| νs(COP) | 1068.22 | 1069.32 | 1068.22 |
| νs(PO2−) | 1087.02 | 1086.95 | 1086.22 |
| νas(PO2−) | 1220.47 | 1215.09 | 1215.48 |
| ν(C=O) | 1731.56 | ND | ND |
| νs(CH2) | 2852.74 | 2852.82 | 2852.70 |
| νs(CH3) | ND | ND | ND |
| νas(CH2) | 2922.06 | 2922.74 | 2922.53 |
| νas(CH3) | 2956.56 | 2956.32 | 2956.45 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pruchnik, H.; Gliszczyńska, A.; Włoch, A. Evaluation of the Physico-Chemical Properties of Liposomes Assembled from Bioconjugates of Anisic Acid with Phosphatidylcholine. Int. J. Mol. Sci. 2021, 22, 13146. https://doi.org/10.3390/ijms222313146
Pruchnik H, Gliszczyńska A, Włoch A. Evaluation of the Physico-Chemical Properties of Liposomes Assembled from Bioconjugates of Anisic Acid with Phosphatidylcholine. International Journal of Molecular Sciences. 2021; 22(23):13146. https://doi.org/10.3390/ijms222313146
Chicago/Turabian StylePruchnik, Hanna, Anna Gliszczyńska, and Aleksandra Włoch. 2021. "Evaluation of the Physico-Chemical Properties of Liposomes Assembled from Bioconjugates of Anisic Acid with Phosphatidylcholine" International Journal of Molecular Sciences 22, no. 23: 13146. https://doi.org/10.3390/ijms222313146
APA StylePruchnik, H., Gliszczyńska, A., & Włoch, A. (2021). Evaluation of the Physico-Chemical Properties of Liposomes Assembled from Bioconjugates of Anisic Acid with Phosphatidylcholine. International Journal of Molecular Sciences, 22(23), 13146. https://doi.org/10.3390/ijms222313146







