Secretome from In Vitro Mechanically Loaded Myoblasts Induces Tenocyte Migration, Transition to a Fibroblastic Phenotype and Suppression of Collagen Production
Abstract
1. Introduction
2. Results
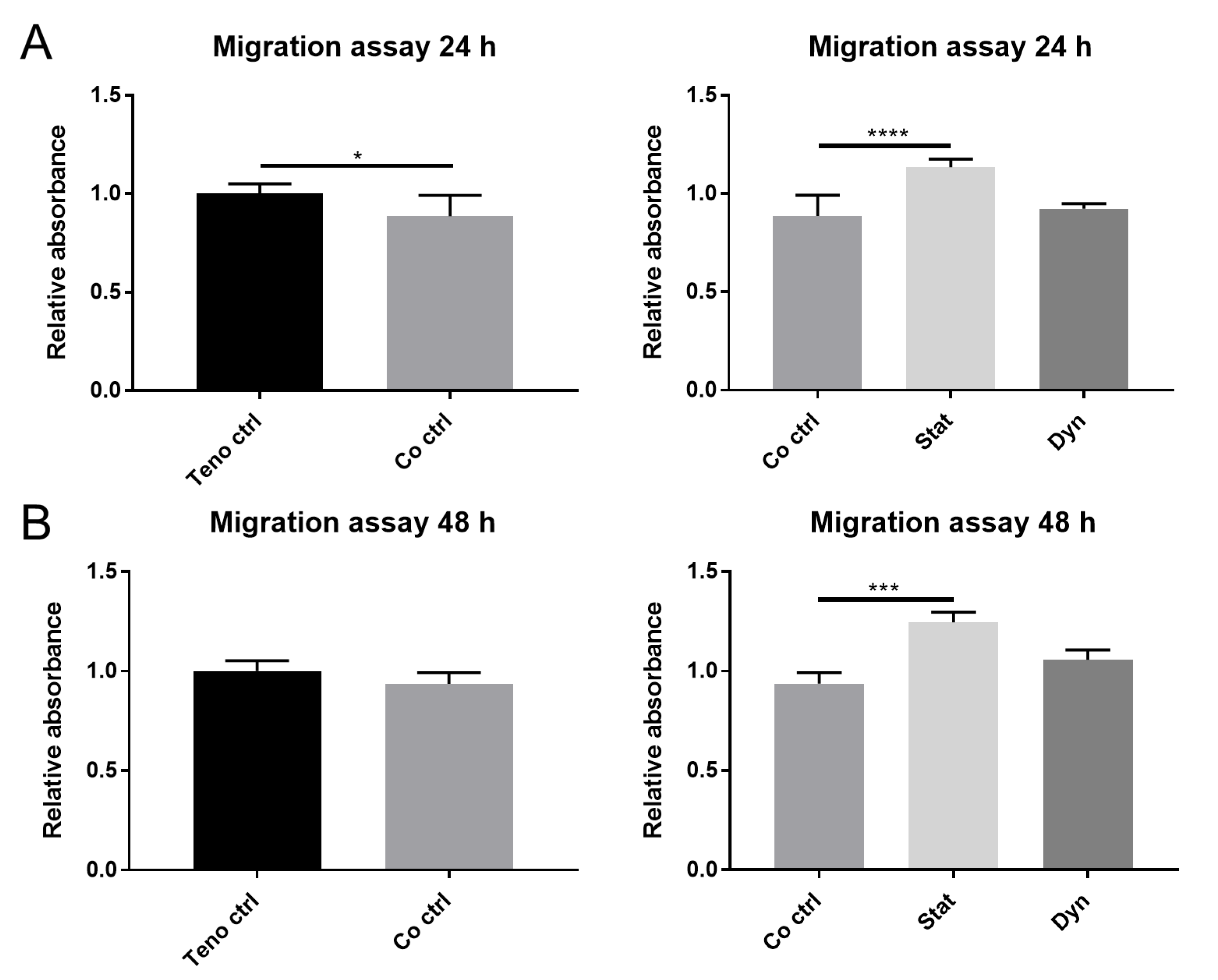
2.1. Secretome from Statically Loaded Myoblasts Enhances Tenocyte Migration
2.2. Mechanically Loaded Myoblasts Have No Effects on Tenocyte Proliferation and Apoptosis
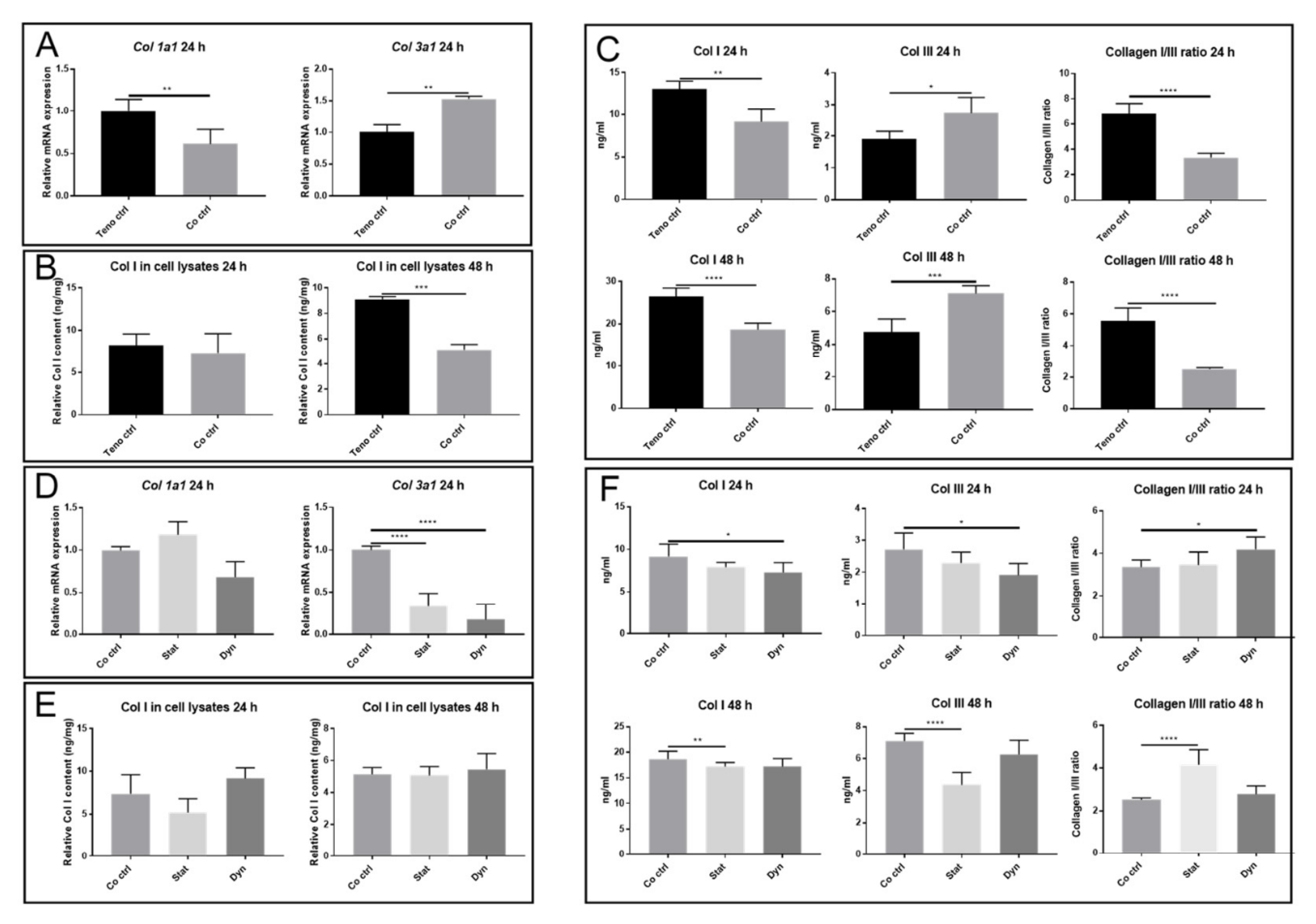
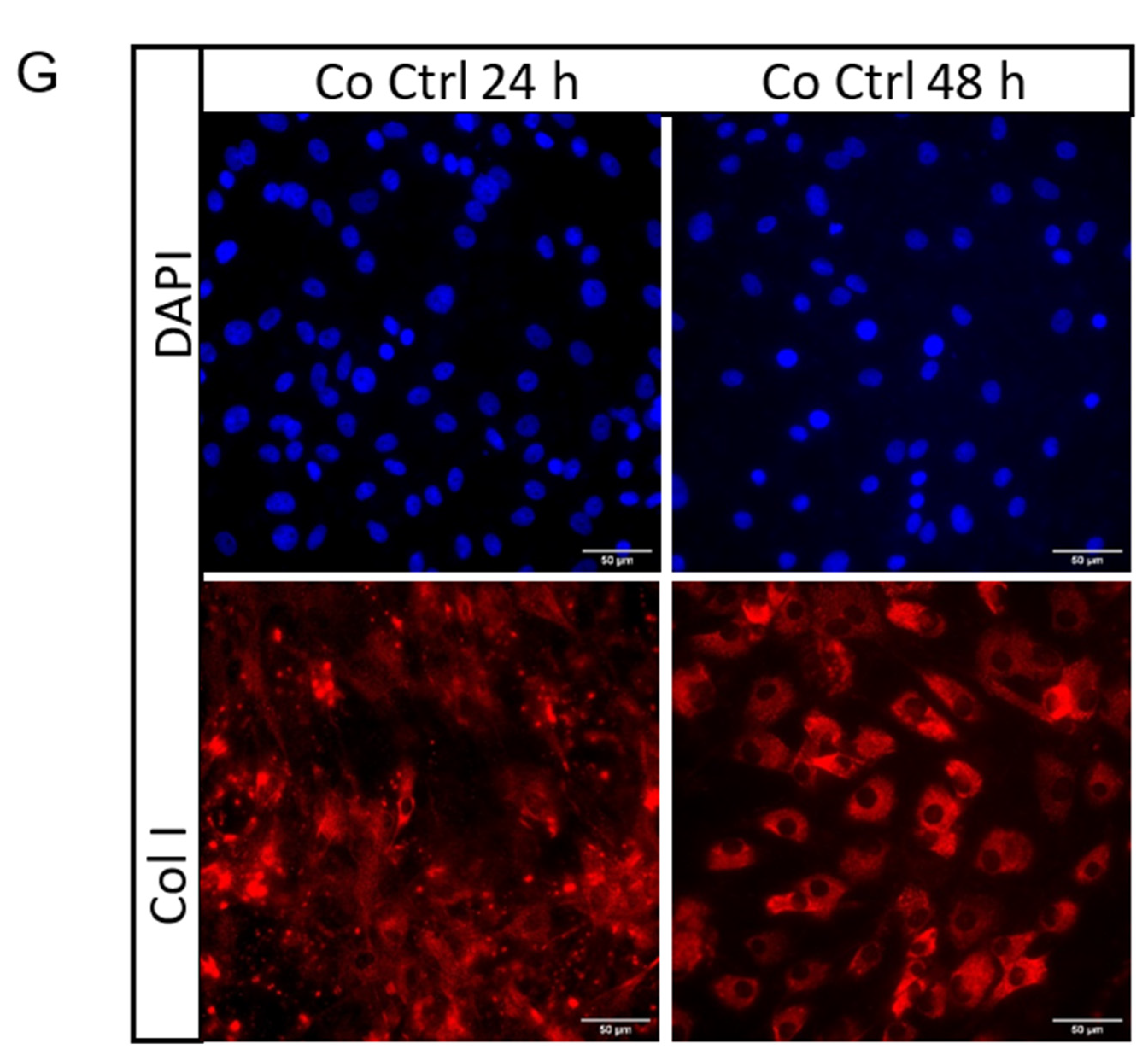
2.3. Secretome from Mechanically Loaded Myoblasts Alters Collagen Expression of Tenocytes
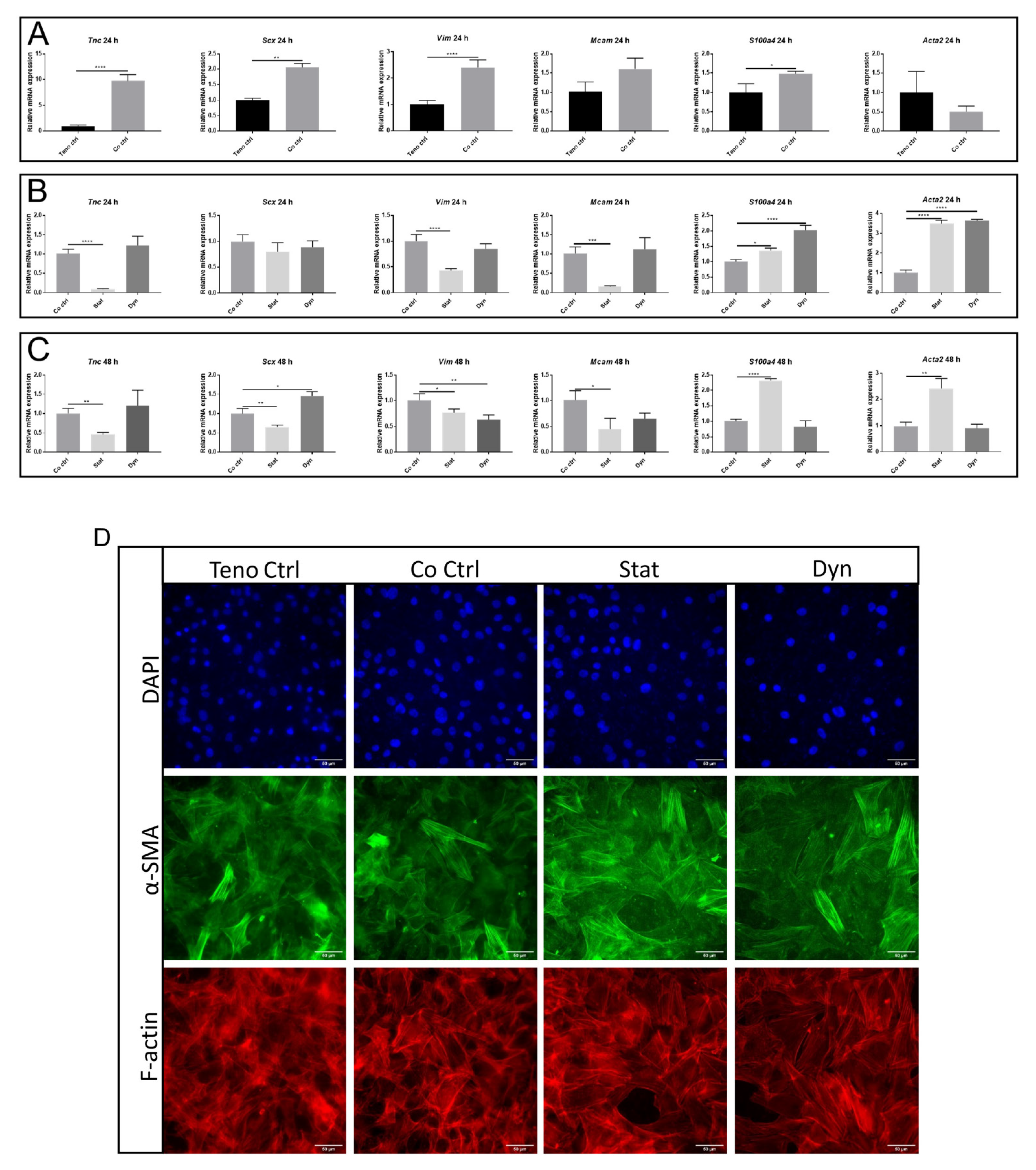
2.4. Statically Loaded Myoblasts Change the Expression of Tenocyte Markers
3. Discussion
4. Materials and Methods
4.1. Isolation and Culture of Primary Cells
4.2. Mechanical Strain
4.3. Indirect Co-Culture
4.4. Migration Assays
4.5. Cell proliferative Assay
4.6. Lactate Dehydrogenase Activity Assay
4.7. RNA Extraction and qRT-PCR
4.8. Western Blot
4.9. ELISA
4.10. Immunocytochemistry
4.11. Statistics
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Andersson, T.; Eliasson, P.; Aspenberg, P. Tissue memory in healing tendons: Short loading episodes stimulate healing. J. Appl. Physiol. 2009, 107, 417–421. [Google Scholar] [CrossRef]
- Eliasson, P.; Andersson, T.; Aspenberg, P. Achilles tendon healing in rats is improved by intermittent mechanical loading during the inflammatory phase. J. Orthopaed. Res. 2012, 30, 274–279. [Google Scholar] [CrossRef]
- Eliasson, P.; Andersson, T.; Aspenberg, P. Rat Achilles tendon healing: Mechanical loading and gene expression. J. Appl. Physiol. 2009, 107, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Nourissat, G.; Berenbaum, F.; Duprez, D. Tendon injury: From biology to tendon repair. Nat. Rev. Rheumatol. 2015, 11, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.H. Mechanobiology of tendon. J. Biomech. 2006, 39, 1563–1582. [Google Scholar] [CrossRef]
- Killian, M.L.; Cavinatto, L.; Galatz, L.M.; Thomopoulos, S. The role of mechanobiology in tendon healing. J. Shoulder Elbow Surg. 2012, 21, 228–237. [Google Scholar] [CrossRef]
- Arnoczky, S.P.; Tian, T.; Lavagnino, M.; Gardner, K. Ex vivo static tensile loading inhibits MMP-1 expression in rat tail tendon cells through a cytoskeletally based mechanotransduction mechanism. J. Orthop. Res. 2004, 22, 328–333. [Google Scholar] [CrossRef]
- Wall, M.E.; Dyment, N.A.; Bodle, J.; Volmer, J.; Loboa, E.; Cederlund, A.; Fox, A.M.; Banes, A.J. Cell Signaling in Tenocytes: Response to Load and Ligands in Health and Disease. Adv. Exp. Med. Biol. 2016, 920, 79–95. [Google Scholar]
- Hammerman, M.; Dietrich-Zagonel, F.; Blomgran, P.; Eliasson, P.; Aspenberg, P. Different mechanisms activated by mild versus strong loading in rat Achilles tendon healing. PLoS ONE 2018, 13, e0201211. [Google Scholar] [CrossRef]
- Dyment, N.A.; Liu, C.F.; Kazemi, N.; Aschbacher-Smith, L.E.; Kenter, K.; Breidenbach, A.P.; Shearn, J.T.; Wylie, C.; Rowe, D.W.; Butler, D.L. The Paratenon Contributes to Scleraxis-Expressing Cells during Patellar Tendon Healing. PLoS ONE 2013, 8, e59944. [Google Scholar] [CrossRef] [PubMed]
- Cariati, I.; Bonanni, R.; Onorato, F.; Mastrogregori, A.; Rossi, D.; Iundusi, R.; Gasbarra, E.; Tancredi, V.; Tarantino, U. Role of Physical Activity in Bone-Muscle Crosstalk: Biological Aspects and Clinical Implications. J. Funct. Morphol. Kinesiol. 2021, 6, 55. [Google Scholar] [CrossRef]
- Bosco, F.; Musolino, V.; Gliozzi, M.; Nucera, S.; Carresi, C.; Zito, M.C.; Scarano, F.; Scicchitano, M.; Reale, F.; Ruga, S.; et al. The muscle to bone axis (and viceversa): An encrypted language affecting tissues and organs and yet to be codified? Pharmacol. Res. 2021, 165, 105427. [Google Scholar] [CrossRef] [PubMed]
- Maurel, D.B.; Jahn, K.; Lara-Castillo, N. Muscle-Bone Crosstalk: Emerging Opportunities for Novel Therapeutic Approaches to Treat Musculoskeletal Pathologies. Biomedicines 2017, 5, 62. [Google Scholar] [CrossRef]
- Norheim, F.; Raastad, T.; Thiede, B.; Rustan, A.C.; Drevon, C.A.; Haugen, F. Proteomic identification of secreted proteins from human skeletal muscle cells and expression in response to strength training. Am. J. Physiol. Endocrinol. Metab. 2011, 301, E1013–E1021. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, A.S.; Cox, J.; Jensen, L.J.; Meissner, F.; Mann, M. Secretome Analysis of Lipid-Induced Insulin Resistance in Skeletal Muscle Cells by a Combined Experimental and Bioinformatics Workflow. J. Proteome Res. 2015, 14, 4885–4895. [Google Scholar] [CrossRef] [PubMed]
- Henningsen, J.; Rigbolt, K.T.; Blagoev, B.; Pedersen, B.K.; Kratchmarova, I. Dynamics of the skeletal muscle secretome during myoblast differentiation. Mol. Cell. Proteom. 2010, 9, 2482–2496. [Google Scholar] [CrossRef]
- Teixeira, F.G.; Panchalingam, K.M.; Assuncao-Silva, R.; Serra, S.C.; Mendes-Pinheiro, B.; Patricio, P.; Jung, S.; Anjo, S.I.; Manadas, B.; Pinto, L.; et al. Modulation of the Mesenchymal Stem Cell Secretome Using Computer-Controlled Bioreactors: Impact on Neuronal Cell Proliferation, Survival and Differentiation. Sci. Rep. 2016, 6, 1–14. [Google Scholar] [CrossRef]
- Ledet, M.M.; Vasquez, A.K.; Rauner, G.; Bichoupan, A.A.; Moroni, P.; Nydam, D.V.; Van de Walle, G.R. The secretome from bovine mammosphere-derived cells (MDC) promotes angiogenesis, epithelial cell migration, and contains factors associated with defense and immunity. Sci. Rep. 2018, 8, 5378. [Google Scholar] [CrossRef]
- Cho, J.W.; Kang, M.C.; Lee, K.S. TGF-beta 1-treated ADSCs-CM promotes expression of type I collagen and MMP-1, migration of human skin fibroblasts, and wound healing in vitro and in vivo. Int. J. Mol. Med. 2010, 26, 901–906. [Google Scholar]
- Infante, A.; Rodriguez, C.I. Secretome analysis of in vitro aged human mesenchymal stem cells reveals IGFBP7 as a putative factor for promoting osteogenesis. Sci. Rep. 2018, 8, 4632. [Google Scholar] [CrossRef]
- Ghebes, C.A.; Groen, N.; Cheuk, Y.C.; Fu, S.C.; Fernandes, H.M.; Saris, D.B.F. Muscle-Secreted Factors Improve Anterior Cruciate Ligament Graft Healing: An In Vitro and In Vivo Analysis. Tissue Eng. Part A 2018, 24, 322–334. [Google Scholar] [CrossRef] [PubMed]
- Harry, L.E.; Sandison, A.; Paleolog, E.M.; Hansen, U.; Pearse, M.F.; Nanchahal, J. Comparison of the healing of open tibial fractures covered with either muscle or fasciocutaneous tissue in a murine model. J. Orthop. Res. 2008, 26, 1238–1244. [Google Scholar] [CrossRef] [PubMed]
- Clarke, M.S.; Feeback, D.L. Mechanical load induces sarcoplasmic wounding and FGF release in differentiated human skeletal muscle cultures. FASEB J. 1996, 10, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Najafbeygi, A.; Fatemi, M.J.; Lebaschi, A.H.; Mousavi, S.J.; Husseini, S.A.; Niazi, M. Effect of Basic Fibroblast Growth Factor on Achilles Tendon Healing in Rabbit. World J. Plast. Surg. 2017, 6, 26–32. [Google Scholar] [PubMed]
- Grigg, N.L.; Wearing, S.C.; Smeathers, J.E. Eccentric calf muscle exercise produces a greater acute reduction in Achilles tendon thickness than concentric exercise. Br. J. Sport Med. 2009, 43, 280–283. [Google Scholar] [CrossRef] [PubMed]
- Kapilevich, L.V.; Zakharova, A.N.; Kabachkova, A.V.; Kironenko, T.A.; Orlov, S.N. Dynamic and Static Exercises Differentially Affect Plasma Cytokine Content in Elite Endurance- and Strength-Trained Athletes and Untrained Volunteers. Front. Physiol. 2017, 8, 35. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.L.; Wang, C.H.; Pan, C.Y.; Chen, F.C. The effects of long-term resistance exercise on the relationship between neurocognitive performance and GH, IGF-1, and hornocysteine levels in the elderly. Front. Behav. Neurosci. 2015, 9, 23. [Google Scholar] [CrossRef]
- Iwanuma, O.; Abe, S.; Hiroki, E.; Kado, S.; Sakiyama, K.; Usami, A.; Ide, Y. Effects of mechanical stretching on caspase and IGF-1 expression during the proliferation process of myoblasts. Zool. Sci. 2008, 25, 242–247. [Google Scholar] [CrossRef][Green Version]
- Perrone, C.E.; Fenwick-Smith, D.; Vandenburgh, H.H. Collagen and stretch modulate autocrine secretion of insulin-like growth factor-1 and insulin-like growth factor binding proteins from differentiated skeletal muscle cells. J. Biol. Chem. 1995, 270, 2099–2106. [Google Scholar] [CrossRef]
- Disser, N.P.; Sugg, K.B.; Talarek, J.R.; Sarver, D.C.; Rourke, B.J.; Mendias, C.L. Insulin-like growth factor 1 signaling in tenocytes is required for adult tendon growth. FASEB J. 2019, 33, 12680–12695. [Google Scholar] [CrossRef]
- Snedeker, J.G.; Foolen, J. Tendon injury and repair—A perspective on the basic mechanisms of tendon disease and future clinical therapy. Acta Biomater. 2017, 63, 18–36. [Google Scholar] [CrossRef]
- Thomopoulos, S.; Parks, W.C.; Rifkin, D.B.; Derwin, K.A. Mechanisms of tendon injury and repair. J. Orthop. Res. 2015, 33, 832–839. [Google Scholar] [CrossRef]
- Woo, S.L.; Hildebrand, K.; Watanabe, N.; Fenwick, J.A.; Papageorgiou, C.D.; Wang, J.H. Tissue engineering of ligament and tendon healing. Clin. Orthop. Relat. Res. 1999, 367, S312–S323. [Google Scholar] [CrossRef]
- Chen, Q.; Liang, Q.; Zhuang, W.; Zhou, J.; Zhang, B.; Xu, P.; Ju, Y.; Morita, Y.; Luo, Q.; Song, G. Tenocyte proliferation and migration promoted by rat bone marrow mesenchymal stem cell-derived conditioned medium. Biotechnol. Lett. 2018, 40, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.Y.; McDermott, J.C.; Siu, K.W. Secretome Analysis of Skeletal Myogenesis Using SILAC and Shotgun Proteomics. Int. J. Proteom. 2011, 2011, 329467. [Google Scholar] [CrossRef]
- Yang, G.; Rothrauff, B.B.; Tuan, R.S. Tendon and Ligament Regeneration and Repair: Clinical Relevance and Developmental Paradigm. Birth Defects Res. C 2013, 99, 203–222. [Google Scholar] [CrossRef] [PubMed]
- Huisman, E.; Lu, A.; McCormack, R.G.; Scott, A. Enhanced collagen type I synthesis by human tenocytes subjected to periodic in vitro mechanical stimulation. BMC Musculoskelet. Disord. 2014, 15, 386. [Google Scholar] [CrossRef] [PubMed]
- Gumucio, J.P.; Sugg, K.B.; Mendias, C.L. TGF-beta Superfamily Signaling in Muscle and Tendon Adaptation to Resistance Exercise. Exerc. Sport Sci. Rev. 2015, 43, 93–99. [Google Scholar] [CrossRef]
- Lee, C.H.; Lee, F.Y.; Tarafder, S.; Kao, K.; Jun, Y.N.; Yang, G.D.; Mao, J.J. Harnessing endogenous stem/progenitor cells for tendon regeneration. J. Clin. Investig. 2015, 125, 2690–2701. [Google Scholar] [CrossRef]
- Jo, C.H.; Lim, H.J.; Yoon, K.S. Characterization of Tendon-Specific Markers in Various Human Tissues, Tenocytes and Mesenchymal Stem Cells. Tissue Eng. Regen. Med. 2019, 16, 151–159. [Google Scholar] [CrossRef]
- Best, K.T.; Loiselle, A.E. Scleraxis lineage cells contribute to organized bridging tissue during tendon healing and identify a subpopulation of resident tendon cells. FASEB J. 2019, 33, 8578–8587. [Google Scholar] [CrossRef]
- Ackerman, J.E.; Nichols, A.E.C.; Studentsova, V.; Best, K.T.; Knapp, E.; Loiselle, A.E. Cell non-autonomous functions of S100a4 drive fibrotic tendon healing. Elife 2019, 8, e45342. [Google Scholar] [CrossRef]
- Premdas, J.; Tang, J.B.; Warner, J.P.; Murray, M.M.; Spector, M. The presence of smooth muscle actin in fibroblasts in the torn human rotator cuff. J. Orthop. Res. 2001, 19, 221–228. [Google Scholar] [CrossRef]
- Cadby, J.A.; Buehler, E.; Godbout, C.; van Weeren, P.R.; Snedeker, J.G. Differences between the Cell Populations from the Peritenon and the Tendon Core with Regard to Their Potential Implication in Tendon Repair. PLoS ONE 2014, 9, e92474. [Google Scholar] [CrossRef]
- Lee, C.; Jun, Y.; Kao, K. Harnessing Endogenous Stem/Progenitor Cells for Tendon Regeneration. Tissue Eng. Part A 2015, 21, S72. [Google Scholar] [CrossRef]
- Blomgran, P.; Blomgran, R.; Ernerudh, J.; Aspenberg, P. A possible link between loading, inflammation and healing: Immune cell populations during tendon healing in the rat. Sci. Rep. 2016, 6, 29824. [Google Scholar] [CrossRef]
- Hammerman, M.; Aspenberg, P.; Eliasson, P. Microtrauma stimulates rat Achilles tendon healing via an early gene expression pattern similar to mechanical loading. J. Appl. Physiol. 2014, 116, 54–60. [Google Scholar] [CrossRef]
- Baccam, A.; Benoni-Sviercovich, A.; Rocchi, M.; Moresi, V.; Seelaender, M.; Li, Z.; Adamo, S.; Xue, Z.; Coletti, D. The Mechanical Stimulation of Myotubes Counteracts the Effects of Tumor-Derived Factors Through the Modulation of the Activin/Follistatin Ratio. Front. Physiol. 2019, 10, 401. [Google Scholar] [CrossRef]
- Moustogiannis, A.; Philippou, A.; Zevolis, E.; Taso, O.; Chatzigeorgiou, A.; Koutsilieris, M. Characterization of Optimal Strain, Frequency and Duration of Mechanical Loading on Skeletal Myotubes’ Biological Responses. In Vivo 2020, 34, 1779–1788. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.L.; Kuang, W.; Jin, Z.L.; Jin, F.; Xu, L.; Yu, Q.J.; Kong, L.; Zeng, G.; Yuan, X.; Duan, Y.Z. Inhibition of NFkappaB by activated c-Jun NH2 terminal kinase 1 acts as a switch for C2C12 cell death under excessive stretch. Apoptosis 2009, 14, 764–770. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Huang, X.; Tian, Y.; Yan, X.; Wang, F.; Chen, J.; Zhang, Q.; Zhang, Q.; Yuan, X. Phosphorylated GSK3beta protects stressinduced apoptosis of myoblasts via the PI3K/Akt signaling pathway. Mol. Med. Rep. 2020, 22, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Park, S.R.; Kim, J.W.; Jun, H.S.; Roh, J.Y.; Lee, H.Y.; Hong, I.S. Stem Cell Secretome and Its Effect on Cellular Mechanisms Relevant to Wound Healing. Mol. Ther. 2018, 26, 606–617. [Google Scholar] [CrossRef] [PubMed]
- Kamprom, W.; Kheolamai, P.; Supokawej, A.; Wattanapanitch, M.; Laowtammathron, C.; Roytrakul, S.; Issaragrisil, S. Endothelial Progenitor Cell Migration-Enhancing Factors in the Secretome of Placental-Derived Mesenchymal Stem Cells. Stem Cells Int. 2016, 2016, 2514326. [Google Scholar] [CrossRef] [PubMed]
- De Donatis, A.; Ranaldi, F.; Cirri, P. Reciprocal control of cell proliferation and migration. Cell Commun. Signal. 2010, 8, 20. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Kim, S.Y.; Kwon, C.H.; Kim, Y.K. Differential effect of FGF and PDGF on cell proliferation and migration in osteoblastic cells. Growth Factors 2007, 25, 77–86. [Google Scholar] [CrossRef]
- De Donatis, A.; Comito, G.; Buricchi, F.; Vinci, M.C.; Parenti, A.; Caselli, A.; Camici, G.; Manao, G.; Ramponi, G.; Cirri, P. Proliferation versus migration in platelet-derived growth factor signaling: The key role of endocytosis. J. Biol. Chem. 2008, 283, 19948–19956. [Google Scholar] [CrossRef]
- Toti, A.; Santi, A.; Pardella, E.; Nesi, I.; Tomasini, R.; Mello, T.; Paoli, P.; Caselli, A.; Cirri, P. Activated fibroblasts enhance cancer cell migration by microvesicles-mediated transfer of Galectin-1. J. Cell. Commun. Signal. 2021, 15, 405–419. [Google Scholar] [CrossRef]
- Smith, R.K.; Birch, H.; Patterson-Kane, J.; Firth, E.C.; Williams, L.; Cherdchutham, W.; van Weeren, W.R.; Goodship, A.E. Should equine athletes commence training during skeletal development?: Changes in tendon matrix associated with development, ageing, function and exercise. Equine Vet. J. Suppl. 1999, 30, 201–209. [Google Scholar] [CrossRef]
- Goncalves-Neto, J.; Witzel, S.S.; Teodoro, W.R.; Carvalho-Junior, A.E.; Fernandes, T.D.; Yoshinari, H.H. Changes in collagen matrix composition in human posterior tibial tendon dysfunction. Joint Bone Spine 2002, 69, 189–194. [Google Scholar] [CrossRef]
- Hartwig, S.; Raschke, S.; Knebel, B.; Scheler, M.; Irmler, M.; Passlack, W.; Muller, S.; Hanisch, F.G.; Franz, T.; Li, X.; et al. Secretome profiling of primary human skeletal muscle cells. Biochim. Biophys. Acta 2014, 1844, 1011–1017. [Google Scholar] [CrossRef]
- Le Bihan, M.C.; Bigot, A.; Jensen, S.S.; Dennis, J.L.; Rogowska-Wrzesinska, A.; Laine, J.; Gache, V.; Furling, D.; Jensen, O.N.; Voit, T.; et al. In-depth analysis of the secretome identifies three major independent secretory pathways in differentiating human myoblasts. J. Proteom. 2012, 77, 344–356. [Google Scholar] [CrossRef] [PubMed]
- Henningsen, J.; Pedersen, B.K.; Kratchmarova, I. Quantitative analysis of the secretion of the MCP family of chemokines by muscle cells. Mol. Biosyst. 2011, 7, 311–321. [Google Scholar] [CrossRef]
- Graham, M.F.; Willey, A.; Adams, J.; Yager, D.; Diegelmann, R.F. Interleukin 1 beta down-regulates collagen and augments collagenase expression in human intestinal smooth muscle cells. Gastroenterology 1996, 110, 344–350. [Google Scholar] [CrossRef]
- Huang, M.; Sharma, S.; Zhu, L.X.; Keane, M.P.; Luo, J.; Zhang, L.; Burdick, M.D.; Lin, Y.Q.; Dohadwala, M.; Gardner, B.; et al. IL-7 inhibits fibroblast TGF-beta production and signaling in pulmonary fibrosis. J. Clin. Investig. 2002, 109, 931–937. [Google Scholar] [CrossRef]
- Reitamo, S.; Remitz, A.; Tamai, K.; Uitto, J. Interleukin-10 modulates type I collagen and matrix metalloprotease gene expression in cultured human skin fibroblasts. J. Clin. Investig. 1994, 94, 2489–2492. [Google Scholar] [CrossRef]
- Dufour, A.M.; Alvarez, M.; Russo, B.; Chizzolini, C. Interleukin-6 and Type-I Collagen Production by Systemic Sclerosis Fibroblasts Are Differentially Regulated by Interleukin-17A in the Presence of Transforming Growth Factor-Beta 1. Front. Immunol. 2018, 9, 1865. [Google Scholar] [CrossRef] [PubMed]
- Aoudjehane, L.; Pissaia, A., Jr.; Scatton, O.; Podevin, P.; Massault, P.P.; Chouzenoux, S.; Soubrane, O.; Calmus, Y.; Conti, F. Interleukin-4 induces the activation and collagen production of cultured human intrahepatic fibroblasts via the STAT-6 pathway. Lab. Investig. 2008, 88, 973–985. [Google Scholar] [CrossRef]
- Kim, Y.M.; Park, S.K.; Xu, J.; Yeon, S.H. Role of IL-25 in Extracellular Matrix and Collagen Production in Nasal Fibroblast. J. Allergy Clin. Immun. 2017, 139, Ab65. [Google Scholar] [CrossRef]
- Franze, E.; Dinallo, V.; Laudisi, F.; Colantoni, A.; Ortenzi, A.; Giuffrida, P.; Di Carlo, S.; Sica, G.; Di Sabatino, A.; Monteleone, G. Interleukin-34 Stimulates Gut Fibroblasts to Produce Collagen Synthesis. Digest. Liver Dis. 2020, 52, S35. [Google Scholar] [CrossRef]
- Dong, K.; Markova, N.; Smiles, K.; Yarosh, D. TNF-alpha and IL-6 regulate collagen 1 and MMP-1 in dermal fibroblasts. Bicyclic monoterpene diols suppress MMP-1 secretion and increase collagen production through TNF-alpha signaling. J. Am. Acad. Dermatol. 2008, 58, Ab64. [Google Scholar]
- Best, K.T.; Lee, F.K.; Knapp, E.; Awad, H.A.; Loiselle, A.E. Deletion of NFKB1 enhances canonical NF-kappaB signaling and increases macrophage and myofibroblast content during tendon healing. Sci. Rep. 2019, 9, 10926. [Google Scholar] [CrossRef]
- Millar, N.L.; Murrell, G.A.C.; McInnes, I.B. Inflammatory mechanisms in tendinopathy—Towards translation. Nat. Rev. Rheumatol. 2017, 13, 110–122. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.T.; Li, J.; Wang, J.H.; Wu, Q.; Yang, P.; Hsu, H.C.; Smythies, L.E.; Mountz, J.D. IL-17 Activates the Canonical NF-kappa B Signaling Pathway in Autoimmune B Cells of BXD2 Mice To Upregulate the Expression of Regulators of G-Protein Signaling 16. J. Immunol. 2010, 184, 2289–2296. [Google Scholar] [CrossRef]
- Li, J.; Lau, G.K.K.; Chen, L.L.; Dong, S.S.; Lan, H.Y.; Huang, X.R.; Li, Y.; Luk, J.M.; Yuan, Y.F.; Guan, X.Y. Interleukin 17A Promotes Hepatocellular Carcinoma Metastasis via NF-kB Induced Matrix Metalloproteinases 2 and 9 Expression. PLoS ONE 2011, 6, e21816. [Google Scholar] [CrossRef]
- Best, K.T.; Nichols, A.E.C.; Knapp, E.; Hammert, W.C.; Ketonis, C.; Jonason, J.H.; Awad, H.A.; Loiselle, A.E. NF-kappa B activation persists into the remodeling phase of tendon healing and promotes myofibroblast survival. Sci. Signal. 2020, 13, eabb7209. [Google Scholar] [CrossRef] [PubMed]
- Ryan, D.G.; Taliana, L.; Sun, L.J.; Wei, Z.G.; Masur, S.K.; Lavker, R.M. Involvement of S100A4 in stromal fibroblasts of the regenerating cornea. Investig. Ophthalmol. Vis. Sci. 2003, 44, 4255–4262. [Google Scholar] [CrossRef][Green Version]
- Droguett, R.; Cabello-Verrugio, C.; Riquelme, C.; Brandan, E. Extracellular proteoglycans modify TGF-beta bio-availability attenuating its signaling during skeletal muscle differentiation. Matrix Biol. 2006, 25, 332–341. [Google Scholar] [CrossRef] [PubMed]
- El-Akkawi, A.I.; Joanroy, R.; Barfod, K.W.; Kallemose, T.; Kristensen, S.S.; Viberg, B. Effect of Early Versus Late Weightbearing in Conservatively Treated Acute Achilles Tendon Rupture: A Meta-Analysis. J. Foot Ankle Surg. 2018, 57, 346–352. [Google Scholar] [CrossRef]
- Huang, J.Z.; Wang, C.; Ma, X.; Wang, X.; Zhang, C.; Chen, L. Rehabilitation Regimen After Surgical Treatment of Acute Achilles Tendon Ruptures A Systematic Review With Meta-analysis. Am. J. Sport Med. 2015, 43, 1008–1016. [Google Scholar] [CrossRef]
- Valkering, K.P.; Aufwerber, S.; Ranuccio, F.; Lunini, E.; Edman, G.; Ackermann, P.W. Functional weight-bearing mobilization after Achilles tendon rupture enhances early healing response: A single-blinded randomized controlled trial. Knee Surg. Sport Tr A 2017, 25, 1807–1816. [Google Scholar] [CrossRef]
- Niehoff, A.; Kersting, U.G.; Helling, S.; Dargel, J.; Maurer, J.; Thevis, M.; Bruggemann, G.P. Different mechanical loading protocols influence serum cartilage oligomeric matrix protein levels in young healthy humans. Eur. J. Appl. Physiol. 2010, 110, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Luc-Harkey, B.A.; Franz, J.R.; Hackney, A.C.; Blackburn, J.T.; Padua, D.A.; Pietrosimone, B. Lesser lower extremity mechanical loading associates with a greater increase in serum cartilage oligomeric matrix protein following walking in individuals with anterior cruciate ligament reconstruction. Clin. Biomech. 2018, 60, 13–19. [Google Scholar] [CrossRef] [PubMed]
- El-Habta, R.; Andersson, G.; Kingham, P.J.; Backman, L.J. Anti-apoptotic effect of adipose tissue-derived stromal vascular fraction in denervated rat muscle. Stem Cell Res. Ther. 2021, 12, 162. [Google Scholar] [CrossRef] [PubMed]

| Gene Symbol | Gene Name | Assay ID |
|---|---|---|
| Mcam | melanoma cell adhesion molecule, CD 146 | Rn00576900_m1 |
| Vim | Vimentin | Rn00579738 |
| Tnc | Tenascin-C | Rn01454948 |
| Acta2 | actin alpha 2, smooth muscle, α-SMA | Rn01759928 |
| S100a4 | S100 calcium-binding protein A4 | Rn01451938 |
| Scx | Scleraxis bHLH transcription factor | Rn01504576 |
| Col1a1 | Collagen type I alpha 1 chain | Rn01463848_m1 |
| Col3a1 | collagen type III alpha 1 chain | Rn01437681_m1 |
| Rpl13a (reference gene) | Ribosomal Protein L13a | Rn00821946_g1 |
| Antibody | Company | Code | Dilution | Species | Molecular Weight (kDa) |
|---|---|---|---|---|---|
| PCNA | Cell Signaling | 2586 | 1:1000 | Mouse | 36 |
| c-PARP | Cell Signaling | 9541 | 1:1000 | Rabbit | 89 |
| β-actin | Cell Signaling | 4967 | 1:2000 | Rabbit | 45 |
| Anti-rabbit IgG, HRP-linked antibody | Cell Signaling | 7074 | 1:2000 | Goat |
| Antibody | Company | Code | Dilution | Species |
|---|---|---|---|---|
| Collagen I | Abcam | 260043 | 1:400 | Rabbit |
| α-SMA | Thermo Fisher | 53976082 | 1:400 | Rabbit |
| Fluorescein-conjugated secondary antibody Goat-anti rabbit | Thermo Fisher | A32740 | 1:400 | Rabbit |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, X.; Li, J.; Giannopoulos, A.; Kingham, P.J.; Backman, L.J. Secretome from In Vitro Mechanically Loaded Myoblasts Induces Tenocyte Migration, Transition to a Fibroblastic Phenotype and Suppression of Collagen Production. Int. J. Mol. Sci. 2021, 22, 13089. https://doi.org/10.3390/ijms222313089
Zhou X, Li J, Giannopoulos A, Kingham PJ, Backman LJ. Secretome from In Vitro Mechanically Loaded Myoblasts Induces Tenocyte Migration, Transition to a Fibroblastic Phenotype and Suppression of Collagen Production. International Journal of Molecular Sciences. 2021; 22(23):13089. https://doi.org/10.3390/ijms222313089
Chicago/Turabian StyleZhou, Xin, Junhong Li, Antonios Giannopoulos, Paul J. Kingham, and Ludvig J. Backman. 2021. "Secretome from In Vitro Mechanically Loaded Myoblasts Induces Tenocyte Migration, Transition to a Fibroblastic Phenotype and Suppression of Collagen Production" International Journal of Molecular Sciences 22, no. 23: 13089. https://doi.org/10.3390/ijms222313089
APA StyleZhou, X., Li, J., Giannopoulos, A., Kingham, P. J., & Backman, L. J. (2021). Secretome from In Vitro Mechanically Loaded Myoblasts Induces Tenocyte Migration, Transition to a Fibroblastic Phenotype and Suppression of Collagen Production. International Journal of Molecular Sciences, 22(23), 13089. https://doi.org/10.3390/ijms222313089






