Author Contributions
Conceptualization, S.S. and S.D.; methodology, S.S., V.D., V.G. and S.D.; software, S.S., V.D., V.G.and S.D.; validation, S.S., V.D., V.G. and S.D.; formal analysis, S.S., V.D., V.G. and S.D.; investigation, S.S., V.D., V.G. and S.D.; resources, S.S.and S.D.; data curation, S.S. and S.D.; writing—original draft preparation, S.S. and S.D.; writing—review and editing, S.S. and S.D.; visualization, S.S. and S.D.; supervision, S.S. and S.D.; project administration, S.S. and S.D.; funding acquisition, S.S. and S.D. All authors have read and agreed to the published version of the manuscript.
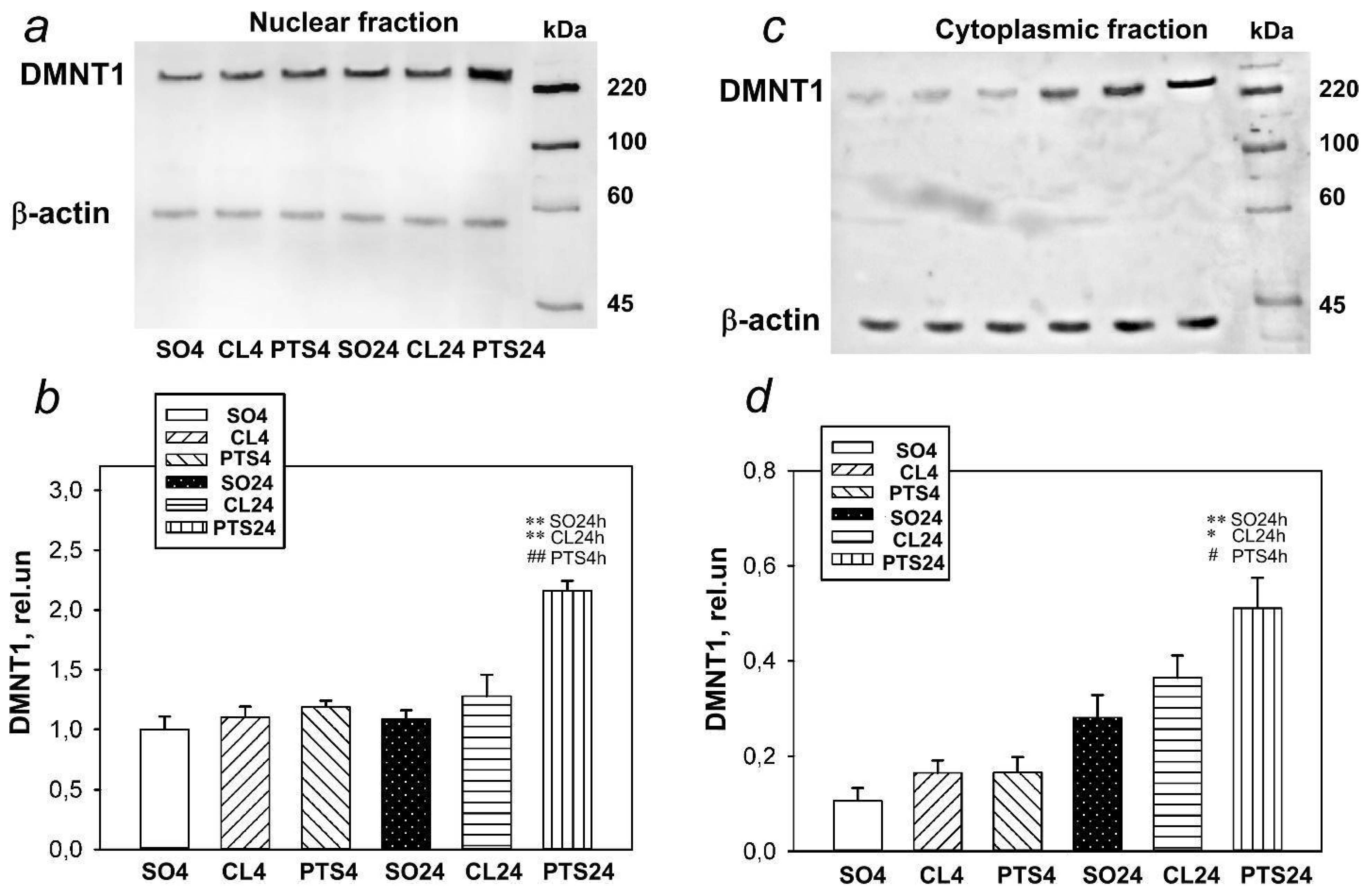
Figure 1.
The expression of DNMT1in nuclear (a,b) and cytoplasmic (c,d) fractions of penumbra tissue at 4 or 24 h after photothrombotic stroke (PTS) in the rat cerebral cortex. The significant differences * p < 0.05 and ** p < 0.01 compared to sham-operated animals (SO) or contralateral cerebral cortex of the same animals (CL4 and CL24), # p < 0.05 and ## p < 0.01 compared to the PTS4 are shown. One-way ANOVA. M ± SEM. n = 7. Rel.un—the ratio of the optical density of the strip of the protein studied to the optical density of the strip of the protein load marker (actin).
Figure 1.
The expression of DNMT1in nuclear (a,b) and cytoplasmic (c,d) fractions of penumbra tissue at 4 or 24 h after photothrombotic stroke (PTS) in the rat cerebral cortex. The significant differences * p < 0.05 and ** p < 0.01 compared to sham-operated animals (SO) or contralateral cerebral cortex of the same animals (CL4 and CL24), # p < 0.05 and ## p < 0.01 compared to the PTS4 are shown. One-way ANOVA. M ± SEM. n = 7. Rel.un—the ratio of the optical density of the strip of the protein studied to the optical density of the strip of the protein load marker (actin).
Figure 2.
The changes in DNMT1 localization and level in the neurons of ischemic penumbra at 4 and 24 h after photothrombotic stroke (PTS) in the rat cerebral cortex. The significant difference in localization and level of DNMT1 at 4 and 24 h after PTS (PTS4 and PTS24, respectively) is shown compared to the contralateral cortex of the same animals (CL4 and CL24), or the cerebral cortex of sham-operated rats (SO). (a) Immunofluorescence of DNMT1 (green), neuronal marker NeuN (red), nuclear chromatin marker Hoechst 33342 (blue), and image overlay. The scale bar is 50 μm. (b) Percent changes (ΔISO) of DNMT1 levels in the penumbra (PTS4 or PTS24) and CL4 or CL24 compared to SO at PTS4 or PTS24. (c) Percent changes (ΔICL) of DNMT1 levels in the penumbra at PTS4 or PTS24 compared to CL4 and CL24. (d) Coefficient M1 of DNMT1 co-localization with the neuronal marker (NeuN) in different control and experimental groups. (e) Immunofluorescence of DNMT1 in cell nuclei. (f) Percentage of cells expressing DNMT1 per 100 cells stained with Hoechst 33342 * p < 0.05 compared to SO, # p < 0.05 compared to CL4 and CL24 of the same animals. One Way ANOVA; M ± SEM; n = 7.
Figure 2.
The changes in DNMT1 localization and level in the neurons of ischemic penumbra at 4 and 24 h after photothrombotic stroke (PTS) in the rat cerebral cortex. The significant difference in localization and level of DNMT1 at 4 and 24 h after PTS (PTS4 and PTS24, respectively) is shown compared to the contralateral cortex of the same animals (CL4 and CL24), or the cerebral cortex of sham-operated rats (SO). (a) Immunofluorescence of DNMT1 (green), neuronal marker NeuN (red), nuclear chromatin marker Hoechst 33342 (blue), and image overlay. The scale bar is 50 μm. (b) Percent changes (ΔISO) of DNMT1 levels in the penumbra (PTS4 or PTS24) and CL4 or CL24 compared to SO at PTS4 or PTS24. (c) Percent changes (ΔICL) of DNMT1 levels in the penumbra at PTS4 or PTS24 compared to CL4 and CL24. (d) Coefficient M1 of DNMT1 co-localization with the neuronal marker (NeuN) in different control and experimental groups. (e) Immunofluorescence of DNMT1 in cell nuclei. (f) Percentage of cells expressing DNMT1 per 100 cells stained with Hoechst 33342 * p < 0.05 compared to SO, # p < 0.05 compared to CL4 and CL24 of the same animals. One Way ANOVA; M ± SEM; n = 7.
![Ijms 22 12483 g002 Ijms 22 12483 g002]()
Figure 3.
The changes of DNMT1 levels in the astrocytes of ischemic penumbra at 4 and 24 h after photothrombotic stroke (PTS) in the rat cerebral cortex. The levels of DNMT1 at 4 and 24 h after PTS are shown (PTS4 and PTS24, respectively) compared to the contralateral cortex of the same animals (CL4 and CL24), or the cortex of sham-operated rats (SO). (a) Immunofluorescence of DNMT1 (green), marker of astrocytes GFAP (red), nuclear chromatin marker Hoechst 33342 (blue), and image overlay. The scale bar is 50 μm. (b) Coefficient M1 of DNMT1 co-localization with astrocyte marker (GFAP) in different control and experimental groups. * p < 0.05 compared to SO, # p < 0.05 compared to CL4 and CL24. One Way ANOVA; M ± SEM; n = 7.
Figure 3.
The changes of DNMT1 levels in the astrocytes of ischemic penumbra at 4 and 24 h after photothrombotic stroke (PTS) in the rat cerebral cortex. The levels of DNMT1 at 4 and 24 h after PTS are shown (PTS4 and PTS24, respectively) compared to the contralateral cortex of the same animals (CL4 and CL24), or the cortex of sham-operated rats (SO). (a) Immunofluorescence of DNMT1 (green), marker of astrocytes GFAP (red), nuclear chromatin marker Hoechst 33342 (blue), and image overlay. The scale bar is 50 μm. (b) Coefficient M1 of DNMT1 co-localization with astrocyte marker (GFAP) in different control and experimental groups. * p < 0.05 compared to SO, # p < 0.05 compared to CL4 and CL24. One Way ANOVA; M ± SEM; n = 7.
Figure 4.
Suv39H1 expression in nuclear (a,b) and cytoplasmic (c) fractions of penumbra tissue at 4 and 24 h after photothrombotic stroke (PTS) in the rat cerebral cortex. The expression of Suv39H1 is shown at 4 and 24 h after PTS (PTS4 and PTS24, respectively) * p < 0.05 compared to sham-operated (SO) animals or contralateral cerebral cortex (CL4 and CL24) of the same animals, # p < 0.05 compared to PTS4. One-way ANOVA. M ± SEM. n = 7. Rel.un—the ratio of the optical density of the strip of the protein studied to the optical density of the strip of the protein load marker (actin).
Figure 4.
Suv39H1 expression in nuclear (a,b) and cytoplasmic (c) fractions of penumbra tissue at 4 and 24 h after photothrombotic stroke (PTS) in the rat cerebral cortex. The expression of Suv39H1 is shown at 4 and 24 h after PTS (PTS4 and PTS24, respectively) * p < 0.05 compared to sham-operated (SO) animals or contralateral cerebral cortex (CL4 and CL24) of the same animals, # p < 0.05 compared to PTS4. One-way ANOVA. M ± SEM. n = 7. Rel.un—the ratio of the optical density of the strip of the protein studied to the optical density of the strip of the protein load marker (actin).
Figure 5.
The changes in Suv39H1 localization and levels in the neurons of ischemic penumbra 4 and 24 h after photothrombotic stroke in the rat cerebral cortex. The localisation and level of Suv39H1 are shown at 4 and 24 h after PTS (PTS4 and PTS24, respectively) compared to the contralateral cortex of the same animals (CL4 and CL24), or the cerebral cortex of sham-operated rats (SO). (a) Immunofluorescence of Suv39H1 (green), neuronal marker NeuN (red), nuclear chromatin marker Hoechst 33342 (blue), and image overlay. The scale bar is 50 μm. (b). Percent changes (ΔISO) of Suv39H1 levels in the penumbra at PTS4 or PTS24 and CL4 or CL24 compared to SO. (c) Percent changes (ΔICL) of Suv39H1 levels in the penumbra at PTS4 or PTS24 compared to the CL4 or CL24. (d) Coefficient M1 of Suv39H1 co-localization with the neuronal marker (NeuN) in different control and experimental groups. (e) Immunofluorescence of Suv39H1 in cell nuclei. (f) Percentage of cells expressing Suv39H1 per 100 cells stained with Hoechst 33342 * p < 0.05 compared to SO, # p < 0.05 compared to theCL4 or CL24. One Way ANOVA; M ± SEM; n = 7.
Figure 5.
The changes in Suv39H1 localization and levels in the neurons of ischemic penumbra 4 and 24 h after photothrombotic stroke in the rat cerebral cortex. The localisation and level of Suv39H1 are shown at 4 and 24 h after PTS (PTS4 and PTS24, respectively) compared to the contralateral cortex of the same animals (CL4 and CL24), or the cerebral cortex of sham-operated rats (SO). (a) Immunofluorescence of Suv39H1 (green), neuronal marker NeuN (red), nuclear chromatin marker Hoechst 33342 (blue), and image overlay. The scale bar is 50 μm. (b). Percent changes (ΔISO) of Suv39H1 levels in the penumbra at PTS4 or PTS24 and CL4 or CL24 compared to SO. (c) Percent changes (ΔICL) of Suv39H1 levels in the penumbra at PTS4 or PTS24 compared to the CL4 or CL24. (d) Coefficient M1 of Suv39H1 co-localization with the neuronal marker (NeuN) in different control and experimental groups. (e) Immunofluorescence of Suv39H1 in cell nuclei. (f) Percentage of cells expressing Suv39H1 per 100 cells stained with Hoechst 33342 * p < 0.05 compared to SO, # p < 0.05 compared to theCL4 or CL24. One Way ANOVA; M ± SEM; n = 7.
![Ijms 22 12483 g005 Ijms 22 12483 g005]()
Figure 6.
The changes of Suv39H1 levels in the astrocytes of ischemic penumbra at 4 and 24 h after photothrombotic stroke (PTS) in the rat cerebral cortex. The level of Suv39H1 is shown at 4 and 24 h after PTS (PTS4 and PTS24, respectively) compared to the contralateral cortex of the same animals (CL4 and CL24), or the cortex of sham-operated rats (SO). (a) Immunofluorescence of Suv39H1 (green), marker of astrocytes GFAP (red), nuclear chromatin marker Hoechst 33342 (blue), and image overlay. The scale bar is 50 μm. (b) Coefficient M1 of Suv39H1 co-localization with astrocyte marker (GFAP) in different control and experimental groups. One Way ANOVA; M ± SEM; n = 7.
Figure 6.
The changes of Suv39H1 levels in the astrocytes of ischemic penumbra at 4 and 24 h after photothrombotic stroke (PTS) in the rat cerebral cortex. The level of Suv39H1 is shown at 4 and 24 h after PTS (PTS4 and PTS24, respectively) compared to the contralateral cortex of the same animals (CL4 and CL24), or the cortex of sham-operated rats (SO). (a) Immunofluorescence of Suv39H1 (green), marker of astrocytes GFAP (red), nuclear chromatin marker Hoechst 33342 (blue), and image overlay. The scale bar is 50 μm. (b) Coefficient M1 of Suv39H1 co-localization with astrocyte marker (GFAP) in different control and experimental groups. One Way ANOVA; M ± SEM; n = 7.
Figure 7.
G9a expression in nuclear (a,c) and cytoplasmic (b) fractions of penumbra tissue 4 or 24 h after photothrombotic stroke (PTS) in the cerebral cortex of rats. * p < 0.05 compared to sham-operated animals (SO) or contralateral cerebral (CL) cortex of the same animals. One-way ANOVA. M ± SEM. n = 7. Rel.un—the ratio of the optical density of the strip of the protein studied to the optical density of the strip of the protein load marker (actin).
Figure 7.
G9a expression in nuclear (a,c) and cytoplasmic (b) fractions of penumbra tissue 4 or 24 h after photothrombotic stroke (PTS) in the cerebral cortex of rats. * p < 0.05 compared to sham-operated animals (SO) or contralateral cerebral (CL) cortex of the same animals. One-way ANOVA. M ± SEM. n = 7. Rel.un—the ratio of the optical density of the strip of the protein studied to the optical density of the strip of the protein load marker (actin).
Figure 8.
The changes in G9a localization and levels in the neurons of ischemic penumbra at 4 and 24 h after photothrombotic stroke (PTS) in the rat cerebral cortex. The localization of G9a at 4 and 24 h after PTS (PTS4 and PTS24, respectively) compared to the contralateral cortex of the same animals (CL4 and CL24), or the cerebral cortex of sham-operated rats (SO). (a) Immunofluorescence of G9a (green), neuronal marker NeuN (red), nuclear chromatin marker Hoechst 33342 (blue), and image overlay. The scale bar is 50 μm. (b) Percent changes (ΔISO) of G9a levels in the penumbra (PTS4 or PTS24) and CL4 or CL24 compared to SO. (c) Percent changes (ΔICL) of G9a levels in the penumbra (PTS4 or PTS24) compared to CL4 or CL24. (d) Coefficient M1 of G9a co-localization with the neuronal marker (NeuN) in different control and experimental groups. (e) Immunofluorescence of G9a in cell nuclei. (f) Percentage of cells expressing G9a per 100 cells stained with Hoechst 33342 * p < 0.05 compared to SO, # p < 0.05 compared to CL4 or CL24. One Way ANOVA; M ± SEM; n = 7.
Figure 8.
The changes in G9a localization and levels in the neurons of ischemic penumbra at 4 and 24 h after photothrombotic stroke (PTS) in the rat cerebral cortex. The localization of G9a at 4 and 24 h after PTS (PTS4 and PTS24, respectively) compared to the contralateral cortex of the same animals (CL4 and CL24), or the cerebral cortex of sham-operated rats (SO). (a) Immunofluorescence of G9a (green), neuronal marker NeuN (red), nuclear chromatin marker Hoechst 33342 (blue), and image overlay. The scale bar is 50 μm. (b) Percent changes (ΔISO) of G9a levels in the penumbra (PTS4 or PTS24) and CL4 or CL24 compared to SO. (c) Percent changes (ΔICL) of G9a levels in the penumbra (PTS4 or PTS24) compared to CL4 or CL24. (d) Coefficient M1 of G9a co-localization with the neuronal marker (NeuN) in different control and experimental groups. (e) Immunofluorescence of G9a in cell nuclei. (f) Percentage of cells expressing G9a per 100 cells stained with Hoechst 33342 * p < 0.05 compared to SO, # p < 0.05 compared to CL4 or CL24. One Way ANOVA; M ± SEM; n = 7.
![Ijms 22 12483 g008 Ijms 22 12483 g008]()
Figure 9.
The changes of G9a levels in the astrocytes of ischemic penumbra 4 and 24 h after photothrombotic stroke (PTS) in the rat cerebral cortex. G9a levels (PTS4 and PTS24, respectively) compared to contralateral cortex of the same animals (CL4 and CL24), or the cortex of sham-operated rats (SO) are shown. (a) Immunofluorescence of G9a (green), marker of astrocytes GFAP (red), nuclear chromatin marker Hoechst 33342 (blue), and image overlay. The scale bar is 50 μm. (b) Coefficient M1 of G9a co-localization with astrocyte marker (GFAP) in different control and experimental groups. One Way ANOVA; M ± SEM; n = 7.
Figure 9.
The changes of G9a levels in the astrocytes of ischemic penumbra 4 and 24 h after photothrombotic stroke (PTS) in the rat cerebral cortex. G9a levels (PTS4 and PTS24, respectively) compared to contralateral cortex of the same animals (CL4 and CL24), or the cortex of sham-operated rats (SO) are shown. (a) Immunofluorescence of G9a (green), marker of astrocytes GFAP (red), nuclear chromatin marker Hoechst 33342 (blue), and image overlay. The scale bar is 50 μm. (b) Coefficient M1 of G9a co-localization with astrocyte marker (GFAP) in different control and experimental groups. One Way ANOVA; M ± SEM; n = 7.
Figure 10.
Expression of lysine 9 dimethylated histone H3 (H3K9diMe) in the penumbra and control samples of the cerebral cortex of sham-operated rats (SO) or the cerebral cortex in the contralateral hemisphere of the same animal (CL4 and CL24) at 4 and 24 h after photothrombotic stroke in the right hemisphere of the rat brain (PTS4 and PTS24). (a) Western blotting of H3K9diMe in the nuclear fraction of rat brain tissue. One Way ANOVA; M ± SEM, N = 8, * p < 0.05; *** p < 0.001 relative to sham-operated animals. (b) Immunofluorescence of H3K9diMe-positive cells in the cerebral cortex of sham-operated rats (SO), in the unirradiated contralateral cortex (CL4 and CL24) and in the penumbra at 4 and 24 h after photothrombotic stroke (PTS4 and PTS24). Scale section: 150 microns. (c) Percentage changes in the fluorescence intensity of H3K9diMe-positive cells in the penumbra and contralateral cortex at 4 or 24 h after PTS relative to that in sham-operated animals (ΔIso). (d) Percentage changes in the fluorescence intensity of H3K9diMe-positive cells in the penumbra at 4 or 24 h after PTS in the rat cerebral cortex relative to the contralateral cortex (ΔICL). ANOVA, n = 8, * p < 0.05 relative to sham-operated animals, # p < 0.05 relative to the contralateral hemisphere. (e,f) Average values of the M1 colocalization coefficient reflecting the proportion of pixels with a red signal (cellular marker, NeuN (e) or GFAP (f)) containing the green signal (H3K4me) in relation to the common signal from the red channel. M ± SEM, ANOVA, n = 8; * p < 0.05 relative to sham-operated animals; # p < 0.05, relative to the contralateral hemisphere. Rel.un—the ratio of the optical density of the strip of the protein studied to the optical density of the strip of the protein load marker (actin).
Figure 10.
Expression of lysine 9 dimethylated histone H3 (H3K9diMe) in the penumbra and control samples of the cerebral cortex of sham-operated rats (SO) or the cerebral cortex in the contralateral hemisphere of the same animal (CL4 and CL24) at 4 and 24 h after photothrombotic stroke in the right hemisphere of the rat brain (PTS4 and PTS24). (a) Western blotting of H3K9diMe in the nuclear fraction of rat brain tissue. One Way ANOVA; M ± SEM, N = 8, * p < 0.05; *** p < 0.001 relative to sham-operated animals. (b) Immunofluorescence of H3K9diMe-positive cells in the cerebral cortex of sham-operated rats (SO), in the unirradiated contralateral cortex (CL4 and CL24) and in the penumbra at 4 and 24 h after photothrombotic stroke (PTS4 and PTS24). Scale section: 150 microns. (c) Percentage changes in the fluorescence intensity of H3K9diMe-positive cells in the penumbra and contralateral cortex at 4 or 24 h after PTS relative to that in sham-operated animals (ΔIso). (d) Percentage changes in the fluorescence intensity of H3K9diMe-positive cells in the penumbra at 4 or 24 h after PTS in the rat cerebral cortex relative to the contralateral cortex (ΔICL). ANOVA, n = 8, * p < 0.05 relative to sham-operated animals, # p < 0.05 relative to the contralateral hemisphere. (e,f) Average values of the M1 colocalization coefficient reflecting the proportion of pixels with a red signal (cellular marker, NeuN (e) or GFAP (f)) containing the green signal (H3K4me) in relation to the common signal from the red channel. M ± SEM, ANOVA, n = 8; * p < 0.05 relative to sham-operated animals; # p < 0.05, relative to the contralateral hemisphere. Rel.un—the ratio of the optical density of the strip of the protein studied to the optical density of the strip of the protein load marker (actin).
![Ijms 22 12483 g010 Ijms 22 12483 g010]()
Figure 11.
Expression of histone H3 monomethylated for lysine 4 (H3K4Me) in the penumbra and control samples of the cerebral cortex of sham-operated rats (SO) or cerebral cortex in the contralateral hemisphere of the same animal (CL4 and CL24) 4 and 24 h after photothrombotic stroke in the right hemisphere of the rat brain (PTS4 and PTS24). (a) Western blotting of H3K4Me in the nuclear fraction of rat brain tissue. One Way ANOVA; M ± SEM, n = 8. (b) Immunofluorescence of H3K4Me-positive cells in the cerebral cortex of SO rats, in the unirradiated contralateral cortex (CL4 and CL24) and in the penumbra at PTS4 and PTS24. Scale section: 150 microns. (c) Percentage changes in the fluorescence intensity of H3K4me-positive cells in the penumbra and contralateral cortex at PTS4 and PTS24 relative to that in SO animals (ΔIso). (d) Percentage changes in the fluorescence intensity of H3K4Me-positive cells in the penumbra at PTS4 and PTS24 in the rat cerebral cortex relative to the contralateral cortex (ΔICL). One Way ANOVA, n = 8. (e,f) Average values of the M1 colocalization coefficient reflecting the proportion of pixels with a red signal (cellular marker, NeuN (e) or GFAP (f)), containing a green signal (H3K4Me), in relation to the common signal from the red channel. M ± SEM, ANOVA, n = 8; Rel.un—the ratio of the optical density of the strip of the protein studied to the optical density of the strip of the protein load marker (actin).
Figure 11.
Expression of histone H3 monomethylated for lysine 4 (H3K4Me) in the penumbra and control samples of the cerebral cortex of sham-operated rats (SO) or cerebral cortex in the contralateral hemisphere of the same animal (CL4 and CL24) 4 and 24 h after photothrombotic stroke in the right hemisphere of the rat brain (PTS4 and PTS24). (a) Western blotting of H3K4Me in the nuclear fraction of rat brain tissue. One Way ANOVA; M ± SEM, n = 8. (b) Immunofluorescence of H3K4Me-positive cells in the cerebral cortex of SO rats, in the unirradiated contralateral cortex (CL4 and CL24) and in the penumbra at PTS4 and PTS24. Scale section: 150 microns. (c) Percentage changes in the fluorescence intensity of H3K4me-positive cells in the penumbra and contralateral cortex at PTS4 and PTS24 relative to that in SO animals (ΔIso). (d) Percentage changes in the fluorescence intensity of H3K4Me-positive cells in the penumbra at PTS4 and PTS24 in the rat cerebral cortex relative to the contralateral cortex (ΔICL). One Way ANOVA, n = 8. (e,f) Average values of the M1 colocalization coefficient reflecting the proportion of pixels with a red signal (cellular marker, NeuN (e) or GFAP (f)), containing a green signal (H3K4Me), in relation to the common signal from the red channel. M ± SEM, ANOVA, n = 8; Rel.un—the ratio of the optical density of the strip of the protein studied to the optical density of the strip of the protein load marker (actin).
![Ijms 22 12483 g011 Ijms 22 12483 g011]()
Figure 12.
(a) Immunofluorescence of DNMT1, Suv39H1, G9a, and TUNEL-positive apoptotic cells, nuclear chromatin marker Hoechst 33342, and image overlay in the penumbra 24 h after photothrombotic stroke in the rat cerebral cortex. The scale bar is 100 μm. Cells containing DNMT1 or G9a co-localized with the TUNEL-positive apoptotic cells (shown by white arrows). (b) Coefficient M1 of DNMT1, Suv39H1, or G9a co-localization with TUNEL in penumbra at 24 h after photothrombotic stroke. M ± SEM; n = 7.
Figure 12.
(a) Immunofluorescence of DNMT1, Suv39H1, G9a, and TUNEL-positive apoptotic cells, nuclear chromatin marker Hoechst 33342, and image overlay in the penumbra 24 h after photothrombotic stroke in the rat cerebral cortex. The scale bar is 100 μm. Cells containing DNMT1 or G9a co-localized with the TUNEL-positive apoptotic cells (shown by white arrows). (b) Coefficient M1 of DNMT1, Suv39H1, or G9a co-localization with TUNEL in penumbra at 24 h after photothrombotic stroke. M ± SEM; n = 7.
Figure 13.
Apoptosis in the peri-infarct (P) area induced by photothrombotic stroke (PTS) in the mouse cerebral cortex. (a,b) Typical images of cortical regions stained with TUNEL (red fluorescence of apoptotic cells) at 4 days (a) and 7 days (b) after PTS. P-peri-infarct area, I-infarct area, N-normal tissue area. Control: sham-operated animals (SO). Experimental groups: The cerebral cortex of mice injected by various inhibitors. Inhibitors: 5-aza-2′-deoxycytidine (5-aza-D), a selective DNMT1 inhibitor; A-366 and BIX01294, a nonspecific inhibitor of Suv39H1 and G9a. Scale bar 200 μm, and the immunofluorescence of TUNEL in the peri-infarct (P) area at bigger magnification (Scale bar 50 μm) (c) Changes in the apoptotic index (AI, %) in the mice of experimental groups at 4 and 7 days after PTS introduction of various inhibitors. (d) The effects of inhibitors of DNMT1 and methyltransferases Suv39H1 and G9a on the infarction core volume in the mouse brain at 4 and 7 days after PTS. Mean values of the infarction core volume (mm3) in the control group (PTS without inhibitors) and in the experimental groups (administration of inhibitors). Scale bar 1 cm. One Way ANOVA; M ± SEM; n = 7–10. * p < 0.05 compared to the PTS in the absence of inhibitors.
Figure 13.
Apoptosis in the peri-infarct (P) area induced by photothrombotic stroke (PTS) in the mouse cerebral cortex. (a,b) Typical images of cortical regions stained with TUNEL (red fluorescence of apoptotic cells) at 4 days (a) and 7 days (b) after PTS. P-peri-infarct area, I-infarct area, N-normal tissue area. Control: sham-operated animals (SO). Experimental groups: The cerebral cortex of mice injected by various inhibitors. Inhibitors: 5-aza-2′-deoxycytidine (5-aza-D), a selective DNMT1 inhibitor; A-366 and BIX01294, a nonspecific inhibitor of Suv39H1 and G9a. Scale bar 200 μm, and the immunofluorescence of TUNEL in the peri-infarct (P) area at bigger magnification (Scale bar 50 μm) (c) Changes in the apoptotic index (AI, %) in the mice of experimental groups at 4 and 7 days after PTS introduction of various inhibitors. (d) The effects of inhibitors of DNMT1 and methyltransferases Suv39H1 and G9a on the infarction core volume in the mouse brain at 4 and 7 days after PTS. Mean values of the infarction core volume (mm3) in the control group (PTS without inhibitors) and in the experimental groups (administration of inhibitors). Scale bar 1 cm. One Way ANOVA; M ± SEM; n = 7–10. * p < 0.05 compared to the PTS in the absence of inhibitors.
![Ijms 22 12483 g013 Ijms 22 12483 g013]()