Estradiol Protects against Noise-Induced Hearing Loss and Modulates Auditory Physiology in Female Mice
Abstract
:1. Introduction
2. Results
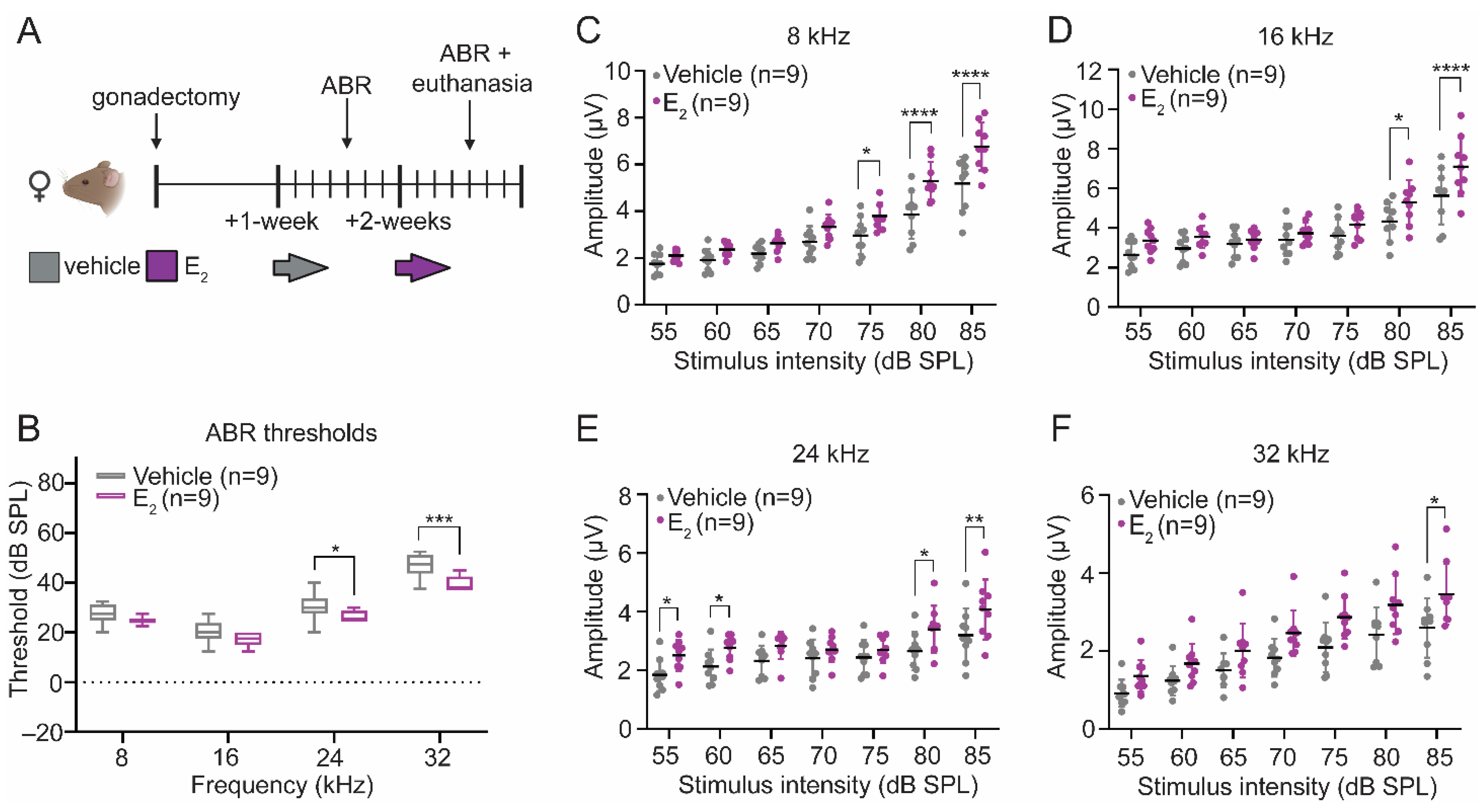
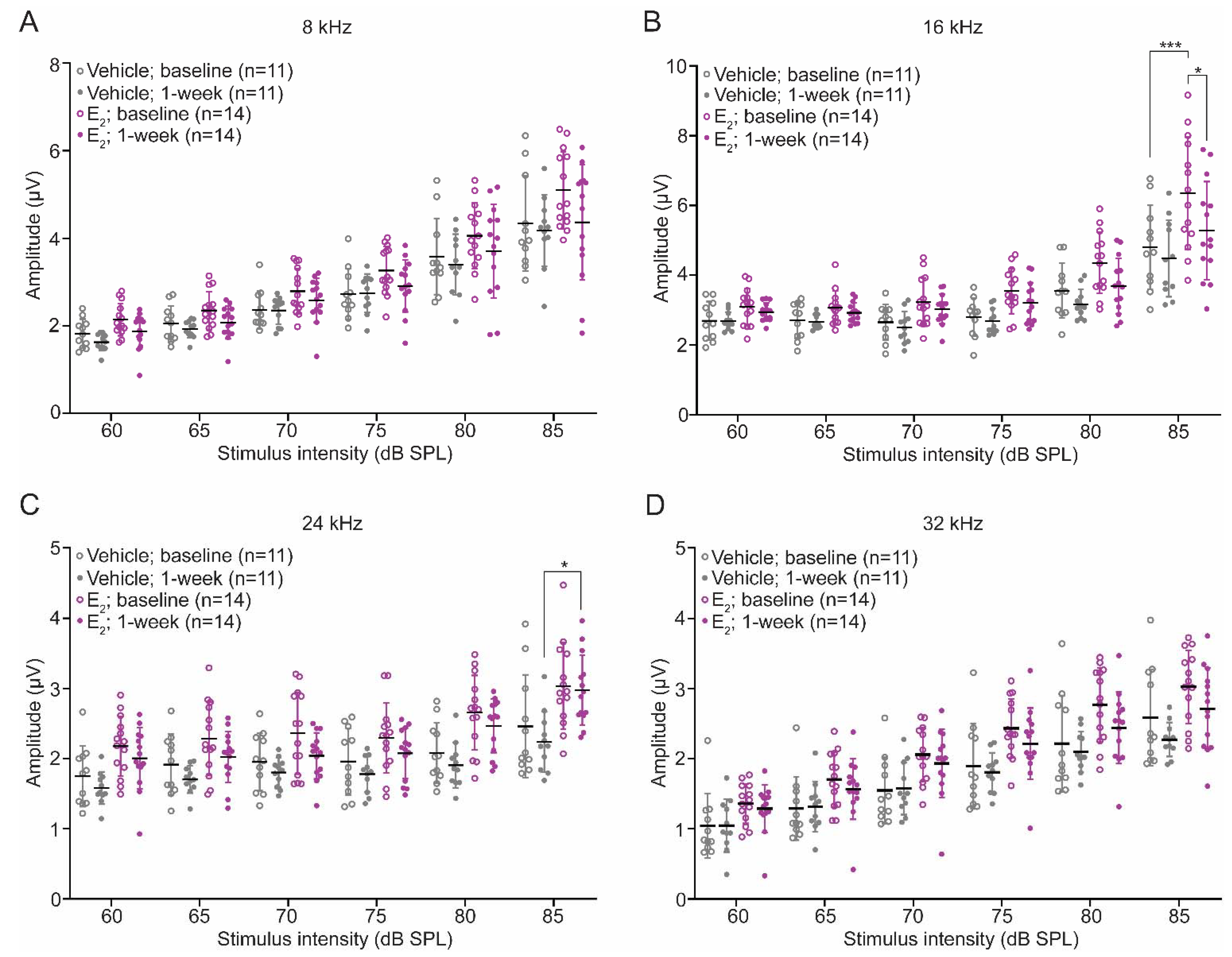
2.1. E2-Replacement Decreases Auditory Thresholds and Increases Wave-1 Amplitude
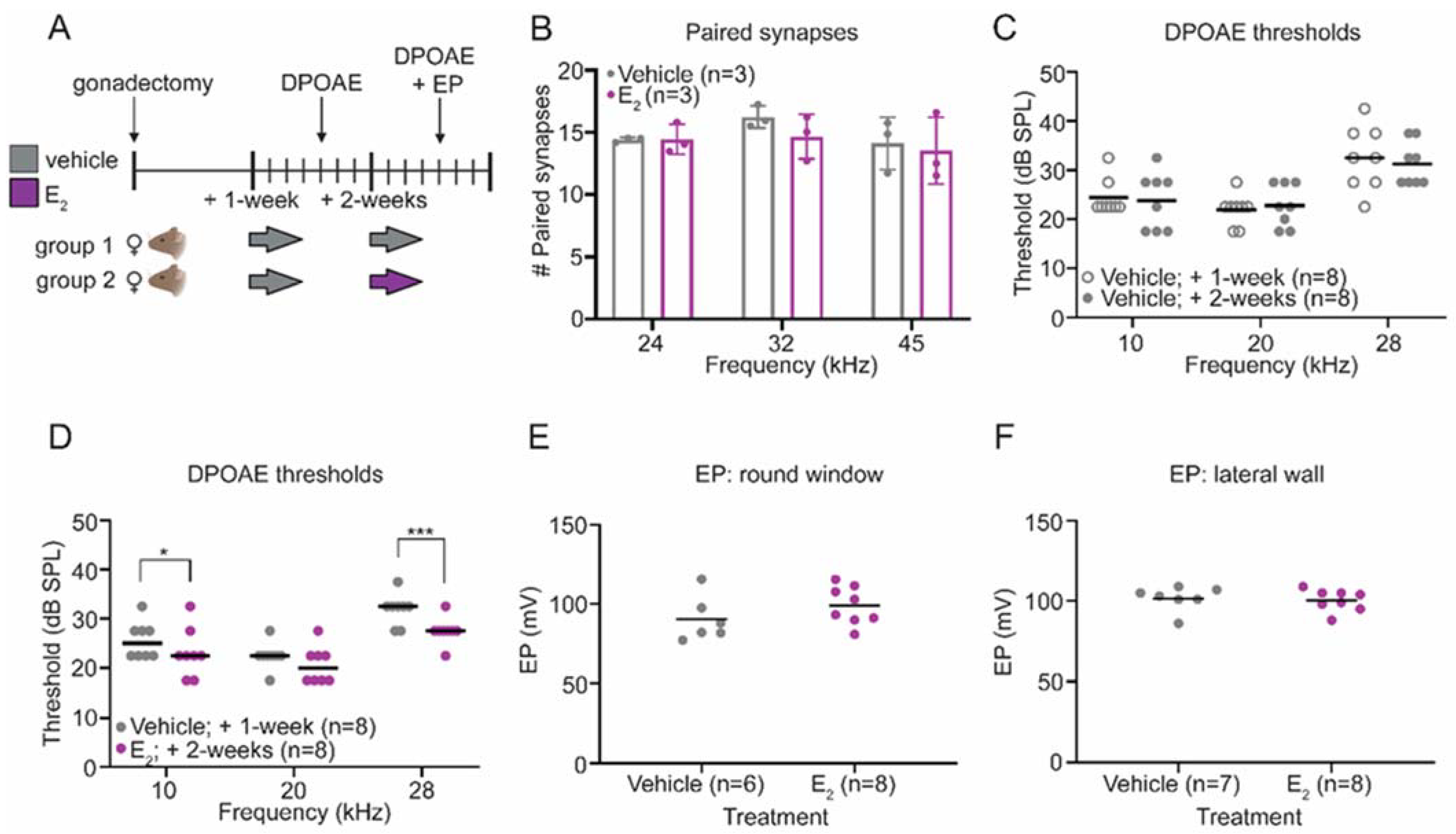
2.2. E2-Replacement Decreases Distortion Product Otoacoustic Emission Thresholds
2.3. E2-Replacementon Does Not Affect the Endocochlear Potential
2.4. Gonadectomy Eliminates Sex Differences in the Response to PTS-Inducing Noise
2.5. E2-Replacement Protects against PTS-Inducing Noise in Female Mice
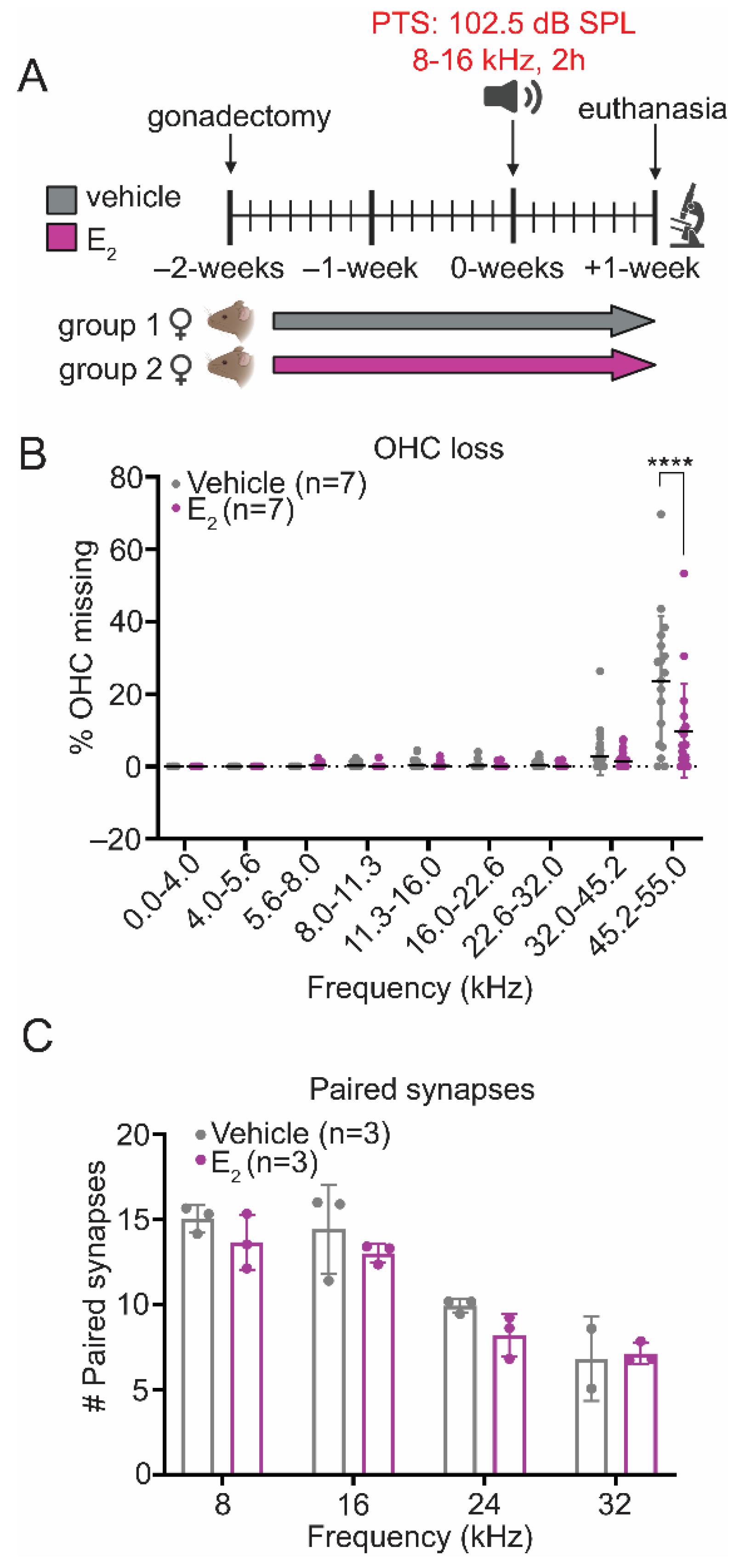
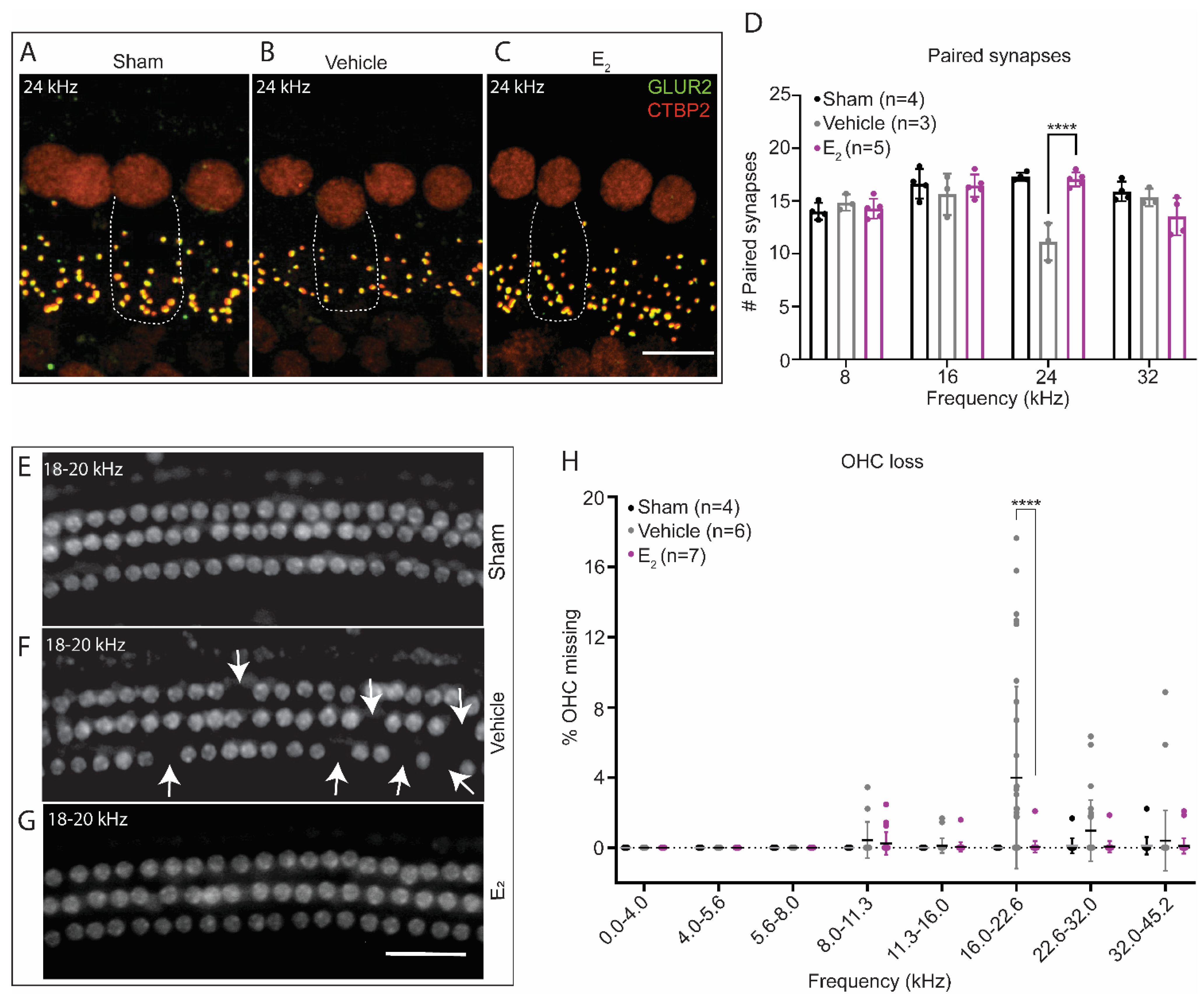
2.6. E2-Replacement Reduces OHC Loss following a PTS-Inducing Noise Exposure
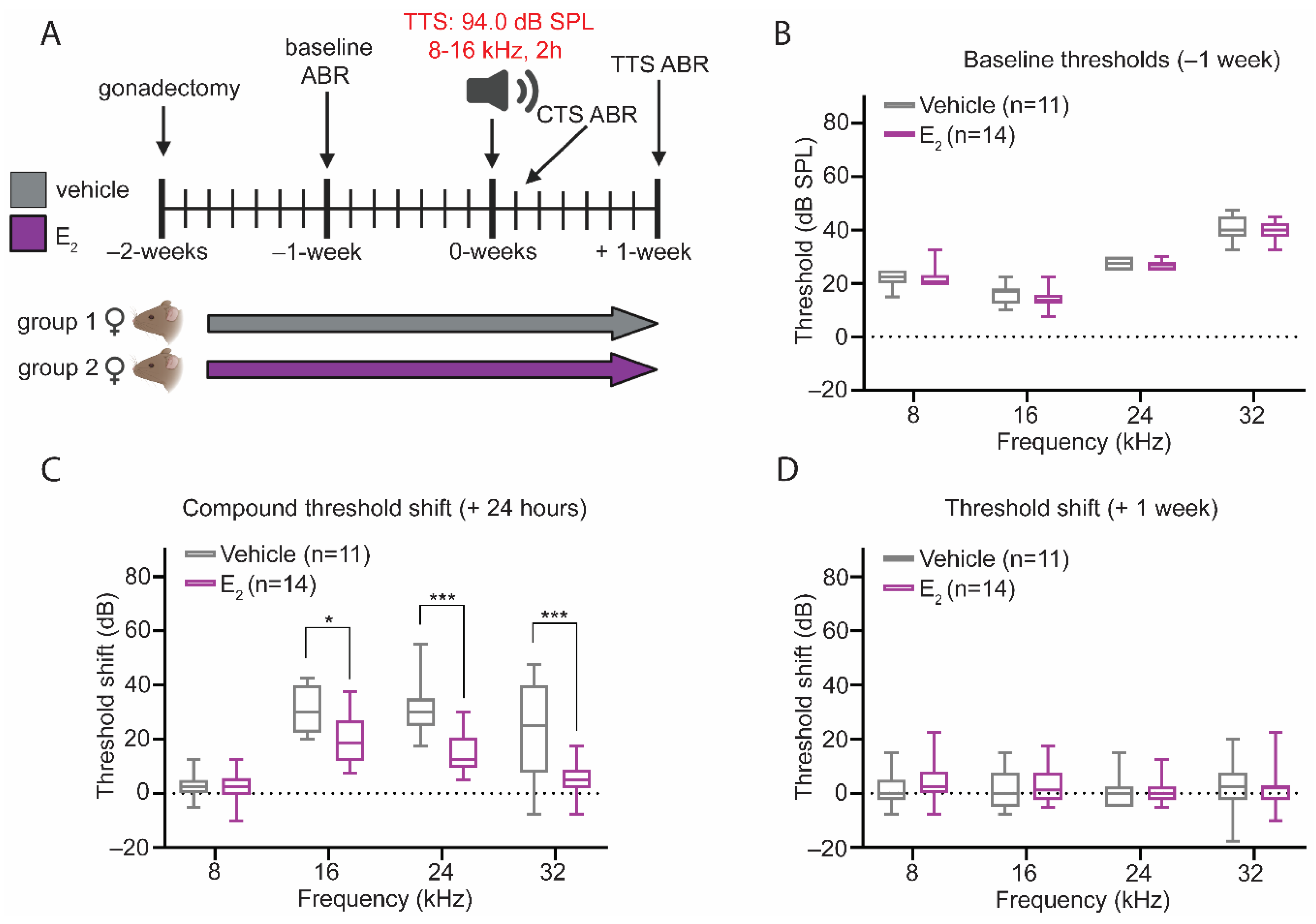
2.7. Protective Effect of E2-Replacement on Hearing Thresholds following TTS-Inducing Noise
2.8. Gonadectomy Increases Susceptibility to OHC Loss
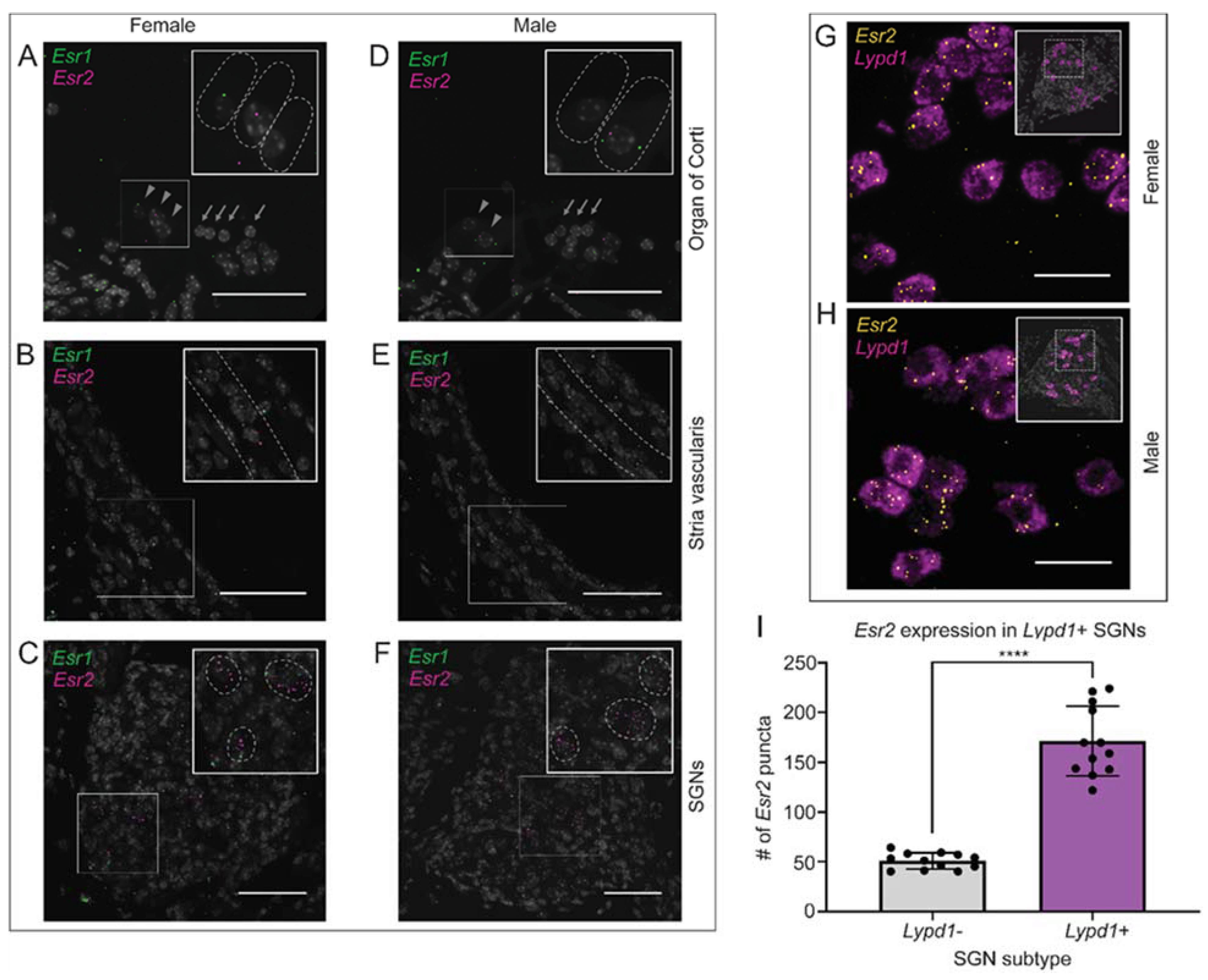
2.9. Canonical Estrogen Receptor Localization
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Gonadectomy
4.3. Subcutaneous Pump Placement
4.4. Hormonal Treatments
4.4.1. Acute Hormonal Supplementation Using Subcutaneous Injections
4.4.2. Continuous Hormonal Supplementation Using Osmotic Pumps
4.4.3. Continuous Hormonal Supplementation Using Subcutaneous Injections
4.5. Noise Exposures
4.6. Auditory Brainstem Response (ABR)
4.7. Distortion Product Otoacoustic Emissions (DPOAE)
4.8. Endocochlear Potential
4.8.1. Round Window
4.8.2. Lateral Wall
4.9. Immunostaining
4.10. Cochlear Frequency Mapping
4.10.1. Synapse Counts
4.10.2. Cytocochleograms
4.11. In Situ Hybridization
4.12. Statistics
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Appendix A

| Main Effect | F-Score | p-Value | ||
|---|---|---|---|---|
| Baseline Physiology | ||||
| ABR Thresholds | Treatment | 28.53 | <0.0001 | |
| Wave-1 Amplitudes | ||||
| 8 kHz | Treatment | 57.07 | <0.0001 | |
| 16 kHz | Treatment | 39.34 | <0.0001 | |
| 24 kHz | Treatment | 43.79 | <0.0001 | |
| 32 kHz | Treatment | 32.35 | <0.0001 | |
| DPOAE Thresholds | ||||
| Vehicle Condition | Treatment | 0.1903 | 0.6671 | |
| E2 Condition | Treatment | 36.11 | <0.0001 | |
| Paired Synapses | Treatment | 0.7923 | 0.3903 | |
| PTS Experiments | ||||
| ABR Thresholds | ||||
| Baseline | Sex | 0.1007 | 0.7516 | |
| Treatment | 0.005573 | 0.9406 | ||
| CTS | Sex | 21.04 | <0.0001 | |
| Treatment | 17.15 | <0.0001 | ||
| PTS | Sex | 27.25 | <0.0001 | |
| Treatment | 33.55 | <0.0001 | ||
| OHC Loss | Treatment | 9.791 | 0.0019 | |
| Paired Synapses | Treatment | 3.009 | 0.1033 | |
| TTS Experiments | ||||
| ABR Thresholds | ||||
| Baseline | Treatment | 2.144 | 0.1465 | |
| CTS | Treatment | 34.03 | <0.0001 | |
| TTS | Treatment | 0.09137 | 0.7631 | |
| Wave-1 Amplitudes | ||||
| 8 kHz | Treatment | 30.99 | <0.0001 | |
| 16 kHz | Treatment | 42.32 | <0.0001 | |
| 24 kHz | Treatment | 59.31 | <0.0001 | |
| 32 kHz | Treatment | 58.97 | <0.0001 | |
| OHC Loss | Treatment | 5.517 | 0.0278 | |
| Paired Synapses | Treatment | 6.705 | 0.0100 |
References
- World Health Organization. Deafness and Hearing Loss. Available online: https://www.who.int/news-room/fact-sheets/detail/deafness-and-hearing-loss (accessed on 8 June 2018).
- Haile, L.M.; Kamenov, K.; Briant, P.S.; Orji, A.U.; Steinmetz, J.D.; Abdoli, A.; Abdollahi, M.; Abu-Gharbieh, E.; Afshin, A.; Ahmed, H.; et al. Hearing loss prevalence and years lived with disability, 1990–2019: Findings from the Global Burden of Disease Study 2019. Lancet 2021, 397, 996–1009. [Google Scholar] [CrossRef]
- Crowson, M.G.; Hertzano, R.; Tucci, D.; Neurotol, O. Emerging Therapies for Sensorineural Hearing Loss HHS Public Access Author manuscript. Otol. Neurotol. 2017, 38, 792–803. [Google Scholar] [CrossRef]
- Ma, Y.; Wise, A.K.; Shepherd, R.K.; Richardson, R.T. New molecular therapies for the treatment of hearing loss. Pharmacol. Ther. 2019, 200, 190–209. [Google Scholar] [CrossRef]
- Le, T.N.; Straatman, L.V.; Lea, J.; Westerberg, B. Current insights in noise-induced hearing loss: A literature review of the underlying mechanism, pathophysiology, asymmetry, and management options. J. Otolaryngol. Head Neck Surg. 2017, 46, 41. [Google Scholar] [CrossRef] [PubMed]
- Ryan, A.F.; Kujawa, S.G.; Hammill, T.; Le Prell, C.; Kil, J. Temporary and Permanent Noise-Induced Threshold Shifts: A Review of Basic and Clinical Observations. Otol. Neurotol. 2016, 37, 271–275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liberman, M.C.; Epstein, M.J.; Cleveland, S.S.; Wang, H.; Maison, S.F. Toward a Differential Diagnosis of Hidden Hearing Loss in Humans. PLoS ONE 2016, 11, e0162726. [Google Scholar] [CrossRef] [PubMed]
- Liberman, M.C.; Kujawa, S.G. Cochlear synaptopathy in acquired sensorineural hearing loss: Manifestations and mechanisms. Hear. Res. 2017, 349, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Furman, A.C.; Kujawa, S.G.; Liberman, M.C. Noise-induced cochlear neuropathy is selective for fibers with low spontaneous rates. J. Neurophysiol. 2013, 110, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, B.R.; Chia, C.; Wu, L.; Kujawa, S.G.; Liberman, M.C.; Goodrich, L.V. Sensory Neuron Diversity in the Inner Ear Is Shaped by Activity. Cell 2018, 174, 1229–1246.e17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Plack, C.J.; Barker, D.; Prendergast, G. Perceptual consequences of ‘hidden’ hearing loss. Trends Hear. 2014, 18, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Nordmann, A.S.; Bohne, B.A.; Harding, G.W. Histopathological differences between temporary and permanent threshold shift. Hear. Res. 2000, 139, 13–30. [Google Scholar] [CrossRef]
- Ou, H.C.; Bohne, B.A.; Harding, G.W. Noise damage in the C57BL/CBA mouse cochlea. Hear. Res. 2000, 145, 111–122. [Google Scholar] [CrossRef]
- Sha, S.H.; Taylor, R.; Forge, A.; Schacht, J. Differential vulnerability of basal and apical hair cells is based on intrinsic susceptibility to free radicals. Hear. Res. 2001, 155, 1–8. [Google Scholar] [CrossRef]
- Shuster, B.Z.; Depireux, D.A.; Mong, J.A.; Hertzano, R. Sex Differences in Hearing: Probing the Role of Estrogen Signaling. J. Acoust. Soc. Am. 2019, 145, 3656–3663. [Google Scholar] [CrossRef]
- Milon, B.; Mitra, S.; Song, Y.; Margulies, Z.; Casserly, R.; Drake, V.; Mong, J.A.; Depireux, D.A.; Hertzano, R. The impact of biological sex on the response to noise and otoprotective therapies against acoustic injury in mice. Biol. Sex Differ. 2018, 9, 12. [Google Scholar] [CrossRef] [PubMed]
- Rouse, S.L.; Matthews, I.R.; Li, J.; Sherr, E.H.; Chan, D.K. Integrated stress response inhibition provides sex-dependent protection against noise-induced cochlear synaptopathy. Sci. Rep. 2020, 10, 18063. [Google Scholar] [CrossRef] [PubMed]
- Caras, M.L. Estrogenic modulation of auditory processing: A vertebrate comparison. Front. Neuroendocrinol. 2013, 34, 285–299. [Google Scholar] [CrossRef] [Green Version]
- Charitidi, K.; Meltser, I.; Tahera, Y.; Canlon, B. Functional responses of estrogen receptors in the male and female auditory system. Hear. Res. 2009, 252, 71–78. [Google Scholar] [CrossRef]
- Delhez, A.; Lefebvre, P.; Péqueux, C.; Malgrange, B.; Delacroix, L. Auditory function and dysfunction: Estrogen makes a difference. Cell. Mol. Life Sci. 2019, 77, 619–635. [Google Scholar] [CrossRef]
- Villavisanis, D.F.; Berson, E.R.; Lauer, A.M.; Cosetti, M.K.; Schrode, K.M. Sex-based Differences in Hearing Loss: Perspectives from Non-clinical Research to Clinical Outcomess. In Otology and Neurotology; Lippincott Williams and Wilkins: Philadelphia, PA, USA, 2020; Volume 41, pp. 290–298. [Google Scholar]
- Meltser, I.; Tahera, Y.; Simpson, E.; Hultcrantz, M.; Charitidi, K.; Gustafsson, J.A.; Canlon, B. Estrogen receptor Beta protects against acoustic trauma in mice. J. Clin. Investig. 2008, 118, 1563–1570. [Google Scholar] [CrossRef] [Green Version]
- Simonoska, R.; Stenberg, A.E.; Duan, M.; Yakimchuk, K.; Fridberger, A.; Sahlin, L.; Gustafsson, J.A.; Hultcrantz, M. Inner ear pathology and loss of hearing in estrogen receptor-beta deficient mice. J. Endocrinol. 2009, 201, 397–406. [Google Scholar] [CrossRef] [PubMed]
- McHenry, J.; Carrier, N.; Hull, E.; Kabbaj, M. Sex differences in anxiety and depression: Role of testosterone. In Frontiers in Neuroendocrinology; Academic Press: Cambridge, MA, USA, 2014; Volume 35, pp. 42–57. [Google Scholar]
- Brinton, R.D. Minireview: Translational Animal Models of Human Menopause: Challenges and Emerging Opportunities. Endocrinology 2012, 153, 3571–3578. [Google Scholar] [CrossRef] [PubMed]
- Komori, T. Animal models for osteoporosis. Eur. J. Pharm. 2015, 759, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Eggermont, J.J. Auditory brainstem response. In Handbook of Clinical Neurology; Elsevier B.V.: Amsterdam, The Netherlands, 2019; Volume 160, pp. 451–464. [Google Scholar]
- Kemp, D. Otoacoustic emissions, their origin in cochlear function, and use. Br. Med. Bull. 2002, 63, 223–241. [Google Scholar] [CrossRef] [PubMed]
- Brownell, W.E. Outer Hair Cell Electromotility and Otoacoustic Emissions. Ear Hear. 1990, 11, 82–92. [Google Scholar] [CrossRef] [Green Version]
- Abdala, C.; Visser-Dumont, L. Distortion Product Otoacoustic Emissions: A Tool for Hearing Assessment and Scientific Study. Volta Rev. 2001, 103, 281–302. [Google Scholar] [PubMed]
- Korrapati, S.; Taukulis, I.; Olszewski, R.; Pyle, M.; Gu, S.; Singh, R.; Griffiths, C.; Martin, D.; Boger, E.; Morell, R.J.; et al. Single Cell and Single Nucleus RNA-Seq Reveal Cellular Heterogeneity and Homeostatic Regulatory Networks in Adult Mouse Stria Vascularis. Front. Mol. Neurosci. 2019, 12, 316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wangemann, P. K+ cycling and the endocochlear potential. Hear. Res. 2002, 165, 1–9. [Google Scholar] [CrossRef]
- Ohlemiller, K.K.; Kaur, T.; Warchol, M.E.; Withnell, R.H. The endocochlear potential as an indicator of reticular lamina integrity after noise exposure in mice. Hear. Res. 2018, 361, 138–151. [Google Scholar] [CrossRef] [Green Version]
- Ohlemiller, K.K. Mechanisms and genes in human strial presbycusis from animal models. Brain Res. 2009, 1277, 70–83. [Google Scholar] [CrossRef] [Green Version]
- Sewell, W.F. The effects of furosemide on the endocochlear potential and auditory-nerve fiber tuning curves in cats. Hear. Res. 1984, 14, 305–314. [Google Scholar] [CrossRef]
- Ohlemiller, K.K.; Jones, S.M.; Johnson, K.R. Application of Mouse Models to Research in Hearing and Balance. J. Assoc. Res. Otolaryngol. 2016, 17, 493–523. [Google Scholar] [CrossRef] [PubMed]
- Kujawa, S.G.; Liberman, M.C. Synaptopathy in the noise-exposed and aging cochlea: Primary neural degeneration in acquired sensorineural hearing loss. Hear. Res. 2015, 330, 191–199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Hirose, K.; Liberman, M.C. Dynamics of Noise-Induced Cellular Injury and Repair in the Mouse Cochlea. J. Assoc. Res. Otolaryngol. 2002, 3, 248–268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, W.-Y.; Ding, D.-L.; Zheng, X.-Y.; Ruan, F.-M.; Liu, Y.-J. A comparison of changes in the stereocilia between temporary and permanent hearing losses in acoustic trauma. Hear. Res. 1992, 62, 27–41. [Google Scholar] [CrossRef]
- Hederstierna, C.; Hultcrantz, M.; Collins, A.; Rosenhall, U. The menopause triggers hearing decline in healthy women. Hear. Res. 2010, 259, 31–35. [Google Scholar] [CrossRef]
- Kim, S.H.; Kang, B.M.; Chae, H.D.; Kim, C.H. The Association between Serum Estradiol Level and Hearing Sensitivity in Postmenopausal Women. Obs. Gynecol. 2002, 99, 726–730. [Google Scholar]
- Seist, R.; Tong, M.; Landegger, L.D.; Vasilijic, S.; Hyakusoku, H.; Katsumi, S.; McKenna, C.E.; Edge, A.S.B.; Stankovic, K.M. Regeneration of Cochlear Synapses by Systemic Administration of a Bisphosphonate. Front. Mol. Neurosci. 2020, 13, 87. [Google Scholar] [CrossRef]
- Brandt, A.; Striessnig, J.; Moser, T. CaV1.3 Channels Are Essential for Development and Presynaptic Activity of Cochlear Inner Hair Cells. J. Neurosci. 2003, 23, 10832–10840. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lösel, R.M.; Falkenstein, E.; Feuring, M.; Schultz, A.; Tillmann, H.-C.; Rossol-Haseroth, K.; Wehling, M. Nongenomic Steroid Action: Controversies, Questions, and Answers. Physiol. Rev. 2003, 83, 965–1016. [Google Scholar] [CrossRef]
- Moser, T.; Beutner, D. Kinetics of exocytosis and endocytosis at the cochlear inner hair cell afferent synapse of the mouse. Proc. Natl. Acad. Sci. USA 2000, 97, 883–888. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rutherford, M.A.; Chapochnikov, N.M.; Moser, T. Spike Encoding of Neurotransmitter Release Timing by Spiral Ganglion Neurons of the Cochlea. J. Neurosci. 2012, 32, 4773–4789. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Björnström, L.; Sjöberg, M. Mechanisms of Estrogen Receptor Signaling: Convergence of Genomic and Nongenomic Actions on Target Genes. Mol. Endocrinol. 2005, 19, 833–842. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Motohashi, R.; Takumida, M.; Shimizu, A.; Konomi, U.; Fujita, K.; Hirakawa, K.; Suzuki, M.; Anniko, M. Effects of age and sex on the expression of estrogen receptor α and β in the mouse inner ear. Acta Oto-Laryngol. 2010, 130, 204–214. [Google Scholar] [CrossRef] [PubMed]
- Stenberg, A.E.; Wang, H.; Sahlin, L.; Hultcrantz, M. Mapping of estrogen receptors α and β in the inner ear of mouse and rat. Hear. Res. 1999, 136, 29–34. [Google Scholar] [CrossRef]
- Saito, K.; Cui, H. Emerging Roles of Estrogen-Related Receptors in the Brain: Potential Interactions with Estrogen Signaling. Int. J. Mol. Sci. 2018, 19, 1091. [Google Scholar] [CrossRef] [Green Version]
- Smith, S.B.; Lichtenhan, J.T.; Cone, B.K. Contralateral inhibition of click- and chirp-evoked human compound action potentials. Front. Neurosci. 2017, 11, 189. [Google Scholar] [CrossRef] [Green Version]
- Hara, Y.; Waters, E.M.; McEwen, B.S.; Morrison, J.H. Estrogen Effects on Cognitive and Synaptic Health over the Lifecourse. Physiol. Rev. 2015, 95, 785–807. [Google Scholar] [CrossRef] [Green Version]
- Hu, R.; Cai, W.; Wu, X.; Yang, Z. Astrocyte-derived estrogen enhances synapse formation and synaptic transmission between cultured neonatal rat cortical neurons. Neuroscience 2007, 144, 1229–1240. [Google Scholar] [CrossRef]
- Lu, Y.; Sareddy, G.R.; Wang, J.; Wang, R.; Li, Y.; Dong, Y.; Zhang, Q.; Liu, J.; O’Connor, J.C.; Xu, J.; et al. Neuron-Derived Estrogen Regulates Synaptic Plasticity and Memory. J. Neurosci. 2019, 39, 2792–2809. [Google Scholar] [CrossRef]
- Spencer-Segal, J.; Tsuda, M.; Mattei, L.; Waters, E.; Romeo, R.; Milner, T.; McEwen, B.; Ogawa, S. Estradiol acts via estrogen receptors alpha and beta on pathways important for synaptic plasticity in the mouse hippocampal formation. Neuroscience 2012, 202, 131–146. [Google Scholar] [CrossRef] [Green Version]
- Hashimoto, K.; Hickman, T.T.; Suzuki, J.; Ji, L.; Kohrman, D.C.; Corfas, G.; Liberman, M.C. Protection from noise-induced cochlear synaptopathy by virally mediated overexpression of NT3. Sci. Rep. 2019, 9, 15362. [Google Scholar] [CrossRef] [Green Version]
- Suzuki, J.; Corfas, G.; Liberman, M.C. Round-window delivery of neurotrophin 3 regenerates cochlear synapses after acoustic overexposure. Sci. Rep. 2016, 6, 24907. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arevalo, M.-A.; Azcoitia, I.; Garcia-Segura, L. The neuroprotective actions of oestradiol and oestrogen receptors. Nat. Rev. Neurosci. 2015, 16, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Gillies, G.E.; McArthur, S. Estrogen Actions in the Brain and the Basis for Differential Action in Men and Women: A Case for Sex-Specific Medicines. Pharm. Rev. 2010, 62, 155–198. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- FDing, F.; Yao, J.; Zhao, L.; Mao, Z.; Chen, S.; Brinton, R.D. Ovariectomy Induces a Shift in Fuel Availability and Metabolism in the Hippocampus of the Female Transgenic Model of Familial Alzheimer’s. PLoS ONE 2013, 8, e59825. [Google Scholar]
- Azcoitia, I.; Fernandez-Galaz, C.; Sierra, A.; Garcia-Segura, L.M. Gonadal hormones affect neuronal vulnerability to excitotoxin-induced degeneration. J. Neurocytol. 1999, 28, 699–710. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.X.; Payne, S.; Yang-Hood, A.; Li, S.-Z.; Davis, B.; Carlquist, J.; V-Ghaffari, B.; Gantz, J.A.; Kallogjeri, D.; Fitzpatrick, J.A.; et al. Vesicular Glutamatergic Transmission in Noise-Induced Loss and Repair of Cochlear Ribbon Synapses. J. Neurosci. 2019, 39, 4434–4447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Puel, J.-L.; Ruel, J.; D’aldin, C.G.; Pujol, R. Excitotoxicity and repair of cochlear synapses after noise-trauma induced hearing loss. NeuroReport 1998, 9, 2109–2114. [Google Scholar] [CrossRef] [PubMed]
- Hu, N.; Rutherford, M.A.; Green, S.H. Protection of cochlear synapses from noise-induced excitotoxic trauma by blockade of Ca2+-permeable AMPA receptors. Proc. Natl. Acad. Sci. USA 2020, 117, 3828–3838. [Google Scholar] [CrossRef]
- Petrone, A.B.; Gatson, J.W.; Simpkins, J.W.; Reed, M.N. Non-feminizing estrogens: A novel neuroprotective therapy. Mol. Cell. Endocrinol. 2014, 389, 40–47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pinkerton, J.V.; Thomas, S. Use of SERMs for treatment in postmenopausal women. J. Steroid Biochem. Mol. Biol. 2014, 142, 142–154. [Google Scholar] [CrossRef] [PubMed]
- Gillies, E.G.; Murray, E.H.; Dexter, D.; McArthur, S. Sex dimorphisms in the neuroprotective effects of estrogen in an animal model of Parkinson’s disease. Pharm. Biochem. Behav. 2004, 78, 513–522. [Google Scholar] [CrossRef] [PubMed]
- Razmara, A.; Duckles, S.P.; Krause, D.N.; Procaccio, V. Estrogen suppresses brain mitochondrial oxidative stress in female and male rats. Brain Res. 2007, 1176, 71–81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rusa, R.; Alkayed, N.J.; Crain, B.J.; Traystman, R.J.; Kimes, A.S.; London, E.D.; Klaus, J.A.; Hurn, P.D. 17β-Estradiol Reduces Stroke Injury in Estrogen-Deficient Female Animals. Stroke 1999, 30, 1665–1670. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Toung, T.J.K.; Traystman, R.J.; Hurn, P.D. Estrogen-Mediated Neuroprotection after Experimental Stroke in Male Rats. Stroke 1998, 29, 1666–1670. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, M.M.; Arnold, A.P. Reframing sexual differentiation of the brain. Nat. Neurosci. 2011, 14, 677–683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhatt, I.; Phillips, S.; Richter, S.; Tucker, D.; Lundgren, K.; Morehouse, R.; Henrich, V. A polymorphism in human estrogen-related receptor beta (ESRRβ) predicts audiometric temporary threshold shift. Int. J. Audiol 2016, 55, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Nolan, L.S.; Maier, H.; Hermans-Borgmeyer, I.; Girotto, G.; Ecob, R.; Pirastu, N.; Cadge, B.A.; Hübner, C.; Gasparini, P.; Strachan, D.P.; et al. Estrogen-related receptor gamma and hearing function: Evidence of a role in humans and mice. Neurobiol. Aging 2013, 34, 2077. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schilit, S.L.; Currall, B.B.; Yao, R.; Hanscom, C.; Collins, R.L.; Pillalamarri, V.; Lee, D.-Y.; Kammin, T.; Zepeda-Mendoza, C.J.; Mononen, T.; et al. Estrogen-related receptor gamma implicated in a phenotype including hearing loss and mild developmental delay. Eur. J. Hum. Genet. 2016, 24, 1622–1626. [Google Scholar] [CrossRef] [Green Version]
- Ben Saïd, M.; Ayedi, L.; Mnejja, M.; Hakim, B.; Khalfallah, A.; Charfeddine, I.; Khifagi, C.; Turki, K.; Ayadi, H.; BenZina, Z.; et al. A novel missense mutation in the ESRRB gene causes DFNB35 hearing loss in a Tunisian family. Eur. J. Med. Genet. 2011, 54, e535–e541. [Google Scholar] [CrossRef] [PubMed]
- Adriztina, I.; Adnan, A.; Adenin, I.; Haryuna, S.H.; Sarumpaet, S. Influence of hormonal changes on audiologic examination in normal ovarian cycle females: An analytic study. Int. Arch. Otorhinolaryngol. 2016, 20, 294–299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jian, H.; Yu, G.; Chen, G.; Lin, N.; Wang, H. Correlation between auditory-vestibular functions and estrogen levels in postmenopausal patients with Meniere’s disease. J. Clin. Lab. Anal. 2019, 33, e22626. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernandez, K.A.; Jeffers, P.W.; Lall, K.; Liberman, M.C.; Kujawa, S.G. Aging after Noise Exposure: Acceleration of Cochlear Synaptopathy in “Recovered” Ears. J. Neurosci. 2015, 35, 7509–7520. [Google Scholar] [CrossRef] [Green Version]
- Wu, P.; O’Malley, J.T.; de Gruttola, V.; Liberman, M.C. Age-Related Hearing Loss Is Dominated by Damage to Inner Ear Sensory Cells, Not the Cellular Battery That Powers Them. J. Neurosci. 2020, 40, 6357–6366. [Google Scholar] [CrossRef] [PubMed]
- Pottoo, F.H.; Bhowmik, M.; Vohora, D. Raloxifene protects against seizures and neurodegeneration in a mouse model mimicking epilepsy in postmenopausal woman. Eur. J. Pharm. Sci. 2014, 65, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Driowo, M.; Landgren, B.-M.; Stenström, B.; Diczfalusy, E. A comparison of the pharmacokinetic properties of three estradiol esters. Contraception 1980, 21, 415–424. [Google Scholar] [CrossRef]
- Lund-Pero, M.; Jeppson, B.; Arneklo-Nobin, B.; Sjögren, H.-O.; Holmgren, K.; Pero, R.W. Non-specific steroidal esterase activity and distribution in human and other mammalian tissues. Clin. Chim. Acta 1994, 224, 9–20. [Google Scholar] [CrossRef]
- Wangemann, P.; Itza, E.M.; Albrecht, B.; Wu, T.; Jabba, S.V.; Maganti, R.J.; Lee, J.H.; Everett, L.A.; Wall, S.M.; E Royaux, I.; et al. Loss of KCNJ10 protein expression abolishes endocochlear potential and causes deafness in Pendred syndrome mouse model. BMC Med. 2004, 2, 30. [Google Scholar] [CrossRef]
- Wangemann, P.; Nakaya, K.; Wu, T.; Maganti, R.J.; Itza, E.M.; Sanneman, J.D.; Harbidge, D.G.; Billings, S.; Marcus, D.C. Loss of cochlear HCO3− secretion causes deafness via endolymphatic acidification and inhibition of Ca2+ reabsorption in a Pendred syndrome mouse model. Am. J. Physiol.-Ren. Physiol. 2007, 292, F1345–F1353. [Google Scholar] [CrossRef] [Green Version]
- Morell, R.J.; Olszewski, R.; Tona, R.; Leitess, S.; Wafa, T.; Taukulis, I.; Schultz, J.M.; Thomason, E.J.; Richards, K.; Whitley, B.N.; et al. Noncoding Microdeletion in Mouse Hgf Disrupts Neural Crest Migration into the Stria Vascularis, Reduces the Endocochlear Potential, and Suggests the Neuropathology for Human Nonsyndromic Deafness DFNB39. J. Neurosci. 2020, 40, 2976–2992. [Google Scholar] [CrossRef] [PubMed]
- Ohlemiller, K.K.; Dahl, A.R.; Gagnon, P.M. Divergent Aging Characteristics in CBA/J and CBA/CaJ Mouse Cochleae. J. Assoc. Res. Otolaryngol. 2010, 11, 605–623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mouse Cochlear Dissection. Available online: https://vimeo.com/144531710 (accessed on 29 September 2020).

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shuster, B.; Casserly, R.; Lipford, E.; Olszewski, R.; Milon, B.; Viechweg, S.; Davidson, K.; Enoch, J.; McMurray, M.; Rutherford, M.A.; et al. Estradiol Protects against Noise-Induced Hearing Loss and Modulates Auditory Physiology in Female Mice. Int. J. Mol. Sci. 2021, 22, 12208. https://doi.org/10.3390/ijms222212208
Shuster B, Casserly R, Lipford E, Olszewski R, Milon B, Viechweg S, Davidson K, Enoch J, McMurray M, Rutherford MA, et al. Estradiol Protects against Noise-Induced Hearing Loss and Modulates Auditory Physiology in Female Mice. International Journal of Molecular Sciences. 2021; 22(22):12208. https://doi.org/10.3390/ijms222212208
Chicago/Turabian StyleShuster, Benjamin, Ryan Casserly, Erika Lipford, Rafal Olszewski, Béatrice Milon, Shaun Viechweg, Kanisa Davidson, Jennifer Enoch, Mark McMurray, Mark A. Rutherford, and et al. 2021. "Estradiol Protects against Noise-Induced Hearing Loss and Modulates Auditory Physiology in Female Mice" International Journal of Molecular Sciences 22, no. 22: 12208. https://doi.org/10.3390/ijms222212208
APA StyleShuster, B., Casserly, R., Lipford, E., Olszewski, R., Milon, B., Viechweg, S., Davidson, K., Enoch, J., McMurray, M., Rutherford, M. A., Ohlemiller, K. K., Hoa, M., Depireux, D. A., Mong, J. A., & Hertzano, R. (2021). Estradiol Protects against Noise-Induced Hearing Loss and Modulates Auditory Physiology in Female Mice. International Journal of Molecular Sciences, 22(22), 12208. https://doi.org/10.3390/ijms222212208






