Proteomic Characterization of Plasma Rich in Growth Factors and Undiluted Autologous Serum
Abstract
1. Introduction
2. Results
2.1. Hematological Characterization of PRGF
2.2. Proteomic Characterization of Blood-Derived Products
2.3. Proteomic Characterization of HK Cells Treated with Blood-Derived Products
3. Discussion
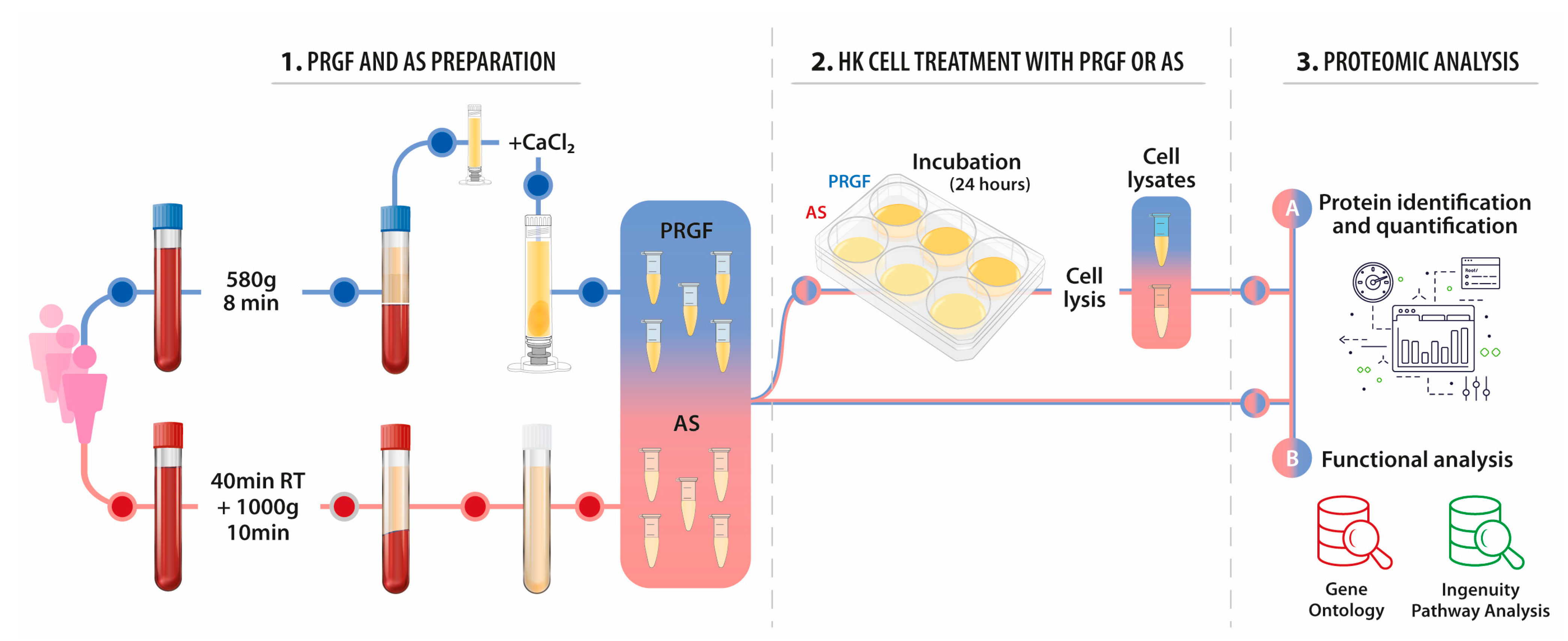
4. Materials and Methods
4.1. PRGF and Autologous Serum (AS) Preparations
4.2. Cells
4.3. Proteomic Analysis
4.4. Functional Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lemp, M.A. Management of dry eye disease. Am. J. Manag. Care 2008, 14, S88–S101. [Google Scholar] [PubMed]
- Blomquist, P.H. Ocular complications of systemic medications. Am. J. Med. Sci. 2011, 342, 62–69. [Google Scholar] [CrossRef]
- Pan, Q.; Angelina, A.; Marrone, M.; Stark, W.J.; Akpek, E.K. Autologous serum eye drops for dry eye. Cochrane Database Syst. Rev. 2017, 2, Cd009327. [Google Scholar] [CrossRef] [PubMed]
- Schultz, C. Safety and efficacy of cyclosporine in the treatment of chronic dry eye. Ophthalmol. Eye Dis. 2014, 6, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Inatomi, T.; Nakamura, T.; Kojyo, M.; Koizumi, N.; Sotozono, C.; Kinoshita, S. Ocular surface reconstruction with combination of cultivated autologous oral mucosal epithelial transplantation and penetrating keratoplasty. Am. J. Ophthalmol. 2006, 142, 757–764. [Google Scholar] [CrossRef]
- Bonini, S.; Lambiase, A.; Rama, P.; Caprioglio, G.; Aloe, L. Topical treatment with nerve growth factor for neurotrophic keratitis. Ophthalmology 2000, 107, 1347–1351. [Google Scholar] [CrossRef]
- Manni, L.; Rocco, M.L.; Bianchi, P.; Soligo, M.; Guaragna, M.; Barbaro, S.P.; Aloe, L. Nerve growth factor: Basic studies and possible therapeutic applications. Growth Factors 2013, 31, 115–122. [Google Scholar] [CrossRef]
- Márquez, E.B.; De Ortueta, D.; Royo, S.B.; Martínez-Carpio, P.A. Epidermal growth factor receptor in corneal damage: Update and new insights from recent reports. Cutan. Ocul. Toxicol. 2011, 30, 7–14. [Google Scholar] [CrossRef]
- Anitua, E.; Muruzabal, F.; Tayebba, A.; Riestra, A.; Perez, V.L.; Merayo-Lloves, J.; Orive, G. Autologous serum and plasma rich in growth factors in ophthalmology: Preclinical and clinical studies. Acta Ophthalmol. 2015, 93, e605–e614. [Google Scholar] [CrossRef]
- Riestra, A.C.; Alonso-Herreros, J.M.; Merayo-Lloves, J. Platelet rich plasma in ocular surface. Arch. Soc. Esp. Oftalmol. 2016, 91, 475–490. [Google Scholar] [CrossRef] [PubMed]
- Pan, Q.; Angelina, A.; Zambrano, A.; Marrone, M.; Stark, W.J.; Heflin, T.; Tang, L.; Akpek, E.K. Autologous serum eye drops for dry eye. Cochrane Database Syst. Rev. 2013, 8, Cd009327. [Google Scholar]
- Giannaccare, G.; Versura, P.; Buzzi, M.; Primavera, L.; Pellegrini, M.; Campos, E.C. Blood derived eye drops for the treatment of cornea and ocular surface diseases. Transfus. Apher. Sci. 2017, 56, 595–604. [Google Scholar] [CrossRef]
- Geerling, G.; Maclennan, S.; Hartwig, D. Autologous serum eye drops for ocular surface disorders. Br. J. Ophthalmol. 2004, 88, 1467–1474. [Google Scholar] [CrossRef]
- Anitua, E. Plasma rich in growth factors: Preliminary results of use in the preparation of future sites for implants. Int. J. Oral Maxillofac. Implant 1999, 14, 529–535. [Google Scholar]
- Anitua, E.; de la Fuente, M.; Muruzabal, F.; Riestra, A.; Merayo-Lloves, J.; Orive, G. Plasma rich in growth factors (PRGF) eye drops stimulates scarless regeneration compared to autologous serum in the ocular surface stromal fibroblasts. Exp. Eye Res. 2015, 135, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Anitua, E.; Zalduendo, M.M.; Alkhraisat, M.H.; Orive, G. Release kinetics of platelet-derived and plasma-derived growth factors from autologous plasma rich in growth factors. Ann. Anat. 2013, 195, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Avila, R.M.; Merayo-Lloves, J.; Riestra, A.C.; Fernandez-Vega Cueto, L.; Anitua, E.; Begoña, L.; Muruzabal, F.; Orive, G. Treatment of patients with neurotrophic keratitis stages 2 and 3 with plasma rich in growth factors (PRGF-Endoret) eye-drops. Int. Ophthalmol. 2018, 38, 1193–1204. [Google Scholar] [CrossRef]
- Merayo-Lloves, J.; Sanchez-Avila, R.M.; Riestra, A.C.; Anitua, E.; Begoña, L.; Orive, G.; Fernandez-Vega, L. Safety and Efficacy of Autologous Plasma Rich in Growth Factors Eye Drops for the Treatment of Evaporative Dry Eye. Ophthalmic Res. 2016, 56, 68–73. [Google Scholar] [CrossRef]
- Sanchez-Avila, R.M.; Merayo-Lloves, J.; Muruzabal, F.; Orive, G.; Anitua, E. Plasma rich in growth factors for the treatment of dry eye from patients with graft versus host diseases. Eur. J. Ophthalmol. 2020, 30, 94–103. [Google Scholar] [CrossRef]
- Freire, V.; Andollo, N.; Etxebarria, J.; Durán, J.A.; Morales, M.C. In vitro effects of three blood derivatives on human corneal epithelial cells. Investig. Ophthalmol. Vis. Sci. 2012, 53, 5571–5578. [Google Scholar] [CrossRef]
- Freire, V.; Andollo, N.; Etxebarria, J.; Hernáez-Moya, R.; Durán, J.A.; Morales, M.C. Corneal wound healing promoted by 3 blood derivatives: An in vitro and in vivo comparative study. Cornea 2014, 33, 614–620. [Google Scholar] [CrossRef] [PubMed]
- Anitua, E.; Muruzabal, F.; de la Fuente, M.; Riestra, A.; Merayo-Lloves, J.; Orive, G. PRGF exerts more potent proliferative and anti-inflammatory effects than autologous serum on a cell culture inflammatory model. Exp. Eye Res. 2016, 151, 115–121. [Google Scholar] [CrossRef]
- Anitua, E.; de la Fuente, M.; Muruzabal, F.; Sánchez-Ávila, R.M.; Merayo-Lloves, J.; Azkargorta, M.; Elortza, F.; Orive, G. Differential profile of protein expression on human keratocytes treated with autologous serum and plasma rich in growth factors (PRGF). PLoS ONE 2018, 13, e0205073. [Google Scholar] [CrossRef]
- Jover Botella, A.; Márquez Peiró, J.F.; Márques, K.; Monts Cambero, N.; Selva Otaolaurruchi, J. Effectiveness of 100% autologous serum drops in ocular surface disorders. Farm. Hosp. 2011, 35, 8–13. [Google Scholar] [CrossRef]
- Lekhanont, K.; Jongkhajornpong, P.; Anothaisintawee, T.; Chuckpaiwong, V. Undiluted Serum Eye Drops for the Treatment of Persistent Corneal Epitheilal Defects. Sci. Rep. 2016, 6, 38143. [Google Scholar] [CrossRef]
- Mishra, A.; Harmon, K.; Woodall, J.; Vieira, A. Sports medicine applications of platelet rich plasma. Curr. Pharm. Biotechnol. 2012, 13, 1185–1195. [Google Scholar] [CrossRef] [PubMed]
- DeLong, J.M.; Russell, R.P.; Mazzocca, A.D. Platelet-rich plasma: The PAW classification system. Arthroscopy 2012, 28, 998–1009. [Google Scholar] [CrossRef]
- Mautner, K.; Malanga, G.A.; Smith, J.; Shiple, B.; Ibrahim, V.; Sampson, S.; Bowen, J.E. A call for a standard classification system for future biologic research: The rationale for new PRP nomenclature. PM&R 2015, 7, S53–S59. [Google Scholar]
- Magalon, J.; Chateau, A.L.; Bertrand, B.; Louis, M.L.; Silvestre, A.; Giraudo, L.; Veran, J.; Sabatier, F. DEPA classification: A proposal for standardising PRP use and a retrospective application of available devices. BMJ Open Sport Exerc. Med. 2016, 2, e000060. [Google Scholar] [CrossRef] [PubMed]
- Harrison, P.; Subcommittee on Platelet, P. The use of platelets in regenerative medicine and proposal for a new classification system: Guidance from the SSC of the ISTH. J. Thromb. Haemost. 2018, 16, 1895–1900. [Google Scholar] [CrossRef]
- Lana, J.; Purita, J.; Paulus, C.; Huber, S.C.; Rodrigues, B.L.; Rodrigues, A.A.; Santana, M.H.; Madureira, J.L., Jr.; Malheiros Luzo, A.C.; Belangero, W.D.; et al. Contributions for classification of platelet rich plasma—Proposal of a new classification: MARSPILL. Regen. Med. 2017, 12, 565–574. [Google Scholar] [CrossRef]
- Kon, E.; Di Matteo, B.; Delgado, D.; Cole, B.J.; Dorotei, A.; Dragoo, J.L.; Filardo, G.; Fortier, L.A.; Giuffrida, A.; Jo, C.H.; et al. Platelet-rich plasma for the treatment of knee osteoarthritis: An expert opinion and proposal for a novel classification and coding system. Expert Opin. Biol. Ther. 2020, 20, 1447–1460. [Google Scholar] [CrossRef] [PubMed]
- Imanishi, J.; Kamiyama, K.; Iguchi, I.; Kita, M.; Sotozono, C.; Kinoshita, S. Growth factors: Importance in wound healing and maintenance of transparency of the cornea. Prog. Retin. Eye Res. 2000, 19, 113–129. [Google Scholar] [CrossRef]
- Noble, B.A.; Loh, R.S.; MacLennan, S.; Pesudovs, K.; Reynolds, A.; Bridges, L.R.; Burr, J.; Stewart, O.; Quereshi, S. Comparison of autologous serum eye drops with conventional therapy in a randomised controlled crossover trial for ocular surface disease. Br. J. Ophthalmol. 2004, 88, 647–652. [Google Scholar] [CrossRef]
- Jeng, B.H.; Dupps, W.J., Jr. Autologous serum 50% eyedrops in the treatment of persistent corneal epithelial defects. Cornea 2009, 28, 1104–1108. [Google Scholar] [CrossRef]
- Anderson, N.L.; Anderson, N.G. The human plasma proteome: History, character, and diagnostic prospects. Mol. Cell Proteom. 2002, 1, 845–867. [Google Scholar] [CrossRef]
- Anderson, N.L.; Polanski, M.; Pieper, R.; Gatlin, T.; Tirumalai, R.S.; Conrads, T.P.; Veenstra, T.D.; Adkins, J.N.; Pounds, J.G.; Fagan, R.; et al. The human plasma proteome: A nonredundant list developed by combination of four separate sources. Mol. Cell Proteom. 2004, 3, 311–326. [Google Scholar] [CrossRef]
- Ma, I.H.; Chen, L.W.; Tu, W.H.; Lu, C.J.; Huang, C.J.; Chen, W.L. Serum components and clinical efficacies of autologous serum eye drops in dry eye patients with active and inactive Sjogren syndrome. Taiwan J. Ophthalmol. 2017, 7, 213–220. [Google Scholar]
- Stenwall, P.A.; Bergström, M.; Seiron, P.; Sellberg, F.; Olsson, T.; Knutson, F.; Berglund, D. Improving the anti-inflammatory effect of serum eye drops using allogeneic serum permissive for regulatory T cell induction. Acta Ophthalmol. 2015, 93, 654–657. [Google Scholar] [CrossRef] [PubMed]
- Read, R.W. Clinical mini-review: Systemic lupus erythematosus and the eye. Ocul. Immunol. Inflamm. 2004, 12, 87–99. [Google Scholar] [CrossRef]
- Stern, M.E.; Schaumburg, C.S.; Siemasko, K.F.; Gao, J.; Wheeler, L.A.; Grupe, D.A.; De Paiva, C.S.; Calder, V.L.; Calonge, M.; Niederkorn, J.Y.; et al. Autoantibodies contribute to the immunopathogenesis of experimental dry eye disease. Investig. Ophthalmol. Vis. Sci. 2012, 53, 2062–2075. [Google Scholar] [CrossRef]
- Tabbara, K.F.; Al-Ghamdi, A.; Al-Mohareb, F.; Ayas, M.; Chaudhri, N.; Al-Sharif, F.; Al-Zahrani, H.; Mohammed, S.Y.; Nassar, A.; Aljurf, M. Ocular findings after allogeneic hematopoietic stem cell transplantation. Ophthalmology 2009, 116, 1624–1629. [Google Scholar] [CrossRef]
- Gan, L.; Fagerholm, P.; Palmblad, J. Vascular endothelial growth factor (VEGF) and its receptor VEGFR-2 in the regulation of corneal neovascularization and wound healing. Acta Ophthalmol. Scand. 2004, 82, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Yadav, U.C.; Kalariya, N.M.; Ramana, K.V. Emerging role of antioxidants in the protection of uveitis complications. Curr. Med. Chem. 2011, 18, 931–942. [Google Scholar] [CrossRef] [PubMed]
- Cejka, C.; Cejkova, J. Oxidative stress to the cornea, changes in corneal optical properties, and advances in treatment of corneal oxidative injuries. Oxid. Med. Cell Longev. 2015, 2015, 591530. [Google Scholar] [CrossRef] [PubMed]
- Ishimoto, S.; Wu, G.S.; Hayashi, S.; Zhang, J.; Rao, N.A. Free radical tissue damages in the anterior segment of the eye in experimental autoimmune uveitis. Investig. Ophthalmol. Vis. Sci. 1996, 37, 630–636. [Google Scholar]
- Augustin, A.J.; Spitznas, M.; Kaviani, N.; Meller, D.; Koch, F.H.; Grus, F.; Göbbels, M.J. Oxidative reactions in the tear fluid of patients suffering from dry eyes. Graefes Arch. Clin. Exp. Ophthalmol. 1995, 233, 694–698. [Google Scholar] [CrossRef]
- Buddi, R.; Lin, B.; Atilano, S.R.; Zorapapel, N.C.; Kenney, M.C.; Brown, D.J. Evidence of oxidative stress in human corneal diseases. J. Histochem. Cytochem. 2002, 50, 341–351. [Google Scholar] [CrossRef]
- Alio, J.L.; Ayala, M.J.; Mulet, M.E.; Artola, A.; Ruiz, J.M.; Bellot, J. Antioxidant therapy in the treatment of experimental acute corneal inflammation. Ophthalmic Res. 1995, 27, 136–143. [Google Scholar] [CrossRef]
- Cejková, J.; Ardan, T.; Cejka, C.; Luyckx, J. Favorable effects of trehalose on the development of UVB-mediated antioxidant/pro-oxidant imbalance in the corneal epithelium, proinflammatory cytokine and matrix metalloproteinase induction, and heat shock protein 70 expression. Graefes Arch. Clin. Exp. Ophthalmol. 2011, 249, 1185–1194. [Google Scholar] [CrossRef]
- Anitua, E.; de la Fuente, M.; Del Olmo-Aguado, S.; Suarez-Barrio, C.; Merayo-Lloves, J.; Muruzabal, F. Plasma rich in growth factors reduces blue light-induced oxidative damage on retinal pigment epithelial cells and restores their homeostasis by modulating vascular endothelial growth factor and pigment epithelium-derived factor expression. Clin. Exp. Ophthalmol. 2020, 48, 830–838. [Google Scholar] [CrossRef]
- Suarez-Barrio, C.; Del Olmo-Aguado, S.; Garcia-Perez, E.; de la Fuente, M.; Muruzabal, F.; Anitua, E.; Baamonde-Arbaiza, B.; Fernandez-Vega-Cueto, L.; Fernandez-Vega, L.; Merayo-Lloves, J. Antioxidant Role of PRGF on RPE Cells after Blue Light Insult as a Therapy for Neurodegenerative Diseases. Int. J. Mol. Sci. 2020, 21, 1021. [Google Scholar] [CrossRef]
- Kitazawa, T.; Kinoshita, S.; Fujita, K.; Araki, K.; Watanabe, H.; Ohashi, Y.; Manabe, R. The mechanism of accelerated corneal epithelial healing by human epidermal growth factor. Investig. Ophthalmol. Vis. Sci. 1990, 31, 1773–1778. [Google Scholar]
- Zieske, J.D.; Takahashi, H.; Hutcheon, A.E.; Dalbone, A.C. Activation of epidermal growth factor receptor during corneal epithelial migration. Investig. Ophthalmol. Vis. Sci. 2000, 41, 1346–1355. [Google Scholar]
- Maldonado, B.A.; Furcht, L.T. Epidermal growth factor stimulates integrin-mediated cell migration of cultured human corneal epithelial cells on fibronectin and arginine-glycine-aspartic acid peptide. Investig. Ophthalmol. Vis. Sci. 1995, 36, 2120–2126. [Google Scholar]
- He, J.; Bazan, H.E. Epidermal growth factor synergism with TGF-beta1 via PI-3 kinase activity in corneal keratocyte differentiation. Investig. Ophthalmol. Vis. Sci. 2008, 49, 2936–2945. [Google Scholar] [CrossRef] [PubMed]
- Netto, M.V.; Mohan, R.R.; Sinha, S.; Sharma, A.; Dupps, W.; Wilson, S.E. Stromal haze, myofibroblasts, and surface irregularity after PRK. Exp. Eye Res. 2006, 82, 788–797. [Google Scholar] [CrossRef]
- Zoncu, R.; Efeyan, A.; Sabatini, D.M. mTOR: From growth signal integration to cancer, diabetes and ageing. Nat. Rev. Mol. Cell Biol. 2011, 12, 21–35. [Google Scholar] [CrossRef]
- Wang, Y.; Gao, G.; Wu, Y.; Wang, Y.; Wu, X.; Zhou, Q. S100A4 Silencing Facilitates Corneal Wound Healing after Alkali Burns by Promoting Autophagy via Blocking the PI3K/Akt/mTOR Signaling Pathway. Investig. Ophthalmol. Vis. Sci. 2020, 61, 19. [Google Scholar] [CrossRef]
- Lee, K.S.; Ko, D.A.; Kim, E.S.; Kim, M.J.; Tchah, H.; Kim, J.Y. Bevacizumab and rapamycin can decrease corneal opacity and apoptotic keratocyte number following photorefractive keratectomy. Investig. Ophthalmol. Vis. Sci. 2012, 53, 7645–7653. [Google Scholar] [CrossRef][Green Version]
- Shin, Y.J.; Hyon, J.Y.; Choi, W.S.; Yi, K.; Chung, E.S.; Chung, T.Y.; Wee, W.R. Chemical injury-induced corneal opacity and neovascularization reduced by rapamycin via TGF-β1/ERK pathways regulation. Investig. Ophthalmol. Vis. Sci. 2013, 54, 4452–4458. [Google Scholar] [CrossRef]
- Milani, B.Y.; Milani, F.Y.; Park, D.W.; Namavari, A.; Shah, J.; Amirjamshidi, H.; Ying, H.; Djalilian, A.R. Rapamycin inhibits the production of myofibroblasts and reduces corneal scarring after photorefractive keratectomy. Investig. Ophthalmol. Vis. Sci. 2013, 54, 7424–7430. [Google Scholar] [CrossRef]
- Kaibuchi, K.; Kuroda, S.; Amano, M. Regulation of the cytoskeleton and cell adhesion by the Rho family GTPases in mammalian cells. Annu. Rev. Biochem. 1999, 68, 459–486. [Google Scholar] [CrossRef]
- Jester, J.V.; Barry-Lane, P.A.; Cavanagh, H.D.; Petroll, W.M. Induction of alpha-smooth muscle actin expression and myofibroblast transformation in cultured corneal keratocytes. Cornea 1996, 15, 505–516. [Google Scholar] [CrossRef]
- Chen, J.; Guerriero, E.; Sado, Y.; SundarRaj, N. Rho-Mediated Regulation of TGF-β1– and FGF-2–Induced Activation of Corneal Stromal Keratocytes. Investig. Ophthalmol. Vis. Sci. 2009, 50, 3662–3670. [Google Scholar] [CrossRef]
- Anitua, E.; Sanchez, M.; Merayo-Lloves, J.; De la Fuente, M.; Muruzabal, F.; Orive, G. Plasma rich in growth factors (PRGF-Endoret) stimulates proliferation and migration of primary keratocytes and conjunctival fibroblasts and inhibits and reverts TGF-beta1-Induced myodifferentiation. Investig. Ophthalmol. Vis. Sci. 2011, 52, 6066–6073. [Google Scholar] [CrossRef]
- Anitua, E.; Muruzabal, F.; Alcalde, I.; Merayo-Lloves, J.; Orive, G. Plasma rich in growth factors (PRGF-Endoret) stimulates corneal wound healing and reduces haze formation after PRK surgery. Exp. Eye Res. 2013, 115, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Maltseva, O.; Folger, P.; Zekaria, D.; Petridou, S.; Masur, S.K. Fibroblast growth factor reversal of the corneal myofibroblast phenotype. Investig. Ophthalmol. Vis. Sci. 2001, 42, 2490–2495. [Google Scholar]
- Wiśniewski, J.R.; Zougman, A.; Nagaraj, N.; Mann, M. Universal sample preparation method for proteome analysis. Nat. Methods 2009, 6, 359–362. [Google Scholar] [CrossRef] [PubMed]
- Cox, J.; Mann, M. MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. Nat. Biotechnol. 2008, 26, 1367–1372. [Google Scholar] [CrossRef] [PubMed]
- Tyanova, S.; Temu, T.; Sinitcyn, P.; Carlson, A.; Hein, M.Y.; Geiger, T.; Mann, M.; Cox, J. The Perseus computational platform for comprehensive analysis of (prote)omics data. Nat. Methods 2016, 13, 731–740. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Bioinformatics enrichment tools: Paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res. 2009, 37, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef] [PubMed]
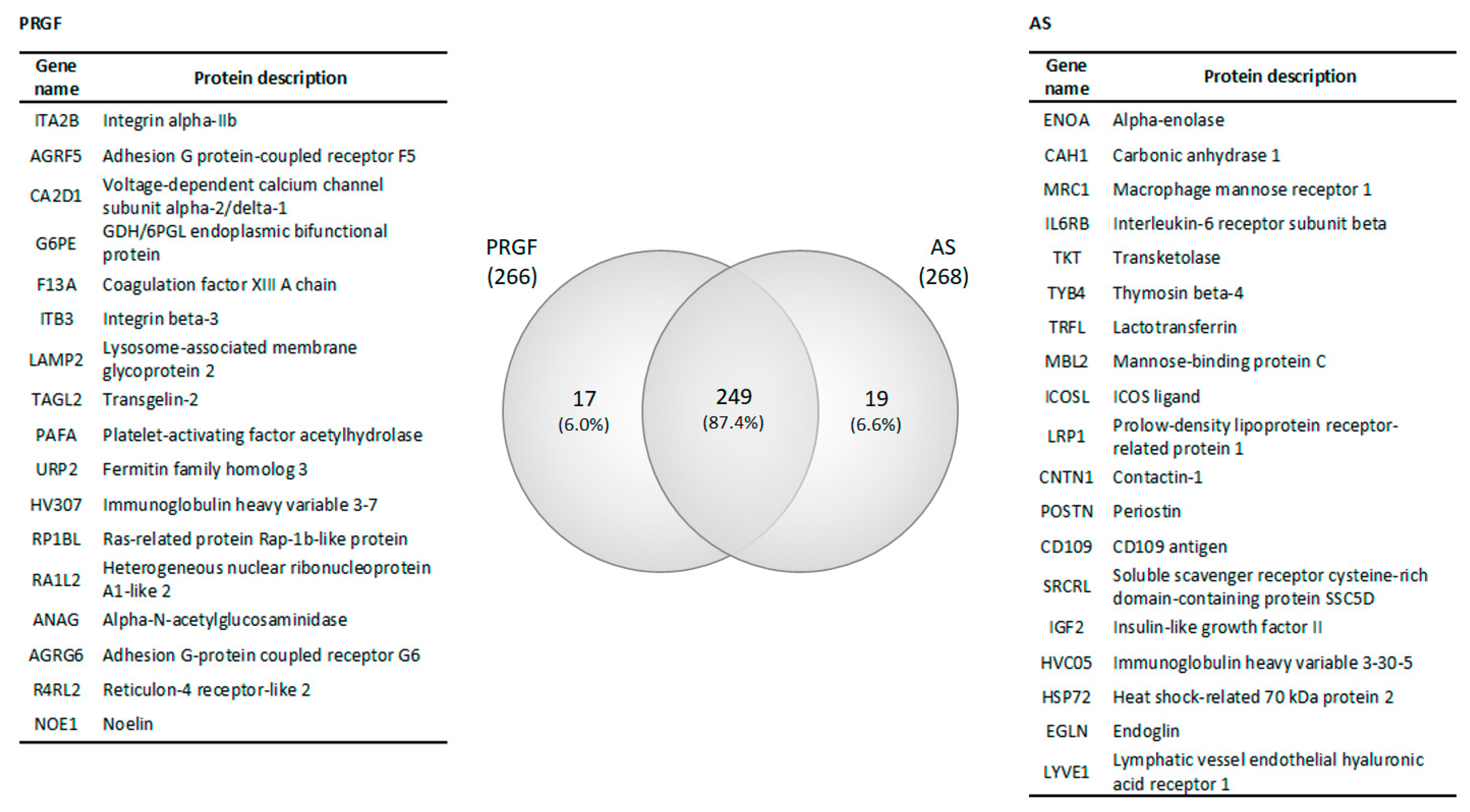

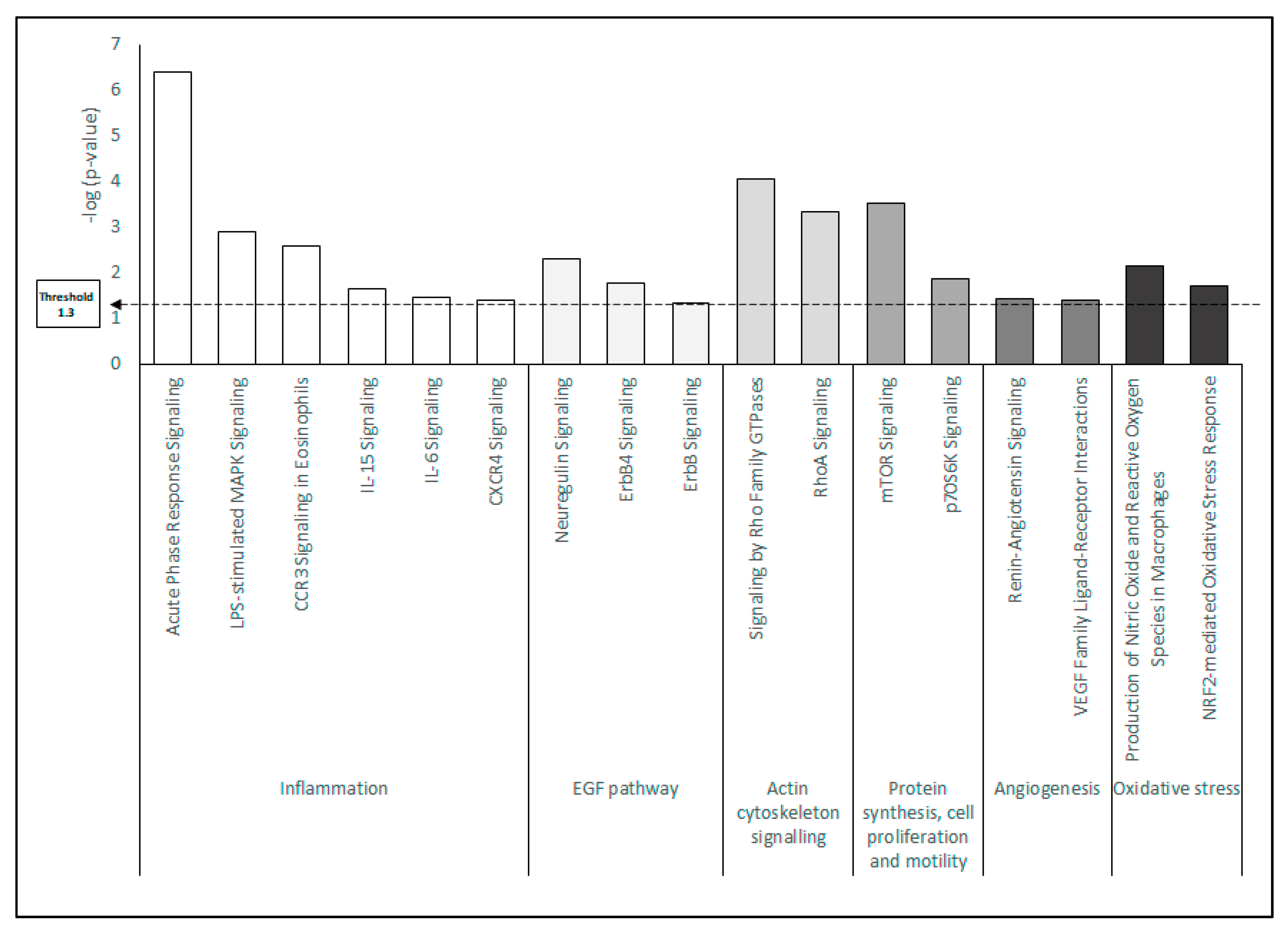

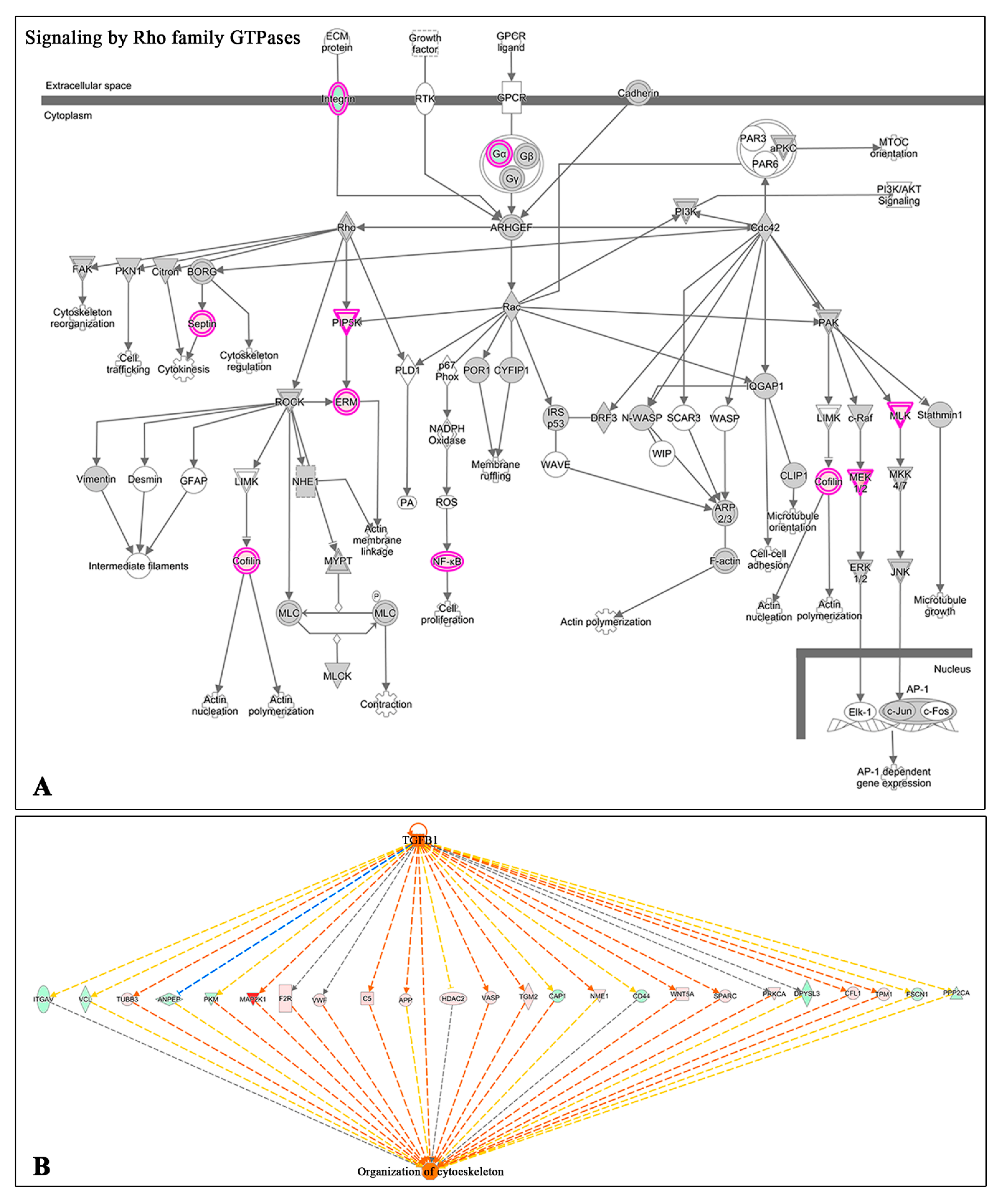
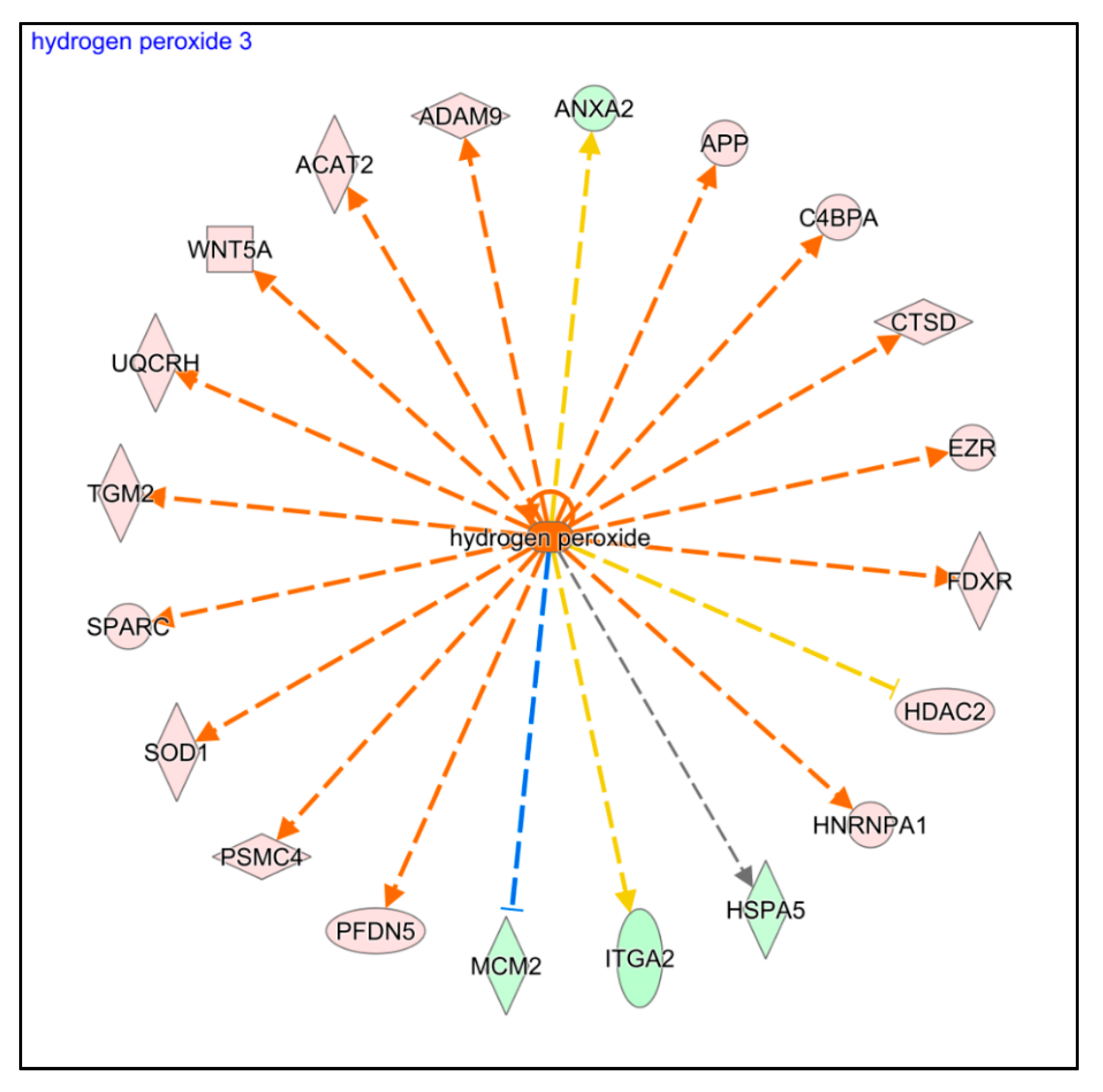
| Classification | Reference | PRGF-Endoret |
|---|---|---|
| MISHRA | [26] | 4 (B) |
| PAW | [27] | P2-x-Bβ |
| PLRA | [28] | P0.5 L-R-A+ |
| DEPA | [29] | CCA |
| ISTH | [30] | PRP IIA1 |
| MARSPILL | [31] | M A+ RBC-P Sp1 PL2-3 Lc-P A- |
| Consensus Experts | [32] | 24-00-11 |
| Protein Accession Number | Gene Name | Protein Description | Fold Change | p-Value |
|---|---|---|---|---|
| Q9UK55 | ZPI | Protein Z-dependent protease inhibitor | 1.2 | 0.0001 |
| P68871 | HBB | Hemoglobin subunit beta | 31.4 | 0.0098 |
| P02788 | TRFL | Lactotransferrin | 2.8 | 0.0156 |
| P03952 | KLKB1 | Plasma kallikrein | 1.1 | 0.0168 |
| P14151 | LYAM1 | L-selectin | 1.1 | 0.0192 |
| P01024 | CO3 | Complement C3 | 1.0 | 0.0193 |
| P08603 | CFAH | Complement factor H | 1.1 | 0.0253 |
| P62328 | TYB4 | Thymosin beta-4 | 3.2 | 0.0255 |
| P00915 | CAH1 | Carbonic anhydrase 1 | 13.2 | 0.0301 |
| Q92496 | FHR4 | Complement factor H-related protein 4 | 1.7 | 0.0329 |
| P08709 | FA7 | Coagulation factor VII | 1.4 | 0.0428 |
| P22105 | TENX | Tenascin-X | 0.9 | 0.0474 |
| P01860 | IGHG3 | Immunoglobulin heavy constant gamma 3 | 1.2 | 0.0491 |
| GO Term | GO Definition | Genes | % | p-Value |
|---|---|---|---|---|
| 0006956 | Complement activation | P08603, P01860, P01024 | 23.1 | 0.0017 |
| 0006508 | Proteolysis | P03952, P02788, P01860, P01024 | 30.8 | 0.0047 |
| 0007596 | Blood coagulation | P68871, P08709, Q9UK55 | 23.1 | 0.0073 |
| 0006957 | Complement activation, alternative pathway | P08603, P01024 | 15.4 | 0.0093 |
| 0030449 | Regulation of complement activation | P08603, P01024 | 15.4 | 0.0212 |
| 0001895 | Retina homeostasis | P02788, P01860 | 15.4 | 0.0282 |
| 0015701 | Bicarbonate transport | P00915, P68871 | 15.4 | 0.0310 |
| 0006958 | Complement activation, classical pathway | P01860, P01024 | 15.4 | 0.068 |
| 0010951 | Negative regulation of endopeptidase activity | Q9UK55, P01024 | 15.4 | 0.0831 |
| 0030036 | Actin cytoskeleton organization | P62328, P22105 | 15.4 | 0.0891 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anitua, E.; Muruzabal, F.; Pino, A.; Prado, R.; Azkargorta, M.; Elortza, F.; Merayo-Lloves, J. Proteomic Characterization of Plasma Rich in Growth Factors and Undiluted Autologous Serum. Int. J. Mol. Sci. 2021, 22, 12176. https://doi.org/10.3390/ijms222212176
Anitua E, Muruzabal F, Pino A, Prado R, Azkargorta M, Elortza F, Merayo-Lloves J. Proteomic Characterization of Plasma Rich in Growth Factors and Undiluted Autologous Serum. International Journal of Molecular Sciences. 2021; 22(22):12176. https://doi.org/10.3390/ijms222212176
Chicago/Turabian StyleAnitua, Eduardo, Francisco Muruzabal, Ander Pino, Roberto Prado, Mikel Azkargorta, Felix Elortza, and Jesús Merayo-Lloves. 2021. "Proteomic Characterization of Plasma Rich in Growth Factors and Undiluted Autologous Serum" International Journal of Molecular Sciences 22, no. 22: 12176. https://doi.org/10.3390/ijms222212176
APA StyleAnitua, E., Muruzabal, F., Pino, A., Prado, R., Azkargorta, M., Elortza, F., & Merayo-Lloves, J. (2021). Proteomic Characterization of Plasma Rich in Growth Factors and Undiluted Autologous Serum. International Journal of Molecular Sciences, 22(22), 12176. https://doi.org/10.3390/ijms222212176






