Platelet-Rich Plasma as an Alternative to Xenogeneic Sera in Cell-Based Therapies: A Need for Standardization
Abstract
1. Introduction
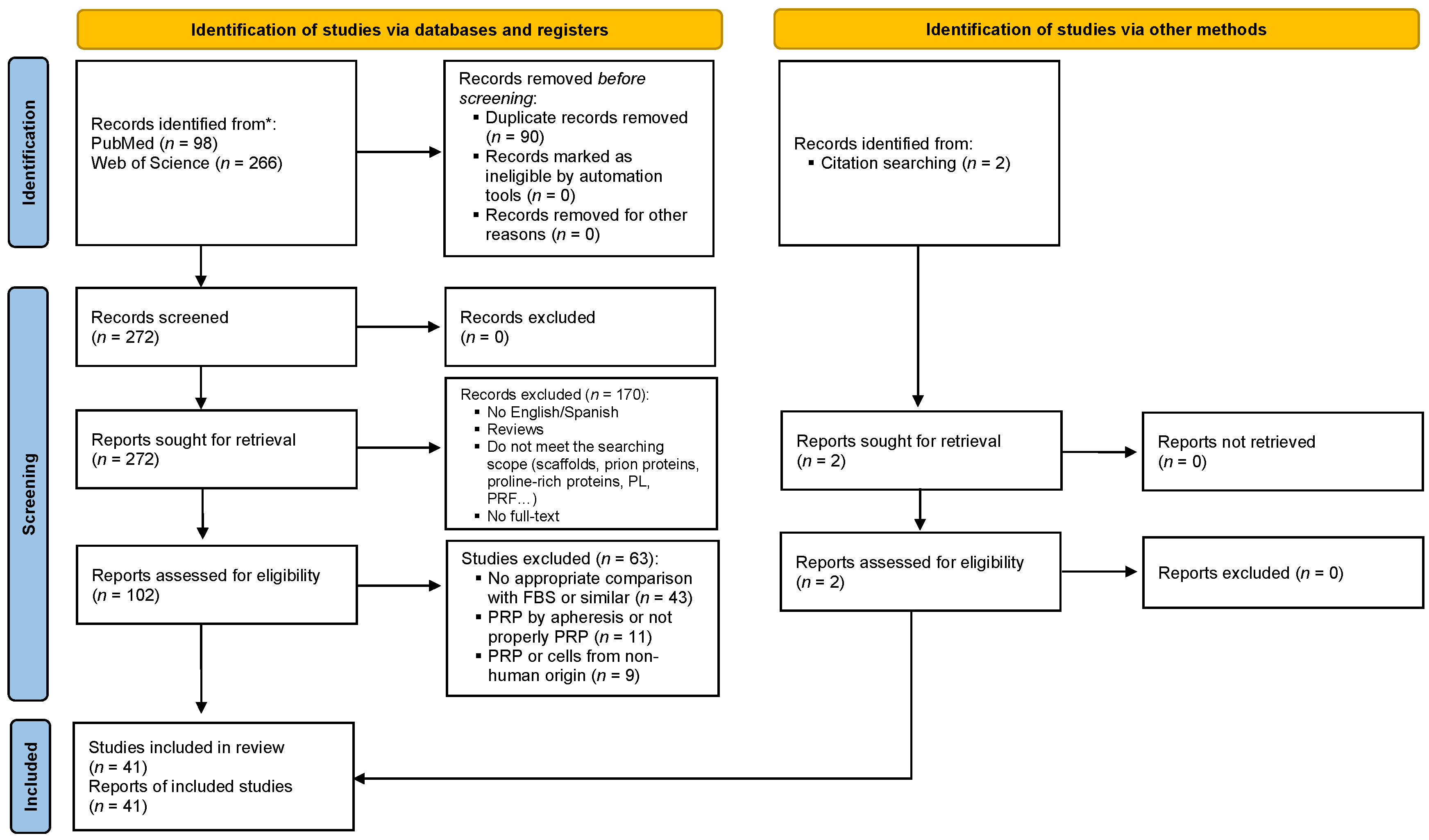
2. Methods
3. Results
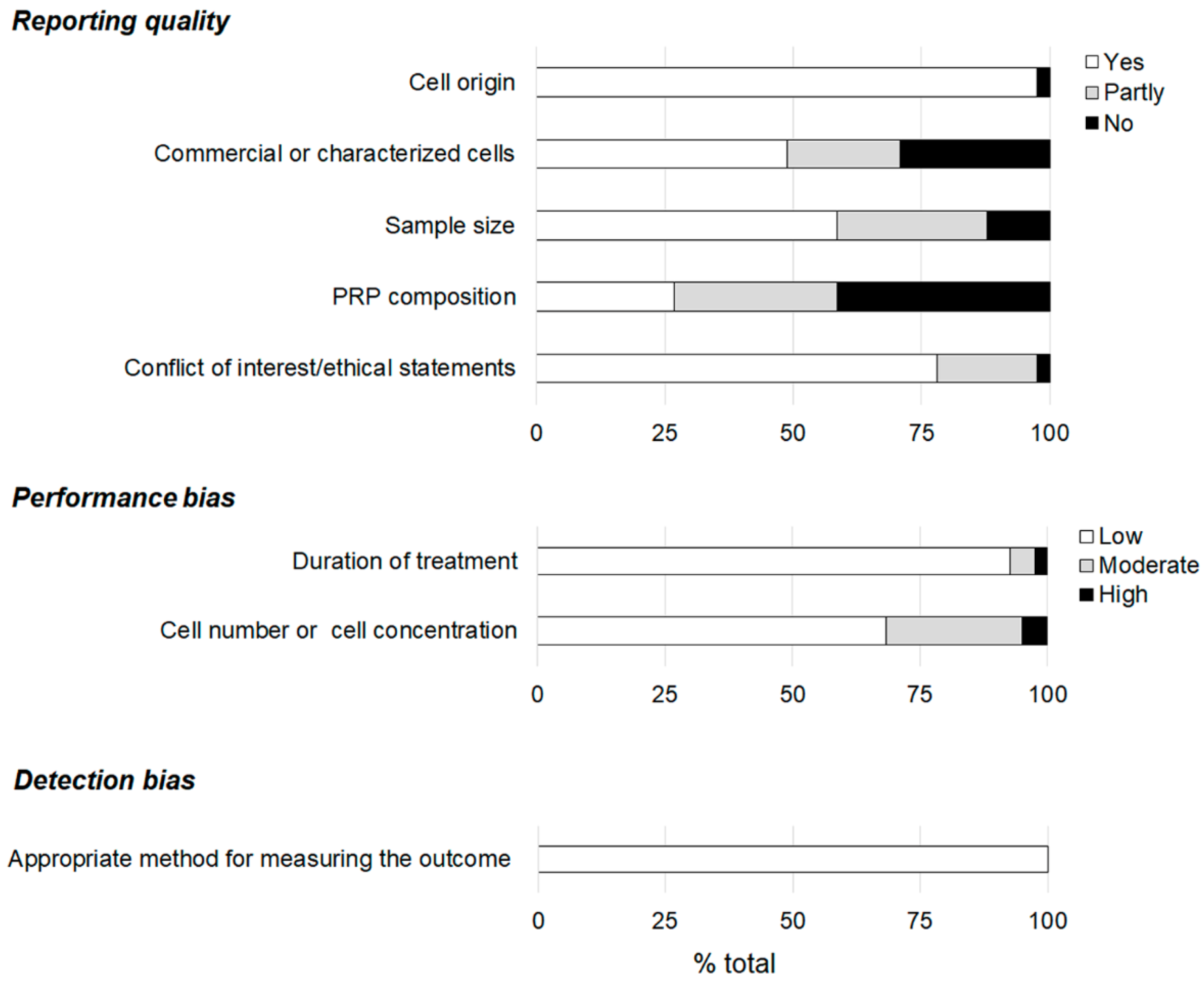
3.1. Reporting Quality and Risk of Bias
3.2. Human Versus Xenogeneic Cullture Medium Supplement
3.2.1. Screening for the Optimal Dose of PRP in the Culture Medium
3.2.2. Adipose Tissue
3.2.3. Oral Tissue
3.2.4. Cartilage Tissue
3.2.5. Bone Marrow Stem Cells (BM-MSCs)
3.2.6. Cells from Skin Tissues
3.2.7. Umbilical Cord Tissue
3.2.8. Miscellaneous
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Alkhraisat, M.H.; Marino, F.T.; Retama, J.R.; Jerez, L.B.; Lopez-Cabarcos, E. Beta-tricalcium phosphate release from brushite cement surface. J. Biomed. Mater. Res. A 2008, 84, 710–717. [Google Scholar] [CrossRef] [PubMed]
- Bunpetch, V.; Wu, H.; Zhang, S.; Ouyang, H. From “Bench to Bedside”: Current Advancement on Large-Scale Production of Mesenchymal Stem Cells. Stem Cells Dev. 2017, 26, 1662–1673. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.; Yazid, M.D.; Yunus, M.H.M.; Chowdhury, S.R.; Lokanathan, Y.; Idrus, R.B.H.; Ng, A.M.H.; Law, J.X. Large-Scale Expansion of Human Mesenchymal Stem Cells. Stem Cells Int. 2020, 2020, 9529465. [Google Scholar] [CrossRef] [PubMed]
- Cimino, M.; Goncalves, R.M.; Barrias, C.C.; Martins, M.C.L. Xeno-Free Strategies for Safe Human Mesenchymal Stem/Stromal Cell Expansion: Supplements and Coatings. Stem Cells Int. 2017, 2017, 6597815. [Google Scholar] [CrossRef]
- Bieback, K.; Fernandez-Munoz, B.; Pati, S.; Schafer, R. Gaps in the knowledge of human platelet lysate as a cell culture supplement for cell therapy: A joint publication from the AABB and the International Society for Cell & Gene Therapy. Cytotherapy 2019, 21, 911–924. [Google Scholar]
- Hemeda, H.; Giebel, B.; Wagner, W. Evaluation of human platelet lysate versus fetal bovine serum for culture of mesenchymal stromal cells. Cytotherapy 2014, 16, 170–180. [Google Scholar] [CrossRef]
- Subbiahanadar Chelladurai, K.; Selvan Christyraj, J.D.; Rajagopalan, K.; Yesudhason, B.V.; Venkatachalam, S.; Mohan, M.; Chellathurai Vasantha, N.; Selvan Christyraj, J.R.S. Alternative to FBS in animal cell culture—An overview and future perspective. Heliyon 2021, 7, e07686. [Google Scholar] [CrossRef]
- Santos, S.; Sigurjonsson, O.E.; Custodio, C.A.; Mano, J. Blood Plasma Derivatives for Tissue Engineering and Regenerative Medicine Therapies. Tissue. Eng. Part B Rev. 2018, 24, 454–462. [Google Scholar] [CrossRef]
- Anitua, E.; Fernandez-de-Retana, S.; Alkhraisat, M.H. Platelet rich plasma in oral and maxillofacial surgery from the perspective of composition. Platelets 2021, 32, 174–182. [Google Scholar] [CrossRef]
- Anitua, E.; Troya, M.; Tierno, R.; Zalduendo, M.; Alkhraisat, M.H. The Effectiveness of Platelet-Rich Plasma as a Carrier of Stem Cells in Tissue Regeneration: A Systematic Review of Pre-Clinical Research. Cells Tissues Organs 2021, 210, 339–350. [Google Scholar] [CrossRef]
- Anitua, E.; Zalduendo, M.; Troya, M.; Tierno, R.; Alkhraisat, M.H. The inclusion of leukocytes into platelet rich plasma reduces scaffold stability and hinders extracellular matrix remodelling. Ann. Anat. 2022, 240, 151853. [Google Scholar] [CrossRef] [PubMed]
- Heldin, C.H.; Westermark, B. Platelet-derived growth factors: A family of isoforms that bind to two distinct receptors. Br. Med. Bull. 1989, 45, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, N.; Lin, W.L.; Kumar, N.M.; Cho, M.I.; Genco, R.J. Mitogenic, chemotactic, and synthetic responses of rat periodontal ligament fibroblastic cells to polypeptide growth factors in vitro. J. Periodontol. 1992, 63, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Betsholtz, C.; Johnsson, A.; Heldin, C.H.; Westermark, B.; Lind, P.; Urdea, M.S.; Eddy, R.; Shows, T.B.; Philpott, K.; Mellor, A.L.; et al. cDNA sequence and chromosomal localization of human platelet-derived growth factor A-chain and its expression in tumour cell lines. Nature 1986, 320, 695–699. [Google Scholar] [CrossRef]
- Anitua, E.; Sanchez, M.; Nurden, A.T.; Nurden, P.; Orive, G.; Andia, I. New insights into and novel applications for platelet-rich fibrin therapies. Trends Biotechnol. 2006, 24, 227–234. [Google Scholar] [CrossRef]
- Anitua, E.; Alkhraisat, M.H.; Orive, G. Perspectives and challenges in regenerative medicine using plasma rich in growth factors. J. Control. Release 2012, 157, 29–38. [Google Scholar] [CrossRef]
- Ornitz, D.M.; Xu, J.; Colvin, J.S.; McEwen, D.G.; MacArthur, C.A.; Coulier, F.; Gao, G.; Goldfarb, M. Receptor specificity of the fibroblast growth factor family. J. Biol. Chem. 1996, 271, 15292–15297. [Google Scholar] [CrossRef]
- Watts, N.B. Clinical utility of biochemical markers of bone remodeling. Clin. Chem. 1999, 45, 1359–1368. [Google Scholar] [CrossRef]
- Gospodarowicz, D.; Neufeld, G.; Schweigerer, L. Fibroblast growth factor: Structural and biological properties. J. Cell. Physiol. Suppl. 1987, 133 (Suppl. S5), 15–26. [Google Scholar] [CrossRef]
- Thrailkill, K.M.; Siddhanti, S.R.; Fowlkes, J.L.; Quarles, L.D. Differentiation of MC3T3-E1 osteoblasts is associated with temporal changes in the expression of IGF-I and IGFBPs. Bone 1995, 17, 307–313. [Google Scholar] [CrossRef]
- Bikle, D.D.; Harris, J.; Halloran, B.P.; Roberts, C.T.; Leroith, D.; Morey-Holton, E. Expression of the genes for insulin-like growth factors and their receptors in bone during skeletal growth. Am. J. Physiol. 1994, 267, E278–E286. [Google Scholar] [CrossRef] [PubMed]
- Meinel, L.; Zoidis, E.; Zapf, J.; Hassa, P.; Hottiger, M.O.; Auer, J.A.; Schneider, R.; Gander, B.; Luginbuehl, V.; Bettschart-Wolfisberger, R.; et al. Localized insulin-like growth factor I delivery to enhance new bone formation. Bone 2003, 33, 660–672. [Google Scholar] [CrossRef]
- Xian, C.J.; Foster, B.K. Repair of injured articular and growth plate cartilage using mesenchymal stem cells and chondrogenic gene therapy. Curr. Stem. Cell Res. Ther. 2006, 1, 213–229. [Google Scholar] [CrossRef] [PubMed]
- Benito, M.; Valverde, A.M.; Lorenzo, M. IGF-I: A mitogen also involved in differentiation processes in mammalian cells. Int. J. Biochem. Cell Biol. 1996, 28, 499–510. [Google Scholar] [CrossRef]
- Bennett, N.T.; Schultz, G.S. Growth factors and wound healing: Part II. Role in normal and chronic wound healing. Am. J. Surg. 1993, 166, 74–81. [Google Scholar] [CrossRef]
- Mosher, D.F.; Furcht, L.T. Fibronectin: Review of its structure and possible functions. J. Investig. Dermatol. 1981, 77, 175–180. [Google Scholar] [CrossRef]
- Anitua, E.; Andia, I.; Ardanza, B.; Nurden, P.; Nurden, A.T. Autologous platelets as a source of proteins for healing and tissue regeneration. Thromb. Haemost. 2004, 91, 4–15. [Google Scholar] [CrossRef]
- Zadeh, G.; Koushan, K.; Baoping, Q.; Shannon, P.; Guha, A. Role of angiopoietin-2 in regulating growth and vascularity of astrocytomas. J. Oncol. 2010, 2010, 659231. [Google Scholar] [CrossRef]
- George, J.N. Platelets. Lancet 2000, 355, 1531–1539. [Google Scholar] [CrossRef]
- Fanburg, B.L.; Lee, S.L. A new role for an old molecule: Serotonin as a mitogen. Am. J. Physiol. 1997, 272, L795–L806. [Google Scholar] [CrossRef]
- Intini, G. The use of platelet-rich plasma in bone reconstruction therapy. Biomaterials 2009, 30, 4956–4966. [Google Scholar] [CrossRef] [PubMed]
- Lawler, J. The functions of thrombospondin-1 and-2. Curr. Opin. Cell Biol. 2000, 12, 634–640. [Google Scholar] [CrossRef]
- Tang, Y.Q.; Yeaman, M.R.; Selsted, M.E. Antimicrobial peptides from human platelets. Infect. Immun. 2002, 70, 6524–6533. [Google Scholar] [CrossRef]
- Bendinelli, P.; Matteucci, E.; Dogliotti, G.; Corsi, M.M.; Banfi, G.; Maroni, P.; Desiderio, M.A. Molecular basis of anti-inflammatory action of platelet-rich plasma on human chondrocytes: Mechanisms of NF-kappaB inhibition via HGF. J. Cell. Physiol. 2010, 225, 757–766. [Google Scholar] [CrossRef] [PubMed]
- Alsberg, E.; Feinstein, E.; Joy, M.P.; Prentiss, M.; Ingber, D.E. Magnetically-guided self-assembly of fibrin matrices with ordered nano-scale structure for tissue engineering. Tissue Eng. 2006, 12, 3247–3256. [Google Scholar] [CrossRef]
- Taus, F.; Meneguzzi, A.; Castelli, M.; Minuz, P. Platelet-Derived Extracellular Vesicles as Target of Antiplatelet Agents. What Is the Evidence? Front. Pharmacol. 2019, 10, 1256. [Google Scholar] [CrossRef]
- Puhm, F.; Boilard, E.; Machlus, K.R. Platelet Extracellular Vesicles: Beyond the Blood. Arterioscler. Thromb. Vasc. Biol. 2021, 41, 87–96. [Google Scholar] [CrossRef]
- Doyle, L.M.; Wang, M.Z. Overview of Extracellular Vesicles, Their Origin, Composition, Purpose, and Methods for Exosome Isolation and Analysis. Cells 2019, 8, 727. [Google Scholar] [CrossRef]
- Jan, A.T.; Rahman, S.; Khan, S.; Tasduq, S.A.; Choi, I. Biology, Pathophysiological Role, and Clinical Implications of Exosomes: A Critical Appraisal. Cells 2019, 8, 99. [Google Scholar] [CrossRef]
- Tao, S.C.; Guo, S.C.; Zhang, C.Q. Platelet-derived Extracellular Vesicles: An Emerging Therapeutic Approach. Int. J. Biol. Sci. 2017, 13, 828–834. [Google Scholar] [CrossRef]
- Anitua, E.; Prado, R.; Orive, G. Safety and efficient ex vivo expansion of stem cells using platelet-rich plasma technology. Ther. Deliv. 2013, 4, 1163–1177. [Google Scholar] [CrossRef] [PubMed]
- Eisinger, F.; Patzelt, J.; Langer, H.F. The Platelet Response to Tissue Injury. Front. Med. 2018, 5, 317. [Google Scholar] [CrossRef] [PubMed]
- Etulain, J. Platelets in wound healing and regenerative medicine. Platelets 2018, 29, 556–568. [Google Scholar] [CrossRef] [PubMed]
- Golebiewska, E.M.; Poole, A.W. Platelet secretion: From haemostasis to wound healing and beyond. Blood Rev. 2015, 29, 153–162. [Google Scholar] [CrossRef]
- Anitua, E.; Tejero, R.; Alkhraisat, M.H.; Orive, G. Platelet-rich plasma to improve the bio-functionality of biomaterials. BioDrugs 2013, 27, 97–111. [Google Scholar] [CrossRef]
- Pinas, L.; Alkhraisat, M.H.; Suarez-Fernandez, R.; Anitua, E. Biomolecules in the treatment of lichen planus refractory to corticosteroid therapy: Clinical and histopathological assessment. Ann. Anat. 2018, 216, 159–163. [Google Scholar] [CrossRef]
- Torres, J.; Tamimi, F.M.; Tresguerres, I.F.; Alkhraisat, M.H.; Khraisat, A.; Lopez-Cabarcos, E.; Blanco, L. Effect of solely applied platelet-rich plasma on osseous regeneration compared to Bio-Oss: A morphometric and densitometric study on rabbit calvaria. Clin. Implant. Dent. Relat. Res. 2008, 10, 106–112. [Google Scholar] [CrossRef]
- Anitua, E.; Alkhraisat, M.H.; Miguel-Sanchez, A.; Orive, G. Surgical correction of horizontal bone defect using the lateral maxillary wall: Outcomes of a retrospective study. J. Oral. Maxillofac. Surg. 2014, 72, 683–693. [Google Scholar] [CrossRef]
- Anitua, E.; Zalduendo, M.; Troya, M. Autologous plasma rich in growth factors technology for isolation and ex vivo expansion of human dental pulp stem cells for clinical translation. Regen. Med. 2019, 14, 97–111. [Google Scholar] [CrossRef]
- Atashi, F.; Jaconi, M.E.E.; Pittet-Cuénod, B.; Modarressi, A. Autologous Platelet-Rich Plasma: A Biological Supplement to Enhance Adipose-Derived Mesenchymal Stem Cell Expansion. Tissue Eng. Part C Methods 2015, 21, 253–262. [Google Scholar] [CrossRef]
- Golbach, L.A.; Portelli, L.A.; Savelkoul, H.F.; Terwel, S.R.; Kuster, N.; de Vries, R.B.; Verburg-van Kemenade, B.M. Calcium homeostasis and low-frequency magnetic and electric field exposure: A systematic review and meta-analysis of in vitro studies. Environ. Int. 2016, 92–93, 695–706. [Google Scholar] [CrossRef] [PubMed]
- Amable, P.R.; Teixeira, M.V.; Carias, R.B.; Granjeiro, J.M.; Borojevic, R. Mesenchymal stromal cell proliferation, gene expression and protein production in human platelet-rich plasma-supplemented media. PLoS ONE 2014, 9, e104662. [Google Scholar] [CrossRef] [PubMed]
- Barlian, A.; Judawisastra, H.; Alfarafisa, N.M.; Wibowo, U.A.; Rosadi, I. Chondrogenic differentiation of adipose-derived mesenchymal stem cells induced by L-ascorbic acid and platelet rich plasma on silk fibroin scaffold. PeerJ 2018, 6, e5809. [Google Scholar] [CrossRef] [PubMed]
- Barlian, A.; Judawisastra, H.; Ridwan, A.; Wahyuni, A.R.; Lingga, M.E. Chondrogenic differentiation of Wharton’s Jelly mesenchymal stem cells on silk spidroin-fibroin mix scaffold supplemented with L-ascorbic acid and platelet rich plasma. Sci. Rep. 2020, 10, 19449. [Google Scholar] [CrossRef]
- Beccia, E.; Carbone, A.; Cecchino, L.R.; Pedicillo, M.C.; Annacontini, L.; Lembo, F.; Di Gioia, S.; Parisi, D.; Angiolillo, A.; Pannone, G.; et al. Adipose Stem Cells and Platelet-Rich Plasma Induce Vascular-like Structures in a Dermal Regeneration Template. Tissue Eng. Part A 2020, 27, 631–641. [Google Scholar] [CrossRef] [PubMed]
- Berndt, S.; Turzi, A.; Pittet-Cuénod, B.; Modarressi, A. Autologous Platelet-Rich Plasma (CuteCell PRP) Safely Boosts In Vitro Human Fibroblast Expansion. Tissue Eng. Part A 2019, 25, 1550–1563. [Google Scholar] [CrossRef]
- Berndt, S.; Turzi, A.; Modarressi, A. Production of Autologous Platelet-Rich Plasma for Boosting In Vitro Human Fibroblast Expansion. J. Vis. Exp. 2021, 168, e60816. [Google Scholar] [CrossRef]
- Bindal, P.; Gnanasegaran, N.; Bindal, U.; Haque, N.; Ramasamy, T.S.; Chai, W.L.; Kasim, N.H.A. Angiogenic effect of platelet-rich concentrates on dental pulp stem cells in inflamed microenvironment. Clin. Oral. Investig. 2019, 23, 3821–3831. [Google Scholar] [CrossRef]
- Brini, A.T.; Ceci, C.; Taschieri, S.; Niada, S.; Lolato, A.; Giannasi, C.; Mortellaro, C.; Del Fabbro, M. Effect of an Activated Platelet Concentrate on Differentiated Cells Involved in Tissue Healing. J. Craniofac. Surg. 2016, 27, 656–661. [Google Scholar] [CrossRef]
- Chieregato, K.; Castegnaro, S.; Madeo, D.; Astori, G.; Pegoraro, M.; Rodeghiero, F. Epidermal growth factor, basic fibroblast growth factor and platelet-derived growth factor-bb can substitute for fetal bovine serum and compete with human platelet-rich plasma in the ex vivo expansion of mesenchymal stromal cells derived from adipose tissue. Cytotherapy 2011, 13, 933–943. [Google Scholar]
- Do Amaral, R.J.; Matsiko, A.; Tomazette, M.R.; Rocha, W.K.; Cordeiro-Spinetti, E.; Levingstone, T.J.; Farina, M.; O’Brien, F.J.; El-Cheikh, M.C.; Balduino, A. Platelet-rich plasma releasate differently stimulates cellular commitment toward the chondrogenic lineage according to concentration. J. Tissue Eng. 2015, 6, 2041731415594127. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, V.K.; de Mulder, E.L.; de Boer, T.; Hannink, G.; van Tienen, T.G.; van Heerde, W.L.; Buma, P. Platelet-rich plasma can replace fetal bovine serum in human meniscus cell cultures. Tissue Eng. Part C Methods 2013, 19, 892–899. [Google Scholar] [CrossRef] [PubMed]
- Hernaez-Moya, R.; Gonzalez, S.; Urkaregi, A.; Pijoan, J.I.; Deng, S.X.; Andollo, N. Expansion of Human Limbal Epithelial Stem/Progenitor Cells Using Different Human Sera: A Multivariate Statistical Analysis. Int. J. Mol. Sci. 2020, 21, 6132. [Google Scholar] [CrossRef]
- Hosseini, L.; Shirazi, A.; Naderi, M.M.; Shams-Esfandabadi, N.; Borjian Boroujeni, S.; Sarvari, A.; Sadeghnia, S.; Behzadi, B.; Akhondi, M.M. Platelet-rich plasma promotes the development of isolated human primordial and primary follicles to the preantral stage. Reprod. Biomed. Online 2017, 35, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Ismail, T.; Lunger, A.; Haumer, A.; Todorov, A.; Menzi, N.; Schweizer, T.; Bieback, K.; Burgin, J.; Schaefer, D.J.; Martin, I.; et al. Platelet-rich plasma and stromal vascular fraction cells for the engineering of axially vascularized osteogenic grafts. J. Tissue Eng. Regen. Med. 2020, 14, 1908–1917. [Google Scholar] [CrossRef] [PubMed]
- Kazemnejad, S.; Najafi, R.; Zarnani, A.H.; Eghtesad, S. Comparative effect of human platelet derivatives on proliferation and osteogenic differentiation of menstrual blood-derived stem cells. Mol. Biotechnol. 2014, 56, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Kinzebach, S.; Dietz, L.; Klüter, H.; Thierse, H.-J.; Bieback, K. Functional and differential proteomic analyses to identify platelet derived factors affecting ex vivo expansion of mesenchymal stromal cells. BMC Cell Biol. 2013, 14, 48. [Google Scholar] [CrossRef]
- Kishimoto, S.; Ishihara, M.; Mori, Y.; Takikawa, M.; Hattori, H.; Nakamura, S.; Sato, T. Effective expansion of human adipose-derived stromal cells and bone marrow-derived mesenchymal stem cells cultured on a fragmin/protamine nanoparticles-coated substratum with human platelet-rich plasma. J. Tissue Eng. Regen. Med. 2013, 7, 955–964. [Google Scholar] [CrossRef]
- Kocaoemer, A.; Kern, S.; Kluter, H.; Bieback, K. Human AB serum and thrombin-activated platelet-rich plasma are suitable alternatives to fetal calf serum for the expansion of mesenchymal stem cells from adipose tissue. Stem. Cells 2007, 25, 1270–1278. [Google Scholar] [CrossRef]
- Lang, S.; Herrmann, M.; Pfeifer, C.; Brockhoff, G.; Zellner, J.; Nerlich, M.; Angele, P.; Prantl, L.; Gehmert, S.; Loibl, M. Leukocyte-reduced platelet-rich plasma stimulates the in vitro proliferation of adipose-tissue derived mesenchymal stem cells depending on PDGF signaling. Clin. Hemorheol. Microcirc. 2017, 67, 183–196. [Google Scholar] [CrossRef]
- Loibl, M.; Lang, S.; Brockhoff, G.; Gueorguiev, B.; Hilber, F.; Worlicek, M.; Baumann, F.; Grechenig, S.; Zellner, J.; Huber, M. The effect of leukocyte-reduced platelet-rich plasma on the proliferation of autologous adipose-tissue derived mesenchymal stem cells 1. Clin. Hemorheol. Microcirc. 2016, 61, 599–614. [Google Scholar] [CrossRef] [PubMed]
- Loibl, M.; Lang, S.; Hanke, A.; Herrmann, M.; Huber, M.; Brockhoff, G.; Klein, S.; Nerlich, M.; Angele, P.; Prantl, L.; et al. Leukocyte-Reduced Platelet-Rich Plasma Alters Protein Expression of Adipose Tissue-Derived Mesenchymal Stem Cells. Plast. Reconstr. Surg. 2016, 138, 397–408. [Google Scholar] [CrossRef] [PubMed]
- Martinez, C.E.; Gomez, R.; Kalergis, A.M.; Smith, P.C. Comparative effect of platelet-rich plasma, platelet-poor plasma, and fetal bovine serum on the proliferative response of periodontal ligament cell subpopulations. Clin. Oral Investig. 2019, 23, 2455–2463. [Google Scholar] [CrossRef]
- McLaughlin, M.; Gagnet, P.; Cunningham, E.; Yeager, R.; D’Amico, M.; Guski, K.; Scarpone, M.; Kuebler, D. Allogeneic platelet releasate preparations derived via a novel rapid thrombin activation process promote rapid growth and increased BMP-2 and BMP-4 expression in human adipose-derived stem cells. Stem Cells Int. 2016, 2016, 7183734. [Google Scholar] [CrossRef] [PubMed]
- Muraglia, A.; Ottonello, C.; Spano, R.; Dozin, B.; Strada, P.; Grandizio, M.; Cancedda, R.; Mastrogiacomo, M. Biological activity of a standardized freeze-dried platelet derivative to be used as cell culture medium supplement. Platelets 2014, 25, 211–220. [Google Scholar] [CrossRef]
- Okada, H.; Takahashi, K.; Ogura, N.; Tomoki, R.; Ito, K.; Kondoh, T. Plasma rich in growth factors stimulates proliferation, migration, and gene expression associated with bone formation in human dental follicle cells. J. Dent. Sci. 2016, 11, 245–252. [Google Scholar] [CrossRef]
- Phetfong, J.; Tawonsawatruk, T.; Seenprachawong, K.; Srisarin, A.; Isarankura-Na-Ayudhya, C.; Supokawej, A. Re-using blood products as an alternative supplement in the optimisation of clinical-grade adipose-derived mesenchymal stem cell culture. Bone Jt. Res. 2017, 6, 414–422. [Google Scholar] [CrossRef]
- Ramos-Torrecillas, J.; de Luna-Bertos, E.; Manzano-Moreno, F.J.; García-Martínez, O.; Ruiz, C. Human Fibroblast–Like Cultures in the Presence of Platelet-Rich Plasma as a Single Growth Factor Source: Clinical Implications. Adv. Skin Wound Care 2014, 27, 114–120. [Google Scholar] [CrossRef]
- Riestra, A.C.; Vazquez, N.; Chacon, M.; Berisa, S.; Sanchez-Avila, R.M.; Orive, G.; Anitua, E.; Meana, A.; Merayo-Lloves, J. Autologous method for ex vivo expansion of human limbal epithelial progenitor cells based on plasma rich in growth factors technology. Ocul. Surf. 2017, 15, 248–256. [Google Scholar] [CrossRef]
- Rosadi, I.; Karina, K.; Rosliana, I.; Sobariah, S.; Afini, I.; Widyastuti, T.; Barlian, A. In vitro study of cartilage tissue engineering using human adipose-derived stem cells induced by platelet-rich plasma and cultured on silk fibroin scaffold. Stem. Cell Res. Ther. 2019, 10, 1–15. [Google Scholar] [CrossRef]
- Simon, M.; Major, B.; Vacz, G.; Kuten, O.; Hornyak, I.; Hinsenkamp, A.; Kardos, D.; Bago, M.; Cseh, D.; Sarkozi, A.; et al. The Effects of Hyperacute Serum on the Elements of the Human Subchondral Bone Marrow Niche. Stem. Cells Int. 2018, 2018, 4854619. [Google Scholar] [CrossRef] [PubMed]
- Suchánek, J.; Suchánková Kleplová, T.; Řeháček, V.; Browne, K.Z.; Soukup, T. Proliferative Capacity and Phenotypical Alteration of Multipotent Ecto-Mesenchymal Stem Cells from Human Exfoliated Deciduous Teeth Cultured in Xenogeneic and Allogeneic Media. Folia Biol. 2016, 62, 1–14. [Google Scholar]
- Suchankova Kleplova, T.; Soukup, T.; Rehacek, V.; Suchanek, J. Human plasma and human platelet-rich plasma as a substitute for fetal calf serum during long-term cultivation of mesenchymal dental pulp stem cells. Acta Med. 2014, 57, 119–126. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sun, X.; Gan, Y.; Tang, T.; Zhang, X.; Dai, K. In vitro proliferation and differentiation of human mesenchymal stem cells cultured in autologous plasma derived from bone marrow. Tissue Eng. Part A 2008, 14, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Talebi, M.; Vatanmakanian, M.; Mirzaei, A.; Barfar, Y.; Hemmatzadeh, M.; Nahayati, M.A.; Velaei, K.; Hoseinzade, A.; Yazdanpanah, B.; Yahyavi, Y.; et al. Platelet-Rich and Platelet-Poor Plasma Might Play Supportive Roles in Cancer Cell Culture: A Replacement for Fetal Bovine Serum? Anti-Cancer Agents Med. Chem. 2021, 21, 2236–2242. [Google Scholar] [CrossRef]
- Tavakolinejad, S.; Khosravi, M.; Mashkani, B.; Ebrahimzadeh Bideskan, A.; Sanjar Mossavi, N.; Parizadeh, M.R.; Hamidi Alamdari, D. The effect of human platelet-rich plasma on adipose-derived stem cell proliferation and osteogenic differentiation. Iran. Biomed. J. 2014, 18, 151–157. [Google Scholar]
- Tchang, L.A.; Pippenger, B.E.; Todorov, A., Jr.; Wolf, F.; Burger, M.G.; Jaquiery, C.; Bieback, K.; Martin, I.; Schaefer, D.J.; Scherberich, A. Pooled thrombin-activated platelet-rich plasma: A substitute for fetal bovine serum in the engineering of osteogenic/vasculogenic grafts. J. Tissue Eng. Regen. Med. 2017, 11, 1542–1552. [Google Scholar] [CrossRef]
- Van Pham, P.; Dang, L.T.-T.; Truong, N.H.; Phan, N.K. Can Activated Platelet Rich Plasma Combined with Adipose-Derived Stem Cells Be Used to Treat Skin Wrinkles?: A Mechanism Study. In Bioinformatics: Concepts, Methodologies, Tools, and Applications; IGI Global: Hershey, PA, USA, 2013; pp. 920–936. [Google Scholar]
- Vogel, J.P.; Szalay, K.; Geiger, F.; Kramer, M.; Richter, W.; Kasten, P. Platelet-rich plasma improves expansion of human mesenchymal stem cells and retains differentiation capacity and in vivo bone formation in calcium phosphate ceramics. Platelets 2006, 17, 462–469. [Google Scholar] [CrossRef]
- Xian, L.J.; Chowdhury, S.R.; Bin Saim, A.; Idrus, R.B. Concentration-dependent effect of platelet-rich plasma on keratinocyte and fibroblast wound healing. Cytotherapy 2015, 17, 293–300. [Google Scholar] [CrossRef]
- Muraglia, A.; Nguyen, V.T.; Nardini, M.; Mogni, M.; Coviello, D.; Dozin, B.; Strada, P.; Baldelli, I.; Formica, M.; Cancedda, R.; et al. Culture Medium Supplements Derived from Human Platelet and Plasma: Cell Commitment and Proliferation Support. Front. Bioeng. Biotechnol. 2017, 5, 66. [Google Scholar] [CrossRef]
- Chisini, L.A.; Conde, M.C.M.; Grazioli, G.; Martin, A.S.S.; Carvalho, R.V.; Nor, J.E.; Demarco, F.F. Venous Blood Derivatives as FBS-Substitutes for Mesenchymal Stem Cells: A Systematic Scoping Review. Braz. Dent. J. 2017, 28, 657–668. [Google Scholar] [CrossRef] [PubMed]
- Bieback, K. Platelet lysate as replacement for fetal bovine serum in mesenchymal stromal cell cultures. Transfus. Med. Hemother. 2013, 40, 326–335. [Google Scholar] [CrossRef] [PubMed]
- Guiotto, M.; Raffoul, W.; Hart, A.M.; Riehle, M.O.; di Summa, P.G. Human platelet lysate to substitute fetal bovine serum in hMSC expansion for translational applications: A systematic review. J. Transl. Med. 2020, 18, 351. [Google Scholar] [CrossRef] [PubMed]
- Kon, E.; Di Matteo, B.; Delgado, D.; Cole, B.J.; Dorotei, A.; Dragoo, J.L.; Filardo, G.; Fortier, L.A.; Giuffrida, A.; Jo, C.H.; et al. Platelet-rich plasma for the treatment of knee osteoarthritis: An expert opinion and proposal for a novel classification and coding system. Expert Opin. Biol. Ther. 2020, 20, 1447–1460. [Google Scholar] [CrossRef]
- Lana, J.; Purita, J.; Paulus, C.; Huber, S.C.; Rodrigues, B.L.; Rodrigues, A.A.; Santana, M.H.; Madureira, J.L., Jr.; Malheiros Luzo, A.C.; Belangero, W.D.; et al. Contributions for classification of platelet rich plasma—Proposal of a new classification: MARSPILL. Regen. Med. 2017, 12, 565–574. [Google Scholar] [CrossRef]
- Magalon, J.; Chateau, A.L.; Bertrand, B.; Louis, M.L.; Silvestre, A.; Giraudo, L.; Veran, J.; Sabatier, F. DEPA classification: A proposal for standardising PRP use and a retrospective application of available devices. BMJ Open Sport Exerc. Med. 2016, 2, e000060. [Google Scholar] [CrossRef]
- Mautner, K.; Malanga, G.A.; Smith, J.; Shiple, B.; Ibrahim, V.; Sampson, S.; Bowen, J.E. A call for a standard classification system for future biologic research: The rationale for new PRP nomenclature. PM&R 2015, 7, S53–S59. [Google Scholar]
- Mishra, A.; Harmon, K.; Woodall, J.; Vieira, A. Sports medicine applications of platelet rich plasma. Curr. Pharm. Biotechnol. 2012, 13, 1185–1195. [Google Scholar] [CrossRef]
- Bowen, R.A.; Remaley, A.T. Interferences from blood collection tube components on clinical chemistry assays. Biochem. Med. 2014, 24, 31–44. [Google Scholar] [CrossRef]
- Cedrone, E.; Neun, B.W.; Rodriguez, J.; Vermilya, A.; Clogston, J.D.; McNeil, S.E.; Barenholz, Y.; Szebeni, J.; Dobrovolskaia, M.A. Anticoagulants influence the performance of in vitro assays intended for characterization of nanotechnology-based formulations. Molecules 2018, 23, 12. [Google Scholar] [CrossRef]
- Anitua, E.; Prado, R.; Troya, M.; Zalduendo, M.; de la Fuente, M.; Pino, A.; Muruzabal, F.; Orive, G. Implementation of a more physiological plasma rich in growth factor (PRGF) protocol: Anticoagulant removal and reduction in activator concentration. Platelets 2016, 27, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Germanovich, K.; Femia, E.A.; Cheng, C.Y.; Dovlatova, N.; Cattaneo, M. Effects of pH and concentration of sodium citrate anticoagulant on platelet aggregation measured by light transmission aggregometry induced by adenosine diphosphate. Platelets 2018, 29, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Hemeda, H.; Kalz, J.; Walenda, G.; Lohmann, M.; Wagner, W. Heparin concentration is critical for cell culture with human platelet lysate. Cytotherapy 2013, 15, 1174–1181. [Google Scholar] [CrossRef] [PubMed]
- Do Amaral, R.J.; da Silva, N.P.; Haddad, N.F.; Lopes, L.S.; Ferreira, F.D.; Filho, R.B.; Cappelletti, P.A.; de Mello, W.; Cordeiro-Spinetti, E.; Balduino, A. Platelet-Rich Plasma Obtained with Different Anticoagulants and Their Effect on Platelet Numbers and Mesenchymal Stromal Cells Behavior In Vitro. Stem. Cells Int. 2016, 2016, 7414036. [Google Scholar] [CrossRef] [PubMed]
- Agostini, F.; Polesel, J.; Battiston, M.; Lombardi, E.; Zanolin, S.; Da Ponte, A.; Astori, G.; Durante, C.; Mazzucato, M. Standardization of platelet releasate products for clinical applications in cell therapy: A mathematical approach. J. Transl. Med. 2017, 15, 107. [Google Scholar] [CrossRef]
- Henschler, R.; Gabriel, C.; Schallmoser, K.; Burnouf, T.; Koh, M.B.C. Human platelet lysate current standards and future developments. Transfusion 2019, 59, 1407–1413. [Google Scholar] [CrossRef]
- Oeller, M.; Laner-Plamberger, S.; Krisch, L.; Rohde, E.; Strunk, D.; Schallmoser, K. Human Platelet Lysate for Good Manufacturing Practice-Compliant Cell Production. Int. J. Mol. Sci. 2021, 22, 5178. [Google Scholar] [CrossRef]
| Reference | Phenotype | Comparison Groups | Assays | Conclusion |
|---|---|---|---|---|
| Amable et al., 2014 [52] | BM-MSC, AT-MSCs and WJ-MSC | 10% FBS vs. 1%, 2.5%, 5%, 10%, 20%, 30%, 40%, 50% PRP | Proliferation, trilineage differentiation, gene expression, and cytokine, growth factor and extracellular matrix quantification | ✓ |
| Anitua et al., 2019 [49] | hDPSCs | 10% FBS vs. 10% PRGF | Isolation, migration, proliferation, osteogenic and adipogenic differentiation, senescence and cryopreservation | ✓ |
| Atashi et al., 2015 [50] | AT-MSCs | 10% FBS vs. 1%, 5%, 10%, 20%, 40%, 60% of either nPRP or tPRP | Cell viability, cell proliferation, cell phenotype, trilineage differentiation, chromosome stability cytogenetic analysis | ✓ |
| Barlian et al., 2018 [53] | ADSCs | 10% FBS (control) vs. 5%, 10%, 20% PRP | Chondrogenic differentiation | ✓ |
| Barlian et al., 2020 [54] | WJ-MSCs | 10% FBS vs. 10% PRP | Chondrogenic differentiation (collagen type II, GAG accumulation). | ✓ |
| Beccia et al., 2021 [55] | ASCs | 10% FBS vs. 2% PRP | Morphology and proliferation | ✕ |
| Berndt et al., 2019 [56] | NHDF | 10% FBS vs. 1%, 5%, 10%, 20%, 30%, 40%, 50% PRP | Cell proliferation, cell cycle analysis, cell morphology, alpha-SMA and vimentin expression, metabolic activity assessment, cell adhesion, wound healing, genomic stability | ✓ |
| Berndt et al., 2021 [57] | NHDF | 10% FBS vs. 1%, 5%, 10%, 20%, 30%, 40%, 50% PRP | NHDF proliferation and activation. | ✓ |
| Bindal et al., 2019 [58] | hiDPSCs | 10% FBS vs. 10%, 20% PRP | Viability, proliferation, proangiogenic gene expression, proangiogenic growth factor release | ✓ |
| Brini et al., 2016 [59] | hDFs / hObs | 10% FBS vs. 5% PRGF (cell proliferation and viability) 10% FBS vs. 2.5% PRGF (osteoblast differentiation) | Cell proliferation and viability, osteogenic differentiation | ✓ |
| Chieregato et al., 2011 [60] | ADSCs | 10% FBS vs. 10% hPRP | Morphology, CFU, proliferation and MEK-1/2 role, multiple differentiation capacity, immunophenotype | ✓ |
| do Amaral et al., 2015 [61] | NCCs and MSCs from bone marrow | 10% FBS vs. 1%, 2.5%, 5%, 10% PRPr | Cell proliferation, GAGs, pellet area measurement, chondrogenic genes quantification, sGAG quantification | ✓ |
| Gonzales et al., 2013 [62] | MFCs | 10% FBS vs. 5%, 10%, 20% PRP | DNA quantification, gene expression (col I, col II and aggrecan), histology (H&E) | ✓ |
| Hernáez-Moya et al., 2020 [63] | LESCs | 5% FBS vs. 10% s-PRGF | Cell growth, cell size and gene expression of stem/progenitor limbal cells markers and K12 marker for corneal epithelial differentiation | ✓ |
| Hosseini et al., 2017 [64] | Human ovarian cells | 10% FBS vs. 10% PRP | Follicle growth and viability assessment. Histological analysis. | ✓ |
| Ismail et al., 2020 [65] | SVF cells | 10% FBS vs. 10% tPRP | Cell number and clonogenicity | ✓ |
| Kazemneja et al., 2014 [66] | MenSCs | 10% FBS vs. PGS vs. PRP vs. HPR (proliferation assays) 15% FBS vs. PGS vs. PRP vs. HPR (differentiation assays) | Cell proliferation and osteogenic differentiation (Alizarin Red, ALP activity, OCN level) | ✓ |
| Kinzebach et al., 2013 [67] | LA-MSC and BM-MSC | 2.5%, 5%, 7.5%, 10% FBS vs. 2.5%, 5%, 7.5%, 10% tPRP | Expansion of MSCs and differential proteomics, proliferation and stimulation assays. Growth factors quantification and cytokine receptors expression. Adipo- and osteogenic differentiation. | ✓ |
| Kishimoto et al., 2013 [68] | ASCs and BMSCs | 0.125%, 0.25%, 0.5%, 1%, 2%, 4% FBS vs. 0.125%, 0.25%, 0.5%, 1%, 2%, 4% PRP (optimal concentration) 2%, 10% FBS vs. 0.5%PRP (proliferation) | Determination of the supplement optimal concentration, proliferation | ✓ |
| Kokaoemer et al., 2007 [69] | AT-MSCs | 10% FCS vs. 10% tPRP | Morphology, adhesion, CFU, cumulative population doubling rates, adipogenic and osteogenic differentiation, immunophenotype | ✓ |
| Lang et al., 2017 [70] | ASCs | 20% FCS vs. 10%, 20% PRP | Cell cycle analysis, expression of PDGF receptorβ, c-MYC, and MEK-1, PDGF receptor β Inhibition | ✓ |
| Loibl et al., 2016 [71] | ASCs | 20% FCS vs. 10%, 20% ACS | Cell cycle analysis | ✓ |
| Loibl et al., 2016 [72] | ASCs | 20% FCS vs. 10%, 20% ACS | Cell cycle analysis and proteomic profile | ✓ |
| Martínez et al., 2019 [73] | PDL cells | 10% FBS vs. 2.5%, 5%,10% PRP | Cell proliferation and clonogenic proliferation | ✓ |
| McLaughlin et al., 2016 [74] | ASCs | 10% FBS vs. 10% tPR | Morphology, growth rate, gene expression (BMP-2, BMP-4, VEGF, TGF-beta, PDGF-B and FGF-2) | ✓ |
| Muraglia et al., 2014 [75] | MSCs from bone marrow/human skin fibroblasts/hObs/human articular chondrocyte | 10% FCS vs. 5% PRP | Clonogenic assay (MSCs) and cell viability of primary cultures | ✓ |
| Okada et al., 2016 [76] | hDFCs | 10% FBS vs. 1%, 5%, 10%, 20% PRGF | Osteogenic differentiation, cell proliferation, cell migration and osteogenic gene expression | ✓ |
| Phetfong et al., 2017 [77] | ADMSCs | FBS vs. Hplasma | Cell morphology, proliferation, CFU, immnuphenotyping, osteogenic and adipogenic differentiation, senescence | ✓ |
| Ramos-Torrecillas et al., 2014 [78] | Human gingival fibroblasts | 10% FBS vs. 10% PRP | Cell growth rate, cell morphology and antigenic expression | ✓ |
| Riestra et al., 2017 [79] | LESCs | 10% FBS vs. 10% PRGF | Measurement of the extent of outgrowths of cultures of LEPCs, number of cells and colony forming efficiency. Morphological analysis and immunocytochemistry and quantification of p63-α HLCE. | ✓ |
| Rosadi et al., 2019 [80] | ADSCs | 10% FBS vs. 10%PRP | Cell proliferation, differentiation assays (GAG levels and mineralization, secretion of TGF-β1, expression of specific stem cell surface protein markers, gene expression) | ✓ |
| Simon et al., 2018 [81] | MSCs | 10% FCS vs. 10% FCS+bFGF vs. 10% PRP | Cell proliferation, lineage-specific markers, gene expression | ✓ |
| Suchánek et al., 2016 [82] | SHED | 2% FCS + GFs (FCS+) vs. 2% PRP + GFs (PRP+) | Proliferative capacity, cumulative population doubling, morphology, viability, expressing cluster of differentiation | ✓ |
| Suchánková et al., 2014 [83] | hDPSC | 2% FCS vs. 2% PRP | Proliferation, population doublings, viability, phenotypic analysis | ✓ |
| Sun, Xiaojiang et al., 2008 [84] | MSCs from bone marrow | 10% FBS vs. 10% APM | Cell morphology, proliferation, surface markers, growth cycle, and apoptosis; osteogenic differentiation; number and area of ALP+CFU-Fs; adipogenic differentiation. | ✓ |
| Talebi et al., 2021 [85] | CCRF-CEM | 10% FBS vs. 2%, 5%, 10%, 15% PRP | Cell viability and YKL-40 mRNA and protein levels. | ✓ |
| Tavakolinejad et al., 2014 [86] | ADSCs | 10% FBS vs. 10%, 15% hPRP | Proliferation and osteogenesis | ✓ |
| Tchang et al., 2017 [87] | SVF cells | 10% FBS vs. 10% tPRP | 2D mineralization assay and 3D angiogenesis | ✓ |
| Van Pham et al., 2014 [88] | UCB-MSCs | 10% FBS vs. 2, 5, 7, 10% PRP | Number of adherent cells and their expansion, percentage of successfully isolated cells in the primary culture, surface marker expression, in vitro differentiation potential following expansion | ✓ |
| Vogel et al., 2006 [89] | BM-MSC | 2% FCS vs. 3% PRP | Growth rate; osteogenic, adipogenic and chondrogenic differentiation capacity | ✓ |
| Xian et al., 2015 [90] | Human keratinocytes and fibroblasts from skin | 5% FBS vs. 10%, 20% PRP | Extracellular matrix gene expression, proliferation, migratory property, soluble factors secretion | ✓ |
| Reference | Type of Anticoagulant | Comparison Groups | Number of Centrifugations | PRP Acquisition | Activation Method |
|---|---|---|---|---|---|
| Amable et al., 2014 [52] | ACD | 10% FBS vs. 1%, 2.5%, 5%, 10%, 20%, 30%, 40%, 50% PRP | Two | Platelet-containing plasma above the buffy coat. Platelets concentrated and suspended in a smaller volume of plasma. | Calcium chloride |
| Anitua et al., 2019 [49] | Sodium citrate | 10% FBS vs. 10% PRGF | One | PRGF: plasma column just above the buffy coat. | Calcium chloride |
| Atashi et al., 2015 [50] | Sodium citrate | 10% FBS vs. 1%, 5%, 10%, 20%, 40%, 60% of either nPRP or tPRP | One | Regenkit: plasma containing platelets above the white and red blood cells. | nPRP: non-activated |
| W/o | 10% FBS (control) vs. 5%, 10%, 20% PRP | Regenkit: plasma over the red and most of the white blood cells formed a clot. The serum extracted from the clot was added 1:10 to PRP to activate the platelets and obtain tPRP. | tPRP: thrombin | ||
| Barlian et al., 2018 [53] | NA | 10% FBS vs. 10% PRP | NA | NA | NA |
| Barlian et al., 2020 [54] | NA | 10% FBS vs. 2% PRP | NA | NA | NA |
| Beccia et al., 2021 [55] | Buffered solution of sodium citrate, theophylline, adenosine and dipyridamole | 10% FBS vs. 1%, 5%, 10%, 20%, 30%, 40%, 50% PRP | Two | Plasma portion separated from cells. | Non-activated |
| Berndt et al., 2019 [56] | NA | 10% FBS vs. 1%, 5%, 10%, 20%, 30%, 40%, 50% PRP | One | Plasma containing platelets, remained above the gel layer, was homogenized by turning the tube five times. | Non-activated |
| Berndt et al., 2021 [57] | NA | 10% FBS vs. 10%, 20% PRP | One | The red and white blood cells are trapped under the gel, and platelets settled on the surface of the gel are resuspended by inverting the tube. | Non-activated |
| Bindal et al., 2019 [58] | NA | 10% FBS vs. 5% PRGF (cell proliferation and viability) 10% FBS vs. 2.5% PRGF (osteoblasts differentiation) | NA | NA | Freezing |
| Brini et al., 2016 [59] | Sodium citrate | 10% FBS vs. 10% hPRP | One | PRGF, the 2 mL plasma just above the buffy coat containing the highest platelets concentration, was collected. | Calcium chloride |
| Chieregato et al., 2011 [60] | Heparin | 10% FBS vs. 1%, 2.5%, 5%, 10% PRPr | One | NA | Freezing and sonication |
| Do Amaral et al., 2015 [61] | Citrate sodium | 10% FBS vs. 5%, 10%, 20% PRP | Two | PRP from protocol 2: The upper plasma fraction without leukocyte and red cells was centrifuged. Platelets pellet was resuspended with the supernatant (platelet-poor plasma). | Calcium chloride |
| Gonzales et al., 2013 [62] | Hirudin | 5% FBS vs. 10% s-PRGF | One | The upper phase containing PRP. | Thrombin?? |
| Hernáez-Moya et al., 2020 [63] | Sodium citrate | 10% FBS vs. 10% PRP | One | The complete supernatant fraction without red and white blood cells. | Calcium chloride |
| Hosseini et al., 2017 [64] | Acid citrate solution | 10% FBS vs. 10% tPRP | Two | The top and middle layers after the first centrifugation were centrifuged again and the remaining 0.5 mL of plasma containing precipitated platelets was mixed evenly and considered to be PRP. | Thrombin |
| Ismail et al., 2020 [65] | NA | 10% FBS vs. 10% PGS vs. 10% PRP vs. 10% HPR (proliferation assays) 15% FBS vs. 15% PGS vs. 15% PRP vs. 15% HPR (differentiation assays) | NA | Two platelet concentrates from buffy coats extracted from whole-blood donations of four AB blood group-typed donors were pooled and suspended in the plasma of one AB donor. | Thrombin |
| Kazemnejad et al., 2014 [66] | NA | 2.5%, 5%, 7.5%, 10% FBS vs. 2.5%, 5%, 7.5%, 10% tPRP | NA | NA | PRP: freezing PGS: thrombinHPR: thrombin |
| Kinzebach et al., 2013 [67] | NA | 0.125%, 0.25%, 0.5%, 1%, 2%, 4% FBS vs. 0.125%, 0.25%, 0.5%, 1%, 2%, 4% PRP (optimal concentration) 2%, 10% FBS vs. 0.5% PRP (proliferation) | NA | Buffy coat-derived pooled platelet concentrates. | Thrombin |
| Kishimoto et al., 2013 [68] | Sodium citrate | 10% FCS vs. 10% tPRP | Two | The upper 1 cm of the erythrocyte layer was collected as the PRP layer. | Freezing |
| Kokaoemer et al., 2007 [69] | NA | 20% FCS vs. 10%, 20% PRP | NA | Pooled platelet concentrate out of buffy coats. | Thrombin |
| Lang et al., 2017 [70] | W/o | 20% FCS vs. 10%, 20% ACS | One | Arthrex: a plasma layer appeared on the top and the red/white blood cell layer was apparent at the bottom. The plasma containing the platelets was isolated. | Freezing |
| Loibl et al., 2016 [72] | NA | 20% FCS vs. 10%, 20% ACS | One | Arthrex | NA |
| Loibl et al., 2016 [71] | NA | 10% FBS vs. 2.5%, 5%, 10% PRP | One | Arthrex: a plasma layer appeared on the top and the red/white blood cell layer was apparent on the bottom. The plasma, containing the platelets, was isolated. | Freezing |
| Martínez et al., 2019 [73] | NA | 10% FBS vs. 10% tPR | One | GPS III | Calcium chloride and thrombin |
| McLaughlin et al., 2016 [74] | NA | 10% FCS vs. 5% PRP | NA | Harvest SmartPrep System | Thrombin |
| Muraglia et al., 2014 [75] | NA | 10% FBS vs. 1%, 5%, 10%, 20% PRGF | Multiple | Buffy coat samples. The platelet pellet was brought to a final volume with PPP to obtain a concentration of 10 × 106 platelets/µL. | Freezing |
| Okada et al., 2016 [76] | Sodium citrate | 10% FBS vs. 10% Hplasma | One | PRGF: the plasma fraction (1 mL over the buffy coat) was collected as F2. | Calcium chloride |
| Pham et al., 2013 [88] | NA | 10% FBS vs. 10% PRP | Two | After the second centrifugation, the platelet pellet was resuspended with the third of the plasma volume. | Calcium chloride |
| Phetfong et al., 2017 [77] | NA | 10% FBS vs. 10% PRGF | NA | It was prepared from FFP. | Calcium chloride |
| Ramos-Torrecillas et al., 2014 [78] | Lithium heparin | 10% FBS vs. 10% PRP | Two | The whole plasma portion and top layer of red blood cells. After a new centrifugation, the upper portion of the plasma was discarded, and the remainder was the PRP. | NA |
| Riestra et al., 2017 [79] | Sodium citrate | 10% FCS vs. 10% FCS + bFGF vs. 10% PRP | One | Liquid PRGF: the lower 2 mL of the plasma column (F2) was discarded. The rest of the plasma column (F1) was drawn off avoiding the buffy coat. | Calcium chloride |
| Rosadi et al., 2019 [80] | NA | 2% FCS + GFs (FCS+) vs. 2% PRP + GFs (PRP+) | NA | NA | NA |
| Simon et al., 2018 [81] | NA | 2% FCS vs. 2% PRP | Two | The platelet-rich layer above the buffy coat was centrifuged and the resulting platelet pellet was resuspended to obtain PRP. | Non-activated |
| Suchánek et al., 2016 [82] | NA | 10% FBS vs. 10% APM | NA | NA | NA |
| Suchánková et al., 2014 [83] | NA | 10% FBS vs. 2%, 5%, 10%, 15% PRP | NA | NA | NA |
| Sun, Xiaojiang et al., 2008 [84] | NA | 10% FBS vs. 10%, 15% hPRP | Two | Bone marrow was concentrated through density gradient centrifugation. After removing the remaining red blood cells and fatty droplets by centrifugation, the APM was collected. | NA |
| Talebi et al., 2021 [85] | Sodium citrate | 10% FBS vs. 10% tPRP | Two | Supernatant from the first centrifugation, including PRP, was again centrifuged and the lower half part, which contains a large number of platelets in the form of the platelet plug, was considered as PRP. | Shaking at 22 °C for nine days. |
| Tavakolinejad et al., 2014 [86] | NA | 10% FBS vs. 2%, 5%, 7%, 10% PRP | Two | The platelets were precipitated and the plasma was removed; then, the platelets were resuspended in 50 mL plasma | Freezing |
| Tchang et al., 2017 [87] | NA | 2% FCS vs. 3% PRP | NA | Platelet concentrates from buffy coats extracted from whole blood. | Thrombin |
| Vogel et al., 2006 [89] | Citrate–phosphate–dextrose | 5% FBS vs. 10%, 20% PRP | One | Allogenic leukocyte-depleted PRP was obtained from a blood bank. Pooled buffy coats were centrifuged and leukocyte-depleted by filtration. | NA |
| Xian et al., 2015 [90] | Sodium citrate | 5% FBS vs. 10%, 20% PRP | Two | Harvested PRP without red cells was concentrated by discarding 2 mL of plasma after the second centrifugation. | Non-activated |
| Item | Description |
|---|---|
| Blood donor features | Age, sex and systemic health status (ASA) |
| Medical device for blood extraction | Type of medical devise (bags, vacutainer, syringe) and commercial information. Type of additives and their concentration Blood to anticoagulant ratio |
| Blood characteristics | Pooled or individually processed Hematocrit and concentration of platelets, leukocytes and red blood cells (RBCs) Blood group (ABO and Rh systems) |
| Blood processing for PRP preparation | Procedure: centrifugation, aphaeresis, microfluidic system Equipment used: commercial information Processing parameters: number of cycles, centrifugation force and time |
| PRP definition | Which specific part of the fractioned blood (plasma and/or buffy coat) is considered PRP? |
| PRP hallmark | Pooled or individually characterized Volume Concentration of platelets, leukocytes and RBCs Post-processing of PRP (lyophilization and freezing) Use of additives (type and concentration) Storage conditions |
| Activation | Yes/no Method of activation: calcium ions, thrombin, light, agitation Concentration of activator solution and ratio to PRP |
| Nomenclature of PRP formulation | Non-activated PRP Activated liquid PRP PRP serum PRP fibrin |
| Storage conditions | Lyophilized or not Temperature |
| Key biomolecules content | Biomolecules identification and kits used for quantifications |
| Origin of PRP relative to the cells | Autologous, allogenic or xenogenic |
| Pathogen detection | Yes/No; if yes, specify microorganism and the assay used |
| Microbial inactivation | Yes/No; if yes, specify the procedure |
| Dose of PRP | Percentage to the volume of cell culture medium |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anitua, E.; Zalduendo, M.; Troya, M.; Alkhraisat, M.H.; Blanco-Antona, L.A. Platelet-Rich Plasma as an Alternative to Xenogeneic Sera in Cell-Based Therapies: A Need for Standardization. Int. J. Mol. Sci. 2022, 23, 6552. https://doi.org/10.3390/ijms23126552
Anitua E, Zalduendo M, Troya M, Alkhraisat MH, Blanco-Antona LA. Platelet-Rich Plasma as an Alternative to Xenogeneic Sera in Cell-Based Therapies: A Need for Standardization. International Journal of Molecular Sciences. 2022; 23(12):6552. https://doi.org/10.3390/ijms23126552
Chicago/Turabian StyleAnitua, Eduardo, Mar Zalduendo, Maria Troya, Mohammad H. Alkhraisat, and Leticia Alejandra Blanco-Antona. 2022. "Platelet-Rich Plasma as an Alternative to Xenogeneic Sera in Cell-Based Therapies: A Need for Standardization" International Journal of Molecular Sciences 23, no. 12: 6552. https://doi.org/10.3390/ijms23126552
APA StyleAnitua, E., Zalduendo, M., Troya, M., Alkhraisat, M. H., & Blanco-Antona, L. A. (2022). Platelet-Rich Plasma as an Alternative to Xenogeneic Sera in Cell-Based Therapies: A Need for Standardization. International Journal of Molecular Sciences, 23(12), 6552. https://doi.org/10.3390/ijms23126552







