Optimized Synthesis of New Thiosemicarbazide Derivatives with Tuberculostatic Activity
Abstract
:1. Introduction
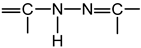 ; indeed, acyl-thiosemicarbazides in their thiol form
; indeed, acyl-thiosemicarbazides in their thiol form 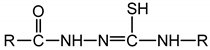 contain this group.
contain this group.2. Results and Discussion
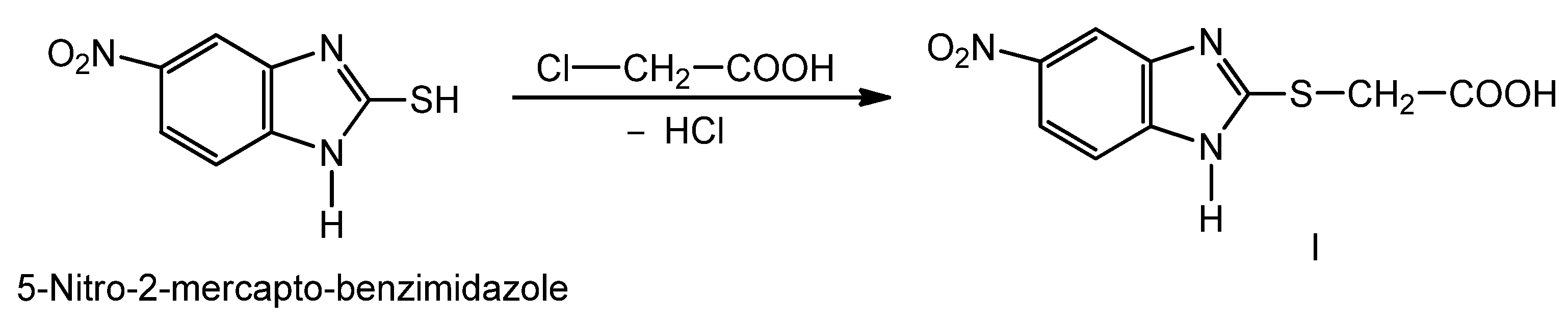
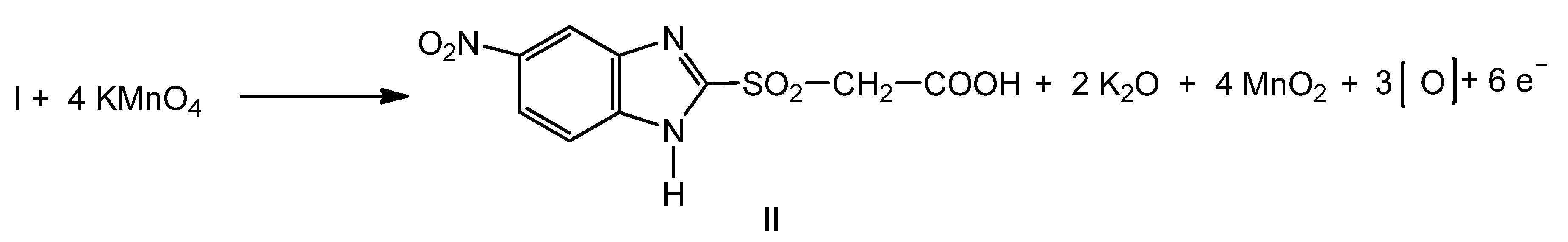
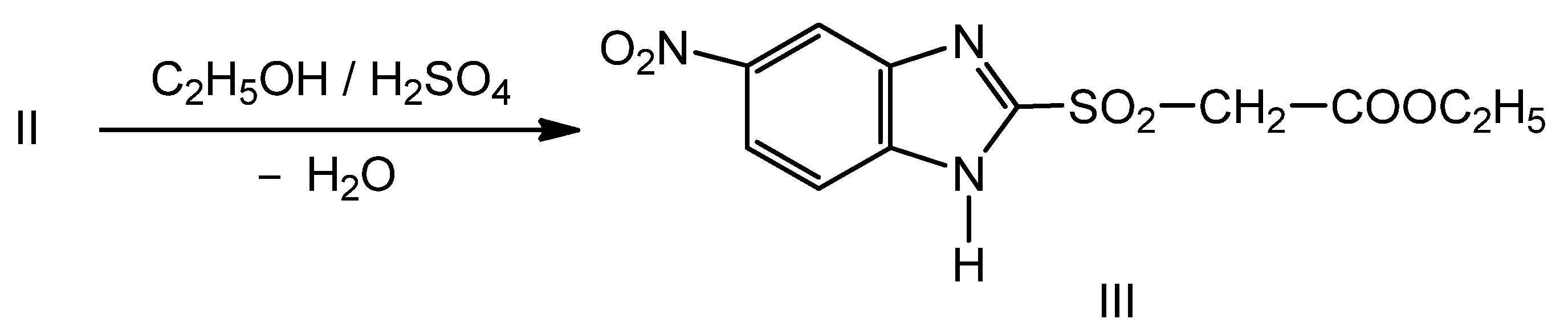
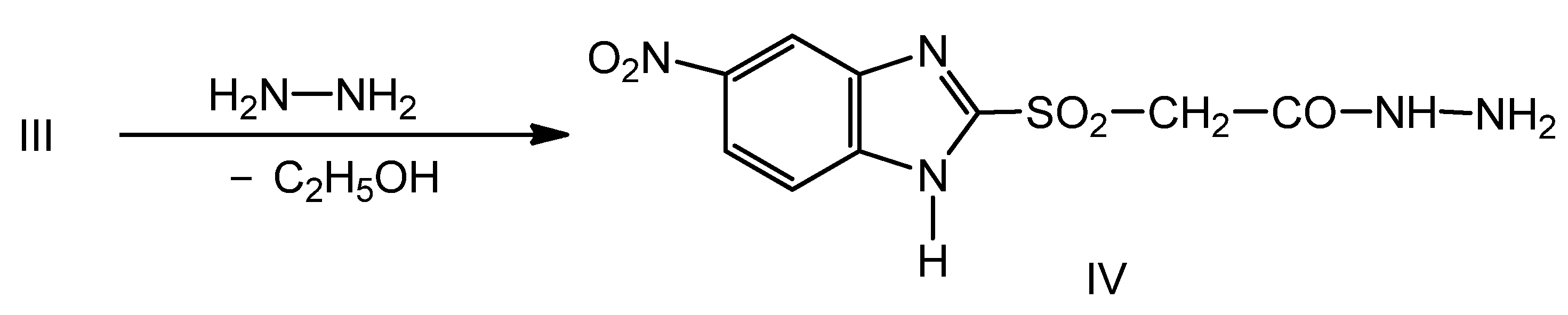
2.1. Synthesis of New Thiosemicarbazides
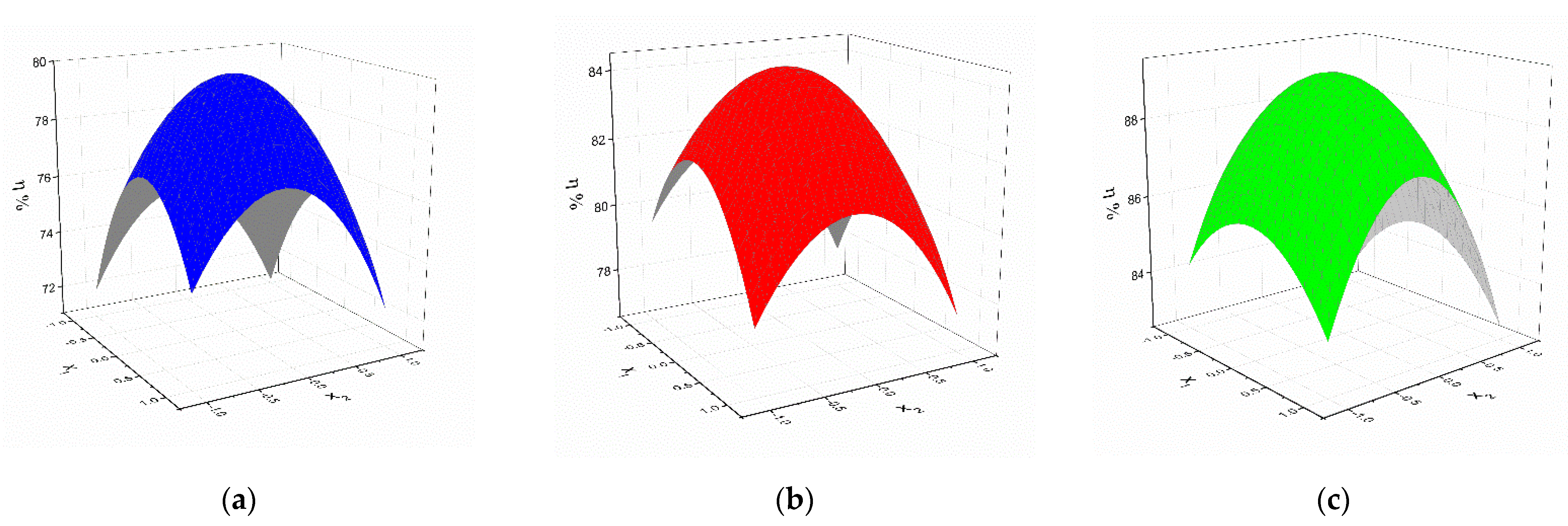
2.2. Optimization of the Chemical Reactions for Obtaining the Thiosemicarbazides V–VII
2.3. Study of Tuberculostatic Activity of Thiosemicarbazides V–VII
3. Materials and Methods
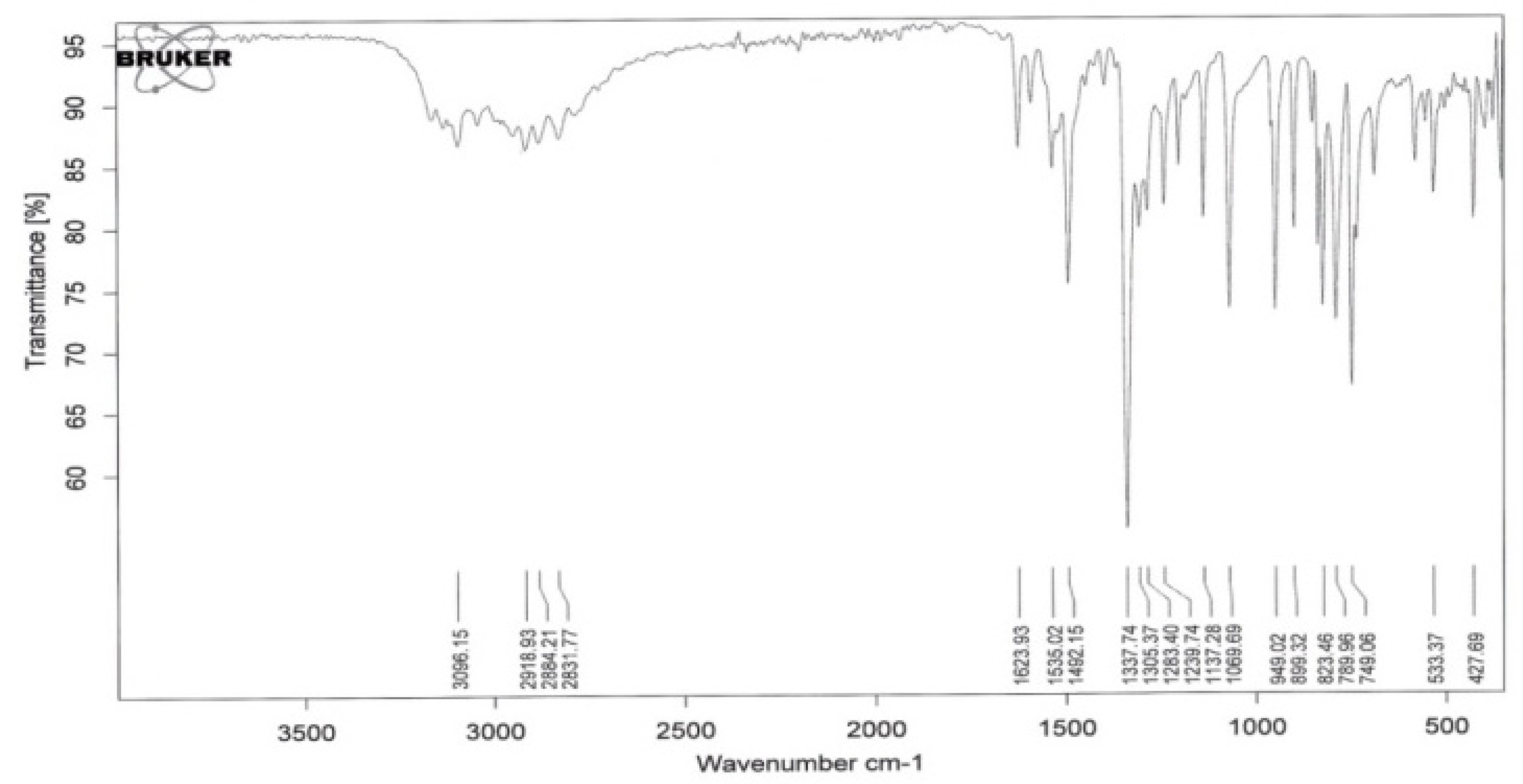
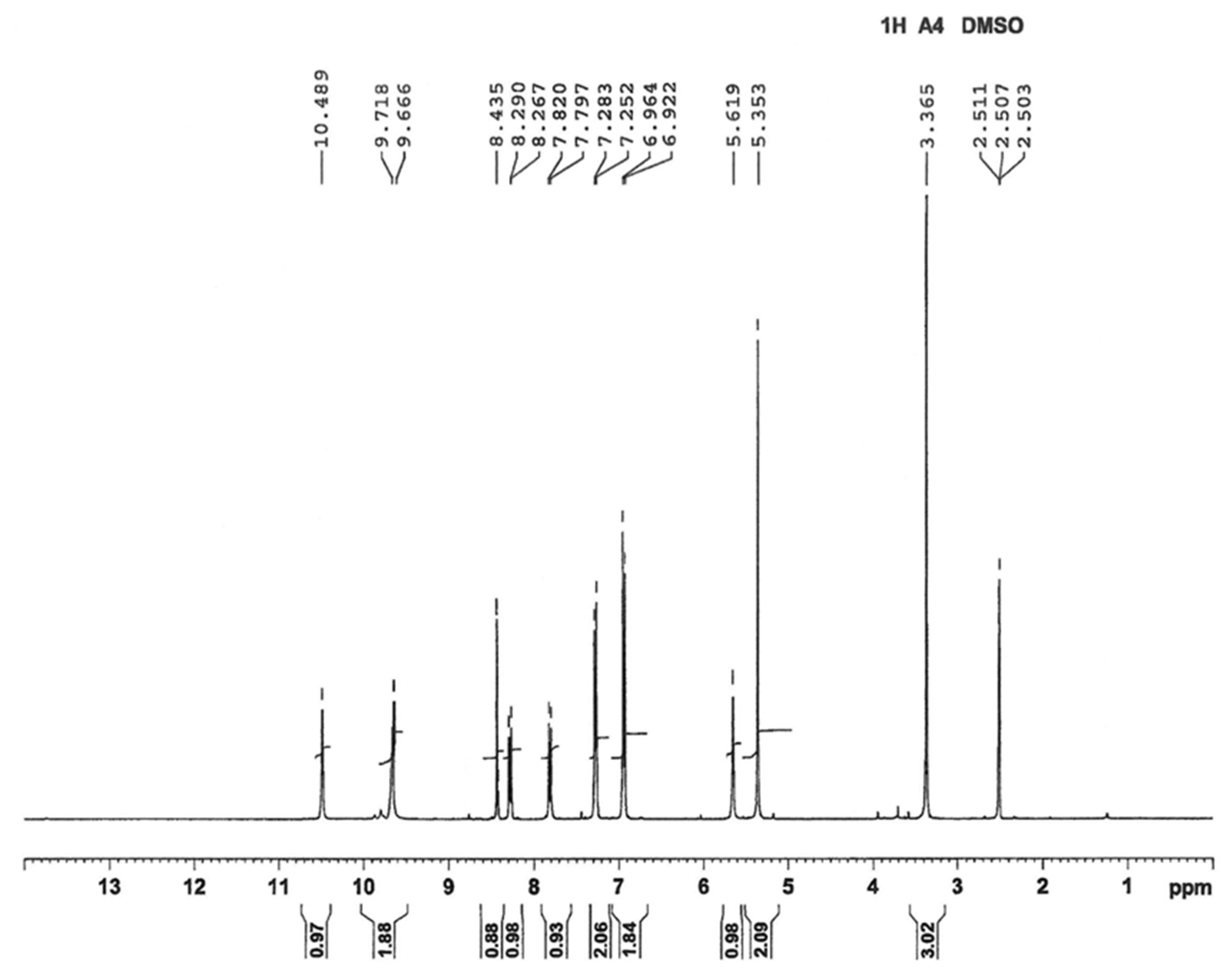
3.1. Chemical Compounds and Analysis
3.2. Optimization of the Chemical Reactions
3.3. Biological Tests
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Garcia-Valverde, M.; Torroba, T. Sulfur-Nitrogen Heterocycles. Molecules 2005, 10, 318–320. [Google Scholar] [CrossRef]
- Moise, M.; Sunel, V.; Profire, L.; Popa, M.; Lionte, C. Synthesis and antimicrobial activity of some new (sulfon-amidophenyl)-amide derivatives of N-(4-nitrobenzoyl)-phenylglycine and N-(4-nitrobenzoyl)-phenylalanine. Farmacia 2008, 56, 283–289. [Google Scholar]
- Bukovski, L.; Janowiec, M.; Zwolska-Kwiec, Z.; Andrezejczyk, Z. Some reactions of 2-cyanomethylimidazo [4,5-b] pyridine with isothiocyanates. Antituberculotic activity of the obtained compounds. Pharmazie 1998, 53, 373–376. [Google Scholar] [CrossRef]
- Karabasanagouda, T.; Adhikari, A.V.; Shetty, N.S. Synthesis and antimicrobial activities of 1,2,4-triazolo[3,4-b]-1,3,4-thidiazoles and 1,2,4-triazolo[3,4-b]-1,3,4-thidiazines carrying thioalkyl and sulphonyl phenoxy moieties. Eur. J. Med. Chem. 2007, 42, 521–529. [Google Scholar] [CrossRef]
- Bhavsar, Z.A.; Acharya, P.T.; Jethava, D.J.; Patel, H.D. Recent advances in development of anthelmintic agents: Synthesis and biological screening. Synth. Commun. 2020, 50, 917–946. [Google Scholar] [CrossRef]
- Cheptea, C.; Sunel, V.; Desbrieres, J.; Popa, M. Synthesis and Antimicrobial Activity of New Derivatives of 1,3,4-Thiadiazoles and 1,2,4-Triazoles with 5-Nitroindazole as Support. J. Heterocycl. Chem. 2013, 50, 366–372. [Google Scholar] [CrossRef]
- Kizilcikli, I.; Mesut, Y. 2-Hydroxy-1-naphthaldehyde S-(Alkyl)-N4-phenylthiosemicarbazone Complexes with Fe(III), Co(III), Ni(II), and Cu(II) Ions. Synth. React. Inorg. Met. Org. Chem. 2003, 33, 949–963. [Google Scholar] [CrossRef]
- Costa, R.F.F.; Rebolledo, A.P.; Matencio, T.; Calado, H.D.; Ardisson, J.D.; Cortés, M.E.; Rodrigues, B.L.; Beraldo, H. Metal complexes of 2-benzoylpyridine-derived thiosemicarbazones: Structural, electrochemical and biological studies. J. Coord. Chem. 2005, 58, 1307–1319. [Google Scholar] [CrossRef]
- Chandra, S.; Raizada, S.; Tyagi, M.; Gautam, A. Synthesis, Spectroscopic, and Antimicrobial Studies on Bivalent Nickel and Copper Complexes of Bis(thiosemicrbazone). Bioinorg. Chem. Appl. 2007, 2007, 051483. [Google Scholar] [CrossRef]
- Beraldo, H.; Gambino, D. The wide pharmacological versatility of semicarbazones, thiosemicarbazones and their metal complexes. Mini Rev. Med. Chem. 2004, 4, 31–39. [Google Scholar] [CrossRef]
- Acharya, P.T.; Bhavsar, Z.A.; Jethava, D.J.; Patel, D.B.; Patel, H.D. A review on development of bio-active thiosemicarbazide derivatives: Recent advances. J. Mol. Struct. 2021, 1226A, 129268. [Google Scholar] [CrossRef]
- Keri, R.S.; Chand, K.; Budagumpi, S.; Somappa, S.B.; Patil, S.A.; Nagaraja, B.M. An overview of benzo[b]thiophene-based medicinal chemistry. Eur. J. Med. Chem. 2017, 138, 1002–1033. [Google Scholar] [CrossRef] [PubMed]
- Küçükgüzel, S.G.; Mazi, A.; Sahin, F.; Öztürk, S.; Stables, J. Synthesis and biological activities of diflunisal hydrazide–hydrazones. Eur. J. Med. Chem. 2003, 38, 1005–1013. [Google Scholar] [CrossRef]
- Papakonstantinou-Garoufalias, S.; Pouli, N.; Marakos, P.; Chytyroglou-Ladas, A. Synthesis antimicrobial and antifungal activity of some new 3-substituted derivatives of 4-(2,4-dichlorophenyl)-5-adamantyl-1H-1,2,4-triazole. Farmaco 2002, 57, 973–977. [Google Scholar] [CrossRef]
- Metwally, M.A.; Bondock, S.; El-Azap, H.; Kandeel, E.-E.M. Thiosemicarbazides: Synthesis and reactions. J. Sulfur Chem. 2011, 32, 489–519. [Google Scholar] [CrossRef]
- Ulusoy Güzeldemirci, N.; Karaman, B.; Küçükbasmaci, Ö. Antibacterial, antitubercular and antiviral activity evaluations of some arylidenehydrazide derivatives bearing imidazo[2,1-b]thiazole moiety. Turk. J. Pharm. Sci. 2017, 14, 157–163. [Google Scholar] [CrossRef]
- Bhowruth, V.; Dover, L.G.; Besra, G.S. 4 Tuberculosis chemotherapy: Recent developments and future perspectives. Prog. Med. Chem. 2007, 45, 169–203. [Google Scholar] [CrossRef] [PubMed]
- Shelke, S.; Mhaske, G.; Gadakh, S.; Gill, C. Green synthesis and biological evaluation of some novel azoles as antimicrobial agents. Bioorg. Med. Chem. Lett. 2010, 20, 7200–7204. [Google Scholar] [CrossRef] [PubMed]
- Bhat, A.R.; Athar, F.; Azam, A. Bis-pyrazolines: Synthesis, characterization and antiamoebic activity as inhibitors of growth of Entamoeba histolytica. Eur. J. Med. Chem. 2009, 44, 426–431. [Google Scholar] [CrossRef]
- Bonde, C.G.; Gaikwad, N.J. Synthesis and preliminary evaluation of some pyrazine containing thiazolines and thiazolidinones as antimicrobial agents. Bioorg. Med. Chem. 2004, 12, 2151–2161. [Google Scholar] [CrossRef] [PubMed]
- Holban, M.; Sunel, V.; Popa, M.; Lionte, C. Synthesis and characterization of a new starch ester with N-[(N’-tiazolyl)-p’-(benzenesulphone)] amide of N-(o-nitrobenzoyl)-D,L-asparagic acid. Cell. Chem. Technol. 2011, 45, 191–196. [Google Scholar]
- Kwara, A.; Flanigan, T.P.; Carter, E.J. Highly active antiretroviral therapy (HAART) in adults with tuberculosis: Current status. Int. J. Tuberc. Lung Dis. 2005, 9, 248–257. [Google Scholar] [PubMed]
- Karali, N.; Illhan, E.; Gürsoy, A.; Kiraz, M. New cyclohexylidenehydrazide and 4-aza-1-thiaspiro[4.5]decan-3-one derivatives of 3-phenyl-4(3H)-quinazolinones. Farmaco 1998, 53, 346–349. [Google Scholar] [CrossRef]
- Bacon, J.; James, B.W.; Wernisch, L.; Williams, A.; Morley, K.A.; Hatch, G.J.; Mangan, J.A.; Hinds, J.; Stoker, N.G.; Butcher, P.D.; et al. The influence of reduced oxygen availability on pathogenicity and gene expression in Mycobacterium tuberculosis. Tuberculosis 2004, 84, 205–217. [Google Scholar] [CrossRef]
- Shakya, B.; Yadav, P.N. Thiosemicarbazones as potent anticancer agents and their modes of action. Mini Rev. Med. Chem. 2020, 20, 638–661. [Google Scholar] [CrossRef]
- Seleem, H.S.; El-Shetary, B.A.; Khalil, S.M.E.; Mostafa, M.; Shebl, M. Structural diversity in copper(II) complexes of bis(thiosemicarbazone) and bis(semicarbazone) ligands. J. Coord. Chem. 2005, 58, 479–493. [Google Scholar] [CrossRef]
- Hussein, M.A.; Iqbal, M.A.; Umar, M.I.; Haque, R.A.; Guan, T.S. Synthesis, structural elucidation and cytotoxicity of new thiosemicarbazone derivatives. Arab. J. Chem. 2019, 12, 3183–3192. [Google Scholar] [CrossRef] [Green Version]
- Gazieva, G.A.; Kravchenko, A.N. Thiosemicarbazides in the synthesis of five- and six-membered heterocyclic compounds. Russ. Chem. Rev. 2012, 81, 494–523. [Google Scholar] [CrossRef]
- Nguyen, D.T.; Le, T.H.; Bui, T.T.T. Antioxidant activities of thiosemicarbazones from substituted benzaldehydes and N-(tetra-O-acetyl-β-D-galactopyranosyl)thiosemicarbazide. Eur. J. Med. Chem. 2013, 60, 199–207. [Google Scholar] [CrossRef]
- Krysin, E.P.; Karel’skii, V.N.; Antonov, A.A.; Rostovskaya, G.E. Preparation of hydrazides of amino acids and peptides. Chem. Nat. Compd. 1979, 15, 601–603. [Google Scholar] [CrossRef]
- Ahmad, A.; Varshney, H.; Rauf, A.; Sherwani, A.; Owais, M. Synthesis and anticancer activity of long chain substituted 1,3,4-oxadiazol-2-thione, 1,2,4-triazol-3-thione and 1,2,4-triazolo[3,4-b]-1,3,4-thiadiazine derivatives. Arab. J. Chem. 2017, 10, S3347–S3357. [Google Scholar] [CrossRef] [Green Version]
- Cheptea, C.; Sunel, V.; Profire, L.; Popa, M.; Lionte, C. New hydrazones of 5-nitroindazol-1-yl-acethydrazide with pharmacological potential. Iasi Polyt. Inst. Bull. 2009, 55, 89–97. [Google Scholar]
- Pintilie, O.; Profire, L.; Sunel, V.; Popa, M.; Pui, A. Synthesis and antimicrobial activity of some new 1,3,4-thiadiazole and 1,2,4-triazole compounds having a D,L-methionine moiety. Molecules 2007, 12, 103–113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Plech, T.; Wujec, M.; Siwek, A.; Kosikowska, U.; Malm, A. Synthesis and antimicrobial activity of thiosemicarbazides, s-triazoles and their Mannich bases bearing 3-chlorophenyl moiety. Eur. J. Med. Chem. 2011, 46, 241–248. [Google Scholar] [CrossRef]
- Grebinisan, D.; Burlea, M.; Cheptea, C.; Lionte, C.; Dorohoi, D.O.; Sunel, V.; Popa, M.; Hurjui, I. Optimization reaction for obtaining some N-[p-R-benzoyl]-1-glutamine derivatives with pharmaceutical action. Dig. J. Nanomater. Bios. 2013, 8, 777–785. [Google Scholar]
- Gicu, T.A.; Nechifor, C.D.; Sunel, V.; Dorohoi, D.O.; Cheptea, C. Optimization reaction for obtaining new hydrazidones with biological activity. Rev. Roum. Chim. 2014, 59, 739–747. [Google Scholar]
- Cheptea, C.; Sunel, V.; Morosanu, A.C.; Dimitriu, D.G.; Dulcescu-Oprea, M.M.; Angheluta, M.D.; Miron, M.; Nechifor, C.D.; Dorohoi, D.O.; Malancus, R.N. Optimized synthesis of new N-mustards based on 2-mercaptobenzoxazole derivatives with antitumor activity. Biomedicines 2021, 9, 476. [Google Scholar] [CrossRef]
- Student. The probable error of a mean. Biometrika 1908, 6, 1–25. [Google Scholar] [CrossRef]
- Hurjui, I.; Cheptea, C.; Dascalu, C.F.; Hurjui, L.; Peptu, C.; Sunel, V.; Dorohoi, D.O. Optimization reaction of some 1,4-disubstituted thiocarbazides with tuberculostatic activity. Dig. J. Nanomater. Bios. 2012, 7, 1747–1756. [Google Scholar]

| No. | x1; T (°C) | x2; t (h) | x1x2 | η (%) | ||
|---|---|---|---|---|---|---|
| 1 | –1 (64) | –1 (2.5) | 1 | 1/3 | 1/3 | 72 |
| 2 | –1 (64) | 0 (3) | 0 | 1/3 | –2/3 | 76 |
| 3 | –1 (64) | 1 (3.5) | –1 | 1/3 | 1/3 | 70 |
| 4 | 0 (64.5) | –1 (2.5) | 0 | –2/3 | 1/3 | 76 |
| 5 | 0 (64.5) | 0 (3) | 0 | –2/3 | –2/3 | 80 |
| 6 | 0 (64.5) | 1 (3.5) | 0 | –2/3 | 1/3 | 77 |
| 7 | 1 (65) | –1 (2.5) | –1 | 1/3 | 1/3 | 75 |
| 8 | 1 (65) | 0 (3) | 0 | 1/3 | –2/3 | 76 |
| 9 | 1 (65) | 1 (3.5) | 1 | 1/3 | 1/3 | 72 |
| No. | x1; T (°C) | x2; t (h) | x1x2 | η (%) | ||
|---|---|---|---|---|---|---|
| 1 | –1 (64) | –1 (3.0) | 1 | 1/3 | 1/3 | 80 |
| 2 | –1 (64) | 0 (3.5) | 0 | 1/3 | –2/3 | 83 |
| 3 | –1 (64) | 1 (4.0) | –1 | 1/3 | 1/3 | 78 |
| 4 | 0 (64.5) | –1 (3.0) | 0 | –2/3 | 1/3 | 82 |
| 5 | 0 (64.5) | 0 (3.5) | 0 | –2/3 | –2/3 | 84 |
| 6 | 0 (64.5) | 1 (4.0) | 0 | –2/3 | 1/3 | 83 |
| 7 | 1 (65) | –1 (3.0) | –1 | 1/3 | 1/3 | 79 |
| 8 | 1 (65) | 0 (3.5) | 0 | 1/3 | –2/3 | 82 |
| 9 | 1 (65) | 1 (4.0) | 1 | 1/3 | 1/3 | 78 |
| No. | x1; T (°C) | x2; t (h) | x1x2 | η (%) | ||
|---|---|---|---|---|---|---|
| 1 | –1 (64) | –1 (3.5) | 1 | 1/3 | 1/3 | 84 |
| 2 | –1 (64) | 0 (4.0) | 0 | 1/3 | –2/3 | 88 |
| 3 | –1 (64) | 1 (4.5) | –1 | 1/3 | 1/3 | 83 |
| 4 | 0 (64.5) | –1 (3.5) | 0 | –2/3 | 1/3 | 86 |
| 5 | 0 (64.5) | 0 (4.0) | 0 | –2/3 | –2/3 | 89 |
| 6 | 0 (64.5) | 1 (4.5) | 0 | –2/3 | 1/3 | 85 |
| 7 | 1 (65) | –1 (3.5) | –1 | 1/3 | 1/3 | 84 |
| 8 | 1 (65) | 0 (4.0) | 0 | 1/3 | –2/3 | 87 |
| 9 | 1 (65) | 1 (4.5) | 1 | 1/3 | 1/3 | 83 |
| Compound | x1; T (°C) | x2; t(h) | η (%) |
|---|---|---|---|
| V | 0.11 (64.55) | –0.09 (3 h 57 min 18 s) | 79.83 |
| VI | –0.05 (64.47) | 0.11 (3 h 33 min 25 s) | 84.22 |
| VII | –0.05 (64.47) | –0.07 (3 h 57 min 54 s) | 89.24 |
| Compound | The Concentration of Thiosemicarbazides in the Culture Medium (μg/mL) | MIC (μg/mL) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 5 | 10 | 20 | 30 | 40 | |||||||
| 6 Days | 15 Days | 6 Days | 15 Days | 6 Days | 15 Days | 6 Days | 15 Days | 6 Days | 15 Days | ||
| V | ++ | +++ | ++ | ++ | ++ | + | – | – | – | – | 35 |
| VI | ++ | +++ | ++ | + | – | – | – | – | – | – | 20 |
| VII | ++ | +++ | + | – | – | – | – | – | – | – | 15 |
| IAH | – | – | – | – | – | – | – | – | – | – | 10 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Popovici, C.; Pavel, C.-M.; Sunel, V.; Cheptea, C.; Dimitriu, D.G.; Dorohoi, D.O.; David, D.; Closca, V.; Popa, M. Optimized Synthesis of New Thiosemicarbazide Derivatives with Tuberculostatic Activity. Int. J. Mol. Sci. 2021, 22, 12139. https://doi.org/10.3390/ijms222212139
Popovici C, Pavel C-M, Sunel V, Cheptea C, Dimitriu DG, Dorohoi DO, David D, Closca V, Popa M. Optimized Synthesis of New Thiosemicarbazide Derivatives with Tuberculostatic Activity. International Journal of Molecular Sciences. 2021; 22(22):12139. https://doi.org/10.3390/ijms222212139
Chicago/Turabian StylePopovici, Corina, Cristina-Maria Pavel, Valeriu Sunel, Corina Cheptea, Dan Gheorghe Dimitriu, Dana Ortansa Dorohoi, Diana David, Valentina Closca, and Marcel Popa. 2021. "Optimized Synthesis of New Thiosemicarbazide Derivatives with Tuberculostatic Activity" International Journal of Molecular Sciences 22, no. 22: 12139. https://doi.org/10.3390/ijms222212139
APA StylePopovici, C., Pavel, C.-M., Sunel, V., Cheptea, C., Dimitriu, D. G., Dorohoi, D. O., David, D., Closca, V., & Popa, M. (2021). Optimized Synthesis of New Thiosemicarbazide Derivatives with Tuberculostatic Activity. International Journal of Molecular Sciences, 22(22), 12139. https://doi.org/10.3390/ijms222212139







