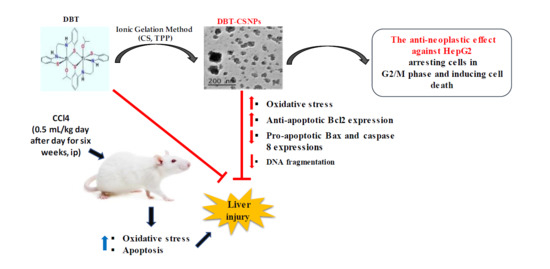
A Titanium (IV)–Dithiophenolate Complex and Its Chitosan Nanocomposite: Their Roles towards Rat Liver Injuries In Vivo and against Human Liver Cancer Cell Lines
Abstract
1. Introduction
2. Results
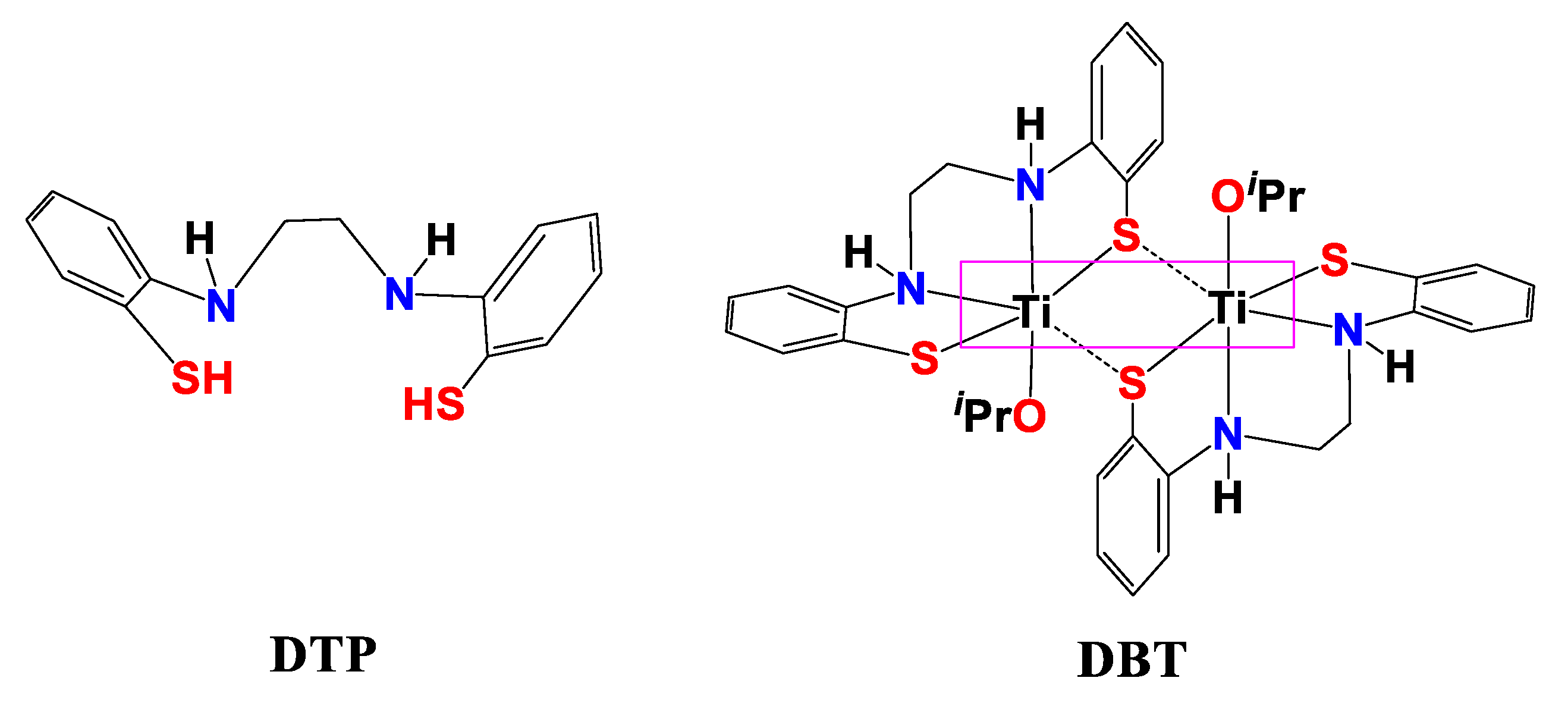
2.1. Thermal Stability of DBT and DBT–CSNPs
2.2. LD50 of DBT and DBT–CSNPs
2.3. Effect of Treatments with DBT, DBT–CSNPs, and Cisplatin on CCl4-Induced Hepatotoxicity
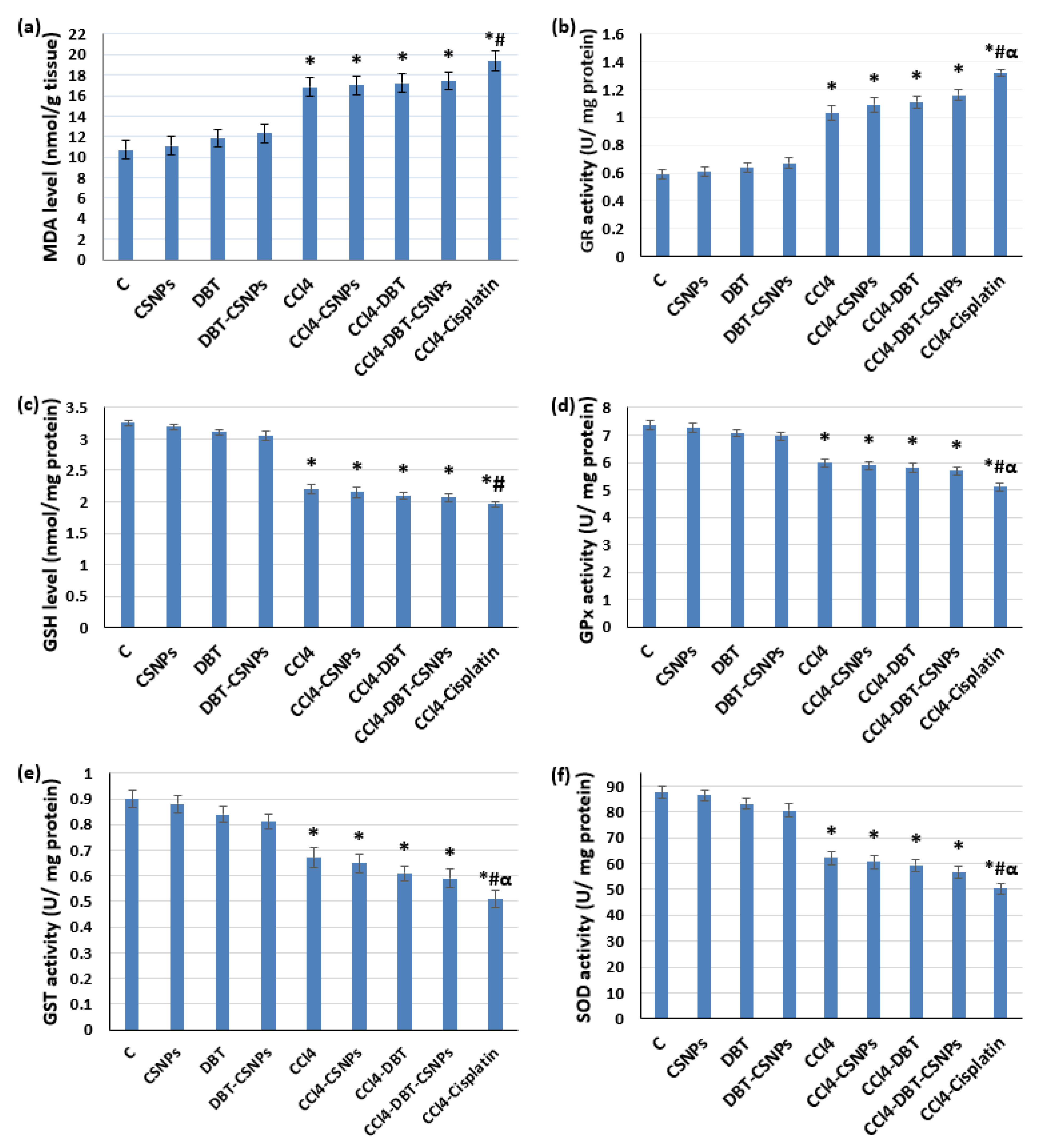
2.3.1. Effect of Different Studied Compounds on OS Markers
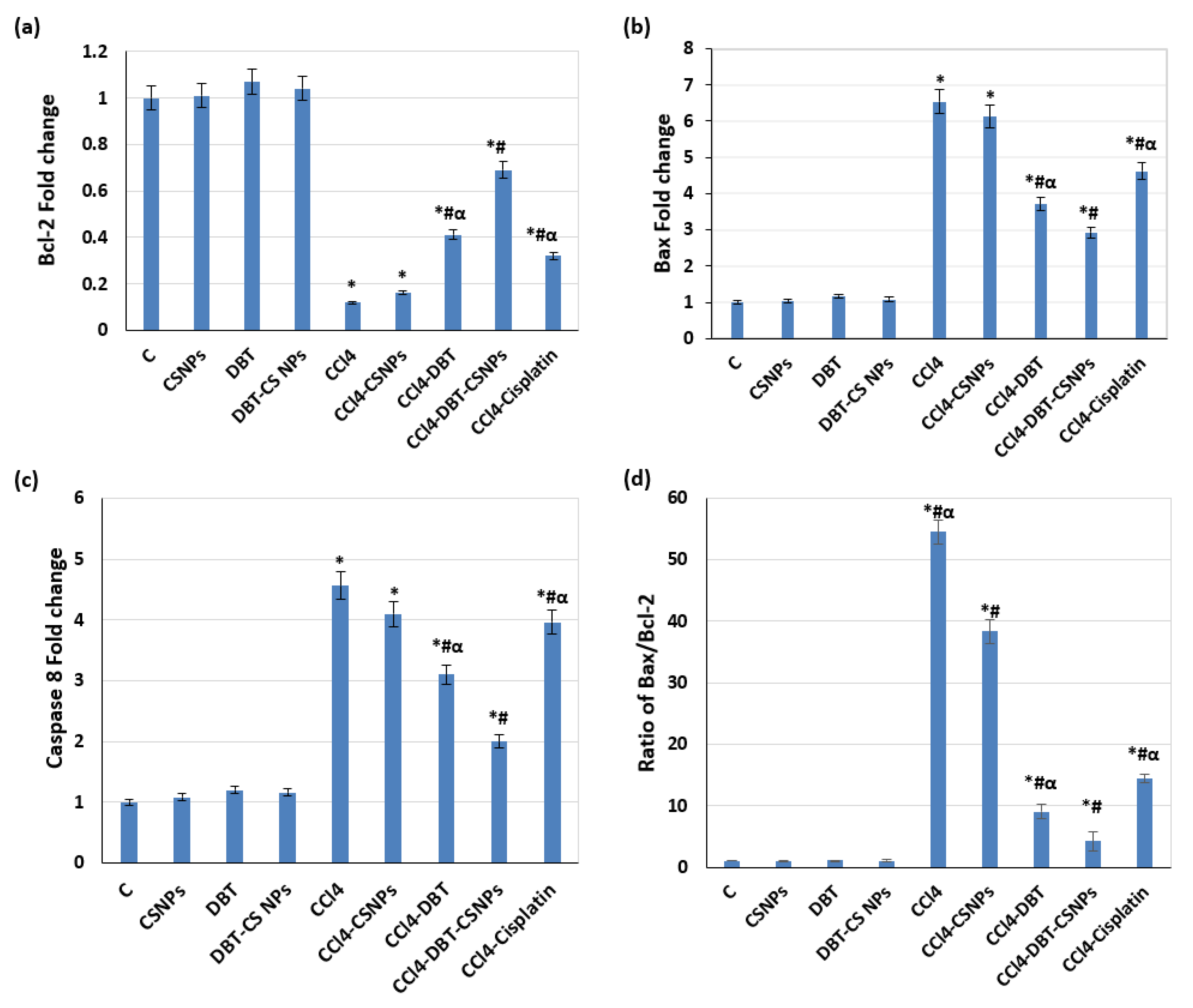
2.3.2. Effect of Different Studied Compounds on Apoptosis
2.4. Effect of Different Studied Compounds on Liver and Kidney Functions and the Lipid Profile
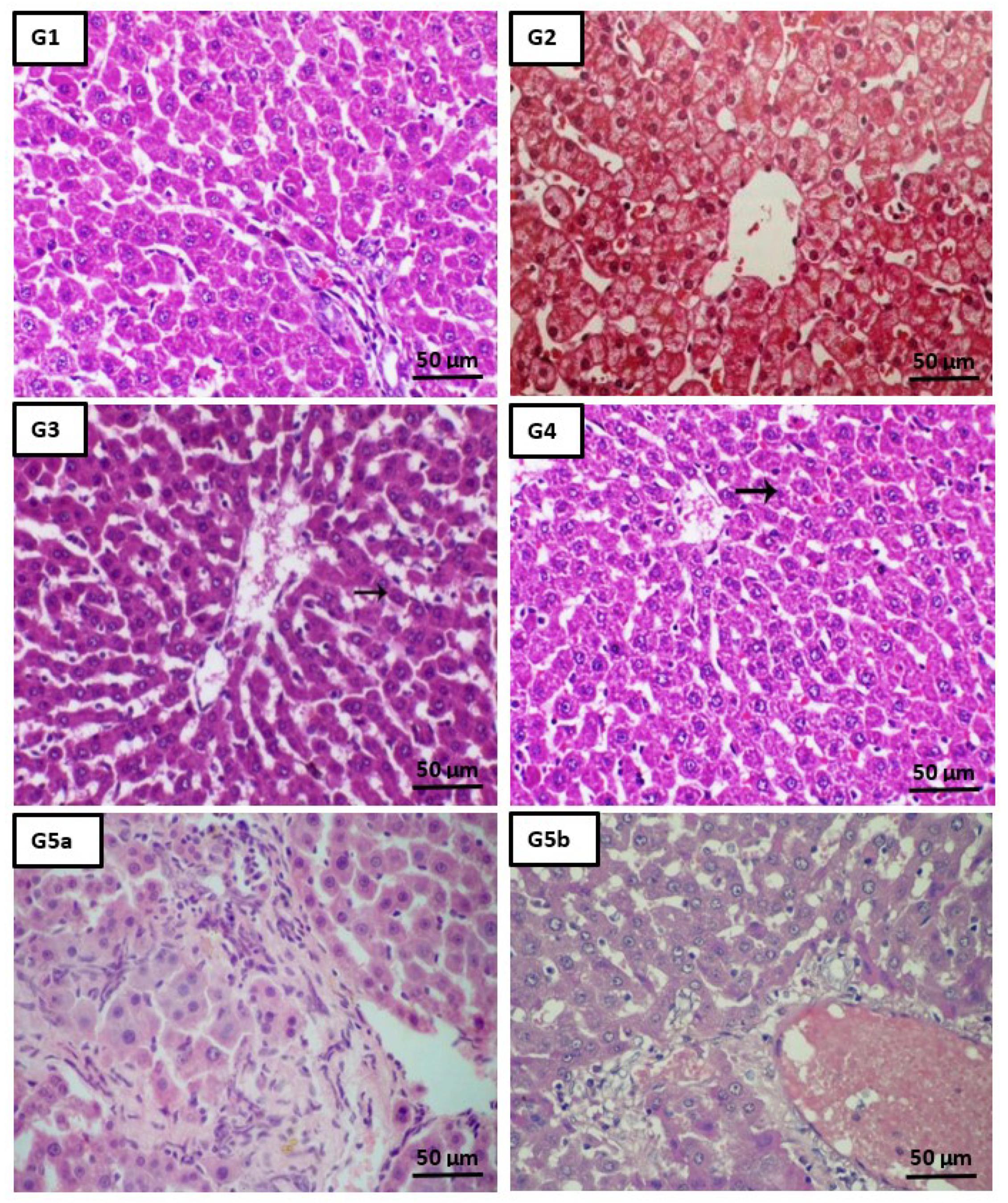
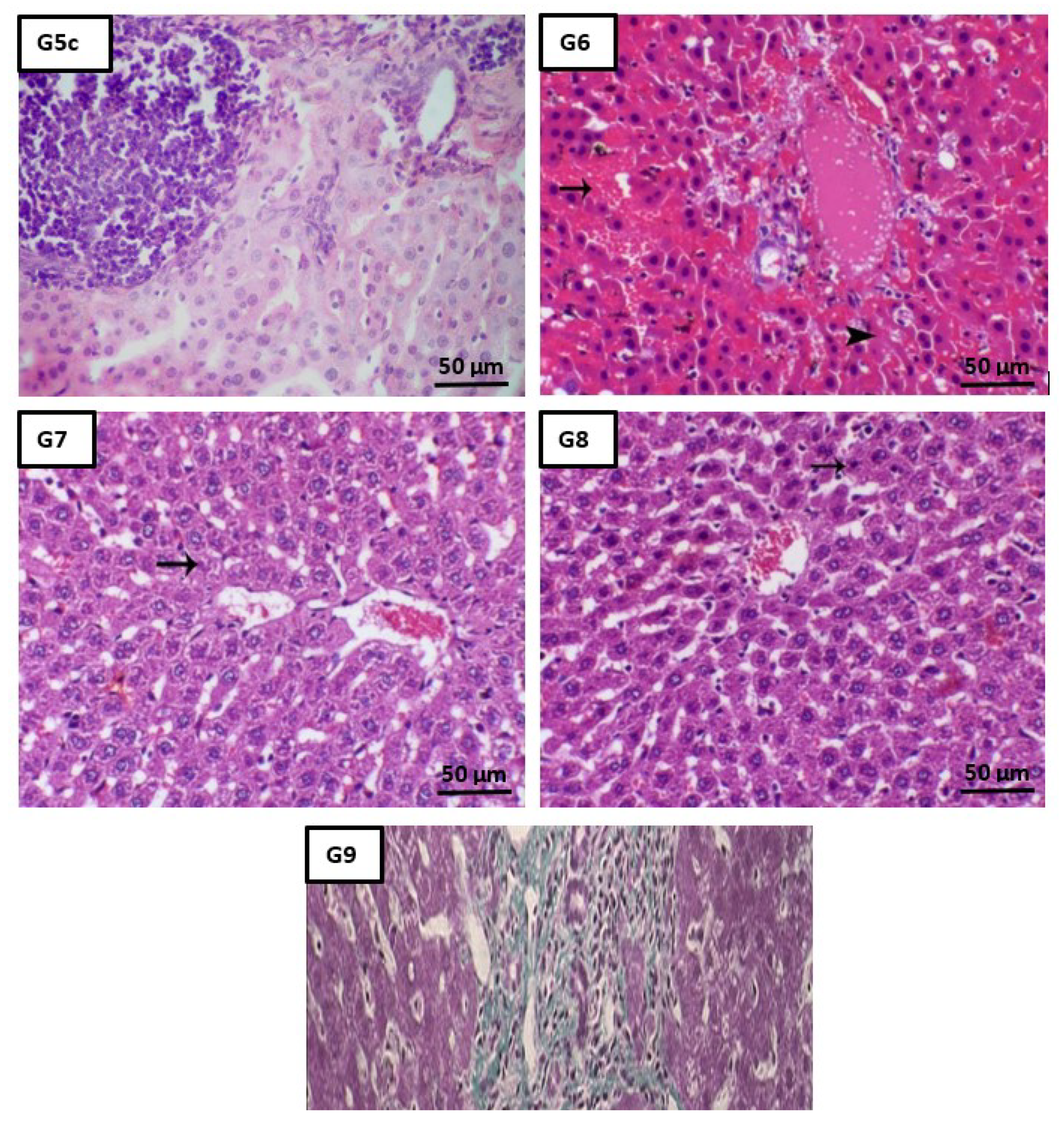
2.5. Histopathological Analysis
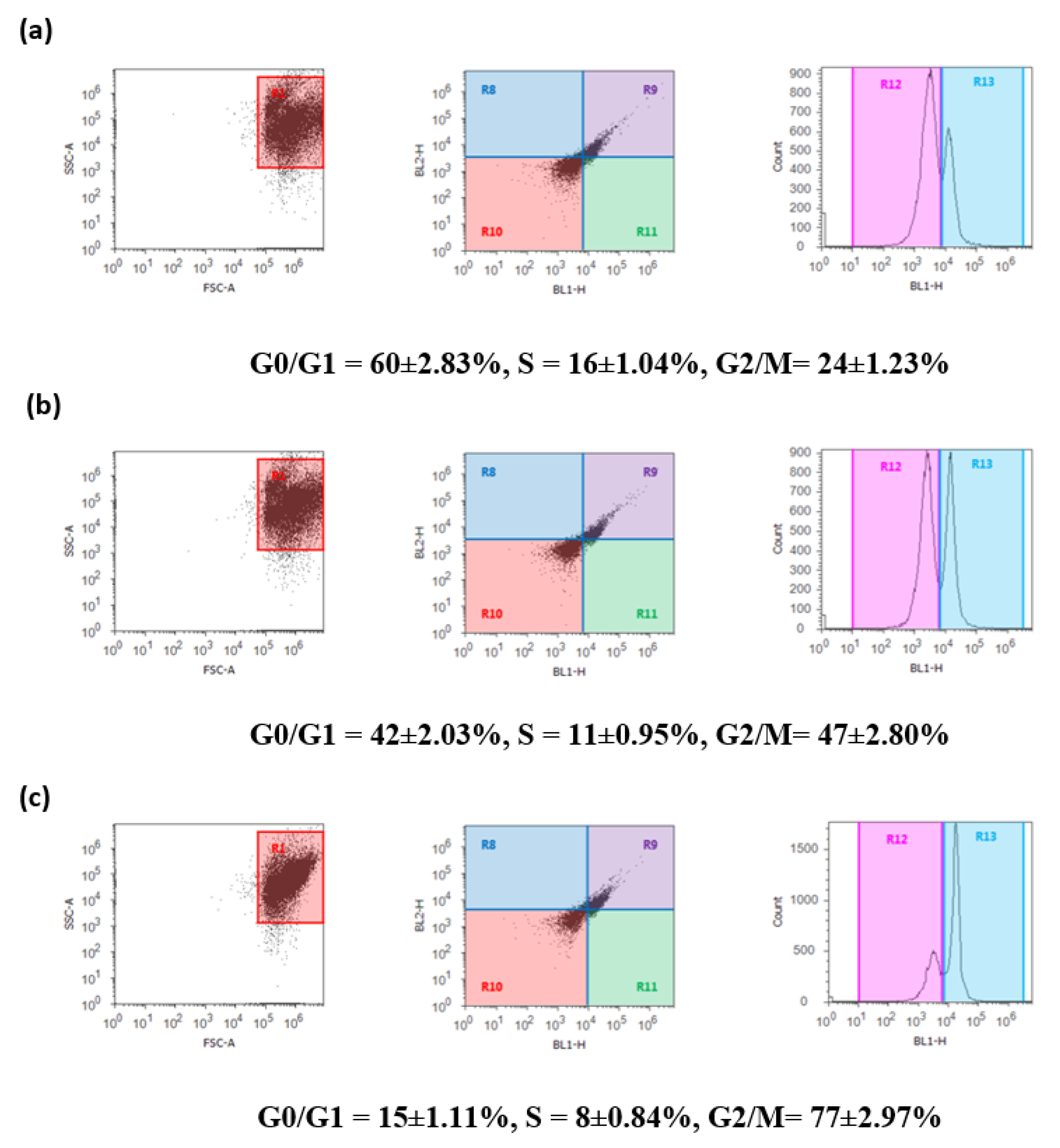
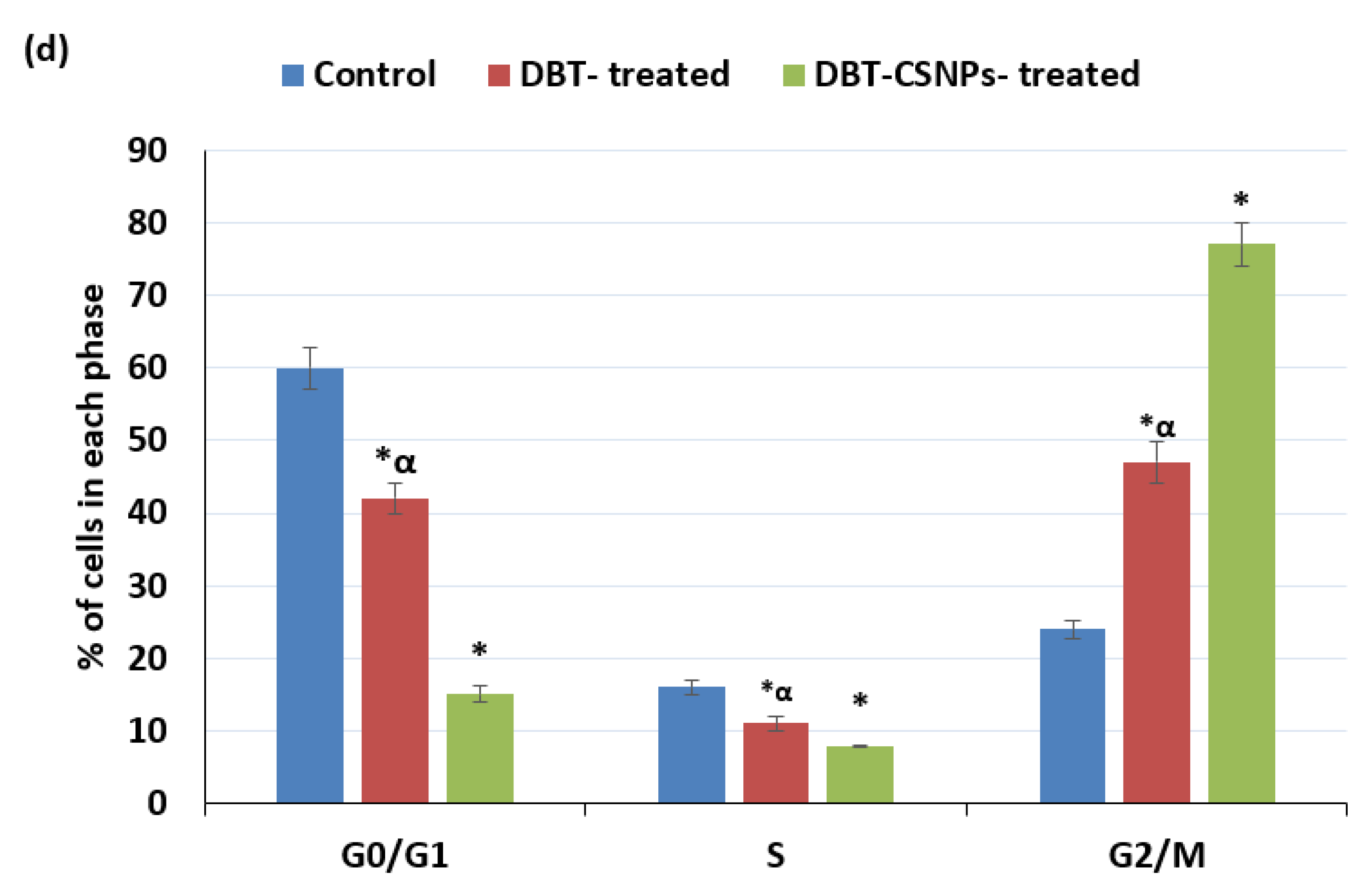
2.6. DBT and DBT–CSNP-Induced Cell Cycle Arrest
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Animals
4.3. Preparation of DBT–CSNPs
4.4. Determination of the LD50 Values of DBT and DBT–CSNPs
4.5. Biological Effects of DBT, DBT–CSNPs, and Cisplatin on CCl4-Induced Liver Injuries
4.6. Effect of the Studied Compounds on OS Markers
4.7. Effect of the Studied Compounds on Apoptotic Markers
4.7.1. Determination of Gene Expressions of Bcl-2, Bax, and Caspase-8
4.7.2. Determination of DNAF
4.8. Effect of the Studied Compounds on Liver and Kidney Functions and the Lipid Profile
4.9. Liver Histopathological Analysis
4.10. Cell Cycle Analysis
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Apaf-1 | apoptosis protease activating factor-1 |
| CCl4 | carbon tetrachloride |
| •CCl3 | trichloromethyl radical |
| CCl3OO• | trichloromethylperoxy radicals |
| CS | chitosan |
| CSNPs | chitosan nanoparticles |
| DBT | titanium (IV)-thiophenolate complex |
| DBT–CSNPS | titanium (IV)–dithiophenolate -Chitosan nanocomposite |
| DMSO | Dimethyl sulfoxide |
| DTP | dithiophenolato ligand |
| GPx | total glutathione peroxidase |
| GR | glutathione reductase |
| GSH | reduced glutathione |
| GST | glutathione-S-transferase |
| GSSG | oxidized glutathione |
| HepG2 | human liver cancer |
| MDA | malondialdehyde |
| RNS | reactive nitrogen species |
| ROS | reactive oxygen speci |
| SOD | superoxide dismutase |
| THLE2 | normal liver cell |
References
- Al-Seeni, M.N.; El Rabey, H.A.; Zamzami, M.A.; Alnefayee, A.M. The hepatoprotective activity of olive oil and Nigella sativa oil against CCl4 induced hepatotoxicity in male rats. BMC Complement. Altern. Med. 2016, 16, 438. [Google Scholar] [CrossRef]
- Shaban, N.Z.; El-Kersh, M.A.R.; Bader-Eldin, M.M.; Kato, S.A.; Hamoda, A.F. Effect of Punica granatum (Pomegranate) juice extract on healthy liver and hepatotoxicity induced by diethylnitrosamine and phenobarbital in male rats. J. Med. Food 2014, 17, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Monem, N.M.; El-Saadani, M.A.; Daba, A.S.; Saleh, S.R.; Aleem, E. Exopolysaccharide-peptide complex from oyster mushroom (Pleurotus ostreatus) protects against hepatotoxicity in rats. Biochem. Biophys. Rep. 2020, 24, 100852. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wen, P.-H.; Zhang, X.-X.; Dai, Y.; He, Q. Breviscapine ameliorates CCl4-induced liver injury in mice through inhibiting inflammatory apoptotic response and ROS generation. Int. J. Mol. Med. 2018, 42, 755–768. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Tang, C.; Zhang, H. Hepatoprotective effects of kaempferol 3-O-rutinoside and kaempferol 3-O-glucoside from Carthamus tinctorius L. on CCl4-induced oxidative liver injury in mice. J. Food Drug Anal. 2015, 23, 310–317. [Google Scholar] [CrossRef]
- Dutta, S.; Chakraborty, A.K.; Dey, P.; Kar, P.; Guha, P.; Sen, S.; Kumar, A.; Sen, A.; Chaudhuri, T.K. Amelioration of CCl4 induced liver injury in swiss albino mice by antioxidant rich leaf extract of Croton bonplandianus Baill. PLoS ONE 2018, 13, e0196411. [Google Scholar] [CrossRef]
- Das, R.; Vupputuri, S.; Hu, Q.; Chen, Y.; Colorado, H.; Guo, Z.; Wang, Z. Synthesis and characterization of antiflammable vinyl ester resin nanocomposites with surface functionalized nanotitania. ES Mater. Manuf. 2020, 8, 46–53. [Google Scholar] [CrossRef]
- El-Sayed, A.; Kamel, M. Advances in nanomedical applications: Diagnostic, therapeutic, immunization, and vaccine production. Environ. Sci. Pollut. Res. 2020, 27, 19200–19213. [Google Scholar] [CrossRef] [PubMed]
- Shaban, N.Z.; Aboelsaad, A.M.; Shoueir, K.R.; Abdulmalek, S.A.; Awad, D.; Shaban, S.Y.; Mansour, H. Chitosan-based dithiophenolato nanoparticles: Preparation, mechanistic information of DNA binding, antibacterial and cytotoxic activities. J. Mol. Liq. 2020, 318, 114252. [Google Scholar] [CrossRef]
- Li, J.; Cai, C.; Sun, T.; Wang, L.; Wu, H.; Yu, G. Chitosan-based nanomaterials for drug delivery. Molecules 2018, 23, 2661. [Google Scholar] [CrossRef] [PubMed]
- Hafez, H.S.; Ghareeb, D.A.; Saleh, S.R.; Abady, M.M.; El Demellawy, M.A.; Hussien, H.; Abdel-Monem, N. Neuroprotective effect of ipriflavone against scopolamine-induced memory impairment in rats. Psychopharmacology 2017, 234, 3037–3053. [Google Scholar] [CrossRef]
- Marcellin, P.; Kutala, B.K. Liver diseases: A major, neglected global public health problem requiring urgent actions and large-scale screening. Liver Int. Off. J. Int. Assoc. Study Liver 2018, 38, 2–6. [Google Scholar] [CrossRef] [PubMed]
- Rashed, W.M.; Kandeil, M.A.M.; Mahmoud, M.O.; Ezzat, S. Hepatocellular Carcinoma (HCC) in Egypt: A comprehensive overview. J. Egypt. Natl. Cancer Inst. 2020, 32, 5. [Google Scholar] [CrossRef]
- Karaca, O.; Paksoy, S.; Hismiogullsri, S.; Kus, M.; Sunay, F.; Gulcen, B.; Ozcan, E.J.A.M.M. Prevention of carbontetrachloride-induced liver injury in rats by omega-3-fatty acids. Acta Med. Mediterr. 2017, 33, 137–144. [Google Scholar]
- Ananth, H.; Kundapur, V.; Mohammed, H.S.; Anand, M.; Amarnath, G.S.; Mankar, S. A review on biomaterials in dental implantology. Int. J. Biomed. Sci. IJBS 2015, 11, 113–120. [Google Scholar]
- Shaban, N.Z.; Aboelsaad, A.M.; Awad, D.; Abdulmalek, S.A.; Shaban, S.Y. Therapeutic effect of dithiophenolato chitosan nanocomposites against carbon tetrachloride-induced hepatotoxicity in rats. Environ. Sci. Pollut. Res. 2021, 8, 1–16. [Google Scholar] [CrossRef]
- Shaban, N.Z.; Yehia, S.A.; Shoueir, K.R.; Saleh, S.R.; Awad, D.; Shaban, S.Y. Design, DNA binding and kinetic studies, antibacterial and cytotoxic activities of stable dithiophenolato titanium (IV)-chitosan Nanocomposite. J. Mol. Liq. 2019, 287, 111002. [Google Scholar] [CrossRef]
- Shaban, S.Y.; Ramadan, A.E.-M.M.; Heinemann, F.W. Titanium isopropoxide complexes containing diamine bis-thiolato based [N2S2]2− ligands; effect of steric bulk on coordination features. Inorg. Chem. Commun. 2012, 20, 135–137. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, G.; Tiancheng, W.; Yu, H.; Wang, T.; Ma, Y.; Jia, G.; Gao, Y.; Li, Y.-F.; Sun, J. Acute toxicity and biodistribution of different sized titanium dioxide particles in mice after oral administration. Toxicol. Lett. 2007, 168, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Alkreathy, H.M.; Khan, R.A.; Khan, M.R.; Sahreen, S. CCl4 induced genotoxicity and DNA oxidative damages in rats: Hepatoprotective effect of Sonchus arvensis. BMC Complement. Altern. Med. 2014, 14, 452. [Google Scholar] [CrossRef] [PubMed]
- Kurutas, E.B. The importance of antioxidants which play the role in cellular response against oxidative/nitrosative stress: Current state. Nutr. J. 2015, 15, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.D.; A Dsouza, U.J.; Iqbal, M. The potential protective effect of Commelina nudiflora L. against carbon tetrachloride (CCl4)-induced hepatotoxicity in rats, mediated by suppression of oxidative stress and inflammation. Environ. Heal. Prev. Med. 2017, 22, 1–19. [Google Scholar] [CrossRef]
- Ighodaro, O.; Akinloye, O. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alex. J. Med. 2018, 54, 287–293. [Google Scholar] [CrossRef]
- Kale, J.; Osterlund, E.J.; Andrews, D.W. BCl-2 family proteins: Changing partners in the dance towards death. Cell Death Differ. 2018, 25, 65–80. [Google Scholar] [CrossRef]
- Hassan, M.; Watari, H.; AbuAlmaaty, A.; Ohba, Y.; Sakuragi, N. Apoptosis and molecular targeting therapy in cancer. Biomed. Res. Int. 2014, 2014, 150845. [Google Scholar] [CrossRef] [PubMed]
- Campbell, K.J.; Tait, S.W.G. Targeting BCl-2 regulated apoptosis in cancer. Open Biol. 2018, 8, 180002. [Google Scholar] [CrossRef]
- Pistritto, G.; Trisciuoglio, D.; Ceci, C.; Garufi, A.; D’Orazi, G. Apoptosis as anticancer mechanism: Function and dysfunction of its modulators and targeted therapeutic strategies. Aging 2016, 8, 603–619. [Google Scholar] [CrossRef]
- Kalkavan, H.; Green, D.R. MOMP, cell suicide as a BCl-2 family business. Cell Death Differ. 2018, 25, 46–55. [Google Scholar] [CrossRef]
- Liu, Z.; Ding, Y.; Ye, N.; Wild, C.; Chen, H.; Zhou, J. Direct Activation of Bax Protein for Cancer Therapy. Med. Res. Rev. 2016, 36, 313–341. [Google Scholar] [CrossRef]
- Eltahir, H.M.; Nazmy, M.H. Esomeprazole ameliorates CCl4 induced liver fibrosis in rats via modulating oxidative stress, inflammatory, fibrogenic and apoptotic markers. Biomed. Pharmacother. 2018, 97, 1356–1365. [Google Scholar] [CrossRef]
- Yokoyama, C.; Sueyoshi, Y.; Ema, M.; Mori, Y.; Takaishi, K.; Hisatomi, H. Induction of oxidative stress by anticancer drugs in the presence and absence of cells. Oncol. Lett. 2017, 14, 6066–6070. [Google Scholar] [CrossRef]
- Fong, C. Free Radical Anticancer Drugs and Oxidative Stress: ORAC and CellROX-Colorectal Cancer Cells by Quantum Chemical Determinations. Ph.D. Thesis, Eigenenergy, Adelaide, Australia, 2018. [Google Scholar]
- Roos, W.P.; Kaina, B. DNA damage-induced cell death: From specific DNA lesions to the DNA damage response and apoptosis. Cancer Lett. 2013, 332, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.L.; Liang, B.; Wang, X.W.; Fan, F.G.; Jin, J.; Lan, R.; Yang, J.H.; Wang, X.C.; Jin, L.; Cao, Q. Glycyrrhizic acid attenuates CCl₄-induced hepatocyte apoptosis in rats via a p53-mediated pathway. World J. Gastroenterol. 2013, 19, 3781–3791. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Wang, S.; Zhou, L.; Sun, L. The potential liver, brain, and embryo toxicity of titanium dioxide nanoparticles on mice. Nanoscale Res. Lett. 2017, 12, 478. [Google Scholar] [CrossRef]
- Pujalté, I.; Passagne, I.; Brouillaud, B.; Tréguer, M.; Durand, E.; Ohayon-Courtès, C.; l’Azou, B. Cytotoxicity and oxidative stress induced by different metallic nanoparticles on human kidney cells. Part. Fibre Toxicol. 2011, 8, 10. [Google Scholar] [CrossRef] [PubMed]
- El-Said, K.S.; Ali, E.M.; Kanehira, K.; Taniguchi, A. Molecular mechanism of DNA damage induced by titanium dioxide nanoparticles in toll-like receptor 3 or 4 expressing human hepatocarcinoma cell lines. J. Nanobiotechnol. 2014, 12, 48. [Google Scholar] [CrossRef]
- Liu, S.; Xu, L.; Zhang, T.; Ren, G.; Yang, Z. Oxidative stress and apoptosis induced by nanosized titanium dioxide in PC12 cells. Toxicology 2010, 267, 172–177. [Google Scholar] [CrossRef]
- Faedmaleki, F.; Shirazi, F.; Ejtemaeimehr, S.; Anjarani, S.; Salarian, A.A.; Ashtiani, H.; Rastegar, H. Study of silymarin and Vitamin E protective effects on silver nanoparticle toxicity on mice liver primary cell culture. Acta Med. Iran. 2016, 54, 85–95. [Google Scholar]
- Jiang, Y.; Yu, X.; Su, C.; Zhao, L.; Shi, Y. Chitosan nanoparticles induced the antitumor effect in hepatocellular carcinoma cells by regulating ROS-mediated mitochondrial damage and endoplasmic reticulum stress. Artif. Cells Nanomed. Biotechnol. 2019, 47, 747–756. [Google Scholar] [CrossRef]
- Miethling-Graff, R.; Rumpker, R.; Richter, M.; Verano-Braga, T.; Kjeldsen, F.; Brewer, J.; Hoyland, J.; Rubahn, H.-G.; Er, H. Exposure to silver nanoparticles induces size- and dose-dependent oxidative stress and cytotoxicity in human colon carcinoma cells. Toxicol. Vitr. 2014, 28, 1280–1289. [Google Scholar] [CrossRef]
- Pratibha, R.; Sameer, R.; Rataboli, P.V.; Bhiwgade, D.A.; Dhume, C.Y. Enzymatic studies of cisplatin induced oxidative stress in hepatic tissue of rats. Eur. J. Pharmacol. 2006, 532, 290–293. [Google Scholar] [CrossRef]
- Abouzeinab, N. Cytoprotective effect and antioxidant properties of silymarin on cisplatin induced hepatotoxicity in rats: A biochemical and histochemical study. Int. J. Cancer Res. 2013, 9, 9–23. [Google Scholar] [CrossRef]
- Katarina, R.; Isidora, S.; Vladimir, Z.; Ivan, S.; Nevena, J.; Vladimir, J.; Dragan, D.; Slobodan, N. The Platinum (II) complexes induced oxidative stress of isolated rat heart. Serb. J. Exp. Clin. Res. 2017, 18, 111–117. [Google Scholar]
- Silici, S.; Ekmekcioglu, O.; Kanbur, M.; Deniz, K. The protective effect of royal jelly against cisplatin-induced renal oxidative stress in rats. World J. Urol. 2011, 29, 127–132. [Google Scholar] [CrossRef]
- Ateyya, H.; Yosef, H.; Nader, M.A. Ameliorative effect of trimetazidine on cisplatin-induced hepatotoxicity in rats. Can. J. Physiol. Pharmacol. 2016, 94, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.H.; Cha, H.-J.; Hwang-Bo, H.; Kim, M.Y.; Kim, S.Y.; Ji, S.Y.; Cheong, J.; Park, C.; Lee, H.; Kim, G.-Y.; et al. Anti-proliferative and pro-apoptotic effects of Licochalcone A through ROS-mediated cell cycle arrest and apoptosis in human bladder cancer cells. Int. J. Mol. Sci. 2019, 20, 3820. [Google Scholar] [CrossRef] [PubMed]
- Shaban, N.Z.; El-Kersh, M.A.L.; El-Rashidy, F.H.; Habashy, N.H. Protective role of Punica granatum (pomegranate) peel and seed oil extracts on diethylnitrosamine and phenobarbital-induced hepatic injury in male rats. Food Chem. 2013, 141, 1587–1596. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, T.; Maishu, S.P.; Akter, R.; Rahman, M.H.; Akhtar, M.F.; Saleem, A.; Bin-Jumah, M.; Kamel, M.; Abdel-Latif, M.A.; Abdel-Daim, M.M. A review on natural sources derived protein nanoparticles as anticancer agents. Curr. Top. Med. Chem. 2021, 21, 1014–1026. [Google Scholar] [CrossRef]
- Weinstein, J.N. Cell lines battle cancer. Nature 2012, 483, 544–545. [Google Scholar] [CrossRef]
- Mirabelli, P.; Coppola, L.; Salvatore, M. Cancer cell lines are useful model systems for medical research. Cancers 2019, 11, 1098. [Google Scholar] [CrossRef]
- Kärber, G. Beitrag zur kollektiven Behandlung pharmakologischer Reihenversuche. Naunyn-Schmiedebergs Arch. für Exp. Pathol. und Pharmakol. 1931, 162, 480–483. [Google Scholar] [CrossRef]
- Baravalia, Y.; Vaghasiya, Y.; Chanda, S. Brine shrimp cytotoxicity, anti-inflammatory and analgesic properties of woodfordia fruticosa Kurz flowers. Iran. J. Pharm. Res. IJPR 2012, 11, 851–861. [Google Scholar]
- Galvao, J.; Davis, B.; Tilley, M.; Normando, E.; Duchen, M.R.; Cordeiro, M.F. Unexpected low-dose toxicity of the universal solvent DMSO. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2014, 28, 1317–1330. [Google Scholar] [CrossRef]
- Wang, Y.; Huang, F.; Cai, H.; Wu, Y.; He, G.; Tan, W.-S. The efficacy of combination therapy using adeno-associated virus-TRAIL targeting to telomerase activity and cisplatin in a mice model of hepatocellular carcinoma. J. Cancer Res. Clin. Oncol. 2010, 136, 1827–1837. [Google Scholar] [CrossRef]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Ellman, G.L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 1959, 82, 70–77. [Google Scholar] [CrossRef]
- Rotruck, J.T.; Pope, A.L.; Ganther, H.E.; Swanson, A.; Hafeman, D.G.; Hoekstra, W. Selenium: Biochemical role as a component of glutathione peroxidase. Science 1973, 179, 588–590. [Google Scholar] [CrossRef]
- Bergmeyer, H.; Bergmeyer, J.; Grassl, M. Methods of Enzymatic Analysis, 3rd ed; Verlag Chemie: Weinheim, Germany, 1983; Volume VI. [Google Scholar]
- Habig, W.H.; Pabst, M.J.; Jakoby, W.B. Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J. Biol. Chem. 1974, 249, 7130–7139. [Google Scholar] [CrossRef]
- Marklund, S.; Marklund, G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 1974, 47, 469–474. [Google Scholar] [CrossRef]
- Reitman, S.; Frankel, S. A colorimetric method for the determination of serum glutamic oxalacetic and glutamic pyruvic transaminases. Am. J. Clin. Pathol. 1957, 28, 56–63. [Google Scholar] [CrossRef]
- Doumas, B.T.; Ard Watson, W.; Biggs, H.G. Albumin standards and the measurement of serum albumin with bromcresol green. Clin. Chim. Ccta; Int. J. Clin. Chem. 1971, 31, 87–96. [Google Scholar] [CrossRef]
- Gornall, A.G.; Bardawill, C.J.; David, M.M. Determination of serum proteins by means of the biuret reaction. J. Biol. Chem. 1949, 177, 751–766. [Google Scholar] [CrossRef]
- Burstein, M.; Scholnick, H.; Morfin, R. Rapid method for the isolation of lipoproteins from human serum by precipitation with polyanions. J. Lipid Res. 1970, 11, 583–595. [Google Scholar] [CrossRef]
- Fossati, P.; Prencipe, L. Serum triglycerides determined colorimetrically with an enzyme that produces hydrogen peroxide. Clin. Chem. 1982, 28, 2077–2080. [Google Scholar] [CrossRef]
- Henry, R.J.; Winkelman, J.W.; Cannon, D.C. Clinical Chemistry; Principles and Technics; Medical Dept., Harper & Row: Hagerstown, MD, USA, 1974; p. 1629. [Google Scholar]
- Patton, C.J.; Crouch, S. Spectrophotometric and kinetics investigation of the Berthelot reaction for the determination of ammonia. Anal. Chem. 1977, 49, 464–469. [Google Scholar] [CrossRef]
- Suzuki, H.; Suzuki, K. Rat hypoplastic kidney (hpk/hpk) induces renal anemia, hyperparathyroidism, and osteodystrophy at the end stage of renal failure. J. Vet. Med Sci. 1998, 60, 1051–1058. [Google Scholar] [CrossRef][Green Version]
- Shanmugasundaram, T.; Radhakrishnan, M.; Gopikrishnan, V.; Kadirvelu, K.; Balagurunathan, R. Biocompatible silver, gold and silver/gold alloy nanoparticles for enhanced cancer therapy: In vitro and in vivo perspectives. Nanoscale 2017, 9, 16773–16790. [Google Scholar] [CrossRef]


| Groups | Dose (mg/kg) | Dose Difference (a) | No. of Rats | LD50 of DBT | LD50 of DBT–CSNPs | ||||
|---|---|---|---|---|---|---|---|---|---|
| No. of Dead | Mean Mortality (b) | Product (a × b) | No. of Dead | Mean Mortality (b) | Product (a × b) | ||||
| 1 | 200 | - | 4 | 0 | - | - | 0 | - | - |
| 2 | 400 | 200 | 4 | 1 | 0.5 | 100 | 0 | - | - |
| 3 | 800 | 400 | 4 | 1 | 1 | 400 | 1 | 0.5 | 200 |
| 4 | 1200 | 400 | 4 | 2 | 1.5 | 600 | 1 | 1 | 400 |
| 5 | 2000 | 800 | 4 | 3 | 2.5 | 2000 | 2 | 1.5 | 1200 |
| 6 | 3000 | 1000 | 4 | 4 | 3.5 | 3500 | 4 | 3 | 3000 |
| Sum of product | 6600 | 4800 | |||||||
| LD50 | LD50 = 3000 ‒ (6600/4) = 1350 mg/kg | LD50 = 3000 − (4800/4) = 1800 mg/kg | |||||||
| Parameters | C | CSNPs | DBT | DBT–CSNPs | CCl4 | CCl4-CSNPs | CCl4-DBT | CCl4-DBT–CSNPs | CCl4-Cisplatin |
|---|---|---|---|---|---|---|---|---|---|
| ALT (U/L) | 117.72 ± 2.54 | 118.3 ± 2.56 | 120.58 ± 3.74 | 122.5 ± 2.81 | 265.52 ± 4.33 * | 258.31 ± 3.89 *α | 188.33 ± 2.74 *#α | 157.92 ± 6.09 *# | 215.8 ± 6.8 *#α |
| AST (U/L) | 146.19 ± 3.86 | 148.3 ± 3.69 | 152.78 ± 5.4 | 150.7 ± 4.7 | 302.1 ± 4.27 * | 293.61 ± 3.89 *α | 224.1 ± 3.25 *#α | 187.49 ± 4.14 *# | 259.58 ± 6.07 *#α |
| ALP (U/L) | 252.85 ± 2.08 | 254.25 ± 2.09 | 257.8 ± 4.1 | 255.85± 4.29 | 456.31 ± 5.58 * | 446.49 ± 5.41 *α | 352.16 ± 4.05 *#α | 314.9 ± 3.44 *# | 375.2 ± 5.72 *#α |
| TP (g/dl) | 5.89 ± 0.042 | 5.87 ± 0.037 | 5.80 ± 0.035 | 5.84 ± 0.037 | 4.99 ± 0.033 * | 5.06 ± 0.039 *α | 5.57 ± 0.045 *# | 5.67 ± 0.035 *# | 5.08 ± 0.095 *α |
| Liver TP (mg/g tissue) | 143.1 ± 7.38 | 143.6 ± 3.92 | 139.8 ± 4.34 | 141.2 ± 3.77 | 91.03 ± 5.88 * | 87.21 ± 5.22 *α | 105.2 ± 7.15 *# | 121.1 ± 5.23 *# | 83.5 ± 4.75 *α |
| Albumin (g/dL) | 4.99 ± 0.33 | 4.91 ± 0.28 | 4.88 ± 0.063 | 4.84 ± 0.057 | 3.48 ± 0.15 * | 3.57 ± 0.14 *α | 3.91 ± 0.025 *# | 3.77 ± 0.057 *# | 3.35 ± 0.054 *α |
| Cholesterol (mg/dL) | 88.75 ± 2.39 | 90.3± 2.31 | 91.17± 3.98 * | 93.08 ± 2.16 | 165.44 ± 2.32 * | 160.7 ± 4.21 *α | 131.11 ± 2.61 *# | 128.94 ± 2.57 *# | 153.78 ± 2.39 *#α |
| TG (mg/dL) | 87.49 ± 1.58 | 89.2 ± 1.62 | 90.08 ± 2.16 | 92.07 ± 2.8 | 148.03 ± 2.27 * | 145.9 ± 3.26 *α | 128.75 ± 1.67 *#α | 123.84 ± 2.78 *# | 133.3 ± 1.9 *#α |
| LDL–cholesterol(mg/dL) | 55.61 ± 1.5 | 56.69 ± 1.54 | 61.19 ± 1.96 | 58.69 ± 1.56 | 93.1± 1.87 * | 90.2 ± 2.83 *α | 78.28 ± 2.2 *#α | 70.79 ± 1.45 *# | 86.59 ± 2.01 *#α |
| HDL–cholesterol (mg/dL) | 47.61 ± 1.45 | 45.2 ± 1.42 | 44.37 ± 1.51 | 42.88 ± 1.86 | 27.95 ± 2.09 * | 28.9 ± 1.93 *α | 40.22 ± 1.17 *# | 41.42 ± 0.87 *# | 33.74 ± 1.74 *#α |
| Urea (mg/dL) | 43.79 ± 2.16 | 45.2 ± 2.19 | 46.74 ± 2.35 * | 45.07 ± 3.96 | 73.21 ± 2.98 * | 71.6 ± 1.09 *α | 65.76 ± 3.19 *#α | 58.74 ± 2.35 *# | 69.74 ± 2.26 *α |
| Creatinine (mg/dL) | 0.95 ± 0.02 | 0.97 ± 0.13 | 0.99 ± 0.108 | 1.03 ± 0.04 | 1.5 ± 0.068 * | 1.42 ± 0.071 *α | 1.38 ± 0.049 *#α | 1.21 ± 0.027 *# | 1.44 ± 0.04 *α |
| Gene | Forward Primer (5′ ------ 3′) | Reverse Primer (5′ ------ 3′) |
|---|---|---|
| Bcl-2 | ATCGCTCTGTGGATGACTGAGTAC | AGAGACAGCCAGGAGAAATCAAAC |
| Bax | ACACCTGAGCTGACCTTG | AGCCCATGATGGTTCTGATC |
| Caspase-8 | CTGGGAAGGATCGACGATTA | CATGTCCTGCATTTTGATGG |
| β-actin | AAGTCCCTCACCCTCCCAAAAG | AAGCAATGCTGTCACCTTCCC |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shaban, N.Z.; Yehia, S.A.; Awad, D.; Shaban, S.Y.; Saleh, S.R. A Titanium (IV)–Dithiophenolate Complex and Its Chitosan Nanocomposite: Their Roles towards Rat Liver Injuries In Vivo and against Human Liver Cancer Cell Lines. Int. J. Mol. Sci. 2021, 22, 11219. https://doi.org/10.3390/ijms222011219
Shaban NZ, Yehia SA, Awad D, Shaban SY, Saleh SR. A Titanium (IV)–Dithiophenolate Complex and Its Chitosan Nanocomposite: Their Roles towards Rat Liver Injuries In Vivo and against Human Liver Cancer Cell Lines. International Journal of Molecular Sciences. 2021; 22(20):11219. https://doi.org/10.3390/ijms222011219
Chicago/Turabian StyleShaban, Nadia Z., Salah A. Yehia, Doaa Awad, Shaban Y. Shaban, and Samar R. Saleh. 2021. "A Titanium (IV)–Dithiophenolate Complex and Its Chitosan Nanocomposite: Their Roles towards Rat Liver Injuries In Vivo and against Human Liver Cancer Cell Lines" International Journal of Molecular Sciences 22, no. 20: 11219. https://doi.org/10.3390/ijms222011219
APA StyleShaban, N. Z., Yehia, S. A., Awad, D., Shaban, S. Y., & Saleh, S. R. (2021). A Titanium (IV)–Dithiophenolate Complex and Its Chitosan Nanocomposite: Their Roles towards Rat Liver Injuries In Vivo and against Human Liver Cancer Cell Lines. International Journal of Molecular Sciences, 22(20), 11219. https://doi.org/10.3390/ijms222011219