The Role of Peritoneal Macrophages in Endometriosis
Abstract
1. Introduction
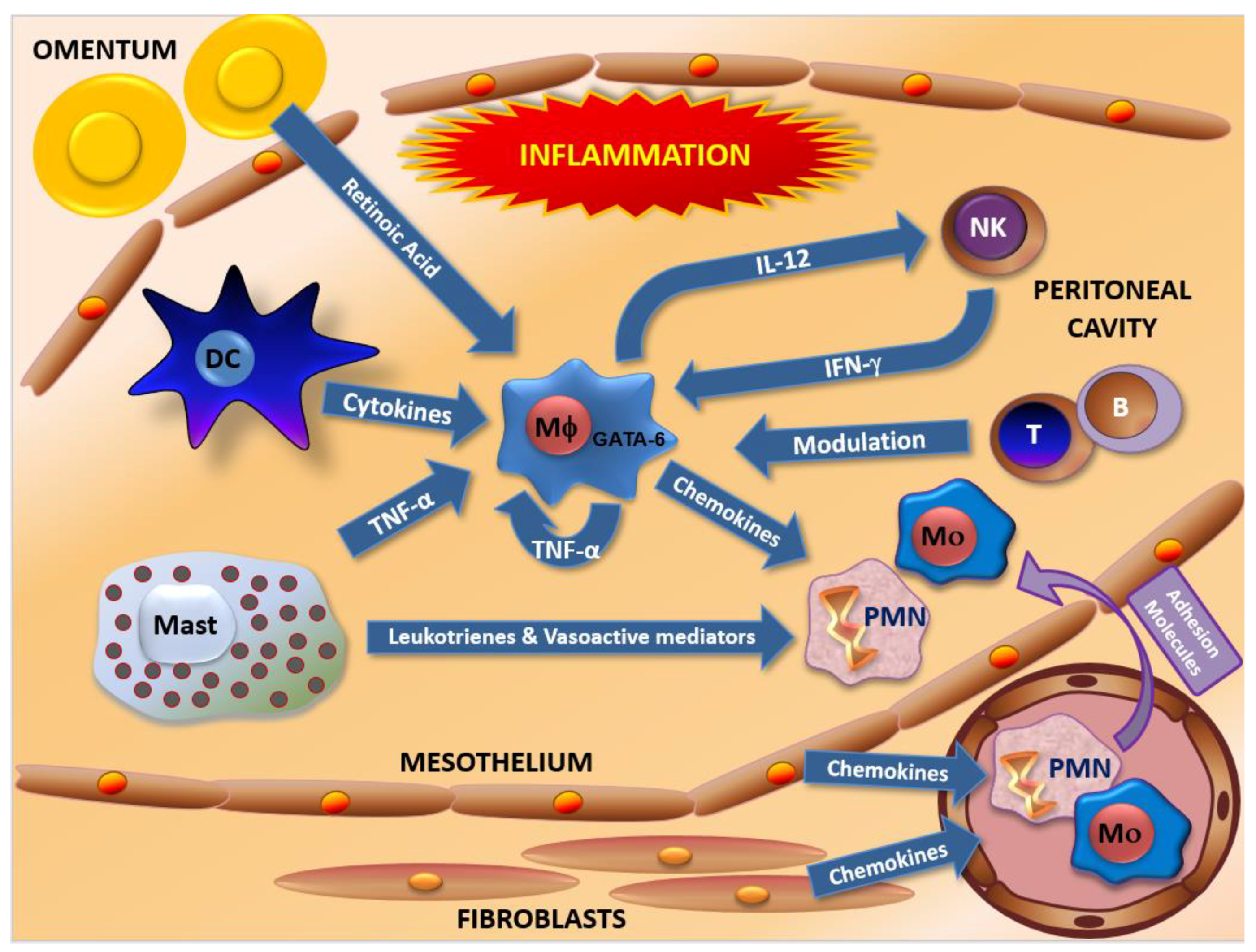
2. Human Peritoneal Host Defenses
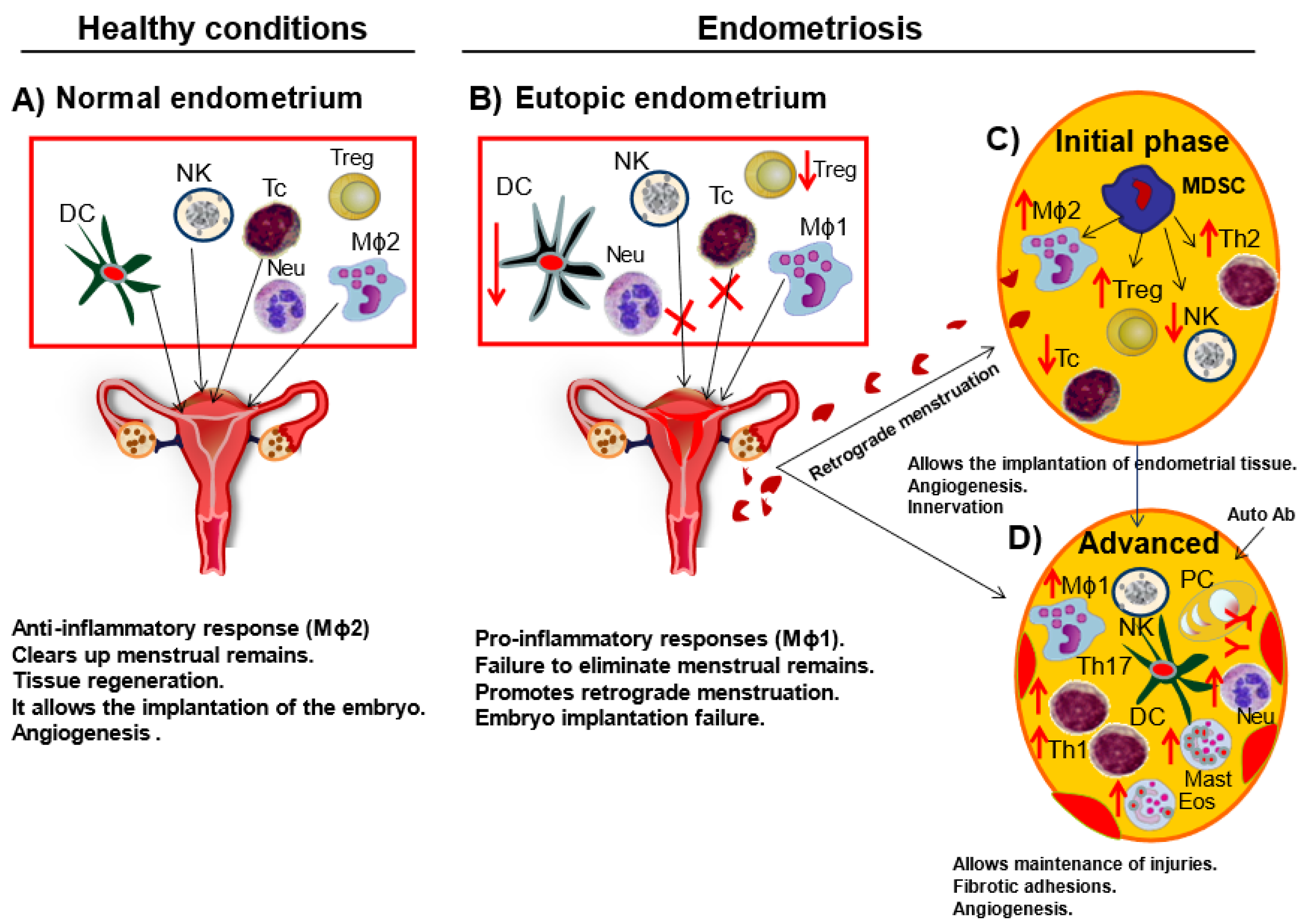
3. Macrophage Phenotype in Endometriosis
4. Phenotype of Macrophages in Animal Models of Endometriosis
5. Role of Cytokines Secreted by Macrophages in Endometriosis
6. Molecules Involved in the Recruitment of Peritoneal Macrophages in Endometriosis
7. Phagocytosis Defects of Macrophages in Endometriosis
8. Dependence of Estrogens and Their Receptors in the Development of Endometriosis
9. Inflammation, Innervation, and Associated Pain in Endometriosis
10. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zondervan, K.T.; Becker, C.M.; Koga, K.; Missmer, S.A.; Taylor, R.N.; Viganò, P. Endometriosis. Nat. Rev. Dis. Primers 2018, 4, 9. [Google Scholar] [CrossRef] [PubMed]
- As-Sanie, S.; Black, R.; Giudice, L.C.; Gray Valbrun, T.; Gupta, J.; Jones, B.; Laufer, M.R.; Milspaw, A.T.; Missmer, S.A.; Norman, A.; et al. Assessing Research Gaps and Unmet Needs in Endometriosis. Am. J. Obstet. Gynecol. 2019, 221, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Da Broi, M.G.; Ferriani, R.A.; Navarro, P.A. Ethiopathogenic Mechanisms of Endometriosis-Related. JBRA Assist. Reprod. 2019, 23, 273. [Google Scholar] [CrossRef]
- Prescott, J.; Farland, L.V.; Tobias, D.K.; Gaskins, A.J.; Spiegelman, D.; Chavarro, J.E.; Rich-Edwards, J.W.; Barbieri, R.L.; Missmer, S.A. A Prospective Cohort Study of Endometriosis and Subsequent Risk of Infertility. Hum. Reprod. 2016, 31, 1475–1482. [Google Scholar] [CrossRef] [PubMed]
- Adoamnei, E.; Morán-Sánchez, I.; Sánchez-Ferrer, M.L.; Mendiola, J.; Prieto-Sánchez, M.T.; Moñino-García, M.; Palomar-Rodríguez, J.A.; Torres-Cantero, A.M. Health-Related Quality of Life in Adult Spanish Women with Endometriomas or Deep Infiltrating Endometriosis: A Case-Control Study. Int. J. Environ. Res. Public Health 2021, 18, 5586. [Google Scholar] [CrossRef]
- Armour, M.; Lawson, K.; Wood, A.; Smith, C.A.; Abbott, J. The Cost of Illness and Economic Burden of Endometriosis and Chronic Pelvic Pain in Australia: A National Online Survey. PLoS ONE 2019, 14, e0223316. [Google Scholar] [CrossRef]
- Simoens, S.; Dunselman, G.; Dirksen, C.; Hummelshoj, L.; Bokor, A.; Brandes, I.; Brodszky, V.; Canis, M.; Colombo, G.L.; Deleire, T.; et al. The Burden of Endometriosis: Costs and Quality of Life of Women with Endometriosis and Treated in Referral Centres. Hum. Reprod. 2012, 27, 1292–1299. [Google Scholar] [CrossRef]
- Kajiyama, H.; Suzuki, S.; Yoshihara, M.; Tamauchi, S.; Yoshikawa, N.; Niimi, K.; Shibata, K.; Kikkawa, F. Endometriosis and Cancer. Free Radic. Biol. Med. 2019, 133, 186–192. [Google Scholar] [CrossRef]
- Guo, S.W. Cancer-Associated Mutations in Endometriosis: Shedding Light on the Pathogenesis and Pathophysiology. Hum. Reprod. Update 2020, 26, 423–449. [Google Scholar] [CrossRef]
- Iwabuchi, T.; Yoshimoto, C.; Shigetomi, H.; Kobayashi, H. Oxidative Stress and Antioxidant Defense in Endometriosis and Its Malignant Transformation. Oxidative Med. Cell. Longev. 2015, 2015, 848595. [Google Scholar] [CrossRef]
- Yamada, Y.; Uchiyama, T.; Ito, F.; Kawahara, N.; Ogawa, K.; Obayashi, C.; Kobayashi, H. Clinical Significance of M2 Macrophages Expressing Heme Oxygenase-1 in Malignant Transformation of Ovarian Endometrioma. Pathol. Res. Pract. 2019, 215, 639–643. [Google Scholar] [CrossRef]
- Akashi, K.; Nagashima, Y.; Tabata, T.; Oda, H. Immunochemical Analysis of Iron Transporters and M2 Macrophages in Ovarian Endometrioma and Clear Cell Adenocarcinoma. Mol. Clin. Oncol. 2021, 15, 159. [Google Scholar] [CrossRef]
- Bedaiwy, M.A.; Alfaraj, S.; Yong, P.; Casper, R. New Developments in the Medical Treatment of Endometriosis. Fertil. Steril. 2017, 107, 555–565. [Google Scholar] [CrossRef]
- Sampson, J.A. The Development of the Implantation Theory for the Origin of Peritoneal Endometriosis. Am. J. Obstet. Gynecol. 1940, 40, 549–557. [Google Scholar] [CrossRef]
- Halme, J.; Hammond, M.G.; Hulka, J.F.; Raj, S.G.; Talbert, L.M. Retrograde Menstruation in Healthy Women and in Patients with Endometriosis. Obstet. Gynecol. 1984, 64, 151–154. [Google Scholar]
- Arafah, M.; Rashid, S.; Akhtar, M. Endometriosis: A Comprehensive Review. Adv. Anat. Pathol. 2021, 28, 30–43. [Google Scholar] [CrossRef] [PubMed]
- Gruenwald, P. Origin of Endometriosis from the Mesenchyme of the Celomic Walls. Am. J. Obstet. Gynecol. 1942, 44, 470–474. [Google Scholar] [CrossRef]
- Bouquet De Joliniere, J.; Ayoubi, J.M.; Lesec, G.; Validire, P.; Goguin, A.; Gianaroli, L.; Dubuisson, J.B.; Feki, A.; Gogusev, J. Identification of Displaced Endometrial Glands and Embryonic Duct Remnants in Female Fetal Reproductive Tract: Possible Pathogenetic Role in Endometriotic and Pelvic Neoplastic Processes. Front. Physiol. 2012, 3, 444. [Google Scholar] [CrossRef]
- Asghari, S.; Valizadeh, A.; Aghebati-Maleki, L.; Nouri, M.; Yousefi, M. Endometriosis: Perspective, Lights, and Shadows of Etiology. Biomed. Pharmacother. 2018, 106, 163–174. [Google Scholar] [CrossRef]
- Sourial, S.; Tempest, N.; Hapangama, D.K. Theories on the Pathogenesis of Endometriosis. Int. J. Reprod. Med. 2014, 2014, 179515. [Google Scholar] [CrossRef] [PubMed]
- Borghese, B.; Zondervan, K.T.; Abrao, M.S.; Chapron, C.; Vaiman, D. Recent Insights on the Genetics and Epigenetics of Endometriosis. Clin. Genet. 2017, 91, 254–264. [Google Scholar] [CrossRef]
- Matalliotakis, M.; Zervou, M.I.; Matalliotaki, C.; Rahmioglu, N.; Koumantakis, G.; Kalogiannidis, I.; Prapas, I.; Zondervan, K.; Spandidos, D.A.; Matalliotakis, I.; et al. The Role of Gene Polymorphisms in Endometriosis. Mol. Med. Rep. 2017, 16, 5881–5886. [Google Scholar] [CrossRef] [PubMed]
- García-Peñarrubia, P.; Ruiz-Alcaraz, A.J.; Martínez-Esparza, M.; Marín, P.; Machado-Linde, F. Hypothetical Roadmap towards Endometriosis: Prenatal Endocrine-Disrupting Chemical Pollutant Exposure, Anogenital Distance, Gut-Genital Microbiota and Subclinical Infections. Hum. Reproduct. Update 2020, 26, 214–246. [Google Scholar] [CrossRef] [PubMed]
- Cano-Sancho, G.; Ploteau, S.; Matta, K.; Adoamnei, E.; Louis, G.B.; Mendiola, J.; Darai, E.; Squifflet, J.; le Bizec, B.; Antignac, J.P. Human Epidemiological Evidence about the Associations between Exposure to Organochlorine Chemicals and Endometriosis: Systematic Review and Meta-Analysis. Environ. Int. 2019, 123, 209–223. [Google Scholar] [CrossRef]
- Wen, X.; Xiong, Y.; Qu, X.; Jin, L.; Zhou, C.; Zhang, M.; Zhang, Y. The Risk of Endometriosis after Exposure to Endocrine-Disrupting Chemicals: A Meta-Analysis of 30 Epidemiology Studies. Gynecol. Endocrinol. 2019, 35, 645–650. [Google Scholar] [CrossRef] [PubMed]
- Symons, L.K.; Miller, J.E.; Kay, V.R.; Marks, R.M.; Liblik, K.; Koti, M.; Tayade, C. The Immunopathophysiology of Endometriosis. Trends Mol. Med. 2018, 24, 748–762. [Google Scholar] [CrossRef]
- Zhang, T.; de Carolis, C.; Man, G.C.W.; Wang, C.C. The Link between Immunity, Autoimmunity and Endometriosis: A Literature Update. Autoimmun. Rev. 2018, 17, 945–955. [Google Scholar] [CrossRef]
- Hogg, C.; Horne, A.W.; Greaves, E. Endometriosis-Associated Macrophages: Origin, Phenotype, and Function. Front. Endocrinol. 2020, 11, 7. [Google Scholar] [CrossRef]
- Akoum, A.; Kong, J.; Metz, C.; Beaumont, M.C. Spontaneous and Stimulated Secretion of Monocyte Chemotactic Protein-1 and Macrophage Migration Inhibitory Factor by Peritoneal Macrophages in Women with and without Endometriosis. Fertil. Steril. 2002, 77, 989–994. [Google Scholar] [CrossRef]
- Xie, Q.; He, H.; Wu, Y.H.; Zou, L.J.; She, X.L.; Xia, X.M.; Wu, X.Q. Eutopic Endometrium from Patients with Endometriosis Modulates the Expression of CD36 and SIRP-α in Peritoneal Macrophages. J. Obstet. Gynaecol. Res. 2019, 45, 1045–1057. [Google Scholar] [CrossRef] [PubMed]
- Khan, K.N.; Kitajima, M.; Hiraki, K.; Fujishita, A.; Sekine, I.; Ishimaru, T.; Masuzaki, H. Immunopathogenesis of Pelvic Endometriosis: Role of Hepatocyte Growth Factor, Macrophages and Ovarian Steroids. Am. J. Reprod. Immunol. 2008, 60, 383–404. [Google Scholar] [CrossRef]
- Takamura, M.; Koga, K.; Izumi, G.; Hirata, T.; Harada, M.; Hirota, Y.; Hiraike, O.; Fujii, T.; Osuga, Y. Simultaneous Detection and Evaluation of Four Subsets of CD4+ T Lymphocyte in Lesions and Peripheral Blood in Endometriosis. Am. J. Reprod. Immunol. 2015, 74, 480–486. [Google Scholar] [CrossRef]
- Da Gama Coelho Riccio, L.; Santulli, P.; Marcellin, L.; Abrão, M.S.; Batteux, F.; Chapron, C. Immunology of Endometriosis. Best Pract. Res. Clin. Obstet. Gynaecol. 2018, 50, 39–49. [Google Scholar] [CrossRef]
- Maeda, N.; Izumiya, C.; Oguri, H.; Kusume, T.; Yamamoto, Y.; Fukaya, T. Aberrant Expression of Intercellular Adhesion Molecule-1 and Killer Inhibitory Receptors Induces Immune Tolerance in Women with Pelvic Endometriosis. Fertil. Steril. 2002, 77, 679–683. [Google Scholar] [CrossRef]
- Wang, J.; Kubes, P. A Reservoir of Mature Cavity Macrophages That Can Rapidly Invade Visceral Organs to Affect Tissue Repair. Cell 2016, 165, 668–678. [Google Scholar] [CrossRef]
- Mutsaers, S.E.; Birnie, K.; Lansley, S.; Herrick, S.E.; Lim, C.B.; Prêle, C.M. Mesothelial Cells in Tissue Repair and Fibrosis. Front. Pharmacol. 2015, 6, 113. [Google Scholar] [CrossRef] [PubMed]
- Broche, F.; Tellado, J.M. Defense Mechanisms of the Peritoneal Cavity. Curr. Opin. Crit. Care 2001, 7, 105–116. [Google Scholar] [CrossRef] [PubMed]
- Jantz, M.A.; Antony, V.B. Pathophysiology of the Pleura. Respiration 2008, 75, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Jonjić, N.; Peri, G.; Bernasconi, S.; Sciacca, F.L.; Colotta, F.; PierGiuseppe, P.; Lanfrancone, L.; Mantovani, A. Expression of Adhesion Molecules and Chemotactic Cytokines in Cultured Human Mesothelial Cells. J. Exp. Med. 1992, 176, 1165–1174. [Google Scholar] [CrossRef]
- Cannistra, S.A.; Ottensmeier, C.; Tidy, J.; DeFranzo, B. Vascular Cell Adhesion Molecule-1 Expressed by Peritoneal Mesothelium Partly Mediates the Binding of Activated Human T Lymphocytes. Exp. Hematol. 1994, 22, 996–1002. [Google Scholar]
- Yung, S.; Chao, T.M. Intrinsic Cells: Mesothelial Cells—Central Players in Regulating Inflammation and Resolution. Proc. Perit. Dial. Int. 2009, 29, 21–27. [Google Scholar]
- Faull, R.J. Peritoneal Defenses against Infection: Winning the Battle but Losing the War? Semin. Dial. 2000, 13, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Okabe, Y.; Medzhitov, R. Tissue-Specific Signals Control Reversible Program of Localization and Functional Polarization of Macrophages. Cell 2014, 157, 832–844. [Google Scholar] [CrossRef] [PubMed]
- Rosas, M.; Davies, L.C.; Giles, P.J.; Liao, C.T.; Kharfan, B.; Stone, C.-T.; O’Donnell, V.B.; Fraser, D.J.; Jones, S.A.; Taylor, P.R. The Transcription Factor Gata6 Links Tissue Macrophage Phenotype and Proliferative Renewal. Science 2014, 344, 645–648. [Google Scholar] [CrossRef] [PubMed]
- Gautier, E.L.; Ivanov, S.; Williams, J.W.; Huang, S.C.C.; Marcelin, G.; Fairfax, K.; Wang, P.L.; Francis, J.S.; Leone, P.; Wilson, D.B.; et al. Gata6 Regulates Aspartoacylase Expression in Resident Peritoneal Macrophages and Controls Their Survival. J. Exp. Med. 2014, 211, 1525–1531. [Google Scholar] [CrossRef] [PubMed]
- Ziegler-Heitbrock, L. Reprint of: Monocyte Subsets in Man and Other Species. Cell. Immunol. 2014, 291, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Alcaraz, A.J.; Martínez-Banaclocha, H.; Marín-Sánchez, P.; Carmona-Martínez, V.; Iniesta-Albadalejo, M.A.; Tristán-Manzano, M.; Tapia-Abellán, A.; García-Peñarrubia, P.; Machado-Linde, F.; Pelegrín, P.; et al. Isolation of Functional Mature Peritoneal Macrophages from Healthy Humans. Immunol. Cell Biol. 2020, 98, 114–126. [Google Scholar] [CrossRef]
- Ruiz-Alcaraz, A.J.; Carmona-Martínez, V.; Tristán-Manzano, M.; Machado-Linde, F.; Sánchez-Ferrer, M.L.; García-Peñarrubia, P.; Martínez-Esparza, M. Characterization of Human Peritoneal Monocyte/Macrophage Subsets in Homeostasis: Phenotype, GATA6, Phagocytic/Oxidative Activities and Cytokines Expression. Sci. Rep. 2018, 8, 12794. [Google Scholar] [CrossRef]
- Kubicka, U.; Olszewski, W.L.; Tarnowski, W.; Bielecki, K.; Ziółkowska, A.; Wierzbicki, Z. Normal Human Immune Peritoneal Cells: Subpopulations and Functional Characteristics. Scand. J. Immunol. 1996, 44, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, C.S.; Bomalaski, J.S.; Zurier, R.B.; Neilson, E.G.; Douglas, S.D. Analysis of Peritoneal Macrophages in Continuous Ambulatory Peritoneal Dialysis Patients. Kidney Int. 1984, 26, 733–740. [Google Scholar] [CrossRef]
- Peterson, P.K.; Gaziano, E.; Suh, H.J.; Devalon, M.; Peterson, L.; Keane, W.F. Antimicrobial Activities of Dialysate-Elicited and Resident Human Peritoneal Macrophages. Infect. Immun. 1985, 49, 212–218. [Google Scholar] [CrossRef]
- McGregor, S.J.; Brock, J.H.; Briggs, J.D.; Junor, B.J.R. Bactericidal Activity of Peritoneal Macrophages from Continuous Ambulatory Dialysis Patients. Nephrol. Dial. Transplant. 1987, 2, 104–108. [Google Scholar] [CrossRef]
- Newman, S.L.; Becker, S.; Halme, J. Phagocytosis by Receptors for C3b (CR1), IC3b (CR3) and IgG (Fc) on Human Peritoneal Macrophages. J. Leukoc. Biol. 1985, 38, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.M.M.; Bomford, A.; Nouri-Aria, K.T.; Davies, T.; Smith, R.; Williams, R. Peritoneal Macrophages from Patients with Cirrhotic Ascites Show Impaired Phagocytosis and Vigorous Respiratory Burst. Results Immunol. 2011, 1, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Schukfeh, N.; Elyas, A.; Viemann, D.; Ure, B.M.; Froemmel, S.; Park, J.K.; Kuebler, J.F.; Vieten, G. Phenotypic Switch of Human Peritoneal Macrophages during Childhood. Eur. J. Pediatr. Surg. 2021, 31, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, J.; Maaßen, V.; Rieber, P.; Fricke, H. T Lymphocytes from Normal Human Peritoneum Are Phenotypically Different from Their Counterparts in Peripheral Blood and CD3−Lymphocyte Subsets Contain MRNA for the Recombination Activating Gene RAG-1. Eur. J. Immunol. 1995, 25, 2626–2631. [Google Scholar] [CrossRef] [PubMed]
- Tangye, S.G. To B1 or Not to B1: That Really Is Still the Question! Blood 2013, 121, 5109–5110. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Alcaraz, A.J.; Tapia-Abellán, A.; Fernández-Fernández, M.D.; Tristán-Manzano, M.; Hernández-Caselles, T.; Sánchez-Velasco, E.; Miras-López, M.; Martínez-Esparza, M.; García-Peñarrubia, P. A Novel CD14high CD16high Subset of Peritoneal Macrophages from Cirrhotic Patients Is Associated to an Increased Response to LPS. Mol. Immunol. 2016, 72, 28–36. [Google Scholar] [CrossRef]
- Nie, M.F.; Xie, Q.; Wu, Y.H.; He, H.; Zou, L.J.; She, X.L.; Wu, X.Q. Serum and Ectopic Endometrium from Women with Endometriosis Modulate Macrophage M1/M2 Polarization via the Smad2/Smad3 Pathway. J. Immunol. Res. 2018, 2018, 6285813. [Google Scholar] [CrossRef]
- Chen, P.; Bonaldo, P. Role of Macrophage Polarization in Tumor Angiogenesis and Vessel Normalization. Implications for New Anticancer Therapies. In International Review of Cell and Molecular Biology; Elsevier Inc.: Amsterdam, The Netherlands, 2013; Volume 301, pp. 1–35. [Google Scholar]
- Laskin, D.L.; Sunil, V.R.; Gardner, C.R.; Laskin, J.D. Macrophages and Tissue Injury: Agents of Defense or Destruction? Annu. Rev. Pharmacol. Toxicol. 2011, 51, 267–288. [Google Scholar] [CrossRef]
- Gou, Y.; Li, X.; Li, P.; Zhang, H.; Xu, T.; Wang, H.; Wang, B.; Ma, X.; Jiang, X.; Zhang, Z. Estrogen Receptor β Upregulates CCL2 via NF-κ B Signaling in Endometriotic Stromal Cells and Recruits Macrophages to Promote the Pathogenesis of Endometriosis. Hum. Reprod. 2019, 34, 646–658. [Google Scholar] [CrossRef]
- Jensen, A.L.; Collins, J.; Shipman, E.P.; Wira, C.R.; Guyre, P.M.; Pioli, P.A. A Subset of Human Uterine Endometrial Macrophages Is Alternatively Activated. Am. J. Reprod. Immunol. 2012, 68, 374–386. [Google Scholar] [CrossRef]
- Ono, Y.; Yoshino, O.; Hiraoka, T.; Akiyama, I.; Sato, E.; Ito, M.; Kobayashi, M.; Nakashima, A.; Wada, S.; Onda, T.; et al. IL-33 Exacerbates Endometriotic Lesions via Polarizing Peritoneal Macrophages to M2 Subtype. Reprod. Sci. 2020, 27, 869–876. [Google Scholar] [CrossRef]
- Miller, J.E.; Monsanto, S.P.; Ahn, S.H.; Khalaj, K.; Fazleabas, A.T.; Young, S.L.; Lessey, B.A.; Koti, M.; Tayade, C. Interleukin-33 Modulates Inflammation in Endometriosis. Sci. Rep. 2017, 7, 17903. [Google Scholar] [CrossRef]
- Martinez, F.O.; Gordon, S. The M1 and M2 Paradigm of Macrophage Activation: Time for Reassessment. F1000Prime Rep. 2014, 6, 13. [Google Scholar] [CrossRef] [PubMed]
- Gibson, D.A.; Collins, F.; de Leo, B.; Horne, A.W.; Saunders, P.T.K. Peritoneal Macrophage Phenotype Correlates with Pain Scores in Women with Suspected Endometriosis. bioRxiv 2020. [Google Scholar] [CrossRef]
- Montagna, P.; Capellino, S.; Villaggio, B.; Remorgida, V.; Ragni, N.; Cutolo, M.; Ferrero, S. Peritoneal Fluid Macrophages in Endometriosis: Correlation between the Expression of Estrogen Receptors and Inflammation. Fertil. Steril. 2008, 90, 156–164. [Google Scholar] [CrossRef]
- Gogacz, M.; Gałczyński, K.; Wojtaś, M.; Winkler, I.; Adamiak, A.; Romanek-Piva, K.; Rechberger, T.; Kotarski, J. Fas-Related Apoptosis of Peritoneal Fluid Macrophages in Endometriosis Patients: Understanding the Disease. J. Immunol. Res. 2017, 2017, 3175394. [Google Scholar] [CrossRef]
- Weng, L.-C.; Hou, S.-H.; Lei, S.-T.; Peng, H.-Y.; Li, M.-Q.; Zhao, D. Estrogen-Regulated CD200 Inhibits Macrophage Phagocytosis in Endometriosis. J. Reprod. Immunol. 2020, 138, 103090. [Google Scholar] [CrossRef] [PubMed]
- Li, M.Z.; Wu, Y.H.; Ali, M.; Wu, X.Q.; Nie, M.F. Endometrial Stromal Cells Treated by Tumor Necrosis Factor-α Stimulate Macrophages Polarized toward M2 via Interleukin-6 and Monocyte Chemoattractant Protein-1. J. Obstet. Gynaecol. Res. 2020, 46, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Hudson, Q.J.; Ashjaei, K.; Perricos, A.; Kuessel, L.; Husslein, H.; Wenzl, R.; Yotova, I. Endometriosis Patients Show an Increased M2 Response in the Peritoneal CD14+low/CD68+low Macrophage Subpopulation Coupled with an Increase in the T-Helper 2 and T-Regulatory Cells. Reprod. Sci. 2020, 27, 1920–1931. [Google Scholar] [CrossRef]
- Clark, D.A.; Dmetrichuk, J.M.; Dhesy-Thind, S.; Crowther, M.A.; Arredondo, J.L. Soluble CD200 in Secretory Phase Endometriosis Endometrial Venules May Explain Endometriosis Pathophysiology and Provide a Novel Treatment Target. J. Reprod. Immunol. 2018, 129, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Epstein, F.H.; Moncada, S.; Higgs, A. The L-Arginine-Nitric Oxide Pathway. N. Engl. J. Med. 1993, 329, 2002–2012. [Google Scholar] [CrossRef]
- Osborn, B.H.; Haney, A.F.; Misukonis, M.A.; Weinberg, J.B. Inducible Nitric Oxide Synthase Expression by Peritoneal Macrophages in Endometriosis-Associated Infertility. Fertil. Steril. 2002, 77, 46–51. [Google Scholar] [CrossRef]
- Rosselli, M.; Dubey, R.K.; Imthurn, B.; Macas, E.; Keller, P.J. Andrology: Effects of Nitric Oxide on Human Spermatozoa: Evidence That Nitric Oxide Decreases Sperm Motility and Induces Sperm Toxicity. Hum. Reprod. 1995, 10, 1786–1790. [Google Scholar] [CrossRef]
- Sun, H.; Li, D.; Yuan, M.; Li, Q.; Zhen, Q.; Li, N.; Wang, G. Macrophages Alternatively Activated by Endometriosis-Exosomes Contribute to the Development of Lesions in Mice. Mol. Hum. Reprod. 2018, 25, 5–16. [Google Scholar] [CrossRef]
- Takebayashi, A.; Kimura, F.; Kishi, Y.; Ishida, M.; Takahashi, A.; Yamanaka, A.; Wu, D.; Zheng, L.; Takahashi, K.; Suginami, H.; et al. Subpopulations of Macrophages within Eutopic Endometrium of Endometriosis Patients. Am. J. Reprod. Immunol. 2015, 73, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Vallvé-Juanico, J.; Santamaria, X.; Vo, K.C.; Houshdaran, S.; Giudice, L.C. Macrophages Display Proinflammatory Phenotypes in the Eutopic Endometrium of Women with Endometriosis with Relevance to an Infectious Etiology of the Disease. Fertil. Steril. 2019, 112, 1118–1128. [Google Scholar] [CrossRef]
- Smith, K.A.; Pearson, C.B.; Hachey, A.M.; Xia, D.L.; Wachtman, L.M. Alternative Activation of Macrophages in Rhesus Macaques (Macaca Mulatta) with Endometriosis. Comp. Med. 2012, 62, 303–310. [Google Scholar]
- Johan, M.Z.; Ingman, W.V.; Robertson, S.A.; Hull, M.L. Macrophages Infiltrating Endometriosis-like Lesions Exhibit Progressive Phenotype Changes in a Heterologous Mouse Model. J. Reprod. Immunol. 2019, 132, 1–8. [Google Scholar] [CrossRef]
- Yuan, M.; Li, D.; An, M.; Li, Q.; Zhang, L.; Wang, G. Rediscovering Peritoneal Macrophages in a Murine Endometriosis Model. Hum. Reprod. 2017, 32, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Hogg, C.; Panir, K.; Dhami, P.; Rosser, M.; Mack, M.; Soong, D.; Pollard, J.W.; Jenkins, S.J.; Horne, A.W.; Greaves, E. Macrophages Inhibit and Enhance Endometriosis Depending on Their Origin. Proc. Natl. Acad. Sci. USA 2021, 118, e2013776118. [Google Scholar] [CrossRef] [PubMed]
- Ono, Y.; Yoshino, O.; Hiraoka, T.; Sato, E.; Furue, A.; Nawaz, A.; Hatta, H.; Fukushi, Y.; Wada, S.; Tobe, K.; et al. CD206+ Macrophage Is an Accelerator of Endometriotic-like Lesion via Promoting Angiogenesis in the Endometriosis Mouse Model. Sci. Rep. 2021, 11, 853. [Google Scholar] [CrossRef] [PubMed]
- Kalu, E.; Sumar, N.; Giannopoulos, T.; Patel, P.; Croucher, C.; Sherriff, E.; Bansal, A. Cytokine Profiles in Serum and Peritoneal Fluid from Infertile Women with and without Endometriosis. J. Obstet. Gynaecol. Res. 2007, 33, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Volpato, L.K.; Horewicz, V.V.; Bobinski, F.; Martins, D.F.; Piovezan, A.P. Annexin A1, FPR2/ALX, and Inflammatory Cytokine Expression in Peritoneal Endometriosis. J. Reprod. Immunol. 2018, 129, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.M.; Ma, Z.Y.; Song, N. Inflammatory Cytokines IL-6, IL-10, IL-13, TNF-α and Peritoneal Fluid Flora Were Associated with Infertility in Patients with Endometriosis. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 2513–2518. [Google Scholar] [CrossRef]
- Sikora, J.; Smycz-Kubańska, M.; Mielczarek-Palacz, A.; Bednarek, I.; Kondera-Anasz, Z. The Involvement of Multifunctional TGF-β and Related Cytokines in Pathogenesis of Endometriosis. Immunol. Lett. 2018, 201, 31–37. [Google Scholar] [CrossRef]
- Jiang, J.; Jiang, Z.; Xue, M. Serum and Peritoneal Fluid Levels of Interleukin-6 and Interleukin-37 as Biomarkers for Endometriosis. Gynecol. Endocrinol. 2019, 35, 571–575. [Google Scholar] [CrossRef]
- Tarokh, M.; Novin, M.G.; Poordast, T.; Tavana, Z.; Nazarian, H.; Norouzian, M.; Gharesi-Fard, B. Serum and Peritoneal Fluid Cytokine Profiles in Infertile Women with Endometriosis. Iran. J. Immunol. 2019, 16, 151–162. [Google Scholar] [CrossRef]
- Bellelis, P.; Frediani Barbeiro, D.; Gueuvoghlanian-Silva, B.Y.; Kalil, J.; Abrão, M.S.; Podgaec, S. Interleukin-15 and Interleukin-7 Are the Major Cytokines to Maintain Endometriosis. Gynecol. Obstet. Investig. 2019, 84, 435–444. [Google Scholar] [CrossRef]
- Voltolini Velho, R.; Halben, N.; Chekerov, R.; Keye, J.; Plendl, J.; Sehouli, J.; Mechsner, S. Functional Changes of Immune Cells: Signal of Immune Tolerance of the Ectopic Lesions in Endometriosis? Reprod. BioMed. Online 2021, 43, 319–328. [Google Scholar] [CrossRef]
- Pizzo, A.; Salmeri, F.M.; Ardita, F.V.; Sofo, V.; Tripepi, M.; Marsico, S. Behaviour of Cytokine Levels in Serum and Peritoneal Fluid of Women with Endometriosis. Gynecol. Obstet. Investig. 2002, 54, 82–87. [Google Scholar] [CrossRef]
- Sikora, J.; Mielczarek-Palacz, A.; Kondera-Anasz, Z. Association of the Precursor of Interleukin-1β and Peritoneal Inflammation-Role in Pathogenesis of Endometriosis. J. Clin. Lab. Anal. 2016, 30, 831–837. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.Y.; Chen, H.Y.; Chen, W.; Liu, Y.N.; Fu, Y.; Wang, L.N. Expression of Inflammatory Cytokines in Serum and Peritoneal Fluid from Patients with Different Stages of Endometriosis. Gynecol. Endocrinol. 2018, 34, 507–512. [Google Scholar] [CrossRef] [PubMed]
- Burns, K.A.; Thomas, S.Y.; Hamilton, K.J.; Young, S.L.; Cook, D.N.; Korach, K.S. Early Endometriosis in Females Is Directed by Immune-Mediated Estrogen Receptor a and IL-6 Cross-Talk. Endocrinology 2018, 159, 103–118. [Google Scholar] [CrossRef] [PubMed]
- Rier, S.E.; Zarmakoupis, P.N.; Hu, X.; Becker, J.L. Dysregulation of Interleukin-6 Responses in Ectopic Endometrial Stromal Cells: Correlation with Decreased Soluble Receptor Levels in Peritoneal Fluid of Women with Endometriosis. J. Clin. Endocrinol. Metab. 1995, 80, 1431–1437. [Google Scholar] [CrossRef] [PubMed]
- Suen, J.L.; Chang, Y.; Chiu, P.R.; Hsieh, T.H.; Hsi, E.; Chen, Y.C.; Chen, Y.F.; Tsai, E.M. Serum Level of IL-10 Is Increased in Patients with Endometriosis, and IL-10 Promotes the Growth of Lesions in a Murine Model. Am. J. Pathol. 2014, 184, 464–471. [Google Scholar] [CrossRef]
- Jeljeli, M.; Riccio, L.G.C.; Chouzenoux, S.; Moresi, F.; Toullec, L.; Doridot, L.; Nicco, C.; Bourdon, M.; Marcellin, L.; Santulli, P.; et al. Macrophage Immune Memory Controls Endometriosis in Mice and Humans. Cell Rep. 2020, 33, 108325. [Google Scholar] [CrossRef]
- Hamilton, G.; Rath, B.; Klameth, L.; Hochmair, M.J. Small Cell Lung Cancer: Recruitment of Macrophages by Circulating Tumor Cells. OncoImmunology 2016, 5, e1093277. [Google Scholar] [CrossRef]
- Lu, H.; Yang, H.-L.; Zhou, W.-J.; Lai, Z.-Z.; Qiu, X.-M.; Fu, Q.; Zhao, J.-Y.; Wang, J.; Li, D.-J.; Li, M.-Q. Rapamycin Prevents Spontaneous Abortion by Triggering Decidual Stromal Cell Autophagy-Mediated NK Cell Residence. Autophagy 2020, 1–17. [Google Scholar] [CrossRef]
- Li, M.O.; Wan, Y.Y.; Sanjabi, S.; Robertson, A.K.L.; Flavell, R.A. Transforming Growth Factor-β Regulation of Immune Responses. Annu. Rev. Immunol. 2006, 24, 99–146. [Google Scholar] [CrossRef]
- Hull, M.L.; Johan, M.Z.; Hodge, W.L.; Robertson, S.A.; Ingman, W.V. Host-Derived TGFB1 Deficiency Suppresses Lesion Development in a Mouse Model of Endometriosis. Am. J. Pathol. 2012, 180, 880–887. [Google Scholar] [CrossRef]
- Young, V.J.; Ahmad, S.F.; Duncan, W.C.; Horne, A.W. The Role of TGF-β in the Pathophysiology of Peritoneal Endometriosis. Hum. Reprod. Update 2017, 23, 548–559. [Google Scholar] [CrossRef]
- Chegini, N. TGF-β System: The Principal Profibrotic Mediator of Peritoneal Adhesion Formation. Semin. Reprod. Med. 2008, 26, 298–312. [Google Scholar] [CrossRef]
- Yang, H.L.; Zhou, W.J.; Chang, K.K.; Mei, J.; Huang, L.Q.; Wang, M.Y.; Meng, Y.; Ha, S.Y.; Li, D.J.; Li, M.Q. The Crosstalk between Endometrial Stromal Cells and Macrophages Impairs Cytotoxicity of NK Cells in Endometriosis by Secreting IL-10 and TGF-β. Reproduction 2017, 154, 815–825. [Google Scholar] [CrossRef]
- Cao, X.; Yang, D.; Song, M.; Murphy, A.; Parthasarathy, S. The Presence of Endometrial Cells in the Peritoneal Cavity Enhances Monocyte Recruitment and Induces Inflammatory Cytokines in Mice: Implications for Endometriosis. Fertil. Steril. 2004, 82, 999–1007. [Google Scholar] [CrossRef]
- Haber, E.; Danenberg, H.D.; Koroukhov, N.; Ron-El, R.; Golomb, G.; Schachter, M. Peritoneal Macrophage Depletion by Liposomal Bisphosphonate Attenuates Endometriosis in the Rat Model. Hum. Reprod. 2009, 24, 398–407. [Google Scholar] [CrossRef] [PubMed]
- Mei, J.; Zhou, W.J.; Li, S.Y.; Li, M.Q.; Sun, H.X. Interleukin-22 Secreted by Ectopic Endometrial Stromal Cells and Natural Killer Cells Promotes the Recruitment of Macrophages through Promoting CCL2 Secretion. Am. J. Reprod. Immunol. 2019, 82, e13166. [Google Scholar] [CrossRef] [PubMed]
- Cakmak, H.; Seval-Celik, Y.; Arlier, S.; Guzeloglu-Kayisli, O.; Schatz, F.; Arici, A.; Kayisli, U.A. P38 Mitogen-Activated Protein Kinase Is Involved in the Pathogenesis of Endometriosis by Modulating Inflammation, but Not Cell Survival. Reprod. Sci. 2018, 25, 587–597. [Google Scholar] [CrossRef]
- Wang, X.Q.; Yu, J.; Luo, X.Z.; Shi, Y.L.; Wang, Y.; Wang, L.; Li, D.J. The High Level of RANTES in the Ectopic Milieu Recruits Macrophages and Induces Their Tolerance in Progression of Endometriosis. J. Mol. Endocrinol. 2010, 45, 291–299. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Liang, Y.; Wang, W.; Huang, J.; Tan, H.; Liu, T.; Shang, C.; Liu, D.; Guo, L.; Yao, S. Potential Role of Semaphorin 3A and Its Receptors in Regulating Aberrant Sympathetic Innervation in Peritoneal and Deep Infiltrating Endometriosis. PLoS ONE 2015, 10, e0146027. [Google Scholar] [CrossRef]
- Casazza, A.; Laoui, D.; Wenes, M.; Rizzolio, S.; Bassani, N.; Mambretti, M.; Deschoemaeker, S.; VanGinderachter, J.A.; Tamagnone, L.; Mazzone, M. Impeding Macrophage Entry into Hypoxic Tumor Areas by Sema3A/Nrp1 Signaling Blockade Inhibits Angiogenesis and Restores Antitumor Immunity. Cancer Cell 2013, 24, 695–709. [Google Scholar] [CrossRef]
- Hou, X.X.; Wang, X.Q.; Zhou, W.J.; Li, D.J. Regulatory T Cells Induce Polarization of Pro-Repair Macrophages by Secreting SFGL2 into the Endometriotic Milieu. Commun. Biol. 2021, 4, 499. [Google Scholar] [CrossRef]
- Barclay, A.N.; van den Berg, T.K. The Interaction between Signal Regulatory Protein Alpha (SIRPα) and CD47: Structure, Function, and Therapeutic Target. Annu. Rev. Immunol. 2014, 32, 25–50. [Google Scholar] [CrossRef]
- de Vos, A.F.; van der Poll, T. SIRP-α Instructs Alveolar Macrophages to Stop Eating after Pneumonia. Nat. Immunol. 2020, 21, 601–604. [Google Scholar] [CrossRef]
- Liu, Y.; Li, M.; Wei, C.; Tang, L.; Sheng, Y.; Liu, Y.; Li, D.; Ding, D.; Qiu, J.; Zhu, X. TSP1-CD47-SIRPα Signaling Facilitates the Development of Endometriosis by Mediating the Survival of Ectopic Endometrium. Am. J. Reprod. Immunol. 2020, 83, e13236. [Google Scholar] [CrossRef]
- Deleon-Pennell, K.Y.; Tian, Y.; Zhang, B.; Cates, C.A.; Iyer, R.P.; Cannon, P.; Shah, P.; Aiyetan, P.; Halade, G.V.; Ma, Y.; et al. CD36 Is a Matrix Metalloproteinase-9 Substrate That Stimulates Neutrophil Apoptosis and Removal during Cardiac Remodeling. Circ. Cardiovasc. Genet. 2016, 9, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Tian, K.; Xu, Y.; Sahebkar, A.; Xu, S. CD36 in Atherosclerosis: Pathophysiological Mechanisms and Therapeutic Implications. Curr. Atheroscler. Rep. 2020, 22, 59. [Google Scholar] [CrossRef] [PubMed]
- Chuang, P.C.; Wu, M.H.; Shoji, Y.; Tsai, S.J. Downregulation of CD36 Results in Reduced Phagocytic Ability of Peritoneal Macrophages of Women with Endometriosis. J. Pathol. 2009, 219, 232–241. [Google Scholar] [CrossRef]
- Chuang, P.C.; Lin, Y.J.; Wu, M.H.; Wing, L.Y.C.; Shoji, Y.; Tsai, S.J. Inhibition of CD36-Dependent Phagocytosis by Prostaglandin E2 Contributes to the Development of Endometriosis. Am. J. Pathol. 2010, 176, 850–860. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.H.; Shoji, Y.; Wu, M.C.; Chuang, P.C.; Lin, C.C.; Huang, M.F.; Tsai, S.J. Suppression of Matrix Metalloproteinase-9 by Prostaglandin E2 in Peritoneal Macrophage Is Associated with Severity of Endometriosis. Am. J. Pathol. 2005, 167, 1061–1069. [Google Scholar] [CrossRef]
- Kuiper, G.G.J.M.; Enmark, E.; Pelto-Huikko, M.; Nilsson, S.; Gustafsson, J.Å. Cloning of a Novel Estrogen Receptor Expressed in Rat Prostate and Ovary. Proc. Natl. Acad. Sci. USA 1996, 93, 5925–5930. [Google Scholar] [CrossRef]
- Liang, Y.; Xie, H.; Wu, J.; Liu, D.; Yao, S. Villainous Role of Estrogen in Macrophage-Nerve Interaction in Endometriosis. Reprod. Biol. Endocrinol. 2018, 16, 122. [Google Scholar] [CrossRef]
- Zeitoun, K.; Takayama, K.; Sasano, H.; Suzuki, T.; Moghrabi, N.; Andersson, S.; Johns, A.; Meng, L.; Putman, M.; Carr, B.; et al. Deficient 17β-Hydroxysteroid Dehydrogenase Type 2 Expression in Endometriosis: Failure to Metabolize 17β-Estradiol. J. Clin. Endocrinol. Metab. 1998, 83, 4474–4480. [Google Scholar] [CrossRef] [PubMed]
- Delvoux, B.; Groothuis, P.; D’Hooghe, T.; Kyama, C.; Dunselman, G.; Romano, A. Increased Production of 17β-Estradiol in Endometriosis Lesions Is the Result of Impaired Metabolism. J. Clin. Endocrinol. Metab. 2009, 94, 876–883. [Google Scholar] [CrossRef]
- Bulun, S.E.; Monsavais, D.; Pavone, M.E.; Dyson, M.; Xue, Q.; Attar, E.; Tokunaga, H.; Su, E.J. Role of Estrogen Receptor-β in Endometriosis. Semin. Reprod. Med. 2012, 30, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Han, S.J.; Lee, J.E.; Cho, Y.J.; Park, M.J.; O’Malley, B.W. Genomic Function of Estrogen Receptor β in Endometriosis. Endocrinology 2019, 160, 2495–2516. [Google Scholar] [CrossRef]
- Han, S.J.; Jung, S.Y.; Wu, S.P.; Hawkins, S.M.; Park, M.J.; Kyo, S.; Qin, J.; Lydon, J.P.; Tsai, S.Y.; Tsai, M.J.; et al. Estrogen Receptor β Modulates Apoptosis Complexes and the Inflammasome to Drive the Pathogenesis of Endometriosis. Cell 2015, 163, 960–974. [Google Scholar] [CrossRef] [PubMed]
- Khan, K.N.; Kitajima, M.; Inoue, T.; Fujishita, A.; Nakashima, M.; Masuzaki, H. 17β-Estradiol and Lipopolysaccharide Additively Promote Pelvic Inflammation and Growth of Endometriosis. Reprod. Sci. 2015, 22, 585–594. [Google Scholar] [CrossRef]
- Greaves, E.; Temp, J.; Esnal-Zufiurre, A.; Mechsner, S.; Horne, A.W.; Saunders, P.T.K. Estradiol Is a Critical Mediator of Macrophage-Nerve Cross Talk in Peritoneal Endometriosis. Am. J. Pathol. 2015, 185, 2286–2297. [Google Scholar] [CrossRef]
- Mechsner, S.; Kaiser, A.; Kopf, A.; Gericke, C.; Ebert, A.; Bartley, J. A Pilot Study to Evaluate the Clinical Relevance of Endometriosis-Associated Nerve Fibers in Peritoneal Endometriotic Lesions. Fertil. Steril. 2009, 92, 1856–1861. [Google Scholar] [CrossRef] [PubMed]
- McKinnon, B.; Bersinger, N.A.; Wotzkow, C.; Mueller, M.D. Endometriosis-Associated Nerve Fibers, Peritoneal Fluid Cytokine Concentrations, and Pain in Endometriotic Lesions from Different Locations. Fertil. Steril. 2012, 97, 373–380. [Google Scholar] [CrossRef]
- Wu, J.; Xie, H.; Yao, S.; Liang, Y. Macrophage and Nerve Interaction in Endometriosis. J. Neuroinflamm. 2017, 14, 53. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, H.; Zhao, Z.; Gao, B.; Meng, L.; Feng, X. MiR-146b Level and Variants Is Associated with Endometriosis Related Macrophages Phenotype and Plays a Pivotal Role in the Endometriotic Pain Symptom. Taiwan. J. Obstet. Gynecol. 2019, 58, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Wright, K.R.; Mitchell, B.; Santanam, N. Redox Regulation of MicroRNAs in Endometriosis-Associated Pain. Redox Biol. 2017, 12, 956–966. [Google Scholar] [CrossRef] [PubMed]
| Cell type | Monocytes/Mϕ (%) | Lymphocytes (%) | NKs (%) | DCs (%) | PMNs (%) | Reference | |||
|---|---|---|---|---|---|---|---|---|---|
| Subjects | Total | T Cells | CD4/CD8 | B Cells | |||||
| Healthy Controls (Women) | 89.0 ± 2.0 | 8.4 ± 1.6 | — | — | — | — | — | — | [53] |
| 90.1 ± 1.9 | — | — | — | — | — | — | — | [54] | |
| 93.2 1 | — | — | — | — | — | — | n.d. | [50] | |
| 85.0–95.0 2 | — | — | — | — | — | — | n.d. | [52] | |
| 53.0 ± 26.5 | — | 23.5 ± 27.4 | — | 0.1 ± 0.2 | 0.7 ± 0.9 | 9.3 ± 11.0 | 2.5 ± 5.1 | [47] | |
| Healthy Controls (Gallbladder) | 21.0–61.0 2 | — | 37.0–49.0 2 | 0.48 | 0.0–2.0 2 | 0.0–18.0 2 | — | <5.0 | [49] |
| CAPD Effluent Patients | 86.5 1 | — | — | — | — | — | — | 3.4 1 | [50] |
| 84.0 1 | 10.0 1 | — | — | — | — | — | 4.0 1 | [51] | |
| >60.0 | — | — | — | — | — | — | — | [52] | |
| Healthy Controls (Infants) | 49.2 ± 7.1 | 22.5 ± 5.4 | 14.5 ± 4.5 | — | 0.7 ± 0.5 | — | — | — | [55] |
| Healthy Controls (Children) | 42.9 ± 11.1 | 33.1 ± 9.6 | 26.6 ± 9.8 | — | 0.8 ± 0.7 | — | — | — | [55] |
| Healthy Controls (Adolescents) | 41.1 ± 8.7 | 36.6 ± 7.1 | 31.5 ± 7.6 | — | 0.3 ± 0.1 | — | — | — | [55] |
| Marker | Endometriosis vs. Health Comparison | Statistical Significance (p value) | Technique Used | Reference |
|---|---|---|---|---|
| CD86 | Diminished | ** | Flow Cytometry | [59] |
| Ratio CD163+/CD86+ | Increased | ** | [59] | |
| CD14high | Increased | * | [67] | |
| CD68 | Increased | *** | Immunohistochemistry | [68] |
| HLA-DR | Without changes | n.s. | Flow Cytometry | [59] |
| Increased | ** | [69] | ||
| NLC-MACRO | Increased | *** | Immunohistochemistry | [68] |
| HAM56 | Increased | *** | [68] | |
| CD95 (Fas) | Increased | * | Flow Cytometry | [69] |
| SIRP-α | Increased | ** | Immunoblot | [30] |
| CD200R | Increased | * | [70] | |
| CD36 | Diminished | ** | [30] |
| Cytokine | Comparison | Endometriosis vs. Health Significance | Reference |
|---|---|---|---|
| IL-6 | Increased | * | [85,86,87] |
| *** | [88,89] | ||
| IL-10 | Without changes | n.s. | [89,90] |
| Increased | *** | [88] | |
| * | [91,92] | ||
| IL-8 | Increased | ** | [85,92] |
| *** | [93] | ||
| MCP-1 | Increased | * | [85,92] |
| *** | [93] | ||
| IL-1β | Without changes | n.s. | [86,90] |
| Increased | ** | [92] | |
| *** | [88,94] | ||
| IL-2 | Without changes | n.s. | [91] |
| Increased | * | [95] | |
| TNF-α | Without changes | n.s. | [85,90] |
| Increased | * | [92,93] | |
| TGF-β | Increased | *** | [88,93] |
| * | [90] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramírez-Pavez, T.N.; Martínez-Esparza, M.; Ruiz-Alcaraz, A.J.; Marín-Sánchez, P.; Machado-Linde, F.; García-Peñarrubia, P. The Role of Peritoneal Macrophages in Endometriosis. Int. J. Mol. Sci. 2021, 22, 10792. https://doi.org/10.3390/ijms221910792
Ramírez-Pavez TN, Martínez-Esparza M, Ruiz-Alcaraz AJ, Marín-Sánchez P, Machado-Linde F, García-Peñarrubia P. The Role of Peritoneal Macrophages in Endometriosis. International Journal of Molecular Sciences. 2021; 22(19):10792. https://doi.org/10.3390/ijms221910792
Chicago/Turabian StyleRamírez-Pavez, Tamara N., María Martínez-Esparza, Antonio J. Ruiz-Alcaraz, Pilar Marín-Sánchez, Francisco Machado-Linde, and Pilar García-Peñarrubia. 2021. "The Role of Peritoneal Macrophages in Endometriosis" International Journal of Molecular Sciences 22, no. 19: 10792. https://doi.org/10.3390/ijms221910792
APA StyleRamírez-Pavez, T. N., Martínez-Esparza, M., Ruiz-Alcaraz, A. J., Marín-Sánchez, P., Machado-Linde, F., & García-Peñarrubia, P. (2021). The Role of Peritoneal Macrophages in Endometriosis. International Journal of Molecular Sciences, 22(19), 10792. https://doi.org/10.3390/ijms221910792







