Therapeutic Effects of an Anti-sialyl Lewis X Antibody in a Murine Model of Allergic Asthma
Abstract
:1. Introduction
2. Results
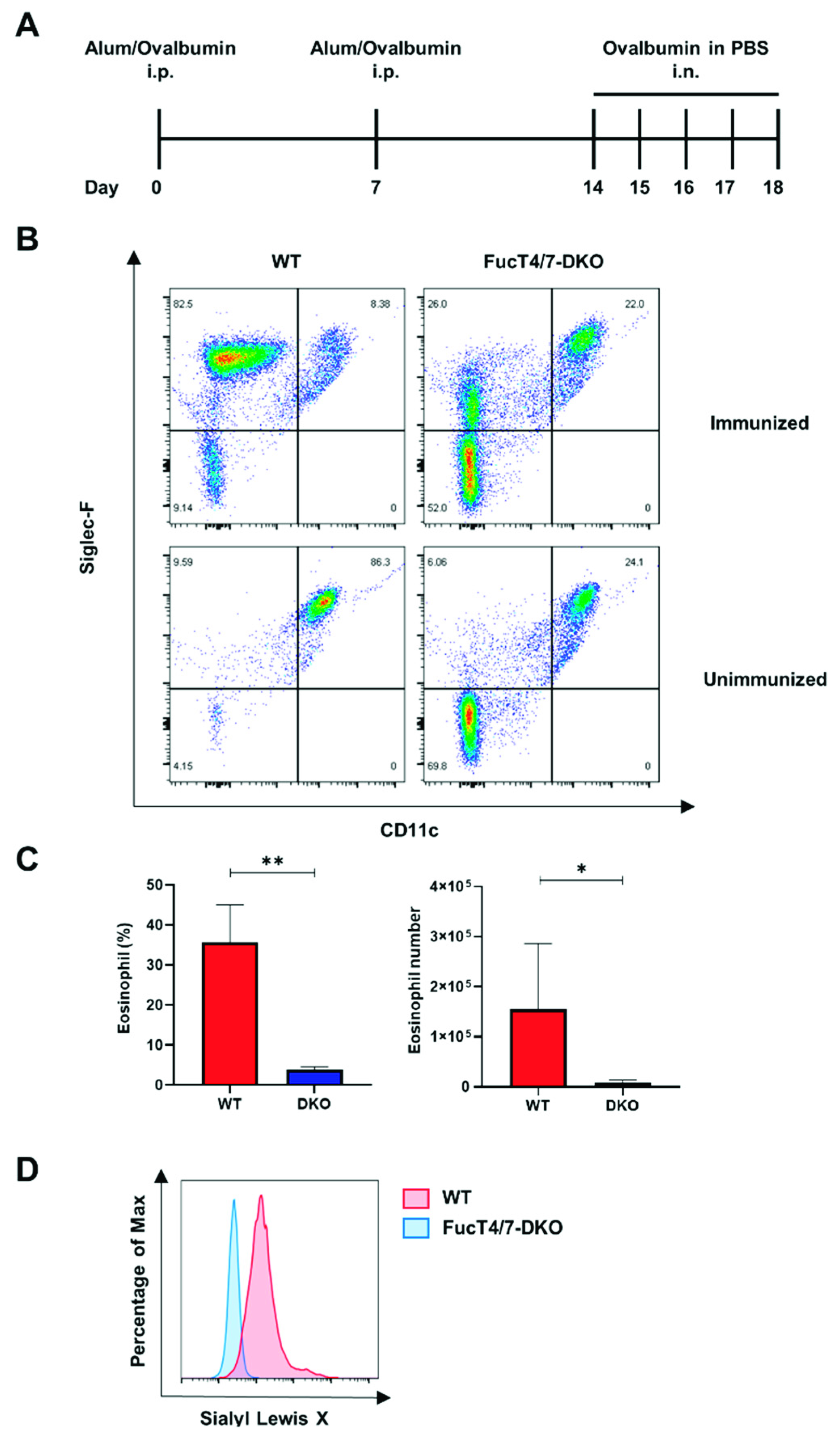
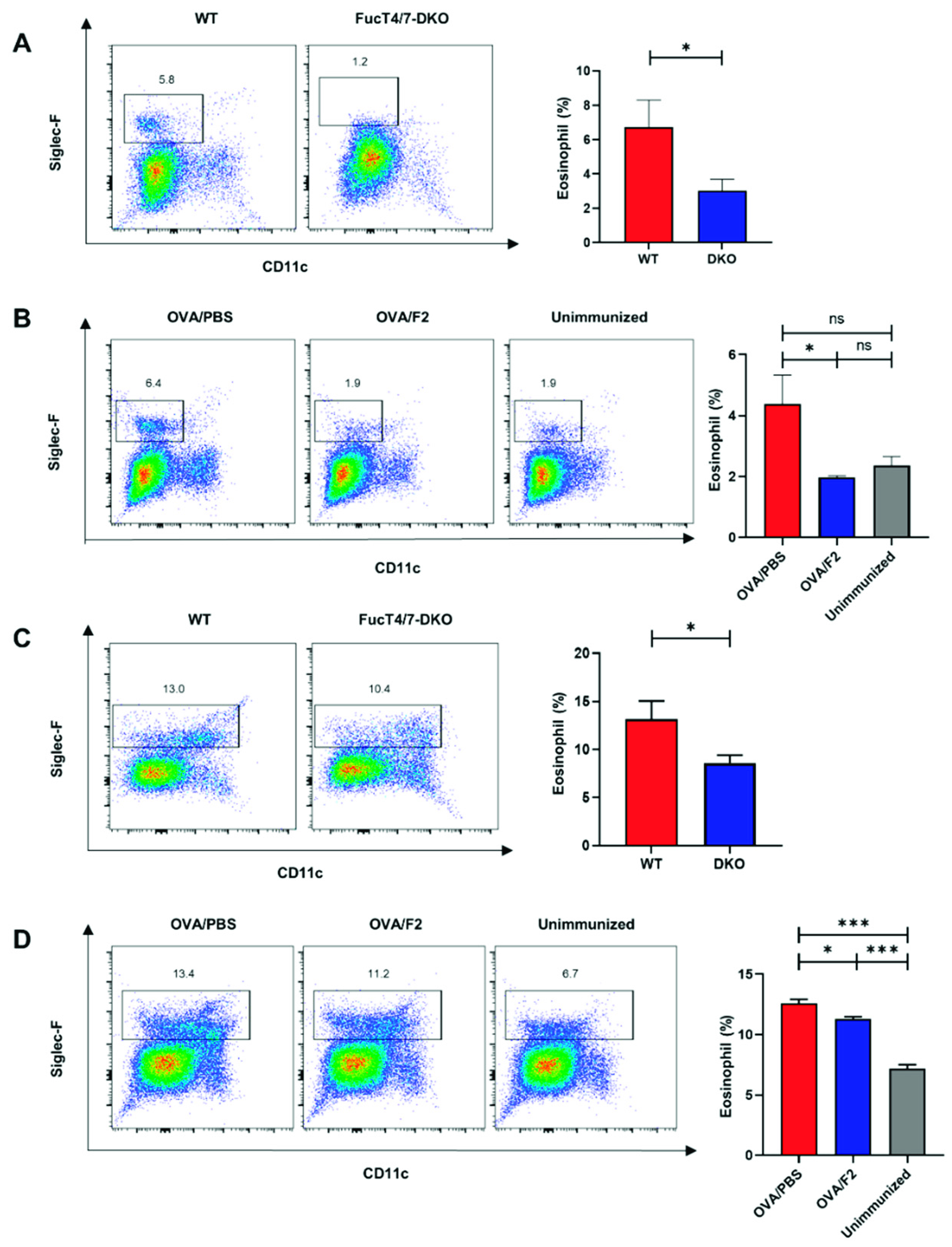
2.1. SLex Deficiency in Eosinophils Resulted in Diminished Infiltration into the Lungs
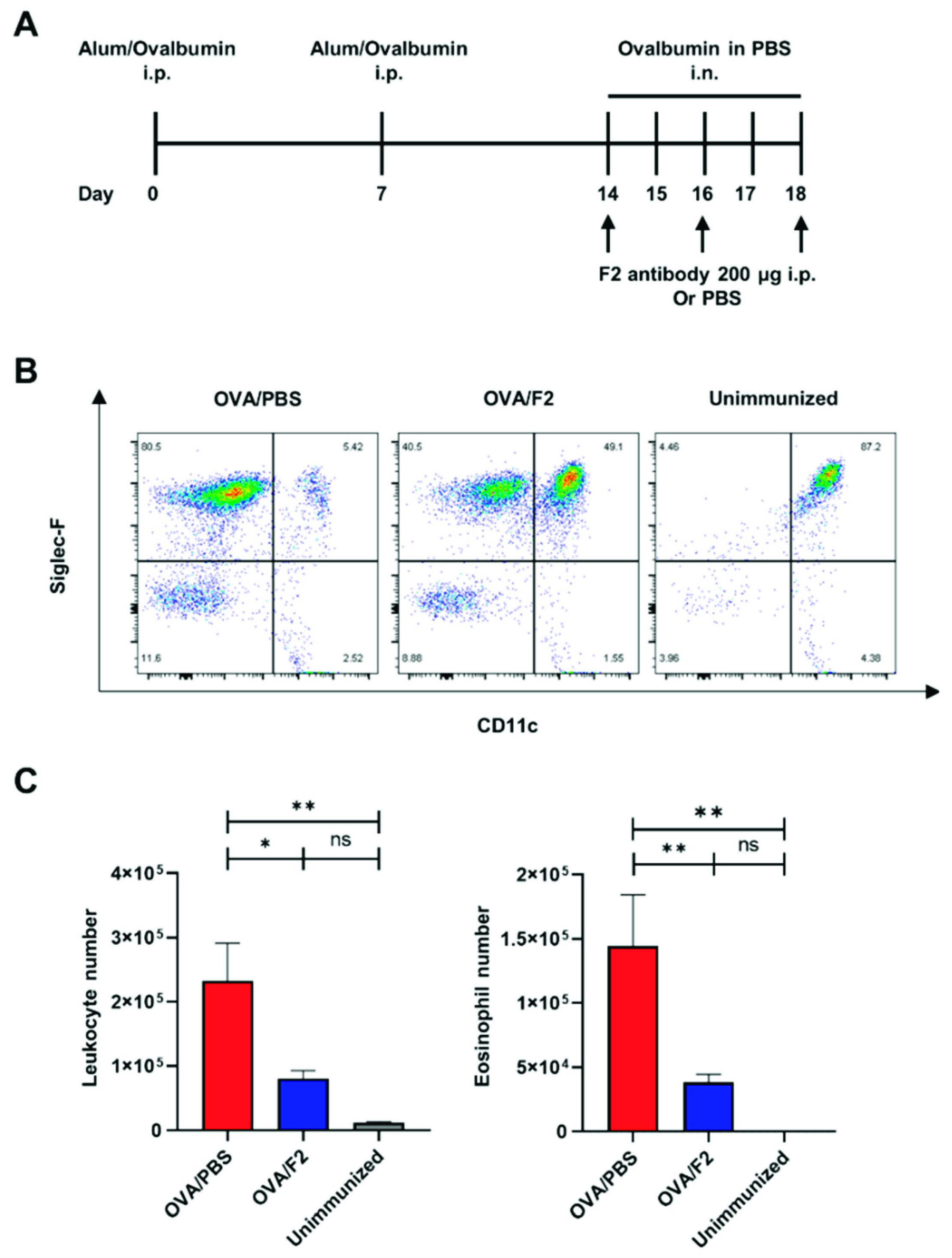
2.2. Administration of mAb F2 Suppressed Eosinophil Infiltration
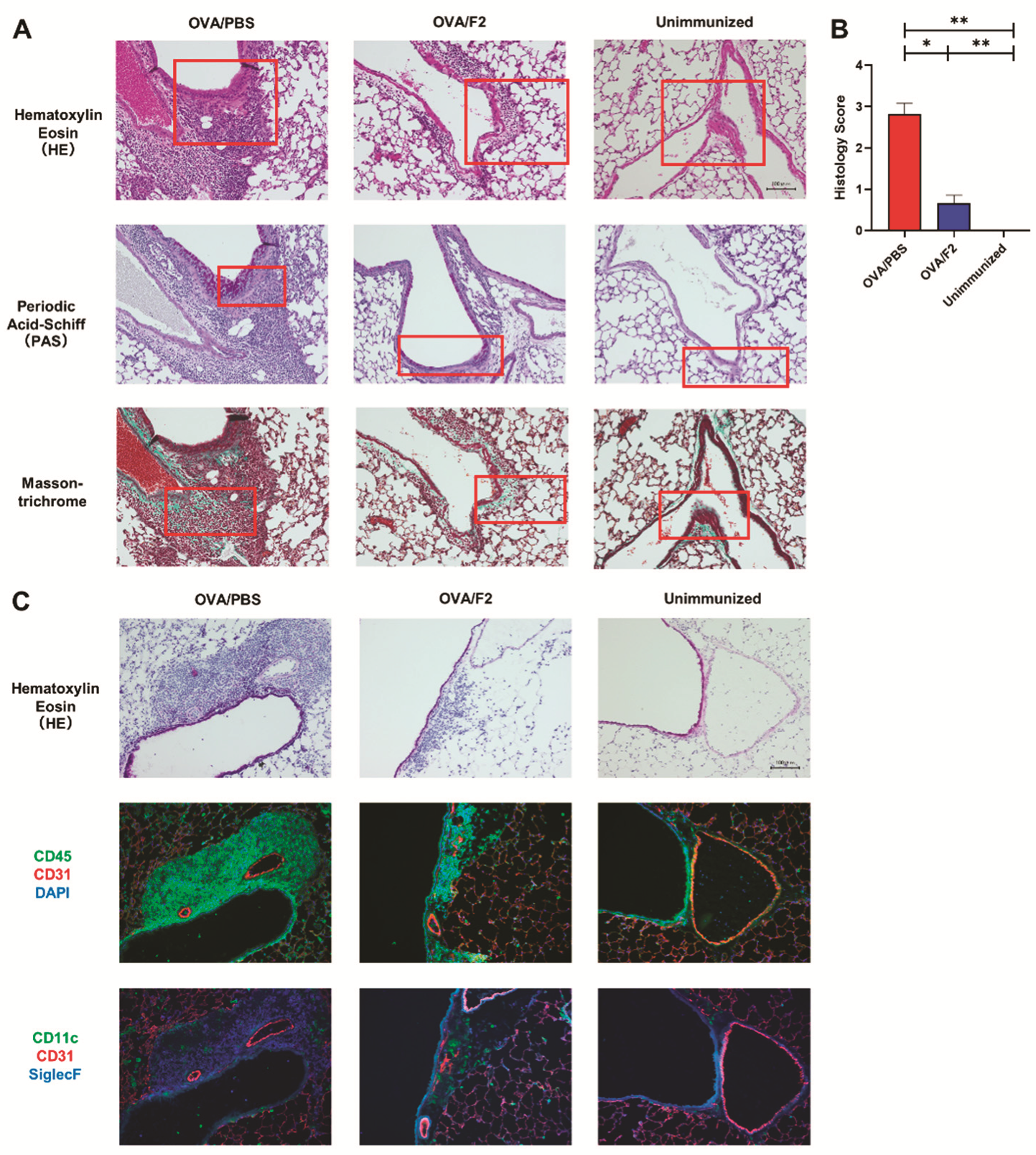
2.3. Decreased Eosinophil Infiltration and Relieved Allergic Responses in mAb F2-Administered Mice
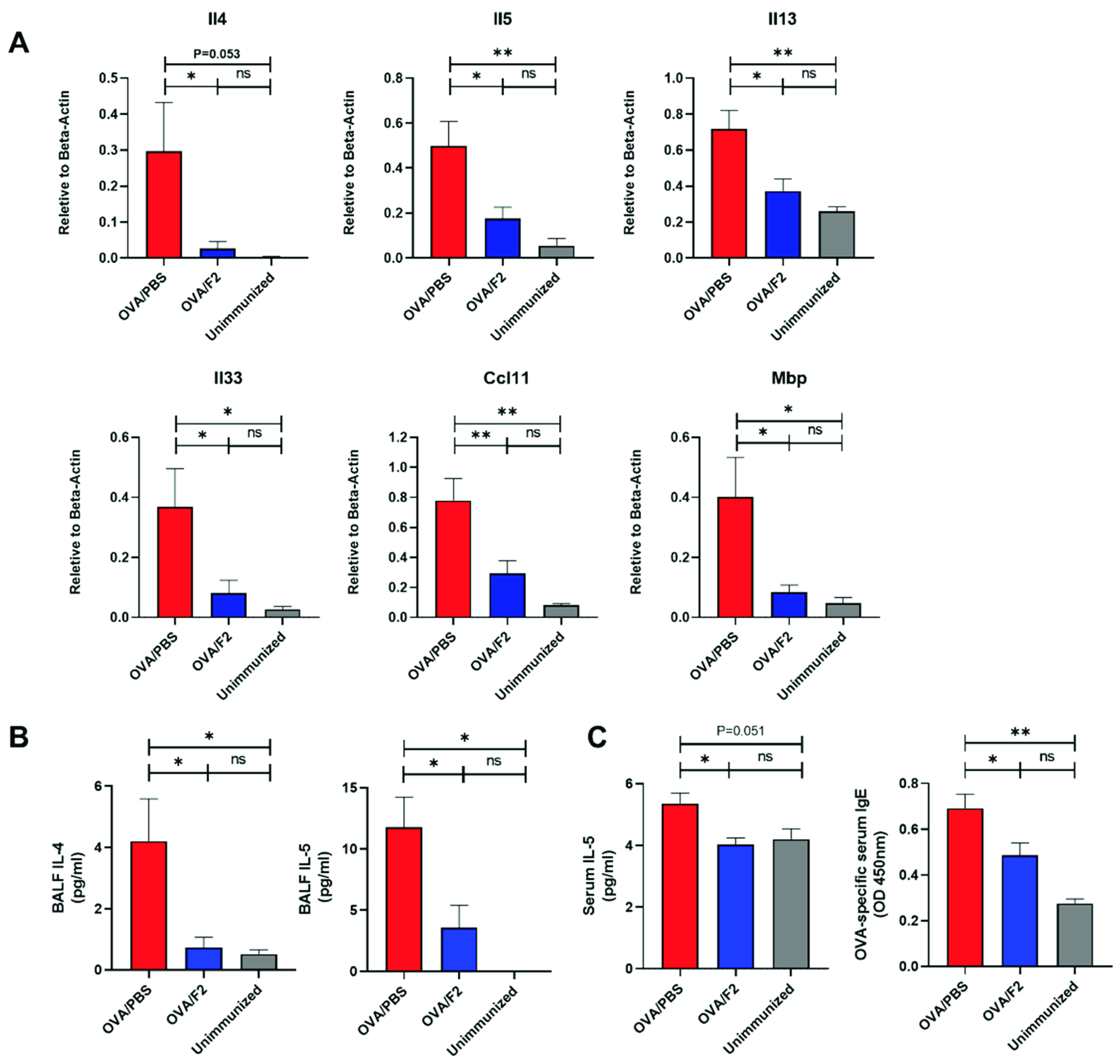
2.4. Administration of mAb F2 Suppressed Allergic Immune Response in the Lungs
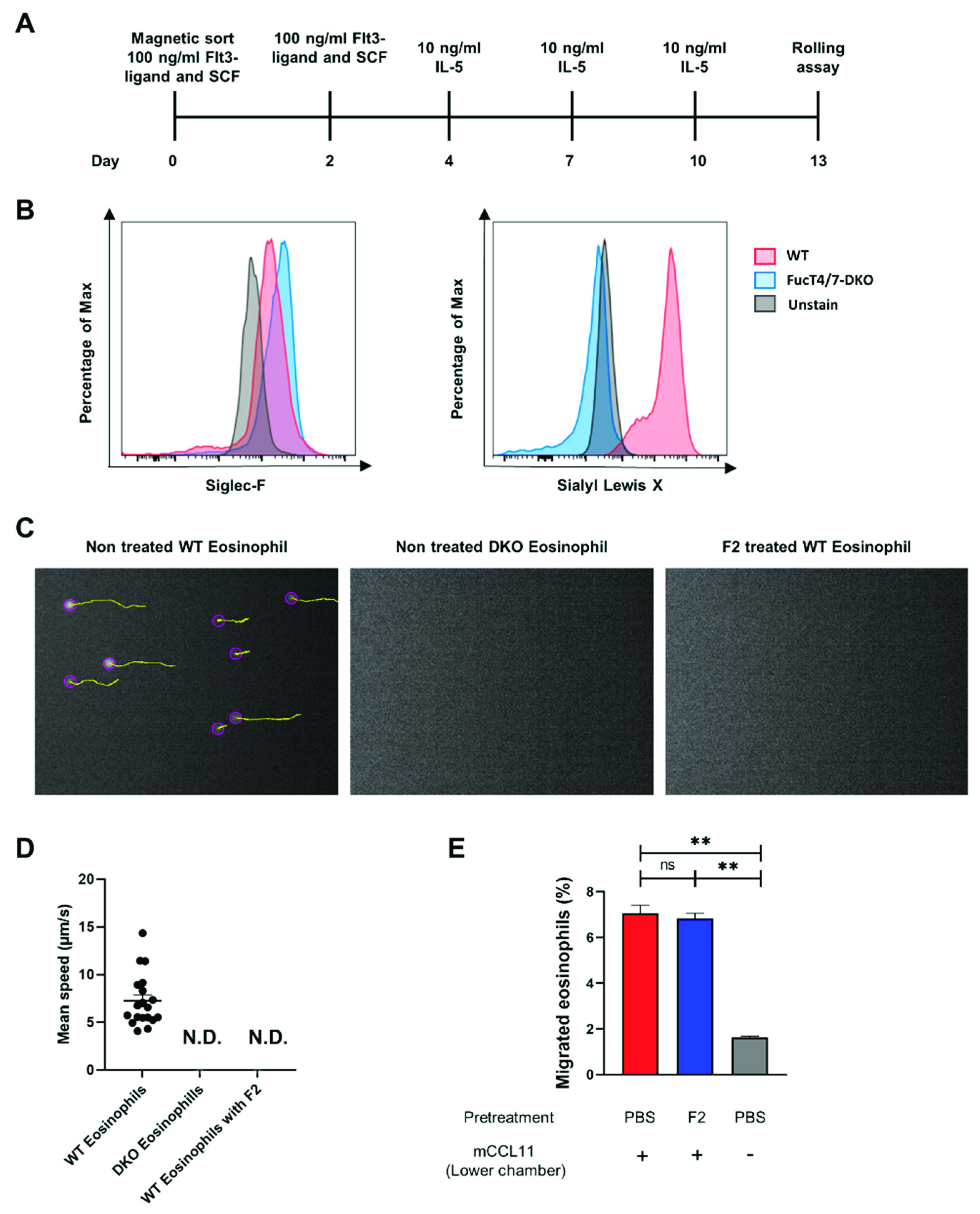
2.5. Blockade of sLex Resulted in Reduced Differentiation of Eosinophils in the Bone Marrow
2.6. SLex Played a Vital Role in the Rolling of Eosinophils by Binding to p-Selectin
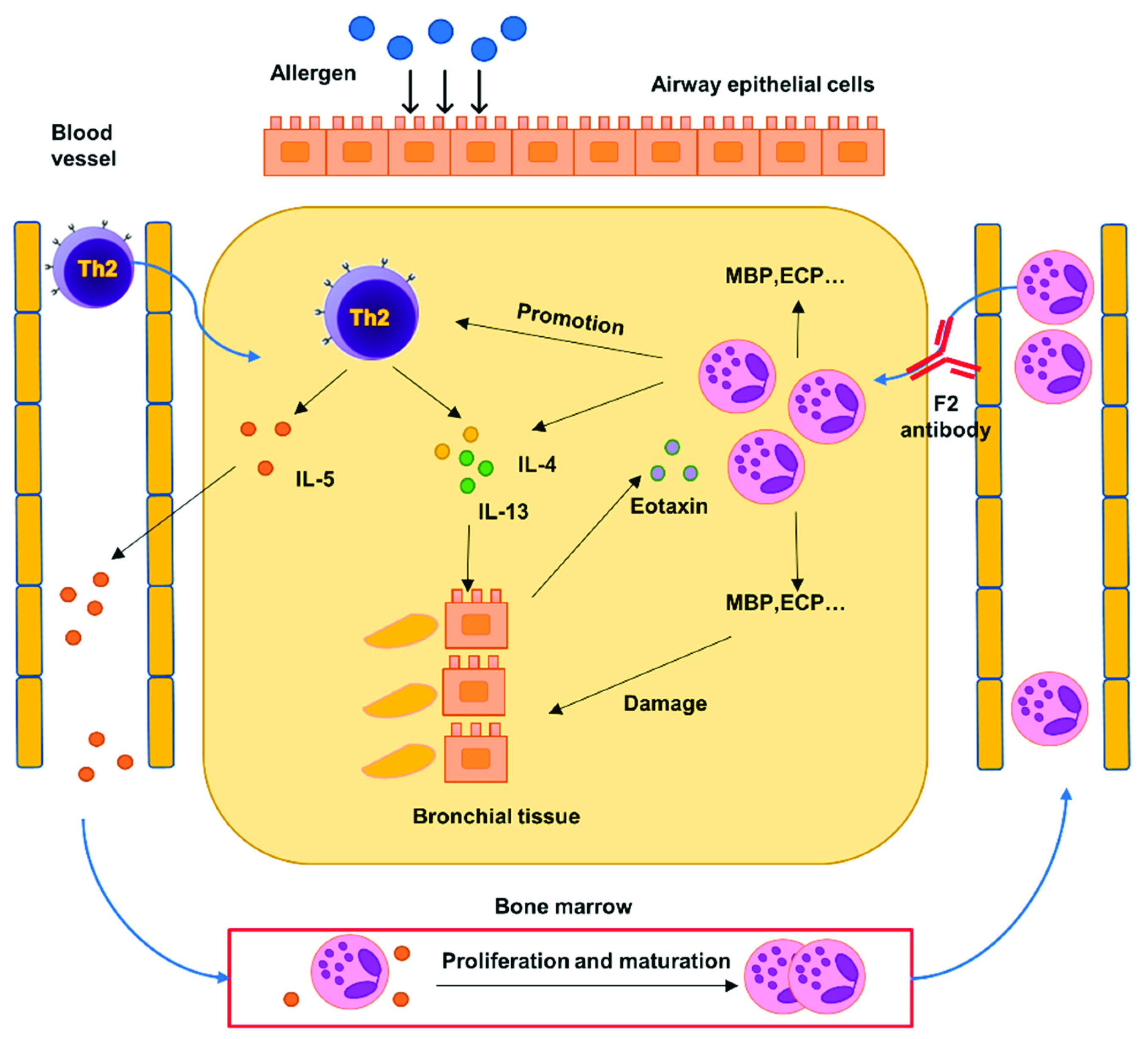
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. OVA-Induced Mouse Model of Asthma
4.3. Harvesting of Bronchoalveolar Lavage Fluid (BALF) and Lung Tissues
4.4. Flow Cytometric Analysis of BALF, Blood and Bone Marrow
4.5. Enzyme-Linked Immunosorbent Assay (ELISA) for the Measurement of Serum IgE and IL-5
4.6. Real-Time Quantitative PCR
4.7. Immunofluorescence
4.8. Histological Analysis
4.9. Ex Vivo Induction of Eosinophils
4.10. Eosinophil Rolling Assay
4.11. Migration Assay
4.12. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [Google Scholar] [CrossRef]
- McGeachie, M.J.; Croteau-Chonka, D.C.; Weiss, S.T. Asthma. In Genomic and Precision Medicine: Primary Care: Third Edition; Elsevier: Amsterdam, The Netherlands, 2017; pp. 231–244. ISBN 9780128006542. [Google Scholar]
- Custovic, A. To what extent is allergen exposure a risk factor for the development of allergic disease? Clin. Exp. Allergy 2015, 45, 54–62. [Google Scholar] [CrossRef]
- Schatz, M.; Rosenwasser, L. The allergic asthma phenotype. J. Allergy Clin. Immunol. Pract. 2014, 2, 645–648. [Google Scholar] [CrossRef] [PubMed]
- Bateman, E.D.; Hurd, S.S.; Barnes, P.J.; Bousquet, J.; Drazen, J.M.; FitzGerald, M.; Gibson, P.; Ohta, K.; O’Byrne, P.; Pedersen, S.E.; et al. Global strategy for asthma management and prevention: GINA executive summary. Eur. Respir. J. 2008, 31, 143–178. [Google Scholar] [CrossRef] [PubMed]
- Kew, K.M.; Karner, C.; Mindus, S.M.; Ferrara, G. Combination formoterol and budesonide as maintenance and reliever therapy versus combination inhaler maintenance for chronic asthma in adults and children. Cochrane Database Syst. Rev. 2013. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parnham, M.J.; Haber, V.E.; Giamarellos-Bourboulis, E.J.; Perletti, G.; Verleden, G.M.; Vos, R. Azithromycin: Mechanisms of action and their relevance for clinical applications. Pharmacol. Ther. 2014, 143, 225–245. [Google Scholar] [CrossRef]
- Chung, K.F.; Wenzel, S.E.; Brozek, J.L.; Bush, A.; Castro, M.; Sterk, P.J.; Adcock, I.M.; Bateman, E.D.; Bel, E.H.; Bleecker, E.R.; et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur. Respir. J. 2014, 43, 343–373. [Google Scholar] [CrossRef] [Green Version]
- Walker, S.; Monteil, M.; Phelan, K.; Lasserson, T.; Walters, E. Anti-IgE for chronic asthma. In The Cochrane Database of Systematic Reviews; John Wiley & Sons, Ltd: Chichester, UK, 2002. [Google Scholar]
- Ha, F.; Wilson, A.; Powell, C.; Bax, L.; Sj, M. Anti-IL5 therapies for asthma (Cochrane Review). Cochrane Database Syst. Rev. 2017. [Google Scholar] [CrossRef]
- Cabon, Y.; Molinari, N.; Marin, G.; Vachier, I.; Gamez, A.S.; Chanez, P.; Bourdin, A. Comparison of anti-interleukin-5 therapies in patients with severe asthma: Global and indirect meta-analyses of randomized placebo-controlled trials. Clin. Exp. Allergy 2017, 47, 129–138. [Google Scholar] [CrossRef]
- Kolaczkowska, E.; Kubes, P. Neutrophil recruitment and function in health and inflammation. Nat. Rev. Immunol. 2013, 13, 159–175. [Google Scholar] [CrossRef]
- Kreisel, D.; Nava, R.G.; Li, W.; Zinselmeyer, B.H.; Wang, B.; Lai, J.; Pless, R.; Gelman, A.E.; Krupnick, A.S.; Miller, M.J. In vivo two-photon imaging reveals monocyte-dependent neutrophil extravasation during pulmonary inflammation. Proc. Natl. Acad. Sci. USA 2010, 107, 18073–18078. [Google Scholar] [CrossRef] [Green Version]
- Norman, K.E.; Katopodis, A.G.; Thoma, G.; Kolbinger, F.; Hicks, A.E.; Cotter, M.J.; Pockley, A.G.; Hellewell, P.G. P-selectin glycoprotein ligand-1 supports rolling on E- and P-selectin in vivo. Blood 2000, 96, 3585–3591. [Google Scholar] [CrossRef]
- Bullard, D.C. Infectious susceptibility and severe deficiency of leukocyte rolling and recruitment in E-selectin and P-selectin double mutant mice. J. Exp. Med. 1996, 183, 2329–2336. [Google Scholar] [CrossRef] [Green Version]
- Huang, C.-C.; Lu, Y.-F.; Wen, S.-N.; Hsieh, W.-C.; Lin, Y.-C.; Liu, M.-R.; Chiang, E.; Chang, C.-N.; Lin, R.-H. A novel apoptosis-inducing anti-PSGL-1 antibody for T cell-mediated diseases. Eur. J. Immunol. 2005, 35, 2239–2249. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, R.; Hirakawa, J.; Sato, K.; Ikeda, T.; Nagai, M.; Fukuda, M.; Imai, Y.; Kawashima, H. Novel antibodies reactive with sialyl Lewis X in both humans and mice define its critical role in leukocyte trafficking and contact hypersensitivity responses. J. Biol. Chem. 2015, 290, 15313–15326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Homeister, J.W.; Thall, A.D.; Petryniak, B.; Malý, P.; Rogers, C.E.; Smith, P.L.; Kelly, R.J.; Gersten, K.M.; Askari, S.W.; Cheng, G.; et al. The α(1,3)fucosyltransferases FucT-IV and FucT-VII exert collaborative control over selectin-dependent leukocyte recruitment and lymphocyte homing. Immunity 2001, 15, 115–126. [Google Scholar] [CrossRef] [Green Version]
- Sokulsky, L.A.; Garcia-Netto, K.; Nguyen, T.H.; Girkin, J.L.N.; Collison, A.; Mattes, J.; Kaiko, G.; Liu, C.; Bartlett, N.W.; Yang, M.; et al. A critical role for the CXCL3/CXCL5/CXCR2 neutrophilic chemotactic axis in the regulation of type 2 responses in a model of rhinoviral-induced asthma exacerbation. J. Immunol. 2020, 205, 2468–2478. [Google Scholar] [CrossRef] [PubMed]
- Magat, J.M.; Thomas, J.L.; Dumouchel, J.P.; Murray, F.; Li, W.X.; Li, J. Endogenous IL-33 and its autoamplification of IL-33/ST2 pathway play an important role in asthma. J. Immunol. 2020, 204, 1592–1597. [Google Scholar] [CrossRef] [Green Version]
- Weissler, J.C. Eosinophilic lung disease. Am. J. Med. Sci. 2017, 354, 339–349. [Google Scholar] [CrossRef]
- Gorczyca, W.; Sun, Z.Y.; Cronin, W.; Li, X.; Mau, S.; Tugulea, S. Immunophenotypic Pattern of Myeloid Populations by Flow Cytometry Analysis; Elsevier Inc.: Amsterdam, The Netherlands, 2011; Volume 103, ISBN 9780123854933. [Google Scholar]
- Reichman, H.; Rozenberg, P.; Munitz, A. Mouse eosinophils: Identification, isolation, and functional analysis. In Current Protocols in Immunology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2017; Volume 2017, pp. 14.43.1–14.43.22. [Google Scholar] [CrossRef]
- Broide, D.H.; Humber, D.; Sriramarao, P. Inhibition of eosinophil rolling and recruitment in P-selectin- and intracellular adhesion molecule-1-deficient mice. Blood 1998, 91, 2847–2856. [Google Scholar] [CrossRef]
- Impellizzeri, D.; Cuzzocrea, S. Targeting selectins for the treatment of inflammatory diseases. Expert Opin. Ther. Targets 2014, 18, 55–67. [Google Scholar] [CrossRef]
- Ridings, P.C.; Holloway, S.; Bloomfield, G.L.; Phillips, M.L.; Fisher, B.J.; Blocher, C.R.; Sugerman, H.J.; Fowler, A.A. Protective role of synthetic sialylated oligosaccharide in sepsis- induced acute lung injury. J. Appl. Physiol. 1997, 82, 644–651. [Google Scholar] [CrossRef]
- Romano, S.J. Selectin antagonists: Therapeutic potential in asthma and COPD. Treat. Respir. Med. 2005, 4, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Kogan, T.P.; Dupré, B.; Bui, H.; McAbee, K.L.; Kassir, J.M.; Scott, I.L.; Hu, X.; Vanderslice, P.; Beck, P.J.; Dixon, R.A.F. Novel synthetic inhibitors of selectin-mediated cell adhesion: Synthesis of 1,6-bis[3-(3-carboxymethylphenyl)-4-(2-α-D-mannopyranosyloxy)phenyl] hexane (TBC1269). J. Med. Chem. 1998, 41, 1099–1111. [Google Scholar] [CrossRef] [PubMed]
- Abraham, W.M.; Ahmed, A.; Sabater, J.R.; Lauredo, I.T.; Botvinnikova, Y.; Bjercke, R.J.; Hu, X.; Mitch Revelle, B.; Kogan, T.P.; Scott, I.L.; et al. Selectin blockade prevents antigen-induced late bronchial responses and airway hyperresponsiveness in allergic sheep. Am. J. Respir. Crit. Care Med. 1999, 159, 1205–1214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beeh, K.M.; Beier, J.; Meyer, M.; Buhl, R.; Zahlten, R.; Wolff, G. Bimosiamose, an inhaled small-molecule pan-selectin antagonist, attenuates late asthmatic reactions following allergen challenge in mild asthmatics: A randomized, double-blind, placebo-controlled clinical cross-over-trial. Pulm. Pharmacol. Ther. 2006, 19, 233–241. [Google Scholar] [CrossRef]
- Avila, P.C.; Boushey, H.A.; Wong, H.; Grundland, H.; Liu, J.; Fahy, J.V. Effect of a single dose of the selectin inhibitor TBC1269 on early and late asthmatic responses. Clin. Exp. Allergy 2004, 34, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Ehrhardt, C.; Kneuer, C.; Bakowsky, U. Selectins—An emerging target for drug delivery. Adv. Drug Deliv. Rev. 2004, 56, 527–549. [Google Scholar] [CrossRef]
- Fulkerson, P.C.; Rothenberg, M.E. Eosinophil Development, Disease Involvement, and Therapeutic Suppression, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2018; Volume 138. [Google Scholar]
- Takatsu, K.; Nakajima, H. IL-5 and eosinophilia. Curr. Opin. Immunol. 2008, 20, 288–294. [Google Scholar] [CrossRef]
- Wang, J.; Palmer, K.; Lǒtvall, J.; Milan, S.; Lei, X.F.; Matthaei, K.I.; Gauldie, J.; Inman, M.D.; Jordana, M.; Xing, Z. Circulating, but not local lung, IL-5 is required for the development of antigen-induced airways eosinophilia. J. Clin. Investig. 1998, 102, 1132–1141. [Google Scholar] [CrossRef]
- Schmitz, J.; Owyang, A.; Oldham, E.; Song, Y.; Murphy, E.; McClanahan, T.K.; Zurawski, G.; Moshrefi, M.; Qin, J.; Li, X.; et al. IL-33, an interleukin-1-like cytokine that signals via the IL-1 receptor-related protein ST2 and induces T helper type 2-associated cytokines. Immunity 2005, 23, 479–490. [Google Scholar] [CrossRef] [Green Version]
- MacKenzie, J.R.; Mattes, J.; Dent, L.A.; Foster, P.S. Eosinophils promote allergic disease of the lung by regulating CD4 + Th2 lymphocyte function. J. Immunol. 2001, 167, 3146–3155. [Google Scholar] [CrossRef] [Green Version]
- Hirahara, K.; Aoki, A.; Nakayama, T. Pathogenic helper T cells. Allergol. Int. 2021, 70, 169–173. [Google Scholar] [CrossRef]
- Altin, J.; Shen, C.; Liston, A. Understanding the genetic regulation of IgE production. Blood Rev. 2010, 24, 163–169. [Google Scholar] [CrossRef]
- Humbles, A.A.; Conroy, D.M.; Marleau, S.; Rankin, S.M.; Palframan, R.T.; Proudfoot, A.E.I.; Wells, T.N.C.; Li, D.; Jeffery, P.K.; Griffiths-Johnson, D.A.; et al. Kinetics of eotaxin generation and its relationship to eosinophil accumulation in allergic airways disease: Analysis in a guinea pig model in vivo. J. Exp. Med. 1997, 186, 601–612. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hogan, S.P.; Rosenberg, H.F.; Moqbel, R.; Phipps, S.; Foster, P.S.; Lacy, P.; Kay, A.B.; Rothenberg, M.E. Eosinophils: Biological Properties and Role in Health and Disease; Blackwell Publishing Ltd.: Hoboken, NJ, USA, 2008; Volume 38, ISBN 9781405157209. [Google Scholar]
- Davoine, F.; Lacy, P. Eosinophil cytokines, chemokines, and growth factors: Emerging roles in immunity. Front. Immunol. 2014, 5, 1–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitroulis, I.; Alexaki, V.I.; Kourtzelis, I.; Ziogas, A.; Hajishengallis, G.; Chavakis, T. Leukocyte integrins: Role in leukocyte recruitment and as therapeutic targets in inflammatory disease. Pharmacol. Ther. 2015, 147, 123–135. [Google Scholar] [CrossRef] [Green Version]
- Kawashima, H.; Petryniak, B.; Hiraoka, N.; Mitoma, J.; Huckaby, V.; Nakayama, J.; Uchimura, K.; Kadomatsu, K.; Muramatsu, T.; Lowe, J.B.; et al. N-acetylglucosamine-6-O-sulfotransferases 1 and 2 cooperatively control lymphocyte homing through L-selectin ligand biosynthesis in high endothelial venules. Nat. Immunol. 2005, 6, 1096–1104. [Google Scholar] [CrossRef]
- Fiscus, L.C.; Van Herpen, J.; Steeber, D.A.; Tedder, T.F.; Tang, M.L.K. L-selectin is required for the development of airway hyperresponsiveness but not airway inflammation in a murine model of asthma. J. Allergy Clin. Immunol. 2001, 107, 1019–1024. [Google Scholar] [CrossRef]
- Rosen, S.D.; Tsay, D.; Singer, M.S.; Hemmerich, S.; Abraham, W.M. Therapeutic targeting of endothelial ligands for L-selectin (PNAd) in a sheep model of asthma. Am. J. Pathol. 2005, 166, 935–944. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiong, W.; Liu, W.; Nishida, S.; Komiyama, D.; Liu, W.; Hirakawa, J.; Kawashima, H. Therapeutic Effects of an Anti-sialyl Lewis X Antibody in a Murine Model of Allergic Asthma. Int. J. Mol. Sci. 2021, 22, 9961. https://doi.org/10.3390/ijms22189961
Xiong W, Liu W, Nishida S, Komiyama D, Liu W, Hirakawa J, Kawashima H. Therapeutic Effects of an Anti-sialyl Lewis X Antibody in a Murine Model of Allergic Asthma. International Journal of Molecular Sciences. 2021; 22(18):9961. https://doi.org/10.3390/ijms22189961
Chicago/Turabian StyleXiong, Wei, Wenxin Liu, Shogo Nishida, Daichi Komiyama, Wei Liu, Jotaro Hirakawa, and Hiroto Kawashima. 2021. "Therapeutic Effects of an Anti-sialyl Lewis X Antibody in a Murine Model of Allergic Asthma" International Journal of Molecular Sciences 22, no. 18: 9961. https://doi.org/10.3390/ijms22189961
APA StyleXiong, W., Liu, W., Nishida, S., Komiyama, D., Liu, W., Hirakawa, J., & Kawashima, H. (2021). Therapeutic Effects of an Anti-sialyl Lewis X Antibody in a Murine Model of Allergic Asthma. International Journal of Molecular Sciences, 22(18), 9961. https://doi.org/10.3390/ijms22189961





