Immunoelectron Microscopic Characterization of Vasopressin-Producing Neurons in the Hypothalamo-Pituitary Axis of Non-Human Primates by Use of Formaldehyde-Fixed Tissues Stored at −25 °C for Several Years
Abstract
:1. Introduction
2. Results
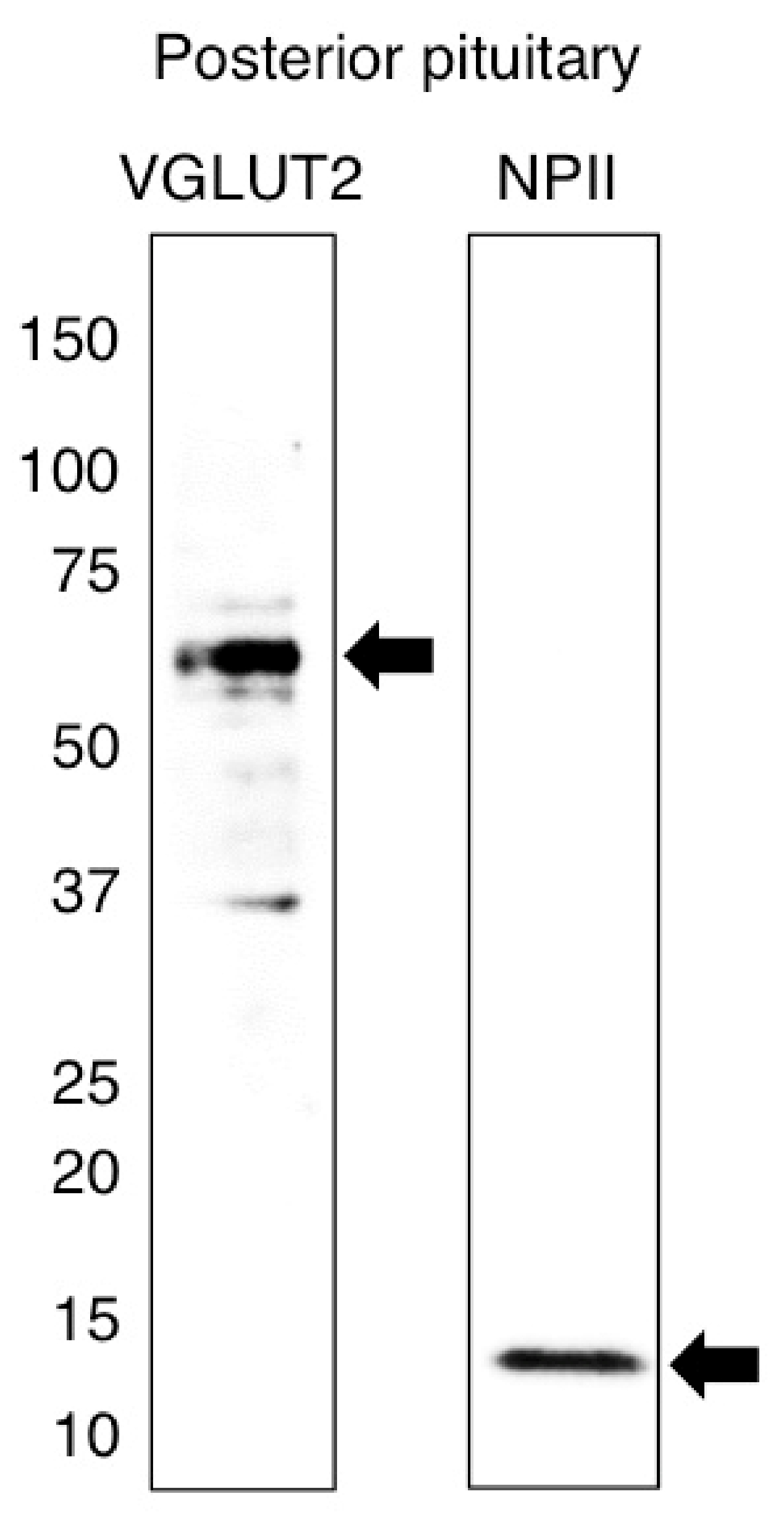
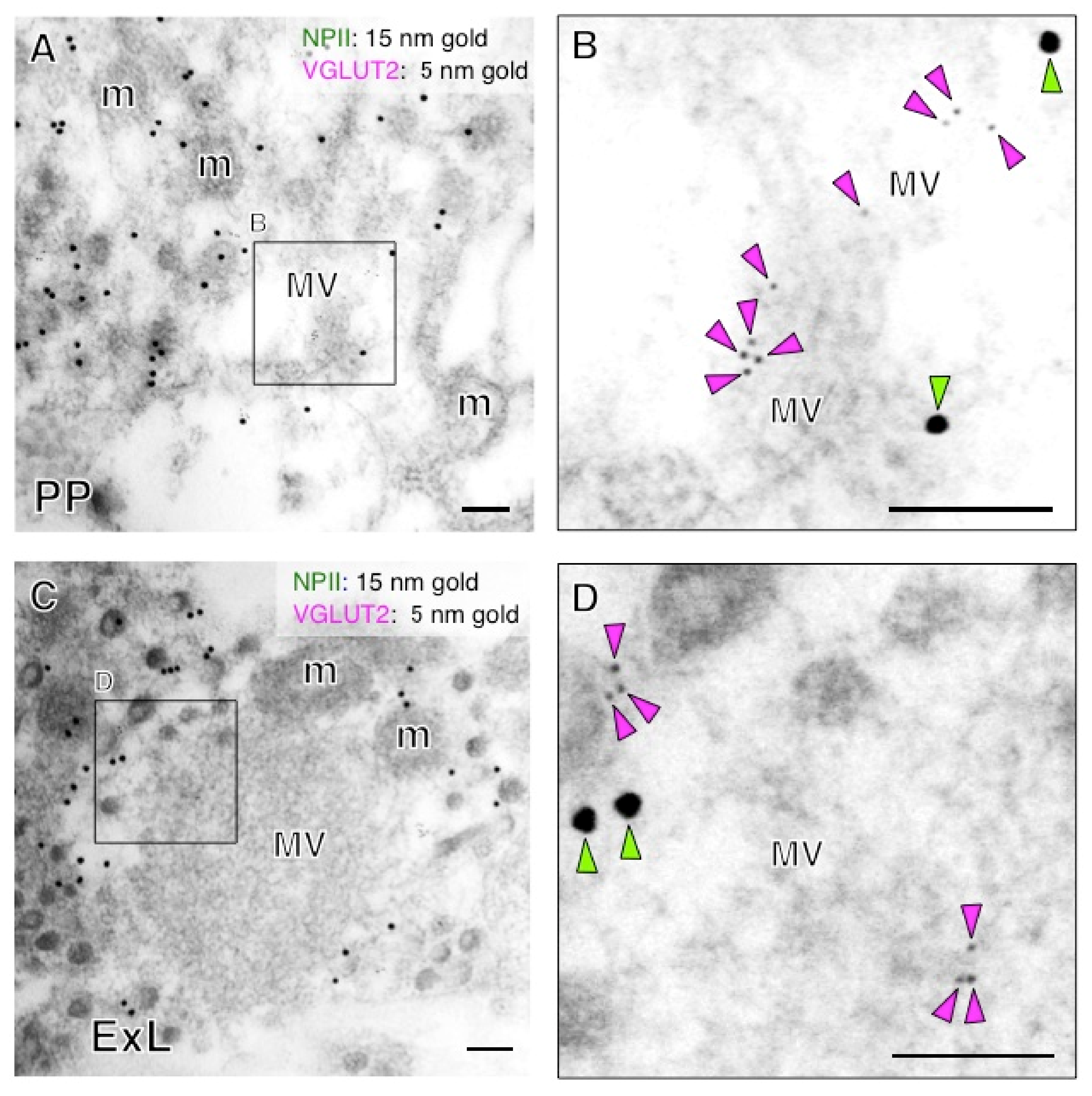
2.1. Antibody Characterization and the Expression of VGLUT2 at the Protein Level in the Posterior Pituitary
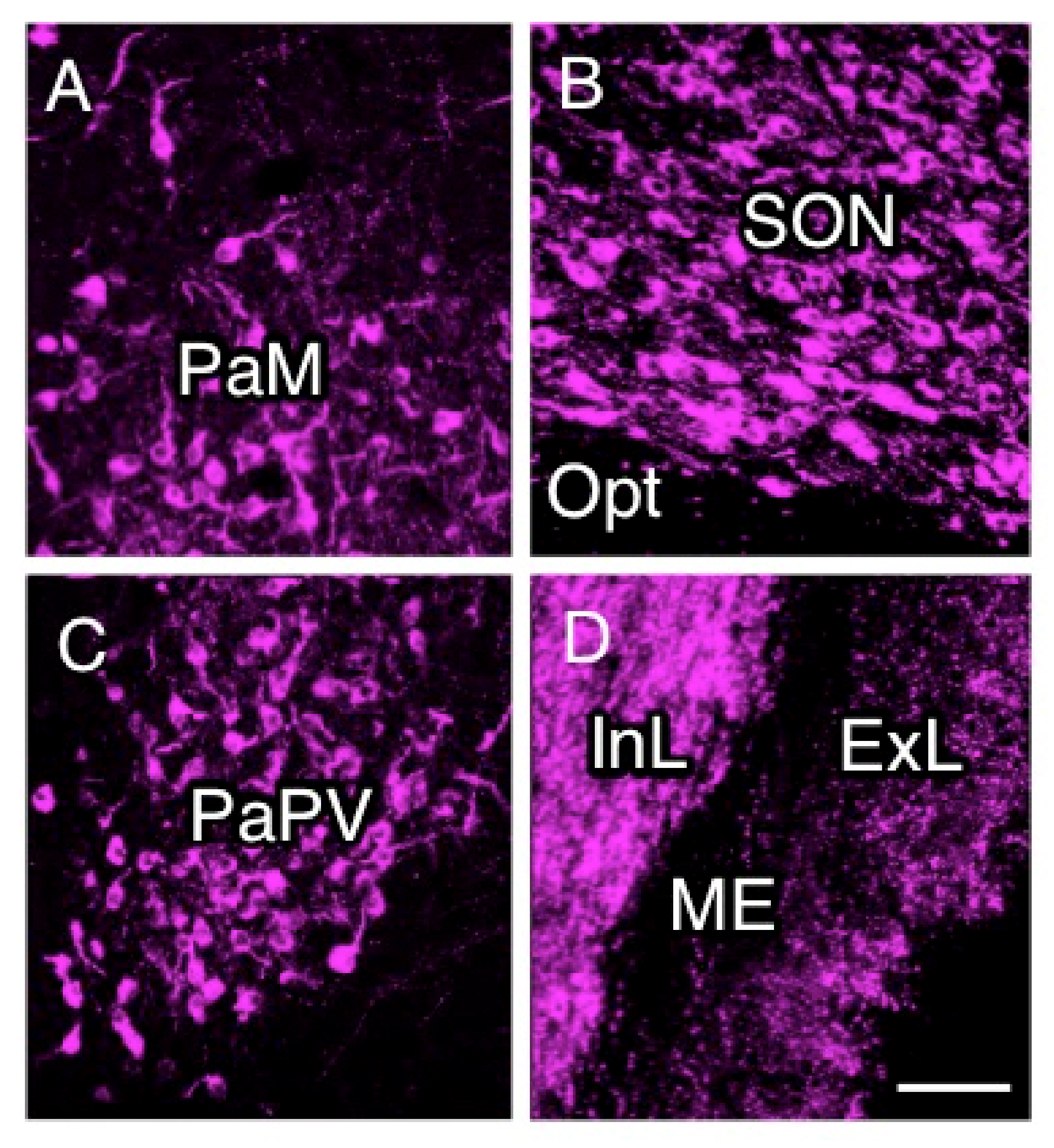
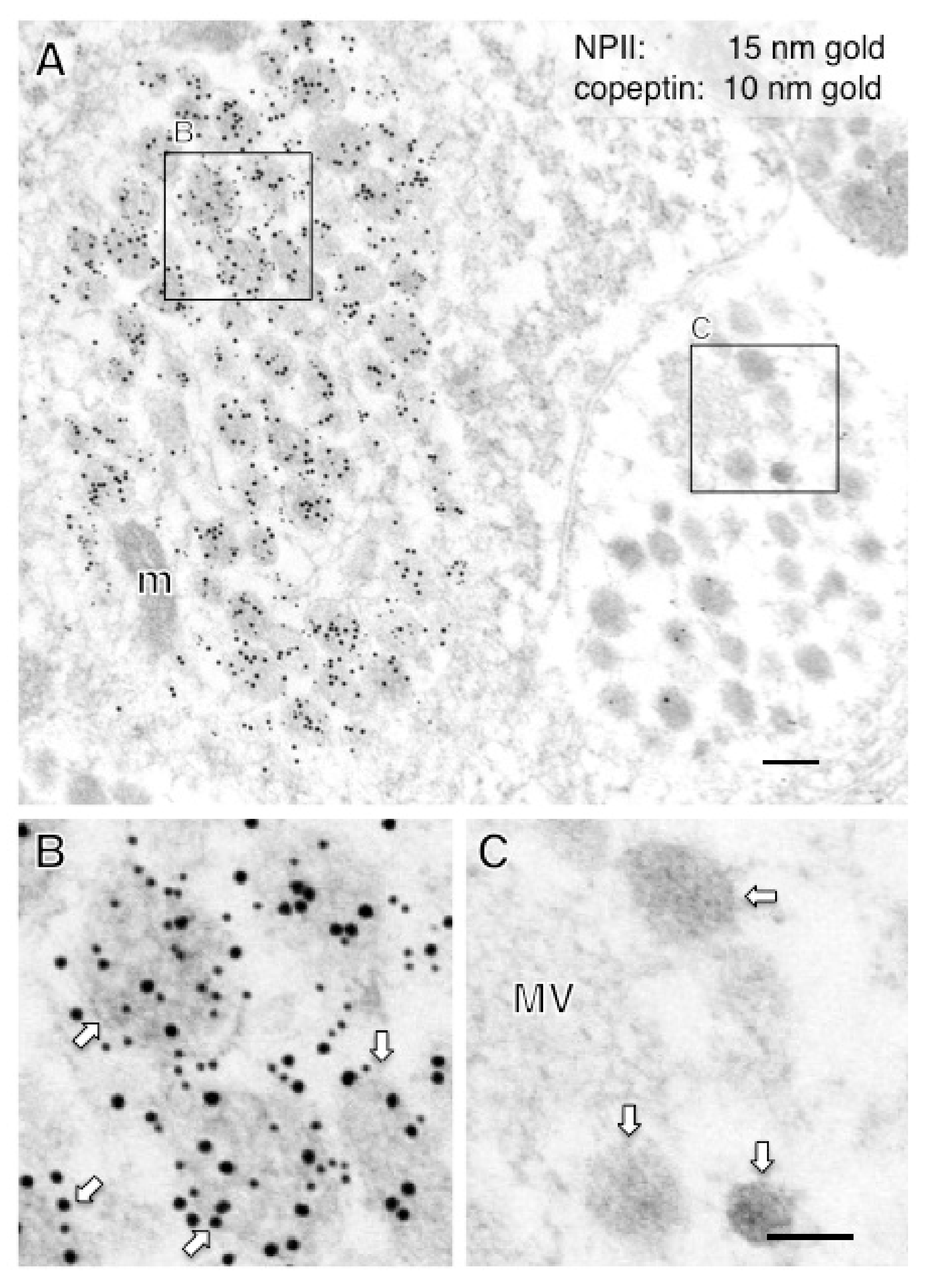
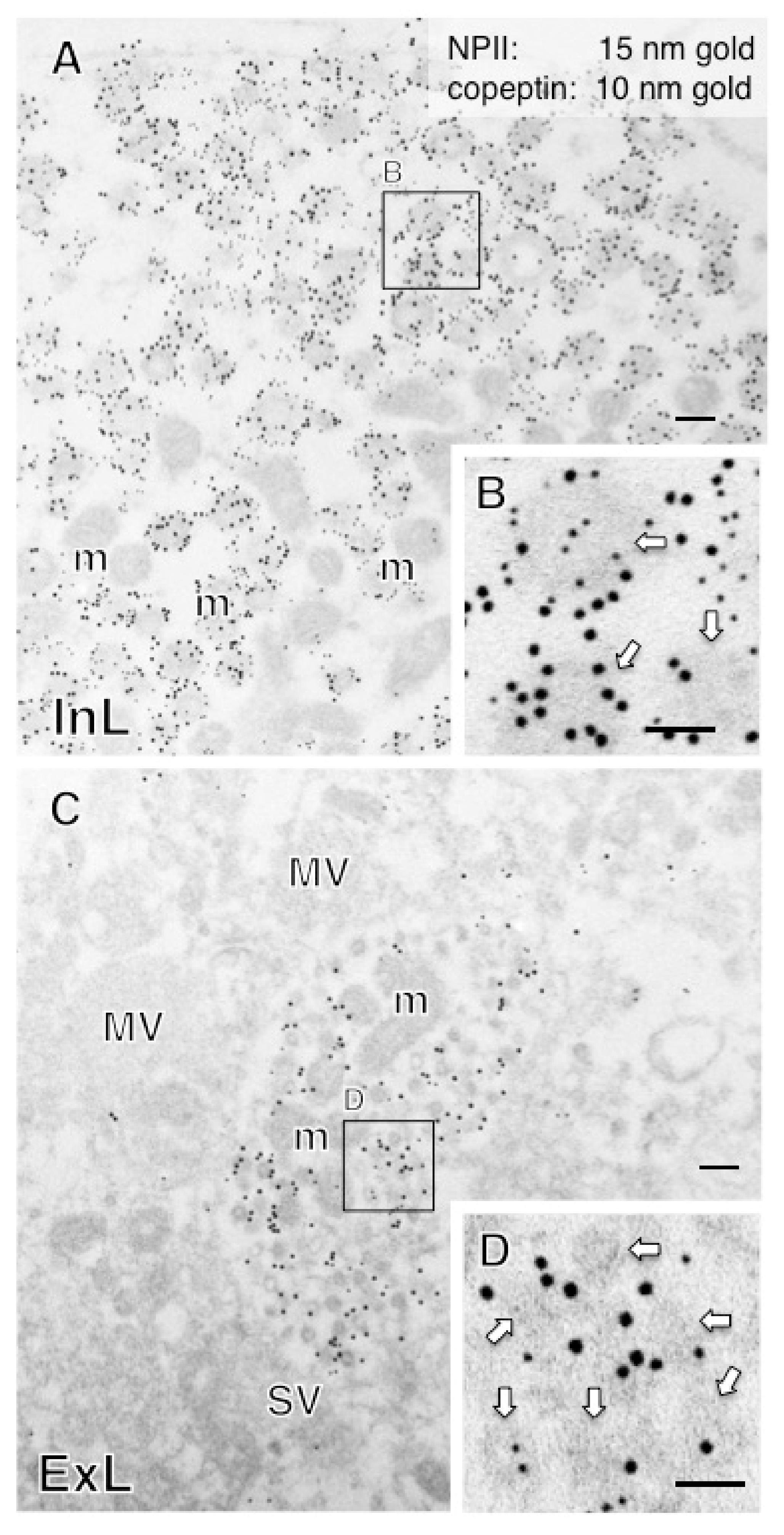
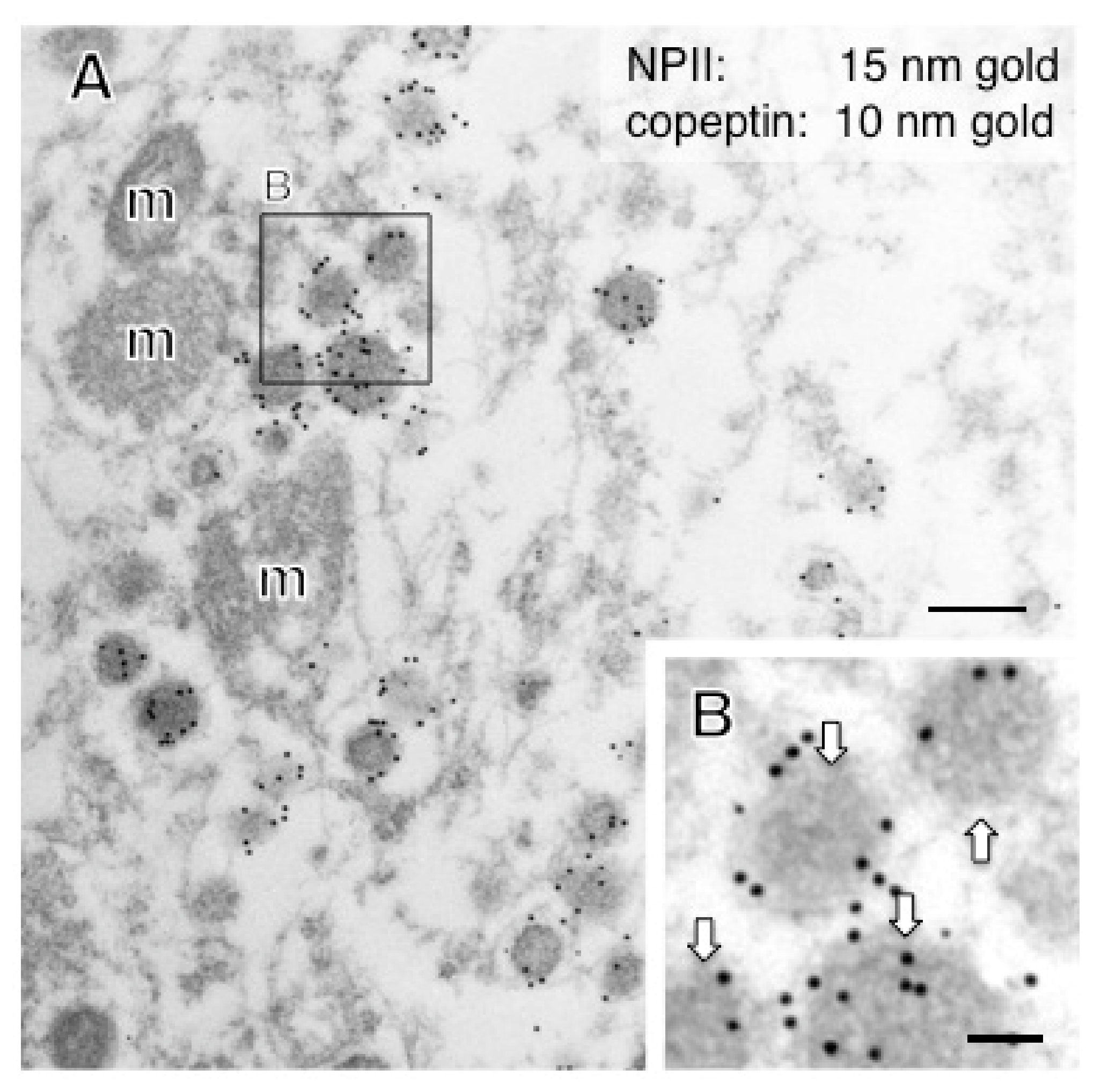
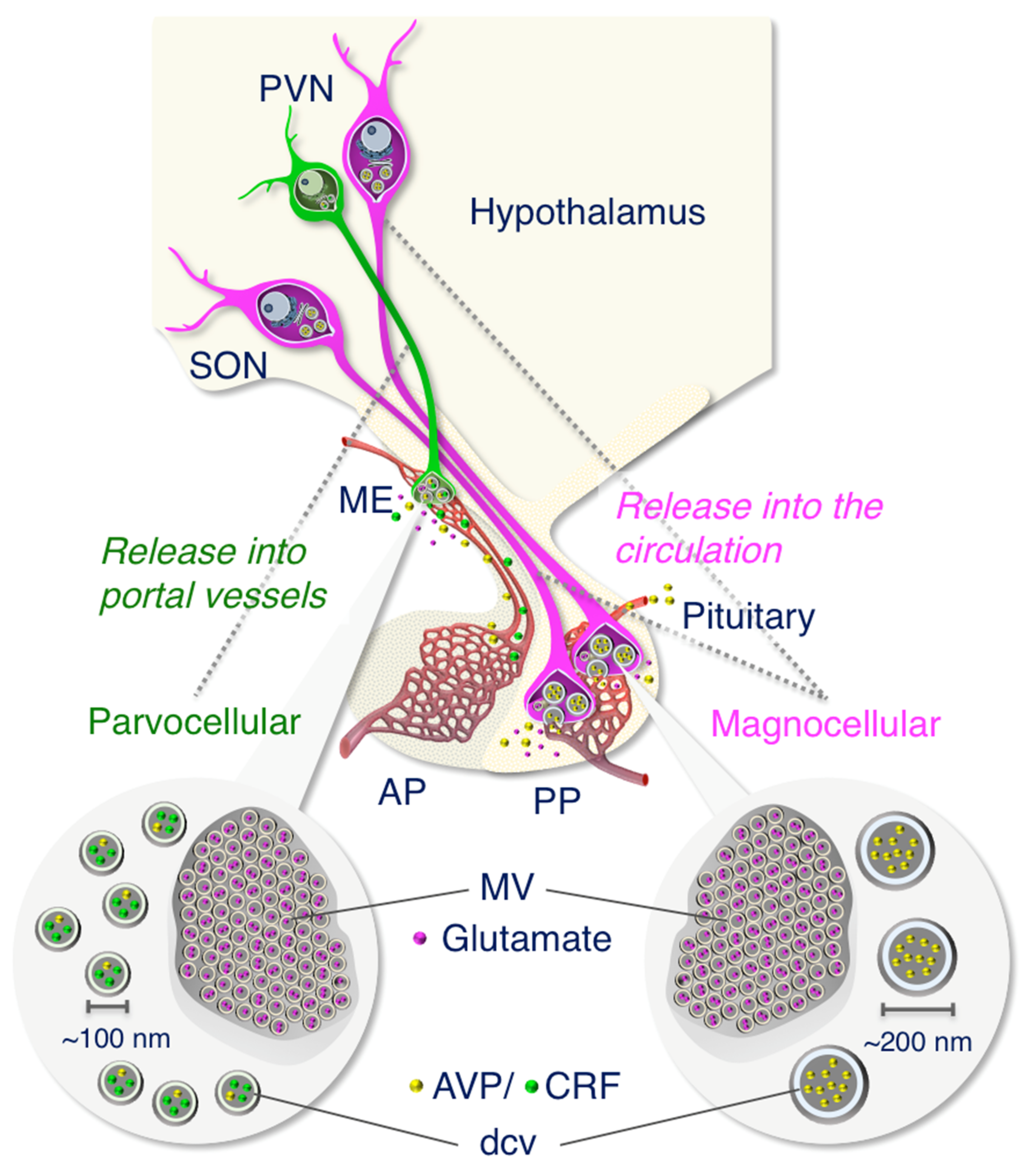
2.2. Anatomy of AVP-Producing Neurons in the Macaque Hypothalamus
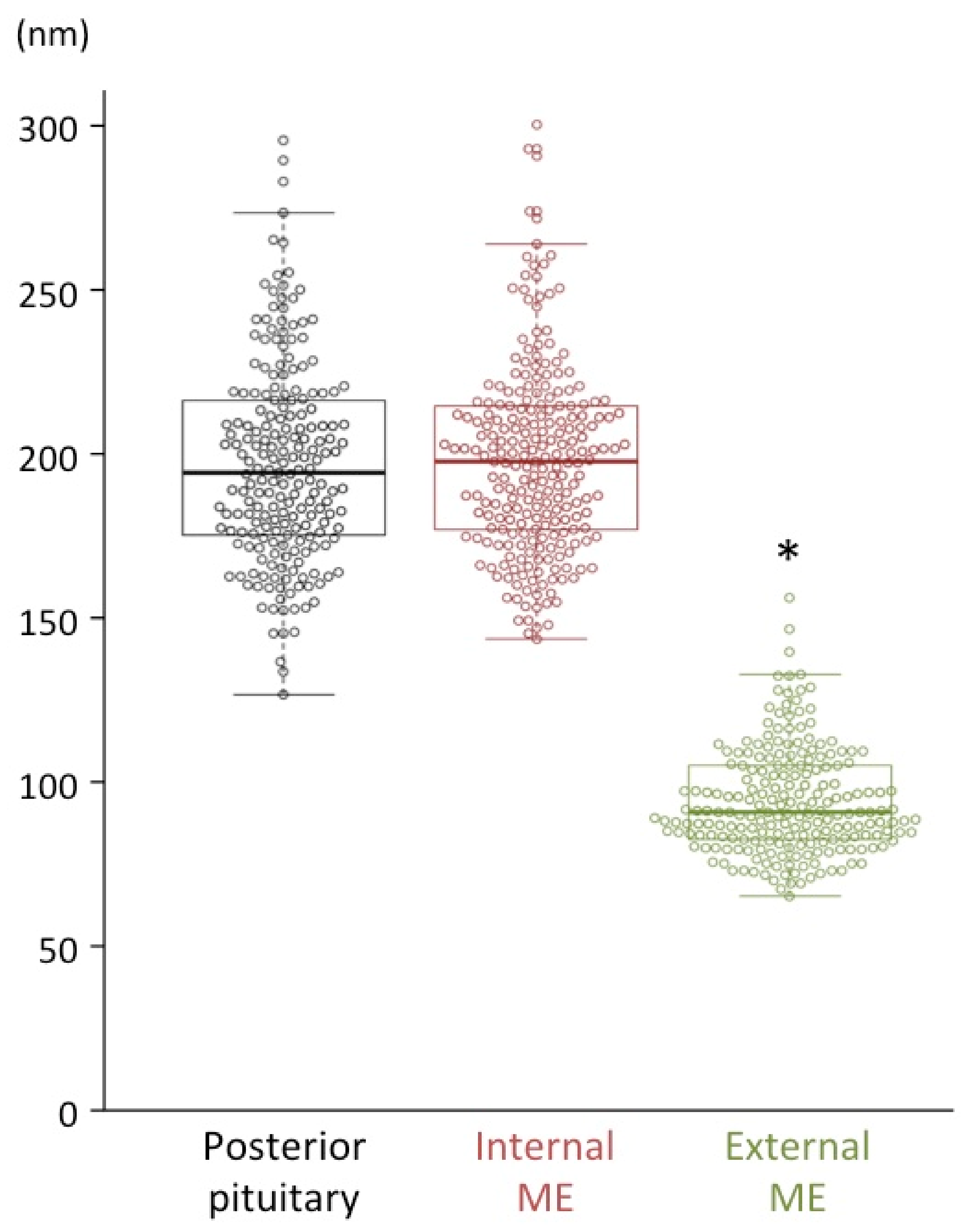
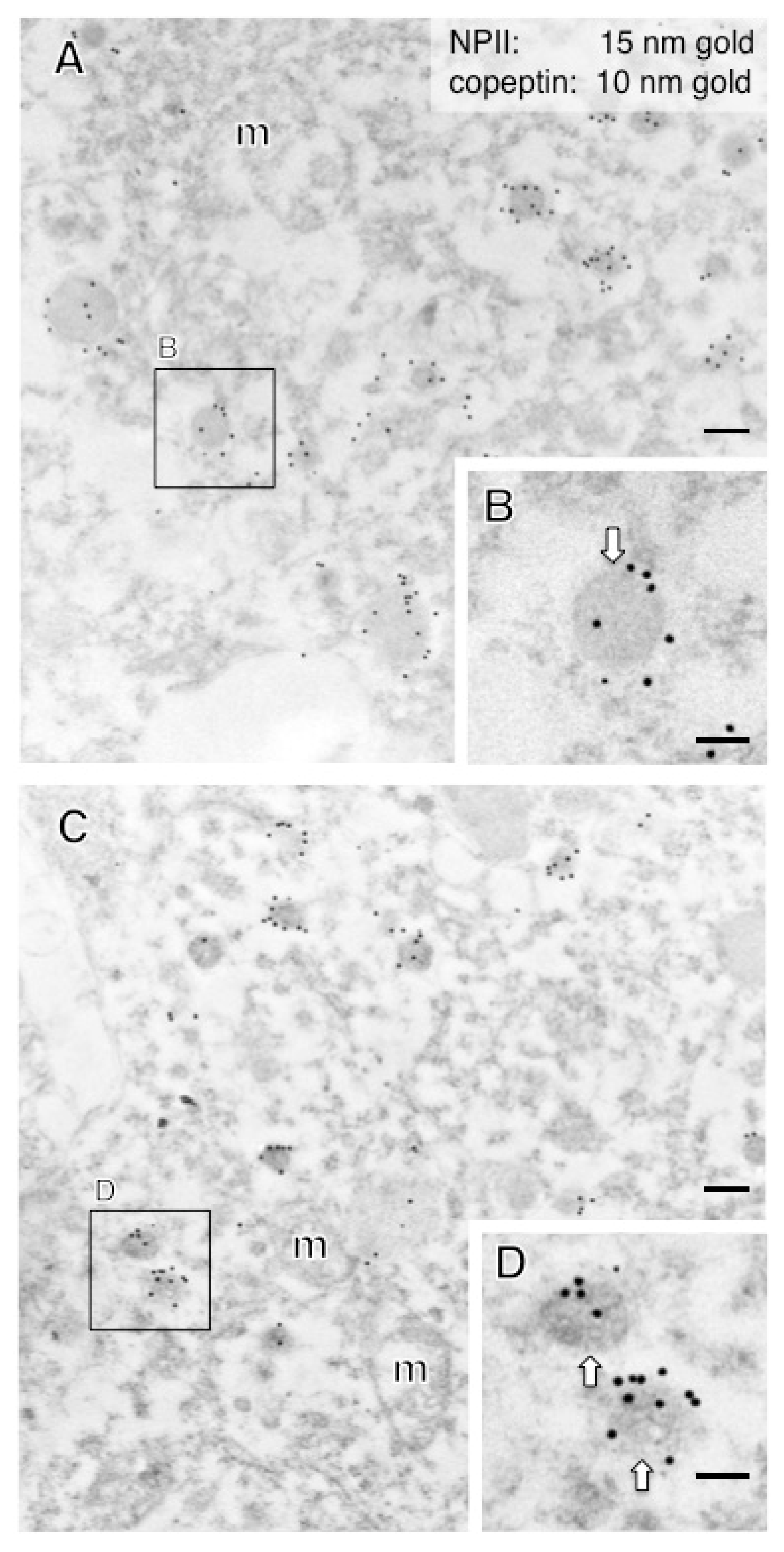
2.3. Size Characterization of Dcv
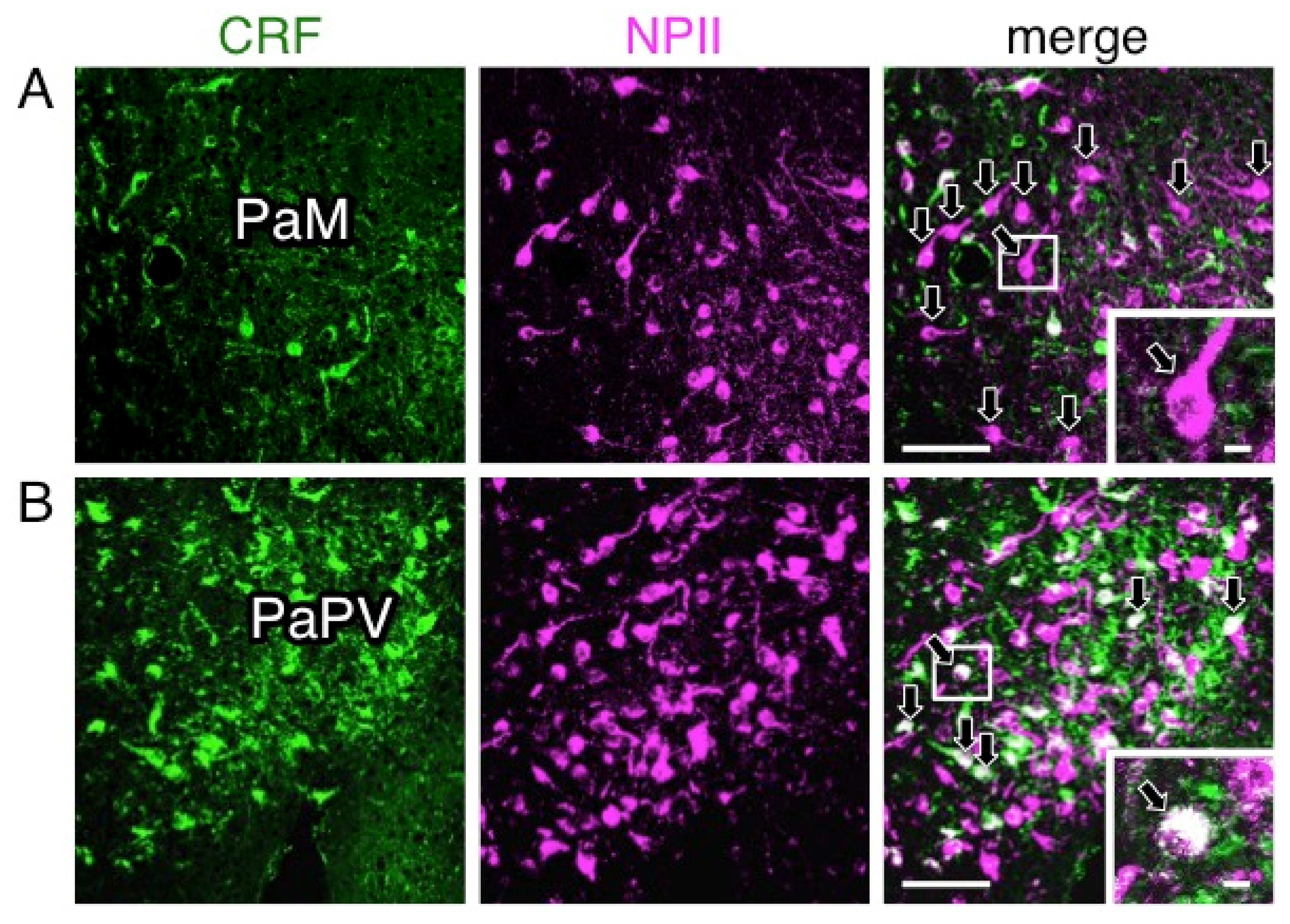
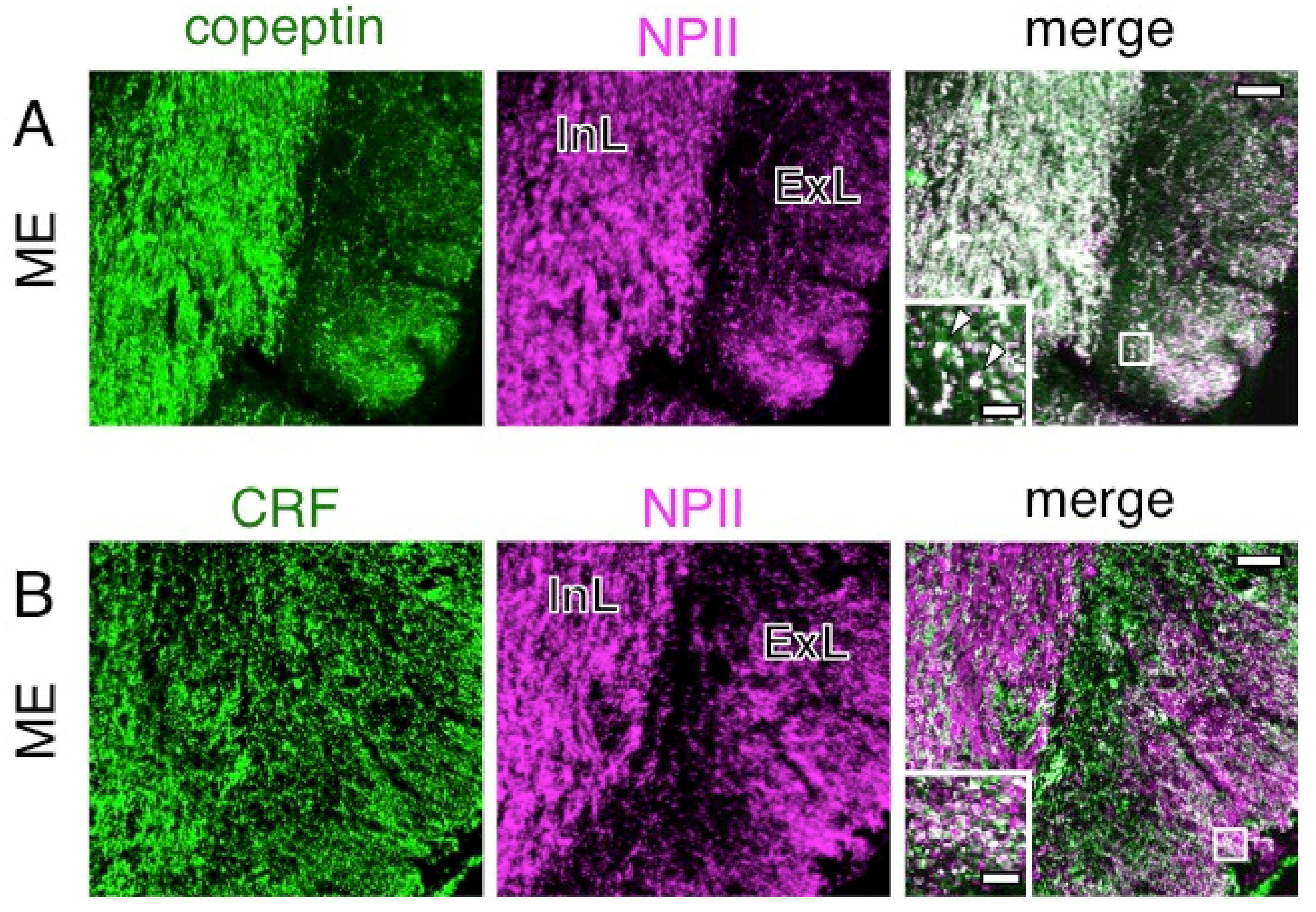
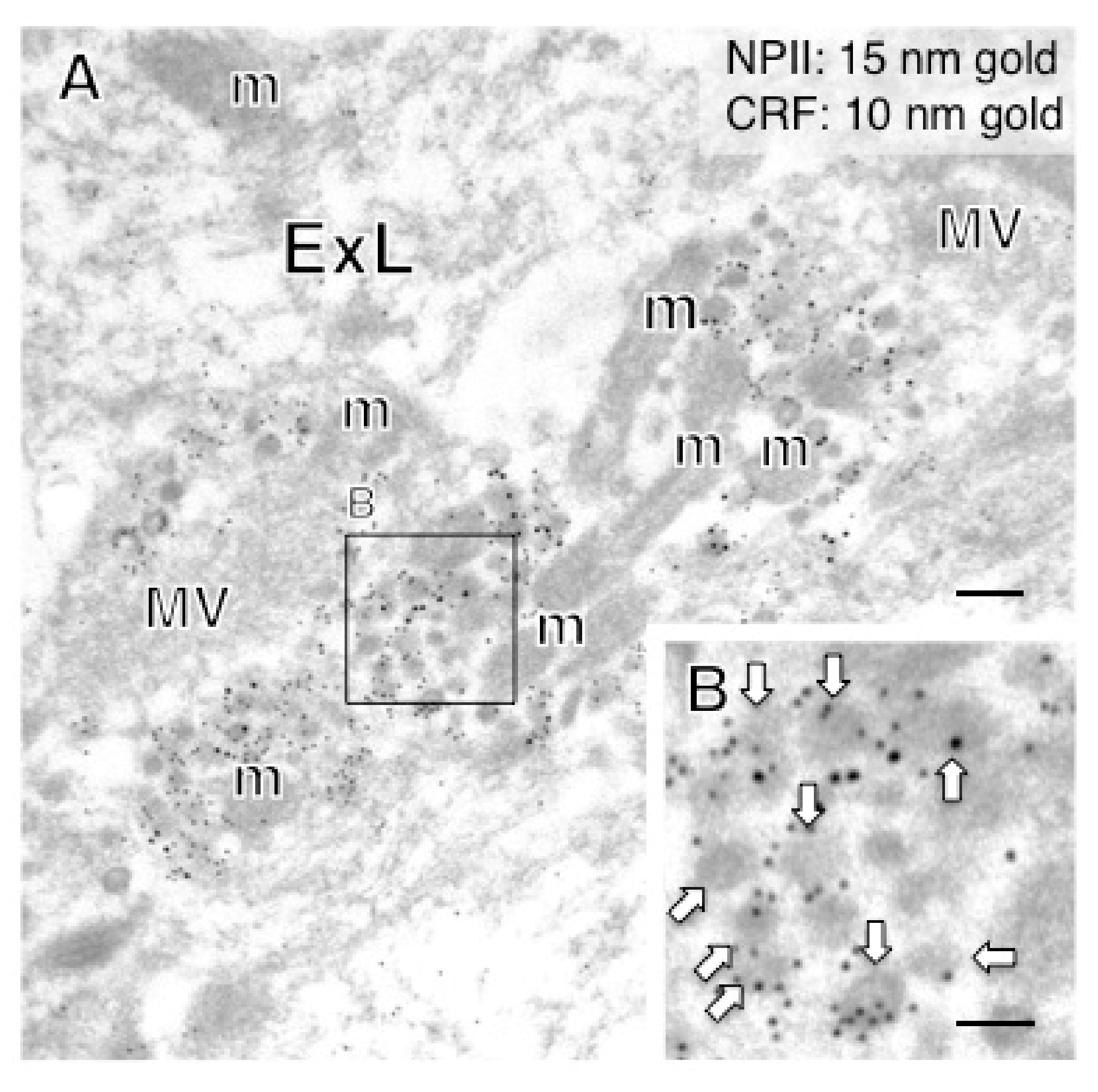
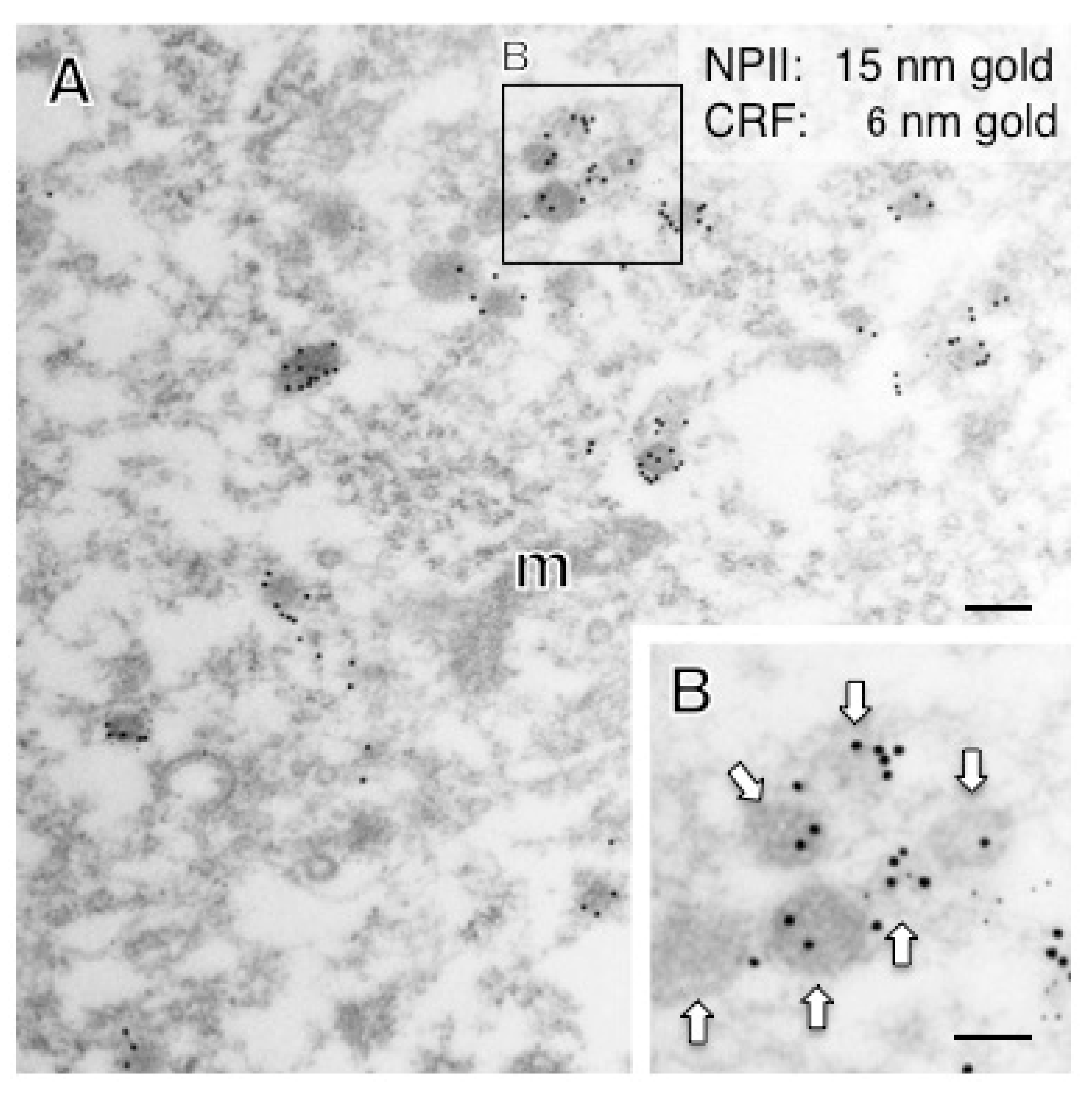
2.4. A Subpopulation of Parvocellular AVP Neurons in the PVN Co-Expresses CRF
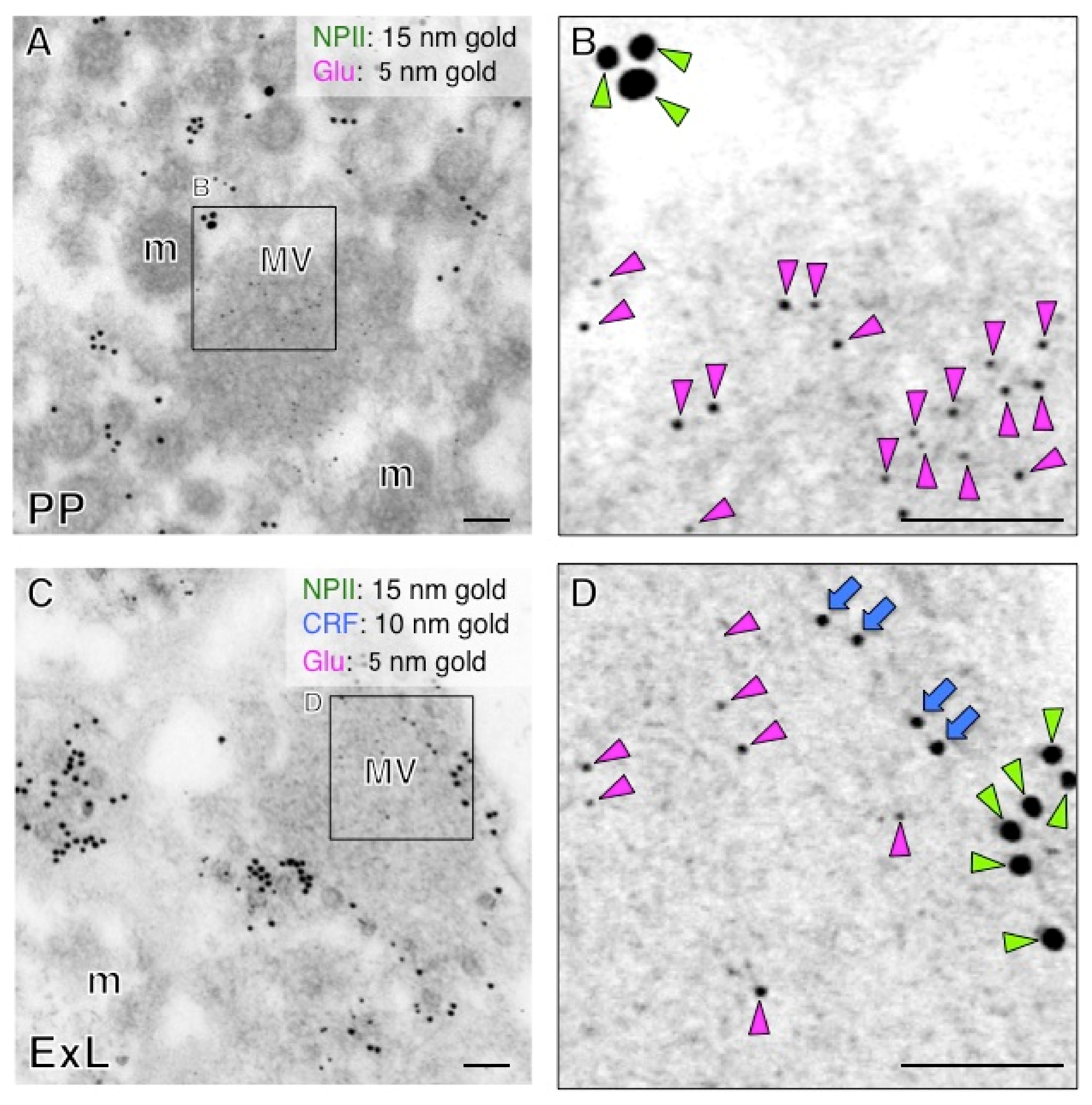
2.5. Both Magno- and Parvocellular AVP Neurons Are Glutamatergic
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Tissue Processing
4.3. Immunofluorescence
4.4. Post-Embedding Immunoelectron Microscopy
4.5. Western Blotting
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ANOVA | analysis of variance |
| AVP | arginine vasopressin |
| CRF | corticotrophin-releasing factor |
| dcv | dense-cored neurosecretory vesicle |
| HRP | horseradish peroxidase |
| NP | neurophysin |
| PB | phosphate buffer |
| PBS | phosphate-buffered saline |
| PFA | paraformaldehyde |
| PVDF | polyvinylidene difluoride |
| PVN | paraventricular nucleus |
| SEM | standard error of the mean |
| SON | supraoptic nucleus |
| Tris-HCl | trishydroxymethyl-aminomethane-HCl |
| TBST | Tris-HCl-buffered saline |
| VGLUT2 | vesicular glutamate transporter 2 |
References
- Abbott, A. Biomedicine: The changing face of primate research. Nature 2014, 506, 24–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walum, H.; Young, L.J. The neural mechanisms and circuitry of the pair bond. Nat. Rev. Neurosci. 2018, 19, 643–654. [Google Scholar] [CrossRef] [PubMed]
- Buijs, R.M.; De Vries, G.J.; Van Leeuwen, F.W.; Swaab, D.F. Vasopressin and oxytocin: Distribution and putative functions in the brain. Prog. Brain Res. 1983, 60, 115–122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Breslow, E. Structure and folding properties of neurophysin and its peptide complexes: Biological implications. Regul. Pept. 1993, 45, 15–19. [Google Scholar] [CrossRef]
- Davies, J.; Waller, S.; Zeng, Q.; Wells, S.; Murphy, D. Further delineation of the sequences required for the expression and physiological regulation of the vasopressin gene in transgenic rat hypothalamic magnocellular neurones. J. Neuroendocrinol. 2003, 15, 42–50. [Google Scholar] [CrossRef]
- Land, H.; Schutz, G.; Schmale, H.; Richter, D. Nucleotide sequence of cloned cDNA encoding bovine arginine vasopressin-neurophysin II precursor. Nature 1982, 295, 299–303. [Google Scholar] [CrossRef]
- Koshimizu, T.A.; Nakamura, K.; Egashira, N.; Hiroyama, M.; Nonoguchi, H.; Tanoue, A. Vasopressin V1a and V1b receptors: From molecules to physiological systems. Physiol. Rev. 2012, 92, 1813–1864. [Google Scholar] [CrossRef] [Green Version]
- Morris, J. Neurosecretory Vesicles: Structure, Distribution, Release and Breakdown. In Neurosecretion: Secretory Mechanisms; Lemons, J.R., Dayanithi, G., Eds.; Springer Nature Switzerland: Cham, Switzerland, 2020; Volume 8, pp. 81–102. [Google Scholar]
- Vale, W.; Spiess, J.; Rivier, C.; Rivier, J. Characterization of a 41-residue ovine hypothalamic peptide that stimulates secretion of corticotropin and beta-endorphin. Science 1981, 213, 1394–1397. [Google Scholar] [CrossRef] [PubMed]
- Fellmann, D.; Bugnon, C.; Bresson, J.L.; Gouget, A.; Cardot, J.; Clavequin, M.C.; Hadjiyiassemis, M. The CRF neuron: Immunocytochemical study. Peptides 1984, 5 (Suppl. 1), 19–33. [Google Scholar] [CrossRef]
- Itoi, K.; Talukder, A.H.; Fuse, T.; Kaneko, T.; Ozawa, R.; Sato, T.; Sugaya, T.; Uchida, K.; Yamazaki, M.; Abe, M.; et al. Visualization of corticotropin-releasing factor neurons by fluorescent proteins in the mouse brain and characterization of labeled neurons in the paraventricular nucleus of the hypothalamus. Endocrinology 2014, 155, 4054–4060. [Google Scholar] [CrossRef]
- Mouri, T.; Itoi, K.; Takahashi, K.; Suda, T.; Murakami, O.; Yoshinaga, K.; Andoh, N.; Ohtani, H.; Masuda, T.; Sasano, N. Colocalization of corticotropin-releasing factor and vasopressin in the paraventricular nucleus of the human hypothalamus. Neuroendocrinology 1993, 57, 34–39. [Google Scholar] [CrossRef]
- Whitnall, M.H.; Mezey, E.; Gainer, H. Co-localization of corticotropin-releasing factor and vasopressin in median eminence neurosecretory vesicles. Nature 1985, 317, 248–250. [Google Scholar] [CrossRef]
- Otubo, A.; Kawakami, N.; Maejima, S.; Ueda, Y.; Morris, J.F.; Sakamoto, T.; Sakamoto, H. Vasopressin gene products are colocalised with corticotrophin-releasing factor within neurosecretory vesicles in the external zone of the median eminence of the Japanese macaque monkey (Macaca fuscata). J. Neuroendocrinol. 2020, 32, e12875. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, E.A.; Carmel, P.W.; Husain, M.K.; Ferin, M.; Tannenbaum, M.; Frantz, A.G.; Robinson, A.G. Vasopressin and neurophysin: High concentrations in monkey hypophyseal portal blood. Science 1973, 182, 925–927. [Google Scholar] [CrossRef] [PubMed]
- Meeker, R.B.; Swanson, D.J.; Greenwood, R.S.; Hayward, J.N. Ultrastructural distribution of glutamate immunoreactivity within neurosecretory endings and pituicytes of the rat neurohypophysis. Brain Res. 1991, 564, 181–193. [Google Scholar] [CrossRef]
- Meeker, R.B.; Swanson, D.J.; Hayward, J.N. Light and electron microscopic localization of glutamate immunoreactivity in the supraoptic nucleus of the rat hypothalamus. Neuroscience 1989, 33, 157–167. [Google Scholar] [CrossRef]
- Valentino, R.J.; Rudoy, C.; Saunders, A.; Liu, X.B.; Van Bockstaele, E.J. Corticotropin-releasing factor is preferentially colocalized with excitatory rather than inhibitory amino acids in axon terminals in the peri-locus coeruleus region. Neuroscience 2001, 106, 375–384. [Google Scholar] [CrossRef]
- Maunsbach, A.B. The influence of different fixatives and fixation methods on the ultrastructure of rat kidney proximal tubule cells. II. Effects of varying osmolality, ionic strength, buffer system and fixative concentration of glutaraldehyde solutions. J. Ultrastruct. Res. 1966, 15, 283–309. [Google Scholar] [CrossRef]
- Maunsbach, A.B. The influence of different fixatives and fixation methods on the ultrastructure of rat kidney proximal tubule cells. I. Comparison of different perfusion fixation methods and of glutaraldehyde, formaldehyde and osmium tetroxide fixatives. J. Ultrastruct. Res. 1966, 15, 242–282. [Google Scholar] [CrossRef]
- Balaram, P.; Hackett, T.A.; Kaas, J.H. Differential expression of vesicular glutamate transporters 1 and 2 may identify distinct modes of glutamatergic transmission in the macaque visual system. J. Chem. Neuroanat. 2013, 50–51, 21–38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kawakami, N.; Otubo, A.; Maejima, S.; Talukder, A.H.; Satoh, K.; Oti, T.; Takanami, K.; Ueda, Y.; Itoi, K.; Morris, J.F.; et al. Variation of pro-vasopressin processing in parvocellular and magnocellular neurons in the paraventricular nucleus of the hypothalamus: Evidence from the vasopressin-related glycopeptide copeptin. J. Comp. Neurol. 2021, 529, 1372–1390. [Google Scholar] [CrossRef] [PubMed]
- Satoh, K.; Oti, T.; Katoh, A.; Ueta, Y.; Morris, J.F.; Sakamoto, T.; Sakamoto, H. In vivo processing and release into the circulation of GFP fusion protein in arginine vasopressin enhanced GFP transgenic rats: Response to osmotic stimulation. FEBS J. 2015, 282, 2488–2499. [Google Scholar] [CrossRef]
- Sawchenko, P.E.; Swanson, L.W.; Vale, W.W. Co-expression of corticotropin-releasing factor and vasopressin immunoreactivity in parvocellular neurosecretory neurons of the adrenalectomized rat. Proc. Natl. Acad. Sci. USA 1984, 81, 1883–1887. [Google Scholar] [CrossRef] [Green Version]
- Foote, S.L.; Cha, C.I. Distribution of corticotropin-releasing-factor-like immunoreactivity in brainstem of two monkey species (Saimiri sciureus and Macaca fascicularis): An immunohistochemical study. J. Comp. Neurol. 1988, 276, 239–264. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Ward, A.R.; Morris, J.F. Oestradiol acutely stimulates exocytosis of oxytocin and vasopressin from dendrites and somata of hypothalamic magnocellular neurons. Neuroscience 1995, 68, 1179–1188. [Google Scholar] [CrossRef]
- Castel, M.; Morris, J.F.; Whitnall, M.H.; Sivan, N. Improved visualization of the immunoreactive hypothalamo-neurohypophysial system by use of immuno-gold techniques. Cell Tissue Res. 1986, 243, 193–204. [Google Scholar] [CrossRef]
- Castel, M.; Morris, J.F. The neurophysin-containing innervation of the forebrain of the mouse. Neuroscience 1988, 24, 937–966. [Google Scholar] [CrossRef]
- Frahm, S.; Antolin-Fontes, B.; Gorlich, A.; Zander, J.F.; Ahnert-Hilger, G.; Ibanez-Tallon, I. An essential role of acetylcholine-glutamate synergy at habenular synapses in nicotine dependence. eLife 2015, 4, e11396. [Google Scholar] [CrossRef]
- Mills, F.; Globa, A.K.; Liu, S.; Cowan, C.M.; Mobasser, M.; Phillips, A.G.; Borgland, S.L.; Bamji, S.X. Cadherins mediate cocaine-induced synaptic plasticity and behavioral conditioning. Nat. Neurosci. 2017, 20, 540–549. [Google Scholar] [CrossRef] [Green Version]
- Sun, D.; Vingrys, A.J.; Kalloniatis, M. Metabolic and functional profiling of the normal rat retina. J. Comp. Neurol. 2007, 505, 92–113. [Google Scholar] [CrossRef] [PubMed]
- Hou, S.; Duale, H.; Cameron, A.A.; Abshire, S.M.; Lyttle, T.S.; Rabchevsky, A.G. Plasticity of lumbosacral propriospinal neurons is associated with the development of autonomic dysreflexia after thoracic spinal cord transection. J. Comp. Neurol. 2008, 509, 382–399. [Google Scholar] [CrossRef] [Green Version]
- Kawata, M.; Sano, Y. Immunohistochemical identification of the oxytocin and vasopressin neurons in the hypothalamus of the monkey (Macaca fuscata). Anat. Embryol. 1982, 165, 151–167. [Google Scholar] [CrossRef]
- Cannata, M.A.; Morris, J.F. Changes in the appearance of hypothalamo-neurohypophysial neurosecretory granules associated with their maturation. J. Endocrinol. 1973, 57, 531–538. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, D.R.; Cullinan, W.E.; Herman, J.P. Distribution of vesicular glutamate transporter mRNA in rat hypothalamus. J. Comp. Neurol. 2002, 448, 217–229. [Google Scholar] [CrossRef]
- Zhang, L.; Hernandez, V.S.; Zetter, M.A.; Eiden, L.E. VGLUT-VGAT expression delineates functionally specialised populations of vasopressin-containing neurones including a glutamatergic perforant path-projecting cell group to the hippocampus in rat and mouse brain. J. Neuroendocrinol. 2020, 32, e12831. [Google Scholar] [CrossRef]
- Hrabovszky, E.; Liposits, Z. Glutamatergic phenotype of hypothalamic neurosecretory systems: A novel aspect of central neuroendocrine regulation. Ideggyogy Sz. 2007, 60, 182–186. [Google Scholar] [PubMed]
- Brann, D.W. Glutamate: A major excitatory transmitter in neuroendocrine regulation. Neuroendocrinology 1995, 61, 213–225. [Google Scholar] [CrossRef] [PubMed]
- Hrabovszky, E.; Csapo, A.K.; Kallo, I.; Wilheim, T.; Turi, G.F.; Liposits, Z. Localization and osmotic regulation of vesicular glutamate transporter-2 in magnocellular neurons of the rat hypothalamus. Neurochem. Int. 2006, 48, 753–761. [Google Scholar] [CrossRef]
- Rehbein, M.; Hillers, M.; Mohr, E.; Ivell, R.; Morley, S.; Schmale, H.; Richter, D. The neurohypophyseal hormones vasopressin and oxytocin. Precursor structure, synthesis and regulation. Biol. Chem. Hoppe-Seyler 1986, 367, 695–704. [Google Scholar] [CrossRef]
- Hara, Y.; Battey, J.; Gainer, H. Structure of mouse vasopressin and oxytocin genes. Brain Res. Mol. Brain Res. 1990, 8, 319–324. [Google Scholar] [CrossRef]
- Naugle, M.M.; Lozano, S.A.; Guarraci, F.A.; Lindsey, L.F.; Kim, J.E.; Morrison, J.H.; Janssen, W.G.; Yin, W.; Gore, A.C. Age and Long-Term Hormone Treatment Effects on the Ultrastructural Morphology of the Median Eminence of Female Rhesus Macaques. Neuroendocrinology 2016, 103, 650–664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holmes, R.L. Comparative Observations on Inclusions in Nerve Fibres of the Mammalian Neurohypophysis. Z Zellforsch Mikrosk Anat. 1964, 64, 474–492. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, W.E.; Tian, M. Separate ultrastructural distributions of neurophysin and C-terminal glycopeptide within dense core vesicles in rat neural lobe. Brain Res. 1991, 562, 144–148. [Google Scholar] [CrossRef]
- Morris, J.F. Hormone storage in individual neurosecretory granules of the pituitary gland: A quantitative ultrastructural approach to hormone storage in the neural lobe. J. Endocrinol. 1976, 68, 209–224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Antigen | Description | Source, Host Species, Cat #, or Code # | Working Dilution | Reference # | RRID |
|---|---|---|---|---|---|
| Copeptin | Synthetic peptide mapping at the amino acids 7–14 of human/mouse copeptin | Generated by our laboratory, rabbit polyclonal, CP8 | 1:10,000 (IF) 1:100 (EM) | [14,22,23] | AB_2722604 |
| CRF | Human/rat CRF coupled to human alpha-globulins via bis-diazotized benzidine | Donated by Dr. W. Vale, rabbit polyclonal, PBL rC70 | 1:20,000 (IF) 1:5000 (EM) | [14,24,25] | AB_2314234 |
| NPII | Soluble proteins extracted from the posterior pituitary of the rat | ATCC, mouse monoclonal, PS41, CRL-1799 | 1:1000 (IF) 1:200 (EM) 1:1000 (WB) | [14,26,27,28] | AB_2313960 |
| VGLUT2 | Recombinant protein corresponding to amino acids 510–582 of the rat VGLUT2 | Synaptic Systems, guinea pig polyclonal, 135 404 | 1:20 (EM) 1:10,000 (WB) | [29,30] | AB_887884 |
| l-Glutamate | l-Glutamate conjugated to glutaraldehyde | Abcam, rabbit polyclonal, ab9440 | 1:10 (EM) | [31,32] | AB_307256 |
| Region | Major Axis (nm) | Magno or Parvo | Number of Vesicles Examined | Identification |
|---|---|---|---|---|
| Posterior pituitary | 196 ± 2 | Magno | 205 | AVP |
| Internal ME | 197 ± 2 | Magno | 237 | AVP without CRF |
| External ME | 93 ± 1 * | Parvo | 212 | CRF |
| Cell Body | Major Axis (nm) | Magno or Parvo | Number of Vesicles Examined | SON or PVN | Identification |
|---|---|---|---|---|---|
| #1 | 187 ± 5 * | Magno | 29 | SON | AVP |
| #2 | 175 ± 6 * | Magno | 35 | SON | AVP |
| #3 | 189 ± 10 * | Magno | 24 | SON | AVP |
| #4 | 181 ± 5 * | Magno | 26 | SON | AVP |
| #5 | 171 ± 7 * | Magno | 25 | SON | AVP |
| #6 | 195 ± 6 * | Magno | 30 | SON | AVP |
| #7 | 205 ± 6 * | Magno | 46 | SON | AVP |
| #8 | 188 ± 5 * | Magno | 49 | PVN | AVP |
| #9 | 170 ± 5 * | Magno | 20 | PVN | AVP |
| #10 | 169 ± 8 * | Magno | 22 | PVN | AVP |
| #11 | 172 ± 4 * | Magno | 24 | PVN | AVP |
| #12 | 184 ± 5 * | Magno | 25 | PVN | AVP |
| #13 | 182 ± 5 * | Magno | 21 | PVN | AVP |
| #14 | 103 ± 5 † | Parvo | 38 | PVN | AVP |
| #15 | 98 ± 4 † | Parvo | 42 | PVN | CRF |
| #16 | 109 ± 4 † | Parvo | 51 | PVN | CRF |
| #17 | 93 ± 5 † | Parvo | 24 | PVN | CRF |
| #18 | 107 ± 5 † | Parvo | 40 | PVN | CRF |
| #19 | 103 ± 4 † | Parvo | 35 | PVN | CRF |
| #20 | 110 ± 7 † | Parvo | 42 | PVN | CRF |
| #21 | 108 ± 5 † | Parvo | 77 | PVN | CRF |
| #22 | 93 ± 4 † | Parvo | 81 | PVN | CRF |
| #23 | 95 ± 5 † | Parvo | 30 | PVN | CRF |
| #24 | 120 ± 6 *,† | (Parvo) | 40 | PVN | CRF |
| #25 | 113 ± 3 *,† | (Parvo) | 89 | PVN | CRF |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Otubo, A.; Maejima, S.; Oti, T.; Satoh, K.; Ueda, Y.; Morris, J.F.; Sakamoto, T.; Sakamoto, H. Immunoelectron Microscopic Characterization of Vasopressin-Producing Neurons in the Hypothalamo-Pituitary Axis of Non-Human Primates by Use of Formaldehyde-Fixed Tissues Stored at −25 °C for Several Years. Int. J. Mol. Sci. 2021, 22, 9180. https://doi.org/10.3390/ijms22179180
Otubo A, Maejima S, Oti T, Satoh K, Ueda Y, Morris JF, Sakamoto T, Sakamoto H. Immunoelectron Microscopic Characterization of Vasopressin-Producing Neurons in the Hypothalamo-Pituitary Axis of Non-Human Primates by Use of Formaldehyde-Fixed Tissues Stored at −25 °C for Several Years. International Journal of Molecular Sciences. 2021; 22(17):9180. https://doi.org/10.3390/ijms22179180
Chicago/Turabian StyleOtubo, Akito, Sho Maejima, Takumi Oti, Keita Satoh, Yasumasa Ueda, John F. Morris, Tatsuya Sakamoto, and Hirotaka Sakamoto. 2021. "Immunoelectron Microscopic Characterization of Vasopressin-Producing Neurons in the Hypothalamo-Pituitary Axis of Non-Human Primates by Use of Formaldehyde-Fixed Tissues Stored at −25 °C for Several Years" International Journal of Molecular Sciences 22, no. 17: 9180. https://doi.org/10.3390/ijms22179180
APA StyleOtubo, A., Maejima, S., Oti, T., Satoh, K., Ueda, Y., Morris, J. F., Sakamoto, T., & Sakamoto, H. (2021). Immunoelectron Microscopic Characterization of Vasopressin-Producing Neurons in the Hypothalamo-Pituitary Axis of Non-Human Primates by Use of Formaldehyde-Fixed Tissues Stored at −25 °C for Several Years. International Journal of Molecular Sciences, 22(17), 9180. https://doi.org/10.3390/ijms22179180






