Effect of Evening Primrose (Oenothera biennis) Oil Cake on the Properties of Polyurethane/Polyisocyanurate Bio-Composites
Abstract
:1. Introduction
2. Results and Discussion
2.1. Foaming Process
2.2. Cell Structure and Thermal Insulation Properties of RPU/PIR Foams
2.3. Physico-Mechanical and Aging Properties of RPU/PIR Foams
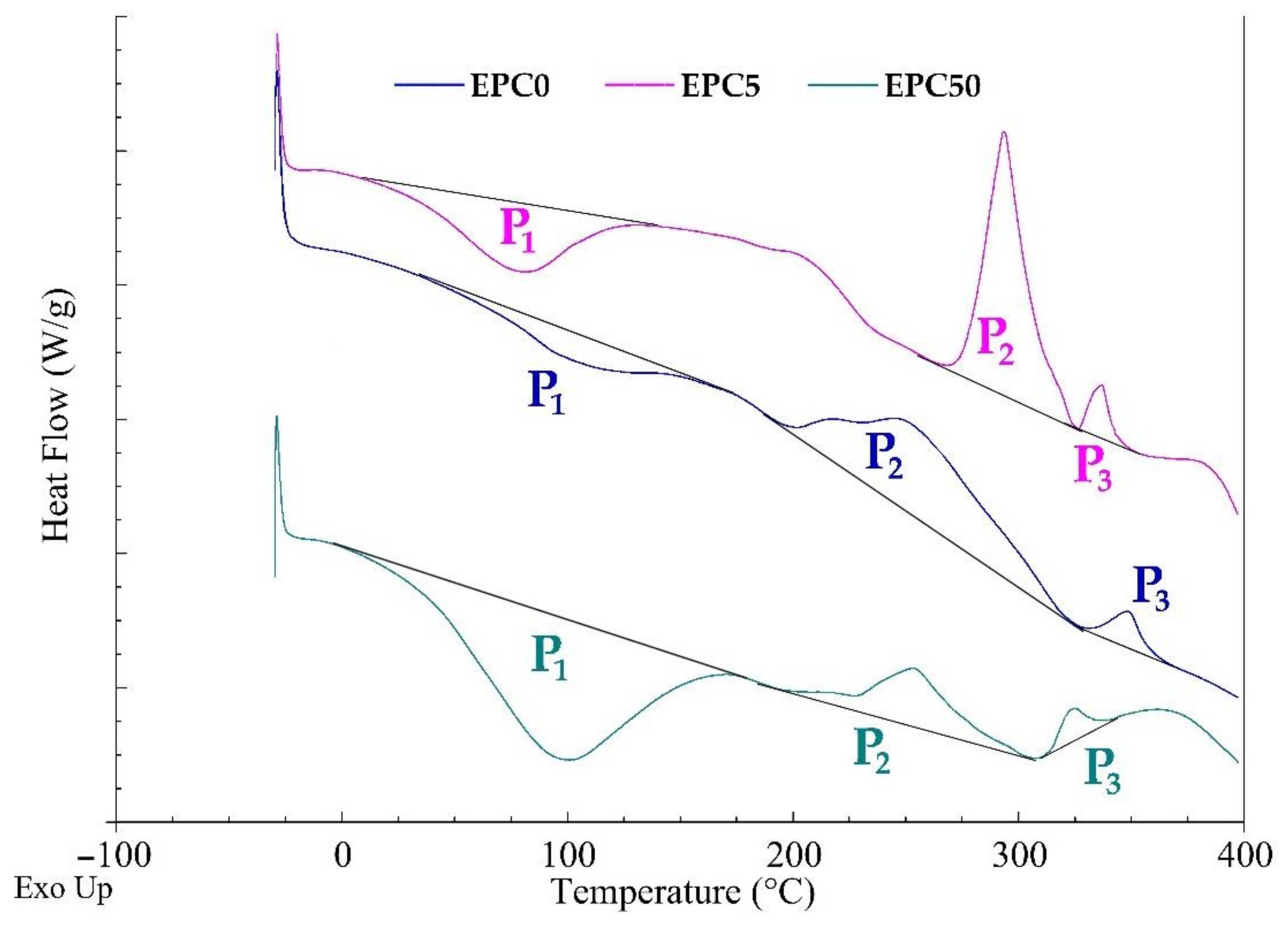
2.4. Flammability and Thermal Resistance of RPU/PIR Foams
3. Materials and Methods
3.1. Raw Materials
3.2. Preparation of Rigid Polyurethane/Polyisocyanurate Foams
3.3. Test Methods
- —
- TTI—time to ignition, (s);
- —
- THR—total heat release from the surface unit of the analyzed material, (MJ/m2);
- —
- HLR—mass loss rate of sample/burning rate, (g/m2·s);
- —
- HRR—heat release rate from the sample during combustion, (kW/m2);
- —
- — tHRRmax—time to reach the maximum value of HRR (HRRmax), (s);
- —
- — CO, CO2—emission of CO and CO2, respectively (kg/kg).
4. Conclusions
5. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wagner, W.L.; Hoch, P.C.; Raven, P.H. Revised classification of the Onagraceae. In Systematic Botany Monographs; Anderson, C.H., Ed.; The American Society of Plant Taxonomists: Ann Arbor, MI, USA, 2007; Volume 83. [Google Scholar]
- Dahiya, S.S.; Kaur, R.; Sharma, S.K. Evaluation of in vitro anthelmintic activity of Oenothera rosea L’Hér. ex Aiton. stem and root. J. Nat. Prod. Plant Resour. 2012, 2, 534–539. [Google Scholar]
- Greiner, S.; Köhl, K. Growing evening primroses (Oenothera). Front. Plant Sci. 2014, 5, 38. [Google Scholar] [CrossRef]
- Thakur, U.; Dutt, B.; Sharma, S.S.; Sharma, K.R.; Thakur, N. Evening Primrose (Oenothera biennis L.): Morphology and Reproductive Biology. Int. J. Curr. Microbiol. Appl. Sci. 2019, 8, 10. [Google Scholar] [CrossRef]
- Rosiak, E. The Market of Oilseeds and Their Processing Products in the European Union. Probl. World Agric. 2019, 19, 151–161. (In Polish) [Google Scholar] [CrossRef] [Green Version]
- Şahin, S.; Elhussein, E.A.A. Valorization of a biomass: Phytochemicals in oilseed by products. Phytochem. Rev. 2018, 17, 657–668. [Google Scholar] [CrossRef]
- Statista 2018. Available online: https://www.statista.com/statistics/263933/production-of-vegetable-oils-worldwide-since-2000/ (accessed on 3 September 2020).
- Cieślikowski, B.; Juliszewski, T.; Łapczyńska-Kordon, B. Utilization of biofuel technology by products for energy purposes. Agric. Eng. 2006, 10, 51–57. [Google Scholar]
- Akindoyo, J.O.; Beg, M.; Ghazali, S.; Islam, M.R.; Jeyaratnam, N.; Yuvaraj, A.R. Polyurethane types, synthesis and applications—A review. RSC Adv. 2016, 6, 114453–114482. [Google Scholar] [CrossRef] [Green Version]
- Szycher, M. Szycher’s Handbook of Polyurethanes, 1st ed.; CRC Press: Boca Raton, FL, USA, 1999. [Google Scholar]
- Gama, N.V.; Ferreira, A.; Barros-Timmons, A. Polyurethane foams: Past, present, and future. Materials 2018, 11, 1841. [Google Scholar] [CrossRef] [Green Version]
- Gama, N.V.; Silva, R.; Costa, M.; Barros-Timmons, A.; Ferreira, A. Statistical evaluation of the effect of formulation on the properties of crude glycerol polyurethane foams. Polym. Test. 2016, 56, 200–206. [Google Scholar] [CrossRef]
- Ferreira, A.; Gama, N.V.; Soares, B.; Freire, C.S.R.; Barros-Timmons, A.; Brandão, I.; Silva, R.; Neto, C.P. Method for Production of Rigid Polyurethane Foams Using Unrefined Crude Glycerol. Patent Application 107,711, 12 June 2014. [Google Scholar]
- Maisonneuve, L.; Chollet, G.; Grau, E.; Cramail, H. Vegetable oils: A source of polyols for polyurethane materials. OCL Oilseeds Fats Crops Lipids 2016, 23, D508–D510. [Google Scholar] [CrossRef] [Green Version]
- Alagi, P.; Choi, Y.J.; Hong, S.C. Preparation of vegetable oil-based polyols with controlled hydroxyl functionalities for thermoplastic polyurethane. Eur. Polym. J. 2016, 78, 46–60. [Google Scholar] [CrossRef]
- Prociak, A.; Kurańska, M.; Cabulis, U.; Ryszkowska, J.; Leszczyńska, M.; Uram, K.; Kirpluks, M. Effect of bio-polyols with different chemical structures on foaming of polyurethane systems and foam properties. Ind. Crops Prod. 2018, 120, 262–270. [Google Scholar] [CrossRef]
- Paciorek-Sadowska, J.; Borowicz, M.; Czupryński, B.; Tomaszewska, E.; Liszkowska, J. Oenothera biennis seed oil as an alternative raw material for production of bio-polyol for rigid polyurethane-polyisocyanurate foams. Ind. Crops Prod. 2018, 126, 208–217. [Google Scholar] [CrossRef]
- Ghasemlou, M.; Daver, F.; Ivanova, E.P.; Adhikari, B. Polyurethanes from seed oil-based polyols: A review of synthesis, mechanical and thermal properties. Ind. Crops Prod. 2019, 142, 111841. [Google Scholar] [CrossRef]
- Członka, S.; Bertino, M.F.; Strzelec, K. Rigid polyurethane foams reinforced with industrial potato protein. Polym. Test. 2018, 68, 135–145. [Google Scholar] [CrossRef]
- Kurańska, M.; Prociak, A.; Michałowski, S.; Cabulis, U.; Kirpluks, M. Microcellulose as a natural filler in polyurethane foams based on the biopolyol from rapeseed oil. Polimery 2016, 61, 625–632. [Google Scholar] [CrossRef]
- Aranguren, M.I.; González, J.F.; Mosiewicki, M.A. Biodegradation of a vegetable oil based polyurethane and wood flour composites. Polym. Test. 2012, 31, 7–15. [Google Scholar] [CrossRef]
- Sienkiewicz, N.; Członka, S.; Kairyte, A.; Vaitkus, S. Curcumin as a natural compound in the synthesis of rigid polyurethane foams with enhanced mechanical, antibacterial and anti-ageing properties. Polym. Test. 2019, 79, 106046. [Google Scholar] [CrossRef]
- Członka, S.; Sienkiewicz, N.; Strąkowska, A.; Strzelec, K. Keratin feathers as a filler for rigid polyurethane foams on the basis of soybean oil polyol. Polym. Test. 2018, 72, 32–45. [Google Scholar] [CrossRef]
- Członka, S.; Strąkowska, A.; Kairytė, A. Effect of walnut shells and silanized walnut shells on the mechanical and thermal properties of rigid polyurethane foams. Polym. Test. 2020, 87, 106534. [Google Scholar] [CrossRef]
- Bryśkiewicz, A.; Zieleniewska, M.; Przyjemska, K.; Chojnacki, P.; Ryszkowska, J. Modification of flexible polyurethane foams by the addition of natural origin fillers. Polym. Degrad. Stab. 2016, 132, 32–40. [Google Scholar] [CrossRef]
- Widya, T.; Macosko, C.W. Nanoclay-modified rigid polyurethane foam. J. Macromol. Sci. Phys. 2005, 44, 897–908. [Google Scholar] [CrossRef]
- Gu, R.; Sain, M.M.; Konar, S.K. A feasibility study of polyurethane composite foam with added hardwood pulp. Ind. Crops Prod. 2013, 42, 273–279. [Google Scholar] [CrossRef]
- Prociak, A.; Kurańska, M.; Malewska, E.; Szczepkowski, L.; Zieleniewska, M.; Ryszkowska, J.; Ficoń, J.; Rząsa, A. Biobased polyurethane foams modified with natural fillers. Polimery 2015, 60, 592–599. [Google Scholar] [CrossRef]
- Kuźnia, M.; Magiera, A.; Pielichowska, K.; Ziąbka, M.; Benko, A.; Szatkowski, P.; Jerzak, W. Fluidized bed combustion fly ash as filler in composite polyurethane materials. J. Waste Manag. 2019, 92, 115–123. [Google Scholar] [CrossRef]
- Paciorek-Sadowska, J.; Borowicz, M.; Isbrandt, M.; Czupryński, B.; Apiecionek, Ł. The use of waste from the production of rapeseed oil for obtaining of new polyurethane composites. Polymers 2019, 11, 1431. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cao, X.; Lee, L.J.; Widya, T.; Macosko, C. Polyurethane/clay nanocomposites foams: Processing, structure and properties. Polymer 2005, 46, 775–783. [Google Scholar] [CrossRef]
- Fan, H.; Tekeei, A.; Suppes, G.J.; Hsieh, F.H. Properties of biobased rigid polyurethane foams reinforced with fillers: Microspheres and nanoclay. Int. J. Polym. Sci. 2012, 2012, 474803. [Google Scholar] [CrossRef] [Green Version]
- Onuegbu, T.U.; Ugwu, L.E.; Ogunfeyitimi, O. Effect of fillers on the density, rising time, creaming time, ignition time, flame duration and thermal conductivity of flexible polyether foam. Int. J. Sci. Eng. Res. 2013, 4, 1691. [Google Scholar]
- Prociak, A. Cell structure and thermal conductivity of rigid polyurethane foams blown with cyclopentane in different moulds. In Proceedings of the 7th International Conference Blowing Agents and Foaming Process 2005, Stuttgart, Germany, 10–11 May 2005. [Google Scholar]
- Faruk, O.; Bledzki, A.K.; Fink, H.P.; Sain, M. Biocomposites reinforced with natural fibers: 2000–2010. Prog. Polym. Sci. 2012, 37, 1552–1596. [Google Scholar] [CrossRef]
- Luo, X.; Mohanty, A.; Misra, M. Lignin as a reactive reinforcing filler for water-blown rigid biofoam composites from soy oil-based polyurethane. Ind. Crops Prod. 2013, 47, 13–19. [Google Scholar] [CrossRef]
- Thirumal, M.; Khastgir, D.; Singha, N.K.; Manjunath, B.S.; Naik, Y.P. Effect of foam density on the properties of water blown rigid polyurethane foam. J. Appl. Polym. Sci. 2008, 108, 1810–1817. [Google Scholar] [CrossRef]
- Fan, H.; Tekeei, A.; Suppes, G.J.; Hsieh, F.H. Rigid polyurethane foams made from high viscosity soy-polyols. J. Appl. Polym. Sci. 2013, 127, 1623–1629. [Google Scholar] [CrossRef]
- Wu, J.W.; Sung, W.F.; Chu, H.S. Thermal conductivity of polyurethane foams. Int. J. Heat Mass Transf. 1999, 42, 2211–2217. [Google Scholar] [CrossRef]
- Galakhova, A.; Santiago-Calvo, M.; Tirado-Mediavilla, J.; Villafañe, F.; Rodríguez-Pérez, M.Á.; Riess, G. Identification and quantification of cell gas evolution in rigid polyurethane foams by novel GCMS methodology. Polymers 2019, 11, 1192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Engels, H.W.; Pirkl, H.G.; Albers, R.; Albach, R.W.; Krause, J.; Hoffmann, A.; Casselmann, H.; Dormish, J. Polyurethanes: Versatile materials and sustainable problem solving for today’s challenges. Angew. Chem. Int. Ed. 2013, 52, 9422–9441. [Google Scholar] [CrossRef] [PubMed]
- Ciecierska, E.; Jurczyk-Kowalska, M.; Bazarnik, P.; Gloc, M.; Kulesza, M.; Kowalski, M.; Krauze, S.; Lewandowska, M. Flammability, mechanical properties and structure of rigid polyurethane foams with different types of carbon reinforcing materials. Compos. Struct. 2016, 140, 67–76. [Google Scholar] [CrossRef]
- Chattopadhyay, D.K.; Webster, D.C. Thermal stability and flame retardancy of polyurethanes. Prog. Polym. Sci. 2009, 34, 1068–1133. [Google Scholar] [CrossRef]
- Badri, K.H.; Ahmad, S.H.; Zakaria, S. Production of a high-functionality RBD palm kernel oil-based polyester polyol. J. Appl. Polym. Sci. 2001, 81, 384–389. [Google Scholar] [CrossRef]
- Kallaos, J.; Bohne, R.A.; Hovde, P.J. Long-term performance of rigid plastic foam building insulation. J. Mater. Civ. Eng. 2014, 26, 374–378. [Google Scholar] [CrossRef]
- Alarie, Y. Toxicity of fire smoke. Crit. Rev. Toxicol. 2002, 32, 259–289. [Google Scholar] [CrossRef]
- Liu, Y.; He, J.; Yang, R. Effects of dimethyl methylphosphonate, aluminum hydroxide, ammonium polyphosphate, and expandable graphite on the flame retardancy and thermal properties of polyisocyanurate–polyurethane foams. Ind. Eng. Chem. Res. 2015, 54, 5876–5884. [Google Scholar] [CrossRef]
- Velencoso, M.M.; Ramos, M.J.; Klein, R.; De Lucas, A.; Rodriguez, J.F. Thermal degradation and fire behaviour of novel polyurethanes based on phosphate polyols. Polym. Degrad. Stab. 2014, 101, 40–51. [Google Scholar] [CrossRef]
- Chen, X.; Li, J.; Gao, M. Thermal degradation and flame retardant mechanism of the rigid polyurethane foam including functionalized graphene oxide. Polymers 2019, 11, 78. [Google Scholar] [CrossRef] [Green Version]
- Paciorek-Sadowska, J.; Czupryński, B.; Borowicz, M.; Liszkowska, J. New polyurethane materials containing biofiller. Polimery 2015, 9, 586–591. [Google Scholar] [CrossRef]
- Liszkowska, J.; Borowicz, M.; Paciorek-Sadowska, J.; Isbrandt, M.; Czupryński, B.; Moraczewski, K. Assessment of Photodegradation and Biodegradation of RPU/PIR Foams Modified by Natural Compounds of Plant Origin. Polymers 2020, 12, 33. [Google Scholar] [CrossRef] [Green Version]
- Mizera, K.; Auguścik, M.; Woronka, D.; Lipert, K.; Kalarus, W.; Ryszkowska, J. Polyurethane composites with mixture of carbon fibers and glass frit. Polimery 2016, 61, 307–315. [Google Scholar] [CrossRef]
- Ghasemnezhad, A.; Honermeier, B. Yield, oil constituents, and protein content of evening primrose (Oenothera biennis L.) seeds depending on harvest time, harvest method and nitrogen application. Ind. Crops Prod. 2008, 28, 17–23. [Google Scholar] [CrossRef]
- Lorusso, C.; Vergaro, V.; Conciauro, F.; Ciccarella, G.; Congedo, P.M. Thermal and mechanical performance of rigid polyurethane foam added with commercial nanoparticles. Nanomater. Nanotechnol. 2017, 7, 1847980416684117. [Google Scholar] [CrossRef]
- Rao, R.R.; Mondy, L.A.; Long, K.N.; Celina, M.C.; Wyatt, N.; Roberts, C.C.; Soehnel, M.M.; Brunini, V.E. The kinetics of polyurethane structural foam formation: Foaming and polymerization. AIChE J. 2017, 63, 2945–2957. [Google Scholar] [CrossRef]
- Kurańska, M. Porous Polyurethane Materials from Renewable Feedstocks. Ph.D. Thesis, Cracow University of Technology, Cracow, Poland, 2014. (In Polish). [Google Scholar]
- ASTM Standard D7487–13e1. Standard Practice for Polyurethane Raw Materials: Polyurethane Foam Cup Test; ASTM International: West Conshohocken, PA, USA, 2016. [Google Scholar] [CrossRef]


| Sample | Cream Time (s) | String Gel Time (s) | Tack Free Time (s) | Free Rise Time (s) |
|---|---|---|---|---|
| EPC0 | 16 ± 1 | 32 ± 1 | 74 ± 1 | 66 ± 1 |
| EPC5 | 16 ± 1 | 32 ± 1 | 75 ± 1 | 67 ± 1 |
| EPC10 | 16 ± 1 | 32 ± 1 | 75 ± 1 | 68 ± 1 |
| EPC15 | 16 ± 1 | 34 ± 1 | 76 ± 1 | 69 ± 1 |
| EPC20 | 16 ± 1 | 34 ± 1 | 81 ± 1 | 75 ± 1 |
| EPC25 | 16 ± 1 | 34 ± 1 | 84 ± 1 | 78 ± 1 |
| EPC30 | 16 ± 1 | 35 ± 1 | 84 ± 1 | 78 ± 1 |
| EPC35 | 16 ± 1 | 36 ± 1 | 89 ± 1 | 78 ± 1 |
| EPC40 | 16 ± 1 | 36 ± 1 | 96 ± 1 | 80 ± 1 |
| EPC45 | 16 ± 1 | 36 ± 1 | 98 ± 1 | 83 ± 1 |
| EPC50 | 16 ± 1 | 38 ± 1 | 98 ± 1 | 83 ± 1 |
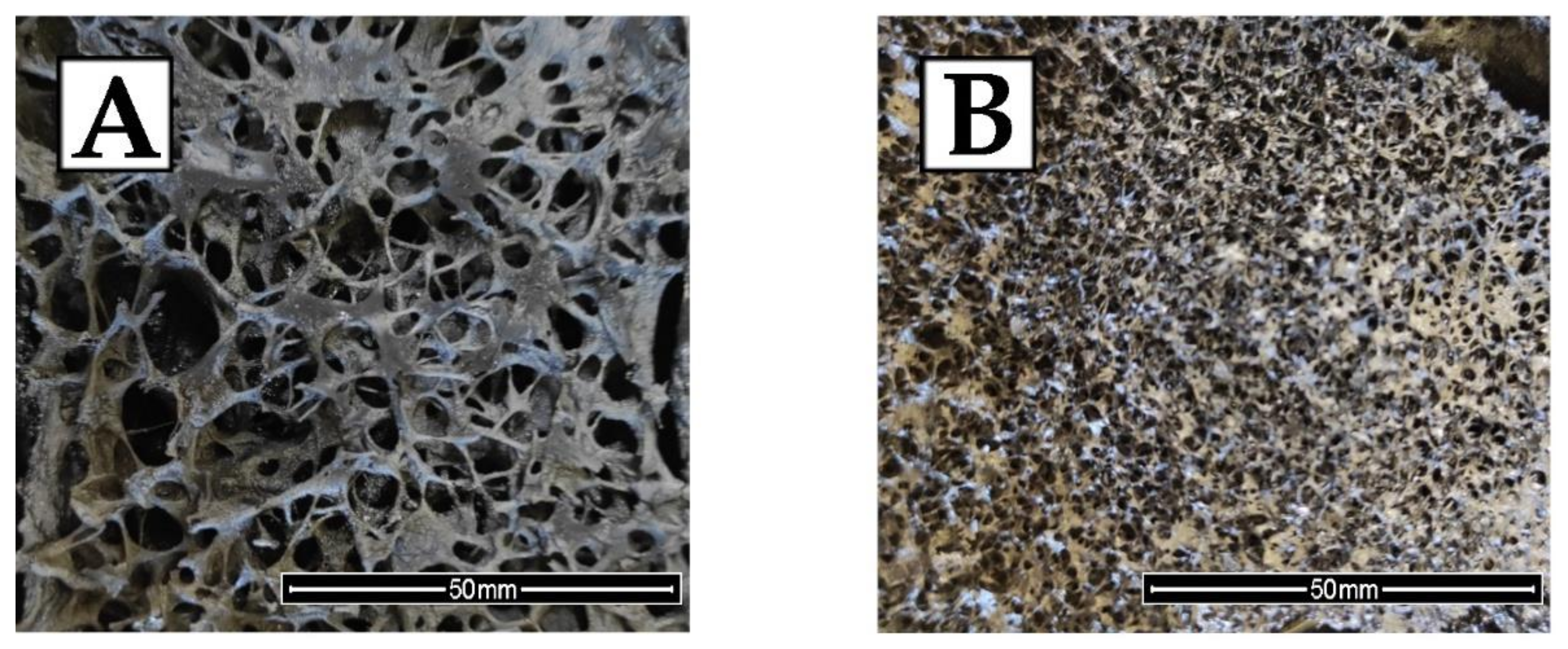
| Sample | Cell Size (μm) | Wall Thickness (μm) | Content of Cells per Area Unit (Cell/mm2) |
|---|---|---|---|
| EPC0 | 319 ± 15 | 17 ± 2 | 10 ± 1 |
| EPC10 | 260 ± 14 | 19 ± 2 | 11 ± 1 |
| EPC20 | 250 ± 14 | 22 ± 2 | 11 ± 1 |
| EPC30 | 240 ± 12 | 23 ± 2 | 12 ± 1 |
| EPC40 | 230 ± 12 | 23 ± 2 | 13 ± 1 |
| EPC50 | 204 ± 12 | 24 ± 2 | 13 ± 1 |
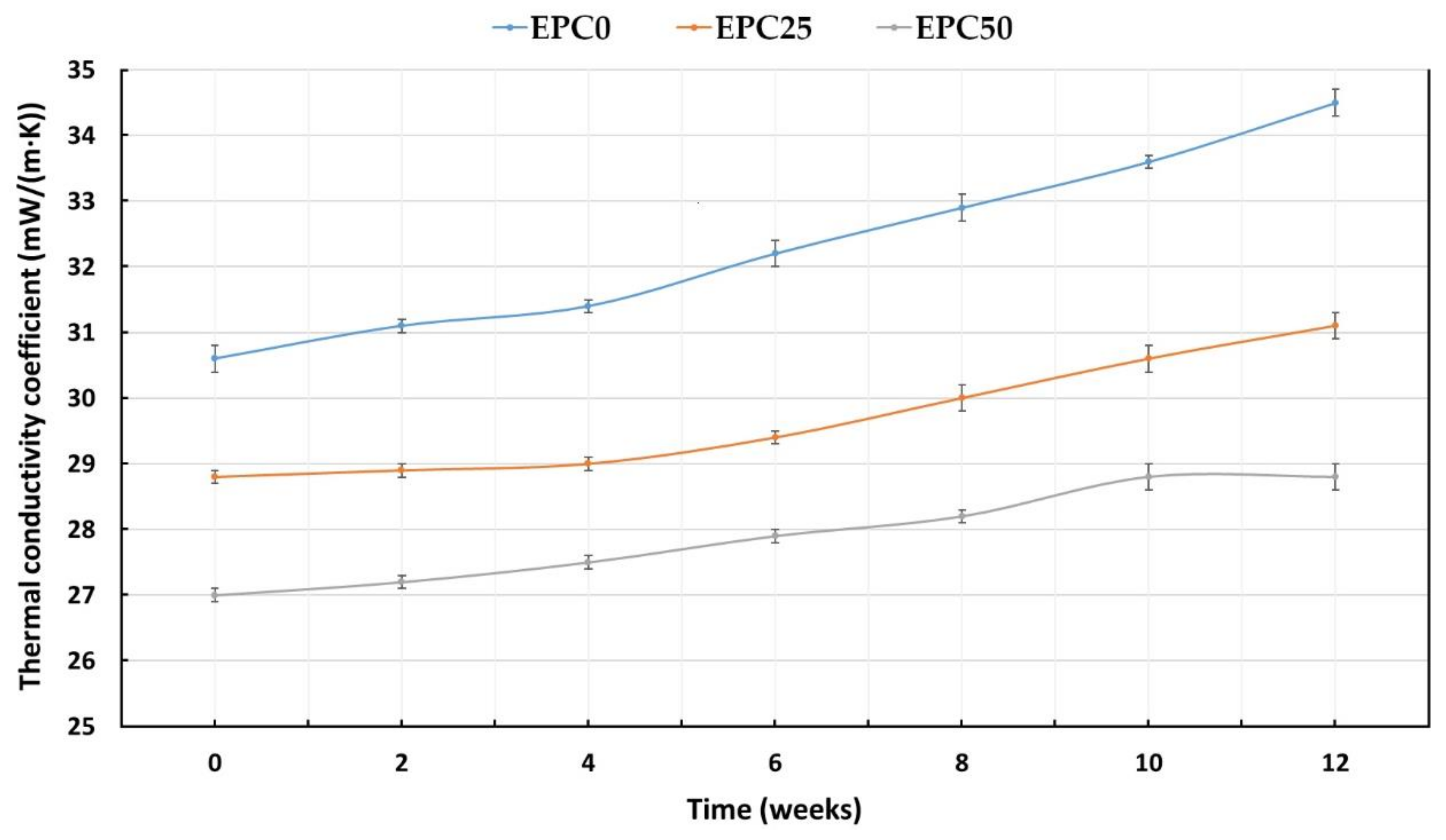
| Sample | Thermal Conductivity Coefficient λ (mW/(m·K)) | Closed Cells Content (%) | Absorbability (%) | Water Absorption (%) |
|---|---|---|---|---|
| EPC0 | 34.6 ± 0.1 | 80.9 ± 0.9 | 10.9 ± 0.2 | 5.5 ± 0.3 |
| EPC5 | 34.2 ± 0.1 | 81.1 ± 0.9 | 10.9 ± 0.2 | 5.5 ± 0.3 |
| EPC10 | 34.0 ± 0.1 | 83.0 ± 0.9 | 10.7 ± 0.3 | 5.1 ± 0.3 |
| EPC15 | 33.6 ± 0.1 | 84.7 ± 1.0 | 10.7 ± 0.3 | 5.0 ± 0.3 |
| EPC20 | 33.5 ± 0.1 | 88.6 ± 0.9 | 10.6 ± 0.5 | 4.8 ± 0.2 |
| EPC25 | 30.6 ± 0.1 | 90.1 ± 0.9 | 10.6 ± 0.2 | 4.6 ± 0.2 |
| EPC30 | 29.1 ± 0.1 | 90.6 ± 1.1 | 10.6 ± 0.6 | 4.2 ± 0.2 |
| EPC35 | 28.8 ± 0.1 | 90.8 ± 0.9 | 9.0 ± 0.5 | 3.5 ± 0.2 |
| EPC40 | 28.1 ± 0.1 | 90.8 ± 0.8 | 8.3 ± 0.5 | 2.2 ± 0.2 |
| EPC45 | 27.7 ± 0.1 | 91.7 ± 1.3 | 8.0 ± 0.8 | 2.0 ± 0.2 |
| EPC50 | 27.4 ± 0.2 | 93.1 ± 1.2 | 6.6 ± 0.2 | 1.8 ± 0.1 |
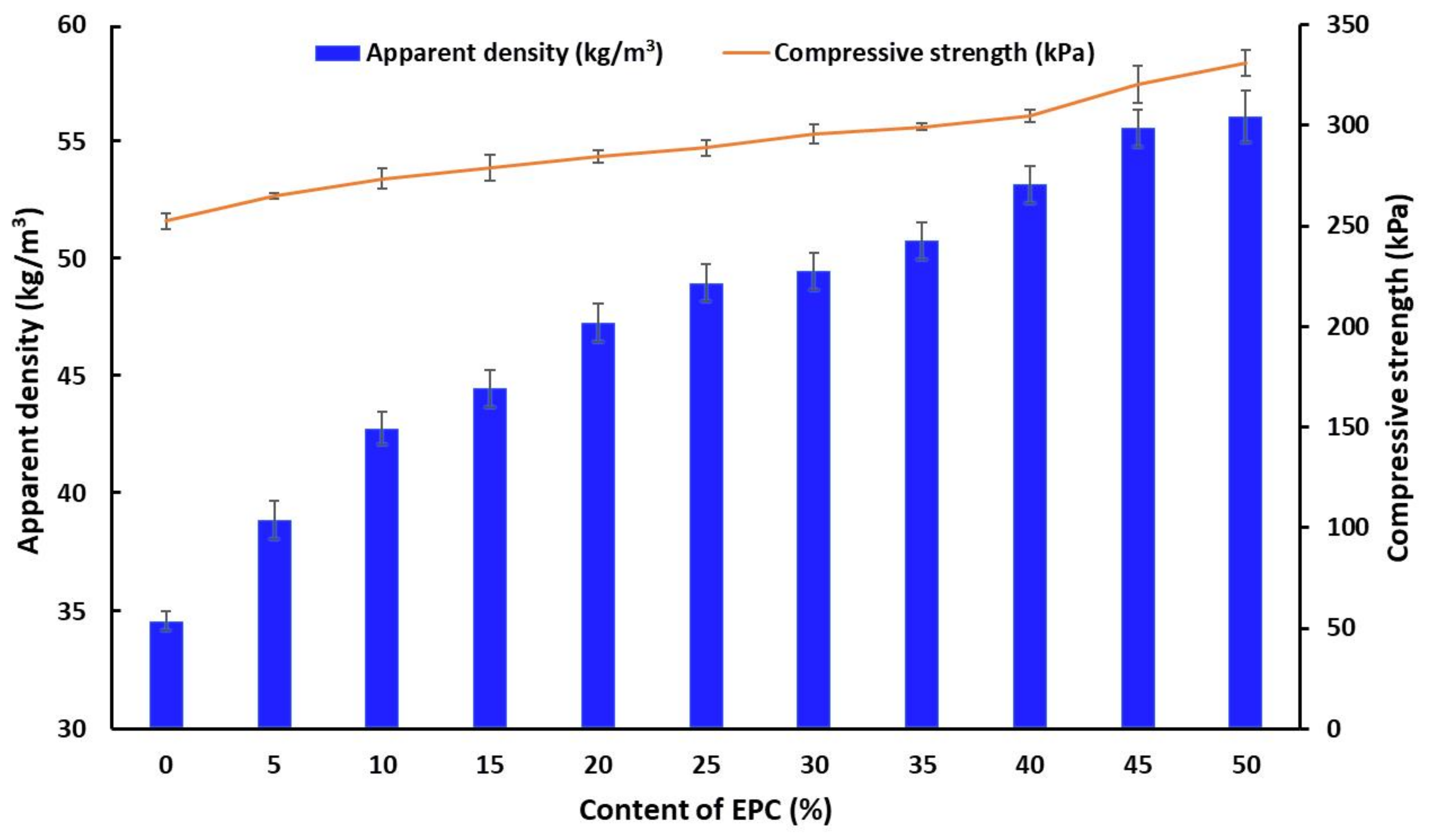
| Sample | Apparent Density (kg/m3) | Compressive Strength (kPa) | Brittleness (%) | Δm (%) | Δl (%) | ΔV (%) |
|---|---|---|---|---|---|---|
| EPC0 | 34.6 ± 0.4 | 252.5 ± 3.9 | 23.8 ± 0.9 | 4.0 ± 0.4 | 5.0 ± 0.4 | 3.0 ± 0.4 |
| EPC5 | 38.9 ± 0.8 | 264.9 ± 1.4 | 23.1 ± 0.8 | 3.0 ± 0.4 | 4.3 ± 0.4 | 2.8 ± 0.4 |
| EPC10 | 42.8 ± 0.7 | 273.5 ± 5.2 | 22.0 ± 0.7 | 2.6 ± 0.4 | 4.0 ± 0.4 | 2.4 ± 0.4 |
| EPC15 | 44.5 ± 0.8 | 278.9 ± 6.6 | 21.4 ± 0.6 | 2.6 ± 0.4 | 3.6 ± 0.4 | 1.9 ± 0.4 |
| EPC20 | 47.3 ± 0.8 | 284.3 ± 3.1 | 20.9 ± 0.6 | 2.1 ± 0.4 | 3.3 ± 0.4 | 1.5 ± 0.4 |
| EPC25 | 49.0 ± 0.8 | 288.9 ± 3.9 | 19.7 ± 0.6 | 1.4 ± 0.4 | 3.1 ± 0.4 | 1.0 ± 0.4 |
| EPC30 | 49.5 ± 0.8 | 295.9 ± 4.8 | 19.1 ± 0.5 | 1.4 ± 0.4 | 3.1 ± 0.4 | 1.0 ± 0.4 |
| EPC35 | 50.8 ± 0.8 | 299.2 ± 1.6 | 17.0 ± 0.5 | 1.2 ± 0.4 | 3.1 ± 0.4 | 0.9 ± 0.4 |
| EPC40 | 53.2 ± 0.8 | 304.7 ± 2.9 | 14.0 ± 0.4 | 1.0 ± 0.4 | 2.9 ± 0.4 | 0.7 ± 0.4 |
| EPC45 | 55.6 ± 0.8 | 320.4 ± 9.3 | 14.8 ± 0.4 | 1.0 ± 0.4 | 2.9 ± 0.4 | 0.6 ± 0.4 |
| EPC50 | 56.1 ± 1.1 | 331.1 ± 6.3 | 13.1 ± 0.4 | 1.0 ± 0.4 | 2.8 ± 0.4 | 0.6 ± 0.4 |
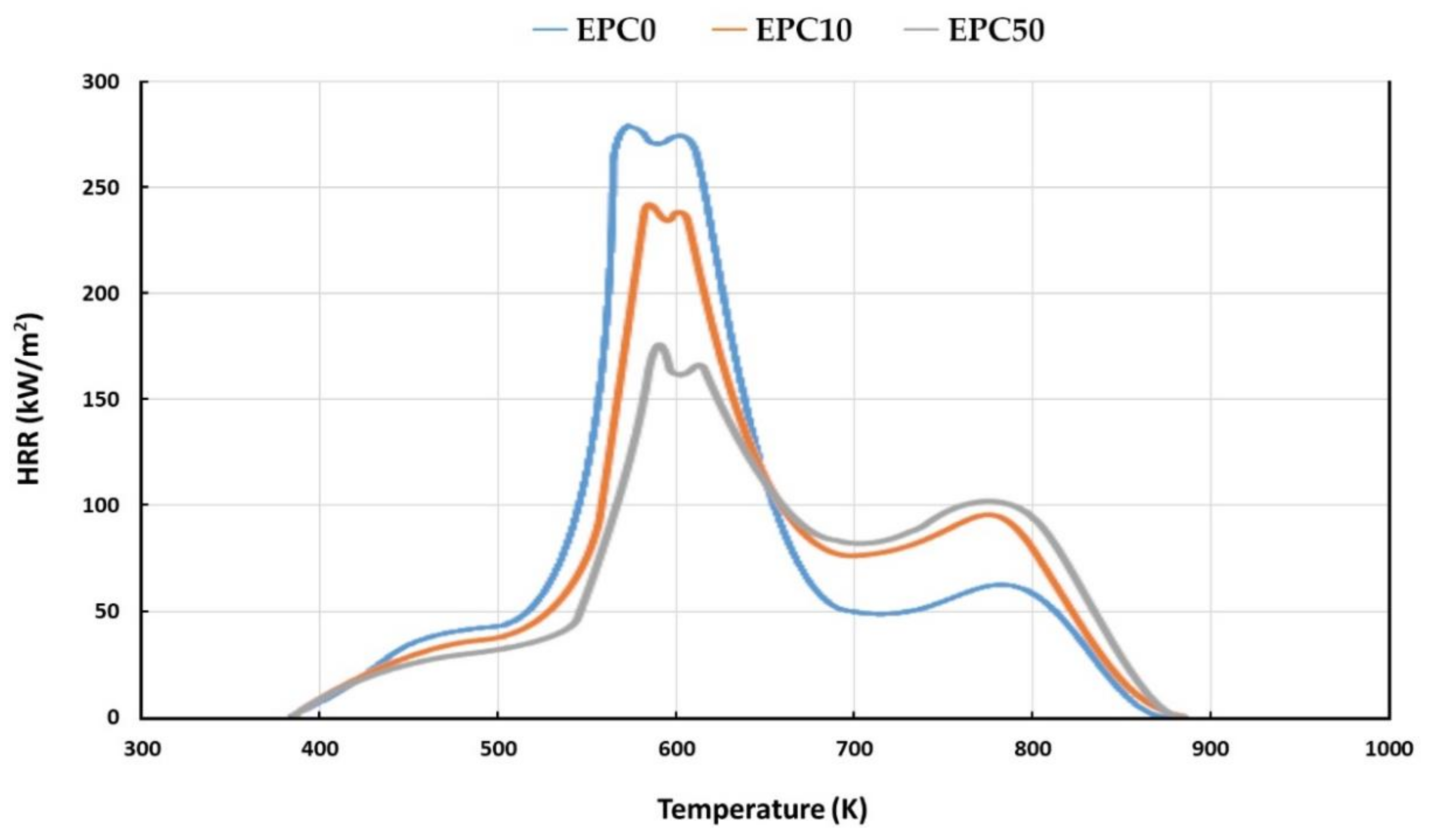
| Sample | TTI (s) | THR (MJ/m2) | HLR (g/m2·s) | HRR (kW/m2) | tHRRmax (s) | Amount of Released CO (kg/kg) | Amount of Released CO2 (kg/kg) | CO/CO2 (-) | LOI (vol.% of O2) |
|---|---|---|---|---|---|---|---|---|---|
| EPC0 | 1.48 | 14.3 | 10.22 | 278.90 | 10 | 0.82 | 6.2 | 0.132 | 19.1 |
| EPC10 | 1.53 | 7.7 | 7.03 | 241.21 | 17 | 0.18 | 1.93 | 0.093 | 19.9 |
| EPC20 | 1.85 | 6.8 | 6.87 | 227.77 | 19 | 0.11 | 1.61 | 0.068 | 20.4 |
| EPC30 | 1.96 | 6.0 | 6.02 | 201.99 | 23 | 0.09 | 1.57 | 0.057 | 20.8 |
| EPC40 | 2.24 | 5.9 | 5.35 | 189.87 | 26 | 0.07 | 1.45 | 0.048 | 21.7 |
| EPC50 | 2.61 | 5.6 | 4.87 | 175.29 | 29 | 0.05 | 1.38 | 0.036 | 22.5 |
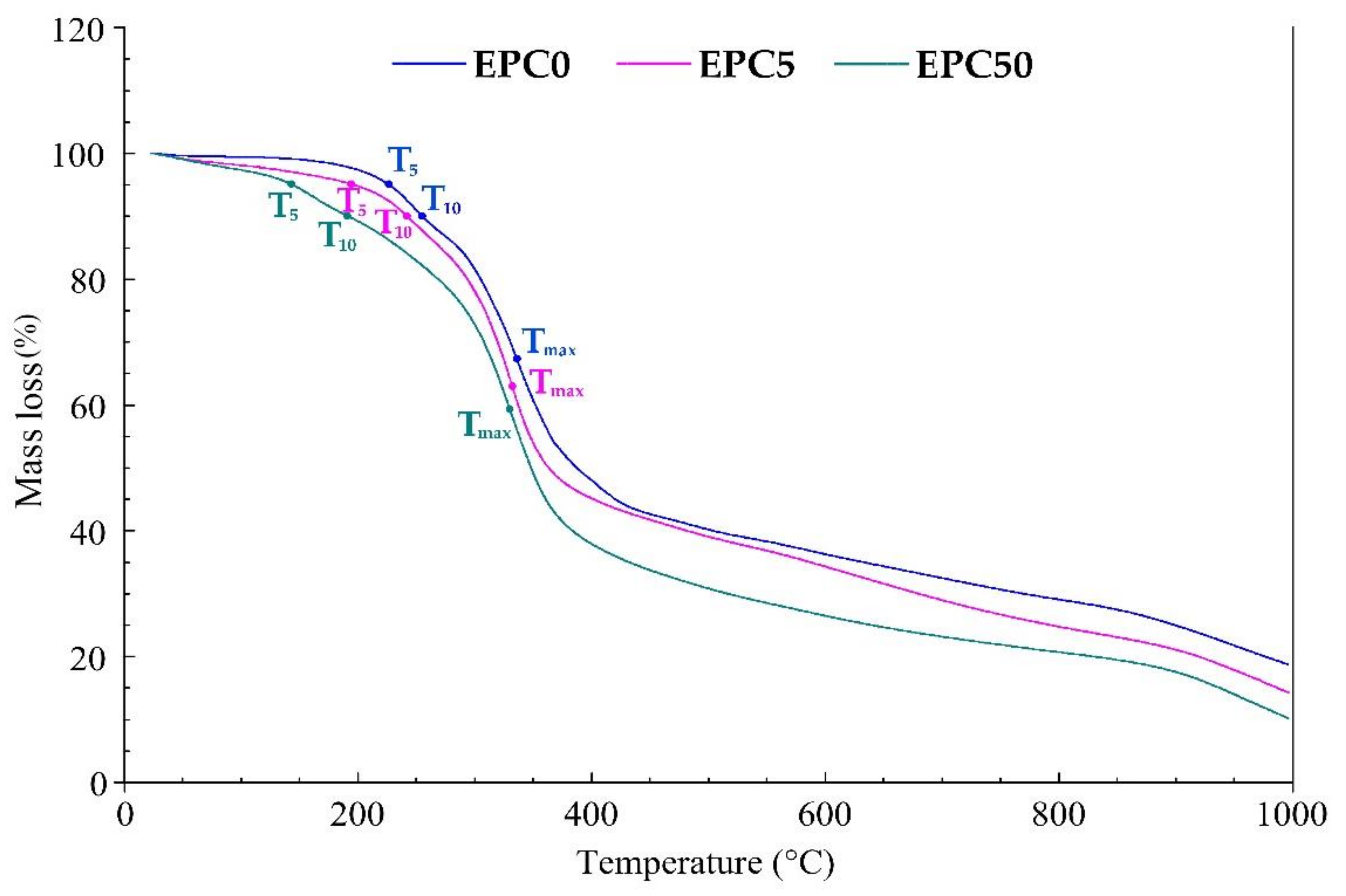
| Sample | T5 (°C) | T10 (°C) | Tmax (°C) | The Highest Mass Loss (%/°C) | The Highest Mass Loss Rate (%/min) | Residue (%) |
|---|---|---|---|---|---|---|
| EPC0 | 222 | 254 | 342 | 0.47 | 4.7 | 21 |
| EPC5 | 196 | 243 | 337 | 0.47 | 4.7 | 16 |
| EPC50 | 145 | 191 | 332 | 0.59 | 5.9 | 12 |
| Components | Dry Weight (%) | Proteins (%) | Minerals (g) | Vitamins (mg) | Fat (%) | Crude Fiber (%) | Tanins (µM/g) |
|---|---|---|---|---|---|---|---|
| Amount | 98.50 | 38.41 * | 52.06 ** | 208.40 *** | 6.40 | 9.20 | 14–17 |
| Sample | Rokopol RF-551 (g) | EPC (g) | Tegostab 8460 (g) | 33% DABCO (g) | 33% Potassium Acetate (g) | Antiblaze TCMP (g) | Distilled Water (g) | Purocyn B (g) |
|---|---|---|---|---|---|---|---|---|
| EPC0 | 66.80 | 0.00 | 5.40 | 3.17 | 7.96 | 53.90 | 3.15 | 250.60 |
| EPC5 | 66.80 | 15.90 | 5.40 | 3.17 | 7.96 | 53.90 | 3.15 | 250.60 |
| EPC10 | 66.80 | 31.80 | 5.40 | 3.17 | 7.96 | 53.90 | 3.15 | 250.60 |
| EPC15 | 66.80 | 47.60 | 5.40 | 3.17 | 7.96 | 53.90 | 3.15 | 250.60 |
| EPC20 | 66.80 | 63.50 | 5.40 | 3.17 | 7.96 | 53.90 | 3.15 | 250.60 |
| EPC25 | 66.80 | 79.30 | 5.40 | 3.17 | 7.96 | 53.90 | 3.15 | 250.60 |
| EPC30 | 66.80 | 95.20 | 5.40 | 3.17 | 7.96 | 53.90 | 3.15 | 250.60 |
| EPC35 | 66.80 | 111.00 | 5.40 | 3.17 | 7.96 | 53.90 | 3.15 | 250.60 |
| EPC40 | 66.80 | 126.90 | 5.40 | 3.17 | 7.96 | 53.90 | 3.15 | 250.60 |
| EPC45 | 66.80 | 142.80 | 5.40 | 3.17 | 7.96 | 53.90 | 3.15 | 250.60 |
| EPC50 | 66.80 | 158.70 | 5.40 | 3.17 | 7.96 | 53.90 | 3.15 | 250.60 |
| Parameter | Viscosity (mPa s) | ||
|---|---|---|---|
| 0.5 rpm | 10 rpm | 50 rpm | |
| EPC0 | 580 | 310 | 270 |
| EPC5 | 620 | 330 | 290 |
| EPC10 | 710 | 360 | 310 |
| EPC15 | 790 | 380 | 345 |
| EPC20 | 820 | 410 | 370 |
| EPC25 | 880 | 440 | 390 |
| EPC30 | 910 | 460 | 405 |
| EPC35 | 940 | 490 | 420 |
| EPC40 | 980 | 515 | 480 |
| EPC45 | 1130 | 535 | 505 |
| EPC50 | 1300 | 550 | 530 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paciorek-Sadowska, J.; Borowicz, M.; Isbrandt, M. Effect of Evening Primrose (Oenothera biennis) Oil Cake on the Properties of Polyurethane/Polyisocyanurate Bio-Composites. Int. J. Mol. Sci. 2021, 22, 8950. https://doi.org/10.3390/ijms22168950
Paciorek-Sadowska J, Borowicz M, Isbrandt M. Effect of Evening Primrose (Oenothera biennis) Oil Cake on the Properties of Polyurethane/Polyisocyanurate Bio-Composites. International Journal of Molecular Sciences. 2021; 22(16):8950. https://doi.org/10.3390/ijms22168950
Chicago/Turabian StylePaciorek-Sadowska, Joanna, Marcin Borowicz, and Marek Isbrandt. 2021. "Effect of Evening Primrose (Oenothera biennis) Oil Cake on the Properties of Polyurethane/Polyisocyanurate Bio-Composites" International Journal of Molecular Sciences 22, no. 16: 8950. https://doi.org/10.3390/ijms22168950
APA StylePaciorek-Sadowska, J., Borowicz, M., & Isbrandt, M. (2021). Effect of Evening Primrose (Oenothera biennis) Oil Cake on the Properties of Polyurethane/Polyisocyanurate Bio-Composites. International Journal of Molecular Sciences, 22(16), 8950. https://doi.org/10.3390/ijms22168950








