Sequential Release of Paclitaxel and Imatinib from Core–Shell Microparticles Prepared by Coaxial Electrospray for Vaginal Therapy of Cervical Cancer
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preparation and Characterization of Microparticles
2.2. In Vitro Release of PTX and IMN
2.3. Degradation of PHIPMPs
2.4. Cytotoxicity Assay
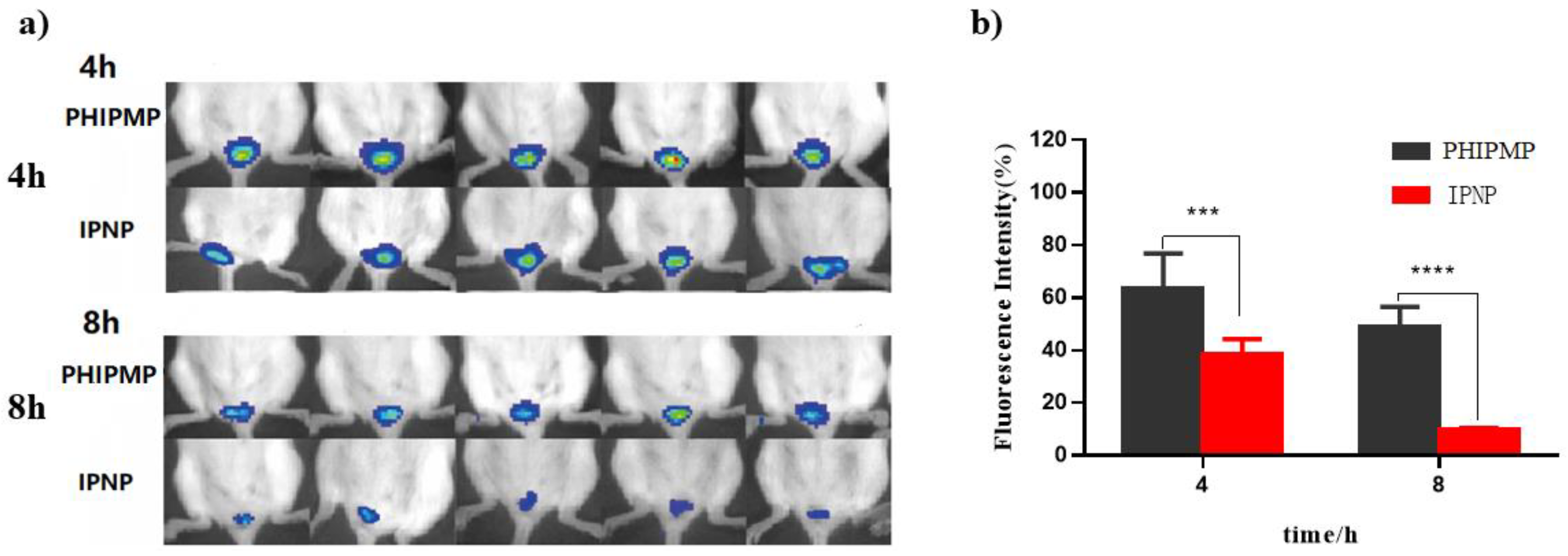
2.5. Vaginal Retention
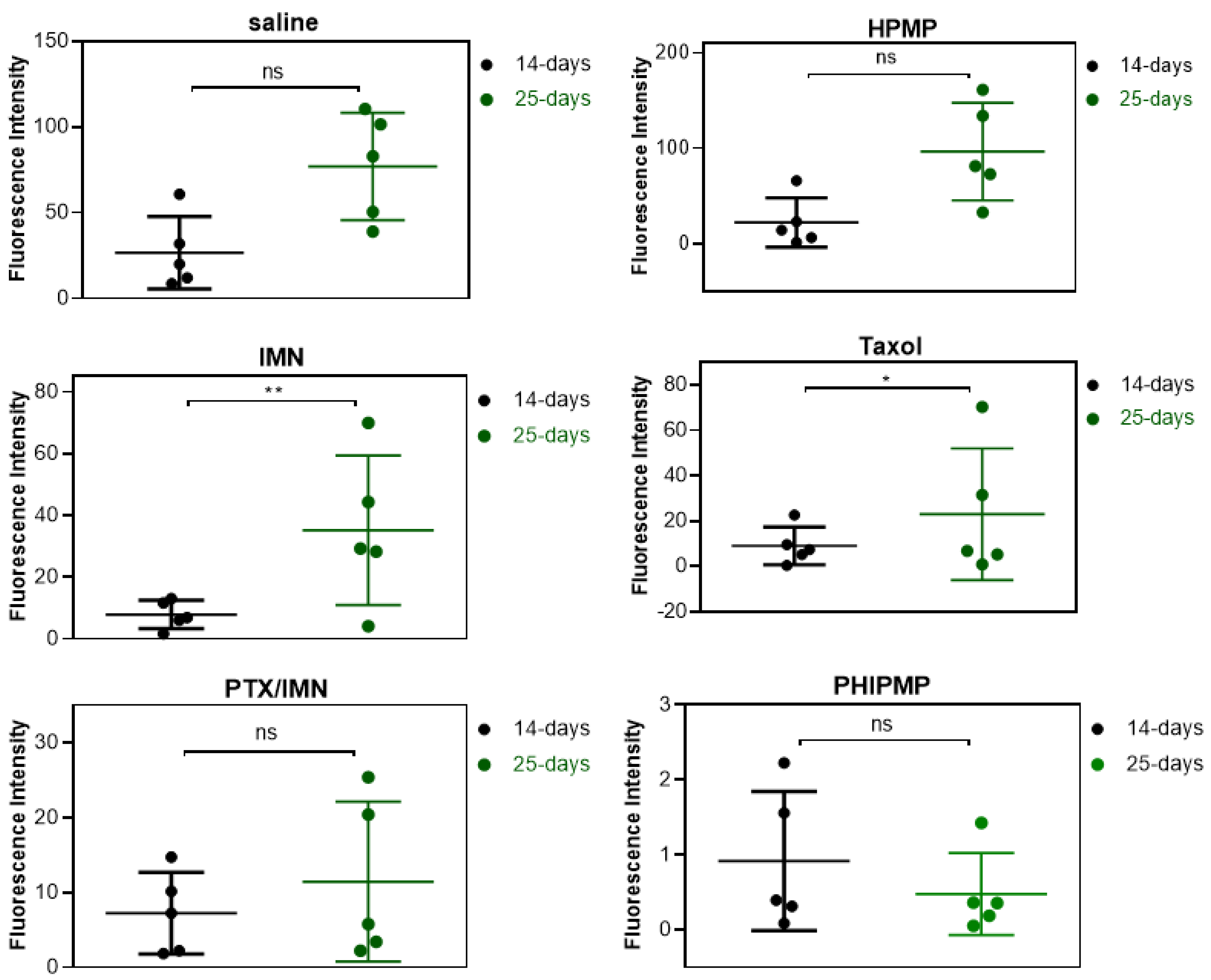
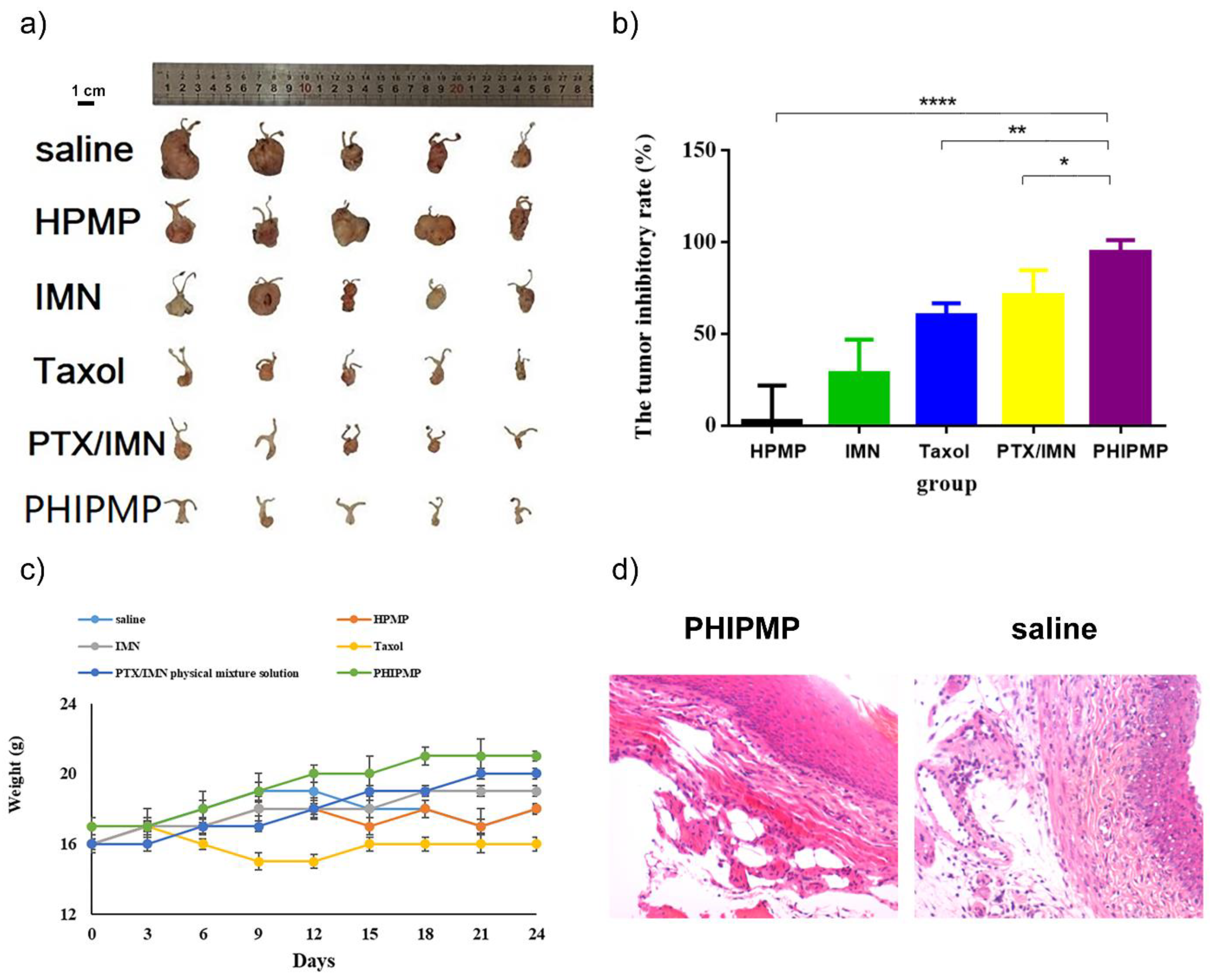
2.6. In Vivo Antitumor Study
3. Materials and Methods
3.1. Materials
3.2. Fabrication of the PTX/HA-IMN/PLGA Microparticles (PHIPMPs), IMN/PLGA Nanoparticles (IPNP), and HA/PLGA Microparticles (HPMPs)
3.3. Structural Morphology and Size Distribution
3.4. Drug Loading (DL) and Encapsulation Efficiency (EE)
3.5. Physical Characterization
3.6. In Vitro Release
3.7. Degradation of PHIPMPs
3.8. Cellular Experiments
3.9. Vaginal Retention Experiment
3.10. In Vivo Antitumor Study
3.11. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nguyen, H.T.; Soe, Z.C.; Yang, K.Y.; Phung, C.D.; Nguyen, L.T.T.; Jeong, J.H.; Jin, S.G.; Choi, H.G.; Ku, S.K.; Yong, C.S.; et al. Transferrin-conjugated pH-sensitive platform for effective delivery of porous palladium nanoparticles and paclitaxel in cancer treatment. Colloids Surf. B Biointerfaces 2019, 176, 265–275. [Google Scholar] [CrossRef]
- Mirrahimi, M.; Abed, Z.; Beik, J.; Shiri, I.; Dezfuli, A.S.; Mahabadi, V.P.; Kamrava, S.K.; Shakeri-Zadeh, A. A thermo-responsive alginate nanogel platform co-loaded with gold nanoparticles and cisplatin for combined cancer chemo-photothermal therapy. Pharmacol. Res. 2019, 143, 178–185. [Google Scholar] [CrossRef]
- Rejhová, A.; Opattová, A.; Čumová, A.; Slíva, D.; Vodička, P. Natural compounds and combination therapy in colorectal cancer treatment. Eur. J. Med. Chem. 2018, 144, 582–594. [Google Scholar] [CrossRef]
- Dong, Q.L.; Zhang, H.Q.; Han, Y.; Djamila, A.; Cheng, H.; Tang, Z.Y.; Zhou, J.P.; Ding, Y. Tumor environment differentiated “nanodepot” programmed for site-specific drug shuttling and combinative therapy on metastatic cancer. J. Control. Release 2018, 283, 59–75. [Google Scholar] [CrossRef]
- Gaber, M.; Elhasany, K.A.; Sabra, S.; Helmy, M.W.; Fang, J.Y.; Khattab, S.N.; Bekhit, A.A.; Teleb, M.; Elkodairy, K.A.; Elzoghby, A.O. Co-Administration of Tretinoin Enhances the Anti-Cancer Efficacy of Etoposide via Tumor-Targeted Green Nano-Micelles. Colloids Surf. B Biointerfaces 2020, 192, 110997. [Google Scholar] [CrossRef] [PubMed]
- Fei, W.D.; Li, C.Q.; Tao, J.Y.; Cai, X.J.; Yao, W.D.; Ye, Y.Q.; Zhang, Y.; Yao, Y.; Song, Q.Q.; Li, F.Z.; et al. Construction of arsenic-metal complexes loaded nanodrugs for solid tumor therapy: A mini review. Int. J. Pharm. 2020, 583, 119385. [Google Scholar] [CrossRef]
- Adityan, S.; Tran, M.; Bhavsar, C.; Wu, S.Y. Nano-therapeutics for modulating the tumour microenvironment: Design, development, and clinical translation. J. Control. Release 2020, 327, 512–532. [Google Scholar] [CrossRef] [PubMed]
- Yuan Zhang, Y.; Bush, X.; Yan, B.F.; Chen, J.A. Gemcitabine nanoparticles promote antitumor immunity against melanoma. Biomaterials 2019, 189, 48–59. [Google Scholar] [CrossRef]
- Sang, C.C.; Ma, L.; Luo, D.; Liu, H.X.; Li, D.; Chen, T.F. Designing bioresponsive metal azolate framework-based nanosystem for efficient cancer therapy. Chem. Eng. J. 2019, 371, 301–305. [Google Scholar] [CrossRef]
- Wu, J. Assessing interactions for fixed-dose drug combinations in subcutaneous tumor xenograft studies. Pharm. Stat. 2013, 12, 115–119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khjauria, V.; Tandon, V.R.; Rani, N.; Roshi Sanjeev, G.; Choudhary, S.; Gilllani, Z. Profile of adverse drug reactions with fixed drug combinations: How big is the problem? JK Sci. 2015, 17, 33–37. [Google Scholar]
- Shim, G.Y.; Kim, M.G.; Kim, D.Y.; Park, J.Y.; Oh, Y.K. Nanoformulation-based sequential combination cancer therapy. Adv. Drug Deliv. Rev. 2017, 115, 57–81. [Google Scholar] [CrossRef]
- Zhang, L.; Su, H.T.; Liu, Y.J.; Pang, N.; Li, J.; Qi, X.R. Enhancing solid tumor therapy with sequential delivery of dexamethasone and docetaxel engineered in a single carrier to overcome stromal resistance to drug delivery. J. Control. Release 2019, 294, 1–16. [Google Scholar] [CrossRef]
- Agrahari, V.; Agrahari, V.; Chou, M.L.; Chew, C.H.; Noll, J.; Burnouf, T. Intelligent micro-/nanorobots as drug and cell carrier devices for biomedical therapeutic advancement: Promising development opportunities and translational challenges. Biomaterials 2020, 260, 120163. [Google Scholar] [CrossRef]
- Wu, D.; Pusuluri, A.; Vogus, D.; Krishnan, V.; Shields, C.W., IV; Kim, J.Y.; Razmi, A.; Mitragotri, S. Design principles of drug combinations for chemotherapy. J. Control. Release 2020, 323, 36–46. [Google Scholar] [CrossRef]
- Hu, H.X.; Lin, Z.Q.; He, B.; Dai, W.B.; Wang, X.Q.; Wang, J.C.; Zhang, X.; Zhang, H.; Zhang, Q. A novel localized co-delivery system with lapatinib microparticles and paclitaxel nanoparticles in a peritumorally injectable in situ hydrogel. J. Control. Release 2015, 220, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Sui, D.Z.; Tang, X.Y.; Ding, J.Q.; Wang, Y.; Qin, Y.; Zhang, N.; Liu, X.R.; Deng, Y.H.; Song, Y.Z. Sequential administration of sialic acid-modified liposomes as carriers for epirubicin and zoledronate elicit stronger antitumor effects with reduced toxicity. Int. J. Pharm. 2021, 602, 120552. [Google Scholar] [CrossRef]
- Zhu, F.; Tan, G.; Zhong, Y.; Jiang, Y.; Cai, L.; Yu, Z.; Liu, S.; Ren, F. Smart nanoplatform for sequential drug release and enhanced chemo-thermal effect of dual drug loaded gold nanorod vesicles for cancer therapy. J. Nanobiotechnol. 2019, 17, 44–59. [Google Scholar] [CrossRef] [PubMed]
- Davoodi, P.; Feng, F.; Xu, Q.; Yan, W.C.; Tong, Y.W.; Srinivasan, M.P. Coaxialelectrohydrodynamic atomization: Microparticles for drug deliveryapplications. J. Control. Release 2015, 205, 70–82. [Google Scholar] [CrossRef] [PubMed]
- Rasekh, M.; Young, C.; Roldo, M.; Lancien, F.; Le Mevel, J.C.; Hafizi, S. Hollow-layered nanoparticles for therapeutic delivery of peptide preparedusing electrospraying. J. Mater. Sci. Mater. Med. 2015, 26, 256. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.M.; Cao, L.H.; Cui, Y.C.; Tu, K.H.; Wang, H.J.; Wang, L.Q. The exploration of endocytic mechanisms of PLA-PEG nanoparticlesprepared by coaxialtri-capillary electrospray-template removal method. Colloids Surf. B Biointerfaces 2018, 161, 10–17. [Google Scholar] [CrossRef]
- George, M.C.; Braun, P.V. Multicompartmental materials by electrohydrodynamic co-jetting. Angew. Chemie Int. Ed. 2009, 48, 8606–8609. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.G.; Li, J.J.; Williams, G.R.; Zhao, M. Electrospun amorphous solid dispersions of poorly water-soluble drugs: A review. J. Control. Release 2018, 292, 91–110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hai, T.; Wan, X.; Yu, D.G.; Wang, K.; Yang, Y.Y.; Liu, Z.P. Electrospun lipid-coated medicated nanocomposites for an improved drug sustained-release profile. Mater. Des. 2019, 162, 70–79. [Google Scholar] [CrossRef]
- Yang, Y.Y.; Zhang, M.; Liu, Z.P.; Wang, K.; Yu, D.G. Meletin sustained-release gliadin nanoparticles prepared via solvent surface modification on blending electrospraying. Appl. Surf. Sci. 2018, 434, 1040–1047. [Google Scholar] [CrossRef]
- Liu, X.K.; Yang, Y.Y.; Yu, D.G.; Zhu, M.J.; Zhao, M.; Williamsc, G.R. Tunable zero-order drug delivery systems created by modified triaxial electrospinning. Chem. Eng. J. 2019, 356, 886–894. [Google Scholar] [CrossRef] [Green Version]
- Zandia, N.; Lotfi, R.; Tamjid, E.; Shokrgozar, M.A.; Simchi, A. Core-sheath gelatin based electrospun nanofibers for dual delivery release of biomolecules and therapeutics. Mater. Sci. Eng. C 2010, 108, 110432. [Google Scholar] [CrossRef]
- Ezrahi, S.; Aserin, A.; Garti, N. Basic principles of drug delivery systems-the case of paclitaxel. Adv. Colloid Interface Sci. 2019, 263, 95–130. [Google Scholar] [CrossRef]
- Yusuf, R.; Duan, Z.; Lamendola, D.; Penson, R.; Seiden, M. Paclitaxel resistance: Molecular mechanisms and pharmacologic manipulation. Curr. Cancer Drug Targets 2003, 3, 1–19. [Google Scholar] [CrossRef]
- Mostoufi, H.; Yousefi, G.; Tamaddon, A.M.; Firuzi, O. Reversing multi-drug tumor resistance to Paclitaxel by well-defined pHsensitive amphiphilic polypeptide block copolymers via induction of lysosomal membrane permeabilization. Colloids Surf. B Biointerfaces 2019, 174, 17–27. [Google Scholar] [CrossRef]
- Choi, B.M.; Kim, Y.M.; Jeong, Y.R.; Pae, H.O.; Song, C.E.; Park, J.E.; Ahn, Y.K.; Chung, H.T. Induction of heme oxygenase-1 is involved in anti-proliferative effects of paclitaxel on rat vascular smooth muscle cells. Biochem. Biophys. Res. Commun. 2004, 321, 132–137. [Google Scholar] [CrossRef]
- Palejwala, A.H.; O’Connor, K.P.; Shi, H.; Villeneuve, L.; Scordino, T.; Glenn, C.A. Chronic myeloid leukemia manifested as myeloid sarcoma: Review of literature and case report. J. Clin. Neurosci. 2019, 64, 269–276. [Google Scholar] [CrossRef]
- Karakas, C.; Christensen, P.; Baek, D.; Jung, M.J.; Ro, J.Y. Dedifferentiated gastrointestinal stromal tumor: Recent advances. Ann. Diagn. Pathol. 2019, 39, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wu, J.Z.; Yang, Y.Q.; Ma, R.; Zhang, J.Y.; Feng, J.F. Expression of growth regulated oncogene1, hepatocyte growth factor, plateletderived growth factorAA and soluble Eselectin and their association with highrisk human papillomavirus infection in squamous cell carcinoma of the uterine cervix. Mol. Med. Rep. 2014, 10, 1013–1024. [Google Scholar] [CrossRef] [Green Version]
- Ci, L.Q.; Huang, Z.G.; Lv, F.M.; Wang, J.; Feng, L.L.; Sun, F.; Cao, S.J.; Liu, Z.P.; Liu, Y.; Wei, G.; et al. Enhanced Delivery of Imatinib into Vaginal Mucosa via a New Positively Charged Nanocrystal-Loaded in Situ Hydrogel Formulation for Treatment of Cervical Cancer. Pharmaceutics 2019, 11, 15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Redondo, A.; Colombo, N.; McCormack, M.; Dreosti, L.; Nogueira-Rodrigues, A.; Scambia, G.; Lorusso, D.; Joly, F.; Schenker, M.; Ruff, P.; et al. Primary results from CECILIA, a global single-arm phase II study evaluating bevacizumab, carboplatin and paclitaxel for advanced cervical cancer. Gynecol. Oncol. 2020, 159, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Symonds, R.P.; Gourley, C.; Davidson, S.; Carty, K.; McCartney, E.; Rai, D.; Banerjee, S.; Jackson, D.; Lord, R.; McCormack, M.; et al. Cediranib combined with carboplatin and paclitaxel in patients with metastatic or recurrent cervical cancer (CIRCCa): A randomised, double-blind, placebo-controlled phase 2 trial. Lancet Oncol. 2015, 16, 1515–1524. [Google Scholar] [CrossRef] [Green Version]
- Corman, S.L.; Nwankwo, C. Years of potential life lost due to cervical and uterine cancer deaths in 2015: Regional and country differences. Gynecol. Oncol. 2020, 159, 2–78. [Google Scholar] [CrossRef]
- Yun, S.K.; Dong, K.S.; Hyemin, K.; Won, H.K.; Young, E.K.; Sei, K.H. Nose-to-brain delivery of hyaluronate—FG loop peptide conjugate for non-invasive hypoxic-ischemic encephalopathy therapy. J. Control. Release 2019, 307, 76–89. [Google Scholar]
- Boni, F.I.; Almeida, A.; Lechanteur, A.; Sarmento, B.; Stringhetti, B.; Cury, F.; Gremião, M.P.D. Mucoadhesive nanostructured polyelectrolytes complexes modulate the intestinal permeability of methotrexate. Eur. J. Pharm. Sci. 2018, 111, 73–82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ensign, L.M.; Cone, R.; Hanes, J. Nanoparticle-based drug delivery to the vagina: A review. J. Control. Release 2014, 190, 500–514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cautela, M.P.; Moshe, H.; Sosnik, A.; Sarmento, B.; das Neves, J. Composite films for vaginal delivery of tenofovir disoproxil fumarate and emtricitabine. Eur. J. Pharm. Biopharm. 2019, 138, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.X.; Li, R.Q.; Li, X.R.; Xie, J.W. Electrospinning: An enabling nanotechnology platform for drug delivery and regenerative medicine. Adv. Drug Deliv. Rev. 2018, 132, 188–213. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Windbergs, M. Influence of polymer composition and drug loading procedure on dual drug release from PLGA:PEG electrospun fibers. Eur. J. Pharm. Sci. 2018, 124, 71–79. [Google Scholar] [CrossRef]
- Zhang, Y.M.; Fang, Q.; Niu, K.; Gan, Z.H.; Yu, Q.S.; Gua, T.X. Time-dependently slow-released multiple-drug eluting external sheath for efficient long-term inhibition of saphenous vein graft failure. J. Control. Release 2019, 293, 172–182. [Google Scholar] [CrossRef]
- Srivastava, A.; Joshi, B.D.; Tandon, P.; Ayala, A.P.; Bansal, A.K.; Damián Grillo, D. Study of polymorphism in imatinib mesylate: A quantum chemical approach using electronic and vibrational spectra. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2013, 103, 325–332. [Google Scholar] [CrossRef]
- Chowdhury, P.; Nagesh, P.K.B.; Hatami, E.; Wagh, S.; Dan, N.; Tripathi, M.K.; Khan, S.; Hafeez, B.B.; Meibohm, B.; Chauhan, S.C.; et al. Tannic acid-inspired paclitaxel nanoparticles for enhanced anticancer effects in breast cancer cells. J. Colloid Interface Sci. 2019, 535, 133–148. [Google Scholar] [CrossRef]
- Yang, C.F.; Xue, Z.L.; Liu, Y.L.; Xiao, J.Y.; Chen, J.R.; Zhang, L.J.; Guo, J.W.; Lin, W.J. Delivery of anticancer drug using pH-sensitive micelles from triblock copolymer MPEG-b-PBAE-b-PLA. Mater. Sci. Eng. C 2018, 84, 254–262. [Google Scholar] [CrossRef]
- Mohamed, E.A.; Hashim, I.I.A.; Yusif, R.M.; Suddek, G.M.; Shaaban, A.A.A.; Badria, F.A.E. Enhanced in vitro cytotoxicity and anti-tumor activity of vorinostat-loaded pluronic micelles with prolonged release and reduced hepatic and renal toxicities. Eur. J. Pharm. Sci. 2017, 96, 232–242. [Google Scholar] [CrossRef]
- Yang, M.; Yu, T.; Wang, Y.Y.; Lai, S.K.; Zeng, Q.; Miao, B.; Tang, B.C.; Simons, B.W.; Ensign, L.M.; Liu, G. Vaginal delivery of paclitaxel via nanoparticles with nonmucoadhesive surfaces suppresses cervical tumor growth. Adv. Healthc. Mater 2014, 3, 1044–1052. [Google Scholar] [CrossRef] [Green Version]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Z.; Chen, H.; Lv, F.; Wang, J.; Zhao, S.; Li, Y.; Xue, X.; Liu, Y.; Wei, G.; Lu, W. Sequential Release of Paclitaxel and Imatinib from Core–Shell Microparticles Prepared by Coaxial Electrospray for Vaginal Therapy of Cervical Cancer. Int. J. Mol. Sci. 2021, 22, 8760. https://doi.org/10.3390/ijms22168760
Liu Z, Chen H, Lv F, Wang J, Zhao S, Li Y, Xue X, Liu Y, Wei G, Lu W. Sequential Release of Paclitaxel and Imatinib from Core–Shell Microparticles Prepared by Coaxial Electrospray for Vaginal Therapy of Cervical Cancer. International Journal of Molecular Sciences. 2021; 22(16):8760. https://doi.org/10.3390/ijms22168760
Chicago/Turabian StyleLiu, Zhepeng, Haini Chen, Fengmei Lv, Jun Wang, Shoujin Zhao, Yijun Li, Xuexin Xue, Yu Liu, Gang Wei, and Weiyue Lu. 2021. "Sequential Release of Paclitaxel and Imatinib from Core–Shell Microparticles Prepared by Coaxial Electrospray for Vaginal Therapy of Cervical Cancer" International Journal of Molecular Sciences 22, no. 16: 8760. https://doi.org/10.3390/ijms22168760
APA StyleLiu, Z., Chen, H., Lv, F., Wang, J., Zhao, S., Li, Y., Xue, X., Liu, Y., Wei, G., & Lu, W. (2021). Sequential Release of Paclitaxel and Imatinib from Core–Shell Microparticles Prepared by Coaxial Electrospray for Vaginal Therapy of Cervical Cancer. International Journal of Molecular Sciences, 22(16), 8760. https://doi.org/10.3390/ijms22168760




