Genetic and Molecular Aspects of Drug-Induced QT Interval Prolongation
Abstract
1. Introduction
2. Congenital Long QT Syndromes
3. Acquired Long QT Syndrome
3.1. Pathophysiology of Drug-Induced Long QT Syndrome
3.1.1. Basic Properties
3.1.2. Molecular Aspects of diLQTS
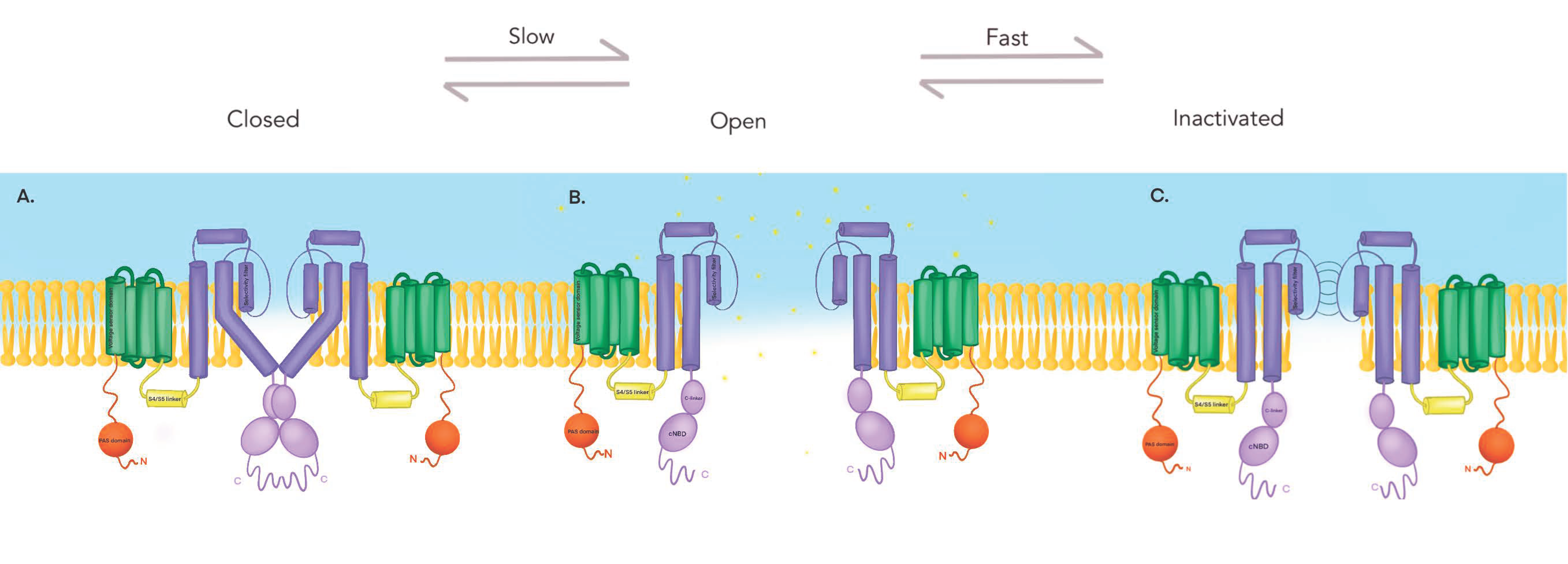
3.1.3. Kv11.1 Gating Kinetics
3.1.4. Molecular Aspects of Drug/Kv11.1 Interaction
3.1.5. The Role of EADs in diLQTS
3.2. Drugs That Cause Acquired Long QT Syndrome
3.3. Risk Factors for Acquired Long QT Syndrome
3.3.1. Drug Regimen and Interactions
3.3.2. Electrolyte Abnormalities
3.3.3. Comorbidities
Heart Disease
Impaired Renal Function
4. Pharmacogenetic Determinants of QT Interval Prolongation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Roden, D.M. Predicting Drug-Induced QT Prolongation and Torsades de Pointes. J. Physiol. 2016, 594, 2459–2468. [Google Scholar] [CrossRef]
- Turner, J.R.; Rodriguez, I.; Mantovani, E.; Gintant, G.; Kowey, P.R.; Klotzbaugh, R.J.; Prasad, K.; Sager, P.T.; Stockbridge, N.; Strnadova, C.; et al. Drug-Induced Proarrhythmia and Torsade de Pointes: A Primer for Students and Practitioners of Medicine and Pharmacy. J. Clin. Pharmacol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Monahan, B.P.; Ferguson, C.L.; Killeavy, E.S.; Lloyd, B.K.; Troy, J.; Cantilena, L.R. Torsades de Pointes Occurring in Association with Terfenadine Use. JAMA 1990, 264, 2788–2790. [Google Scholar] [CrossRef]
- Woosley, R.L.; Chen, Y.; Freiman, J.P.; Gillis, R.A. Mechanism of the Cardiotoxic Actions of Terfenadine. JAMA 1993, 269, 1532–1536. [Google Scholar] [CrossRef]
- Shah, S.R.; Park, K.; Alweis, R. Long QT Syndrome: A Comprehensive Review of the Literature and Current Evidence. Curr. Probl. Cardiol. 2019, 44, 92–106. [Google Scholar] [CrossRef]
- Nachimuthu, S.; Assar, M.D.; Schussler, J.M. Drug-Induced QT Interval Prolongation: Mechanisms and Clinical Management. Ther. Adv. Drug Saf. 2012, 3, 241–253. [Google Scholar] [CrossRef] [PubMed]
- Klotzbaugh, R.J.; Martin, A.; Turner, J.R. Drug-Induced Proarrhythmia: Discussion and Considerations for Clinical Practice. J. Am. Assoc. Nurse Pract. 2020, 32, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Smulyan, H. QT Interval: Bazett’s Correction Corrected. J. Electrocardiol. 2018, 51, 1009–1010. [Google Scholar] [CrossRef]
- Taran, L.M.; Szilagyi, N. The Duration of the Electrical Systole, Q-T, in Acute Rheumatic Carditis in Children. Am. Heart J. 1947, 33, 14–26. [Google Scholar] [CrossRef]
- Drew, B.J.; Ackerman, M.J.; Funk, M.; Gibler, W.B.; Kligfield, P.; Menon, V.; Philippides, G.J.; Roden, D.M.; Zareba, W.; American Heart Association Acute Cardiac Care Committee of the Council on Clinical Cardiology; et al. Prevention of Torsade de Pointes in Hospital Settings: A Scientific Statement from the American Heart Association and the American College of Cardiology Foundation. J. Am. Coll. Cardiol. 2010, 55, 934–947. [Google Scholar] [CrossRef]
- Priori, S.G.; Blomström-Lundqvist, C.; Mazzanti, A.; Blom, N.; Borggrefe, M.; Camm, J.; Elliott, P.M.; Fitzsimons, D.; Hatala, R.; Hindricks, G.; et al. 2015 ESC Guidelines for the Management of Patients with Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death: The Task Force for the Management of Patients with Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death of the European Society of Cardiology (ESC). Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC). Eur. Heart J. 2015, 36, 2793–2867. [Google Scholar] [CrossRef]
- El-Sherif, N.; Turitto, G.; Boutjdir, M. Acquired Long QT Syndrome and Torsade de Pointes. Pacing Clin. Electrophysiol. 2018, 41, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Titier, K.; Girodet, P.-O.; Verdoux, H.; Molimard, M.; Bégaud, B.; Haverkamp, W.; Lader, M.; Moore, N. Atypical Antipsychotics: From Potassium Channels to Torsade de Pointes and Sudden Death. Drug Saf. 2005, 28, 35–51. [Google Scholar] [CrossRef] [PubMed]
- Giudicessi, J.R.; Wilde, A.A.M.; Ackerman, M.J. The Genetic Architecture of Long QT Syndrome: A Critical Reappraisal. Trends Cardiovasc. Med. 2018, 28, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Khan, Q.; Ismail, M.; Haider, I.; Ali, Z. Prevalence of the Risk Factors for QT Prolongation and Associated Drug–Drug Interactions in a Cohort of Medical Inpatients. J. Formos. Med. Assoc. 2019, 118, 109–115. [Google Scholar] [CrossRef]
- Barsheshet, A.; Dotsenko, O.; Goldenberg, I. Genotype-Specific Risk Stratification and Management of Patients with Long QT Syndrome. Ann. Noninvasive Electrocardiol. 2013, 18, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Neira, V.; Enriquez, A.; Simpson, C.; Baranchuk, A. Update on Long QT Syndrome. J. Cardiovasc. Electrophysiol. 2019, 30, 3068–3078. [Google Scholar] [CrossRef]
- Bohnen, M.S.; Peng, G.; Robey, S.H.; Terrenoire, C.; Iyer, V.; Sampson, K.J.; Kass, R.S. Molecular Pathophysiology of Congenital Long QT Syndrome. Physiol. Rev. 2017, 97, 89–134. [Google Scholar] [CrossRef]
- Turker, I.; Ai, T.; Itoh, H.; Horie, M. Drug-Induced Fatal Arrhythmias: Acquired Long QT and Brugada Syndromes. Pharmacol. Ther. 2017, 176, 48–59. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, P.J.; Woosley, R.L. Predicting the Unpredictable: Drug-Induced QT Prolongation and Torsades de Pointes. J. Am. Coll. Cardiol. 2016, 67, 1639–1650. [Google Scholar] [CrossRef]
- Molokhia, M.; Pathak, A.; Lapeyre-Mestre, M.; Caturla, L.; Montastruc, J.L.; L’Association Française des Centres Régionaux de Pharmacovigilance (CRPV); McKeigue, P. Case Ascertainment and Estimated Incidence of Drug-Induced Long-QT Syndrome: Study in Southwest France. Br. J. Clin. Pharmacol. 2008, 66, 386–395. [Google Scholar] [CrossRef] [PubMed]
- Mahida, S.; Hogarth, A.J.; Cowan, C.; Tayebjee, M.H.; Graham, L.N.; Pepper, C.B. Genetics of Congenital and Drug-Induced Long QT Syndromes: Current Evidence and Future Research Perspectives. J. Interv. Card. Electrophysiol. 2013, 37, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Heemskerk, C.P.M.; Pereboom, M.; van Stralen, K.; Berger, F.A.; van den Bemt, P.M.L.A.; Kuijper, A.F.M.; van der Hoeven, R.T.M.; Mantel-Teeuwisse, A.K.; Becker, M.L. Risk Factors for QTc Interval Prolongation. Eur. J. Clin. Pharmacol. 2018, 74, 183–191. [Google Scholar] [CrossRef]
- CredibleMeds: Home. Available online: https://www.crediblemeds.org/ (accessed on 21 March 2021).
- Leishman, D.J.; Abernathy, M.M.; Wang, E.B. Revisiting the HERG Safety Margin after 20 Years of Routine HERG Screening. J. Pharmacol. Toxicol. Methods 2020, 105, 106900. [Google Scholar] [CrossRef]
- Itoh, H.; Crotti, L.; Aiba, T.; Spazzolini, C.; Denjoy, I.; Fressart, V.; Hayashi, K.; Nakajima, T.; Ohno, S.; Makiyama, T.; et al. The Genetics Underlying Acquired Long QT Syndrome: Impact for Genetic Screening. Eur. Heart J. 2016, 37, 1456–1464. [Google Scholar] [CrossRef] [PubMed]
- Ridder, B.J.; Leishman, D.J.; Bridgland-Taylor, M.; Samieegohar, M.; Han, X.; Wu, W.W.; Randolph, A.; Tran, P.; Sheng, J.; Danker, T.; et al. A Systematic Strategy for Estimating HERG Block Potency and Its Implications in a New Cardiac Safety Paradigm. Toxicol. Appl. Pharmacol. 2020, 394, 114961. [Google Scholar] [CrossRef]
- Barsheshet, A.; Dotsenko, O.; Goldenberg, I. Congenital Long QT Syndromes: Prevalence, Pathophysiology and Management. Paediatr. Drugs 2014, 16, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Priori, S.G.; Wilde, A.A.; Horie, M.; Cho, Y.; Behr, E.R.; Berul, C.; Blom, N.; Brugada, J.; Chiang, C.-E.; Huikuri, H.; et al. Executive Summary: HRS/EHRA/APHRS Expert Consensus Statement on the Diagnosis and Management of Patients with Inherited Primary Arrhythmia Syndromes. J. Arrhythmia 2014, 30, 29–47. [Google Scholar] [CrossRef]
- Schwartz, P.J.; Crotti, L. QTc Behavior during Exercise and Genetic Testing for the Long-QT Syndrome. Circulation 2011, 124, 2181–2184. [Google Scholar] [CrossRef]
- Adler, A.; Novelli, V.; Amin, A.S.; Abiusi, E.; Care, M.; Nannenberg, E.A.; Feilotter, H.; Amenta, S.; Mazza, D.; Bikker, H.; et al. An International, Multicentered, Evidence-Based Reappraisal of Genes Reported to Cause Congenital Long QT Syndrome. Circulation 2020, 141, 418–428. [Google Scholar] [CrossRef]
- Nakano, Y.; Shimizu, W. Genetics of Long-QT Syndrome. J. Hum. Genet. 2016, 61, 51–55. [Google Scholar] [CrossRef]
- El-Sherif, N.; Turitto, G.; Boutjdir, M. Congenital Long QT Syndrome and Torsade de Pointes. Ann. Noninvasive Electrocardiol. 2017, 22, e12481. [Google Scholar] [CrossRef] [PubMed]
- Kallergis, E.M.; Goudis, C.A.; Simantirakis, E.N.; Kochiadakis, G.E.; Vardas, P.E. Mechanisms, Risk Factors, and Management of Acquired Long QT Syndrome: A Comprehensive Review. Sci. World J. 2012, 2012, 212178. [Google Scholar] [CrossRef]
- Tisdale, J.E.; Chung, M.K.; Campbell, K.B.; Hammadah, M.; Joglar, J.A.; Leclerc, J.; Rajagopalan, B.; American Heart Association. Clinical Pharmacology Committee of the Council on Clinical Cardiology and Council on Cardiovascular and Stroke Nursing Drug-Induced Arrhythmias: A Scientific Statement from the American Heart Association. Circulation 2020, 142, e214–e233. [Google Scholar] [CrossRef] [PubMed]
- Roden, D.M.; Viswanathan, P.C. Genetics of Acquired Long QT Syndrome. J. Clin. Investig. 2005, 115, 2025–2032. [Google Scholar] [CrossRef]
- Hodge, C.; Marra, F.; Marzolini, C.; Boyle, A.; Gibbons, S.; Siccardi, M.; Burger, D.; Back, D.; Khoo, S. Drug Interactions: A Review of the Unseen Danger of Experimental COVID-19 Therapies. J. Antimicrob. Chemother. 2020, 75, 3417–3424. [Google Scholar] [CrossRef] [PubMed]
- Chorin, E.; Wadhwani, L.; Magnani, S.; Dai, M.; Shulman, E.; Nadeau-Routhier, C.; Knotts, R.; Bar-Cohen, R.; Kogan, E.; Barbhaiya, C.; et al. QT Interval Prolongation and Torsade de Pointes in Patients with COVID-19 Treated with Hydroxychloroquine/Azithromycin. Heart Rhythm 2020, 17, 1425–1433. [Google Scholar] [CrossRef] [PubMed]
- Lazzerini, P.E.; Boutjdir, M.; Capecchi, P.L. COVID-19, Arrhythmic Risk, and Inflammation. Circulation 2020, 142, 7–9. [Google Scholar] [CrossRef]
- Cubeddu, L.X. Drug-Induced Inhibition and Trafficking Disruption of Ion Channels: Pathogenesis of QT Abnormalities and Drug-Induced Fatal Arrhythmias. Curr. Cardiol. Rev. 2016, 12, 141–154. [Google Scholar] [CrossRef]
- El-Sherif, N.; Turitto, G.; Boutjdir, M. Acquired Long QT Syndrome and Electrophysiology of Torsade de Pointes. Arrhythm Electrophysiol. Rev. 2019, 8, 122–130. [Google Scholar] [CrossRef]
- Antzelevitch, C. Cardiac Repolarization. The Long and Short of It. Europace 2005, 7, 3–9. [Google Scholar] [CrossRef]
- Lee, W.; Windley, M.J.; Vandenberg, J.I.; Hill, A.P. In vitro and In silico Risk Assessment in Acquired Long QT Syndrome: The Devil Is in the Details. Front. Physiol. 2017, 8, 934. [Google Scholar] [CrossRef]
- Vandenberg, J.I.; Perry, M.D.; Perrin, M.J.; Mann, S.A.; Ke, Y.; Hill, A.P. HERG K(+) Channels: Structure, Function, and Clinical Significance. Physiol. Rev. 2012, 92, 1393–1478. [Google Scholar] [CrossRef]
- Perrin, M.J.; Subbiah, R.N.; Vandenberg, J.I.; Hill, A.P. Human Ether-a-Go-Go Related Gene (HERG) K+ Channels: Function and Dysfunction. Prog. Biophys. Mol. Biol. 2008, 98, 137–148. [Google Scholar] [CrossRef]
- Wang, W.; MacKinnon, R. Cryo-EM Structure of the Open Human Ether-à-Go-Go-Related K+ Channel HERG. Cell 2017, 169, 422–430.e10. [Google Scholar] [CrossRef] [PubMed]
- Cabral, J.H.M.; Lee, A.; Cohen, S.L.; Chait, B.T.; Li, M.; Mackinnon, R. Crystal Structure and Functional Analysis of the HERG Potassium Channel N Terminus: A Eukaryotic PAS Domain. Cell 1998, 95, 649–655. [Google Scholar] [CrossRef]
- Robertson, G.A.; Morais-Cabral, J.H. HERG Function in Light of Structure. Biophys. J. 2020, 118, 790–797. [Google Scholar] [CrossRef]
- de la Peña, P.; Domínguez, P.; Barros, F. Gating Mechanism of Kv11.1 (HERG) K+ Channels without Covalent Connection between Voltage Sensor and Pore Domains. Pflug. Arch. Eur. J. Physiol. 2018, 470, 517–536. [Google Scholar] [CrossRef] [PubMed]
- Hardman, R.M.; Stansfeld, P.J.; Dalibalta, S.; Sutcliffe, M.J.; Mitcheson, J.S. Activation Gating of HERG Potassium Channels: S6 Glycines Are Not Required as Gating Hinges*. J. Biol. Chem. 2007, 282, 31972–31981. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Shen, R.; Reddy, B.; Perozo, E.; Roux, B. Mechanism of C-Type Inactivation in the HERG Potassium Channel. Sci. Adv. 2021, 7, eabd6203. [Google Scholar] [CrossRef] [PubMed]
- Durdagi, S.; Deshpande, S.; Duff, H.J.; Noskov, S.Y. Modeling of Open, Closed, and Open-Inactivated States of the HERG1 Channel: Structural Mechanisms of the State-Dependent Drug Binding. J. Chem. Inf. Model. 2012, 52, 2760–2774. [Google Scholar] [CrossRef]
- Chen, J.; Seebohm, G.; Sanguinetti, M.C. Position of Aromatic Residues in the S6 Domain, Not Inactivation, Dictates Cisapride Sensitivity of HERG and Eag Potassium Channels. Proc. Natl. Acad. Sci. USA 2002, 99, 12461–12466. [Google Scholar] [CrossRef] [PubMed]
- Butler, A.; Helliwell, M.V.; Zhang, Y.; Hancox, J.C.; Dempsey, C.E. An Update on the Structure of HERG. Front. Pharmacol. 2019, 10, 1572. [Google Scholar] [CrossRef]
- Macdonald, L.C.; Kim, R.Y.; Kurata, H.T.; Fedida, D. Probing the Molecular Basis of HERG Drug Block with Unnatural Amino Acids. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Gardner, A.; Sanguinetti, M.C. The Link between Inactivation and High-Affinity Block of HERG1 Channels. Mol. Pharmacol. 2015, 87, 1042–1050. [Google Scholar] [CrossRef] [PubMed]
- De Waard, S.; Montnach, J.; Ribeiro, B.; Nicolas, S.; Forest, V.; Charpentier, F.; Mangoni, M.E.; Gaborit, N.; Ronjat, M.; Loussouarn, G.; et al. Functional Impact of BeKm-1, a High-Affinity HERG Blocker, on Cardiomyocytes Derived from Human-Induced Pluripotent Stem Cells. Int. J. Mol. Sci. 2020, 21, 7167. [Google Scholar] [CrossRef]
- Saxena, P.; Zangerl-Plessl, E.-M.; Linder, T.; Windisch, A.; Hohaus, A.; Timin, E.; Hering, S.; Stary-Weinzinger, A. New Potential Binding Determinant for HERG Channel Inhibitors. Sci. Rep. 2016, 6, 24182. [Google Scholar] [CrossRef]
- Helliwell, M.V.; Zhang, Y.; El Harchi, A.; Du, C.; Hancox, J.C.; Dempsey, C.E. Structural Implications of HERG K+ Channel Block by a High-Affinity Minimally Structured Blocker. J. Biol. Chem. 2018, 293, 7040–7057. [Google Scholar] [CrossRef]
- Dickson, C.J.; Velez-Vega, C.; Duca, J.S. Revealing Molecular Determinants of HERG Blocker and Activator Binding. J. Chem. Inf. Model. 2020, 60, 192–203. [Google Scholar] [CrossRef]
- Zhao, Z.; Xie, Y.; Wen, H.; Xiao, D.; Allen, C.; Fefelova, N.; Dun, W.; Boyden, P.A.; Qu, Z.; Xie, L.-H. Role of the Transient Outward Potassium Current in the Genesis of Early Afterdepolarizations in Cardiac Cells. Cardiovasc. Res. 2012, 95, 308–316. [Google Scholar] [CrossRef]
- Qu, Z.; Xie, L.-H.; Olcese, R.; Karagueuzian, H.S.; Chen, P.-S.; Garfinkel, A.; Weiss, J.N. Early Afterdepolarizations in Cardiac Myocytes: Beyond Reduced Repolarization Reserve. Cardiovasc. Res. 2013, 99, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Chu, Z.; Yang, D.; Huang, X. Conditions for the Genesis of Early Afterdepolarization in a Model of a Ventricular Myocyte. Chaos 2020, 30, 043105. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Han, D.; Sun, X.; Tan, H.; Wang, Z.; Liu, C.; Zhang, Y.; Li, B.; Sun, C.; Shi, R.; et al. Prevalence and Risk Factors of Acquired Long QT Syndrome in Hospitalized Patients with Chronic Kidney Disease. J. Investig. Med. 2019, 67, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Madhvani, R.V.; Angelini, M.; Xie, Y.; Pantazis, A.; Suriany, S.; Borgstrom, N.P.; Garfinkel, A.; Qu, Z.; Weiss, J.N.; Olcese, R. Targeting the Late Component of the Cardiac L-Type Ca2+ Current to Suppress Early Afterdepolarizations. J. Gen. Physiol. 2015, 145, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.B.; Vandersickel, N.; Panfilov, A.; Qu, Z. R-from-T as a Common Mechanism of Arrhythmia Initiation in Long QT Syndromes. Circ. Arrhythmia Electrophysiol. 2019, 12. [Google Scholar] [CrossRef] [PubMed]
- Woosley, R.L.; Romero, K.; Heise, C.W.; Gallo, T.; Tate, J.; Woosley, R.D.; Ward, S. Adverse Drug Event Causality Analysis (ADECA): A Process for Evaluating Evidence and Assigning Drugs to Risk Categories for Sudden Death. Drug Saf. 2017, 40, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Iwata, N.; Karasawa, M.; Omine, M.; Maekawa, T.; Suzuki, T.; Kawai, Y. Aclarubicin-Associated QTc Prolongation and Ventricular Fibrillation. Cancer Treat. Rep. 1984, 68, 527–529. [Google Scholar]
- Van Leeuwen, M.T.; Luu, S.; Gurney, H.; Brown, M.R.; Pearson, S.-A.; Webber, K.; Hunt, L.; Hong, S.; Delaney, G.P.; Vajdic, C.M. Cardiovascular Toxicity of Targeted Therapies for Cancer: An Overview of Systematic Reviews. JNCI Cancer Spectr. 2020, 4, pkaa076. [Google Scholar] [CrossRef]
- Mourad, A.; Stiber, J.A.; Perfect, J.R.; Johnson, M.D. Real-World Implications of QT Prolongation in Patients Receiving Voriconazole and Amiodarone. J. Antimicrob. Chemother. 2019, 74, 228–233. [Google Scholar] [CrossRef]
- Whittaker, D.G.; Ni, H.; Benson, A.P.; Hancox, J.C.; Zhang, H. Computational Analysis of the Mode of Action of Disopyramide and Quinidine on HERG-Linked Short QT Syndrome in Human Ventricles. Front. Physiol. 2017, 8. [Google Scholar] [CrossRef]
- Wenzel-Seifert, K.; Wittmann, M.; Haen, E. QTc Prolongation by Psychotropic Drugs and the Risk of Torsade de Pointes. Dtsch. Arztebl. Arztl. Mitt. Ausg. B 2011, 108, 687–693. [Google Scholar] [CrossRef]
- Cubeddu, L.X. Iatrogenic QT Abnormalities and Fatal Arrhythmias: Mechanisms and Clinical Significance. Curr. Cardiol. Rev. 2009, 5, 166–176. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, P.J.; Menezes, P.A.; Vu, H.T.; Rivera, A.L.; Ramaswamy, K. Fluconazole- and Levofloxacin-Induced Torsades de Pointes in an Intensive Care Unit Patient. Am. J. Health Syst. Pharm. 2003, 60, 2479–2483. [Google Scholar] [CrossRef] [PubMed]
- Michaud, V.; Dow, P.; Rihani, S.B.A.; Deodhar, M.; Arwood, M.; Cicali, B.; Turgeon, J. Risk Assessment of Drug-Induced Long QT Syndrome for Some COVID-19 Repurposed Drugs. medRxiv 2020. [Google Scholar] [CrossRef]
- Juurlink, D.N. Safety Considerations with Chloroquine, Hydroxychloroquine and Azithromycin in the Management of SARS-CoV-2 Infection. CMAJ 2020, 192, E450–E453. [Google Scholar] [CrossRef] [PubMed]
- Beach, S.R.; Celano, C.M.; Sugrue, A.M.; Adams, C.; Ackerman, M.J.; Noseworthy, P.A.; Huffman, J.C. QT Prolongation, Torsades de Pointes, and Psychotropic Medications: A 5-Year Update. Psychosomatics 2018, 59, 105–122. [Google Scholar] [CrossRef]
- Su, Z.; Martin, R.; Cox, B.F.; Gintant, G. Mesoridazine: An Open-Channel Blocker of Human Ether-a-Go-Go-Related Gene K+ Channel. J. Mol. Cell Cardiol. 2004, 36, 151–160. [Google Scholar] [CrossRef]
- Lee, H.-A.; Kim, K.-S.; Park, S.-J.; Kim, E.-J. Cellular Mechanism of the QT Prolongation Induced by Sulpiride. Int. J. Toxicol. 2009, 28, 207–212. [Google Scholar] [CrossRef]
- Spevak, C.; Hamsher, C.; Brown, C.Q.; Wedam, E.F.; Haigney, M.C. The Clinical Significance of QT Interval Prolongation in Anesthesia and Pain Management: What You Should and Should Not Worry About. Pain Med. 2012, 13, 1072–1080. [Google Scholar] [CrossRef]
- Rossi, M.; Giorgi, G. Domperidone and Long QT Syndrome. Curr. Drug Saf. 2010, 5, 257–262. [Google Scholar] [CrossRef]
- Ellermann, C.; Könemann, H.; Wolfes, J.; Rath, B.; Wegner, F.K.; Willy, K.; Dechering, D.G.; Reinke, F.; Eckardt, L.; Frommeyer, G. Propofol Abolishes Torsade de Pointes in Different Models of Acquired Long QT Syndrome. Sci. Rep. 2020, 10, 12133. [Google Scholar] [CrossRef]
- Kuryshev, Y.A.; Ficker, E.; Wang, L.; Hawryluk, P.; Dennis, A.T.; Wible, B.A.; Brown, A.M.; Kang, J.; Chen, X.-L.; Sawamura, K.; et al. Pentamidine-Induced Long QT Syndrome and Block of HERG Trafficking. J. Pharmacol. Exp. Ther. 2005, 312, 316–323. [Google Scholar] [CrossRef]
- Malone, K.; Hancox, J.C. QT Interval Prolongation and Torsades de Pointes with Donepezil, Rivastigmine and Galantamine. Ther. Adv. Drug Saf. 2020, 11. [Google Scholar] [CrossRef]
- Wieneke, H.; Conrads, H.; Wolstein, J.; Breuckmann, F.; Gastpar, M.; Erbel, R.; Scherbaum, N. Levo-α-Acetylmethadol (LAAM) Induced QTc-Prolongation—Results from a Controlled Clinical Trial. Eur. J. Med. Res. 2009, 14, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Koenig, X.; Kovar, M.; Rubi, L.; Mike, A.K.; Lukacs, P.; Gawali, V.S.; Todt, H.; Hilber, K.; Sandtner, W. Anti-Addiction Drug Ibogaine Inhibits Voltage-Gated Ionic Currents: A Study to Assess the Drug’s Cardiac Ion Channel Profile. Toxicol. Appl. Pharmacol. 2013, 273, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Vandael, E.; Vandenberk, B.; Vandenberghe, J.; Willems, R.; Foulon, V. Risk Factors for QTc-Prolongation: Systematic Review of the Evidence. Int. J. Clin. Pharm. 2017, 39, 16–25. [Google Scholar] [CrossRef]
- Tisdale, J.E.; Jaynes, H.A.; Kingery, J.R.; Mourad, N.A.; Trujillo, T.N.; Overholser, B.R.; Kovacs, R.J. Development and Validation of a Risk Score to Predict QT Interval Prolongation in Hospitalized Patients. Circ. Cardiovasc. Qual. Outcomes 2013, 6, 479–487. [Google Scholar] [CrossRef]
- Lexi-Interact Data Fields. Available online: http://webstore.lexi.com/Information/Product-Information/Lexi-Interact-Fields (accessed on 10 June 2021).
- Berger, F.A.; van der Sijs, H.; van Gelder, T.; Kuijper, A.F.M.; van den Bemt, P.M.L.A.; Becker, M.L. Comparison of Two Algorithms to Support Medication Surveillance for Drug-Drug Interactions between QTc-Prolonging Drugs. Int. J. Med Inform. 2021, 145, 104329. [Google Scholar] [CrossRef] [PubMed]
- Cytochrome P450 Drug Interaction Table—Drug Interactions. Available online: https://drug-interactions.medicine.iu.edu/MainTable.aspx (accessed on 10 June 2021).
- Chhabra, S.T.; Mehta, S.; Chhabra, S.; Singla, M.; Aslam, N.; Mohan, B.; Wander, G.S. Hypocalcemia Presenting as Life Threatening Torsades de Pointes with Prolongation of QTc Interval. Ind. J. Clin. Biochem. 2018, 33, 235–238. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Snyders, D.J.; Roden, D.M. Rapid Inactivation Determines the Rectification and [K+]o Dependence of the Rapid Component of the Delayed Rectifier K+ Current in Cardiac Cells. Circ. Res. 1997, 80, 782–789. [Google Scholar] [CrossRef]
- Yu, H.; Zhang, L.; Liu, J.; Liu, Y.; Kowey, P.R.; Zhang, Y.; Chen, Y.; Wei, Y.; Gao, L.; Li, H.; et al. Acquired Long QT Syndrome in Hospitalized Patients. Heart Rhythm 2017, 14, 974–978. [Google Scholar] [CrossRef]
- Němec, J.; Kim, J.J.; Salama, G. The Link between Abnormal Calcium Handling and Electrical Instability in Acquired Long QT Syndrome—Does Calcium Precipitate Arrhythmic Storms? Prog. Biophys. Mol. Biol. 2016, 120, 210–221. [Google Scholar] [CrossRef]
- El-Sherif, N.; Turitto, G. Electrolyte Disorders and Arrhythmogenesis. Cardiol. J. 2011, 18, 233–245. [Google Scholar] [PubMed]
- Liu, M.; Zhang, L.; Li, S.; Yang, T.; Liu, L.; Zhao, J.; Liu, H. Prediction of HERG Potassium Channel Blockage Using Ensemble Learning Methods and Molecular Fingerprints. Toxicol. Lett. 2020, 332, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Laecke, S.V. Hypomagnesemia and Hypermagnesemia. Acta Clin. Belg. 2019, 74, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Madias, C.; Fitzgibbons, T.P.; Alsheikh-Ali, A.A.; Bouchard, J.L.; Kalsmith, B.; Garlitski, A.C.; Tighe, D.A.; Estes, N.A.M.; Aurigemma, G.P.; Link, M.S. Acquired Long QT Syndrome from Stress Cardiomyopathy Is Associated with Ventricular Arrhythmias and Torsades de Pointes. Heart Rhythm 2011, 8, 555–561. [Google Scholar] [CrossRef]
- Manolis, A.S.; Manolis, A.A.; Manolis, T.A.; Apostolopoulos, E.J.; Papatheou, D.; Melita, H. COVID-19 Infection and Cardiac Arrhythmias. Trends Cardiovasc. Med. 2020, 30, 451–460. [Google Scholar] [CrossRef]
- Si, D.; Du, B.; Ni, L.; Yang, B.; Sun, H.; Jiang, N.; Liu, G.; Massé, S.; Jin, L.; Nanthakumar, J.; et al. Death, Discharge and Arrhythmias among Patients with COVID-19 and Cardiac Injury. CMAJ 2020, 192, E791–E798. [Google Scholar] [CrossRef] [PubMed]
- Anupama, B.K.; Adhikari, S.; Chaudhuri, D. Prolonged QT Interval in a Patient with Coronavirus Disease-2019: Beyond Hydroxychloroquine and Azithromycin. J. Investig. Med. High Impact Case Rep. 2020, 8. [Google Scholar] [CrossRef]
- Lazzerini, P.E.; Laghi-Pasini, F.; Bertolozzi, I.; Morozzi, G.; Lorenzini, S.; Simpatico, A.; Selvi, E.; Bacarelli, M.R.; Finizola, F.; Vanni, F.; et al. Systemic Inflammation as a Novel QT-Prolonging Risk Factor in Patients with Torsades de Pointes. Heart 2017, 103, 1821–1829. [Google Scholar] [CrossRef]
- Wang, J.; Wang, H.; Zhang, Y.; Gao, H.; Nattel, S.; Wang, Z. Impairment of HERG K(+) Channel Function by Tumor Necrosis Factor-Alpha: Role of Reactive Oxygen Species as a Mediator. J. Biol. Chem. 2004, 279, 13289–13292. [Google Scholar] [CrossRef]
- Liu, P.; Wang, L.; Han, D.; Sun, C.; Xue, X.; Li, G. Acquired Long QT Syndrome in Chronic Kidney Disease Patients. Ren. Fail. 2020, 42, 54–65. [Google Scholar] [CrossRef]
- Tarquini, R.; Lazzeri, C.; Pala, L.; Rotella, C.M.; Gensini, G.F. The Diabetic Cardiomyopathy. Acta Diabetol. 2011, 48, 173–181. [Google Scholar] [CrossRef]
- Lopez-Medina, A.I.; Campos-Staffico, A.M.; Luzum, J.A. QT Prolongation with Hydroxychloroquine and Azithromycin for the Treatment of COVID-19: The Need for Pharmacogenetic Insights. J. Cardiovasc. Electrophysiol. 2020, 31, 2793–2794. [Google Scholar] [CrossRef]
- Strauss, D.G.; Vicente, J.; Johannesen, L.; Blinova, K.; Mason, J.W.; Weeke, P.; Behr, E.R.; Roden, D.M.; Woosley, R.; Kosova, G.; et al. Common Genetic Variant Risk Score Is Associated with Drug-Induced QT Prolongation and Torsade de Pointes Risk. Circulation 2017, 135, 1300–1310. [Google Scholar] [CrossRef] [PubMed]
- Niemeijer, M.N.; van den Berg, M.E.; Eijgelsheim, M.; Rijnbeek, P.R.; Stricker, B.H. Pharmacogenetics of Drug-Induced QT Interval Prolongation: An Update. Drug Saf. 2015, 38, 855–867. [Google Scholar] [CrossRef] [PubMed]
- Avery, C.L.; Sitlani, C.M.; Arking, D.E.; Arnett, D.K.; Bis, J.C.; Boerwinkle, E.; Buckley, B.M.; Ida Chen, Y.-D.; de Craen, A.J.M.; Eijgelsheim, M.; et al. Drug-Gene Interactions and the Search for Missing Heritability: A Cross-Sectional Pharmacogenomics Study of the QT Interval. Pharm. J. 2014, 14, 6–13. [Google Scholar] [CrossRef] [PubMed]
- van Noord, C.; Eijgelsheim, M.; Stricker, B.H.C. Drug- and Non-Drug-Associated QT Interval Prolongation. Br. J. Clin. Pharmacol. 2010, 70, 16–23. [Google Scholar] [CrossRef]
- Ramirez, A.H.; Shaffer, C.M.; Delaney, J.T.; Sexton, D.P.; Levy, S.E.; Rieder, M.J.; Nickerson, D.A.; George, A.L.; Roden, D.M. Novel Rare Variants in Congenital Cardiac Arrhythmia Genes Are Frequent in Drug-Induced Torsades de Pointes. Pharm. J. 2013, 13, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Weeke, P.E. Chapter Thirteen—Pharmacogenetics in Cardiovascular Medicine. In Pharmacogenetics; Advances in Pharmacology Series; Brøsen, K., Damkier, P., Eds.; Academic Press: Cambridge, MA, USA, 2018; Volume 83, pp. 333–360. [Google Scholar]
- Visscher, H.; Amstutz, U.; Sistonen, J.; Ross, C.J.; Hayden, M.R.; Carleton, B.C. Pharmacogenomics of Cardiovascular Drugs and Adverse Effects in Pediatrics. J. Cardiovasc. Pharmacol. 2011, 58, 228–239. [Google Scholar] [CrossRef]
- van Noord, C.; Aarnoudse, A.-J.L.H.J.; Eijgelsheim, M.; Sturkenboom, M.C.J.M.; Straus, S.M.J.M.; Hofman, A.; Kors, J.A.; Newton-Cheh, C.; Witteman, J.C.M.; Stricker, B.H.C. Calcium Channel Blockers, NOS1AP, and Heart-Rate-Corrected QT Prolongation. Pharm. Genom. 2009, 19, 260–266. [Google Scholar] [CrossRef]
- Judson, R.S.; Salisbury, B.A.; Reed, C.R.; Ackerman, M.J. Pharmacogenetic Issues in Thorough QT Trials. Mol. Diagn. Ther. 2006, 10, 153–162. [Google Scholar] [CrossRef]
- Behr, E.R.; Roden, D. Drug-Induced Arrhythmia: Pharmacogenomic Prescribing? Eur. Heart J. 2013, 34, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Shah, R.R. Drug-Induced QT Interval Prolongation: Does Ethnicity of the Thorough QT Study Population Matter? Br. J. Clin. Pharmacal. 2013, 75, 347–358. [Google Scholar] [CrossRef] [PubMed]
- Rautaharju, P.M.; Prineas, R.J.; Kadish, A.; Larson, J.C.; Hsia, J.; Lund, B. Normal Standards for QT and QT Subintervals Derived from a Large Ethnically Diverse Population of Women Aged 50 to 79 Years (the Women’s Health Initiative [WHI]). Am. J. Cardiol. 2006, 97, 730–737. [Google Scholar] [CrossRef] [PubMed]
- Fugate, T., II; Moss, A.J.; Jons, C.; McNitt, S.; Mullally, J.; Ouellet, G.; Goldenberg, I.; Zareba, W.; Robinson, J.L. Long QT Syndrome in African-Americans. Ann. Noninvasive Electrocardiol. 2010, 15, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Manini, A.F.; Stimmel, B.; Vlahov, D. Racial Susceptibility for QT Prolongation in Acute Drug Overdoses. J. Electrocardiol. 2014, 47, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Olatunde, A.; Price Evans, D.A. Blood Quinidine Levels and Cardiac Effects in White British and Nigerian Subjects. Br. J. Clin. Pharmacol. 1982, 14, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.-G.; Kang, W.; Shon, J.-H.; Arefayene, M.; Yoon, Y.-R.; Kim, K.-A.; Kim, D.-I.; Kim, D.-S.; Cho, K.-H.; Woosley, R.L.; et al. Possible Interethnic Differences in Quinidine-Induced QT Prolongation between Healthy Caucasian and Korean Subjects. Br. J. Clin. Pharmacol. 2007, 63, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues-Soares, F.; Peñas-Lledó, E.M.; Tarazona-Santos, E.; Sosa-Macías, M.; Terán, E.; López-López, M.; Rodeiro, I.; Moya, G.E.; Calzadilla, L.R.; Ramírez-Roa, R.; et al. Genomic Ancestry, CYP2D6, CYP2C9, and CYP2C19 Among Latin Americans. Clin. Pharmacol. Ther. 2020, 107, 257–268. [Google Scholar] [CrossRef]
- Aberg, K.; Adkins, D.E.; Liu, Y.; McClay, J.L.; Bukszár, J.; Jia, P.; Zhao, Z.; Perkins, D.; Stroup, T.S.; Lieberman, J.A.; et al. Genome-Wide Association Study of Antipsychotic-Induced QTc Interval Prolongation. Pharm. J. 2012, 12, 165–172. [Google Scholar] [CrossRef]
- Karczewski, K.J.; Francioli, L.C.; Tiao, G.; Cummings, B.B.; Alföldi, J.; Wang, Q.; Collins, R.L.; Laricchia, K.M.; Ganna, A.; Birnbaum, D.P.; et al. The Mutational Constraint Spectrum Quantified from Variation in 141,456 Humans. Nature 2020, 581, 434–443. [Google Scholar] [CrossRef] [PubMed]
| Gene (Subtype) † | Locus † | Protein † | Functional Effect † | Mode of Inheritance † | Frequency † | Level of Evidence ‡ |
|---|---|---|---|---|---|---|
| Long QT syndrome (major) | ||||||
| KCNQ1 (LQT1) | 11p15.5 | Kv7.1 | Reduced IKs | AD; AR | 30–35% | Definitive |
| KCNH2 (LQT2) | 7q35-46 | Kv11.1 | Reduced IKr | AD | 25–30% | Definitive |
| SCN5A (LQT3) | 3p21-p24 | Nav1.5 | Increased INa | AD | 5–10% | Definitive |
| Long QT syndrome (minor) | ||||||
| AKAP9 (AKAP9-LQTS) | 7q21-q22 | Yotiao | Reduced IKs | AD | <1% | Disputed |
| CACNA1C (CACNA1C-LQTS) | 12p13.3 | Cav1.2 | Increased ICa-L | AD | <1% | Moderate |
| CAV3 (CAV3-LQTS) | 3p25 | Caveolin 3 | Increased INa | AD | <1% | Limited |
| KCNE1 (KCNE1-LQTS) | 21q22.1 | MinK | Reduced IKs | AD | <1% | Limited |
| KCNE2 (KCNE2-LQTS) | 21q22.1 | MiRP1 | Reduced IKr | AD | <1% | Disputed |
| KCNJ5 (KCNJ5-LQTS) | 11q24 | Kir3.4 | Reduced IK,ACh | AD | <1% | Disputed |
| SCN4B (SCN4B-LQTS) | 11q23.3 | Nav1.5 β4-subunit | Increased INa | AD | <1% | Disputed |
| SNTA1 (SNTA-LQTS) | 20q11.2 | Syntrophin-α1 | Increased INa | AD | <1% | Disputed |
| Jervell and Lange-Nielsen syndrome | ||||||
| KCNQ1 (JLNS1) | 11p15.5 | Kv7.1 | Reduced IKs | AR | Very rare | NA |
| KCNE1 (JLNS2) | 21q22.1 | MinK | Reduced IKs | AR | Very rare | NA |
| Ankyrin-B syndrome | ||||||
| ANKB (ABS) | 4q25-q27 | Ankyrin B | Aberrant ion channel /transporter localization | AD | <1% | NA |
| Andersen-Tawil syndrome | ||||||
| KCNJ2 (ATS) | 17q23 | Kir2.1 | Reduced IK1 | AD | <1% | Definitive 1 |
| Timothy syndrome | ||||||
| CACNA1C (TS) | 12p13.3 | Cav1.2 | Increased ICa-L | Sporadic | Very rare | Definitive 2 |
| Recurrent infantile cardiac arrest syndrome | ||||||
| CALM1 | 14q24-q31 | Calmodulin 1 | Dysfunctional Ca2+ signaling | Sporadic | <1% | Definitive |
| CALM2 | 2p21 | Calmodulin 2 | Dysfunctional Ca2+ signaling | Sporadic | <1% | Definitive |
| Drug Class (with Known Risk for TdP) | Drug | Molecular Mechanism for Prolonged QT Interval | Comments | Reference |
|---|---|---|---|---|
| Anticancer | Aclarubicin | - | Acute cardiotoxicity and heart failure coupled with QT prolontation in case reports and case series | [68] |
| Arsenic trioxide | Inhibition of Kv11.1 trafficking | QT prolongation >500 ms (up to 40% of patients) | [35] | |
| Oxaliplatin | - | - | ||
| Vandetanib | IKr block | Incidence of QTc interval prolongation with 300 mg/day: 16.4% (all grades), 3.7% (high-grade) | [69] | |
| Antiarrhythmic | ||||
| Amiodarone | IKr block, binds to the closed state of the channel | Inactivation-removing mutations (S631A) reduce inhibitory effects on Kv11.1 channel | [70] | |
| Disopyramide | IKr block, INa, | Dose-dependent prolongation | [71] | |
| Dofetilide | IKr block, INa-L augmentation | High risk of ventricular arrythmias, requires inpatient monitoring for approximately 3 days Adjust dose in CKD | [35] | |
| Dronedarone | IKr block | - | - | |
| Flecainide | IKr block Inhibition of CYP3A4 | The risk of TdP correlates with the extent of prolongation of the QT interval. | [72] | |
| Hydroquinidine | IKr block | - | - | |
| Ibutilide | IKr block, INa-L augmentation | |||
| Nifekalant | - | - | - | |
| Procainamide | IKr block | |||
| Quinidine | IKr block, IK1, Ito inhibition | [71] | ||
| Sotalol | IKr block, INa-L augmentation | - | - | |
| Antihistamine | ||||
| Astemizole | IKr block | High mortality related to concomitant administration with azole compounds | [73] | |
| Terfenadine | IKr block | [73] | ||
| Antibiotic | ||||
| Azithromycin | IKr block, INa-L augmentation | |||
| Ciprofloxacin | IKr block | [73] | ||
| Clarithromycin | IKr block Inhibition of CYP3A4 | Greater risk of TdP at high doses and during IV administration | [73] | |
| Erythromycin | IKr block, INa-L augmentation Inhibition of CYP3A4 | Greater risk of TdP at high doses and during IV administration | [73] | |
| Grepafloxacin | ||||
| Gatifloxacin | IK block, hERG block | Should be avoided in patients with risk factors of QT prolongation | [73] | |
| Levofloxacin | IKr block, hERG block | [74] | ||
| Moxifloxacin | IKr block | Predictable 10–15 ms QTc prolongation | [1] | |
| Roxithromycin | IKr block | |||
| Sparfloxacin | Direct inhibition of hERG, IKr block | Potent compound blocking hERG and increases the instability of repolarization | [73] | |
| Antianginal | ||||
| Bepridil | - | - | - | |
| Antimalarial | ||||
| Chloroquine | IKr block, hERG block | Risk of death is proportional to drug exposure | [75] | |
| Halofantrine | IKr block | |||
| Hydroxychloroquine | IKr block, hERG block | Risk of TdP is dependent on dose | [76] | |
| Antipsychotic | ||||
| Chlorpromazine | IKr block, hERG block | Least potent blocking hERG K+ channels | [73] | |
| Chlorprothixene | ||||
| Droperidol | IKr block | Leads to transient increases in QT and clinically significant QT prolongation in 1–9% of patients with other risk factors for QT prolongation | [77] | |
| Haloperidol | IKr block | Despite the relatively mild QTc prolongation associated with the oral and IM forms of haloperidol, this medication has clearly been linked to TdP | [77] | |
| Levomepromazine | - | - | - | |
| Levosulpiride | - | - | - | |
| Mesoridazine | hERG block | Prolongs the QT interval in a dose-dependent manner | [78] | |
| Pimozide | IKr block | Clear association with TdP | [77] | |
| Sertindole | The frequency of QTc intervals of 500 ms or longer is 3.1% to 7.8% | [72] | ||
| Sulpiride | hERG block IKr block | Dose-dependent | [79] | |
| Sultopride | ||||
| Thioridazine | IKr block, INa-L augmentation | Most associated with QTc prolongation among phenothiazines | [77] | |
| Antidepressant | ||||
| Citalopram | IKr block, and Inhibition of Kv11.1 trafficking | Prolongs the QT on the order of 10-20 ms | [77] | |
| Escitalopram | IKr block, and Inhibition of Kv11.1 trafficking | May carry some risk of mild QT prolongation, but not enough to be clinically significant | [77] | |
| Anesthetic, general | ||||
| Propofol | IKr block, Ito block | - | - | |
| Sevoflurane | IKr block, Ito block | Mean increase in maximal rate-corrected QT prolongation of 46 ms while patients are exposed to clinically relevant concentrations of the inhaled agent | [80] | |
| Antiemetic | ||||
| Domperidone | IKr block, hERG block | Dose-dependent risk factor of LQTS | [81] | |
| Ondansetron | IKr block | 36 episodes of TdP were observed p < 0.02 | [82] | |
| Antifungal | ||||
| Fluconazole | IKr block, and Inhibition of Kv11.1 trafficking | Implicated in three cases of QTc-interval prolongation when given alone or in combination with other drugs | [74] | |
| Pentamidine | Inhibition of Kv11.1 trafficking | Prolongs the cardiac action potential by blocking hERG trafficking and reduction of the number of functional hERG channels at the cell surface | [83] | |
| Antiparasitic | ||||
| Meglumine antimoniate | - | - | - | |
| Antilipemic | ||||
| Probucol | Inhibition of hERG trafficking | [73] | ||
| Cholinesterase inhibitors | ||||
| Donepezil | IKr block, hERG block and trafficking inhibition | Long-term use of donepezil is associated with prolongation of the QT interval. | [84] | |
| GI stimulant | ||||
| Cisapride | hERG block | Risk of cisapride-related LQTS may be minimized by avoiding cofactors. | [73] | |
| Local anesthetic | ||||
| Cocaine | - | - | - | |
| Muscle relaxant | ||||
| Terodiline | - | - | - | |
| Opioid agonist | ||||
| Levomethadyl acetate | Higher potential to induce cardiac arrhythmias than does methadone in comparable dosages | [85] | ||
| Phosphodiesterase 3 inhibitor | ||||
| Anagrelide | - | - | - | |
| Cilostazol | - | - | - | |
| Psychedelic | ||||
| Ibogaine | hERG block, calcium channel inhibition | Besides QT prolongation, a powerful psychoactive drug such as ibogaine may also lead to adverse cardiac effects related to its central nervous activity | [86] | |
| Toxin | ||||
| Cesium chloride | - | - | - | |
| Vasodilator | ||||
| Papaverine HCl | - | - | - |
| System | Risk Factor |
|---|---|
| Autonomic nervous system | Pheochromocytoma Head-up-tilt Pure autonomic failure Sleep deprivation |
| Cardiovascular disease | Bradycardia Stress cardiomyopathy Stroke Aortic stenosis |
| Electrolyte disorders | Hypokalemia Hypomagnesemia Hypocalcemia Gitelman syndrome Blood transfusion |
| Endocrine disorders | Hypothyroidism Hyperparathyroidism |
| Environmental effects | Hypothermia Carbon monoxide Grapefruit juice Synthetic cannabinoids |
| General | Female sex, age > 65, |
| Inflammation/auto-immune | Rheumatoid arthritis Celiac disease Ankylosing spondylitis |
| Miscellaneous | Genotypic association Propionic academia Liquid protein diet Sickle cell disease |
| CYP Metabolizer | Drug with a Known TdP Risk | Type X Interactions When Combined with (High Risk of QT Prolongation) |
|---|---|---|
| CYP3A4 | Amiodarone | Voriconazol |
| Indinavir | ||
| Ritonavir | ||
| Saquinavir | ||
| Clarithromycin | ||
| Disopyramide | Itraconazol | |
| Clarithromycin | ||
| Ketoconazol (systemic) | ||
| Saquinavir | ||
| Telithromycin | ||
| Dofetilide | Clarithromycin | |
| Itraconazole | ||
| Ketoconazole | ||
| Dronedarone | Clarithromycin | |
| Ceritinib | ||
| Indinavir | ||
| Itraconazole | ||
| Nefazodone | ||
| Ritonavir | ||
| Saquinavir | ||
| Telithromycin | ||
| Voriconazole | ||
| Ibutilide | Clarithromycin | |
| Quinidine | Clarithromycin | |
| Itraconazol | ||
| Ketoconazol | ||
| Ritonavir | ||
| Saquinavir | ||
| Voriconazole | ||
| Azithromycin | Certinib | |
| Clarithromycin | ||
| Saquinavir | ||
| Voriconazole | ||
| Clarithromycin | Saquinavir | |
| Erythromycin | Saquinavir | |
| Cloroquine | Ceritinib | |
| Clarithromycin | ||
| Saquinavir | ||
| Voriconazole | ||
| Chlorpromazine | Certinib Saquinavir | |
| Voriconazol | ||
| Clarithromycin | ||
| Haloperidol | Saquinavir | |
| Levomepromazine | Ritonavir Saquinavir | |
| Escitalopram | - | |
| Ondansetron | - | |
| Cisapride | Ceritinib | |
| Clarithromycin | ||
| Indinavir | ||
| Itraconazole | ||
| Ketoconazole | ||
| Nefazodone | ||
| 2D6 | Flecainide | |
| Ibutilide | Quinidine | |
| Procainamide | Quinidine | |
| Chloroquine | - | |
| Hydroxychloroquine | - | |
| Chlorpromazine | - | |
| Haloperidol | - | |
| 2C19 | Citalopram | - |
| Escitalopram | - | |
| Chlorpromazine | - |
| Drug | Increased Risk in the Setting of | Use Causes Hypokalemia and/or Hypomagnesemia | Reduces Elimination of a QT/TdP Drug | Use in cLQTS | ||||
|---|---|---|---|---|---|---|---|---|
| Bradycardia | Hypokalemia/ Hypomagnesemia | Excesive Dose | Use with Concomitant QT/TdP Drug | Impaired Drug Elimination | ||||
| Abiraterone | X | |||||||
| Amantadine | X | X | X | X | ||||
| Amisulpride | X | X | X | |||||
| Amitriptyline | X | X | X | X | X | |||
| Amphotericin B | X | X | ||||||
| Amsacrine | X | |||||||
| Atazanavir | X | |||||||
| Bendroflumethiazide | X | X | ||||||
| Chloral hydrate | X | |||||||
| Cimetidine | X | X | X | X | ||||
| Clomipramine | X | X | X | |||||
| Diphenhydramine | X | |||||||
| Doxepin | X | X | X | X | ||||
| Eperisone | X | |||||||
| Esomeprazole | X | X | ||||||
| Famotidine | X | X | X | X | ||||
| Fluoxetine | X | X | X | X | X | |||
| Fluvoxamine | X | X | X | X | ||||
| Furosemide | X | X | ||||||
| Galantamine | X | X | X | X | ||||
| Garenoxacin | X | X | ||||||
| Hydrochlorothiazide | X | |||||||
| Hydroxyzine | X | X | X | X | ||||
| Indapamide | X | X | ||||||
| Itraconazole | X | X | ||||||
| Ivabradine | X | X | X | X | ||||
| Ketoconazole | X | X | X | |||||
| Lansoprazol | X | X | ||||||
| Loperamide | X | X | ||||||
| Metoclopramide | X | X | X | X | ||||
| Metolazone | X | X | X | X | ||||
| Metronidazole | X | X | ||||||
| Nelfinavir | X | X | X | |||||
| Olanzapine | X | X | ||||||
| Omeprazole | X | X | ||||||
| Pantoprazole | X | X | ||||||
| Paroxetine | X | |||||||
| Piperacilin/tazobactam | X | X | X | X | ||||
| Posaconazole | X | X | X | X | X | |||
| Propafenone | X | X | X | |||||
| Quetiapine | X | X | X | X | X | |||
| Quinine sulfate | X | X | X | |||||
| Ranolazine | X | X | X | X | ||||
| Risperidone | X | X | X | X | ||||
| Sertraline | X | X | X | X | ||||
| Solifenacin | X | X | X | |||||
| Telaprevir | X | X | ||||||
| Torsemide | X | X | ||||||
| Trazodone | X | X | X | X | ||||
| Voriconazole | X | X | X | |||||
| Ziprasidone | X | X | X | X | ||||
| Antibiotics | Cloroquine/Hydroxycloroquine Macrolides Quinolones |
| Antiviral agents | Lopinavir/Ritonavir Favipiravir Tocilizumab Fingolimod |
| Anesthetics | Propofol |
| Antiemetics | Domperidone |
| Antiarrythmics | Class IA Class III |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baracaldo-Santamaría, D.; Llinás-Caballero, K.; Corso-Ramirez, J.M.; Restrepo, C.M.; Dominguez-Dominguez, C.A.; Fonseca-Mendoza, D.J.; Calderon-Ospina, C.A. Genetic and Molecular Aspects of Drug-Induced QT Interval Prolongation. Int. J. Mol. Sci. 2021, 22, 8090. https://doi.org/10.3390/ijms22158090
Baracaldo-Santamaría D, Llinás-Caballero K, Corso-Ramirez JM, Restrepo CM, Dominguez-Dominguez CA, Fonseca-Mendoza DJ, Calderon-Ospina CA. Genetic and Molecular Aspects of Drug-Induced QT Interval Prolongation. International Journal of Molecular Sciences. 2021; 22(15):8090. https://doi.org/10.3390/ijms22158090
Chicago/Turabian StyleBaracaldo-Santamaría, Daniela, Kevin Llinás-Caballero, Julián Miguel Corso-Ramirez, Carlos Martín Restrepo, Camilo Alberto Dominguez-Dominguez, Dora Janeth Fonseca-Mendoza, and Carlos Alberto Calderon-Ospina. 2021. "Genetic and Molecular Aspects of Drug-Induced QT Interval Prolongation" International Journal of Molecular Sciences 22, no. 15: 8090. https://doi.org/10.3390/ijms22158090
APA StyleBaracaldo-Santamaría, D., Llinás-Caballero, K., Corso-Ramirez, J. M., Restrepo, C. M., Dominguez-Dominguez, C. A., Fonseca-Mendoza, D. J., & Calderon-Ospina, C. A. (2021). Genetic and Molecular Aspects of Drug-Induced QT Interval Prolongation. International Journal of Molecular Sciences, 22(15), 8090. https://doi.org/10.3390/ijms22158090







