Carbon Monoxide and Nitric Oxide as Examples of the Youngest Class of Transmitters
Abstract
1. Introduction
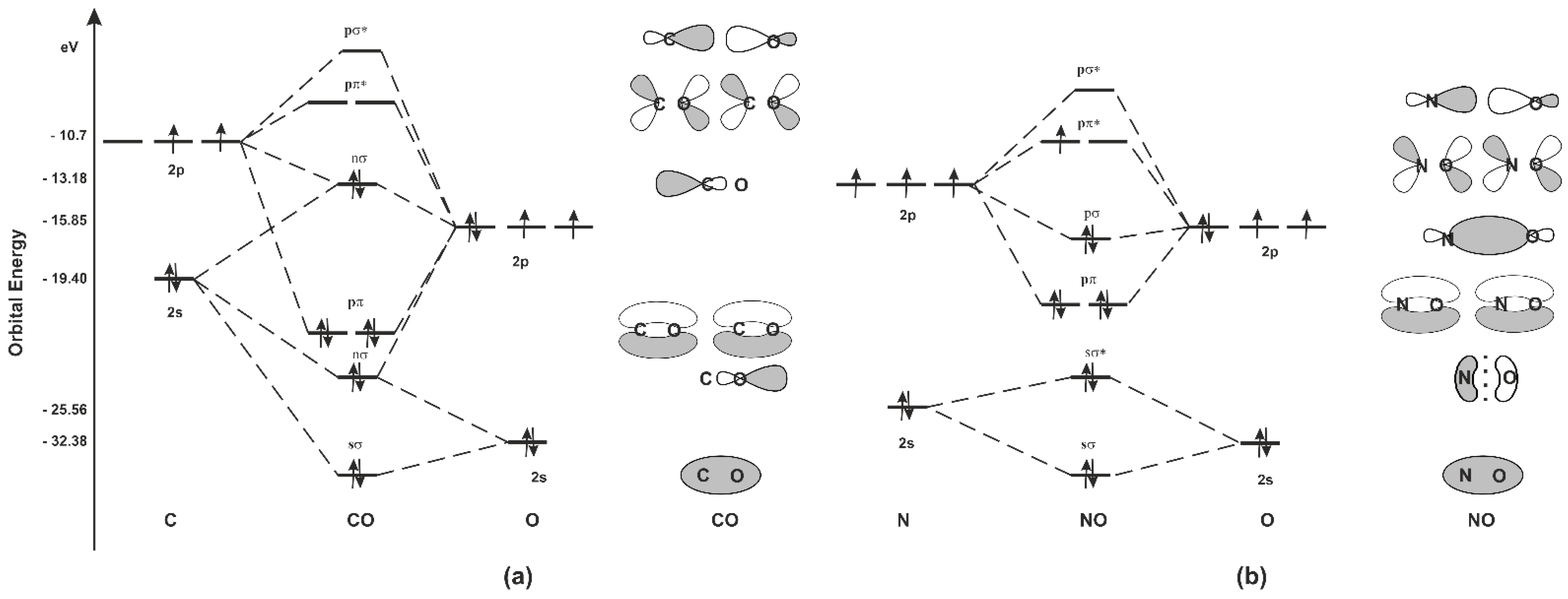
2. CO and NO—Chemistry
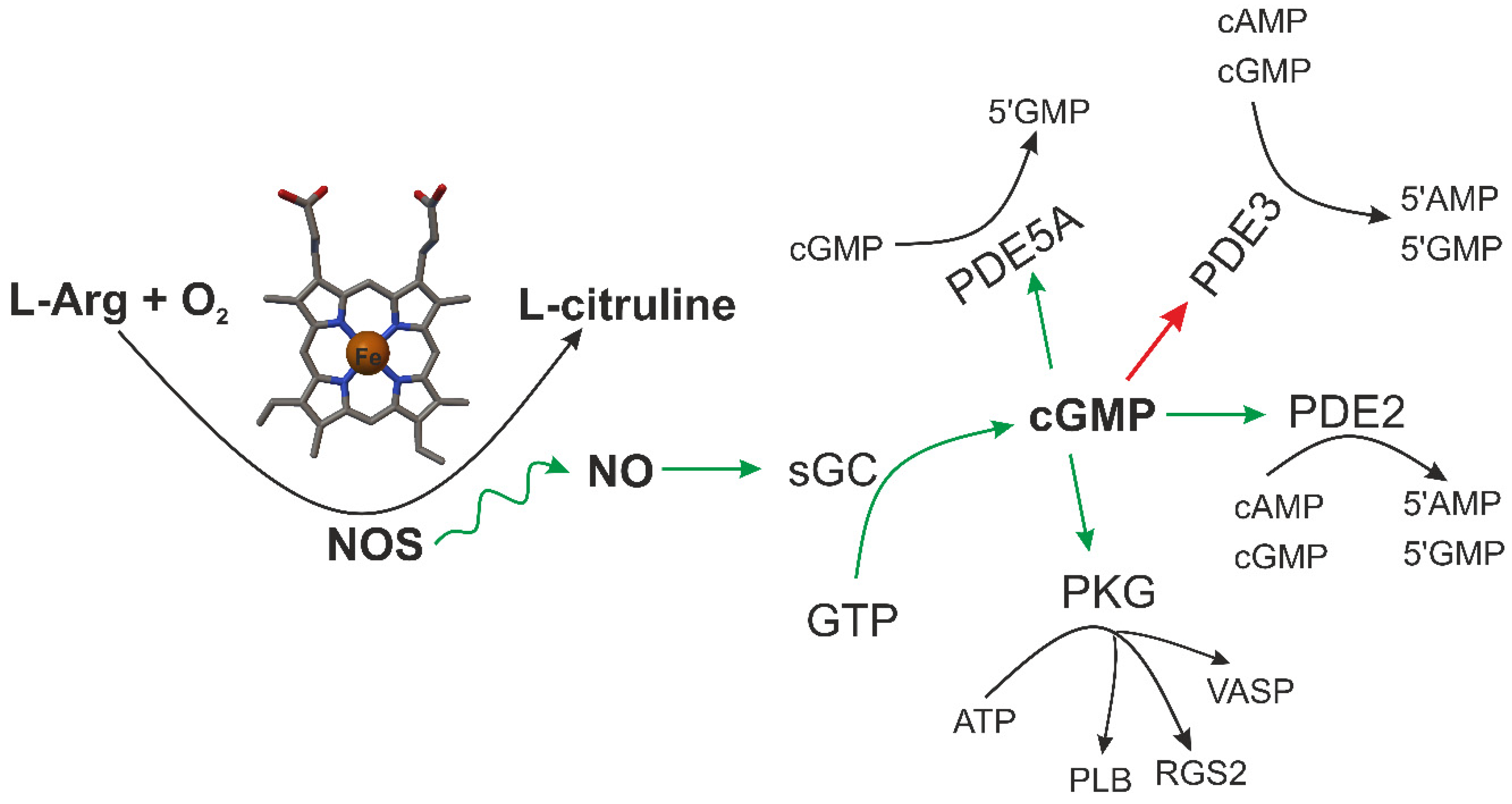
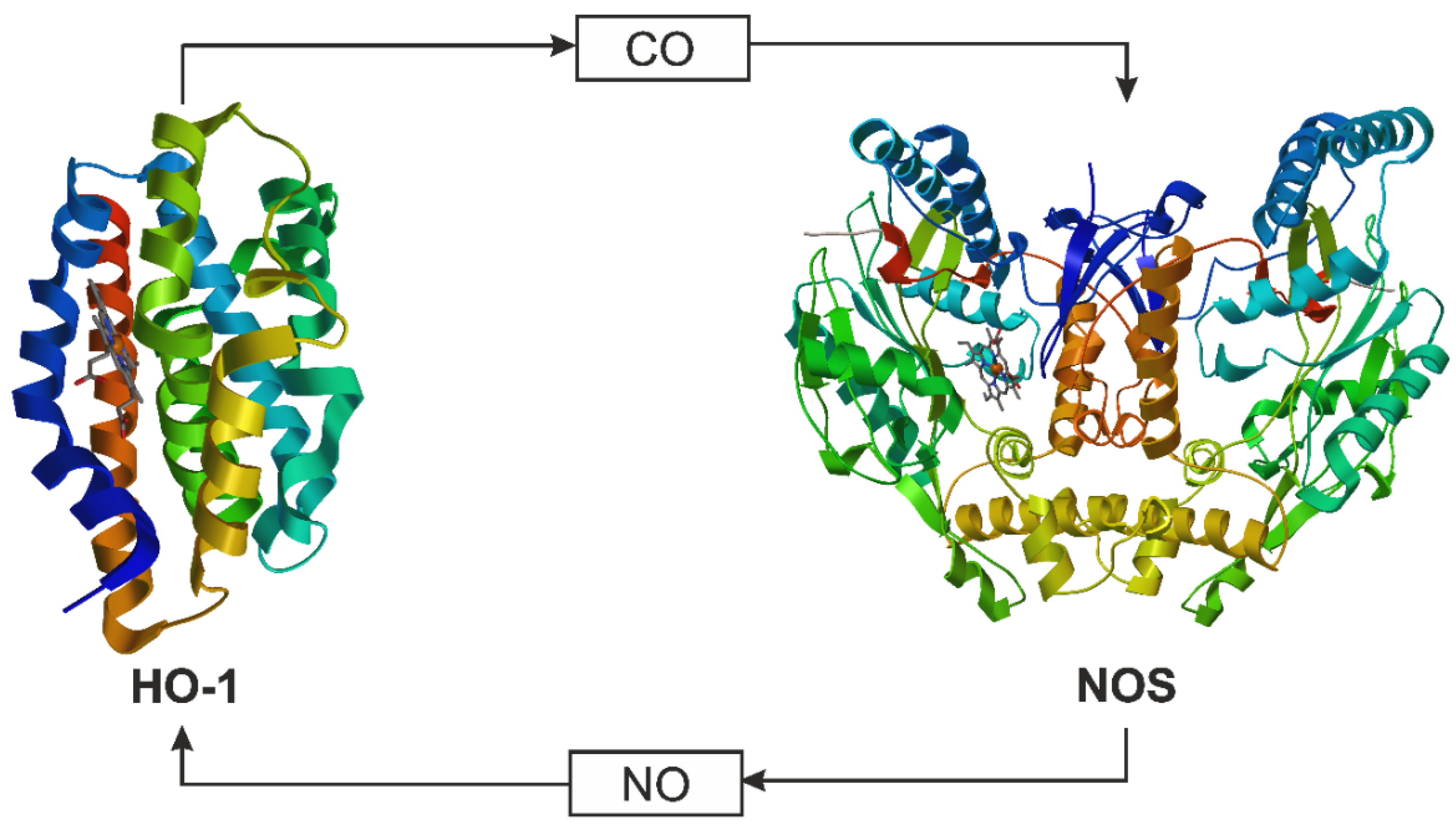
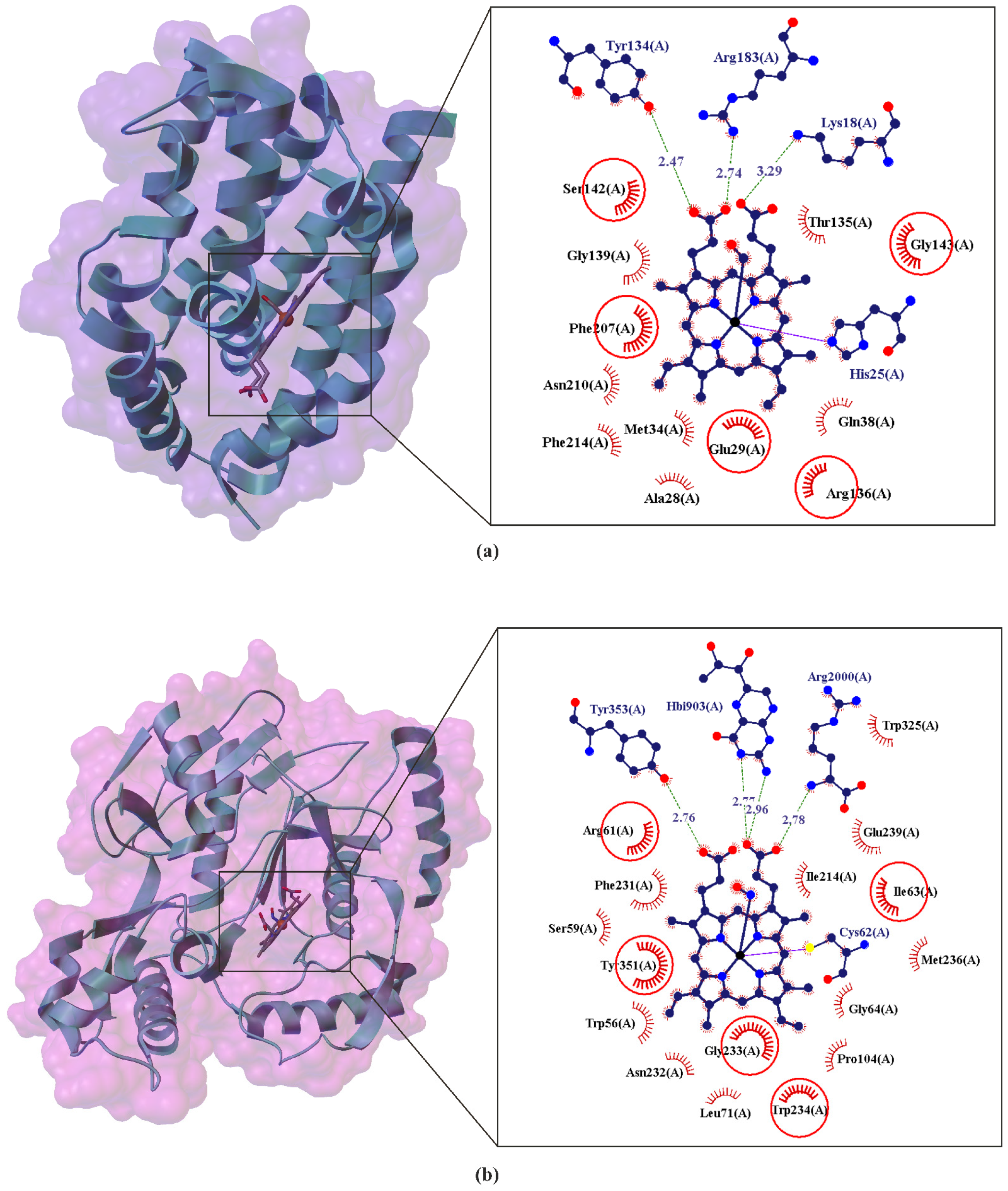
3. CO and NO—Endogenous Production
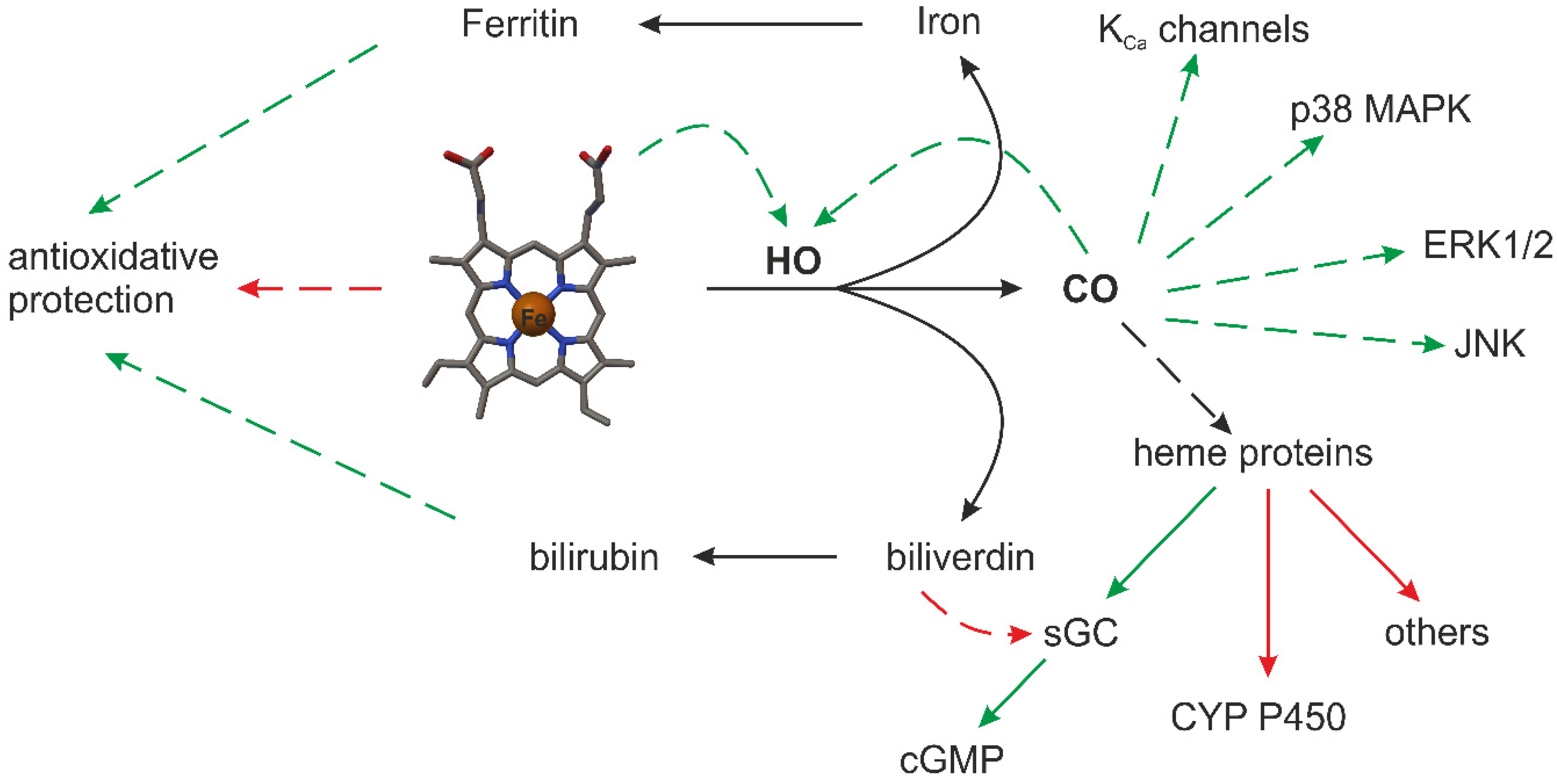
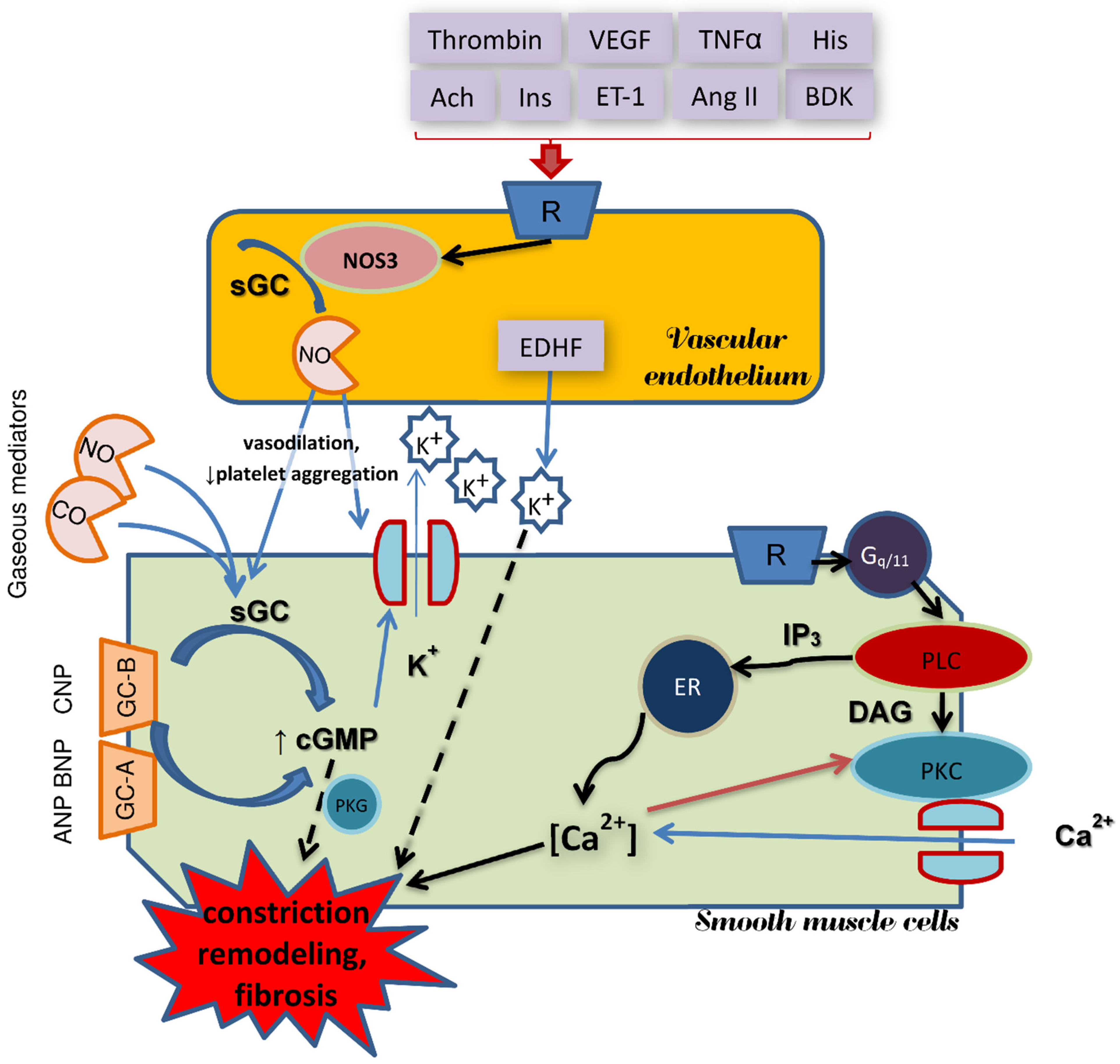
4. CO and NO—Biological Properties
| Biological Property | Carbon Monoxide | Nitric Oxide |
|---|---|---|
| Main substrates | Heme [4] | l-arginine [4] |
| Generating enzymes | Heme oxygenases [80] | NO synthases [80] |
| Inducer | Free radicals [4] | Acetylcholine, endotoxin |
| Inhibitor | zinc protoporphyrin-IX [4] | NG-nitro-l-arginine methyl ester |
| air to lung partition coefficient (log Plung) | −1.69 [81] | |
| blood to lung partition coefficient (log Kblood) | −1.69 [81] | |
| Protein targets | cGMP, KCa channel [4] | cGMP, KCa channel |
| Amino acid targets | histidine [4] | cysteine |
| Production tissue source | endothelial cell < smooth muscle cell [4] | endothelial cell > smooth muscle cell |
| Production rate in the human body | 16.4 μmol/h, 500 μmol/daily [82] | between 0.15 and 2.2 μmol/h, between 4 and 53 μmol/daily [83] |
| Half-life in solution | Minutes [4] | <1 s [83] |
| Lethal concentration | 1000 ppm [84] | |
| Metabolism | None [33] | Rapid conversion to nitrite/nitrate [33] |
| Preclinical efficacy | ||
| Pulmonary hypertension | 250 ppm for 1 h/d has long-term efficacy [85] | 20–80 ppm has rapid efficacy [66,86] |
| Sepsis/acute respiratory distress syndrome | 250 ppm for 4 h promotes bacteria clearance and decreases inflammation [86] | 0.2–20 ppm for 4 h decreases pulmonary hypertension but has no effect on inflammation [87] |
| Myocardial ischemia | 250–1000 ppm for 24 h prevents ischemia reperfusion injury [12] | 80 ppm for 60 min prevents ischemia reperfusion injury [88] |
| pharmacological application | antiinflammatory; protection from tissue reperfusion injury; vasodilatation; antiapoptotic; anti-proliferative; anti-hypoxia; antiaggregatory; anti-ischaemi; [89,90] | antiinflammatory; antiatherogenic; antihyperglycemic; antioxidative; antihypertensive; antimicrobial; antinociception, anticancer; gastric cytoprotection; antithrombogenicity; [74,91] |
5. CO and NO—Gaseous Neurotransmitters
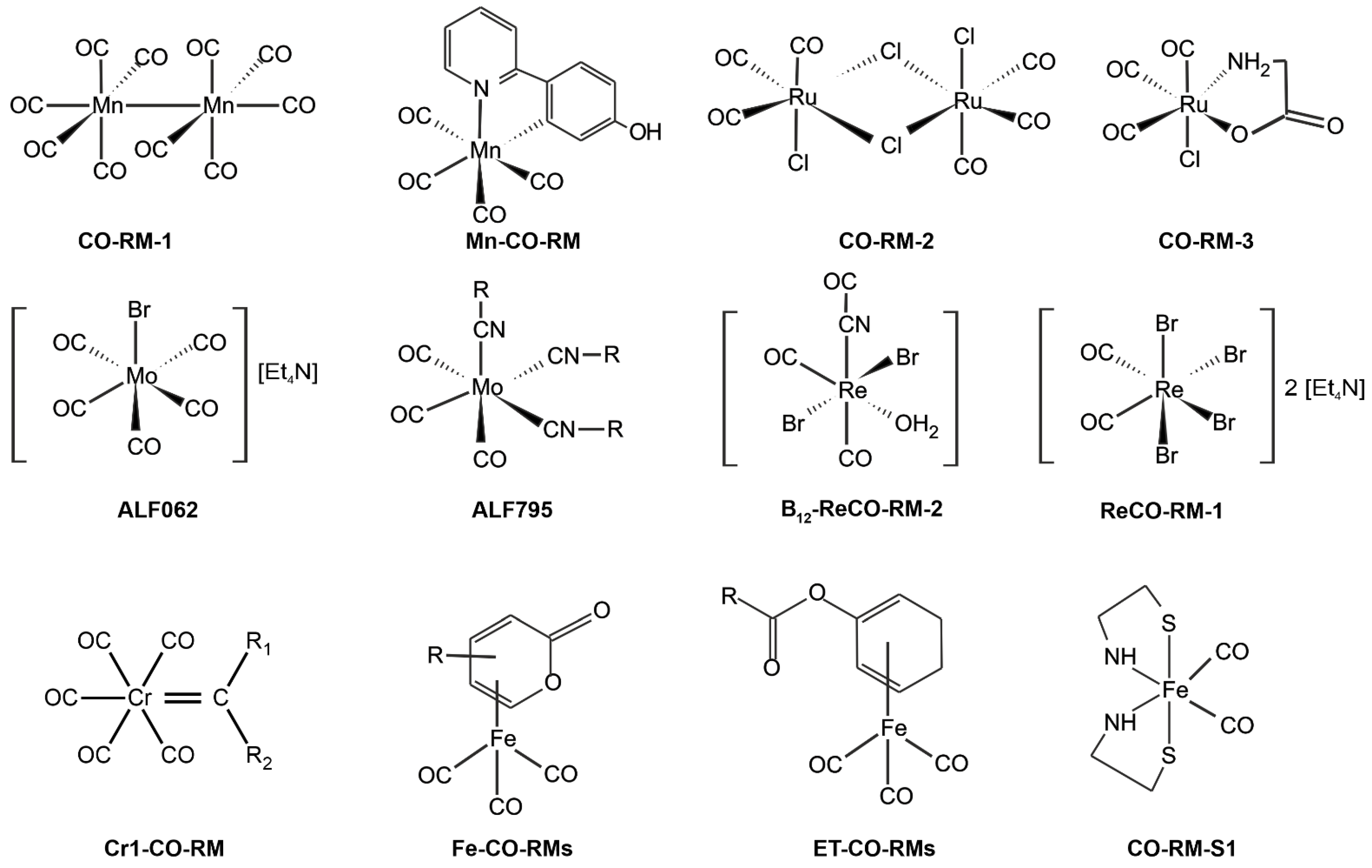
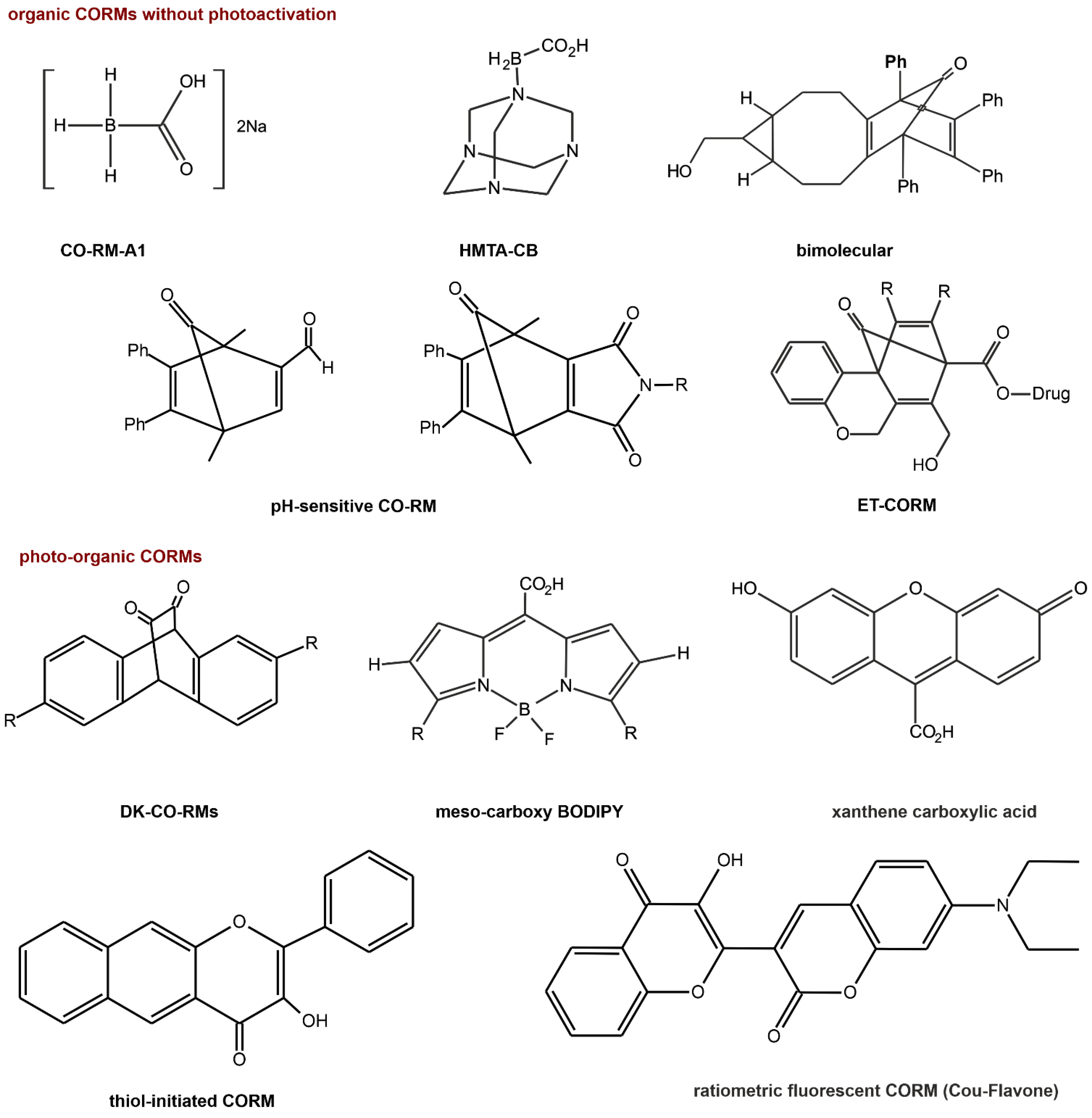
6. CO and NO as Potential Therapeutic Agent
6.1. CO as a Prodrug
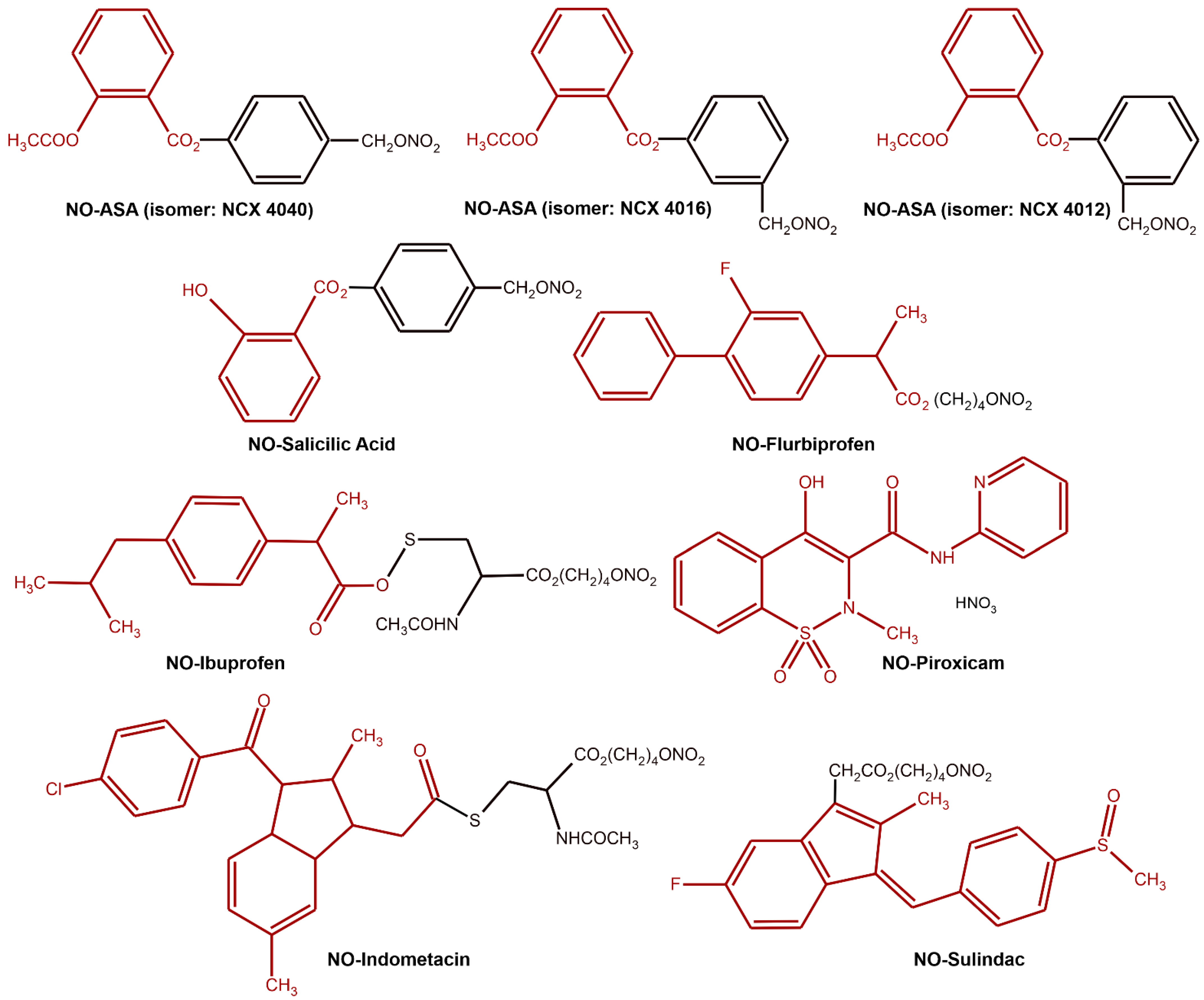
6.2. NO as a Prodrug
7. Conclusion and Perspective
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Ach | acetylcholine |
| Akt | protein kinase B |
| Ang II | angiotensin II |
| ASA | aspirin |
| ATC | Anatomical Therapeutic Chemical Classification System |
| BDK | bradykinin |
| cAMP | cyclic adenosine monophosphate |
| Ca2+ | calcium ions |
| CH3CHO | acetaldehyde |
| CHEBI | Chemical Entities of Biological Interest |
| CO | carbon monoxide |
| CORM | CO releasing molecule |
| COHb | carboxyhemoglobin |
| COX-2 | cyclooxygenase 2 |
| DAG | diacylglycerol |
| EDRF | endothelium-derived relaxing factor |
| EDHF | Endothelium-derived hyperpolarizing factor |
| ET-1 | Endothelin type 1 |
| EMA | European Medicines Agency |
| ER | endoplasmic reticulum |
| ERK | extracellular signal-regulated kinases |
| ERK1/2 | extracellular signal-regulated kinases 1/2 |
| FDA | US Food and Drug Administration |
| FhlA | formate hydrogenlyase transcriptional activator |
| FAD | flavin adenine dinucleotide |
| Gq/11 | Gq alpha subunit family |
| G | G protein type Gq/11 |
| GAF | small-molecule-binding domains of cGMP, cAMP, FhlA |
| GC | guanylate cyclase |
| GC-A | membrane guanylate cyclase type A |
| GC-B | membrane guanylate cyclase type B |
| sGC | soluble guanylate cyclase |
| GMP | guanosine monophosphate |
| cGMP | cyclic guanosine monophosphate |
| GTP | guanosine triphosphate |
| H2S | hydrogen sulfide |
| Hb | hemoglobin |
| His | histamine |
| HMBD | Human Metabolome Database |
| HO-x | heme oxygenase type x |
| HOMO | highest occupied molecular orbital |
| HPS | heat shock protein |
| ICAM-1 | intercellular adhesion molecule 1 |
| Ins | insulin |
| IL-x | interleukin type x |
| IFN-γ | interferons type II |
| IP3 | inositol 1,4,5-trisphosphate |
| ISMN, ISDN | isosorbide mono- and dinitrate |
| JNK | c-Jun N-terminal kinase |
| K+ | potassium ions |
| KCa | large-conductance potassium channels |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| L-Arg | L-arginine |
| LPS | lipopolysaccharide/endotoxin |
| LUMO | lowest unoccupied molecular orbital |
| M | metal |
| metHb | methemoglobin |
| ANP | natriuretic peptide type A |
| BNP | natriuretic peptide type B |
| CNP | natriuretic peptide type C |
| N2O | dinitrogen oxide |
| NF-κB | nuclear factor kappa-light-chain-enhancer of activated B cells |
| NADPH | nicotinamide adenine dinucleotide phosphate |
| NH3 | ammonia |
| NO | nitric oxide |
| NO-NSAIDs | nitric oxide donating non-steroidal antiinflammatory drugs |
| NSAIDs | non-steroidal antiinflammatory drugs |
| NOS | nitric oxide synthase |
| NOS1 | neuronal nitric oxide synthase |
| NOS2 | cytokine-inducible nitric oxide synthase |
| NOS3 | endothelial nitric oxide synthase |
| NTG | glyceryl trinitrate |
| mTOR | mammalian target of rapamycin |
| p38 MAPK | p38 mitogen activated protein kinases |
| PDEx | phosphodiesterase type x |
| PDB ID | Protein Data Bank Identifier |
| PGE-2 | prostaglandin-2 |
| PKC | protein kinase type C |
| PKG | cGMP dependent protein kinase G |
| PLB | phospholamban |
| PLC | phospholipase type C |
| R | G protein-coupled receptor |
| Rb | retinoblastoma |
| RANKL | the receptor activator of nuclear factor κB ligand |
| RGS2 | regulator of G protein signaling 2 |
| SO2 | sulfur dioxide |
| SOMO | single occupied molecular orbital |
| SNP | sodium nitroprusside |
| BH4 | tetrahydrobiopterin |
| TNFα | tumor necrosis factor α |
| VASP | vasodilatory-stimulated phosphoprotein |
| VEGF | Vascular endothelial growth factor |
References
- Valenstein, E.S. The discovery of chemical neurotransmitters. Brain Cogn. 2002, 49, 73–95. [Google Scholar] [CrossRef]
- McCoy, A.N.; Tan, Y.S. Otto loewi (1873–1961): Dreamer and nobel laureate. Singap. Med. J. 2014, 55, 3. [Google Scholar] [CrossRef]
- Garthwaite, J. Nitric oxide as a multimodal brain transmitter. Brain Neurosci. Adv. 2018, 2. [Google Scholar] [CrossRef] [PubMed]
- Wang, R. Two’s company, three’s a crowd: Can H2S be the third endogenous gaseous transmitter? Faseb J. 2002, 16, 1792–1798. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.ebi.ac.uk/chebi/searchId.do?chebiId=CHEBI:16480 (accessed on 26 April 2021).
- Ignarro, L.J. After 130 years, the molecular mechanism of action of nitroglycerin is revealed. Proc. Nat. Acad. Sci. USA 2002, 99, 7816–7817. [Google Scholar] [CrossRef]
- Picón-Pagès, P.; Garcia-Buendia, J.; Muñoz, F.J. Functions and dysfunctions of nitric oxide in brain. BBA-Mol. Basis Dis. 2019, 1865, 1949–1967. [Google Scholar] [CrossRef] [PubMed]
- Cinelli, M.A.; Do, H.T.; Miley, G.P.; Silverman, R.B. Inducible nitric oxide synthase: Regulation, structure, and inhibition. Med. Res. Rev. 2020, 40, 158–189. [Google Scholar] [CrossRef] [PubMed]
- Lisi, F.; Zelikin, A.N.; Chandrawati, R. Nitric Oxide to Fight Viral Infections. Adv. Sci. 2021, 2003895. [Google Scholar] [CrossRef] [PubMed]
- Fagone, P.; Mazzon, E.; Bramanti, P.; Bendtzen, K.; Nicoletti, F. Gasotransmitters and the immune system: Mode of action and novel therapeutic targets. Eur. J. Pharmacol. 2018, 834, 92–102. [Google Scholar] [CrossRef]
- Mukherjee, P.; Cinelli, M.A.; Kang, S.; Silverman, R.B. Development of nitric oxide synthase inhibitors for neurodegeneration and neuropathic pain. Chem. Soc. Rev. 2014, 43, 6814–6838. [Google Scholar] [CrossRef]
- Fujimoto, H.; Ohno, M.; Ayabe, S.; Kobayashi, H.; Ishizaka, N.; Kimura, H.; Yoshida, K.-I.; Nagai, R. Carbon monoxide protects against cardiac ischemia—reperfusion injury in vivo via MAPK and Akt—eNOS pathways. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1848–1853. [Google Scholar] [CrossRef]
- Available online: https://www.ebi.ac.uk/chebi/searchId.do?chebiId=CHEBI:17245 (accessed on 26 April 2021).
- Available online: https://spectrabase.com/spectrum/KlmI6BZQQsa (accessed on 26 April 2021).
- Available online: https://spectrabase.com/spectrum/GeuwXWi3T6m (accessed on 26 April 2021).
- Keller-Rudek, H.; Moortgat, G.; Sander, R.; Sörensen, R. The MPI-Mainz UV/VIS spectral atlas of gaseous molecules of atmospheric interest. Earth Syst. Sci. Data 2013, 5, 365–373. [Google Scholar] [CrossRef]
- Available online: http://satellite.mpic.de/spectral_atlas/cross_sections/Carbon-oxides/CO.spc (accessed on 26 April 2021).
- Available online: http://satellite.mpic.de/spectral_atlas/cross_sections/Nitrogen%20oxides/NO.spc (accessed on 26 April 2021).
- Available online: https://webbook.nist.gov/cgi/cbook.cgi?ID=C630080&Mask=200#Mass-Spec (accessed on 26 April 2021).
- Available online: https://webbook.nist.gov/cgi/cbook.cgi?ID=C10102439&Mask=200#Mass-Spec (accessed on 26 April 2021).
- Romão, C.C.; Blättler, W.A.; Seixas, J.D.; Bernardes, G.J. Developing drug molecules for therapy with carbon monoxide. Chem. Soc. Rev. 2012, 41, 3571–3583. [Google Scholar] [CrossRef]
- Tadeusiewicz, J.; Olas, B. Tlenek azotu i tlenek węgla-dwa ważne gazotransmitery. Kosmos 2014, 63, 543–554. [Google Scholar]
- Motterlini, R.; Mann, B.E.; Johnson, T.R.; Clark, J.E.; Foresti, R.; Green, C.J. Bioactivity and pharmacological actions of carbon monoxide-releasing molecules. Curr. Pharm. Des. 2003, 9, 2525–2539. [Google Scholar] [CrossRef]
- Gell, D.A. Structure and function of haemoglobins. Blood Cells Mol. Dis. 2018, 70, 13–42. [Google Scholar] [CrossRef] [PubMed]
- Toledo, J.J.C.; Ohara, A. Connecting the Chemical and Biological Properties of Nitric Oxide. Chem. Res. Toxicol. 2012, 25, 975–989. [Google Scholar] [CrossRef] [PubMed]
- McCleverty, J. Chemistry of nitric oxide relevant to biology. Chem. Rev. 2004, 104, 403–418. [Google Scholar] [CrossRef]
- Wolak, M.; van Eldik, R. To be or not to be NO in coordination chemistry? A mechanistic approach. Coordin. Chem. Rev. 2002, 230, 263–282. [Google Scholar] [CrossRef]
- Ford, P.C.; Laverman, L.E. Reaction mechanisms relevant to the formation of iron and ruthenium nitric oxide complexes. Coordin. Chem. Rev. 2005, 249, 391–403. [Google Scholar] [CrossRef]
- Hsia, C.C. Respiratory function of hemoglobin. N. Engl. J. Med. 1998, 338, 239–248. [Google Scholar] [CrossRef]
- Piantadosi, C.A. Biological chemistry of carbon monoxide. Antioxid. Redox Signal. 2002, 4, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Allen, T.A.; Root, W.S. Partition of carbon monoxide and oxygen between air and whole blood of rats, dogs and men as affected by plasma pH. J. Appl. Physiol. 1957, 10, 186–190. [Google Scholar] [CrossRef]
- Wu, L.; Wang, R. Carbon monoxide: Endogenous production, physiological functions, and pharmacological applications. Pharmacol. Rev. 2005, 57, 585–630. [Google Scholar] [CrossRef]
- Motterlini, R.; Otterbein, L.E. The therapeutic potential of carbon monoxide. Nat. Rev. Drug Discov. 2010, 9, 728–743. [Google Scholar] [CrossRef] [PubMed]
- Otterbein, L.E. The evolution of carbon monoxide into medicine. Resp. Care 2009, 54, 925–932. [Google Scholar] [CrossRef]
- Mayer, S. Estimation of activation energies for nitrous oxide, carbon dioxide, nitrogen dioxide, nitric oxide, oxygen, and nitrogen reactions by a bond-energy method. J. Phys. Chem. 1969, 73, 3941–3946. [Google Scholar] [CrossRef]
- Shriver, D. Activation of Carbon Monoxide by Carbon and Oxygen Coordination: Lewis Acid and Proton Induced Reduction of Carbon Monoxide. In Catalytic Activation of Carbon Monoxide; Ford, P.C., Ed.; ACS: Washington, DC, USA, 1981; Volume 152, pp. 1–18. [Google Scholar] [CrossRef]
- Gadzhiev, O.B.; Ignatov, S.K.; Gangopadhyay, S.; Masunov, A.E.; Petrov, A.I. Mechanism of nitric oxide oxidation reaction (2NO + O2→ 2NO2) revisited. J. Chem. Theory Comput. 2011, 7, 2021–2024. [Google Scholar] [CrossRef] [PubMed]
- Found, C.G. Ionization potentials of argon, nitrogen, carbon monoxide, helium, hydrogen and mercury and iodine vapors. Phys. Rev. 1920, 16, 41–53. [Google Scholar] [CrossRef]
- Tejero, J.; Hunt, A.P.; Santolini, J.; Lehnert, N.; Stuehr, D.J. Mechanism and regulation of ferrous heme-nitric oxide (NO) oxidation in NO synthases. J. Biol. Chem. 2019, 294, 7904–7916. [Google Scholar] [CrossRef]
- Available online: https://www.genome.jp/dbget-bin/www_bget?ec:1.14.13.39 (accessed on 26 April 2021).
- Leone, A.; Gustafsson, L.; Francis, P.; Persson, M.; Wiklund, N.; Moncada, S. Nitric oxide is present in exhaled breath in humans: Direct GC-MS confirmation. Biochem. Bioph. Res. Commun. 1994, 201, 883–887. [Google Scholar] [CrossRef] [PubMed]
- Chachlaki, K.; Garthwaite, J.; Prevot, V. The gentle art of saying NO: How nitric oxide gets things done in the hypothalamus. Nat. Rev. Endocrinol. 2017, 13, 1–15. [Google Scholar] [CrossRef]
- O’Connor, D.M.; O’Brien, T. Nitric oxide synthase gene therapy: Progress and prospects. Expert Opin. Biol. Ther. 2009, 9, 867–878. [Google Scholar] [CrossRef]
- Alderton, W.K.; Cooper, C.E.; Knowles, R.G. Nitric oxide synthases: Structure, function and inhibition. Biochem. J. 2001, 357, 593–615. [Google Scholar] [CrossRef]
- Yang, C.; Jeong, S.; Ku, S.; Lee, K.; Park, M.H. Use of gasotransmitters for the controlled release of polymer-based nitric oxide carriers in medical applications. J. Control. Release 2018, 279, 157–170. [Google Scholar] [CrossRef] [PubMed]
- Tsai, E.J.; Kass, D.A. Cyclic GMP signaling in cardiovascular pathophysiology and therapeutics. Pharmacol. Therapeut. 2009, 122, 216–238. [Google Scholar] [CrossRef] [PubMed]
- Feil, R.; Kemp-Harper, B. cGMP signalling: From bench to bedside: Conference on cGMP generators, effectors and therapeutic implications. EMBO Rep. 2006, 7, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Martinez, S.E.; Bruder, S.; Schultz, A.; Zheng, N.; Schultz, J.E.; Beavo, J.A.; Linder, J.U. Crystal structure of the tandem GAF domains from a cyanobacterial adenylyl cyclase: Modes of ligand binding and dimerization. Proc. Nat. Acad. Sci. USA 2005, 102, 3082–3087. [Google Scholar] [CrossRef]
- Available online: https://www.genome.jp/dbget-bin/www_bget?ec:1.14.99.3 (accessed on 26 April 2021).
- Motterlini, R.; Foresti, R. Biological signaling by carbon monoxide and carbon monoxide-releasing molecules. Am. J. Physiol. Cell Physiol. 2017, 312, C302–C313. [Google Scholar] [CrossRef]
- Abraham, N.G.; Kappas, A. Pharmacological and clinical aspects of heme oxygenase. Pharmacol. Rev. 2008, 60, 79–127. [Google Scholar] [CrossRef] [PubMed]
- Fischmann, T.O.; Hruza, A.; Da Niu, X.; Fossetta, J.D.; Lunn, C.A.; Dolphin, E.; Prongay, A.J.; Reichert, P.; Lundell, D.J.; Narula, S.K. Structural characterization of nitric oxide synthase isoforms reveals striking active-site conservation. Nat. Struct. Biol. 1999, 6, 233–242. [Google Scholar] [CrossRef]
- Brunet, L.R. Nitric oxide in parasitic infections. Int. Immunopharmacol. 2001, 1, 1457–1467. [Google Scholar] [CrossRef]
- Bruckdorfer, R. The basics about nitric oxide. Mol. Asp. Med. 2005, 26, 3–31. [Google Scholar] [CrossRef] [PubMed]
- Michel, T.; Feron, O. Nitric oxide synthases: Which, where, how, and why? J. Clin. Investig. 1997, 100, 2146–2152. [Google Scholar] [CrossRef] [PubMed]
- Förstermann, U.; Li, H. Nitric Oxide: Biological Synthesis and Functions. In Gasotransmitters: Physiology and Pathophysiology; Hermann, A., Sitdikova, G.F., Weiger, T.M., Eds.; Springer: Heidelberg, Germany, 2012; pp. 1–36. [Google Scholar] [CrossRef]
- Calabrese, V.; Butterfield, D.A.; Scapagnini, G.; Stella, A.G.; Maines, M.D. Redox regulation of heat shock protein expression by signaling involving nitric oxide and carbon monoxide: Relevance to brain aging, neurodegenerative disorders, and longevity. Antioxid. Redox Sign. 2006, 8, 444–477. [Google Scholar] [CrossRef] [PubMed]
- Sugishima, M.; Moffat, K.; Noguchi, M. Discrimination between CO and O2 in heme oxygenase: Comparison of static structures and dynamic conformation changes following CO photolysis. Biochemistry 2012, 51, 8554–8562. [Google Scholar] [CrossRef]
- Available online: https://hmdb.ca/ (accessed on 26 April 2021).
- Available online: https://hmdb.ca/metabolites/HMDB0001361 (accessed on 26 April 2021).
- Available online: https://hmdb.ca/metabolites/HMDB0003378 (accessed on 26 April 2021).
- Available online: https://www.genome.jp/kegg/ (accessed on 26 April 2021).
- Available online: https://www.genome.jp/dbget-bin/www_bget?cpd:C00237 (accessed on 26 April 2021).
- Available online: https://www.genome.jp/dbget-bin/www_bget?C00533 (accessed on 26 April 2021).
- West, J.B. Joseph Priestley, oxygen, and the enlightenment. Am. J. Physiol. Lung C. 2014, 306, L111–L119. [Google Scholar] [CrossRef]
- Li, H.; Igarashi, J.; Jamal, J.; Yang, W.; Poulos, T.L. Structural studies of constitutive nitric oxide synthases with diatomic ligands bound. J. Biol. Inorg. Chem. 2006, 11, 753–768. [Google Scholar] [CrossRef]
- Frostell, C.; Fratacci, M.; Wain, J.; Jones, R.; Zapol, W. Inhaled nitric oxide. A selective pulmonary vasodilator reversing hypoxic pulmonary vasoconstriction. Circulation 1991, 83, 2038–2047. [Google Scholar] [CrossRef]
- Ford, P.C. Reactions of NO and nitrite with heme models and proteins. Inorg. Chem. 2010, 49, 6226–6239. [Google Scholar] [CrossRef]
- Flores-Santana, W.; Switzer, C.; Ridnour, L.A.; Basudhar, D.; Mancardi, D.; Donzelli, S.; Thomas, D.D.; Miranda, K.M.; Fukuto, J.M.; Wink, D.A. Comparing the chemical biology of NO and HNO. Arch. Pharm. Res. 2009, 32, 1139–1153. [Google Scholar] [CrossRef]
- Wang, P.G.; Xian, M.; Tang, X.; Wu, X.; Wen, Z.; Cai, T.; Janczuk, A.J. Nitric oxide donors: Chemical activities and biological applications. Chem. Rev. 2002, 102, 1091–1134. [Google Scholar] [CrossRef] [PubMed]
- Ignarro, L.J. Endothelium-derived nitric oxide: Pharmacology and relationship to the actions of organic nitrate esters. Pharm. Res. 1989, 6, 651–659. [Google Scholar] [CrossRef]
- Radomski, M.W.; Palmer, R.; Moncada, S. The anti-aggregating properties of vascular endothelium: Interactions between prostacyclin and nitric oxide. Brit. J. Pharmacol. 1987, 92, 639–646. [Google Scholar] [CrossRef] [PubMed]
- Marsh, N.; Marsh, A. A short history of nitroglycerine and nitric oxide in pharmacology and physiology. Clin. Exp. Pharmacol. Physiol. 2000, 27, 313–319. [Google Scholar] [CrossRef]
- Levine, A.B.; Punihaole, D.; Levine, T.B. Characterization of the role of nitric oxide and its clinical applications. Cardiology 2012, 122, 55–68. [Google Scholar] [CrossRef]
- Parent, M.; Boudier, A.; Fries, I.; Gostyńska, A.; Rychter, M.; Lulek, J.; Leroy, P.; Gaucher, C. Nitric oxide-eluting scaffolds and their interaction with smooth muscle cells in vitro. J. Biomed. Mater. Res. A 2015, 103, 3303–3311. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Fu, J.; Zhang, Y. Nitric oxide donor-based cancer therapy: Advances and prospects. J. Med. Chem. 2017, 60, 7617–7635. [Google Scholar] [CrossRef] [PubMed]
- Basudhar, D.; Ridnour, L.A.; Cheng, R.; Kesarwala, A.H.; Heinecke, J.; Wink, D.A. Biological signaling by small inorganic molecules. Coordin. Chem. Rev. 2016, 306, 708–723. [Google Scholar] [CrossRef]
- Leemann, T.; Bonnabry, P.; Dayer, P. Selective inhibition of major drug metabolizing cytochrome P450 isozymes in human liver microsomes by carbon monoxide. Life Sci. 1994, 54, 951–956. [Google Scholar] [CrossRef]
- Alonso, J.R.; Cardellach, F.; López, S.; Casademont, J.; Miró, Ò. Carbon monoxide specifically inhibits cytochrome c oxidase of human mitochondrial respiratory chain. Pharm. Toxicol. 2003, 93, 142–146. [Google Scholar] [CrossRef] [PubMed]
- Kasparek, M.S.; Linden, D.R.; Kreis, M.E.; Sarr, M.G. Gasotransmitters in the gastrointestinal tract. Surgery 2008, 143, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Abraham, M.H.; Ibrahim, A.; Acree, W.E., Jr. Air to lung partition coefficients for volatile organic compounds and blood to lung partition coefficients for volatile organic compounds and drugs. Eur. J. Med. Chem. 2008, 43, 478–485. [Google Scholar] [CrossRef]
- Coburn, R.F. The carbon monoxide body stores. Ann. N. Y. Acad. Sci. 1970, 174, 11–22. [Google Scholar] [CrossRef]
- Luiking, Y.C.; Engelen, M.P.; Deutz, N.E. Regulation of nitric oxide production in health and disease. Curr. Opin. Clin. Nutr. 2010, 13, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, K.T. Carbon Monoxide. In Small Animal Toxicology; Elsevier: Amsterdam, The Netherlands, 2013; pp. 479–487. [Google Scholar]
- Zuckerbraun, B.S.; Chin, B.Y.; Wegiel, B.; Billiar, T.R.; Czsimadia, E.; Rao, J.; Shimoda, L.; Ifedigbo, E.; Kanno, S.; Otterbein, L.E. Carbon monoxide reverses established pulmonary hypertension. J. Exp. Med. 2006, 203, 2109–2119. [Google Scholar] [CrossRef] [PubMed]
- Bloch, K.D.; Ichinose, F.; Roberts, J.D., Jr.; Zapol, W.M. Inhaled NO as a therapeutic agent. Cardiovas. Res. 2007, 75, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Middelveld, R.; Alving, K. Endotoxin-induced shock in the pig–limited effects of low and high concentrations of inhaled nitric oxide. Acta Physiol. Scand. 2003, 179, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Nagasaka, Y.; Fernandez, B.O.; Garcia-Saura, M.F.; Petersen, B.; Ichinose, F.; Bloch, K.D.; Feelisch, M.; Zapol, W.M. Brief periods of nitric oxide inhalation protect against myocardial ischemia–reperfusion injury. J. Am. Soc. Anesthesiol. 2008, 109, 675–682. [Google Scholar] [CrossRef]
- Mann, B.E. Carbon monoxide: An essential signalling molecule. In Medicinal Organometallic Chemistry; Jaouen, G., Metzler-Nolte, N., Eds.; Springer: Berline, Franch; Heidelberg, Germany, 2010; pp. 247–285. [Google Scholar]
- Ling, K.; Men, F.; Wang, W.-C.; Zhou, Y.-Q.; Zhang, H.-W.; Ye, D.-W. Carbon Monoxide and Its Controlled Release: Therapeutic Application, Detection, and Development of Carbon Monoxide Releasing Molecules (CORMs) Miniperspective. J. Med. Chem. 2017, 61, 2611–2635. [Google Scholar] [CrossRef]
- Chatterjee, A.; Catravas, J.D. Endothelial nitric oxide (NO) and its pathophysiologic regulation. Vasc. Pharmacol. 2008, 49, 134–140. [Google Scholar] [CrossRef]
- Wang, R.; Wang, Z.; Wu, L. Carbon monoxide-induced vasorelaxation and the underlying mechanisms. Brit. J. Pharmacol. 1997, 121, 927–934. [Google Scholar] [CrossRef]
- Brüne, B.; Ullrich, V. Inhibition of platelet aggregation by carbon monoxide is mediated by activation of guanylate cyclase. Mol. Pharmacol. 1987, 32, 497–504. [Google Scholar]
- Utz, J.; Ullrich, V. Carbon monoxide relaxes ileal smooth muscle through activation of guanylate cyclase. Biochem. Pharmacol. 1991, 41, 1195–1201. [Google Scholar] [CrossRef]
- Mann, B.E.; Motterlini, R. CO and NO in medicine. Chem. Commu. 2007, 41, 4197–4208. [Google Scholar] [CrossRef] [PubMed]
- Otterbein, L.E.; Foresti, R.; Motterlini, R. Heme oxygenase-1 and carbon monoxide in the heart: The balancing act between danger signaling and pro-survival. Circ. Res. 2016, 118, 1940–1959. [Google Scholar] [CrossRef]
- Ryter, S.W.; Choi, A.M. Carbon monoxide: Present and future indications for a medical gas. Korean J. Intern. Med. 2013, 28, 123–140. [Google Scholar] [CrossRef] [PubMed]
- Gardner, P.R. Hemoglobin: A nitric-oxide dioxygenase. Scientifica 2012, 2012, 1–34. [Google Scholar] [CrossRef]
- Wang, R. Gasotransmitters: Growing pains and joys. Trends Biochem. Sci. 2014, 39, 227–232. [Google Scholar] [CrossRef]
- Kolluru, G.K.; Shen, X.; Yuan, S.; Kevil, C.G. Gasotransmitter heterocellular signaling. Antioxid. Redox Sign. 2017, 26, 936–960. [Google Scholar] [CrossRef]
- Piepoli, M.F.; Hoes, A.W.; Agewall, S.; Albus, C.; Brotons, C.; Catapano, A.L.; Cooney, M.-T.; Corrà, U.; Cosyns, B.; Deaton, C.; et al. ESC Scientific Document Group. 2016 European Guidelines on cardiovascular disease prevention in clinical practice: The Sixth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of 10 societies and by invited experts) Developed with the special contribution of the European Association for Cardiovascular Prevention & Rehabilitation (EACPR). Eur. Heart J. 2016, 37, 2315–3281. [Google Scholar]
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.; Coats, A.J.; Falk, V.; González-Juanatey, J.R.; Harjola, V.-P.; Jankowska, E.A.; et al. ESC Scientific Document Group. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2016, 37, 2129–2200. [Google Scholar]
- Cuspidi, C.; Tadic, M.; Grassi, G.; Mancia, G. Treatment of hypertension: The ESH/ESC guidelines recommendations. Pharmacol. Res. 2018, 128, 315–321. [Google Scholar] [CrossRef]
- Armstrong, P.W.; Pieske, B.; Anstrom, K.J.; Ezekowitz, J.; Hernandez, A.F.; Butler, J.; Lam, C.S.; Ponikowski, P.; Voors, A.A.; Jia, G. Vericiguat in patients with heart failure and reduced ejection fraction. N. Engl. J. Med. 2020, 382, 1883–1893. [Google Scholar] [CrossRef]
- Borghi, C.; Omboni, S.; Novo, S.; Vinereanu, D.; Ambrosio, G.; Ambrosioni, E. Efficacy and safety of zofenopril versus ramipril in the treatment of myocardial infarction and heart failure: A review of the published and unpublished data of the randomized double-blind SMILE-4 study. Adv. Ther. 2018, 35, 604–618. [Google Scholar] [CrossRef] [PubMed]
- Grześk, E.; Malinowski, B.; Wiciński, M.; Szadujkis-Szadurska, K.; Sinjab, T.A.; Manysiak, S.; Tejza, B.; Słupski, M.; Odrowąż-Sypniewska, G.; Grześk, G. Cyclosporine-A, but not tacrolimus significantly increases reactivity of vascular smooth muscle cells. Pharmacol. Rep. 2016, 68, 201–205. [Google Scholar] [CrossRef]
- Magierowski, M.; Jasnos, K.; Sliwowski, Z.; Surmiak, M.; Krzysiek-Maczka, G.; Ptak-Belowska, A.; Kwiecien, S.; Brzozowski, T. Exogenous asymmetric dimethylarginine (ADMA) in pathogenesis of ischemia-reperfusion-induced gastric lesions: Interaction with protective nitric oxide (NO) and calcitonin gene-related peptide (CGRP). Int. J. Mol. Sci. 2014, 15, 4946–4964. [Google Scholar] [CrossRef] [PubMed]
- Anatomical Therapeutic Chemical Classification System. Available online: https://www.whocc.no/atc_ddd_index/?code=V04CX08 (accessed on 26 April 2021).
- Anatomical Therapeutic Chemical Classification System. Available online: https://www.whocc.no/atc_ddd_index/?code=R07AX01 (accessed on 26 April 2021).
- Available online: https://go.drugbank.com/drugs/DB11588 (accessed on 26 April 2021).
- Available online: https://go.drugbank.com/drugs/DB00435 (accessed on 26 April 2021).
- Gullotta, F.; Masi, A.D.; Ascenzi, P. Carbon monoxide: An unusual drug. Iubmb Life 2012, 64, 378–386. [Google Scholar] [CrossRef]
- Ji, X.; Damera, K.; Zheng, Y.; Yu, B.; Otterbein, L.E.; Wang, B. Toward carbon monoxide–based therapeutics: Critical drug delivery and developability issues. J. Pharm. Sci. 2016, 105, 406–416. [Google Scholar] [CrossRef]
- Rezk-Hanna, M.; Mosenifar, Z.; Benowitz, N.L.; Rader, F.; Rashid, M.; Davoren, K.; Moy, N.B.; Doering, L.; Robbins, W.; Sarna, L. High carbon monoxide levels from charcoal combustion mask acute endothelial dysfunction induced by hookah (waterpipe) smoking in young adults. Circulation 2019, 139, 2215–2224. [Google Scholar] [CrossRef] [PubMed]
- Lal, A.; Patterson, L.; Goldrich, A.; Marsh, A. Point-of-care end-tidal carbon monoxide reflects severity of hemolysis in sickle cell anemia. Pediatr. Blood Cancer 2015, 62, 912–914. [Google Scholar] [CrossRef] [PubMed]
- Bathoorn, E.; Slebos, D.-J.; Postma, D.S.; Koeter, G.H.; van Oosterhout, A.J.; van der Toorn, M.; Boezen, H.M.; Kerstjens, H.A. Anti-inflammatory effects of inhaled carbon monoxide in patients with COPD: A pilot study. Eur. Respir. J. 2007, 30, 1131–1137. [Google Scholar] [CrossRef] [PubMed]
- Barisione, G.; Brusasco, V. Lung diffusing capacity for nitric oxide and carbon monoxide following mild-to-severe COVID-19. Physiol. Rep. 2021, 9, e14748. [Google Scholar] [CrossRef]
- Fredenburgh, L.E.; Perrella, M.A.; Barragan-Bradford, D.; Hess, D.R.; Peters, E.; Welty-Wolf, K.E.; Kraft, B.D.; Harris, R.S.; Maurer, R.; Nakahira, K. A phase I trial of low-dose inhaled carbon monoxide in sepsis-induced ARDS. JCI Insight 2018, 3, e124039. [Google Scholar] [CrossRef]
- Zobi, F. CO and CO-releasing molecules in medicinal chemistry. Future Med. Chem. 2013, 5, 175–188. [Google Scholar] [CrossRef] [PubMed]
- Motterlini, R.; Mann, B.E.; Foresti, R. Therapeutic applications of carbon monoxide-releasing molecules. Expert Opin. Inv. Drug. 2005, 14, 1305–1318. [Google Scholar] [CrossRef] [PubMed]
- Motterlini, R. Carbon monoxide-releasing molecules (CO-RMs): Vasodilatory, anti-ischaemic and anti-inflammatory activities. Biochem. Soc. T. 2007, 35, 1142–1146. [Google Scholar] [CrossRef]
- Ward, J.S.; Lynam, J.M.; Moir, J.W.; Sanin, D.E.; Mountford, A.P.; Fairlamb, I.J. A therapeutically viable photo-activated manganese-based CO-releasing molecule (photo-CO-RM). Dalton 2012, 41, 10514–10517. [Google Scholar] [CrossRef]
- Schatzschneider, U. Novel lead structures and activation mechanisms for CO-releasing molecules (CORMs). Brit. J. Pharm. 2015, 172, 1638–1650. [Google Scholar] [CrossRef]
- Kourti, M.; Jiang, W.G.; Cai, J. Aspects of carbon monoxide in form of CO-releasing molecules used in cancer treatment: More light on the way. Oxid. Med. Cell. Longev. 2017, 2017, 9326454. [Google Scholar] [CrossRef] [PubMed]
- Heinemann, S.H.; Hoshi, T.; Westerhausen, M.; Schiller, A. Carbon monoxide–physiology, detection and controlled release. Chem. Commun. 2014, 50, 3644–3660. [Google Scholar] [CrossRef]
- Magierowska, K.; Magierowski, M.; Surmiak, M.; Adamski, J.; Mazur-Bialy, A.I.; Pajdo, R.; Sliwowski, Z.; Kwiecien, S.; Brzozowski, T. The protective role of carbon monoxide (CO) produced by heme oxygenases and derived from the CO-releasing molecule CORM-2 in the pathogenesis of stress-induced gastric lesions: Evidence for non-involvement of nitric oxide (NO). Int. J. Mol. Sci. 2016, 17, 442. [Google Scholar] [CrossRef]
- Wang, D.; Viennois, E.; Ji, K.; Damera, K.; Draganov, A.; Zheng, Y.; Dai, C.; Merlin, D.; Wang, B. A click-and-release approach to CO prodrugs. Chem. Commun. 2014, 50, 15890–15893. [Google Scholar] [CrossRef] [PubMed]
- Abeyrathna, N.; Washington, K.; Bashur, C.; Liao, Y. Nonmetallic carbon monoxide releasing molecules (CORMs). Org. Biom. Chem. 2017, 15, 8692–8699. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.-X.; Ke, B.-W.; Lu, W.; Wang, B.-H. CO as a therapeutic agent: Discovery and delivery forms. Chin. J. Nat. Med. 2020, 18, 284–295. [Google Scholar]
- Ji, X.; Wang, B. Strategies toward organic carbon monoxide prodrugs. Accounts Chem. Res. 2018, 51, 1377–1385. [Google Scholar] [CrossRef] [PubMed]
- De La Cruz, L.K.C.; Benoit, S.p.L.; Pan, Z.; Yu, B.; Maier, R.J.; Ji, X.; Wang, B. Click, release, and fluoresce: A chemical strategy for a cascade prodrug system for codelivery of carbon monoxide, a drug payload, and a fluorescent reporter. Org. Lett. 2018, 20, 897–900. [Google Scholar] [CrossRef]
- Feng, W.; Feng, S.; Feng, G. CO release with ratiometric fluorescence changes: A promising visible-light-triggered metal-free CO-releasing molecule. Chem. Commun. 2019, 55, 8987–8990. [Google Scholar] [CrossRef]
- Adach, W.; Olas, B. Carbon monoxide and its donors–their implications for medicine. Future Med. Chem. 2019, 11, 61–73. [Google Scholar] [CrossRef] [PubMed]
- Bakalarz, D.; Surmiak, M.; Yang, X.; Wójcik, D.; Korbut, E.; Śliwowski, Z.; Ginter, G.; Buszewicz, G.; Brzozowski, T.; Cieszkowski, J. Organic carbon monoxide prodrug, BW-CO-111, in protection against chemically-induced gastric mucosal damage. Acta Pharmaceutica Sinica B 2021, 11, 456–475. [Google Scholar] [CrossRef]
- Magierowska, K.; Bakalarz, D.; Wójcik, D.; Korbut, E.; Danielak, A.; Głowacka, U.; Pajdo, R.; Buszewicz, G.; Ginter, G.; Surmiak, M. Evidence for cytoprotective effect of carbon monoxide donor in the development of acute esophagitis leading to acute esophageal epithelium lesions. Cells 2020, 9, 1203. [Google Scholar] [CrossRef] [PubMed]
- Magierowska, K.; Brzozowski, T.; Magierowski, M. Emerging role of carbon monoxide in regulation of cellular pathways and in the maintenance of gastric mucosal integrity. Pharmacol. Res. 2018, 129, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Jasnos, K.; Magierowski, M.; Kwiecień, S.; Brzozowski, T. Carbon monoxide in human physiology--its role in the gastrointestinal tract. Postepy Higieny Medycyny Doswiadczalnej 2014, 68, 101–109. [Google Scholar] [CrossRef]
- Committee for Medicinal Products for Human Use. European Public Assessment Report (EPAR) Summary for the Public INOmax Nitric Oxide. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/inomax (accessed on 26 April 2021).
- Pieretti, J.C.; Junho, C.V.C.; Ramos, M.S.C.; Seabra, A.B. H2S-and NO-releasing gasotransmitter platform: A crosstalk signaling pathway in the treatment of acute kidney injury. Pharmacol. Res. 2020, 105121. [Google Scholar] [CrossRef] [PubMed]
- Divakaran, S.; Loscalzo, J. The role of nitroglycerin and other nitrogen oxides in cardiovascular therapeutics. J. Am. Coll. Cardiol. 2017, 70, 2393–2410. [Google Scholar] [CrossRef]
- Holme, M.R.; Sharman, T. Sodium Nitroprusside. [Internet]. 2020. Available online: https://www.ncbi.nlm.nih.gov/books/NBK557487/ (accessed on 26 April 2021).
- Kim, K.H.; Kerndt, C.; Schaller, D.J. Nitroglycerin. [Internet]. 2020. Available online: https://pubmed.ncbi.nlm.nih.gov/29494004/ (accessed on 26 April 2021).
- Fung, H.-L. Biochemical mechanism of nitroglycerin action and tolerance: Is this old mystery solved? Annu. Rev. Pharmacol. Toxicol. 2004, 44, 67–85. [Google Scholar] [CrossRef]
- Hottinger, D.G.; Beebe, D.S.; Kozhimannil, T.; Prielipp, R.C.; Belani, K.G. Sodium nitroprusside in 2014: A clinical concepts review. J. Anaesthesiol. Clin. Pharmacol. 2014, 30, 462–471. [Google Scholar]
- Somasundaram, V.; Basudhar, D.; Bharadwaj, G.; No, J.H.; Ridnour, L.A.; Cheng, R.Y.S.; Fujita, M.; Thomas, D.D.; Anderson, S.K.; McVicar, D.W.; et al. Molecular mechanisms of nitric oxide in cancer progression, signal transduction, and metabolism. Antioxid. Redox Sign. 2019, 30, 1124–1143. [Google Scholar] [CrossRef]
- Bonavida, B. Nitric Oxide (NO) and Cancer; Springer: New York, NY, USA, 2010; pp. 459–477. [Google Scholar]
- Aisa, M.C.; Datti, A.; Orlacchio, A.; Di Renzo, G.C. COX inhibitors and bone: A safer impact on osteoblasts by NO-releasing NSAIDs. Life Sci. 2018, 208, 10–19. [Google Scholar] [CrossRef]
- Privett, B.J.; Broadnax, A.D.; Bauman, S.J.; Riccio, D.A.; Schoenfisch, M.H. Examination of bacterial resistance to exogenous nitric oxide. Nitric. Oxide 2012, 26, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Danielak, A.; Wallace, J.L.; Brzozowski, T.; Magierowski, M. Gaseous mediators as a key molecular targets for the development of gastrointestinal-safe anti-inflammatory pharmacology. Front. Pharmacol. 2021, 12, 657457. [Google Scholar] [CrossRef] [PubMed]
- Yeh, R.K.; Chen, J.; Williams, J.L.; Baluch, M.; Hundley, T.R.; Rosenbaum, R.E.; Kalala, S.; Traganos, F.; Benardini, F.; del Soldato, P. NO-donating nonsteroidal antiinflammatory drugs (NSAIDs) inhibit colon cancer cell growth more potently than traditional NSAIDs: A general pharmacological property? Biochem. Pharmacol. 2004, 67, 2197–2205. [Google Scholar] [CrossRef] [PubMed]
- McIlhatton, M.A.; Tyler, J.; Burkholder, S.; Ruschoff, J.; Rigas, B.; Kopelovich, L.; Fishel, R. Nitric Oxide–Donating Aspirin Derivatives Suppress Microsatellite Instability in Mismatch Repair–Deficient and Hereditary Nonpolyposis Colorectal Cancer Cells. Cancer Res. 2007, 67, 10966–10975. [Google Scholar] [CrossRef][Green Version]
- Serafim, R.A.; Pernichelle, F.G.; Ferreira, E.I. The latest advances in the discovery of nitric oxide hybrid drug compounds. Expert Opin. Drug Dis. 2017, 12, 941–953. [Google Scholar] [CrossRef]
- Kashfi, K.; Duvalsaint, P.L. Nitric oxide donors and therapeutic applications in cancer. In Nitric Oxide Donors; Elsevier: Amsterdam, The Netherlands, 2017; pp. 75–119. [Google Scholar]
- Van’t Hof, R.J.; Ralston, S.H. Nitric oxide and bone. Immunology 2001, 103, 255–261. [Google Scholar] [CrossRef]
- Hollas, M.A.; Aissa, M.B.; Lee, S.H.; Gordon-Blake, J.M.; Thatcher, G.R. Pharmacological manipulation of cGMP and NO/cGMP in CNS drug discovery. Nitric Oxide 2019, 82, 59–74. [Google Scholar] [CrossRef]
- Seabra, A.B.; Durán, N. Nitric oxide donors for prostate and bladder cancers: Current state and challenges. Eur. J. Pharmacol. 2018, 826, 158–168. [Google Scholar] [CrossRef]
- Wallace, J.L. Nitric oxide in the gastrointestinal tract: Opportunities for drug development. Brit. J. Pharmacol. 2019, 176, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.-G.; Leung, W.K. Potential strategies in the prevention of nonsteroidal anti-inflammatory drugs-associated adverse effects in the lower gastrointestinal tract. Gut Liver 2020, 14, 179. [Google Scholar] [CrossRef] [PubMed]
- Tziona, P.; Theodosis-Nobelos, P.; Rekka, E.A. Medicinal chemistry approaches of controlling gastrointestinal side effects of non-steroidal anti-inflammatory drugs. Endogenous protective mechanisms and drug design. Med. Chem. 2017, 13, 408–420. [Google Scholar] [CrossRef]
- Kashfi, K.; Esmaili, M. NO-H2S-releasing chimeras as a multifaceted approach to cancer therapy. In Nitric Oxide (Donor/Induced) in Chemosensitizing; Elsevier: Amsterdam, The Netherlands, 2017; pp. 105–142. [Google Scholar]
- Magierowski, M.; Magierowska, K.; Kwiecien, S.; Brzozowski, T. Gaseous mediators nitric oxide and hydrogen sulfide in the mechanism of gastrointestinal integrity, protection and ulcer healing. Molecules 2015, 20, 9099–9123. [Google Scholar] [CrossRef]
- Rigas, B.; Kashfi, K. Nitric-oxide-donating NSAIDs as agents for cancer prevention. Trends Mol. Med. 2004, 10, 324–330. [Google Scholar] [CrossRef] [PubMed]
- Pereira-Leite, C.; Nunes, C.; Jamal, S.K.; Cuccovia, I.M.; Reis, S. Nonsteroidal anti-inflammatory therapy: A journey toward safety. Med. Res. Rev. 2017, 37, 802–859. [Google Scholar] [CrossRef]
- Fiorucci, S.; Santucci, L.; Wallace, J.L.; Sardina, M.; Romano, M.; del Soldato, P.; Morelli, A. Interaction of a selective cyclooxygenase-2 inhibitor with aspirin and NO-releasing aspirin in the human gastric mucosa. Proc. Nat. Acad. Sci. USA 2003, 100, 10937–10941. [Google Scholar] [CrossRef]
- Fiorucci, S.; Santucci, L.; Gresele, P.; Faccino, R.M.; Del Soldato, P.; Morelli, A. Gastrointestinal safety of NO-aspirin (NCX-4016) in healthy human volunteers: A proof of concept endoscopic study. Gastroenterology 2003, 124, 600–607. [Google Scholar] [CrossRef] [PubMed]
- Fiorucci, S.; Mencarelli, A.; Meneguzzi, A.; Lechi, A.; Renga, B.; del Soldato, P.; Morelli, A.; Minuz, P. Co-administration of nitric oxide-aspirin (NCX-4016) and aspirin prevents platelet and monocyte activation and protects against gastric damage induced by aspirin in humans. J. Am. Coll. Cardiol. 2004, 44, 635–641. [Google Scholar] [CrossRef]
- Gresele, P.; Migliacci, R.; Procacci, A.; De Monte, P.; Bonizzoni, E. Prevention by NCX 4016, a nitric oxide-donating aspirin, but not by aspirin, of the acute endothelial dysfunction induced by exercise in patients with intermittent claudication. Thromb. Haemost. 2007, 97, 444–450. [Google Scholar] [PubMed]
- Gresele, P.; Marzotti, S.; Guglielmini, G.; Momi, S.; Giannini, S.; Minuz, P.; Lucidi, P.; Bolli, G.B. Hyperglycemia-induced platelet activation in type 2 diabetes is resistant to aspirin but not to a nitric oxide–donating agent. Diabetes Care 2010, 33, 1262–1268. [Google Scholar] [CrossRef] [PubMed]
- Gresele, P.; Migliacci, R.; Arosio, E.; Bonizzoni, E.; Minuz, P.; Violi, F.; Group, N.-X.-S. Effect on walking distance and atherosclerosis progression of a nitric oxide-donating agent in intermittent claudication. J. Vasc. Surg. 2012, 56, 1622–1628.e5. [Google Scholar] [CrossRef] [PubMed]
- Davies, S.L.; Moral, M.A.; Bozzo, J. Salicylates targeting insulin resistance. Drug. Future 2007, 32, 361–365. [Google Scholar] [CrossRef]
- Schnitzer, T.J.; Kivitz, A.J.; Lipetz, R.S.; Sanders, N.; Hee, A. Comparison of the COX-inhibiting nitric oxide donator AZD3582 and rofecoxib in treating the signs and symptoms of osteoarthritis of the knee. Arthritis Care Res. Off. J. Am. Coll. Rheumatol. 2005, 53, 827–837. [Google Scholar] [CrossRef] [PubMed]
- Schnitzer, T.; Kivitz, A.; Frayssinet, H.; Duquesroix, B. Efficacy and safety of naproxcinod in the treatment of patients with osteoarthritis of the knee: A 13-week prospective, randomized, multicenter study. Osteoarthr. Cartilage 2010, 18, 629–639. [Google Scholar] [CrossRef] [PubMed]
- Schnitzer, T.J.; Hochberg, M.C.; Marrero, C.E.; Duquesroix, B.; Frayssinet, H.; Beekman, M. Efficacy and Safety of Naproxcinod in Patients with Osteoarthritis of the Knee: A 53-Week Prospective Randomized Multicenter Study, Seminars in Arthritis and Rheumatism; Elsevier: Amsterdam, The Netherlands, 2011; pp. 285–297. [Google Scholar]
- Available online: http://www.nicox.com/wp-content/uploads/pr2010072200en.pdf?aeb643 (accessed on 26 April 2021).
- Magierowski, M.; Magierowska, K.; Surmiak, M.; Hubalewska-Mazgaj, M.; Kwiecien, S.; Wallace, J.; Brzozowski, T. The effect of hydrogen sulfide-releasing naproxen (ATB-346) versus naproxen on formation of stress-induced gastric lesions, the regulation of systemic inflammation, hypoxia and alterations in gastric microcirculation. J. Physiol. Pharmacol 2017, 68, 749–756. [Google Scholar] [PubMed]
- del Soldato, P.; Sorrentino, R.; Pinto, A. NO-aspirins: A class of new anti-inflammatory and antithrombotic agents. Trends Pharmacol. Sci. 1999, 20, 319–323. [Google Scholar] [CrossRef]
- Wang, R. Overview of Gasotransmitters and the Related Signaling Network. Gasotransmitters 2018, 1–28. [Google Scholar] [CrossRef]
| Physical Property | Carbon Monoxide | Nitric Oxide |
|---|---|---|
| Molecular weight | 28.01 | 30.06 |
| Boiling point (°C) | −191.5 | −151.8 |
| Melting point (°C) | −205 | −163.6 |
| refractive index | 1.0003 | 1.0002697 |
| water solubility | 354 mL/dL; 44.3 ppm by mass at STP [30] | 67 mg/L [30] |
| Density (kg/m3 vapor) | 788.6 [32] | 3.027 [32] |
| Specific gravity (g/L) | 1.250 [34] | 1.037 [33] |
| Bond order | 2.5 [35] | 3 [35] |
| Bond length, re × 10−8 cm | 1.151 [35] | 1.128 [35] |
| Reactivity | inert, except binds to hemoproteins; free CO does not readily react with reducing agents, including hydrogen; the coordinated CO has greater reactivity than the free gas, and the reduction of CO can be greatly facilitated by transition metals [36] | highly reactive, very short half life [34] |
| Vibrational wave number (cm−1) | 1220 [35] | 1180 [35] |
| Energy activation of oxidation reaction kJ/mol | 213 [30] | 6.47 [37] |
| Dissociation energy (kcal/mol) | 152.8 [35] | 258.9 [35] |
| Ionization potential (eV) | 15.0 [38] | 9.27 |
| Feature | Neurotransmitter | Gasotransmitters |
|---|---|---|
| Criteria of definition |
|
|
| Examples | Acetylcholine, catecholamines, serotonin, histamine, glutamate, glycine, GABA, ATP and other | NO, CO, and H2S |
| Chemical nature | Lipophilic or lipophobic | Amphiphilic |
| Action modes: | ||
| Release | Exocytotic vesicle | Cytoplasm release |
| Re-uptake | + | - |
| Removal mechanism | Enzyme-dependent | Nonenzymatic: oxidation, scavenging, methylation, etc. |
| Revert direction | Pre- to postsynaptic membrane (unidirectional) | Bidirectional |
| Membrane receptors | Necessary | Not necessary |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nowaczyk, A.; Kowalska, M.; Nowaczyk, J.; Grześk, G. Carbon Monoxide and Nitric Oxide as Examples of the Youngest Class of Transmitters. Int. J. Mol. Sci. 2021, 22, 6029. https://doi.org/10.3390/ijms22116029
Nowaczyk A, Kowalska M, Nowaczyk J, Grześk G. Carbon Monoxide and Nitric Oxide as Examples of the Youngest Class of Transmitters. International Journal of Molecular Sciences. 2021; 22(11):6029. https://doi.org/10.3390/ijms22116029
Chicago/Turabian StyleNowaczyk, Alicja, Magdalena Kowalska, Jacek Nowaczyk, and Grzegorz Grześk. 2021. "Carbon Monoxide and Nitric Oxide as Examples of the Youngest Class of Transmitters" International Journal of Molecular Sciences 22, no. 11: 6029. https://doi.org/10.3390/ijms22116029
APA StyleNowaczyk, A., Kowalska, M., Nowaczyk, J., & Grześk, G. (2021). Carbon Monoxide and Nitric Oxide as Examples of the Youngest Class of Transmitters. International Journal of Molecular Sciences, 22(11), 6029. https://doi.org/10.3390/ijms22116029