Molecular Genetics of Conjunctival Melanoma and Prognostic Value of TERT Promoter Mutation Analysis
Abstract
1. Introduction
2. Results
2.1. Clinical and Histopathological Characteristics
2.2. Mutation Analysis
2.3. Immunohistochemistry
3. Discussion
4. Materials and Methods
4.1. Material Selection
4.2. DNA Isolation
4.3. Targeted Next-Generation Sequencing
4.4. Mutation Analysis
4.5. Immunohistochemistry
4.6. Survival Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cao, J.; Heijkants, R.C.; Jochemsen, A.G.; Dogrusöz, M.; De Lange, M.J.; Van Der Velden, P.A.; Van Der Burg, S.H.; Jager, M.J.; Verdijk, R.M. Targeting of the MAPK and AKT pathways in conjunctival melanoma shows potential synergy. Oncotarget 2017, 8, 58021–58036. [Google Scholar] [CrossRef] [PubMed]
- Larsen, A.C.; Dahmcke, C.M.; Dahl, C.; Siersma, V.D.; Toft, P.B.; Coupland, S.E.; Prause, J.U.; Guldberg, P.; Heegaard, S. A retrospective review of conjunctival malignant melanoma presentation, treatment and outcome and an investigation of features associated with BRAF mutations. JAMA Ophthalmol. 2015, 133, 1295–1303. [Google Scholar] [CrossRef]
- Griewank, K.G.; Westekemper, H.; Murali, R.; Mach, M.; Schilling, B.; Wiesner, T.; Schimming, T.; Livingstone, E.; Sucker, A.; Grabellus, F.; et al. Conjunctival Melanomas Harbor BRAF and NRAS Mutations and Copy Number Changes Similar to Cutaneous and Mucosal Melanomas. Clin. Cancer Res. 2013, 19, 3143–3152. [Google Scholar] [CrossRef] [PubMed]
- Dalvin, L.A.; Salomão, D.R.; Patel, S.V. Population-based incidence of conjunctival tumours in Olmsted County, Minnesota. Br. J. Ophthalmol. 2018, 102, 1728–1734. [Google Scholar] [CrossRef]
- Missotten, G.S.; Keijser, S.; De Keizer, R.J.W.; De Wolff-Rouendaal, D. Conjunctival Melanoma in The Netherlands: A Nationwide Study. Investig. Opthalmol. Vis. Sci. 2005, 46, 75–82. [Google Scholar] [CrossRef]
- Rossi, E.; Schinzari, G.; Maiorano, B.A.; Pagliara, M.M.; Di Stefani, A.; Bria, E.; Peris, K.; Blasi, M.A.; Tortora, G. Conjunctival Melanoma: Genetic and Epigenetic Insights of a Distinct Type of Melanoma. Int. J. Mol. Sci. 2019, 20, 5447. [Google Scholar] [CrossRef]
- Zhou, C.; Wang, Y.; Jia, R.; Fan, X. Conjunctival Melanoma in Chinese Patients: Local Recurrence, Metastasis, Mortality, and Comparisons with Caucasian Patients. Investig. Opthalmol. Vis. Sci. 2017, 58, 5452–5459. [Google Scholar] [CrossRef]
- Wong, J.R.; Nanji, A.A.; Galor, A.; Karp, C.L. Management of conjunctival malignant melanoma: A review and update. Expert Rev. Oph-Thalmol. 2014, 9, 185–204. [Google Scholar] [CrossRef] [PubMed]
- Kujala, E.; Tuomaala, S.; Eskelin, S.; Kivelä, T. Mortality after uveal and conjunctival melanoma: Which tumour is more deadly? Acta Ophthalmol. 2009, 87, 149–153. [Google Scholar] [CrossRef]
- Esmaeli, B.; Wang, X.; Youssef, A.; Gershenwald, J.E. Patterns of regional and distant metastasis in patients with conjunctival melanoma: Experience at a cancer center over four decades. Ophthalmology 2001, 108, 2101–2105. [Google Scholar] [CrossRef]
- Paridaens, A.D.; Minassian, D.C.; McCartney, A.C.; Hungerford, J.L. Prognostic factors in primary malignant melanoma of the conjunctiva: A clinicopathological study of 256 cases. Br. J. Ophthalmol. 1994, 78, 252–259. [Google Scholar] [CrossRef]
- Smit, K.N.; Van Poppelen, N.M.; Vaarwater, J.; Verdijk, R.; van Marion, R.; Kalirai, H.; Coupland, S.E.; Thornton, S.; Farquhar, N.; Dubbink, H.J.; et al. Combined mutation and copy-number variation detection by targeted next-generation sequencing in uveal melanoma. Mod. Pathol. 2018, 31, 763–771. [Google Scholar] [CrossRef] [PubMed]
- van Ipenburg, J.A.; Naus, N.C.; Dubbink, H.J.; van Ginderdeuren, R.; Missotten, G.S.; Paridaens, D.; Verdijk, R.M. Prognostic value of TERT promoter mutations in conjunctival melanomas in addition to clinicopathological features. Br. J. Ophthalmol. 2020. [Google Scholar] [CrossRef]
- Koopmans, A.E.; Ober, K.; Dubbink, H.J.; Paridaens, D.; Naus, N.C.; Belunek, S.; Krist, B.; Post, E.; Zwarthoff, E.C.; de Klein, A.; et al. Prevalence and Implications of TERT Promoter Mutation in Uveal and Conjunc-tival Melanoma and in Benign and Premalignant Conjunctival Melanocytic Lesions. Invest. Ophthalmol. Vis. Sci. 2014, 55, 6024–6030. [Google Scholar] [CrossRef]
- Scholz, S.L.; Cosgarea, I.; Süßkind, D.; Murali, R.; Möller, I.; Reis, H.; Leonardelli, S.; Schilling, B.; Schimming, T.; Hadaschik, E.; et al. NF1 mutations in conjunctival melanoma. Br. J. Cancer 2018, 118, 1243–1247. [Google Scholar] [CrossRef]
- Cui, C.; Lian, B.; Zhou, L.; Song, X.; Zhang, X.; Wu, D.; Chi, Z.; Si, L.; Sheng, X.; Kong, Y.; et al. Multifactorial Analysis of Prognostic Factors and Survival Rates Among 706 Mucosal Melanoma Patients. Ann. Surg. Oncol. 2018, 25, 2184–2192. [Google Scholar] [CrossRef] [PubMed]
- Ellerhorst, J.A.; Greene, V.R.; Ekmekcioglu, S.; Warneke, C.L.; Johnson, M.M.; Cooke, C.P.; Wang, L.E.; Prieto, V.G.; Gershenwald, J.E.; Wei, Q.; et al. Clinical correlates of NRAS and BRAF mutations in primary human melanoma. Clin. Cancer Res. 2011, 17, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Griewank, K.G.; Murali, R.; Schilling, B.E.; Scholz, S.; Sucker, A.; Song, M.; Susskind, D.; Grabellus, F.; Zimmer, L.O.; Hillen, U.; et al. TERT promoter mutations in ocular melanoma distinguish between conjunctival and uveal tumours. Br. J. Cancer 2013, 109, 497–501. [Google Scholar] [CrossRef] [PubMed]
- Van Raamsdonk, C.D.; Griewank, K.G.; Crosby, M.B.; Garrido, M.C.; Vemula, S.; Wiesner, T.; Obenauf, A.C.; Wackernagel, W.; Green, G.; Bouvier, N.; et al. Mutations in GNA11 in uveal melanoma. N. Engl. J. Med. 2010, 363, 2191–2199. [Google Scholar] [CrossRef]
- Jain, P.; Finger, P.T.; Damato, B.; Coupland, S.E.; Heimann, H.; Kenawy, N.; Brouwer, N.J.; Marinkovic, M.; Van Duinen, S.G.; Caujolle, J.P.; et al. Multicenter, International Assessment of the Eighth Edition of the American Joint Committee on Cancer Cancer Staging Manual for Conjunctival Melanoma. JAMA Ophthalmol. 2019, 137, 905–911. [Google Scholar] [CrossRef]
- Dratwa, M.; Wysoczańska, B.; Lacina, P.; Kubik, T.; Bogunia-Kubik, K. TERT-Regulation and Roles in Cancer Formation. Front. Immunol. 2020, 11, 589929. [Google Scholar] [CrossRef] [PubMed]
- Torres-Cabala, C.; Li-Ning-Tapia, E.; Hwu, W.-J. Pathology-based Biomarkers Useful for Clinical Decisions in Melanoma. Arch. Med Res. 2020, 51, 827–838. [Google Scholar] [CrossRef] [PubMed]
- Akincilar, S.C.; Chan, C.H.T.; Ng, Q.F.; Fidan, K.; Tergaonkar, V. Non-canonical roles of canonical telomere binding proteins in cancers. Cell. Mol. Life Sci. 2021, 1–23. [Google Scholar] [CrossRef]
- Colebatch, A.J.; Dobrovic, A.; Cooper, W.A. TERT gene: Its function and dysregulation in cancer. J. Clin. Pathol. 2019, 72, 281–284. [Google Scholar] [CrossRef] [PubMed]
- Dono, M.; Angelini, G.; Cecconi, M.; Amaro, A.; Esposito, A.I.; Mirisola, V.; Maric, I.; Lanza, F.; Nasciuti, F.; Viaggi, S.; et al. Mutation frequencies of GNAQ, GNA11, BAP1, SF3B1, EIF1AX and TERT in uveal melanoma: Detection of an activating mutation in the TERT gene promoter in a single case of uveal melanoma. Br. J. Cancer 2014, 110, 1058–1065. [Google Scholar] [CrossRef]
- Macerola, E.; Loggini, B.; Giannini, R.; Garavello, G.; Giordano, M.; Proietti, A.; Niccoli, C.; Basolo, F.; Fontanini, G. Coexistence of TERT promoter and BRAF mutations in cutaneous melanoma is associated with more clinicopathological features of aggressiveness. Virchows Archiv. 2015, 467, 177–184. [Google Scholar] [CrossRef]
- Triay, E.; Bergman, L.; Nilsson, B.; All-Ericsson, C.; Seregard, S. Time trends in the incidence of conjunctival melanoma in Sweden. Br. J. Ophthalmol. 2009, 93, 1524–1528. [Google Scholar] [CrossRef]
- Larsen, A.C.; Dahl, C.; Dahmcke, C.M.; Lade-Keller, J.; Siersma, V.D.; Toft, P.B.; Coupland, S.E.; Prause, J.U.; Guldberg, P.; Heegaard, S. BRAF mutations in conjunctival melanoma: Investigation of incidence, clinico-pathological features, prognosis and paired premalignant lesions. Acta Ophthalmol. 2016, 94, 463–470. [Google Scholar] [CrossRef]
- Ascierto, P.A.; Kirkwood, J.M.; Grob, J.-J.; Simeone, E.; Grimaldi, A.M.; Maio, M.; Palmieri, G.; Testori, A.; Marincola, F.M.; Mozzillo, N. The role of BRAF V600 mutation in melanoma. J. Transl. Med. 2012, 10, 1–9. [Google Scholar] [CrossRef]
- Pasquali, S.; Hadjinicolaou, A.V.; Sileni, V.C.; Rossi, C.R.; Mocellin, S. Systemic treatments for metastatic cutaneous melanoma. Cochrane Database Syst. Rev. 2018, 2, CD011123. [Google Scholar] [CrossRef]
- Kenawy, N.; Kalirai, H.; Sacco, J.J.; Lake, S.L.; Heegaard, S.; Larsen, A.-C.; Finger, P.T.; Milman, T.; Chin, K.; Mosci, C.; et al. Conjunctival melanoma copy number alterations and correlation with mutation status, tumor features, and clinical outcome. Pigment. Cell Melanoma Res. 2019, 32, 564–575. [Google Scholar] [CrossRef]
- Mor, J.M.; Heindl, L.M. Systemic BRAF/MEK Inhibitors as a Potential Treatment Option in Metastatic Conjunctival Melanoma. Ocul. Oncol. Pathol. 2017, 3, 133–141. [Google Scholar] [CrossRef]
- Shields, C.L.; Chang, M.; Lally, S.E.; Dalvin, L.A.; Orloff, M.M. Conjunctival melanoma with orbital invasion and liver metastasis managed with systemic immune checkpoint inhibitor therapy. Indian J. Ophthalmol. 2019, 67, 2071–2073. [Google Scholar] [CrossRef]
- Heppt, M.V.; Siepmann, T.; Engel, J.; Schubert-Fritschle, G.; Eckel, R.; Mirlach, L.; Kirchner, T.; Jung, A.; Gesierich, A.; Ruzicka, T.; et al. Prognostic significance of BRAF and NRAS mutations in melanoma: A German study from routine care. BMC Cancer 2017, 17, 536. [Google Scholar] [CrossRef] [PubMed]
- Adam, C.; Fusi, L.; Weiss, N.; Goller, S.G.; Meder, K.; Frings, V.G.; Kneitz, H.; Goebeler, M.; Houben, R.; Schrama, D.; et al. Efficient Suppression of NRAS-Driven Melanoma by Co-Inhibition of ERK1/2 and ERK5 MAPK Pathways. J. Investig. Dermatol. 2020, 140, 2455–2465. [Google Scholar] [CrossRef] [PubMed]
- Quek, C.; Rawson, R.V.; Ferguson, P.M.; Shang, P.; Silva, I.; Saw, R.P.; Shannon, K.; Thompson, J.F.; Hayward, N.K.; Long, G.V.; et al. Recurrent hotspot SF3B1 mutations at codon 625 in vulvovaginal muco-sal melanoma identified in a study of 27 Australian mucosal melanomas. Oncotarget 2019, 10, 930–941. [Google Scholar] [CrossRef]
- Van Poppelen, N.M.; Vaarwater, J.; Mudhar, H.S.; Sisley, K.; Rennie, I.G.; Rundle, P.; Brands, T.; Van Den Bosch, Q.C.; Mensink, H.W.; de Klein, A.; et al. Genetic Background of Iris Melanomas and Iris Melanocytic Tumors of Uncertain Malignant Potential. Ophthalmology 2018, 125, 904–912. [Google Scholar] [CrossRef] [PubMed]
- Kong, Y.; Krauthammer, M.; Halaban, R. Rare SF3B1 R625 mutations in cutaneous melanoma. Melanoma Res. 2014, 24, 332–334. [Google Scholar] [CrossRef] [PubMed]
- Hintzsche, J.D.; Gorden, N.T.; Amato, C.M.; Kim, J.; Wuensch, K.E.; Robinson, S.E.; Applegate, A.J.; Couts, K.L.; Medina, T.M.; Wells, K.R.; et al. Whole-exome sequencing identifies recurrent SF3B1 R625 mutation and comutation of NF1 and KIT in mucosal melanoma. Melanoma Res. 2017, 27, 189–199. [Google Scholar] [CrossRef]
- Yang, H.M.; Hsiao, S.J.; Schaeffer, D.F.; Lai, C.; Remotti, H.E.; Horst, D.; Mansukhani, M.M.; Horst, B.A. Identification of recurrent mutational events in anorectal melanoma. Mod. Pathol. 2016, 30, 286–296. [Google Scholar] [CrossRef]
- Yavuzyigitoglu, S.; Koopmans, A.E.; Verdijk, R.M.; Vaarwater, J.; Eussen, B.; Van Bodegom, A.; Paridaens, D.; Kiliç, E.; de Klein, A. Uveal Melanomas with SF3B1 Mutations: A Distinct Subclass Associated with Late-Onset Metastases. Ophthalmology 2016, 123, 1118–1128. [Google Scholar] [CrossRef] [PubMed]
- Pham, D.M.; Guhan, S.; Tsao, H. KIT and Melanoma: Biological Insights and Clinical Implications. Yonsei Med. J. 2020, 61, 562–571. [Google Scholar] [CrossRef] [PubMed]
- Piris, A.; Mihm, M.C., Jr.; Hoang, M.P. BAP1 and BRAFV600E expression in benign and malignant melanocytic proliferations. Hum. Pathol. 2015, 46, 239–245. [Google Scholar] [CrossRef]
- Chau, C.; van Doorn, R.; van Poppelen, N.M.; van der Stoep, N.; Mensenkamp, A.R.; Sijmons, R.H.; van Paassen, B.W.; van den Ouweland, A.M.; Naus, N.C.; van der Hout, A.H.; et al. Families with BAP1-Tumor Predisposition Syndrome in The Nether-lands: Path to Identification and a Proposal for Genetic Screening Guidelines. Cancers 2019, 11, 1114. [Google Scholar] [CrossRef] [PubMed]
- Koopmans, A.E.; Vaarwater, J.; Paridaens, D.; Naus, N.C.; Kiliç, E.; De Klein, A. Patient survival in uveal melanoma is not affected by oncogenic mutations in GNAQ and GNA11. Br. J. Cancer 2013, 109, 493–496. [Google Scholar] [CrossRef]
- Bol, K.F.; Donia, M.; Heegaard, S.; Kiilgaard, J.F.; Svane, I.M. Genetic Biomarkers in Melanoma of the Ocular Region: What the Medical Oncologist Should Know. Int. J. Mol. Sci. 2020, 21, 5231. [Google Scholar] [CrossRef]
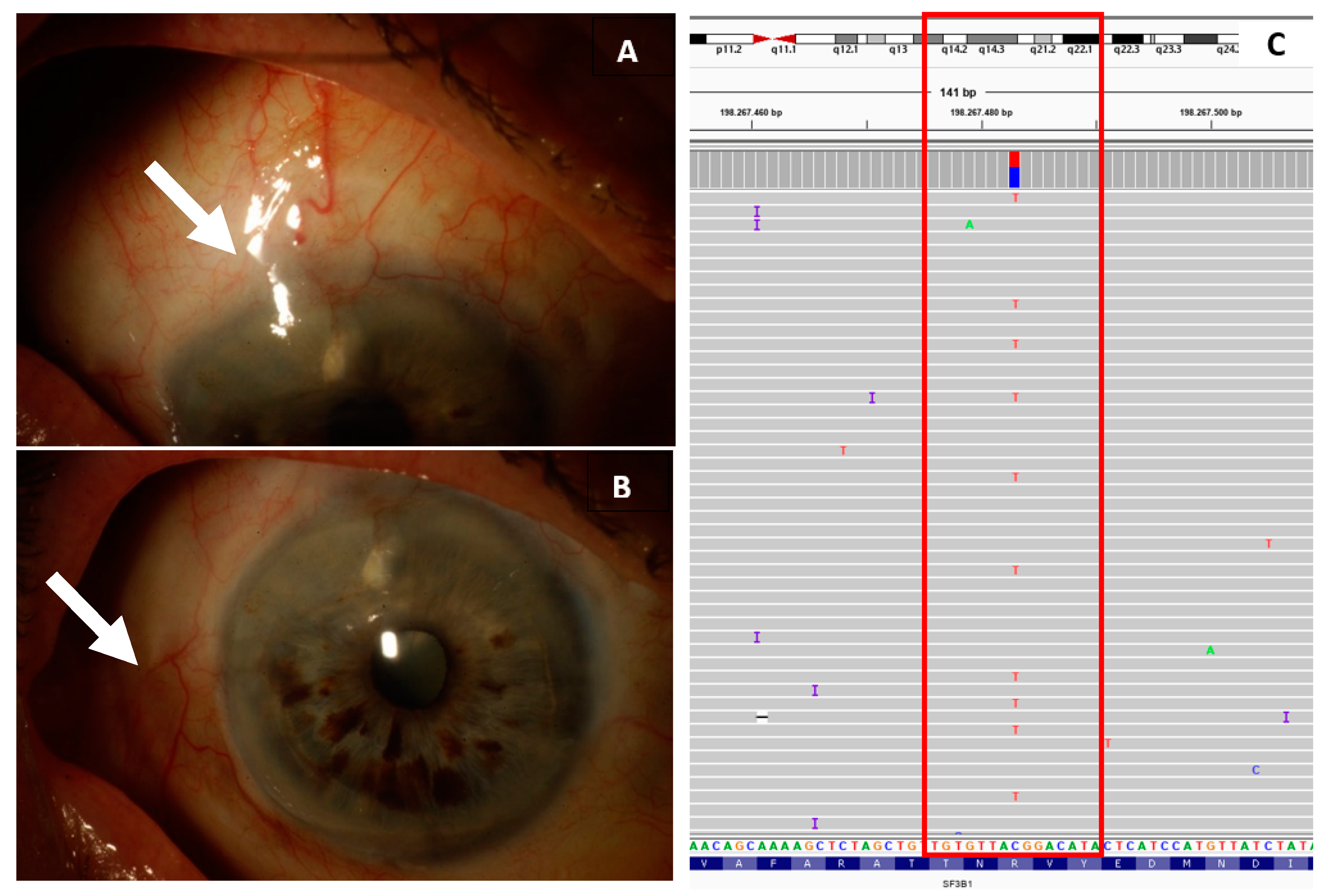
| Clinical Characteristics | |
|---|---|
| Median age at diagnosis (years) | 63 (16–89) |
| Gender | |
| Male | 14 (50%) |
| Female | 14 (50%) |
| Location | |
| Bulbar | 16 (57%) |
| Palpebral | 10 (36%) |
| Fornix | 5 (18%) |
| Caruncle | 1 (4%) |
| Metastasis | |
| No | 19 (68%) |
| Yes | 9 (32%) |
| Local recurrence | |
| No | 20 (71%) |
| Yes | 8 (29%) |
| Histopathological Characteristics | |
| Median diameter (cm) | 0.7 (0.05–1.8) |
| Median tumor thickness (mm) | 3.0 (0.18–7.70) |
| pT status | |
| pT1a | 6 (21%) |
| pT1b | 5 (18%) |
| pT2a | 1 (4%) |
| pT2b | 11 (39%) |
| pTx | 5 (18%) |
| Origin | |
| PAM | 18 (64%) |
| Nevus | 4 (14%) |
| De novo | 3 (11%) |
| Unknown | 3 (11%) |
| Gene | Presence of a Mutation | n (%) | Metastasis n (%) | MFS p-Value | Recurrences n (%) | RFS p-Value |
|---|---|---|---|---|---|---|
| SF3B1 | 0.45 | 0.45 | ||||
| Yes | 1 (4) | 0 (0) | 0 (0) | |||
| No | 27 (96) | 9 (33) | 8 (30) | |||
| BAP1 | 0.46 | 0.69 | ||||
| Yes | 5 (18) | 1 (20) | 2 (40) | |||
| No | 23 (82) | 8 (35) | 6 (26) | |||
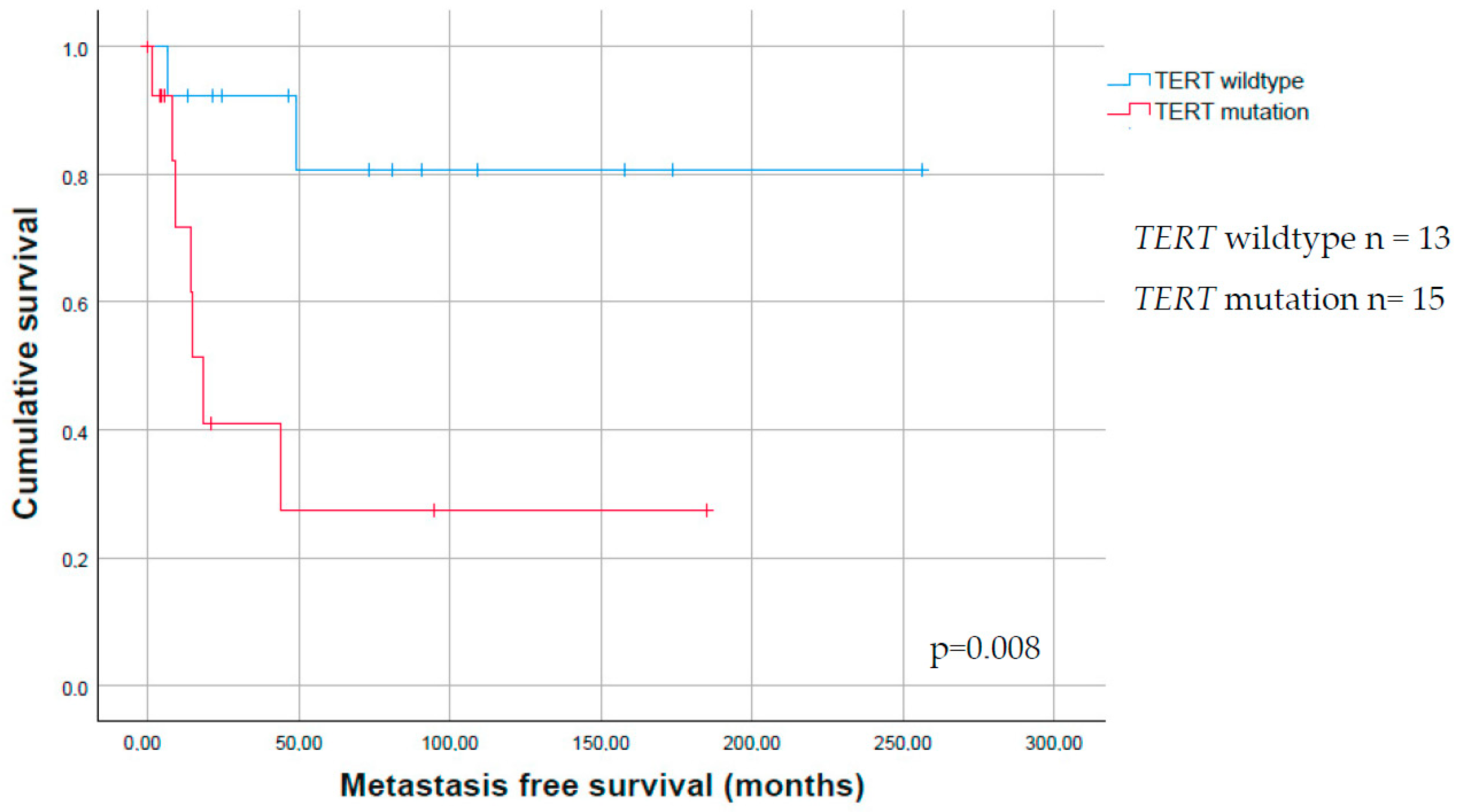
| TERT | 0.008 | 0.20 | ||||
| Yes | 15 (54) | 7 (47) | 2 (13) | |||
| No | 13 (46) | 2 (15) | 6 (46) | |||
| NRAS | 0.17 | 0.82 | ||||
| Yes | 6 (21) | 4 (67) | 2 (33) | |||
| No | 22 (79) | 5 (23) | 6 (27) | |||
| KIT | 0.26 | 0.88 | ||||
| Yes | 2 (7) | 0 (0) | 1 (50) | |||
| No | 26 (93) | 9 (35) | 7 (28) | |||
| PTEN | 0.53 | 0.25 | ||||
| Yes | 4 (14) | 1 (25) | 2 (50) | |||
| No | 24 (86) | 8 (33) | 6 (25) | |||
| BRAF | 0.052 | 0.76 | ||||
| Yes | 13 (46) | 5 (38) | 2 (15) | |||
| No | 15 (54) | 4 (27) | 6 (40) |
| TERT n = 15 (%) | P | BRAF n = 13 (%) | P | BAP1 n = 5 (%) | P | NRAS n = 6 (%) | p | PTEN n = 4 (%) | P | c-KIT n= 2 (%) | P | SF3B1 n = 1 (%) | P | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gender | 0.26 | 0.71 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | ||||||||
| Male | 6 (40) | 6 (46) | 3 (60) | 3 (50) | 3 (75) | 1 (50) | 1 (100) | ||||||||
| Female | 9 (60) | 7 (54) | 2 (40) | 3 (50) | 1 (25) | 1 (50) | 0 (0) | ||||||||
| Age | 0.91 | 0.91 | 0.52 | 0.32 | 0.92 | 0.24 | 0.50 | ||||||||
| <50y | 2 (13) | 2 (15) | 0 (0) | 2 (33) | 0 (0) | 0 (0) | 0 (0) | ||||||||
| 50–65y | 7 (47) | 6 (46) | 3 (60) | 2 (33) | 2 (50) | 2 (100) | 1 (100) | ||||||||
| >65y | 6 (40) | 5 (38) | 2 (40) | 2 (33) | 2 (50) | 0 (0) | 0 (0) | ||||||||
| Location | 0.16 | 0.85 | 1.00 | 1.00 | 0.59 | 1.00 | 0.48 | ||||||||
| Bulbar | 8 (53) | 6 (46) | 2 (40) | 2 (33) | 1 (25) | 1 (50) | 1 (100) | ||||||||
| Forniceal/ palpebral/ caruncular involvement | 5 (33) | 6 (46) | 2 (40) | 2 (33) | 2 (50) | 1 (50) | 0 (0) | ||||||||
| Tumor thick-ness | 0.67 | 0.68 | 1.00 | 0.63 | 0.56 | 0.53 | 0.31 | ||||||||
| Tumor thickness ≤2 mm | 5 (33) | 3 (23) | 1 (20) | 2 (33) | 2 (50) | 1 (50) | 1 (100) | ||||||||
| Tumor thickness >2mm | 8 (53) | 9 (69) | 4 (80) | 3 (50) | 2 (50) | 1 (50) | 0 (0) | ||||||||
| pT status | 0.16 | 0.85 | 1.00 | 1.00 | 0.59 | 1.00 | 0.48 | ||||||||
| pT1 | 8 (53) | 6 (46) | 2 (40) | 2 (33) | 1 (25) | 1 (50) | 1 (100) | ||||||||
| pT2 | 5 (33) | 6 (46) | 2 (40) | 2 (33) | 2 (50) | 1 (50) | 0 (0) | ||||||||
| Origin | 0.01 | 1.00 | 1.00 | 0.30 | 1.00 | 1.00 | 1.00 | ||||||||
| PAM | 6 (40) | 7 (54) | 3 (60) | 3 (50) | 3 (80) | 2 (100) | 1 (100) | ||||||||
| Non PAM (nevus/de novo) | 7 (47) | 3 (23) | 1 (20) | 3 (50) | 1 (25) | 0 (0) | 0 (0) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
van Poppelen, N.M.; van Ipenburg, J.A.; van den Bosch, Q.; Vaarwater, J.; Brands, T.; Eussen, B.; Magielsen, F.; Dubbink, H.J.; Paridaens, D.; Brosens, E.; et al. Molecular Genetics of Conjunctival Melanoma and Prognostic Value of TERT Promoter Mutation Analysis. Int. J. Mol. Sci. 2021, 22, 5784. https://doi.org/10.3390/ijms22115784
van Poppelen NM, van Ipenburg JA, van den Bosch Q, Vaarwater J, Brands T, Eussen B, Magielsen F, Dubbink HJ, Paridaens D, Brosens E, et al. Molecular Genetics of Conjunctival Melanoma and Prognostic Value of TERT Promoter Mutation Analysis. International Journal of Molecular Sciences. 2021; 22(11):5784. https://doi.org/10.3390/ijms22115784
Chicago/Turabian Stylevan Poppelen, Natasha M., Jolique A. van Ipenburg, Quincy van den Bosch, Jolanda Vaarwater, Tom Brands, Bert Eussen, Frank Magielsen, Hendrikus J. Dubbink, Dion Paridaens, Erwin Brosens, and et al. 2021. "Molecular Genetics of Conjunctival Melanoma and Prognostic Value of TERT Promoter Mutation Analysis" International Journal of Molecular Sciences 22, no. 11: 5784. https://doi.org/10.3390/ijms22115784
APA Stylevan Poppelen, N. M., van Ipenburg, J. A., van den Bosch, Q., Vaarwater, J., Brands, T., Eussen, B., Magielsen, F., Dubbink, H. J., Paridaens, D., Brosens, E., Naus, N., de Klein, A., Kiliç, E., & Verdijk, R. M. (2021). Molecular Genetics of Conjunctival Melanoma and Prognostic Value of TERT Promoter Mutation Analysis. International Journal of Molecular Sciences, 22(11), 5784. https://doi.org/10.3390/ijms22115784







