Antimicrobial and Anti-Biofilm Peptide Octominin for Controlling Multidrug-Resistant Acinetobacter baumannii
Abstract
1. Introduction
2. Results
2.1. Isolation and Molecular Identification of A. baumannii
2.2. Antibiotic Sensitivity Spectrum of A. baumannii
2.3. MIC and MBC of A. baumannii Treated with Octominin
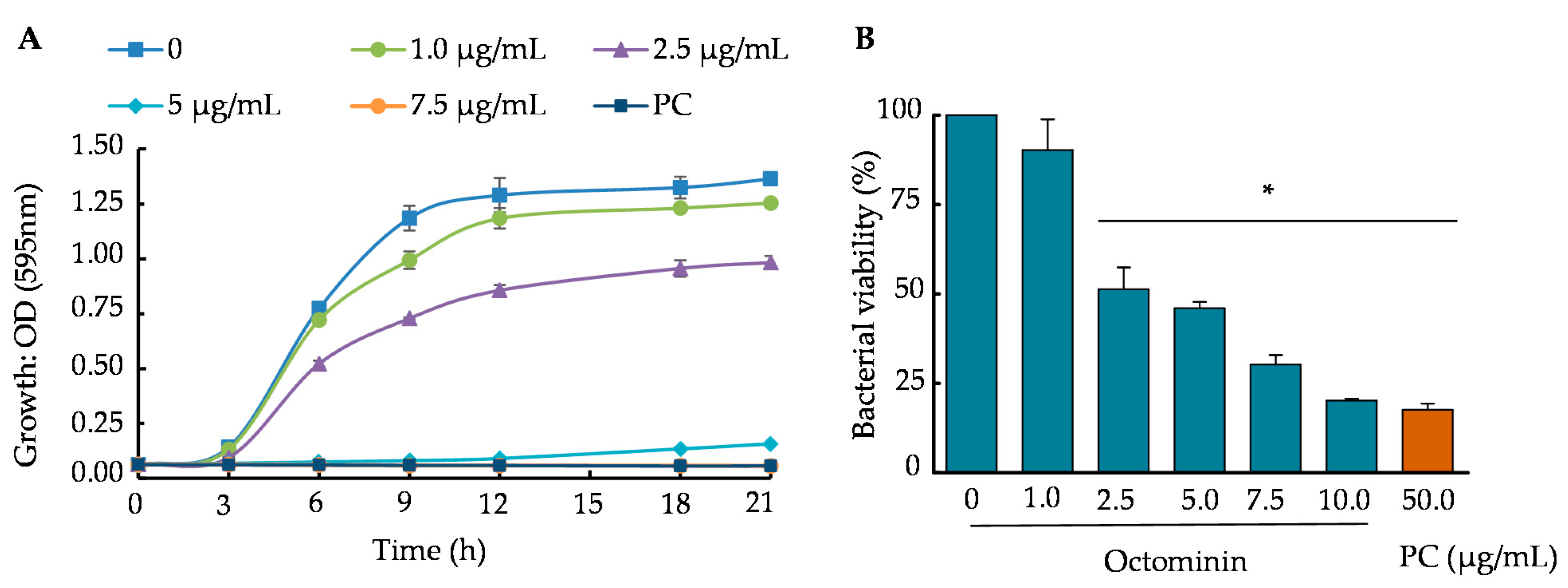
2.4. Time-Kill Kinetics and Bacterial Viability of A. baumannii upon Octominin Treatment
2.5. Morphological Changes in A. baumannii Treated with Octominin
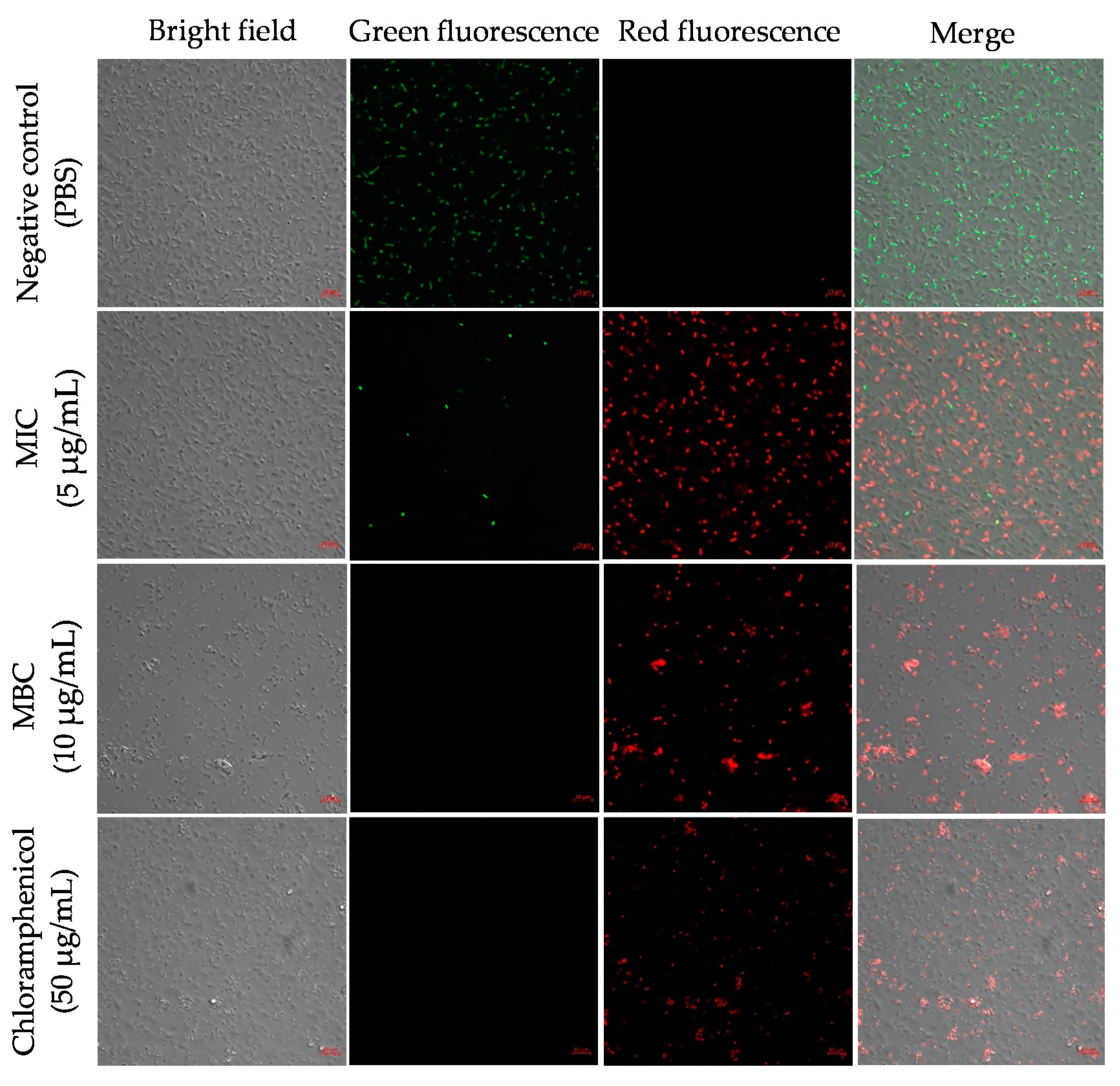
2.6. Octominin Effects on A. baumannii Membrane Permeability and Cell Death
2.7. ROS Generation in A. baumannii upon Exposure to Octominin
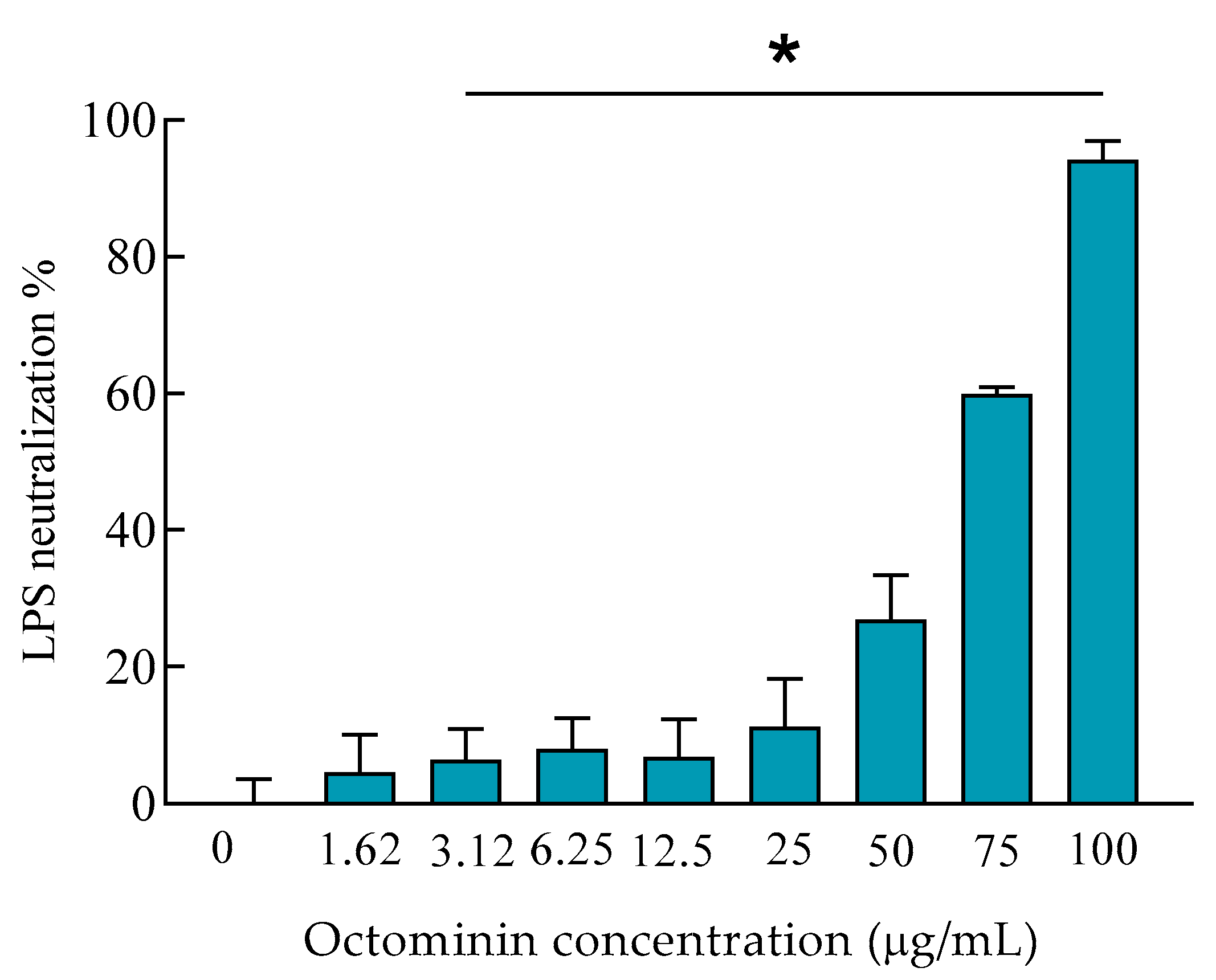
2.8. LPS Neutralization Activity of Octominin
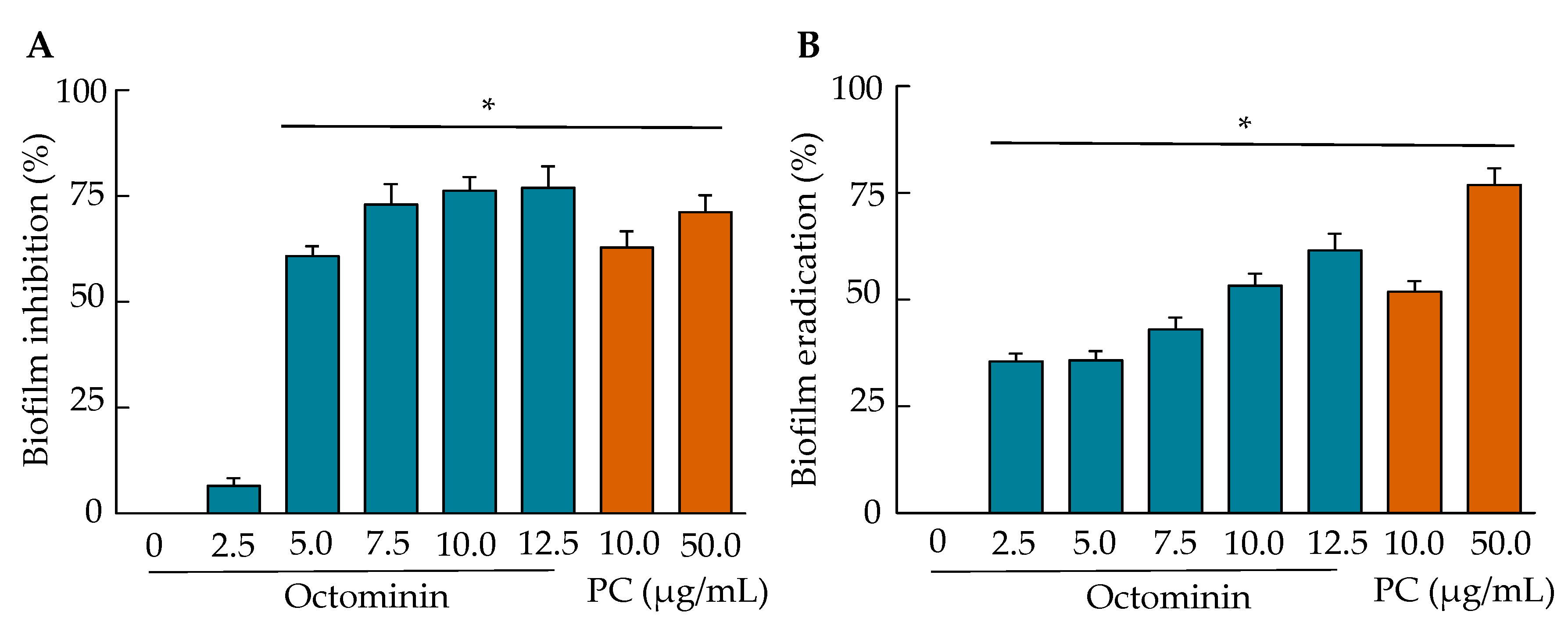
2.9. Biofilm Inhibition and Biofilm Eradication Activities of Octominin
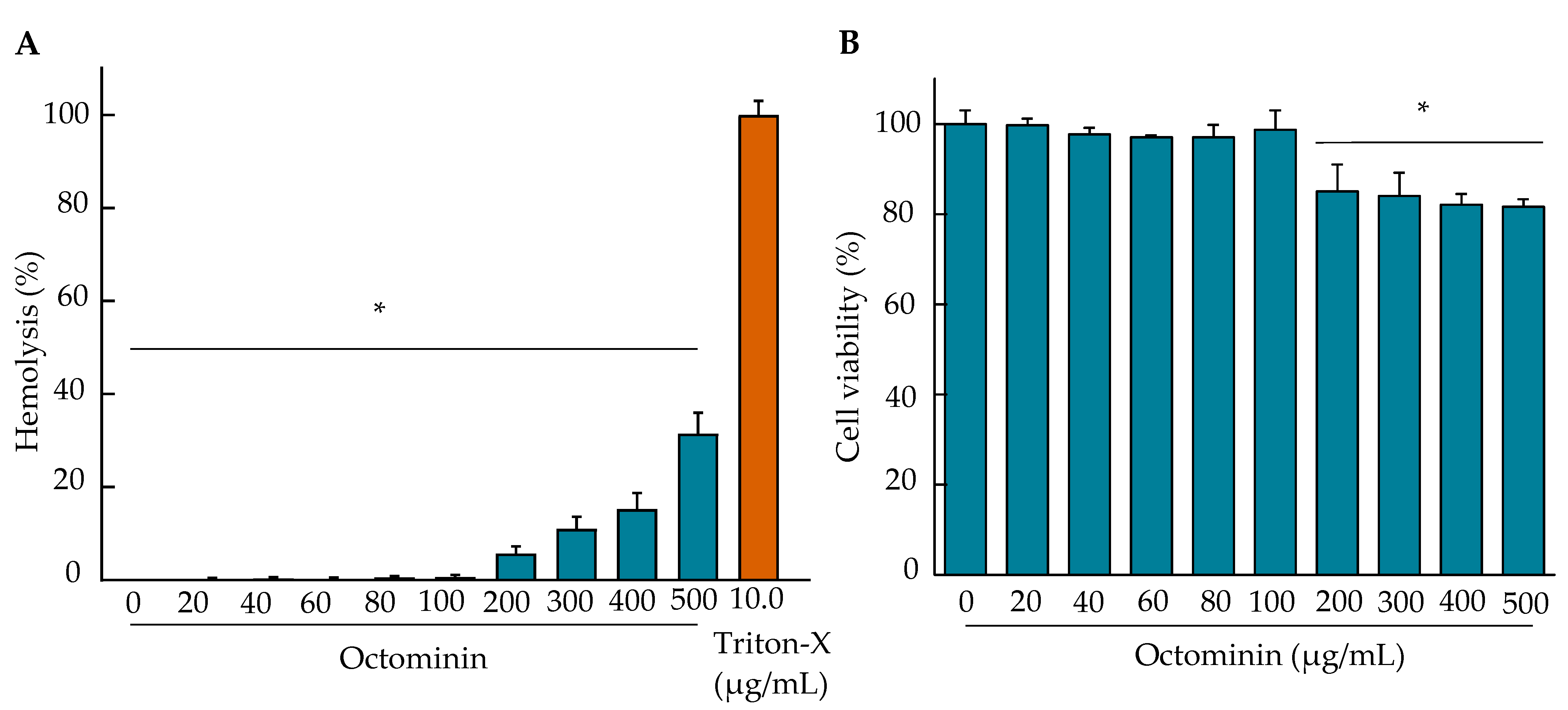
2.10. Hemolysis Activity and Cytotoxicity of Octominin
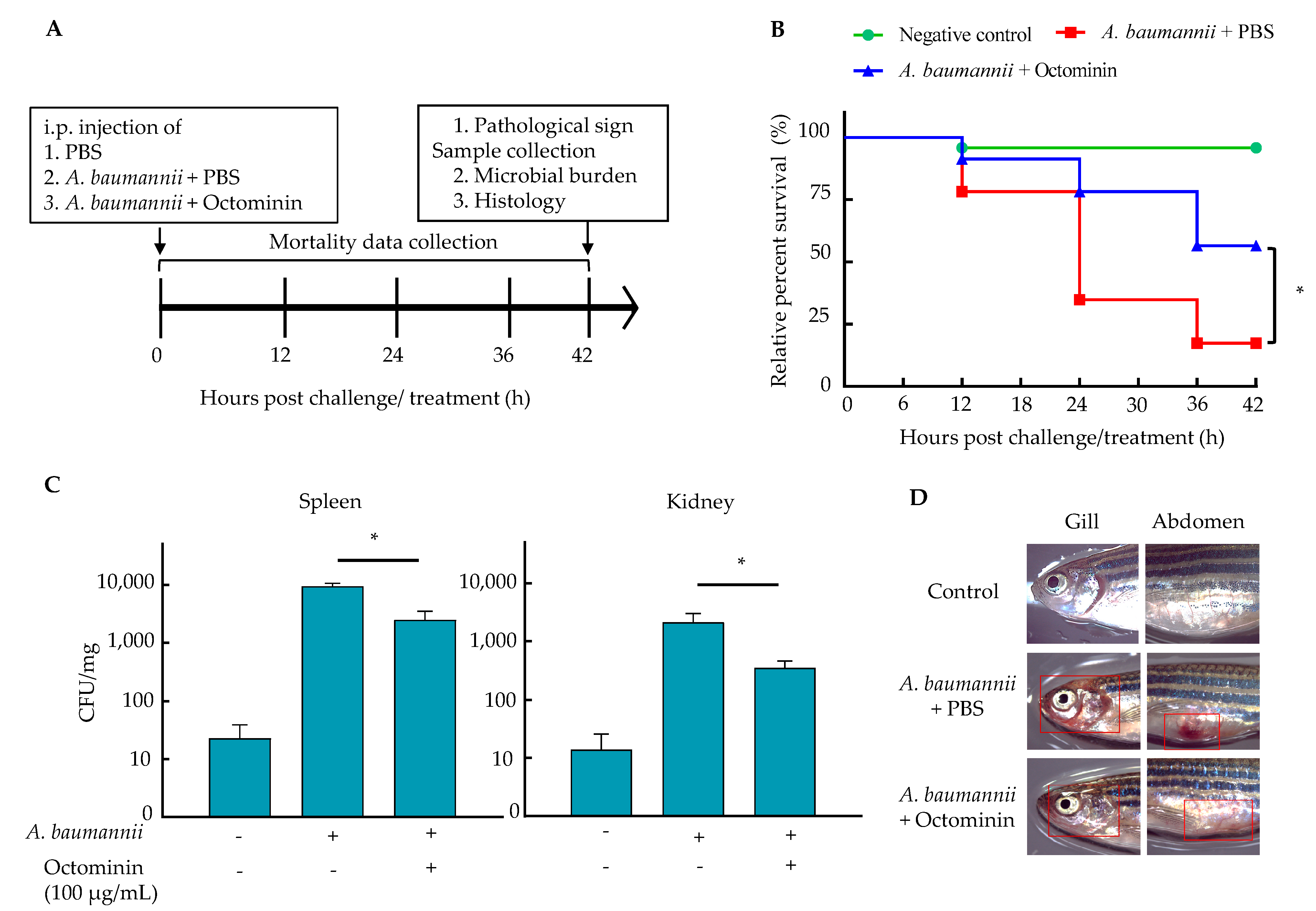
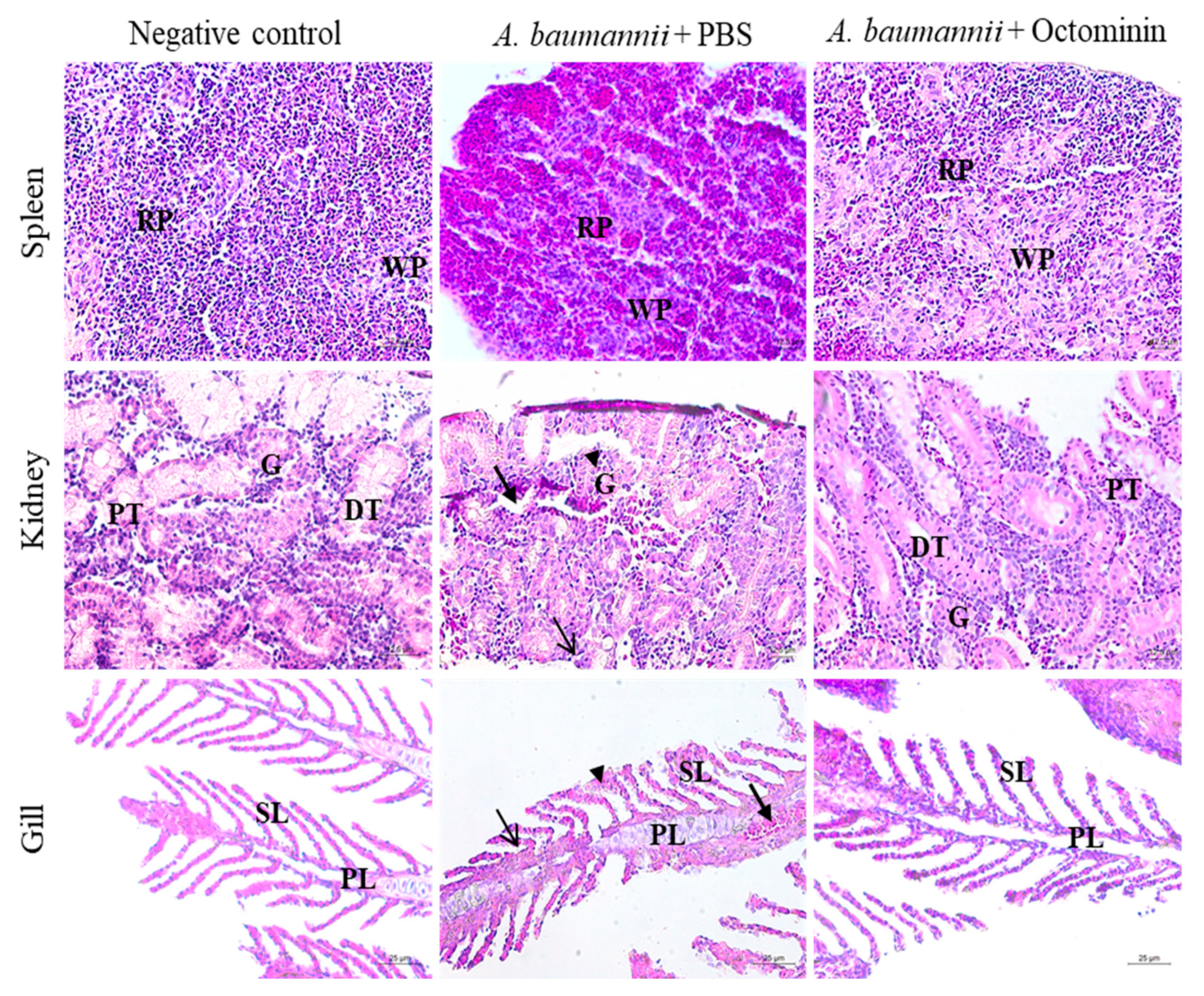
2.11. Efficiency of A. baumannii Control by Octominin in a Zebrafish Model
3. Discussion
4. Materials and Methods
4.1. A. baumannii (Lab Strain 1), Media, and Growth Conditions
4.2. Determination of the Antibiotic Sensitivity Spectrum of A. baumannii
4.3. Determination of the Time-Kill Kinetics, MIC, and MBC of Octominin for A. baumannii
4.4. Determination of A. baumannii Cell Viability after Octominin Treatment
4.5. Effect of Octominin on Morphological Changes in A. baumannii
4.6. Effect of Octominin on Membrane Permeability Alteration in A. baumannii
4.7. Effect of Octominin on ROS Generation in A. baumannii
4.8. Effect of Octominin on LPS Neutralization
4.9. Effect of Octominin on A. baumannii Biofilm Formation Inhibition and Eradication
4.10. Determination of Octominin Toxicity
4.10.1. Hemolysis Assay of Octominin
4.10.2. Cytotoxicity of Octominin against Murine Macrophage Cells
4.11. Determination of the In Vivo Efficiency of Octominin in Controlling A. baumannii Infection in a Zebrafish Model
4.12. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Peleg, A.Y.; Seifert, H.; Paterson, D.L. Acinetobacter baumannii: Emergence of a successful pathogen. Clin. Microbiol. Rev. 2008, 21, 538–582. [Google Scholar] [CrossRef]
- Gerner-Smidt, P. Ribotyping of the Acinetobacter calcoaceticus-Acinetobacter baumannii complex. J. Clin. Microbiol. 1992, 30, 2680–2685. [Google Scholar] [CrossRef] [PubMed]
- Wong, D.; Nielsen, T.B.; Bonomo, R.A.; Pantapalangkoor, P.; Luna, B.; Spellberg, B. Clinical and Pathophysiological Overview of Acinetobacter Infections: A Century of Challenges. Clin. Microbiol. Rev. 2017, 30, 409–447. [Google Scholar] [CrossRef]
- Vázquez-López, R.; Solano-Gálvez, S.G.; Juárez Vignon-Whaley, J.J.; Abello Vaamonde, J.A.; Padró Alonzo, L.A.; Rivera Reséndiz, A.; Muleiro Álvarez, M.; Vega López, E.N.; Franyuti-Kelly, G.; Álvarez-Hernández, D.A.; et al. Acinetobacter baumannii Resistance: A Real Challenge for Clinicians. Antibiotics 2020, 9, 205. [Google Scholar] [CrossRef] [PubMed]
- Kurihara, M.N.L.; Sales, R.O.; Silva, K.E.D.; Maciel, W.G.; Simionatto, S. Multidrug-resistant Acinetobacter baumannii outbreaks: A global problem in healthcare settings. Rev. Soc. Bras. Med. Trop. 2020, 53. [Google Scholar] [CrossRef]
- Kyriakidis, I.; Vasileiou, E.; Pana, Z.D.; Tragiannidis, A. Acinetobacter baumannii Antibiotic Resistance Mechanisms. Pathogens 2021, 10, 373. [Google Scholar] [CrossRef] [PubMed]
- Xie, R.; Zhang, X.D.; Zhao, Q.; Peng, B. Analysis of global prevalence of antibiotic resistance in Acinetobacter baumannii infections disclosed a faster increase in OECD countries. Emerg. Microbes Infect. 2018, 14, 31. [Google Scholar] [CrossRef]
- Inchai, J.; Pothirat, C.; Bumroongkit, C.; Limsukon, A.; Khositsakulchai, W. Prognostic factors associated with mortality of drug-resistant Acinetobacter baumannii ventilator-associated pneumonia. J. Intensive Care. 2015, 2, 1–8. [Google Scholar] [CrossRef]
- Chen, C.; Lin, L.; Chang, Y.; Chen, Y.; Chang, C. Infection control programs and antibiotic control programs to limit transmission of multi-drug resistant Acinetobacter baumannii infections: Evolution of old problems and new challenges for institutes. Int. J. Environ. Res. Public Health. 2015, 12, 8871–8882. [Google Scholar] [CrossRef] [PubMed]
- Biswaro, L.S.; Mauricio, G.; Sousa, C.; Rezende, T.M.B.; Dias, S.C.; Franco, O.L. Antimicrobial peptides and nanotechnology, recent advances and challenges. Front. Microbiol. 2018, 9, 1–14. [Google Scholar] [CrossRef]
- Bahar, A.A.; Ren, D. Antimicrobial peptides. Pharmaceuticals 2013, 28, 1543–1575. [Google Scholar] [CrossRef]
- Saikia, K.; Sravani, Y.D.; Ramakrishnan, V.; Chaudhary, N. Highly potent antimicrobial peptides from N-terminal membrane-binding region of E. coli MreB. Nat. Publ. Gr. 2017, 23, 1–9. [Google Scholar] [CrossRef]
- Nguyen, L.T.; Haney, E.F.; Vogel, H.J. The expanding scope of antimicrobial peptide structures and their modes of action. Trends Biotechnol. 2011, 29, 464–472. [Google Scholar] [CrossRef]
- Lehmann, J.; Retz, M.; Sidhu, S.S.; Suttmann, H.; Sell, M.; Unteregger, G.; Sto, M.; Paulsen, F. Antitumor activity of the antimicrobial peptide magainin II against bladder cancer cell lines. Eur. Urol. 2006, 50, 141–147. [Google Scholar] [CrossRef]
- Guarna, M.M.; Coulson, R.; Rubinchik, E. Anti-inflammatory activity of cationic peptides: Application to the treatment of acne vulgaris. FEMS Microbiol. Lett. 2006, 257, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Ramos, R.; Pedro, J.; Cristina, A.; Costa, R.; Guardão, L.; Schmitt, F.; Soares, R.; Vilanova, M.; Domingues, L.; Gama, M. Wound healing activity of the human antimicrobial peptide LL37. Peptides 2011, 32, 1469–1476. [Google Scholar] [CrossRef]
- Yang, N.; Liu, X.; Teng, D.; Li, Z.; Wang, X.; Mao, R. Antibacterial and detoxifying activity of NZ17074 analogues with multi-layers of selective antimicrobial actions against Escherichia coli and Salmonella enteritidis. Sci. Rep. 2017, 7, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Bulet, P.; Hetru, C.; Dimarcq, J. Antimicrobial peptides in insects: Structure and function. Dev. Comp. Immunol. 1999, 23, 4–5. [Google Scholar] [CrossRef]
- Wang, G. Database-guided discovery of potent peptides to combat HIV-1 or superbugs. Pharmaceuticals 2013, 6, 728–758. [Google Scholar] [CrossRef]
- Zorko, M.; Hafner-bratkovi, I.; Jerala, R. Expression, purification and structural studies of a short antimicrobial peptide. Biochim Biophys Acta. 2009, 1788, 314–323. [Google Scholar] [CrossRef]
- Lei, J.; Sun, L.; Huang, S.; Zhu, C.; Li, P.; He, J.; Mackey, V.; Coy, D.H. The antimicrobial peptides and their potential clinical applications. Am. J. Transl. Res. 2019, 11, 3919–3931. [Google Scholar] [PubMed]
- Nikapitiya, C.; Dananjaya, S.H.S.; Chandrarathna, H.P.S.U.; De Zoysa, M.; Whang, I. Octominin: A novel synthetic anticandidal peptide derived from defense protein of Octopus minor. Mar. Drugs 2020, 18, 56. [Google Scholar] [CrossRef] [PubMed]
- Mwangi, J.; Yin, Y.; Wang, G.; Yang, M.; Li, Y.; Zhang, Z.; Lai, R. The antimicrobial peptide ZY4 combats multidrug- resistant Pseudomonas aeruginosa and Acinetobacter baumannii infection. Proc. Natl. Acad. Sci. USA 2019, 116, 1–7. [Google Scholar] [CrossRef]
- Lee, C.; Lee, J.H.; Park, M.; Park, K.S.; Bae, I.K. Biology of Acinetobacter baumannii: Pathogenesis, antibiotic resistance mechanisms, and prospective treatment options. Front. Cell. Infect. Microbiol. 2017, 7. [Google Scholar] [CrossRef]
- Son, Y.W.; Jung, I.Y.; Ahn, M.Y.; Jeon, Y.D.; Ann, H.W.; Ahn, J.Y.; Ku, N.S.; Han, S.H.; Choi, J.Y.; Song, Y.G.; et al. A case of community-acquired pneumonia caused by multidrug-resistant Acinetobacter baumannii in Korea. Infect. Chemother. 2017, 49, 297–300. [Google Scholar] [CrossRef]
- Fair, R.J.; Tor, Y. Perspectives in medicinal chemistry antibiotics and bacterial resistance in the 21st century. Perspect. Medicin. Chem. 2014, 6, 25–64. [Google Scholar] [CrossRef]
- Asanka Sanjeewa, K.K.; Nagahawatta, D.P.; Yang, H.W.; Oh, J.Y.; Jayawardena, T.U.; Jeon, Y.J.; De Zoysa, M.; Whang, I.; Ryu, B. Octominin inhibits LPS-induced chemokine and pro-inflammatory cytokine secretion from raw 264.7 macrophages via blocking TLRS/nf-κb signal transduction. Biomolecules 2020, 10, 511. [Google Scholar] [CrossRef]
- Guo, Y.; Wang, L.; Lei, J.; Xu, J.; Han, L. Antimicrobial and antibiofilm activity of human cationic Antibacterial peptide (LL-37) and its analogs against pan-drug-resistant Acinetobacter baumannii. Jundishapur J. Microbiol. 2017, 10, 4–10. [Google Scholar] [CrossRef]
- Jaśkiewicz, M.; Neubauer, D.; Kazor, K.; Bartoszewska, S.; Kamysz, W. Antimicrobial activity of selected antimicrobial peptides against planktonic culture and biofilm of Acinetobacter baumannii. Probiotics Antimicrob. Proteins. 2019, 1, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Traczewski, M.M.; Katz, B.D.; Steenbergen, J.N.; Brown, S.D. Inhibitory and bactericidal activities of daptomycin, vancomycin, and teicoplanin against methicillin-resistant Staphylococcus aureus isolates collected from 1985 to 2007. Antimicrob. Agents Chemother. 2009, 53, 1735–1738. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.K.; Kang, N.; Ko, S.J.; Park, J.; Park, E.; Shin, D.W.; Kim, S.H.; Lee, S.A.; Lee, J.I.; Lee, S.H.; et al. Antibacterial and antibiofilm activity and mode of action of magainin 2 against drug-resistant Acinetobacter baumannii. Int. J. Mol. Sci. 2018, 19, 3041. [Google Scholar] [CrossRef]
- Thennarasu, S.; Tan, A.; Penumatchu, R.; Shelburne, C.E.; Heyl, D.L.; Ramamoorthy, A. Antimicrobial and membrane-disrupting activities of a peptide derived from the human cathelicidin antimicrobial peptide ll37. Biophys. J. 2010, 98, 248–257. [Google Scholar] [CrossRef]
- Vila-Farrés, X.; López-Rojas, R.; Pachón-Ibánez, M.E.; Teixidó, M.; Pachón, J.; Vila, J.; Giralt, E. Sequence-activity relationship, and mechanism of action of mastoparan analogues against extended-drug resistant Acinetobacter baumannii. Eur. J. Med. Chem. 2015, 101, 34–40. [Google Scholar] [CrossRef]
- Nagarajan, D.; Roy, N.; Kulkarni, O.; Nanajkar, N.; Datey, A.; Ravichandran, S.; Thakur, C.; Sandeep, T.; Aprameya, I.V.; Sarma, S.P. A designed antimicrobial peptide to combat carbapenem- and tigecycline-resistant Acinetobacter baumannii. Sci. Adv. 2019, 5. [Google Scholar] [CrossRef]
- Zhao, X.; Drlica, K. Reactive oxygen species and the bacterial response to lethal stress. Curr. Opin. Microbiol. 2014, 21, 1–6. [Google Scholar] [CrossRef]
- Wang, C.; Zhao, G.; Wang, S.; Chen, Y.; Gong, Y.; Chen, S.; Xu, Y.; Hu, M.; Wang, X.; Zeng, H.; et al. A simplified derivative of human defensin 5 with potent and efficient activity against multidrug-resistant Acinetobacter baumannii. Antimicrob. Agents Chemother. 2018, 62, 1–14. [Google Scholar] [CrossRef]
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Pessarakli, M. Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J. Bot. 2012, 2012. [Google Scholar] [CrossRef]
- Singh, A.K.; Jiang, Y. Lipopolysaccharide (LPS) induced activation of the immune system in control rats and rats chronically exposed to a low level of the organ thiophosphate insecticide, acephate. Toxicol. Ind. Health 2003, 19, 93–108. [Google Scholar] [CrossRef]
- Pulido, D.; Nogués, M.V.; Boix, E.; Torrent, M. Lipopolysaccharide neutralization by antimicrobial peptides: A gambit in the innate host defense strategy. J Innate Immun. 2012, 4, 327–336. [Google Scholar] [CrossRef]
- Unno, Y.; Sato, Y.; Nishida, S.; Nakano, A.; Nakano, R.; Ubagai, T.; Ono, Y. Acinetobacter baumannii Lipopolysaccharide Influences Adipokine Expression in 3T3-L1 Adipocytes. Mediat. Inflamm. 2017, 2017. [Google Scholar] [CrossRef]
- Donlan, R.M. Biofilms: Microbial life on surfaces. Emerg. Infect. Dis. 2002, 8, 881–890. [Google Scholar] [CrossRef] [PubMed]
- Stewart, P.S. Mechanisms of antibiotic resistance in bacterial biofilms. Int. J. Med. Microbiol. 2002, 292, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.K.; Yeung, A.T.Y.; Hancock, R.E.W. Antibiotic resistance in Pseudomonas aeruginosa biofilms: Towards the development of novel anti-biofilm therapies. J. Biotechnol. 2014, 191, 121–130. [Google Scholar] [CrossRef]
- Pletzer, D.; Hancock, R.E.W. Antibiofilm peptides: Potential as broad-spectrum agents. J. Bacteriol. 2016, 198, 2572–2578. [Google Scholar] [CrossRef]
- Peng, J.; Long, H.; Liu, W.; Wu, Z.; Wang, T.; Zeng, Z.; Guo, G.; Wu, J. Antibacterial mechanism of peptide Cec4 against Acinetobacter baumannii. Infect. Drug Resist. 2019, 12, 2417–2428. [Google Scholar] [CrossRef]
- Cruz, C.D.; Shah, S.; Tammela, P. Defining conditions for biofilm inhibition and eradication assays for gram-positive clinical reference strains. BMC Microbiol. 2018, 18, 1–9. [Google Scholar] [CrossRef]
- Laverty, G.; Gilmore, B. Cationic antimicrobial peptide cytotoxicity. SOJ Microbiol. Infect. Dis. 2014, 2, 1–8. [Google Scholar] [CrossRef]
- Naomi, R.; Kishi, I.; Stach-machado, D.; Singulani, J.D.L.; Tavares, C.; Fusco-almeida, A.M.; Cilli, E.M.; Picchi, S.C.; Machado, M.A.; Freitas-astu, J. Evaluation of cytotoxicity features of antimicrobial peptides with potential to control bacterial diseases of citrus. PLoS ONE 2018, 13, 1–18. [Google Scholar] [CrossRef]
- Harris, G.; Lee, K.; Lam, C.K.; Kanzaki, G.; Patel, G.B.; Xu, H.H. A mouse model of Acinetobacter baumannii-associated pneumonia using a clinically isolated hypervirulent strain. Antimicrob. Agents. Chemother. 2013, 57, 3601–3613. [Google Scholar] [CrossRef]
- Abdelhamed, H.; Ibrahim, I.; Baumgartner, W.; Lawrence, M.L.; Shaw, J. Characterization of histopathological and ultrastructural changes in channel catfish experimentally infected with virulent Aeromonas hydrophila. Front. Microbiol. 2017, 8, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Dananjaya, S.H.S.; Udayangani, R.M.C.; Oh, C.; Nikapitiya, C.; Lee, J.; De Zoysa, M. Green synthesis, physio-chemical characterization and anti-candidal function of a biocompatible chitosan gold nanocomposite as a promising antifungal therapeutic agent. RSC Adv. 2017, 15, 9182–9193. [Google Scholar] [CrossRef]
- Thulshan Jayathilaka, E.H.T.; Liyanage, T.D.; Rajapaksha, D.C.; Dananjaya, S.H.S.; Nikapitiya, C.; Whang, I.; De Zoysa, M. Octominin: An antibacterial and anti-biofilm peptide for controlling the multidrug resistance and pathogenic Streptococcus parauberis. Fish Shellfish. Immunol. 2021, 110, 23–34. [Google Scholar] [CrossRef]
- Madanchi, H.; Kiasari, R.E.; Mousavi, S.J.S.; Johari, B.; Shabani, A.A.; Sardari, S. Design and Synthesis of Lipopolysaccharide-Binding Antimicrobial Peptides Based on Truncated Rabbit and Human CAP18 Peptides and Evaluation of Their Action Mechanism. Probiotics Antimicrob. Proteins 2020, 12, 1582–1593. [Google Scholar] [CrossRef]
- Liyanage, T.D.; Nikapitiya, C.; Lee, J.; De Zoysa, M. Molecular insight into regulation of miRNAs in the spleen of zebrafish (Danio rerio) upon pathogenic Streptococcus parauberis infection. Fish Shellfish. Immunol. 2020, 106, 898–909. [Google Scholar] [CrossRef]


| Antibiotic Class | Antibiotic | Diameter of Zone of Inhibition (mm) | Antibiotic Sensitivity Level |
|---|---|---|---|
| Aminoglycoside | Streptomycin (300 µg) | 13 | Resistant |
| Gentamycin (120 µg) | 14 | Resistant | |
| Amikacin (30 µg) | 15 | Intermediate | |
| Tetracyclines | Tetracycline (30 µg) | 16 | Intermediate |
| Doxycycline (30 µg) | 20 | Susceptible | |
| Fluoroquinolone | Ciprofloxacin (5 µg) | 19 | Intermediate |
| Ofloxacin (5 µg) | 17 | Susceptible | |
| Glycopeptide | Vancomycin (30 µg) | 0 | Resistant |
| Macrolides | Erythromycin (15 µg) | 0 | Resistant |
| Tobramycin (10 µg) | 11 | Resistant | |
| Cephalosporin | Cefotaxime (30 µg) | 10 | Resistant |
| Penicillin | Penicillin (10 U) | 0 | Resistant |
| Lincosamide | Clindamycin (2 µg) | 0 | Resistant |
| Carbapenem | Imipenem (10 µg) | 25 | Susceptible |
| Folic acid synthesis inhibitor | Sulfamethoxazole/ Trimethoprim (23.75/1.25 µg) | 15 | Intermediate |
| Diaminopyrimidine | Trimethoprim (5 µg) | 0 | Resistant |
| Rifamycin | Rifampin (5 µg) | 0 | Resistant |
| Chloramphenicol | Chloramphenicol (30 µg) | 22 | Susceptible |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jayathilaka, E.H.T.T.; Rajapaksha, D.C.; Nikapitiya, C.; De Zoysa, M.; Whang, I. Antimicrobial and Anti-Biofilm Peptide Octominin for Controlling Multidrug-Resistant Acinetobacter baumannii. Int. J. Mol. Sci. 2021, 22, 5353. https://doi.org/10.3390/ijms22105353
Jayathilaka EHTT, Rajapaksha DC, Nikapitiya C, De Zoysa M, Whang I. Antimicrobial and Anti-Biofilm Peptide Octominin for Controlling Multidrug-Resistant Acinetobacter baumannii. International Journal of Molecular Sciences. 2021; 22(10):5353. https://doi.org/10.3390/ijms22105353
Chicago/Turabian StyleJayathilaka, E. H. T. Thulshan, Dinusha C. Rajapaksha, Chamilani Nikapitiya, Mahanama De Zoysa, and Ilson Whang. 2021. "Antimicrobial and Anti-Biofilm Peptide Octominin for Controlling Multidrug-Resistant Acinetobacter baumannii" International Journal of Molecular Sciences 22, no. 10: 5353. https://doi.org/10.3390/ijms22105353
APA StyleJayathilaka, E. H. T. T., Rajapaksha, D. C., Nikapitiya, C., De Zoysa, M., & Whang, I. (2021). Antimicrobial and Anti-Biofilm Peptide Octominin for Controlling Multidrug-Resistant Acinetobacter baumannii. International Journal of Molecular Sciences, 22(10), 5353. https://doi.org/10.3390/ijms22105353






