Vibrational Spectroscopy in Assessment of Early Osteoarthritis—A Narrative Review
Abstract
1. Introduction
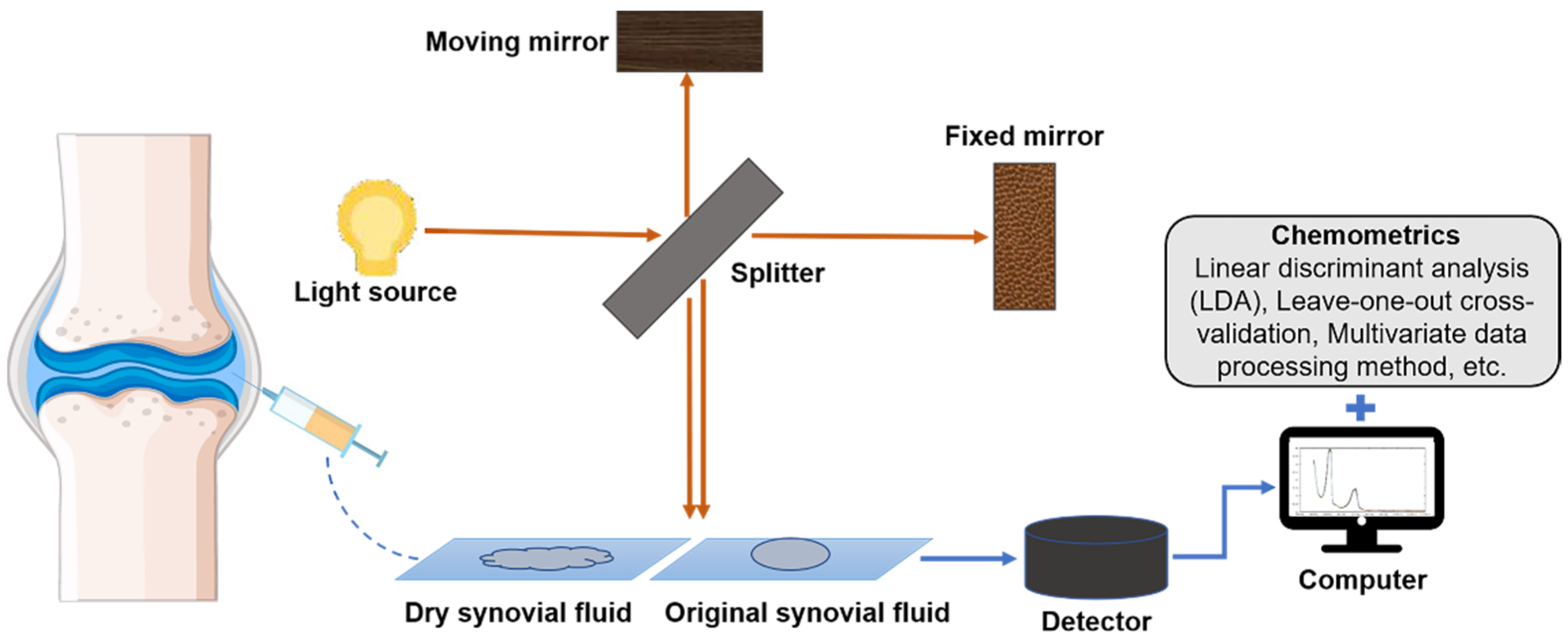
2. NIR Spectroscopy
2.1. Application of NIR Spectroscopy on OA SF Analysis
2.2. Application of NIR Spectroscopy on OA Cartilage Analysis
3. Infrared Spectroscopy
3.1. Application of IR Spectroscopy to SF Analysis in OA Subsection
3.2. Application of FTIR Imaging on Articular Cartilage Analysis in OA
3.3. Application of FTIR in OA In Situ Analysis
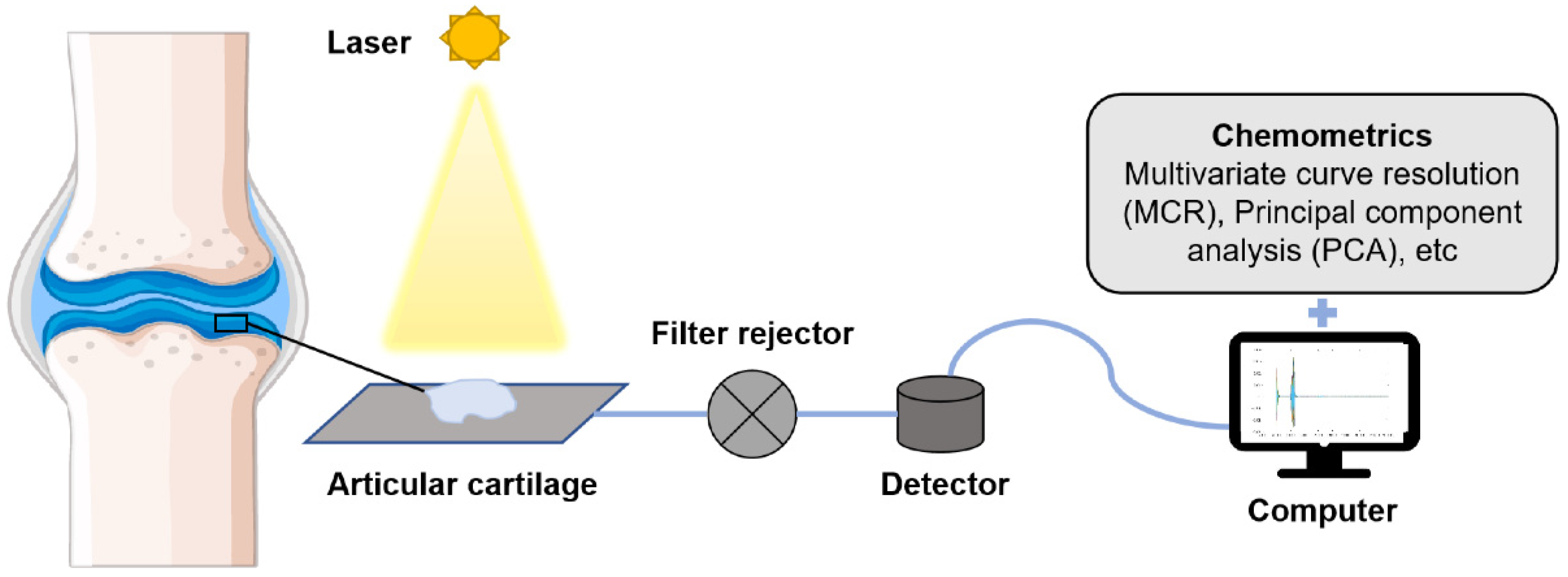
4. Raman Spectroscopy
4.1. Application of Raman Spectroscopy on OA SF Analysis
4.2. Application of Raman Spectroscopy in Subchondral Bone Analysis in OA
4.3. Raman Spectroscopy Analysis of Articular Cartilage in OA
5. Summary and Prospect
Author Contributions
Funding
Conflicts of Interest
References
- Wang, Q.; Rozelle, A.L.; Lepus, C.M.; Scanzello, C.R.; Song, J.J.; Larsen, D.M.; Crish, J.F.; Bebek, G.; Ritter, S.Y.; Lindstrom, T.M.; et al. Identification of a central role for complement in osteoarthritis. Nat. Med. 2011, 17, 1674–1679. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.; Sharma, A.R.; Chakraborty, C.; Saibaba, B.; Ahn, M.E.; Lee, S.S. Review of Prospects of Biological Fluid Biomarkers in Osteoarthritis. Int. J. Mol. Sci. 2017, 18, 601. [Google Scholar] [CrossRef] [PubMed]
- Safiri, S.; Kolahi, A.A.; Smith, E.; Hill, C.; Bettampadi, D.; Mansournia, M.A.; Hoy, D.; Ashrafi-Asgarabad, A.; Sepidarkish, M.; Almasi-Hashiani, A.; et al. Global, regional and national burden of osteoarthritis 1990–2017: A systematic analysis of the Global Burden of Disease Study 2017. Ann. Rheum. Dis. 2020, 79, 819–828. [Google Scholar] [CrossRef]
- Collins, J.E.; Losina, E.; Nevitt, M.C.; Roemer, F.W.; Guermazi, A.; Lynch, J.A.; Katz, J.N.; Kent Kwoh, C.; Kraus, V.B.; Hunter, D.J. Semiquantitative Imaging Biomarkers of Knee Osteoarthritis Progression: Data From the Foundation for the National Institutes of Health Osteoarthritis Biomarkers Consortium. Arthritis Rheumatol. 2016, 68, 2422–2431. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, K.V.; Sanmartin, C.; Langlois, K.; Marshall, D.A. Symptom onset, diagnosis and management of osteoarthritis. Health Rep. 2014, 25, 10–17. [Google Scholar] [PubMed]
- Bitton, R. The economic burden of osteoarthritis. Am. J. Manag. Care 2009, 15, S230–S235. [Google Scholar]
- Gamble, R.; Wyeth-Ayerst, J.; Johnson, E.L. Recommendations for the medical management of osteoarthritis of the hip and knee: 2000 update. American College of Rheumatology Subcommittee on Osteoarthritis Guidelines. Arthritis Rheum. 2000, 43, 1905–1915. [Google Scholar] [CrossRef]
- Bobinac, D.; Spanjol, J.; Zoricic, S.; Maric, I. Changes in articular cartilage and subchondral bone histomorphometry in osteoarthritic knee joints in humans. Bone 2003, 32, 284–290. [Google Scholar] [CrossRef]
- Garvican, E.R.; Vaughan-Thomas, A.; Clegg, P.D.; Innes, J.F. Biomarkers of cartilage turnover. Part 2: Non-collagenous markers. Vet. J. 2010, 185, 43–49. [Google Scholar] [CrossRef]
- Neame, P.J.; Tapp, H.; Azizan, A. Noncollagenous, nonproteoglycan macromolecules of cartilage. Cell Mol. Life Sci. 1999, 55, 1327–1340. [Google Scholar] [CrossRef]
- Chen, Y.; Jiang, W.; Yong, H.; He, M.; Yang, Y.; Deng, Z.; Li, Y. Macrophages in osteoarthritis: Pathophysiology and therapeutics. Am. J. Transl. Res. 2020, 12, 261–268. [Google Scholar]
- Dall’Ara, E.; Ohman, C.; Baleani, M.; Viceconti, M. Reduced tissue hardness of trabecular bone is associated with severe osteoarthritis. J. Biomech. 2011, 44, 1593–1598. [Google Scholar] [CrossRef]
- Yang, Y.T.; Li, P.R.; Zhu, S.S.; Bi, R.Y. Comparison of early-stage changes of osteoarthritis in cartilage and subchondral bone between two different rat models. Peerj 2020, 8. [Google Scholar] [CrossRef]
- Le, T.K.; Montejano, L.B.; Cao, Z.; Zhao, Y.; Ang, D. Healthcare costs associated with osteoarthritis in US patients. Pain Pract. 2012, 12, 633–640. [Google Scholar] [CrossRef]
- Zhao, J.; Link, T.M. MRI in degenerative arthritides: Structural and clinical aspects. Ann. N. Y. Acad. Sci. 2009, 1154, 115–135. [Google Scholar] [CrossRef]
- Chaudhari, A.S.; Kogan, F.; Pedoia, V.; Majumdar, S.; Gold, G.E.; Hargreaves, B.A. Rapid Knee MRI Acquisition and Analysis Techniques for Imaging Osteoarthritis. J. Magn. Reson. Imaging 2020, 52, 1321–1339. [Google Scholar] [CrossRef]
- Zhang, X.M.; Tong, H.Y.; Zhang, J.; Xu, J.Y.; Xia, S.Y. Diagnostic Value of 3.0T MRI in Cartilage Injury Grading of Knee Osteoarthritis. J. Med. Imaging Health Inform. 2020, 10, 2979–2984. [Google Scholar] [CrossRef]
- Pishgar, F.; Guermazi, A.; Roemer, F.W.; Link, T.M.; Demehri, S. Conventional MRI-based subchondral trabecular biomarkers as predictors of knee osteoarthritis progression: Data from the osteoarthritis initiative. Eur. Radiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Herz, P.; Bourquin, S.; Hsiung, P.L.; Ko, T.; Schneider, K.; Fujimoto, J.; Adams, S.; Roberts, M.; Patel, N.; Brezinski, M. Imaging of cartilage degeneration in vivo using ultrahigh resolution OCT. Opt. Coherence Tomogr. Coherence Tech. 2003, 5140, 152–154. [Google Scholar] [CrossRef]
- Kushida, Y.; Ozeki, N.; Mizuno, M.; Katano, H.; Otabe, K.; Tsuji, K.; Koga, H.; Kishima, K.; Soma, Y.; Sekiya, I. Two- and three-dimensional optical coherence tomography to differentiate degenerative changes in a rat meniscectomy model. J. Orthop. Res. 2020, 38, 2592–2600. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Eltit, F.; Yang, X.; Maloufi, S.; Alousaimi, H.; Liu, Q.H.; Huang, L.; Wang, R.Z.; Tang, S. Detecting human articular cartilage degeneration in its early stage with polarization-sensitive optical coherence tomography. Biomed. Opt. Express 2020, 11, 2745–2760. [Google Scholar] [CrossRef] [PubMed]
- Radunovic, G.L.; Pilipovic, N.; Stanisic, M.; Damjanov, N.S. Assessment of knee osteoarthritis: X-ray or ultrasonography? Ann. Rheum. Dis. 2003, 62, 262. [Google Scholar]
- Novakofski, K.D.; Pownder, S.L.; Koff, M.F.; Williams, R.M.; Potter, H.G.; Fortier, L.A. High-Resolution Methods for Diagnosing Cartilage Damage In Vivo. Cartilage 2016, 7, 39–51. [Google Scholar] [CrossRef]
- Rocha, B.D.; Torres, R.C.S. Ultrasonic and radiographic study of laxity in hip joints of young dogs. Arq. Bras. Med. Vet. Zoo. 2007, 59, 90–96. [Google Scholar] [CrossRef]
- Saarakkala, S.; Toyras, J.; Hirvonen, J.; Laasanen, M.S.; Lappalainen, R.; Jurvelin, J.S. Ultrasonic quantitation of superficial degradation of articular cartilage. Ultrasound Med. Biol. 2004, 30, 783–792. [Google Scholar] [CrossRef] [PubMed]
- Viren, T.; Timonen, M.; Tyrvainen, H.; Tiitu, V.; Jurvelin, J.S.; Toyras, J. Ultrasonic evaluation of acute impact injury of articular cartilage in vitro. Osteoarthr. Cartil. 2012, 20, 719–726. [Google Scholar] [CrossRef]
- Shamir, L.; Ling, S.M.; Scott, W.W.; Bos, A.; Orlov, N.; Macura, T.J.; Eckley, D.M.; Ferrucci, L.; Goldberg, I.G. Knee X-Ray Image Analysis Method for Automated Detection of Osteoarthritis. IEEE Trans. Bio-Med. Eng. 2009, 56, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Saleem, M.; Farid, M.S.; Saleem, S.; Khan, M.H. X-ray image analysis for automated knee osteoarthritis detection. Signal. Image Video Process. 2020, 14, 1079–1087. [Google Scholar] [CrossRef]
- Chen, S.B.; Lin, S.B.; Li, Y.Q.; Liu, Y.T. Characteristics of musculoskeletal ultrasound versus X-ray in their differential diagnosis of knee osteoarthritis. Int. J. Clin. Exp. Med. 2020, 13, 8734–8739. [Google Scholar]
- Yong, C.W.; Teo, K.; Murphy, B.P.; Hum, Y.C.; Tee, Y.K.; Xia, K.J.; Lai, K.W. Knee osteoarthritis severity classification with ordinal regression module. Multimed. Tools Appl. 2021. [Google Scholar] [CrossRef]
- Chu, C.R.; Williams, A.; Tolliver, D.; Kwoh, C.K.; Bruno, S.; Irrgang, J.J. Clinical Optical Coherence Tomography of Early Articular Cartilage Degeneration in Patients With Degenerative Meniscal Tears. Arthritis Rheum. 2010, 62, 1412–1420. [Google Scholar] [CrossRef]
- Adarmes, H.; Croxatto, A.; Galleguillos, M.; Gonzalez, E. Concentration of glycosaminoglycan, aldehydes and protein in synovial fluid from normal and damaged equine metacarpophalangeal joints. Arch. Med. Vet. 2006, 38, 47–52. [Google Scholar] [CrossRef]
- dos Santos, C.A.T.; Pascoa, R.N.M.J.; Lopes, J.A. A review on the application of vibrational spectroscopy in the wine industry: From soil to bottle. Trac-Trend Anal. Chem. 2017, 88, 100–118. [Google Scholar] [CrossRef]
- Bocsa, C.D.; Moisoiu, V.; Stefancu, A.; Leopold, L.F.; Leopold, N.; Fodor, D. Knee osteoarthritis grading by resonant Raman and surface-enhanced Raman scattering (SERS) analysis of synovial fluid. Nanomed. Nanotechnol. 2019, 20. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.Y.; Zhao, Y.; Shang, L.W.; Zhu, Y.K.; Fu, J.J.; Lu, Y.F.; Yin, J.H. Research Progress of Raman Spectroscopy Application for Articular Cartilage and Osteoarthritis. Spectrosc. Spect. Anal. 2020, 40, 2029–2034. [Google Scholar] [CrossRef]
- Takahashi, Y.; Sugano, N.; Takao, M.; Sakai, T.; Nishii, T.; Pezzotti, G. Raman spectroscopy investigation of load-assisted microstructural alterations in human knee cartilage: Preliminary study into diagnostic potential for osteoarthritis. J. Mech. Behav. Biomed. Mater. 2014, 31, 77–85. [Google Scholar] [CrossRef]
- Li, L.; Zang, H.; Li, J.; Chen, D.; Li, T.; Wang, F. Identification of anisodamine tablets by Raman and near-infrared spectroscopy with chemometrics. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 127, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Song, Y.; Leng, J.; Jiang, Z.D. Near Infrared Spectroscopy Analysis Technology. Chin. J. Spectrosc. Lab. 2007, 24, 388–395. [Google Scholar]
- Badaro, A.T.; Garcia-Martin, J.F.; Lopez-Barrera, M.D.C.; Barbin, D.F.; Alvarez-Mateos, P. Determination of pectin content in orange peels by near infrared hyperspectral imaging. Food Chem. 2020, 323, 126861. [Google Scholar] [CrossRef]
- Schopf, M.; Wehrli, M.C.; Becker, T.; Jekle, M.; Scherf, K.A. Fundamental characterization of wheat gluten. Eur. Food Res. Technol. 2021, 247, 985–997. [Google Scholar] [CrossRef]
- Thomson, A.L.; Karunaratne, S.B.; Copland, A.; Stayches, D.; McNabb, E.M.; Jacobs, J. Use of traditional, modern, and hybrid modelling approaches for in situ prediction of dry matter yield and nutritive characteristics of pasture using hyperspectral datasets. Anim. Feed. Sci. Tech. 2020, 269. [Google Scholar] [CrossRef]
- Xia, J.A.; Zhang, W.Y.; Zhang, W.X.; Yang, Y.W.; Hu, G.Y.; Ge, D.K.; Liu, H.; Cao, H.X. A cloud computing-based approach using the visible near-infrared spectrum to classify greenhouse tomato plants under water stress. Comput. Electron. Agr. 2021, 181. [Google Scholar] [CrossRef]
- Arndt, M.; Rurik, M.; Drees, A.; Ahlers, C.; Feldmann, S.; Kohlbacher, O.; Fischer, M. Food authentication: Determination of the geographical origin of almonds (Prunus dulcis MILL.) via near-infrared spectroscopy. Microchem. J. 2021, 160. [Google Scholar] [CrossRef]
- Chang, Y.T.; Hsueh, M.C.; Hung, S.P.; Lu, J.M.; Peng, J.H.; Chen, S.F. Prediction of specialty coffee flavors based on near-infrared spectra using machine- and deep-learning methods. J. Sci. Food Agr. 2021. [Google Scholar] [CrossRef]
- Gao, B.; Xu, X.D.; Han, L.J.; Liu, X. A novel near infrared spectroscopy analytical strategy for meat and bone meal species discrimination based on the insight of fraction composition complexity. Food Chem. 2021, 344. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, S.; Genkawa, T.; Miyamoto, A.; Ikehata, A. Useful tissues in cabbage head for freshness evaluation with visible and near infrared spectroscopy. Food Chem. 2021, 339, 128058. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, H.T.; Aoki, W.; Kotani, T.; Kuzuhara, M.; Omiya, M.; Reiners, A.; Zechmeister, M. Elemental abundances of M dwarfs based on high-resolution near-infrared spectra: Verification by binary systems. Publ. Astron. Soc. Jpn. 2020, 72. [Google Scholar] [CrossRef]
- Rubini, M.; Feuillerat, L.; Cabaret, T.; Leroyer, L.; Leneveu, L.; Charrier, B. Comparison of the performances of handheld and benchtop near infrared spectrometers: Application on the quantification of chemical components in maritime pine (Pinus Pinaster) resin. Talanta 2021, 221. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, Y.; Umemura, K. Sensing of epigallocatechin gallate and tannic acid based on near infrared optical spectroscopy of DNA-wrapped single-walled carbon nanotube hybrids. J. Near Infrared Spec. 2020. [Google Scholar] [CrossRef]
- Chen, X.Y.; Sun, X.F.; Hua, H.M.; Yi, Y.; Li, H.L.; Chen, C. Quality evaluation of decoction pieces of Rhizoma Atractylodis Macrocephalae by near infrared spectroscopy coupled with chemometrics. Spectrochim. Acta A 2019, 221. [Google Scholar] [CrossRef] [PubMed]
- Li, L.Q.; Pan, X.P.; Chen, W.L.; Wei, M.M.; Feng, Y.C.; Yin, L.H.; Hu, C.Q.; Yang, H.H. Multi-manufacturer drug identification based on near infrared spectroscopy and deep transfer learning. J. Innov. Opt. Health Sci. 2020, 13. [Google Scholar] [CrossRef]
- Mishra, P.; Nordon, A.; Roger, J.M. Improved prediction of tablet properties with near-infrared spectroscopy by a fusion of scatter correction techniques. J. Pharm. Biomed. 2021, 192. [Google Scholar] [CrossRef]
- Sun, F.; Zhao, W.J.; Wang, K.Y.; Wang, S.M.; Liang, S.W. Near-infrared spectroscopy to assess typhaneoside and isorhamnetin-3-O-glucoside in different processed products of pollen typhae. Spectrosc. Lett. 2019, 52, 423–430. [Google Scholar] [CrossRef]
- Moros, J.; Garrigues, S.; de la Guardia, M. Vibrational spectroscopy provides a green tool for multi-component analysis. Trac-Trend Anal. Chem. 2010, 29, 578–591. [Google Scholar] [CrossRef]
- Bec, K.B.; Grabska, J.; Huck, C.W. Near-Infrared Spectroscopy in Bio-Applications. Molecules 2020, 25, 2948. [Google Scholar] [CrossRef]
- Biancolillo, A.; Marini, F.; Ruckebusch, C.; Vitale, R. Chemometric Strategies for Spectroscopy-Based Food Authentication. Appl. Sci. 2020, 10, 6544. [Google Scholar] [CrossRef]
- Afara, I.O.; Prasadam, I.; Arabshahi, Z.; Xiao, Y.; Oloyede, A. Monitoring osteoarthritis progression using near infrared (NIR) spectroscopy. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Shaw, R.A.; Kotowich, S.; Eysel, H.H.; Jackson, M.; Thomson, G.T.D.; Mantsch, H.H. Arthritis diagnosis based upon the near infrared spectrum of synovial fluid. Rheumatol. Int. 1995, 15, 159–165. [Google Scholar] [CrossRef]
- Mickiewicz, B.; Kelly, J.J.; Ludwig, T.E.; Weljie, A.M.; Wiley, J.P.; Schmidt, T.A.; Vogel, H.J. Metabolic analysis of knee synovial fluid as a potential diagnostic approach for osteoarthritis. J. Orthop. Res. 2015, 33, 1631–1638. [Google Scholar] [CrossRef]
- Mickiewicz, B.; Heard, B.J.; Chau, J.K.; Chung, M.; Hart, D.A.; Shrive, N.G.; Frank, C.B.; Vogel, H.J. Metabolic Profiling of Synovial Fluid in a Unilateral Ovine Model of Anterior Cruciate Ligament Reconstruction of the Knee Suggests Biomarkers for Early Osteoarthritis. J. Orthop. Res. 2015, 33, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Esmonde-White, K.A.; Mandair, G.S.; Raaii, F.; Jacobson, J.A.; Miller, B.S.; Urquhart, A.G.; Roessler, B.J.; Morris, M.D. Raman spectroscopy of synovial fluid as a tool for diagnosing osteoarthritis. J. Biomed. Opt. 2009, 14. [Google Scholar] [CrossRef] [PubMed]
- Chhol, K.Z.; Bykov, V.A.; Nikolaeva, S.S.; Rebrova, G.A.; Roshina, A.A.; Rumjantseva, N.V.; Yakovleva, L.B.; Korolyova, O.A.; Rebrov, L.B. The changes of biochemical characteristics of collagen and nature of water in human osteoarthrotic cartilage. Vopr. Med. Khimii 2001, 47, 498–505. [Google Scholar]
- Hofmann, G.O.; Marticke, J.; Grossstuck, R.; Hoffmann, M.; Lange, M.; Plettenberg, H.K.; Braunschweig, R.; Schilling, O.; Kaden, I.; Spahn, G. Detection and evaluation of initial cartilage pathology in man: A comparison between MRT, arthroscopy and near-infrared spectroscopy (NIR) in their relation to initial knee pain. Pathophysiology 2010, 17, 1–8. [Google Scholar] [CrossRef]
- Murat, N.; Karadam, B.; Ozkal, S.; Karatosun, V.; Gidener, S. Quantification of papain-induced rat osteoarthritis in relation to time with the Mankin score. Acta Orthop. Traumatol. Turc. 2007, 41, 233–237. [Google Scholar]
- van der Sluijs, J.A.; Geesink, R.G.; van der Linden, A.J.; Bulstra, S.K.; Kuyer, R.; Drukker, J. The reliability of the Mankin score for osteoarthritis. J. Orthop. Res. 1992, 10, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Watanabe, H.; Kimata, K. The roles of proteoglycans for cartilage. Clin. Calcium 2006, 16, 1029–1033. [Google Scholar]
- Palukuru, U.P.; Hanifi, A.; McGoverin, C.M.; Devlin, S.; Lelkes, P.I.; Pleshko, N. Near infrared spectroscopic imaging assessment of cartilage composition: Validation with mid infrared imaging spectroscopy. Anal. Chim. Acta 2016, 926, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Buck, R.J.; Wirth, W.; Dreher, D.; Nevitt, M.; Eckstein, F. Frequency and spatial distribution of cartilage thickness change in knee osteoarthritis and its relation to clinical and radiographic covariates—Data from the osteoarthritis initiative. Osteoarthr. Cartil. 2013, 21, 102–109. [Google Scholar] [CrossRef]
- Sarin, J.K.; Te Moller, N.C.R.; Mancini, I.A.D.; Brommer, H.; Visser, J.; Malda, J.; van Weeren, P.R.; Afara, I.O.; Toyras, J. Arthroscopic near infrared spectroscopy enables simultaneous quantitative evaluation of articular cartilage and subchondral bone in vivo. Sci. Rep. 2018, 8, 13409. [Google Scholar] [CrossRef]
- Afara, I.O.; Sarin, J.K.; Ojanen, S.; Finnila, M.A.J.; Herzog, W.; Saarakkala, S.; Korhonen, R.K.; Toyras, J. Machine Learning Classification of Articular Cartilage Integrity Using Near Infrared Spectroscopy. Cell. Mol. Bioeng. 2020, 13, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Sarin, J.K.; Torniainen, J.; Prakash, M.; Rieppo, L.; Afara, I.O.; Toyras, J. Dataset on equine cartilage near infrared spectra, composition, and functional properties. Sci. Data 2019, 6, 164. [Google Scholar] [CrossRef]
- Chen, Y.; Li, C.; Wang, X.; Chu, Q.; Long, Z. Detection of knee osteoarthritis with near infrared spectroscopy in vivo. J. Optoelectron. Laser 2014, 25, 1023–1026. [Google Scholar]
- Kafian-Attari, I.; Semenov, D.; Nippolainen, E.; Hauta-Kasari, M.; Toyras, J.; Afara, I.O. Optical properties of articular cartilage in the near-infrared spectral range are related to its proteoglycan content. Tissue Opt. Photonics 2020, 11363. [Google Scholar] [CrossRef]
- Maddams, W.F.; Willis, H.A. The principles and applications of mathematical peak finding procedures in vibrational spectra IR spectroscopy. Proc. SPIE Int. Soc. Opt. Eng. 1988, 917, 35–46. [Google Scholar]
- Ichimura, K.T.T.; Suzuki, Y. Fourier transform technique and infrared analysis. J. Jpn. Soc. Infrared Sci. Technol. 1995, 5, 36–48. [Google Scholar]
- Tiernan, H.; Byrne, B.; Kazarian, S.G. ATR-FTIR spectroscopy and spectroscopic imaging for the analysis of biopharmaceuticals. Spectrochim. Acta A 2020, 241. [Google Scholar] [CrossRef]
- Bunaciu, A.A.; Hoang, V.D.; Aboul-Enein, H.Y. Vibrational Micro-Spectroscopy of Human Tissues Analysis: Review. Crit. Rev. Anal. Chem. 2017, 47, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Oinas, J.; Rieppo, L.; Finnila, M.A.J.; Valkealahti, M.; Lehenkari, P.; Saarakkala, S. Imaging of Osteoarthritic Human Articular Cartilage using Fourier Transform Infrared Microspectroscopy Combined with Multivariate and Univariate Analysis. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Eysel, H.H.; Jackson, M.; Nikulin, A.; Somorjai, R.L.; Thomson, G.T.D.; Mantsch, H.H. A novel diagnostic test for arthritis: Multivariate analysis of infrared spectra of synovial fluid. Biospectroscopy 1997, 3, 161–167. [Google Scholar] [CrossRef]
- Hou, S.Y. Development of diagnostic models for canine osteoarthritis based on serum and joint fluid mid-infrared spectral data using five different discrimination and classification methods. J. Chemom. 2016, 30, 663–681. [Google Scholar] [CrossRef]
- Ren, P.L.; Niu, H.J.; Cen, H.P.; Jia, S.W.; Gong, H.; Fan, Y.B. Biochemical and Morphological Abnormalities of Subchondral Bone and Their Association with Cartilage Degeneration in Spontaneous Osteoarthritis. Calcif. Tissue Int. 2021. [Google Scholar] [CrossRef]
- Yin, J.H.; Xia, Y.; Xiao, Z.Y. Comparison of Macromolecular Component Distributions in Osteoarthritic and Healthy Cartilages by Fourier Transform Infrared Imaging. J. Innov. Opt. Health Sci. 2013, 6. [Google Scholar] [CrossRef]
- Rieppo, L.; Saarakkala, S.; Jurvelin, J.S.; Rieppo, J. Optimal variable selection for Fourier transform infrared spectroscopic analysis of articular cartilage composition. J. Biomed. Opt. 2014, 19. [Google Scholar] [CrossRef] [PubMed]
- Rieppo, L.; Saarakkala, S.; Narhi, T.; Helminen, H.J.; Jurvelin, J.S.; Rieppo, J. Application of second derivative spectroscopy for increasing molecular specificity of fourier transform infrared spectroscopic imaging of articular cartilage. Osteoarthr. Cartil. 2012, 20, 451–459. [Google Scholar] [CrossRef] [PubMed]
- David-Vaudey, E.; Burghardt, A.; Keshari, K.; Brouchet, A.; Ries, M.; Majumdar, S. Fourier Transform Infrared Imaging of focal lesions in human osteoarthritic cartilage. Eur. Cell Mater. 2005, 10, 51–60. [Google Scholar] [CrossRef]
- Mao, Z.H.; Zhang, X.X.; Wu, Y.C.; Yin, J.H.; Xia, Y. Fourier Transform Infrared Microscopic Imaging and Fisher Discriminant Analysis for Identification of Healthy and Degenerated Articular Cartilage. Chin. J. Anal. Chem. 2015, 43, 518–522. [Google Scholar] [CrossRef]
- Zhang, X.X.; Yin, J.H.; Mao, Z.H.; Xia, Y. Discrimination of healthy and osteoarthritic articular cartilages by Fourier transform infrared imaging and partial least squares-discriminant analysis. J. Biomed. Opt. 2015, 20. [Google Scholar] [CrossRef]
- Mao, Z.H.; Wu, Y.C.; Zhang, X.X.; Gao, H.; Yin, J.H. Comparative study on identification of healthy and osteoarthritic articular cartilages by fourier transform infrared imaging and chemometrics methods. J. Innov. Opt. Health Sci. 2017, 10. [Google Scholar] [CrossRef]
- Johansson, A.; Sundqvist, T.; Kuiper, J.H.; Oberg, P.A. A spectroscopic approach to imaging and quantification of cartilage lesions in human knee joints. Phys. Med. Biol. 2011, 56, 1865–1878. [Google Scholar] [CrossRef]
- Hanifi, A.; Bi, X.H.; Yang, X.; Kavukcuoglu, B.; Lin, P.C.; DiCarlo, E.; Spencer, R.G.; Bostrom, M.P.G.; Pleshko, N. Infrared Fiber Optic Probe Evaluation of Degenerative Cartilage Correlates to Histological Grading. Am. J. Sport Med. 2012, 40, 2853–2861. [Google Scholar] [CrossRef]
- West, P.A.; Torzilli, P.A.; Chen, C.; Lin, P.; Camacho, N.P. Fourier transform infrared imaging spectroscopy analysis of collagenase-induced cartilage degradation. J. Biomed. Opt. 2005, 10. [Google Scholar] [CrossRef]
- Yang, L.P.; Liu, J.L.; Song, Q.H.; Zhu, J.; Zhang, W.Q.; Kong, H.Y.; Zhao, T.J. FTIR Microspectroscopic Investigation of the Age-Related Changes of Subchondral Bone of the Knee in Guinea Pig. Spectrosc. Spect. Anal. 2013, 33, 2369–2373. [Google Scholar] [CrossRef]
- Zhao, Y.; Lu, Y.F.; Zhu, Y.K.; Wu, Y.C.; Zhai, M.Y.; Wang, X.; Yin, J.H. Submillimetric FTIR detection of articular cartilage by home-made ATR-MIR-Hollow optical fiber probe. Infrared Phys. Tech. 2019, 98, 236–239. [Google Scholar] [CrossRef]
- Akhmanov, S.A.; Koroteyev, N.I. Spectroscopy of Light-Scattering and Nonlinear Optics, Nonlinear Optical Methods in Active Spectroscopy of Raman and Rayleigh-Scattering. Sov. Phys. Uspekhi 1977, 123, 405–471. [Google Scholar] [CrossRef]
- Cialla-May, D.; Schmitt, M.; Popp, J. Theoretical principles of Raman spectroscopy. Phys. Sci. Rev. 2019, 4. [Google Scholar] [CrossRef]
- Brenan, C.J.; Hunter, I.W. Chemical imaging with a confocal scanning Fourier-transform-Raman microscope. Appl. Opt. 1994, 33, 7520–7528. [Google Scholar] [CrossRef]
- Lu, J.; Zhu, S.S.; Cui, X.Y.; Chen, S.; Yao, Y.D. Raman Spectroscopic Imaging Technology and Its Biomedical Applications. Chin. J. Lasers 2018, 45. [Google Scholar] [CrossRef]
- Chernenko, T.; Sawant, R.R.; Miljkovic, M.; Quintero, L.; Diem, M.; Torchilin, V. Raman microscopy for noninvasive imaging of pharmaceutical nanocarriers: Intracellular distribution of cationic liposomes of different composition. Mol. Pharm. 2012, 9, 930–936. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.L.; Heglund, D.L.; Ray, M.D.; Harder, D.; Dobert, R.; Leung, K.P.; Wu, M.T.; Sedlacek, A.J. Application of resonance Raman lidar for chemical species identification. Proc. SPIE Int. Soc. Opt. Eng. 1997, 3065, 279–285. [Google Scholar]
- Fikiet, M.A.; Khandasammy, S.R.; Mistek, E.; Ahmed, Y.; Halamkova, L.; Bueno, J.; Lednev, I.K. Surface enhanced Raman spectroscopy: A review of recent applications in forensic science. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2018, 197, 255–260. [Google Scholar] [CrossRef]
- Alvarez-Figueroa, M.J.; Narvaez-Araya, D.; Armijo-Escalona, N.; Carrasco-Flores, E.A.; Gonzalez-Aramundiz, J.V. Design of Chitosan Nanocapsules with Compritol 888 ATO (R) for Imiquimod Transdermal Administration. Evaluation of Their Skin Absorption by Raman Microscopy. Pharm. Res. Dordr. 2020, 37. [Google Scholar] [CrossRef]
- Dadou, S.M.; Tian, Y.W.; Li, S.; Jones, D.S.; Andrews, G.P. The optimization of process analytical technology for the inline quantification of multiple drugs in fixed dose combinations during continuous processing. Int. J. Pharm. 2021, 592. [Google Scholar] [CrossRef]
- Starciuc, T.; Guinet, Y.; Hedoux, A.; Shalaev, E. Water content thresholds in glycerol/water system: Low- and high-wavenumber Raman spectroscopy study. J. Mol. Liq. 2021, 321. [Google Scholar] [CrossRef]
- Dohrn, S.; Luebbert, C.; Lehmkemper, K.; Kyeremateng, S.O.; Degenhardt, M.; Sadowski, G. Solvent influence on the phase behavior and glass transition of Amorphous Solid Dispersions. Eur. J. Pharm. Biopharm. 2021, 158, 132–142. [Google Scholar] [CrossRef]
- Franzen, L.; Anderski, J.; Windbergs, M. Quantitative detection of caffeine in human skin by confocal Raman spectroscopy—A systematic in vitro validation study. Eur. J. Pharm. Biopharm. 2015, 95, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Stella, A.; Bonnier, F.; Tfayli, A.; Yvergnaux, F.; Byrne, H.J.; Chourpa, I.; Munnier, E.; Tauber, C. Raman mapping coupled to self-modelling MCR-ALS analysis to estimate active cosmetic ingredient penetration profile in skin. J. Biophotonics 2020, 13, e202000136. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, J.H.; Kang, Y.S.; Jang, K.T.; Im, J.; Seong, M.J. Evolution of amorphous carbon films into nano-crystalline graphite with increasing growth temperature in plasma-enhanced chemical vapor deposition. Curr. Appl Phys. 2021, 23, 52–56. [Google Scholar] [CrossRef]
- Lee, J.; Kim, H.S.; Osawa, E.; Hoang, G.C.; Lee, K.H. Predicting the Number of Graphene-Like Layers on Surface for Commercial Fumed Nanodiamonds with Raman Spectra and Model Calculations. J. Nanosci. Nanotechnol. 2021, 21, 1815–1819. [Google Scholar] [CrossRef]
- Gibbons, E.; Leveille, R.; Berlo, K. Data fusion of laser-induced breakdown and Raman spectroscopies: Enhancing clay mineral identification. Spectrochim. Acta B 2020, 170. [Google Scholar] [CrossRef]
- Yao, C.; Song, H.; Li, Q.; Li, N.; Zhang, G.Y. Micro Raman Spectral Characteristics and Implication of Pyrite in the Jiaojia Gold Deposit, Jiaodong Area, Shandong Province, China. Spectrosc. Spect. Anal. 2020, 40, 2479–2483. [Google Scholar] [CrossRef]
- Esmonde-White, K.A.; Mandair, G.S.; Esmonde-White, F.W.L.; Raaii, F.; Roessler, B.J.; Morris, M.D. Osteoarthritis Screening using Raman Spectroscopy of Dried Human Synovial Fluid Drops. Opt. Bone Biol. Diagn. 2009, 7166. [Google Scholar] [CrossRef]
- Matisioudis, N.; Rizos, E.; Tyrnenopoulou, P.; Papazoglou, L.; Diakakis, N.; Aggeli, A. Comparative Studies of Hyaluronic Acid Concentration in Normal and Osteoarthritic Equine Joints. Fluids 2019, 4, 193. [Google Scholar] [CrossRef]
- Sun, Z.P.; Wu, S.P.; Liang, C.D.; Zhao, C.X.; Sun, B.Y. The synovial fluid neuropeptide PACAP may act as a protective factor during disease progression of primary knee osteoarthritis and is increased following hyaluronic acid injection. Innate Immun. 2019, 25, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Timchenko, E.; Timchenko, P.; Volova, L.; Dolgushkin, D.; Markova, M.; Yagofarova, E. The synovial fluid analysis by using Raman Scattering spectroscopy in order to educe the synovial joint pathology. Int. Conf. Phys. 2018, 1038. [Google Scholar] [CrossRef]
- Chaudhari, A.; Dhonde, S.B. A Review on Speech Enhancement Techniques. In Proceedings of the 2015 International Conference on Pervasive Computing (ICPC), Pune, India, 8–10 January 2015. [Google Scholar]
- Mandair, G.S.; Dehring, K.A.; Roessler, B.J.; Morris, M.D. Detection of potential osteoarthritis biomarkers using surface-enhanced Raman spectroscopy in the near-infrared. Biomed. Vib. Spectrosc. Adv. Res. Ind. 2006, 6093. [Google Scholar] [CrossRef]
- Galli, M.M.; Protzman, N.M.; Bleazey, S.T.; Brigido, S.A. Role of Demineralized Allograft Subchondral Bone in the Treatment of Shoulder Lesions of the Talus: Clinical Results With Two-Year Follow-Up. J. Foot Ankle Surg. 2015, 54, 717–722. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Chu, L.Y.; Liu, X.Q.; He, Z.H.; Han, X.Q.; Yan, M.N.; Qu, X.H.; Li, X.F.; Yu, Z.F. Subchondral Trabecular Microstructure and Articular Cartilage Damage Variations Between Osteoarthritis and Osteoporotic Osteoarthritis: A Cross-sectional Cohort Study. Front. Med. 2021, 8. [Google Scholar] [CrossRef]
- Kerns, J.G.; Buckley, K.; Gikas, P.D.; Birch, H.L.; McCarthy, I.D.; Keen, R.; Parker, A.W.; Matousek, P.; Goodship, A.E. Raman spectroscopy reveals evidence for early bone changes in osteoarthritis. Int. J. Exp. Pathol. 2015, 96, A3. [Google Scholar]
- Dehring, K.A.; Roessle, B.J.; Morris, M.D. Correlating chemical changes in subchondral bone mineral due to aging or defective type II collagen by Raman spectroscopy. Adv. Biomed. Clin. Diagn. Syst. V 2007, 6430. [Google Scholar] [CrossRef]
- Dehring, K.A.; Crane, N.J.; Smukler, A.R.; McHugh, J.B.; Roessler, B.J.; Morris, M.D. Identifying chemical changes in subchondral bone taken from murine knee joints using Raman spectroscopy. Appl. Spectrosc. 2006, 60, 1134–1141. [Google Scholar] [CrossRef] [PubMed]
- Stack, J.; McCarthy, G.M. Cartilage calcification and osteoarthritis: A pathological association? Osteoarthr. Cartil. 2020, 28, 1301–1302. [Google Scholar] [CrossRef] [PubMed]
- Das Gupta, S.; Finnila, M.A.J.; Karhula, S.S.; Kauppinen, S.; Joukainen, A.; Kroger, H.; Korhonen, R.K.; Thambyah, A.; Rieppo, L.; Saarakkala, S. Raman microspectroscopic analysis of the tissue-specific composition of the human osteochondral junction in osteoarthritis: A pilot study. Acta Biomater. 2020, 106, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Esmonde-White, K.A.; Esmonde-White, F.W.; Morris, M.D.; Roessler, B.J. Fiber-optic Raman spectroscopy of joint tissues. Analyst 2011, 136, 1675–1685. [Google Scholar] [CrossRef]
- Mason, D.; Murugkar, S.; Speirs, A.D. Measurement of cartilage sub-component distributions through the surface by Raman spectroscopy-based multivariate analysis. J. Biophotonics 2021, 14. [Google Scholar] [CrossRef]
- Jensen, M.; Horgan, C.C.; Vercauteren, T.; Albro, M.B.; Bergholt, M.S. Multiplexed polarized hypodermic Raman needle probe for biostructural analysis of articular cartilage. Opt. Lett. 2020, 45, 2890–2893. [Google Scholar] [CrossRef]
- Lim, N.S.J.; Hamed, Z.; Yeow, C.H.; Chan, C.; Huang, Z.W. Early detection of biomolecular changes in disrupted porcine cartilage using polarized Raman spectroscopy. J. Biomed. Opt. 2011, 16. [Google Scholar] [CrossRef]
- Kumar, R.; Gronhaug, K.M.; Afseth, N.K.; Isaksen, V.; Davies, C.D.; Drogset, J.O.; Lilledahl, M.B. Optical investigation of osteoarthritic human cartilage (ICRS grade) by confocal Raman spectroscopy: A pilot study. Anal. Bioanal. Chem. 2015, 407, 8067–8077. [Google Scholar] [CrossRef]
- de Souza, R.A.; Xavier, M.; Mangueira, N.M.; Santos, A.P.; Pinheiro, A.L.B.; Villaverde, A.B.; Silveira, L. Raman spectroscopy detection of molecular changes associated with two experimental models of osteoarthritis in rats. Laser Med. Sci. 2014, 29, 797–804. [Google Scholar] [CrossRef]
- Kumar, R.; Singh, G.P.; Gronhaug, K.M.; Afseth, N.K.; Davies, C.D.; Drogset, J.O.; Lilledahl, M.B. Single Cell Confocal Raman Spectroscopy of Human Osteoarthritic Chondrocytes: A Preliminary Study. Int. J. Mol. Sci. 2015, 16, 9341–9353. [Google Scholar] [CrossRef] [PubMed]
- Oshima, Y.; Akehi, M.; Kiyomatsu, H.; Miura, H. Label-free characterization of degenerative changes in articular cartilage by Raman spectroscopy. Biomed. Imaging Sens. Conf. 2017, 10251. [Google Scholar] [CrossRef]
- Sirleto, L.; Ferrara, M.A. Fiber Amplifiers and Fiber Lasers Based on Stimulated Raman Scattering: A Review. Micromachines 2020, 11, 247. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.H.; Song, B.; Li, X.; Zou, Y.; Wang, Y.; Yu, Z.G.; Huang, M.Z. A smart optical fiber probe for Raman spectrometry and its application. J. Opt. 2017, 46, 62–67. [Google Scholar] [CrossRef]
- Xu, H.; Zhu, Y.K.; Lu, Y.F.; Yin, J.H. Development and Biomedical Application of Raman Probe. Laser Optoelectron. Prog. 2019, 56. [Google Scholar] [CrossRef]
- Bergholt, M.S.; Albro, M.B.; Stevens, M.M. Online quantitative monitoring of live cell engineered cartilage growth using diffuse fiber-optic Raman spectroscopy. Biomaterials 2017, 140, 128–137. [Google Scholar] [CrossRef] [PubMed]
- Oshima, Y.; Ishimaru, Y.; Kiyomatsu, H.; Hino, K.; Miura, H. Evaluation of degenerative changes in articular cartilage of osteoarthritis by Raman spectroscopy. Imaging Manip. Anal. Biomol. Cells Tissues Xvi 2018, 10497. [Google Scholar] [CrossRef]
- Pester, J.K.; Stumpfe, S.T.; Steinert, S.; Marintschev, I.; Plettenberg, H.K.; Aurich, M.; Hofmann, G.O. Histological, Biochemical and Spectroscopic Changes of Articular Cartilage in Osteoarthritis: Is There a Chance for Spectroscopic Evaluation? Z. Orthop. Unf. 2014, 152, 469–479. [Google Scholar] [CrossRef]
- Slooter, M.D.; Bierau, K.; Chan, A.B.; Lowik, C.W. Near infrared fluorescence imaging for early detection, monitoring and improved intervention of diseases involving the joint. Connect. Tissue Res. 2015, 56, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Paraskevaidi, M.; Hook, P.D.; Morais, C.L.M.; Anderson, J.R.; White, R.; Martin-Hirsch, P.L.; Peffers, M.J.; Martin, F.L. Attenuated total reflection Fourier-transform infrared (ATR-FTIR) spectroscopy to diagnose osteoarthritis in equine serum. Equine Vet. J. 2020, 52, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Bartick, E. Forensic analysis by Raman spectroscopy: An emerging technology. Meet. Int. Assoc. Forensic Sci. 2002, 45–50. [Google Scholar]
- Downes, A.; Elfick, A. Raman Spectroscopy and Related Techniques in Biomedicine. Sensors 2010, 10, 1871–1889. [Google Scholar] [CrossRef]

| Name | Main Inspection Site | Advantages | Disadvantages | Reference |
|---|---|---|---|---|
| MRI | Structure of cartilage | Observe structural features related to cartilage integrity, no radiation | Expensive, long scan time | [15,16,17,18] |
| OCT | Transverse view of articular cartilage | Good reproducibility and no radiation to living tissue | Depth limitations allow only surface cross-sectional analysis | [18,19,20,21] |
| Ultrasonic diagnosis | Periarticular soft tissue | Visible image information, low cost | Poor penetration of bone, inability to observe deeper structures, poor reproducibility | [22,23,24,25,26] |
| X-ray | Joint appearance | Low cost, high benefit, combined with arthroscopy to assist doctors in diagnosis | Unable to show early symptoms of OA | [27,28,29] |
| Vibrational Spectroscopy | Working Principle | Advantages | Disadvantages | Application in OA |
|---|---|---|---|---|
| NIR spectroscopy | Reflects the overtones and/or combination bands of stretching and bending vibrations of C–H, N–H, and O–H bonds ranging from 12,500 to 4000 cm−1 | Fast, accurate, nondestructive, labor-saving | Wide band, high spectral overlap, difficult to distinguish characteristic peak | Has a high penetration depth but can only provide a full spectral signal of the cartilage |
| IR spectroscopy | Studies the structural changes in the range (4000–400 cm−1) caused by the transition of vibrational and rotational energy levels of molecules | Fast, accurate, and nondestructive; reflects information of most organic matter | Low sensitivity, complex band, sample limitations | Detects a variety of components in the cartilage and can achieve high-speed imaging |
| Raman spectroscopy | Reflects the vibrational information between molecules based on the principle of Raman scattering | Weak water signal, fast, simple, reflects biological signal | Raman scattering area can be affected by the optical system, fluorescence interference | Reflects physiological changes of OA at tissue, cell, and molecular levels |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, C.; Zhao, B.; Li, Y.; Zang, H.; Li, L. Vibrational Spectroscopy in Assessment of Early Osteoarthritis—A Narrative Review. Int. J. Mol. Sci. 2021, 22, 5235. https://doi.org/10.3390/ijms22105235
Yu C, Zhao B, Li Y, Zang H, Li L. Vibrational Spectroscopy in Assessment of Early Osteoarthritis—A Narrative Review. International Journal of Molecular Sciences. 2021; 22(10):5235. https://doi.org/10.3390/ijms22105235
Chicago/Turabian StyleYu, Chen, Bing Zhao, Yan Li, Hengchang Zang, and Lian Li. 2021. "Vibrational Spectroscopy in Assessment of Early Osteoarthritis—A Narrative Review" International Journal of Molecular Sciences 22, no. 10: 5235. https://doi.org/10.3390/ijms22105235
APA StyleYu, C., Zhao, B., Li, Y., Zang, H., & Li, L. (2021). Vibrational Spectroscopy in Assessment of Early Osteoarthritis—A Narrative Review. International Journal of Molecular Sciences, 22(10), 5235. https://doi.org/10.3390/ijms22105235







