Bone Disease in Nephropathic Cystinosis: Beyond Renal Osteodystrophy
Abstract
1. Introduction
1.1. The Emerging Concept of Cystinosis Metabolic Bone Disease
1.2. Bone Impairment in Patients Nephropathic Cystinosis: Clinical Facts
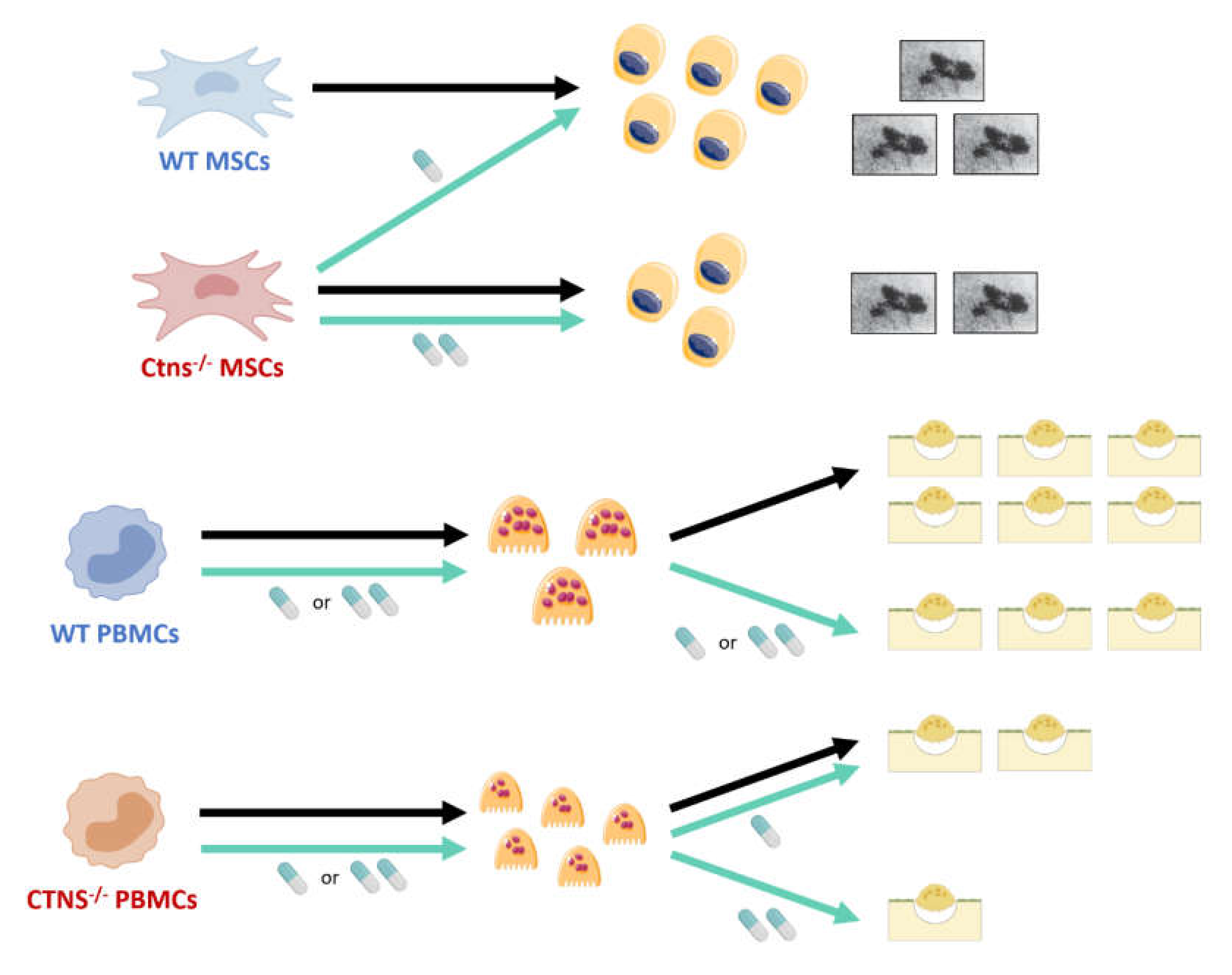
1.3. Bone Disease and Nephropathic Cystinosis: A Functional Impairment of Both Osteoblasts and Osteoclasts
1.4. Future Research Axes: Toward Damaged Mitochondrial Function in Osteoclasts Derived From Patients with Nephropathic Cystinosis?
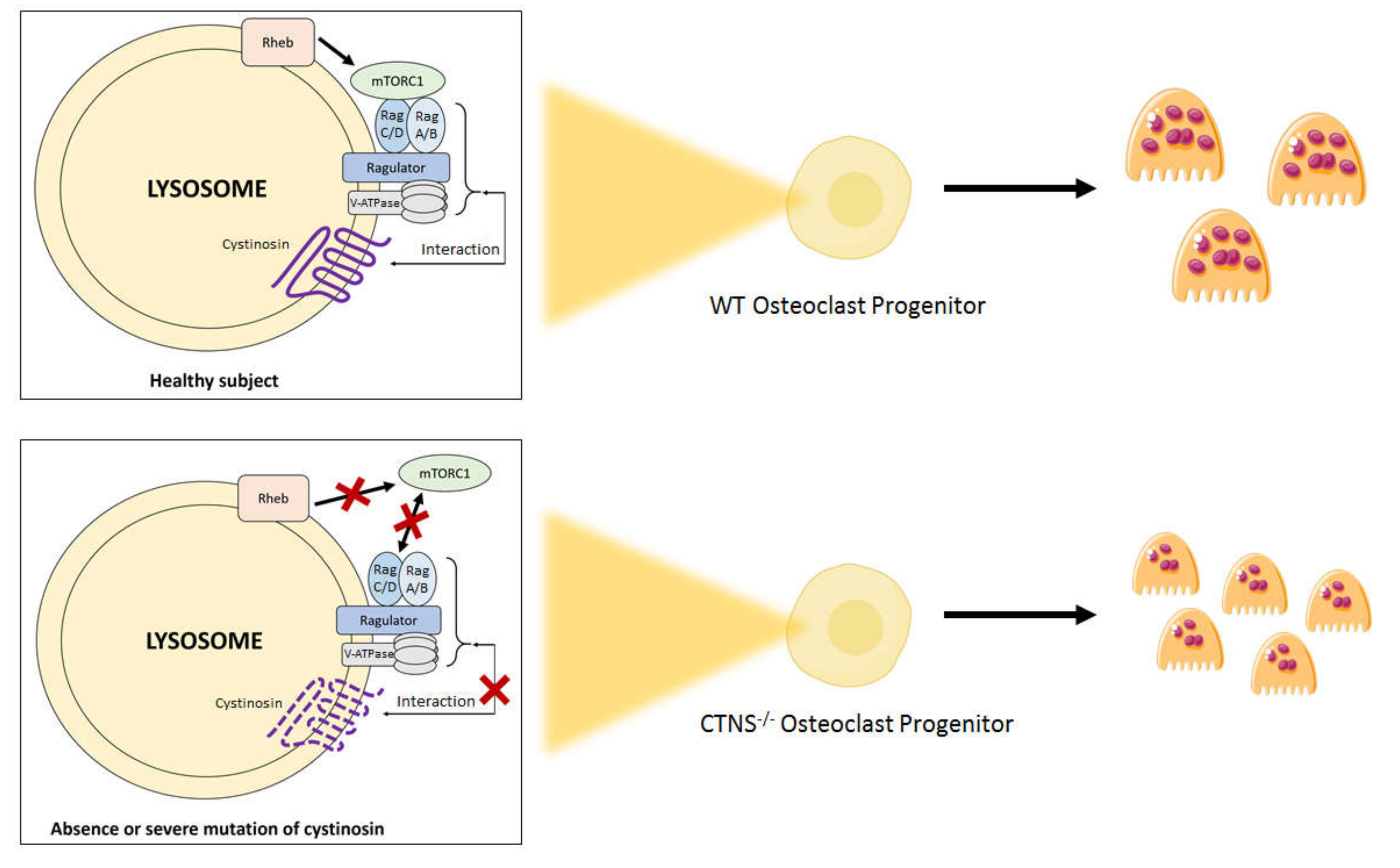
1.5. Future Research Axes: Toward an Impaired mTor Signaling in Osteoclasts Derived from Patients with Nephropathic Cystinosis?
2. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CKD | chronic kidney disease |
| CKD-MBD | chronic kidney disease mineral and bone disorders |
| GH | growth hormone |
| IGF1 | insulin-like growth factor 1 |
| ROD | renal osteodystrophy |
| PTH | parathyroid hormone |
| CTNS | lysosomal cystine transporter |
| CMBD | cystinosis metabolic bone disease |
| ALP | alkaline phosphatase |
| 25-D | 25 OH-vitamin D |
| FGF23 | fibroblast growth factor 23 |
| DXA | dual X-ray absorptiometry |
| HR-pQCT | high resolution peripheral quantitative computed tomography |
| BMD | bone mineral density |
| TV | trabecular volume |
| MSC | mesenchymal stem cell |
| BrdU | bromodeoxyuridine |
| LDH | lactate dehydrogenase |
| RUNX2 | runt-related transcription factor 2 |
| COL1A2 | collagen type I alpha 2 chain |
| M-CSF | macrophage colony-stimulating factor |
| RANKL | receptor activator of nuclear factor kappa-B ligand |
| TRAP | tartrate-resistant acid phosphatase |
| PBMC | peripheral blood mononuclear cell |
| TCIRG1 | T-cell immune regulator 1 |
| ClCN7 | chloride voltage-gated channel 7 |
| OSTM1 | osteopetrosis associated transmembrane protein 1 |
| SLC29A3 | solute carrier family 29, member 3 |
| MMP9 | matrix metallopeptidase 9 |
| mTORC1 | mammalian target of rapamycin complex 1 |
| TSC1 | tuberous sclerosis complex 1 |
References
- Bacchetta, J.; Harambat, J.; Cochat, P.; Salusky, I.B.; Wesseling-Perry, K. The consequences of chronic kidney disease on bone metabolism and growth in children. Nephrol. Dial. Transplant. 2012, 27, 3063–3071. [Google Scholar] [CrossRef]
- Moe, S.; Drüeke, T.; Cunningham, J.; Goodman, W.; Martin, K.; Olgaard, K.; Eknoyan, G. Definition, evaluation, and classification of renal osteodystrophy: A position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int. 2006, 69, 1945–1953. [Google Scholar] [CrossRef]
- Ketteler, M.; Block, G.A.; Evenepoel, P.; Fukagawa, M.; Herzog, C.A.; McCann, L.; Vervloet, M.G. Executive summary of the 2017 KDIGO Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD) Guideline Update: What’s changed and why it matters. Kidney Int. 2017, 92, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Groothoff, J.W.; Offringa, M.; Van Eck-Smit, B.L.F.; Gruppen, M.P.; Van De Kar, N.J.; Wolff, E.D.; Dekker, F.W. Severe bone disease and low bone mineral density after juvenile renal failure. Kidney Int. 2003, 63, 266–275. [Google Scholar] [CrossRef] [PubMed]
- Denburg, M.R.; Kumar, J.; Jemielita, T.; Brooks, E.R.; Skversky, A.; Portale, A.A.; Leonard, M.B. Fracture Burden and Risk Factors in Childhood CKD: Results from the CKiD Cohort Study. J. Am. Soc. Nephrol. 2016, 27, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Bacchetta, J.; Ranchin, B.; Demède, D.; Allard, L. The consequences of pediatric renal transplantation on bone metabolism and growth. Curr. Opin. Organ Transplant. 2013, 18, 555–562. [Google Scholar] [CrossRef]
- Cochat, P.; Rumsby, G. Primary hyperoxaluria. N. Engl. J. Med. 2013, 369, 649–658. [Google Scholar] [CrossRef]
- Nesterova, G.; Gahl, W.A. Cystinosis: The evolution of a treatable disease. Pediatr. Nephrol. 2013, 28, 51–59. [Google Scholar] [CrossRef]
- Beaufils, C.; Farlay, D.; Machuca-Gayet, I.; Fassier, A.; Zenker, M.; Freychet, C.; Bacchetta, J. Skeletal impairment in Pierson syndrome: Is there a role for lamininβ2 in bone physiology? Bone 2018, 106, 187–193. [Google Scholar] [CrossRef]
- Evenepoel, P.; Claes, K.; Cavalier, E.; Meijers, B.; Stenvinkel, P.; Behets, G.; Bammens, B. A distinct bone phenotype in ADPKD patients with end-stage renal disease. Kidney Int. 2019, 95, 412–419. [Google Scholar] [CrossRef]
- David, D.; Princiero Berlingerio, S.; Elmonem, M.A.; Oliveira Arcolino, F.; Soliman, N.; van den Heuvel, B.; Levtchenko, E. Molecular Basis of Cystinosis: Geographic Distribution, Functional Consequences of Mutations in the CTNS Gene, and Potential for Repair. Nephron 2019, 141, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Cherqui, S.; Courtoy, P.J. The renal Fanconi syndrome in cystinosis: Pathogenic insights and therapeutic perspectives. Nat. Rev. Nephrol. 2017, 13, 115–131. [Google Scholar] [CrossRef] [PubMed]
- Bertholet-Thomas, A.; Bacchetta, J.; Tasic, V.; Cochat, P. Nephropathic cystinosis--a gap between developing and developed nations. N. Engl. J. Med. 2014, 370, 1366–1367. [Google Scholar] [CrossRef] [PubMed]
- Gahl, W.A.; Thoene, J.G.; Schneider, J.A. Cystinosis. N. Engl. J. Med. 2002, 347, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Pisoni, R.L.; Thoene, J.G.; Christensen, H.N. Detection and characterization of carrier-mediated cationic amino acid transport in lysosomes of normal and cystinotic human fibroblasts. Role in therapeutic cystine removal? J. Biol. Chem. 1985, 260, 4791–4798. [Google Scholar] [PubMed]
- Langman, C.B. Bone Complications of Cystinosis. J. Pediatr. 2017, 183, S2–S4. [Google Scholar] [CrossRef] [PubMed]
- Besouw, M.T.P.; Bowker, R.; Dutertre, J.-P.; Emma, F.; Gahl, W.A.; Greco, M.; Skovby, F. Cysteamine toxicity in patients with cystinosis. J. Pediatr. 2011, 159, 1004–1011. [Google Scholar] [CrossRef]
- Bertholet-Thomas, A.; Claramunt-Taberner, D.; Gaillard, S.; Deschênes, G.; Sornay-Rendu, E.; Szulc, P.; Bacchetta, J. Teenagers and young adults with nephropathic cystinosis display significant bone disease and cortical impairment. Pediatr. Nephrol. 2018, 33, 1165–1172. [Google Scholar] [CrossRef]
- Florenzano, P.; Ferreira, C.; Nesterova, G.; Roberts, M.S.; Tella, S.H.; De Castro, L.F.; Gafni, R.I. Skeletal Consequences of Nephropathic Cystinosis. J. Bone Miner Res. 2018, 33, 1870–1880. [Google Scholar] [CrossRef]
- Bacchetta, J.; Greco, M.; Bertholet-Thomas, A.; Nobili, F.; Zustin, J.; Cochat, P.; Boivin, G. Skeletal implications and management of cystinosis: Three case reports and literature review. BoneKEy Rep. 2016, 5, 828. [Google Scholar] [CrossRef]
- Hohenfellner, K.; Rauch, F.; Ariceta, G.; Awan, A.; Bacchetta, J.; Bergmann, C.; Gahl, W.A. Management of bone disease in cystinosis: Statement from an international conference. J. Inherit Metab. Dis. 2019, 42, 1019–1029. [Google Scholar] [CrossRef] [PubMed]
- Conforti, A.; Taranta, A.; Biagini, S.; Starc, N.; Pitisci, A.; Bellomo, F.; Emma, F. Cysteamine treatment restores the in vitro ability to differentiate along the osteoblastic lineage of mesenchymal stromal cells isolated from bone marrow of a cystinotic patient. J. Transl. Med. 2015, 13, 143. [Google Scholar] [CrossRef] [PubMed]
- Claramunt-Taberner, D.; Flammier, S.; Gaillard, S.; Cochat, P.; Peyruchaud, O.; Machuca-Gayet, I.; Bacchetta, J. Bone disease in nephropathic cystinosis is related to cystinosin-induced osteoclastic dysfunction. Nephrol. Dial. Transplant. 2017, 33, 1525–1532. [Google Scholar] [CrossRef] [PubMed]
- Besouw, M.T.P.; Schneider, J.; Janssen, M.C.; Greco, M.; Emma, F.; Cornelissen, E.A.; De Paepe, A. Copper deficiency in patients with cystinosis with cysteamine toxicity. J. Pediatr. 2013, 163, 754–760. [Google Scholar] [CrossRef]
- Cherqui, S.; Sevin, C.; Hamard, G.; Kalatzis, V.; Sich, M.; Pequignot, M.O.; Antignac, C. Intralysosomal cystine accumulation in mice lacking cystinosin, the protein defective in cystinosis. Mol. Cell. Biol. 2002, 22, 7622–7632. [Google Scholar] [CrossRef]
- Klusmann, M.; Van’t Hoff, W.; Monsell, F.; Offiah, A.C. Progressive destructive bone changes in patients with cystinosis. Skeletal Radiol. 2013, 43, 387–391. [Google Scholar] [CrossRef]
- Sirrs, S.; Munk, P.; Mallinson, P.I.; Ouellette, H.; Horvath, G.; Cooper, S.; Hoang, L.N. Cystinosis with sclerotic bone lesions. JIMD Rep. 2014, 13, 27–31. [Google Scholar]
- Tucker, T.; Nelson, T.; Sirrs, S.; Roughley, P.; Glorieux, F.H.; Moffatt, P.; Rauch, F. A co-occurrence of osteogenesis imperfecta type VI and cystinosis. Am. J. Med. Genet. A 2012, 158, 1422–1426. [Google Scholar] [CrossRef]
- Drube, J.; Wan, M.; Bonthuis, M.; Wühl, E.; Bacchetta, J.; Santos, F.; Tönshoff, B. Clinical practice recommendations for growth hormone treatment in children with chronic kidney disease. Nat. Rev. Nephrol. 2019, 15, 577–589. [Google Scholar] [CrossRef]
- Battafarano, G.; Rossi, M.; Rega, L.R.; Di Giovamberardino, G.; Pastore, A.; D’Agostini, M.; Del Fattore, A. Intrinsic Bone Defects in Cystinotic Mice. Am. J. Pathol. 2019, 189, 1053–1064. [Google Scholar] [CrossRef]
- Baron, R.; Kneissel, M. WNT signaling in bone homeostasis and disease: From human mutations to treatments. Nat. Med. 2013, 19, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Georgess, D.; Mazzorana, M.; Terrado, J.; Delprat, C.; Chamot, C.; Guasch, R.M.; Machuca-Gayet, I. Comparative transcriptomics reveals RhoE as a novel regulator of actin dynamics in bone-resorbing osteoclasts. Mol. Biol. Cell. 2014, 25, 380–396. [Google Scholar] [CrossRef]
- Lacombe, J.; Karsenty, G.; Ferron, M. Regulation of lysosome biogenesis and functions in osteoclasts. Cell Cycle 2013, 12, 2744–2752. [Google Scholar] [CrossRef] [PubMed]
- Festa, B.P.; Chen, Z.; Berquez, M.; Debaix, H.; Tokonami, N.; Prange, J.A.; Giles, R.H. Impaired autophagy bridges lysosomal storage disease and epithelial dysfunction in the kidney. Nat. Commun. 2018, 9, 161. [Google Scholar] [CrossRef] [PubMed]
- Bellomo, F.; Signorile, A.; Tamma, G.; Ranieri, M.; Emma, F.; De Rasmo, D. Impact of atypical mitochondrial cyclic-AMP level in nephropathic cystinosis. Cell Mol. Life Sci. 2018, 75, 3411–3422. [Google Scholar] [CrossRef] [PubMed]
- De Rasmo, D.; Signorile, A.; De Leo, E.; Polishchuk, E.V.; Ferretta, A.; Raso, R.; Bellomo, F. Mitochondrial Dynamics of Proximal Tubular Epithelial Cells in Nephropathic Cystinosis. Int. J. Mol. Sci. 2019, 21, 192. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.; Kwon, J.-O.; Kim, M.K.; Song, M.-K.; Kim, B.; Lee, Z.H.; Kim, H.H. Mitofusin 2, a mitochondria-ER tethering protein, facilitates osteoclastogenesis by regulating the calcium-calcineurin-NFATc1 axis. Biochem. Biophys. Res. Commun. 2019, 516, 202–208. [Google Scholar] [CrossRef]
- Ballard, A.; Zeng, R.; Zarei, A.; Shao, C.; Cox, L.; Yan, H.; Veis, D.J. The tethering function of mitofusin2 controls osteoclast differentiation by modulating the Ca2+-NFATC1 axis. J. Biol. Chem. 2020. [Google Scholar] [CrossRef]
- Ivanova, E.A.; van den Heuvel, L.P.; Elmonem, M.A.; De Smedt, H.; Missiaen, L.; Pastore, A.; Levtchenko, E.N. Altered mTOR signalling in nephropathic cystinosis. J. Inherit. Metab. Dis. 2016, 39, 457–464. [Google Scholar] [CrossRef]
- Huynh, H.; Wan, Y. mTORC1 impedes osteoclast differentiation via calcineurin and NFATc1. Commun. Biol. 2018, 1, 29. [Google Scholar] [CrossRef]
- Langman, C.B. Oh cystinosin: Let me count the ways! Kidney Int. 2019, 96, 275–277. [Google Scholar] [CrossRef] [PubMed]
- Andrzejewska, Z.; Nevo, N.; Thomas, L.; Chhuon, C.; Bailleux, A.; Chauvet, V.; Antignac, C. Cystinosin is a Component of the Vacuolar H+-ATPase-Ragulator-Rag Complex Controlling Mammalian Target of Rapamycin Complex 1 Signaling. J. Am. Soc. Nephrol. 2016, 27, 1678–1688. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Machuca-Gayet, I.; Quinaux, T.; Bertholet-Thomas, A.; Gaillard, S.; Claramunt-Taberner, D.; Acquaviva-Bourdain, C.; Bacchetta, J. Bone Disease in Nephropathic Cystinosis: Beyond Renal Osteodystrophy. Int. J. Mol. Sci. 2020, 21, 3109. https://doi.org/10.3390/ijms21093109
Machuca-Gayet I, Quinaux T, Bertholet-Thomas A, Gaillard S, Claramunt-Taberner D, Acquaviva-Bourdain C, Bacchetta J. Bone Disease in Nephropathic Cystinosis: Beyond Renal Osteodystrophy. International Journal of Molecular Sciences. 2020; 21(9):3109. https://doi.org/10.3390/ijms21093109
Chicago/Turabian StyleMachuca-Gayet, Irma, Thomas Quinaux, Aurélia Bertholet-Thomas, Ségolène Gaillard, Débora Claramunt-Taberner, Cécile Acquaviva-Bourdain, and Justine Bacchetta. 2020. "Bone Disease in Nephropathic Cystinosis: Beyond Renal Osteodystrophy" International Journal of Molecular Sciences 21, no. 9: 3109. https://doi.org/10.3390/ijms21093109
APA StyleMachuca-Gayet, I., Quinaux, T., Bertholet-Thomas, A., Gaillard, S., Claramunt-Taberner, D., Acquaviva-Bourdain, C., & Bacchetta, J. (2020). Bone Disease in Nephropathic Cystinosis: Beyond Renal Osteodystrophy. International Journal of Molecular Sciences, 21(9), 3109. https://doi.org/10.3390/ijms21093109




