Cannabidiol and Other Non-Psychoactive Cannabinoids for Prevention and Treatment of Gastrointestinal Disorders: Useful Nutraceuticals?
Abstract
1. Introduction
2. Use of Hemp and Non-Psychoactive Phytocannabinoids as Nutraceuticals and Food Ingredients
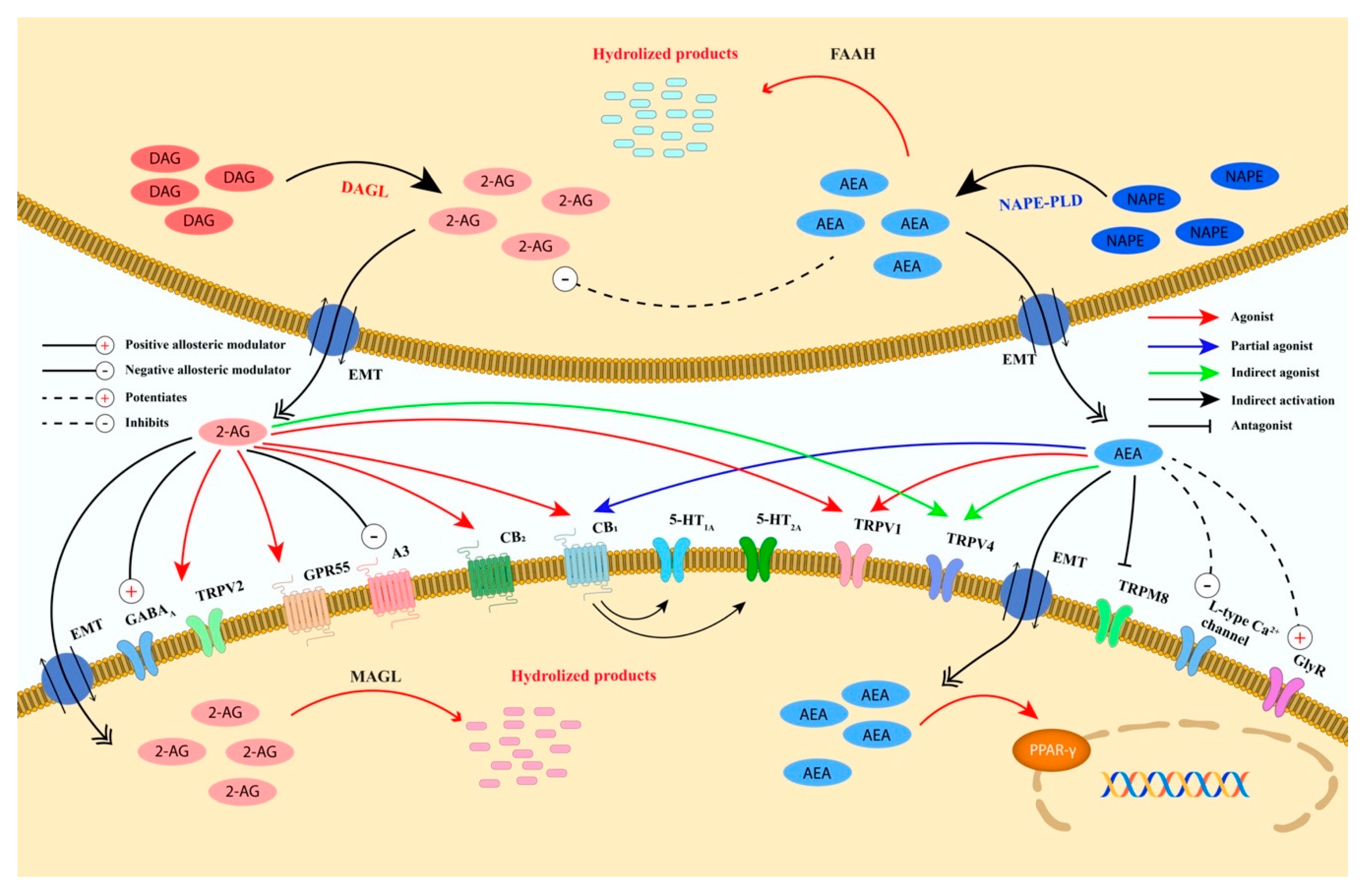
3. The Endogenous Cannabinoid System
4. Cannabis sativa and its Phytocannabinoids
Psychoactive and Non-Psychoactive Phytocannabinoids Molecular Targets
5. Cannabidiol and Other Non-Psychoactive Phytocannabinoids for Gastrointestinal Health
5.1. Role in Irritable Bowel Syndrome
5.2. Role in Inflammatory Bowel Disease
5.3. Role in Gastrointestinal Cancer
5.4. Other Disorders and Diseases of the Gastrointestinal System
5.4.1. Nausea and Emesis
5.4.2. Gastric Secretion and Gastroprotection
5.4.3. Gastroesophageal Reflux Disease
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 2-AG | 2-arachidonoylglycerol |
| 2-AGE | Noladin ether, 2-arachidonoylglycerylether |
| 5-HT1A | 5-hydroxytryptamine 1A receptor |
| 5-HT2A | 5-hydroxytryptamine 2A receptor |
| 5HT3A | 5-hydroxytryptamine 3A receptor |
| A | Adenosine receptor |
| ABHD6/12 | α/β -hydrolase domain 6/12 |
| AEA | Anandamide, arachidonylethanolamide |
| AIDS | Acquired immunodeficiency syndrome |
| CB1 | Cannabinoid receptor 1 |
| CB2 | Cannabinoid receptor 2 |
| CBC | Cannabichromene |
| CBCA | Cannabichromenic acid |
| CBD | Cannabidiol |
| CBD-BDS | Cannabidiol-botanical drug substance |
| CBDA | Cannabidiolic acid |
| CBDV | Cannabidivarin |
| CBG | Cannabigerol |
| CBGA | Cannabigerolic acid |
| CBN | Cannabinol |
| CBNA | Cannabinolic acid |
| CBNDA | Cannabinodiolic acid |
| CD | Crohn’s disease |
| CDAI | Crohn’s disease activity index |
| CHS | Cannabinoid hyperemesis syndrome |
| CNS | Central nervous system |
| COX | Cyclooxygenase |
| CRC | Colorectal cancer |
| DAG | Diacylglycerol |
| DAGL | Diacylglycerol lipase |
| DEA | Drug Enforcement Administration |
| DNBS | Dinitrobenzene sulphonic acid |
| DSS | Dextrane sulfate sodium |
| ECS | Endocannabinoid system, endogenous cannabinoid system |
| EFSA | European Food Safety Authority’s |
| EMT | Endocannabinoid membrane transporter |
| ENS | Enteric nervous system |
| EU | European Union |
| FAAH | Fatty acid amide hydrolase |
| FABP | Fatty acid binding protein |
| FDA | Food and Drug Administration |
| FUFOSE | Functional Food Science in Europe |
| GABA | γ -aminobutyric acid |
| GABAA | γ-aminobutyric acid type A receptor |
| GI | Gastrointestinal |
| GLP-1 | Glucagon like peptide 1 |
| GlyR | Glycine receptor |
| GPR | G protein-coupled receptor |
| GPX | Glutathione peroxidase |
| IBD | Inflammatory bowel disease |
| IBS | Irritable bowel syndrome |
| IBS-C | Irritable bowel syndrome with constipation |
| IBS-D | Irritable bowel syndrome with diarrhea |
| IBS-M | Irritable bowel syndrome with mixed bowel habits |
| IFN | Interferon |
| IL | Interleukine |
| i.p. | Intraperitoneal |
| iNOS | Inducible nitric oxide synthase |
| LES | Lower esophageal sphincter |
| LOX | Lipooxygenase |
| LPI | Lysophosphatidylinositol |
| LPS | Lipopolysaccharide |
| MAGL | Monoacylglycerol lipase |
| MAPK | Mitogen-activated protein kinase |
| MCP-1α | Monocyte chemoattractant protein-1α |
| MPO | Myeloperoxidase |
| NAAA | N-acylethanolamine acid amide hydrolase, N-acylethanolamine hydrolyzing acid amidase |
| NADA | N-arachidonoyldopamine |
| NAPE | N-acyl-phosphatidylethanolamine |
| NAPE-PLD | N-acyl-phosphatidylethanolamine-specific phospholipase D |
| NO | Nitric oxide |
| O-AEA | Virodhamine, O-arachidonoylethanolamine |
| OEA | Oleylethanolamide |
| PEA | Palmithoylethanolamide |
| PNS | Peripheral nervous system |
| PPARα | Peroxisome proliferator-activated receptor type-α |
| PPARγ | Peroxisome proliferator-activated receptor type-γ |
| ROS | Reactive oxygen species |
| SOD | Superoxide dismutase |
| THC | Δ9-tetrahydrocannabinol |
| THCA | Tetrahydrocannabinolic acid |
| TNBS | Trinitrobenzene sulfonic acid |
| TNF | Tumor necrosis factor |
| TRP | Transient receptor potential |
| TRPA1 | Transient receptor potential cation channel subfamily ankyrin 1-type |
| TRPM8 | Transient receptor potential cation channel subfamily M member 8 |
| TRPV1 | Transient receptor potential vanilloid type-1 channel |
| TRPV2 | Transient receptor potential cation channel subfamily V member 2 |
| TRPV3 | Transient receptor potential cation channel subfamily V member 3 |
| TRPV4 | Transient receptor potential cation channel subfamily V member 4 |
| UC | Ulcerative colitis |
| UGT | Upper gastrointestinal transit |
| USDA | United States Department of Agriculture |
| XIAP | X-linked inhibitor apoptosis |
| Δ9-THC-BDS | Δ9-tetrahydrocannabinol-botanical drug substance |
References
- Gao, X.; Liu, J.; Li, L.; Liu, W.; Sun, M. A brief review of nutraceutical ingredients in gastrointestinal disorders: Evidence and suggestions. Int. J. Mol. Sci. 2020, 21, 1822. [Google Scholar] [CrossRef] [PubMed]
- Larussa, T.; Imeneo, M.; Luzza, F. Potential role of nutraceutical compounds in inflammatory bowel disease. World J. Gastroenterol. 2017, 23, 2483–2492. [Google Scholar] [CrossRef] [PubMed]
- Larussa, T.; Rossi, M.; Suraci, E.; Marasco, R.; Imeneo, M.; Abenavoli, L.; Luzza, F. Use of complementary and alternative medicine by patients with irritable bowel syndrome according to the roma IV criteria: A single-center italian survey. Medicina 2019, 55, 46. [Google Scholar] [CrossRef] [PubMed]
- McPartland, J.M. Cannabis Systematics at the Levels of Family, Genus, and Species. Cannabis Cannabinoid Res. 2018, 3, 203–212. [Google Scholar] [CrossRef]
- Morales, P.; Hurst, D.P.; Reggio, P.H. Molecular Targets of the Phytocannabinoids: A Complex Picture. In Phytocannabinoids. Unraveling the Complex Chemistry and Pharmacology of Cannabis sativa; Kinghorn, A.D., Falk, H., Gibbons, S., Kobayashi, J., Eds.; Springer International Publishing: Cham, Switzerland, 2017; Volume 103, pp. 103–131. ISBN 9783319455396. [Google Scholar]
- Gertsch, J. Cannabimimetic phytochemicals in the diet—An evolutionary link to food selection and metabolic stress adaptation? Br. J. Pharmacol. 2017, 174, 1464–1483. [Google Scholar] [CrossRef]
- Johnson, R. Defining Hemp: A Fact Sheet; Congressional Research Service: Washington, DC, USA, 2019. [Google Scholar]
- Leonard, W.; Zhang, P.; Ying, D.; Fang, Z. Hempseed in food industry: Nutritional value, health benefits, and industrial applications. Compr. Rev. Food Sci. Food Saf. 2019, 1–27. [Google Scholar] [CrossRef]
- Salentijn, E.M.J.; Zhang, Q.; Amaducci, S.; Yang, M.; Trindade, L.M. New developments in fiber hemp (Cannabis sativa L.) breeding. Ind. Crops Prod. 2015, 68, 32–41. [Google Scholar] [CrossRef]
- Garcia, A.G.M. Hemp: A Composition Review Plus; Food Science and Nutrition Department, California Polytechnic State University: San Luis Obispo, CA, USA, 2017; Volume 102. [Google Scholar]
- Audu, B.S.; Ofojekwu, P.C.; Ujah, A.; Ajima, M.N.O. Phytochemical, proximate composition, amino acid profile and characterisation of Marijuana (Cannabis sativa L.). Phytopharmacology 2014, 3, 35–43. [Google Scholar]
- Andre, C.M.; Hausman, J.F.; Guerriero, G. Cannabis sativa: The plant of the thousand and one molecules. Front. Plant Sci. 2016, 7, 1–17. [Google Scholar] [CrossRef]
- Pacifico, D.; Miselli, F.; Carboni, A.; Moschella, A.; Mandolino, G. Time course of cannabinoid accumulation and chemotype development during the growth of Cannabis sativa L. Euphytica 2008, 160, 231–240. [Google Scholar] [CrossRef]
- Callaway, J.C. Hempseed as a nutritional resource: An overview. Euphytica 2004, 140, 65–72. [Google Scholar] [CrossRef]
- Vonapartis, E.; Aubin, M.P.; Seguin, P.; Mustafa, A.F.; Charron, J.B. Seed composition of ten industrial hemp cultivars approved for production in Canada. J. Food Compos. Anal. 2015, 39, 8–12. [Google Scholar] [CrossRef]
- Wang, M.; Wang, Y.H.; Avula, B.; Radwan, M.M.; Wanas, A.S.; Van Antwerp, J.; Parcher, J.F.; Elsohly, M.A.; Khan, I.A. Decarboxylation Study of Acidic Cannabinoids: A Novel Approach Using Ultra-High-Performance Supercritical Fluid Chromatography/Photodiode Array-Mass Spectrometry. Cannabis Cannabinoid Res. 2016, 1, 262–271. [Google Scholar] [CrossRef] [PubMed]
- Cardenia, V.; Gallina Toschi, T.; Scappini, S.; Rubino, R.C.; Rodriguez-Estrada, M.T. Development and validation of a Fast gas chromatography/mass spectrometry method for the determination of cannabinoids in Cannabis sativa L. J. Food Drug Anal. 2018, 26, 1283–1292. [Google Scholar] [CrossRef] [PubMed]
- Kalra, E.K. Nutraceutical-Definition and introduction. AAPS J. 2003, 5, 1–2. [Google Scholar] [CrossRef]
- Martirosyan, D.M.; Singh, J. A new definition of functional food by FFC: What makes a new definition unique? Funct. Foods Heal. Dis. 2015, 5, 209–223. [Google Scholar] [CrossRef]
- The European Commission. Functional Foods; Publications Office of the European Union: Luxembourg, 2010. [Google Scholar]
- King, J.W. The relationship between cannabis/hemp use in foods and processing methodology. Curr. Opin. Food Sci. 2019, 28, 32–40. [Google Scholar] [CrossRef]
- USDA (United States Department of Agriculture USA) FoodData Central. Available online: https://fdc.nal.usda.gov/fdc-app.html#/?query=hemp (accessed on 25 March 2020).
- European Comission Novel Food Catalogue. Available online: https://ec.europa.eu/food/safety/novel_food/catalogue_en (accessed on 27 January 2020).
- EFSA Panel on Dietetic Products; Nutrition and Allergies (NDA); Turck, D.; Bresson, J.L.; Burlingame, B.; Dean, T.; Fairweather-Tait, S.; Heinonen, M.; Hirsch-Ernst, K.I.; Mangelsdorf, I.; et al. Guidance on the preparation and presentation of an application for authorisation of a novel food in the context of Regulation (EU) 2015/2283. EFSA J. 2016, 14, e04594. [Google Scholar]
- Másílko, M.; Sadílek, T.; Martin, T.; Jurado, E. Cannabidiol (CBD) and Hemp: Novel Food Status in the EU; Int. Cannabis Cannabinoids Inst: Prague, Czech Republic, 2019. [Google Scholar]
- Di Marzo, V. New approaches and challenges to targeting the endocannabinoid system. Nat. Rev. Drug Discov. 2018, 17, 623–639. [Google Scholar] [CrossRef]
- Chye, Y.; Christensen, E.; Solowij, N.; Yücel, M. The endocannabinoid system and cannabidiol’s promise for the treatment of substance use disorder. Front. Psychiatry 2019, 10, 1–12. [Google Scholar] [CrossRef]
- Devane, W.A.; Dysarz, F.A.; Johnson, M.R.; Melvin, L.S.; Howlett, A.C. Determination and characterization of a cannabinoid receptor in rat brain. Mol. Pharmacol. 1988, 34, 605–613. [Google Scholar] [PubMed]
- Salio, C.; Doly, S.; Fischer, J.; Franzoni, M.F.; Conrath, M. Neuronal and astrocytic localization of the cannabinoid receptor-1 in the dorsal horn of the rat spinal cord. Neurosci. Lett. 2002, 329, 13–16. [Google Scholar] [CrossRef]
- Stella, N. Cannabinoid and cannabinoid-like receptors in microglia, astrocytes, and astrocytomas. Glia 2010, 58, 1017–1030. [Google Scholar] [CrossRef] [PubMed]
- Mackie, K. Distribution of Cannabinoid Receptors in the Central and Peripheral Nervous System. In Cannabinoids; Springer-Verlag: Berlin/Heidelberg, 2005; Volume 20, pp. 299–325. ISBN 354022565X. [Google Scholar]
- Pesce, M.; D’Alessandro, A.; Borrelli, O.; Gigli, S.; Seguella, L.; Cuomo, R.; Esposito, G.; Sarnelli, G. Endocannabinoid-related compounds in gastrointestinal diseases. J. Cell. Mol. Med. 2018, 22, 706–715. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, S.E. Endocannabinoids and the Cardiovascular System in Health and Disease. In Endocannabinoids; Springer: Cham, Switzerland, 2015; pp. 393–422. ISBN 9783319208251. [Google Scholar]
- Kaschina, E. Cannabinoid CB1/CB2 Receptors in the Heart: Expression, Regulation, and Function. In Cannabinoids in Health and Disease; InTech: London, UK, 2016; Volume i, p. 13. [Google Scholar]
- Dhopeshwarkar, A.; Mackie, K. CB 2 Cannabinoid Receptors as a Therapeutic Target—What Does the Future Hold? Mol. Pharmacol. 2014, 86, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, L.A.; Lolait, S.J.; Brownstein, M.J.; Young, A.C.; Bonner, T.I. Structure of a cannabinoid receptor and functional expression of the cloned cDNA. Nature 1990, 346, 561–564. [Google Scholar] [CrossRef] [PubMed]
- Zou, S.; Kumar, U. Cannabinoid receptors and the endocannabinoid system: Signaling and function in the central nervous system. Int. J. Mol. Sci. 2018, 19, 833. [Google Scholar]
- Iannotti, F.A.; Di Marzo, V.; Petrosino, S. Endocannabinoids and endocannabinoid-related mediators: Targets, metabolism and role in neurological disorders. Prog. Lipid Res. 2016, 62, 107–128. [Google Scholar] [CrossRef]
- Frassinetti, S.; Moccia, E.; Caltavuturo, L.; Gabriele, M.; Longo, V.; Bellani, L.; Giorgi, G.; Giorgetti, L. Nutraceutical potential of hemp (Cannabis sativa L.) seeds and sprouts. Food Chem. 2018, 262, 56–66. [Google Scholar] [CrossRef]
- Brighenti, V.; Pellati, F.; Steinbach, M.; Maran, D.; Benvenuti, S. Development of a new extraction technique and HPLC method for the analysis of non-psychoactive cannabinoids in fibre-type Cannabis sativa L. (hemp). J. Pharm. Biomed. Anal. 2017, 143, 228–236. [Google Scholar] [CrossRef]
- Pellati, F.; Borgonetti, V.; Brighenti, V.; Biagi, M.; Benvenuti, S.; Corsi, L. Cannabis sativa L. and Nonpsychoactive Cannabinoids: Their Chemistry and Role against Oxidative Stress, Inflammation, and Cancer. Biomed Res. Int. 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Bayewitch, M.; Rhee, M.H.; Avidor-Reiss, T.; Breuer, A.; Mechoulam, R.; Vogel, Z. (-)-Δ9-Tetrahydrocannabinol antagonizes the peripheral cannabinoid receptor-mediated inhibition of adenylyl cyclase. J. Biol. Chem. 1996, 271, 9902–9905. [Google Scholar] [CrossRef] [PubMed]
- Lauckner, J.E.; Jensen, J.B.; Chen, H.Y.; Lu, H.C.; Hille, B.; Mackie, K. GPR55 is a cannabinoid receptor that increases intracellular calcium and inhibits M current. Proc. Natl. Acad. Sci. USA 2008, 105, 2699–2704. [Google Scholar] [CrossRef] [PubMed]
- McHugh, D.; Page, J.; Dunn, E.; Bradshaw, H.B. Δ 9-tetrahydrocannabinol and N-arachidonyl glycine are full agonists at GPR18 receptors and induce migration in human endometrial HEC-1B cells. Br. J. Pharmacol. 2012, 165, 2414–2424. [Google Scholar] [CrossRef] [PubMed]
- Console-Bram, L.; Brailoiu, E.; Brailoiu, G.C.; Sharir, H.; Abood, M.E. Activation of GPR18 by cannabinoid compounds: A tale of biased agonism. Br. J. Pharmacol. 2014, 171, 3908–3917. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.H.S.; Isaev, D.; Morales, M.; Petroianu, G.; Galadari, S.; Oz, M. The effect of δ9-tetrahydrocannabinol on 5-HT3 receptors depends on the current density. Neuroscience 2010, 171, 40–49. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, S.E.; Tarling, E.J.; Bennett, A.J.; Kendall, D.A.; Randall, M.D. Novel time-dependent vascular actions of Δ9- tetrahydrocannabinol mediated by peroxisome proliferator-activated receptor gamma. Biochem. Biophys. Res. Commun. 2005, 337, 824–831. [Google Scholar] [CrossRef]
- O’Sullivan, S.E.; Kendall, D.A.; Randall, M.D. Further characterization of the time-dependent vascular effects of Δ9-tetrahydrocannabinol. J. Pharmacol. Exp. Ther. 2006, 317, 428–438. [Google Scholar] [CrossRef]
- De Petrocellis, L.; Vellani, V.; Schiano-Moriello, A.; Marini, P.; Magherini, P.C.; Orlando, P.; Di Marzo, V. Plant-derived cannabinoids modulate the activity of transient receptor potential channels of ankyrin type-1 and melastatin type-8. J. Pharmacol. Exp. Ther. 2008, 325, 1007–1015. [Google Scholar] [CrossRef]
- Qin, N.; Neeper, M.P.; Liu, Y.; Hutchinson, T.L.; Lubin, M.L.; Flores, C.M. TRPV2 is activated by cannabidiol and mediates CGRP release in cultured rat dorsal root ganglion neurons. J. Neurosci. 2008, 28, 6231–6238. [Google Scholar] [CrossRef]
- De Petrocellis, L.; Orlando, P.; Moriello, A.S.; Aviello, G.; Stott, C.; Izzo, A.A.; di Marzo, V. Cannabinoid actions at TRPV channels: Effects on TRPV3 and TRPV4 and their potential relevance to gastrointestinal inflammation. Acta Physiol. 2012, 204, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Vaysse, P.J.; Gardner, E.L.; Zukin, R.S. Modulation of Rat Brain Opioid Receptors by Cannabinoids1. J. Pharmacol. Exp. Ther. 1987, 241, 534–539. [Google Scholar] [PubMed]
- Kathmann, M.; Flau, K.; Redmer, A.; Tränkle, C.; Schlicker, E. Cannabidiol is an allosteric modulator at mu- and delta-opioid receptors. Naunyn. Schmiedebergs. Arch. Pharmacol. 2006, 372, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Xiong, W.; Cheng, K.; Cui, T.; Godlewski, G.; Rice, K.C.; Xu, Y.; Zhang, L. Cannabinoid potentiation of glycine receptors contributes to cannabis-induced analgesia. Nat. Chem. Biol. 2011, 7, 296–303. [Google Scholar] [CrossRef] [PubMed]
- Bisogno, T.; Hanuš, L.; De Petrocellis, L.; Tchilibon, S.; Ponde, D.E.; Brandi, I.; Moriello, A.S.; Davis, J.B.; Mechoulam, R.; Di Marzo, V. Molecular targets for cannabidiol and its synthetic analogues: Effect on vanilloid VR1 receptors and on the cellular uptake and enzymatic hydrolysis of anandamide. Br. J. Pharmacol. 2001, 134, 845–852. [Google Scholar] [CrossRef] [PubMed]
- De Petrocellis, L.; Ligresti, A.; Moriello, A.S.; Allarà, M.; Bisogno, T.; Petrosino, S.; Stott, C.G.; Di Marzo, V. Effects of cannabinoids and cannabinoid-enriched Cannabis extracts on TRP channels and endocannabinoid metabolic enzymes. Br. J. Pharmacol. 2011, 163, 1479–1494. [Google Scholar] [CrossRef] [PubMed]
- Elmes, M.W.; Kaczocha, M.; Berger, W.T.; Leung, K.N.; Ralph, B.P.; Wang, L.; Sweeney, J.M.; Miyauchi, J.T.; Tsirka, S.E.; Ojima, I.; et al. Fatty acid-binding proteins (FABPs) are intracellular carriers for Δ9-tetrahydrocannabinol (THC) and cannabidiol (CBD). J. Biol. Chem. 2015, 290, 8711–8721. [Google Scholar] [CrossRef] [PubMed]
- Carrier, E.J.; Auchampach, J.A.; Hillard, C.J. Inhibition of an equilibrative nucleoside transporter by cannabidiol: A mechanism of cannabinoid immunosuppression. Proc. Natl. Acad. Sci. USA 2006, 103, 7895–7900. [Google Scholar] [CrossRef]
- Pertwee, R.G. Handbook of Cannabis; Pertwee, R.G., Ed.; Oxford University Press: Oxford, UK, 2014. [Google Scholar]
- Laprairie, R.B.; Bagher, A.M.; Kelly, M.E.M.; Denovan-Wright, E.M. Cannabidiol is a negative allosteric modulator of the cannabinoid CB1 receptor. Br. J. Pharmacol. 2015, 172, 4790–4805. [Google Scholar] [CrossRef]
- Martínez-Pinilla, E.; Varani, K.; Reyes-Resina, I.; Angelats, E.; Vincenzi, F.; Ferreiro-Vera, C.; Oyarzabal, J.; Canela, E.I.; Lanciego, J.L.; Nadal, X.; et al. Binding and signaling studies disclose a potential allosteric site for cannabidiol in cannabinoid CB2 receptors. Front. Pharmacol. 2017, 8, 1–10. [Google Scholar] [CrossRef]
- Ryberg, E.; Larsson, N.; Sjögren, S.; Hjorth, S.; Hermansson, N.O.; Leonova, J.; Elebring, T.; Nilsson, K.; Drmota, T.; Greasley, P.J. The orphan receptor GPR55 is a novel cannabinoid receptor. Br. J. Pharmacol. 2007, 152, 1092–1101. [Google Scholar] [CrossRef] [PubMed]
- Laun, A.S.; Song, Z.H. GPR3 and GPR6, novel molecular targets for cannabidiol. Biochem. Biophys. Res. Commun. 2017, 490, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.J.; Laun, A.S.; Song, Z.H. Cannabidiol, a novel inverse agonist for GPR12. Biochem. Biophys. Res. Commun. 2017, 493, 451–454. [Google Scholar] [CrossRef] [PubMed]
- Gonca, E.; Darici, F. The Effect of Cannabidiol on ischemia/reperfusion-induced ventricular arrhythmias: The role of adenosine a1 receptors. J. Cardiovasc. Pharmacol. Ther. 2015, 20, 76–83. [Google Scholar] [CrossRef]
- Russo, E.B.; Burnett, A.; Hall, B.; Parker, K.K. Agonistic properties of cannabidiol at 5-HT1a receptors. Neurochem. Res. 2005, 30, 1037–1043. [Google Scholar] [CrossRef]
- Yang, K.H.; Galadari, S.; Isaev, D.; Petroianu, G.; Shippenberg, T.S.; Oz, M. The nonpsychoactive cannabinoid cannabidiol inhibits 5- hydroxytryptamine3A receptor-mediated currents in xenopus laevis oocytes. J. Pharmacol. Exp. Ther. 2010, 333, 547–554. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, S.E.; Sun, Y.; Bennett, A.J.; Randall, M.D.; Kendall, D.A. Time-dependent vascular actions of cannabidiol in the rat aorta. Eur. J. Pharmacol. 2009, 612, 61–68. [Google Scholar] [CrossRef]
- Bakas, T.; van Nieuwenhuijzen, P.S.; Devenish, S.O.; McGregor, I.S.; Arnold, J.C.; Chebib, M. The direct actions of cannabidiol and 2-arachidonoyl glycerol at GABAA receptors. Pharmacol. Res. 2017, 119, 358–370. [Google Scholar] [CrossRef]
- Ahrens, J.; Demir, R.; Leuwer, M.; De La Roche, J.; Krampfl, K.; Foadi, N.; Karst, M.; Haeseler, G. The nonpsychotropic cannabinoid cannabidiol modulates and directly activates alpha-1 and alpha-1-beta glycine receptor function. Pharmacology 2009, 83, 217–222. [Google Scholar] [CrossRef]
- Xiong, W.; Cui, T.; Cheng, K.; Yang, F.; Chen, S.R.; Willenbring, D.; Guan, Y.; Pan, H.L.; Ren, K.; Xu, Y.; et al. Cannabinoids suppress inflammatory and neuropathic pain by targeting α3 glycine receptors. J. Exp. Med. 2012, 209, 1121–1134. [Google Scholar] [CrossRef]
- Rosenthaler, S.; Pöhn, B.; Kolmanz, C.; Nguyen Huu, C.; Krewenka, C.; Huber, A.; Kranner, B.; Rausch, W.D.; Moldzio, R. Differences in receptor binding affinity of several phytocannabinoids do not explain their effects on neural cell cultures. Neurotoxicol. Teratol. 2014, 46, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Navarro, G.; Varani, K.; Reyes-Resina, I.; de Medina, V.S.; Rivas-Santisteban, R.; Callado, C.S.C.; Vincenzi, F.; Casano, S.; Ferreiro-Vera, C.; Canela, E.I.; et al. Cannabigerol action at cannabinoid CB1 and CB2 receptors and at CB1-CB2 heteroreceptor complexes. Front. Pharmacol. 2018, 9, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Anavi-Goffer, S.; Baillie, G.; Irving, A.J.; Gertsch, J.; Greig, I.R.; Pertwee, R.G.; Ross, R.A. Modulation of L-α-lysophosphatidylinositol/GPR55 mitogen-activated protein kinase (MAPK) signaling by cannabinoids. J. Biol. Chem. 2012, 287, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Cascio, M.G.; Gauson, L.A.; Stevenson, L.A.; Ross, R.A.; Pertwee, R.G. Evidence that the plant cannabinoid cannabigerol is a highly potent α 2-adrenoceptor agonist and moderately potent 5HT 1A receptor antagonist. Br. J. Pharmacol. 2010, 159, 129–141. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.J.; Jones, N.A.; Smith, I.; Hill, C.L.; Williams, C.M.; Stephens, G.J.; Whalley, B.J. Voltage-gated sodium (NaV) channel blockade by plant cannabinoids does not confer anticonvulsant effects per se. Neurosci. Lett. 2014, 566, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Udoh, M.; Santiago, M.; Devenish, S.; McGregor, I.S.; Connor, M. Cannabichromene is a cannabinoid CB2 receptor agonist. Br. J. Pharmacol. 2019, 176, 4537–4547. [Google Scholar] [CrossRef] [PubMed]
- Bolognini, D.; Rock, E.M.; Cluny, N.L.; Cascio, M.G.; Limebeer, C.L.; Duncan, M.; Stott, C.G.; Javid, F.A.; Parker, L.A.; Pertwee, R.G. Cannabidiolic acid prevents vomiting in Suncus murinus and nausea-induced behaviour in rats by enhancing 5-HT1A receptor activation. Br. J. Pharmacol. 2013, 168, 1456–1470. [Google Scholar] [CrossRef]
- Maroon, J.; Bost, J. Review of the neurological benefits of phytocannabinoids. Surg. Neurol. Int. 2018, 9, 91. [Google Scholar] [CrossRef]
- Boggs, D.L.; Nguyen, J.D.; Morgenson, D.; Taffe, M.A.; Ranganathan, M. Clinical and Preclinical Evidence for Functional Interactions of Cannabidiol and Δ9-Tetrahydrocannabinol. Neuropsychopharmacology 2018, 43, 142–154. [Google Scholar] [CrossRef]
- Hudson, R.; Renard, J.; Norris, C.; Rushlow, W.J.; Laviolette, S.R. Cannabidiol Counteracts the Psychotropic Side-Effects of Δ-9-Tetrahydrocannabinol in the Ventral Hippocampus through Bidirectional Control of ERK1–2 Phosphorylation. J. Neurosci. 2019, 39, 8762–8777. [Google Scholar] [CrossRef]
- Pisanti, S.; Malfitano, A.M.; Ciaglia, E.; Lamberti, A.; Ranieri, R.; Cuomo, G.; Abate, M.; Faggiana, G.; Proto, M.C.; Fiore, D.; et al. Cannabidiol: State of the art and new challenges for therapeutic applications. Pharmacol. Ther. 2017, 175, 133–150. [Google Scholar] [CrossRef] [PubMed]
- Noreen, N.; Muhammad, F.; Akhtar, B.; Azam, F.; Anwar, M.I. Is cannabidiol a promising substance for new drug development? A review of its potential therapeutic applications. Crit. Rev. Eukaryot. Gene Expr. 2018, 28, 73–86. [Google Scholar] [CrossRef] [PubMed]
- Chagas, M.H.N.; Eckeli, A.L.; Zuardi, A.W.; Pena-Pereira, M.A.; Sobreira-Neto, M.A.; Sobreira, E.T.; Camilo, M.R.; Bergamaschi, M.M.; Schenck, C.H.; Hallak, J.E.C.; et al. Cannabidiol can improve complex sleep-related behaviours associated with rapid eye movement sleep behaviour disorder in Parkinson’s disease patients: A case series. J. Clin. Pharm. Ther. 2014, 39, 564–566. [Google Scholar] [CrossRef] [PubMed]
- Shannon, S.; Lewis, N.; Lee, H.; Hughes, S. Cannabidiol in Anxiety and Sleep: A Large Case Series. Perm. J. 2019, 23, 18–041. [Google Scholar] [CrossRef] [PubMed]
- Pinto, J.V.; Saraf, G.; Frysch, C.; Vigo, D.; Keramatian, K.; Chakrabarty, T.; Lam, R.W.; Kauer-Sant’Anna, M.; Yatham, L.N. Cannabidiol as a Treatment for Mood Disorders: A Systematic Review. Can. J. Psychiatry 2020, 65, 213–227. [Google Scholar] [CrossRef] [PubMed]
- Peres, F.F.; Lima, A.C.; Hallak, J.E.C.; Crippa, J.A.; Silva, R.H.; Abílio, V.C. Cannabidiol as a promising strategy to treat and prevent movement disorders? Front. Pharmacol. 2018, 9, 1–12. [Google Scholar] [CrossRef]
- Aso, E.; Fernández-Dueñas, V.; López-Cano, M.; Taura, J.; Watanabe, M.; Ferrer, I.; Luján, R.; Ciruela, F. Adenosine A2A-Cannabinoid CB1 Receptor Heteromers in the Hippocampus: Cannabidiol Blunts Δ9-Tetrahydrocannabinol-Induced Cognitive Impairment. Mol. Neurobiol. 2019, 56, 5382–5391. [Google Scholar] [CrossRef]
- Samanta, D. Cannabidiol: A Review of Clinical Efficacy and Safety in Epilepsy. Pediatr. Neurol. 2019, 96, 24–29. [Google Scholar] [CrossRef]
- Premoli, M.; Aria, F.; Bonini, S.A.; Maccarinelli, G.; Gianoncelli, A.; Della Pina, S.; Tambaro, S.; Memo, M.; Mastinu, A. Cannabidiol: Recent advances and new insights for neuropsychiatric disorders treatment. Life Sci. 2019, 224, 120–127. [Google Scholar] [CrossRef]
- Hill, A.J.; Mercier, M.S.; Hill, T.D.M.; Glyn, S.E.; Jones, N.A.; Yamasaki, Y.; Futamura, T.; Duncan, M.; Stott, C.G.; Stephens, G.J.; et al. Cannabidivarin is anticonvulsant in mouse and rat. Br. J. Pharmacol. 2012, 167, 1629–1642. [Google Scholar] [CrossRef]
- Vigli, D.; Cosentino, L.; Raggi, C.; Laviola, G.; Woolley-Roberts, M.; De Filippis, B. Chronic treatment with the phytocannabinoid Cannabidivarin (CBDV) rescues behavioural alterations and brain atrophy in a mouse model of Rett syndrome. Neuropharmacology 2018, 140, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Zamberletti, E.; Gabaglio, M.; Woolley-Roberts, M.; Bingham, S.; Rubino, T.; Parolaro, D. Cannabidivarin Treatment Ameliorates Autism-Like Behaviors and Restores Hippocampal Endocannabinoid System and Glia Alterations Induced by Prenatal Valproic Acid Exposure in Rats. Front. Cell. Neurosci. 2019, 13, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Pretzsch, C.M.; Freyberg, J.; Voinescu, B.; Lythgoe, D.; Horder, J.; Mendez, M.A.; Wichers, R.; Ajram, L.; Ivin, G.; Heasman, M.; et al. Effects of cannabidiol on brain excitation and inhibition systems; a randomised placebo-controlled single dose trial during magnetic resonance spectroscopy in adults with and without autism spectrum disorder. Neuropsychopharmacology 2019, 44, 1398–1405. [Google Scholar] [CrossRef] [PubMed]
- Izzo, A.A.; Sharkey, K.A. Cannabinoids and the gut: New developments and emerging concepts. Pharmacol. Ther. 2010, 126, 21–38. [Google Scholar] [CrossRef] [PubMed]
- Sharkey, K.A.; Wiley, J.W. The Role of the Endocannabinoid System in the Brain–Gut Axis. Gastroenterology 2016, 151, 252–266. [Google Scholar] [CrossRef]
- Taschler, U.; Hasenoehrl, C.; Storr, M.; Schicho, R. Cannabinoid Receptors in Regulating the GI Tract: Experimental Evidence and Therapeutic Relevance. In Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2016; Volume 239, pp. 343–362. ISBN 9783319563596. [Google Scholar]
- Joshi, N.; Onaivi, E.S. Endocannabinoid System Components: Overview and Tissue Distribution. In Advances in Experimental Medicine and Biology; Springer: Berlin/Heidelberg, Germany, 2019; Volume 1162, pp. 1–12. ISBN 9783030217365. [Google Scholar]
- Vera, G.; López-Pérez, A.E.; Uranga, J.A.; Girón, R.; Martín-Fontelles, M.I.; Abalo, R. Involvement of cannabinoid signaling in vincristine-induced gastrointestinal dysmotility in the rat. Front. Pharmacol. 2017, 8, 1–16. [Google Scholar] [CrossRef]
- Salaga, M.; Zatorski, H.; Zielińska, M.; Mosinska, P.; Timmermans, J.P.; Kordek, R.; Storr, M.; Fichna, J. Highly selective CB2 receptor agonist A836339 has gastroprotective effect on experimentally induced gastric ulcers in mice. Naunyn. Schmiedebergs. Arch. Pharmacol. 2017, 390, 1015–1027. [Google Scholar] [CrossRef]
- Abalo, R.; Vera, G.; López-Pérez, A.E.; Martnez-Villaluenga, M.; Martín-Fontelles, M.I. The gastrointestinal pharmacology of cannabinoids: Focus on motility. Pharmacology 2012, 90, 1–10. [Google Scholar] [CrossRef]
- Malik, Z.; Baik, D.; Schey, R. The Role of Cannabinoids in Regulation of Nausea and Vomiting, and Visceral Pain. Curr. Gastroenterol. Rep. 2015, 17, 9. [Google Scholar] [CrossRef]
- Gyires, K.; S Zádori, Z. Role of Cannabinoids in Gastrointestinal Mucosal Defense and Inflammation. Curr. Neuropharmacol. 2016, 14, 935–951. [Google Scholar] [CrossRef]
- Hasenoehrl, C.; Taschler, U.; Storr, M.; Schicho, R. The gastrointestinal tract—A central organ of cannabinoid signaling in health and disease. Neurogastroenterol. Motil. 2016, 28, 1765–1780. [Google Scholar] [CrossRef] [PubMed]
- Acharya, N.; Penukonda, S.; Shcheglova, T.; Hagymasi, A.T.; Basu, S.; Srivastava, P.K. Endocannabinoid system acts as a regulator of immune homeostasis in the gut. Proc. Natl. Acad. Sci. USA 2017, 114, 5005–5010. [Google Scholar] [CrossRef] [PubMed]
- Abalo, R.; Martín-Fontelles, M.I. Cannabis, cannabinoids, and visceral pain. In Handbook of Cannabis and Related Pathologies, Biology, Pharmacology, Diagnosis, and Treatment; Preedy, V., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 439–449. ISBN 9780128007563. [Google Scholar]
- Camilleri, M. Cannabinoids and gastrointestinal motility: Pharmacology, clinical effects, and potential therapeutics in humans. Neurogastroenterol. Motil. 2018, 30, e13370. [Google Scholar] [CrossRef] [PubMed]
- Uranga, J.A.; Vera, G.; Abalo, R. Cannabinoid pharmacology and therapy in gut disorders. Biochem. Pharmacol. 2018, 157, 134–147. [Google Scholar] [CrossRef] [PubMed]
- Pinto, L.; Izzo, A.A.; Cascio, M.G.; Bisogno, T.; Hospodar-Scott, K.; Brown, D.R.; Mascolo, N.; Di Marzo, V.; Capasso, F. Endocannabinoids as physiological regulators of colonic propulsion in mice. Gastroenterology 2002, 123, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Aviello, G.; Romano, B.; Izzo, A.A. Cannabinoids and gastrointestinal motility: Animal and human studies. Eur. Rev. Med. Pharmacol. Sci. 2008, 12, 81–93. [Google Scholar]
- Fioramonti, J.; Bueno, L. Role of cannabinoid receptors in the control of gastrointestinal motility and perception. Expert Rev. Gastroenterol. Hepatol. 2008, 2, 385–397. [Google Scholar] [CrossRef]
- Salama, R.A.A.; Abdelsalam, R.M.; Abdel-Salam, O.M.E.; Khattab, M.M.; Salem, N.A.; El-Khyat, Z.A.; Morsy, F.A.; Eldenshary, E.E.D.S. Modulation of gastric acid secretion by cannabinoids in rats. J. Biochem. Mol. Toxicol. 2019, 33. [Google Scholar] [CrossRef]
- Brusberg, M.; Arvidsson, S.; Kang, D.; Larsson, H.; Lindström, E.; Martinez, V. CB1 receptors mediate the analgesic effects of cannabinoids on colorectal distension-induced visceral pain in rodents. J. Neurosci. 2009, 29, 1554–1564. [Google Scholar] [CrossRef]
- Alhouayek, M.; Muccioli, G.G. The endocannabinoid system in inflammatory bowel diseases: From pathophysiology to therapeutic opportunity. Trends Mol. Med. 2012, 18, 615–625. [Google Scholar] [CrossRef]
- Amin, M.R.; Ali, D.W. Pharmacology of Medical Cannabis. In Recent Advances in Cannabinoid Physiology and Pathology. Advances in Experimental Medicine and Biology; Bukiya, A., Ed.; Springer: Cham, Switzerland, 2019; pp. 151–165. [Google Scholar]
- Cogan, P.S. On healthcare by popular appeal: Critical assessment of benefit and risk in cannabidiol based dietary supplements. Expert Rev. Clin. Pharmacol. 2019, 12, 501–511. [Google Scholar] [CrossRef] [PubMed]
- Williamson, E.M.; Liu, X.; Izzo, A.A. Trends in use, pharmacology, and clinical applications of emerging herbal nutraceuticals. Br. J. Pharmacol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Drossman, D.A. Functional gastrointestinal disorders: History, pathophysiology, clinical features, and Rome IV. Gastroenterology 2016, 150, 1262–1279.e2. [Google Scholar] [CrossRef] [PubMed]
- Hadjivasilis, A.; Tsioutis, C.; Michalinos, A.; Ntourakis, D.; Christodoulou, D.K.; Agouridis, A.P. New insights into irritable bowel syndrome: From pathophysiology to treatment. Ann. Gastroenterol. 2019, 32, 554–564. [Google Scholar] [CrossRef] [PubMed]
- Hellström, P.M. Pathophysiology of the irritable bowel syndrome—Reflections of today. Best Pract. Res. Clin. Gastroenterol. 2019, 40–41. [Google Scholar] [CrossRef]
- Labanski, A.; Langhorst, J.; Engler, H.; Elsenbruch, S. Stress and the brain-gut axis in functional and chronic-inflammatory gastrointestinal diseases: A transdisciplinary challenge. Psychoneuroendocrinology 2020, 111. [Google Scholar] [CrossRef]
- Grad, S.; Dumitrascu, D.L. Irritable Bowel Syndrome Subtypes: New Names for Old Medical Conditions. Dig. Dis. 2020, 38, 122–127. [Google Scholar] [CrossRef]
- Chester, G.B.; Dahl, C.J.; Everingham, M.; Jackson, D.M.; Marchant-Williams, H.; Starmer, G.A. The effect of cannabinoids on intestinal motility and their antinociceptive effect in mice. Br. J. Pharmacol. 1973, 49, 588–594. [Google Scholar] [CrossRef]
- Shook, J.E.; Burks, T.F. Psychoactive cannabinoids reduce gastrointestinal propulsion and motility in rodents. J. Pharmacol. Exp. Ther. 1989, 249, 444–449. [Google Scholar]
- Fride, E.; Ponde, D.; Breuer, A.; Hanuš, L. Peripheral, but not central effects of cannabidiol derivatives: Mediation by CB1 and unidentified receptors. Neuropharmacology 2005, 48, 1117–1129. [Google Scholar] [CrossRef]
- Izzo, A.A.; Capasso, R.; Aviello, G.; Borrelli, F.; Romano, B.; Piscitelli, F.; Gallo, L.; Capasso, F.; Orlando, P.; Di Marzo, V. Inhibitory effect of cannabichromene, a major non-psychotropic cannabinoid extracted from Cannabis sativa, on inflammation-induced hypermotility in mice. Br. J. Pharmacol. 2012, 166, 1444–1460. [Google Scholar] [CrossRef] [PubMed]
- Capasso, R.; Borrelli, F.; Aviello, G.; Romano, B.; Scalisi, C.; Capasso, F.; Izzo, A.A. Cannabidiol, extracted from Cannabis sativa, selectively inhibits inflammatory hypermotility in mice. Br. J. Pharmacol. 2008, 154, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- Pagano, E.; Romano, B.; Iannotti, F.A.; Parisi, O.A.; D’Armiento, M.; Pignatiello, S.; Coretti, L.; Lucafò, M.; Venneri, T.; Stocco, G.; et al. The non-euphoric phytocannabinoid cannabidivarin counteracts intestinal inflammation in mice and cytokine expression in biopsies from UC pediatric patients. Pharmacol. Res. 2019, 149, 104464. [Google Scholar] [CrossRef] [PubMed]
- Izzo, A.A.; Pinto, L.; Borrelli, F.; Capasso, R.; Mascolo, N.; Capasso, F. Central and peripheral cannabinoid modulation of gastrointestinal transit in physiological states or during the diarrhoea induced by croton oil. Br. J. Pharmacol. 2000, 129, 1627–1632. [Google Scholar] [CrossRef] [PubMed]
- Izzo, A.A.; Fezza, F.; Capasso, R.; Bisogno, T.; Pinto, L.; Iuvone, T.; Esposito, G.; Mascolo, N.; Di Marzo, V.; Capasso, F. Cannabinoid CB1-receptor mediated regulation of gastrointestinal motility in mice in a model of intestinal inflammation. Br. J. Pharmacol. 2001, 134, 563–570. [Google Scholar] [CrossRef] [PubMed]
- Pagano, E.; Capasso, R.; Piscitelli, F.; Romano, B.; Parisi, O.A.; Finizio, S.; Lauritano, A.; Di Marzo, V.; Izzo, A.A.; Borrelli, F. An orally active Cannabis extract with high content in cannabidiol attenuates chemically-induced intestinal inflammation and hypermotility in the mouse. Front. Pharmacol. 2016, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.H.; Yuece, B.; Li, Y.Y.; Feng, Y.J.; Feng, J.Y.; Yu, L.Y.; Li, K.; Li, Y.N.; Storr, M. A novel CB receptor GPR55 and its ligands are involved in regulation of gut movement in rodents. Neurogastroenterol. Motil. 2011, 23. [Google Scholar] [CrossRef]
- Wong, B.S.; Camilleri, M.; Busciglio, I.; Carlson, P.; Szarka, L.A.; Burton, D.; Zinsmeister, A.R. Pharmacogenetic Trial of a Cannabinoid Agonist Shows Reduced Fasting Colonic Motility in Patients With Nonconstipated Irritable Bowel Syndrome. Gastroenterology 2011, 141, 1638–1647.e7. [Google Scholar] [CrossRef]
- Wong, B.S.; Camilleri, M.; Eckert, D.; Carlson, P.; Ryks, M.; Burton, D.; Zinsmeister, A.R. Randomized pharmacodynamic and pharmacogenetic trial of dronabinol effects on colon transit in irritable bowel syndrome-diarrhea. Neurogastroenterol. Motil. 2012, 24. [Google Scholar] [CrossRef]
- Formukong, E.A.; Evans, A.T.; Evans, F.J. Analgesic and antiinflammatory activity of constituents of Cannabis sativa L. Inflammation 1988, 12, 361–371. [Google Scholar] [CrossRef]
- Sanson, M.; Bueno, L.; Fioramonti, J. Involvement of cannabinoid receptors in inflammatory hypersensitivity to colonic distension in rats. Neurogastroenterol. Motil. 2006, 18, 949–956. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, A.; Ohashi, K.; Sugie, Y.; Sugimoto, H.; Omura, H. Pharmacological evaluation of a novel cannabinoid 2 (CB2) ligand, PF-03550096, in vitro and in vivo by using a rat model of visceral hypersensitivity. J. Pharmacol. Sci. 2008, 106, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Booker, L.; Naidu, P.S.; Razdan, R.K.; Mahadevan, A.; Lichtman, A.H. Evaluation of prevalent phytocannabinoids in the acetic acid model of visceral nociception. Drug Alcohol Depend. 2009, 105, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Fan, J.; Kemmerer, E.S.; Evans, S.; Li, Y.; Wiley, J.W. Reciprocal changes in vanilloid (TRPV1) and endocannabinoid (CB1) receptors contribute to visceral hyperalgesia in the water avoidance stressed rat. Gut 2009, 58, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Ravnefjord, A.; Brusberg, M.; Kang, D.; Bauer, U.; Larsson, H.; Lindström, E.; Martinez, V. Involvement of the transient receptor potential vanilloid 1 (TRPV1) in the development of acute visceral hyperalgesia during colorectal distension in rats. Eur. J. Pharmacol. 2009, 611, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Naidu, P.S.; Booker, L.; Cravatt, B.F.; Lichtman, A.H. Synergy between enzyme inhibitors of fatty acid amide hydrolase and cyclooxygenase in visceral nociception. J. Pharmacol. Exp. Ther. 2009, 329, 48–56. [Google Scholar] [CrossRef]
- Iwata, Y.; Ando, K.; Taniguchi, K.; Koba, N.; Sugiura, A.; Sudo, M. Identification of a highly potent and selective CB2 agonist, RQ-00202730, for the treatment of irritable bowel syndrome. Bioorganic Med. Chem. Lett. 2015, 25, 236–240. [Google Scholar] [CrossRef]
- Klooker, T.K.; Leliefeld, K.E.M.; Van Den Wijngaard, R.M.; Boeckxstaens, G.E.E. The cannabinoid receptor agonist delta- 9- tetrahydrocannabinol does not affect visceral sensitivity to rectal distension in healthy volunteers and IBS patients. Neurogastroenterol. Motil. 2011, 23, 30. [Google Scholar] [CrossRef]
- De Vries, M.; van Rijckevorsel, D.C.M.; Vissers, K.C.P.; Wilder-Smith, O.H.G.; van Goor, H. Tetrahydrocannabinol Does Not Reduce Pain in Patients With Chronic Abdominal Pain in a Phase 2 Placebo-controlled Study. Clin. Gastroenterol. Hepatol. 2017, 15, 1079–1086.e4. [Google Scholar] [CrossRef]
- Buggy, D.J.; Toogood, L.; Maric, S.; Sharpe, P.; Lambert, D.G.; Rowbotham, D.J. Lack of analgesic efficacy of oral δ-9-tetrahydrocannabinol in postoperative pain. Pain 2003, 106, 169–172. [Google Scholar] [CrossRef]
- Guan, Q. A Comprehensive Review and Update on the Pathogenesis of Inflammatory Bowel Disease. J. Immunol. Res. 2019, 2019. [Google Scholar] [CrossRef] [PubMed]
- Lamb, C.A.; Kennedy, N.A.; Raine, T.; Hendy, P.A.; Smith, P.J.; Limdi, J.K.; Hayee, B.; Lomer, M.C.E.; Parkes, G.C.; Selinger, C.; et al. British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults. Gut 2019, 68, s1–s106. [Google Scholar] [CrossRef] [PubMed]
- Eichele, D.D.; Young, R. Medical Management of Inflammatory Bowel Disease. Surg. Clin. North Am. 2019, 99, 1223–1235. [Google Scholar] [CrossRef] [PubMed]
- Tamilarasan, A.G.; Cunningham, G.; Irving, P.M.; Samaan, M.A. Recent advances in monoclonal antibody therapy in IBD: Practical issues. Frontline Gastroenterol. 2019, 10, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Quezada, S.M.; McLean, L.P.; Cross, R.K. Adverse events in IBD therapy: The 2018 update. Expert Rev. Gastroenterol. Hepatol. 2018, 12, 1183–1191. [Google Scholar] [CrossRef]
- Picardo, S.; Kaplan, G.G.; Sharkey, K.A.; Seow, C.H. Insights into the role of cannabis in the management of inflammatory bowel disease. Therap. Adv. Gastroenterol. 2019, 12, 175628481987097. [Google Scholar] [CrossRef]
- Kienzl, M.; Storr, M.; Schicho, R. Cannabinoids and Opioids in the Treatment of Inflammatory Bowel Diseases. Clin. Transl. Gastroenterol. 2020, 11, e00120. [Google Scholar] [CrossRef]
- Lal, S.; Prasad, N.; Ryan, M.; Tangri, S.; Silverberg, M.S.; Gordon, A.; Steinhart, H. Cannabis use amongst patients with inflammatory bowel disease. Eur. J. Gastroenterol. Hepatol. 2011, 23, 891–896. [Google Scholar] [CrossRef]
- Ravikoff Allegretti, J.; Courtwright, A.; Lucci, M.; Korzenik, J.R.; Levine, J. Marijuana Use Patterns Among Patients with Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2013, 19, 2809–2814. [Google Scholar] [CrossRef]
- Storr, M.; Devlin, S.; Kaplan, G.G.; Panaccione, R.; Andrews, C.N. Cannabis use provides symptom relief in patients with inflammatory bowel disease but is associated with worse disease prognosis in patients with Crohn’s disease. Inflamm. Bowel Dis. 2014, 20, 472–480. [Google Scholar] [CrossRef]
- Esposito, G.; Filippis, D.D.; Cirillo, C.; Iuvone, T.; Capoccia, E.; Scuderi, C.; Steardo, A.; Cuomo, R.; Steardo, L. Cannabidiol in Inflammatory Bowel Diseases: A Brief Overview. Phyther. Res. 2013, 27, 633–636. [Google Scholar] [CrossRef]
- Leinwand, K.L.; Gerich, M.E.; Hoffenberg, E.J.; Collins, C.B. Manipulation of the Endocannabinoid System in Colitis. Inflamm. Bowel Dis. 2017, 23, 192–199. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Couch, D.G.; Maudslay, H.; Doleman, B.; Lund, J.N.; O’Sullivan, S.E. The Use of Cannabinoids in Colitis: A Systematic Review and Meta-Analysis. Inflamm. Bowel Dis. 2018, 24, 680–697. [Google Scholar] [CrossRef]
- Turcotte, C.; Chouinard, F.; Lefebvre, J.S.; Flamand, N. Regulation of inflammation by cannabinoids, the endocannabinoids 2-arachidonoyl-glycerol and arachidonoyl-ethanolamide, and their metabolites. J. Leukoc. Biol. 2015, 97, 1049–1070. [Google Scholar] [CrossRef] [PubMed]
- Zoppi, S.; Madrigal, J.L.M.; Pérez-Nievas, B.G.; Marín-Jiménez, I.; Caso, J.R.; Alou, L.; García-Bueno, B.; Colón, A.; Manzanares, J.; Luisa Gómez-Lus, M.; et al. Endogenous cannabinoid system regulates intestinal barrier function in vivo through cannabinoid type 1 receptor activation. Am. J. Physiol.-Gastrointest. Liver Physiol. 2012, 302, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Izzo, A.A.; Capasso, F.; Costagliola, A.; Bisogno, T.; Marsicano, G.; Ligresti, A.; Matias, I.; Capasso, R.; Pinto, L.; Borrelli, F.; et al. An endogenous cannabinoid tone attenuates cholera toxin-induced fluid accumulation in mice. Gastroenterology 2003, 125, 765–774. [Google Scholar] [CrossRef]
- Mathison, R.; Ho, W.; Pittman, Q.J.; Davison, J.S.; Sharkey, K.A. Effects of cannabinoid receptor-2 activation on accelerated gastrointestinal transit in lipopolysaccharide-treated rats. Br. J. Pharmacol. 2004, 142, 1247–1254. [Google Scholar] [CrossRef]
- De Filippis, D.; Esposito, G.; Cirillo, C.; Cipriano, M.; de Winter, B.Y.; Scuderi, C.; Sarnelli, G.; Cuomo, R.; Steardo, L.; de Man, J.G.; et al. Cannabidiol reduces intestinal inflammation through the control of neuroimmune axis. PLoS ONE 2011, 6, e28159. [Google Scholar] [CrossRef]
- Schicho, R.; Storr, M. Topical and systemic cannabidiol improves trinitrobenzene sulfonic acid colitis in mice. Pharmacology 2012, 89, 149–155. [Google Scholar] [CrossRef]
- Jamontt, J.M.; Molleman, A.; Pertwee, R.G.; Parsons, M.E. The effects of Δ 9-tetrahydrocannabinol and cannabidiol alone and in combination on damage, inflammation and in vitro motility disturbances in rat colitis. Br. J. Pharmacol. 2010, 160, 712–723. [Google Scholar] [CrossRef]
- Borrelli, F.; Aviello, G.; Romano, B.; Orlando, P.; Capasso, R.; Maiello, F.; Guadagno, F.; Petrosino, S.; Capasso, F.; Di Marzo, V.; et al. Cannabidiol, a safe and non-psychotropic ingredient of the marijuana plant Cannabis sativa, is protective in a murine model of colitis. J. Mol. Med. 2009, 87, 1111–1121. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, F.; Fasolino, I.; Romano, B.; Capasso, R.; Maiello, F.; Coppola, D.; Orlando, P.; Battista, G.; Pagano, E.; Di Marzo, V.; et al. Beneficial effect of the non-psychotropic plant cannabinoid cannabigerol on experimental inflammatory bowel disease. Biochem. Pharmacol. 2013, 85, 1306–1316. [Google Scholar] [CrossRef] [PubMed]
- Romano, B.; Borrelli, F.; Fasolino, I.; Capasso, R.; Piscitelli, F.; Cascio, M.G.; Pertwee, R.G.; Coppola, D.; Vassallo, L.; Orlando, P.; et al. The cannabinoid TRPA1 agonist cannabichromene inhibits nitric oxide production in macrophages and ameliorates murine colitis. Br. J. Pharmacol. 2013, 169, 213–229. [Google Scholar] [CrossRef] [PubMed]
- Greco, M.; Varriale, G.; Coppola, G.; Operto, F.; Verrotti, A.; Iezzi, M.L. Investigational small molecules in phase II clinical trials for the treatment of epilepsy. Expert Opin. Investig. Drugs 2018, 27, 971–979. [Google Scholar] [CrossRef] [PubMed]
- De Petrocellis, L.; Di Marzo, V. Non-CB1, Non-CB2 receptors for endocannabinoids, plant cannabinoids, and synthetic cannabimimetics: Focus on G-protein-coupled receptors and transient receptor potential channels. J. Neuroimmune Pharmacol. 2010, 5, 103–121. [Google Scholar] [CrossRef] [PubMed]
- Kun, J.; Szitter, I.; Kemény, Á.; Perkecz, A.; Kereskai, L.; Pohóczky, K.; Vincze, Á.; Gódi, S.; Szabó, I.; Szolcsányi, J.; et al. Upregulation of the transient receptor potential ankyrin 1 ion channel in the inflamed human and mouse colon and its protective roles. PLoS ONE 2014, 9, e108164. [Google Scholar] [CrossRef]
- Khalil, M.; Alliger, K.; Weidinger, C.; Yerinde, C.; Wirtz, S.; Becker, C.; Engel, M.A. Functional role of transient receptor potential channels in immune cells and epithelia. Front. Immunol. 2018, 9, 1–7. [Google Scholar] [CrossRef]
- Rizopoulos, T.; Papadaki-Petrou, H.; Assimakopoulou, M. Expression Profiling of the Transient Receptor Potential Vanilloid (TRPV) Channels 1, 2, 3 and 4 in Mucosal Epithelium of Human Ulcerative Colitis. Cells 2018, 7, 61. [Google Scholar] [CrossRef]
- Ligresti, A.; Cascio, M.G.; Pryce, G.; Kulasegram, S.; Beletskaya, I.; De Petrocellis, L.; Saha, B.; Mahadevan, A.; Visintin, C.; Wiley, J.L.; et al. New potent and selective inhibitors of anandamide reuptake with antispastic activity in a mouse model of multiple sclerosis. Br. J. Pharmacol. 2006, 147, 83–91. [Google Scholar] [CrossRef]
- Naftali, T.; Mechulam, R.; Marii, A.; Gabay, G.; Stein, A.; Bronshtain, M.; Laish, I.; Benjaminov, F.; Konikoff, F.M. Low-Dose Cannabidiol Is Safe but Not Effective in the Treatment for Crohn’s Disease, a Randomized Controlled Trial. Dig. Dis. Sci. 2017, 62, 1615–1620. [Google Scholar] [CrossRef]
- Irving, P.M.; Iqbal, T.; Nwokolo, C.; Subramanian, S.; Bloom, S.; Prasad, N.; Hart, A.; Murray, C.; Lindsay, J.O.; Taylor, A.; et al. A Randomized, Double-blind, Placebo-controlled, Parallel-group, Pilot Study of Cannabidiol-rich Botanical Extract in the Symptomatic Treatment of Ulcerative Colitis. Inflamm. Bowel Dis. 2018, 24, 714–724. [Google Scholar] [CrossRef] [PubMed]
- Kafil, T.S.; Nguyen, T.M.; MacDonald, J.K.; Chande, N. Cannabis for the treatment of Crohn’s disease. Cochrane Database Syst. Rev. 2018. [Google Scholar] [CrossRef] [PubMed]
- Kafil, T.S.; Nguyen, T.M.; MacDonald, J.K.; Chande, N. Cannabis for the treatment of ulcerative colitis. Cochrane Database Syst. Rev. 2018. [Google Scholar] [CrossRef] [PubMed]
- Dekker, E.; Tanis, P.J.; Vleugels, J.L.A.; Kasi, P.M.; Wallace, M.B. Colorectal cancer. Lancet 2019, 394, 1467–1480. [Google Scholar] [CrossRef]
- Rawla, P.; Sunkara, T.; Barsouk, A. Epidemiology of colorectal cancer: Incidence, mortality, survival, and risk factors. Prz. Gastroenterol. 2019, 14, 89–103. [Google Scholar] [CrossRef] [PubMed]
- Bar-Lev Schleider, L.; Mechoulam, R.; Lederman, V.; Hilou, M.; Lencovsky, O.; Betzalel, O.; Shbiro, L.; Novack, V. Prospective analysis of safety and efficacy of medical cannabis in large unselected population of patients with cancer. Eur. J. Intern. Med. 2018, 49, 37–43. [Google Scholar] [CrossRef]
- Kleckner, A.S.; Kleckner, I.R.; Kamen, C.S.; Tejani, M.A.; Janelsins, M.C.; Morrow, G.R.; Peppone, L.J. Opportunities for cannabis in supportive care in cancer. Ther. Adv. Med. Oncol. 2019, 11, 175883591986636. [Google Scholar] [CrossRef]
- Venerito, M.; Link, A.; Rokkas, T.; Malfertheiner, P. Review: Gastric cancer—Clinical aspects. Helicobacter 2019, 24, 1–5. [Google Scholar] [CrossRef]
- Wang, D.; Wang, H.; Ning, W.; Backlund, M.G.; Dey, S.K.; DuBois, R.N. Loss of Cannabinoid Receptor 1 Accelerates Intestinal Tumor Growth. Cancer Res. 2008, 68, 6468–6476. [Google Scholar] [CrossRef]
- Cianchi, F.; Papucci, L.; Schiavone, N.; Lulli, M.; Magnelli, L.; Vinci, M.C.; Messerini, L.; Manera, C.; Ronconi, E.; Romagnani, P.; et al. Cannabinoid receptor activation induces apoptosis through tumor necrosis factor α-mediated ceramide de novo synthesis in colon cancer cells. Clin. Cancer Res. 2008, 14, 7691–7700. [Google Scholar] [CrossRef]
- Velasco, G.; Sánchez, C.; Guzmán, M. Towards the use of cannabinoids as antitumour agents. Nat. Rev. Cancer 2012, 12, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Dariš, B.; Verboten, M.T.; Knez, Ž.; Ferk, P. Cannabinoids in cancer treatment: Therapeutic potential and legislation. Bosn. J. Basic Med. Sci. 2019, 19, 14–23. [Google Scholar] [CrossRef] [PubMed]
- McAllister, S.D.; Soroceanu, L.; Desprez, P.-Y. The Antitumor Activity of Plant-Derived Non-Psychoactive Cannabinoids. J. Neuroimmune Pharmacol. 2015, 10, 255–267. [Google Scholar] [CrossRef] [PubMed]
- Kis, B.; Ifrim, F.C.; Buda, V.; Avram, S.; Pavel, I.Z.; Antal, D.; Paunescu, V.; Dehelean, C.A.; Ardelean, F.; Diaconeasa, Z.; et al. Cannabidiol—From plant to human body: A promising bioactive molecule with multi-target effects in cancer. Int. J. Mol. Sci. 2019, 20, 5905. [Google Scholar] [CrossRef] [PubMed]
- Sharafi, G.; He, H.; Nikfarjam, M. Potential Use of Cannabinoids for the Treatment of Pancreatic Cancer. J. Pancreat. Cancer 2019, 5, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kosgodage, U.S.; Uysal-Onganer, P.; MacLatchy, A.; Mould, R.; Nunn, A.V.; Guy, G.W.; Kraev, I.; Chatterton, N.P.; Thomas, E.L.; Inal, J.M.; et al. Cannabidiol Affects Extracellular Vesicle Release, miR21 and miR126, and Reduces Prohibitin Protein in Glioblastoma Multiforme Cells. Transl. Oncol. 2019, 12, 513–522. [Google Scholar] [CrossRef]
- Scott, K.A.; Shah, S.; Dalgleish, A.G.; Liu, W.M. Enhancing the activity of cannabidiol and other cannabinoids in vitro through modifications to drug combinations and treatment schedules. Anticancer Res. 2013, 33, 4373–4380. [Google Scholar]
- Sultan, A.S.; Marie, M.A.; Sheweita, S.A. Novel mechanism of cannabidiol-induced apoptosis in breast cancer cell lines. Breast 2018, 41, 34–41. [Google Scholar] [CrossRef]
- Kargl, J.; Andersen, L.; Hasenöhrl, C.; Feuersinger, D.; Stančic, A.; Fauland, A.; Magnes, C.; El-Heliebi, A.; Lax, S.; Uranitsch, S.; et al. GPR55 promotes migration and adhesion of colon cancer cells indicating a role in metastasis. Br. J. Pharmacol. 2016, 173, 142–154. [Google Scholar] [CrossRef]
- Kargl, J.; Haybaeck, J.; Stančić, A.; Andersen, L.; Marsche, G.; Heinemann, A.; Schicho, R. O-1602, an atypical cannabinoid, inhibits tumor growth in colitis-associated colon cancer through multiple mechanisms. J. Mol. Med. 2013, 91, 449–458. [Google Scholar] [CrossRef]
- Sreevalsan, S.; Joseph, S.; Jutooru, I.; Chadalapaka, G.; Safe, S.H. Induction of apoptosis by cannabinoids in prostate and colon cancer cells is phosphatase dependent. Anticancer Res. 2011, 31, 3799–3807. [Google Scholar] [PubMed]
- Aviello, G.; Romano, B.; Borrelli, F.; Capasso, R.; Gallo, L.; Piscitelli, F.; Di Marzo, V.; Izzo, A.A. Chemopreventive effect of the non-psychotropic phytocannabinoid cannabidiol on experimental colon cancer. J. Mol. Med. 2012, 90, 925–934. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.; Kim, B.G.; Kim, D.Y.; Kim, B.R.; Kim, J.L.; Park, S.H.; Na, Y.J.; Jo, M.J.; Yun, H.K.; Jeong, Y.A.; et al. Cannabidiol overcomes oxaliplatin resistance by enhancing NOS3- and SOD2-Induced autophagy in human colorectal cancer cells. Cancers (Basel) 2019, 11, 781. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.; Yun, H.K.; Jeong, Y.A.; Jo, M.J.; Kang, S.H.; Kim, J.L.; Kim, D.Y.; Park, S.H.; Kim, B.R.; Na, Y.J.; et al. Cannabidiol-induced apoptosis is mediated by activation of Noxa in human colorectal cancer cells. Cancer Lett. 2019, 447, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Romano, B.; Borrelli, F.; Pagano, E.; Cascio, M.G.; Pertwee, R.G.; Izzo, A.A. Inhibition of colon carcinogenesis by a standardized Cannabis sativa extract with high content of cannabidiol. Phytomedicine 2014, 21, 631–639. [Google Scholar] [CrossRef]
- Honarmand, M.; Namazi, F.; Mohammadi, A.; Nazifi, S. Can cannabidiol inhibit angiogenesis in colon cancer? Comp. Clin. Path. 2019, 28, 165–172. [Google Scholar] [CrossRef]
- Zhang, X.; Qin, Y.; Pan, Z.; Li, M.; Liu, X.; Chen, X.; Qu, G.; Zhou, L.; Xu, M.; Zheng, Q.; et al. Cannabidiol induces cell cycle arrest and cell apoptosis in human gastric cancer SGC-7901 cells. Biomolecules 2019, 9, 1–17. [Google Scholar] [CrossRef]
- Jeong, S.; Jo, M.J.; Yun, H.K.; Kim, D.Y.; Kim, B.R.; Kim, J.L.; Park, S.H.; Na, Y.J.; Jeong, Y.A.; Kim, B.G.; et al. Cannabidiol promotes apoptosis via regulation of XIAP/Smac in gastric cancer. Cell Death Dis. 2019, 10. [Google Scholar] [CrossRef]
- Nallathambi, R.; Mazuz, M.; Namdar, D.; Shik, M.; Namintzer, D.; Vinayaka, A.C.; Ion, A.; Faigenboim, A.; Nasser, A.; Laish, I.; et al. Identification of synergistic interaction between cannabis-derived compounds for cytotoxic activity in colorectal cancer cell lines and colon polyps that induces apoptosis-related cell death and distinct gene expression. Cannabis Cannabinoid Res. 2018, 3, 120–135. [Google Scholar] [CrossRef]
- Hijiya, N.; Shibata, T.; Daa, T.; Hamanaka, R.; Uchida, T.; Matsuura, K.; Tsukamoto, Y.; Nakada, C.; Iha, H.; Inomata, M.; et al. Overexpression of cannabinoid receptor 1 in esophageal squamous cell carcinoma is correlated with metastasis to lymph nodes and distant organs, and poor prognosis. Pathol. Int. 2017, 67, 83–90. [Google Scholar] [CrossRef]
- Duran, M.; Pérez, E.; Abanades, S.; Vidal, X.; Saura, C.; Majem, M.; Arriola, E.; Rabanal, M.; Pastor, A.; Farré, M.; et al. Preliminary efficacy and safety of an oromucosal standardized cannabis extract in chemotherapy-induced nausea and vomiting. Br. J. Clin. Pharmacol. 2010, 70, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Mersiades, A.J.; Tognela, A.; Haber, P.S.; Stockler, M.; Lintzeris, N.; Simes, J.; McGregor, I.; Olver, I.; Allsop, D.J.; Gedye, C.; et al. Oral cannabinoid-rich THC/CBD cannabis extract for secondary prevention of chemotherapy-induced nausea and vomiting: A study protocol for a pilot and definitive randomised double-blind placebo-controlled trial (CannabisCINV). BMJ Open 2018, 8, e020745. [Google Scholar] [CrossRef] [PubMed]
- Parker, L.A.; Rock, E.M.; Limebeer, C.L. Regulation of nausea and vomiting by cannabinoids. Br. J. Pharmacol. 2011, 163, 1411–1422. [Google Scholar] [CrossRef] [PubMed]
- Sharkey, K.A.; Darmani, N.A.; Parker, L.A. Regulation of nausea and vomiting by cannabinoids and the endocannabinoid system. Eur. J. Pharmacol. 2014, 722, 134–146. [Google Scholar] [CrossRef] [PubMed]
- Rock, E.M.; Goodwin, J.M.; Limebeer, C.L.; Breuer, A.; Pertwee, R.G.; Mechoulam, R.; Parker, L.A. Interaction between non-psychotropic cannabinoids in marihuana: Effect of cannabigerol (CBG) on the anti-nausea or anti-emetic effects of cannabidiol (CBD) in rats and shrews. Psychopharmacology (Berl.) 2011, 215, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Galli, J.A.; Sawaya, R.A.; Friedenberg, F.K. Cannabinoid hyperemesis syndrome. Curr. Drug Abuse Rev. 2011, 4, 241–249. [Google Scholar] [CrossRef]
- Ruffle, J.K.; Bajgoric, S.; Samra, K.; Chandrapalan, S.; Aziz, Q.; Farmer, A.D. Cannabinoid hyperemesis syndrome: An important differential diagnosis of persistent unexplained vomiting. Eur. J. Gastroenterol. Hepatol. 2015, 27, 1403–1408. [Google Scholar] [CrossRef]
- Hasler, W.L.; Levinthal, D.J.; Tarbell, S.E.; Adams, K.A.; Li, B.U.K.; Issenman, R.M.; Sarosiek, I.; Jaradeh, S.S.; Sharaf, R.N.; Sultan, S.; et al. Cyclic vomiting syndrome: Pathophysiology, comorbidities, and future research directions. Neurogastroenterol. Motil. 2019, 31, 1–10. [Google Scholar] [CrossRef]
- Abalo, R.; Cabezos, P.A.; Vera, G.; López-miranda, V.; Herradón, E.; Martín-fontelles, M.I. Cannabinoid-induced delayed gastric emptying is selectively increased upon intermittent administration in the rat: Role of CB1 receptors. Neurogastroenterol. Motil. 2011, 23, 457–468. [Google Scholar] [CrossRef]
- Abalo, R.; Cabezos, P.A.; López-Miranda, V.; Vera, G.; González, C.; Castillo, M.; Fernández-Pujol, R.; Martín, M.I. Selective lack of tolerance to delayed gastric emptying after daily administration of WIN 55,212-2 in the rat. Neurogastroenterol. Motil. 2009, 21, 22–24. [Google Scholar] [CrossRef]
- Cuba, L.F.; Salum, F.G.; Cherubini, K.; Figueiredo, M.A.Z. Cannabidiol: An alternative therapeutic agent for oral mucositis? J. Clin. Pharm. Ther. 2017, 42, 245–250. [Google Scholar] [CrossRef]
- Klein, M.; de Quadros De Bortolli, J.; Guimarães, F.S.; Salum, F.G.; Cherubini, K.; de Figueiredo, M.A.Z. Effects of cannabidiol, a Cannabis sativa constituent, on oral wound healing process in rats: Clinical and histological evaluation. Phyther. Res. 2018, 32, 2275–2281. [Google Scholar] [CrossRef] [PubMed]
- Gotfried, J.; Kataria, R.; Schey, R. Review: The Role of Cannabinoids on Esophageal Function-What We Know Thus Far. Cannabis Cannabinoid Res. 2017, 2, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Beaumont, H.; Jensen, J.; Carlsson, A.; Ruth, M.; Lehmann, A.; Boeckxstaens, G.E. Effect of Δ 9-tetrahydrocannabinol, a cannabinoid receptor agonist, on the triggering of transient lower oesophageal sphincter relaxations in dogs and humans. Br. J. Pharmacol. 2009, 156, 153–162. [Google Scholar] [CrossRef] [PubMed]
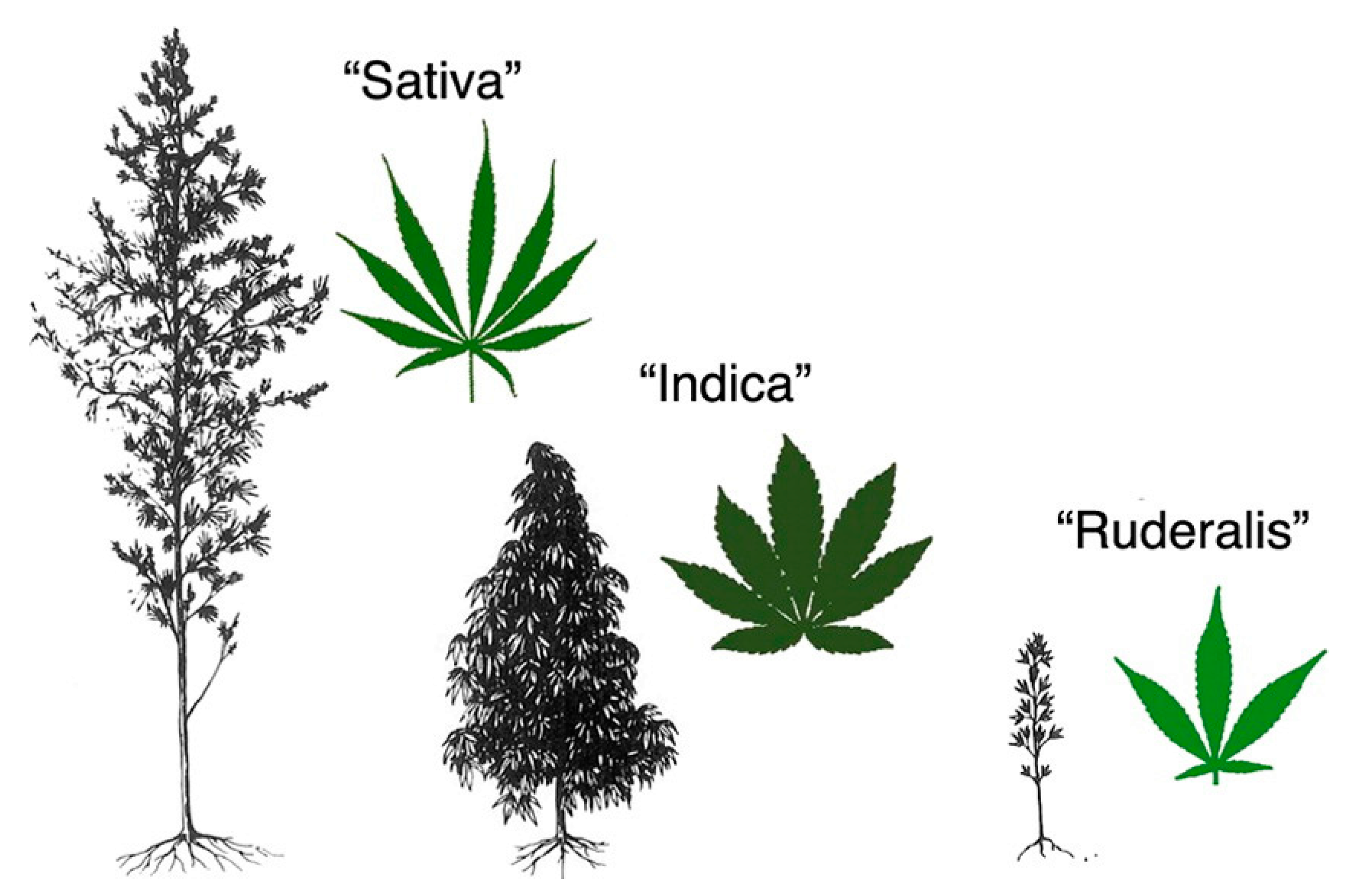
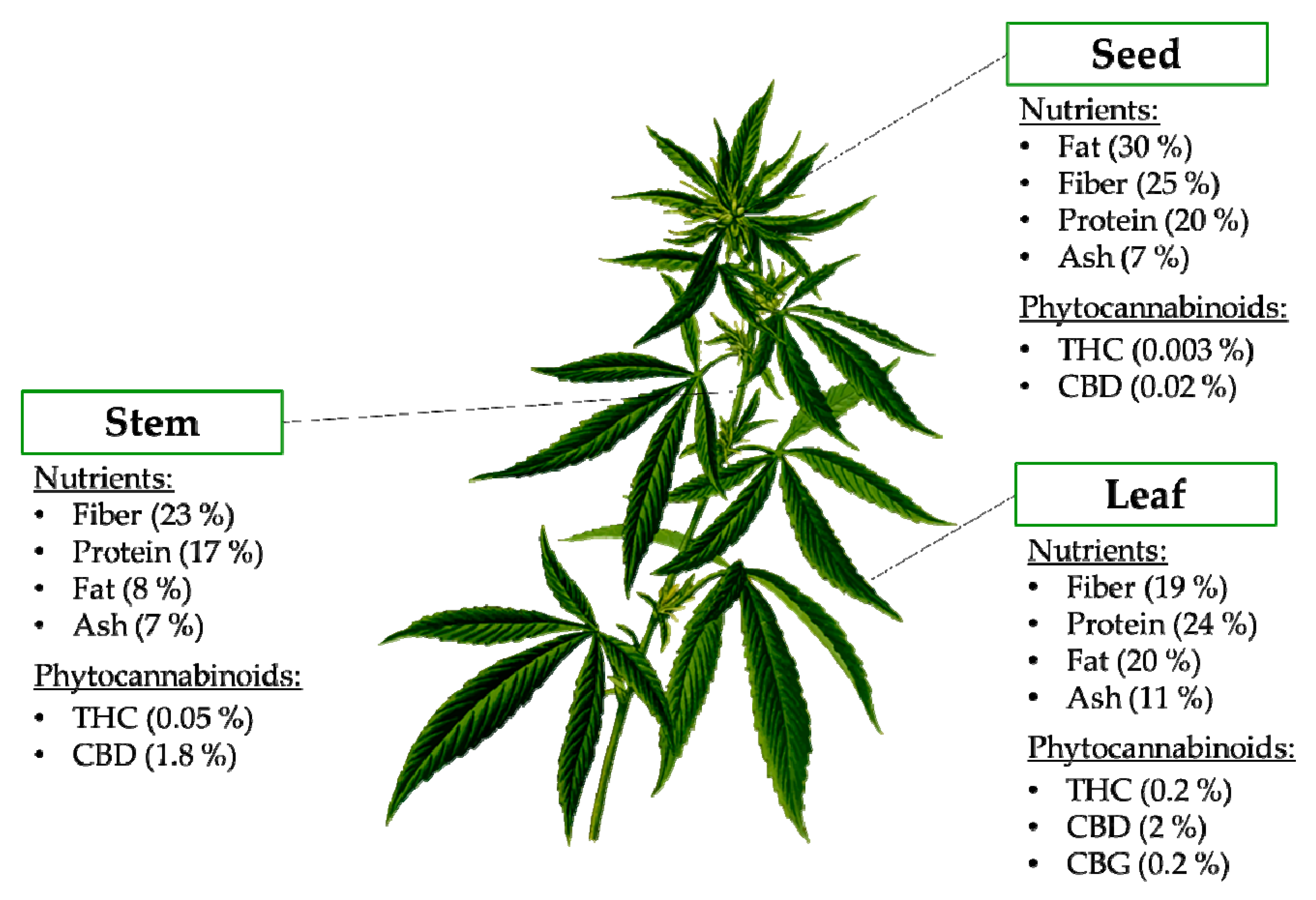
| Type of Ingredient | Type of Food | Number of Products |
|---|---|---|
| Hemp seed | Seeds | 56 |
| Chocolate | 13 | |
| Cereal | 80 | |
| Bars and snacks | 178 | |
| Bakery products | 37 | |
| Beverage | 52 | |
| Dressings, butter | 51 | |
| Prepared food | 24 | |
| Yogurt, cheese, ice-cream (vegan) | 13 | |
| Hemp protein extract | Beverage | 78 |
| Bars and snacks | 40 | |
| Powder | 24 | |
| Capsules | 1 | |
| Hemp oil | Oil | 9 |
| Beverage | 7 | |
| Hemp flour | Pasta | 2 |
| Veggie burger or sausage | 12 | |
| Bakery products | 3 |
| Δ9-TETRAHYDROCANNABINOL | ||
| Chemical structure | Molecular Targets | References |
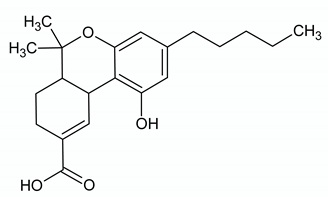 | CB1 partial agonist | [42] |
| CB2 partial agonist | [42] | |
| GPR55 agonist | [43] | |
| GPR18 agonist | [44,45] | |
| 5HT3A antagonist | [46] | |
| PPARγ agonist | [47,48] | |
| TRPA1 agonist | [49] | |
| TRPV2 agonist | [50] | |
| TRPV3 agonist | [51] | |
| TRPV4 agonist | [51] | |
| TRPM8 antagonist | [49] | |
| µ and δ opioid allosteric modulator | [52,53] | |
| GlyR α1 and α3 positive allosteric modulator | [54] | |
| AEA uptake inhibition by targeting FABPs | [55,56,57] | |
| Adenosine reuptake inhibitor | [58] | |
| CANNABIDIOL | ||
| Chemical structure | Molecular Targets | References |
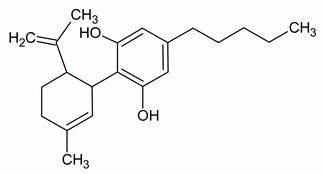 | CB1 inverse agonist and negative allosteric modulator | [59,60] |
| CB2 partial agonist and negative allosteric modulator | [59,61] | |
| GPR55 antagonist | [62] | |
| GPR18 antagonist | [44] | |
| GPR3 inverse agonist | [63] | |
| GPR6 inverse agonist | [63] | |
| GPR12 inverse agonist | [64] | |
| A1Aagonist | [65] | |
| 5HT1A agonist | [66] | |
| 5HT2A partial agonist | [66] | |
| 5HT3A antagonist | [67] | |
| PPARγ agonist | [68] | |
| TRPA1 agonist | [56] | |
| TRPV1 agonist | [55,56] | |
| TRPV2 agonist | [50] | |
| TRPV3 agonist | [51] | |
| TRPM8 antagonist | [56] | |
| GABAA positive allosteric modulator | [69] | |
| µ and δ opioid allosteric modulator | [52,53] | |
| GlyRα1 and α3 positive allosteric modulator | [70,71] | |
| AEA uptake inhibition by targeting FABPs | [55,56,57] | |
| Adenosine reuptake inhibitor | [58] | |
| CANNABIGEROL | ||
| Chemical structure | Molecular Targets | References |
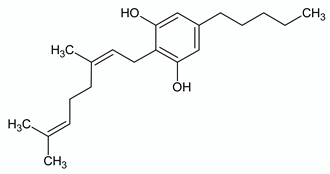 | CB1 weak partial agonist | [72] |
| CB2 weak partial agonist | [72,73] | |
| GPR55 antagonist | [74] | |
| α2-adrenoceptor agonist | [75] | |
| 5HT1A antagonist | [75] | |
| TRPA1 agonist | [49,56] | |
| TRPV1 agonist | [56] | |
| TRPV2 agonist | [56] | |
| TRPV4 antagonist | [51] | |
| TRPM8 antagonist | [49,56] | |
| Voltage-gated sodium channels Nav blocker | [76] | |
| AEA uptake inhibitor | [56] | |
| MAGL inhibitor | [56] | |
| CANNABICHROMENE | ||
| Chemical structure | Molecular Targets | References |
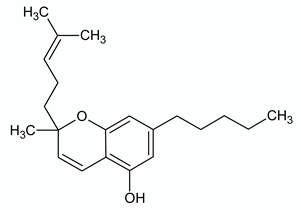 | CB2 agonist | [77] |
| TRPA1 agonist | [49,56] | |
| TRPV1 agonist | [56] | |
| TRPV3 agonist | [51] | |
| TRPV4 agonist | [51] | |
| TRPM8 weak antagonist | [56] | |
| AEA uptake inhibitor | [56] | |
| MAGL inhibitor | [56] | |
| CANNABIDIVARIN | ||
| Chemical structure | Molecular Targets | References |
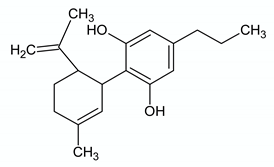 | GPR55 antagonist | [74] |
| TRPA1 agonist | [56] | |
| TRPV1 agonist | [56] | |
| TRPV2 agonist | [56] | |
| TRPV3 agonist | [51] | |
| TRPV4 agonist | [51] | |
| TRPM8 antagonist | [56] | |
| DAGL inhibitor | [56] | |
| AEA uptake inhibitor | [56] | |
| CANNABIDIOLIC ACID | ||
| Chemical structure | Molecular Targets | References |
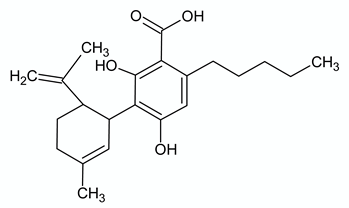 | GPR55 antagonist | [74] |
| 5HT1A agonist (potentiation) | [78] | |
| TRPA1 agonist | [49,56] | |
| TRPV1 weak agonist | [56] | |
| TRPM8 antagonist | [56] | |
| NAAA inhibitor | [56] | |
| DAGL inhibitor | [56] | |
| AEA uptake inhibitor | [56] | |
| CANNABIGEROLIC ACID | ||
| Chemical structure | Molecular Targets | References |
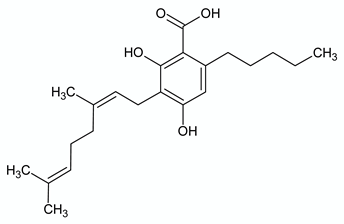 | GPR55 inhibition | [74] |
| TRPA1 weak agonist | [56] | |
| TRPV3 antagonist | [51] | |
| TRPV4 antagonist | [51] | |
| TRPM8 antagonist | [56] | |
| AEA uptake inhibitor | [56] | |
| DAGL inhibitor | [56] | |
| RECEPTORS | ||
|---|---|---|
| COMPONENT | LOCATION | FUNCTION |
| CB1 (binds AEA, 2-AG) | ENS: cholinergic neurons Mucosa: epithelial and plasma cells Vascular smooth muscle cells Lamina propria: macrophages and plasma cells | Reduces GI motility and secretion Modulates immune function |
| CB2 (binds AEA, 2-AG) | ENS (under inflammatory conditions) Mucosa: epithelial cells, macrophages > plasma cells Lamina propria: macrophages and plasma cells | Reduces GI motility and secretion Modulates immune function |
| TRPV1 (binds AEA > OEA) | Extrinsic afferent fibers, running through the muscle layers Immune cells adjacent to blood vessels. | Visceral hypersensitivity signaling Increase in intestinal contractility (under inflammatory conditions) |
| PPAR-α (binds AEA, 2-AG, OEA, PEA, others) | Enterocytes of the small intestine ENS Vagal afferent fibers Enteric glial cells | |
| GPR55 (binds PEA) | Epithelial cells and ENS of the small intestine | |
| GPR119 (binds OEA, PEA > AEA) | Villi: enteroendocrine L cells | Regulates the release of GLP-1 |
| METABOLIC ENZYMES | ||
| COMPONENT | LOCATION | FUNCTION |
| FAAH | Cells of the myenteric plexus in stomach and intestine | Degrades AEA, PEA, OEA |
| MAGL | Nerve cells and fibers in the muscle layers and mucosa of duodenum, ileum and colon | Degrades 2-AG Activity decreases from proximal to distal locations |
| LIGANDS | ||
| COMPONENT | LOCATION | FUNCTION |
| AEA | Higher levels in colon than in ileum | Increased levels in IBD, celiac disease, diverticulitis, CRC |
| 2-AG | Higher levels in ileum than in colon | Increased levels in CRC |
| Compound | Model | Species (Strain, Sex) | Type of Treatment | Main Effects on Inflammation-Related Parameters | Reference |
|---|---|---|---|---|---|
| CBD | LPS | Mouse (Swiss OF1, males) | Preventive | ↓ Enteric glial activation ↓ Mast cell activation ↓ macrophages activation ↓ TNF-α ↓ cleaved caspase-3 (↓ apoptosis) | [163] |
| CBD | LPS | Rat (Sprague Dawley, N.S.) | Preventive | ↓ Histopathological alterations ↓ TNF-α ↓ IL-6 Restoration of smooth muscle myoelectrical activity | [132] |
| CBD | LPS | Mouse (C57/BL, N.S.) | Preventive | ↓ IL-6 Restoration of UGT | [132] |
| CBD | TNBS | Mouse (CD1, males) | Preventive | ↓ Histopathological alterations (i.p. treatment) No effects on histopathological alterations (oral treatment) ↓ Histopathological alterations (intrarectal treatment) | [164] |
| CBD | TNBS | Rat (Wistar, males) | Preventive | No effect on indices of inflammation ↓ MPO activity | [165] |
| CBD + THC | TNBS | Rat (Wistar, males) | Preventive | ↓ Indices of inflammation ↓ MPO activity | [165] |
| CBD | DNBS | Mouse (ICR, males) | Preventive | ↓ Indices of inflammation ↓ Histopathological alterations ↓ iNOS expression Normalization of IL-1β and IL-10 levels | [166] |
| CBD | DNBS | Mouse (ICR, males) | Curative | No effects on indices of inflammation | [131] |
| CBD (enriched botanical extract) | DNBS | Mouse (ICR, males) | Curative | ↓ Indices of inflammation ↓ MPO activity | [131] |
| CBD (enriched botanical extract) | Croton oil | Mouse (ICR, males) | Curative | ↓ Hypermotility (UGT) | [131] |
| CBD | Croton oil | Mouse (ICR, males) | Curative | ↓ Hypermotility (UGT) | [131] |
| CBD | Croton oil | Mouse (ICR, males) | Curative | Normalization of hypermotility (UGT) | [127] |
| CBD | Croton oil | Mouse (ICR, males) | Curative | ↓ Hypermotility (UGT) | [130] |
| CBG | DNBS | Mouse (ICR, males) | Preventive | ↓ Indices of colitis | [167] |
| CBG | DNBS | Mouse (ICR, males) | Curative | ↓ Indices of colitis ↓ Histopathological alterations Restoration of epithelial barrier function Normalization of IL-1β, IL-10 and IFN-γ levels Normalization of MPO activity Normalization of SOD activity | [167] |
| CBC | Croton oil | Mouse (ICR, males) | Curative | Normalization of UGT | [126] |
| CBC | DNBS | Mouse (ICR, males) | Curative | ↓ Indices of colitis ↓ Histopathological alterations Amelioration of epithelial barrier function ↓ MPO activity | [168] |
| CBC | DNBS | Mouse (ICR, males) | Preventive | ↓ Indices of colitis | [168] |
| CBDV | DNBS | Mouse (CD1, males) | Preventive | ↓ Indices of colitis ↓ MPO activity Amelioration of epithelial barrier function ↓ Histopathological alterations | [128] |
| CBDV | DNBS | Mouse (CD1, males) | Curative | ↓ Indices of colitis ↓ MPO activity Amelioration of epithelial barrier function ↓ Histopathological alterations Normalization of IL-1β, IL-6, and MCP-1α expression ↓ Inflammation-associated dysbiosis | [128] |
| CBDV | DSS | Mouse (CD1, males) | Curative | ↓ Indices of colitis ↓ MPO activity ↓ IL-1β levels | [128] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez, V.; Iriondo De-Hond, A.; Borrelli, F.; Capasso, R.; del Castillo, M.D.; Abalo, R. Cannabidiol and Other Non-Psychoactive Cannabinoids for Prevention and Treatment of Gastrointestinal Disorders: Useful Nutraceuticals? Int. J. Mol. Sci. 2020, 21, 3067. https://doi.org/10.3390/ijms21093067
Martínez V, Iriondo De-Hond A, Borrelli F, Capasso R, del Castillo MD, Abalo R. Cannabidiol and Other Non-Psychoactive Cannabinoids for Prevention and Treatment of Gastrointestinal Disorders: Useful Nutraceuticals? International Journal of Molecular Sciences. 2020; 21(9):3067. https://doi.org/10.3390/ijms21093067
Chicago/Turabian StyleMartínez, Vicente, Amaia Iriondo De-Hond, Francesca Borrelli, Raffaele Capasso, María Dolores del Castillo, and Raquel Abalo. 2020. "Cannabidiol and Other Non-Psychoactive Cannabinoids for Prevention and Treatment of Gastrointestinal Disorders: Useful Nutraceuticals?" International Journal of Molecular Sciences 21, no. 9: 3067. https://doi.org/10.3390/ijms21093067
APA StyleMartínez, V., Iriondo De-Hond, A., Borrelli, F., Capasso, R., del Castillo, M. D., & Abalo, R. (2020). Cannabidiol and Other Non-Psychoactive Cannabinoids for Prevention and Treatment of Gastrointestinal Disorders: Useful Nutraceuticals? International Journal of Molecular Sciences, 21(9), 3067. https://doi.org/10.3390/ijms21093067










