Thermogenesis in Adipose Tissue Activated by Thyroid Hormone
Abstract
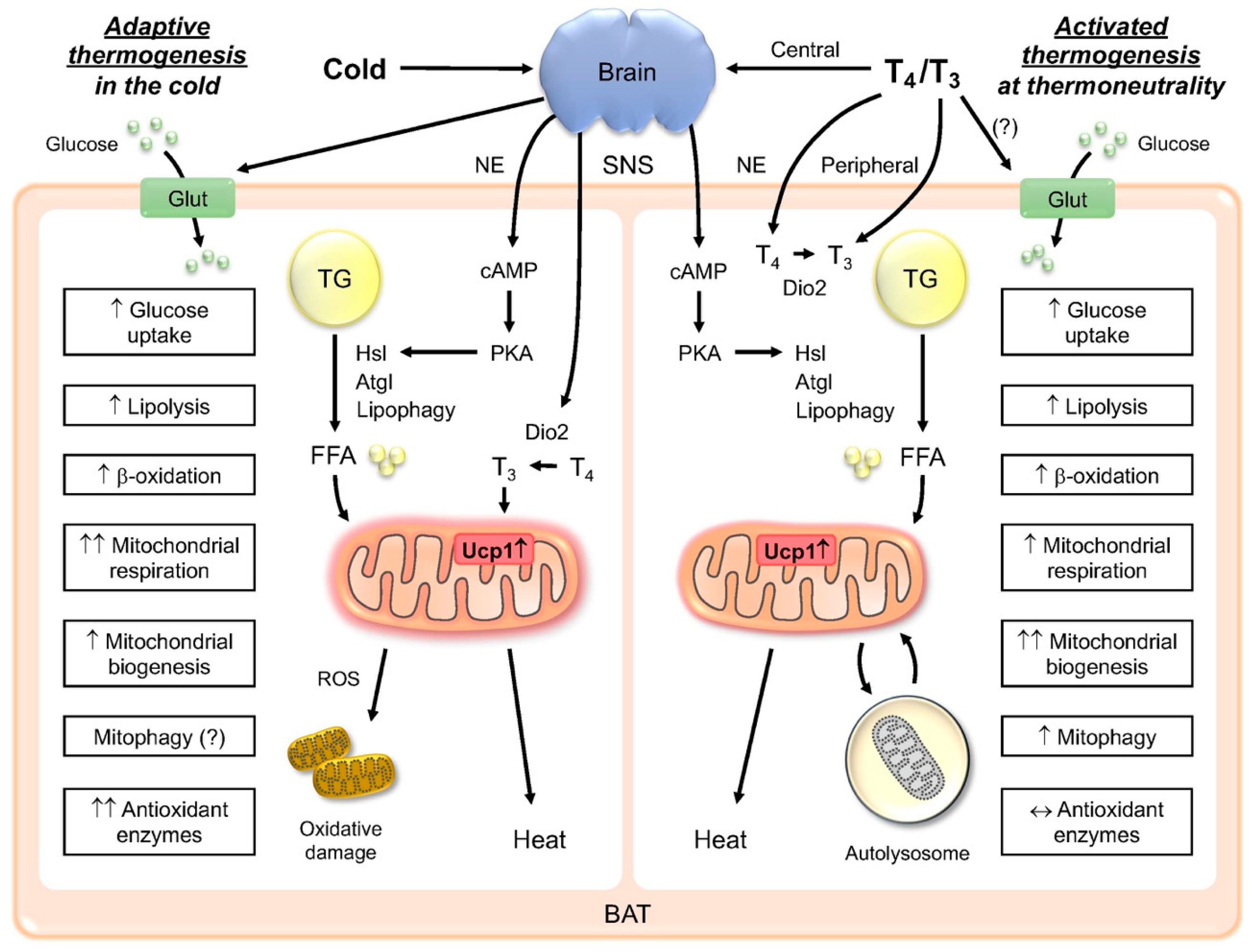
1. Introduction
1.1. Thermogenesis and Adipose Tissue
1.2. TH induction of Thermogenesis in BAT
1.3. Central and Peripheral Regulation of Thermogenesis
2. Metabolic Actions of TH in BAT
2.1. TH Increases Glucose Uptake
2.2. TH Increases Fatty Acid β-Oxidation
2.3. TH Increases Lipogenesis
3. TH effects on Mitochondria in BAT
3.1. TH Increases Mitochondrial Biogenesis
3.2. TH Increases Mitophagy
4. TH Induces Browning/Beiging in WAT
5. Role of TH Metabolites in Thermogenesis
6. Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Atg5 | Autophagy-related protein 5 |
| BAT | Brown adipose tissue |
| cAMP | Cyclic adenosine monophosphate |
| DAG | Diacylglycerol |
| Dgat1 | Diacylglycerol O-acyltransferase 1 |
| Dio2 | 5-Deiodinase type 2 |
| FADH2 | Flavin adenine dinucleotide |
| 18F-FDG | 18F-Fluorodeoxyglucose |
| FFA | Free fatty acids |
| Gpat4 | Gycerol-3-phosphate acyltransferase 4 |
| Glut | Glucose transporter |
| KO | Knockout |
| mTOR | Mammalian target of rapamycin |
| NADH | Nicotinamide adenine dinucleotide (NADH) |
| OXPHOS | Oxidative phosphorylation |
| PET | Positron emission tomography |
| PKA | Protein kinase A |
| TH | Thyroid hormone |
| TR | Thyroid hormone receptor |
| TG | Triacylglycerol |
| Ucp1 | Uncoupling protein 1 |
| SNS | Sympathetic nervous system |
| WAT | White adipose tissue |
References
- Silva, J.E. The thermogenic effect of thyroid hormone and its clinical implications. Ann. Intern. Med. 2003, 139, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Eyolfson, D.A.; Tikuisis, P.; Xu, X.; Weseen, G.; Giesbrecht, G.G. Measurement and prediction of peak shivering intensity in humans. Eur. J. Appl. Physiol. 2001, 84, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Periasamy, M.; Herrera, J.L.; Reis, F.C.G. Skeletal Muscle Thermogenesis and Its Role in Whole Body Energy Metabolism. Diabetes Metab. J. 2017, 41, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Celi, F.S.; Le, T.N.; Ni, B. Physiology and relevance of human adaptive thermogenesis response. Trends Endocrinol. Metab. 2015, 26, 238–247. [Google Scholar] [CrossRef] [PubMed]
- Cannon, B.; Nedergaard, J. Metabolic consequences of the presence or absence of the thermogenic capacity of brown adipose tissue in mice (and probably in humans). Int. J. Obes (Lond.) 2010, 34, S7–S16. [Google Scholar] [CrossRef]
- Cannon, B.; Nedergaard, J. Brown adipose tissue: Function and physiological significance. Physiol. Rev. 2004, 84, 277–359. [Google Scholar] [CrossRef]
- van der Lans, A.A.; Hoeks, J.; Brans, B.; Vijgen, G.H.; Visser, M.G.; Vosselman, M.J.; Hansen, J.; Jorgensen, J.A.; Wu, J.; Mottaghy, F.M.; et al. Cold acclimation recruits human brown fat and increases nonshivering thermogenesis. J. Clin. Investig. 2013, 123, 3395–3403. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, M.; Xu, M.; Gu, W.; Xi, Y.; Qi, L.; Li, B.; Wang, W. Brown adipose tissue activation is inversely related to central obesity and metabolic parameters in adult human. PLoS ONE 2015, 10, e0123795. [Google Scholar] [CrossRef]
- Yilmaz, Y.; Ones, T.; Purnak, T.; Ozguven, S.; Kurt, R.; Atug, O.; Turoglu, H.T.; Imeryuz, N. Association between the presence of brown adipose tissue and non-alcoholic fatty liver disease in adult humans. Aliment. Pharmacol. Ther. 2011, 34, 318–323. [Google Scholar] [CrossRef]
- Wu, J.; Bostrom, P.; Sparks, L.M.; Ye, L.; Choi, J.H.; Giang, A.H.; Khandekar, M.; Virtanen, K.A.; Nuutila, P.; Schaart, G.; et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 2012, 150, 366–376. [Google Scholar] [CrossRef]
- Stock, M.J. Thermogenesis and brown fat: Relevance to human obesity. Infusionstherapie 1989, 16, 282–284. [Google Scholar] [CrossRef] [PubMed]
- Townsend, K.L.; Tseng, Y.H. Brown fat fuel utilization and thermogenesis. Trends Endocrinol. Metab. 2014, 25, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Blondin, D.P.; Labbe, S.M.; Tingelstad, H.C.; Noll, C.; Kunach, M.; Phoenix, S.; Guerin, B.; Turcotte, E.E.; Carpentier, A.C.; Richard, D.; et al. Increased brown adipose tissue oxidative capacity in cold-acclimated humans. J. Clin. Endocrinol. Metab. 2014, 99, E438–E446. [Google Scholar] [CrossRef]
- Blondin, D.P.; Daoud, A.; Taylor, T.; Tingelstad, H.C.; Bezaire, V.; Richard, D.; Carpentier, A.C.; Taylor, A.W.; Harper, M.E.; Aguer, C.; et al. Four-week cold acclimation in adult humans shifts uncoupling thermogenesis from skeletal muscles to brown adipose tissue. J. Physiol. 2017, 595, 2099–2113. [Google Scholar] [CrossRef]
- Kajimura, S.; Spiegelman, B.M.; Seale, P. Brown and Beige Fat: Physiological Roles beyond Heat Generation. Cell Metab. 2015, 22, 546–559. [Google Scholar] [CrossRef]
- Lizcano, F. The Beige Adipocyte as a Therapy for Metabolic Diseases. Int. J. Mol. Sci. 2019, 20, 5058. [Google Scholar] [CrossRef]
- Hanssen, M.J.; Hoeks, J.; Brans, B.; van der Lans, A.A.; Schaart, G.; van den Driessche, J.J.; Jorgensen, J.A.; Boekschoten, M.V.; Hesselink, M.K.; Havekes, B.; et al. Short-term cold acclimation improves insulin sensitivity in patients with type 2 diabetes mellitus. Nat. Med. 2015, 21, 863–865. [Google Scholar] [CrossRef]
- Hanssen, M.J.; van der Lans, A.A.; Brans, B.; Hoeks, J.; Jardon, K.M.; Schaart, G.; Mottaghy, F.M.; Schrauwen, P.; van Marken Lichtenbelt, W.D. Short-term Cold Acclimation Recruits Brown Adipose Tissue in Obese Humans. Diabetes 2016, 65, 1179–1189. [Google Scholar] [CrossRef]
- Zoico, E.; Rubele, S.; De Caro, A.; Nori, N.; Mazzali, G.; Fantin, F.; Rossi, A.; Zamboni, M. Brown and Beige Adipose Tissue and Aging. Front. Endocrinol. (Lausanne) 2019, 10, 368. [Google Scholar] [CrossRef]
- Yoneshiro, T.; Aita, S.; Matsushita, M.; Okamatsu-Ogura, Y.; Kameya, T.; Kawai, Y.; Miyagawa, M.; Tsujisaki, M.; Saito, M. Age-related decrease in cold-activated brown adipose tissue and accumulation of body fat in healthy humans. Obesity (Silver Spring) 2011, 19, 1755–1760. [Google Scholar] [CrossRef]
- Matsushita, M.; Yoneshiro, T.; Aita, S.; Kameya, T.; Sugie, H.; Saito, M. Impact of brown adipose tissue on body fatness and glucose metabolism in healthy humans. Int. J. Obes. (Lond.) 2014, 38, 812–817. [Google Scholar] [CrossRef] [PubMed]
- Yau, W.W.; Singh, B.K.; Lesmana, R.; Zhou, J.; Sinha, R.A.; Wong, K.A.; Wu, Y.; Bay, B.H.; Sugii, S.; Sun, L.; et al. Thyroid hormone (T3) stimulates brown adipose tissue activation via mitochondrial biogenesis and MTOR-mediated mitophagy. Autophagy 2019, 15, 131–150. [Google Scholar] [CrossRef] [PubMed]
- Lopez, M.; Varela, L.; Vazquez, M.J.; Rodriguez-Cuenca, S.; Gonzalez, C.R.; Velagapudi, V.R.; Morgan, D.A.; Schoenmakers, E.; Agassandian, K.; Lage, R.; et al. Hypothalamic AMPK and fatty acid metabolism mediate thyroid regulation of energy balance. Nat. Med. 2010, 16, 1001–1008. [Google Scholar] [CrossRef]
- Bianco, A.C.; Maia, A.L.; da Silva, W.S.; Christoffolete, M.A. Adaptive activation of thyroid hormone and energy expenditure. Biosci. Rep. 2005, 25, 191–208. [Google Scholar] [CrossRef]
- Obregon, M.J. Adipose tissues and thyroid hormones. Front. Physiol. 2014, 5, 479. [Google Scholar] [CrossRef]
- Sellers, E.A.; You, S.S. Role of the thyroid in metabolic responses to a cold environment. Am. J. Physiol. 1950, 163, 81–91. [Google Scholar] [CrossRef]
- Silva, J.E.; Larsen, P.R. Adrenergic activation of triiodothyronine production in brown adipose tissue. Nature 1983, 305, 712–713. [Google Scholar] [CrossRef]
- Fernandez, J.A.; Mampel, T.; Villarroya, F.; Iglesias, R. Direct assessment of brown adipose tissue as a site of systemic tri-iodothyronine production in the rat. Biochem. J. 1987, 243, 281–284. [Google Scholar] [CrossRef]
- Christoffolete, M.A.; Linardi, C.C.; de Jesus, L.; Ebina, K.N.; Carvalho, S.D.; Ribeiro, M.O.; Rabelo, R.; Curcio, C.; Martins, L.; Kimura, E.T.; et al. Mice with targeted disruption of the Dio2 gene have cold-induced overexpression of the uncoupling protein 1 gene but fail to increase brown adipose tissue lipogenesis and adaptive thermogenesis. Diabetes 2004, 53, 577–584. [Google Scholar] [CrossRef]
- de Jesus, L.A.; Carvalho, S.D.; Ribeiro, M.O.; Schneider, M.; Kim, S.W.; Harney, J.W.; Larsen, P.R.; Bianco, A.C. The type 2 iodothyronine deiodinase is essential for adaptive thermogenesis in brown adipose tissue. J. Clin. Investig. 2001, 108, 1379–1385. [Google Scholar] [CrossRef]
- Wikstrom, L.; Johansson, C.; Salto, C.; Barlow, C.; Campos Barros, A.; Baas, F.; Forrest, D.; Thoren, P.; Vennstrom, B. Abnormal heart rate and body temperature in mice lacking thyroid hormone receptor alpha 1. EMBO J. 1998, 17, 455–461. [Google Scholar] [CrossRef]
- Liu, Y.Y.; Schultz, J.J.; Brent, G.A. A thyroid hormone receptor alpha gene mutation (P398H) is associated with visceral adiposity and impaired catecholamine-stimulated lipolysis in mice. J. Biol. Chem. 2003, 278, 38913–38920. [Google Scholar] [CrossRef]
- Ribeiro, M.O.; Carvalho, S.D.; Schultz, J.J.; Chiellini, G.; Scanlan, T.S.; Bianco, A.C.; Brent, G.A. Thyroid hormone—Sympathetic interaction and adaptive thermogenesis are thyroid hormone receptor isoform—Specific. J. Clin. Investig. 2001, 108, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, M.O.; Bianco, S.D.; Kaneshige, M.; Schultz, J.J.; Cheng, S.Y.; Bianco, A.C.; Brent, G.A. Expression of uncoupling protein 1 in mouse brown adipose tissue is thyroid hormone receptor-beta isoform specific and required for adaptive thermogenesis. Endocrinology 2010, 151, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Johansson, C.; Gothe, S.; Forrest, D.; Vennstrom, B.; Thoren, P. Cardiovascular phenotype and temperature control in mice lacking thyroid hormone receptor-beta or both alpha1 and beta. Am. J. Physiol. 1999, 276, H2006–H2012. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Crespo, M.; Csikasz, R.I.; Martinez-Sanchez, N.; Dieguez, C.; Cannon, B.; Nedergaard, J.; Lopez, M. Essential role of UCP1 modulating the central effects of thyroid hormones on energy balance. Mol. Metab. 2016, 5, 271–282. [Google Scholar] [CrossRef]
- Martinez-Sanchez, N.; Moreno-Navarrete, J.M.; Contreras, C.; Rial-Pensado, E.; Ferno, J.; Nogueiras, R.; Dieguez, C.; Fernandez-Real, J.M.; Lopez, M. Thyroid hormones induce browning of white fat. J. Endocrinol. 2017, 232, 351–362. [Google Scholar] [CrossRef]
- Mohacsik, P.; Erdelyi, F.; Baranyi, M.; Botz, B.; Szabo, G.; Toth, M.; Haltrich, I.; Helyes, Z.; Sperlagh, B.; Toth, Z.; et al. A Transgenic Mouse Model for Detection of Tissue-Specific Thyroid Hormone Action. Endocrinology 2018, 159, 1159–1171. [Google Scholar] [CrossRef]
- Dittner, C.; Lindsund, E.; Cannon, B.; Nedergaard, J. At thermoneutrality, acute thyroxine-induced thermogenesis and pyrexia are independent of UCP1. Mol. Metab. 2019, 25, 20–34. [Google Scholar] [CrossRef]
- Warner, A.; Rahman, A.; Solsjo, P.; Gottschling, K.; Davis, B.; Vennstrom, B.; Arner, A.; Mittag, J. Inappropriate heat dissipation ignites brown fat thermogenesis in mice with a mutant thyroid hormone receptor alpha1. Proc. Natl. Acad. Sci. USA 2013, 110, 16241–16246. [Google Scholar] [CrossRef]
- Vaitkus, J.A.; Farrar, J.S.; Celi, F.S. Thyroid Hormone Mediated Modulation of Energy Expenditure. Int. J. Mol. Sci. 2015, 16, 16158–16175. [Google Scholar] [CrossRef]
- Silva, J.E. Thermogenic mechanisms and their hormonal regulation. Physiol. Rev. 2006, 86, 435–464. [Google Scholar] [CrossRef] [PubMed]
- Kazak, L.; Rahbani, J.F.; Samborska, B.; Lu, G.Z.; Jedrychowski, M.P.; Lajoie, M.; Zhang, S.; Ramsay, L.C.; Dou, F.Y.; Tenen, D.; et al. Ablation of adipocyte creatine transport impairs thermogenesis and causes diet-induced obesity. Nat. Metab. 2019, 1, 360–370. [Google Scholar] [CrossRef] [PubMed]
- Kazak, L.; Chouchani, E.T.; Jedrychowski, M.P.; Erickson, B.K.; Shinoda, K.; Cohen, P.; Vetrivelan, R.; Lu, G.Z.; Laznik-Bogoslavski, D.; Hasenfuss, S.C.; et al. A creatine-driven substrate cycle enhances energy expenditure and thermogenesis in beige fat. Cell 2015, 163, 643–655. [Google Scholar] [CrossRef] [PubMed]
- Grimpo, K.; Volker, M.N.; Heppe, E.N.; Braun, S.; Heverhagen, J.T.; Heldmaier, G. Brown adipose tissue dynamics in wild-type and UCP1-knockout mice: In vivo insights with magnetic resonance. J. Lipid Res. 2014, 55, 398–409. [Google Scholar] [CrossRef]
- Keipert, S.; Kutschke, M.; Ost, M.; Schwarzmayr, T.; van Schothorst, E.M.; Lamp, D.; Brachthauser, L.; Hamp, I.; Mazibuko, S.E.; Hartwig, S.; et al. Long-Term Cold Adaptation Does Not Require FGF21 or UCP1. Cell Metab. 2017, 26, 437–446. [Google Scholar] [CrossRef]
- Greco-Perotto, R.; Zaninetti, D.; Assimacopoulos-Jeannet, F.; Bobbioni, E.; Jeanrenaud, B. Stimulatory effect of cold adaptation on glucose utilization by brown adipose tissue. Relationship with changes in the glucose transporter system. J. Biol. Chem. 1987, 262, 7732–7736. [Google Scholar]
- Vallerand, A.L.; Perusse, F.; Bukowiecki, L.J. Stimulatory effects of cold exposure and cold acclimation on glucose uptake in rat peripheral tissues. Am. J. Physiol. 1990, 259, R1043–R1049. [Google Scholar] [CrossRef]
- Shimizu, Y.; Nikami, H.; Saito, M. Sympathetic activation of glucose utilization in brown adipose tissue in rats. J. Biochem. 1991, 110, 688–692. [Google Scholar] [CrossRef]
- Liu, X.; Perusse, F.; Bukowiecki, L.J. Chronic norepinephrine infusion stimulates glucose uptake in white and brown adipose tissues. Am. J. Physiol. 1994, 266, R914–R920. [Google Scholar] [CrossRef]
- Shimizu, Y.; Nikami, H.; Tsukazaki, K.; Machado, U.F.; Yano, H.; Seino, Y.; Saito, M. Increased expression of glucose transporter GLUT-4 in brown adipose tissue of fasted rats after cold exposure. Am. J. Physiol. 1993, 264, E890–E895. [Google Scholar] [CrossRef]
- Dallner, O.S.; Chernogubova, E.; Brolinson, K.A.; Bengtsson, T. Beta3-adrenergic receptors stimulate glucose uptake in brown adipocytes by two mechanisms independently of glucose transporter 4 translocation. Endocrinology 2006, 147, 5730–5739. [Google Scholar] [CrossRef] [PubMed]
- Weiner, J.; Kranz, M.; Kloting, N.; Kunath, A.; Steinhoff, K.; Rijntjes, E.; Kohrle, J.; Zeisig, V.; Hankir, M.; Gebhardt, C.; et al. Thyroid hormone status defines brown adipose tissue activity and browning of white adipose tissues in mice. Sci. Rep. 2016, 6, 38124. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Cheng, W.; Sun, Y.; Dang, Y.; Gong, F.; Zhu, H.; Li, N.; Li, F.; Zhu, Z. Activating brown adipose tissue for weight loss and lowering of blood glucose levels: A microPET study using obese and diabetic model mice. PLoS ONE 2014, 9, e113742. [Google Scholar] [CrossRef]
- Lahesmaa, M.; Orava, J.; Schalin-Jantti, C.; Soinio, M.; Hannukainen, J.C.; Noponen, T.; Kirjavainen, A.; Iida, H.; Kudomi, N.; Enerback, S.; et al. Hyperthyroidism increases brown fat metabolism in humans. J. Clin. Endocrinol. Metab. 2014, 99, E28–E35. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Miao, Q.; Ye, H.; Zhang, Z.; Zuo, C.; Hua, F.; Guan, Y.; Li, Y. The effects of thyroid hormones on brown adipose tissue in humans: A PET-CT study. Diabetes Metab. Res. Rev. 2014, 30, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Skarulis, M.C.; Celi, F.S.; Mueller, E.; Zemskova, M.; Malek, R.; Hugendubler, L.; Cochran, C.; Solomon, J.; Chen, C.; Gorden, P. Thyroid hormone induced brown adipose tissue and amelioration of diabetes in a patient with extreme insulin resistance. J. Clin. Endocrinol. Metab. 2010, 95, 256–262. [Google Scholar] [CrossRef][Green Version]
- Olsen, J.M.; Sato, M.; Dallner, O.S.; Sandstrom, A.L.; Pisani, D.F.; Chambard, J.C.; Amri, E.Z.; Hutchinson, D.S.; Bengtsson, T. Glucose uptake in brown fat cells is dependent on mTOR complex 2-promoted GLUT1 translocation. J. Cell Biol. 2014, 207, 365–374. [Google Scholar] [CrossRef]
- Richardson, J.M.; Pessin, J.E. Identification of a skeletal muscle-specific regulatory domain in the rat GLUT4/muscle-fat gene. J. Biol. Chem. 1993, 268, 21021–21027. [Google Scholar]
- Broeders, E.P.; Vijgen, G.H.; Havekes, B.; Bouvy, N.D.; Mottaghy, F.M.; Kars, M.; Schaper, N.C.; Schrauwen, P.; Brans, B.; van Marken Lichtenbelt, W.D. Thyroid Hormone Activates Brown Adipose Tissue and Increases Non-Shivering Thermogenesis—A Cohort Study in a Group of Thyroid Carcinoma Patients. PLoS ONE 2016, 11, e0145049. [Google Scholar] [CrossRef]
- Yu, X.X.; Lewin, D.A.; Forrest, W.; Adams, S.H. Cold elicits the simultaneous induction of fatty acid synthesis and beta-oxidation in murine brown adipose tissue: Prediction from differential gene expression and confirmation in vivo. FASEB J. 2002, 16, 155–168. [Google Scholar] [CrossRef] [PubMed]
- Blondin, D.P.; Frisch, F.; Phoenix, S.; Guerin, B.; Turcotte, E.E.; Haman, F.; Richard, D.; Carpentier, A.C. Inhibition of Intracellular Triglyceride Lipolysis Suppresses Cold-Induced Brown Adipose Tissue Metabolism and Increases Shivering in Humans. Cell Metab. 2017, 25, 438–447. [Google Scholar] [CrossRef] [PubMed]
- Flores-Morales, A.; Gullberg, H.; Fernandez, L.; Stahlberg, N.; Lee, N.H.; Vennstrom, B.; Norstedt, G. Patterns of liver gene expression governed by TRbeta. Mol. Endocrinol. 2002, 16, 1257–1268. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, T.L.; Werneck-De-Castro, J.P.; Castillo, M.; Bocco, B.M.; Fernandes, G.W.; McAninch, E.A.; Ignacio, D.L.; Moises, C.C.; Ferreira, A.R.; Gereben, B.; et al. Tissue-specific inactivation of type 2 deiodinase reveals multilevel control of fatty acid oxidation by thyroid hormone in the mouse. Diabetes 2014, 63, 1594–1604. [Google Scholar] [CrossRef] [PubMed]
- Labbe, S.M.; Caron, A.; Bakan, I.; Laplante, M.; Carpentier, A.C.; Lecomte, R.; Richard, D. In vivo measurement of energy substrate contribution to cold-induced brown adipose tissue thermogenesis. FASEB J. 2015, 29, 2046–2058. [Google Scholar] [CrossRef] [PubMed]
- Richard, M.A.; Blondin, D.P.; Noll, C.; Lebel, R.; Lepage, M.; Carpentier, A.C. Determination of a pharmacokinetic model for [(11)C]-acetate in brown adipose tissue. EJNMMI Res. 2019, 9, 31. [Google Scholar] [CrossRef]
- Cooper, D.E.; Grevengoed, T.J.; Klett, E.L.; Coleman, R.A. Glycerol-3-phosphate Acyltransferase Isoform-4 (GPAT4) Limits Oxidation of Exogenous Fatty Acids in Brown Adipocytes. J. Biol. Chem. 2015, 290, 15112–15120. [Google Scholar] [CrossRef]
- Irshad, Z.; Dimitri, F.; Christian, M.; Zammit, V.A. Diacylglycerol acyltransferase 2 links glucose utilization to fatty acid oxidation in the brown adipocytes. J. Lipid Res. 2017, 58, 15–30. [Google Scholar] [CrossRef]
- Chen, H.C.; Smith, S.J.; Ladha, Z.; Jensen, D.R.; Ferreira, L.D.; Pulawa, L.K.; McGuire, J.G.; Pitas, R.E.; Eckel, R.H.; Farese, R.V., Jr. Increased insulin and leptin sensitivity in mice lacking acyl CoA:diacylglycerol acyltransferase 1. J. Clin. Investig. 2002, 109, 1049–1055. [Google Scholar] [CrossRef]
- Stone, S.J.; Myers, H.M.; Watkins, S.M.; Brown, B.E.; Feingold, K.R.; Elias, P.M.; Farese, R.V., Jr. Lipopenia and skin barrier abnormalities in DGAT2-deficient mice. J. Biol. Chem. 2004, 279, 11767–11776. [Google Scholar] [CrossRef]
- Oppenheimer, J.H.; Schwartz, H.L.; Lane, J.T.; Thompson, M.P. Functional relationship of thyroid hormone-induced lipogenesis, lipolysis, and thermogenesis in the rat. J. Clin. Investig. 1991, 87, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Goodridge, A.G. Regulation of malic enzyme synthesis by thyroid hormone and glucagon: Inhibitor and kinetic experiments. Mol. Cell. Endocrinol. 1978, 11, 19–29. [Google Scholar] [CrossRef]
- Towle, H.C.; Mariash, C.N.; Oppenheimer, J.H. Changes in the hepatic levels of messenger ribonucleic acid for malic enzyme during induction by thyroid hormone or diet. Biochemistry 1980, 19, 579–585. [Google Scholar] [CrossRef] [PubMed]
- Miksicek, R.J.; Towle, H.C. Changes in the rates of synthesis and messenger RNA levels of hepatic glucose-6-phosphate and 6-phosphogluconate dehydrogenases following induction by diet or thyroid hormone. J. Biol. Chem. 1982, 257, 11829–11835. [Google Scholar] [PubMed]
- Yeh, W.J.; Leahy, P.; Freake, H.C. Regulation of brown adipose tissue lipogenesis by thyroid hormone and the sympathetic nervous system. Am. J. Physiol. 1993, 265, E252–E258. [Google Scholar] [CrossRef] [PubMed]
- Mishra, A.; Zhu, X.G.; Ge, K.; Cheng, S.Y. Adipogenesis is differentially impaired by thyroid hormone receptor mutant isoforms. J. Mol. Endocrinol. 2010, 44, 247–255. [Google Scholar] [CrossRef]
- Ying, H.; Araki, O.; Furuya, F.; Kato, Y.; Cheng, S.Y. Impaired adipogenesis caused by a mutated thyroid hormone alpha1 receptor. Mol. Cell. Biol. 2007, 27, 2359–2371. [Google Scholar] [CrossRef]
- Lu, C.; Cheng, S.Y. Thyroid hormone receptors regulate adipogenesis and carcinogenesis via crosstalk signaling with peroxisome proliferator-activated receptors. J. Mol. Endocrinol. 2010, 44, 143–154. [Google Scholar] [CrossRef]
- Hall, J.A.; Ribich, S.; Christoffolete, M.A.; Simovic, G.; Correa-Medina, M.; Patti, M.E.; Bianco, A.C. Absence of thyroid hormone activation during development underlies a permanent defect in adaptive thermogenesis. Endocrinology 2010, 151, 4573–4582. [Google Scholar] [CrossRef]
- Bianco, A.C.; Carvalho, S.D.; Carvalho, C.R.; Rabelo, R.; Moriscot, A.S. Thyroxine 5’-deiodination mediates norepinephrine-induced lipogenesis in dispersed brown adipocytes. Endocrinology 1998, 139, 571–578. [Google Scholar] [CrossRef][Green Version]
- Klingenspor, M.; Ivemeyer, M.; Wiesinger, H.; Haas, K.; Heldmaier, G.; Wiesner, R.J. Biogenesis of thermogenic mitochondria in brown adipose tissue of Djungarian hamsters during cold adaptation. Biochem. J. 1996, 316, 607–613. [Google Scholar] [CrossRef] [PubMed]
- Iteldmaier, G. Temperature Adaptation and Brown Adipose Tissue in Hairless and Albino Mice. J. Comp. Physiol. 1974, 92, 281–292. [Google Scholar] [CrossRef]
- Wu, Z.; Puigserver, P.; Andersson, U.; Zhang, C.; Adelmant, G.; Mootha, V.; Troy, A.; Cinti, S.; Lowell, B.; Scarpulla, R.C.; et al. Mechanisms controlling mitochondrial biogenesis and respiration through the thermogenic coactivator PGC-1. Cell 1999, 98, 115–124. [Google Scholar] [CrossRef]
- Puigserver, P.; Wu, Z.; Park, C.W.; Graves, R.; Wright, M.; Spiegelman, B.M. A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell 1998, 92, 829–839. [Google Scholar] [CrossRef]
- Leone, T.C.; Lehman, J.J.; Finck, B.N.; Schaeffer, P.J.; Wende, A.R.; Boudina, S.; Courtois, M.; Wozniak, D.F.; Sambandam, N.; Bernal-Mizrachi, C.; et al. PGC-1alpha deficiency causes multi-system energy metabolic derangements: Muscle dysfunction, abnormal weight control and hepatic steatosis. PLoS Biol. 2005, 3, e101. [Google Scholar] [CrossRef]
- Lin, J.; Wu, P.H.; Tarr, P.T.; Lindenberg, K.S.; St-Pierre, J.; Zhang, C.Y.; Mootha, V.K.; Jager, S.; Vianna, C.R.; Reznick, R.M.; et al. Defects in adaptive energy metabolism with CNS-linked hyperactivity in PGC-1alpha null mice. Cell 2004, 119, 121–135. [Google Scholar] [CrossRef]
- Maushart, C.I.; Loeliger, R.; Gashi, G.; Christ-Crain, M.; Betz, M.J. Resolution of Hypothyroidism Restores Cold-Induced Thermogenesis in Humans. Thyroid 2019, 29, 493–501. [Google Scholar] [CrossRef]
- Gavrila, A.; Hasselgren, P.O.; Glasgow, A.; Doyle, A.N.; Lee, A.J.; Fox, P.; Gautam, S.; Hennessey, J.V.; Kolodny, G.M.; Cypess, A.M. Variable Cold-Induced Brown Adipose Tissue Response to Thyroid Hormone Status. Thyroid 2017, 27, 1–10. [Google Scholar] [CrossRef]
- Sato, T.; Imura, E.; Murata, A.; Igarashi, N. Thyroid hormone-catecholamine interrelationship during cold acclimation in rats. Compensatory role of catecholamine for altered thyroid states. Acta Endocrinol. (Copenh.) 1986, 113, 536–542. [Google Scholar] [CrossRef]
- Hernandez, A.; Obregon, M.J. Triiodothyronine amplifies the adrenergic stimulation of uncoupling protein expression in rat brown adipocytes. Am. J. Physiol. Endocrinol. Metab. 2000, 278, E769–E777. [Google Scholar] [CrossRef]
- Liu, X.T.; Li, Q.F.; Huang, C.X.; Sun, R.Y. Effects of thyroid status on cold-adaptive thermogenesis in Brandt’s vole, Microtus brandti. Physiol. Zool. 1997, 70, 352–361. [Google Scholar] [PubMed]
- Weitzel, J.M.; Iwen, K.A.; Seitz, H.J. Regulation of mitochondrial biogenesis by thyroid hormone. Exp. Physiol. 2003, 88, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Wrutniak-Cabello, C.; Casas, F.; Cabello, G. Thyroid hormone action in mitochondria. J. Mol. Endocrinol. 2001, 26, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Wulf, A.; Harneit, A.; Kroger, M.; Kebenko, M.; Wetzel, M.G.; Weitzel, J.M. T3-mediated expression of PGC-1alpha via a far upstream located thyroid hormone response element. Mol. Cell. Endocrinol. 2008, 287, 90–95. [Google Scholar] [CrossRef]
- de Quiroga, G.B.; Lopez-Torres, M.; Perez-Campo, R.; Abelenda, M.; Paz Nava, M.; Puerta, M.L. Effect of cold acclimation on GSH, antioxidant enzymes and lipid peroxidation in brown adipose tissue. Biochem. J. 1991, 277, 289–292. [Google Scholar] [CrossRef]
- Buzadzic, B.; Korac, B.; Petrovic, V.M. The effect of adaptation to cold and re-adaptation to room temperature on the level of glutathione in rat tissues. J. Ther. Biol. 1999, 24, 373–377. [Google Scholar] [CrossRef]
- Zhang, Y.; Goldman, S.; Baerga, R.; Zhao, Y.; Komatsu, M.; Jin, S. Adipose-specific deletion of autophagy-related gene 7 (atg7) in mice reveals a role in adipogenesis. Proc. Natl. Acad. Sci. USA 2009, 106, 19860–19865. [Google Scholar] [CrossRef]
- Mottillo, E.P.; Desjardins, E.M.; Crane, J.D.; Smith, B.K.; Green, A.E.; Ducommun, S.; Henriksen, T.I.; Rebalka, I.A.; Razi, A.; Sakamoto, K.; et al. Lack of Adipocyte AMPK Exacerbates Insulin Resistance and Hepatic Steatosis through Brown and Beige Adipose Tissue Function. Cell Metab. 2016, 24, 118–129. [Google Scholar] [CrossRef]
- Lu, Y.; Fujioka, H.; Joshi, D.; Li, Q.; Sangwung, P.; Hsieh, P.; Zhu, J.; Torio, J.; Sweet, D.; Wang, L.; et al. Mitophagy is required for brown adipose tissue mitochondrial homeostasis during cold challenge. Sci. Rep. 2018, 8, 8251. [Google Scholar] [CrossRef]
- Cairo, M.; Villarroya, J.; Cereijo, R.; Campderros, L.; Giralt, M.; Villarroya, F. Thermogenic activation represses autophagy in brown adipose tissue. Int. J. Obes. (Lond.) 2016, 40, 1591–1599. [Google Scholar] [CrossRef]
- Herz, C.T.; Kiefer, F.W. Adipose tissue browning in mice and humans. J. Endocrinol. 2019, 241, R97–R109. [Google Scholar] [CrossRef] [PubMed]
- Krause, K. Novel Aspects of White Adipose Tissue Browning by Thyroid Hormones. Exp. Clin. Endocrinol. Diabetes 2019. [Google Scholar] [CrossRef] [PubMed]
- Finan, B.; Clemmensen, C.; Zhu, Z.; Stemmer, K.; Gauthier, K.; Muller, L.; De Angelis, M.; Moreth, K.; Neff, F.; Perez-Tilve, D.; et al. Chemical Hybridization of Glucagon and Thyroid Hormone Optimizes Therapeutic Impact for Metabolic Disease. Cell 2016, 167, 843–857. [Google Scholar] [CrossRef] [PubMed]
- Johann, K.; Cremer, A.L.; Fischer, A.W.; Heine, M.; Pensado, E.R.; Resch, J.; Nock, S.; Virtue, S.; Harder, L.; Oelkrug, R.; et al. Thyroid-Hormone-Induced Browning of White Adipose Tissue Does Not Contribute to Thermogenesis and Glucose Consumption. Cell Rep. 2019, 27, 3385–3400. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Takahashi, N.; Yasubuchi, M.; Kim, Y.I.; Hashizaki, H.; Kim, M.J.; Sakamoto, T.; Goto, T.; Kawada, T. Triiodothyronine induces UCP-1 expression and mitochondrial biogenesis in human adipocytes. Am. J. Physiol. Cell Physiol. 2012, 302, C463–C472. [Google Scholar] [CrossRef]
- Marrif, H.; Schifman, A.; Stepanyan, Z.; Gillis, M.A.; Calderone, A.; Weiss, R.E.; Samarut, J.; Silva, J.E. Temperature homeostasis in transgenic mice lacking thyroid hormone receptor-alpha gene products. Endocrinology 2005, 146, 2872–2884. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lin, J.Z.; Martagon, A.J.; Cimini, S.L.; Gonzalez, D.D.; Tinkey, D.W.; Biter, A.; Baxter, J.D.; Webb, P.; Gustafsson, J.A.; Hartig, S.M.; et al. Pharmacological Activation of Thyroid Hormone Receptors Elicits a Functional Conversion of White to Brown Fat. Cell Rep. 2015, 13, 1528–1537. [Google Scholar] [CrossRef]
- Lombardi, A.; Senese, R.; De Matteis, R.; Busiello, R.A.; Cioffi, F.; Goglia, F.; Lanni, A. 3,5-Diiodo-L-thyronine activates brown adipose tissue thermogenesis in hypothyroid rats. PLoS ONE 2015, 10, e0116498. [Google Scholar] [CrossRef]
- Gachkar, S.; Oelkrug, R.; Martinez-Sanchez, N.; Rial-Pensado, E.; Warner, A.; Hoefig, C.S.; Lopez, M.; Mittag, J. 3-Iodothyronamine Induces Tail Vasodilation Through Central Action in Male Mice. Endocrinology 2017, 158, 1977–1984. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yau, W.W.; Yen, P.M. Thermogenesis in Adipose Tissue Activated by Thyroid Hormone. Int. J. Mol. Sci. 2020, 21, 3020. https://doi.org/10.3390/ijms21083020
Yau WW, Yen PM. Thermogenesis in Adipose Tissue Activated by Thyroid Hormone. International Journal of Molecular Sciences. 2020; 21(8):3020. https://doi.org/10.3390/ijms21083020
Chicago/Turabian StyleYau, Winifred W., and Paul M. Yen. 2020. "Thermogenesis in Adipose Tissue Activated by Thyroid Hormone" International Journal of Molecular Sciences 21, no. 8: 3020. https://doi.org/10.3390/ijms21083020
APA StyleYau, W. W., & Yen, P. M. (2020). Thermogenesis in Adipose Tissue Activated by Thyroid Hormone. International Journal of Molecular Sciences, 21(8), 3020. https://doi.org/10.3390/ijms21083020



