Hydroxyapatite Formation Coexists with Amyloid-like Self-Assembly of Human Amelogenin
Abstract
1. Introduction
2. Results
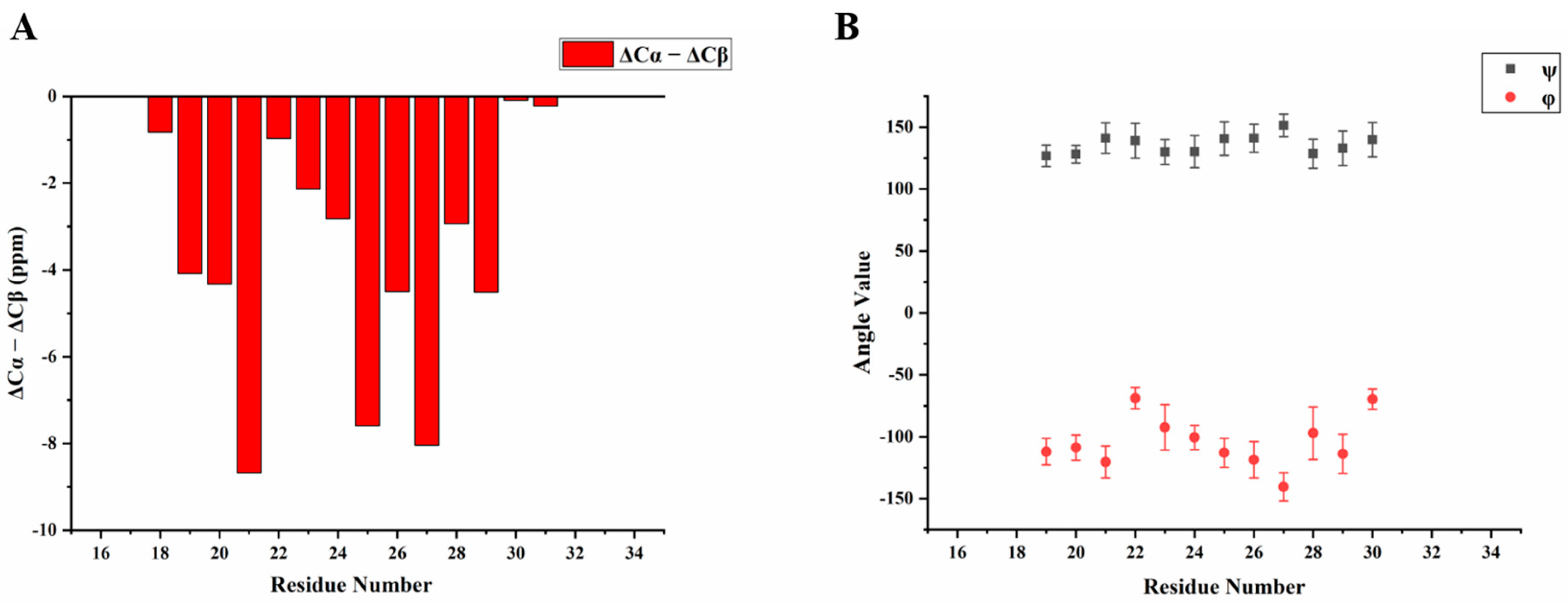
2.1. H175 could Adopt Amyloid-Like Structure at Acidic pH Conditions
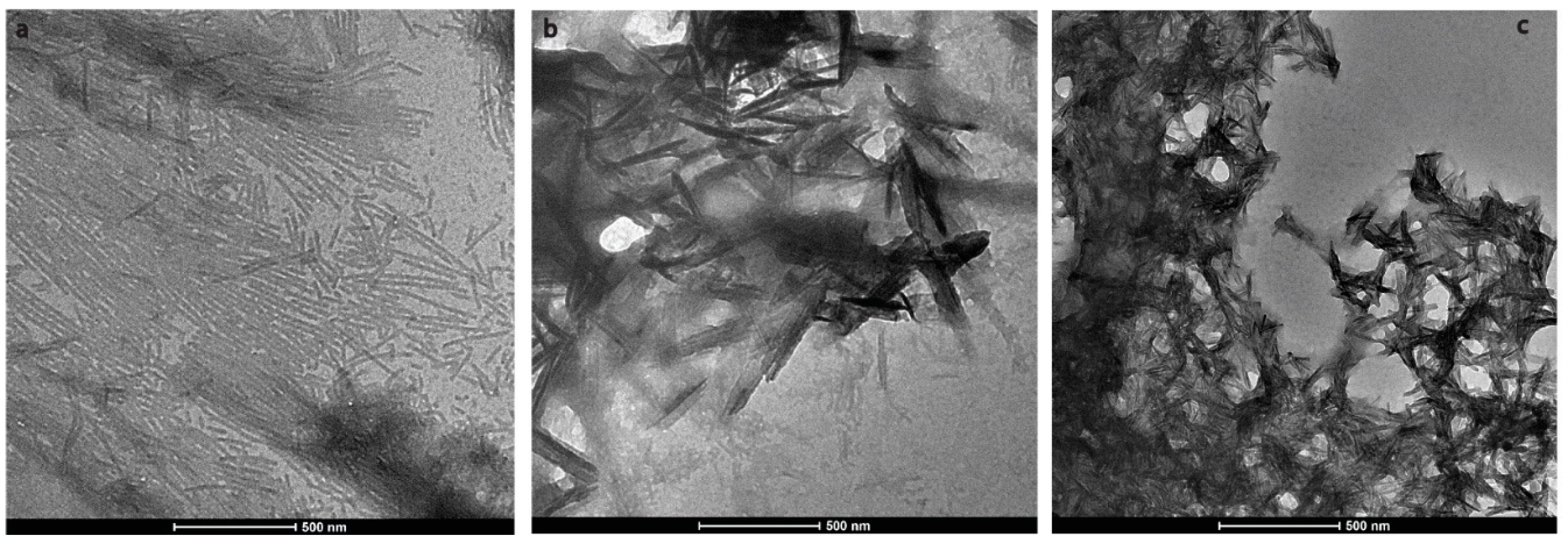
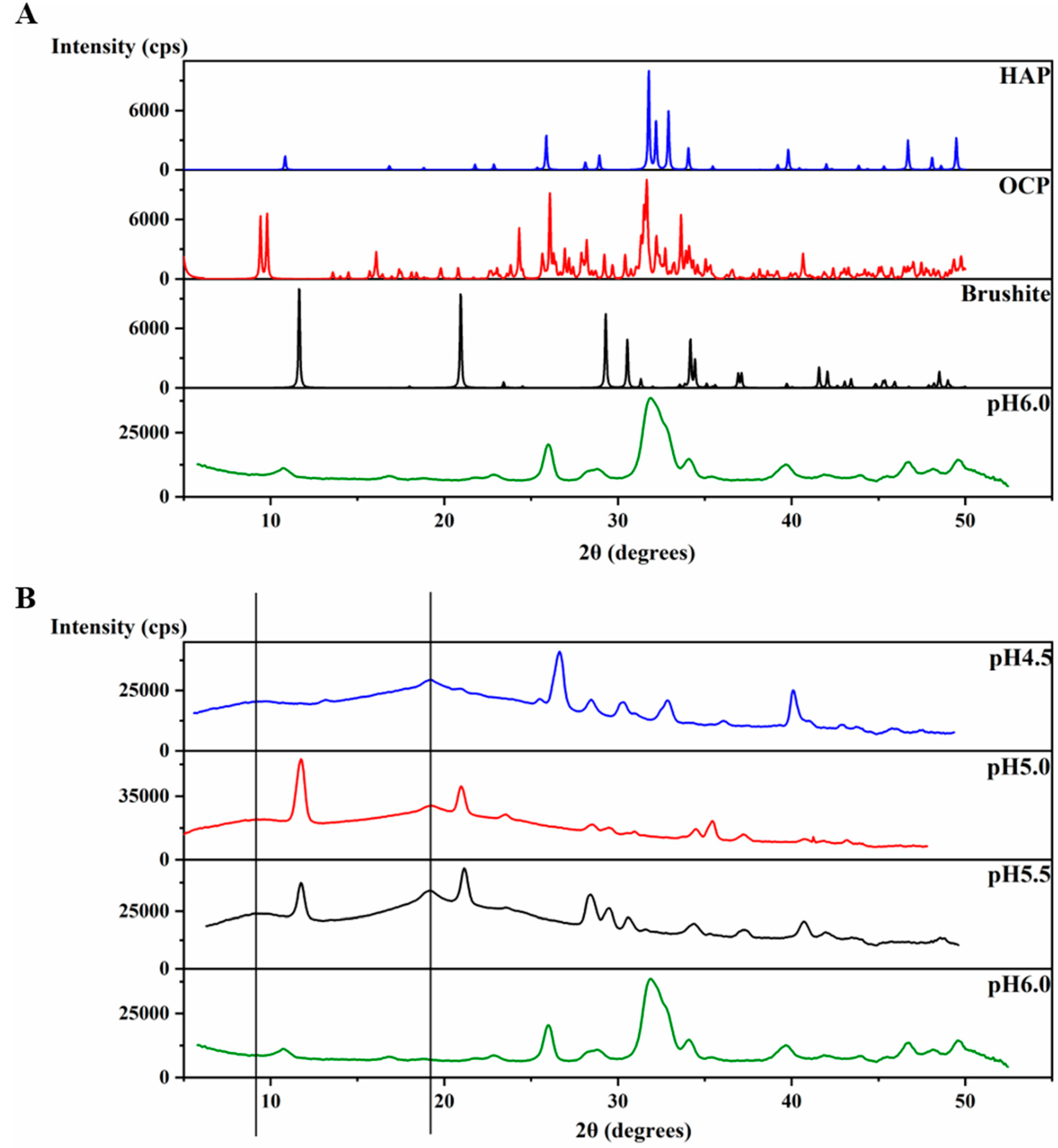
2.2. HAP is Formed at pH 6.0 in the Presence of H175 Amyloid
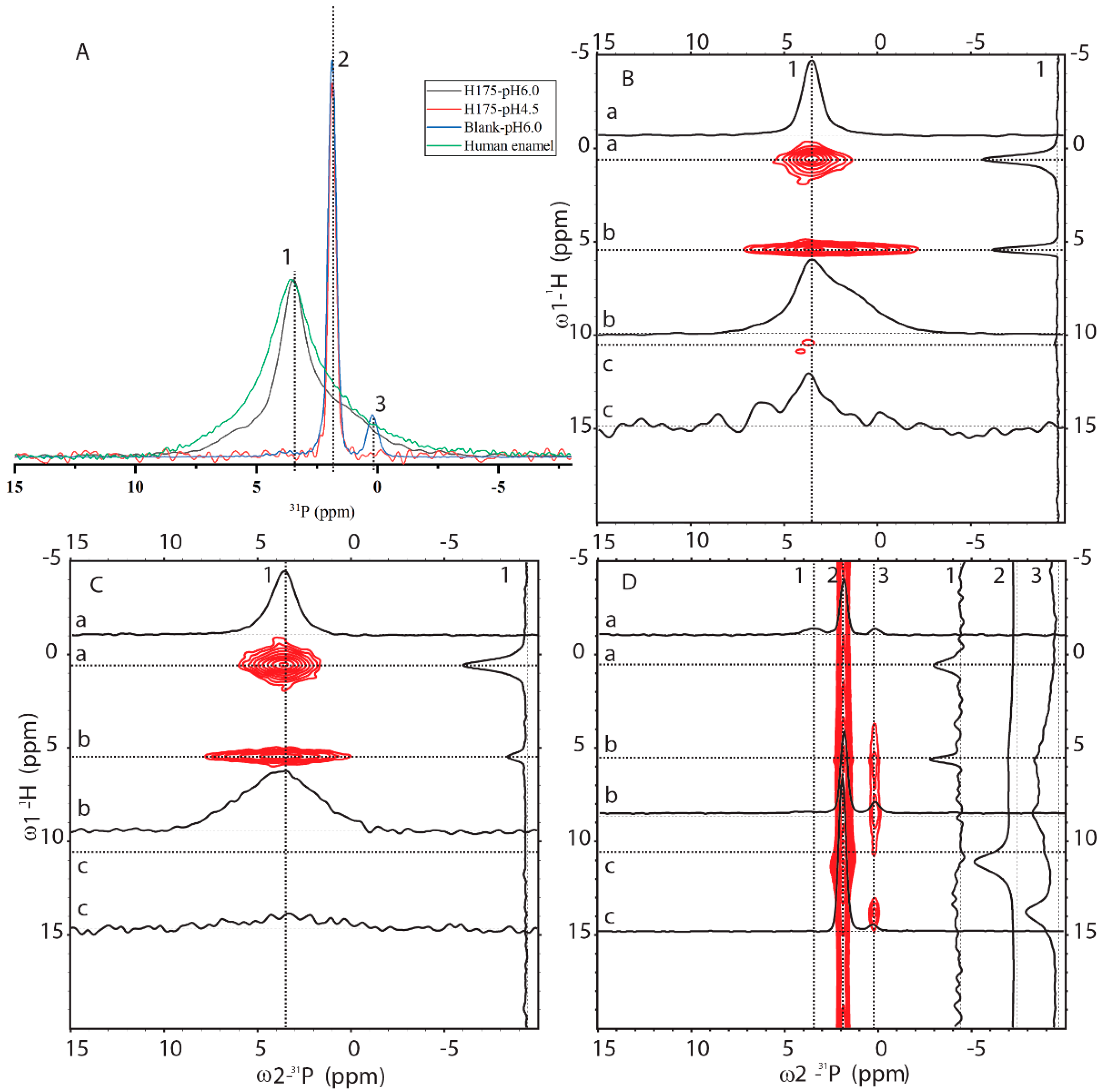
2.3. 31P Solid-State NMR (SSNMR) Compared HAP Formed at pH 6.0 with the Natural Enamel
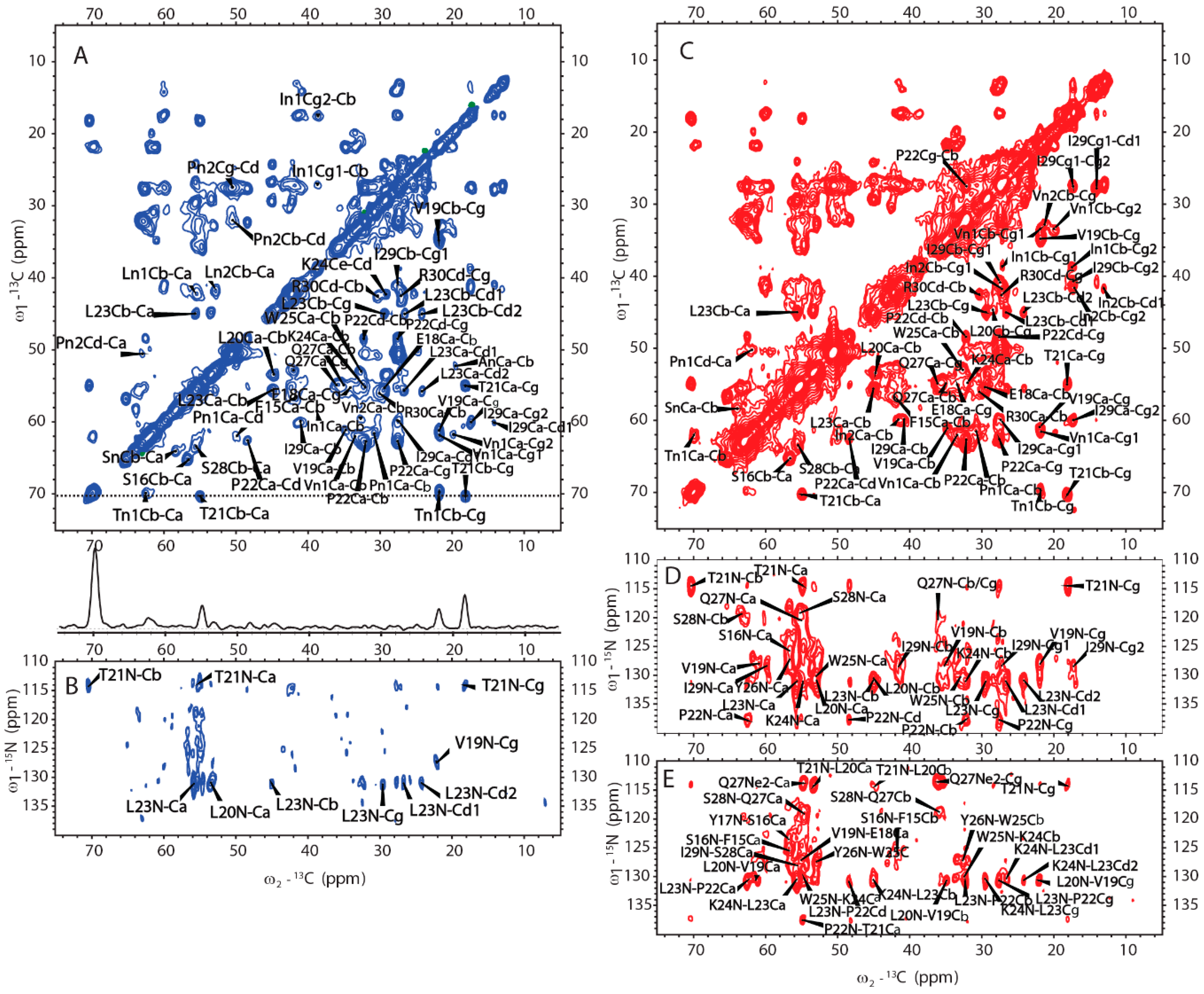
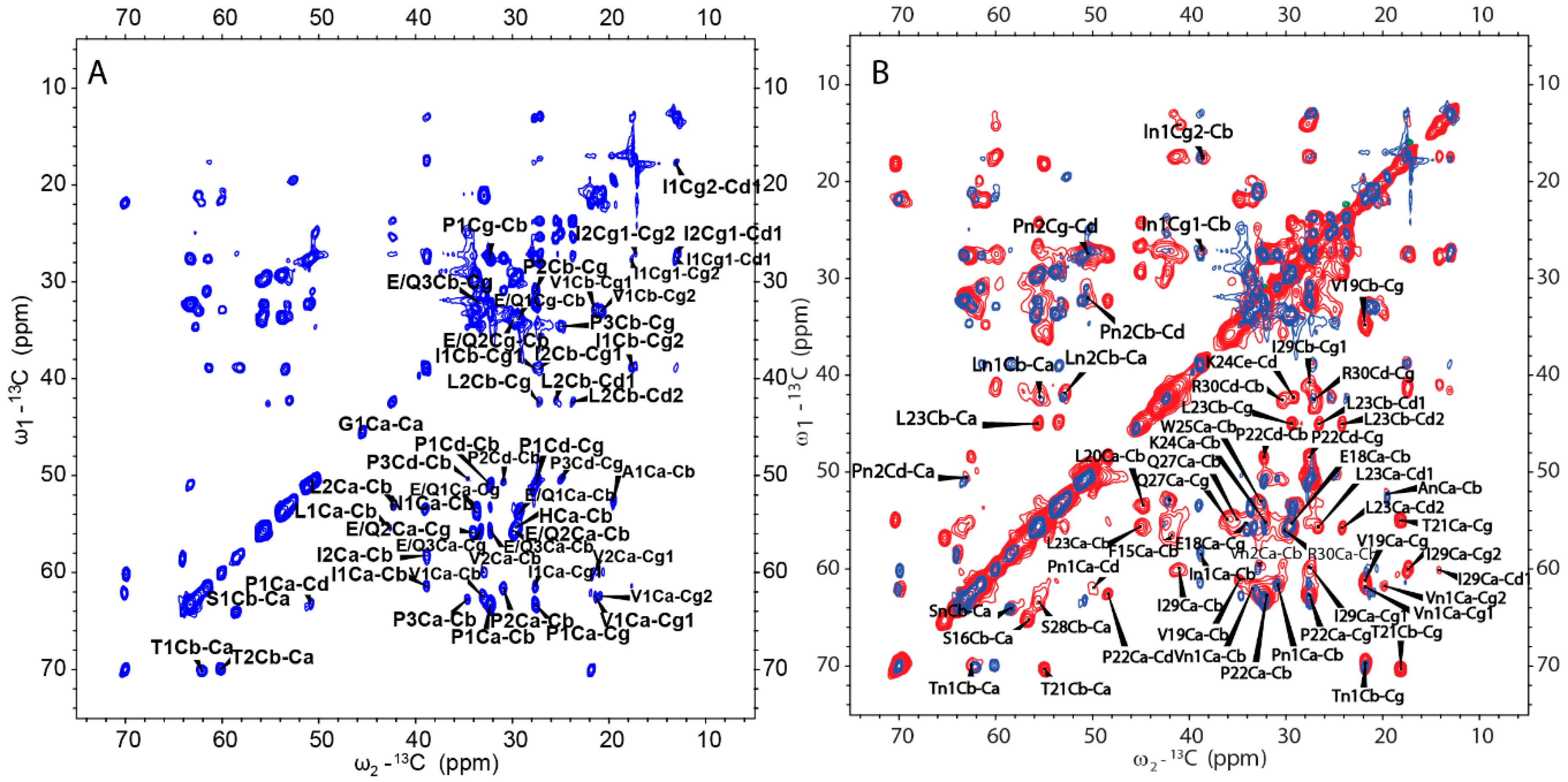
2.4. Amyloid-like Assemblies of H175 are Facilitated by a Short Segment of Sequence
2.5. H175 has Other Structural Components beside β-amyloid
3. Discussion
4. Materials and Methods
4.1. Recombinant Amelogenin Protein Expression and Purification
4.2. Amelogenin Protein H175 Self-assembly Preparation
4.3. Enamel Isolation from Dental Tissues
4.4. X-Ray Diffraction (XRD)
4.5. Atomic Force Microscopy (AFM)
4.6. Transmission Electron Microscopy (TEM)
4.7. Magic Angle Spinning (MAS) Solid-State NMR Experiments
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| HAP | hydroxyapatite |
| OCP | octacalcium phosphate |
| XRD | x-ray diffraction |
| SSNMR | solid-state NMR |
| HETCOR | heteronuclear correlation |
| AFM | atomic force microscope |
| TEM | transmission electron microscope |
| LRAP | leucine-rich amelogenin peptide |
| CP | cross-polarization |
| TEV INEPT TOBSY | tobacco etch virus insensitive nuclei enhanced by polarization transfer total through bond correlation spectroscopy |
References
- Skinner, H.C.W.; Jahren, A.H. Biomineralization. Treatise Geochem. 2003, 8, 117–184. [Google Scholar]
- Moradian-Oldak, J. The regeneration of tooth enamel. Dimens Dent. Hyg. 2009, 7, 12–15. [Google Scholar] [PubMed]
- Simmer, J.P.; Fincham, A.G. Molecular Mechanisms of Dental Enamel Formation. Crit. Rev. Oral. Biol. Med. 1995, 6, 84–108. [Google Scholar] [CrossRef] [PubMed]
- Gadhia, K.; McDonald, S.; Arkutu, N.; Malik, K. Amelogenesis imperfecta: An introduction. Br. Dent. J. 2012, 212, 377–379. [Google Scholar] [CrossRef] [PubMed]
- Gibson, C.W.; Yuan, Z.A.; Hall, B.; Longenecker, G.; Chen, E.; Thyagarajan, T.; Sreenath, T.; Wright, J.T.; Decker, S.; Piddington, R.; et al. Amelogenin-deficient mice display an amelogenesis imperfecta phenotype. J. Biol. Chem. 2001, 276, 31871–31875. [Google Scholar] [CrossRef] [PubMed]
- Ruan, Q.; Moradian-Oldak, J. Amelogenin and Enamel Biomimetics. J. Mater. Chem. B 2015, 3, 3112–3129. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Sun, Z.; Moradian-Oldak, J. Controlled remineralization of enamel in the presence of amelogenin and fluoride. Biomaterials 2009, 30, 478–483. [Google Scholar] [CrossRef]
- Kwak, S.Y.; Litman, A.; Margolis, H.C.; Yamakoshi, Y.; Simmer, J.P. Biomimetic Enamel Regeneration Mediated by Leucine-Rich Amelogenin Peptide. J. Dent. Res. 2017, 96, 524–530. [Google Scholar] [CrossRef]
- Dogan, S.; Fong, H.; Yucesoy, D.T.; Cousin, T.; Gresswell, C.; Dag, S.; Huang, G.; Sarikaya, M. Biomimetic Tooth Repair: Amelogenin-Derived Peptide Enables in Vitro Remineralization of Human Enamel. ACS Biomater. Sci. Eng. 2018, 4, 1788–1796. [Google Scholar] [CrossRef]
- Buchko, G.W.; Tarasevich, B.J.; Bekhazi, J.; Snead, M.L.; Shaw, W.J. A solution NMR investigation into the early events of amelogenin nanosphere self-assembly initiated with sodium chloride or calcium chloride. Biochemistry 2008, 47, 13215–13222. [Google Scholar] [CrossRef]
- Fincham, A.G.; Moradian-Oldak, J.; Simmer, J.P.; Sarte, P.; Lau, E.C.; Dieswisch, T.; Slavkin, H.C. Self-assembly of a recombinant amelogenin protein generates supramolecular structures. J. Struct. Biol. 1994, 112, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Sun, Z.; Ma, R.; Fan, D.; Moradian-Oldak, J. Amelogenin “nanorods” formation during proteolysis by Mmp-20. J. Struct Biol. 2011, 176, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Engelberth, S.A.; Bacino, M.S.; Sandhu, S.; Li, W.; Bonde, J.; Habelitz, S. Progression of Self-Assembly of Amelogenin Protein Supramolecular Structures in Simulated Enamel Fluid. Biomacromolecules 2018, 19, 3917–3924. [Google Scholar] [CrossRef] [PubMed]
- Fincham, A.G.; Moradian-Oldak, J.; Diekwisch, T.G.H.; Lyaruu, D.M.; Wright, J.T.; Bringas, P.J.; Slavkin, H.C. Evidence for Amelogenin “nanospheres” as functional components of secretory-stage enamel matrix. J. Struct Biol. 1995, 115, 50–59. [Google Scholar] [CrossRef]
- Jodaikin, A.; Traub, W.; Weiner, S. Protein conformation in rat tooth enamel. Arch. Oral. Biol. 1986, 31, 685–689. [Google Scholar] [CrossRef]
- Carneiro, K.M.; Zhai, H.; Zhu, L.; Horst, J.A.; Sitlin, M.; Nguyen, M.; Wagner, M.; Simpliciano, C.; Milder, M.; Chen, C.L.; et al. Amyloid-like ribbons of amelogenins in enamel mineralization. Sci. Rep. 2016, 6, 23105. [Google Scholar] [CrossRef]
- Lu, J.X.; Xu, Y.S.; Buchko, G.W.; Shaw, W.J. Mineral association changes the secondary structure and dynamics of murine amelogenin. J. Dent. Res. 2013, 92, 1000–1004. [Google Scholar] [CrossRef]
- Lacruz, R.S.; Habelitz, S.; Wright, J.T.; Paine, M.L. Dental Enamel Formation and Implications for Oral Health and Disease. Physiol. Rev. 2017, 97, 939–993. [Google Scholar] [CrossRef]
- Martinez-Avila, O.M.; Wu, S.; Cheng, Y.; Lee, R.; Khan, F.; Habelitz, S. Self-assembly of amelogenin proteins at the water-oil interface. Eur. J. Oral. Sci. 2011, 119 (Suppl. 1), 75–82. [Google Scholar] [CrossRef]
- He, X.; Wu, S.; Martinez-Avila, O.; Cheng, Y.; Habelitz, S. Self-aligning amelogenin nanoribbons in oil-water system. J. Struct Biol. 2011, 174, 203–212. [Google Scholar] [CrossRef]
- Ma, C.W.; Zhang, J.; Dong, X.Q.; Lu, J.X. Amyloid structure of high-order assembly of Leucine-rich amelogenin revealed by solid-state NMR. J. Struct Biol. 2019, 206, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Sanii, B.; Martinez-Avila, O.; Simpliciano, C.; Zuckermann, R.N.; Habelitz, S. Matching 4.7-angstrom XRD Spacing in Amelogenin Nanoribbons and Enamel Matrix. J. Dent. Res. 2014, 93, 918–922. [Google Scholar] [CrossRef] [PubMed]
- Maji, S.K.; Wang, L.; Greenwald, J.; Riek, R. Structure-activity relationship of amyloid fibrils. Febs. Lett. 2009, 583, 2610–2617. [Google Scholar] [CrossRef] [PubMed]
- Yesinowski, J.P.; Eckert, H. Hydrogen Environments in Calcium Phosphates:1H MAS NMR at High Spinning Speeds. J. Am. Chem. Soc. 1987, 109, 6274–6282. [Google Scholar] [CrossRef]
- Leroy, C.; Aussenac, F.; Bonhomme-Coury, L.; Osaka, A.; Hayakawa, S.; Babonneau, F.; Coelho-Diogo, C.; Bonhomme, C. Hydroxyapatites: Key Structural Questions and Answers from Dynamic Nuclear Polarization. Anal. Chem. 2017, 89, 10201–10207. [Google Scholar] [CrossRef]
- Cho, G.; Wu, Y.; Ackerman, J.L. Detection of hydroxyl ions in bone mineral by solid-state NMR spectroscopy. Science 2003, 300, 1123–1127. [Google Scholar] [CrossRef]
- Shen, Y.; Bax, A. Protein backbone and sidechain torsion angles predicted from NMR chemical shifts using artificial neural networks. J. Biomol. NMR 2013, 56, 227–241. [Google Scholar] [CrossRef]
- Chow, L.C. Solubility of calcium phosphates. Monogr. Oral. Sci. 2001, 18, 94–111. [Google Scholar]
- Tarasevich, B.J.; Lea, S.; Bernt, W.; Engelhard, M.; Shaw, W.J. Adsorption of amelogenin onto self-assembled and fluoroapatite surfaces. J. Phys. Chem. B 2009, 113, 1833–1842. [Google Scholar] [CrossRef]
- Tarasevich, B.J.; Lea, S.; Bernt, W.; Engelhard, M.H.; Shaw, W.J. Changes in the quaternary structure of amelogenin when adsorbed onto surfaces. Biopolymers 2009, 91, 103–107. [Google Scholar] [CrossRef]
- Baldus, M.; Meier, B.H. Total Correlation Spectroscopy in the Solid State. The Use of Scalar Couplings to Determine the Through-Bond Connectivity. J. Magn. Reson. 1996, 121, 65–69. [Google Scholar] [CrossRef]
- Baldus, M.; Iuliucci, R.J.; Meier, B.H. Probing Through-Bond Connectivities and Through-Space Distances in Solids by Magic-Angle-Spinning Nuclear Magnetic Resonance. J. Am. Chem. Soc. 1997, 119, 1121–1124. [Google Scholar] [CrossRef][Green Version]
- Pauli, J.; Baldus, M.; van Rossum, B.; de Groot, H.; Oschkinat, H. Backbone and side-chain 13C and 15N signal assignments of the alpha-spectrin SH3 domain by magic angle spinning solid-state NMR at 17.6 Tesla. Chembiochem 2001, 2, 272–281. [Google Scholar] [CrossRef]
- Igumenova, T.I.; Wand, A.J.; McDermott, A.E. Assignment of the backbone resonances for microcrystalline ubiquitin. J. Am. Chem. Soc. 2004, 126, 5323–5331. [Google Scholar] [CrossRef]
- Marulanda, D.; Tasayco, M.L.; McDermott, A.; Cataldi, M.; Arriaran, V.; Polenova, T. Magic angle spinning solid-state NMR spectroscopy for structural studies of protein interfaces. resonance assignments of differentially enriched Escherichia coli thioredoxin reassembled by fragment complementation. J. Am. Chem. Soc. 2004, 126, 16608–16620. [Google Scholar] [CrossRef]
- Shi, L.; Ahmed, M.A.; Zhang, W.; Whited, G.; Brown, L.S.; Ladizhansky, V. Three-dimensional solid-state NMR study of a seven-helical integral membrane proton pump--structural insights. J. Mol. Biol. 2009, 386, 1078–1093. [Google Scholar] [CrossRef]
- Sperling, L.J.; Berthold, D.A.; Sasser, T.L.; Jeisy-Scott, V.; Rienstra, C.M. Assignment strategies for large proteins by magic-angle spinning NMR: The 21-kDa disulfide-bond-forming enzyme DsbA. J. Mol. Biol. 2010, 399, 268–282. [Google Scholar] [CrossRef]
- Baldus, M.; Petkova, A.T.; Herzfeld, J.; Griffin, R.G. Cross polarization in the tilted frame: Assignment and spectral simplification in heteronuclear spin systems. Mol. Phys. 1998, 95, 1197–1207. [Google Scholar] [CrossRef]
- Fung, B.M.; Khitrin, A.K.; Ermolaev, K. An improved broadband decoupling sequence for liquid crystals and solids. J. Magn. Reson. 2000, 142, 97–101. [Google Scholar] [CrossRef]
- Mathew, R.; Gunawidjaja, P.N.; Izquierdo-Barba, I.; Jansson, K.; Garcia, A.; Arcos, D.; Vallet-Regi, M.; Eden, M. Solid-State P and H NMR Investigations of Amorphous and Crystalline Calcium Phosphates Grown Biomimetically From a Mesoporous Bioactive Glass. J. Phys. Chem. C Nanomater. Interfaces 2011, 115, 20572–20582. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Wang, J.; Ma, C.; Lu, J. Hydroxyapatite Formation Coexists with Amyloid-like Self-Assembly of Human Amelogenin. Int. J. Mol. Sci. 2020, 21, 2946. https://doi.org/10.3390/ijms21082946
Zhang J, Wang J, Ma C, Lu J. Hydroxyapatite Formation Coexists with Amyloid-like Self-Assembly of Human Amelogenin. International Journal of Molecular Sciences. 2020; 21(8):2946. https://doi.org/10.3390/ijms21082946
Chicago/Turabian StyleZhang, Jing, Jian Wang, Chengwei Ma, and Junxia Lu. 2020. "Hydroxyapatite Formation Coexists with Amyloid-like Self-Assembly of Human Amelogenin" International Journal of Molecular Sciences 21, no. 8: 2946. https://doi.org/10.3390/ijms21082946
APA StyleZhang, J., Wang, J., Ma, C., & Lu, J. (2020). Hydroxyapatite Formation Coexists with Amyloid-like Self-Assembly of Human Amelogenin. International Journal of Molecular Sciences, 21(8), 2946. https://doi.org/10.3390/ijms21082946




