Biocatalytic CO2 Absorption and Structural Studies of Carbonic Anhydrase under Industrially-Relevant Conditions
Abstract
1. Introduction
2. Results and Discussion
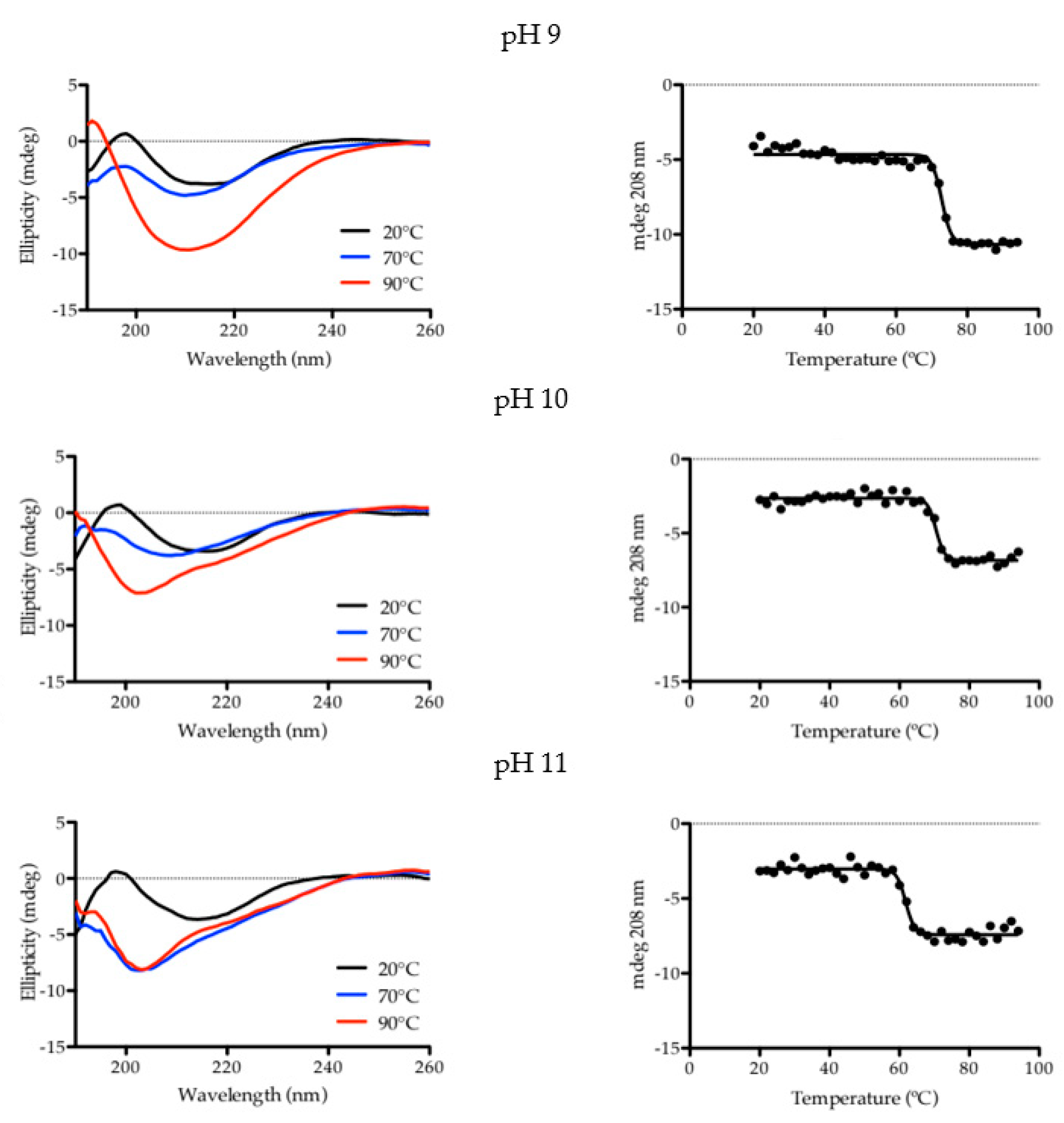
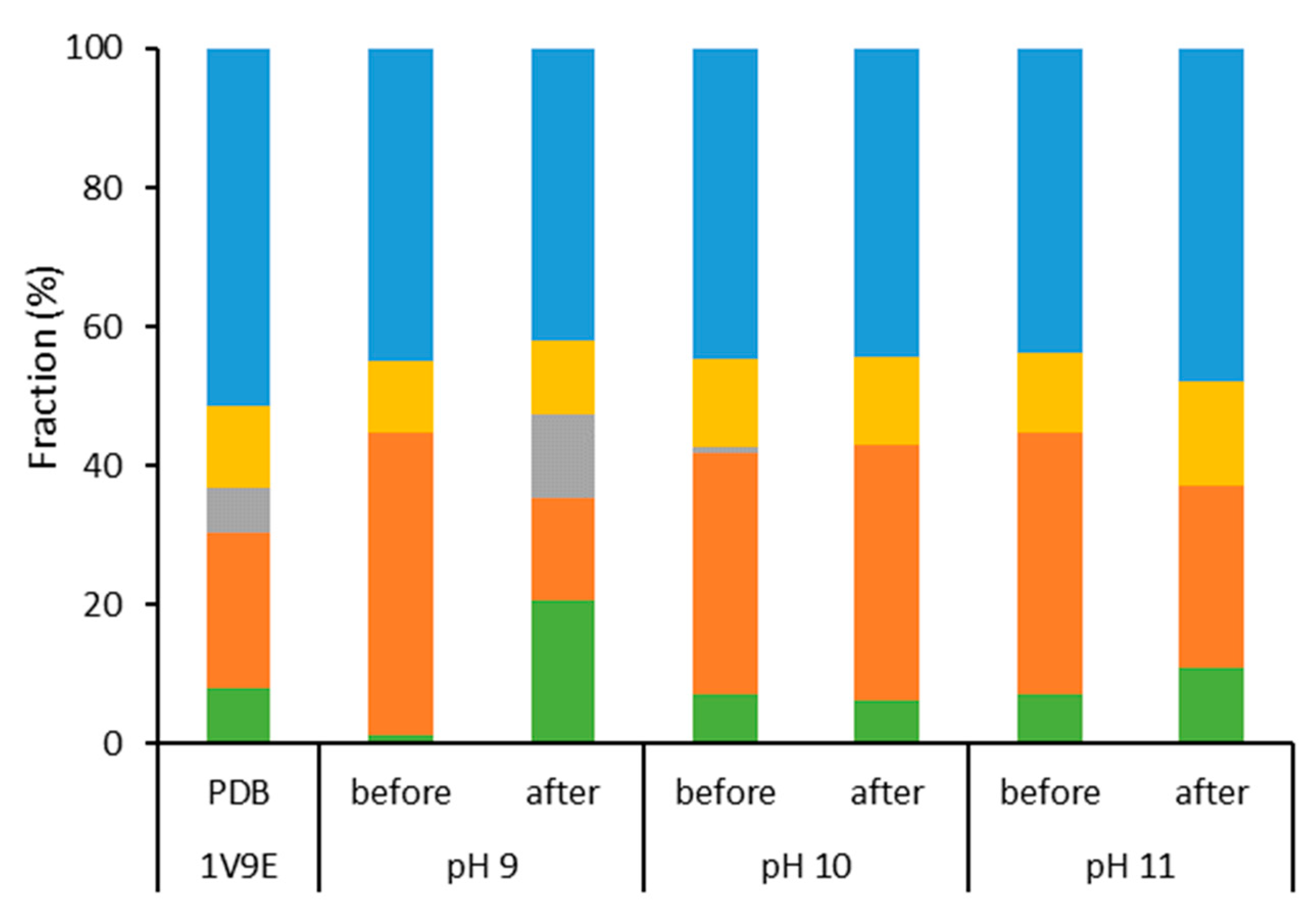
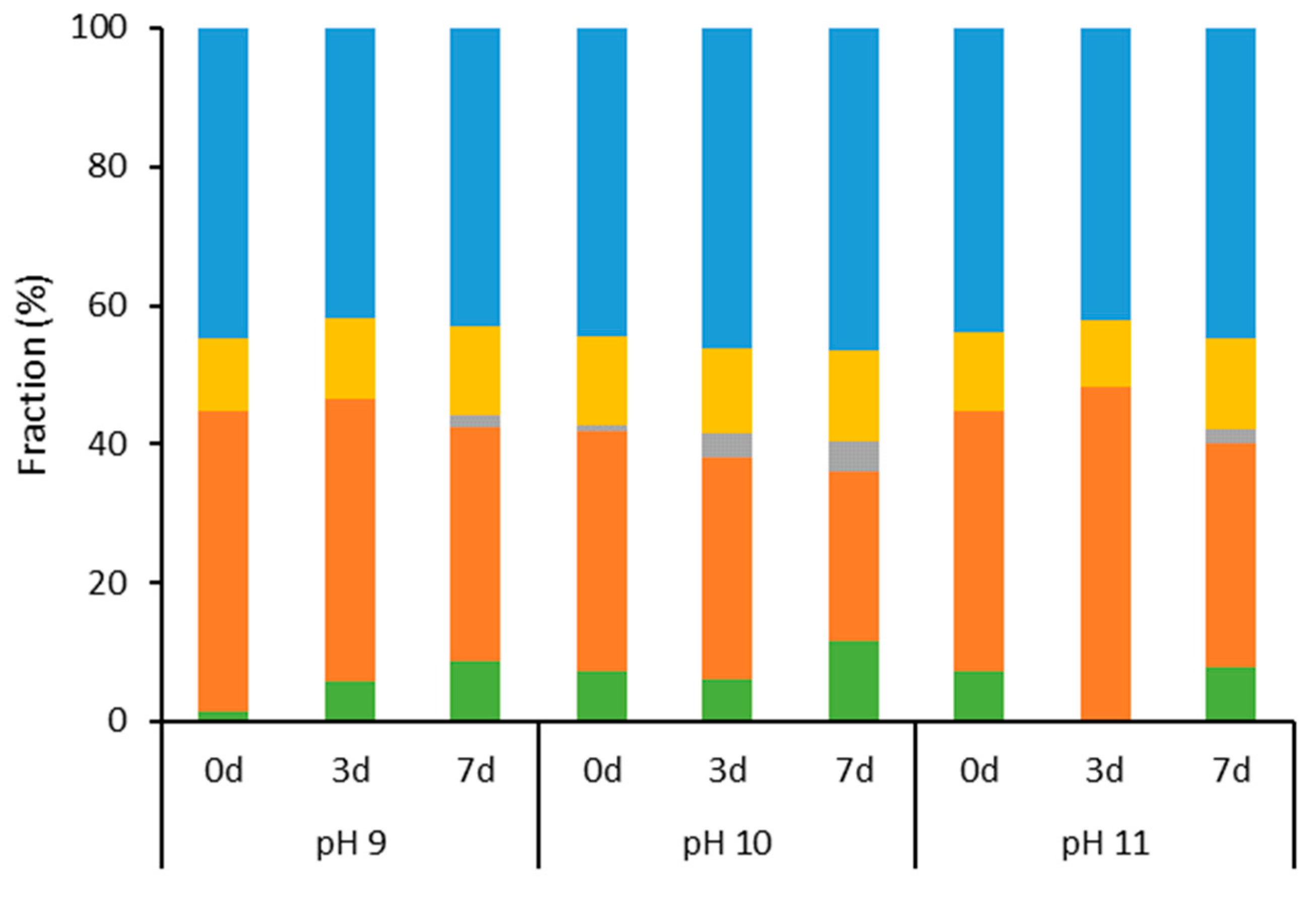
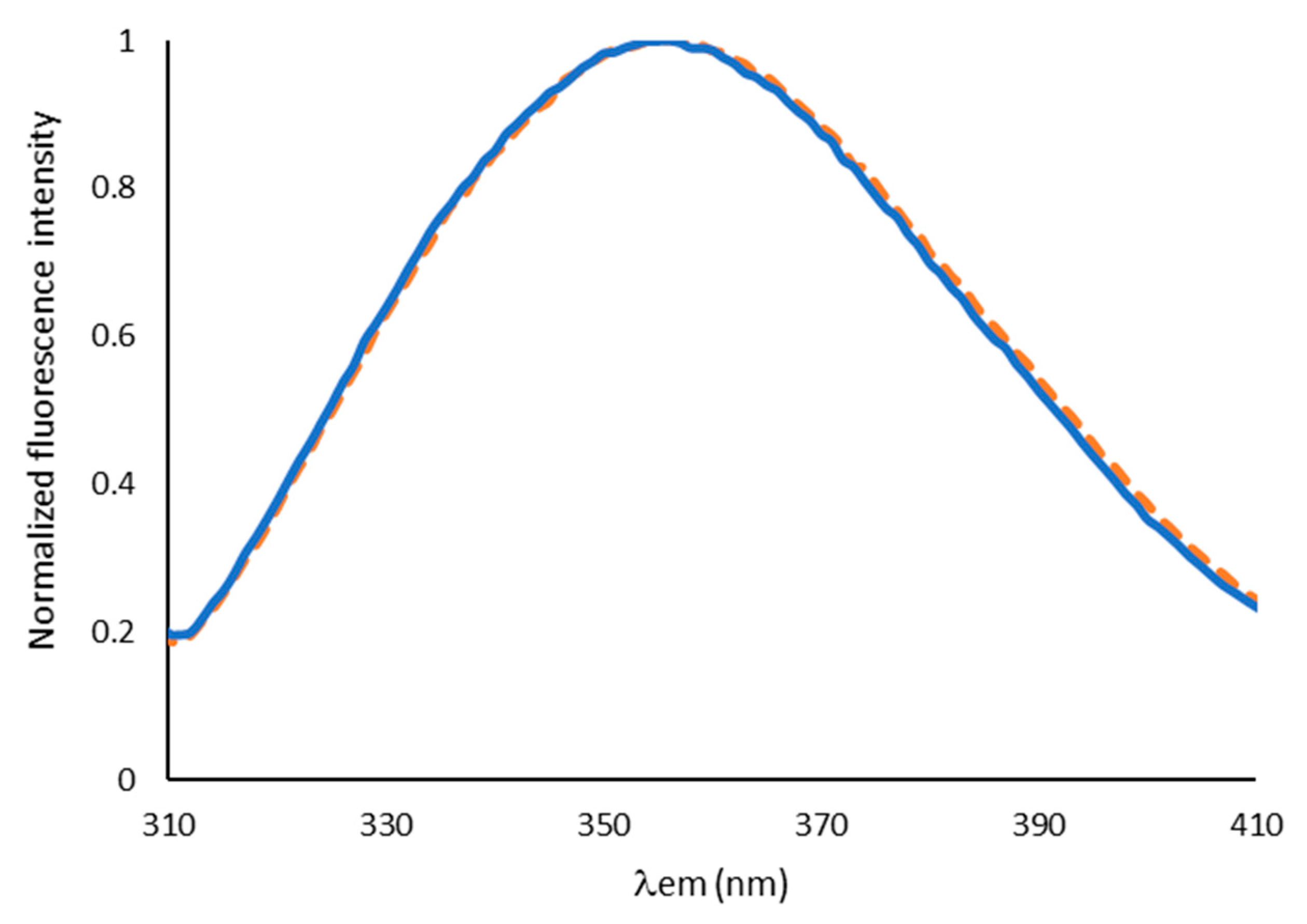
2.1. Characterization of Aqueous Media and BCA
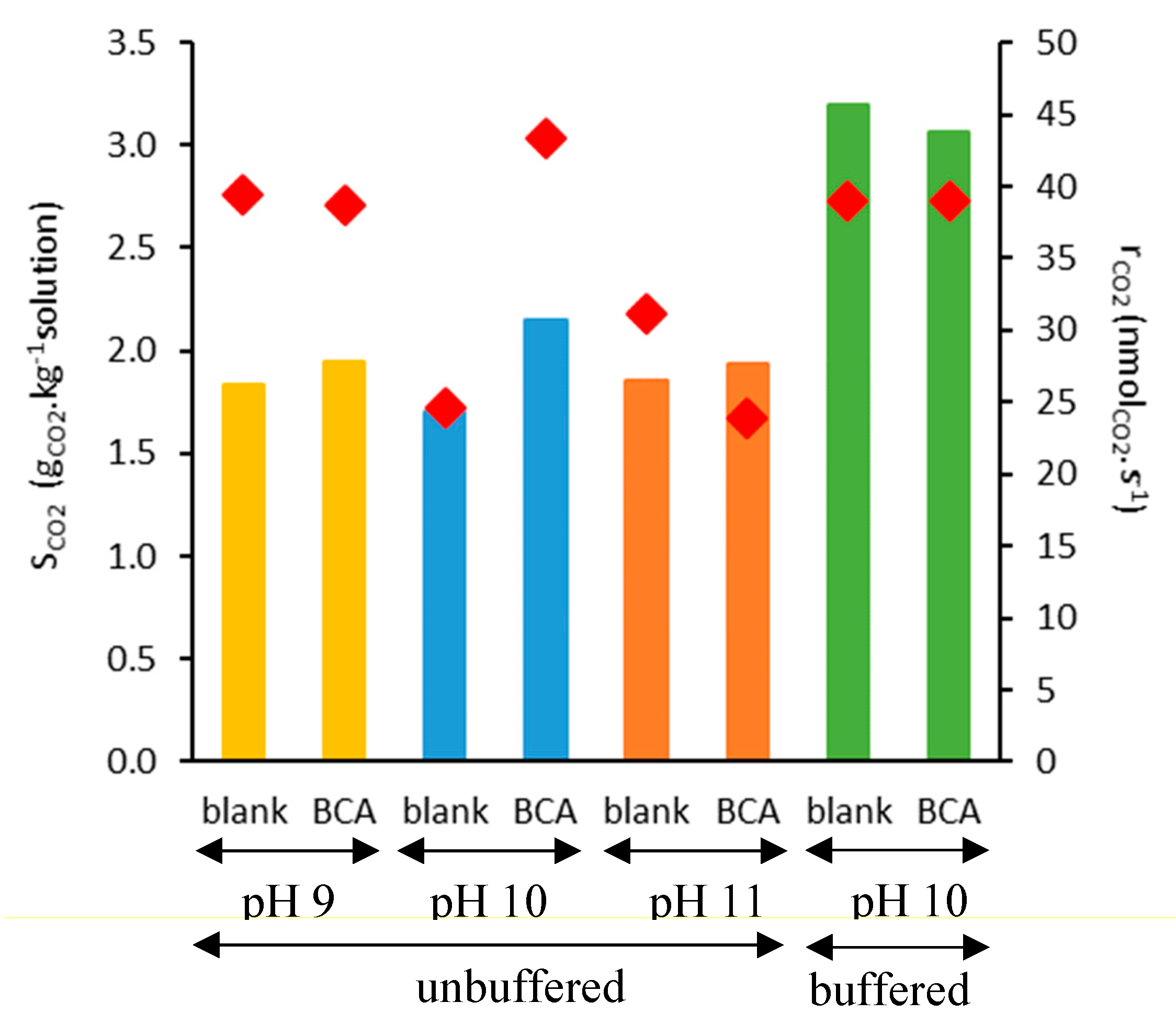
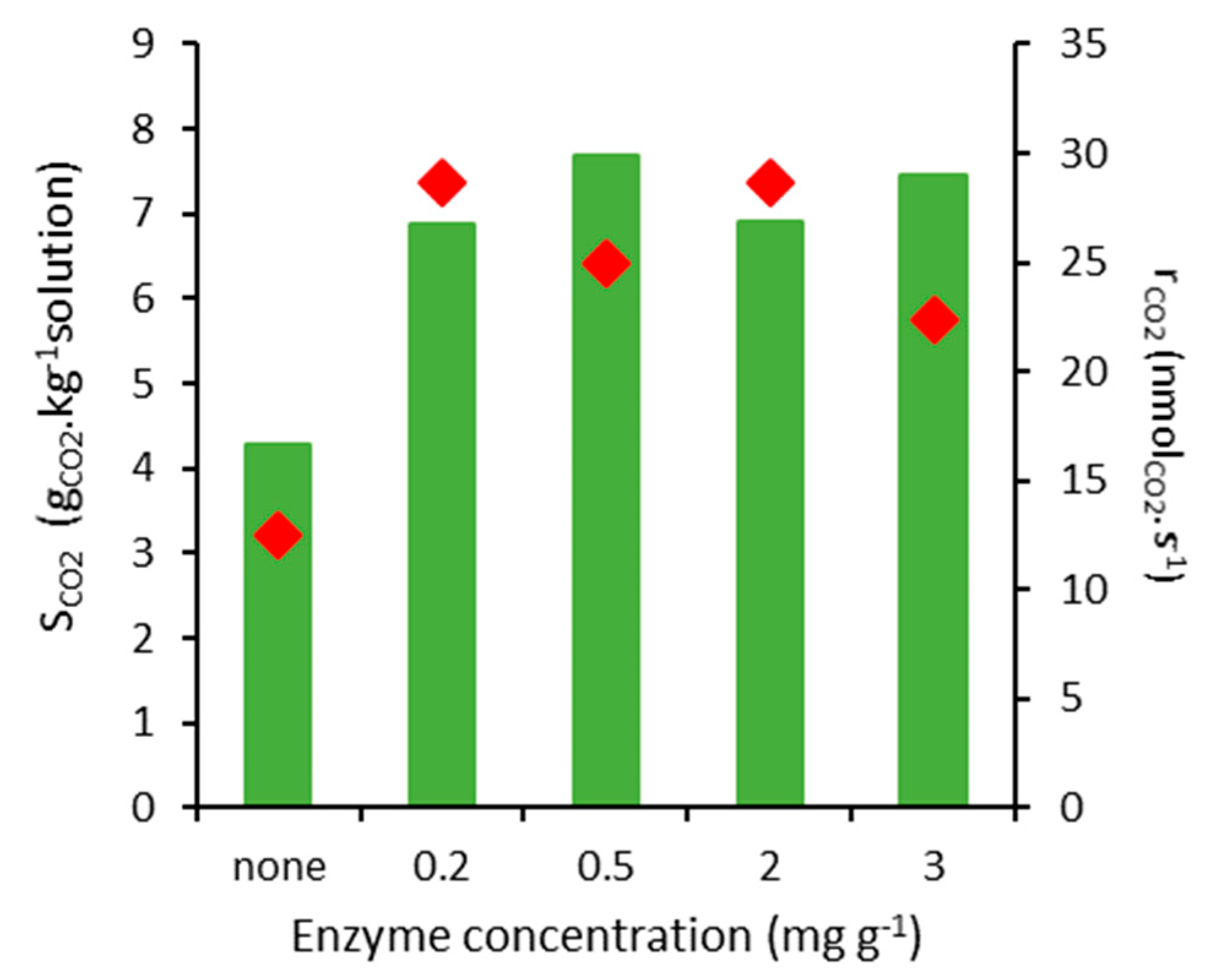
2.2. CO2 Absorption Tests
3. Materials and Methods
3.1. Materials
3.2. Solution Characterization
3.3. Enzyme Characterization
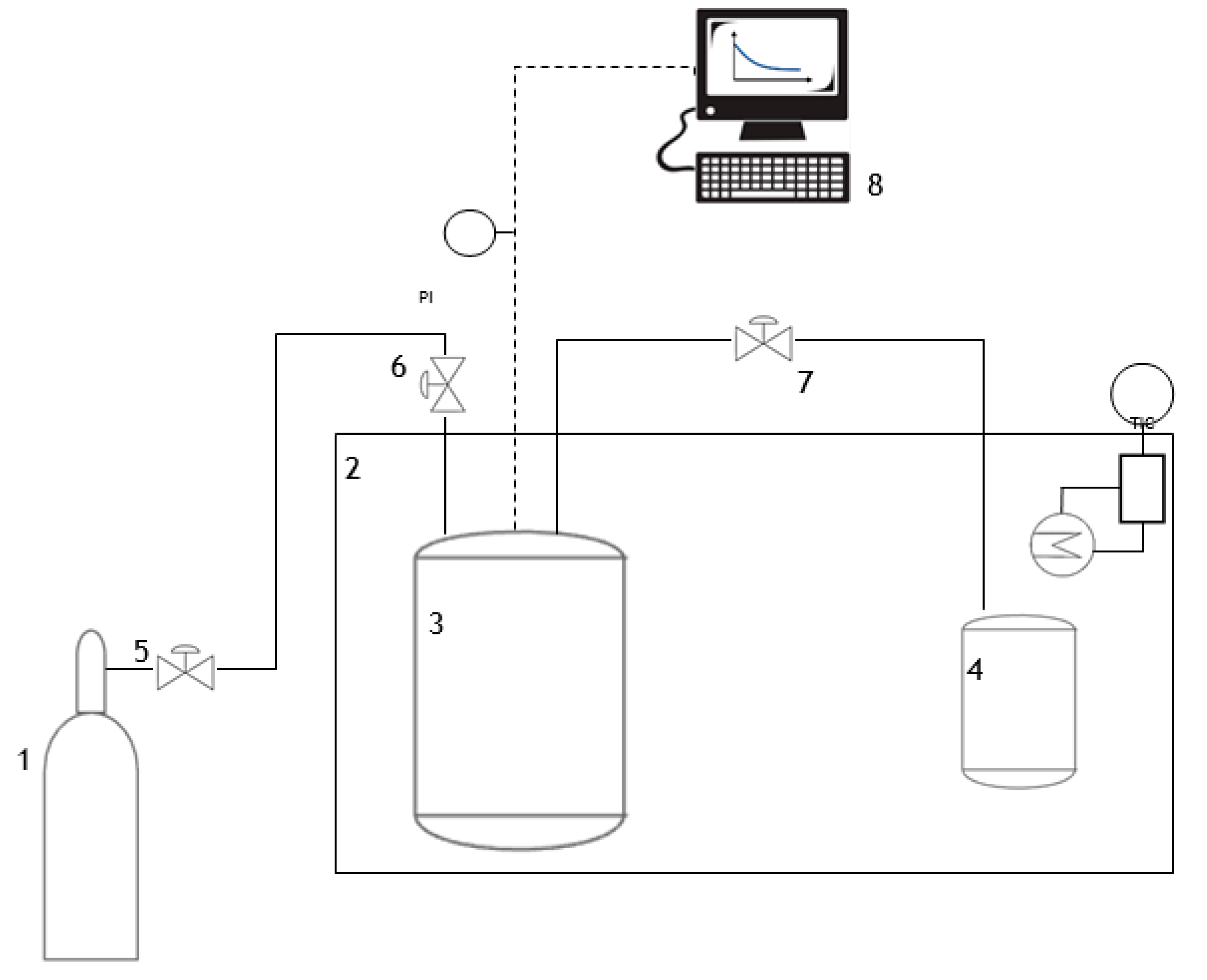
3.4. Sorption Tests
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Sahoo, P.C.; Kumar, M.; Puri, S.K.; Ramakumar, S.S.V. Enzyme inspired complexes for industrial CO2 capture: Opportunities and challenges. J. CO2 Util. 2018, 24, 419–429. [Google Scholar] [CrossRef]
- Bhattacharyya, D.; Miller, D.C. Post-combustion CO2 capture technologies—A review of processes for solvent-based and sorbent-based CO2 capture. Curr. Opin. Chem. Eng. 2017, 17, 78–92. [Google Scholar] [CrossRef]
- Effendi, S.S.W.; Ng, I.-S. The prospective and potential of carbonic anhydrase for carbon dioxide sequestration: A critical review. Proc. Biochem. 2019, 87, 55–65. [Google Scholar] [CrossRef]
- Boone, C.D.; Habibzadegan, A.; Gill, S.; McKenna, R. Carbonic anhydrases and their biotechnological applications. Biomolecules 2013, 3, 553–562. [Google Scholar] [CrossRef] [PubMed]
- Di Fiore, A.; Alterio, V.; Monti, S.M.; De Simone, G.; D’Ambrosio, K. Thermostable carbonic anhydrases in biotechnological applications. Int. J. Mol. Sci. 2015, 16, 15456–15480. [Google Scholar] [CrossRef] [PubMed]
- Aspatwar, A.; Kairys, V.; Rala, S.; Parikka, M.; Bozdag, M.; Carta, F.; Supuran, C.T.; Parkkila, S. Mycobacterium tuberculosis β-carbonic anhydrases: Novel targets for developing antituberculosis drugs. Int. J. Mol. Sci. 2019, 20, 5153. [Google Scholar] [CrossRef]
- Bose, H.; Satyanarayana, T. Microbial carbonic anhydrases in biomimetic carbon sequestration for mitigating global warming: Prospects and perspectives. Front. Microbiol. 2017, 8, 1615. [Google Scholar] [CrossRef]
- Montich, G.G. Partly folded states of bovine carbonic anhydrase interact with zwitterionic and anionic lipid membranes. Biochim. Biophys. Acta 2000, 1468, 115–126. [Google Scholar] [CrossRef]
- Bushmarina, N.A.; Kuznetsova, I.M.; Biktashev, A.G.; Turoverov, K.K.; Uversky, V.N. Partially folded conformations in the folding pathway of bovine carbonic anhydrase II: A fluorescence spectroscopic analysis. ChemBioChem 2001, 2, 813–821. [Google Scholar] [CrossRef]
- Gladis, A.; Lomholdt, N.F.; Fosbol, P.L.; Woodley, J.M.; von Solms, N. Pilot scale absorption experiments with carbonic anhydrase-enhanced MDEA—Benchmarking with 30 wt% MEA. Int. J. Greenh. Gas Control 2019, 82, 69–85. [Google Scholar] [CrossRef]
- Penders van-Elk, N.J.M.C.; Hamborg, E.S.; Huttenhuis, P.J.G.; Fradette, S.; Carley, J.A.; Versteeg, G.F. Kinetics of absorption of carbon dioxide in aqueous amine and carbonate solutions with carbonic anhydrase. Int. J. Greenh. Gas Control 2013, 12, 259–268. [Google Scholar] [CrossRef]
- Saunier, F.; Fradette, S.; Clerveaux, F.; Lefebvre, S.; Madore, E.; Veilleux, G.; Bulle, C.; Surprenant, R. Comparison of life-cycle assessment between bio-catalyzed and promoted potassium carbonate processes and amine-based carbon capture technologies. Int. J. Greenh. Gas Control 2019, 88, 134–155. [Google Scholar] [CrossRef]
- Abdelrahim, M.Y.M.; Martins, C.F.; Neves, L.A.; Capasso, C.; Supuran, C.T.; Coelhoso, I.M.; Crespo, J.G.; Barboiu, M. Supported ionic liquid membranes immobilized with carbonic anhydrases for CO2 transport at high temperatures. J. Membr. Sci. 2017, 528, 225–230. [Google Scholar] [CrossRef]
- Sarraf, N.S.; Sabourny, A.A.; Ranjbar, B.; Moosavi-Movahedi, A.A. Structural and functional changes of bovine carbonic anhydrase as a consequence of temperature. Acta Biochim. Pol. 2004, 51, 665–671. [Google Scholar] [CrossRef] [PubMed]
- Es-haghi, A.; Ebrahim-Habibi, A.; Sabbaghian, M.; Nemat-Gorgani, M. Amyloid-like aggregates formation by bovine apo-carbonic anhydrase in various alcohols: A comparative study. Int. J. Biol. Macromol. 2016, 92, 573–580. [Google Scholar] [CrossRef]
- Russo, M.E.; Bareschino, P.; Pepe, F.; Marzocchella, A.; Salatino, P. Modelling of enzymatic reactive CO2 absorption. Chem. Eng. Trans. 2018, 69, 457–462. [Google Scholar] [CrossRef]
- Dolgikh, D.A.; Kolomiets, A.P.; Bolotina, I.A.; Ptitsyn, O.B. “Molten-globule” state accumulates in carbonic anhydrase folding. FEBS Lett. 1984, 165, 88–92. [Google Scholar] [CrossRef]
- Kuwajima, K. The molten globule state as a clue for understanding the folding and cooperativity of globular-protein structure. Proteins Struct. Funct. Bioinform. 1989, 6, 87–103. [Google Scholar] [CrossRef]
- Ohgushi, M.; Wada, A. “Molten-globule state”: A compact form of globular proteins with mobile side-chains. FEBS Lett. 1983, 164, 21–24. [Google Scholar] [CrossRef]
- Qi, G.; Liu, K.; Frimpong, R.A.; House, A.; Salmon, S.; Liu, K. Integrated bench-scale parametric study on CO2 capture using a carbonic anhydrase promoted K2CO3 solvent with low temperature vacuum stripping. Ind. Eng. Chem. Res. 2016, 55, 12452–12459. [Google Scholar] [CrossRef]
- Russo, M.E.; Bareschino, P.; Olivieri, G.; Chirone, R.; Salatino, P.; Marzocchella, A. Modeling of slurry staged bubble column for biomimetic CO2 capture. Int. J. Greenh. Gas Control 2016, 47, 200–209. [Google Scholar] [CrossRef]
- Zhang, S.; Zhang, Z.; Lu, Y.; Rostam-Abadi, M.; Jones, A. Activity and stability of immobilized carbonic anhydrase for promoting CO2 absorption into a carbonate solution for post-combustion CO2 capture. Bioresour. Technol. 2011, 102, 10194–10201. [Google Scholar] [CrossRef] [PubMed]
- Gudiksen, K.L.; Gitlin, I.; Moustakas, D.T.; Whitesides, G.M. Increasing the net charge and decreasing the hydrophobicity of bovine carbonic anhydrase decreases the rate of denaturation with sodium dodecyl sulphate. Biophys. J. 2006, 91, 298–310. [Google Scholar] [CrossRef] [PubMed]
- Wojtasik, J.; Gladyszewski, K.; Skiborowski, M.; Gorak, A.; Piatkowski, M. Enzyme-enhanced CO2 absorption process in rotating packed bed. Chem. Pap. 2019, 73, 861–869. [Google Scholar] [CrossRef]
- Koytsoumpa, E.I.; Bergins, C.; Kakaras, E. The CO2 economy: Review of CO2 capture and reuse technologies. J. Supercrit. Fluids 2018, 132, 3–16. [Google Scholar] [CrossRef]
- Faridi, S.; Satynarayana, T. Novel alkalistable α-carbonic anhydrase from the polyextremophilic bacterium Bacillus halodurans: Characteristics and applicability in flue gas CO2 sequestration. Environ. Sci. Pollut. Res. 2016, 23, 15236–15249. [Google Scholar] [CrossRef]
- Li, W.; Chen, W.-S.; Zhou, P.-P.; Yu, L.-J. Influence of enzyme concentration on bio-sequestration of CO2 in carbonate form using bacterial carbonic anhydrase. Chem. Eng. J. 2013, 232, 149–156. [Google Scholar] [CrossRef]
- Russo, M.E.; Olivieri, G.; Marzocchella, A.; Salatino, P.; Caramuscio, P.; Cavaleiro, C. Post-combustion carbono capture mediated by carbonic anhydrase. Sep. Purif. Technol. 2013, 107, 331–339. [Google Scholar] [CrossRef]
- Ye, X.; Lu, Y. CO2 absorption into catalyzed potassium carbonate-bicarbonate solutions: Kinetics and stability of the enzyme carbonic anhydrase as a biocatalyst. Chem. Eng. Sci. 2014, 116, 567–575. [Google Scholar] [CrossRef]
- Micsonai, A.; Wien, F.; Bulyaki, E.; Kun, J.; Moussong, E.; Lee, Y.H.; Goto, Y.; Refregiers, M.; Kardos, J. BeStSel: A web server for accurate protein secondary structure prediction and fold recognition from the circular dichroism spectra. Nucleic Acids Res. 2018, 46, W315–W322. [Google Scholar] [CrossRef]
- Micsonai, A.; Wien, F.; Kernya, L.; Lee, Y.H.; Goto, Y.; Refregiers, M.; Kardos, J. Accurate secondary structure prediction and fold recognition for circular dichroism. Proc. Nat. Acad. Sci. USA. 2015, 112, E3095–E3103. [Google Scholar] [CrossRef] [PubMed]
- Wilbur, K.M.; Anderson, N.G. Electrometric and colorimetric determination of carbonic anhydrase. J. Biol. Chem. 1948, 176, 147–154. [Google Scholar] [PubMed]
- Neves, L.A.; Afonso, C.; Coelhoso, I.M.; Crespo, J.G. Integrated CO2 capture and enzymatic bioconversion in supported ionic liquid membranes. Sep. Purif. Technol. 2012, 97, 34–41. [Google Scholar] [CrossRef]
| Ion | Concentration (mg L−1) |
|---|---|
| Ca+2 | 0.386 ± 0.031 |
| K+ | 2.253 ± 0.053 |
| Mg+2 | 0.055 ± 0.007 |
| Na+ | 8.871 ± 0.267 |
| Al+3 | n.d. |
| Fe+2 | n.d. |
| Cl− | 38.976 ± 0.071 |
| NO2− | 4.804 ± 0.200 |
| SO4−2 | 54.911 ± 0.205 |
| pH | Tm (°C) | R2 |
|---|---|---|
| 9 | 72.82 ± 0.29 | 0.9783 |
| 10 | 70.45 ± 0.32 | 0.9738 |
| 11 | 61.78 ± 0.36 | 0.9707 |
| Initial pH | Enzyme Addition | pH Drop | Conductivity Change (μS cm−1) |
|---|---|---|---|
| 9 | no | 2.78 | +124 |
| yes | 2.37 | +17 | |
| 10 | no | 3.42 | −70 |
| yes | 3.45 | +78 | |
| 11 | no | 4.23 | −533 |
| yes | 4.26 | −488 |
| Enzyme Concentration (mg g−1) | Net Specific CO2 Absorption (molCO2 mol−1specie) |
|---|---|
| Blank | 0.00180 |
| 0.2 | 8014 |
| 0.5 | 5071 |
| 2 | 1005 |
| 3 | 761 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castro, A.M.d.; Ferreira, E.; Portugal, C.; Neves, L.A.; Crespo, J.G. Biocatalytic CO2 Absorption and Structural Studies of Carbonic Anhydrase under Industrially-Relevant Conditions. Int. J. Mol. Sci. 2020, 21, 2918. https://doi.org/10.3390/ijms21082918
Castro AMd, Ferreira E, Portugal C, Neves LA, Crespo JG. Biocatalytic CO2 Absorption and Structural Studies of Carbonic Anhydrase under Industrially-Relevant Conditions. International Journal of Molecular Sciences. 2020; 21(8):2918. https://doi.org/10.3390/ijms21082918
Chicago/Turabian StyleCastro, Aline M. de, Elisabete Ferreira, Carla Portugal, Luisa A. Neves, and João G. Crespo. 2020. "Biocatalytic CO2 Absorption and Structural Studies of Carbonic Anhydrase under Industrially-Relevant Conditions" International Journal of Molecular Sciences 21, no. 8: 2918. https://doi.org/10.3390/ijms21082918
APA StyleCastro, A. M. d., Ferreira, E., Portugal, C., Neves, L. A., & Crespo, J. G. (2020). Biocatalytic CO2 Absorption and Structural Studies of Carbonic Anhydrase under Industrially-Relevant Conditions. International Journal of Molecular Sciences, 21(8), 2918. https://doi.org/10.3390/ijms21082918






